1. Introduction
In recent decades, the agricultural sector has witnessed a shift from synthetic pesticides to biopesticides, driven by concerns about chemical residues, pest resistance, and ecological damage [
1,
2]. Among these alternatives, plant-derived biopesticides have shown promise in controlling pests such as whiteflies and caterpillars while maintaining populations of beneficial insects [
3]. Given that crop losses induced by pathogens and pests can range 8–40% globally [
4], there is an urgent need for effective pest control strategies that are both effective and environmentally benign.
Despite growing interest in biopesticides development, significant challenges remain in large-scale production and efficacy assessment against different phytopathogens. These challenges stem primarily from the lack of established agronomic practices for cultivating biopesticide source plants and the lower acute toxicity of biopesticides relative to synthetic alternatives [
5].
A promising candidate for large-scale biopesticide production is yam bean (
Pachyrhizus spp.), which is a herbaceous climbing vine legume of the family Fabaceae. Pachyrhizus comprises five identified species:
Pachyrhizus tuberosus,
P. erosus,
P. ahipa,
P. ferrugineus, and
P. panamensis [
6]. These species are adapted to the nutrient-poor soils of the Amazon and Andean regions [
7,
8]. They produce edible tuberous roots, with yields up to 108 t·ha⁻¹ [
9,
10], while the seeds contain toxic compounds.
Yam bean seeds have demonstrated potential as biopesticide. They have proven effective against plant fungi such as
Sclerotium rolfsii [
11,
12],
Colletotrichum gloeosporioides,
Fusarium oxysporum,
Rhizopus stolonifera [
13] and
Peronophythora litchi [
14], bacteria such as
Ralstonia solanacearum [
12,
14]; and various insect pests across multiple orders (Lepidoptera, Coleoptera, Hemiptera, and Diptera), notably
Spodoptera frugiperda [
15],
Helicoverpa armigera [
16],
Bemisia tabaci [
17], and
Plutella xylostella [
18].
These toxic properties are attributed to diverse bioactive compounds, including rotenoids (rotenone, erosone, pachyrrhizone, dolineone, pachyrrhizine), isoflavonoids (neotenone and dehydroneotenone), and other secondary metabolites [
13,
19,
20,
21,
22,
23], as well as defensins such a SPE10 protein [
24]. Among these, rotenone already was used as a commercial pesticide until the 1940s, when DDT (Dichlorodiphenyltrichloroethane) became dominant.
In recent decades, rotenone has regained commercial interest due to its high efficacy, rapid biodegradability [
25,
26], and relatively low acute oral toxicity (LD
50 = 100 mg·kg⁻¹ in rats and 300–500 mg·kg⁻¹ in humans) [
27,
28]. Complete degradation occurs within 7-8 days in plant and within 10 days in soil [
29,
30].
Therefore, the
Pachyrhizus breeding program at National Institute for Amazonian Research (INPA, Brazil) expanded to include a research line targeting seed toxicity and yield, in addition to the traditional focus on tuberous root yield improvement [
10,
31]. Of the 64 INPA progenies nine showed high efficacy against
Sclerotium rolfsii in vitro [
11], but have not yet been evaluated for seed yield or screened against other economically important Amazonian pathogens.
Ralstonia solanacearum (RS) is another critical pathogen in Amazonian agriculture, causing bacterial wilt in economically important crops such as tomato, banana, and hot peppers [
32,
33]. This pathogen represents the primary constraint to tomato production in Amazonas State, Brazil, where efficient control methods remain unavailable. Therefore, this study evaluated the nine promising progenies identified by de Lima [
11] for their ability to control RS at two concentrations (0.5% and 5% w/v) and assessed seed yield under trellis cultivation to maximize seed production.
2. Results
2.1. Agronomical Performance
Seed yield among progenies ranged from 0.56 to 0.98 t·ha⁻¹, with no statistically significant differences detected (
Table 1). Similarly, stand and pod yield showed no significant variation. However, three morphological traits exhibited significant differences (
p < 0.05): mass of 100 seeds, pod length, and pod width (
Table 1).
Progenies P14, P20, and P23 exhibited the highest numerical seed yields (0.83-0.98 t·ha⁻¹) and pod yields (1.84-2.21 t·ha⁻¹) (
Table 1). Broad-sense heritability (H²) ranged from 0.77 to 0.85 for pod morphological traits (pod length, pod width, and 100-seed mass) (
Table 1). However, H
2 for stand, pod yield, pods per plant and seed yield was considered zero because mean square of error exceeded to mean square of progenies, preventing estimation of genetic variance.
2.2. Effect of 0.5% Yam Bean Extracts
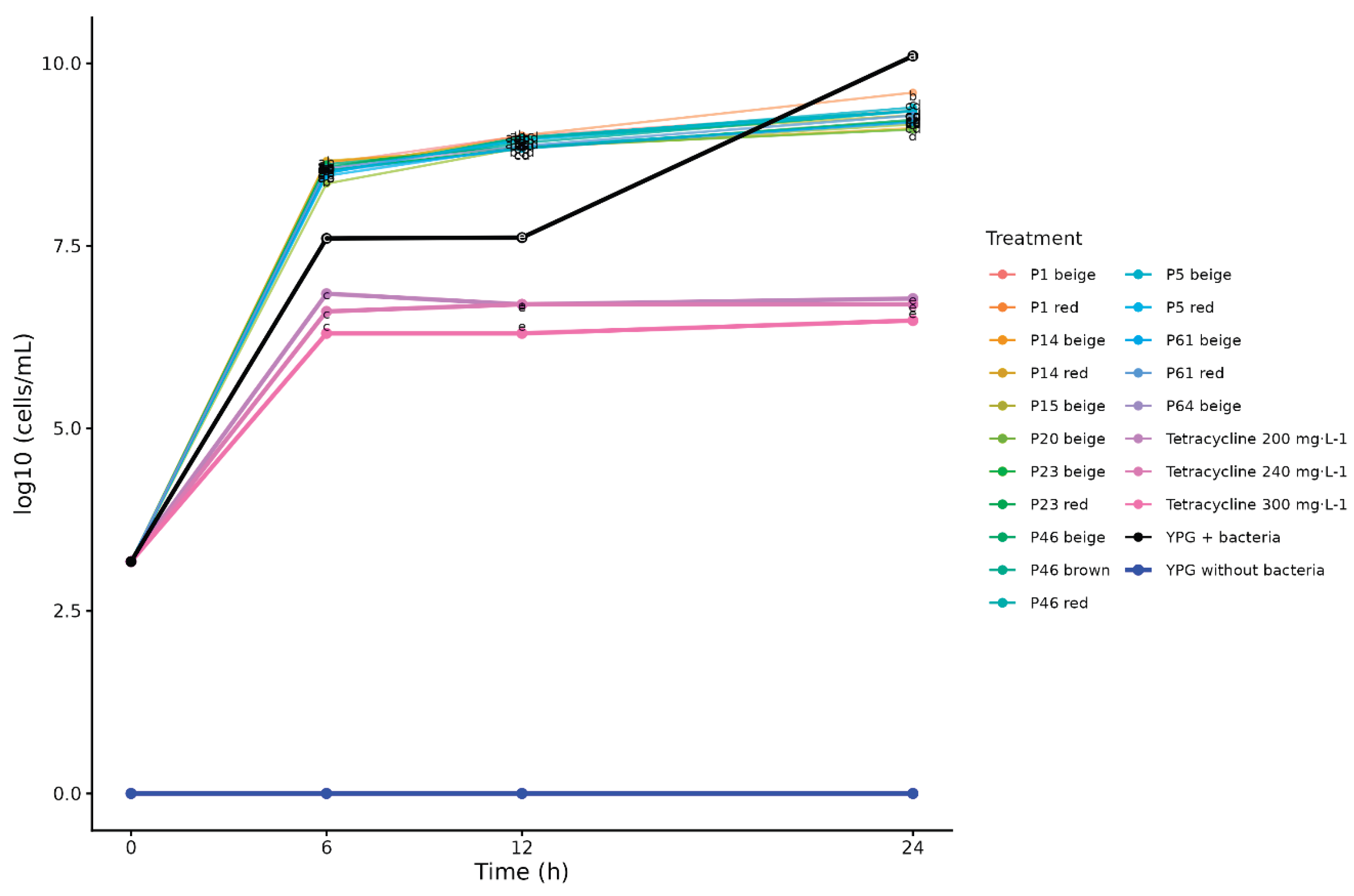
At 6 and 12 h of incubation, Duncan's test (
p < 0.05) showed that the YPG (yeast-peptone-glucose) media with extracts had higher bacterial cell counts than the bacterial growth control (YPG + bacteria, no extracts) (
Figure 1). In contrast, at 24 h, the bacterial growth control exhibited significantly higher cell numbers than the extract treatments (
Figure 1). These results indicate that the extracts initially promoted bacterial multiplication up to 12 h, but after 24 h, they exhibited toxic effects on RS. While the bacterial growth control increased exponentially from 7.06 to 10.10 log
10 cells·mL⁻¹ between 6 and 24 h, extract-treated samples showed limited growth from ~8,54 to ~9,30 log₁₀ cells·mL⁻¹ on average, representing an approximately 6-fold reduction in bacterial multiplication.
Regarding the effect of tetracycline (200, 240, and 300 mg·L⁻¹), bacterial cell counts at 6 h were significantly lower than those in the bacterial growth control (
Figure 1) and remained stable at approximately 6.60 log₁₀ cells·mL⁻¹ across all time points (6, 12, and 24 h). These results indicate that tetracycline exerted rapid antibacterial activity, with inhibition occurring within the first 6 h of incubation and being maintained thereafter.
Among all progenies tested, P14 beige and P23 beige exhibited the highest antibacterial efficacy at 24 h, with cell counts of 9.11 and 9.09 log₁₀ cells·mL⁻¹, respectively (p < 0.05). These extracts reduced RS multiplication by approximately 10-fold compared to the bacterial growth control. In contrast, P1 red (9.60 log10 cells·mL⁻¹) showed the lowest antibacterial activity, limiting bacterial growth by only 3-fold.
2.3. Effect of 5% Yam Bean Extracts
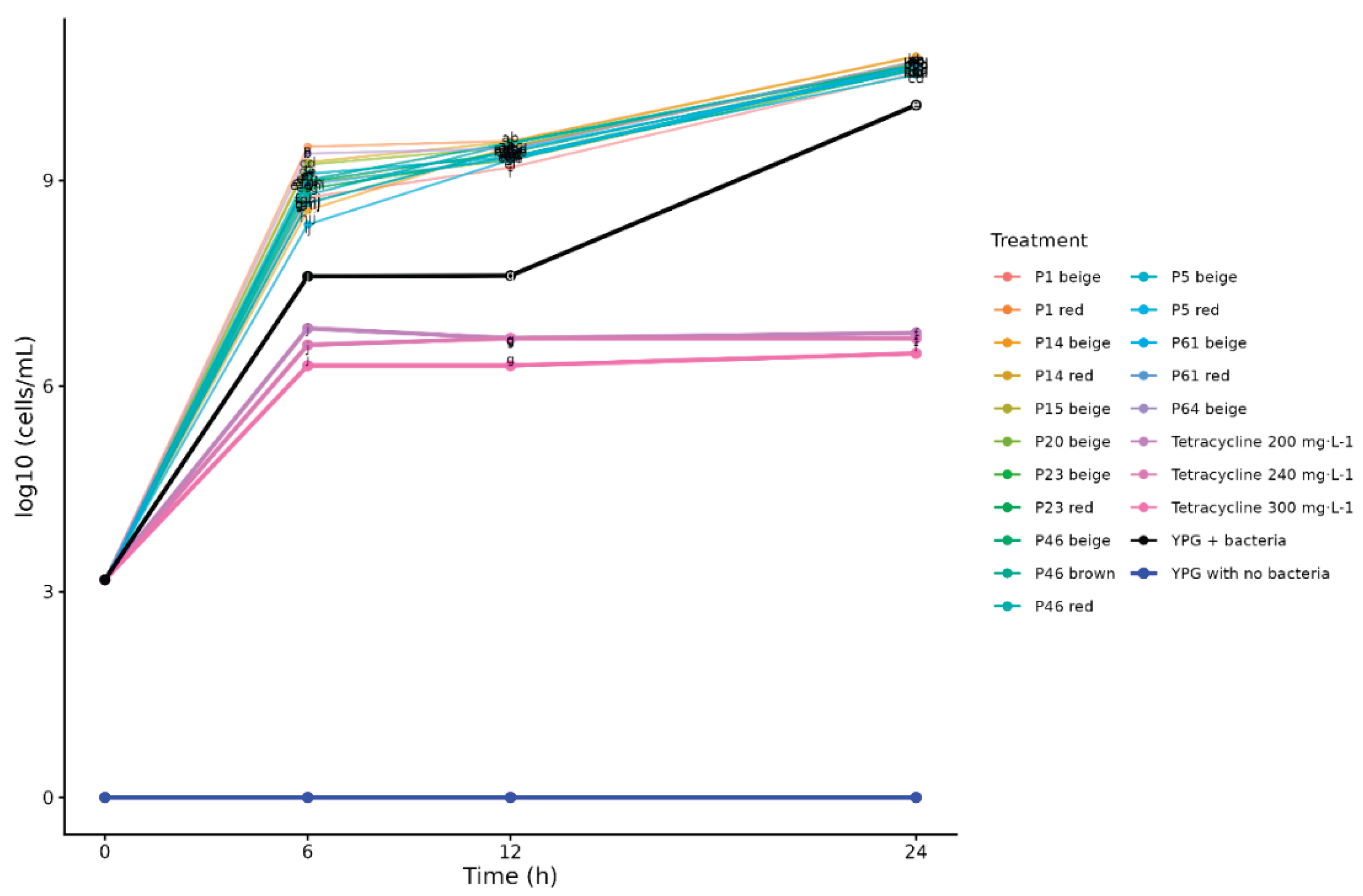
At 6, 12, and 24 h of incubation, Duncan's test (
p < 0.05) showed that the YPG culture media with 5% extracts had higher bacterial cell counts than the bacterial growth control (YPG + bacteria) (
Figure 2). This indicates that the 5% extracts promoted bacterial multiplication throughout the 24 h incubation period.
Figure 2 also shows that cell numbers in these extracts at 24 h had reaching up 10.82 log
10 cells mL⁻¹ and bacterial growth control (10.10 log
10 cells·mL⁻¹), indicating that the toxic effect on RS remained minimal at 24 h post-inoculation.
At 24 h, progenies differed significantly in cell concentration: P5 beige yielded the lowest values (10.54 log10 cells mL⁻¹) and P1 red and P14 red the highest (10.82 and 10.81 log10 cells mL⁻¹, respectively). This pattern may reflect differences in extract composition that affect RS growth, potentially including nutrient availability.
Despite their strong inhibition at 0.5% after 24 h, P14 beige and P23 beige supported only modest multiplication at 5%, reaching 10.69 and 10.63 log10 cells mL⁻¹, respectively. This suggests that these progenies may still exhibit comparatively greater antibacterial potential after 24 h, even at the higher extract concentration.
3. Discussion
3.1. Seed Yield and Agronomical Traits
The results showed seed yields of nine yam bean progenies ranging from 0.56 to 0.98 t·ha⁻¹ when grown in Spodosol in the Central Amazon. Broad-sense heritability (H2) was high to pod length (H2=0,77), pod width (H2=0,81), and 100-seed mass (H2=0.85), whereas H2 for pod yield and seed yield was zero.
Duncan’s test showed no significant differences in seed yield among the nine progenies, whose yields ranged from 0.56 to 0.98 t ha⁻¹ under the evaluated conditions. However, the highest mean yield approached 1 t ha⁻¹, which is greater than the seed yields reported for about 36% of other grain legume species [
34]. The evaluated progenies had not been specifically bred for seed yield, and genetic variability was observed within progenies, for example in flower (white and violet) and seed color. These findings indicate that the species has considerable potential for increasing seed yield if breeding methods appropriate for allogamous plants are applied, such as population improvement or selection of pure lines and hybrids.
The present study also highlighted the advantage of growing
Pachyrhizus using a trellis system. Mean pod yield reached 1.74 t ha⁻
1 (
Table 1), which was 164% higher than the 0.66 t ha⁻¹ reported by [
31] for 20 INPA progenies cultivated at the same site without staking. The use of stakes for vertical plant support was the main management difference between experiments. This substantial yield improvement suggests that trellis systems are essential for maximizing pod and seed production in
Pachyrhizus spp.
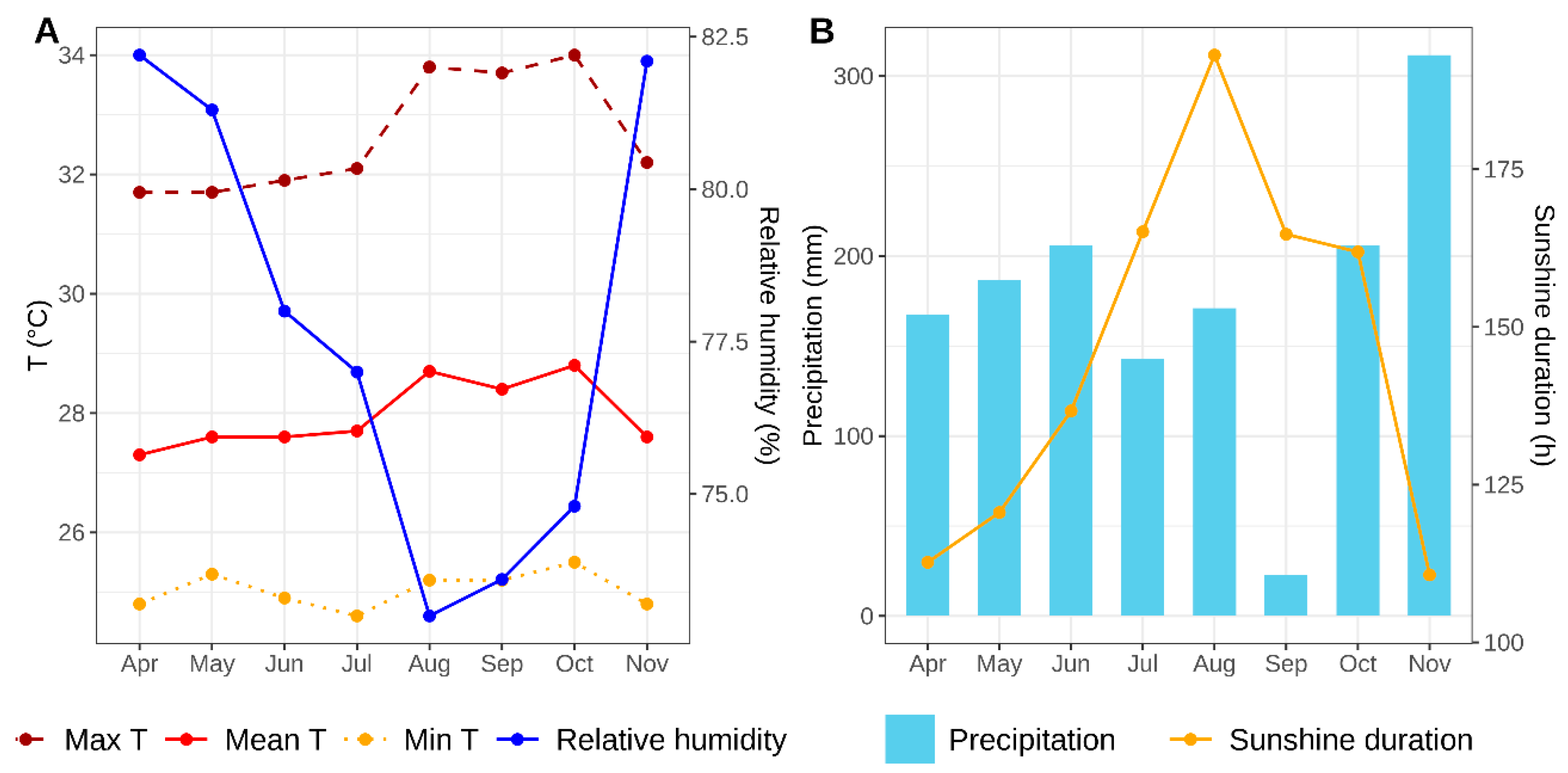
Water availability critically influences seed productivity in
Pachyrhizus spp. This study evaluated progenies on sandy Spodosol without supplemental irrigation (except the first month post-transplanting) during a seven-month growing cycle (April–November) spanning the Central Amazon dry season (June–October). Zanklan et al [
9] demonstrated seed yield reductions under water stress, with
P. ahipa declining from 3.04 to 1.10 t ha⁻¹ (64% reduction) and
P. erosus from 5.66 to 4.69 t ha⁻¹ (17% reduction). Cultivation on Latosols during the rainy season (beginning November–December) could enhance seed yields, as these soils offer better fertility and structure than sandy Spodosols, and natural precipitation would eliminate the need for supplemental irrigation.
The estimates of H2 of pod and seed yield was zero. It indicates that the selection cannot be performed. However, high H2 was observed among progenies for pod morphological traits (size and shape), it indicates that these traits can be phenotypically selected with gain by selection.
3.2. Effect of Pachyrhizus Extracts
Antibacterial assays revealed that 0.5% (w/v) yam bean extracts significantly inhibited RS multiplication by 3- to 10-fold at 24 h post-inoculation, while 5.0% (w/v) extracts stimulated bacterial growth throughout the evaluation period (6, 12 and 24 h).
After 6 and 12 hours of incubation, 0.5% yam bean extracts stimulated bacterial proliferation 10-fold and 25-fold, respectively, compared to the bacterial growth control (YPG medium + bacteria, no extract). However, at 24 hours, the extracts reduced bacterial growth 10-fold relative to the same control. These findings corroborate Silva et al [
12], who tested yam bean extracts at various concentrations (0.01%, 0.05%, and 0.5%) against RS and observed that 0.5% extracts provided effective bacterial control at 48 h, confirming the time-dependent antibacterial activity observed in the present study.
This biphasic pattern suggests that water-soluble nutrients in the extract (amino acids, simple sugars, minerals) initially support bacterial metabolism and growth, while antimicrobial secondary metabolites exert delayed inhibitory effects, possibly due to time-dependent accumulation or metabolic activation.
One of these metabolites is rotenone, which demonstrates sustained cytotoxic activity lasting up to 7 or 8 days [
29,
30]. This extended activity profile suggests that antibacterial effects may continue to intensify beyond 24 hours. Therefore, while 24-hour assessments provide valuable initial toxicity data, extended monitoring at multiple timepoints (48-168 hours) is warranted to determine the full spectrum of extract antimicrobial efficacy.
It was also observed that tetracycline inhibits bacterial multiplication from 6 to 24 hours with the same intensity and with a greater inhibitory capacity than the yam bean extracts. These results confirm the efficiency of tetracycline on RS in doses from 240 to 700 mg L-1 [
12,
35]. Despite this efficacy, soil application is not recommended because tetracycline significantly disrupts soil microbial communities, with effects observed at concentrations as low as 5 mg·kg⁻¹ and increasing substantially at 25-500 mg·kg⁻¹ [
36].
At 24 h and 0.5% concentration, progenies P23 beige, P14 beige, and P20 beige reduced the bacterial population by up to 90%, 89%, and 88%, respectively, relative to the bacterial growth control. These findings indicate that yam bean progenies may be promising candidates for developing biocontrol strategies against RS.
The 5% extracts promoted bacterial growth at 6, 12 and 24 h compared with the bacterial growth control (YPG + bacteria, no extract), suggesting that the nutritional effects outweigh antimicrobial activity at this concentration. Therefore, extended incubation studies (48-192 hours) are needed to determine whether antimicrobial effects emerge at later timepoints, given that rotenone persists for 7 or 8 days [
29,
30].
3.3. Limitations and Implications
The failure to detect genetic variation for seed yield can be attributed to insufficient sampling, as only four plants per experimental unit were evaluated. Previous researches indicate that 14 to 30 plants per progeny are required to adequately capture genetic diversity in half-sibling families [
37,
38].
This study evaluated the antibacterial activity of
Pachyrhizus extracts against RS phylotype II isolate FIO104B collected from tomato in Iranduba, Amazonas, Brazil. Given the high genetic diversity and recombination within RS, particularly in virulence-related genes [
39], these findings require validation for others phylotypes.
Another limitation of the present study was the restricted 24 h incubation period. Future studies should evaluate extract efficacy over extended timeframes (48-168 hours) to determine the complete duration of antibacterial activity.
These findings indicate that progenies P14, P20, and P23 are promising materials for breeding programs focused on dual purposes: seed production and biopesticide development. The observed high genetic variability and frequent cross-pollination suggest that breeding methods for allogamous (cross-pollinating) plants should be applied to yam bean.
The 0.5% concentration was more effective than 5%, indicating that lower concentrations can be used in commercial formulations to reduce production costs. The biphasic activity (initial stimulation then inhibition) suggests that application timing is critical for maximizing antimicrobial effects.
4. Materials and Methods
4.1. Field Experiment
4.1.1. Location
The work was carried out at the Horticultural Research Station of INPA at Km 14 of highway AM-010, municipality of Manaus, Amazonas, Brazil. The soil was classified as a Spodosol, with a sandy texture. Climatic variables during the experimental period were obtained from INMET (2025) and are presented in
Figure 3.
4.1.2. Material
Seeds from nine yam bean progenies (P1, P5, P14, P15, P20, P23, P46, P61, and P64), previously selected for toxicity against
Sclerotium rolfsii [
11], were obtained from the INPA Vegetable Germplasm Bank. Due to within-progeny color variation, seeds were grouped by color: beige (P1, P5, P14, P15, P20, P23, P64), brown (P46), and red (P61).
4.1.3. Soil Preparation
First, a composite soil sample was collected and analyzed at the Soil Laboratory of the Federal University of Amazonas. The soil exhibited the following characteristics: pH 4.8; P 168 mg dm⁻³; K 22 mg dm⁻³; Ca 2.8 cmolc dm⁻³; Mg 0.4 cmolc dm⁻³; Na 9.0 mg dm⁻³; H+Al 1.82 cmolc dm⁻³; CEC (pH 7) 5.12 cmolc dm⁻³; effective CEC 3.3 cmolc dm⁻³; and base saturation 64.5%. On the day of transplanting, the soil preparation was conducted using a heavy tractor (Massey Ferguson 250X) equipped with disc plows, making three passes. Immediately, furrows were created at 1-m intervals using a micro-tractor (Yanmar TC-14) and mineral fertilization (N-P-K at 0-40-40 kg·ha⁻¹) (Stamford et al. 1999) and dolomitic lime (2 t·ha⁻¹) were applied in the furrows and incorporated into the soil using the micro-tractor.
4.1.4. Seedling Preparation and Field Establishment
Seeds were pre-soaked in water for 24 h at room temperature and sown in April 2021 in 300 mL cups filled with commercial substrate (Tropstrato HT Hortaliças, Vida Verde, Mogi Mirim, São Paulo, Brazil). Thirty-day-old seedlings were transplanted to the field in a randomized complete block design with nine treatments (progenies), four replications, and four plants per plot arranged in double rows (1 × 1 m spacing)
4.1.5. Cultivation
Plants were trained using 2.5-m gliricídia stakes (Gliricidia sepium (Jack.) Kunth) positioned adjacent to each plant. Stakes were secured with twine to galvanized wire (No. 12) that was stretched longitudinally along the planting rows and supported by bamboo poles at 2-m height. Manual weeding was conducted at 15-day intervals, and irrigation management was restricted to the first month following field transplanting.
4.1.6. Harvest and Evaluation
Pods were harvested three times between October and November 2021 upon reaching full maturity and dryness. For each plot, the following agronomic traits were measured: plant stand, pod yield, seed yield (both in t ha⁻¹), 100-seed mass (g), number of pods per plant, and pod dimensions. Pod length and width (cm) were determined from ten randomly selected pods per plot. Yield calculations followed the formula: Yield (t ha⁻¹) = [Average mass per plant (kg) × 10,000 plants ha⁻¹] ÷ 1,000. Broad-sense heritability (H
2) also was estimated by formula (1).
where
: = Genetic variance,
=Error variance, and r=number of blocks=4
4.2. In Vitro Experiment
4.2.1. Material
Seeds from nine
Pachyrhizus spp. progenies obtained from the field experiment were separated by color (beige, red, and brown) (
Figure 4). Due to limited seed availability, P20 (red) and P64 (red) were excluded, resulting in 16 samples for toxicity assay.
The Ralstonia solanacearum phylotype II isolate FIO104B was obtained from the INPA Phytopathogenic Microorganism Collection. This isolate was originally collected from tomato in Iranduba, Amazonas, Brazil. The bacterial culture was grown on YPGA (yeast-peptone-glucose-agar) medium in Petri dishes at 28°C for 48 hours.
4.2.2. Preparation of Aqueous Seed Extracts
Seeds were sterilized as follows: (i) immersion in 70% ethanol for five minutes; (ii) immersion in commercial bleach for five minutes; and (iii) ten washes with distilled water. Seeds were dried with paper towels, stored in aluminum foil envelopes, and then dried in an oven at 50°C for 48 hours. They were then ground in a ball mill for five minutes at 15 Hz·s⁻¹. The resulting powder was stored in airtight plastic containers treated with ultraviolet light for 20 minutes, placed in aluminum envelopes, and kept at -17°C.
The extract was prepared by macerating the powder from each progeny with sterilized water (1:10, w/v) at 10 °C for 48 hours. After maceration, the extract was purified with a 3.0 µm membrane filter followed by centrifugation at 5,500 rpm for 15 minutes. The supernatant was filtered again through 0.45 µm and 0.22 µm mesh membranes. The purified extract at a concentration of 10% was stored in sterile Erlenmeyer flasks and frozen at -17°C.
The extracts were then adjusted to 0.5% (w/v) and 5% (w/v) concentrations by mixing them with sterile YPG (yeast-peptone-glucose) medium. Controls consisted of antibiotic control (tetracycline® mixed with the YPG medium to obtain final concentrations of 200, 240, and 300 mg·L⁻¹), bacterial growth control (YPG + bacteria, no extract), and sterility control (YPG without bacteria and extract). All treatments were adjusted to a 20 mL final volume.
4.2.3. In Vitro Toxicity Assay
Bacterial inoculation was performed by adding 30 µL of the suspension (1 × 10⁶ cells mL⁻¹) to 20 mL of extract (Oliveira and Vidor 1984), resulting in a final concentration of 1.5 × 10³ cells mL⁻¹. Samples were incubated in a shaker at 150 rpm and 25 °C. Aliquots were collected at 6, 12, and 24 h and stored at −17 °C for subsequent cell counting in a Neubauer chamber.
4.2.4. Statistical Analysis
Data were subjected to analysis of variance (ANOVA), and means were compared using Duncan’s multiple range test (p < 0.05). All analyses were conducted using the PROC GLM procedure of SAS 9.4 software (SAS Institute Inc. 2013).
5. Conclusions
Progenies P14, P20 and P23 are promising for seed production and for the development of biopesticides targeting Ralstonia solanacearum. These progenies should be evaluated in experimental designs with larger plot sizes (>14 plants per plot) for seed yield following breeding methods for half-sib families.
The 0.5% (w/v) extracts exhibited time-dependent activity, stimulating bacterial multiplication at earlier time points but reducing R. solanacearum growth at 24 h. Conversely, the 5% (w/v) extracts promoted bacterial growth at 24 h, highlighting the need for assessments beyond 24 h to evaluate any delayed inhibitory effects.
Author Contributions
For research articles with several authors, a short paragraph specifying their individual contributions must be provided. The following statements should be used “Conceptualization, C.A.T.B.; methodology, C.A.T.B., L.A.O.; software, C.A.T.B.; validation, R.G.G.V.; formal analysis, C.A.T.B.; investigation, R.G.G.V.; resources, C.A.T.B. and L.A.O.; data curation, R.G.G.V; writing—original draft preparation, R.G.G.V.; writing—review and editing, R.G.G.V., C.A.T.B.; visualization, R.G.G.V., C.A.T.B.; supervision, C.A.T.B.; project administration, C.A.T.B. , L.A.O.; funding acquisition, C.A.T.B. , L.A.O. All authors have read and agreed to the published version of the manuscript.”.
Funding
This research received no external funding.
Data Availability Statement
Acknowledgments
The authors would like to thank the Plant Breeding Laboratory team at INPA for its logistical and institutional support. Financial support for this publication was provided by the Fundação de Amparo à Pesquisa do Estado do Amazonas (FAPEAM) through the PROGRAMA DE APOIO À DISSEMINAÇÃO DO CONHECIMENTO CIENTÍFICO, TECNOLÓGICO E INOVADOR NO ÂMBITO DA PÓS-GRADUAÇÃO STRICTO SENSU—DIVULGA CT&I (Edital N.º 017/2024) and POSGRAD/FAPEAM.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Abbreviations
The following abbreviations are used in this manuscript:
| RS |
Ralstonia solanacearum |
| YPG |
yeast-peptone-glucose |
| INPA |
National Institute for Amazonian Research |
| LD50
|
median lethal dose |
References
- Verma, N.S.; Kuldeep, D.K.; Chouhan, M.; Prajapati, R.; Singh, S.K. A Review on Eco-Friendly Pesticides and Their Rising Importance in Sustainable Plant Protection Practices. Int. J. Plant Soil Sci. 2023, 35, 200–214. [Google Scholar] [CrossRef]
- Gundreddy, R.R.; Bv, J.; M, S.; Darjee, S.; Alekhya, G. Recent Advances in Biopesticide Formulations for Targeted Insect Pest Management: A Review. J. Exp. Agric. Int. 2024, 46, 876–889. [Google Scholar] [CrossRef]
- Tembo, Y.; Mkindi, A.G.; Mkenda, P.A.; Mpumi, N.; Mwanauta, R.; Stevenson, P.C.; Ndakidemi, P.A.; Belmain, S.R. Pesticidal Plant Extracts Improve Yield and Reduce Insect Pests on Legume Crops Without Harming Beneficial Arthropods. Front. Plant Sci. 2018, 9, 1425. [Google Scholar] [CrossRef] [PubMed]
- Savary, S.; Willocquet, L.; Pethybridge, S.J.; Esker, P.; McRoberts, N.; Nelson, A. The Global Burden of Pathogens and Pests on Major Food Crops. Nat. Ecol. Evol. 2019, 3, 430–439. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, J.; Amin, H. Implication of Biopesticides for Insect Pest Management: An Alternative of Chemical Pesticides. Bangladesh J. Entomol. 2023, 31. [Google Scholar] [CrossRef]
- Sørensen, M. Yam Bean: Pachyrhizus DC.—Promoting the Conservation and Use of Underutilized and Neglected Crops. 4; Bioversity International (IPGRI): Rome, Italy, 1996; ISBN 92-9043-293-8. [Google Scholar]
- Nielsen, P.E.; Sørensen, M.; Halafihi, M. Yield Potential of Yam Bean Pachyrhizus erosus (L.) Urban Accessions in the Kingdom of Tonga, South Pacific. Trop. Agric. 2000, 77, 174–179. [Google Scholar]
- Tapia, C.; Sørensen, M. Morphological Characterization of the Genetic Variation Existing in a Neotropical Collection of Yam Bean, Pachyrhizus tuberosus (Lam.) Spreng. Genet. Resour. Crop Evol. 2003, 50, 681–692. [Google Scholar] [CrossRef]
- Zanklan, A.S.; Ahouangonou, S.; Becker, H.C.; Pawelzik, E.; Grüneberg, W.J. Evaluation of the Storage Root-Forming Legume Yam Bean (Pachyrhizus spp.) under West African Conditions. Crop Sci. 2007, 47, 1934–1946. [Google Scholar] [CrossRef]
- Silva, E.S.D.; Silva Filho, D.F.D.; Ticona-Benavente, C.A. Diversity of Yam Bean (Pachyrhizus spp. Fabaceae) Based on Morphoagronomic Traits in the Brazilian Amazon. Acta Amaz. 2016, 46, 233–240. [Google Scholar] [CrossRef]
- de Lima, R.G.; Ticona-Benavente, C.E.A.; da Silva Filho, D.F.; e Silva, L.S.; Guimarães, R.G.V.; Hanada, R.E. Pachyrhizus Spp. Seed Extracts Control the Sclerotium rolfsii Sacc. Mycelial Growth. J. Med. Plants Res. 2021, 15, 289–296. [Google Scholar] [CrossRef]
- Silva, T.M.; Guimarães, R.G.; Martins, A.K.; Silva Filho, D.F.; Ticona-Benavente, C.A. Pachyrhizus Toxicity: Genetic Variability of Mature and Immature Seeds and Its Effects on Sclerotium rolfsii and Ralstonia solanacearum. J. Med. Plants Res. 2023, 17, 8–15. [Google Scholar] [CrossRef]
- Barrera-Necha, L.; Bautista-Baños, S.; Bravo-Luna, L.; García-Suárez, F.; Alavez-Solano, D.; Reyes-Chilpa, R. Antifungal Activity of Seed Powders, Extracts, and Secondary Metabolites of Pachyrhizus erosus (L.) Urban (Fabaceae) against Three Postharvest Fungi. Rev. Mex. Fitopatol. 2004, 22, 356–361. [Google Scholar]
- Zhang, H.; Xu, D.; Pan, R.; Xu, H. Screening for antimicrobial activities in some plant extracts. Acta Phytopathol. Sin. 2008, 38, 441–444. [Google Scholar]
- Agustini, M.; Sartiami, D. Dadang Effectiveness of soursop leaves, yam bean seeds, and javanese long pepper fruits extracts against larvae of fall armyworm (Spodoptera frugiperda (Smith)) (Lepidoptera: Noctuidae). J. Entomol. Indones. Indones. J. Entomol. 2024, 21, 224–233. [Google Scholar] [CrossRef]
- Ravindranath, N.; Samanta, A.; Samanta, S. Dose-Dependent Toxicity and Field Efficacy of PEG-Emulsified Botanical Formulations against Chickpea Pod Borer (Helicoverpa Armigera). J. Entomol. Res. 2025, 49, 632–637. [Google Scholar] [CrossRef]
- Kumar, R.; Singh, P. Efficacy of Certain Insecticides against Bemisia tabaci (Gennadius) in Okra. Indian J. Entomol. 2022, 84, 348–349. [Google Scholar] [CrossRef]
- Basukriadi, A.; Wilkins, R. Oviposition Deterrent Activities of Pachyrhizus erosus Seed Extract and Other Natural Products on Plutella xylostella (Lepidoptera: Plutellidae). J. Insect Sci. 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Phrutivorapongkul, A.; Lipipun, V.; Ruangrungsi, N.; Watanabe, T.; Ishikawa, T. Studies on the Constituents of Seeds of Pachyrrhizus erosus and Their Anti Herpes Simplex Virus (HSV) Activities. Chem. Pharm. Bull. (Tokyo) 2002, 50, 534–537. [Google Scholar] [CrossRef]
- Lautié, E.; Rozet, E.; Hubert, P.; Leclercq, J.Q. Quantification of Rotenone in Seeds of Different Species of Yam Bean (Pachyrhizus sp.) by a SPE HPLC–UV Method. Food Chem. 2012, 131, 1531–1538. [Google Scholar] [CrossRef]
- Lautié, E.; Rozet, E.; Hubert, P.; Vandelaer, N.; Billard, F.; Zum Felde, T.; Grüneberg, W.J.; Quetin-Leclercq, J. Fast Method for the Simultaneous Quantification of Toxic Polyphenols Applied to the Selection of Genotypes of Yam Bean (Pachyrhizus sp.) Seeds. Talanta 2013, 117, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Leuner, O.; Havlik, J.; Budesinsky, M.; Vrkoslav, V.; Chu, J.; Bradshaw, T.D.; Hummelova, J.; Miksatkova, P.; Lapcik, O.; Valterova, I.; et al. Cytotoxic Constituents of Pachyrhizus tuberosus from Peruvian Amazon. Nat. Prod. Commun. 2013, 8, 1423–1426. [Google Scholar] [CrossRef]
- Catteau, L.; Lautié, E.; Koné, O.; Coppée, M.; Hell, K.; Pomalegni, C.; Quetin-Leclercq, J. Degradation of Rotenone in Yam Bean Seeds (Pachyrhizus sp.) through Food Processing. J. Agric. FOOD Chem. 2013, 61, 11173–11179. [Google Scholar] [CrossRef]
- Song, X.; Wang, J.; Wu, F.; Li, X.; Teng, M.; Gong, W. cDNA Cloning, Functional Expression and Antifungal Activities of a Dimeric Plant Defensin SPE10 from Pachyrrhizus erosus Seeds. Plant Mol. Biol. 2005, 57, 13–20. [Google Scholar] [CrossRef]
- Cabras, P.; Caboni, P.; Cabras, M.; Angioni, A.; Russo, M. Rotenone Residues on Olives and in Olive Oil. J. Agric. Food Chem. 2002, 50, 2576–2580. [Google Scholar] [CrossRef] [PubMed]
- Isman, M.B. Botanical Insecticides in the Twenty-First Century—Fulfilling Their Promise? Annu. Rev. Entomol. 2020, 65, 233–249. [Google Scholar] [CrossRef] [PubMed]
- De Wilde, A.R.; Heyndrickx, A.; Carton, D. A Case of Fatal Rotenone Poisoning in a Child. J. Forensic Sci. 1986, 31, 1492–1498. [Google Scholar] [CrossRef] [PubMed]
- Mascaro, U.C.P.; Rodrigues, L.A.; Bastos, J.K.; Santos, E.; Costa, J.P. Valores de DL50 Em Peixes e No Rato Tratados Com Pó de Raízes de Derris spp. e Suas Implicações Ecotoxicológicas. Pesqui. Veterinária Bras. 1998, 18, 53–56. [Google Scholar] [CrossRef]
- Cavoski, I.; Caboni, P.; Sarais, G.; Miano, T. Degradation and Persistence of Rotenone in Soils and Influence of Temperature Variations. J. Agric. Food Chem. 2008, 56, 8066–8073. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zhang, N.; Wang, K.; Li, W.; Li, H.; Zhang, Z. Dissipation and Residue of Rotenone in Cabbage and Soil under Field Conditions. Bull. Environ. Contam. Toxicol. 2013, 91, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Silva, E.S.; Atroch, A.L.; de Jesus, R.S.; Bitencourt, M.A.F.; Ticona-Benavente, C.A. Genotype x Year Interaction for Agronomical Traits and Proximate Composition in Yam Beans (Pachyrhizus spp., Fabaceae) in the Brazilian Amazon. Genet. Mol. Res. 2022, 21, 1–12. [Google Scholar] [CrossRef]
- Netto, R.A.C.; Pereira, B.G.; Noda, H.; Boher, B. Murcha Bacteriana No Estado Do Amazonas, Brasil. Fitopatol. Bras. 2004, 29, 17–23. [Google Scholar] [CrossRef]
- Costa, S.B.; Ferreira, M.A.S.V.; Lopes, C.A. Diversidade Patogênica e Molecular de Ralstonia solanacearum Da Região Amazônica Brasileira. Fitopatol. Bras. 2007, 32, 285–294. [Google Scholar] [CrossRef]
- Cernay, C.; Pelzer, E.; Makowski, D. A Global Experimental Dataset for Assessing Grain Legume Production. Sci. Data 2016, 3, 160084. [Google Scholar] [CrossRef]
- Bawari, M.R.; Narendrappa, T. In-Vitro Evaluation of Bioagents and Antibiotics against Ralstonia solanacearum Causing Brinjal Wilt. J. Pharmacogn. Phytochem. 2019, 8, 2038–2041. [Google Scholar]
- Santás-Miguel, V.; Díaz-Raviña, M.; Martín, A.; García-Campos, E.; Barreiro, A.; Núñez-Delgado, A.; Álvarez-Rodríguez, E.; Arias-Estévez, M.; Fernández-Calviño, D. Soil Enzymatic Activities and Microbial Community Structure in Soils Polluted with Tetracycline Antibiotics. Agronomy 2021, 11, 906. [Google Scholar] [CrossRef]
- Leipold, M.; Tausch, S.; Hirtreiter, M.; Poschlod, P.; Reisch, C. Sampling for Conservation Genetics: How Many Loci and Individuals Are Needed to Determine the Genetic Diversity of Plant Populations Using AFLP? Conserv. Genet. Resour. 2020, 12, 99–108. [Google Scholar] [CrossRef]
- McGlaughlin, M.E.; Riley, L.; Brandsrud, M.; Arcibal, E.; Helenurm, M.K.; Helenurm, K. How Much Is Enough? Minimum Sampling Intensity Required to Capture Extant Genetic Diversity in Ex Situ Seed Collections: Examples from the Endangered Plant Sibara filifolia (Brassicaceae). Conserv. Genet. 2015, 16, 253–266. [Google Scholar] [CrossRef]
- Castillo, J.A.; Greenberg, J.T. Evolutionary Dynamics of Ralstonia solanacearum. Appl. Environ. Microbiol. 2007, 73, 1225–1238. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).