1. Introduction
Gestational diabetes mellitus (GDM) is one of the most common metabolic disorders occurring during pregnancy and is characterized by first-diagnosed hyperglycemia, the severity of which ranges from mild glucose tolerance impairment to overt diabetes requiring insulin therapy. The prevalence of this pathology is steadily increasing worldwide due to the obesity epidemic, advancing maternal age, and lifestyle changes, reaching 7% to 20% of all pregnancies across different populations [
1,
2,
3]. Beyond its direct impact on the course of gestation, GDM is associated with a wide range of adverse maternal and perinatal outcomes, among which fetal macrosomia (FM) holds a prominent place. FM occurs in 15–45% of women with this pathology and represents a risk factor for birth trauma, operative delivery, and metabolic disorders in offspring during postnatal development [
4,
5,
6,
7].
Traditionally, research on the pathogenesis of GDM and its complications has focused primarily on carbohydrate and lipid metabolism disturbances, whereas the role of amino acids—which serve not only as building blocks for protein synthesis but also as signaling molecules modulating insulin secretion, tissue glucose sensitivity, and epigenetic processes—has been studied to a considerably lesser extent [
7,
8]. Amino acids play a critical role in fetal development, meeting the demand for nitrogenous compounds and participating in the formation of the nervous system, muscle tissue, and internal organs. Their transport across the placenta is mediated by specialized active transport systems, the functional activity of which may be altered under conditions of hyperglycemia and hyperinsulinemia [
9,
10,
11]. In recent years, emerging studies have indicated associations between concentrations of specific amino acids, particularly branched-chain amino acids, and the risk of developing insulin resistance [
12,
13]. Comprehensive analysis of the amino acid profile in the mother-fetus system in GDM, including comparative data from maternal serum, cord blood, and amniotic fluid, has been performed in only a few studies [
14,
15].
Of particular interest is the investigation of amino acid status in GDM complicated by FM, since excessive amino acid supply to the fetus may represent a key factor stimulating pancreatic β-cell hyperplasia, insulin hypersecretion, and consequently, excessive adipose tissue accumulation, independent of maternal glycemia levels. Amniotic fluid, serving as an integrative compartment that reflects fetal metabolic status and excretory function, may contain unique information regarding amino acid metabolism patterns in different GDM phenotypes; however, its diagnostic potential in this context remains largely unexplored. Correlation analysis between amino acid levels in various biological compartments and clinical parameters of mothers and newborns enables not only the identification of the most informative markers but also the reconstruction of pathogenetic mechanisms underlying adverse pregnancy outcomes.
Modern multivariate statistical methods, including machine learning algorithms and SHAP value calculation, offer new opportunities for identifying metabolic signatures associated with the development of GDM and its complications, overcoming the limitations of traditional approaches based on the analysis of individual parameters. Metabolic pathway enrichment analysis using specialized databases allows not only to detect alterations in individual amino acid concentrations but also to understand which biochemical processes are involved in disease pathogenesis—knowledge that is crucial for developing targeted therapeutic strategies.
The aim of the present study was to perform a comparative analysis of amino acid concentrations in maternal serum, cord blood, and amniotic fluid from women with physiological pregnancies and GDM, and to assess their association with the development of FM, in order to identify potential metabolic biomarkers and elucidate the pathogenetic mechanisms underlying this pathology.
2. Results
2.1. Clinical and Anamnestic Parameters
Clinical and anamnestic data were analyzed from 62 mother–newborn dyads. Comparative analysis of women with GDM (n=37) and without GDM (n=25) revealed a number of differences, presented in
Table S1.
The median age in the GDM group was 36 years, whereas in the control group, the age was lower at 33 years. Although these differences did not reach statistical significance, they demonstrate an important clinical trend. The median age in the GDM group exceeds the 35-year threshold, which is a well-established risk factor. IVF rates were comparable between groups (8% vs. 11%, p=1.00), allowing exclusion of conception method as a confounding factor for GDM development in this cohort. High risk of preeclampsia (PE) according to first-trimester screening was detected six times more frequently (27% vs. 4% of cases) in the GDM group compared to the control group (p=0.047). This finding is likely related to shared pathogenetic mechanisms between GDM and PE, including endothelial dysfunction, systemic inflammation, and insulin resistance, and supports the need for comprehensive risk assessment [
16]. Pre-pregnancy BMI was significantly higher in the GDM group (24.45 kg/m² vs. 22.15 kg/m², p=0.03). Notably, the upper quartile in the GDM group (28.72 kg/m²) already corresponds to overweight status. Importantly, 54% of women with GDM had normal baseline weight, indicating heterogeneity in pathogenesis and the need for universal screening. BMI at delivery was also significantly higher in the GDM group (30.09 kg/m² vs. 27.89 kg/m², p=0.03), with the median value in the GDM group corresponding to class I obesity. Pathological weight gain was observed only in the GDM group (in 14% of women), emphasizing the importance of monitoring gestational weight gain. Family history of diabetes mellitus was identified in 43% of women with GDM (43% vs. 16%, p=0.048), confirming the significant role of genetic predisposition [
17]. Family history of diabetes and pre-pregnancy BMI are among the factors most strongly influencing the odds of developing GDM. The combined contribution of BMI and family history of diabetes to GDM development is presented in
Figure S1.
Dietary therapy was received by 89% of women with GDM, and insulin therapy by 32%, with insulin prescribed to 19% in the second trimester and 32% in the third trimester, reflecting the natural progression of insulin resistance.
A history of macrosomia was observed only in the GDM group (16% vs. 0%, p=0.09), consistent with data indicating an increased risk of recurrence. Newborn birth weight did not differ significantly between groups (3600 g vs. 3518 g, p=0.37). Apgar scores at 1 and 5 minutes were equally high in both groups (8 and 9 points, respectively). Only newborns from the GDM group required observation and treatment in the neonatal intensive care unit (5%) and the neonatal pathology department (8%). However, these differences were not statistically significant.
Next, clinical and anamnestic data from 62 mother–newborn dyads were analyzed according to the presence of GDM and FM. The dyads were stratified into four subgroups: Subgroup 1 (GDM–, fetal normosomia, n=18), Subgroup 2 (GDM–, FM, n=7), Subgroup 3 (GDM+, fetal normosomia, n=24), and Subgroup 4 (GDM+, FM, n=13). Comparison of the four subgroups revealed a number of statistically significant intergroup differences, as well as several clinically important trends that did not reach statistical significance but warrant discussion in the context of current evidence (
Table S2).
A tendency toward higher maternal age was observed in subgroups with GDM, consistent with data from the HAPO study [
18], which identified age over 35 years as a risk factor for GDM and FM. IVF rates were comparable across all subgroups (p=0.85), ranging from 6–14%. Pre-pregnancy BMI differed significantly between subgroups (p=0.02), reaching minimum values in Subgroup 1 (21.8 kg/m²) and maximum values in Subgroup 4 (26.5 kg/m², p=0.007). In Subgroup 4, 38% of women were overweight and 23% had class I obesity. These findings align with data published in Lancet Diabetes & Endocrinology regarding the association between elevated BMI and risk of GDM and FM [
19], and further support the concept of GDM heterogeneity with distinct insulin-resistant and secretory subtypes. Median weight gain was 12 kg in Subgroups 1, 2, and 4, whereas Subgroup 3 had the lowest median gain at 9.5 kg (p=0.16). The frequency of pathological weight gain (p=0.008) was absent in Subgroups 1 and 2, was 4% in Subgroup 3, and reached 31% in Subgroup 4. This is consistent with findings from the LIFE-Moms Prospective Meta-Analysis [
20], demonstrating that excessive gestational weight gain in GDM increases the risk of FM by 2.5–3-fold. The highest BMI at delivery was observed in Subgroups 2 and 4 (30.8 and 30.9 kg/m², p<0.001), further confirming data linking BMI >30 kg/m² to a twofold increased risk of FM. Family history of diabetes (p=0.07) was observed across all subgroups, reaching 50% and 31% in Subgroups 3 and 4, respectively. Dietary therapy was prescribed to 96% of women in Subgroup 3 and 77% in Subgroup 4 (p34=0.03), while insulin therapy was administered to 33% and 31%, respectively (p=0.02), underscoring the importance of timely treatment initiation. History of macrosomia (p=0.03) was present only in GDM subgroups: in 8% of Subgroup 3 and 31% of Subgroup 4, consistent with evidence of high recurrence risk for FM [
21]. Male fetal sex was significantly more prevalent in subgroups with FM, reaching 86% and 77% in Subgroups 2 and 4 (compared to 39–38% in subgroups without FM, p=0.02), aligning with published data indicating an increased risk of FM in male infants [
22].
Fetal weight was most strongly influenced by maternal pre-pregnancy BMI and infant sex (
Figure S2), with male infants having higher birth weights than female infants born to women with equivalent BMI.
2.2. Aminoacid Profiling
Next, we analyzed alterations in the quantitative amino acid (AA) composition within the mother–fetus system in relation to GDM and newborn birth weight, aiming to identify potential markers of these conditions.
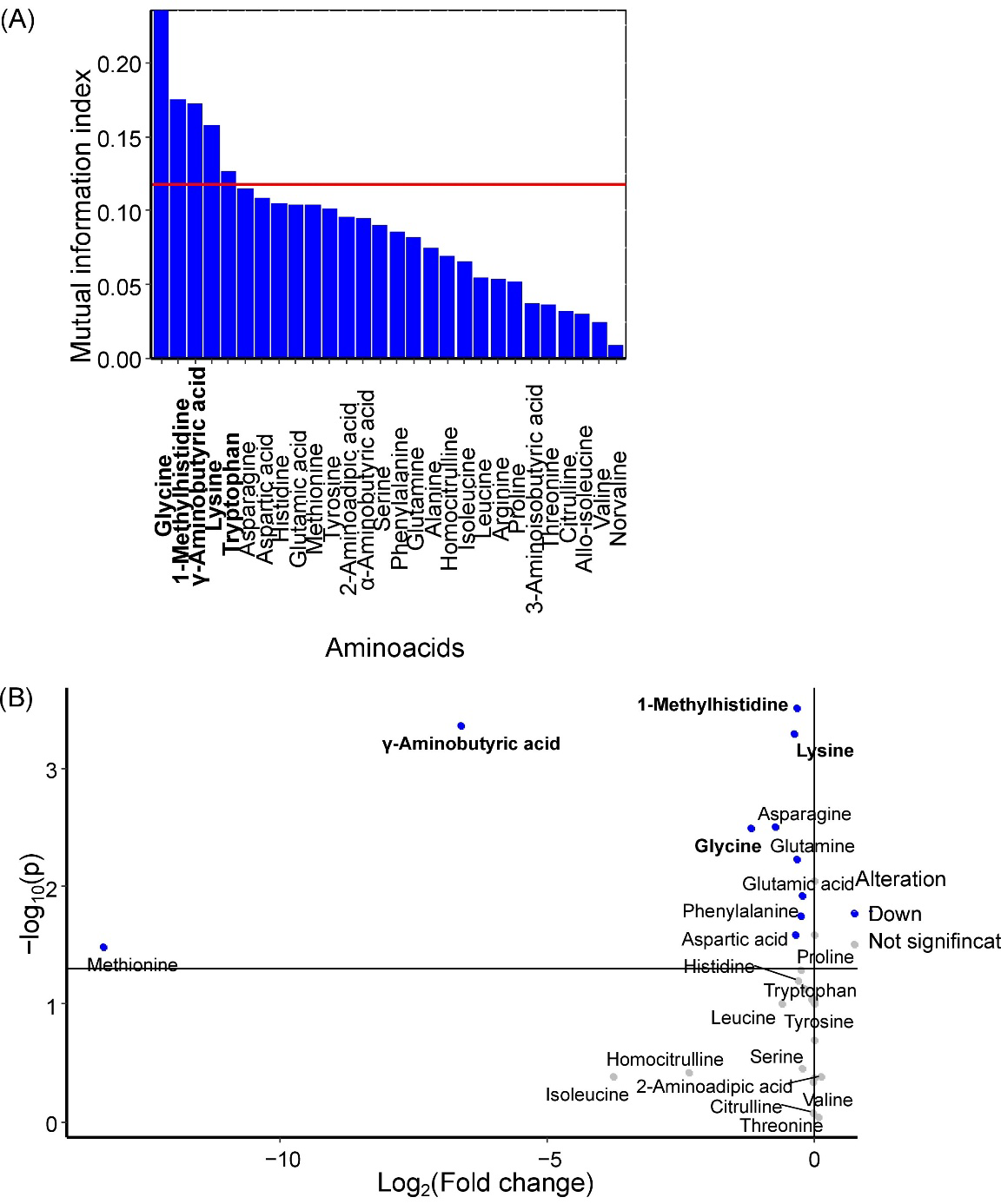
Glycine, 1-methylhistidine, γ-aminobutyric acid, lysine, and tryptophan were identified as potential GDM markers in maternal venous serum (
Figure 1A), with all amino acids except tryptophan showing statistically significant decreases in GDM (
Figure 1B).
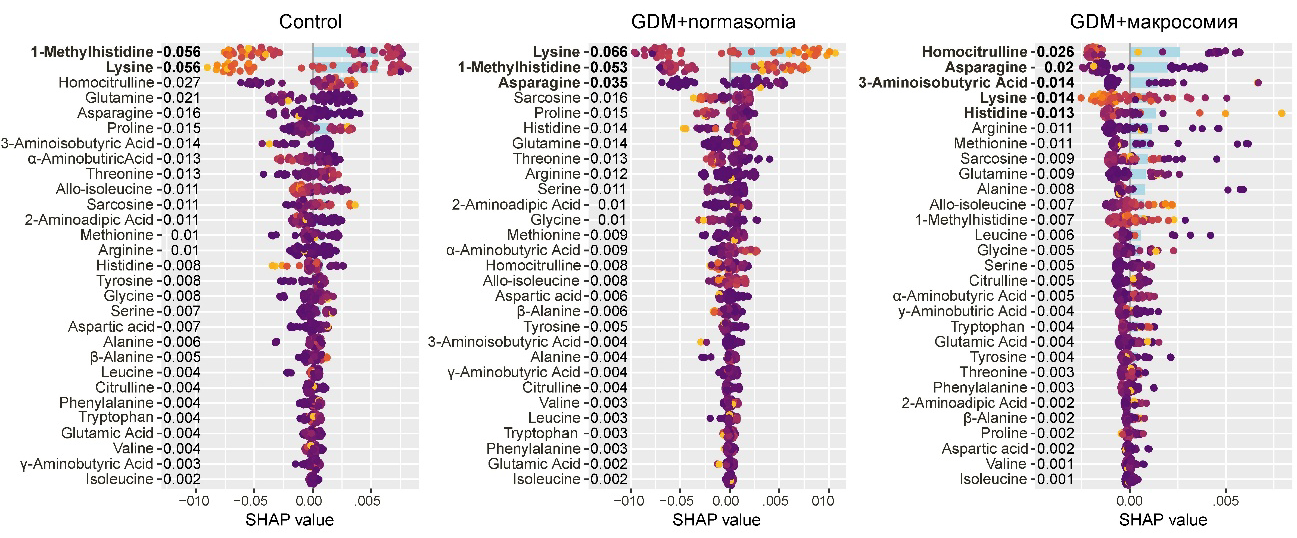
Lysine and 1-methylhistidine were characteristic of the control group, with lysine showing a specific decrease in GDM pregnancies resulting in normal-weight newborns (
Figure 2).
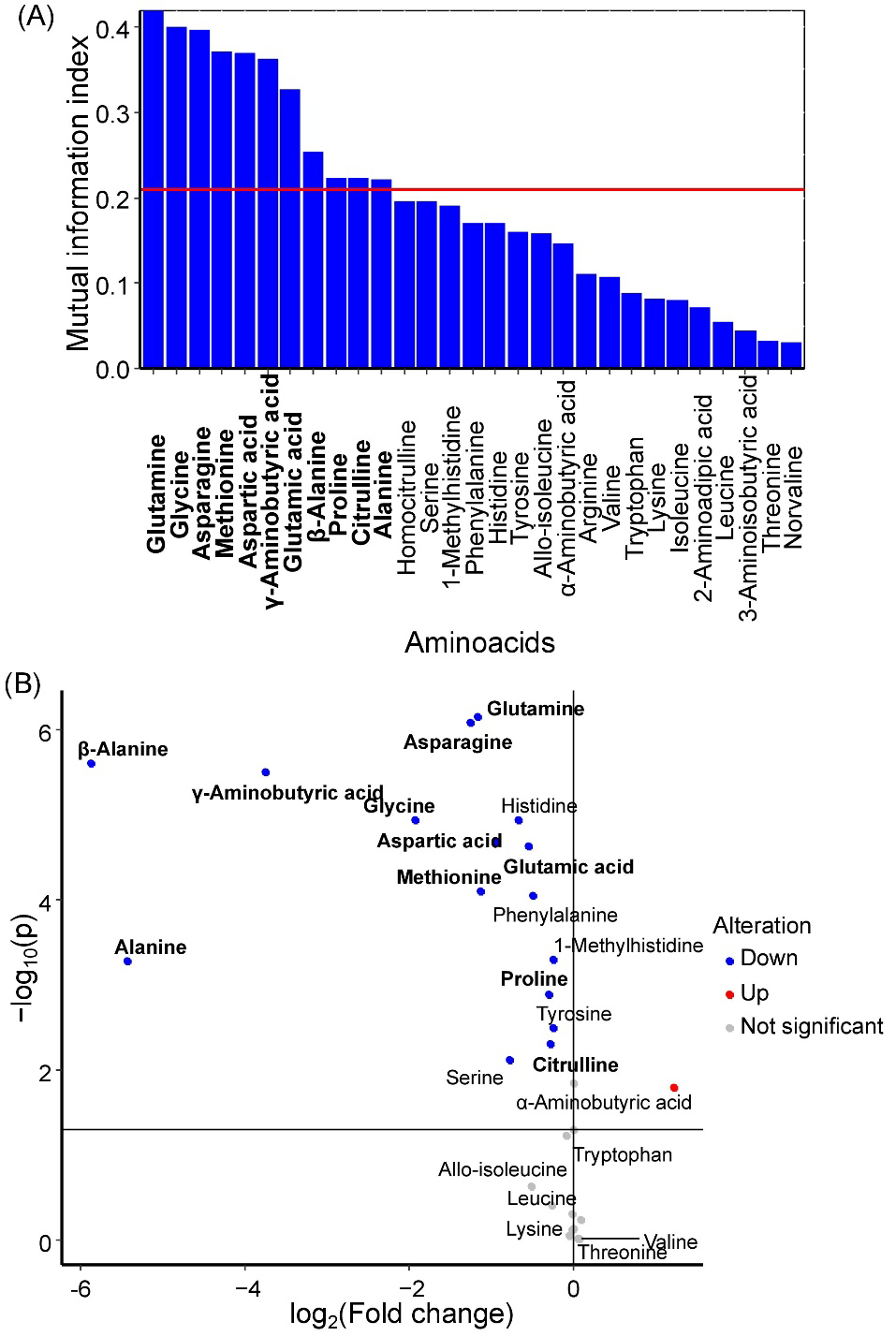
Glutamine, glycine, asparagine, methionine, aspartic acid, γ-aminobutyric acid, glutamic acid, β-alanine, proline, citrulline, and alanine were identified as potential GDM markers in umbilical cord serum (
Figure 3A). All of these amino acids exhibited statistically significant decreases in concentration during GDM development (
Figure 3B).
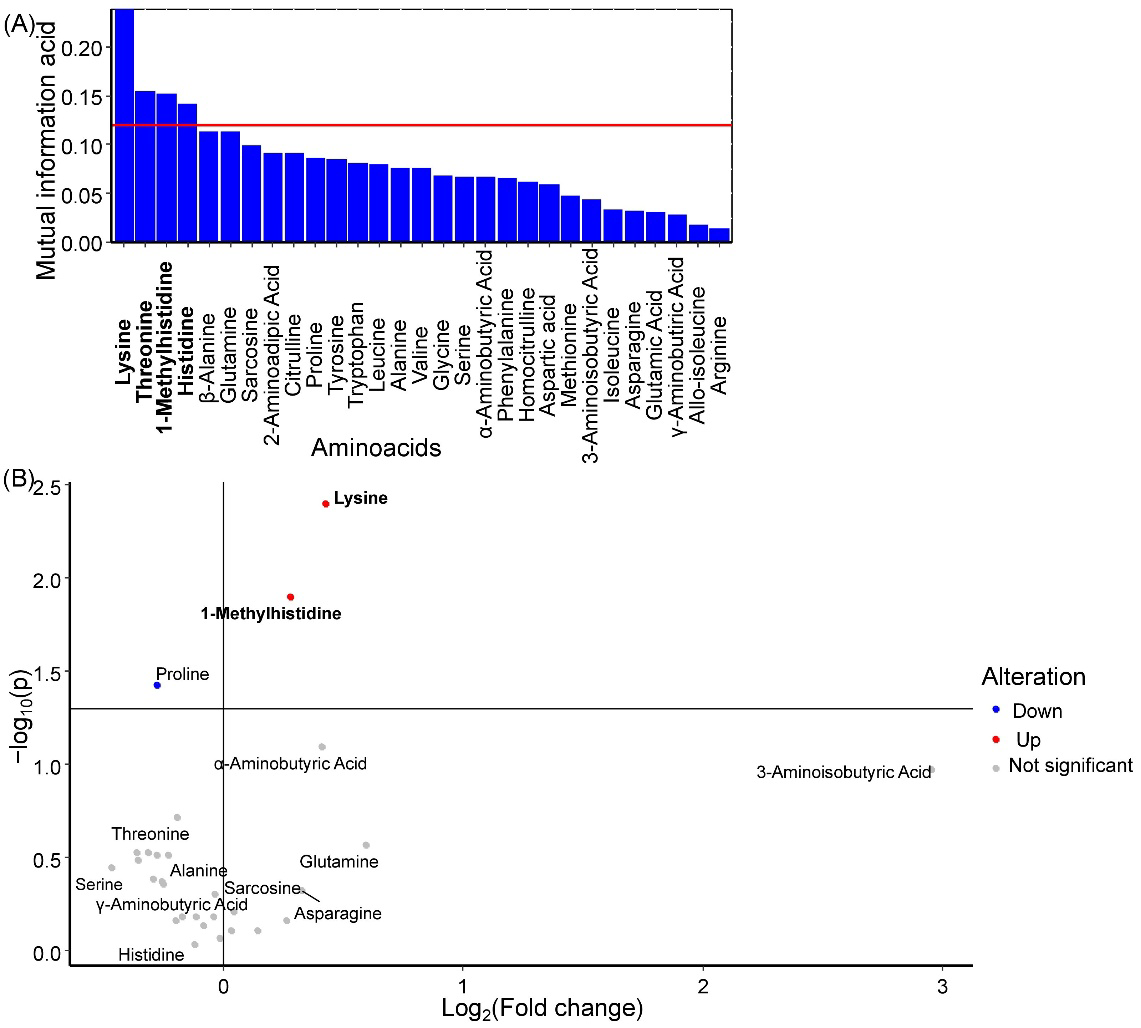
Lysine, 1. methylhistidine, and histidine were identified as potential GDM markers in amniotic fluid (
Figure 4A). Among these, lysine and 1-methylhistidine levels showed statistically significant increases during GDM development (
Figure 4B).
Decreased levels of lysine and 1-methylhistidine characterized the control group, whereas increased levels of these amino acids characterized the GDM group with normal-weight newborns. For GDM with normal-weight newborns, low levels of homocitrulline, asparagine, aminobutyric acid, and lysine, along with high levels of histidine, were characteristic (
Figure 5).
Total protein and fibrinogen concentrations in maternal blood prior to delivery exhibited strong inverse correlations with amino acid levels in maternal venous serum. The strongest associations were observed between total protein and proline (R = -0.56, p < 0.001), tryptophan (R = -0.52, p < 0.001), methionine (R = -0.49, p < 0.001), aspartic acid (R = -0.48, p < 0.001), alanine (R = -0.47, p < 0.001), asparagine (R = -0.46, p < 0.001), glutamine (R = -0.45, p < 0.001), tyrosine (R = -0.45, p < 0.001), phenylalanine (R = -0.43, p < 0.001), isoleucine (R = -0.43, p < 0.001), and citrulline (R = -0.41, p < 0.001). Fibrinogen showed strong inverse correlations with 1-methylhistidine (R = -0.44, p < 0.001), histidine (R = -0.42, p < 0.001), and γ-aminobutyric acid (R = -0.42, p < 0.001).
Pre-pregnancy BMI demonstrated weak positive correlations with 3-aminoisobutyric acid (R = 0.29, p = 0.02). Maternal BMI at delivery showed weak positive correlations with 3-aminoisobutyric acid (R = 0.28, p = 0.02) and 2-aminoadipic acid (R = 0.28, p = 0.02). Gestational weight gain exhibited weak inverse correlations with 3-aminoisobutyric acid (R = -0.26, p = 0.047) and homocitrulline (R = -0.29, p = 0.02), and weak positive correlations with proline (R = 0.26, p = 0.046), tyrosine (R = 0.27, p = 0.03), phenylalanine (R = 0.26, p = 0.045), and methionine (R = 0.33, p = 0.008) (
Figure S3).
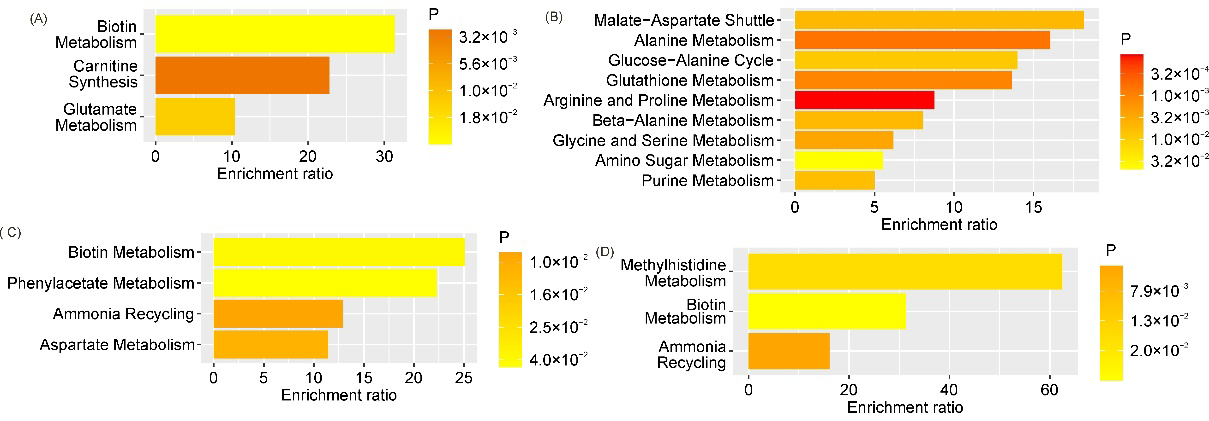
GDM markers in maternal venous serum significantly enriched pathways related to biotin metabolism, glutamate metabolism, and carnitine synthesis (
Figure 6A), whereas GDM markers in cord blood predominantly enriched pathways associated with amino acid metabolism, purine metabolism, and amino sugar metabolism (
Figure 6B). GDM markers in amniotic fluid significantly enriched pathways related to biotin metabolism, phenylacetate metabolism, aspartate metabolism, and the ammonia cycle (
Figure 6C). Similarly, markers of GDM with macrosomia significantly enriched ammonia cycle and biotin metabolism pathways. Additionally, the methylhistidine metabolism pathway was significantly enriched (
Figure 6D).
3. Discussion
The main risk factors for GDM in the studied cohort were high risk of PE (OR ≈ 6.75), family history of diabetes (OR ≈ 3.9), and elevated pre-pregnancy and delivery BMI. The heterogeneity of GDM is supported by the substantial proportion of women with normal weight (54%). Shared pathogenetic mechanisms with PE, evidence of systemic inflammation, and residual metabolic imbalance despite treatment underscore the need for improved therapeutic strategies. The absence of differences in most obstetric outcomes reflects the effectiveness of timely diagnosis and therapy.
These findings are fully consistent with the contemporary concept of GDM as a heterogeneous condition requiring a personalized approach. The Hyperglycemia and Adverse Pregnancy Outcome (HAPO) study [
23] demonstrated that the long-term consequences of GDM for mothers (risk of type 2 diabetes mellitus, cardiovascular disease) and offspring (obesity, impaired glucose tolerance) depend not only on the degree of hyperglycemia but also on baseline metabolic status, gestational weight gain, and genetic factors. The differences observed between subgroups 3 and 4 in the present study confirm the existence of distinct GDM phenotypes: “compensated” (with weight control and dietary therapy) and “decompensated” (with pathological weight gain, elevated glucose levels, and FM). This aligns with recent work in this area [
24,
25].
Current international guidelines (NICE, ACOG, FIGO) emphasize the need for a comprehensive approach to managing pregnant women with GDM, including not only glycemic control but also monitoring of gestational weight gain, screening for urinary tract infections, assessment of coagulation status, and consideration of risk factors such as history of macrosomia and family history of diabetes. The data obtained in this study confirm the importance of each of these components and justify the need for further prospective studies with larger sample sizes to develop more accurate prognostic models and therapeutic strategies.
This study revealed significant differences in the amino acid profiles of maternal serum, umbilical cord blood, and amniotic fluid between GDM and physiological pregnancies, and established an association between these alterations and the development of FM, opening new perspectives for understanding the pathogenesis of metabolic disturbances and their prediction. The findings demonstrate that GDM is accompanied by profound remodeling of amino acid metabolism, affecting not only the maternal organism but also the fetoplacental unit, with the pattern of these changes differing according to the presence or absence of FM. This allows consideration of specific amino acids as potential biomarkers for different clinical outcomes. Analysis of maternal serum identified glycine, 1-methylhistidine, γ-aminobutyric acid, lysine, and tryptophan as the most informative GDM markers, with all except tryptophan showing statistically significant concentration decreases during GDM development. This finding aligns with current understanding of enhanced amino acid consumption by the fetoplacental complex and impaired amino acid synthesis under conditions of insulin resistance [
26,
27].
Of particular note is the observed differential dynamics of lysine and 1-methylhistidine, which not only decreased in GDM overall but also exhibited specific alterations in the subgroup of women delivering normal-weight newborns, indicating the existence of distinct metabolic phenotypes within the GDM group and underscoring the heterogeneity of this condition. Analysis of the amino acid profile in umbilical cord blood revealed a substantially broader spectrum of alterations, including glutamine, glycine, asparagine, methionine, aspartic acid, γ-aminobutyric acid, glutamic acid, β-alanine, proline, citrulline, and alanine, all of which also showed decreased concentrations in GDM. These processes reflect impaired placental transport and altered metabolism in fetal tissues under conditions of hyperglycemia and hyperinsulinemia [
28]. Of particular interest is the identified association between levels of branched-chain amino acids, specifically leucine and allo-isoleucine, in cord blood and the development of FM, confirming the key role of these compounds in stimulating insulin secretion and activating anabolic processes in the fetal organism, leading to excessive adipose tissue accumulation and increased birth weight [
14].
Analysis of amniotic fluid identified lysine, threonine, 1-methylhistidine, and histidine as GDM markers; however, a different dynamic was observed here: lysine and 1-methylhistidine levels were significantly increased in GDM, which may be related to enhanced excretion of these amino acids by the fetal kidneys or to alterations in amniotic fluid composition resulting from polyuria characteristic of fetal hyperglycemia [
29,
30]. Interestingly, the GDM group subsequently delivering macrosomic infants was characterized by low levels of homocitrulline, asparagine, aminobutyric acid, and lysine alongside high histidine concentrations, whereas the opposite pattern predominated in the GDM group without macrosomia. This suggests distinct metabolic trajectories leading to excessive fetal weight gain and opens opportunities for early risk stratification.
Correlation analysis revealed a complex network of relationships between clinical parameters and amino acid profiles, with the strongest negative correlations observed between total protein and fibrinogen concentrations in maternal blood and the levels of numerous amino acids in both maternal and umbilical cord serum. This reflects the tight integration of protein and amino acid metabolism and their coordinated regulation under conditions of metabolic stress [
31].
Of particular interest are the observed correlations between newborn birth weight and concentrations of specific amino acids. Interestingly, maternal BMI both before pregnancy and at delivery showed weak but statistically significant correlations with a limited spectrum of amino acids, predominantly 3-aminoisobutyric acid and 2-aminoadipic acid, whereas gestational weight gain was associated with a broader range of metabolites, including proline, tyrosine, phenylalanine, and methionine. This underscores the importance of dynamic changes in body weight, rather than baseline anthropometric measures alone, in shaping the amino acid profile [
32]. Summary
Table S3 presents alterations in amino acid levels across the three biological compartments (maternal serum, umbilical cord serum, amniotic fluid) in GDM, as well as features characteristic of GDM cases complicated by FM.
The metabolic pathway enrichment analysis using the SMPDB database identified key biochemical processes disrupted in GDM and in GDM with FM. Markers of GDM in maternal serum were primarily associated with biotin metabolism, glutamate metabolism, and carnitine synthesis, whereas markers in cord blood enriched a broader spectrum of pathways, including amino acid metabolism, purine metabolism, and amino sugar metabolism, reflecting the systemic nature of metabolic disturbances in the fetus. In amniotic fluid, GDM markers were associated with biotin metabolism, phenylacetate metabolism, aspartate metabolism, and the ammonia cycle, whereas in GDM with FM, the methylhistidine metabolism pathway was additionally enriched. This may be directly related to altered muscle metabolism and changes in body composition in macrosomic newborns. Summary data are presented in
Table S4.
The amino acid profile undergoes systemic changes across compartments (mother, umbilical cord, AF); however, these changes are not mirror-like. For example, 1-methylhistidine decreases in maternal blood but increases in AF, highlighting the importance of investigating multiple compartments to understand the complete picture. Decreases in glycine and GABA indicate oxidative stress, inflammation, and impaired neuro/metabolic regulation. Reduced levels of glutamine, asparagine, and citrulline in umbilical cord blood reflect fetoplacental unit dysfunction, including impaired nutrient transport, endothelial dysfunction, and possibly intrauterine fetal metabolic stress.
Amniotic fluid serves as a unique “window” into fetal status. Alterations in AF (increased 1-methylhistidine and lysine) may reflect not so much placental transport as fetal renal function, skin and lung condition, and meconium composition [
33]. This makes AF a valuable specimen for assessing the fetal response to maternal hyperglycemia. The observed changes (particularly glycine and glutamine deficiency) point to potential targets for nutritional intervention or biochemical monitoring aimed at reducing fetal risks. However, this requires further investigation.
An important methodological limitation for interpretation: all conclusions drawn from this study are preliminary (hypothesis-generating) in nature due to the small sample size. They indicate interesting trends and biological mechanisms that should be confirmed in an independent, larger-scale cohort. It is important to note that in 2025, we published an article entitled “Amino Acid Profile Alterations in the Mother-Fetus System in Gestational Diabetes Mellitus and Macrosomia” [
15], based on the results of a two-center study (Moscow + Tomsk) conducted from January 2024 to March 2025, which included 94 mother-fetus dyads (GDM group: 53, control group: 41; GDM+FM: 23, GDM+normosomia: 30, control: 36). Comparative analysis of data from the Tomsk patient cohort and the Moscow cohort (the present publication) revealed high consensus on key markers: glutamine, histidine, and asparagine were consistently decreased in GDM in maternal blood; isoleucine, leucine, and serine were consistently increased in FM in cord blood and AF; GABA decrease was confirmed in both studies (marker of metabolic dysregulation). Both studies confirm the involvement of amino acid transport, glutamate metabolism, and the urea cycle. Lysine and isoleucine were the most consistent positive correlates of fetal weight; homocitrulline was a consistent negative correlate of fetal weight. Both studies demonstrate high consistency of results, despite different cohorts and sample sizes. The identified markers (glutamine, histidine, asparagine in maternal blood; isoleucine, leucine, serine in cord blood and AF) can be considered valid candidates for the development of diagnostic panels. The differential dynamics of amino acids across different biological compartments underscore the need for a comprehensive approach to assessing the mother-fetus system and open prospects for early diagnosis and personalized correction of metabolic disturbances in GDM and FM.
Author Contributions
Conceptualization, N.F. and A.T.; methodology, N.F.; software, A.T. and V.C.; validation, A.D. and V.F.; formal analysis, A.T. and V.C.; investigation, N.F., A.D., A.N., and V.C.; resources, V.F. and G.S.; data curation, A.T. and V.F.; writing—original draft preparation, N.F.; writing—review and editing, N.F., A.T., and V.F.; visualization, A.D.; supervision, V.F.; project administration, G.S.; funding acquisition, V.F. and G.S. All authors have read and agreed to the published version of the manuscript.