1. Introduction
After several decades of global efforts, it is becoming obvious that renewable sources of energy are not capable of meeting modern energy needs. Their share in the global energy is unlikely to significantly exceed 5–6%. For quite a long period, the global energy will be dominated by fossil hydrocarbons, the shortage of which will not threaten the world in the foreseeable future. Huge unconventional natural gas resources are capable of providing global energy up to the advent of thermonuclear energy [
1].
Limited oil resources and the lower price of natural gas make it more and more attractive not only as a fuel, but also as a petrochemical feedstock. But to use the vast unconventional resources of natural gas, cost-effective technologies for its chemical processing are needed. The conversion of methane into thermodynamically less stable petrochemicals requires complex multistage processes and a lot of energy. The basis of most processes of modern large–tonnage gas chemistry is the preliminary conversion of natural gas into syngas [
2,
3,
4]. Due to the complexity and high energy consumption for converting natural gas into syngas, which accounts for up to 60% of all costs for obtaining basic petrochemicals [
5], specific capital investments in gas chemistry several times higher than in petrochemistry, which is a serious barrier to its more intensive development.
To overcome this barrier, various alternative approaches to the conversion of natural gas into syngas have recently been proposed. Some of them use various modifications of the traditional catalytic approach. For example, the use of ion-permeable membranes made of non–porous multicomponent metal oxides (Ion Transport Membrane, ITM) allows oxygen to be transported to the reactor directly from the air, thereby eliminating the complex and expensive process of its preliminary separation from nitrogen [
6,
7]. The Microprocessing Technology (MPT) developed by Velocys Inc. [
8] and CompactGTL [
9] dramatically increases the intensity of heat transfer in a catalytic MPT reformer consisting of a large number of successive layers of microchannels less than 1 mm in size, and thereby dramatically reduces the weight and size characteristics of the reformer, its cost and increases the efficiency of using the catalyst.
There is a growing interest in non-catalytic processes, the main advantages of which are relative technological simplicity, higher specific capacity and significantly less rigid requirements to the composition and preparation of the feedstock used. Unlike catalytic processes carried out at temperatures, as a rule, no higher than 1200 K [
4,
10,
11], in non-catalytic processes it is necessary to maintain a temperature of 1400–1800 K, at which gas phase processes of partial oxidation, steam and carbon dioxide reforming of hydrocarbons proceed at a rate acceptable for real technological processes [
12].
Such processes as Partial Oxidation (POX) [
13,
14] and Autothermal Reforming (ATR) [
3,
15] have long been used as industrial technologies. ATR accounts for a significant part of the newly constructed large-capacity facilities. However, to ensure a high yield of syngas during the non-catalytic partial oxidation of methane, the main component of natural gas, by reaction
which has too low thermal effect, requires additional heat supply to expand the rich limit of its oxidation (combustion). Approximation to the desired oxygen excess coefficient α = [O
2]/2[CH
4] = 0.25, which is 4 times lower than during complete oxidation of methane
can be achieved either by preheating the reagents, increasing their pressure, using oxygen instead of air, or a combination of these methods. POX and ATR industrial processes usually use a combination of these methods, which makes them very complex and costly, and their practical application is justified only at very large production scales. Therefore, the search continues for simpler ways to convert rich mixtures of methane into syngas, having an oxygen excess coefficient as close as possible to α = 0.25.
The most obvious and easily implemented method of expanding the rich oxidation (combustion) limit of methane is preheating of reagents. However, the use of external heaters or heat exchangers for this purpose significantly complicates technology, increases its cost and the specific energy consumption for processing. Since natural gas itself is the cheapest and most affordable source of energy for gas-chemical processes, autothermal processes, in which, along with the target reaction (1) of partial oxidation of methane into syngas, some part of it undergoes complete oxidation in parallel reaction (2), thereby providing the necessary heat, are most attractive for gas chemistry.
A classic example of such processes is the production of acetylene by partial oxidation of natural gas [
16,
17]. However, even the theoretical yield of acetylene in this process only slightly exceeds 30% [
16]. This value is too low to make this process predominant in large-scale gas chemical conversion of natural gas. The possibility of joint production of acetylene and syngas [
18], two basic products that can be obtained directly from methane with the highest selectivity, is considered as one of the possibilities for increasing this value.
The same type of process can be attributed the oxidation of hydrocarbons at short-time catalysis, which occurs at their millisecond contact time with a noble metal catalyst [
19,
20,
21]. In these processes, the role of the catalyst is reduced to providing autothermal heating of the reagents due to the complete oxidation of part of hydrocarbons upon contact with it, while the subsequent conversion of their remaining part takes place already in the gas phase due to the high temperature reached.
Another group are processes in which fresh reagents are heated by transferring heat from hot products directly in the reactor in one way or another. One of such possibilities is to carry out the conversion in a reactor with a porous medium that provides heat transfer towards the flow of reagents [
22,
23], or its implementation during filtration combustion under conditions of filtration of the reacting gas mixture through an inert porous medium acting as a heat carrier [
24,
25]. Under certain conditions, the so-called superadiabatic combustion can be realized, in which the temperature of the products can exceed the adiabatic temperature of their oxidative conversion [
26,
27].
A separate group of non-catalytic processes is the production of syngas based on various energy-chemical technologies, for example, gas piston engines [
28,
29,
30], gas turbines [
31,
32] or combustion chambers of jet engines [
33,
34]. They usually combine various factors contributing to the expansion of the rich combustion limit and stabilization of the conversion of rich mixtures, including preheating of the mixture, its turbulization, and high pressure.
However, despite significant progress in the creation of large-scale natural gas conversion processes based on autothermal reforming [
35] and active attempts to involve carbon dioxide as an oxidizer in the conversion process (for example, tri-reforming [
36]), none of these alternative methods has so far led to the creation of industrial technologies. Therefore, the problem of effective conversion of natural gas into syngas remains open.
2. Matrix Reforming of Hydrocarbon Gases: Principles and Some Examples of the Implementation
A fundamentally new promising approach to the conversion of natural gas into syngas can be matrix reforming – gas–phase partial oxidation of hydrocarbons with internal heat recuperation from heated conversion products back into the stream of fresh reagents directly during the conversion [
37]. This is one of the variants of the implementation of the so-called "superadiabatic" combustion, in which, due to the heat recuperation of the products, the temperature in the flame front can exceed the adiabatic burning temperature of a mixture of this composition, another example of which is filtration combustion [
26,
27].
In matrix reforming, recuperation of heat is realized by organizing flameless surface combustion, in which the combustion front is stabilized at a distance of 1–2 mm from the surface of a matrix made of heat-resistant material permeable to a mixture of hydrocarbon gas and oxidizer. Such materials can be perforated ceramics, foam metals, metal felt or pressed metal wire. Recuperation of heat occurs due to intensive convective and (in the presence of a screen or in a geometrically closed matrix) radiative heat transfer between the flame front and the solid matrix. As a result, the temperature of the outgoing conversion products is reduced by 400–5000C, and the working surface of the matrix is heated to 900–10000C. Due to this, the fresh reaction mixture, passing through the matrix, heats up and enters the flame front already preheated. Preheating significantly expands the combustion limits of hydrocarbons and allows stable conversion of their very rich mixtures with an oxidizer, thereby significantly increasing the yield of syngas.
Similarly, the matrix combustion of very lean mixtures increases the completeness of their combustion, thereby increasing the efficiency of fuel use, reducing the emission of incomplete combustion products (CO, polyaromatic hydrocarbons (PAHs), soot, etc.) and (due to a decreasing in the temperature of combustion) the formation of NOx.
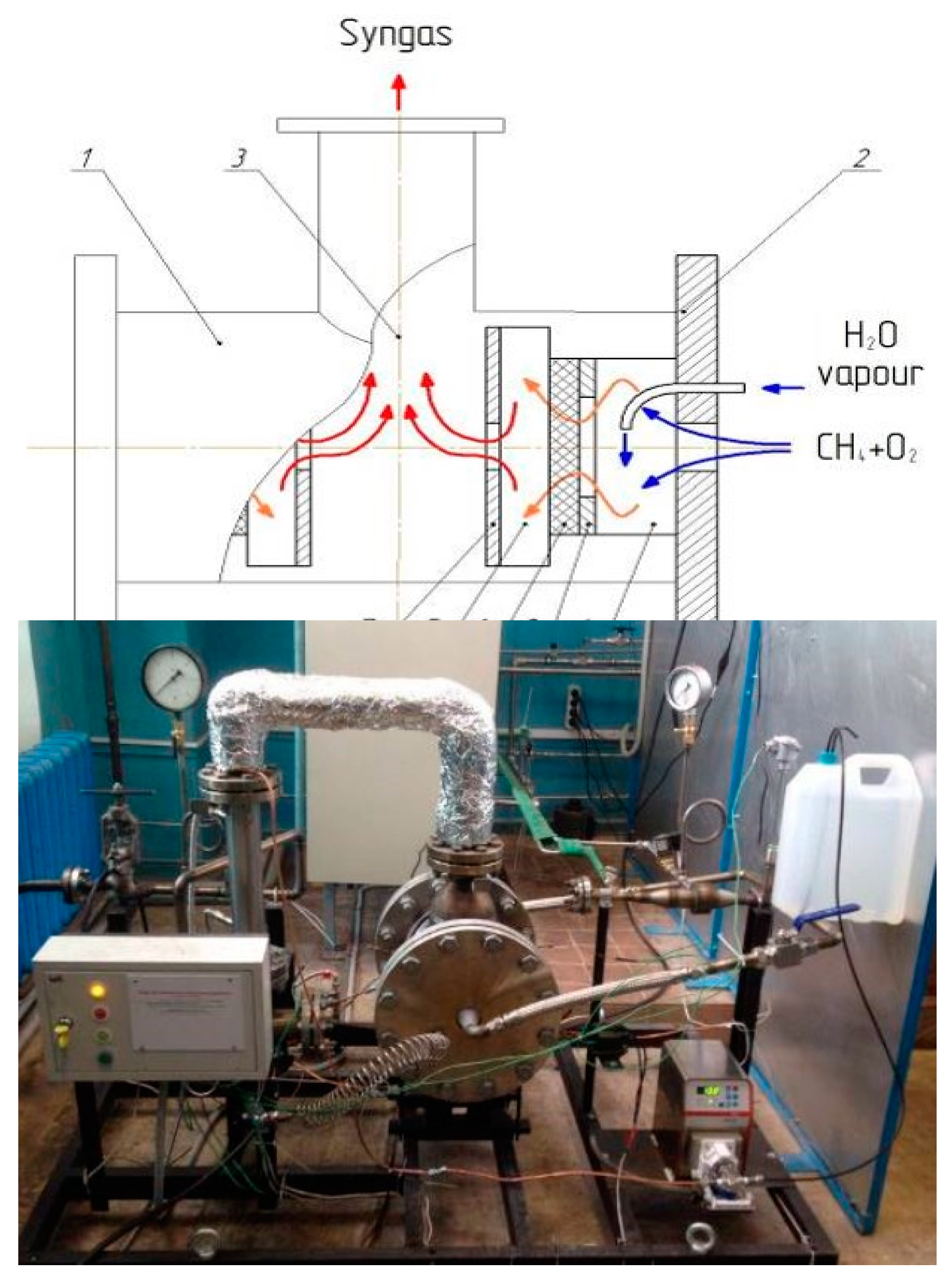
The scheme of one of the versions of the practical implementation of the matrix reformer and the demonstration unit with a gas capacity of up to 10 m
3/h is shown in
Figure 1.
Heat recuperation of conversion products allows stable conversion of very rich mixtures of methane with atmospheric air, oxygen-enriched air and oxygen at very low values of the oxygen excess coefficient
α = [O
2]/2[CH
4], up to
α = 0.30. However, the most complete conversion of methane and the maximum yield of syngas components H
2 and CO are achieved at values
α = 0.34–0.36, which are optimal for this process. At lower values of
α, the syngas yield decreases due to a decrease in methane conversion. The H
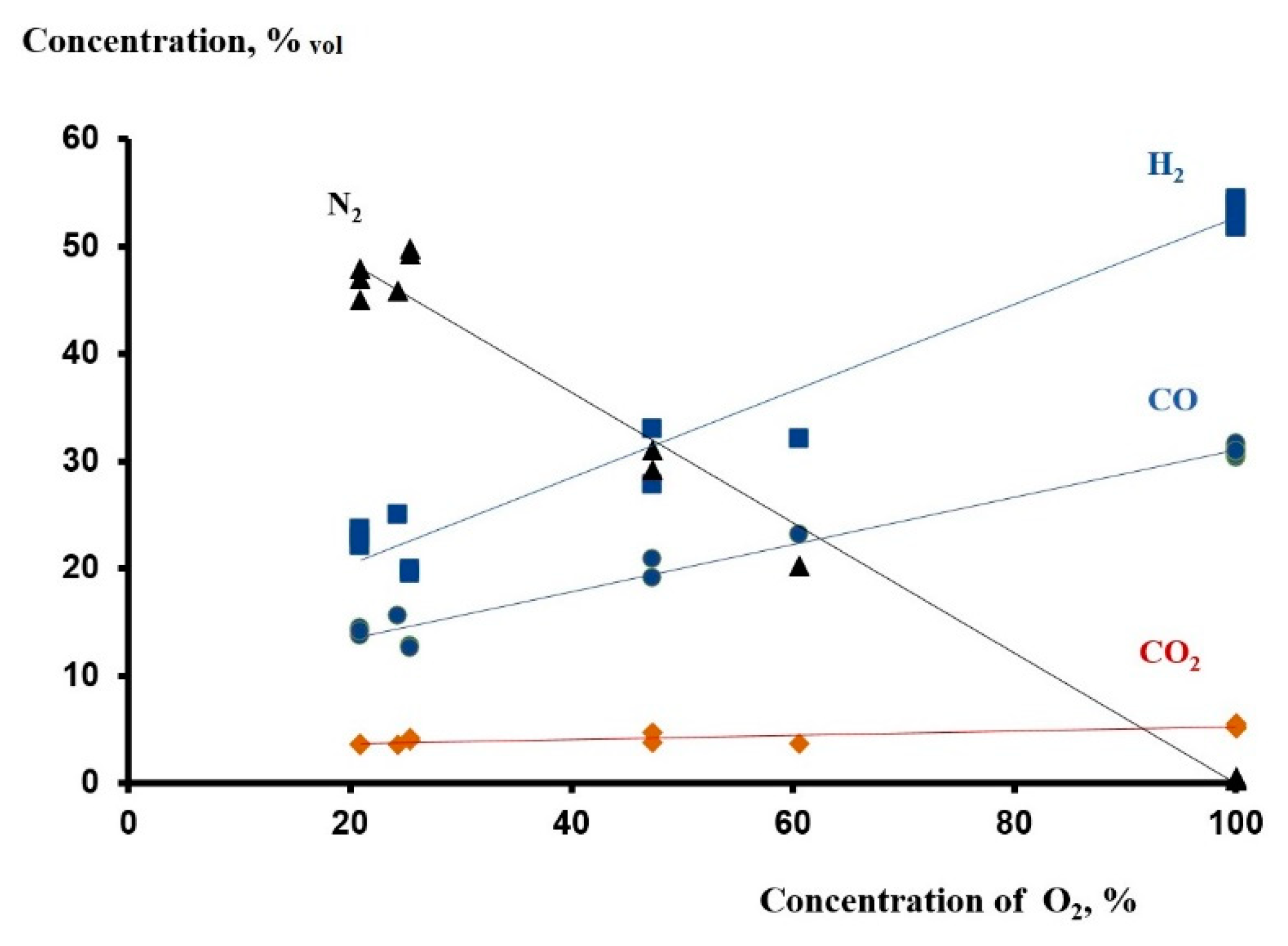
2/CO ratio, which is very important for the subsequent use of syngas, does not strongly depend on the value of α and is in the range of 1.7–1.8. The process of conversion, with the exception of the thermal regime of the matrix, practically does not depend on the presence of nitrogen, the concentration of which in the oxidizer affects only the concentration of the components of the resulting syngas (
Figure 2). When oxidized by atmospheric air, the following concentrations of syngas components are achieved: [H
2] ~23%, [CO] ~14%, [CO
2] ~ 3.6%. The conversion of both reagents is almost complete, over 95–97%. When oxidized with oxygen, similar values reach the values: [H
2] ~54%, [CO] ~31%, [CO
2] ~5.1%, oxygen conversion is complete, methane conversion is ~90%.
3. Kinetics of Matrix Conversion of Hydrocarbon Gases
The conversion of hydrocarbon gases in the matrix reformer is not limited to their rapid partial oxidation near the surface of the matrix and continues further in the post-flame zone (PFZ) of the reactor. In a series of papers [
12,
38,
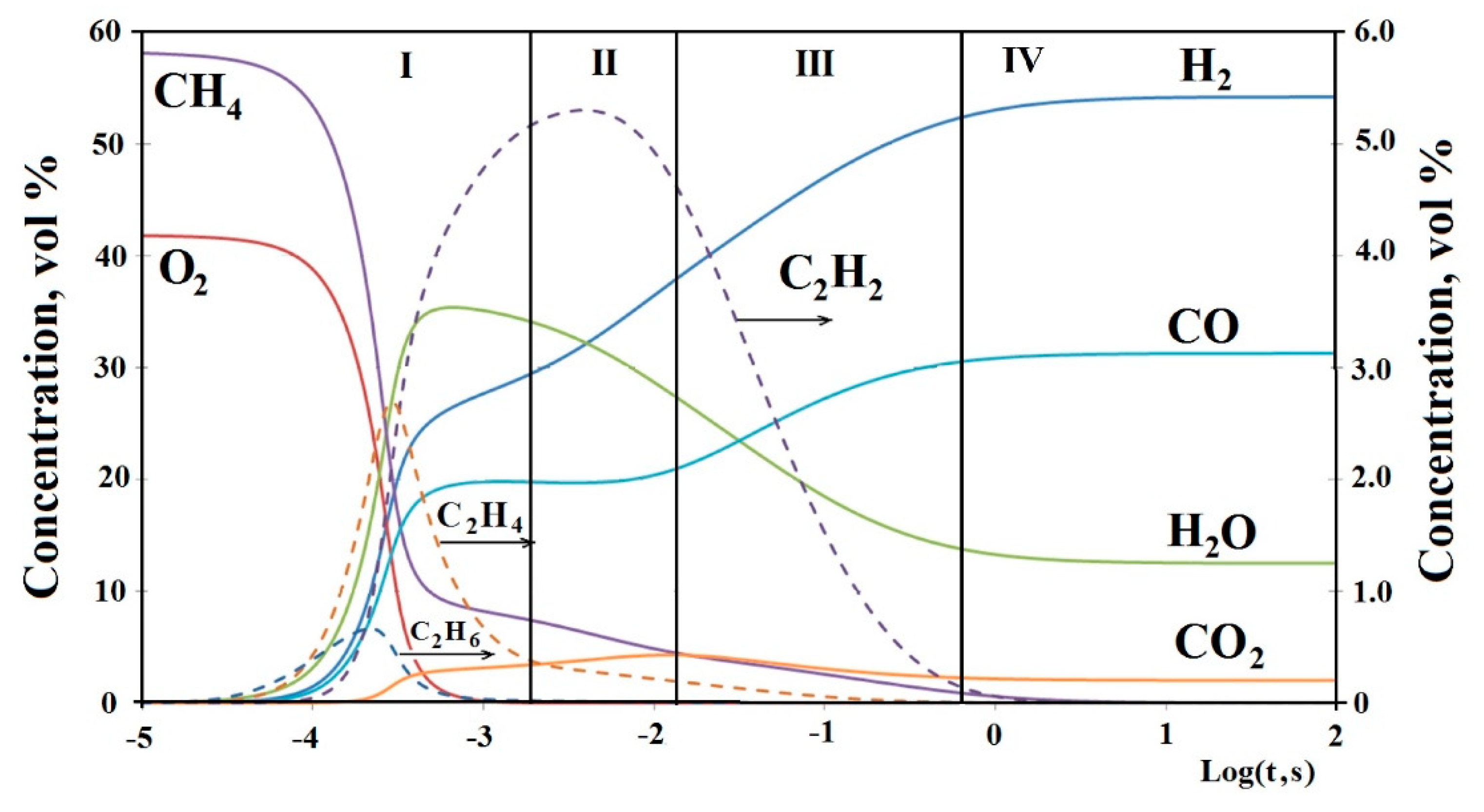
39], the following sequence of stages of matrix reforming was revealed by kinetic modeling (
Figure 3):
1. A rapid stage of partial oxidation of hydrocarbons occurring near the surface of the matrix (flame zone);
2. The stage of oxygen-free thermal pyrolysis of residual hydrocarbons, in which acetylene is the main product formed;
3. The stage of steam and carbon dioxide reforming of acetylene, at which there is a more than twofold increase in the yield of H2, and CO and a thermodynamic equilibrium is established between the components of the water gas shift reaction (WGSR) H2, H2O, CO, CO2;
4. The stage of subsequent gradual approach to the equilibrium composition of the products.
Of course, these stages cannot be clearly separated and partially overlap with each other, so the corresponding processes take place at neighboring stages. At matrix partial oxidation of rich mixtures of hydrocarbon gas and oxidizer, almost complete conversion of oxygen supplied in a significant shortage occurs in a narrow flame zone. The gas stream leaving the flame zone has a composition far from thermodynamic equilibrium, with a relatively low concentrations of H2, CO and a low H2/CO ratio. Already in the post-flame zone, in the absence of oxygen, due to the pyrolysis of unreacted hydrocarbons and the subsequent steam and carbon dioxide reforming of acetylene formed from them as a result of pyrolysis, an increase in the concentrations of H2, CO and the ratio of H2/CO occurs, as well as an approximation of the gas composition to the equilibrium composition. Therefore, the processes in the post-flame zone have a great influence on the formation of the final composition of the gas mixture and the technical parameters of the process. Kinetic modeling has shown that the most intense change in the composition of the gas mixture occurs at the initial stage of the processes in the PFZ at reaction times of 0.1–0.5 s. The temperature at which the gas mixture enters the PFZ has the most significant effect. The higher the temperature, the higher the conversion of unreacted hydrocarbons and the resulting C2H2, the higher the subsequent concentrations of H2, CO and the ratio of H2/CO, the higher the yield of H2 per mole of hydrocarbons supplied with the initial mixture. The active reforming agents in PFZ are H2O and CO2, the addition of which to the initial mixture significantly improves the performance of the process.
One of the most important results of the kinetic analysis carried out is the first established sequence of methane conversion, which is characteristic for all non-catalytic partial oxidation processes. It was found that after the completion of the oxidizing stage, at heating temperatures of the mixture
T < 1800 K, even in the presence of a significant concentration of H
2O and CO
2, pyrolysis of methane into acetylene proceeds at a much higher rate compared to its steam and carbon dioxide reforming. At the same time, the formation of a significant part of syngas components occurs already during the subsequent steam and carbon dioxide reforming of the formed acetylene. The following processes play the most important role in the post-flame zone:
In contrast to the conversion of methane, during the non–catalytic conversion of C2+ hydrocarbons, their pyrolysis initially proceeds at a high rate, and then at a lower rate oxidative processes involving O2, H2O, CO2.
Kinetic modeling has revealed the possibility of optimizing processes in the post-flame zone of the matrix reformer, including increasing the yield of syngas and optimizing its composition for subsequent catalytic processes. One of the ways to increase the yield of syngas is an additional supply of CO
2 released from products or supplied from the side, which can be considered as an effective way of its utilization [
39].
4. Main Advantages of Matrix Reforming
Let's briefly consider the main advantages of the matrix reforming of hydrocarbon gases into syngas, which make it attractive for many technological applications in which natural gas is used. First of all, it is an autothermal process that does not require external sources of heat or energy. The conversion of rich mixtures of hydrocarbon gas (methane) with an oxidizer occurs due to the fact that in parallel with the reaction (1) of its partial oxidation into syngas, some part of it undergoes complete oxidation by reaction (2), thereby providing the necessary energy to maintain the autothermal nature of the process. As already noted, autothermal processes that do not require additional heat exchangers and are not accompanied by heat losses ensure maximum efficiency of using the energy of hydrocarbon feedstock in technological processes.
The conversion takes place completely in the gas phase behind the matrix surface, which makes the process easily manageable and eliminates the problem of carbon formation, which is critical for many catalytic processes.
The absence of a catalyst not only simplifies the process, but also dramatically reduces the requirements for gas preparation and purification, making process insensitive to many impurities that are catalytic poisons, and allowing the direct use of hydrocarbon gases of almost any composition as feedstock, including associated and refinery gases, as well as low-boiling liquid hydrocarbons up to the kerosene fraction.
The use of atmospheric air, enriched air or oxygen does not change the nature and kinetics of the process and even the H2/CO ratio, affecting only the content of ballast nitrogen in the resulting syngas. The ability to use atmospheric air not only dramatically reduces the cost of the process, but also makes it safe to use directly in places of production and processing of flammable hydrocarbons.
Effective recuperation of heat of products makes it possible to convert gases with a high content of inert components (N2, CO2, H2O), including biogas, into syngas, opening up the possibility of obtaining petrochemicals from renewable bioproducts or wastes.
One of the problems of all reforming processes based on partial oxidation (POX) is the relatively low H2/CO ratio, usually not exceeding 1.8. Traditionally, this is solved by the subsequent partial catalytic steam reforming of part of the formed CO into hydrogen by the water gas shift reaction (6). The advantage of matrix reforming in this regard is that the process remains autothermal even taking into account the heat consumption for steam generation in a volume sufficient for the complete conversion the formed carbon monoxide into hydrogen.
The gas-phase nature of the matrix reforming makes it stable and easy to control, and the design of the converters is extremely simple and easily scalable. Compared with traditional catalytic steam reforming, matrix reforming has a very high specific volumetric capacity, since it proceeds at a distance of less than 1 cm compared to the length of tubes with catalyst in catalytic reformers, usually exceeding 10 m. This makes it possible to create compact low-tonnage installations with low gas capacity starting from several cubic meters per hour.
5. Current Level of Research in the Field of Matrix Reforming
Currently, several demonstration units for the matrix conversion of natural, associated, refinery gases and biogas with an inlet gas flow of up to 10 m3/h and an operating pressure of up to 2.5 MPa have been launched and tested. The tests carried out confirmed high efficiency and productivity, as well as good controllability and stability of the matrix reforming with atmospheric air, oxygen-enriched air and oxygen of various hydrocarbon gases: methane, model associated petroleum gas (APG), model refinery gas, propane-butane fraction, model biogas. Several patents have been obtained for various versions of syngas and hydrogen production processes and design of reformers.
Based on the matrix reforming by atmospheric air, a low-tonnage commercial process of APG conversion into liquid products is proposed [
40]. The use of atmospheric air instead of oxygen and the refuse of syngas recirculation in favor of a cascade of 2–3 consistent reactors dramatically simplify the process and makes its use in the oil and gas producing fields safe. The subsequent conversion of the resulting nitrogen-containing syngas into synthetic liquid hydrocarbons, methanol or dimethyl ether (DME) can be the optimal solution for using the resources of remote low-yield gas fields. Since the connection of such fields to the gas pipeline system requires too high costs, it is more economically justified to convert the produced natural gas or APG directly on site into liquid products that are more convenient for transportation. In addition, one of these products, methanol, is widely in demand in natural gas producing fields as an inhibitor of hydrate formation. The technologies used should be as simple as possible, cheap and do not require numerous highly qualified personnel to service. The matrix reforming fully meets all these requirements.
To implement such low-tonnage processes, in addition to matrix reformers using atmospheric air as an oxidizer, for the subsequent conversion of the resulting nitrogen-containing syngas into liquid products, its high conversion per pass is required in order to abandon the recycling of unreacted gas. Among the liquid products obtained from syngas, the highest degree of conversion, about 90%, is currently achieved in the synthesis of DME on combined Cu–Zn–Al-zeolite catalysts. The conversion of syngas into methanol or synthetic hydrocarbons proceeds with a lower conversion per pass and requires a cascade of two or three consecutive reactors. However, due to dilution of the syngas with nitrogen, the specific heat release in the catalytic layer is reduced, which allows the use of shell-and–tube Fischer-Tropsch synthesis reactors with an increased diameter. Thus, the capital costs for the manufacture of two reactors optimized for nitrogen-diluted syngas may not be higher than for the manufacture of one reactor operating on undiluted syngas [
41,
42].
A serious problem is the low-tonnage processing of various types of bio-products into syngas. Low energy density in agricultural or biotechnological feedstocks dictates the need for their local processing. Small-scale agricultural products or wastes processing plants can`t profitably use technologies common in large-scale petrochemistry, including the complex and expensive steam reforming of biogas into syngas for its subsequent conversion to liquid fuel [
43]. Matrix reforming allows using small-scale sources of inexpensive and easily obtained biogas and other renewable hydrocarbon gases for the production of liquid biofuel by their air reforming into low-cost nitrogen-rich syngas, followed by Fischer-Tropsch synthesis in a cascade of sequential reactors [
44]. The possibility of matrix reforming of biogas with a CO
2 concentration of up to 50% into syngas has been experimentally demonstrated.
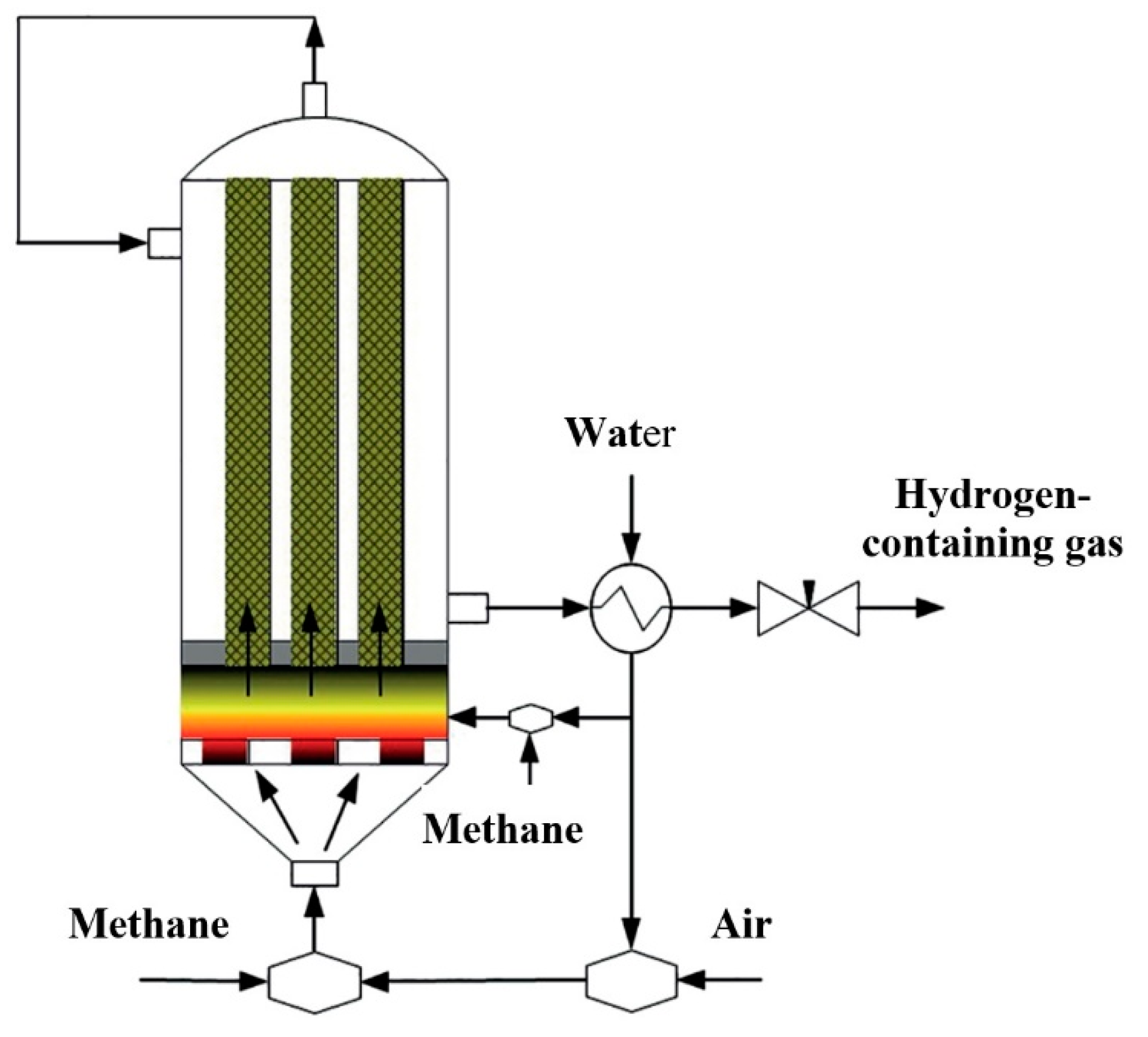
Hydrogen is becoming one of the most popular products of natural gas processing. For low-tonnage production of hydrogen-containing gas, matrix reforming should be supplemented by subsequent catalytic steam reforming of CO (WGSR) into hydrogen. Steam is generated by the heat of a hot hydrogen-containing gas (
Figure 4). Thus, the process remains completely autothermal. A comparison of such combined methane-to-hydrogen matrix reforming with the traditional steam reforming of methane is presented in
Table 1.
Figure 5 shows two pilot units for producing hydrogen-containing gas by matrix reforming of natural gas followed by catalytic steam conversion of CO to H
2. Such low-tonnage production of hydrogen, including from liquid hydrocarbons directly at hydrogen supply stations, eliminates the problems of its transportation and storage. On their basis, reducing gas for non-ferrous and ferrous metallurgy can also be supplied.
A combined autothermal process for the production of methanol and hydrogen with very low CO
2 emissions has been developed, in which the vast majority the carbon contained in CH
4 passes into methanol [
45]. The high temperature of the syngas after matrix reforming makes it possible to add an additional amount of fresh natural gas and steam to it and direct the resulting gas mixture to the catalytic stage, in which, as the temperature gradually decreases from 1223 K to 723 K, the endothermic processes of steam reforming of methane and partial steam reforming of CO to produce hydrogen sequentially proceed. The scheme of the reactor for the implementation of such a combined autothermal process is shown in
Figure 6. There is no need for a complete conversion of CO, since the remaining part of it is used in the subsequent synthesis of methanol. According to the preliminary material balance of the process, with a complete flow of incoming methane of 4,301 nm
3/h (2,826 kg/h), the yield of methanol will be 5,397 kg/h and that of hydrogen – 141 kg/h, when the CO
2 output is less than 20 kg/h.
A combination of a matrix reformer of hydrocarbon gases with solid oxide fuel cells (SOFC) has been proposed [
46]. It allows simultaneously provide SOFC with syngas and high temperature, necessary for its operation, thus eliminating the cumbersome and energy-consuming steam reforming, which also requires a water treatment system.
6. Matrix Combustion of Lean Mixtures
Matrix combustion of lean gas-air mixtures opens up interesting prospects in energy, allowing very lean mixtures to be burned, which increases the completeness of their combustion, thereby increasing fuel efficiency and reducing the emission of incomplete combustion products and the formation of NOx. A conceptual model of a low-emission matrix combustion chamber (MCC) for ground-based gas turbine engines (GTE) with record low NOx and CO emissions < 5 ppm, which also does not require a fire pipe and secondary air supply systems, has been proposed and tested [
47]. One of advantages of MCC is the replacement the turbulent flare combustion of fuel with its laminar combustion near the surface of the matrix. The uniquely low (1200–1300°C) temperature of the flame front in MCC with high combustion completeness, provided by the possibility of combustion a very lean mixtures, makes it possible to reduce the volume of environmentally problematic emissions to a level inaccessible to other known types of burners.
It can also be predicted that due to the laminar combustion mode in and lower maximum temperatures, MCC will provide higher combustion stability and, accordingly, a significantly lower level of noise and flow pulsation, which will reduce the requirements for the structural materials of the combustion chamber and increase its working life. The new type of combustion chamber makes it possible to abandon such complex and unreliable components of traditional combustion chambers of gas turbines, such as a fire pipe and its cooling system, as well as a supply system for secondary air, which will not only facilitate and simplify the design, but also further increase the service life of the combustion chamber. The new type of combustion chambers also expands the possibility of using low-quality and alternative fuels in GTE. Due to the simplification of the combustion chamber design and the absence of a fuel burn-out zone, it is possible to expect a decrease in the overall weight characteristics of the MCC compared to the combustion chambers of standard gas turbine engines.
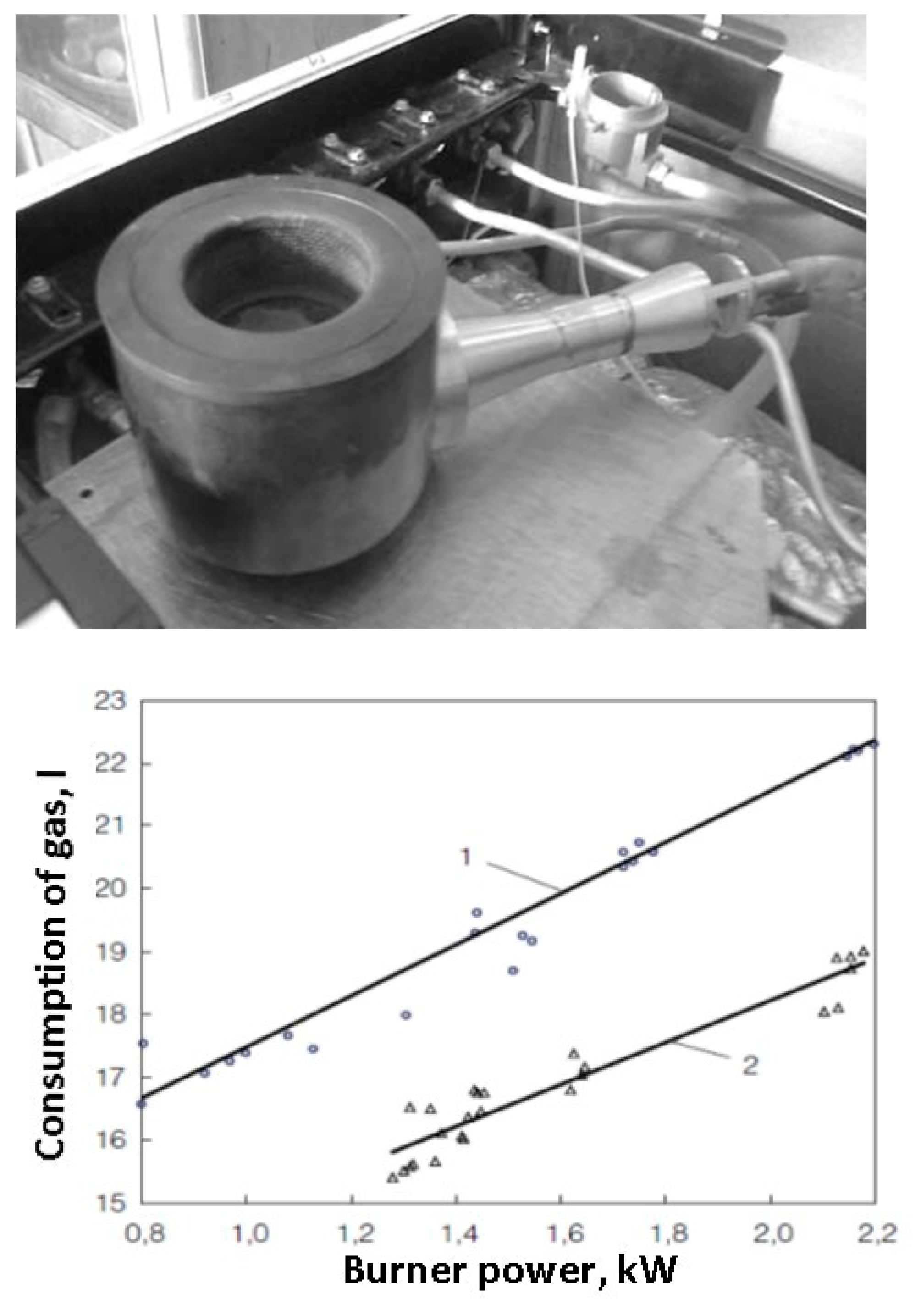
The domestic use of various types of matrix burners is also a promising direction of their utilization. Infrared burners with a volumetric matrix for household gas stoves are superior to typical burners in terms of efficiency and reduction of emissions of environmentally problematic combustion products. Models of low-emission infrared burners for household gas stoves with NOx and CO emissions less than 10 ppm have been developed, allowing for ~50% reduction in gas consumption (
Figure 7) [
48].
7. Conclusions
The energy and petrochemical industries are in urgent need of new, more efficient and more compact technologies for converting huge natural gas resources into more convenient and in-demand liquid fuels, hydrogen and other petrochemicals. Non-catalytic conversion offers great opportunities for creating such processes. One of the most promising among such processes may be non-catalytic matrix reforming, which has a number of undeniable advantages over traditional natural gas reforming and has already shown its real potential. For its further development, it is necessary to transfer from demonstration units to larger pilot plants with a gas consumption of hundreds and thousands of cubic meters per hour.
Nevertheless, there are still a number of important tasks to be solved. Conversion modes to be investigated at higher pressures and higher gas flow rates. It is also necessary to develop principles for scaling the process and to study the degradation of the matrix material in conditions of high temperature and a sufficiently aggressive environment, as well as ways to increase the resource of heat-resistant materials used for their manufacture. Based on CFD modeling of the process, it is necessary to optimize the design of the reformer and its operating modes, optimize the thermophysical properties, thickness and permeability of the matrix, increase the service life of its material, consider various options for using composite matrices, investigate the distribution of gas flows in the matrix in order to minimize heat loss through the side surface and structural elements.
However, it is already clear that matrix reforming makes it possible to solve a number of fundamental issues, for example, the creation of distributed low-tonnage hydrogen production directly at the places of its consumption, including the vehicle fueling stations. Any local liquid or gaseous hydrocarbon feedstocks, including biogas, can be used for this purpose. Such distributed low-tonnage hydrogen production can make it possible to bypass the problems of transporting and storing large volumes of hydrogen [
49,
50] that do not yet have a practical solution.
References
- Arutyunov, V.S.; Lisichkin, G.V. Energy resources of the 21st century: problems and forecasts. Can renewable energy sources replace fossil fuels? Russ. Chem. Rev. 2017, 86, 777–804. http://iopscience.iop.org/article/10.1070/RCR4723/pdf.
- Aasberg-Petersen, K.; Dybkjær, I.; Ovesen, C.V.; Schjødt, N.C.; Sehested, J.; Thomsen, S.G. Natural gas to synthesis gas – Catalysts and catalytic processes. J. Nat. Gas Sci. Eng. 2011, 3, 423–459. [CrossRef]
- Dybkjær, I.; Aasberg-Petersen, K. Synthesis Gas Technology Large-Scale Applications. Can. J. Chem. Eng. 2016, 94, 607–612. [CrossRef]
- Speight, J.G. Handbook of industrial hydrocarbon processes (Second ed.). Cambridge, MA. 2020. ISBN 9780128099230. OCLC 1129385226.
- Aasberg-Petersen, K.; Hansen, J.-H. B.; Christensen, T.S.; Dybkjaer, I.; Christensen, P.S.; Nielsen, C.S.; Madsen, S.E.L.W.; Rostrup-Nielsen J.R. Technologies for large-scale gas conversion. Applied Catalysis A: General 2001, 221, 379–387.
- Mazanets, Е.А. In Proceedings of the 6th Natural Gas Conversion Symposium. Girdwood, Alaska, 2001 // Studies in Surface Science and Catalysis. Vol. 136. 2001. Natural Gas Conversion VI / Ed. E. Iglesia, T. H. Fleish. Elsevier Science B. V. Amsterdam; London; New York; Oxford; Paris; Shannon; Tokyo. 2001.
- Chen, C.M.; Bennett, D.L.; Carolan, M.F.; Foster, E.P.; Schinski, W.L.; Taylor, D.M. ITM syngas ceramic membrane technology for synthesis gas production // Proceedings of the 7th Natural Gas Conversion Symposium, June 6–10, Dalian, China. Studies in Surface Science and Catalysis. Vol. 147. Natural Gas Conversion VII. X. Bao and Y. Xu (Editors). Elsevier. Amsterdam et al. 2004. P. 55.
- Mazanec, T.; Perry, S.; Tonkovich, L.; Wang Y. Microchannel gas-to-liquids conversion – thinking big by thinking small // Proceedings of the 7th Natural Gas Conversion Symposium, June 6–10, Dalian, China. Studies in Surface Science and Catalysis. Vol. 147. Natural Gas Conversion VII. X. Bao and Y. Xu (Editors). Elsevier. Amsterdam et al. 2004. P. 169.
- CompactGTL https://www.cbinsights.com/company/compactgtl (Accessed January 15, 2025).
- Zhang, L.; Wu, W.; Zhang, Y.; Zhou, X. Clean synthesis gas production from municipal solid waste via catalytic gasification and reforming technology. Catalysis Today. 2018, 318, 39–45. [CrossRef]
- Zhang, H.; Sun, Z.; Hu, Y.H. Steam reforming of methane: Current states of catalyst design and process upgrading. Renewable and Sustainable Energy Reviews. 2021, 149, 111330. [CrossRef]
- Savchenko, V.I.; Nikitin, A.V.; Zimin, Ya.S.; Ozerskii, A.V.; Sedov, I.V.; Arutyunov, V.S. Impact of post-flame processes on the hydrogen yield in matrix partial oxidation reformer. Chem. Eng. Res. Des. 2021, 175, 250–258. [CrossRef]
- Eastman, D.B. Synthesis gas by partial oxidation. Ind. Eng. Chem. 1956, 48, 1118–1122.
- Kuhre, C.J.; Shearer, C.J. Partial oxidation grows stronger in U. S. The Oil and Gas Journal. September 1971. Р. 85–90.
- Dahl, P.J.; Christensen, T.S.; Winter-Madsen, S.; King, S.M. Proven autothermal reforming technology for modern large-scale methanol plants. Nitrogen + Syngas. International Conference and Exibition. 2014. https://www.academia.edu/11329078/Proven_autothermal_reforming_technology_for_modern_large_scale_methanol_plants (Accessed January 2, 2025).
- Zhang, Q.; Liu, Y.; Chen, T.; Yu, X.; Wang, J.; Wang, T. Simulations of methane partial oxidation by CFD coupled with detailed chemistry at industrial operating conditions. Chem. Eng. Sci. 2016, 142, 126–136. [CrossRef]
- Chen, D.; Chen, X.; Luo, C.; Liu, Z.; Gan, L.-H. Structural design and performance evaluation of industrial-scale C2H2 reactor by partial oxidation of natural gas. Chem. Eng. J. 2021, 426, 130871. [CrossRef]
- Liu, Y.; Zhang, Q.; Wang, T. Detailed chemistry modeling of partial combustion of natural gas for coproducing acetylene and syngas. Combust. Sci. Technol. 2017, 189, 908–922. [CrossRef]
- Bodke, A.S.; Olschki, D.A.; Schmidt, L.D.; Ranzi, E. High Selectivities to Ethylene by Partial Oxidation of Ethane. Science. 1999, 285 (5428), 712–715. doi: 10.1126/science.285.5428.712.
- Schmidt, L.D. Modeling millisecond reactors // Proceedings of the 6th Natural Gas Conversion Symposium. Girdwood, Alaska, 2001. Studies in Surface Science and Catalysis. Vol. 136. 2001. Natural Gas Conversion VI /Ed. E. Iglesia, T. H. Fleish. Elsevier Science B. V. Amsterdam; London; New York; Oxford; Paris; Shannon; Tokyo. 2001.
- Basini, L.E.; Guarinoni, A. Short Contact Time Catalytic Partial Oxidation (SCT-CPO) for Synthesis Gas Processes and Olefins Production. Ind. Eng. Chem. Res. 2013, 52, 17023−17037. dx.doi.org/10.1021/ie402463m.
- Al-Hamamre, Z.; Al-Zoubi, A. The use of inert porous media based reactors for hydrogen production. Int. J. Hydrog. Energy. 2010, 35, 1971–1986. [CrossRef]
- Nourbakhsh, H.; Shahrouzi, J.R.; Ebrahimi, H.; Zamaniyan, A.; Nasr, M.R.J. Experimental and numerical study of syngas production during premixed and ultra-rich partial oxidation of methane in a porous reactor. Int. J. Hydrog. Energy. 2019, 44, 31757–31771. [CrossRef]
- Toledo, M.; Bubnovich, V.; Saveliev, A.; Kennedy, L. Hydrogen production in ultrarich combustion of hydrocarbon fuel in porous media. Int. J. Hydrogen Energy. 2009, 34, 1818–1827. DOI: 10.1016/j.ijhydene.2008.12.001.
- Toledo, M.; Utria, K.S.; Gonzalez, F.A.; Zuniga, J.P.; Saveliev, A.V. Hybrid filtration combustion of natural gas and coal. Int. J. Hydrogen Energ. 2012, 37, 6942−6948. [CrossRef]
- Dorofeenko, S.O.; Polianczyk, E.V. Conversion of hydrocarbon gases to synthesis gas in a reversed low filtration combustion reactor. Chem. Eng. J. 2016, 292, 183–189. [CrossRef]
- Dorofeenko, S.O.; Polianczyk, E.V.; Tsvetkov M.V. Toward the ultimate efficiency of methane to syngas conversion by partial oxidation: A moving bed reactor with parallel preheating of reactants. Fuel. 2024, 363, 131005. [CrossRef]
- Patent RU2136580, 1998.
- Lim, E.G.; Dames, E.E.; Cedrone, K.D.; Acocella, A.J.; Needham, T.R.; Arce, A.; Cohn, D.R.; Bromberg, L.; Cheng, W.K.; Green, W.H. The Engine Reformer: Syngas Production in an Engine for Compact Gas-to-Liquids Synthesis. Can. J. Chem. Eng. 2016, 94, 623–635. [CrossRef]
- Cho, M.; Oh, W.; Park, N.; Song, H.H. Experimental study on the effects of gas additives on uncatalyzed partial oxidation of methane in a spark-ignition engine. Int. J. Engine Research. 2024, 1–12. [CrossRef]
- Cornelissen, R.; Tober, E.; Kok, J.; van de Meer, T. Generation of synthesis gas by partial oxidation of natural gas in a gas turbine. Energy. 2006, 31, 3199–3207.
- Granovskiy, M. Integrated Coproduction of Power and Syngas from Natural Gas to Abate Greenhouse Gas Emissions without Economic Penalties. ACS Omega. 2021, 6, 16336−16342.
- Buravtsev, N.N.; Kolbanovskii, Y.A.; Rossikhin, I.V.; Bilera, I.V. Effect of the Calorific Intensity of Combustion Chamber on Production of Synthesis Gas in Partial Oxidation of Methane–Oxygen Mixtures in the Combustion Mode. Russ. J. Appl. Chem. 2018, 91, 1588–1596. [CrossRef]
- Levikhin, A.A.; Boryaev, A.A. High-temperature reactor for hydrogen production by partial oxidation of hydrocarbons. Int. J. Hydrogen Energy. 2023, 48, 28187–28204. [CrossRef]
- Aasberg-Petersen, K.; Christensen, T.S.; Dybkjaer, I.; Sehested, J.; Шstberg, M.; Coertzen, R.M.; Keyser, M. J.; Steynberg, A.P. Synthesis gas production for FT synthesis. Stud Surf. Sci. Cat. 2004, 152, 258–405. [CrossRef]
- Soleimani, S.; Lehner, M. Tri-Reforming of Methane: Thermodynamics, Operating Conditions, Reactor Technology and Efficiency Evaluation – A Review. Energies. 2022, 15, 7159. [CrossRef]
- Nikitin, A.; Ozersky, A.; Savchenko, V.; Sedov, I.; Shmelev, V.; Arutyunov, V. Matrix conversion of natural gas to syngas: the main parameters of the process and possible applications. Chem. Eng. J. 2019, 377. ISCRE 25 Special Issue: Bridging Science and Technology. 1 December 2019, article 120883. DOI: 10.1016/j.cej.2019.01.162.
- Savchenko, V.I.; Nikitin, A.V.; Sedov, I.V.; Ozerskii, A.A.; Arutyunov, V.S. The role of homogeneous steam reforming of acetylene in the partial oxidation of methane to syngas in matrix type converters. Chem. Eng. Sci. 2019, 207, 744–751. DOI: 10.1016/j.ces.2019.07.012.
- Savchenko, V.I.; Zimin, Ya.S.; Nikitin, A.V.; Sedov, I.V.; Arutyunov, V.S. Utilization of CO2 in non-catalytic dry reforming of C1–C4 hydrocarbons. Journal of CO2 Utilization. 2021, 47, 101490. [CrossRef]
- Arutyunov, V.S.; Strekova, L.N.; Savchenko, V.I.; Sedov, I.V.; Nikitin, A.V.; Eliseev, O.L.; Kryuchkov, M.V.; Lapidus, A.L. Prospects of Conversion of Hydrocarbon Gases to Liquid Products Based on Nitrogen-Rich Synthesis Gas (Review). Petroleum Chemistry. 2019, 59, 370–379. [CrossRef]
- Hedden, K.; Jess, A.; Kuntze, T. From Natural gas to liquid hydrocarbons (1). A new concept for the production of liquid hydrocarbons from natural gas in remote areas. OIL GAS – European Magazine. 1994, 3, 42–44.
- Jess, A.; Popp, R.; Hedden, K. Fischer–Tropsch-synthesis with nitrogen-rich syngas. Fundamentals and reactor design aspects. Appl. Catal. A. 1999, 186, 321–342.
- Kertthong, T.; Schmid, M.; Scheffknecht, G. Non-catalytic partial oxidation of methane in biomass-derived syngas with high steam and hydrogen content optimal for subsequent synthesis process. J. Energy Inst. 2022, 105, 251–261. [CrossRef]
- Arutyunov, V.; Nikitin, A.; Strekova, L.; Savchenko, V.; Sedov, I. Renewable biogas as a source for small-scale production of liquid fuels. Catalysis Today. 2021, 379, 23–27. [CrossRef]
- Arutyunov, V.S.; Nikitin, A.V.; Savchenko, V.I.; Sedov, I.V. Combined Production of Hydrogen and Methanol without CO2 Emission Based on Matrix Conversion of Natural Gas. Doklady Chemistry. 2023, 513 (2), 361–366. DOI: 10.1134/S0012500823601018.
- Patent RU 2608749 С1, 2017.
- Arutyunov, V.S.; Shmelev, V.M.; Rakhmetov, A.N.; Shapovalova, O.V.; Zakharov, A.A.; Roschin, A.V. New approaches to development of low-emission combustion chambers for gas turbine engines. International Scientific Journal for Alternative Energy and Ecology. 2013, № 06 (128), 105–120.
- Lyalin, D.A.; Vasilik, N.Ya.; Zaharov, A.A.; Arutyunov, V.S.; Shmelev, V.M. Low-emission efficient infrared burner for cooking applications. Oil & Gas Chemistry. 2018, No. 4, 5–9. [CrossRef]
- Qazi, U.Y. Future of Hydrogen as an Alternative Fuel for Next-Generation Industrial Applications; Challenges and Expected Opportunities. Energies 2022, 15, 4741. [CrossRef]
- Lamari, F.; Weinberger, B.; Langlois, P.; Fruchart, D. Instances of Safety-Related Advances in Hydrogen as Regards Its Gaseous Transport and Buffer Storage and Its Solid-State Storage. Hydrogen 2024, 5, 387–402. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).