Submitted:
02 March 2026
Posted:
03 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
- 1.
- A critical assessment of the influence of the equation of state on the prediction of thermophysical properties for supercritical and its mixtures,
- 2.
- rich mixtures depressurization analysis of the flow rate, and pressure depression and void fraction profiles with different outlet diameters and geometries,
- 3.
- An evaluation of the effect of impurities by simulating binary mixture on the decompression wave propagation and the resulting critical mass flow rate.
2. Numerical Methodology
2.1. Governing Equations
2.2. Thermodynamic Modeling
- Property evaluation as function ,
- Reverse lookup after solving for internal energy.
2.3. Numerical Schemes
3. Computational Setup
3.1. Setup Description
3.2. Mesh Sensitivity
4. Results and Discussions
4.1. Heat Transfer and Boundary Conditions
4.2. Effect of Equation of State
4.3. Effect of Impurity
4.3.1. Comparison with Pure
4.3.2. Comparison Between Different Impurities
4.3.3. Role of Impurity Mass Fraction
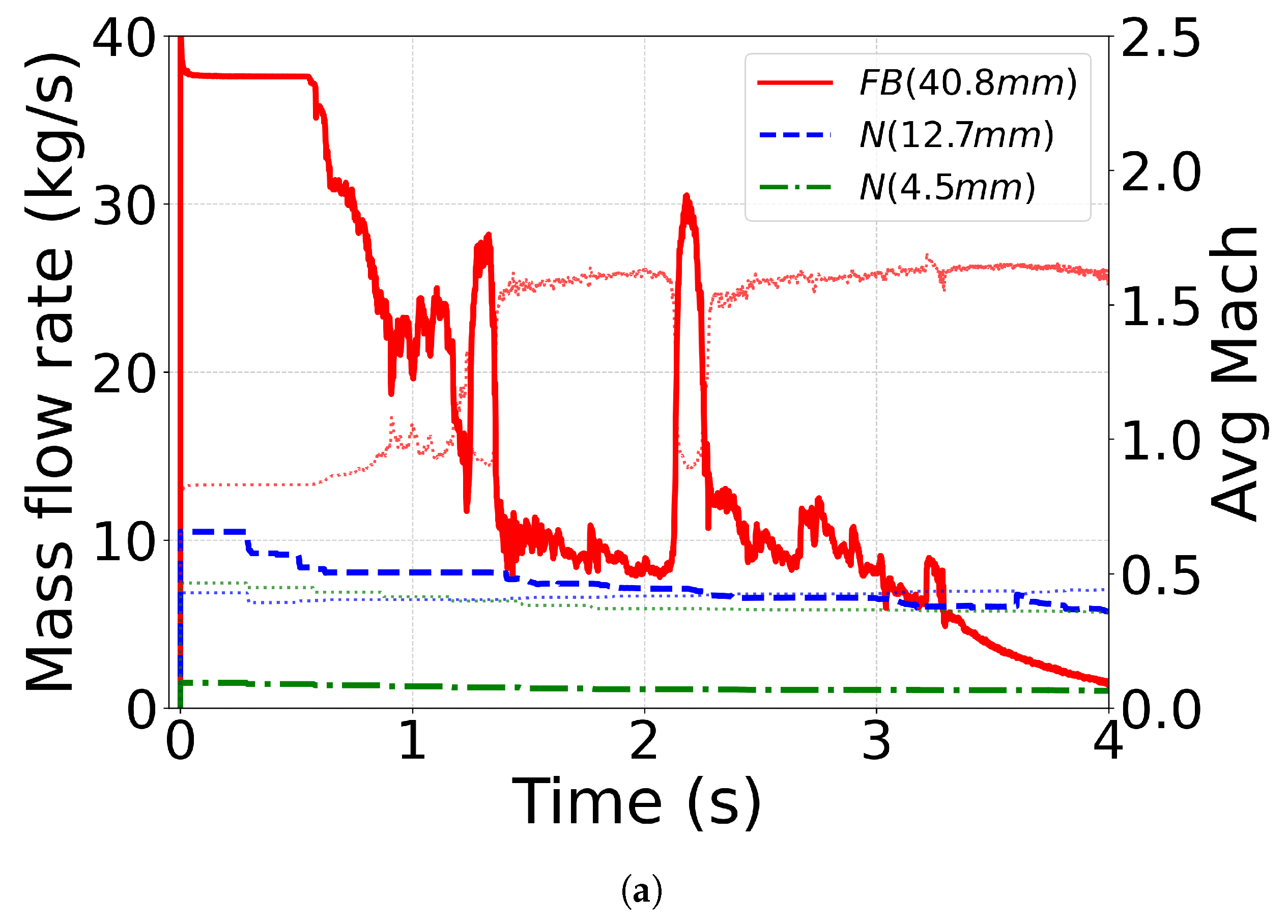
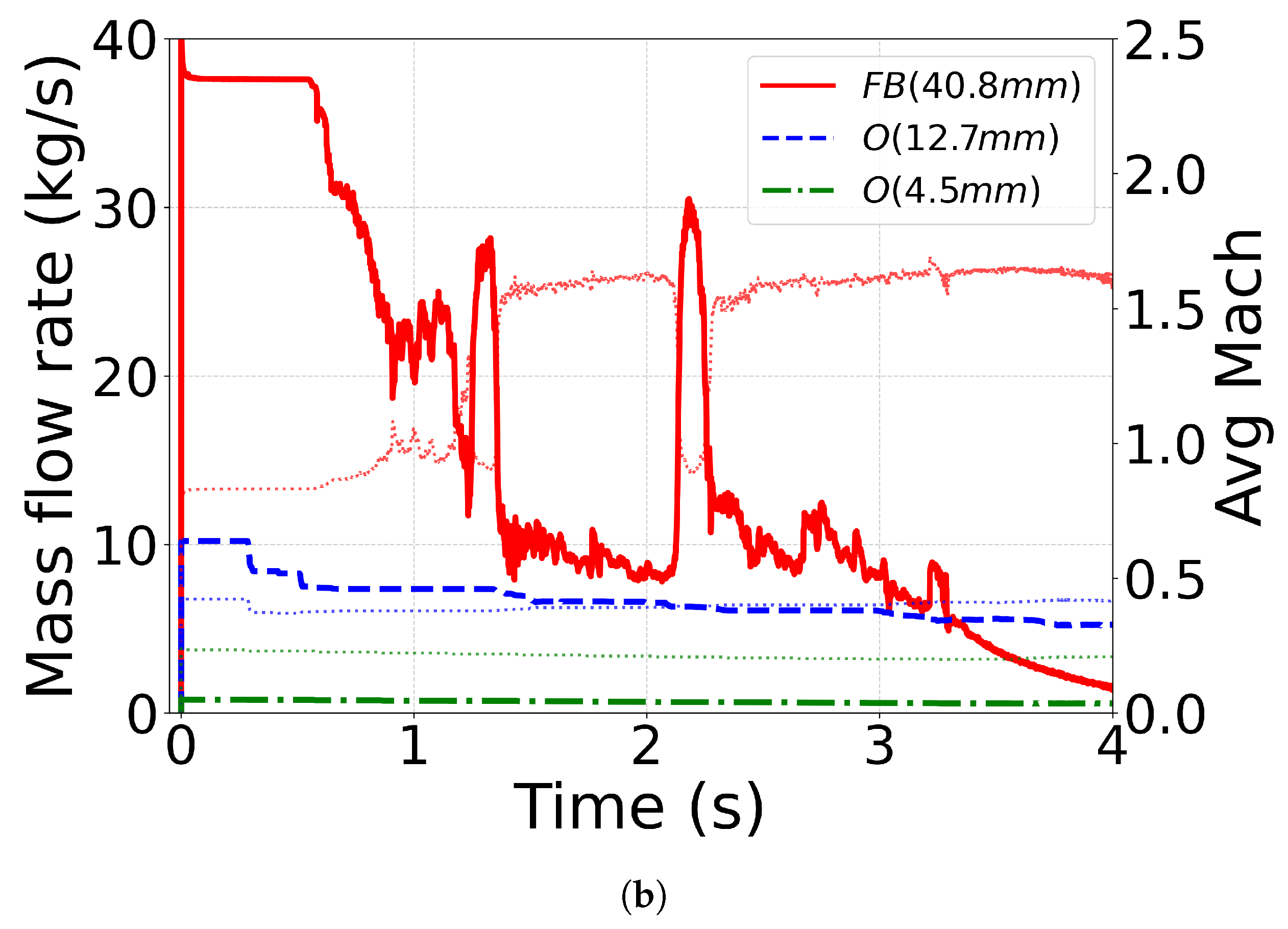
4.4. Effect of Outlet Geometry and Diameter
4.5. Mass-Flow Rate and Mach Number
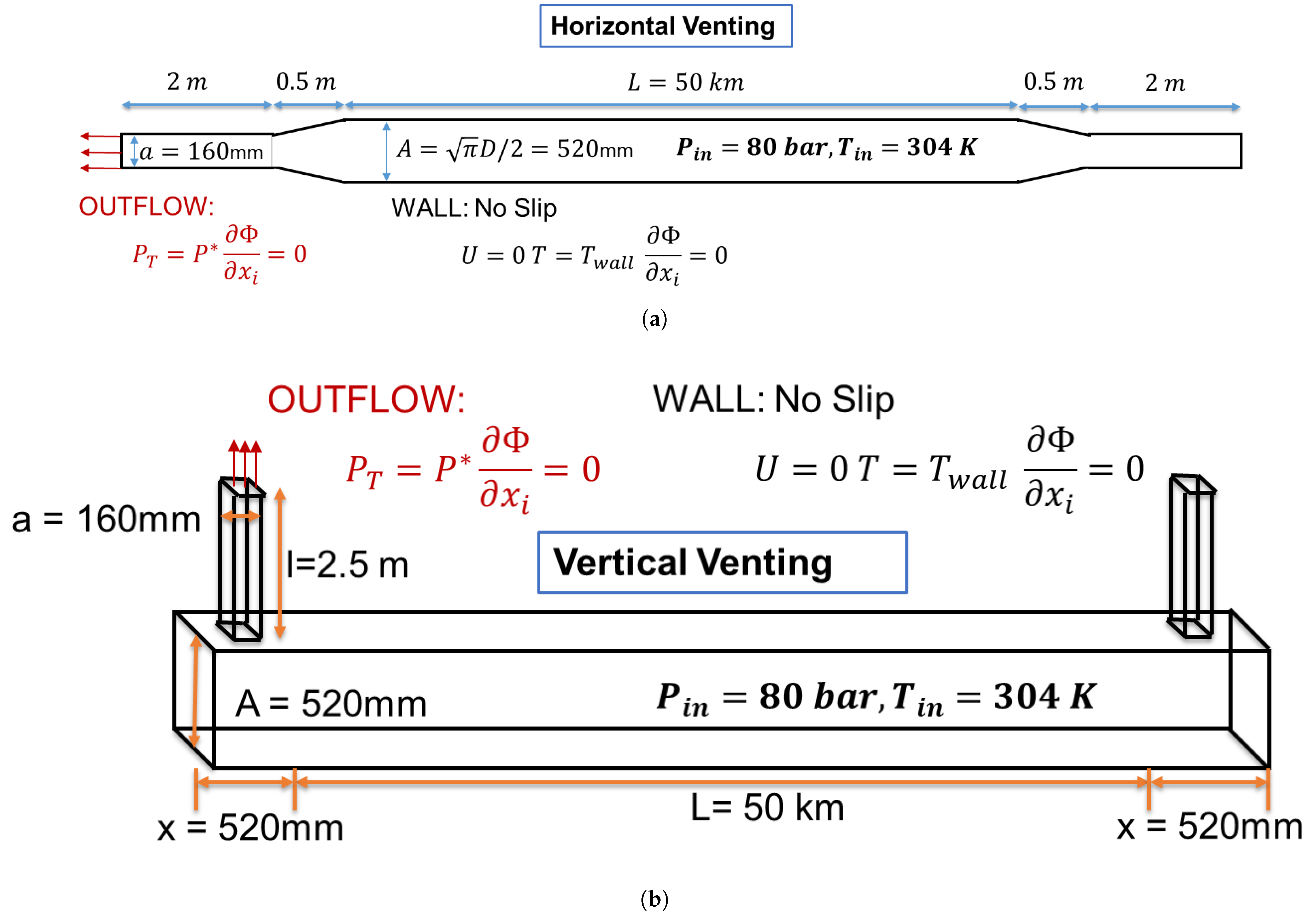
5. Pipeline Transport: Industrial Scale Simulation
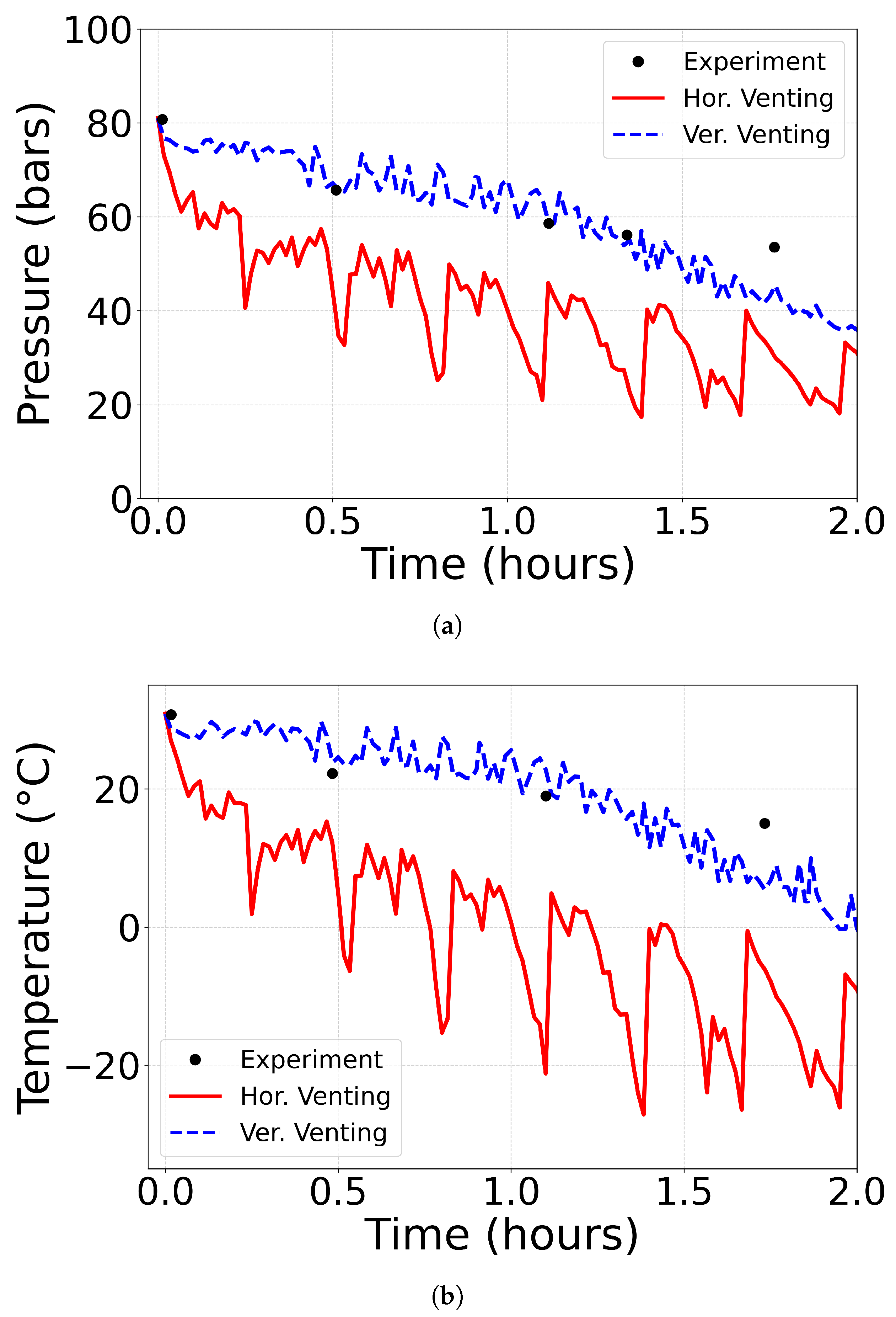
5.1. Setup of the 50km Pipeline with Venting
5.2. Pressure and Temperature Profiles
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CCUS | Carbon Capture and Storage |
| RFM | Real Fluid Method |
| Supercritical | |
| VLE | Vapor Liquid Equilibrium |
| EoS | Equation of State |
| PR | Peng Robinson |
| CPA | Cubic Plus Association |
| SAFT | Perturbed-Chain Statistical Associating Fluid Theory |
| Mass flow rate at the transition from dense phase to two-phase | |
| Mass flow rate at the transition from two-phase to gaseous phase |
References
- Eide, L.I.; Batum, M.; Dixon, T.; Elamin, Z.; Graue, A.; Hagen, S.; Hovorka, S.; Nazarian, B.; Nøkleby, P.H.; Olsen, G.I.; et al. Enabling large-scale carbon capture, utilisation, and storage (CCUS) using offshore carbon dioxide (CO2) infrastructure developments—a review. Energies 2019, 12, 1945. [Google Scholar] [CrossRef]
- Rui, Z.; Zeng, L.; Dindoruk, B. Challenges in the large-scale deployment of CCUS. Engineering 2025, 44, 17–20. [Google Scholar] [CrossRef]
- Boot-Handford, M.E.; Abanades, J.C.; Anthony, E.J.; Blunt, M.J.; Brandani, S.; Mac Dowell, N.; Fernández, J.R.; Ferrari, M.C.; Gross, R.; Hallett, J.P.; et al. Carbon capture and storage update. Energy & Environmental Science 2014, 7, 130–189. [Google Scholar]
- Leung, D.Y.; Caramanna, G.; Maroto-Valer, M.M. An overview of current status of carbon dioxide capture and storage technologies. Renewable and sustainable energy reviews 2014, 39, 426–443. [Google Scholar] [CrossRef]
- Haszeldine, R.S. Carbon capture and storage: how green can black be? Science 2009, 325, 1647–1652. [Google Scholar] [CrossRef]
- Wareing, C.J.; Fairweather, M.; Falle, S.A.; Woolley, R.M. Modelling ruptures of buried high pressure dense phase CO2 pipelines in carbon capture and storage applications—Part I. Validation. International Journal of Greenhouse Gas Control 2015, 42, 701–711. [Google Scholar] [CrossRef]
- Martynov, S.; Zheng, W.; Mahgerefteh, H.; Brown, S.; Hebrard, J.; Jamois, D.; Proust, C. Computational and experimental study of solid-phase formation during the decompression of high-pressure CO2 pipelines. Industrial & Engineering Chemistry Research 2018, 57, 7054–7063. [Google Scholar]
- Brown, S.; Martynov, S.; Mahgerefteh, H.; Chen, S.; Zhang, Y. Modelling the non-equilibrium two-phase flow during depressurisation of CO2 pipelines. International Journal of Greenhouse Gas Control 2014, 30, 9–18. [Google Scholar] [CrossRef]
- Peletiri, S.P.; Rahmanian, N.; Mujtaba, I.M. CO2 Pipeline design: A review. Energies 2018, 11, 2184. [Google Scholar] [CrossRef]
- Bielka, P.; Kuczyński, S.; Włodek, T.; Nagy, S. Risks and safety of CO2 pipeline transport: a case study of the analysis and modeling of the risk of accidental release of CO2 into the atmosphere. Energies 2024, 17, 3943. [Google Scholar] [CrossRef]
- Lu, H.; Ma, X.; Huang, K.; Fu, L.; Azimi, M. Carbon dioxide transport via pipelines: A systematic review. Journal of Cleaner Production 2020, 266, 121994. [Google Scholar] [CrossRef]
- Mahgerefteh, H.; Brown, S.; Martynov, S. A study of the effects of friction, heat transfer, and stream impurities on the decompression behavior in CO2 pipelines. Greenhouse gases: science and technology 2012, 2, 369–379. [Google Scholar] [CrossRef]
- Woolley, R.M.; Fairweather, M.; Wareing, C.J.; Falle, S.A.; Mahgerefteh, H.; Martynov, S.; Brown, S.; Narasimhamurthy, V.D.; Storvik, I.E.; Saelen, L.; et al. CO2PipeHaz: quantitative hazard assessment for next generation CO2 pipelines. Energy Procedia 2014, 63, 2510–2529. [Google Scholar] [CrossRef]
- Wareing, C.J.; Fairweather, M.; Woolley, R.M.; Falle, S.A. Numerical simulation of CO2 dispersion from punctures and ruptures of buried high-pressure dense phase CO2 pipelines with experimental validation. Energy Procedia 2014, 63, 2500–2509. [Google Scholar] [CrossRef]
- Pham, L.H.H.P.; Rusli, R. A review of experimental and modelling methods for accidental release behaviour of high-pressurised CO2 pipelines at atmospheric environment. Process Safety and Environmental Protection 2016, 104, 48–84. [Google Scholar] [CrossRef]
- Munkejord, S.T.; Hammer, M.; Løvseth, S.W. CO2 transport: Data and models–A review. Applied Energy 2016, 169, 499–523. [Google Scholar] [CrossRef]
- Vitali, M.; Corvaro, F.; Marchetti, B.; Terenzi, A. Thermodynamic challenges for CO2 pipelines design: A critical review on the effects of impurities, water content, and low temperature. International Journal of Greenhouse Gas Control 2022, 114, 103605. [Google Scholar] [CrossRef]
- Shang, Y.; Chen, X.; Yang, M.; Xing, X.; Jiao, J.; An, G.; Li, X.; Xiong, X. Comprehensive review on leakage characteristics and diffusion laws of carbon dioxide pipelines. Energy & Fuels 2024, 38, 10456–10493. [Google Scholar]
- Gu, S.; Li, Y.; Teng, L.; Hu, Q.; Zhang, D.; Ye, X.; Wang, C.; Wang, J.; Iglauer, S. A new model for predicting the decompression behavior of CO2 mixtures in various phases. Process Safety and Environmental Protection 2018, 120, 237–247. [Google Scholar] [CrossRef]
- Magen, O.; Kozak, Y.; Di Lucchio, L.; Marengo, M.; Bar-Kohany, T. Predicting nucleation pressure under rapid depressurization: Bridging positive and negative pressure regions. International Journal of Heat and Mass Transfer 2025, 251, 127309. [Google Scholar] [CrossRef]
- He, X.; Gao, P.; Wang, J.; Zhang, Z. Experimental investigation on hot water rapid depressurization process characteristics inside and outside of the ruptured pipeline. Nuclear Engineering and Design 2024, 425, 113341. [Google Scholar] [CrossRef]
- Liao, H.; Wang, X.; Yang, K.; Hou, Z.; Wang, H. Impurity-driven variations in CO2 critical flow dynamics: Modeling approaches for enhanced CCS safety. Energy 2025, 323, 135850. [Google Scholar] [CrossRef]
- Cao, Q.; Yan, X.; Liu, S.; Yu, J.; Chen, S.; Zhang, Y. Temperature and phase evolution and density distribution in cross section and sound evolution during the release of dense CO2 from a large-scale pipeline. International Journal of Greenhouse Gas Control 2020, 96, 103011. [Google Scholar] [CrossRef]
- Hansen, P.M.; Gaathaug, A.V.; Bjerketvedt, D.; Vaagsaether, K. The behavior of pressurized liquefied CO2 in a vertical tube after venting through the top. International Journal of Heat and Mass Transfer 2017, 108, 2011–2020. [Google Scholar] [CrossRef]
- Hansen, P.M.; Gaathaug, A.V.; Bjerketvedt, D.; Vaagsaether, K. Rapid depressurization and phase transition of CO2 in vertical ducts–small-scale experiments and Rankine-Hugoniot analyses. Journal of Hazardous Materials 2019, 365, 16–25. [Google Scholar] [CrossRef]
- Bhuvankar, P.; Cihan, A.; Birkholzer, J. A framework to simulate the blowout of CO2 through wells in geologic carbon storage. International Journal of Greenhouse Gas Control 2023, 127, 103921. [Google Scholar] [CrossRef]
- Yin, B.; Huang, W.; Ouyang, X.; Zhao, X.; Meng, L.; Zhang, L.; Hu, Q.; Li, Y. A transient model considering non-equilibrium phase transition in CO2 pipe decompression. Journal of Pipeline Science and Engineering 2025, 100324. [Google Scholar] [CrossRef]
- Log, A.M.; Hammer, M.; Deng, H.; Austegard, A.; Hafner, A.; Munkejord, S.T. Depressurization of CO2 in a pipe: Effect of initial state on non-equilibrium two-phase flow. International Journal of Multiphase Flow 2024, 170, 104624. [Google Scholar] [CrossRef]
- Wang, J.; Lu, Z.; Wu, S.; Zhang, Z.; Liu, J. Research on non-equilibrium characteristics and relaxation time parameter optimization in high-pressure CO2 pipeline depressurization. International Communications in Heat and Mass Transfer 2025, 169, 109527. [Google Scholar] [CrossRef]
- Log, A.M.; Hammer, M.; Munkejord, S.T. A flashing flow model for the rapid depressurization of CO2 in a pipe accounting for bubble nucleation and growth. International Journal of Multiphase Flow 2024, 171, 104666. [Google Scholar] [CrossRef]
- Munkejord, S.T.; Deng, H.; Austegard, A.; Hammer, M.; Aasen, A.; Skarsvåg, H.L. Depressurization of CO2-N2 and CO2-He in a pipe: Experiments and modelling of pressure and temperature dynamics. International Journal of Greenhouse Gas Control 2021, 109, 103361. [Google Scholar] [CrossRef]
- Yu, S.; Yan, X.; He, Y.; Chen, L.; Hu, Y.; Yang, K.; Cao, Z.; Yu, J.; Chen, S. Study on the decompression behavior during large-scale pipeline puncture releases of CO2 with various N2 compositions: Experiments and mechanism analysis. Energy 2024, 296, 131180. [Google Scholar] [CrossRef]
- Zhu, J.; Wu, J.; Xie, N.; Li, Z.; Hu, Q.; Li, Y. Study on water hammer phase transition characteristics of dense/liquid phase CO2 pipeline. Energy 2024, 311, 133470. [Google Scholar] [CrossRef]
- Liao, H.; Yang, K.; Liang, Z.; Hu, H.; Wang, X.; Wang, H. A new paradigm in critical flow analysis: Combining Buckingham Pi theorem with neural network for improved predictions in microchannels. Chemical Engineering Science 2024, 299, 120483. [Google Scholar] [CrossRef]
- Yu, S.; Yan, X.; He, Y.; Hu, Y.; Qiao, F.; Yang, K.; Cao, Z.; Chen, L.; Liu, Z.; Yu, J.; et al. Study on the effect of valve openings and multi-stage throttling structures on the pressure and temperature during CO2 pipeline venting processes. Energy 2024, 308, 132967. [Google Scholar] [CrossRef]
- Ding, Y.; Xu, P.; Lu, Y.; Yang, M.; Zhang, J.; Liu, K. Research on pipeline leakage calculation and correction method based on numerical calculation method. Energies 2023, 16, 7255. [Google Scholar] [CrossRef]
- Zhang, Z.; Lu, Z.; Yan, L.; Wang, J.; Yao, S. Experiment and numerical investigation on flow characteristics and near-field structure of dense phase CO2 pipeline leakage. Process Safety and Environmental Protection 2024, 182, 327–344. [Google Scholar] [CrossRef]
- Yu, S.; Yan, X.; He, Y.; Yu, J.; Chen, S. Study on the leakage morphology and temperature variations in the soil zone during large-scale buried CO2 pipeline leakage. Energy 2024, 288, 129674. [Google Scholar] [CrossRef]
- Shang, Y.; Xing, X.; Chen, X.; Yang, M.; Shah, R.K.; Pang, X. Developing Transient Model and Simulating the Effects of Soil Properties on a Small Hole Leakage and Diffusion Characteristics in the Buried CO2 Pipelines. Energy & Fuels 2025. [Google Scholar]
- Chen, L.; Yan, X.; Hu, Y.; Yang, K.; Yu, S.; Yu, J.; Chen, S. Depressurization and heat transfer during leakage of supercritical CO2 from a pipeline. Greenhouse Gases: Science and Technology 2022, 12, 616–628. [Google Scholar] [CrossRef]
- Hu, Y.; Han, H.; Jing, R.; Hu, Q.; Li, Y. Experimental analysis of pressure response and jet behavior in underwater supercritical CO2 pipeline leakage. Ocean Engineering 2025, 341, 122469. [Google Scholar] [CrossRef]
- Span, R.; Wagner, W. A new equation of state for carbon dioxide covering the fluid region from the triple-point temperature to 1100 K at pressures up to 800 MPa. Journal of physical and chemical reference data 1996, 25, 1509–1596. [Google Scholar] [CrossRef]
- Nazeri, M.; Chapoy, A.; Burgass, R.; Tohidi, B. Measured densities and derived thermodynamic properties of CO2-rich mixtures in gas, liquid and supercritical phases from 273 K to 423 K and pressures up to 126 MPa. The Journal of Chemical Thermodynamics 2017, 111, 157–172. [Google Scholar] [CrossRef]
- Demetriades, T.A.; Graham, R.S. A new equation of state for CCS pipeline transport: Calibration of mixing rules for binary mixtures of CO2 with N2, O2 and H2. The Journal of Chemical Thermodynamics 2016, 93, 294–304. [Google Scholar] [CrossRef]
- Kunz, O.; Wagner, W. The GERG-2008 wide-range equation of state for natural gases and other mixtures: An expansion of GERG-2004. Journal of chemical & engineering data 2012, 57, 3032–3091. [Google Scholar]
- Petropoulou, E.; Voutsas, E.; Westman, S.F.; Austegard, A.; Stang, H.G.J.; Løvseth, S.W. Vapor-liquid equilibrium of the carbon dioxide/methane mixture at three isotherms. Fluid Phase Equilibria 2018, 462, 44–58. [Google Scholar] [CrossRef]
- Li, H.; Yan, J. Evaluating cubic equations of state for calculation of vapor–liquid equilibrium of CO2 and CO2-mixtures for CO2 capture and storage processes. Applied Energy 2009, 86, 826–836. [Google Scholar] [CrossRef]
- Diamantonis, N.I.; Boulougouris, G.C.; Mansoor, E.; Tsangaris, D.M.; Economou, I.G. Evaluation of cubic, SAFT, and PC-SAFT equations of state for the vapor–liquid equilibrium modeling of CO2 mixtures with other gases. Industrial & Engineering Chemistry Research 2013, 52, 3933–3942. [Google Scholar]
- Gross, J.; Sadowski, G. Perturbed-chain SAFT: An equation of state based on a perturbation theory for chain molecules. Industrial & engineering chemistry research 2001, 40, 1244–1260. [Google Scholar]
- Avendano, C.; Lafitte, T.; Galindo, A.; Adjiman, C.S.; Jackson, G.; Müller, E.A. SAFT-γ force field for the simulation of molecular fluids. 1. A single-site coarse grained model of carbon dioxide. The Journal of Physical Chemistry B 2011, 115, 11154–11169. [Google Scholar] [CrossRef]
- Lymperiadis, A.; Adjiman, C.S.; Galindo, A.; Jackson, G. A group contribution method for associating chain molecules based on the statistical associating fluid theory (SAFT-γ). The Journal of chemical physics 2007, 127. [Google Scholar] [CrossRef]
- Bahadori, A.; Vuthaluru, H.B. Predictive tool for an accurate estimation of carbon dioxide transport properties. International Journal of Greenhouse Gas Control 2010, 4, 532–536. [Google Scholar] [CrossRef]
- Nazeri, M.; Chapoy, A.; Valtz, A.; Coquelet, C.; Tohidi, B. New experimental density data and derived thermophysical properties of carbon dioxide–Sulphur dioxide binary mixture (CO2-SO2) in gas, liquid and supercritical phases from 273 K to 353 K and at pressures up to 42 MPa. Fluid Phase Equilibria 2017, 454, 64–77. [Google Scholar] [CrossRef]
- Jarrahian, A.; Heidaryan, E. A novel correlation approach to estimate thermal conductivity of pure carbon dioxide in the supercritical region. The Journal of Supercritical Fluids 2012, 64, 39–45. [Google Scholar] [CrossRef]
- Rostami, A.; Arabloo, M.; Ebadi, H. Genetic programming (GP) approach for prediction of supercritical CO2 thermal conductivity. Chemical Engineering Research and Design 2017, 122, 164–175. [Google Scholar] [CrossRef]
- Hellmann, R.; Bich, E.; Vesovic, V. Cross second virial coefficients and dilute gas transport properties of the (CH4+ CO2),(CH4+ H2S), and (H2S+ CO2) systems from accurate intermolecular potential energy surfaces. The Journal of Chemical Thermodynamics 2016, 102, 429–441. [Google Scholar] [CrossRef]
- Li, H.; Wilhelmsen; Lv, Y.; Wang, W.; Yan, J. Viscosities, thermal conductivities and diffusion coefficients of CO2 mixtures: Review of experimental data and theoretical models. International Journal of Greenhouse Gas Control 2011, 5, 1119–1139. [Google Scholar] [CrossRef]
- Trusler Martin, J. Equation of state for solid phase I of carbon dioxide valid for temperatures up to 800 K and pressures up to 12 GPa. Journal of Physical and Chemical Reference Data 2011, 40. [Google Scholar] [CrossRef]
- Trusler Martin, J. Erratum: Equation of state for solid phase I of carbon dioxide valid for temperatures up to 800 K and pressures up to 12 GPa [J. Phys. Chem. Ref. Data 40, 043105 (2011)]. Journal of Physical and Chemical Reference Data2012, 41, 039901..
- Jäger, A.; Span, R. Equation of state for solid carbon dioxide based on the Gibbs free energy. Journal of Chemical & Engineering Data 2012, 57, 590–597. [Google Scholar] [CrossRef]
- Nikolaidis, I.K.; Boulougouris, G.C.; Peristeras, L.D.; Economou, I.G. Equation-of-State Modeling of Solid–Liquid–Gas Equilibrium of CO2 Binary Mixtures. Industrial & engineering chemistry research 2016, 55, 6213–6226. [Google Scholar]
- Løvseth, S.W.; Austegard, A.; Westman, S.F.; Stang, H.G.J.; Herrig, S.; Neumann, T.; Span, R. Thermodynamics of the carbon dioxide plus argon (CO2+ ar) system: An improved reference mixture model and measurements of vapor-liquid, vapor-solid, liquid-solid and vapor-liquid-solid phase equilibrium data at the temperatures 213–299 k and pressures up to 16 mpa. Fluid Phase Equilibria 2018, 466, 48–78. [Google Scholar]
- Tang, L.; Li, C.; Lim, S. Solid–liquid–vapor equilibrium model applied for a CH4–CO2 binary mixture. Industrial & Engineering Chemistry Research 2019, 58, 18355–18366. [Google Scholar]
- Maltby, T.W.; Aasen, A.; Hammer, M.; Wilhelmsen. Review of Experimental Data and Evaluation of Equations of State for Modeling Formation of Solid CO2 in CCS and Natural Gas Applications. Industrial & Engineering Chemistry Research 2025. [Google Scholar]
- Bhatia, H.; Habchi, C. Real Fluid Modeling and Simulation of the Structures and Dynamics of Condensation in CO2 Flows Shocked Inside a de Laval Nozzle, Considering the Effects of Impurities. Applied Sciences 2023, 13, 10863. [Google Scholar] [CrossRef]
- Yang, S.; Yi, P.; Habchi, C. Real-fluid injection modeling and LES simulation of the ECN Spray A injector using a fully compressible two-phase flow approach. International Journal of Multiphase Flow 2020, 122, 103145. [Google Scholar] [CrossRef]
- Yi, P.; Yang, S.; Habchi, C.; Lugo, R. A multicomponent real-fluid fully compressible four-equation model for two-phase flow with phase change. Physics of Fluids 2019, 31. [Google Scholar] [CrossRef]
- Gaballa, H.; Jafari, S.; Habchi, C.; de Hemptinne, J.C. Numerical investigation of droplet evaporation in high-pressure dual-fuel conditions using a tabulated real-fluid model. International Journal of Heat and Mass Transfer 2022, 189, 122671. [Google Scholar] [CrossRef]
- de Hemptinne, J.C.; Ferrando, N.; Hajiw-Riberaud, M.; Lachet, V.; Maghsoodloo, S.; Mougin, P.; Ngo, T.D.; Pigeon, L.; Yanes, J.R.; Wender, A. Carnot: a thermodynamic library for energy industries. Science and Technology for Energy Transition 2023, 78, 30. [Google Scholar] [CrossRef]
- Munkejord, S.T.; Austegard, A.; Deng, H.; Hammer, M.; Stang, H.J.; Løvseth, S.W. Depressurization of CO2 in a pipe: High-resolution pressure and temperature data and comparison with model predictions. Energy 2020, 211, 118560. [Google Scholar] [CrossRef]
- Hammer, M.; Deng, H.; Austegard, A.; Log, A.M.; Munkejord, S.T. Experiments and modelling of choked flow of CO2 in orifices and nozzles. International Journal of Multiphase Flow 2022, 156, 104201. [Google Scholar] [CrossRef]
- GRTgaz, S.A. Proposition de spécifications dioxyde de carbone. Technical report, GRTgaz S.A. Technical report. 2023. [Google Scholar]
- Habchi, C.; Gaballa, H.; de Hemptinne, J.C. A new real-fluid modelling framework applied to cavitation simulation. In Proceedings of the 12th International Cavitation – CAV2024, CHANIA, GREECE, 2024; pp. 2–7 June. [Google Scholar]
- Jafari, S.; Gaballa, H.; Habchi, C.; de Hemptinne, J.C. Towards understanding the structure of subcritical and transcritical liquid–gas interfaces using a tabulated real fluid modeling approach. Energies 2021, 14, 5621. [Google Scholar] [CrossRef]
- Jafari, S.; Gaballa, H.; Habchi, C.; De Hemptinne, J.C.; Mougin, P. Exploring the interaction between phase separation and turbulent fluid dynamics in multi-species supercritical jets using a tabulated real-fluid model. The Journal of Supercritical Fluids 2022, 184, 105557. [Google Scholar] [CrossRef]
- Gaballa, H.; Habchi, C.; de Hemptinne, J.C. Modeling and LES of high-pressure liquid injection under evaporating and non-evaporating conditions by a real fluid model and surface density approach. International Journal of Multiphase Flow 2023, 160, 104372. [Google Scholar] [CrossRef]
- Chung, T.H.; Ajlan, M.; Lee, L.L.; Starling, K.E. Generalized multiparameter correlation for nonpolar and polar fluid transport properties. Industrial & engineering chemistry research 1988, 27, 671–679. [Google Scholar]
- Nicoud, F.; Toda, H.B.; Cabrit, O.; Bose, S.; Lee, J. Using singular values to build a subgrid-scale model for large eddy simulations. Physics of fluids 2011, 23. [Google Scholar] [CrossRef]
- Peng, D.Y.; Robinson, D.B. A new two-constant equation of state. Industrial & Engineering Chemistry Fundamentals 1976, 15, 59–64. [Google Scholar] [CrossRef]
- Kontogeorgis, G.M.; Voutsas, E.C.; Yakoumis, I.V.; Tassios, D.P. An equation of state for associating fluids. Industrial & engineering chemistry research 1996, 35, 4310–4318. [Google Scholar]
- Ware, C.; Knight, W.; Wells, D. Memory intensive statistical algorithms for multibeam bathymetric data. Computers & Geosciences 1991, 17, 985–993. [Google Scholar] [CrossRef]
- Gaballa, H.; Jafari, S.; Di-Lella, A.; Habchi, C.; et al. A tabulated real-fluid modeling approach applied to renewable dual-fuel evaporation and mixing: Paper 216. In Proceedings of the International Conference on Liquid Atomization and Spray Systems (ICLASS), 2021; Vol. 1. [Google Scholar]
- Senecal, P.; Pomraning, E.; Richards, K.; Som, S. Grid-convergent spray models for internal combustion engine computational fluid dynamics simulations. Journal of Energy Resources Technology 2014, 136, 012204. [Google Scholar] [CrossRef]
- Clausen, S.; Oosterkamp, A.; Strøm, K.L. Depressurization of a 50 km long 24 inches CO2 pipeline. Energy Procedia 2012, 23, 256–265. [Google Scholar] [CrossRef]
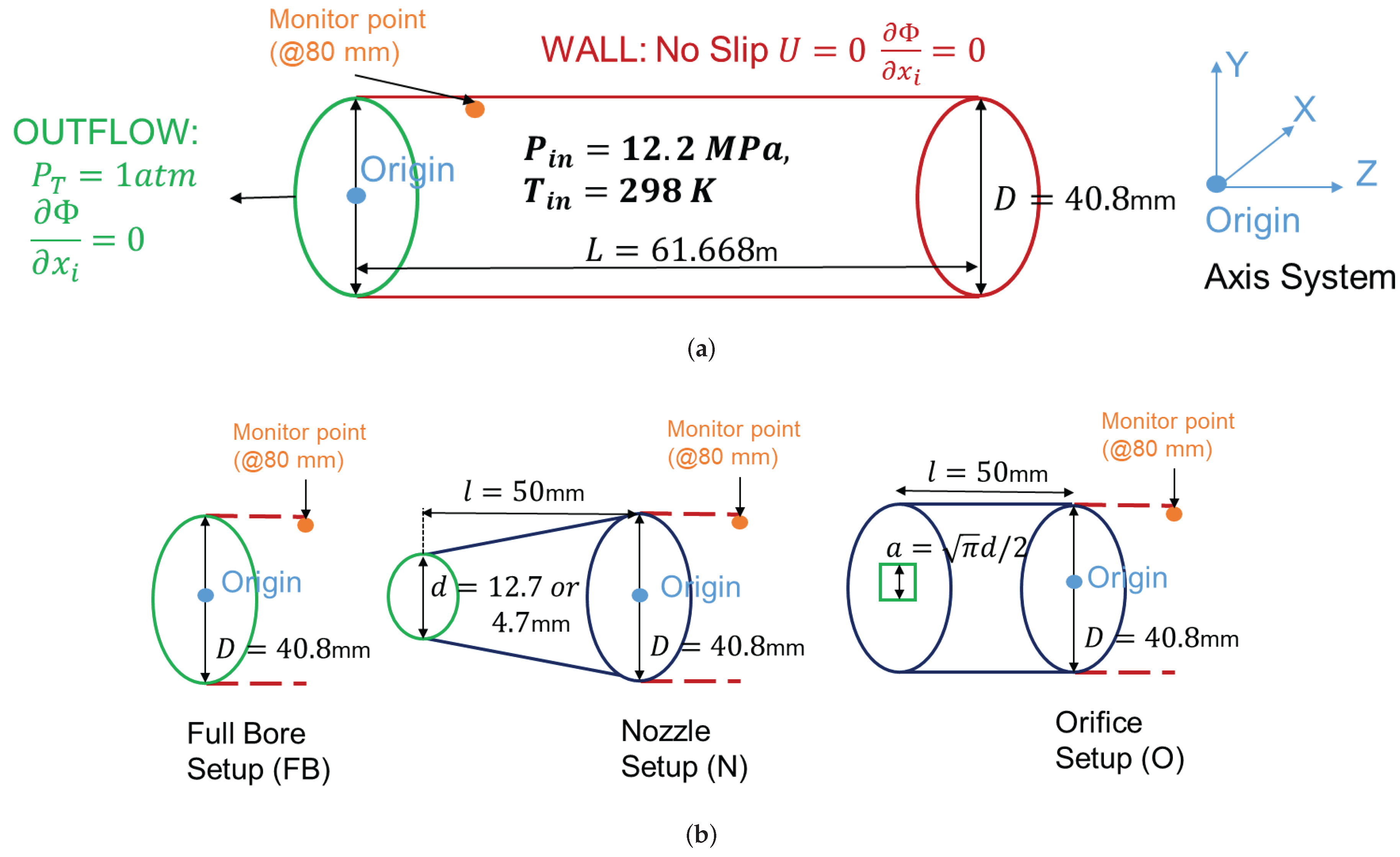
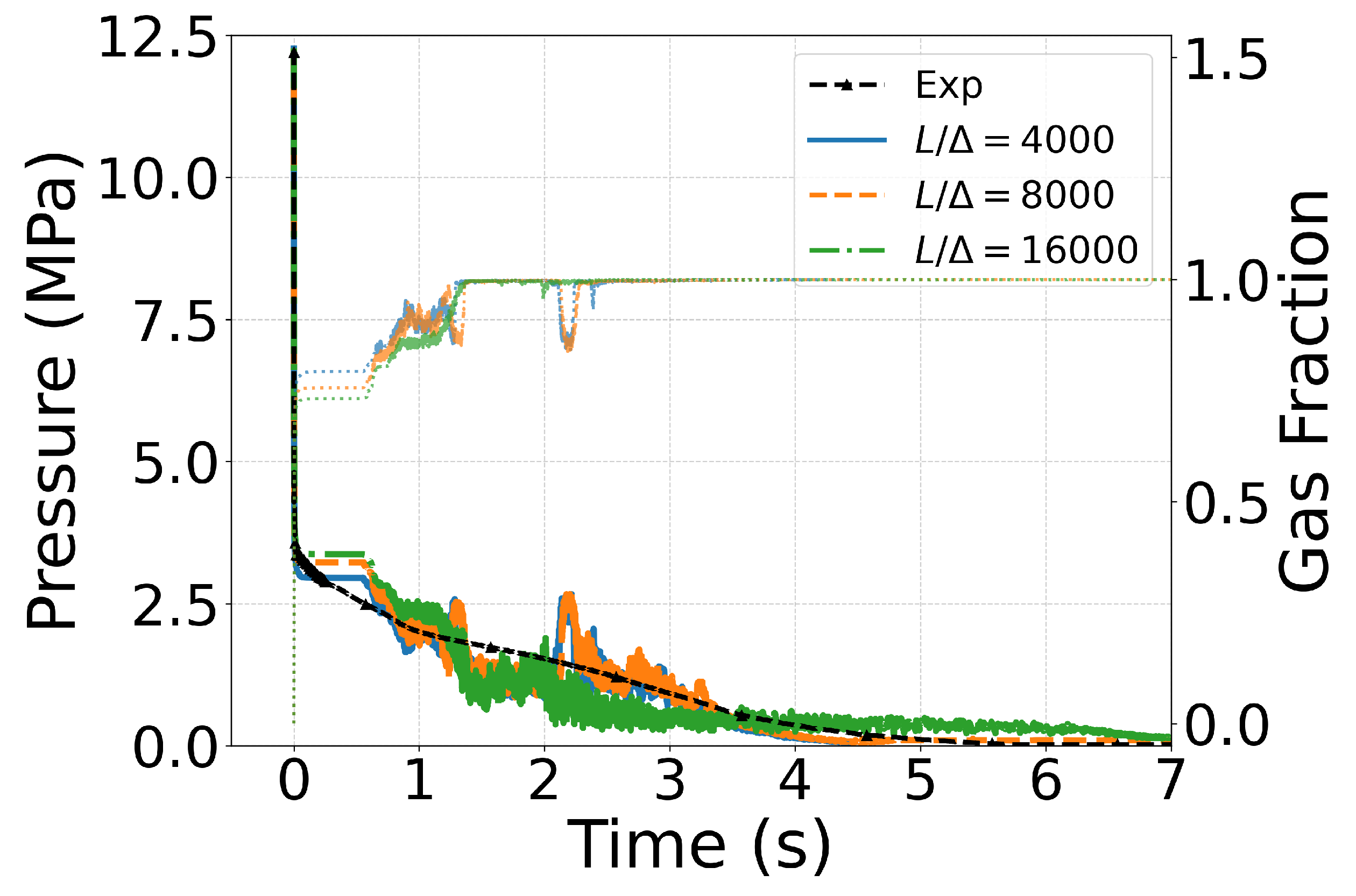
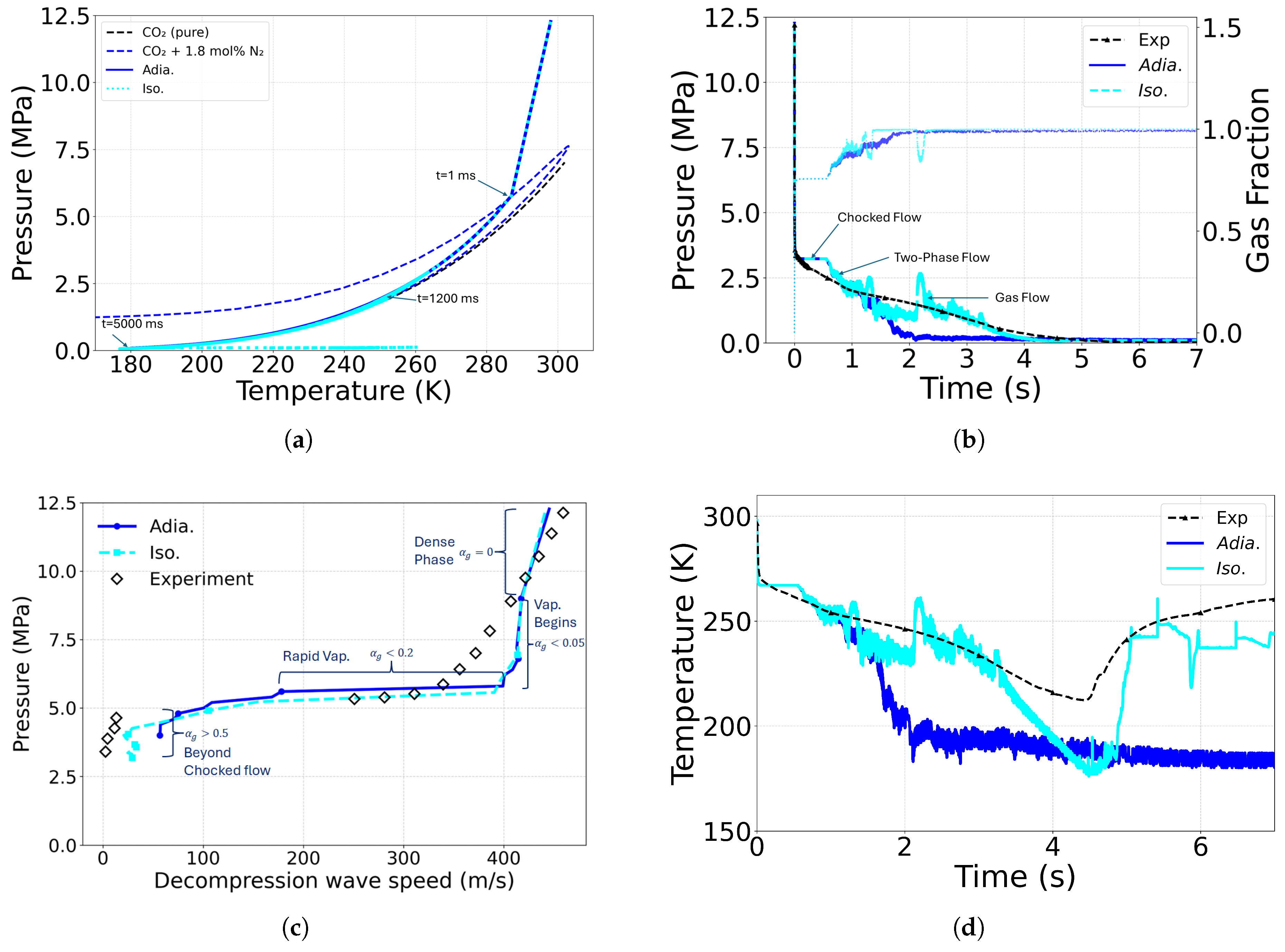
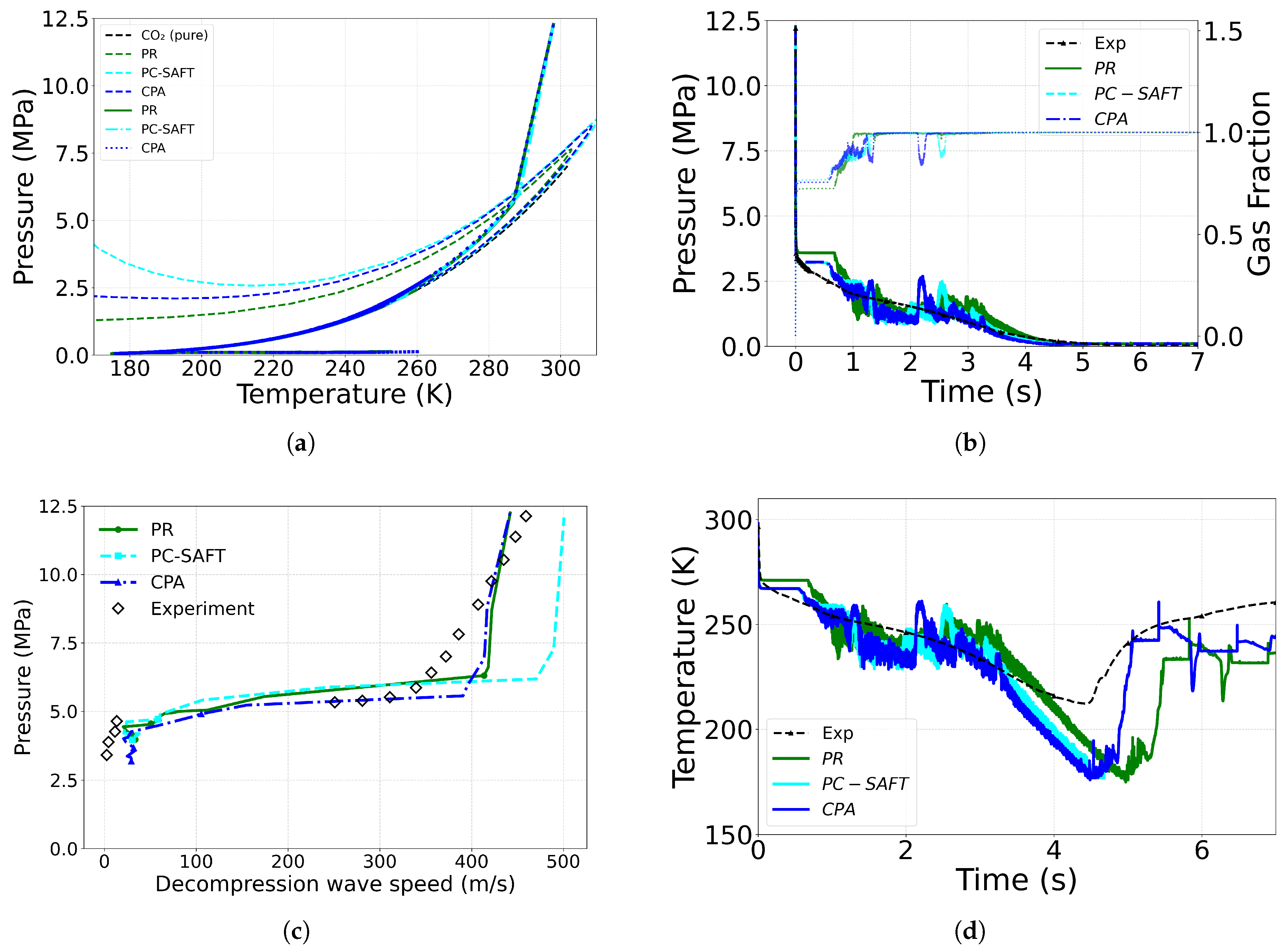
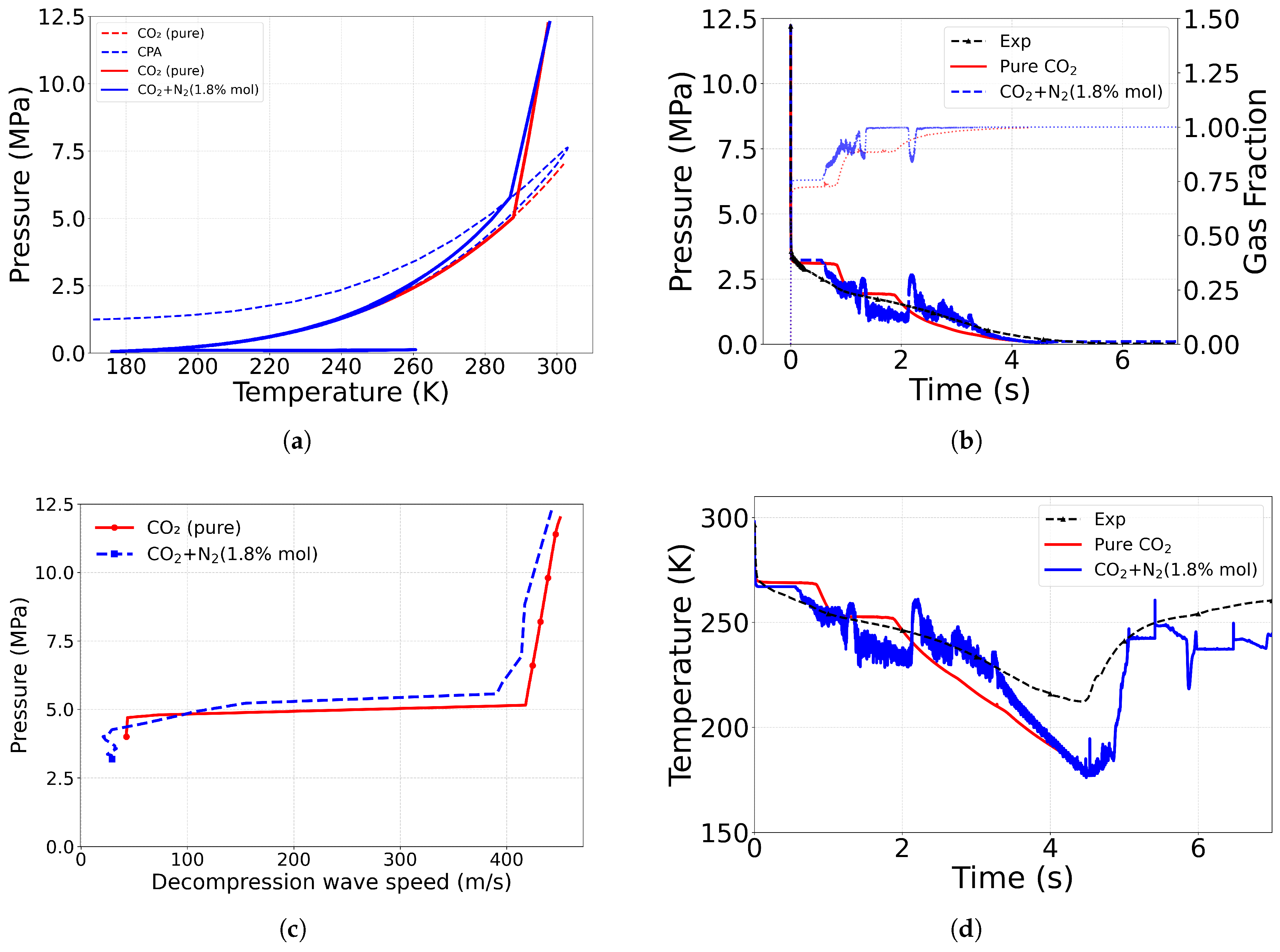
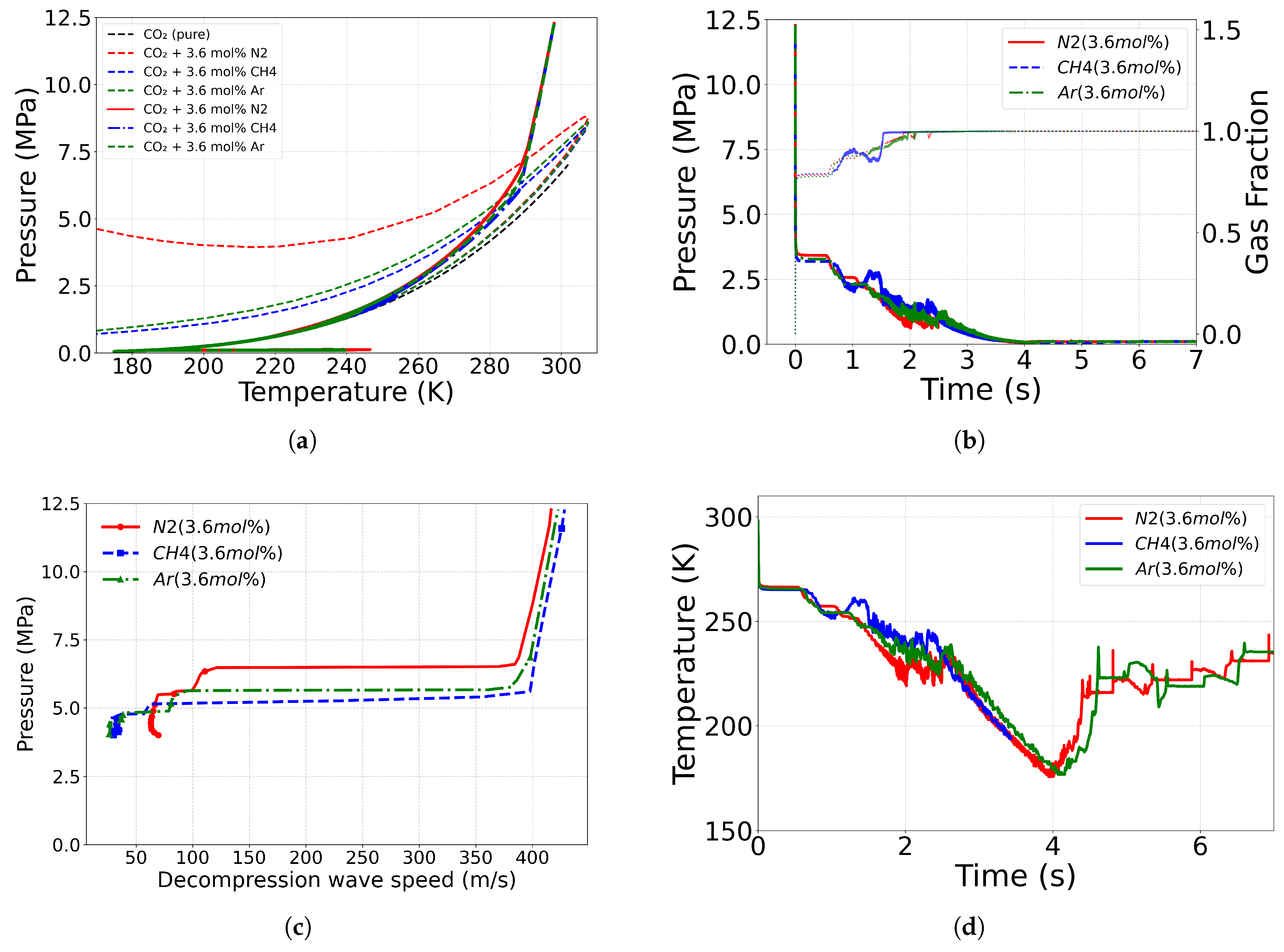
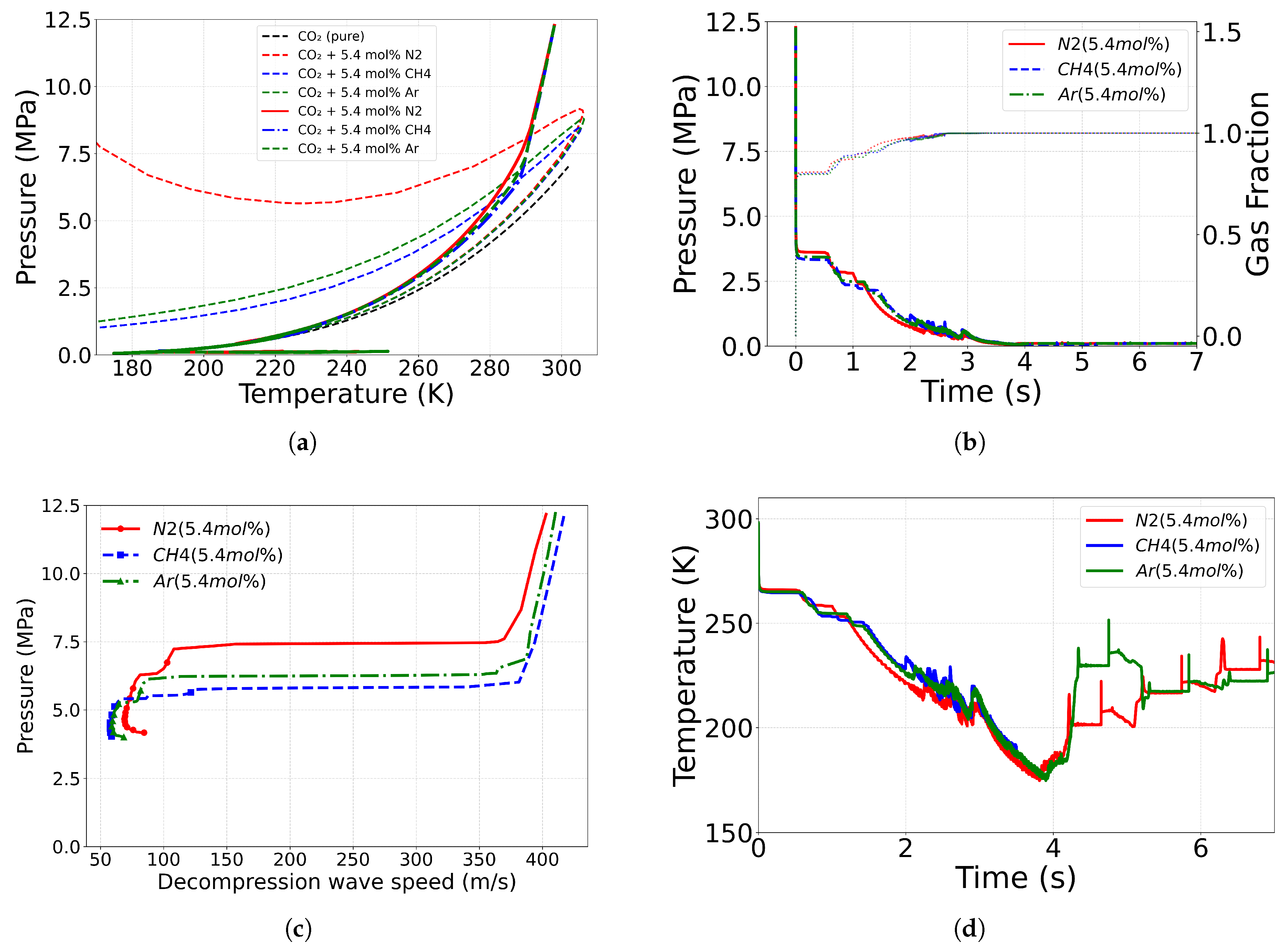
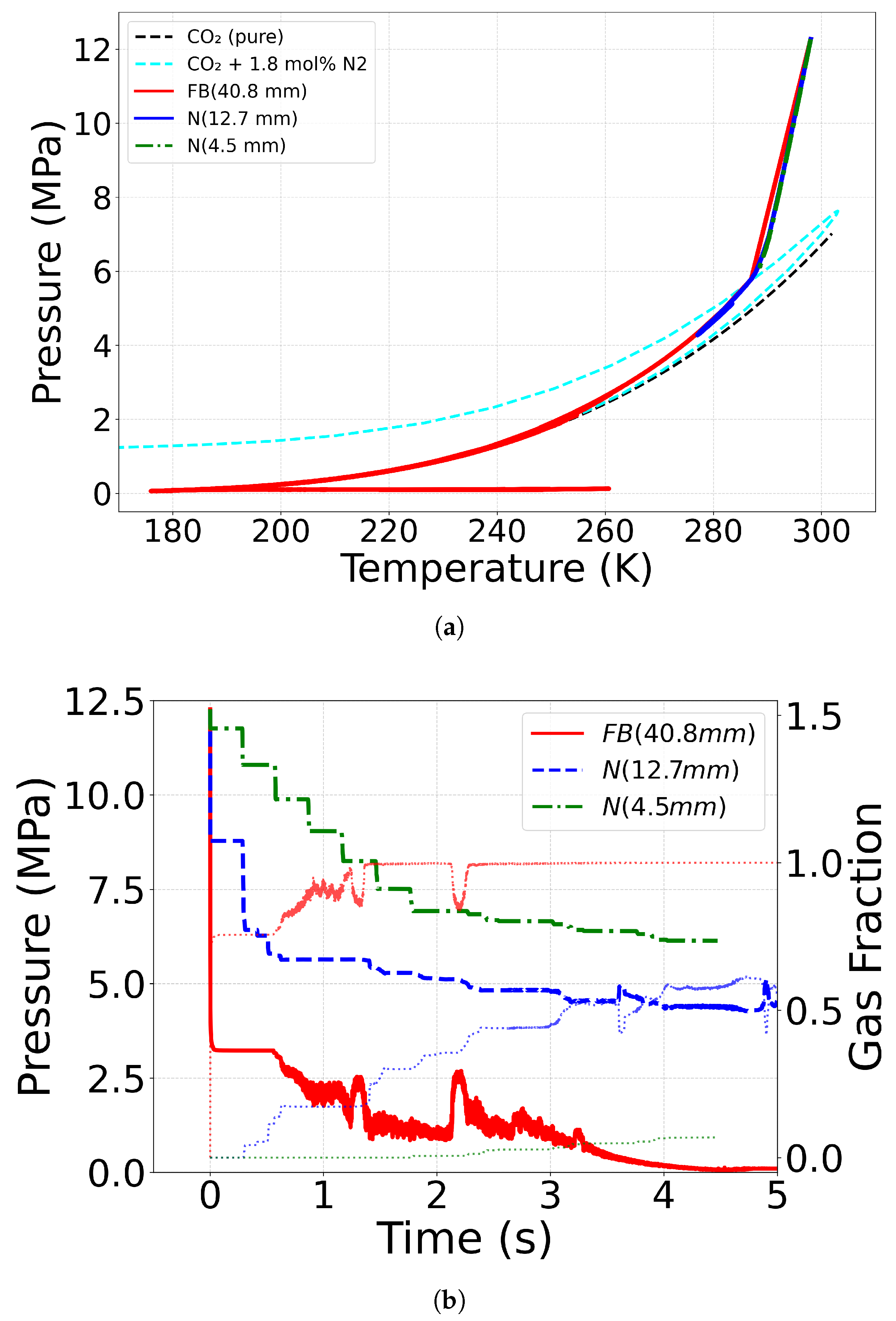
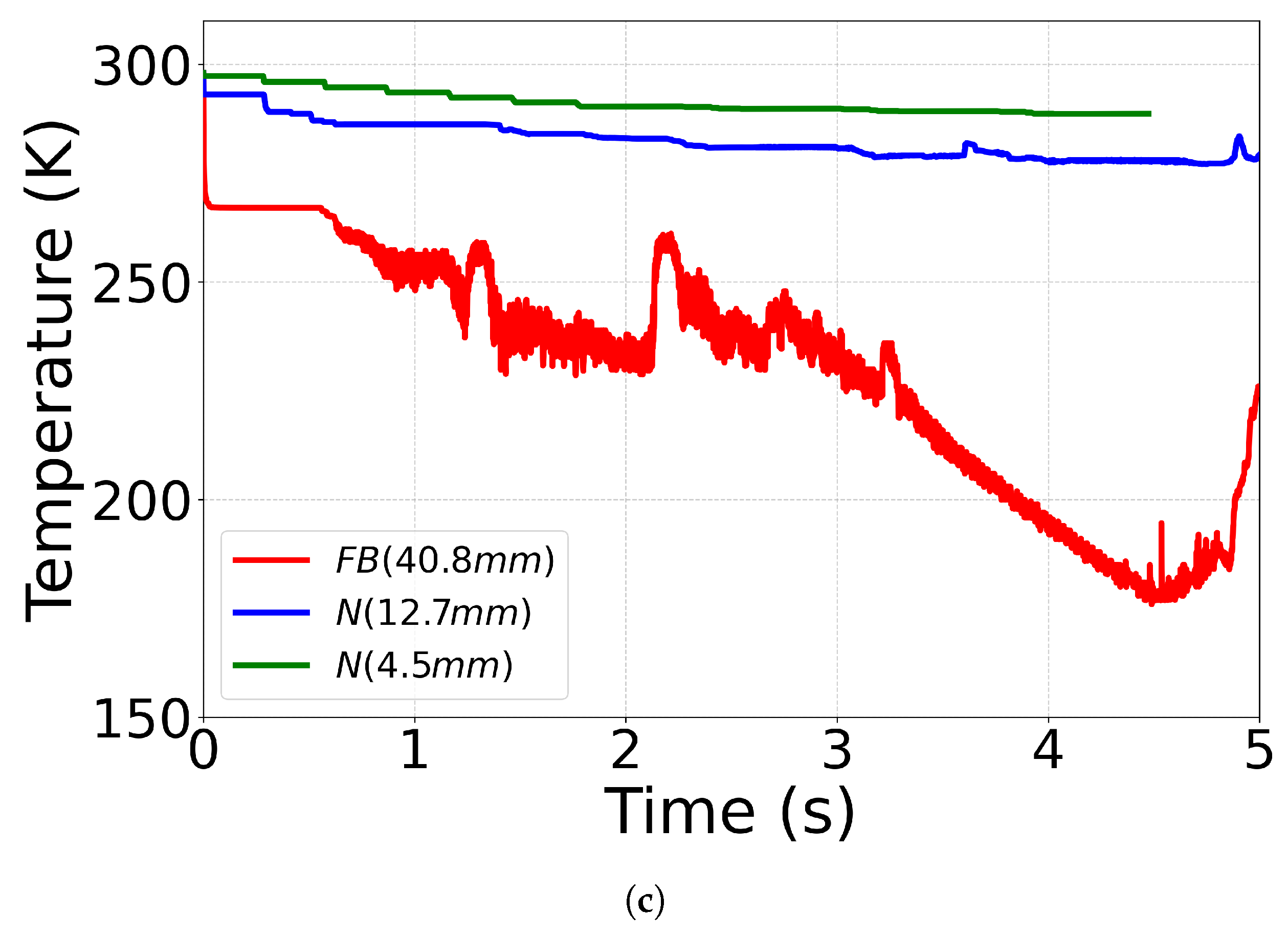
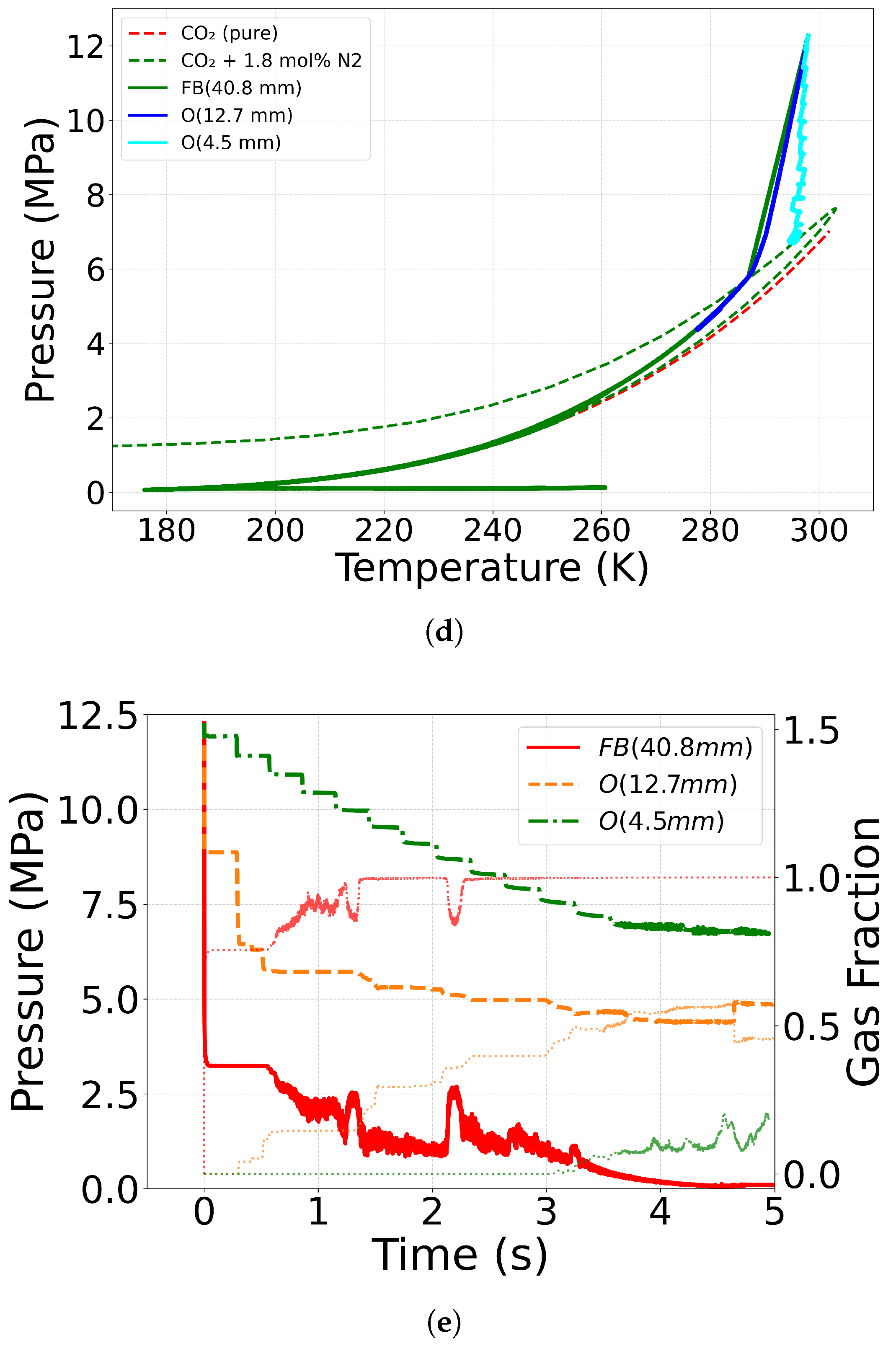
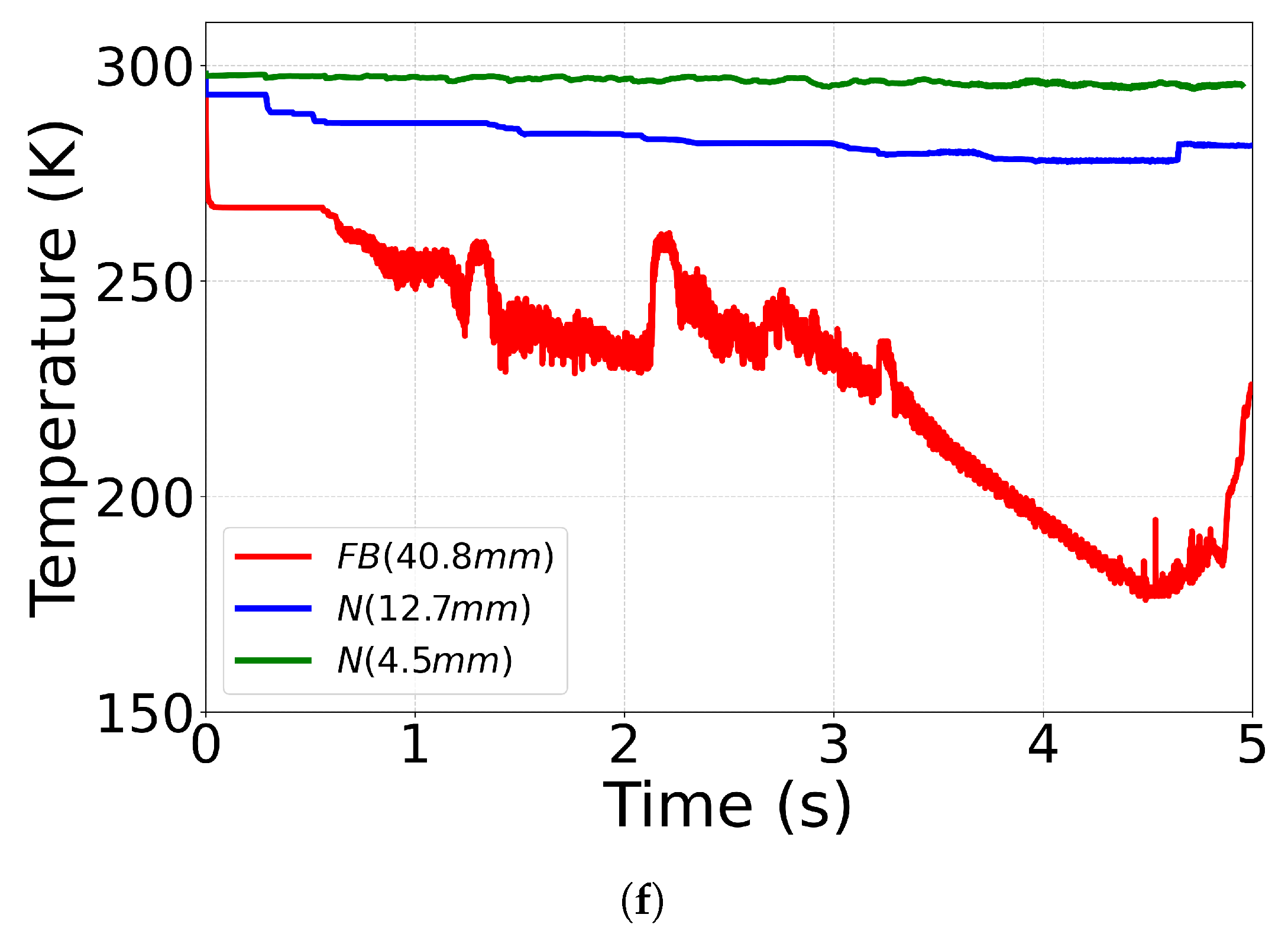
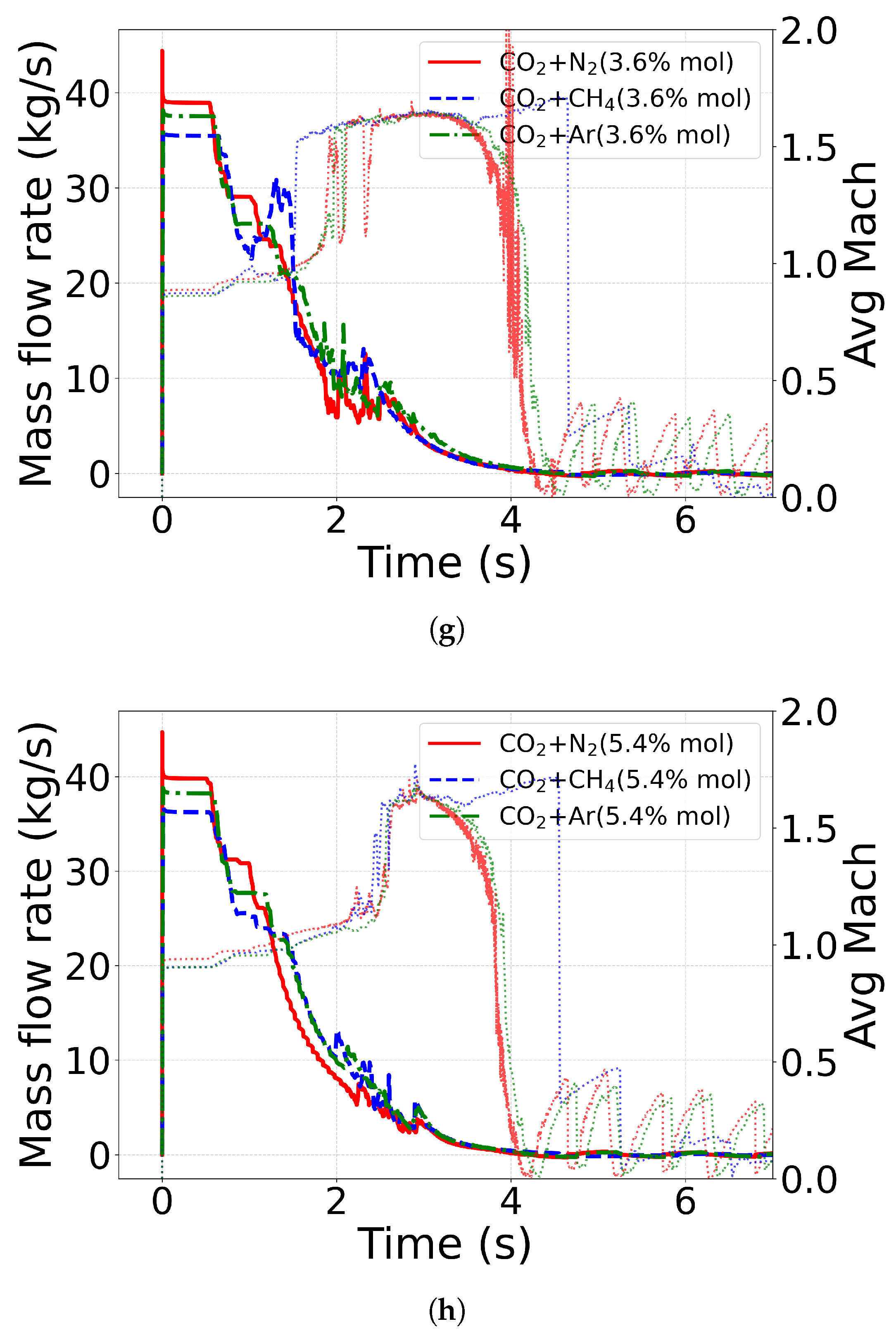
| Case ID | EoS | Outlet Type | Impurity (mol%) | Impurity (mass%) |
|---|---|---|---|---|
| C1(No. 9[31]) | CPA | FB | N2 (1.8) | N2 (1.15) |
| C2(No. 9[31]) | PR | FB | N2 (1.8) | N2 (1.15) |
| C3(No. 9[31]) | SAFT | FB | N2 (1.8) | N2 (1.15) |
| Case ID | EoS | Outlet Type | Impurity (mol%) | Impurity (mass%) |
|---|---|---|---|---|
| C4 | CPA | FB | N2 (3.6) | N2 (2.3) |
| C5 | CPA | FB | N2 (5.4) | N2 (3.5) |
| C6 | CPA | FB | CH4 (3.6) | CH4 (1.3) |
| C7 | CPA | FB | CH4 (5.4) | CH4 (2.0) |
| C8 | CPA | FB | Ar (3.6) | Ar (3.2) |
| C9 | CPA | FB | Ar (5.4) | Ar (4.9) |
| Case ID | EoS | Outlet Type | Impurity (mol%) | Impurity (mass%) |
|---|---|---|---|---|
| C10(No. 13 [71]) | CPA | O (12.7 mm) | N2 (1.8) | N2 (1.15) |
| C11(No. 16 [71]) | CPA | O (4.5 mm) | N2 (1.8) | N2 (1.15) |
| C12(No. 18 [71]) | CPA | N (12.7 mm) | N2 (1.8) | N2 (1.15) |
| C13(No. 17 [71]) | CPA | N (4.5 mm) | N2 (1.8) | N2 (1.15) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





