Submitted:
02 March 2026
Posted:
03 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Glass Preparation and Heat Treatment
2.2. Characterisation
2.2.1. Differential Scanning Calorimetry
2.2.2. ATR-FTIR Spectroscopy
2.2.3. X-Ray Powder Diffraction
2.2.4. 31P MAS-NMR Spectroscopy
2.2.5. 19F MAS-NMR Spectroscopy
2.2.6. Transmission Electron Microscopy
3. Results
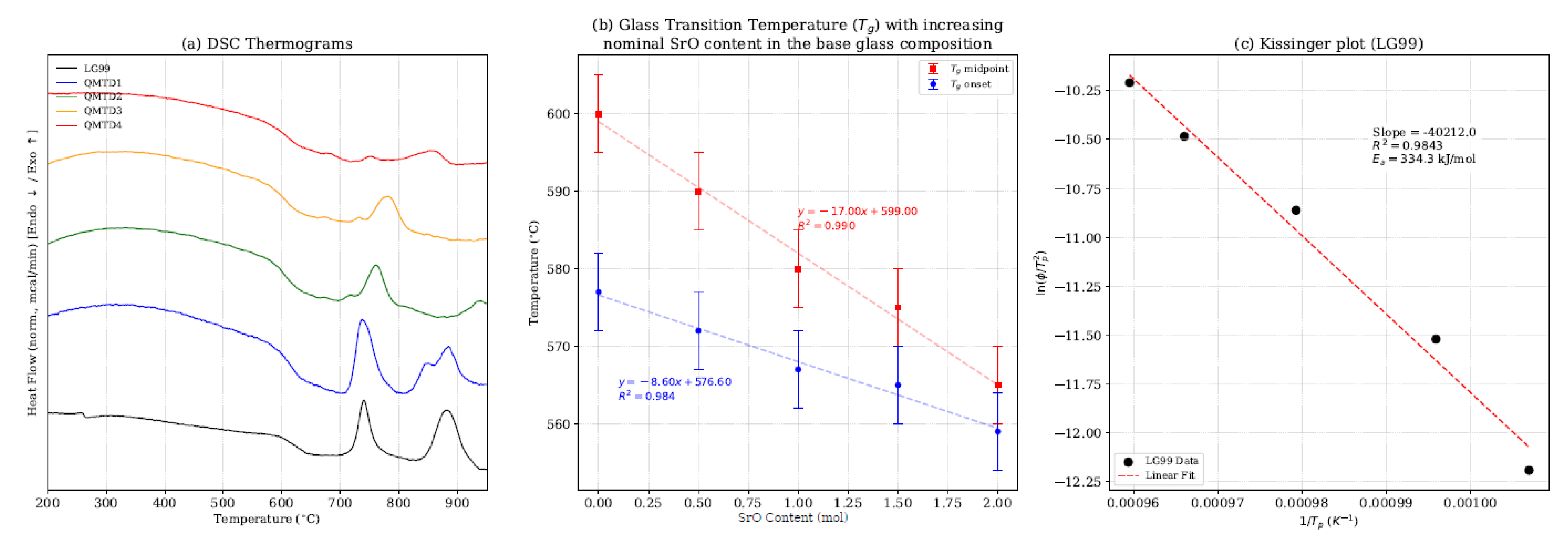
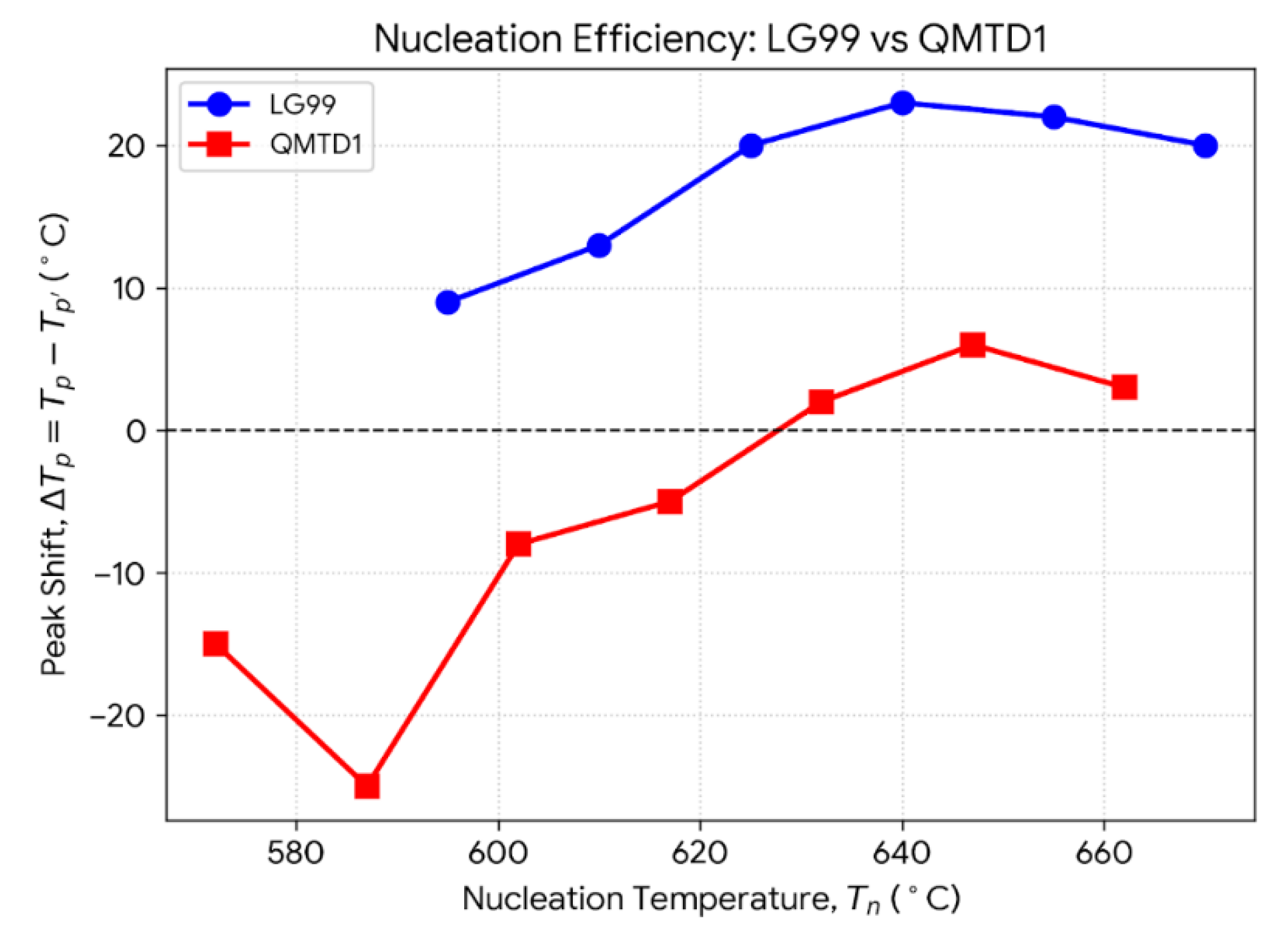
3.1. Thermal Behaviour, Nucleation and Crystallisation Tendency (DSC)
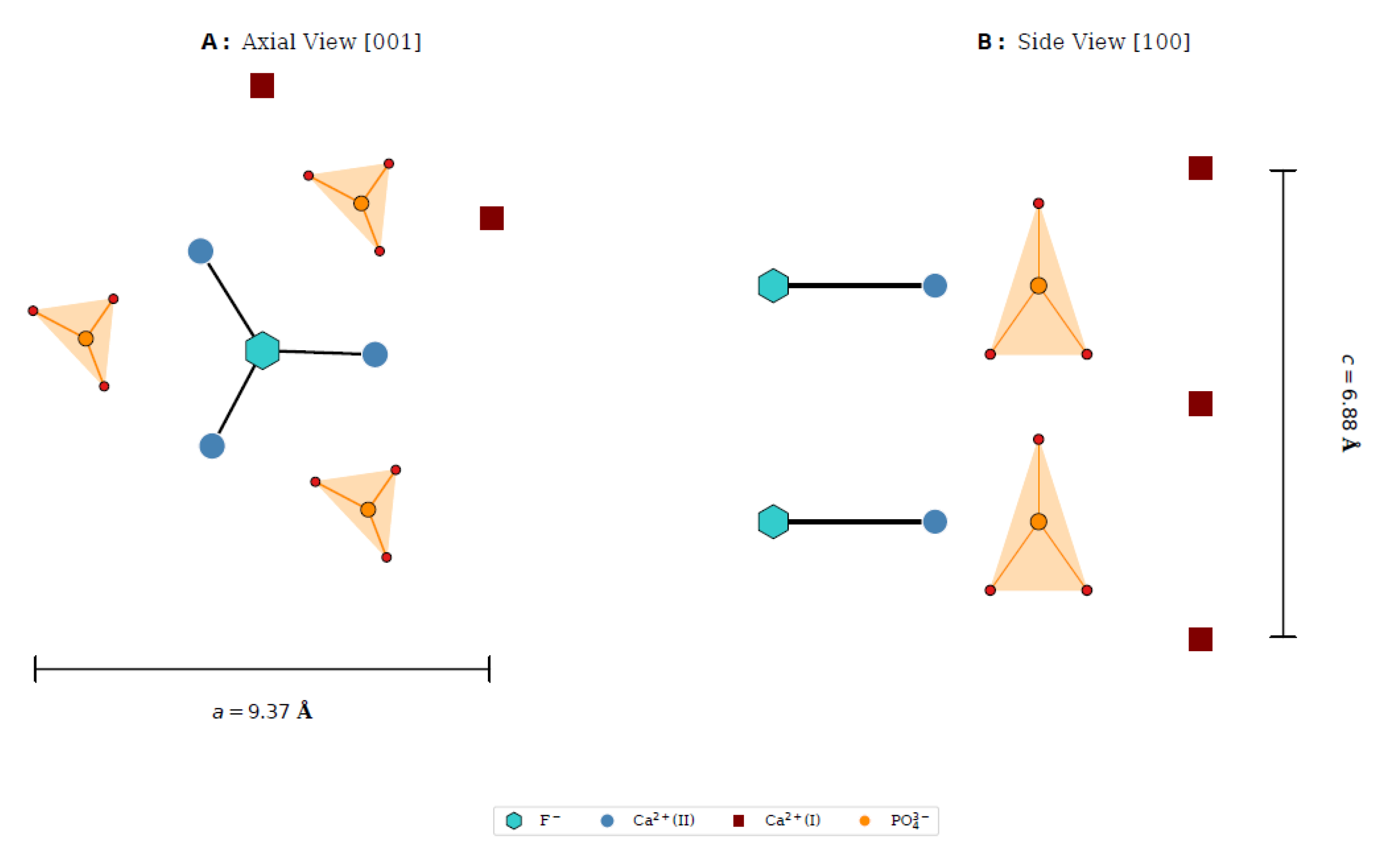
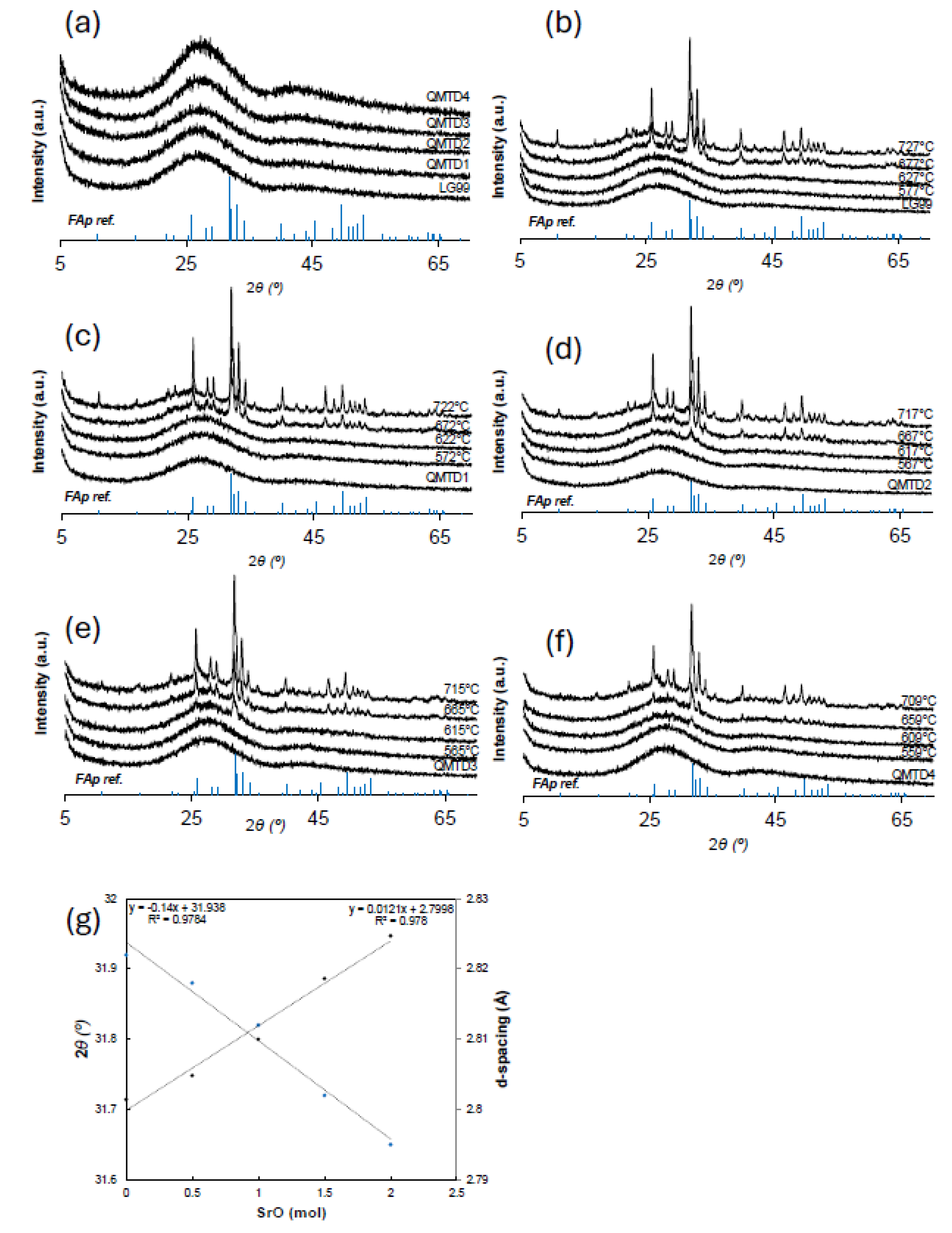
3.2. Phase Formation and Lattice Expansion (XRD)
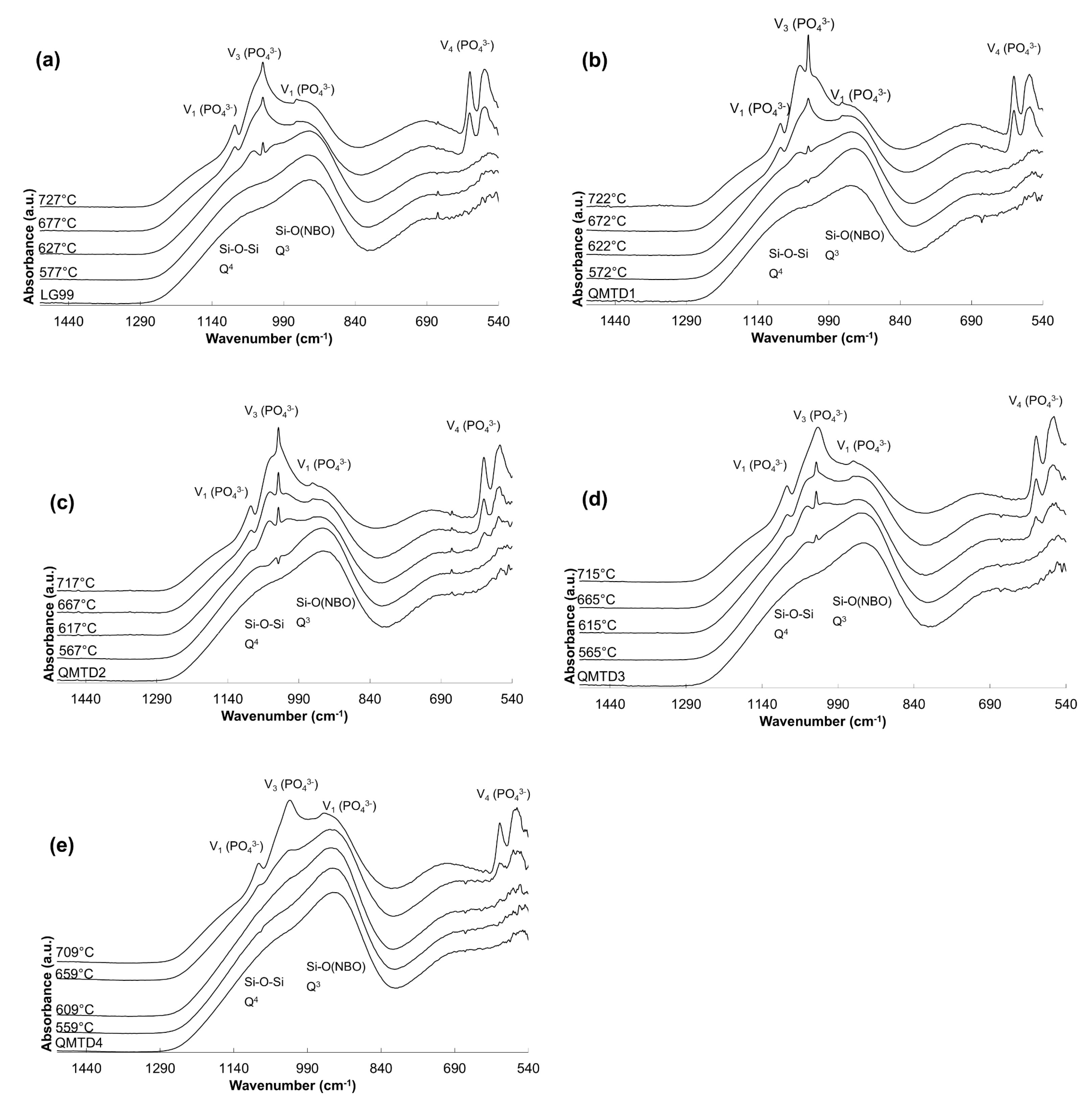
3.3. ATR-FTIR Spectroscopy
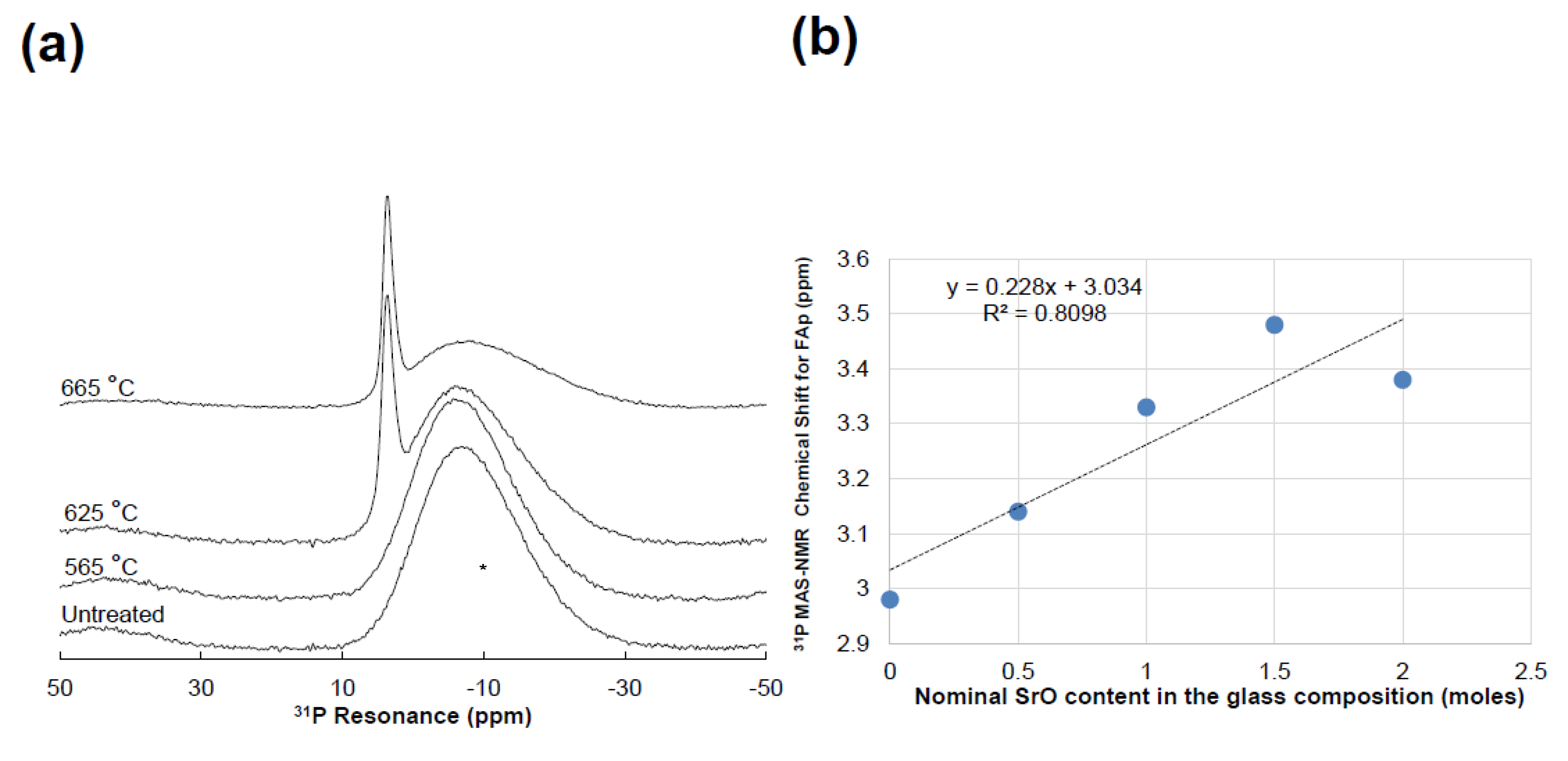
3.4. ³¹P MAS-NMR: Phosphate Coordination Environments
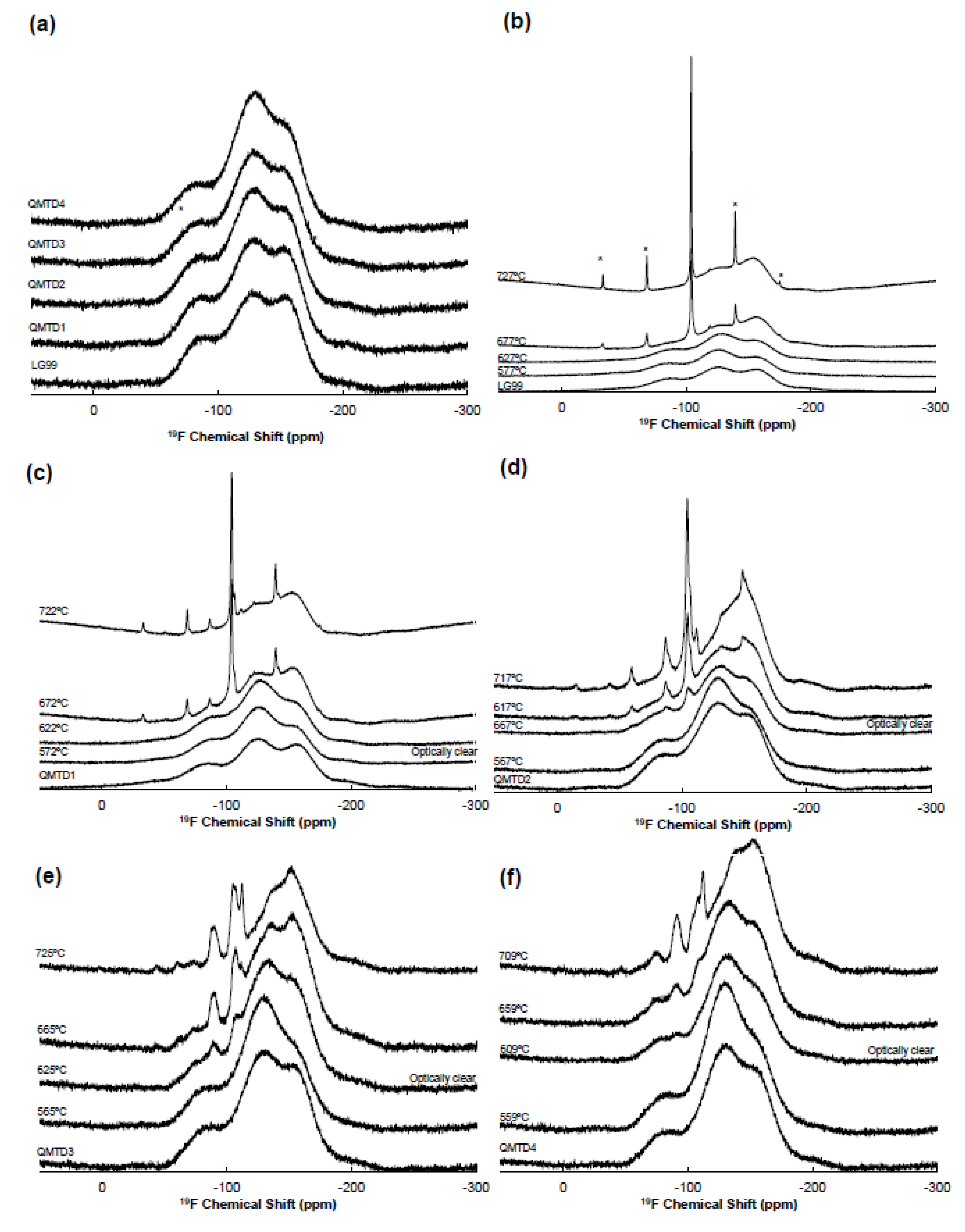
3.5. ¹⁹F MAS-NMR: Ca(II)/Sr(II) Site Occupancy
- As-Quenched Glass Structure
- Evolution of Crystalline Phases
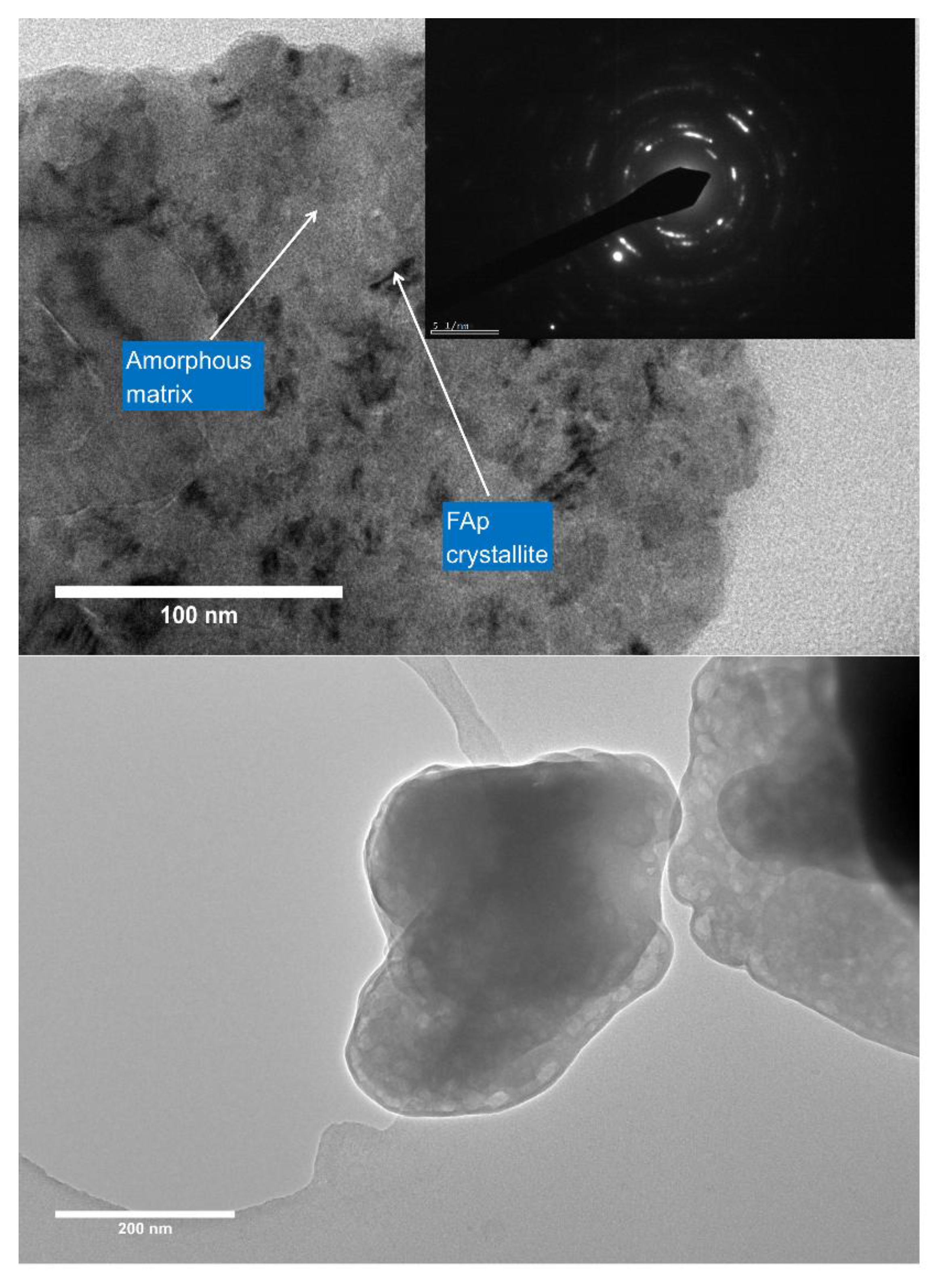
3.6. Nanostructure (TEM)
4. Discussion
4.1. Glass Structure and Nucleation Kinetics
4.2. Structural Site Preference and NMR Sensitivity
4.3. Secondary Phases and Nanoscale Structure
4.4. The Kinetic Barrier to Sr Incorporation
5. Conclusions
- CaO/SrO substitution in the glass compositions studied reduces Tg, network polymerisation and suppresses homogeneous nucleation of fluorapatite.
- A mixed Ca5−xSrx(PO4)3F solid solution is the primary crystalline phase formed within the investigated temperature range, with minor contributions from fluorite, Al-F-Ca(2) and Al-F-Sr(2) crystalline environments.
- Sr2+ ions partially incorporate into Ca(II) sites, inducing a systematic expansion of the fluorapatite lattice.
- 31P MAS-NMR reveals significant phosphorus deshielding, reflecting local PO4 tetrahedral distortion, rather than the total extent of strontium substitution.
- Limited Sr incorporation into the fluorapatite is attributed to a kinetic barrier arising from Sr2+ preference for Si-F-Sr(n) speciation in the parent glass. This sequestering effect constrains Sr and F availability during nucleation and crystallisation mechanism, despite the thermodynamic favourability of Sr-substituted apatite formation.
Acknowledgments
CRediT Statement
References
- Hench, L.L.; Wilson, J. Surface-active biomaterials. Science 1984, 226, 630–636. [Google Scholar] [CrossRef] [PubMed]
- Hill, R.G. An alternative view of the degradation of bioglass. J Mater Sci Lett. 1996, 15, 1122–1125. [Google Scholar] [CrossRef]
- Rehman, I.; Bonfield, W. Characterization of hydroxyapatite and carbonated apatite by photoacoustic FTIR spectroscopy. J Mater Sci Mater Med. 1997, 8, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, J.W. Structural and chemical aspects of dental materials. Dent Mater. 2010, 26, 93–102. [Google Scholar]
- Hill, R.G. Structural aspects of bioactive glasses: influence on degradation and cell response. J Non-Cryst Solids 2011, 357, 388–394. [Google Scholar]
- Serra, J.; González, P.; Liste, S.; Chiussi, S.; León, B.; Pérez-Amor, M. Composition, microstructure, and bioactivity of novel bioactive glass-derived ceramics. J Eur Ceram Soc. 2014, 34, 3913–3923. [Google Scholar]
- Jones, J.R.; et al. Bioactive glasses: from concept to clinical applications. Acta Biomater. 2011, 7, 2214–2225. [Google Scholar]
- Marie, P.J. Role of osteoblasts and osteocytes in bone remodeling. Bone 2010, 46, 7–15. [Google Scholar]
- Christoffersen, J.; et al. Regulation of mineralization in calcified tissues. Calcif Tissue Int. 1997, 60, 59–63. [Google Scholar]
- Bigi, A.; Panzavolta, S.; Roveri, N.; Ripamonti, A.; Fassina, L. Synthetic hydroxyapatites: intrinsic crystals and composites. J Inorg Biochem. 2007, 101, 1749–1757. [Google Scholar]
- Vahabzadeh, S.; et al. Sr-substituted fluorapatite for dental and orthopedic applications. Mater Sci Eng C. 2015, 48, 237–246. [Google Scholar]
- Elliott, J.C. Structure and Chemistry of the Apatites; Elsevier: Amsterdam, 1994. [Google Scholar]
- Koutsopoulos, S. Synthesis and characterization of hydroxyapatite crystals: A review study on the analytical methods. J Biomed Mater Res. 2002, 62, 600–612. [Google Scholar] [CrossRef] [PubMed]
- Terra, J.; et al. Tailoring chemical composition and nanostructure of bioactive materials. Chem Mater. 2009, 21, 550–557. [Google Scholar]
- Bohner, M. Design of ceramic biomaterials and bone substitutes. Biomaterials 2000, 21, 1301–1309. [Google Scholar]
- Grover, L.M.; et al. Strontium substitution in hydroxyapatite: structural and biological effects. J Mater Sci Mater Med. 2012, 23, 2627–2637. [Google Scholar]
- Landi, E.; Tampieri, A.; Celotti, G.; Sprio, S.; Sandri, M. Hydroxyapatite for biomedical applications: tailoring microstructure and mechanical properties. Biomaterials 2007, 28, 3649–3657. [Google Scholar]
- Kim, H.W.; Song, S.H.; Suh, H.; Knowles, J.C. Bioactive glass ceramics for bone regeneration. Biomaterials 2004, 25, 2533–2539. [Google Scholar] [CrossRef]
- Tampieri, A.; Sprio, S.; Sandri, M.; Bronzini, I.; Gepi, A.; Bracci, B. Biomimetic hydroxyapatite scaffolds. Acta Biomater. 2008, 4, 1626–1633. [Google Scholar]
- Zhou, H.; Lee, J. Nanoscale hydroxyapatite particles for bone tissue engineering. Acta Biomater. 2011, 7, 2769–2778. [Google Scholar] [CrossRef]
- Landi, E.; Tampieri, A.; Celotti, G.; Sprio, S. Influence of synthesis and sintering parameters on the characteristics of carbonate apatite. Biomaterials 2004, 25, 1763–1770. [Google Scholar] [CrossRef]
- Yesinowski, J.P.; Eckert, H. Hydrogen environments in calcium phosphates: 1H MAS NMR at high spinning speeds. J Am Chem Soc. 1987, 109, 6274–6282. [Google Scholar] [CrossRef]
- Smith, M.E. Applications of nuclear magnetic resonance spectroscopy in materials chemistry. Prog Nucl Magn Reson Spectrosc. 1993, 25, 159–190. [Google Scholar]
- Wu, Y.; Michaelis, V.K.; Danielson, D.C.; Grandinetti, P.J. Solid-state NMR of calcium phosphates and bioactive glasses. Solid State Nucl Magn Reson. 2010, 38, 49–56. [Google Scholar]
- Chen, Q.; et al. MAS-NMR studies on phosphate environments in Sr-substituted apatites. Solid State Nucl Magn Reson. 2019, 98, 25–34. [Google Scholar]
- Shelby, J.E. Introduction to Glass Science and Technology; Royal Society of Chemistry: Cambridge, 2005. [Google Scholar]
- Sun, L.; et al. Strontium effects on glass network depolymerisation and crystallisation. J Non-Cryst Solids 2020, 534, 119996. [Google Scholar]
- Liu, Y.; et al. Lattice expansion and site-specific substitution in Sr-fluorapatites. J Mater Sci. 2015, 50, 12345–12355. [Google Scholar]
- Mao, J.; et al. Surface nucleation and growth of fluorapatite nanocrystals in bioactive glasses. J Non-Cryst Solids 2017, 463, 87–96. [Google Scholar]
- Kaur, H.; et al. Coordination chemistry and NMR characterisation of substituted apatites. J Phys Chem C. 2020, 124, 11245–11255. [Google Scholar]
- Shen, D.; et al. Effects of Sr substitution on MAS-NMR phosphate chemical shifts. Solid State Nucl Magn Reson. 2016, 78, 12–21. [Google Scholar]
- Zeng, Q.; Stebbins, J.F. Fluoride sites in aluminosilicate glasses: High-resolution 19F NMR results. Am Mineral. 2000, 85, 863–867. [Google Scholar] [CrossRef]
- Kiczenski, T.J.; Stebbins, J.F. Fluoride sites in calcium and barium oxyfluorides: 19F NMR on crystalline model compounds and glasses. J Non-Cryst Solids 2002, 306, 160–168. [Google Scholar]
- Stebbins, J.F.; Zeng, Q. Cation ordering at fluoride sites in silicate glasses: a high-resolution 19F NMR study. J Non-Cryst Solids 2000, 262, 1–5. [Google Scholar]
- Wang, X.; et al. Nanoscale lattice expansion in Sr-fluorapatite verified by TEM. J Mater Chem B 2017, 5, 2345–2354. [Google Scholar]
- Song, Y.; et al. Surface-dominated nucleation in Sr-fluorapatite glasses. Ceram Int. 2018, 44, 18203–18212. [Google Scholar]
- Yang, F.; et al. Surface vs bulk crystallisation in phosphate glasses containing SrO. J Non-Cryst Solids 2019, 517, 1–9. [Google Scholar]
- O’Donnell, M.D.; Hill, R.G. Influence of strontium and the importance of glass chemistry and structure when designing bioactive glasses for bone regeneration. Acta Biomater. 2010, 6, 2382–2385. [Google Scholar] [CrossRef] [PubMed]
- Hill, R.G.; Stamboulis, A.; Law, R.V.; Clifford, A.; Towler, M.R.; Crowley, C. The influence of strontium substitution in fluorapatite glasses and glass ceramics. J Non Cryst Solids 2004, 336, 223–229. [Google Scholar] [CrossRef]
- Duminis, T. Evidence for new fluoride coordination in SiO₂-Al₂O₃-P₂O₅-CaO/SrO-CaF₂ compositions used in ion-leachable ionomer-type cements: A ¹⁹F MAS-NMR investigation. Mater Lett. 2024, 359, 135919. [Google Scholar] [CrossRef]
- Hill, R.G.; Calver, A.; Skinner, S.; Stamboulis, A.; Law, R.V. A MAS-NMR and Combined Rietveldt Study of Mixed Calcium/Strontium Fluorapatite Glass-Ceramics. Key Engineering Materials 2006, 309, 305–308. [Google Scholar]
- Marotta, A.; Buri, A.; Branda, F. Nucleation in glass and differential thermal analysis. Journal of Materials Science 1981, 16, 341–344. [Google Scholar] [CrossRef]
- Duminis, T.; Shahid, S.; Hill, R.G. Apatite glass-ceramics: a review. Front Mater. 2016, 3, 59. [Google Scholar]
- Shahid, S.; Hassan, U.; Billington, R.W.; Hill, R.G.; Anderson, P. Glass ionomer cements: Effect of strontium substitution on esthetics, radiopacity and fluoride release. Dent Mater. 2014, 30, 308–313. [Google Scholar] [CrossRef]
- Kissinger, H.E. Reaction Kinetics in Differential Thermal Analysis. Anal. Chem. 1957, 29, 1702–1706. [Google Scholar] [CrossRef]
- Augis, J.A.; Bennett, J.E. Calculation of the Avrami parameters for heterogeneous solid state reactions using a linear heating rate. J. Therm. 1978, 13, 283–292. [Google Scholar]
- Hill, R.G.; Wood, D.J. The crystallization of bioactive glasses in the system SiO2-P2O5-CaO-Na2O-CaF2. J Mater Sci Mater Med. 1995, 6, 311–318. [Google Scholar]
- Fredholm, Y.C.; Brauer, D.S.; Hill, R.G.; Law, R.V. Influence of strontium substitution for calcium on the crystallization of bioactive glass. J Non-Cryst Solids 2010, 356, 2546–2551. [Google Scholar] [CrossRef]
- Matusita, K.; Sakka, S. Kinetic study of crystallization of glass by differential scanning calorimetry—criterion for crystallization mechanism. Phys Chem Glasses 1982, 23, 116–120. [Google Scholar]
- Donald, I.W. Crystallization kinetics of some silicate glasses. J Non-Cryst Solids 2004, 345, 120–126. [Google Scholar]
| Glass Code | SiO₂ | Al₂O₃ | P₂O₅ | CaO | SrO | CaF₂ |
| LG99 | 4.5 | 3 | 1.5 | 2 | 0 | 3 |
| QMTD1 | 4.5 | 3 | 1.5 | 1.5 | 0.5 | 3 |
| QMTD2 | 4.5 | 3 | 1.5 | 1 | 1 | 3 |
| QMTD3 | 4.5 | 3 | 1.5 | 0.5 | 1.5 | 3 |
| QMTD4 | 4.5 | 3 | 1.5 | 0 | 2 | 3 |
| LG99 (Reference) | QMTD1 (Sr-Substituted) | ||||
|---|---|---|---|---|---|
| Tn (°C) | Tp′ (°C) | ΔTp (°C) | Tn (°C) | Tp′ (°C) | ΔTp (°C) |
| 595 | 740 | 9 | 572 | 744 | -15 |
| 610 | 736 | 13 | 587 | 754 | -25 |
| 625 | 729 | 20 | 602 | 737 | -8 |
| 640 | 726 | 23 (Max) | 617 | 734 | -5 |
| 655 | 727 | 22 | 632 | 727 | 2 |
| 670 | 729 | 20 | 647 | 723 | 6 (Max) |
| 662 | 726 | 3 |
| Sample | SrO (mol) | Tg Onset (°C) | Tg Midpoint (°C) | Tp (°C) | Avrami parameter, n | Core Mechanism | Residual Glass Mechanism |
|---|---|---|---|---|---|---|---|
| LG99 | 0.0 | 577 | 600 | 740 | 3.01 | 3D Volume (homogenous) nucleation | - |
| QMTD1 | 0.5 | 572 | 590 | 740 | 2.08 | 1D/3D Mixed (volume + surface) | 2D Interface-Controlled Growth |
| QMTD2 | 1.0 | 567 | 580 | 760 | 1.92 | 1D Surface-dominated | 2D Interface-Controlled Growth |
| QMTD3 | 1.5 | 565 | 575 | 780 | 1.63 | 1D Surface-exclusive | 3D Diffusion-Controlled Growth |
| QMTD4 | 2.0 | 559 | 565 | 850 | 1.49 | 1D Surface-exclusive | 1D Diffusion-Controlled Growth |
| Fluoride Environment | Peak Position (ppm) | Assignment | Reference |
|---|---|---|---|
| Amorphous (Glass) | |||
| Network Modifier | -90 | F-M(n) | [32,34] |
| Mixed Silicon | -125 | Si-F-M(n) | [33,40] |
| Mixed Aluminum | -150 | Al-F-M(n) | [32] |
| Crystalline | |||
| Ca-FAp | -104 | F-Ca(3) | [41] |
| Mixed FAp | -90 | F-Ca(2)Sr | [41] |
| Mixed FAp | -70 | F-Sr(2)Ca | [41] |
| Sr-FAp | -60 | F-Sr(3) | [41] |
| Ca-Fluoroaluminate | -139 | Al-F-Ca(2) | [33] |
| Sr-Fluoroaluminate | -112 | Al-F-Sr(2) | Present study |
| Mixed fluorite Ca(1-x)SrxF2 | -106 | - | Present study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




