1. Introduction
Postbiotics, which comprise inanimate microorganisms and/or their cellular components and metabolites, are gaining increased attention for their potential health benefits [
1]. Although these beneficial actions resemble those of probiotics, the stability and safety of postbiotics make them an appealing alternative in particular for vulnerable populations, including newborns, elderly people, and patients with clinical states for whom the use of probiotics may be not recommended [
2,
3,
4]. Emerging findings suggest that the health benefits of selected postbiotics can match, and my even exceed those reported for the probiotic counterparts [
2,
5,
6]. Extensive research has uncovered that selected postbiotic products can exert beneficial modulatory actions on microbiome, epithelial barrier, nervous system, immune and metabolic pathways [
5,
7,
8].
Processes commonly used for producing postbiotics include fermentation and microbial inactivation. The fermentation process could increase the microorganisms’ population and the production of several cellular components and metabolites, all of which can exhibit functional activities [
3]. Microbial inactivation can be achieved through various methods, including heat treatment, sonication, high-pressure, and enzymatic treatment [
9]. The effectiveness of postbiotics can vary significantly comparing different strains and preparation processes, thus testing the efficacy of each single postbiotic product is mandatory to ensure it can deliver the intended health benefits [
10]. The majority of available data derives from studies on single bacterial strain, but it has been suggested that the presence of multiple microorganisms can increase the beneficial health effects compared to single-strain preparations thanks to the synergistic action of different microorganisms [
11,
12]. However, most of the products are obtained by growing the microorganisms separately and then combining them in the formulation of the finished product or by co-fermenting two or more microorganisms simultaneousl [
12,
13]. Co-fermentation is based on the simultaneous processing of different living microorganisms to take advantage of the metabolic cooperation, which can lead to improved fermentation performances or the development of products with increased beneficial potential. The dark side of this process could be the possible competition between microorganisms [
13].
An innovative postbiotic product deriving from the sequential fermentation of two Lacticaseibacillus strains, the L. rhamnosus GG and L. paracasei NPB-01 has of increasingly developed with the aim to increase the beneficial modulation of body functions. Here we comparatively evaluated the beneficial actions exerted by this innovative postbiotic product vs. the two same single heat-inactivated probiotic strains on epithelial barrier and innate immunity in human enterocytes.
2. Materials and Methods
Postbiotic Products
Three different postbiotic products were tested in the study. The first one was obtained from the fermentation of L. rhamnosus GG (ATCC 53103) on MRS broth (24 h, 37 °C). At the end of the fermentation process, a mild heat treatment was conducted (90 °C, 1 min) to inactivate the bacterial biomass, yielding the postbiotic preparation (LGGp). The second one was obtained from the fermentation of L. paracasei NPB-01 (DSM 34367) on MRS broth (24 h, 37 °C). At the end of the fermentation process, a mild heat treatment (90 °C, 1 min) was applied to inactivate the bacterial biomass, obtaining the postbiotic preparation (Lpp). The third one was obtained from the sequential fermentation process using L. rhamnosus GG and L. paracasei NPB-01 (iPB). Briefly, after the first fermentation of LGG, a mild heat treatment (90 °C, 1 min) was applied for inactivating the microbial population. Then, a second fermentation was started by inoculating L. paracasei NPB-01. Following the second fermentation, a mild heat treatment (90 °C, 1 min) was performed. Both fermentations were carried out on MRS broth for 24h at 37 °C. All postbiotics preparation (LGGp, Lpp and iPB) used in this study were dried. All postbiotic products utilized in the study were provided as food grade powder by Science Power SrL (Milan, Italy).
Human Enterocytes Cell Line
For all experiments we used Caco-2 cell line (American Type Culture Collection, Middlesex, UK; accession number: HTB-37). Cells were grown in Dulbecco’s modified Eagle’s medium with a high glucose concentration (4.5 g/L) (Gibco, Berlin, Germany). The medium was supplemented with 10% fetal bovine serum, 1% l-glutamine, 1% non-essential amino acids, and 1% penicillin/ streptomycin (Thermo Fisher Scientifics). Cells were incubated at 37 °C and 5% CO2. The medium was changed every 2 days.
Postbiotic Stimulation Protocol
Caco-2 cells were seeded (0.1 × 106 cells/well) in 12-well plates (Corning). After 15 days post-confluence cells were stimulated with the postbiotic products. Caco-2 cells stimulated with medium alone were used as controls. Experiments were carried out in triplicate.
Modulation of Epithelial Gut Barrier
The potential modulatory action on epithelial barrier elicited by the study products was investigated using different tools, as previously adopted [
14,
15]. Enterocytes cell growth and differentiation were assessed using proliferation assay and brush border enzyme lactase gene expression, respectively. The modulatory action exerted on mucous thickness was explored evaluating mucin 2 (Muc2) gene expression. The effect on tight junctions’ network was explored by analyzing the gene expression of two major proteins, occludin and zonula occludens 1 (ZO-1). Lastly, the potential modulatory action on innate immunity was explored analyzing the production of the major innate immunity peptide, Human Beta Defensin (HBD-2), by enterocytes.
Cell Proliferation Assay
Human enterocyte proliferation assays were performed using MTT (the bromide salt of 3-(4,5-dimethylthiazol-2-yl)- 2,5-diphenyl tetrazolium) (Sigma-Aldrich, Milan, Italy). Cells (104 cells/well) were seeded in 24-well plates (Corning, Inc., New York, NY, USA) with or without the postbiotic products at 37 °C in a humidified atmosphere containing 5% CO2. The cell viability was monitored by adding 5 mg/mL of MTT solution followed by 1 h incubation. The medium was then removed, and the converted dye was solubilized with acidic isopropanol (0.04-0.1 N HCl in absolute isopropanol). Absorbance was read at 570 nm using an Epoch Microplate Spectrophotometer (Bioteck, Winooski, VT, USA).
Real Time PCR
Total RNA from Caco-2 cells was extracted using TRIzol reagent (Gibco BRL, Paisley, UK). The reverse transcription to cDNA was performed with the High-Capacity RNA-to-cDNATM Kit (Life Technologies, Waltham, MA, USA) following the manufacturer’s instructions. The cDNAs were stored at −80 °C until analysis. Quantitative real-time PCR (qRT-PCR) analysis, carried out with the TaqMan Gene Expression Master Mix (Applied Biosystems, Vilnius, Lithuania), was used to evaluate the gene expression occludin (Hs05465837_g1) and ZO-1 (Hs01551871_m1). Gene expression of the mucin 2 (Muc2), and lactase enzyme, were evaluated using a SYBR green Master Mix (Applied Biosystems, Grand Island, NY, USA). The primers used for Muc2 were forward (5′-CTCCGCATGAGTGTGAGT-3′) and reverse (5′-TAGCAGCCACACTTGTCTG-3′). The primers used for lactase were forward (5′-ACACGGTCGATTTCCTCTCT-3′) and reverse (5′-TGGGTTCTTCATGGTGGAGG-3′). The amplification protocol was 40 cycles of 15 s of denaturation at 95 °C, 60 s of annealing at 60 °C, and 60 s of elongation at 60 °C in a Light Cycler 7900HT (Applied Biosystems, Grand Island, NY, USA). Data were analyzed using the comparative threshold cycle method. We used the glucuronidase beta (GUS-B) gene to normalize the level of mRNA expression (TaqMan probes: Hs00939627_m1; SYBR green Forward primer: 5′-GAAAATATGTGGTTGGAGAGCTCATT-3′; SYBR green Reverse primer: 5′-CCGAGTGAAGATCCCCTTTTTA-3′).
Modulation of Innate Immunity
After stimulation with the postbiotics, cell supernatants were collected and stored at −80 °C until analysis. Production of HBD-2 was determined by ELISA test, using commercial enzymatic kits (Human DEFβ2/DEFB2, Elabscience Biotechnology Inc. Wuhan, Hubei. Detection limit of 62.5 pg/mL).
Statistical Analysis
The Kolmogorov-Smirnov test was used to determine whether variables were normally distributed. Data were analysed using unpaired t-test. The level of significance for all statistical tests was two-sided, p < 0.05. All data were collected in a dedicated database and analysed by a statistician using GraphPad Prism 9.3.0 (La Jolla, CA USA).
3. Results
3.1. Modulation of Human Enterocytes Cell Growth and Differentiation
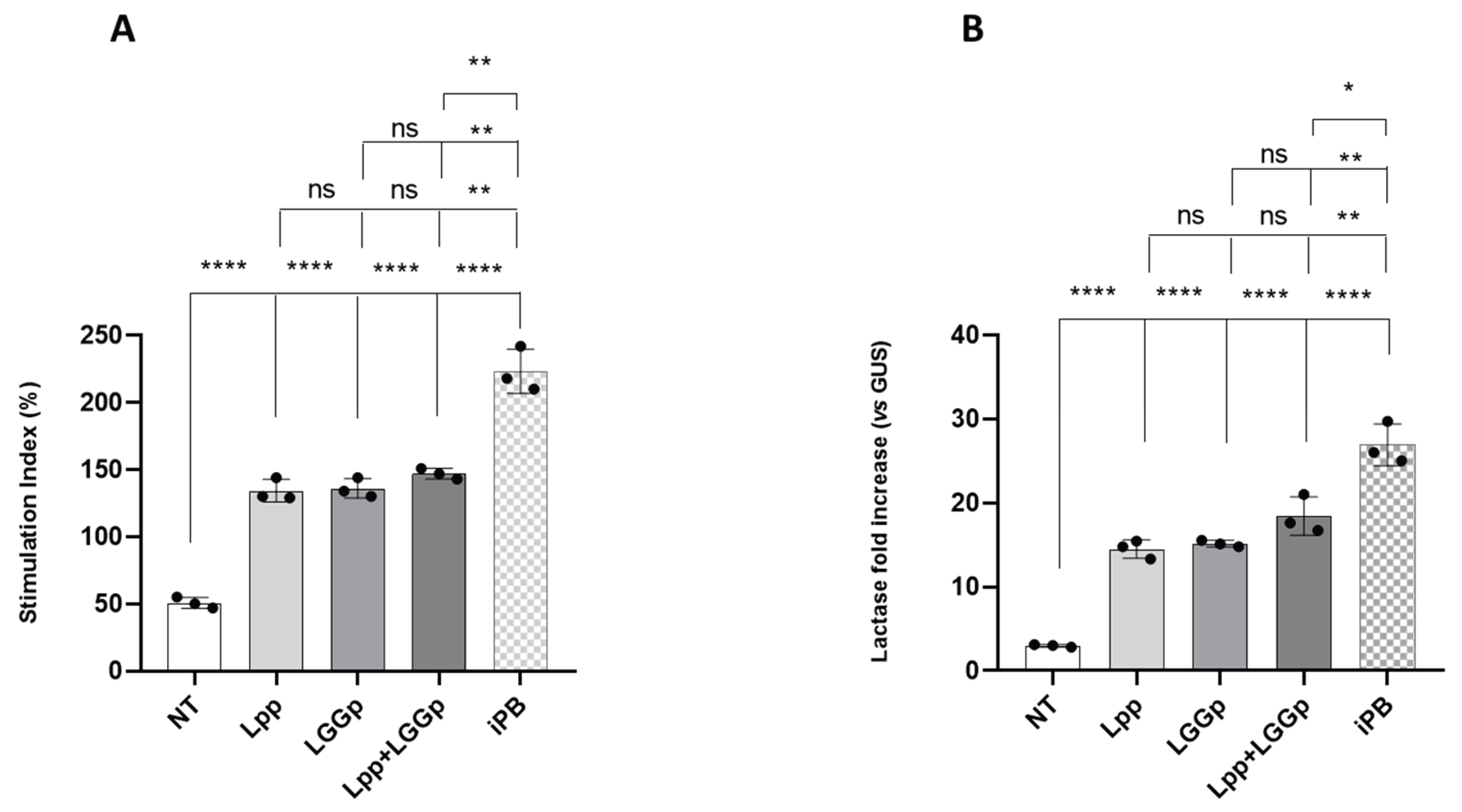
In preliminary dose-response and time course experiments we defined the best experimental conditions for each postbiotic product. As shown in
Figure 1 (panels A and B), both products obtained through the single strain fermentation process (Lpp and LGGp) promoted the growth and the differentiation of human enterocytes. No further increase in cell growth and differentiation was observed adding Lpp and LGGp simultaneously at the maximum dose. On the contrary, the modulatory actions elicited by the postbiotic product deriving from sequential fermentation (iPB) were significantly higher for both parameters, if compared with those obtained by the two single strain postbiotic products alone or in combination.
3.2. Modulation of Tight Junction Proteins and Mucous Production in Human Enterocytes
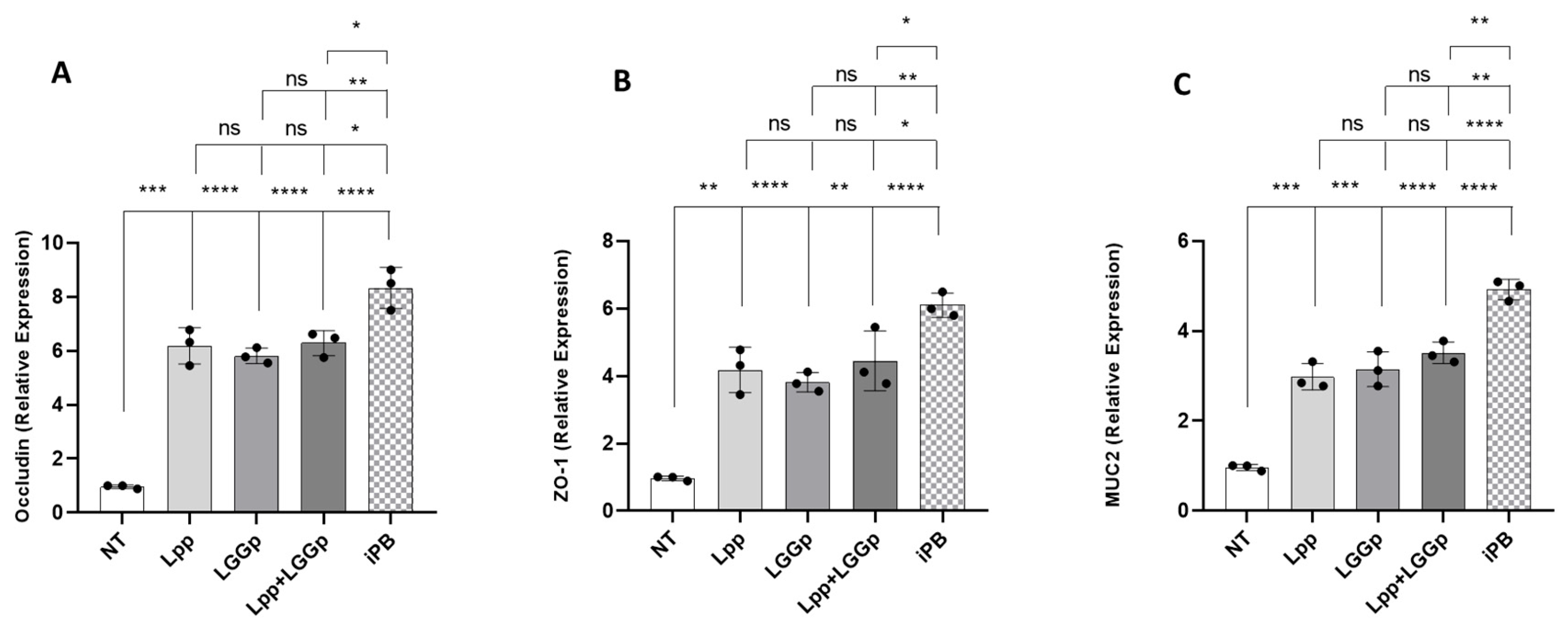
As shown in
Figure 2, the incubation with all the three postbiotic products led to the increase in the expression of the two major tight junction proteins, occludin and ZO-1 (panels A and B). Also, the expression of the epithelial mucus layer protein MUC2, resulted modulated by these two postbiotic products (panel C). No further stimulatory action for all three parameters was observed adding Lpp and LGGp simultaneously at the maximum respective dose. On the contrary, the iPB consistently induced a significantly greater stimulatory effect on all biomarkers compared to the other postbiotics alone or in combination.
3.3. Modulation of Innate Immunity
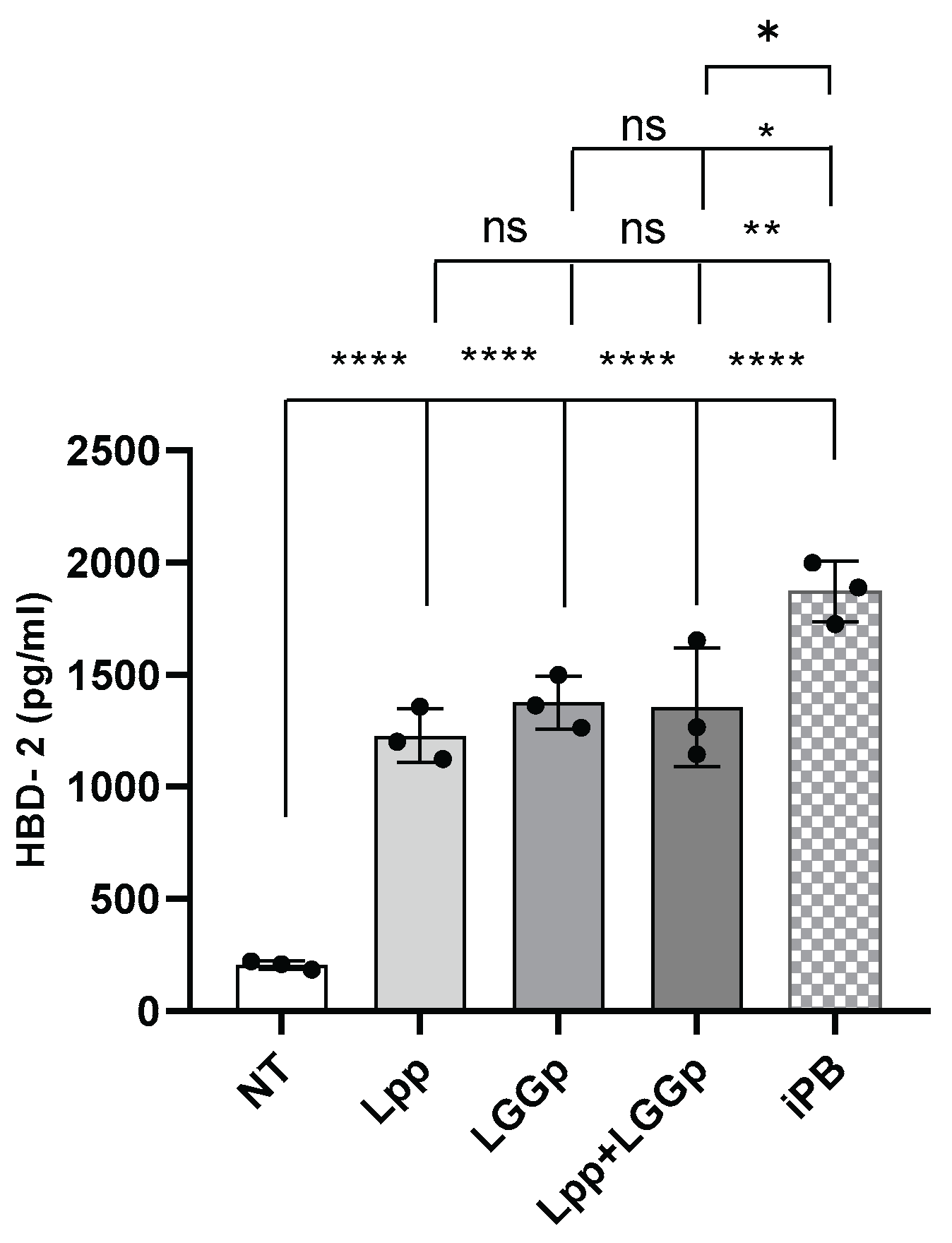
To investigate the potential modulatory action elicited by the postbiotic products evaluated in this study, we measured the production of the major innate immunity peptide HBD-2 by human enterocytes stimulated with the study products for 48h. As shown in
Figure 3, the incubation with all the three postbiotic products led to the increase in the production of HBD-2 by human enterocytes. No further stimulatory action in HBD-2 production was observed stimulating the cells simultaneously with Lpp and LGGp at the maximum respective dose. On the contrary, exposing human enterocytes to iPB resulted in a significantly higher stimulatory effect on HBD-2 production if compared with those observed stimulating the cells with Lpp or with LGGp alone or in combination.
4. Discussion
Fermentation is a primary and essential process for producing postbiotics [
16], it could significantly affect composition and function of these products [
17,
18,
19,
20]. Several studies have highlighted the beneficial effects of different postbiotic products derived from single or multiple bacterial strains, either co-fermented or mixed post-fermentation [
21,
22,
23]. In this study, we provided the first evidence on the beneficial actions on human enterocytes elicited by an innovative postbiotic (iPB) produced through sequential fermentation of two probiotic strains (i.e., L. rhamnosus GG and L. paracasei NPB-01). Our findings demonstrated that iPB exerts a more potent regulatory action on human enterocytes proliferation and differentiation, as demonstrated by MTT and brush border enzyme lactase increases, respectively. A parallel more potent effect was also observed on epithelial gut barrier architecture, as suggested by increased expression of the two major tight-junctions’ proteins, occludin and ZO-1. We also found that the iPB exposure resulted in a higher increase in the expression of Muc2 and the innate immunity peptide HBD-2 by human enterocytes. Muc2 is a key mucin protein forming the protective mucus layer in the intestine essential for epithelial gut barrier function [
24]. Muc2 mucus layer is essential in protecting the host not only against pathogens but also against the occurrence of allergies limiting the exposure to environmental allergens [
25]. In addition, stimulating the production of key regulatory factors such as IL-10, Transforming Growth Factor-β (TGF-β), Retinoic Acid (RA), and thymic stromal lymphopoietin (TSLP) by epithelial and dendritic cells (DCs), Muc2 promotes the expansion of Treg cells and reduces inflammation, thereby restoring immune tolerance and homeostasis [
25].
Beyond their structural and metabolic roles, intestinal epithelial cells actively participate in mucosal immunity by producing antimicrobial peptides like HBD-2. This small, cationic, antimicrobial peptide “farms” the microbiome, influencing its composition and preventing dysbiosis which can lead to inflammatory conditions. The HBD-2 possesses a broad-spectrum activity against bacteria, viruses, fungi and some parasites. However, in addition HBD-2 activates a variety of immune cells and regulates cytokine/chemokine production, cell migration, proliferation, differentiation, angiogenesis, the wound healing process and maintenance of the epithelial barrier function. Alterations in the level of HBD-2 have been associated with the initiation and development of various inflammatory and allergic diseases [
26,
27]. Thus, this molecule could be considered as cornerstone of mucosal innate immune system, providing a first line of defence against infections, dysbiosis, inflammation and allergic sensitization [
14,
28].
For all variables investigated into the study, we found a higher modulatory action elicited by iPB if compared with those obtained by single postbiotics alone or in combination, suggesting that the sequential fermentation process likely induces a unique metabolic signature. We hypothesize that the first fermentation step with LGG modifies the substrate and releases primary metabolites that, following heat inactivation, serve as a specialized matrix for the subsequent fermentation by L. paracasei NBP01. This ‘metabolic priming’ may trigger the synthesis of a more complex array of bioactive compounds, such as specific peptides or metabolites. This study introduces the concept of sequential postbiotic design as a strategy to maximize the health-promoting potential of lactic acid bacteria.
While these results are promising, some limitations should be acknowledged. First, this study focused on two specific probiotic strains, both individually and in a sequential combination; therefore, the reproducibility and generalizability of these effects to other bacterial taxa remain to be established. Additionally, the sequential fermentation was performed exclusively by inoculating LGG followed by L.paracasei NBP01. Future research exploring the reversed inoculation order or different timing intervals could provide deeper insights into how microbial succession shapes the final postbiotic activity.
Furthermore, although the biological impact of iPB is highlighted by the results of this study, the specific effector molecules—whether they be secreted metabolites, cell wall components (such as the capsule), or genomic DNA fragments— as well the respective mechanisms of action, have yet to be fully characterized. It has been suggested that many postbiotics’ effects involve the activation of toll like receptors and related intracellular pathways, such as NF-κB signalling [
28]. Thus, the results of this study suggest that the sequential fermentation optimizes the concentration of ‘Microbe-Associated Molecular Patterns’ (MAMPs) or secreted factors that are recognized by TLRs on human cells [
29]. This enhanced modulatory action on epithelial gut barrier and innate immunity suggest that iPB could provide a more efficient protective effect against conditions characterized by alteration of intestinal permeability, dysbiosis, immune tolerance and inflammation. Nevertheless, our findings highlight the potential of sequential fermentation as a novel and superior biotechnological platform for postbiotic production. This approach could pave the way for a new generation of functional ingredients with enhanced efficacy in modulating human health. Further investigations are warranted to test different strain combinations and to broaden the evaluation of intestinal barrier integrity, systemic immunity, and cellular differentiation pathways.
5. Conclusions
In conclusion, our data support the use of sequential fermentation to produce a high-performance postbiotic. iPB stands out as a promising candidate for functional food applications and clinical interventions aimed at strengthening the intestinal barrier and immunity. Future studies involving ‘omics’ technologies (metabolomics and proteomics) will be essential to pinpoint the exact molecular effectors responsible for these observed synergistic benefits.
Author Contributions
Conceptualization, F.O. and L.C.; methodology, F.O., A.C., M.D.A. and A.L.; validation, F.O., S.C. and L.C.; formal analysis, F.O. and A.C.; investigation, F.O.; resources, M.M., C.L. and A.A.; data curation, F.O. and A.C.; writing—original draft preparation, F.O.; writing—review and editing, L.C.; visualization, F.O.; supervision, L.C. All authors have read and agreed to the published version of the manuscript.
Funding
The research was funded under the National Recovery and Resilience Plan (NRRP), Mission 4 Component 2 Investment 1.3—Call for tender No. 341 of 15 March 2022 of Italian Ministry of University and Research funded by the European Union—NextGenerationEU. Project code PE00000003, Concession Decree No. 1550 of 11 October 2022, adopted by the Italian Ministry of University and Research, CUP E63C22002030007, Project title “ON Foods—Research and innovation network on food and nutrition Sustainability, Safety and Security—Working ON Foods.”.
Acknowledgments
All postbiotic products utilized in the study were a kind gift from Science Power SrL, Via Senato, 8, 20121 Milan (Italy). Science Power had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Conflicts of Interest
None reported.
Abbreviations
The following abbreviations are used in this manuscript:
| iPB |
Innovative postbiotic |
| ZO-1 |
Zonula occludens-1 |
| MUC-2 |
Mucin-2 |
| HBD-2 |
Beta-Defensin 2 |
| LGG |
Lactocticaseibacillus GG |
| LGGp |
Lactocticaseibacillus GG postbiotic |
| Lpp |
Lactocticaseibacillus paracasei NPB-01 postbiotic |
| qRT-PCR |
Quantitative real-time PCR |
| GUS-B |
Glucuronidase beta |
| TGF-β |
Transforming Growth Factor-β |
| RA |
Retinoic Acid |
| TSLP |
thymic stromal lymphopoietin |
| DCs |
dendritic cells |
References
- Aguilar-Toalá, J.E.; Arioli, S.; Behare, P.; Belzer, C.; Berni Canani, R.; Chatel, J.M.; et al. Postbiotics—When simplification fails to clarify. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 825–826. [Google Scholar] [CrossRef]
- Calvanese, C.M.; Villani, F.; Ercolini, D.; De Filippis, F. Postbiotics versus probiotics: Possible new allies for human health. Food Res. Int. 2025, 217, 116869. [Google Scholar] [CrossRef]
- Prajapati, N.; Patel, J.; Singh, S.; Yadav, V.K.; Joshi, C.; Patani, A.; et al. Postbiotic production: Harnessing the power of microbial metabolites for health applications. Front. Microbiol. 2023, 14, 1306192. [Google Scholar] [CrossRef]
- Yeşilyurt, N.; Yılmaz, B.; Ağagündüz, D.; Capasso, R. Involvement of Probiotics and Postbiotics in the Immune System Modulation. Biologics 2021, 1, 89–110. [Google Scholar] [CrossRef]
- Roggero, P.; Liotto, N.; Pozzi, C.; Braga, D.; Troisi, J.; Menis, C.; et al. Analysis of immune, microbiota and metabolome maturation in infants in a clinical trial of Lactobacillus paracasei CBA L74-fermented formula. Nat. Commun. 2020, 11, 2703. [Google Scholar] [CrossRef] [PubMed]
- Salminen, S.; Collado, M.C.; Endo, A.; Hill, C.; Lebeer, S.; Quigley, E.M.M.; et al. The International Scientific Association of Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of postbiotics. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 649–667. [Google Scholar] [CrossRef] [PubMed]
- Scott, E.; De Paepe, K.; Van de Wiele, T. Postbiotics and Their Health Modulatory Biomolecules. Biomolecules 2022, 12, 1640. [Google Scholar] [CrossRef] [PubMed]
- Yolmeh, M.; Xavier-Santos, D.; Sant’Ana, A.S. Modulating gut microbiota by paraprobiotics: Mechanisms, advantages, and challenges. Food Biosci. 2024, 60, 104305. [Google Scholar] [CrossRef]
- Thorakkattu, P.; Khanashyam, A.C.; Shah, K.; Babu, K.S.; Mundanat, A.S.; Deliephan, A.; et al. Postbiotics: Current Trends in Food and Pharmaceutical Industry. Foods 2022, 11, 3094. [Google Scholar] [CrossRef]
- Balakrishna, K.; Naveena, G.; Kingston, J.J. Postbiotics at the interface of microbial biotechnology and therapeutics: Industrial production, functional mechanisms, and clinical potentials. Arch. Microbiol. 2026, 208, 123. [Google Scholar] [CrossRef]
- Maniya, H.; Modasiya, I.; Chauhan, M.; Mori, P.; Kumar, V. Developing Robust Probiotic Consortia: A Methodological Optimization Approach. Curr. Microbiol. 2024, 81, 407. [Google Scholar] [CrossRef] [PubMed]
- Roberts, K.D.; Ahmed, S.; San Valentin, E.; Di Martino, L.; McCormick, T.S.; Ghannoum, M.A. Immunomodulatory Properties of Multi-Strain Postbiotics on Human CD14+ Monocytes. Life 2024, 14, 1673. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Luo, X.; Huang, Y.; Zhao, M.; Liu, T.; Wang, J.; et al. Influence of cooking techniques on food quality, digestibility, and health risks regarding lipid oxidation. Food Res. Int. 2023, 167, 112685. [Google Scholar] [CrossRef]
- Paparo, L.; Aitoro, R.; Nocerino, R.; Fierro, C.; Bruno, C.; Canani, B.B. Direct effects of fermented cow’s milk product with Lactobacillus paracasei CBA L74 on human enterocytes. Benef. Microbes 2018, 9, 165–172. [Google Scholar] [CrossRef]
- Nocerino, R.; Bedogni, G.; Carucci, L.; Cosenza, L.; Cozzolino, T.; Paparo, L.; et al. The Impact of Formula Choice for the Management of Pediatric Cow’s Milk Allergy on the Occurrence of Other Allergic Manifestations: The Atopic March Cohort Study. J. Pediatr. 2021, 232, 183–191.e3. [Google Scholar] [CrossRef] [PubMed]
- Bueno, E.B.T.; Silva Kde, O.; Mendes, M.E.F.; de Oliveira, L.B.; Menezes FPde Imperador, A.C.; et al. Postbiotics Derived from Lactic Acid Bacteria Fermentation: Therapeutic Potential in the Treatment of Muscular Complications in Inflammatory Bowel Disease. Fermentation 2025, 11, 362. [Google Scholar] [CrossRef]
- Alrosan, M.; Al-Massad, M.; Obeidat, H.J.; Maghaydah, S.; Alu’datt, M.H.; Tan, T.C.; et al. Fermentation-induced modifications to the structural, surface, and functional properties of quinoa proteins. Food Sci. Biotechnol. 2025, 34, 3317–3329. [Google Scholar] [CrossRef]
- Oglio, F.; Paparo, L.; Carucci, L.; Gaeta, A.; Armiento, S.; Coppola, S.; et al. Postbiotic effects elicited by heat-inactivated Lacticaseibacillus rhamnosus GG against cow’s milk allergy in human cells. Front. Immunol. 2026, 16, 1671729. [Google Scholar] [CrossRef]
- Pimentel, T.C.; Cruz, A.G.; Pereira, E.; Almeida da Costa, W.K.; da Silva Rocha, R.; Targino de Souza Pedrosa, G.; et al. Postbiotics: An overview of concepts, inactivation technologies, health effects, and driver trends. Trends Food Sci. Technol. 2023, 138, 199–214. [Google Scholar] [CrossRef]
- Sawant, S.S.; Park, H.Y.; Sim, E.Y.; Kim, H.S.; Choi, H.S. Microbial Fermentation in Food: Impact on Functional Properties and Nutritional Enhancement—A Review of Recent Developments. Fermentation 2025, 11, 15. [Google Scholar] [CrossRef]
- Kerksick, C.M. Acute Alpha-Glycerylphosphorylcholine Supplementation Enhances Cognitive Performance in Healthy Men. Nutrients 2024, 16, 4240. [Google Scholar] [CrossRef] [PubMed]
- Kienesberger, S.; Cosic, A.; Kitsera, M.; Raffl, S.; Hiesinger, M.; Leitner, E.; et al. Enterotoxin tilimycin from gut-resident Klebsiella promotes mutational evolution and antibiotic resistance in mice. Nat. Microbiol. 2022, 7, 1834–1848. [Google Scholar] [CrossRef] [PubMed]
- Rezaie, A.; Chang, B.W.; de Freitas Germano, J.; Leite, G.; Mathur, R.; Houser, K.; et al. Effect, Tolerability, and Safety of Exclusive Palatable Elemental Diet in Patients With Intestinal Microbial Overgrowth. Clin. Gastroenterol. Hepatol. 2025, 23, 2306–2317.e7. [Google Scholar] [CrossRef] [PubMed]
- Parrish, A.; Boudaud, M.; Kuehn, A.; Ollert, M.; Desai, M.S. Intestinal mucus barrier: A missing piece of the puzzle in food allergy. Trends Mol. Med. 2022, 28, 36–50. [Google Scholar] [CrossRef]
- Wehkamp, J.; Schmid, M.; Stange, E.F. Defensins and other antimicrobial peptides in inflammatory bowel disease. Curr. Opin. Gastroenterol. 2007, 23, 370–378. [Google Scholar] [CrossRef]
- Meade, K.G.; O’Farrelly, C. β-Defensins: Farming the Microbiome for Homeostasis and Health. Front. Immunol. 2019, 9, 3072. [Google Scholar] [CrossRef]
- Pinkerton, J.W.; Kim, R.Y.; Koeninger, L.; Armbruster, N.S.; Hansbro, N.G.; Brown, A.C.; et al. Human beta-defensin-2 suppresses key features of asthma in murine models of allergic airways disease. Clin. Exp. Allergy 2021, 51, 120–131. [Google Scholar] [CrossRef]
- Lia, F.; Baron, B.; Lia, F.; Baron, B. Analysis of Polyphenolic Composition, Antioxidant Power and Stress-Response Effects of Fractionated Perilla Leaf Extract on Cells In Vitro. Biologics 2025, 5, 2. [Google Scholar] [CrossRef]
- Furnari, S.; Ciantia, R.; Garozzo, A.; Furneri, P.M.; Fuochi, V. Lactobacilli-Derived Microbe-Associated Molecular Patterns (MAMPs) in Host Immune Modulation. Biomolecules 2025, 15, 1609. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).