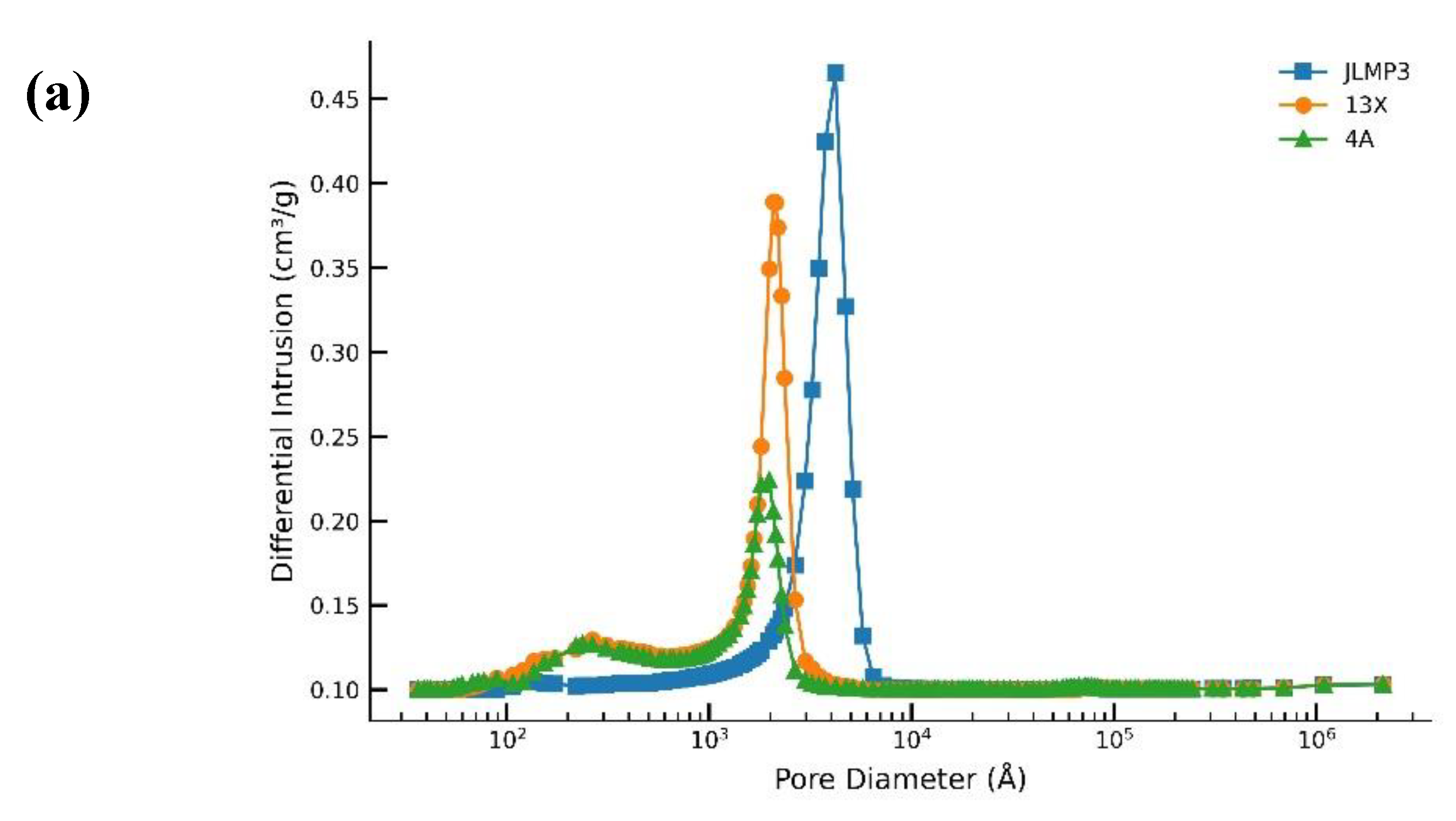
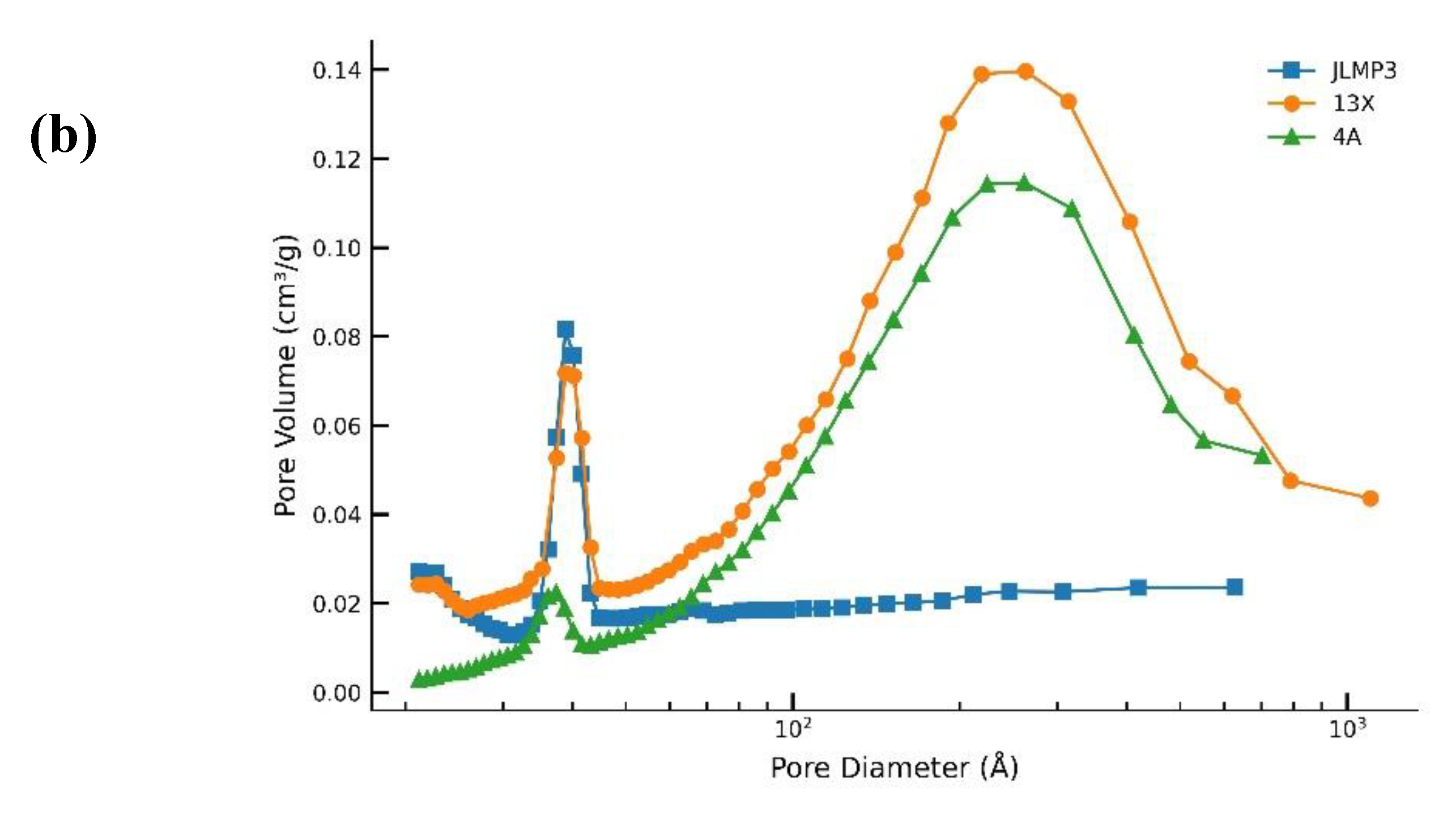
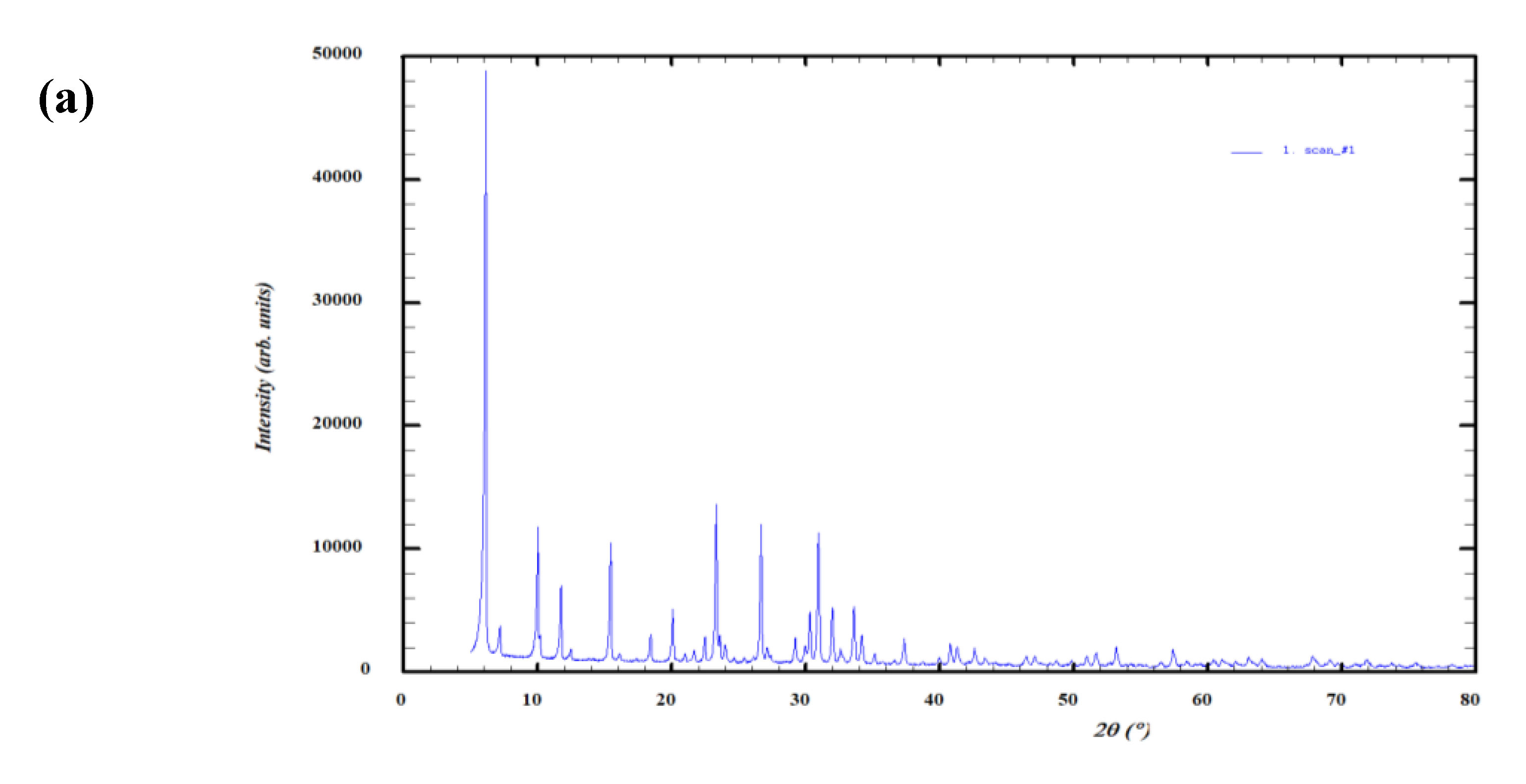
3.1. 40 g Packed Bed of Molecular Sieves JLPM3, 13X and 4A (No Pre-treatment)
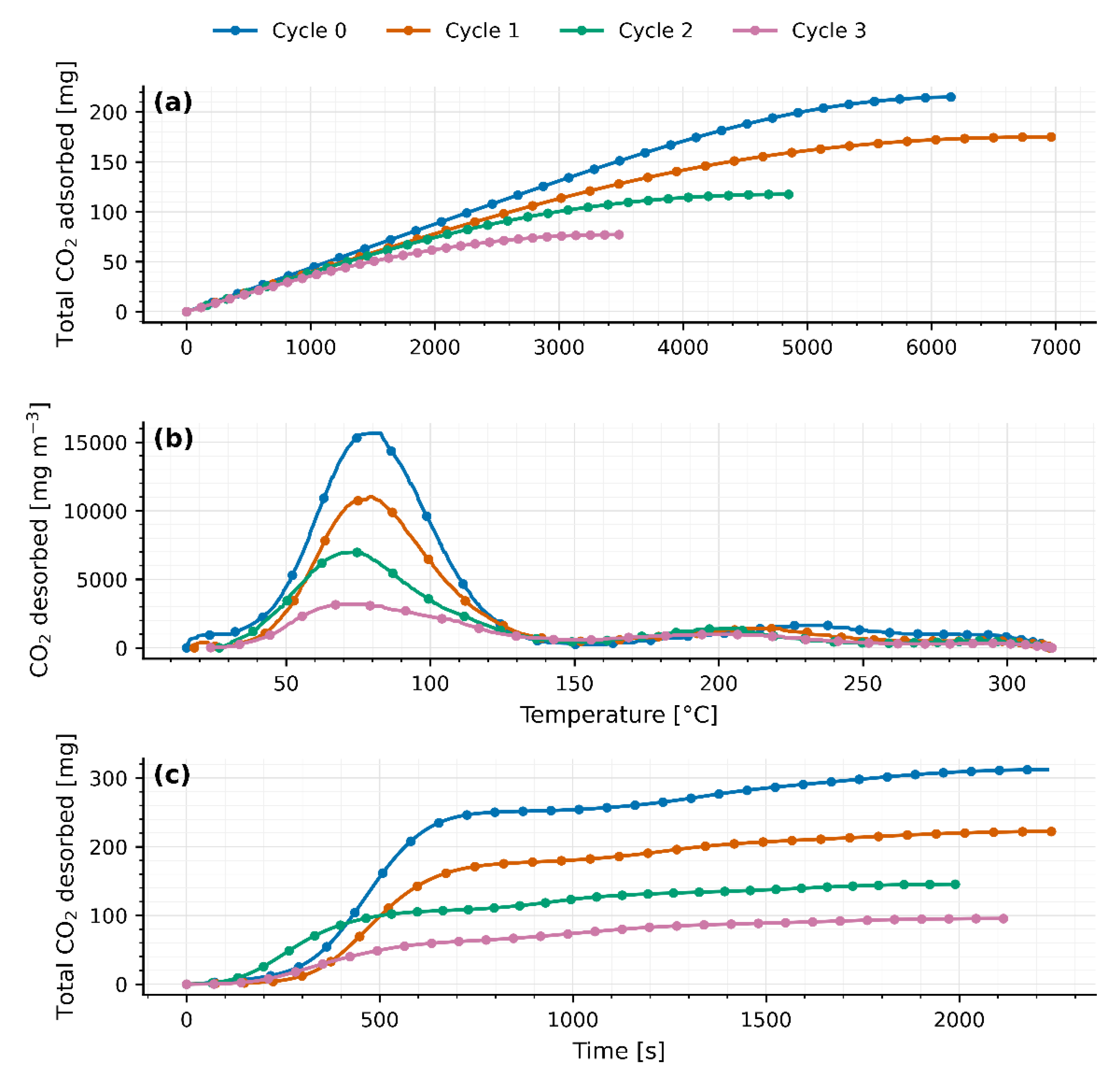
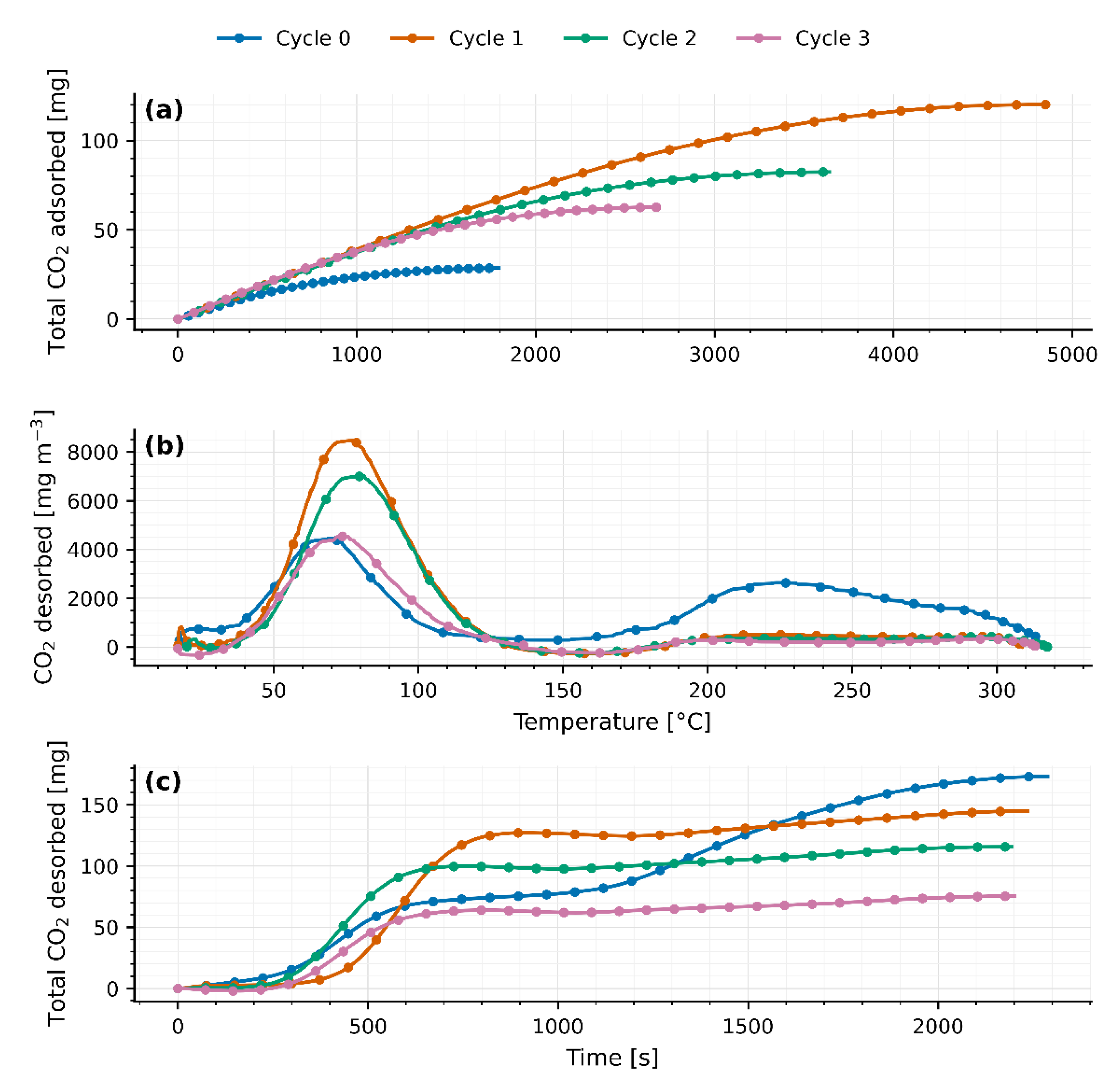
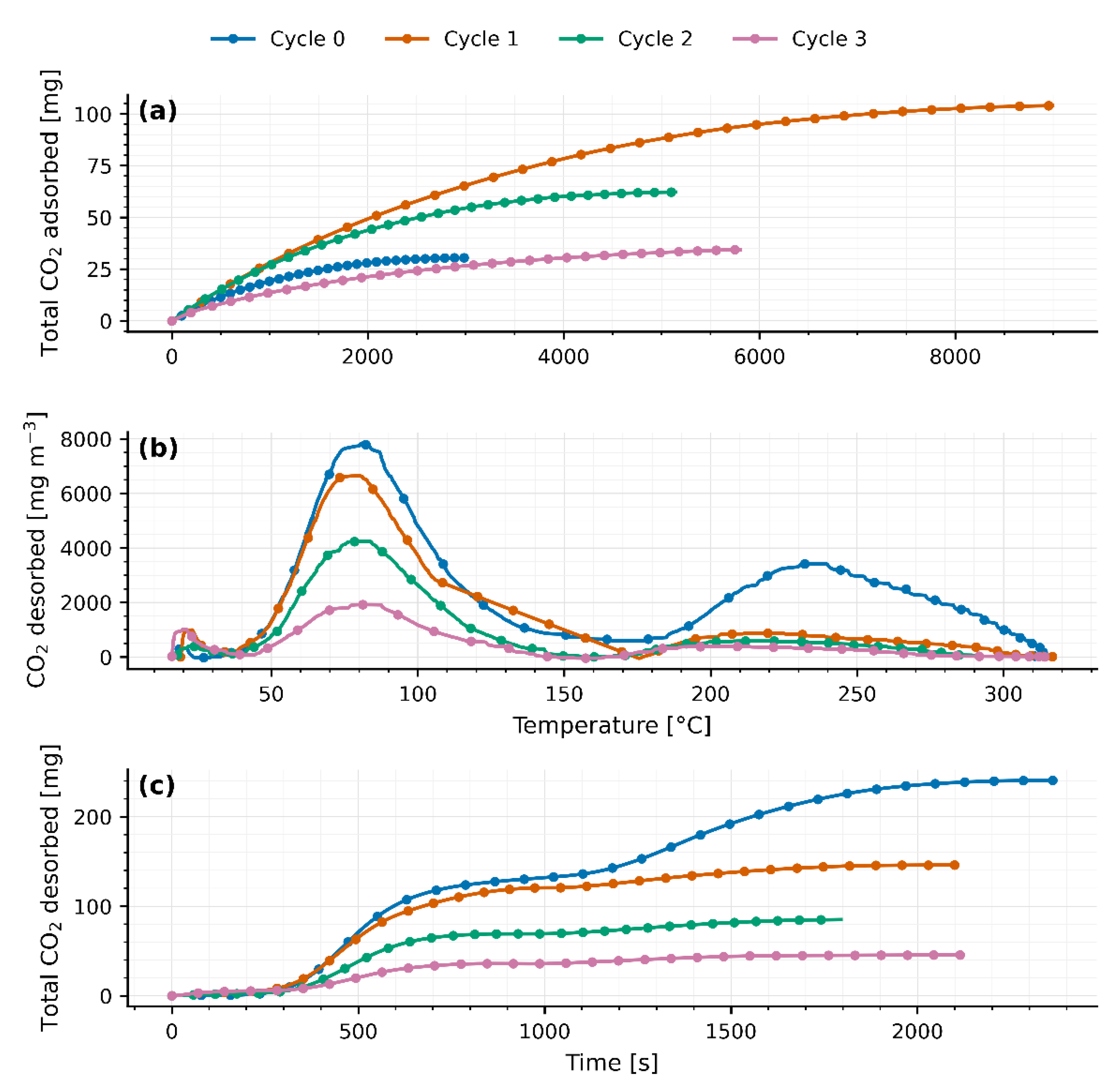
Four TSA cycles were carried out on separate days in a fixed-bed reactor packed with 40 g of JLPM3, 13X and 4A molecular sieves. The first adsorption cycle (cycle 0) served as a pretreatment to activate the zeolites. Adsorption was performed under ambient conditions, while desorption was induced by a temperature swing from ambient () up to 310–315 °C. The packed beds had a radius () of 90 mm and lengths () of 0.8 cm, 0.8 cm, and 0.7 cm, respectively.
Four TSA cycles were performed on separate days with 40 g beds of JLPM3, 13X, and 4A using humid ambient air as feed (
Section 2.1). Adsorption proceeded at ambient conditions; desorption used a temperature swing to 310–315 °C. The corresponding breakthrough and temperature-programmed desorption (TPD) profiles are shown in
Figure 3,
Figure 4 and
Figure 5, and cycle-wise uptakes in
Table 1,
Table 2 and
Table 3.
Typically, zeolites are considered physical adsorbents, but formation of chemisorbed species (like carbonates or bicarbonates) on zeolite cation sites is known under certain conditions [
7]. For example,
Amrit Kumar et al. (2015) observed that some physisorbents can bind CO
2 strongly enough to require elevated temperatures for release, hinting at chemisorptive interactions in MOFs/zeolites during DAC [
15]. For instance
: “Similar dual desorption behavior has been reported for CO2 on Na-X zeolites, where a low-temperature desorption corresponds to weak physisorption and a high-temperature feature is attributed to CO2 binding as bicarbonate/carbonate on cation sites” [
7]. In all three sieves, the CO₂ TPD curves show a reproducible bi-modal shape: a low-temperature peak (≈75–82 °C) followed by a broader peak at ≈214–220 °C (
Figure 3b,
Figure 4b and
Figure 5b). We attribute the first to readily reversible physisorption in micropores and the second to stronger binding (e.g., bicarbonate/carbonate-like species on cationic/defect sites) that requires higher temperature to desorb, consistent with reports on FAU/LTA zeolites that separate weakly vs. strongly bound CO₂ in TPD features [
6,
7]. A fraction of stronger sites can also be introduced or amplified by composition/defects or metal modification (e.g., Fe@13X), which shifts part of the desorption to higher T [
14].
As heat is applied from ambient up to 310–315 °C, a first peak appears between 75 °C and 82 °C (77.9 ± 1.2 °C for JLPM3; 75.3 ± 2.8 °C for 13X; 81.9 ± 0.6 °C for 4A), signaling the release of weakly adsorbed CO₂ (physisorption or surface interactions). Following this peak, the CO₂ concentration falls sharply (flush-out of residual gas) until roughly 156–168 °C. Then, a second, broader peak emerges at 214–220 °C (214.1 ± 7.3 °C for JLPM3; 216.6 ± 4.5 °C for 13X; 219.6 ± 5.1 °C for 4A), attributed to the desorption of more strongly bound CO₂ (chemisorption or internal zeolite interactions). A small, marginal rise near 300 °C is observed in all cases, likely due to combustion of trace organics rather than continued desorption.
Integrating the breakthrough curves, the initial CO₂ uptakes rank JLPM3 > 13X > 4A (
Table 1,
Table 2 and
Table 3). JLPM3 shows the steepest front, indicative of faster kinetics, consistent with its higher porosity and smaller mesopores that shorten diffusion paths to CO₂-selective micropores (
Section 4). Recent studies confirm that appropriately engineered zeolites can capture CO₂ at air-level concentrations with fast kinetics and modest heats of adsorption, particularly MOR-type frameworks under dry or controlled-humidity conditions [
16]. These results align with our ranking (JLPM3 ≥ 13X ≫ 4A) and the observed sensitivity to moisture. All three materials, however, display substantial co-adsorption of H₂O; the measured H₂O/CO₂ selectivity increases across cycles (
Figure 3,
Figure 4 and
Figure 5), reflecting the strong hydrophilicity of low-Si/Al zeolites and site blocking by water under ambient humidity [
6,
11]. The particularly low CO₂ capacity of 4A is consistent with its small LTA windows that limit CO₂ access and its strong affinity for H₂O [
6,
11,20]. Notably, while competitive adsorption by water is generally dominant in hydrophilic zeolites, specific K-MER sites have been shown to allow CO₂ to displace pre-adsorbed H₂O at 30 °C, highlighting framework/cation site effects under humid DAC conditions [
17].
Quantitatively, the fraction of CO₂ released via the first mechanism (“low temperature” peak) varies by sorbent and cycle. For JLPM3 it remains around 80 % across cycles (with no statistically significant change) whereas 13X rises from 43 % in cycle 0 (preloaded and burning off organics) to about 85 % thereafter, and 4A from 55 % up to ~80–82 %. Weight measurements (
Table 1,
Table 2 and
Table 3) confirm a large drop after the first cycle (40.01 g → 37.49 g for 13X; 40.01 g → 37.85 g for 4A), then only minor losses in subsequent cycles, corroborating removal of pre-existing CO₂/organics and stabilization of the material. Across all sorbents, after pre-treatment (after cycle 0), adsorption/desorption capacities (
and
match within margin of error each cycle, aside from cycle 0, but this is because during the desorption in cycle 0 there was still CO
2 adsorbed in the zeolites and also part of the excess of CO
2 comes from organic matter being burnt, demonstrating full reversibility once pre-treatment effects are accounted for.
Except for the first cycle, the integrated CO₂ desorbed () matches the CO₂ adsorbed () within the combined experimental uncertainty of the flow, baseline subtraction, and analyzer calibration. The apparent > observed in cycle 0 arises from (i) pre-loaded CO₂ in the as-received zeolites (CO₂ weakly bound and/or bicarbonate/carbonate species accumulated) that is released upon the first heat-up, and (ii) oxidation of trace organics on the pellets/binder, evidenced by the small shoulder near ~300 °C, which contributes additional CO₂ not associated with the immediately preceding adsorption step. After this conditioning cycle, baseline-corrected desorption profiles close the CO₂ mass balance in every cycle, confirming reversible uptake once pre-treatment effects are removed.
Despite this reversibility, total CO₂ uptake and release steadily decline with each TSA cycle (
Figure 3a/3c, 4a/4c, 5a/5c), indicative of thermal degradation at 310–315 °C. This is consistent with thermally induced degradation of hydrophilic zeolites under repeated TSA exposure in humid feeds [
10]. As high-quality CO₂ sites/accessibility decline, the H₂O/CO₂ selectivity rises because polar H₂O increasingly dominates competitive adsorption [
6,
11]. JLPM3 shows the greatest performance drop and physical fragmentation, producing dust, whereas 13X retains its structure best, and 4A lies in between. By cycle 3, JLPM3 and 13X converge to similar uptake levels, underscoring how JLPM3’s structural breakdown drives its loss. When comparing all three sorbents (
Figure 6), JLPM3 initially delivers the highest CO₂ uptake, followed by 13X and then 4A, but all three diminish over cycles. Water uptake likewise converges, and the H₂O/CO₂ selectivity ratio increases cycle-by-cycle: as the sorbents’ electromagnetic adsorption strength weakens (affecting non-polar CO₂ more than polar H₂O), given that they preferentially capture water. These trends highlight that while the dual-peak desorption signature and reversible adsorption mechanisms persist, high-temperature cycling inexorably erodes sorbent capacity and selectivity, especially for JLPM3, suggesting a trade-off between initial performance and thermal durability.
The humidity penalty observed here and in literature [
11,
16,
17,
18] underscores the need for water management (e.g., layered beds or pre-drying) to enable moderate-temperature TSA (≈100–120 °C) with zeolites [
11]. Framework topology and extra-framework cations critically tune DAC performance at 400 ppm, especially in LTA/4A where cation charge/size governs low-ppm uptake and moisture sensitivity [
18]. These comparisons support our mechanistic interpretation and guide process/material strategies to mitigate water competition and thermal stress in DAC cycles.
3.2. Comparison of Molecular Sieves JLPM3, 13X and 4A (No-Pretreatment)
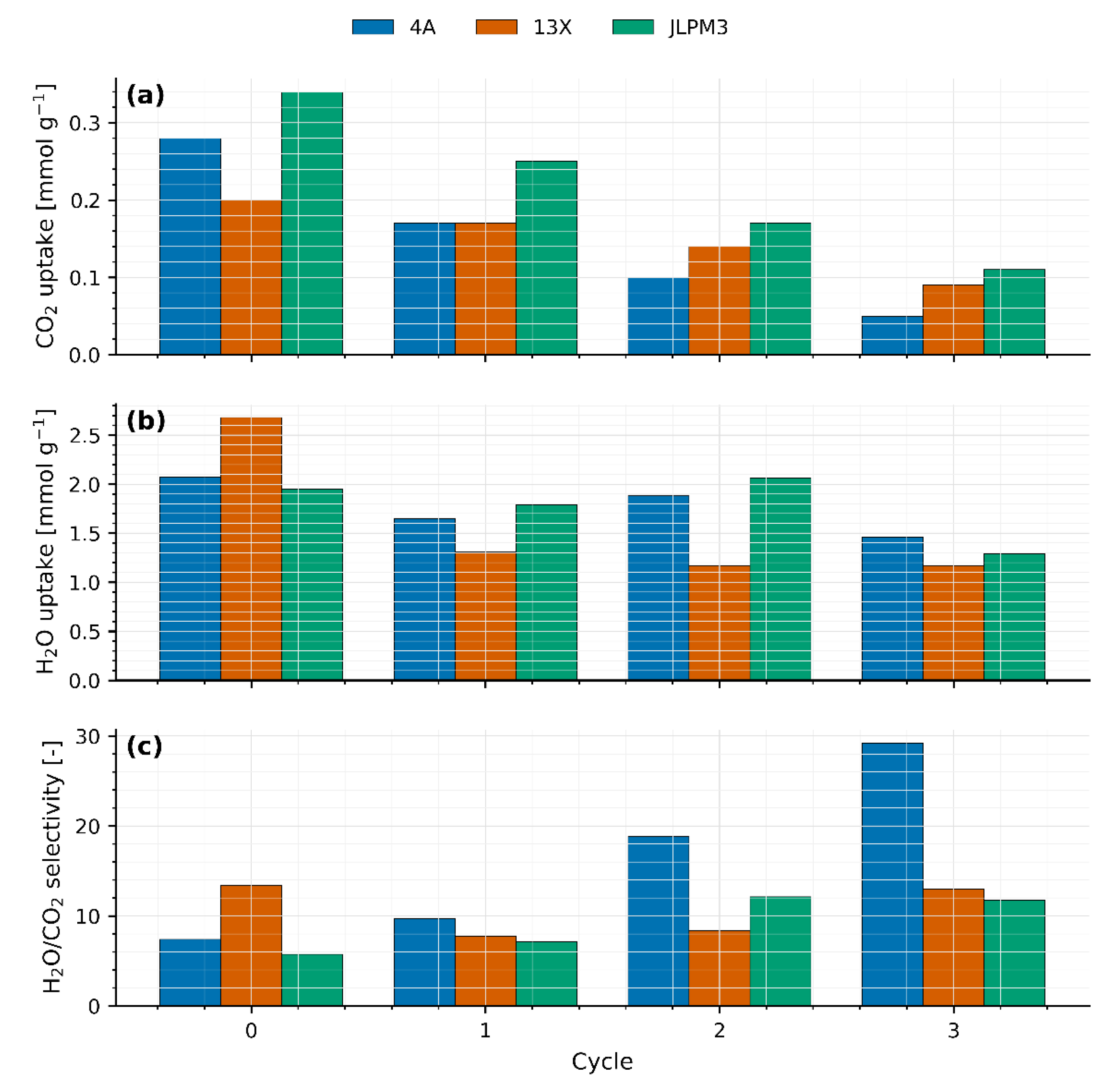
Figure 6 consolidates the cycle-wise CO₂ uptake, H₂O uptake, and H₂O/CO₂ selectivity for the three sieves (see
Table 1,
Table 2 and
Table 3 for values and uncertainties). Trends are discussed in terms of framework topology, extra-framework cations, and competitive adsorption with water, which dominate DAC performance at ~400 ppm CO₂.
CO₂ uptake (
Figure 6a): In the first cycle, the ranking is JLPM3 > 13X > 4A. JLPM3’s advantage is consistent with its higher accessible porosity and shorter diffusion paths to CO₂-selective micropores (
Section 4), which sharpen the breakthrough front and improve bed utilization. 13X (FAU) provides large pore windows (~0.74 nm) and a high density of Na⁺ sites that polarize CO₂ effectively, explaining its robust, though lower, uptake. By contrast, 4A (LTA) has ~0.40–0.42 nm apertures that restrict CO₂ access and are readily occupied by H₂O under ambient humidity, depressing its effective capacity [
6,
11,
16]. These observations align with DAC-specific studies showing zeolites can capture air-level CO₂ with fast kinetics under dry/controlled-humidity feeds (e.g., MOR- and FAU-type) and that framework/cation pairing strongly governs low-ppm performance [
6,
12,
16].
H₂O uptake (
Figure 6b): All three sieves co-adsorb substantial water, with 4A and 13X exhibiting the strongest moisture loading, consistent with their low Si/Al and hydrophilicity [
6,
11]. Preferential H₂O occupation of cationic sites reduces the number of electrostatically favorable CO₂ sites and lengthens mass-transfer paths, explaining earlier CO₂ breakthrough and lower integrated uptake relative to dry conditions [
6,
11]. While water typically “wins” on hydrophilic zeolites, site-specific exceptions exist (e.g., K-MER), where CO₂ can displace pre-adsorbed H₂O at 30 °C; this underscores that cation identity and local ring geometry can flip CO₂/H₂O selectivity under certain conditions [
17].
H
2O/CO
2 (
Figure 6c) increases with cycle number for all sieves. Two effects compound: (i) progressive loss of high-quality CO₂ sites and/or pore accessibility due to thermal/mechanical stress at 310–315 °C in humid feeds, well documented for 4A/13X-class materials [
10], and (ii) H₂O “hogging” of the remaining cationic sites as the CO₂-affine population shrinks [
6,
11]. The net result is a rising water fraction in the working capacity even as both absolute uptakes decline.
The monotonic decrease in CO₂ capacity across cycles, the visible fragmentation in JLPM3, and the relative robustness of 13X are consistent with literature on thermo-humid cyclic degradation of hydrophilic zeolites [
10]. By the last cycle, JLPM3 and 13X converge to similar CO₂ uptakes, implicating mechanical attrition and/or partial amorphization in JLPM3 as the cause of its steeper decline (see
Section 4). 4A remains transport-limited and water-poisoned throughout, consistent with LTA access constraints and strong cation–H₂O interactions [
6,
11,
16]. The pronounced cation sensitivity of LTA-type zeolites at 400 ppm reported elsewhere further explains 4A’s low working capacity under humid air [
18].
Mechanistic link to desorption profiles. The bi-modal CO₂-TPD response discussed in
Section 3.1, low-T, readily reversible physisorption followed by a higher-T contribution from stronger binding on cationic/defect sites, rationalizes the selective loss of CO₂ capacity under harsh cycling. Introducing or exposing a stronger-site fraction (e.g., via composition/defects or metal incorporation) shifts part of the desorption to higher temperature, as seen for Fe-modified FAU (Fe@13X) [
14].
These results point to two levers for DAC with zeolites: (1) Water management, e.g., upstream drying or layered beds with a desiccant pre-layer [
11]. (2) Framework/cation tuning, especially in LTA/FAU, controls the CO₂/H₂O balance at 400 ppm; cations with appropriate charge density and placement can improve low-ppm CO₂ uptake yet should avoid excessive water affinity [
6,
16,
18]. These principles are consistent with the stronger initial performance of FAU-type materials here and with recent demonstrations of low-ppm CO₂ capture on optimized zeolites [
12,
16].
3.3. Effect of Prolonged Thermal Conditioning (12 Days at 100 ºC) on CO2 Uptake and Mechanism
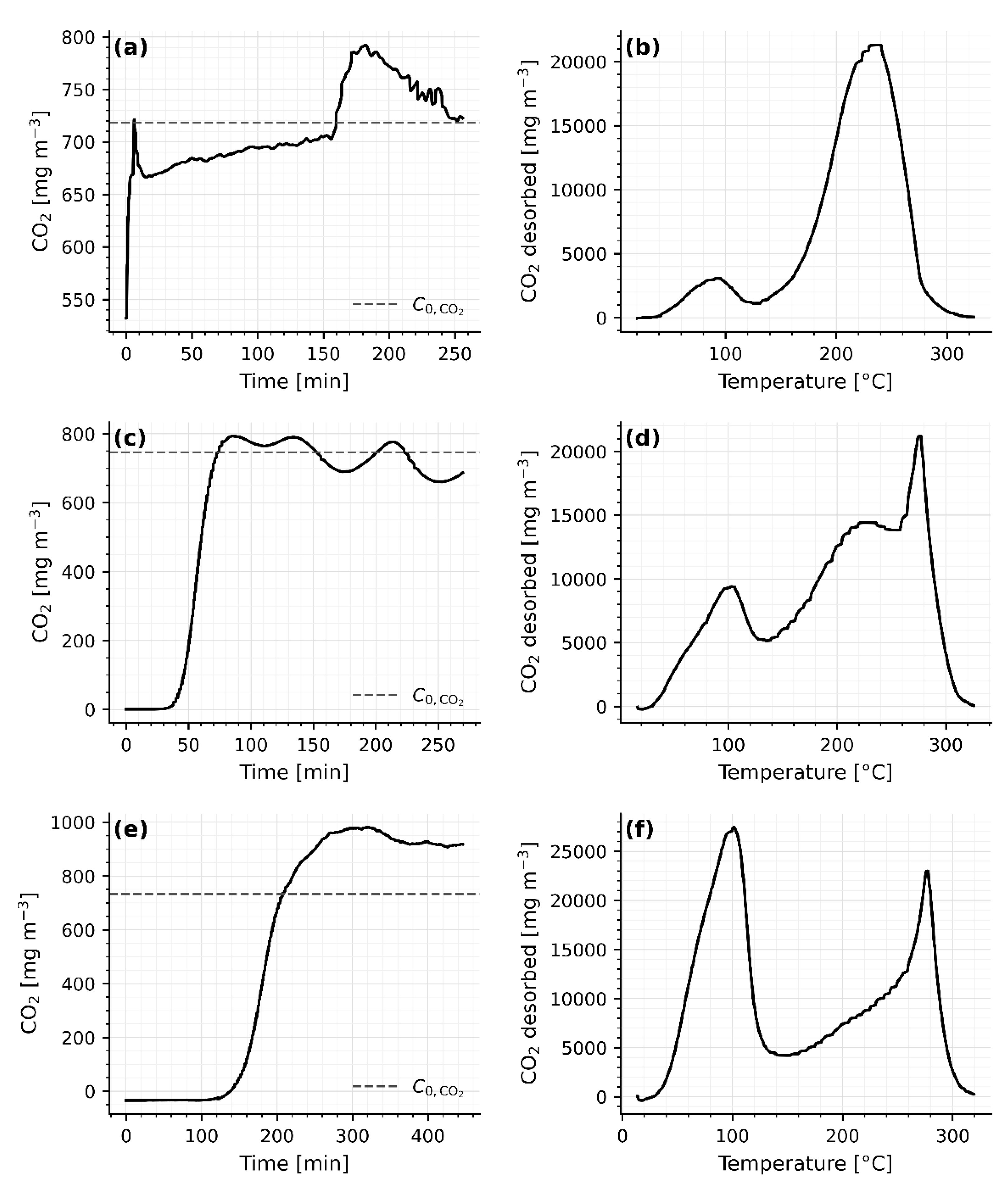
A 12-day conditioning at 100 °C was applied to 200 g beds of JLPM3, 13X, and 4A to emulate cumulative desorption exposure and obtain cycle-stabilized behavior. After this treatment, one TSA cycle was performed under humid ambient air. The results (
Figure 7a–f;
Table 4) show (i) similar total CO₂ uptake for JLPM3 and 13X relative to their first cycles in section 3.1, (ii) a redistribution of CO₂ between weak and strong binding (a larger high-T desorption contribution), and (iii) persistent H₂O co-adsorption that continues to limit working capacity under humidity.
From
Figure 7.b, 7.d and 7.f, the desorption profiles of the three molecular sieves as a function of temperature exhibit a distinct pattern: an initial peak (
), followed by a relative minimum (
), and finally, another peak (
). These temperature points, detailed in
Table 4, correspond to different desorption mechanisms. At
, the zeolites release all CO
2 stored via physisorption. The intermediate point
marks the onset of desorption for CO
2 bound by stronger interactions, while
indicates the complete release of all stored CO
2.
In the pre-treated 200 g beds, the CO₂-TPD traces display a low-temperature release centered near 95–105 °C (JLPM3 103.1 °C; 13X 103.7 °C; 4A 92.3 °C), followed by an intermediate minimum (~137–153 °C) and a pronounced high-temperature tail/peak extending to ~235–275 °C (
Table 4;
Figure 7b,d,f). We attribute the first feature to readily reversible physisorption in micropores and the higher-T release to stronger binding (bicarbonate/carbonate-like species on cationic/defect sites) that requires additional thermal input, consistent with established assignments for FAU/LTA zeolites and other physisorbents showing dual-mode CO₂ retention [
6,
7,
15]. The growth of the high-T fraction after conditioning indicates that mild, extended heating can activate or expose stronger sites (e.g., by cation re-distribution or defect formation) [
6,
15], a trend aligned with observations that metal-modified FAU (e.g., Fe@13X) holds a portion of CO₂ more strongly and desorbs at higher T [
14].
The total CO
2 and H
2O uptakes in
Table 4 were calculated using the post-desorption weights of the molecular sieves, representing the fully regenerated samples.
Table 4 shows that the weight of the molecular sieves after desorption is significantly lower than before adsorption, confirming the presence of CO
2 and H
2O stored in the materials even after 12 days at 100°C. Post-conditioning CO₂ uptakes are 0.24 ± 0.01 mmol g⁻¹ (JLPM3), 0.20 ± 0.01 mmol g⁻¹ (13X), 0.14 ± 0.01 mmol g⁻¹ (4A) (
Table 4), preserving the rank JLPM3 > 13X > 4A seen in 40 g tests. This hierarchy matches expectations from framework access and micropore volume (FAU ≫ LTA) and the kinetic advantage of hierarchical/mesoporous architectures [
6,
12,
16]. Nevertheless, H₂O uptake remains substantial (2.51, 2.14, and 1.89 mmol g⁻¹ for JLPM3, 13X, and 4A), keeping H₂O/CO₂ selectivity ≈ 10–14 (
Table 4). This confirms that under humid feeds, water preferentially occupies cationic sites, depressing CO₂ working capacity unless moisture is managed [
6,
9,
11]. While water usually predominates on hydrophilic, low-Si/Al zeolites, site-specific motifs (e.g., K-MER) can allow CO₂ to displace pre-adsorbed H₂O at 30 °C, underscoring the role of cation identity and ring geometry in tuning competition [
17].
Figure 7.a,c,e provide insights into the adsorption behavior after extended pre-conditioning. For JLPM3 (
Figure 7.a), the CO
2 concentration surpasses the inlet concentration (
) before stabilizing at a higher level. This phenomenon may result from the heat released during adsorption, which partially desorbs CO
2, and from competition between H
2O and CO
2 for adsorption sites due to JLPM3's strong electromagnetic interactions. For 13X (
Figure 7.c), the CO
2 concentration initially exceeds
before oscillating around this value, suggesting lower heat being emitted during adsorption and fast cooling of the sorbent, which gives a rapid equilibration. In contrast, the behavior of 4A (
Figure 7.e) reflects a significant loss of adsorption capacity following thermal pre-treatment, with a markedly anomalous adsorption curve compared to JLPM3, 13X, and the results from
Section 3.1 for the 40g packed bed of molecular sieves 4A.
Prolonged exposure at 100 °C under humid air likely dehydrates residual hydroxyls, drives limited cation migration, and stabilizes defect-adjacent sites, modestly increasing the population of stronger CO₂-binding sites while slightly reducing purely physisorptive capacity [
6,
7,
15]. This dual effect explains the larger high-T desorption contribution in
Figure 7, and the observation that JLPM3 and 13X have similar capacities relative to their unconditioned cycle 1 (section 3.1), whereas 4A remains transport-limited by LTA apertures under humid air [
6,
11,
16,
18]. The trend is consistent with literature where framework/cation tuning at DAC concentrations adjusts the balance between weak and strong binding, particularly for LTA/FAU families [
6,
18,19].