1. Introduction
Orchardgrass (
Dactylis glomerata L.) is a perennial herbaceous species belonging to the genus
Dactylis within the family Poaceae. Wild orchardgrass originated in temperate regions of Europe, North Africa, and Asia and is now widely distributed along forest margins, shrublands, and montane grasslands throughout temperate zones worldwide. It is recognized as one of the most important cool-season forage grasses globally [
1]. Following more than 200 years of domestication and cultivation, orchardgrass has become a high-yielding and high-quality cultivated forage due to its high protein and lipid contents and excellent palatability [
2]. Orchardgrass exhibits strong adaptability and stress tolerance, including resistance to drought, heat, poor soils, and shade. Moreover, it is particularly suitable for ecological restoration programs such as conversion of cropland to forest or grassland, establishment of agroforestry systems, and grassland reseeding, thereby playing a critical role in global agricultural production and ecological rehabilitation [
2,
3].
China is one of the major production regions of orchardgrass worldwide, with a wide distribution range. Among these regions, Xinjiang is an important production area where orchardgrass is extensively distributed along forest margins of the Tianshan Mountains and dominates montane grasslands at elevations of 1000 – 3000 m [
4].
However, during orchardgrass growth, various diseases frequently occur and seriously affect forage yield and quality. Among these, rust disease is the most extensively studied [
5,
6], followed by smut disease [
7], zonate leaf spot [
8], root rot [
9], and bacterial leaf streak [
10]. Fulcher et al. [
9] identified
Fusarium graminearum as the causal agent of orchardgrass root rot from wild plants collected in Essex County, New York, USA. Root rot is highly destructive to plant health, as it damages the normal structure and function of roots, leading to water and nutrient deficiency in aerial tissues. Affected plants typically exhibit stunted growth, reduced plant height, leaf yellowing and curling, and, in severe cases, plant death. In forage crop research, root rot diseases of cultivated forages such as alfalfa, clover, and sainfoin—particularly alfalfa—have been extensively investigated, whereas studies on root rot of wild forage species remain limited and are mostly restricted to pathogen identification.
Bipolaris sorokiniana is known to infect a wide range of plant species, including orchardgrass [
11].
During field surveys conducted from May to August in both 2024 and 2025, root rot symptoms characterized by dark brown to black necrosis of roots accompanied by leaf blight and plant dieback were consistently observed on wild orchardgrass plants in multiple townships of the Ili region of Xinjiang. To date, no studies have reported orchardgrass root rot in Xinjiang. Therefore, the present study aimed to identify the causal pathogen through morphological and molecular analyses, verify its pathogenicity, characterize its biological traits, and screen effective fungicides under laboratory conditions. The results provide a theoretical basis for future studies on disease epidemiology, pathogenic mechanisms, and integrated management strategies for orchardgrass root rot.
2. Materials and Methods
2.1. Disease Survey and Sample Collection
From May to August in 2024 and 2025, field surveys were conducted on mountain slopes, forest margins, and natural grasslands in the Ili region of Xinjiang, specifically in Zhaosu County, Xinyuan County and Tekes County. In each township three plots were established; within each plot three 1 m 1 m quadrats were sampled at random to record disease incidence. Symptomatic plants were photographed and GPS and collection metadata recorded. Diseased specimens were collected and transported to the laboratory for isolation and further analysis.
Disease incidence was calculated as:
2.2. Pathogen Isolation and Morphological Identification
Pathogens were isolated using tissue isolation. Root samples were washed with running water, and 3-mm pieces were excised from the margin between diseased and healthy tissue. Tissue pieces were surface sterilized in 75 % ethanol for 30 s, rinsed in 2 % sodium hypochlorite for 3 min, washed three times in sterile distilled water, blotted dry on sterile filter paper, and placed on PDA (60 mm diameter) plates. Plates were incubated at 25 °C in the dark for 5 days. Single-conidium isolation was performed to obtain pure cultures; purified strains were stored in 20 % glycerol at − 80 °C.
For morphological observation, isolates were activated on PDA at 25 °C for 5 – 7 days. Colony characteristics (color, texture, radial growth) were recorded. Conidiophores and conidia were observed and measured (n = 30) under a Nikon microscope (Nikon Corporation; model ECLIPSE Ni-U). Measurements included conidiophore length and conidial size. Morphological identification followed descriptions in references [
11,
12,
13,
14].
2.3. Molecular Identification
Genomic DNA was extracted from fresh mycelium using a fungal genomic DNA extraction kit (Tiangen Biotech, Beijing, China; Tiangen Biotech) according to the manufacturer’s instructions. Three loci were amplified for molecular identification: ITS (primers ITS1/ITS4), GAPDH (primers GDF1/GDR1), and TEF (primers EF1-728F/EF1-986R) [
15,
16,
17]. PCR reactions (25 µL total volume) contained 12.5 µL Taq Master Mix, 1.0 µL of each primer (forward and reverse), 1.0 µL DNA template, and sterile ultrapure water to 25 µL. PCR cycling conditions were: initial denaturation at 94 °C for 5 min; 35 cycles of 94 °C for 40 s, 58 °C for 40 s, and 72 °C for 30 s; and a final extension at 72 °C for 7 min. PCR products were checked by electrophoresis on 2 % agarose gel and Sanger-sequenced by Shanghai Bioengineering Co., Ltd. Obtained sequences were compared against the NCBI database (BLAST), and a maximum-likelihood phylogeny was constructed in MEGA 11.
2.4. Pathogenicity Tests
Forty-five-day-old potted orchardgrass plants grown at room temperature (20 – 25 °C) were used for pathogenicity assays. Roots were gently wounded with a sterile needle, and plants were immersed in a conidial suspension (1
10⁶ spores·mL
-1) for 15 min. After transplanting, 15 mL of the same suspension was applied as a soil drench to each pot. Controls consisted of wounded plants treated with sterile water by immersion and drench. Each treatment comprised three pots with five plants per pot. Disease development was recorded 15 days post-inoculation. Re-isolation from symptomatic roots was performed and morphological characteristics of re-isolated strains were compared with the original inoculum to fulfil Koch’s postulates [
18].
2.5. Biological Characteristics of the Pathogen
Mycelial plugs (5 mm diameter) were taken from the actively growing margin of 5- to 7-day-old colonies and placed mycelium-side down at the center of test plates. Plates were incubated for 5 days under the following conditions:
Temperature: 5, 10, 15, 20, 25, 30, and 35 °C.
pH: media adjusted to pH 4, 5, 6, 7, 8, 9, 10 and 11 using 1 M HCl or 1 M NaOH.
Light regime: continuous light (24 h), 12 h light / 12 h dark, and continuous dark (24 h).
Culture media: PDA, PSA, PCA, WA, CA, CMA and Czapek agar (see
Table 1 for composition).
Carbon and nitrogen sources: Czapek medium was used as the basal medium. Carbon sources tested (substituted on an equal-carbon basis) were soluble starch, glucose, lactose, mannitol, inulin, maltose and D-fructose; a no-carbon control (basal medium without added carbon) was included. Nitrogen sources tested (substituted on an equal-nitrogen basis) were ammonium sulfate, yeast extract, yeast paste, urea, potassium nitrate, beef extract and peptone; a no-nitrogen control (basal medium without added nitrogen) was included.
Each treatment had three replicates and colony diameter was measured using the crossed-line method. All data were recorded as mean colony diameters (mm) ± standard deviation. Statistical comparisons were performed as appropriate (see Section X for statistical methods).
2.6. In Vitro Fungicide Sensitivity Assays
The inhibitory activity of fungicides against the pathogen was evaluated using the mycelial growth rate method [
19]. Fungicides at different concentrations were incorporated into PDA to prepare amended media with five concentration gradients (
Table 2). PDA without fungicides served as the control. A 5-mm-diameter mycelial plug taken from the actively growing margin of the colony was placed at the center of each plate. Each treatment consisted of three replicate plates, and the experiment was conducted once. Plates were incubated at 25 °C for 5 days, after which colony diameters were measured using the crossed-line method. The percentage inhibition of mycelial growth was calculated relative to the control.
2.7. Statistical Analysis
Microsoft Excel 2019 was used to calculate EC50 values and corresponding regression equations. Statistical analyses were performed using SPSS 23.0 software. Differences among treatments were evaluated by analysis of variance (ANOVA), and mean comparisons were conducted using the least significant difference (LSD) test at p < 0.05.
3. Results
3.1. Field Disease Symptoms and Incidence
Root rot of orchardgrass initially caused poor plant vigor and stunting; symptomatic plants were markedly shorter than adjacent healthy individuals (
Figure 1). Older leaves of affected plants showed chlorosis and necrosis; leaf margins and tips became brown and necrotic, leaves folded downward, and basal stems became brown and rotted. Belowground, root systems were reduced. At advanced stages, plants were strongly stunted and chlorotic, leaves browned and desiccated over large areas, stem bases became blackened, constricted and rotten, and severe cases resulted in wilting, collapse and plant death; roots exhibited extensive black-brown rot.
Field surveys conducted in the Ili region of Ili Kazakh Autonomous Prefecture (three counties) recorded disease incidences ranging from 20.00 % to 71.32 % (Table 3). The highest incidence was recorded at Tügürqën Bulak stud farm in Zhaosu County (71.32 %), and the lowest incidence was recorded in Tuerken Township, Xinyuan County (20.00 %).
3.2. Morphological Identification
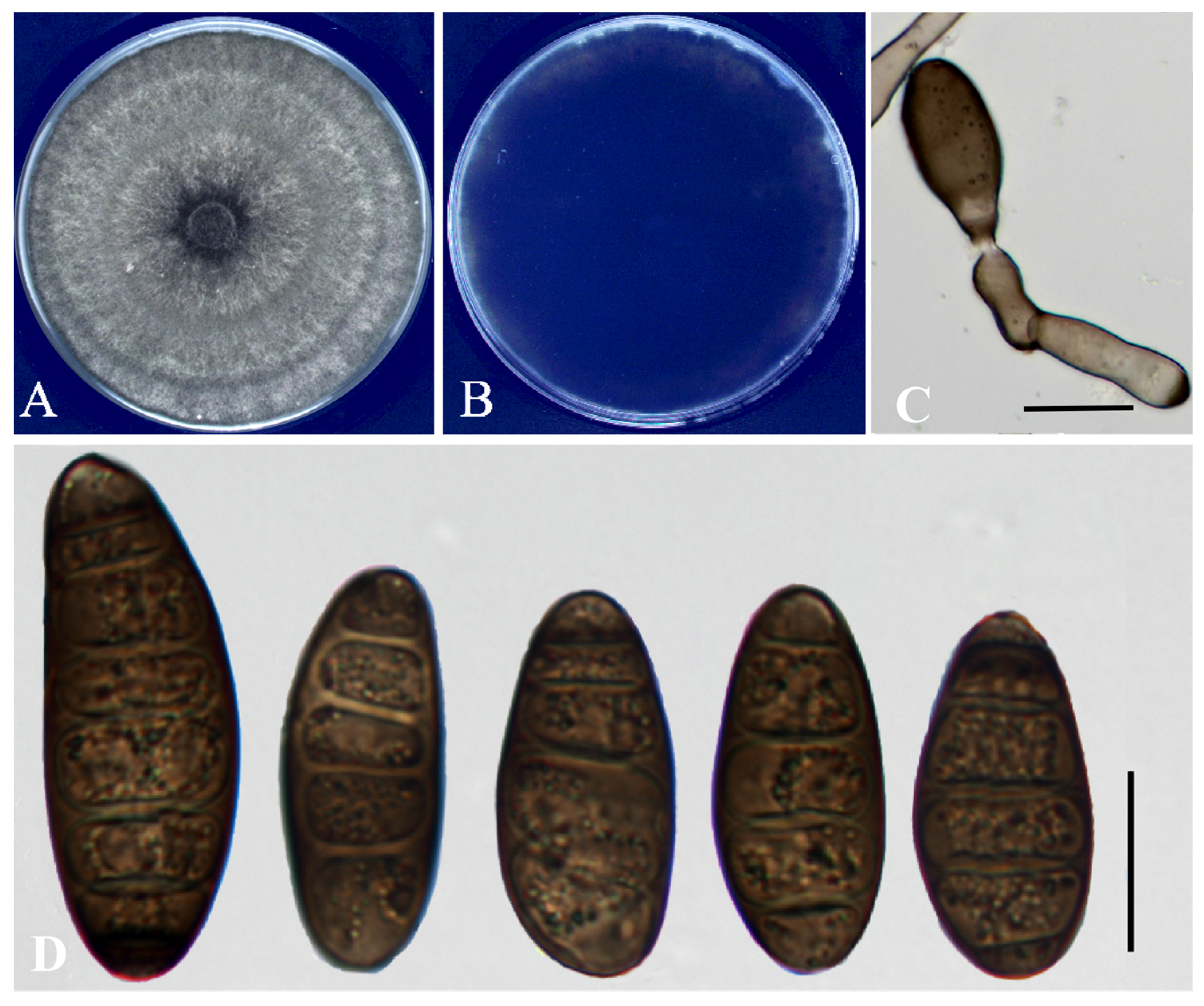
From 135 diseased orchardgrass plants sampled, 89 isolates were recovered and morphologically assigned to the genus Bipolaris. A representative isolate (YM1) was selected for detailed morphological description and subsequent experiments. After 7 days on PDA at 25 °C, colonies of YM1 were circular, gray-brown and felted. Conidiophores were olivaceous-brown, geniculate (bent at points), typically solitary and unbranched, 6 – 9 µm wide. Conidia were dark brown, ellipsoid to oblong, widest at the middle and tapered to rounded ends, with 4 – 7 transverse septa; conidial size ranged from 30.75 to 62.15 µm 10.22 to 18.31 µm. Based on these morphological characters, the isolate was preliminarily identified as a Bipolaris species.
Figure 2.
Morphological features of isolate YM1 after 7 d on PDA. (A) colony front; (B) colony reverse; (C) conidiophores and conidia; (D) conidia. Scale bars in Panels C–D = 20 µm.
Figure 2.
Morphological features of isolate YM1 after 7 d on PDA. (A) colony front; (B) colony reverse; (C) conidiophores and conidia; (D) conidia. Scale bars in Panels C–D = 20 µm.
3.3. Molecular Identification and Phylogeny
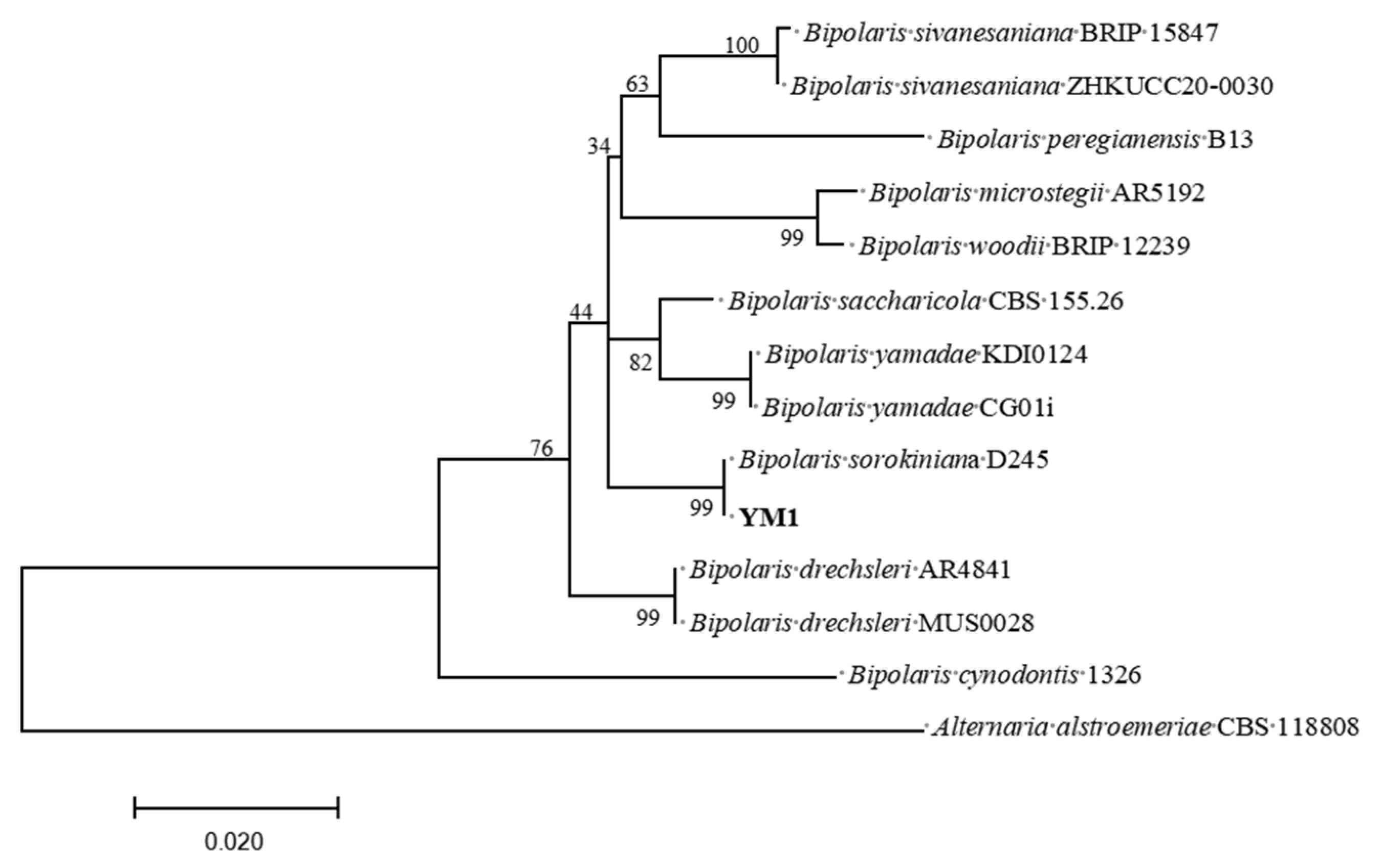
PCR amplification of ITS, GAPDH and TEF from isolate YM1 produced fragments of ≈568 bp, 420 bp and 321 bp, respectively. A concatenated ITS–GAPDH–TEF maximum-likelihood tree (rooted with Alternaria alstroemeriae CBS 118808) placed YM1 in a strongly supported clade (99 % bootstrap) with
B. sorokiniana strain D245 (GenBank accessions: ITS PX671498, GAPDH PX879951, TEF PX879952;
Figure 3). Combining morphological and multilocus molecular evidence, isolate YM1 was identified as
B. sorokiniana.
3.4. Pathogenicity of Isolate YM1
Inoculated potted orchardgrass plants developed foliar chlorosis beginning at leaf tips by 15 days post-inoculation (dpi). Symptoms progressed with increasing foliar necrosis and desiccation; by 30 dpi stem-base tissues were brown and constricted, fine roots showed brown rot, and aerial parts displayed extensive leaf desiccation—closely resembling symptoms observed in the field. Water-treated wounded controls remained healthy at 30 dpi, with stem-base tissues remaining white and only occasional tip desiccation of older leaves. Re-isolation from symptomatic roots recovered a fungus morphologically identical to the inoculated YM1 strain, satisfying Koch’s postulates and confirming YM1 as the causal agent.
Figure 4.
Disease development in orchardgrass following inoculation with YM1 at 30 dpi. (A–B) water control plants (aboveground and whole plant); (C–D) inoculated plants showing leaf desiccation and brown necrosis at the stem base. Scale bars = 10 cm.
Figure 4.
Disease development in orchardgrass following inoculation with YM1 at 30 dpi. (A–B) water control plants (aboveground and whole plant); (C–D) inoculated plants showing leaf desiccation and brown necrosis at the stem base. Scale bars = 10 cm.
3.5. Biological Characteristics of YM1
YM1 grew at all tested temperatures (5 – 35 °C), but growth differed significantly among temperatures (P < 0.05). Maximum radial growth was observed at 25 °C (mean colony diameter 62.15 mm), with reduced growth at 20 °C and 30 °C, and very slow growth (colony diameter < 11 mm) at ≤ 15 °C (
Table 4). YM1 grew across pH 4 – 11, with colony diameters > 34 mm for all pH values tested. Growth was optimal under neutral to slightly alkaline conditions: the largest colony diameter occurred at pH 7 (53.15 mm), followed by pH 8 (50.21 mm). Growth on different media indicated that YM1 utilized all tested media well (colony diameter > 46 mm). The fastest mycelial growth occurred on PSA (68.53 mm), followed by PDA (62.54 mm) and OMA (62.33 mm). When Czapek medium carbon and nitrogen sources were substituted on an equal-element basis, soluble starch supported the greatest growth among carbon sources (58.43 mm), whereas D-fructose was the least favorable carbon source (41.22 mm). For nitrogen sources, peptone supported the best growth (46.28 mm) and ammonium sulfate the poorest (35.85 mm). Light regime affected growth: colonies were largest in continuous dark (47.53 mm), intermediate under 12 h light/12 h dark (43.16 mm), and smallest under continuous light (34.68 mm).
3.6. In Vitro Fungicide Sensitivity
All nine tested fungicides inhibited mycelial growth of YM1 to varying degrees (
Table 5). Among them, 25 % difenoconazole (EW) exhibited the strongest inhibitory activity (EC
50 = 0.0706 mg·L
-1). Tebuconazole (430 g·L
-1 SC) and 0.15 % tetramycin (AS) also showed strong activity with EC
50 values of 0.3606 mg·L
-1 and 0.6815 mg·L
-1, respectively. Hymexazol (30 % AS), pyraclostrobin (25 % SC) and eugenol (20 % EW) showed moderate activity with EC
50 values in the range of ~ 5 – 11 mg·L
-1. Thiophanate-methyl (500 g·L
-1 SC), carbendazim (50 % WP) and ethylicin (80 % EC) were the least effective, with EC
50 values in the ~ 18 – 21 mg·L
-1 range.
Figure 6.
Dual-culture plates showing YM1 antagonism with fungicide-amended PDA at 7 d. (mg·L-1) Panels: (A) 50 % carbendazim; (B) 0.15 % tetramycin; (C) 25 % pyraclostrobin; (D) 25 % difenoconazole; (E) 430 g·L-1 tebuconazole; (F) 500 g·L-1 thiophanate-methyl; (G) 30 % hymexazol; (H) 80 % ethylicin; (I) 20 % eugenol.
Figure 6.
Dual-culture plates showing YM1 antagonism with fungicide-amended PDA at 7 d. (mg·L-1) Panels: (A) 50 % carbendazim; (B) 0.15 % tetramycin; (C) 25 % pyraclostrobin; (D) 25 % difenoconazole; (E) 430 g·L-1 tebuconazole; (F) 500 g·L-1 thiophanate-methyl; (G) 30 % hymexazol; (H) 80 % ethylicin; (I) 20 % eugenol.
4. Discussion
Field surveys revealed that orchardgrass root rot occurred across mountain slopes, forest margins and natural grasslands in multiple locations of Xinjiang, and that incidence was generally higher in open grassland than under forest canopy. We hypothesize that this pattern reflects differences in microenvironmental water availability and host vigor: mixed forest–grass systems typically provide more stable moisture and shading that favor more vigorous orchardgrass growth and greater disease tolerance, whereas grassland sites are relatively drier and support weaker plants with reduced resistance. This hypothesis should be tested directly by correlating soil moisture and other abiotic variables with disease incidence in future field studies.
Combining morphological characters with multilocus phylogenetic analysis, we identified the causal agent of the orchardgrass root rot in this study as
B.
sorokiniana. The morphological characters observed here are consistent with previous descriptions of
B. sorokiniana recovered from orchardgrass in Jilin Province [
11], yet to our knowledge this is the first report of
B. sorokiniana infecting wild orchardgrass in Xinjiang.
B.
sorokiniana has a broad host range spanning more than 30 plant families and is commonly associated with leaf blight diseases; however, it can infect roots and multiple other tissues on many hosts, including cereals, legumes and various woody taxa [
11,
20]. Notably, this pathogen is an important wheat pathogen that can infect multiple wheat organs throughout development, causing seedling blight, root rot, stem-base rot, foliar necrosis and reduced grain filling, and has been reported to cause yield losses approaching ~ 40 % under severe epidemics [
12,
21]. The broad host range and potential for severe crop losses indicate that the detection of
B. sorokiniana on orchardgrass expands the pathogen’s known ecological impact and highlights a possible phytosanitary risk for adjacent crops and vegetation.
Biological-characteristic assays showed that YM1 grew best at 25 °C and pH 7, with soluble starch and peptone supporting the most rapid mycelial expansion, and that growth was greater in darkness than under continuous light. These results largely agree with prior work on
B. sorokiniana from wheat in Xinjiang [
22] and from maize leaf-spot isolates [
23], which report optimal growth in the mid-20s °C and neutral to slightly alkaline pH. Minor discrepancies among studies (e.g., some reports indicating optimal temperatures of 28 – 30 °C [
24,
25]) likely reflect intraspecific variability among isolates, host-associated adaptation or regional climatic differences; nevertheless, the general conclusion is that
B. sorokiniana isolates tolerate and grow well across a broad temperate range ( ≈ 25 – 30 °C). Regarding media preference, our isolate grew well on PSA, PDA and OMA, consistent with previous reports that different isolates can show modestly different preferences for complex media [
26]. These biological data provide a basis for predicting seasonal risk windows and for designing in vitro assays that approximate field conditions.
Laboratory fungicide assays showed that the tested fungicides—representing benzimidazoles, triazoles, strobilurins, organosulfur compounds, plant-derived phenolics and an antibiotic—exerted varying degrees of in vitro inhibition on YM1. Difenoconazole exhibited the strongest inhibitory effect (lowest EC
50), followed by tebuconazole and tetramycin, while thiophanate-methyl, carbendazim and ethylicin were least effective. These results align with prior fungicide-screening studies on
B. sorokiniana from wheat and other hosts [
23,
27]. Notably, tetramycin (a biologically derived antibiotic) demonstrated promising inhibitory activity, suggesting potential for integration into low-residue or “green” management strategies. However, in vitro sensitivity does not always predict field performance: systemic uptake, persistence in soil or plant tissue, application method, timing, and environmental conditions will influence control efficacy in situ. Therefore, promising candidates identified here (e.g., difenoconazole, tebuconazole, tetramycin) should be evaluated in controlled greenhouse and field trials to determine effective application rates, optimal timing, and non-target effects.
This study has several limitations that merit acknowledgement. First, surveys and sampling were geographically limited to several counties within the Ili region; broader geographic sampling would better define the current distribution and genetic diversity of B. sorokiniana on orchardgrass in Xinjiang and adjacent regions. Second, we relied on three loci (ITS, GAPDH, TEF) for molecular identification; although these loci are robust for species-level resolution in many taxa, population-level diversity, pathogenicity variation, and fungicide-resistance mechanisms would benefit from higher-resolution genotyping (e.g., microsatellites, multilocus sequence typing, or whole-genome sequencing). Third, environmental drivers of incidence (soil moisture, temperature microclimate, soil texture, and host nutritional status) were not measured concurrently with disease surveys; integrating those measurements into future epidemiological studies will test the hypotheses proposed here about habitat-driven differences in disease incidence. Finally, assessment of potential inoculum sources and transmission pathways (e.g., seedborne contamination, infested crop residues, or alternate hosts) is necessary to design effective prevention and containment strategies.
From an applied perspective, our findings indicate that B. sorokiniana represents a novel root pathogen of wild orchardgrass in this region and that several registered fungicides display in vitro activity. Immediate next steps should include (1) evaluating the efficacy and phytotoxicity of the most active fungicides under greenhouse and field conditions, (2) quantifying isolate diversity and potential fungicide-resistance alleles, (3) surveying adjacent cereal and pasture crops for cross-infection risk, and (4) testing cultural measures (e.g., irrigation management, residue removal, crop rotation or mixed-species sowing) that might reduce disease incidence in grassland settings.
5. Conclusions
This study documents, for the first time in Xinjiang, root-rot disease of wild orchardgrass caused by B. sorokiniana. The pathogen was confirmed by combined morphological and multilocus molecular analyses and by fulfilling Koch’s postulates. Biological assays indicate that the isolate YM1 grows optimally at ~ 25 °C and neutral pH, prefers complex carbon and nitrogen sources (soluble starch and peptone), and shows greater radial growth in darkness. In vitro fungicide screening identified difenoconazole and tebuconazole as the most effective chemical inhibitors and highlighted tetramycin as a promising biological control candidate. These results provide foundational information for subsequent epidemiological studies and for the development of integrated disease-management strategies. Future work should expand geographic and genetic surveys, verify fungicide efficacy under field conditions, and explore cultural and biological control measures to mitigate disease risk in orchardgrass populations and to reduce potential spillover to agricultural crops.
Author Contributions
Conceptualization, L.S. and L.L.W; methodology, S.L., Z.Y.Y. and S.S; software, Z.Y.Y; validation, L.S., Z.Y.Y. and L.L.W.; formal analysis, L.S.; investigation, L.S. and Z.Y.Y.; resources, Y.J.Z.; data curation, L.S. and Z.Y.Y; writing—original draft preparation, S.L. and Z.Y.Y; writing—review and editing, L.S. and B.Z.F; visualization, L.S.; supervision, M.J.; project administration, L.L.W.; funding acquisition, L.L.W. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by National Key Research and Development Program of China (2022YFD1401103); Xinjiang Agricultural University High-level Talent Research Cultivation Initiative (2524GCCRC); Xinjiang Autonomous Region Training Project for Key Talents in Agriculture, Rural Areas, and Farmers (2023SNGGGCC016).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in this study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author. The original gene sequences data presented in the study are openly available in NCBI with accession number provided in the article.
Acknowledgments
Yu-Yuan Li and Meng-Hao Dang are thanked for their help with sample collection.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Hirata, M.; Yuyama, N.; Cai, H. Isolation and characterization of simple sequence repeat markers for the tetraploid forage grass Dactylis glomerata: Simple sequence repeatmarkers for Dactylis. Plant Breeding 2011, 130, 503–506. [Google Scholar] [CrossRef]
- Lolicato, S.; Rumball, W.; Sutherland, G. Past and present improvement of cocksfoot (Dactylis glomerata L.) in Australia and New Zealand. New Zealand Journal of Agricultural Research 1994, 37, 379–390. [Google Scholar] [CrossRef]
- Probert, R.J. Germination studies in European populations of Dactylis glomerata L. Journal of Agronomy 2016, 195, e993–994. [Google Scholar]
- Pen, Y.; Zhang, X.Q. Progress in studies on genetic diversity of Dactylis glomerata L. Journal of Plant Genetic Resources 2003, 4, 179–183. [Google Scholar]
- Yan, H.D.; Zhang, X.Q.; Zeng, B.; Cheng, L.; Nie, G.; Ji, Y.; Huang, L.K. Resistance evaluation of Dactylis glomerata germplasm resources to rust and field investigation on summer survival. Acta Agrestia Sinica 2013, 21, 720–728. [Google Scholar]
- Cécile, L.; Karen, C.G.S.; Hugo, G.; Arnaud, H. Advances in understanding obligate biotrophy in rust fungi. New Phytologist 2019, 222, 1190–1206. [Google Scholar]
- Cagaš, B.; Macháč, R. Effect of some factors on the incidence of choke (Epichloë typhina) in grass seed stands in the Czech Republic. Plant Protection Science 2012, 48, 10–16. [Google Scholar] [CrossRef]
- Zaffarano, P.L.; McDonald, B.A.; Linde, C.C. Two new species of Rhynchosporium. Mycologia 2011, 103, 195–202. [Google Scholar] [CrossRef]
- Fulcher, M.R.; Winans, J.B.; Bergstrom, G.C. Fusarium graminearum isolates obtained from wheat and wild grasses in northeastern New York display comparable range of phenotypes, including virulence on crop hosts. Journal of Plant Pathology 2021, 103, 71–77. [Google Scholar] [CrossRef]
- Hartman, T.; Tharnish, B.; Harbour, J.; Yuen, G.Y.; Jackson-Ziems, T.A. Alternative Hosts in the Families Poaceae and Cyperaceae for Xanthomonas vasicola pv. vasculorum, causal agent of bacterial leaf streak of corn. Phytopathology 2020, 110, 1147–1152. [Google Scholar]
- Deng, H. Taxonomic studies on the genus Bipolaris Shoemaker in China; Shandong Agricultural University: Jinan, China, 2002. [Google Scholar]
- Al-Sadi, A.M. Bipolaris sorokiniana-Induced black point, common root rot, and spot blotch diseases of wheat: a review. Front. Cell. Infect. Microbiol. 2021, 11, 584899. [Google Scholar] [CrossRef] [PubMed]
- Kouadri, A.E.M.; Bekkar, A.A.; Zaim, S. First characterization of Bipolaris sorokiniana isolated from lentil (Lens culinaris) in Algeria. Journal of Plant Pathology 2025, 107, 4–14. [Google Scholar] [CrossRef]
- Neves, D.; Bruening, B.; Knott, C.; Lee, C.A.; Bradley, C.A. First report of spot blotch caused by Bipolaris sorokiniana on winter rye in Kentucky. Plant Disease 2021, 105, 4160. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: San Diego, USA, 1990; pp. 315–322. [Google Scholar]
- Guerber, J.C.; Liu, B.; Correll, J.C.; Johnston, P.R. Characterization of diversity in Colletotrichum acutatum sensu lato by sequence analysis of two gene introns, mtDNA and intron RFLPs, and mating compatibility. Mycologia 2003, 95, 872–895. [Google Scholar] [CrossRef]
- Wang, L.; Wang, N.; Yu, J.L.; Wu, J.; Liu, H.; Lin, K.J.; Zhang, Y.Y. Identification of pathogens causing alfalfa Fusarium root rot in Inner Mongolia, China. Agronomy. 2023, 13, 456–469. [Google Scholar] [CrossRef]
- Fang, X.L.; Zhang, C.X.; Nan, Z.B. Research advances in Fusarium root rot of alfalfa (Medicago sativa). Acta Prataculturae Sinica 2019, 28, 169–183. [Google Scholar]
- Aaihaiti, N.; Wei, R.; Cai, C.Y.; Zhang, Q.; Wang, H.Y.; Guo, Q.Y.; Wang, L.L. Identification, biological characteristics and fungicides of pathogen causing marigold brown spot disease in Xinjiang. Journal of Xinjiang Agricultural University 2023, 46, 42–49. [Google Scholar]
- Farr, D.F.; Bills, G.F.; Chamuris, G.P.; Rossman, A.Y. Fungi on Plants and Plant Products in the United States; The American Phytopathological Society Press: St. Paul, MN, USA, 1989; p. pp. 1252. [Google Scholar]
- Sharma, B.A.; Singh, A.; Singh, P.T. Effect of black point on seed germination parameters in popular wheat cultivars of Northern India. Indian Phytopathology 2021, 74, 1–5. [Google Scholar] [CrossRef]
- Yan, R.; Zhou, X.Y.; Zhang, J.G.; Wang, L.; Li, J.; Liang, L.; Gong, J.Y.; Du, Y.; Li, K.M.; Lei, B. Pathogen identification and biological characteristics analysis of wheat root rot caused by Bipolaris sorokiniana in Xinjiang. Xinjiang Agricultural Sciences 2024, 61, 1209–1217. [Google Scholar]
- Guo, N.; Hu, Q.Y.; Liu, Y.Y.; Shi, J.; Zhang, H.J.; Liu, S.S. Biological characteristics and fungicide sensitivity of Bipolaris sorokiniana causing maize leaf spot. Acta Agriculturae Boreali-Sinica 2019, 34, 289–295. [Google Scholar]
- Kouadri, M.E.A.; Bekkar, A.A.; Zaim, S. First report of Bipolaris sorokiniana causing spot blotch of lentil in Algeria. New Dis Rep. 2021, 43, e12009. [Google Scholar] [CrossRef]
- Naresh, P.; Biswas, K.S.; Kumar, U.; Rajik, M. Effect of media, pH, temperature, host range and fungicides on Bipolaris sorokiniana. Annals of Plant Protection Sciences 2009, 17, 394–397. [Google Scholar]
- Sinijadas, K.; Dhar, A.; Bhaumik, P.; Chowdhury, A.K. Evaluation of culture medium for the growth of Bipolaris sorokiniana the causal agent of spot blotch of wheat. Int. J. Curr. Microbiol. App. Sci. 2018, 7, 579–583. [Google Scholar] [CrossRef]
- Sun, H.; Wang, R.R.; Shi, C.C.; Zhang, L.R; Liu, D.Q.; Yang, W.X. Fungicide screening and variety resistance identification of wheat root rot. Northeast Agricultural Sciences 2023, 48, 58–61+82. [Google Scholar]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).