Submitted:
13 April 2024
Posted:
16 April 2024
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Definitions
3. Special Features of Earth and CBEs
3.1. Special Features of Earth
3.2. Special Features of H1-CBEs (Carbon Atoms)
3.3. Special Features of Other CBEs
4. The Driving Force Mechanism
5. The Structural Mechanism
6. The Natural Selection Mechanism
7. The CBEP from the Lens of the CBET
7.1. The Core Viewpoints of the CBET
7.2. Chemical Evolution from the Lens of the CBET
7.3. Biological Evolution from the Lens of the CBET
7.4. Social Evolution from the Lens of the CBET
7.5. The Natural Roots of Multiple Important Social Management Notions
8. Reliability of the CBET
9. Novelties of the CBET
9. Conclusions and Perspectives
Acknowledgments
Conflicts of Interest
References
- Xie, P. The aufhebung and breakthrough of the theories on the origin and evolution of life (Science Press, 2014).
- Schreiber, A.; Gimbel, S. Evolution and the second law of thermodynamics: Effectively communicating to non-technicians. Evo Edu Outreach 2010, 3, 99–106. [Google Scholar] [CrossRef]
- Schrodinger, E. What is life (Cambridge University Press, 2012).
- Prigogine, I. Time, structure and fluctuation (Nobel Lecture). Science 1978, 201, 777–785. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.M.; Chen, J.W. Root of science?the driving force and mechanisms of the extensive evolution (Science Press, 2000).
- Oparin, A.I. Chemistry and the origin of life. R. Inst. Chem. Rev. 1969, 2, 1–12. [Google Scholar] [CrossRef]
- Guo, X.; Fu, S.; Ying, J.; Zhao, Y. Prebiotic chemistry: a review of nucleoside phosphorylation and polymerization. Open Biol. 2023, 13, 220234. [Google Scholar] [CrossRef] [PubMed]
- Sumie, Y.; Sato, K.; Kakegawa, T.; Furukawa, Y. Boron-assisted abiotic polypeptide synthesis. Commun. Chem. 2023, 6, 89. [Google Scholar] [CrossRef] [PubMed]
- de Graaf, R.; De Decker, Y.; Sojo, V.; Hudson, R. Quantifying catalysis at the origin of life. Chemistry 2023, 29, e202301447. [Google Scholar] [CrossRef] [PubMed]
- Nogal, N.; Sanz-Sánchez, M.; Vela-Gallego, S.; Ruiz-Mirazo, K.; de la Escosura, A. The protometabolic nature of prebiotic chemistry. Chem Soc Rev. 2023, 52, 7359–7388. [Google Scholar] [CrossRef] [PubMed]
- Chieffo, C.; Shvetsova, A.; Skorda, F.; Lopez, A.; Fiore, M. The origin and early evolution of life: Homochirality emergence in prebiotic environments. Astrobiology 2023, 23, 1368–1382. [Google Scholar] [CrossRef]
- Fiore, M. Prebiotic chemistry and life's origin (Royal Society of Chemistry, 2022). [CrossRef]
- Anna Neubeck, A.; McMahon, S. Prebiotic chemistry and the origin of life (Springer, 2021). [CrossRef]
- Farías-Rico, J.A.; Mourra-Díaz, C.M. A short tale of the origin of proteins and ribosome evolution. Microorganisms 2022, 10, 2115. [Google Scholar] [CrossRef]
- Ershov, B. Natural radioactivity and chemical evolution on the early Earth: Prebiotic chemistry and oxygenation. Molecules 2022, 27, 8584. [Google Scholar] [CrossRef]
- Eigen, M.; Schuster, P. Stages of emerging life — Five principles of early organization. J. Mol. Evo. 1982, 19, 47–61. [Google Scholar] [CrossRef] [PubMed]
- Futuyma, D.J.; Kirkpatrick, M. Evolution (Sinauer Associates, 2017).
- White, L.A. The evolution of culture: the development of civilization to the fall of Rome (Routledge, 2010).
- Richerson, P.J.; Christiansen, M.H. Cultural evolution: Society, technology, language, and religion (The MIT Press, 2013).
- Laland, K.N. Darwin's unfinished symphony: How culture made the human mind (Princeton University Press, 2017).
- Seager, S. Exoplanet habitability. Science 2013, 340, 577–581. [Google Scholar] [CrossRef] [PubMed]
- Cohen, K.M.; Harper, D.A.T.; Gibbard, P.L.; Car, N. International chronostratigraphic chart (https://stratigraphy.org/ICSchart/ChronostratChart2023-09.pdf, accessed 9 April 2024).
- Charette, M.A. , & Smith, W.H.F. The volume of Earth's ocean. Oceanography 2010, 23, 112–114. [Google Scholar] [CrossRef]
- Carbon (https://en.wikipedia.org/wiki/carbon, accessed 9 April 2024).
- Roston, E. The carbon age: How life's core element has become civilization's greatest threat (Walker & Company, 2008).
- Li, Z.H.; Bailleul, A.M.; Stidham, T.A.; Wang, M.; Teng, T. Exceptional preservation of an extinct ostrich from the Late Miocene Linxia Basin of China. Vertebrata PalAsiatica 2021, 59, 229. [Google Scholar] [CrossRef]
- Heck, P.R.; Greer, J.; Kööp, L.; et al. Lifetimes of interstellar dust from cosmic ray exposure ages of presolar silicon carbide. Proc. Natl. Acad. Sci. U. S. A. 2020, 117, 1884–1889. [Google Scholar] [CrossRef] [PubMed]
- Borgnakke, C.; Sonntag, R.E. Fundamentals of thermodynamics (Wiley, 2022).
- Thermodynamics and chemistry https://www2.chem.umd.edu/thermobook/v10-screen.pdf (DeVoe, H., accessed 9 April 2024).
- Martin, W.F.; Sousa, F.L.; Lane, N. Energy at life's origin. Science 2014, 344, 1092–1093. [Google Scholar] [CrossRef] [PubMed]
- von Bertalanffy, L. General system theory: Foundations, development, applications (George Braziller, 1968).
- Morrell, D.G. Catalysis of organic reactions (CRC Press, 2019).
- Stone, E.A.; Cutrona, K.J.; Miller, S.J. Asymmetric catalysis upon helically chiral loratadine analogues unveils enantiomer-dependent antihistamine activity. J. Am. Chem. Soc. 2020, 142, 12690–12698. [Google Scholar] [CrossRef]
- de Graaf, R.; De Decker, Y.; Sojo, V.; Hudson, R. Quantifying catalysis at the origin of life. Chemistry. 2023, 29, e202301447. [Google Scholar] [CrossRef]
- Fitzgerald, D.M.; Rosenberg, S.M. What is mutation? A chapter in the series: How microbes “jeopardize” the modern synthesis. PLoS Genet. 2019, 15, e1007995. [Google Scholar] [CrossRef]
- Olivieri, D.N.; Mirete-Bachiller, S.; Gambón-Deza, F. Insights into the evolution of IG genes in amphibians and reptiles. Dev. Comp. Immunol. 2021, 114, 103868. [Google Scholar] [CrossRef]
- Sabarís, G.; Fitz-James, M.H.; Cavalli, G. Epigenetic inheritance in adaptive evolution. Ann. N. Y. Acad. Sci. 2023, 1524, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Sun, Y. Variation in the analysis of positively selected sites using nonsynonymous/synonymous rate ratios: An example using influenza virus. PLoS One. 2011, 6, e19996. [Google Scholar] [CrossRef]
- McGee, M.D.; Borstein, S.R.; Meier, J.I. , et al. The ecological and genomic basis of explosive adaptive radiation. Nature. 2020, 586, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Mayr, E. Ecological factors in speciation. Evolution. 1947, 1, 263–288. [Google Scholar] [CrossRef]
- Gómez-Schiavon, M.; Buchler, N.E. Epigenetic switching as a strategy for quick adaptation while attenuating biochemical noise. PLoS Comput Biol. 2019, 15, e1007364. [Google Scholar] [CrossRef] [PubMed]
- Benton, M.J. The red queen and the Court Jester: species diversity and the role of biotic and abiotic factors through time. Science 2009, 323:728–732. [CrossRef]
- Olejarz, J.; Iwasa, Y.; Knoll, A.H. , et al. The Great Oxygenation Event as a consequence of ecological dynamics modulated by planetary change. Nat. Commun. 2021, 12, 3985. [Google Scholar] [CrossRef] [PubMed]
- Schmitt-Kopplin, P.; Gabelica, Z.; Gougeonm, R.D. , et al. High molecular diversity of extraterrestrial organic matter in Murchison meteorite revealed 40 years after its fall. Proc. Natl. Acad. Sci. U. S. A. 2010, 107, 2763–2768. [Google Scholar] [CrossRef]
- Percival, L.M.E.; Ruhl, M.; Hesselbo, S.P. , et al. Mercury evidence for pulsed volcanism during the end-Triassic mass extinction. Proc. Natl. Acad. Sci. U. S. A. 2017, 114, 7929–7934. [Google Scholar] [CrossRef] [PubMed]
- Dodd, M.S.; Papineau, D.; Grenne, T.; et al. Evidence for early life in earth's oldest hydrothermal vent precipitates. Nature 2017, 543, 60–64. [Google Scholar] [CrossRef]
- Weiss, M.C.; Sousa, F.L.; Mrnjavac, N. , et al. The physiology and habitat of the last universal common ancestor. Nat. Microb. 2016, 1, 16116. [Google Scholar] [CrossRef]
- Stobart, C.C.; Moore, M.; L. RNA virus reverse genetics and vaccine design. Viruses 2014, 6, 2531–2550. [Google Scholar] [CrossRef] [PubMed]
- Hutchison, C.A. , 3rd.; Chuang, R.Y.; Noskov, V.N., et al. Design and synthesis of a minimal bacterial genome. Science 2016, 351, aad6253. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Zhu, M.; Knoll, A. Decimetre-scale multicellular eukaryotes from the 1.56-billion-year-old Gaoyuzhuang formation in North China. Nat. Commun. 2016, 7, 11500. [Google Scholar] [CrossRef] [PubMed]
- Han, T.M.; Runnegar, B. Megascopic eukaryotic algae from the 2.1-billion-year-old negaunee iron-formation, Michigan. Science 1992, 257, 232–235. [Google Scholar] [CrossRef] [PubMed]
- Robertson, M.P.; Joyce, G.F. The origins of the RNA world. Cold Spring Harb. Perspect. Biol. 2012, 4, a003608. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.P.; Woltz, C.R.; Tosca, N.J.; Porter, S.M.; Briggs, D.E.G. Fossilisation processes and our reading of animal antiquity. Trends Ecol Evol. 2023, 38, 1060–1071. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Yin, X.; Shih, C.; Gao, T.; Ren, D. Termite colonies from mid-Cretaceous Myanmar demonstrate their early eusocial lifestyle in damp wood. Natl. Sci. Rev. 2020, 7, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Mera-Rodríguez, D.; Jourdan, H.; Ward, P.S.; Shattuck, S.; Cover, S.P.; Wilson, E.O.; Rabeling, C. Biogeography and evolution of social parasitism in Australian Myrmecia bulldog ants revealed by phylogenomics. Mol. Phyogenet. Evol. 2023, 186, 107825. [Google Scholar] [CrossRef] [PubMed]
- Nowak, M.; Tarnita, C.; Wilson, E. The evolution of eusociality. Nature 2010, 466, 1057–1062. [Google Scholar] [CrossRef]
- Plowers, N. An introduction to eusociality. Nature Education Knowledge 2010, 3, 7. [Google Scholar]
- Moffett, M.W. Adventures among ants (University of California Press, 2010).
- 59. SIPRI Military Expenditure Database, (https://www.sipri.org/databases/milex, accessed 9 April 2024).
- Ramstead, M.J.D.; Badcock, P.B.; Friston, K.J. Answering Schrödinger's question: A free-energy formulation. Phys. Life Rev. 2018, 24, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Skene, K.R. Systems theory, thermodynamics and life: Integrated thinking across ecology, organization and biological evolution. Biosystems 2024, 236, 105123. [Google Scholar] [CrossRef] [PubMed]
- Rott, P.; Grinstead, S.; Dallot, S. , et al. Genetic diversity, evolution, and diagnosis of sugarcane yellow leaf virus from 19 sugarcane-producing locations worldwide. Plant Dis. 2023, 107, 3437–3447. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Liu, X.; Wang, C. , et al. The pig pangenome provides insights into the roles of coding structural variations in genetic diversity and adaptation. Genome Res. 2023, 33, 1833–1847. [Google Scholar] [CrossRef]
- Tang, R.; Zhu, Y.; Yang, S. , et al. Genome-wide identification and analysis of WRKY gene family in Melastoma dodecandrum. Int. J. Mol. Sci. 2023, 24, 14904. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, X.; Feng, Y. Autotetraploid origin of Chinese cherry revealed by chromosomal karyotype and in situ hybridization of seedling progenies. Plants (Basel) 2023, 12, 3116. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Guo, Y.; Liu, D. , et al. Genome-wide analysis of the membrane attack complex and perforin genes and their expression pattern under stress in the Solanaceae. Int. J. Mol. Sci. 2023, 24, 13193. [Google Scholar] [CrossRef]
- Chen, J.M.; Chen, J.W. Disproving two widely accepted notions regarding entropy. Preprints 2024. https://www.preprints.org/manuscript/202404.0655/v1.
- Rudman, L.A.; Saud, L.H. Justifying social inequalities: The role of social Darwinism. Pers. Soc. Psychol. B. 2020, 46, 1139–1155. [Google Scholar] [CrossRef]
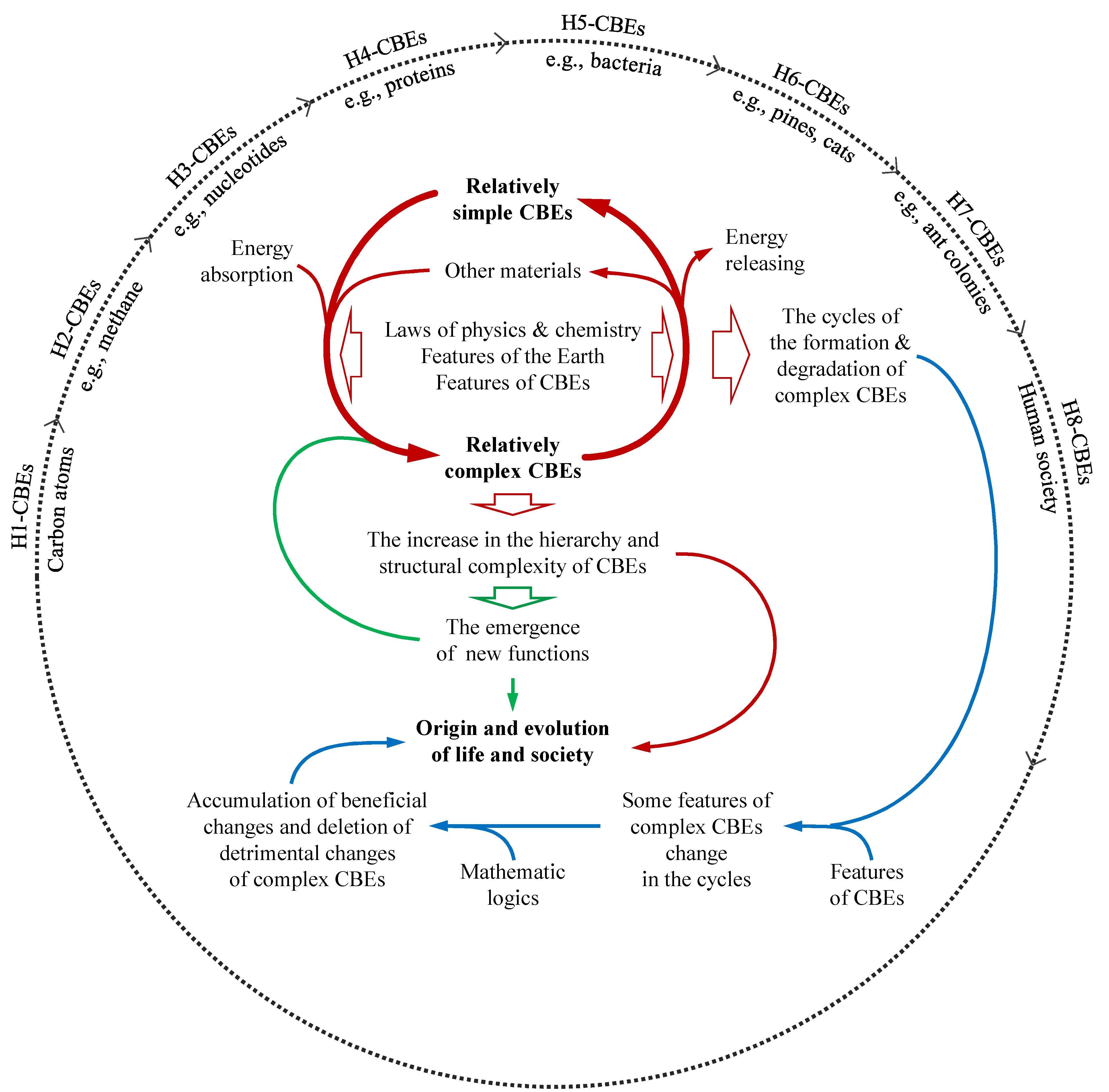

| Definition and inner structures | Features or functions associated with the evolution of CBEs |
| H1-CBEs refer to carbon atoms. Carbon atoms are composed of protons, neutrons, and electrons that are restricted by laws of physics and chemistry | Carbon atoms are abundant on Earth and in the universe. Carbon atoms can form some small molecules and myriad intermediate molecules with other atoms. Among all atoms, only carbon atoms can act as the backbone of myriad large molecules. |
| H2-CBEs refer to small carbon-containing molecules (CCMs), such as methane and carbon dioxide, composed of carbon atoms and other atoms that are restricted by laws of physics and chemistry | The production of many H2-CBEs can be aided by catalyzers; some H2-CBEs can participate in the evolution of CBEs as catalyzers, protectors, energy carriers, or constituent materials for the production of other molecules; some H2-CBEs, along with other molecules, can form H3-CBEs when they absorb energy. |
| H3-CBEs refer to intermediate CCMs, such as lysine and glucose, composed of carbon atoms and other atoms that are restricted by laws of physics and chemistry | The production of many H3-CBEs can be aided by catalyzers; and many H3-CBEs can participate in the evolution of CBEs as catalyzers, protectors, energy carriers, or constituent materials for the production of other molecules; some H3-CBEs, along with other molecules, can form H4-CBEs when they absorb energy. |
| H4-CBEs refer to large CCMs, such as proteins and nucleic acids; composed of H3-CBE residues and some functional groups that are restricted by laws of physics and chemistry | The production of H4-CBEs can be aided by catalyzers; and many H4-CBEs can participate in the evolution of CBEs as catalyzers, protectors, energy carriers, or constituent materials for the production of other molecules; some H4-CBEs, along with other materials, can form H5-CBEs through myriad spontaneous random formations of multiple-molecular structures. |
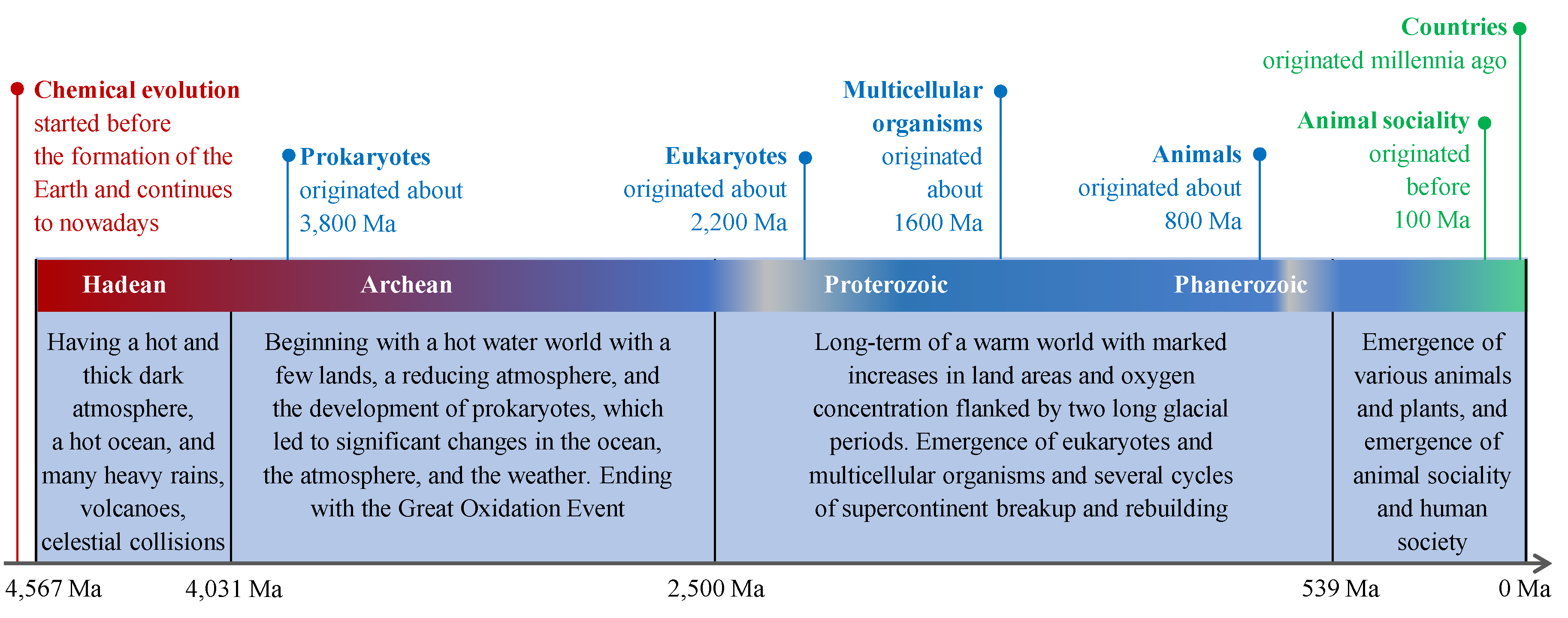
| H5-CBEs refer to cells, such as bacteria, composed of various H4-CBEs and other molecules that are restricted by the laws of physics, chemistry, and biology for cells and their nucleic acids | Cells have complex functions stemming from their complex structures to obtain relevant materials and energy to reproduce and maintain themselves with variations and confront natural selection; some H5-CBEs can form H6-CBEs through major variations. |
| H6-CBEs refer to multicellular organisms, such as pines and rabbits, composed of some H5-CBEs (cells) and other materials that are restricted by laws of physics, chemistry, and biology for the H5-CBE and their reproductive cells | Multicellular organisms have complex functions stemming from their complex structures to obtain relevant materials and energy to reproduce and maintain themselves with variations and confront natural selection; some multicellular organisms formed H7-CBEs through major variations. |
| H7-CBEs refer to animal social collectives, such as ant colonies, composed of some H6-CBEs (animal individuals) that are restricted by laws of physics, chemistry, and biology and collaborate with different duties for the collectives | Animal social collectives utilize collective advantages and hence have great power in obtaining relevant materials and energy to maintain and rejuvenate themselves and reduce inner competition and conflicts. They have reduced the competition inside animal social collectives and elevated the competition among animal social collectives. |
| H8-CBEs refer to human social collectives, which are restricted by laws of physics, chemistry, and social morals and rules; they form multiple hierarchies (e.g., clans, tribes, and kingdoms, or towns, counties, provinces, countries, and country allies); low-hierarchy collectives collaborate with different duties for high-hierarchy collectives | Humans have established multiple hierarchies of social collectives due to their intelligence, knowledge accumulation, and collaboration spirits. Some H8-CBEs utilize collective advantages and hence have great power in obtaining material and energy to maintain and rejuvenate themselves and confronting competition and conflicts within or between human collectives. Technological development has elevated the destructive power of these competition and conflicts to the extent of destroying humanity and Earth, which underpins the integration of all countries into a global harmonious social collective. |
| Core viewpoints | Explanations |
| 1. The energy dissipated from the permanent energy sources, such as sunlight, geothermal energy, cosmic radiation, water flow, wind, etc., is adjusted by the atmosphere and the abundant water on Earth. Many substances on Earth spontaneously or actively absorb energy from these sources under some principles of physics and chemistry (e.g., the second law of thermodynamics). Some carbon-based entities (CBEs) can form more complex structures due to their special features after energy absorption. This constitutes the driving force mechanism that provides energy for the evolution of CBEs on Earth. | Energy is essential for the synthesis of organic molecules, the growth of plants, the movement of animals, the reproduction of organisms, and the development of human society. The second law of thermodynamics has been mistaken by many people (including some scientists) for decades to contradict biological evolution, which have retarded the development of evolutionary theories and have been employed by creationists to challenge evolutionary theories.Among all atoms, only carbon can lead other atoms to form multiple hierarchies of structures due to some special features of carbon atoms and other CBEs. Among all known planets, only Earth has been found to support life due to its rare habitable features. These special features that have been overlooked in previous evolutionary theories are essential for a theory to provide direct, and explicit explanations for CBE evolution. |
| 2. Some complex CBEs possess new functions that less complex CBEs do not. This constitutes the structural mechanism that generates new functions for the evolution of CBEs on Earth. Some complex CBEs can hence obtain the reproduction and self-protection functions. | This mechanism stems from the logic that a new structure can engender new functions. For example, birds can fly, but bird cells cannot. This logic is equivalent to the core principle of systems theory: the whole exceeds the sum of its parts. This mechanism is aided by the driving force mechanism and reinforced by the natural selection mechanism. |
| 3. The above two mechanisms lead to the formation and accumulation of various complex CBEs on Earth. | The driving mechanism and the structural mechanism are more obvious in chemical evolution and biological evolution, respectively. |
| 4. Almost all complex CBEs will degrade, and regenerated complex CBEs usually carry variations due to some features of CBEs. Therefore, there are cycles of formation and degradation of complex CBEs with variations. 5. The cycles, in mathematics, lead to the accumulation of the variations beneficial to the formation and maintenance of complex CBEs and the deletion of detrimental variations, which constitutes the natural selection mechanism. |
Natural selection was explained by previous theories with the phenomenon of survival competition in organisms. It is explained in the CBET using its mathematical essence. The CBET also extends natural selection from biological evolution to the competition among organic molecules (chemical evolution) and the competition of animal and human social collectives (social evolution). The driving mechanism and the structural mechanism explain why complex CBEs emerge on Earth and the natural selection explain why some complex CBEs can exist less than others or cannot exist. |
| 6. The synergistic action of the above three mechanisms results in the progression from chemical to biological and social evolution, marked by the escalating hierarchy of CBEs and the increase in the quantity, diversity, and orderliness of high-hierarchy CBEs. | Previous theories overlooked the energy driving the evolution of CBEs and addressed only the natural selection or another single mechanism of evolution. Meanwhile, chemical evolution, biological evolution, and social evolution were largely investigated separately in previous theories. |
| Issue | Previous explanations | Explanations of the CBET |
| The mechanisms of chemical, biological, and social evolution | The three phases of the carbon-based evolution were largely investigated separately, and no theories have explicitly interpreted them from a panorama view | The CBET provides new, direct, and explicit explanations for the carbon-based evolution in its entirety from a panorama view and elucidates the mechanisms shared by chemical evolution, biological evolution, and social evolution |
| Evolution and physics | Using elusive concepts (e.g., negative entropy, dissipative systems, or maximum entropy production) to explain the contradiction between the second law of thermodynamics and evolution | Using the concept of eight hierarchies of carbon-based entities (CBEs) and three mechanisms to provide direct, and explicit explanations for the CBEP from a panoramic view, accepting the new notion that no contradiction exists between the second law of thermodynamics and evolution, and clarifying that this law is highly associated with the driving force of evolution |
| The driving force of evolution | Natural selection, genetic drift, competition, or mutation, none of which directly involve energy | Many CBEs on Earth can absorb energy from energy sources on Earth, which supports them in forming more complex CBEs in terms of energy |
| The mechanisms of evolution | Natural selection, sexual selection, and epigenetic changes, which cannot explain macroevolution | The synergistic action of the driving force mechanism, the structural mechanism, and the natural selection mechanism, which can explain microevolution and macroevolution |
| Origin of life | Highlighting the roles of RNA, autocatalysis, and inorganic catalyzers | Highlighting the role of the collaboration of various molecules, allocatalysis, and organic catalyzers |
| Natural selection | Being explained with survival competition among organisms (the phenomenon), highlighting the selection targets of a single trait, genetic changes, individuals, or populations, highlighting competition rather than inclusiveness in natural selection | Being explained with mathematical logic (the essence), highlighting the selection targets of the overall fitness of complex CBEs and thus allowing the existence of disadvantageous traits, highlighting the roles of inheritable changes and non-inheritable changes in natural selection, highlighting the roles of multiple hierarchies of CBEs in natural selection, and highlighting both fierce competition and inclusiveness in natural selection |
| Natural roots of key social management notions | Only highlighting the importance of fierce competition and selfishness in the evolution | The natural roots of multiple pivotal and seemingly paradoxical social management notions, such as inclusiveness and competition, altruism and selfishness, freedom and restriction, inherited advantages and acquired strengths, as well as specialized development and all-around development, are revealed |
| Inclusion of evolutionary facts | Natural selection, non-random mutations, neutral mutations, epigenetic changes, and acquired strengths, cannot be integrated into a previous evolutionary theory | Natural selection, non-random mutations, neutral mutations, epigenetic changes, and acquired strengths are integrated into the cohesive framework of the CBET |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




