1. Introduction
Small-scale artisanal fisheries constitute the livelihood of millions of people in coastal communities of developing countries (FAO, 2020). However, these fisheries typically operate under data-limited conditions ("data-poor fisheries"), characterized by the absence of historical catch and effort time series, the lack of species-specific stock assessments, and the scarcity of basic biological information (Cochrane et al., 2011; Hilborn & Ovando, 2014). This limitation represents one of the most significant challenges for the assessment and sustainable management of fishery resources globally, particularly in high-priority conservation regions (Newman et al., 2015).
Coastal sharks of the family Carcharhinidae are fundamental components of tropical and subtropical marine ecosystems, where they act as mid-to-high trophic level predators and contribute to the regulation of food webs (Cortés, 1999; Heithaus et al., 2008). Within this group, the Atlantic sharpnose shark Rhizoprionodon terraenovae (Richardson, 1837) is a small-sized shark (<150 cm) traditionally considered of "high biological productivity" due to its relatively fast growth rates, early maturity (3-4 years), and high reproductive potential (Cortés, 2000; Márquez-Farías & Castillo-Géniz, 1998). However, this perception of resilience has been challenged by evidence of overexploitation in various regions of the Gulf of Mexico (Aguirre-García et al., 2004; Worm et al., 2013), generating a debate on the actual vulnerability of species with "fast" life histories facing intense, unregulated fishing pressures (Walker, 2005; Dulvy et al., 2014).
The southern coast of Tamaulipas hosts two Marine Priority Regions for conservation in Mexico: Region 45 "La Pesca-Rancho Nuevo" and Region 46 "Laguna de San Andrés", designated in the Gulf of Mexico and Caribbean Sea Marine and Regional Ecological Zoning Program (DOF, 2012). These regions belong to the Conservation Sector, whose objective is "the promotion of protection, restoration, and conservation of ecosystems, natural resources, environmental goods and services, aimed at fostering their use while guaranteeing sustainable development" (DOF, 2012). Additionally, the Playa Tortuguera Rancho Nuevo site has been designated as a Wetland of International Importance (Ramsar Site No. 1326) due to its critical role in the nesting of the Kemp's ridley sea turtle (Lepidochelys kempii), an endemic species of the Gulf of Mexico and critically endangered (CONANP, 2004; IUCN, 2023).
In this high-priority conservation area, intense artisanal fishing activity targets multiple species, including R. terraenovae, which is caught both in directed longline fisheries and as bycatch in scale fish fisheries (DOF, 2012; CONANP, 2014). A crucial aspect, scarcely addressed in the literature, is the role of R. terraenovae as a natural predator of sea turtle hatchlings. The Management Program of the Playa Rancho Nuevo Sanctuary (CONANP, 2014) explicitly recognizes that species such as the sharpnose shark exert predation pressure on hatchlings during the hatching season (August-November), when they emerge from nests and head to the sea facing high mortality (Witherington et al., 2012; Pilcher et al., 2021).
Assessing the population status of R. terraenovae in this context becomes relevant not only for fisheries management but also for the conservation of L. kempii. However, controversies persist regarding the true status of the species' populations in the Gulf of Mexico, with exploitation rate estimates ranging from 0.14 in Campeche (Bada-Sánchez et al., 2016) to 0.77 in the same region (Aguirre-García et al., 2004), suggesting a complex spatio-temporal dynamic that could be due to methodological differences, regional variations in fishing pressure, or the existence of multiple fishery stocks (Heist et al., 1996; Loefer & Sedberry, 2003).
The hypotheses guiding this study were: (H₁) the population of R. terraenovae in Marine Priority Regions 45-46 is overexploited as a result of artisanal fishing pressure; (H₂) the overexploitation of this predator has potential implications for the conservation of L. kempii and for the fulfillment of the Conservation Sector objectives; and (H₃) despite the limitations inherent to a data-limited fishery, it is possible to obtain robust estimates of population parameters using length-frequency based methods.
The main objective of this study was to estimate growth and mortality parameters, and to determine the exploitation rate of R. terraenovae in Marine Priority Regions 45-46 and the influence area of the Playa Tortuguera Rancho Nuevo Wetland of International Importance, to assess its population status and analyze the implications of its exploitation for sea turtle conservation and fisheries management in a data-limited context. Our results reveal severe overexploitation (E = 0.78), evidencing the urgent need to implement ecosystem-based management in this priority region.
2. Materials and Methods
2.1. Study Area
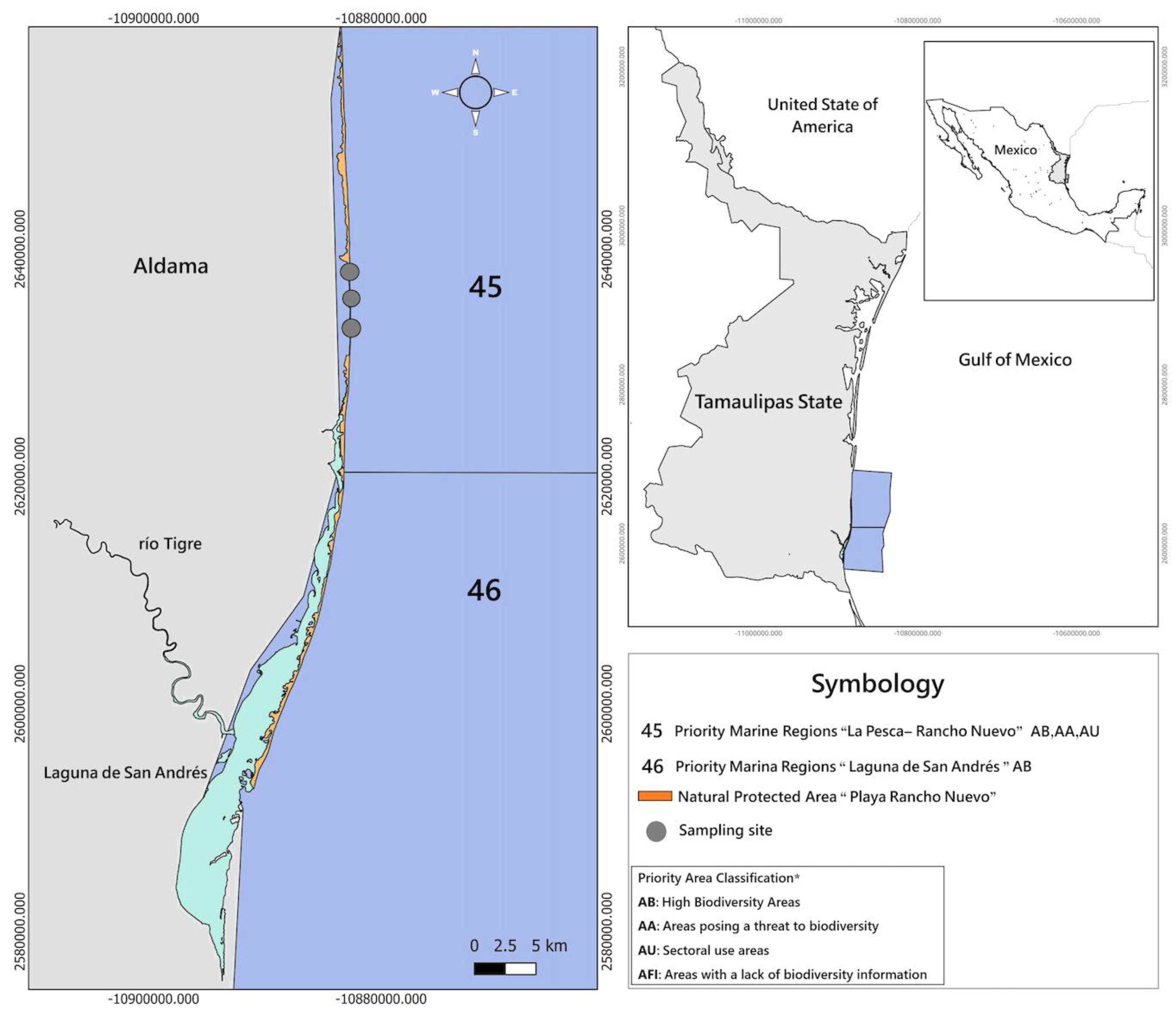
The study was conducted in the locality of La Barra del Tordo, municipality of Aldama, Tamaulipas, Mexico (23°03' N, 97°46' W), located within the area comprising two Marine Priority Regions for conservation in Mexico: Region 45 "La Pesca-Rancho Nuevo" and Region 46 "Laguna de San Andrés" (Arriaga et al., 1998), and within the influence zone of the Playa Tortuguera Rancho Nuevo Wetland of International Importance (Ramsar Site No. 1326) (CONANP, 2004) (
Figure 1).
Marine Priority Region 45 "La Pesca-Rancho Nuevo" is located between 23°30' and 22°54' N latitude and 97°48'36'' and 97°18'36'' W longitude, with an area of 2,955 km² (Arriaga et al., 1998). This region is characterized by a warm semi-arid climate with summer rains, mean annual temperature of 22-26°C, and occurrence of tropical storms, hurricanes, and northerly winds. Geologically, it presents sedimentary rocks, gentle slope continental shelf, and wide platform. It includes coastal lagoons, beaches, coastal dunes, marshes, and estuaries. Biodiversity includes mollusks, polychaetes, echinoderms, crustaceans, fish, turtles, birds, marine mammals, and mangroves, with turtle endemism (L. kempii) and constitutes a turtle nesting and reproduction zone (Arriaga et al., 1998).
Marine Priority Region 46 "Laguna de San Andrés" is located between 22°54' and 22°25'48'' N latitude and 97°56'24'' and 97°23'24'' W longitude, with an area of 2,384 km² (Arriaga et al., 1998). It presents a subhumid temperate climate, mean annual precipitation of 635-791 mm, evaporation of 1,475-1,876 mm annually, mean annual temperature of 17-24°C, with hurricanes in summer and northerly winds in winter. Geologically, it presents marginal sea coasts with sedimentary rocks and floodplain depression. It includes lagoons, swamps, grasslands, and beaches, with medium eutrophication. Biodiversity includes mollusks, crustaceans, fish, turtles, birds, seagrasses (Ruppia maritima), and mangroves (white mangrove Avicennia tinctoria and red mangrove Rhizophora mangle), with high ecological integrity in seagrass beds, dunes, and beaches. It constitutes a growth and feeding zone for crustaceans and fish, and a growth zone for mollusks (Arriaga et al., 1998).
The Playa Tortuguera Rancho Nuevo site was designated as a Wetland of International Importance (Ramsar Site No. 1326) on February 2, 2004, with an area of 30 hectares in its main polygon and an influence zone encompassing marine areas up to 30 nautical miles (CONANP, 2004). This designation recognizes the critical importance of this site for the nesting of the Kemp's ridley sea turtle (L. kempii), an endemic species of the Gulf of Mexico and critically endangered.
The Gulf of Mexico and Caribbean Sea Marine and Regional Ecological Zoning Program (DOF, 2012) classifies these regions within the Conservation Sector, whose objective includes "the promotion of protection, restoration, and conservation of ecosystems, natural resources, environmental goods and services, aimed at fostering their use while guaranteeing sustainable development." This instrument recognizes the need to reconcile productive activities, such as artisanal fisheries, with conservation objectives.
Sea surface temperature in the region has a mean annual average of 24°C, with summer maxima (29-30°C) and winter minima (18-20°C) (INAPESCA, 2013). The Kemp's ridley sea turtle nesting season extends from April to August, with the hatching and hatchling emergence season occurring between August and November (CONANP, 2014).
Artisanal and coastal fisheries constitute the main economic activity of La Barra del Tordo and other surrounding communities such as Tepehuajes and Carbonera. Small fiberglass vessels ("pangas") with outboard motors operate from the shoreline up to 30 nautical miles, employing various fishing gears: gillnets (anchored), bottom longlines (1000-1200 hooks), and handlines for shark fishing (SEMARNAT, 2014; CONANP, 2014). The fleet operates year-round, with higher landings in spring-summer. The main target species are spotted seatrout (Cynoscion nebulosus), red snapper (Lutjanus campechanus), robalo, corvina, and various small shark species, including R. terraenovae, locally known as "cazón" (DOF, 2012; CONANP, 2014). According to the characterization of the Marine Priority Regions, "artisanal fisheries (oyster, crab, scale fish)" of low impact occur in this area, although without previous quantitative assessments (Arriaga et al., 1998).
2.2. Biological Sampling
Monthly samplings were conducted during the period from November 2018 to February 2020. Organisms were obtained directly from artisanal fishery landings in La Barra del Tordo, with the collaboration of local fishermen. The following data were recorded for each specimen: (1) total length (TL) with 1 mm precision, measured from the tip of the snout to the end of the caudal fin in natural position; (2) eviscerated weight (EW) with 1 g precision using a Royal Universal digital scale (10 kg capacity); and (3) sex determined by the presence (male) or absence (female) of claspers.
2.3. Data Analysis
Given that this fishery is classified as data-limited, without historical catch and effort time series or previous assessments, analysis methods based exclusively on length frequencies were selected, which have been widely validated for such contexts (Sparre & Venema, 1998; Hordyk et al., 2015).
2.3.1. Sex Ratio and Size Structure
The observed sex ratio (females:males) was compared with the expected 1:1 ratio using a Chi-square test (X²) with a significance level of α = 0.05 (Zar, 2010). A Student's t-test for independent samples was applied to evaluate significant differences in mean total lengths between sexes (Zar, 2010).
2.3.2. Total Length-Eviscerated Weight Relationship
The relationship between total length (L, cm) and eviscerated weight (P, g) was modeled using the power function P = a·Lᵇ (Ricker, 1975), where "a" is the condition factor and "b" is the allometry coefficient. Parameters were estimated by linear regression of logarithmically transformed data: log(P) = log(a) + b·log(L). Growth type was evaluated using a Student's t-test to determine whether the b value differed significantly from 3 (isometric growth), with H₀: b = 3 and Hₐ: b ≠ 3 (Walpole et al., 2012). Curves for females and males were compared using the coincident curves analysis proposed by Chen et al. (1992).
2.3.3. Growth Parameters
Length growth was modeled using the von Bertalanffy equation (1938): Lₜ = L∞[1 - exp{-k(t - t₀)}]. The parameters L∞ and k were estimated using three length-frequency based methods included in the FISAT II software package (Gayanilo et al., 2005): (1) ELEFAN I routine (Electronic Length Frequency Analysis); (2) Shepherd's method; and (3) Powell-Wetherall method. These methods are particularly appropriate for data-limited fisheries, as they require only length information and do not depend on chronological age data (Pauly, 1984).
The parameter t₀ was estimated using Pauly's empirical equation (1984). To assess uncertainty in L∞ and k estimates, a non-parametric bootstrap procedure (Efron & Tibshirani, 1993) was applied with 3000 replicates, using the resampling module of SPSS software (PASW Statistics 18, IBM Corp., Armonk, NY, USA). Medians of bootstrap distributions are reported as point estimates, and the 2.5 and 97.5 percentiles as 95% confidence intervals.
The quality of growth estimates was evaluated using the Phi prime growth performance index (Ø' = log₁₀k + 2·log₁₀L∞) proposed by Pauly & Munro (1984). This index allows comparison of estimates from this study with those reported in the literature, under the assumption that phylogenetically close species exhibit similar Ø' values (Pauly, 1991).
2.3.4. Total Mortality (Z)
Total mortality (Z, yr⁻¹) was estimated using two methods appropriate for data-limited contexts: (1) linearized catch curve (Sparre & Venema, 1998); and (2) the Jones and van Zalinge method (Jones & van Zalinge, 1981), which is an extension of the catch curve method that uses grouped length-frequency data and does not require complete age conversion.
Since Z estimates are sensitive to the k value used in length-age conversion (Pauly, 1984), two k values were employed: that obtained by ELEFAN I (k = 0.59) and that obtained by Shepherd's method (k = 0.78). This generated four Z estimates: Z₁ (catch curve with kELEFAN), Z₂ (catch curve with kShepherd), Z₃ (Jones-van Zalinge with kELEFAN), and Z₄ (Jones-van Zalinge with kShepherd).
2.3.5. Natural Mortality (M)
Natural mortality (M, yr⁻¹) was estimated using Pauly's empirical equation (1980) for fish, which relates M to growth parameters and environmental temperature: log₁₀M = -0.0066 - 0.279·log₁₀L∞ + 0.6543·log₁₀k + 0.4634·log₁₀T, where T is the mean annual water temperature (24°C for the study area; INAPESCA, 2013). Two M estimates were obtained using the two k values: M₁ (with kELEFAN) and M₂ (with kShepherd).
2.3.6. Fishing Mortality (F) and Exploitation Rate (E)
Fishing mortality (F, yr⁻¹) was calculated by subtraction: F = Z - M (Ricker, 1975). Using the four Z estimates and the two M estimates, eight F values were generated. The exploitation rate (E) was calculated as E = F/Z (Ricker, 1975), obtaining 32 estimates. Following Gulland's criterion (1971), E = 0.5 was considered as the target reference point (maximum sustainable yield), where F = M.
2.3.7. Uncertainty Analysis and Selection of Robust Estimates
To identify E estimates with lower uncertainty, the confidence interval length (CIL = upper limit - lower limit) was calculated for each combination. Those with the smallest CIL were selected as the most robust estimates. Two-way ANOVA was applied to evaluate the effect of the factors "Z estimation method" and "F estimation method" on E, and one-way ANOVA to compare estimates with lower variability (α = 0.05).
2.3.8. Declaration on the Use of Artificial Intelligence
During the preparation of this manuscript, no generative artificial intelligence tools were used for the generation of text, data, graphics, or for study design, data collection, analysis, or interpretation of results. Statistical and length-frequency analysis software (SPSS, FISAT II) were used in a conventional manner.
3. Results
3.1. Sample Composition and Population Structure
A total of 541 R. terraenovae organisms caught by artisanal fisheries in Marine Priority Regions 45-46 and the influence area of the Playa Tortuguera Rancho Nuevo Wetland of International Importance, Tamaulipas, Mexico, were analyzed during the period November 2018 - February 2020. The sample consisted of 260 females (48.1%) and 281 males (51.9%). The female:male sex ratio (0.93:1) did not differ significantly from the expected 1:1 ratio (X² = 0.642, df = 1, p = 0.423), indicating sexual balance in the exploited population.
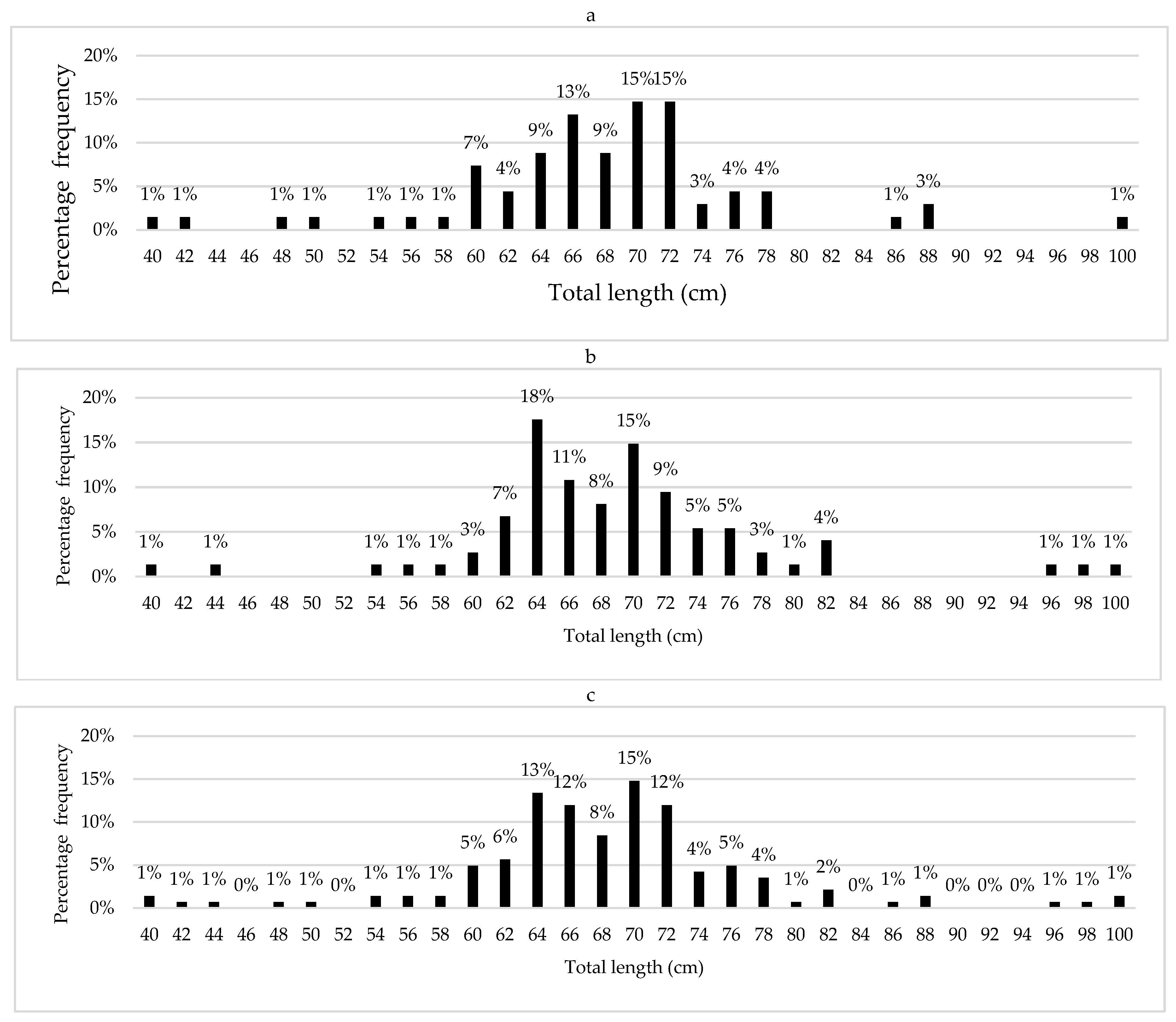
The total length frequency distribution showed a similar unimodal pattern for both sexes (
Figure 2). Females exhibited a length range of 40 to 100 cm, with a mean of 66.99 ± 12.3 cm SD and a mode in the 66-68 cm class (
Figure 2a). Males showed a slightly smaller range (40-97 cm), with a mean of 66.98 ± 11.9 cm SD and the highest concentration of individuals in the 64-70 cm classes (
Figure 2b). For combined sexes (n=541), the range was 40 to 100 cm with a mean of 66.99 ± 12.1 cm SD, showing a pronounced mode in the 64-68 cm interval, where approximately 25% of individuals were concentrated, with a gradual decrease towards larger (>85 cm) and smaller (<50 cm) sizes (
Figure 2c).
No significant differences were found in mean total length between sexes (t = 0.005, df = 539, p = 0.996). Sizes smaller than 50 cm and larger than 85 cm were scarcely represented (<5% of the total), suggesting a low proportion of hatchlings and long-lived organisms in the catches, with a higher percentage of individuals in juvenile and young adult stages.
3.2. Length-Weight Relationship
Length-weight relationship parameters are presented in
Table 1. The b values were close to 3 for both sexes and combined sexes, and the t-test did not reject the isometry hypothesis (p > 0.05). The coincident curves analysis (Chen et al., 1992) detected no significant differences between sexes (F = -12.39, df = 2,537, p < 0.05), justifying the combined analysis.
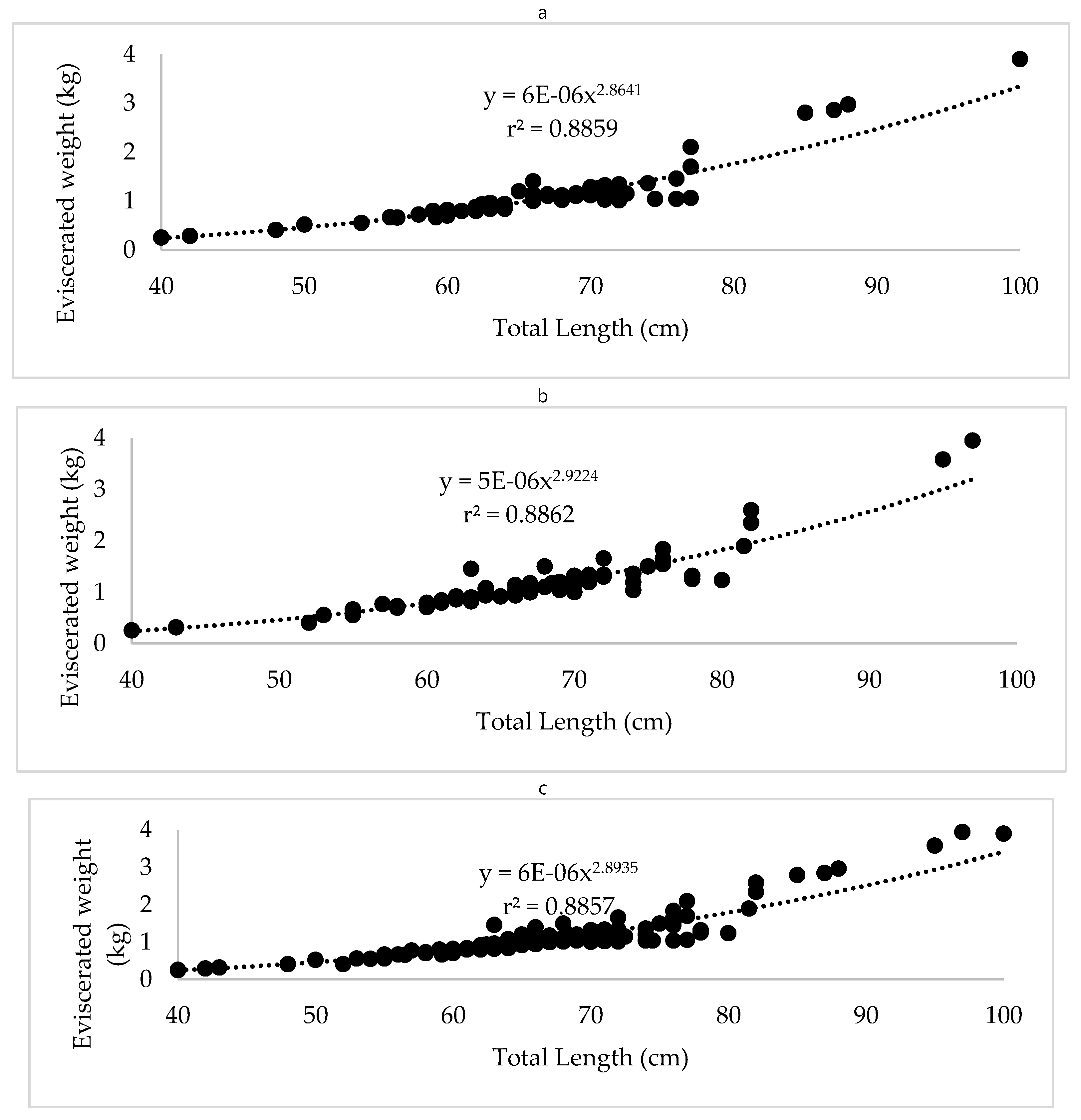
The total length-eviscerated weight relationship for
R. terraenovae showed a solid fit to the power model in all analyzed groups (r² ≥ 0.905;
Figure 3). Regression parameters and associated statistical tests are detailed in
Table 1. The allometry coefficient (b) values were close to 3 for females (b = 2.86), males (b = 2.92), and combined sexes (b = 2.89). Student’s t-test showed no significant differences from theoretical isometry (p > 0.05 in all cases), confirming isometric growth type for the species in the study area. Likewise, the coincident curves analysis detected no significant differences between sexes (F = -12.39, df = 2,537, p < 0.05), which justifies the use of combined analysis to describe the population biometry in Marine Priority Regions 45 and 46 of Tamaulipas.
3.3. Growth Parameters
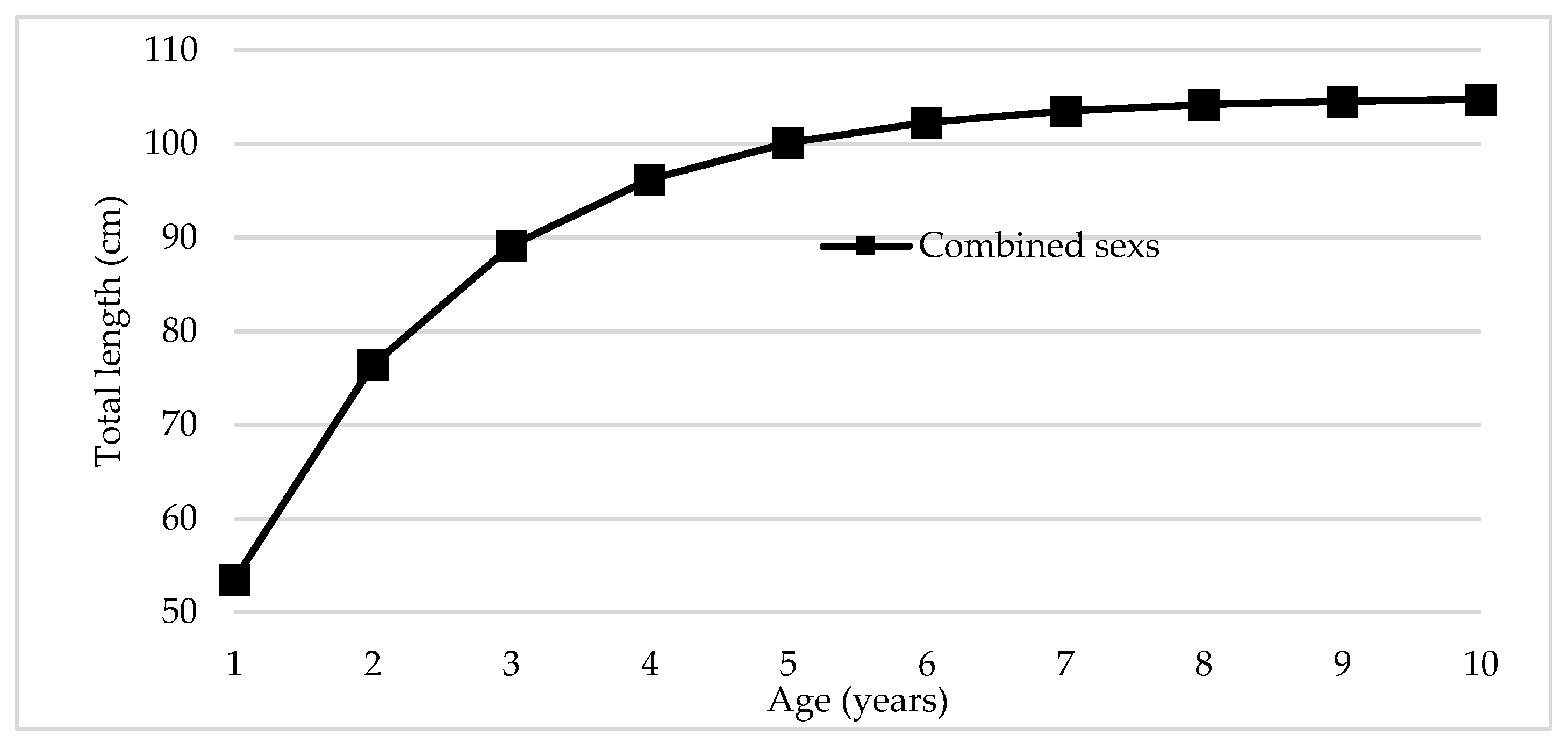
The von Bertalanffy growth curve in length for combined sexes of R.
terraenovae showed a pattern of rapid growth during the first years of life, with subsequent gradual deceleration when approaching the asymptotic length (
Figure 4). The observed points, corresponding to lengths converted to age (n=541), were adequately distributed around the fitted curve, indicating a good fit of the model to the data.
During the first 3-4 years, individuals reached approximately 60-70% of their asymptotic length (L∞ = 105 cm), evidencing accelerated growth characteristic of species with a "fast" life history strategy. At 5 years, the mean length was approximately 85 cm, and at 8 years it exceeded 95 cm, with a marked reduction in annual growth rate observed after 6-7 years. The asymptote of 105 cm represents the theoretical maximum length that individuals can reach in the region, although the low representation of organisms >90 cm in the catches (
Figure 2) suggests that few individuals fully express their growth potential due to fishing pressure.
Growth parameter estimates are presented in
Table 2. ANOVA detected no significant differences between sexes for L∞ (F = 0.144, p = 0.714) nor for k (F = 0.319, p = 0.585); therefore, combined sex parameters were used for mortality analyses. The growth performance index (Ø' = 3.81) falls within the range reported for the species (3.34-3.86;
Table 5).
3.4. Mortality
Mortality estimates for combined sexes are presented in
Table 3. The Z values obtained by Jones-van Zalinge (Z₃ = Z₄ = 4.13 yr⁻¹) were higher than those from catch curve and presented lower uncertainty (CIL = 1.05). Mean natural mortality was M = 0.467 yr⁻¹ (M₁ = 0.461, M₂ = 0.473). Mean fishing mortality with lower uncertainty was F = 3.22 yr⁻¹ (95% CI: 2.69-3.74).
3.5. Exploitation Rate
The 32 E estimates ranged from 0.42 to 1.21. Two-way ANOVA revealed significant effects of the Z method (F = 6.56, p = 0.002) and F method (F = 4.52, p = 0.001) (
Table 4).
The estimates with lowest uncertainty corresponded to combinations with Z₃-Z₄ and F₅-F₈ (
Table 5), with E values ranging from 0.76 to 0.80 and CIL between 0.05-0.07. One-way ANOVA detected no significant differences among these four estimates (F = 2.57, p = 0.089), yielding a mean value: E = 0.78 (95% CI: 0.745 - 0.805).
This value significantly exceeds the reference point of E = 0.5 (Gulland, 1971), indicating that the population of R. terraenovae in Marine Priority Regions 45 and 46 and in the influence area of the Playa Tortuguera Rancho Nuevo Wetland of International Importance is in a state of severe overexploitation.
Table 5.
Selected exploitation rate (E) estimates with lowest uncertainty.
Table 5.
Selected exploitation rate (E) estimates with lowest uncertainty.
| Combination |
Z |
F |
E |
95% CI E |
CIL |
| E₍₅,₃₎ |
Z₃ (4.13) |
F₅ (3.30) |
0.80 |
0.77-0.82 |
0.05 |
| E₍₆,₃₎ |
Z₃ (4.13) |
F₆ (3.13) |
0.76 |
0.72-0.79 |
0.07 |
| E₍₇,₄₎ |
Z₄ (4.13) |
F₇ (3.30) |
0.80 |
0.77-0.82 |
0.05 |
| E₍₈,₄₎ |
Z₄ (4.13) |
F₈ (3.13) |
0.76 |
0.72-0.79 |
0.07 |
| Mean |
- |
- |
0.78 |
0.745-0.805 |
0.06 |
4. Discussion
The main objective of this study was to estimate growth and mortality parameters, and to determine the exploitation rate of R. terraenovae in the Aldama region, within Marine Priority Regions 45 "La Pesca-Rancho Nuevo" and 46 "Laguna de San Andrés" and in the influence area of the Playa Tortuguera Rancho Nuevo Wetland of International Importance (Ramsar Site No. 1326), in the State of Tamaulipas, Mexico; to assess its population status and analyze the implications of its exploitation for sea turtle conservation and for fulfillment of the objectives of the Conservation Sector of the Gulf of Mexico and Caribbean Sea Marine and Regional Ecological Zoning Program (DOF, 2012). This work constitutes the first quantitative assessment for the species on the Tamaulipas coast and, more broadly, represents a case study on how to approach artisanal fishery assessment in data-limited contexts operating in high-priority conservation areas.
The main finding of this study is the documentation of a state of severe overexploitation (E = 0.78) of R. terraenovae in this high-priority conservation region. This result acquires particular relevance not only for its significance in terms of fisheries management, but also for its potential ecological implications in predator-prey dynamics with sea turtle hatchlings, an interaction explicitly recognized in the Management Program of the Playa Rancho Nuevo Sanctuary (CONANP, 2014) and in the characterization of Marine Priority Region 45, which identifies "pressure on turtle species" as a problem (Arriaga et al., 1998). These findings are discussed below in the context of previous studies, implications for turtle conservation and fulfillment of Conservation Sector objectives, ecosystem-based management, and lessons learned for data-limited fishery assessment in priority conservation areas.
4.1. Overexploitation of Rhizoprionodon terraenovae in Marine Priority Regions 45 and 46
The estimated exploitation rate (E = 0.78) is comparable to the value of 0.77 reported by Aguirre-García et al. (2004) for Campeche, but substantially higher than the values of 0.37-0.39 reported by Worm et al. (2013) and Zea-de-la-Cruz (2012) for the southern Gulf of Mexico and Veracruz, respectively (
Table 6). The estimated fishing mortality (F = 3.22 yr⁻¹) is the highest documented for the species in the Gulf of Mexico, reflecting intense fishing pressure in the region. These regional differences could be explained by variations in fishing intensity, selectivity of the fishing gears used, and local oceanographic characteristics affecting population productivity (Loefer & Sedberry, 2003).
Particularly notable is the difference with the study by Bada-Sánchez et al. (2016), who reported an exploitation rate of only 0.14 for the Campeche Bank, suggesting an underexploited population. This discrepancy could be due to methodological differences: Bada-Sánchez et al. (2016) used the Hoenig model (1983) modified by Then et al. (2014) to estimate M, while we employed Pauly's method (1980). Additionally, the natural mortality estimated in this study (M = 0.46 yr⁻¹) is slightly lower than that reported by Bada-Sánchez et al. (2016) (M = 0.50 yr⁻¹), but similar to values documented by other authors for the species in the Gulf of Mexico (Márquez-Farías & Castillo-Géniz, 1998: M = 0.39 yr⁻¹; Aguirre-García et al., 2004: M = 0.39 yr⁻¹; Zea-de-la-Cruz, 2012: M = 0.27-0.70 yr⁻¹). The main difference lies in fishing mortality (F), which in our study (F = 3.22 yr⁻¹) is considerably higher than that reported by Bada-Sánchez et al. (2016) (F = 0.19 yr⁻¹), reflecting substantially greater fishing pressure in Marine Priority Regions 45-46 of Tamaulipas compared to the Campeche Bank.
It is possible that real differences exist in population status between regions, which would support the hypothesis of multiple fishery stocks in the Gulf of Mexico (Heist et al., 1996). While the Campeche Bank could harbor a population with lower fishing pressure, our results indicate that the R. terraenovae population in Marine Priority Regions 45-46 is overexploited (E = 0.78) as a result of intense artisanal fishing pressure in this high-priority conservation region, confirming hypothesis H₁.
4.2. Implications for Sea Turtle Conservation and Fulfillment of Conservation Sector Objectives
The overexploitation of R. terraenovae in Marine Priority Regions 45 and 46 acquires a particularly relevant dimension due to the ecological role of this species as a predator of sea turtle hatchlings, and due to the conservation status of these areas. The Gulf of Mexico and Caribbean Sea Marine and Regional Ecological Zoning Program (DOF, 2012) classifies these regions within the Conservation Sector, whose objective is "the promotion of protection, restoration, and conservation of ecosystems, natural resources, environmental goods and services, aimed at fostering their use while guaranteeing sustainable development." Additionally, the Ramsar designation of the Playa Tortuguera Rancho Nuevo site (CONANP, 2004) implies international commitments for the conservation of wetlands and their associated biodiversity.
The characterization of Marine Priority Region 45 (Arriaga et al., 1998) explicitly identifies sea turtles as part of its biodiversity, highlighting the endemism of Lepidochelys kempii and the importance of the area as a nesting and reproduction zone. Likewise, it identifies "pressure on turtle species" as part of the environmental problems and the need for "expansion of the conservation area to 120 km, between La Pesca and Barra de Chavarría, to cover the entire nesting zone." Our results, by documenting the overexploitation of a turtle hatchling predator, provide relevant information for managing this problem.
From a sea turtle conservation perspective, the reduction of a natural predator could initially be interpreted as a benefit, by reducing hatchling mortality in their first moments of life, which is critically high (Witherington et al., 2012; Pilcher et al., 2021). Sea turtle hatchling survival rates during the first hours at sea are extremely low, with estimates of less than 1% survival to adulthood (Bjorndal et al., 2011). Therefore, the reduction of predation pressure could, theoretically, increase recruitment and benefit the recovery of L. kempii, thus contributing to the objectives of the Conservation Sector.
However, this simplistic interpretation ignores the complex trophic interactions in which top predators participate (Heithaus et al., 2008). Sharks, as mid-to-high trophic level predators, fulfill fundamental ecological functions in coastal ecosystems, including regulation of mesopredator populations and maintenance of food web structure (Cortés, 1999; Estes et al., 2011). The elimination of native predators can trigger trophic cascade effects, with unpredictable consequences for the ecosystem (Myers et al., 2007; Ferretti et al., 2010). These effects could compromise other conservation objectives of the Conservation Sector, such as protection of biodiversity in general and maintenance of ecosystem services.
In the specific case of Marine Priority Regions 45 and 46, the overexploitation of R. terraenovae could have at least three potential ecological effects: (1) direct effect on turtle hatchlings (reduction of predation mortality); (2) effect on mesopredators (release of medium-sized fish populations that are prey of R. terraenovae); and (3) effect on ecosystem structure (alteration of trophic interactions in the coastal lagoons, beaches, dunes, marshes, and estuaries that characterize these regions). These possible ecological consequences, not yet empirically evaluated in the study area, represent a source of uncertainty for the management of these priority areas and confirm the relevance of our hypothesis H₂.
4.3. Biological Consistency of Estimates
Despite the high fishing mortality rates, our growth and natural mortality parameters show biological consistency with those reported for the species in other regions. The growth performance index (Ø' = 3.81) falls within the range of 3.34-3.86 documented for R. terraenovae in the Gulf of Mexico (Pauly & Munro, 1984; Loefer & Sedberry, 2003; SEDAR, 2007). The M/k ratio = 0.79 (using M = 0.467 yr⁻¹ and k = 0.59 yr⁻¹ from ELEFAN) approximates the range of 0.5-1.0 typically observed in exploited elasmobranch populations (Prince et al., 2014; Cortés, 2016), and is consistent with values reported for the species in other Gulf of Mexico localities (Márquez-Farías & Castillo-Géniz, 1998: M/k = 0.65; Aguirre-García et al., 2004: M/k = 0.46-0.57). The estimated asymptotic length (L∞ = 105 cm) is similar to that reported by Zea-de-la-Cruz (2012) for Veracruz (104.3 cm) and by Branstetter (1987) for Galveston Bay (108 cm). This consistency suggests that, although our fishing mortality values (F = 3.22 yr⁻¹) are the highest documented for the species, the growth and natural mortality parameters are biologically plausible and reflect exceptionally intense fishing pressure in Marine Priority Regions 45-46 of Tamaulipas.
4.4. Implications for Ecosystem-Based Management in the Conservation Sector
Our results have direct implications for the management of Marine Priority Regions 45 and 46 and the Playa Tortuguera Rancho Nuevo Wetland of International Importance, in line with the objectives of the Conservation Sector of the Marine Ecological Zoning Program (DOF, 2012) and with the strategies established in the Sanctuary Management Program (CONANP, 2014). Specifically, the latter establishes as a priority action: "Promote with the competent authority the best commercial, coastal, and artisanal fishing practices for those fisheries that have any negative impact on sea turtle populations" (CONANP, 2014, p. 125). The R. terraenovae fishery, due to its trophic interaction with hatchlings, qualifies as a fishery with potential impact on turtles, although the direction and magnitude of this impact requires further research.
The Ecosystem-Based Fisheries Management (EBFM) approach explicitly recognizes the need to consider interactions between target and non-target species, as well as the trophic effects of fishing (Pikitch et al., 2004; Link, 2010). Within the framework of the Conservation Sector, which seeks to reconcile ecosystem protection with sustainable resource use, we propose the following integrated management measures for the area:
Establishment of a minimum legal size (70 cm TL) based on the species' size at first sexual maturity (Márquez-Farías & Castillo-Géniz, 1998; Oviedo-Pérez et al., 2014).
Temporary closure during the reproductive period of R. terraenovae (spring-summer), which partially coincides with the turtle nesting season.
Fishing zoning establishing temporary or permanent exclusion areas for gillnet fishing during the turtle hatching season (August-November), particularly in zones near nesting beaches. This measure would address the recommendation to expand the conservation area to cover the entire nesting zone (Arriaga et al., 1998).
Participatory monitoring with local fishermen to obtain catch time series and evaluate the effect of management measures, within the framework of recommendations for Marine Priority Region 46, which suggests evaluating pressure on crab, shrimp, and fish species (Arriaga et al., 1998).
Interdisciplinary research to evaluate the real impact of R. terraenovae predation on turtle hatchlings, through stomach content analysis, telemetry studies, and trophic network models.
Implementation of best fishing practices as established by the Sanctuary Management Program, including training fishermen in techniques for releasing incidentally caught turtles and adopting selective fishing gears.
Strengthening surveillance at Rancho Nuevo for turtle protection, addressing the identified problem of "lack of surveillance at Rancho Nuevo for turtle (Kemp's ridley) protection" (Arriaga et al., 1998).
These measures should be implemented through a participatory process involving fishermen, fisheries authorities (CONAPESCA), and environmental authorities (CONANP), within the framework of the Protected Area advisory councils and environmental policy instruments such as the Marine Ecological Zoning Program.
4.5. Lessons for Data-Limited Fishery Assessment in Priority Conservation Areas
This study constitutes a case of successful application of length-frequency based methods for assessing an artisanal data-limited fishery in a context of high conservation relevance (Marine Priority Regions and Wetland of International Importance). Several lessons can be drawn from this experience:
Methodological uncertainty can and should be explicitly quantified. By generating multiple estimates using different methods and k values, and by applying bootstrap to estimate confidence intervals, we were able to identify the most robust combinations (lowest CIL) and obtain a point estimate with its corresponding uncertainty. This approach is recommended for all data-limited fishery assessments (Hilborn & Walters, 1992; Punt et al., 2016).
Consistency with comparative indices (Ø') and theoretical relationships (M/k) provides indirect validation. Despite the absence of independent age validation, the consistency of our growth parameters with those reported in the literature and with Beverton-Holt invariants suggests that the estimates are biologically plausible.
Participatory monitoring with fishermen is viable and generates quality data. Collaboration with local fishermen allowed obtaining a sample of 541 organisms over 16 months, demonstrating that it is possible to generate valuable information even without large investments in monitoring infrastructure.
Length-based methods are appropriate for artisanal fisheries in priority conservation areas. Since these methods do not require sacrificing organisms or sophisticated equipment, they are particularly suitable for protected natural areas and priority regions where minimizing research impact is sought.
Integration of fisheries management and conservation objectives is possible even with limited information. Our study demonstrates that, even in data-limited contexts, it is possible to generate relevant information for decision-making that integrates both fisheries sustainability and priority species conservation, as well as fulfillment of Conservation Sector objectives (confirming H₃).
4.6. Study Limitations and Future Perspectives
We acknowledge the limitations inherent to the length-frequency based methods used in this study. These methods assume that the population is in a steady state, that recruitment is constant, and that the sample is representative of the population (Sparre & Venema, 1998). Although we sought to minimize biases through systematic sampling over 16 months, we cannot rule out partial violations of these assumptions. The absence of independent age validation (through vertebral reading) constitutes another limitation, as growth estimates could be influenced by data grouping into size classes (Campana, 2001). Additionally, the estimation of M using Pauly's empirical formula (1980) introduces uncertainty, as this equation was developed for teleost fish and its application to elasmobranchs should be interpreted with caution.
Future research should focus on: (1) validating age and growth through vertebral reading and microchemistry analysis to determine population connectivity; (2) quantifying R. terraenovae predation on turtle hatchlings through stable isotope analysis and stomach contents; (3) evaluating the movement and residency of the species in the Marine Priority Regions through acoustic telemetry; (4) modeling trophic interactions in the coastal ecosystem using Ecopath or Ecosim models; (5) applying ecological risk assessment (ERA) models that integrate uncertainty in biological and fishery parameters (Cortés et al., 2010; Hobday et al., 2011); and (6) assessing the status of other elasmobranch species caught in the region, such as Sphyrna tiburo and Carcharhinus acronotus, to determine if they face similar pressures.
5. Conclusions
This study provides the first quantitative assessment of the population status of R. terraenovae in Marine Priority Regions 45 "La Pesca-Rancho Nuevo" and 46 "Laguna de San Andrés", and in the influence area of the Playa Tortuguera Rancho Nuevo Wetland of International Importance (Ramsar Site No. 1326), Tamaulipas, demonstrating that it is possible to obtain robust estimates in data-limited fishery contexts in high-priority conservation areas.
The estimated exploitation rate (E = 0.78; 95% CI: 0.745-0.805) significantly exceeds the reference point of E = 0.5, indicating that the population is in a state of severe overexploitation (confirming H₁).
The high fishing mortality rates (F = 3.22 yr⁻¹) reflect the intense artisanal fishing pressure in these priority regions, where fishermen from communities such as La Barra del Tordo, Tepehuajes, and Carbonera operate, within the framework of the Conservation Sector of the Marine Ecological Zoning Program.
The overexploitation of R. terraenovae has potential implications for sea turtle conservation, given that this species acts as a natural predator of hatchlings during the hatching season (August-November). The ecological effects of this population reduction could be positive (reduced predation) or negative (food web alteration), which requires specific research for fulfillment of Conservation Sector objectives (confirming the relevance of H₂).
This study demonstrates that, despite the limitations inherent to data-limited fisheries, it is possible to generate quantitative information relevant for decision-making through length-frequency based methods, uncertainty analysis, and validation through comparative indices (confirming H₃).
Urgent implementation of ecosystem-based fisheries management in Marine Priority Regions 45 and 46 is required, considering the interactions between the R. terraenovae fishery and sea turtle conservation, including minimum legal sizes (70 cm TL), temporary closures coordinated with the hatching season, fishing zoning, participatory monitoring, and strengthening of surveillance, addressing the specific recommendations of environmental policy instruments.
This study contributes to fulfillment of the objectives of the Conservation Sector of the Gulf of Mexico and Caribbean Sea Marine and Regional Ecological Zoning Program (DOF, 2012) and of the commitments derived from the Ramsar designation of the Playa Tortuguera Rancho Nuevo site, by providing scientific bases to reconcile fisheries exploitation with biodiversity conservation in these priority areas.
Author Contributions
For research articles with several authors, a short paragraph specifying their individual contributions must be provided. The following statements should be used “Conceptualization, J.H.R.C. and S.E.O.F.; methodology, J.H.R.C. and S.E.O.F.; software, J.H.R.C.; S.E.O.F.; J.A.R.O.; U.J.B.M. and J.T.O.; validation, J.H.R.C.; J.A.R.O.; U.J.B.M.; J.A.R.L. and L.A.V.O.; formal analysis, J.H.R.C.; S.E.O.F. and J.A.R.L.; investigation, J.H.R.C.; S.E.O.F.; U.J.B.M. and J.A.R.L.; resources, J.H.R.C.; S.E.O.F.; J.A.R.O.; J.T.O. J.A.R.L. and L.A.V.O.; data curation, J.H.R.C. ; U.J.B.M. ; J.T.O. and L.A.V.O.; writing—original draft preparation, J.H.R.C. and S.E.O.F.; writing—review and editing, ; S.E.O.F.; J.A.R.O. ; U.J.B.M. and J.A.R.L.; visualization, J.H.R.C.; J.A.R.O.; U.J.B.M.; J.A.R.L. and L.A.V.O.; supervision, J.H.R.C.; J.A.R.O. ; U.J.B.M. ; J.T.O. and L.A.V.O.; project administration, J.H.R.C. ; J.T.O. and L.A.V.O.; Funding acquisition, J.H.R.C.; S.E.O.F. and L.A.V.O. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The authors will make the raw data that support the conclusions of this article available to others without undue reservation.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- Aguirre-García, B.; Medina-Quej, A.; López-García, A. Análisis de la pesquería artesanal del "cazón de ley" Rhizoprionodon terraenovae (Richardson, 1836) en Campeche, México. Proceedings of the 60th Gulf and Caribbean Fisheries Institute 2007, 60, 153-160.
- Arriaga, L.; Vázquez, E.; González, J.; Jiménez, R.; Muñoz, E.; Aguilar, V. Regiones Marinas Prioritarias de México; Comisión Nacional para el Conocimiento y Uso de la Biodiversidad (CONABIO): México D.F., México, 1998.
- Bada-Sánchez, E.; Pérez-Jiménez, J.C.; Méndez-Loaeza, I. Estado del stock pesquero del cazón tutzún Rhizoprionodon terraenovae (Richardson, 1836) en el Banco de Campeche. In Proceedings of the VII Simposio Nacional de Tiburones y Rayas, Puerto Vallarta, Jalisco, México, 18-22 April 2016; Anislado-Tolentino, V., Pérez-Jiménez, J.C., Cabrera-Sánchez, M.G., Eds.; Sociedad Mexicana de Peces Cartilaginosos, A.C.: México D.F., México, 2016; pp. 53-55.
- Beverton, R.J.H.; Holt, S.J. A review of the lifespans and mortality rates of fish in nature, and their relation to growth and other physiological characteristics. Ciba Foundation Symposium - The Lifespan of Animals (Colloquia on Ageing) 1959, 5, 142-180.
- Bjorndal, K.A.; Bowen, B.W.; Chaloupka, M.; et al. Better science needed for restoration in the Gulf of Mexico. Science 2011, 331(6017), 537-538. [CrossRef]
- Branstetter, S. Age and growth validation of newborn sharks held in laboratory aquaria, with comments on the life history of the Atlantic sharpnose shark, Rhizoprionodon terraenovae. Copeia 1987, 1987(2), 291-300. [CrossRef]
- Campana, S.E. Accuracy, precision and quality control in age determination, including a review of the use and abuse of age validation methods. Journal of Fish Biology 2001, 59(2), 197-242. [CrossRef]
- Carlson, J.K.; Baremore, I.E. Changes in biological parameters of Atlantic sharpnose shark Rhizoprionodon terraenovae in the Gulf of Mexico: evidence for density-dependent growth and maturity? Marine and Freshwater Research 2003, 54(3), 227-234. [CrossRef]
- Chen, Y.; Jackson, A.; Harvey, H. A comparison of von Bertalanffy and polynomial functions in modelling fish growth data. Canadian Journal of Fisheries and Aquatic Sciences 1992, 49(6), 1228-1235. [CrossRef]
- Cochrane, K.L.; Andrew, N.L.; Parma, A.M. Primary fisheries management: a minimum requirement for provision of sustainable human benefits in small-scale fisheries. Fish and Fisheries 2011, 12(3), 275-288. [CrossRef]
- CONABIO. Sitios Prioritarios Acuáticos Epicontinentales para la Conservación de la Biodiversidad en México; Comisión Nacional para el Conocimiento y Uso de la Biodiversidad: México D.F., México, 2008.
- CONANP. Ficha Informativa de los Humedales de Ramsar - Santuario Playa Rancho Nuevo; Comisión Nacional de Áreas Naturales Protegidas: Ciudad de México, México, 2004.
- CONANP. Programa de Manejo del Santuario Playa Rancho Nuevo; Comisión Nacional de Áreas Naturales Protegidas: Ciudad de México, México, 2014.
- Cortés, E. Demographic analysis of the Atlantic sharpnose shark, Rhizoprionodon terraenovae, in the Gulf of Mexico. Fishery Bulletin 1995, 93(1), 57-66.
- Cortés, E. Standardized diet compositions and trophic levels of sharks. ICES Journal of Marine Science 1999, 56(5), 707-717. [CrossRef]
- Cortés, E. Life history patterns and correlations in sharks. Reviews in Fisheries Science 2000, 8(4), 299-344. [CrossRef]
- Cortés, E.; Arocha, F.; Beerkircher, L.; Carvalho, F.; Domingo, A.; Heupel, M.; Holtzhausen, H.; Santos, M.N.; Ribera, M.; Simpfendorfer, C. Ecological risk assessment of pelagic sharks caught in Atlantic pelagic longline fisheries. Aquatic Living Resources 2010, 23(1), 25-34. [CrossRef]
- Cortés, E. Perspectives on the intrinsic rate of population increase of elasmobranchs and consequences for sustainable management. Marine and Freshwater Research 2016, 67(12), 1843-1852.
- DOF. Acuerdo por el que se expide la parte marina del Programa de Ordenamiento Ecológico Marino y Regional del Golfo de México y Mar Caribe; Diario Oficial de la Federación: Ciudad de México, México, 24 de agosto de 2012.
- Dulvy, N.K.; Fowler, S.L.; Musick, J.A.; Cavanagh, R.D.; Kyne, P.M.; Harrison, L.R.; Carlson, J.K.; Davidson, L.N.K.; Fordham, S.V.; Francis, M.P.; et al. Extinction risk and conservation of the world's sharks and rays. eLife 2014, 3, e00590. [CrossRef]
- Efron, B.; Tibshirani, R.J. An Introduction to the Bootstrap; Chapman & Hall: New York, NY, USA, 1993.
- Estes, J.A.; Terborgh, J.; Brashares, J.S.; Power, M.E.; Berger, J.; Bond, W.J.; Carpenter, S.R.; Essington, T.E.; Holt, R.D.; Jackson, J.B.C.; et al. Trophic downgrading of planet Earth. Science 2011, 333(6040), 301-306. [CrossRef]
- FAO. The State of World Fisheries and Aquaculture 2020; Food and Agriculture Organization of the United Nations: Rome, Italy, 2020.
- Ferretti, F.; Worm, B.; Britten, G.L.; Heithaus, M.R.; Lotze, H.K. Patterns and ecosystem consequences of shark declines in the ocean. Ecology Letters 2010, 13(8), 1055-1071. [CrossRef]
- Gayanilo, F.C.; Sparre, P.; Pauly, D. FAO-ICLARM Stock Assessment Tools II (FiSAT II). Revised version. User's guide; FAO Computerized Information Series (Fisheries) No. 8; Food and Agriculture Organization of the United Nations: Rome, Italy, 2005.
- Gulland, J.A. Manual de Métodos para la Evaluación de las Poblaciones de Peces; Acribia: Zaragoza, España, 1971.
- Heist, E.J.; Musick, J.A.; Graves, J.E. Mitochondrial DNA diversity and divergence among sharpnose sharks, Rhizoprionodon terraenovae, from the Gulf of Mexico and Mid-Atlantic Bight. Fishery Bulletin 1996, 94(4), 664-668.
- Heithaus, M.R.; Frid, A.; Wirsing, A.J.; Worm, B. Predicting ecological consequences of marine top predator declines. Trends in Ecology & Evolution 2008, 23(4), 202-210.
- Hilborn, R.; Ovando, D. Reflections on the success of traditional fisheries management. ICES Journal of Marine Science 2014, 71(5), 1040-1046. [CrossRef]
- Hilborn, R.; Walters, C.J. Quantitative Fisheries Stock Assessment: Choice, Dynamics and Uncertainty; Chapman and Hall: New York, NY, USA, 1992.
- Hobday, A.J.; Smith, A.D.; Stobutzki, I.C.; Bulman, C.; Daley, R.; Dambacher, J.M.; Deng, R.A.; Dowdney, J.; Fuller, M.; Furlani, D.; et al. Ecological risk assessment for the effects of fishing. Fisheries Research 2011, 108(2-3), 372-384. [CrossRef]
- Hoenig, J.M. Empirical use of longevity data to estimate mortality rates. Fishery Bulletin 1983, 82(1), 898-903.
- Hordyk, A.; Ono, K.; Valencia, S.; Loneragan, N.; Prince, J. A novel length-based empirical estimation method of spawning potential ratio (SPR), and tests of its performance, for small-scale, data-poor fisheries. ICES Journal of Marine Science 2015, 72(1), 217-231. [CrossRef]
- INAPESCA. Boletín Hidrometeorológico - Temperatura Superficial del Mar; Instituto Nacional de Pesca: Ciudad de México, México, 2013.
- IUCN. The IUCN Red List of Threatened Species, Version 2023-1; International Union for Conservation of Nature: Gland, Switzerland, 2023.
- Jones, R.; van Zalinge, N.P. Estimations of mortality rate and population size for shrimp in Kuwait waters. Kuwait Bulletin of Marine Science 1981, 2, 273-288.
- Link, J.S. Ecosystem-Based Fisheries Management: Confronting Tradeoffs; Cambridge University Press: Cambridge, UK, 2010.
- Loefer, J.K.; Sedberry, G.R. Life history of the Atlantic sharpnose shark (Rhizoprionodon terraenovae) (Richardson, 1836) off the southeastern United States. Fishery Bulletin 2003, 101(1), 75-88.
- Márquez-Farías, J.F.; Castillo-Géniz, J.L. Fishery biology and demography of the Atlantic sharpnose shark, Rhizoprionodon terraenovae, in the southern Gulf of Mexico. Fisheries Research 1998, 39(2), 183-198. [CrossRef]
- Myers, R.A.; Baum, J.K.; Shepherd, T.D.; Powers, S.P.; Peterson, C.H. Cascading effects of the loss of apex predatory sharks from a coastal ocean. Science 2007, 315(5820), 1846-1850. [CrossRef]
- Newman, D.; Berkson, J.; Suatoni, L. Current methods for setting catch limits for data-limited fish stocks in the United States. Fisheries Research 2015, 164, 86-93. [CrossRef]
- NOM-059-SEMARNAT-2010. Norma Oficial Mexicana que determina las especies en riesgo; Secretaría de Medio Ambiente y Recursos Naturales: Ciudad de México, México, 2010.
- Oviedo-Pérez, J.L.; Zea-de-la-Cruz, H.; Aguirre-Villaseño, D.H.; Meiners-Mandujano, C.; Jiménez-Badillo, L.; González-Ocaranza, L. Talla de madurez sexual del tiburón Rhizoprionodon terraenovae en Veracruz, México. Ciencia Pesquera 2014, 22(1), 37-45.
- Parsons, G.R. Growth and age estimation of the Atlantic sharpnose shark, Rhizoprionodon terraenovae: a comparison of techniques. Copeia 1985, 1985(1), 80-85. [CrossRef]
- Pauly, D. On the interrelationships between natural mortality, growth parameters, and mean environmental temperature in 175 fish stocks. ICES Journal of Marine Science 1980, 39(2), 175-192. [CrossRef]
- Pauly, D. Fish population dynamics in tropical waters: a manual for use with programmable calculators. ICLARM Studies and Reviews 1984, 8, 1-325.
- Pauly, D. Growth performance in fishes: rigorous description of patterns as a basis for understanding causal mechanisms. Aquabyte 1991, 4(3), 3-6.
- Pauly, D.; Munro, J.L. Once more on the comparison of growth in fish and invertebrates. Fishbyte 1984, 2(1), 21.
- Pikitch, E.K.; Santora, C.; Babcock, E.A.; Bakun, A.; Bonfil, R.; Conover, D.O.; Dayton, P.; Doukakis, P.; Fluharty, D.; Heneman, B.; et al. Ecosystem-based fishery management. Science 2004, 305(5682), 346-347. [CrossRef]
- Pilcher, N.J.; Antonopoulou, M.; Rodriguez-Zarate, C.J. Marine turtle nest predation. Endangered Species Research 2021, 45, 123-136.
- Prince, J.; Hordyk, A.; Valencia, S.R.; Loneragan, N.; Sainsbury, K. Revisiting the concept of Beverton–Holt life-history invariants with the aim of informing data-poor fisheries assessment. ICES Journal of Marine Science 2014, 72(1), 194-203. [CrossRef]
- Punt, A.E.; Butterworth, D.S.; de Moor, C.L.; De Oliveira, J.A.; Haddon, M. Management strategy evaluation: best practices. Fish and Fisheries 2016, 17(2), 303-334. [CrossRef]
- Ricker, W.E. Computation and interpretation of biological statistics of fish populations. Bulletin of the Fisheries Research Board of Canada 1975, 191, 1-382.
- SEDAR. *SEDAR 13 - Small Coastal Shark Complex, Atlantic Sharpnose, Blacknose, Bonnethead, and Finetooth Shark Stock Assessment Report*; Southeast Data, Assessment, and Review: North Charleston, SC, USA, 2007.
- SEMARNAT. Diagnóstico del Sector Pesquero en la Costa Sur de Tamaulipas; Secretaría de Medio Ambiente y Recursos Naturales, Delegación Tamaulipas: Ciudad Victoria, Tamaulipas, México, 2014.
- SMPC. Código de Ética para la Investigación con Peces Cartilaginosos; Sociedad Mexicana de Peces Cartilaginosos, A.C.: México D.F., México, 2012.
- Sparre, P.; Venema, S.C. Introducción a la Evaluación de Recursos Pesqueros Tropicales. Parte 1. Manual; FAO Documento Técnico de Pesca No. 306.1, Rev. 2; Food and Agriculture Organization of the United Nations: Rome, Italy, 1998.
- Then, A.Y.; Hoenig, J.M.; Hall, N.G.; Hewitt, D.A. Evaluating the predictive performance of empirical estimators of natural mortality rate using information on over 200 fish species. ICES Journal of Marine Science 2014, 72(1), 82-92. [CrossRef]
- von Bertalanffy, L. A quantitative theory of organic growth. Human Biology 1938, 10(2), 181-213.
- Walker, T.I. Management measures. In Management Techniques for Elasmobranch Fisheries; Musick, J.A., Bonfil, R., Eds.; FAO Fisheries Technical Paper No. 474; Food and Agriculture Organization of the United Nations: Rome, Italy, 2005; pp. 216-242.
- Walpole, R.E.; Myers, R.H.; Myers, S.L.; Ye, K. Probability & Statistics for Engineers & Scientists, 9th ed.; Pearson: Boston, MA, USA, 2012.
- Witherington, B.; Kubilis, P.; Brost, B.; Meylan, A. Decreasing annual nest counts in a globally important loggerhead sea turtle population. Ecological Applications 2012, 22(5), 1533-1545. [CrossRef]
- Worm, B.; Davis, B.; Kettemer, L.; Ward-Paige, C.A.; Chapman, D.; Heithaus, M.R.; Kessel, S.T.; Gruber, S.H. Global catches, exploitation rates, and rebuilding options for sharks. Marine Policy 2013, 40, 194-204. [CrossRef]
- Zar, J.H. Biostatistical Analysis, 5th ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2010.
- Zea-de-la-Cruz, H. Estatus y evolución del tiburón Rhizoprionodon terraenovae (Richardson, 1836) en la costa central del Estado de Veracruz. Tesis de Maestría, Universidad Veracruzana, Boca del Río, Veracruz, México, 2012.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).