Submitted:
24 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Fruit flies (Diptera: Tephritidae) are major pests of crops, requiring effective monitoring for management. This study evaluated locally available food baits for monitoring fruit flies in guava orchards in Maputo, Mozambique. It also assessed infestation levels, examined the relationship between trap catches and adult/kg from incubated fruits. A randomized block design with four treatments (palm sap, molasses, torula yeast, and water) and four replications were used. Tephri traps were installed on four trees per block and inspected weekly, while guava fruits from trees and the ground were collected and incubated to estimate infestation indices. Data on flies’ density was subjected to ANOVA for analysis, to compare the treatments. Three genera (Bactrocera, Dacus, and Ceratitis) were recorded, with high relative abundance (90.37%) for Bactrocera dorsalis. Torula and palm sap were the best attractants, with no significant difference between them. Guava fruits showed high infestation (208.46±13.34 adults/kg). Trap catches of B. dorsalis were positively correlated with adults/kg, explaining 42.5% of infestation variation, highlighting the effect of the “outside the orchard” factors in the infestation indices. Results show that palm sap is a promising low-cost alternative bait and highlight the importance of considering area wide management of fruit flies.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Description of the Study Area
2.2. Sampling Procedures
2.2.1. Description of Treatments
- Molasses: a byproduct of sugar refining rich in fermentable sugars, was prepared by diluting 100 ml of molasses with 900 ml of clean water to make a 10% solution of molasses. The same procedure was used by [19]. Molasses were obtained from sugar factory located in Xinavane (Açucareira de Xinavane) (Manhiça district, Maputo province) in bottles of 20 Litres.
- Palm sap was obtained from the inflorescence of the palm tree (Phoenix reclinata), [20]. Fresh Palm sap was used and it was chosen because it is readily available in Mozambique. It was got from the local vendors (at Incoluane, Gaza province) and was kept fresh by keeping it in a deep freezer.
- For water, fresh and clean tap water was put in the tephri traps and used as a negative control.
2.2.2. Trap Placement and Monitoring
2.3. Guava Fruits Sampling
2.4. Identification of the Species of Fruit Flies
2.5. Determination of Variables
2.5.1. Estimation of the Absolute and Relative Abundance of Fruit Fly Species
2.5.2. Determination of Sex Ratios
2.5.3. Estimation of Population Density of Fruit Fly Species During the Study Period
2.5.4. Estimation of the Adult Infestation Indices of Guava Fruits from the Orchards
2.5.5. Correlation Between Trapped Fruit Flies and Adults/kg of Incubated Fruits
2.6. Data Analysis
3. Results
3.1. Absolute and Relative Abundance of the Different Fruit Fly Species and the Sex Ratio
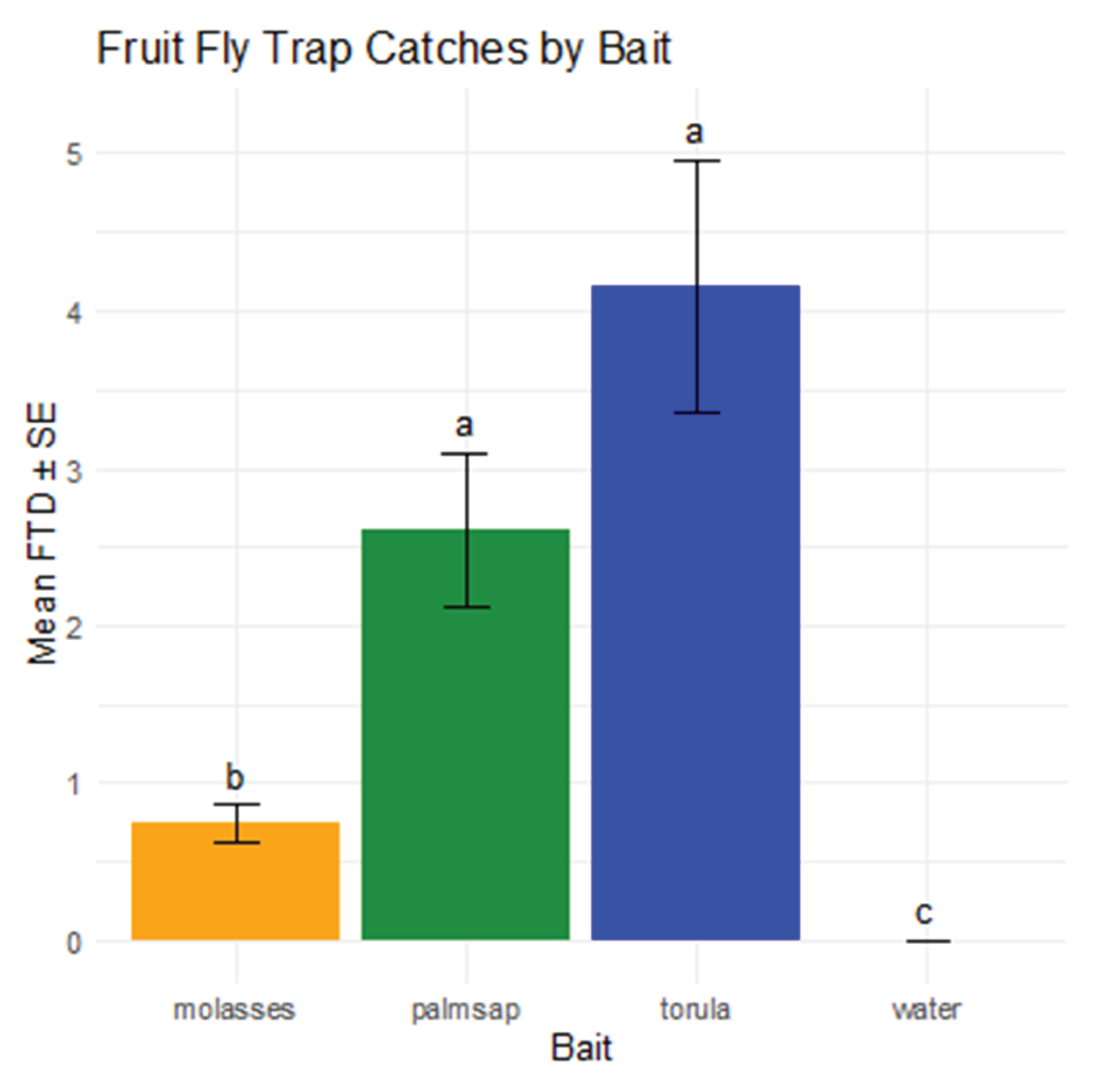
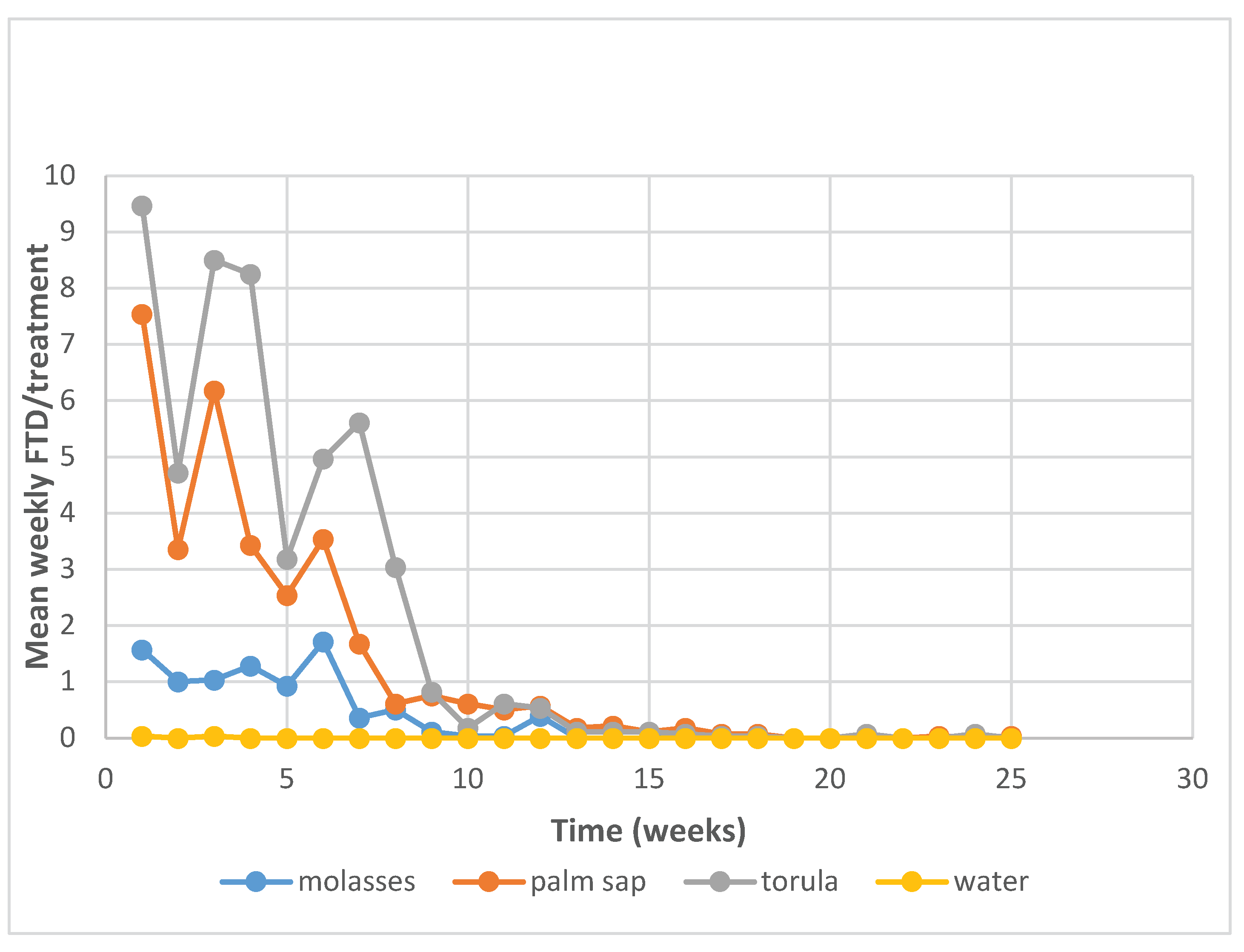
3.2. Population Density of Fruit Fly Species in Each Treatment
4.4. Infestation Indices of the Fruits
4.5. Correlation Between Trapped Flies (B. dorsalis) and B. dorsalis Infestation Index
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CABI | Center for Agriculture and Bioscience International |
| FAEF | Faculty of Agronomy and Forestry Engineering |
| FAO | Food and Agriculture Organisation |
| FTD | Fruit fly per trap per day |
| IAEA | International Atomic Energy Agency |
References
- Food and Agriculture Organization of the United Nations. FAO Agrifood Systems Employment Database. Available online: https://openknowledge.fao.org/items/44a3712c-85c6-4a63-a0e5-edfa8f15b937 (accessed on 14 November 2025).
- Angulo-López, J.E.; Flores-Gallegos, A.C.; Torres-León, C.; Ramírez-Guzmán, K.N.; Martínez, G.A.; Aguilar, C.N. Guava (Psidium Guajava L.) Fruit and Valorization of Industrialization By-Products. Processes 2021, 9, 1075. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations FAOSTAT – Production of Mangoes; Mangosteens and Guavas in Mozambique. Available online: https://www.fao.org/faostat/en/#data/QCL (accessed on 15 November 2025).
- Teshome, E.; Teka, T.A.; Nandasiri, R.; Rout, J.R.; Harouna, D.V.; Astatkie, T.; Urugo, M.M. Fruit By-Products and Their Industrial Applications for Nutritional Benefits and Health Promotion: A Comprehensive Review. Sustainability 2023, 15, 7840. [Google Scholar] [CrossRef]
- Papadopoulos, N.T.; De Meyer, M.; Terblanche, J.S.; Kriticos, D.J. Fruit Flies: Challenges and Opportunities to Stem the Tide of Global Invasions. Annu. Rev. Entomol. 2024, 69, 355–373. [Google Scholar] [CrossRef] [PubMed]
- Niassy, S.; Ekesi, S.; Migiro, L.; Otieno, W. Sustainable Management of Invasive Pests in Africa; 2020; ISBN 978-3-030-41082-7. [Google Scholar]
- José, L.; Cugala Domingos; Luisa, Santos. Assessment of Invansive Fruit Fly, Fruit Infestation and Damage in Cabo Delgado. African Crop Science Journal 2013, 21, 21–28. [Google Scholar]
- Canhanga, L.; De Meyer, M.; Cugala, D.R.; Virgilio, M; Bota, L.; Mwatawala, M. Perception of Fruit Farmers on the Occurrence of the Oriental Fruit Fly Bactrocera Dorsalis (Diptera: Tephritidae) and Its Associated Economic Impact in Manica Province, Mozambique. Fruits 2021, 76, 295–302. [Google Scholar] [CrossRef]
- De Meyer, M.; Mohamed, S.; White, I.M. Invasive Fruit Fly Pests in Africa. Available online: https://www.africamuseum.be/en/research/scientific-collections/zoology/fruit-flies (accessed on 11 January 2025).
- Vayssières, J.-F.; De Meyer, M.; Ouagoussounon, I.; Sinzogan, A.; Adandonon, A.; Korie, S.; Wargui, R.; Anato, F.; Houngbo, H.; Didier, C.; et al. Seasonal Abundance of Mango Fruit Flies (Diptera: Tephritidae) and Ecological Implications for Their Management in Mango and Cashew Orchards in Benin (Centre & North). J Econ Entomol 2015, 108, 2213–2230. [Google Scholar] [CrossRef]
- Opoku, E.; Haseeb, M.; Rodriguez, E.J.; Steck, G.J.; Cabral, M.J.S. Economically Important Fruit Flies (Diptera: Tephritidae) in Ghana and Their Regulatory Pest Management. Insects 2025, 16, 285. [Google Scholar] [CrossRef]
- Ekesi, S.; Billah, M.K.; Nderitu, P.W.; Lux, S.A.; Rwomushana, I. Evidence for Competitive Displacement of Ceratitis Cosyra by the Invasive Fruit Fly Bactrocera Invadens (Diptera: Tephritidae) on Mango and Mechanisms Contributing to the Displacement. ec 2009, 102, 981–991. [Google Scholar] [CrossRef]
- Ekesi, S.; Mohamed, S.; Tanga, C.M. Comparison of Food-Based Attractants for Bactrocera Invadens (Diptera: Tephritidae) and Evaluation of Mazoferm-Spinosad Bait Spray for Field Suppression in Mango. jnl. econ. entom. 2014, 107, 299–309. [Google Scholar] [CrossRef]
- Nanga Nanga, S.; Hanna, R.; Fotso Kuate, A.; Fiaboe, K.K.M.; Nchoutnji, I.; Ndjab, M.; Gnanvossou, D.; Mohamed, S.A.; Ekesi, S.; Djieto-Lordon, C. Tephritid Fruit Fly Species Composition, Seasonality, and Fruit Infestations in Two Central African Agro-Ecological Zones. Insects 2022, 13, 1045. [Google Scholar] [CrossRef]
- Enkerlin, W.; Arevalo, E.; Caballero, J.E.; Fezza, T.; Garavelli, E.; Martinez, D.B.; Rodriguez, P.A.; Shelly, T.; Thomas, M.E.; Villaseñor, A.; et al. Searching for More Effective Food Baits for Tephritid Fruit Flies (Diptera: Tephritidae): Performance of Newly Developed Vial-Lures Relative to Torula Yeast Borax. Insects 2025, 16, 53. [Google Scholar] [CrossRef]
- Ekesi, S. Billah A Field Guide to the Management of Economically Important Tephritid Fruit Flies in Africa; International Centre of Insect Physiology and Ecology (ICIPE): Nairobi, Kenya, 2006. [Google Scholar]
- Bortoli, L.C.; Machota, R.; Garcia, F.R.M.; Botton, M. Evaluation of Food Lures for Fruit Flies (Diptera: Tephritidae) Captured in a Citrus Orchard of the Serra Gaúcha. Florida Entomologist 2016, 99, 381–384. [Google Scholar] [CrossRef]
- Bota, L.; Fabião, B.; De Meyer, M.; Manuel, L.; Mwatawala, M.; Virgilio, M.; Canhanga, L.; Cugala, D. Fine-Scale Infestation Pattern of Bactrocera Dorsalis (Diptera: Tephritidae) in a Mango Orchard in Central Mozambique. Int J Trop Insect Sci 2020, 40, 943–950. [Google Scholar] [CrossRef]
- Alves, J.C.G.; De Brito, C.H.; Oliveira, R.D.; Corsato, C.D.A.; Silva, J.F.D.; Barbosa, V.D.O.; Batista, J.D.L. Food Attractants Used in the Fruit Fly Monitoring (Diptera: Tephritidae) in a Commercial Orchard of Psidium Guajava. JEAI 2019, 1–10. [Google Scholar] [CrossRef]
- Hai, A.; Rambabu, K.; Al Dhaheri, A.S.; Kurup, S.S.; Banat, F. Tapping into Palm Sap: Insights into Extraction Practices, Quality Profiles, Fermentation Chemistry, and Preservation Techniques. Heliyon 2024, 10, e35611. [Google Scholar] [CrossRef]
- Shelly, T.; Fezza, T.; Kurashima, R. Captures of Oriental Fruit Flies and Melon Flies (Diptera: Tephritidae) in Traps Baited With Torula Yeast Borax Solution Or 2- Or 3-Component Synthetic Food Cones in Hawaii. Florida Entomologist 2022, 105. [Google Scholar] [CrossRef]
- García-Mendoza, P.J.; Morales Valles, P.A.; Pérez-Almeida, I.; Taramona Ruíz, L.A.; Marín-Rodríguez, C.A. Efficiency of Handmade Attractants in Fruit Fly Control. Bioagro 2024, 36, 335–346. [Google Scholar] [CrossRef]
- Cugala, D.R.; Mahumane, W.; Gabriel, A.; Mausse, S. Assessment of the Status of Fruit Flies with Emphasis on the Invasive Exotic Fruit Fly *Bactrocera Invadens* in Mozambique; Faculty of Agronomy and Forest Engineering (FAEF): Mozambique, 2008. [Google Scholar]
- Billah, M.K.; Afreh-Nuamah, K. Species Range of Fruit Flies Associated with Mango from Three Agro-Ecological Zones in Ghana. Unpublished manuscript, available on ResearchGate 2012. [Google Scholar]
- Santos, J.P.D.; Arioli, C.J.; Rosa, J.M.D.; Menezes-Netto, A.C. EFFICIENCY OF FOOD LURES FOR CAPTURE AND MONITORING OF SOUTH AMERICAN FRUIT FLY IN ASIAN PEAR ORCHARD. Rev. Caatinga 2022, 35, 722–729. [Google Scholar] [CrossRef]
- FAO/IAEA Trapping Guidelines for Area-Wide Fruit Fly Programmes, Second Edition; Enkerlin, W.R., Reyes-Flores, J., Eds.; Food and Agriculture Organization of the United Nations / International Atomic Energy Agency: Rome, Italy, 2018. [Google Scholar]
- Nankinga, C.M.; Isabirye, B.E.; Muyinza, H.; Rwomushana, I.; Stevenson, P.C.; Mayamba, A.; Aool, W.; Akol, A.M. Fruit Fly Infestation in Mango: A Threat to the Horticultural Sector in Uganda; 2014. [Google Scholar]
- De, Meyer; Mohamed, S.; White, I.M. Invasive Fruit Fly Pests in Africa: A Diagnostic Tool and Information Reference for the Four Asian Species of Fruit Flies (Diptera: Tephritidae) That Have Become Accidentally Established as Pests in Africa, Including the Indian Ocean Islands; International Centre of Insect Physiology and Ecology (icipe) and Royal Museum for Central Africa: Nairobi, Kenya and Tervuren, Belgium, 2012. [Google Scholar]
- Virgilio, M.; White, I.; De Meyer, M. A Set of Multi-Entry Identification Keys to African Frugivorous Flies (Diptera, Tephritidae). ZK 2014, 428, 97–108. [Google Scholar] [CrossRef]
- Hanna, R.; Gnanvossou, D.; Goergen, G.; Bokonon-Ganta, A.H.; Mohamed, S.A.; Ekesi, S. Efficiency of Food-Based Attractants for Monitoring Tephritid Fruit Fly Diversity and Abundance in Mango Systems across Three West African Agro-Ecological Zones. Journal of Economic Entomology 2019, 113, 860–871. [Google Scholar] [CrossRef]
- Howse, E.T.; Van Klinken, R.D.; Beeton, N.J.; Spafford, H.; James, K.P.; Hill, M.P. Field Evaluation of Female- and Male-Targeted Traps for Ceratitis Capitata (Diptera: Tephritidae). Journal of Economic Entomology 2024, 117, 1459–1467. [Google Scholar] [CrossRef] [PubMed]
- Duyck, P.F.; Sterlin, J.F.; Quilici, S. Survival and Development of Different Life Stages of Bactrocera Zonata (Diptera: Tephritidae) Reared at Five Constant Temperatures Compared to Other Fruit Fly Species. Bull. Entomol. Res. 2004, 94, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Duyck, P.F.; Quilici, S.; Peyre, G. Survival and Development of Three Invasive Tephritidae Fruit Fly Species in the Presence of a Natural Enemy. Biocontrol Science and Technology 2011, 21, 429–440. [Google Scholar] [CrossRef]
- Raga, A.; Paula, L.; Souza-Filho, M.; Castro, J. Population Dynamics and Infestation Rate of Fruit Flies in Stone Fruits in São Paulo State, Brazil. ARRB 2017, 14, 1–11. [Google Scholar] [CrossRef]
- Jenkins, D.A.; Epsky, N.D.; Kendra, P.E.; Heath, R.R.; Goenaga, R. Food-Based Lure Performance in Three Locations in Puerto Rico: Attractiveness to Anastrepha Suspensa and A. Obliqua (Diptera: Tephritidae). Florida Entomologist 2011, 94, 186–194. [Google Scholar] [CrossRef]
- Khan, M.H.; Khuhro, N.H.; Awais, M.; Asif, M.U.; Muhammad, R. Seasonal Abundance of Fruit Fly, Bactrocera Species (Diptera: Tephritidae) with Respect to Environmental Factors in Guava and Mango Orchards. PJAR 2021, 34. [Google Scholar] [CrossRef]
- Crawley, M.J. The R Book, 2nd ed.; John Wiley & Sons: Chichester, UK, 2012; ISBN 978-0-470-97392-9. [Google Scholar]
- Grechi, I.; Preterre, A.-L.; Lardenois, M.; Ratnadass, A. Bactrocera Dorsalis Invasion Increased Fruit Fly Incidence on Mango Production in Reunion Island. Crop Protection 2022, 161, 106056. [Google Scholar] [CrossRef]
- Ndayizeye, L.; Odette, M.; Nduwarugira, D.; Sibomana, C.; Dieudonné, N. Dominance of Bactrocera Dorsalis Hendel on Ceratitis Cosyra Walker (Diptera: Tephritidae) in Orchards in Bujumbura, Burundi. Arch. Agric. Environ. Sci. 2024, 9, 345–349. [Google Scholar] [CrossRef]
- CABI Bactrocera Dorsalis (Oriental Fruit Fly). Available online: https://www.cabi.org/cpc (accessed on 10 May 2025).
- Shelly, T.; Kurashima, R.; Fezza, T. Field Evaluation of Three-Component Solid Food-Based Dispenser versus Torula Yeast for Capturing Mediterranean and Oriental Fruit Flies (Diptera: Tephritidae). Journal of Asia-Pacific Entomology 2020, 23, 825–831. [Google Scholar] [CrossRef]
- Shelly, T. Distant-Dependent Capture Probabilities of Mediterranean and Oriental Fruit Flies (Diptera: Tephritidae) in a Food-Based Trap in a Hawaiian Mango Orchard. Florida Entomologist 2021, 104. [Google Scholar] [CrossRef]
- Delgado, S.; Calvo, M.V.; Duarte, F.; Borges, A.; Scatoni, I.B. Food Attractants for Mass Trapping of Fruit Flies (Diptera: Tephritidae) and Its Selectivity for Beneficial Arthropods. Florida Entomologist 2022, 105. [Google Scholar] [CrossRef]
- Shelly, T.; Kurashima, R.; Nishimoto, J.; Andress, E. Capture of Zeugodacus Cucurbitae (Diptera: Tephritidae) in Traps Baited with Torula Yeast Solution Versus Cucumber Volatile Plugs. Florida Entomologist 2017, 100, 15–20. [Google Scholar] [CrossRef]
- Siderhurst, M.S.; Jang, E.B. Cucumber Volatile Blend Attractive to Female Melon Fly, Bactrocera Cucurbitae (Coquillett). J Chem Ecol 2010, 36, 699–708. [Google Scholar] [CrossRef]
- Royer, J.E.; De Faveri, S.G.; Lowe, G.E.; Wright, C.L. Cucumber Volatile Blend, a Promising Female-biased Lure for B Actrocera Cucumis ( F Rench 1907) ( D Iptera: T Ephritidae: D Acinae), a Pest Fruit Fly That Does Not Respond to Male Attractants. Austral Entomology 2014, 53, 347–352. [Google Scholar] [CrossRef]
- Katiyar, K.P.; Molina, J.C.; Matheus, R. Fruit Flies (Diptera: Tephritidae) Infesting Fruits of the Genus Psidium (Myrtaceae) and Their Altitudinal Distribution in Western Venezuela. The Florida Entomologist 2000, 83, 480. [Google Scholar] [CrossRef]
- Abraham, J.; Amissah, C.; Kuffour, F.O.; Abraham, J.D. Palm Wine as a Food-Based Bait for Monitoring Adult Ceratitis Ditissima (Munro) (Diptera: Tephritidae) in Citrus Orchards. African Entomology 2023, 31, e12637. [Google Scholar] [CrossRef]
- Salvi, J.; Katewa, S.S. Chemical Composition and Nutritive Value of Sap of Phoenix Sylvestris Roxb. Electronic Journal of Environmental, Agricultural and Food Chemistry 2012, 11, 578–583. [Google Scholar]
- Makhlouf-Gafsi, I.; Mokni-Ghribi, A.; Bchir, B.; Attia, H.; Blecker, C.; Besbes, S. Physico-Chemical Properties and Amino Acid Profiles of Sap from Tunisian Date Palm. Sci. agric. (Piracicaba, Braz.) 2016, 73, 85–90. [Google Scholar] [CrossRef]
- Vargas, R.; Piñero, J.; Leblanc, L. An Overview of Pest Species of Bactrocera Fruit Flies (Diptera: Tephritidae) and the Integration of Biopesticides with Other Biological Approaches for Their Management with a Focus on the Pacific Region. Insects 2015, 6, 297–318. [Google Scholar] [CrossRef]
- Deconninck, G.; Boulembert, M.; Eslin, P.; Couty, A.; Dubois, F.; Gallet-Moron, E.; Pincebourde, S.; Chabrerie, O. Fallen Fruit: A Backup Resource during Winter Shaping Fruit Fly Communities. Agri and Forest Entomology 2024, 26, 232–248. [Google Scholar] [CrossRef]
- Vayssieres, J.; Adandonon, A.; Sinzogan, A.; Korie, S. Diversity of Fruit Fly Species (Diptera: Tephritidae) Associated with Citrus Crops (Rutaceae) in Southern Benin in 2008–2009. International Journal of Biological and Chemical Sciences 2011, 4. [Google Scholar] [CrossRef]
- Vayssières, J.-F.; Korie, S.; Ayegnon, D. Correlation of Fruit Fly (Diptera Tephritidae) Infestation of Major Mango Cultivars in Borgou (Benin) with Abiotic and Biotic Factors and Assessment of Damage. Crop Protection 2009, 28, 477–488. [Google Scholar] [CrossRef]
- Wen-Hua, C.; Tsui-Ying, C.; Rou-Tong, L.; Li-Hsin, H. Correlation between Bactrocera Dorsalis (Hendel) (Diptera: Tephritidae) Trap Catches and Guava Fruit Infestation in Taiwan. Journal of Entomological Research 2023, 47, 28–34. [Google Scholar] [CrossRef]
- Manrakhan, A.; Daneel, J. -H.; Beck, R.; Virgilio, M.; Meganck, K.; De Meyer, M. Efficacy of Trapping Systems for Monitoring of Afrotropical Fruit Flies. J Applied Entomology 2017, 141, 825–840. [Google Scholar] [CrossRef]
|
SPECIES |
TREATMENT | ||||||||
| Molasses | Palm sap | Torula | Water | Grand total | |||||
| Total (A/A) | R/A (%) | Total (A/A) | R/A (%) | Total (A/A) | R/A (%) | Total (A/A) | R/A (%) | ||
| Bactrocera dorsalis | 252 | 84.28 | 903 | 92.33 | 1,413 | 90.40 | 2 | 50.00 | 2,570 |
| Dacus bivitatus | 27 | 9.03 | 11 | 1.12 | 98 | 6.27 | 0 | 0.00 | 136 |
| Dacus punctatifrons | 1 | 0.33 | 8 | 0.82 | 8 | 0.51 | 1 | 25.00 | 18 |
| Dacus frontalis | 17 | 5.69 | 26 | 2.66 | 9 | 0.58 | 1 | 25.00 | 53 |
| Dacus vertebratus | 2 | 0.67 | 24 | 2.45 | 2 | 0.13 | 0 | 0.00 | 28 |
| Dacus ciliatus | 0 | 0.00 | 1 | 0.10 | 1 | 0.06 | 0 | 0.00 | 2 |
| Ceratitis quilicii | 0 | 0.00 | 1 | 0.10 | 15 | 0.96 | 0 | 0.00 | 16 |
| Ceratitis rosa | 0 | 0.00 | 2 | 0.20 | 8 | 0.51 | 0 | 0.00 | 10 |
| Ceratitis capitata | 0 | 0.00 | 1 | 0.10 | 7 | 0.45 | 0 | 0.00 | 8 |
| Ceratitis punctata | 0 | 0.00 | 1 | 0.10 | 1 | 0.06 | 0 | 0.00 | 2 |
| Ceratitis cosyra | 0 | 0.00 | 0 | 0.00 | 1 | 0.06 | 0 | 0.00 | 1 |
| Total | 299 | 100 | 978 | 100 | 1563 | 100 | 4 | 100 | 2,844 |
| SPECIES | TREATMENT | |||||||
| Molasses | Palm sap | torula | water | |||||
| F (%) | M (%) | F (%) | M (%) | F (%) | M (%) | F (%) | M (%) | |
| Bactrocera dorsalis | 70.63 | 29.37 | 56.70 | 43.30 | 63.98 | 36.02 | 0.00 | 100 |
| Dacus bivitatus | 0.00 | 100 | 33.36 | 63.64 | 31.63 | 68.37 | 0.00 | 0.00 |
| Dacus punctatifrons | 0.00 | 100 | 12.50 | 87.50 | 25.00 | 75.00 | 0.00 | 100 |
| Dacus frontalis | 17.65 | 82.35 | 26.92 | 73.08 | 44.44 | 55.56 | 0.00 | 100 |
| Dacus vertebratus | 0.00 | 100 | 20.83 | 79.17 | 50.00 | 50.00 | 0.00 | 0.00 |
| Dacus ciliatus | 0.00 | 0.00 | 100 | 0.00 | 100 | 0.00 | 0.00 | 0.00 |
| Ceratitis quilicii | 0.00 | 0.00 | 100 | 0.00 | 93.33 | 6.67 | 0.00 | 0.00 |
| Ceratitis rosa | 0.00 | 0.00 | 50.00 | 50.00 | 100 | 0.00 | 0.00 | 0.00 |
| Ceratitis capitata | 0.00 | 0.00 | 100 | 0.00 | 100 | 0.00 | 0.00 | 0.00 |
| Ceratitis punctata | 0.00 | 0.00 | 100 | 0.00 | 100 | 0.00 | 0.00 | 0.00 |
| Ceratiti cosyra | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 100 | 0.00 | 0.00 |
| Fruit source | Total number of fruits | Total weight (kg) | Number of pupae | Emerged adults | Bactrocera dorsalis | Females | Males | pupae/kg | Adult/kg |
| Guava (ground) | 456 | 29.568 | 6154 | 5120 | 5115 | 2719 | 2401 | 210.00±16.6 | 175.00 ± 13.7 |
| Guava (from tree) | 364 | 22.013 | 6713 | 5808 | 5799 | 2888 | 2920 | 287.00±28.3 | 248 ± 23.1 |
| Total (overall) | 820 | 51.581 | 12,867 | 10,928 | 10,914 | 5,607 | 5,321 | 245.06±16.10 | 208.46 ± 13.34 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




