1. Introduction
Malaria remains one of the most devastating global health challenges, with an estimated 263 million cases and 597,000 deaths reported in 2023, the majority occurring in sub-Saharan Africa [
1]. The disease is caused by
Plasmodium parasites transmitted through infected
Anopheles mosquitoes. Following transmission, sporozoites travel to the liver where they invade hepatocytes and undergo extensive replication, producing thousands of merozoites that subsequently initiate the symptomatic blood-stage infection [
2,
3,
4]. Among the five
Plasmodium species that infect humans,
P. falciparum is the most virulent, and the emergence of drug resistance particularly to artemisinin-based therapies continues to undermine control efforts [
5]. These challenges highlight the urgent need for novel intervention strategies targeting understudied aspects of the parasite life cycle, particularly the clinically silent liver stage.
During liver stage infection, only a small number of hepatocytes become infected. Within these cells, the parasite undergoes extensive asexual schizogony, producing thousands of new daughter merozoites [
3,
4]. While this replication process is remarkably efficient, the parasite relies entirely on host cell resources for its growth and development. During its development within hepatocytes, the parasite resides within a parasitophorous vacuole (PV) enclosed by parasitophorous vacuole membrane (PVM). This specialized compartment is crucial for nutrient acquisition, immune evasion, and parasite egress [
6,
7,
8,
9]. The host hepatocyte serves as a protective niche, shielding the parasite from immune detection while supplying essential nutrients. For instance,
Plasmodium lacks the ability to synthesize sterols
de novo and instead scavenges host-derived cholesterol, either produced internally or acquired via low-density lipoproteins [
10]. Additionally, the parasite relies on soluble host factors, such as proteins and solutes, to support its replication during schizogony. These nutrients may be delivered via the host cell’s canonical autophagy and endolysosomal pathways. Various host vesicles including autophagosomes, amphisomes, late endosomes, and lysosomes have been observed associated or near the PVM, and disrupting these compartments impairs robust liver stage development [
8,
11,
12,
13,
14,
15]. Despite the well-established importance of the PVM, the molecular mechanisms underlying its interactions with host organelles, particularly lysosomes and late endosomes, remain poorly understood. Intriguingly, many intracellular pathogens manipulate host vesicle trafficking to promote their survival and replication [
16,
17,
18]. This raises the possibility that
Plasmodium similarly exploits host membrane trafficking machinery, particularly SNARE-mediated fusion events, to facilitate its growth within hepatocytes.
SNARE proteins are a highly conserved family of membrane-associated proteins that mediate vesicle fusion in eukaryotic cells, playing essential roles in endocytosis, exocytosis, autophagy, and lysosomal delivery [
19,
20]. They are broadly classified into vesicle-associated SNAREs (v-SNAREs) and target membrane-associated SNAREs (t-SNAREs) [
21,
22]. v-SNAREs typically contribute an arginine (R) residue to the fusion process, whereas the t-SNAREs typically contribute a glutamine (Q) residue. During fusion, a v-SNARE on the vesicle forms a stable four-helix bundle with three t-SNAREs on the target membrane, driving membrane proximity and overcoming the energy barrier to fusion [
23]. SNAREs are widely distributed across eukaryotes, with 38 members in humans, 25 in
Saccharomyces cerevisiae, and 18 SNARE-like proteins in
Plasmodium [
24,
25]. In mammalian cells, specific SNAREs including VAMP7, VAMP8, and Vti1B regulate endolysosomal trafficking. These proteins often function in precise combinations to ensure the specificity of membrane fusion and cargo delivery [
26,
27]. SNARE proteins, as key regulators of vesicle trafficking, are frequently exploited by intracellular pathogens. Many parasites, bacteria and viruses have developed strategies to manipulate SNARE function, diverting vesicular transport to facilitate their replication or avoid immune detection [
16,
28,
29,
30,
31,
32]. However, the role of host SNARE proteins in
Plasmodium liver stage development remains poorly understood. Notably, none of the SNARE-like proteins investigated so far in
Plasmodium localize to the PVM, suggesting that the parasite may instead co-opt host SNARE machinery to mediate the fusion of nutrient-carrying vesicles. Investigating how
Plasmodium engages these host factors could yield crucial insights into the molecular mechanisms driving its intracellular survival and a novel target for antimalarial intervention.
Using a CRISPR/Cas9-engineered knockout system combined with quantitative imaging, this study aims to elucidate the functional roles of key SNARE proteins involved in early-to-late endosomal fusion and autophagosome-lysosome fusion pathways in mediating vesicle-PVM interactions during Plasmodium liver stage development.
2. Materials and Methods
2.1. Ethics Statement
The experiments were conducted in strict accordance with the Swiss Animal Protection Act (TSchG) and received approval from the Animal Experimentation Commission of the Canton of Bern (Authorization BE118/22). Research staff involved in animal handling possessed certifications accredited by the Federation of European Laboratory Animal Science Associations (FELASA). Female C57BL/6 and BALB/c mice, aged 6 to 12 weeks and weighing 20–30 g at the time of infection, were obtained from Janvier Labs (France) or bred in-house at the University of Bern’s Institute of Cell Biology (IZB). Housing was provided in individually ventilated cages (IVCs) containing standard bedding and nesting material. Environmental conditions were maintained at a 12h light/dark cycle, 20–22 °C, and 55 ± 10% relative humidity. Ad libitum access to chow and water was provided, and animal well-being was ensured through daily monitoring by trained personnel.
2.2. Plasmodium Parasite Lines
The study used
P. berghei ANKA strain, including fluorescent reporter lines
PbmCherry (expressing mCherry under
P. berghei hsp70; [
9].
2.3. Cell Culture and In Vitro Infection of HeLa Cells
Human epithelial HeLa cells (European Cell Culture Collection) were cultured in complete Minimum Essential Medium (cMEM; MEM with Earle's salts (1-31F01-I, BioConcept, Allschwil, Switzerland) supplemented with 10% heat-inactivated fetal bovine serum [Sigma-Aldrich], 100 U penicillin, 100 ug/ml streptomycin and 2 mM L-Glutamine (all from BioConcept, Allschwil, Switzerland)) at 37°C under 5% CO₂ atmosphere. For infection assays, 40,000 HeLa cells were seeded onto glass coverslips in 24-well plates, allowed to adhere overnight, then infected with P. berghei sporozoites in cMEM containing 2.5 μg/mL amphotericin B (E437, Bioconcept, Allschwil, Switzerland) to prevent fungal contamination, with medium changes performed every 24 hours.
2.4. Stable Expression of GFP-Tagged SNARE Constructs in HeLa Cells
To generate HeLa cells stably expressing GFP-tagged SNAREs, SNARE-GFP constructs were subcloned into lentiviral backbone plasmids (pLX307, Addgene #98343 or pLVX, Addgene #58263) as previously described [
33]. Briefly, the open reading frames of VAMP7, VAMP8, and Stx7 were amplified using specific primers (VAMP7-GFP: 5’-GATCCGCTAGCGCTACCGG-3’ and 5’-CTATTTCTTCACACAGCTTGGCCATG-3’; VAMP8-GFP: 5’-GATCCGCTAGCGCTACCGG-3’ and 5’-TTACTTGTACAGCTCGTCCATGCC-3’; Stx7-GFP: 5’-ATGTTTACACTCCAGGAGTTGG-3’ and 5’-AGATATCTTTACTTGTACAGCTCGTCCATG-3’) and cloned via NheI and EcoRV restriction sites. The lentiviral plasmid pLX307 (Addgene #98343) was kindly provided by William Hahn and Sefi Rosenbluh [
34,
35], while the VSV-G envelope plasmid pMD2.G (Addgene #12259) and packaging plasmid psPAX2 (Addgene #12260) were gifts from Didier Trono. Lentiviruses (Addgene #58263) were produced in human embryonic kidney cells (HEK293T) cells, and HeLa cells were transduced following established protocols [
33]. Transduced cells were selected with 1 µg/mL puromycin from 2 days post-transduction for 3-day treatment, and single-cell clones were isolated and validated by live imaging and immunofluorescence assay (IFA) using anti-GFP antibodies.
2.5. Generation of SNARE Knockout Cell Lines Using CRISPR/Cas9
SNARE knockout (KO) cell lines were generated using the CRISPR/Cas9 GeCKO v2 lentiviral system [
36]. The system employed two vectors: pLentiCas9-Blast (blasticidin resistance; encoding hSpCas9; Addgene #52962) and pLentiGuide-Puro (puromycin resistance; encoding sgRNAs; Addgene #52963), both gifts from Feng Zhang. Target-specific guide RNAs were designed to disrupt exon 1 of human VAMP7 (gRNA: caccgAACAGCAAAAAGAATCGCCA), exon 2 of VAMP8 (gRNA: caccgTGTGCGGAACCTGCAAAGTG), exon 1 of Vti1B (gRNA: caccgAAGCTGCACGAGATCTTCCG), and exon 3 of Stx7 (gRNA: caccgGATGTTAGAAGAGATCCTCT), and were cloned into the pLentiGuide-Puro backbone. For double (2KO) or triple (3KO) knockouts, we additionally used lentiGuide-Hygro-eGFP (hygromycin resistance; Addgene #99375) or lentiGuide-zeo (zeocin resistance; Addgene #99375). Lentiviruses were produced as previously described [
33], and HeLa cells were transduced with both Cas9- and sgRNA-encoding viruses. Transduced cells were selected with puromycin (1 μg/mL) and blasticidin (4 μg/mL) for 3 days, or with hygromycin/zeocin (500 μg/mL) for 5 days when using hygro-eGFP or zeocin-based constructs. For combinations of resistance markers (Puro/Hygro/Zeo/Blast), antibiotics were applied simultaneously with daily media changes and cell death monitoring. Single-cell clones were isolated and validated by Western blotting and Sanger sequencing of target loci using gene-specific primers (VAMP7: 5′- CTTCTGTTGAACTATTTCTCTAGG-3′ and 5′-ACCAGATATATTTTCTAAGTTGGTG-3′; VAMP8: 5′-CATCTCACCTTCTGGGGCTTACAAATGC-3′ and 5′-GTCATGGGATCCATTCCCTGGGATCC-3′; Vti1B: 5′-CTTTCGCTGCGGCCTTTCCCCAAC-3′ and 5′-GGACCGCAGATGCAAAAGCCCCTC-3′; Stx7: 5′-GACTCTGTCCTTTGAAGTCCAG-3′ and 5′-GTAGTGCTACCTGGTTCCAAAC-3′). Editing efficiency was quantified using TIDE (
https://tide.nki.nl) where applicable.
2.6. Western Blotting
Confluent HeLa cells in 6-well plates were rinsed with PBS and lysed in 200 μL of preheated (95°C) Laemmli buffer (25 mM Tris-HCl pH 6.8, 2% glycerol, 0.8% SDS, 0.004% bromophenol blue, 2% 2-mercaptoethanol). Lysates were collected, treated with Universal Nuclease (Thermo Fisher #88701; 5 min, RT), and heat-denatured at 95°C for 5 min as previously described [
35]. Proteins were resolved on 15% SDS-PAGE gels alongside PageRuler Pre-stained NIR markers (Thermo Fisher #26635) and transferred to nitrocellulose membranes using a Hoefer tank blot system. After blocking for 30 min with 5% non-fat milk in PBST (PBS containing 0.05% Tween-20), membranes were incubated for 1 h at room temperature with primary antibodies: : mouse anti-VAMP7 (1:500; Synaptic Systems #232011), mouse anti-VAMP8 (1:500; Santa Cruz #sc-166820), mouse anti-Vti1B (1:1000; BD Biosciences #611404), mouse anti-Stx7 (1:500; Santa Cruz #sc-514017), chicken anti-GAPDH (1:3000; Millipore #AB2302) and mouse anti-GAPDH (1:3000; DSHB #2G7), and mouse anti-α-tubulin (1:1000; Cell Signaling #2125). Membranes were then incubated for 1 h with HRP-conjugated secondary antibodies: goat anti-mouse IgG (HRP) (1:3000; Sigma #A5278), and goat anti-chicken IgY (HRP) (1:5000; Invitrogen #A16054). Protein signals were then detected using SuperSignal West Femto substrate (Thermo Fisher #34096), and chemiluminescence was captured with an Amersham Imager 600 in semi-automated mode.
2.7. Sporozoite Isolation from Mosquito Salivary Glands
Sporozoites were isolated from
Anopheles mosquitoes using established protocols [
37,
38]. Mosquitoes were fed on
Plasmodium-infected mice with 0.2–1% gametocytemia, and isolations were initiated from day 18 post-infection. For salivary gland sporozoite collection, mosquitoes were anesthetized with chloroform, surface-sterilized in 70% ethanol, and rinsed twice with PBS. Glands were dissected under a binocular microscope in Iscove’s Modified Dulbecco’s Medium (IMDM), pooled in 50 μL ice-cold IMDM, and homogenized using a motorized pestle system (Z359947/Z359971, Sigma, Buchs, Switzerland). The sporozoite suspension was quantified via Neubauer hemacytometer by enumerating sporozoites in four corner grids. Total yields (sporozoites/mL and per mosquito) were calculated based on dilution factors and dissection counts. Freshly isolated sporozoites were immediately used to infect seeded HeLa cells and was centrifuged (1000 × g, 1 min) before been incubated at 37°C with 5% CO
2.
2.8. Live Cell Imaging
HeLa cells were seeded in 96-well plates and allowed to adhere overnight before infection with
P. berghei salivary gland sporozoites during a 2-hour incubation at 37°C. Following infection, cells were washed twice with MEM medium to remove uninternalized sporozoites and salivary gland debris, the cells were then rinsed with 1XPBS and incubate with accutase for 10 mins before been transferred to 24-well glass-bottom imaging plates (P24-1.5H-N, CellVis, California, USA) for maintenance in complete cMEM at 37°C/5% CO₂ as previously described [
8,
39]. After 1-hour incubation in fresh cMEM, time-lapse imaging was performed using a Nikon W1 LIPSI spinning-disk confocal system with a 100 oil-immersion objective (NA 1.45), capturing infected cells at defined intervals to simultaneously to monitor the infected cells dynamics and parasite tagged protein fluorescence signal development throughout the intracellular lifecycle.
2.9. Immunofluorescence Analysis
Immunofluorescence was carried out as previously described [
40,
41]. Infected HeLa cells on glass coverslips were fixed with 4% PFA (F8775, Sigma, Buchs, Switzerland) in PBS at room temperature for 15 min, PBS-washed, and blocked with 10% FCS/PBS for 20 min, at room temperature. The cells were then incubated in 10% FCS/PBS for 1 h, at room temperature with primary antibodies: Vti1B (mouse mAb 1:1000 (1:500 PS-ExM), BD Transduction Laboratories #611404), UIS4 (rabbit 1:1000, P. Sinnis; chicken 1:10000, Proteogenix), hLAMP1 (mouse mAb 1:1000 (1:500 PS-ExM), DSHB H4A3; rabbit pAb 1:1000, Cell Signaling #9091), GFP (rabbit pAb 1:1000, Origene TA100030; mouse mAb 1:1000, Roche AQ160), and α-tubulin (guinea pig pAb 1:500, ABCD AA345). After PBS washes, the cells were incubated in secondary antibodies (1:1000, 1 h room temperature) included: anti-rabbit/mouse Alexa Fluor 488/594 (Invitrogen A11034/A21207/A11001/A11032), anti-guinea pig Alexa Fluor 647 (Sigma SAB4600033), and anti-chicken Cy5 (Jackson ImmunoResearch #AB_2340365). Nuclei were stained with DAPI before mounting with ProLong Gold (Invitrogen #P36930). The cells were imaged with a Nikon W1 LIPSI Confocal microscope with DAPI/FITC/TRITC/Cy5 filters. The single-staining controls were conducted to exclude crosstalk.
2.10. Pre-Staining Expansion Microscopy (PS-ExM)
PS-ExM of infected HeLa cells expressing SNARE proteins was performed as previously described [
42]. Briefly, cells grown on 13 mm glass coverslips in 24-well plates were fixed with 4% PFA in PBS. Pre-expansion immunofluorescence staining was carried out as described above. The cells were then anchored in PBS containing 0.7% formaldehyde and 1% acrylamide at 37°C for 2 h. Gelation was carried out using a monomer solution (MS) containing 19% sodium acrylate (408220, Sigma, Buchs, Switzerland), 10% acrylamide (A4058, Sigma, Buchs, Switzerland), and 0.1% bisacrylamide (M1533, Sigma, Buchs, Switzerland). The MS was mixed with 10% TEMED (17919, Thermo Fisher, Reinach, Switzerland) and 10% APS (5 µL each per 90 µL monomer solution). Samples on coverslips were inverted onto 35 µL monomer droplets on parafilm in a humid chamber, incubated on ice for 5 min to improve penetration, and polymerized at 37°C for 30 min. Gel-embedded samples were denatured in SDS buffer (200 mM SDS, 200 mM NaCl, 50 mM Tris-HCl, pH 9) with agitation (200 rpm, 15 min at room temperature), followed by incubation at 95°C for 1 h. Initial expansion was performed in MilliQ water for 1 h, with optional water exchange. The gels were incubated with DAPI (1 µg/mL in 2% BSA) to stain the DNA at 37°C for 2 h. Fully expanded samples were mounted in 0.2% propyl gallate (02370, Sigma, Buchs, Switzerland) on poly-D-lysine-coated glass-bottom dishes (Gibco A38904, Buchs, Switzerland) and imaged using a Nikon W1 LIPSI confocal microscope.
2.11. Liver Stage Parasite Size Measurement
To assess liver stage parasite growth dynamics, HeLa cells were seeded at 40,000 cells/well (triplicate) in 96-well plates and infected 24 hours later with 15,000
PbmCherry sporozoites/well. Following a 2-hour infection period at 37°C, cells were washed, accutase treated and redistributed across eight wells of two 96-well plates (4 wells/plate per replicate) to achieve optimal imaging density [
4,
43]. Parasite development was monitored at 6, 24, and 48 hpi using an InCell Analyzer 2000 high-content imaging system (10x objective). Image analysis parameters (kernel size, sensitivity, and size threshold) were optimized in InCell Developer software to accurately segment parasites from background. Growth metrics were derived from cross-sectional area measurements, while survival rates were normalized to 6 hpi parasite counts (set as 100% viability baseline).
2.12. Quantification of Lysosome (LE) and Late Endosome Association with Plasmodium PVM
To quantify lysosome (LE) and late endosome associations with the
Plasmodium PVM, 3D rendering of UIS4 (PVM marker) and LAMP1 (lysosomal/LE marker) from confocal Z-stacks (10–15 µm; 20 parasites/condition; 3 replicates) was performed in infected HeLa cells using Imaris. Cells infected with
PbmCherry sporozoites were fixed with 4% PFA in PBS at 24 hpi, stained for UIS4, LAMP1, DAPI (DNA), and either α-tubulin or Alexa Fluor 488-phalloidin to delineate cell boundaries, then imaged with spinning disk confocal microscopy. UIS4 signals were reconstructed as isosurfaces (detail level 0.215; absolute intensity), while LAMP1-positive vesicles were detected as isospots (local contrast mode; non-expanded cells: XY = 0.4 µm, Z = 0.5 µm; expanded cells: XY = 1.5 µm, Z = 2.4 µm). PVM-proximal LAMP1-positive isospots (≤0 µm distance) were quantified using α-tubulin defined cell borders to exclude neighboring uninfected cells. Data were exported with the attached LE proportion calculated as (LAMP1-positive isospots at PVM) divided by (total LAMP1-positive isospots per infected cell) [
42].
2.13. Image Acquisition and Analysis
The live-cell imaging, expansion microscopy, and immunofluorescence (IFA) data were acquired using a Nikon W1 LIPSI spinning disk confocal system, with images processed and analyzed in FIJI (Java 1.8.0_322 (64-bit), USA), Huygens Professional deconvolution software (v3.8; Scientific Volume Imaging, Netherlands), or IMARIS 3D/4D analysis software (v10.2.0). Data were quantified and visualized using GraphPad Prism v10.4.1 (GraphPad Software, San Diego, California, USA), with statistical significance determined by one-way ANOVA and appropriate post-hoc tests (*p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001). All experiments in this study were performed in triplicate.
4. Discussion
During
Plasmodium liver infection, the parasite exhibits one of the fastest growth rates observed in eukaryotic cells, driven by parasite scavenging of host cell nutrients. Recent studies demonstrate that host lysosomal - and late endosomal vesicles fuse with the PVM during the liver stage [
11,
12,
13,
15]. While these vesicles are known to be critical for parasite development, the molecular mechanisms by which the parasite targets and manipulates host organelles to acquire nutrients remain poorly understood. Given the fundamental role of SNARE proteins in overcoming the energetic barrier to membrane fusion, our study investigated whether host SNARE machinery mediates lysosome–PVM fusion to support parasite development. The PVM is a critical interface for
Plasmodium liver stage development, uniquely adapted to recruit host-derived vesicles while minimizing parasite protein exposure to the host cytoplasm [
51]. The liver stage PVM may actively hijack host trafficking pathways through parasite-encoded SNAREs or lipid-based signals. Transcriptomic analyses revealed upregulation of
Plasmodium SNARE homologs during liver infection [
52], including Q-SNAREs (Stx1) predicted to localize to the parasite plasma membrane [
53]. However, their direct presence at the PVM remains undetected [
25,
54], suggesting alternative mechanisms where the parasite hijacks host derived SNAREs for vesicle recruitment. Using CRISPR/Cas9 KO approach and advanced microscopy tools, our study elucidates how host SNARE proteins mediate vesicular trafficking between lysosomes and the PVM, revealing a host-dependent nutrient acquisition pathway essential for parasite development.
Our data support a two-phase model of SNARE involvement at the PVM. Early recruitment of VAMP7 and Vti1B occurs shortly after invasion and likely primes the PVM for subsequent vesicle interactions, whereas later recruitment of VAMP8 and Stx7 coincides with the onset of rapid parasite growth during schizogony. The remarkable speed of this process within 30 minutes mirrors rapid host machinery co-option seen in other pathogens.
Salmonella similarly recruits LC3-positive autophagic structures within 10 minutes of infection [
55]. This model is supported by the delayed recruitment (24 hpi) of VAMP8, a v-SNARE participating in lysosomal fusion complexes with Vti1B and Stx7 coinciding with the onset of rapid parasite nuclear replication and biomass expansion [
56,
57]. The t-SNARE Stx7 shows weak PVM association at 6 hpi but becomes more prominent by 24 hpi (
Figure 1 and
Figure S1), consistent with its known role in late endosomal/lysosomal fusion when partnered with Vti1B and VAMP8. Functional genetic studies demonstrated significant redundancy among SNARE proteins [
31,
58,
59,
60]. Functional redundancy among SNARE proteins likely explains why single knockouts had limited phenotypic impact, whereas combinatorial depletion revealed pronounced defects. In particular, disruption of Vti1B-containing complexes severely impaired parasite survival and lysosome–PVM association, identifying Vti1B as a central node in this pathway. The failure to generate viable Stx7-containing combinatorial knockouts further underscores the essential role of this SNARE in core lysosomal and autophagic processes required for host cell viability.
These findings integrate well with existing models of host–parasite interactions at the PVM. During liver stage infection, parasites activate the PAAR response and recruit components such as the V-type ATPase to the PVM, processes traditionally associated with parasite elimination [
35,
61,
62]. Our data instead support a model in which
Plasmodium selectively exploits lysosomal fusion to access host-derived resources while avoiding destructive acidification. This balance is likely facilitated by PVM properties, including pores that allow nutrient exchange while dissipating proton gradients.
Lysosomes thus represent not only degradative organelles but also reservoirs of lipids, amino acids, and metabolites that can be repurposed to support parasite growth. By hijacking host SNARE-mediated trafficking, Plasmodium converts a potentially hostile compartment into a supportive source for its fast intracellular development.
Our SNARE-related findings complement these understandings, as vesicle trafficking defects in knockout cells might disrupt these processes. The potential interaction between our identified SNARE candidates and previously identified Rab GTPases provides additional mechanistic insight.
Plasmodium-essential Rab5b and Rab11a orchestrate vesicle sorting, transport, and fusion, processes critical for parasite development [
6,
63]. The convergence of SNARE-mediated fusion with Rab-regulated vesicle trafficking suggests coordinated host pathway subversion, where
Plasmodium simultaneously manipulates multiple aspects of vesicle dynamics to support intracellular growth.
In conclusion, this study demonstrates how Plasmodium exploits sequential SNARE interactions to redirect lysosomal cargo (lipids, metabolites) to the PVM. The staged nature of this process with early VAMP7/Vti1B incorporation enabling later VAMP8/Stx7-mediated fusion creates a druggable host dependency.
Future work should identify specific vesicle cargo via proteomic and metabolomic analysis and validate the implicated mechanisms in primary hepatocytes. Although no specific, side-effect-free SNARE-targeting drugs are currently available to directly validate our findings in vitro, subsequent studies must focus on using in vivo models to assess therapeutic potential. This study not only advances our understanding of malaria liver stage biology but also provides a paradigm for how intracellular pathogens manipulate conserved membrane trafficking pathways.
Author Contributions
Conceptualization, K.A., V.H., N.K., A.B., J.S., and R.C.; methodology, K.A., V.H., N.K., A.B., J.S., and R.C.; validation, K.A., V.H.; A.B., and N.K.; formal analysis, K.A.; investigation, K.A., and V.H.; resources, K.A., and V.H.; data curation, K.A; writing—original draft preparation, K.A.; writing—review and editing, K.A., V.H., N.K., A.B., J.S., and R.C.; visualization, K.A., A.B., and N.K.; supervision, V.H.; project administration, V.H.; K.A.; funding acquisition, V.H.; All authors have read and agreed to the published version of the manuscript.”
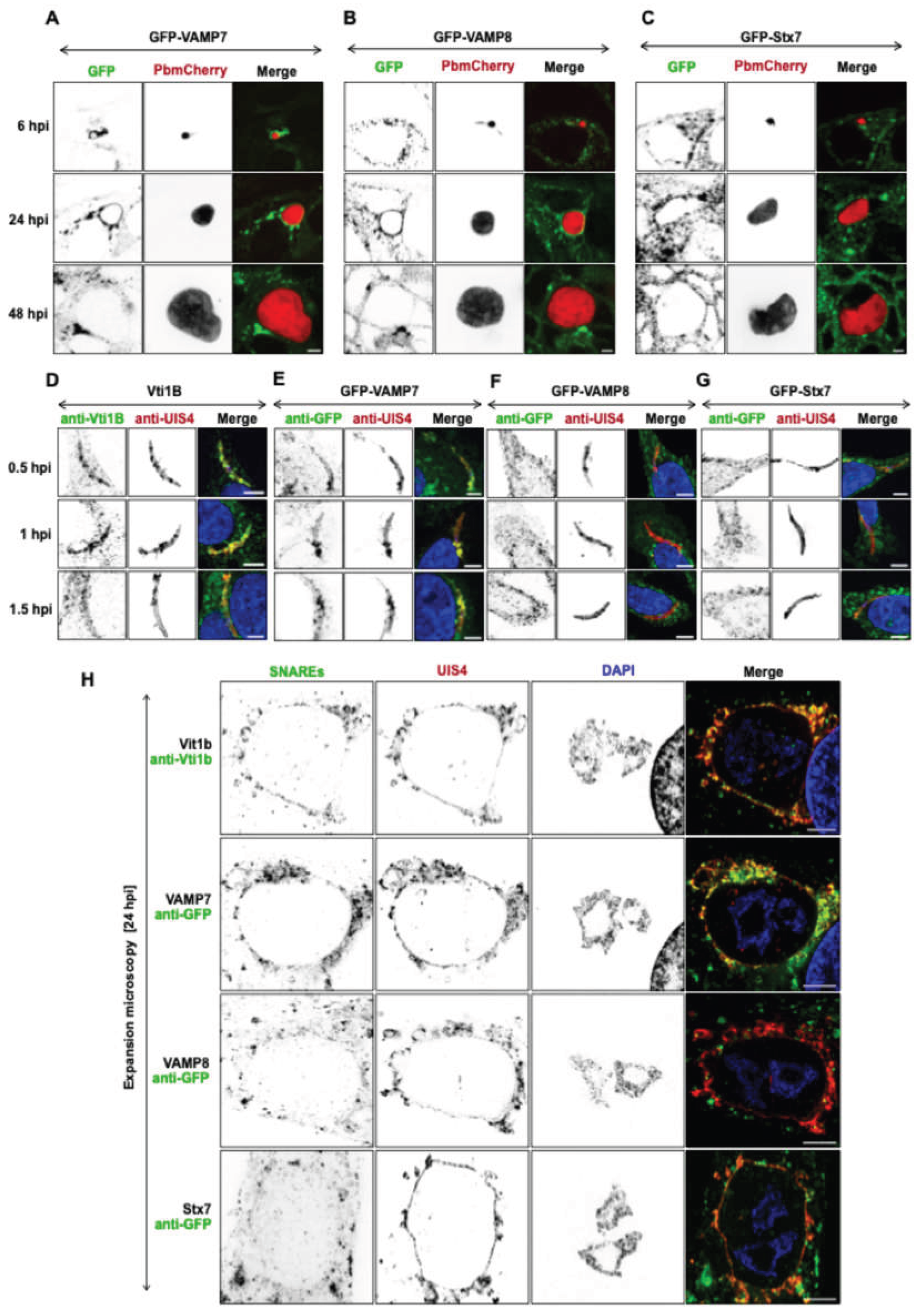
Figure 1.
Localization of host SNARE proteins at the Plasmodium PVM during liver stage infection. (A–C) Confocal live cell imaging of GFP-tagged SNARE proteins VAMP7-GFP (A), VAMP8-GFP (B), and Stx7-GFP (C) in P. berghei-infected HeLa cells expressing mCherry (parasite cytoplasm, red) at 6, 24, and 48 hpi. SNARE localization is shown in green. (D–G) Immunofluorescence analysis of Vti1B (D), VAMP7 (E), VAMP8 (F), and Stx7 (G) in HeLa cells fixed at 0.5, 1, and 1.5 hpi. SNAREs were detected with anti-GFP (green) or anti-Vti1B (green); the PVM was labeled with anti-UIS4 (red); and DAPI (blue) stained nuclei. (H) Localization of SNARE proteins (anti-Vti1B/anti-GFP in green) with the PVM marker UIS4 (red) at 24 hpi, visualized by expansion microscopy (5-fold expanded). Merged channels (yellow) highlight PVM-SNARE protein association. Nuclei were counterstained with DAPI (blue). Scale bars: 5 μm (A–G); 10 μm (H).
Figure 1.
Localization of host SNARE proteins at the Plasmodium PVM during liver stage infection. (A–C) Confocal live cell imaging of GFP-tagged SNARE proteins VAMP7-GFP (A), VAMP8-GFP (B), and Stx7-GFP (C) in P. berghei-infected HeLa cells expressing mCherry (parasite cytoplasm, red) at 6, 24, and 48 hpi. SNARE localization is shown in green. (D–G) Immunofluorescence analysis of Vti1B (D), VAMP7 (E), VAMP8 (F), and Stx7 (G) in HeLa cells fixed at 0.5, 1, and 1.5 hpi. SNAREs were detected with anti-GFP (green) or anti-Vti1B (green); the PVM was labeled with anti-UIS4 (red); and DAPI (blue) stained nuclei. (H) Localization of SNARE proteins (anti-Vti1B/anti-GFP in green) with the PVM marker UIS4 (red) at 24 hpi, visualized by expansion microscopy (5-fold expanded). Merged channels (yellow) highlight PVM-SNARE protein association. Nuclei were counterstained with DAPI (blue). Scale bars: 5 μm (A–G); 10 μm (H).
Figure 2.
Role of SNARE proteins in LAMP1-positive lysosomal vesicle trafficking. (A) Expansion microscopy (VAMP7, VAMP8, and Stx7) and standard IFA (Vti1B) confocal images showing colocalization of the SNARE proteins (green) with LAMP1 (red). Merged channels (yellow) highlight SNARE-LAMP1 positive vesicles association. Nuclei were stained with DAPI (blue). Scale bars: 10 µm. (B) Quantification of SNARE-LAMP1 colocalization using Pearson’s correlation coefficient (B): VAMP7 (r = 0.7), VAMP8 (r = 0.6), Vti1B (r = 0.9), Stx7 (r = 0.5). Each dot represents one cell; horizontal lines indicate mean values. (C) Immunoblot validation of SNARE knockout (KO) cell lines (VAMP7KO, VAMP8KO, Vti1BKO, Stx7KO) showing protein molecular weights. GAPDH (~37 kDa) and ⍺-tubulin (~55 kDa) served as a loading control. (D) Quantification of LAMP1-positive vesicles using 3D and 4D IMARIS software over time in WT cells, with an average of 1073 LAMP1-positive vesicles per cell at 24-hour post-seeding. Data represent mean ± SD; n = 25 cells (5 per experiment) per time point; ns = not significant. (E) Quantification of LAMP1-positive vesicles per cell at 24 H in WT (1152) vs. SNARE KO cell lines: VAMP7KO (817), VAMP8KO (845), Vti1BKO (722), Stx7KO (768). Data represent mean ± SD; n = 15 cells per cell lines. Statistical significance was determined by one-way ANOVA with Dunnett’s multiple comparisons test (*p < 0.05, **p < 0.01, ***p < 0.001; ns, not significant).
Figure 2.
Role of SNARE proteins in LAMP1-positive lysosomal vesicle trafficking. (A) Expansion microscopy (VAMP7, VAMP8, and Stx7) and standard IFA (Vti1B) confocal images showing colocalization of the SNARE proteins (green) with LAMP1 (red). Merged channels (yellow) highlight SNARE-LAMP1 positive vesicles association. Nuclei were stained with DAPI (blue). Scale bars: 10 µm. (B) Quantification of SNARE-LAMP1 colocalization using Pearson’s correlation coefficient (B): VAMP7 (r = 0.7), VAMP8 (r = 0.6), Vti1B (r = 0.9), Stx7 (r = 0.5). Each dot represents one cell; horizontal lines indicate mean values. (C) Immunoblot validation of SNARE knockout (KO) cell lines (VAMP7KO, VAMP8KO, Vti1BKO, Stx7KO) showing protein molecular weights. GAPDH (~37 kDa) and ⍺-tubulin (~55 kDa) served as a loading control. (D) Quantification of LAMP1-positive vesicles using 3D and 4D IMARIS software over time in WT cells, with an average of 1073 LAMP1-positive vesicles per cell at 24-hour post-seeding. Data represent mean ± SD; n = 25 cells (5 per experiment) per time point; ns = not significant. (E) Quantification of LAMP1-positive vesicles per cell at 24 H in WT (1152) vs. SNARE KO cell lines: VAMP7KO (817), VAMP8KO (845), Vti1BKO (722), Stx7KO (768). Data represent mean ± SD; n = 15 cells per cell lines. Statistical significance was determined by one-way ANOVA with Dunnett’s multiple comparisons test (*p < 0.05, **p < 0.01, ***p < 0.001; ns, not significant).
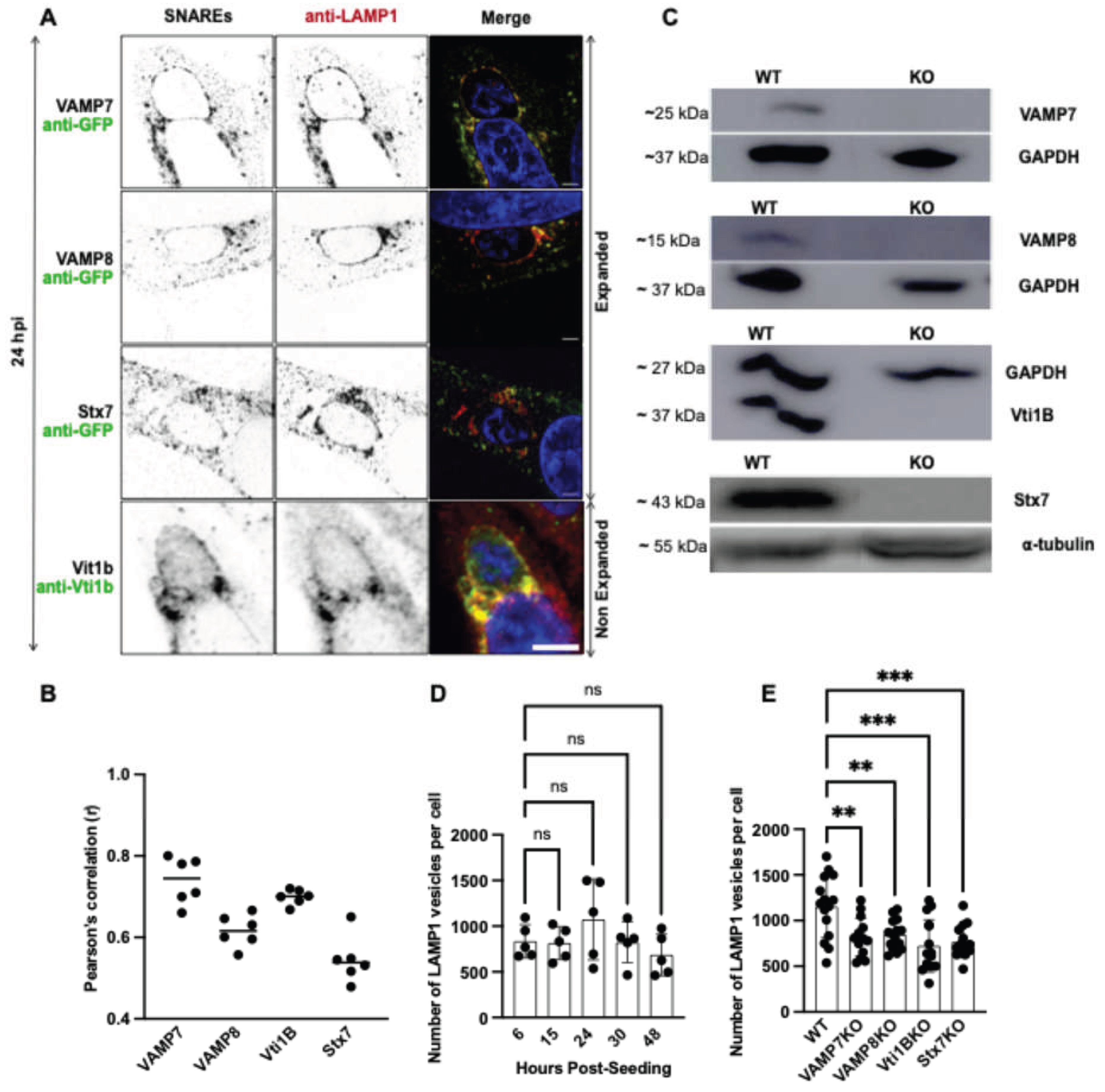
Figure 3.
Role of SNARE candidates in P. berghei liver stage infection and development in HeLa cells. (A–D) Infection rates at 6 hpi in WT HeLa cells and SNARE knockout (KO) or rescue (Ex) lines infected with P. berghei sporozoites. No significant difference was observed in VAMP7KO cells (9.3%) (A), whereas infection was significantly reduced in VAMP8KO (8.3%) (B), Vti1BKO (6.5%) (C), and Stx7KO (5.2%) (D) cells compared to WT (10%). Over-expression with the respective SNARE proteins partially restored infection rates. (E–H) Parasite size quantified at 48 hpi in WT and KO/Ex cell lines. No significant differences in parasite size were observed in the SNARE KO or Ex lines except of Vti1B. (I–L) Parasite survival rate (6-48 hpi) in WT and KO/Ex lines. Parasite survival was significantly reduced in VAMP7KO (I), VAMP8KO (J), and Stx7KO (L) cells, but not in Vti1BKO cells (K), indicating that these SNAREs are critical for parasite survival during liver stage development. Data are presented as mean ± SD. Statistical significance was determined by one-way ANOVA with Dunnett’s multiple comparisons, and appropriate post-hoc test, (C, G, K) Students t-Test. *p < 0.05, **p < 0.01, ***p < 0.001; ns, not significant.
Figure 3.
Role of SNARE candidates in P. berghei liver stage infection and development in HeLa cells. (A–D) Infection rates at 6 hpi in WT HeLa cells and SNARE knockout (KO) or rescue (Ex) lines infected with P. berghei sporozoites. No significant difference was observed in VAMP7KO cells (9.3%) (A), whereas infection was significantly reduced in VAMP8KO (8.3%) (B), Vti1BKO (6.5%) (C), and Stx7KO (5.2%) (D) cells compared to WT (10%). Over-expression with the respective SNARE proteins partially restored infection rates. (E–H) Parasite size quantified at 48 hpi in WT and KO/Ex cell lines. No significant differences in parasite size were observed in the SNARE KO or Ex lines except of Vti1B. (I–L) Parasite survival rate (6-48 hpi) in WT and KO/Ex lines. Parasite survival was significantly reduced in VAMP7KO (I), VAMP8KO (J), and Stx7KO (L) cells, but not in Vti1BKO cells (K), indicating that these SNAREs are critical for parasite survival during liver stage development. Data are presented as mean ± SD. Statistical significance was determined by one-way ANOVA with Dunnett’s multiple comparisons, and appropriate post-hoc test, (C, G, K) Students t-Test. *p < 0.05, **p < 0.01, ***p < 0.001; ns, not significant.
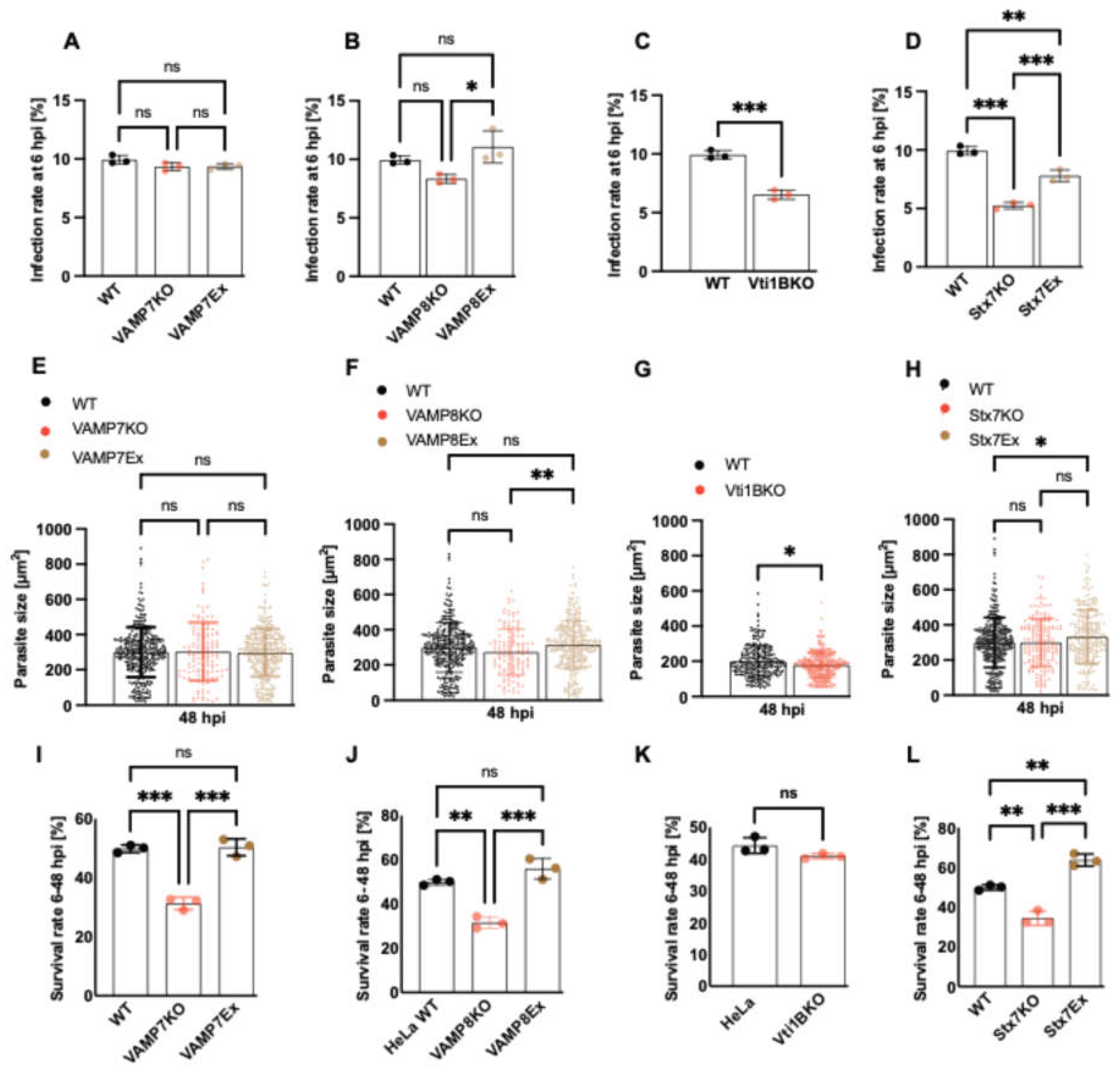
Figure 4.
Impact of SNARE depletion on LAMP1-positive vesicle association with the P. berghei PVM and intracellular distribution. (A) Representative confocal microscopy images of HeLa cells infected with P. berghei sporozoites and fixed with 4% paraformaldehyde (PFA) at 24 hpi. Immunofluorescence staining was performed using anti-LAMP1 (lysosomes, green) and anti-UIS4 (PVM, red). Nuclei were counterstained with DAPI (blue). Note the altered vesicle distribution in KO cells, with LAMP1-positive vesicles showing perinuclear clustering (arrows in A) compared to the dispersed pattern in WT cells. Z-stack images were processed using IMARIS software for 3D/4D reconstruction: LAMP1-positive vesicles were quantified as isospots, while the PVM was rendered as an isosurface. The percentage of LAMP1 vesicles associated with the PVM was calculated based on isospots located within 0-0.5 µm of the PVM isosurface. (B-E) Quantification of LAMP1-positive vesicles in contact with the PVM (normalized to total LAMP1 vesicles per cell) at 24 and 48 hpi. No significant differences were observed between wild-type (WT) and VAMP7KO (B), VAMP8KO (C), Vti1BKO (D), or Stx7KO (E) cells.. Scale bar: 5 µm. Data represent mean ± SD; n = 25 cells per condition. Statistical significance between time points and conditions was determined by one-way ANOVA with Dunnett's multiple comparisons test *p < 0.05, **p < 0.01, ***p < 0.001; ns, not significant.
Figure 4.
Impact of SNARE depletion on LAMP1-positive vesicle association with the P. berghei PVM and intracellular distribution. (A) Representative confocal microscopy images of HeLa cells infected with P. berghei sporozoites and fixed with 4% paraformaldehyde (PFA) at 24 hpi. Immunofluorescence staining was performed using anti-LAMP1 (lysosomes, green) and anti-UIS4 (PVM, red). Nuclei were counterstained with DAPI (blue). Note the altered vesicle distribution in KO cells, with LAMP1-positive vesicles showing perinuclear clustering (arrows in A) compared to the dispersed pattern in WT cells. Z-stack images were processed using IMARIS software for 3D/4D reconstruction: LAMP1-positive vesicles were quantified as isospots, while the PVM was rendered as an isosurface. The percentage of LAMP1 vesicles associated with the PVM was calculated based on isospots located within 0-0.5 µm of the PVM isosurface. (B-E) Quantification of LAMP1-positive vesicles in contact with the PVM (normalized to total LAMP1 vesicles per cell) at 24 and 48 hpi. No significant differences were observed between wild-type (WT) and VAMP7KO (B), VAMP8KO (C), Vti1BKO (D), or Stx7KO (E) cells.. Scale bar: 5 µm. Data represent mean ± SD; n = 25 cells per condition. Statistical significance between time points and conditions was determined by one-way ANOVA with Dunnett's multiple comparisons test *p < 0.05, **p < 0.01, ***p < 0.001; ns, not significant.
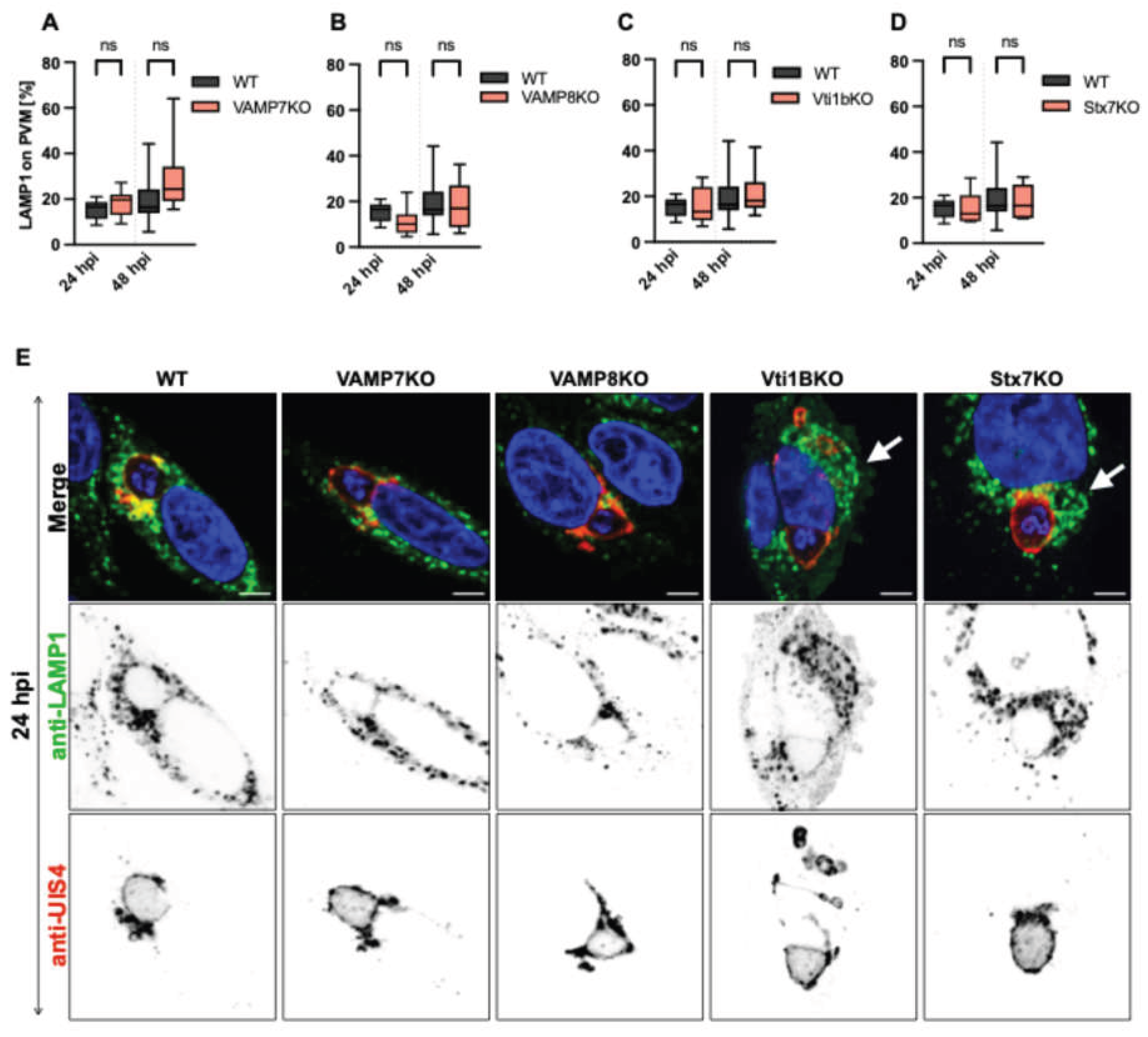
Figure 5.
Double knockout of SNARE candidates impairs P. berghei liver stage development. (A-B) Generation and validation of VAMP7-VAMP8 and VAMP7-Vti1B 2KO HeLa cell lines. (A) Western blot analysis confirms efficient depletion of VAMP7 and VAMP8 proteins in VAMP7-VAMP8 2KO cells; (B) Validation of Vti1B knockout in VAMP7KO cells. Due to their similar molecular weights (~23 - 27 kDa), confirming the VAMP7-Vti1B 2KO by western blot was challenging. Instead, a genetic analysis was performed using TIDE (Tracking of Indels by Decomposition). The TIDE plot confirms successful Vti1B knockout in VAMP7KO cells, with a total editing efficiency of 86.1%. (C) Infection rates of P. berghei sporozoites at 6 hours post-infection (hpi). Both, 2KO lines showed significantly reduced infection (VAMP7-VAMP8 2KO: 9%; VAMP7-Vti1B 2KO: 7.8%) compared to wild-type (WT) cells (10.5%). (D) Parasite survival (6-48 hpi,) was significantly decreased in VAMP7-Vti1B 2KO cells (30%) compared to WT (51.2%), while VAMP7-VAMP8 2KO cells showed a non-significant reduction (47.2%). Each experiment was conducted in triplicate. (E) Parasite size quantification at 48 hpi. Both, 2KO lines exhibited impaired growth: VAMP7-VAMP8 2KO: 66 µm² (24 hpi), 140 µm² (48 hpi); VAMP7-Vti1B 2KO: 67 µm² (24 hpi), 150 µm² (48 hpi) compared to WT: 73 µm² (24 hpi), 203 µm² (48 hpi). Each dot represent a single measured parasite (C-E), WT: black bots, VAMP7-VAMP8 2KO: red dots and VAMP7-Vti1B: magenta dots. (F) Confocal microscopy images of infected cells at 24 hpi stained with anti-UIS4 (PVM, red) and anti-LAMP1 (lysosomes, green). VAMP7-VAMP8 2KO cells showed reduced LAMP1 signal, while VAMP7-Vti1B 2KO cells exhibited perinuclear accumulation of LAMP1-positive vesicles near parasites (arrow). Nuclei were counterstained with DAPI (blue). Scale bar: 5 µm. (G) Quantification of LAMP1-positive vesicle association with the PVM at 24 hpi using 3D/4D IMARIS. Both, 2KO lines showed significantly reduced attachment: VAMP7-VAMP8 2KO: 18%; VAMP7-Vti1B 2KO: 9.1% compared to WT: 36%. Data represent mean ± SD. Statistical significance was determined by one-way ANOVA with appropriate post-hoc tests: *p < 0.05, **p < 0.01, ***p < 0.001; ns, not significant.
Figure 5.
Double knockout of SNARE candidates impairs P. berghei liver stage development. (A-B) Generation and validation of VAMP7-VAMP8 and VAMP7-Vti1B 2KO HeLa cell lines. (A) Western blot analysis confirms efficient depletion of VAMP7 and VAMP8 proteins in VAMP7-VAMP8 2KO cells; (B) Validation of Vti1B knockout in VAMP7KO cells. Due to their similar molecular weights (~23 - 27 kDa), confirming the VAMP7-Vti1B 2KO by western blot was challenging. Instead, a genetic analysis was performed using TIDE (Tracking of Indels by Decomposition). The TIDE plot confirms successful Vti1B knockout in VAMP7KO cells, with a total editing efficiency of 86.1%. (C) Infection rates of P. berghei sporozoites at 6 hours post-infection (hpi). Both, 2KO lines showed significantly reduced infection (VAMP7-VAMP8 2KO: 9%; VAMP7-Vti1B 2KO: 7.8%) compared to wild-type (WT) cells (10.5%). (D) Parasite survival (6-48 hpi,) was significantly decreased in VAMP7-Vti1B 2KO cells (30%) compared to WT (51.2%), while VAMP7-VAMP8 2KO cells showed a non-significant reduction (47.2%). Each experiment was conducted in triplicate. (E) Parasite size quantification at 48 hpi. Both, 2KO lines exhibited impaired growth: VAMP7-VAMP8 2KO: 66 µm² (24 hpi), 140 µm² (48 hpi); VAMP7-Vti1B 2KO: 67 µm² (24 hpi), 150 µm² (48 hpi) compared to WT: 73 µm² (24 hpi), 203 µm² (48 hpi). Each dot represent a single measured parasite (C-E), WT: black bots, VAMP7-VAMP8 2KO: red dots and VAMP7-Vti1B: magenta dots. (F) Confocal microscopy images of infected cells at 24 hpi stained with anti-UIS4 (PVM, red) and anti-LAMP1 (lysosomes, green). VAMP7-VAMP8 2KO cells showed reduced LAMP1 signal, while VAMP7-Vti1B 2KO cells exhibited perinuclear accumulation of LAMP1-positive vesicles near parasites (arrow). Nuclei were counterstained with DAPI (blue). Scale bar: 5 µm. (G) Quantification of LAMP1-positive vesicle association with the PVM at 24 hpi using 3D/4D IMARIS. Both, 2KO lines showed significantly reduced attachment: VAMP7-VAMP8 2KO: 18%; VAMP7-Vti1B 2KO: 9.1% compared to WT: 36%. Data represent mean ± SD. Statistical significance was determined by one-way ANOVA with appropriate post-hoc tests: *p < 0.05, **p < 0.01, ***p < 0.001; ns, not significant.
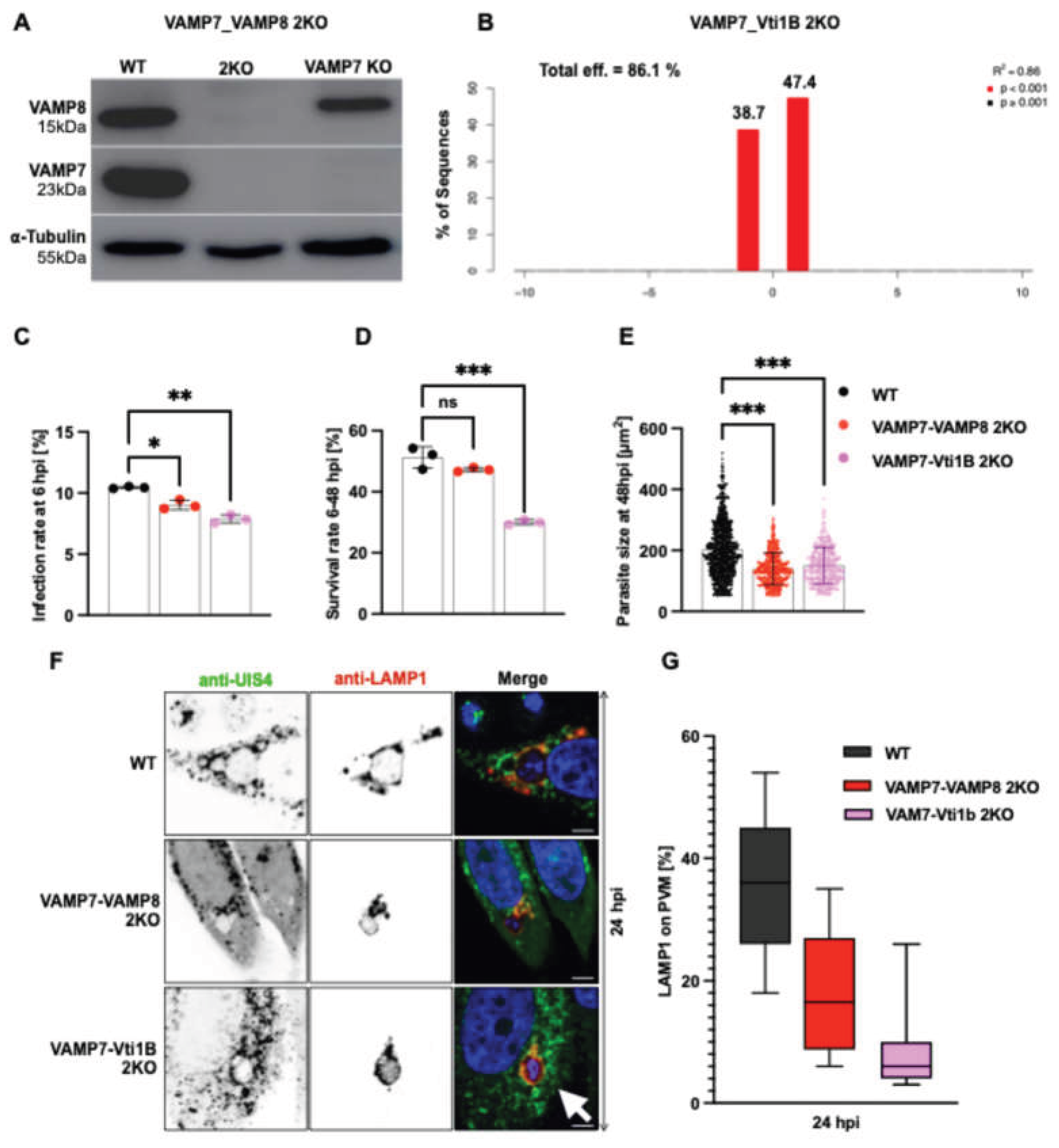
Figure 6.
Triple SNARE knockout exacerbates impairment of P. berghei liver stage development and lysosome-PVM interaction. (A) Western blot validation of VAMP7-VAMP8-Vti1B triple knockout (3KO) HeLa cells. Efficient depletion of all three SNARE proteins in the 3KO cell line is shown compared to wild-type (WT) controls. Vti1B (~27 kDa) deletion was first confirmed in a VAMP7-VAMP8 double knockout (2KO) background, with α-Tubulin and GAPDH serving as loading controls. The subsequent absence of VAMP7 (~23-25 kDa) and VAMP8 (~15 kDa) in the 3KO line was also verified, using GAPDH as a loading control, prior to further experiments. (B) Infection rates at 6 hpi showing significant reduction in VAMP7-Vti1B 2KO and VAMP7-Vti1B-VAMP8 3KO compared to the WT. No significant difference was observed between 2KO and 3KO. (C) Parasite survival (6-48 hpi) was significantly decreased in both 2KO and 3KO compared to the WT, with no additional significant effect in 3KO versus 2KO. (D) Parasite size quantification at 48 hpi (significant impairment in both genotypes). No significant difference was observed between 2KO and 3KO at any timepoint. (E) Immunofluorescence confocal images at 24 hpi showing LAMP1 (lysosomes, green), UIS4 (PVM, red), and DAPI (nuclei, blue). Note perinuclear LAMP1 accumulation in 2KO and 3KO versus dispersed distribution in WT (Arrow). (F-H) Fluorescence intensity profiles across infected cells (PVM=red line and LAMP1 signal = green lines) demonstrating strong LAMP1-UIS4 colocalization at PVM in WT (F, red arrow), partial colocalization in VAMP7-Vti1B 2KO, and (G) Nearly absent colocalization in 3KO (H, red arrow). Lysosome accumulation within the cytoplasm was also visible as broadening of the curves (G-H). Scale bars: 5 µm (B-D). Data represent mean ± SD; statistical significance (one-way ANOVA with Dunnett's test): *p < 0.05, **p < 0.01, ***p < 0.001; ns, not significant.
Figure 6.
Triple SNARE knockout exacerbates impairment of P. berghei liver stage development and lysosome-PVM interaction. (A) Western blot validation of VAMP7-VAMP8-Vti1B triple knockout (3KO) HeLa cells. Efficient depletion of all three SNARE proteins in the 3KO cell line is shown compared to wild-type (WT) controls. Vti1B (~27 kDa) deletion was first confirmed in a VAMP7-VAMP8 double knockout (2KO) background, with α-Tubulin and GAPDH serving as loading controls. The subsequent absence of VAMP7 (~23-25 kDa) and VAMP8 (~15 kDa) in the 3KO line was also verified, using GAPDH as a loading control, prior to further experiments. (B) Infection rates at 6 hpi showing significant reduction in VAMP7-Vti1B 2KO and VAMP7-Vti1B-VAMP8 3KO compared to the WT. No significant difference was observed between 2KO and 3KO. (C) Parasite survival (6-48 hpi) was significantly decreased in both 2KO and 3KO compared to the WT, with no additional significant effect in 3KO versus 2KO. (D) Parasite size quantification at 48 hpi (significant impairment in both genotypes). No significant difference was observed between 2KO and 3KO at any timepoint. (E) Immunofluorescence confocal images at 24 hpi showing LAMP1 (lysosomes, green), UIS4 (PVM, red), and DAPI (nuclei, blue). Note perinuclear LAMP1 accumulation in 2KO and 3KO versus dispersed distribution in WT (Arrow). (F-H) Fluorescence intensity profiles across infected cells (PVM=red line and LAMP1 signal = green lines) demonstrating strong LAMP1-UIS4 colocalization at PVM in WT (F, red arrow), partial colocalization in VAMP7-Vti1B 2KO, and (G) Nearly absent colocalization in 3KO (H, red arrow). Lysosome accumulation within the cytoplasm was also visible as broadening of the curves (G-H). Scale bars: 5 µm (B-D). Data represent mean ± SD; statistical significance (one-way ANOVA with Dunnett's test): *p < 0.05, **p < 0.01, ***p < 0.001; ns, not significant.
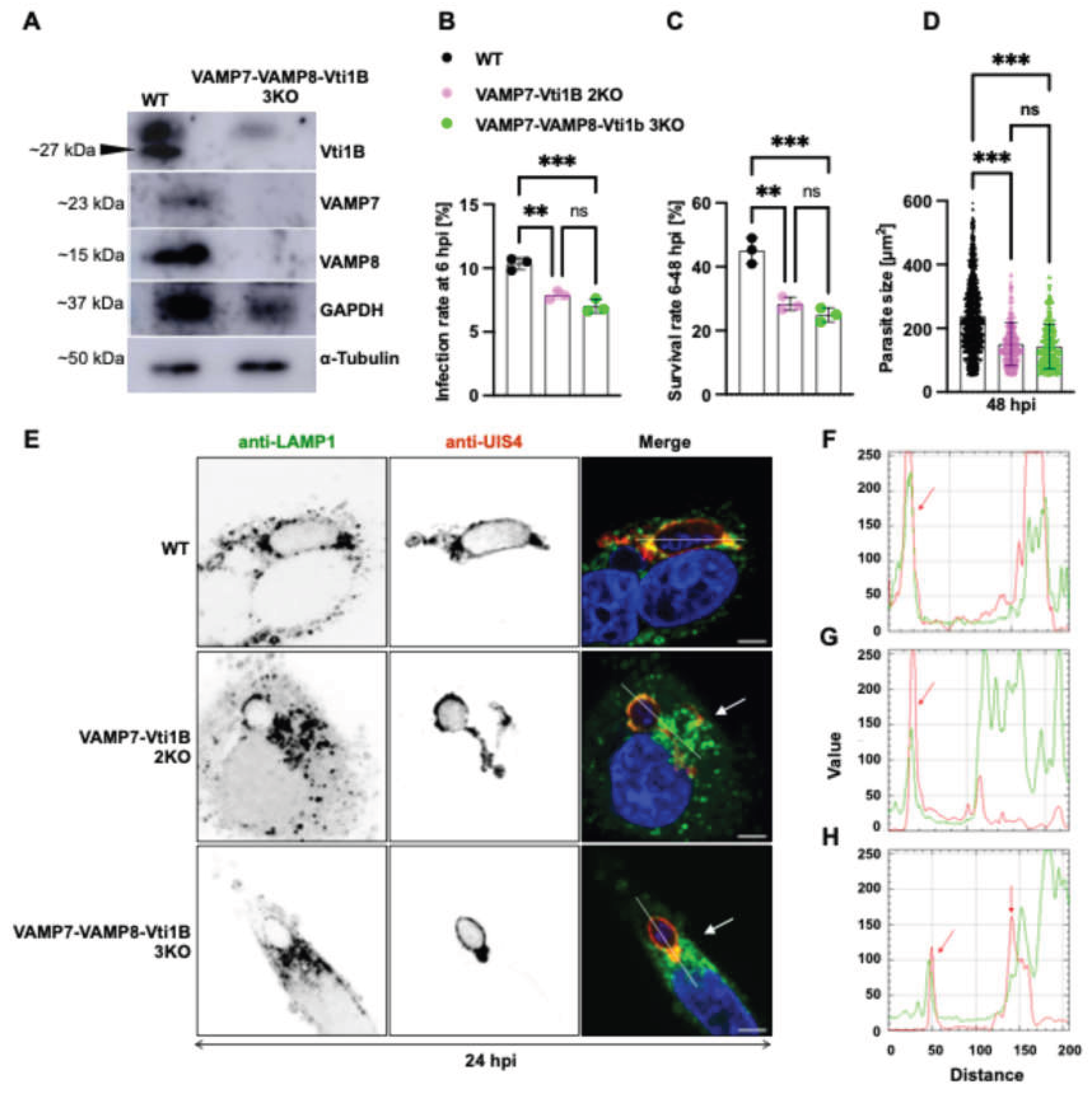
Figure 7.
SNARE-mediated lysosome-PVM fusion supports Plasmodium liver stage development. This schematic illustrates how host SNARE proteins mediate membrane fusion between host lysosomal vesicles and the Plasmodium PVM, facilitating parasite development in hepatocytes. (A) In uninfected hepatocytes, cytosolic material is degraded through the endolysosomal and autophagic pathways. Fusion events between phagophores, autophagosomes, late endosomes, and lysosomes are orchestrated by specific v-SNAREs (vesicle SNAREs, shown in orange) and t-SNAREs (target membrane SNAREs, shown in green). LAMP1 serves as a lysosomal marker. (B–D) Upon infection with P. berghei sporozoites expressing cytosolic mCherry, the parasite resides within a PVM and undergoes liver stage development. (B) At early stages (6 hpi), host v-SNARE VAMP7 is recruited to the PVM formed through invagination of the host plasma membrane, bringing with it the associated t-SNARE Vti1B. (C) As the infection progresses (24 hpi), additional host SNAREs including VAMP8 (v) and Stx7 (t) are found on LAMP1 positive lysosomal vesicles that dock at the PVM, likely facilitating membrane fusion. This delivers lysosome-associated cargo (cytosolic material) to support schizogony (D). Together, these observations support a model where Plasmodium hijacks the host's SNARE-dependent lysosomal trafficking machinery to import host-derived nutrients across the PVM. This process is essential for the parasite's intracellular growth and successful liver stage development. v = v-SNARE; t = t-SNARE. Created with BioRender.com.
Figure 7.
SNARE-mediated lysosome-PVM fusion supports Plasmodium liver stage development. This schematic illustrates how host SNARE proteins mediate membrane fusion between host lysosomal vesicles and the Plasmodium PVM, facilitating parasite development in hepatocytes. (A) In uninfected hepatocytes, cytosolic material is degraded through the endolysosomal and autophagic pathways. Fusion events between phagophores, autophagosomes, late endosomes, and lysosomes are orchestrated by specific v-SNAREs (vesicle SNAREs, shown in orange) and t-SNAREs (target membrane SNAREs, shown in green). LAMP1 serves as a lysosomal marker. (B–D) Upon infection with P. berghei sporozoites expressing cytosolic mCherry, the parasite resides within a PVM and undergoes liver stage development. (B) At early stages (6 hpi), host v-SNARE VAMP7 is recruited to the PVM formed through invagination of the host plasma membrane, bringing with it the associated t-SNARE Vti1B. (C) As the infection progresses (24 hpi), additional host SNAREs including VAMP8 (v) and Stx7 (t) are found on LAMP1 positive lysosomal vesicles that dock at the PVM, likely facilitating membrane fusion. This delivers lysosome-associated cargo (cytosolic material) to support schizogony (D). Together, these observations support a model where Plasmodium hijacks the host's SNARE-dependent lysosomal trafficking machinery to import host-derived nutrients across the PVM. This process is essential for the parasite's intracellular growth and successful liver stage development. v = v-SNARE; t = t-SNARE. Created with BioRender.com.
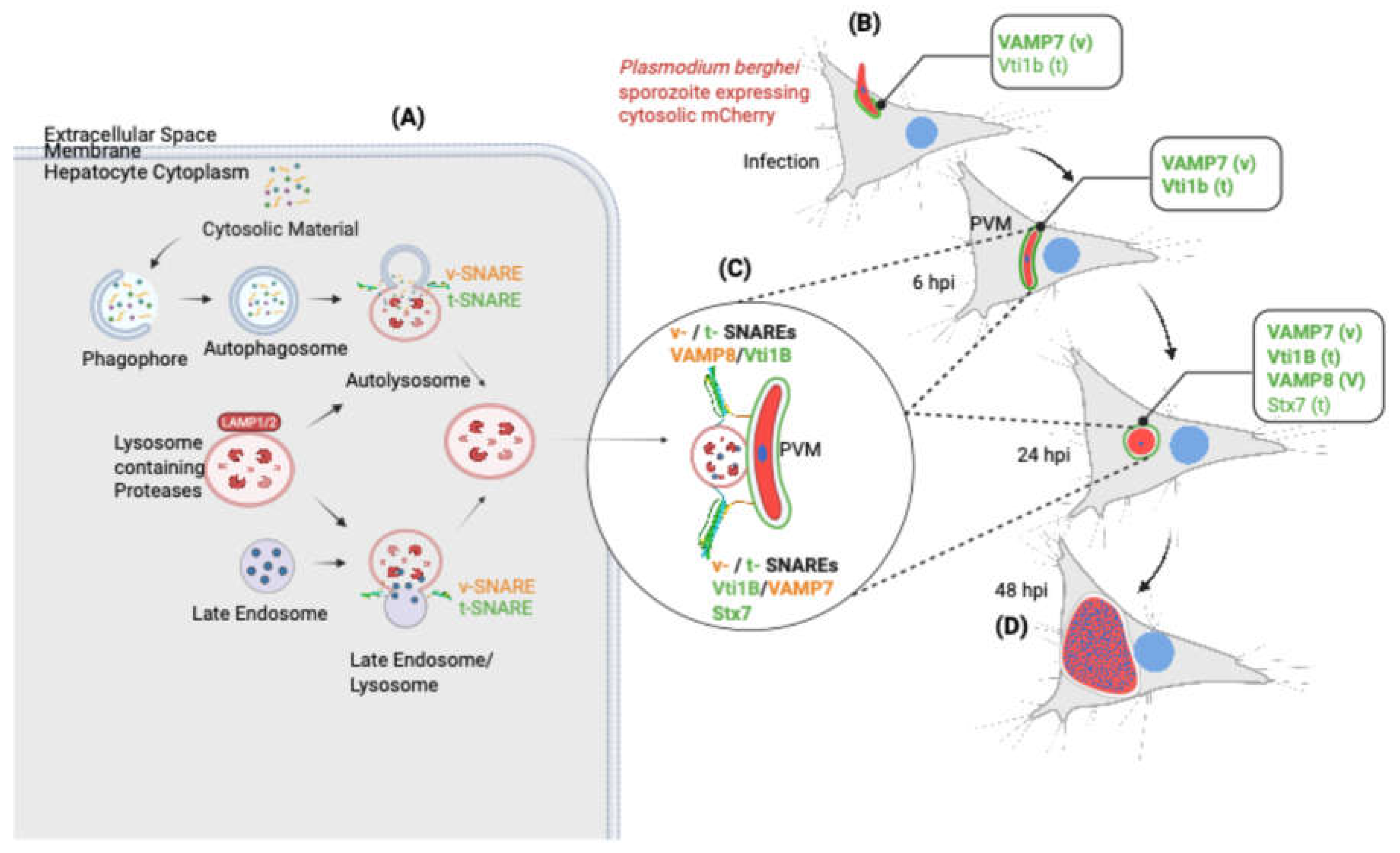
Table 1.
SNARE protein localization at the PVM during Liver stage Infection.
Table 1.
SNARE protein localization at the PVM during Liver stage Infection.
SNARE
Protein |
Subcellular localization [24] |
Construct |
(*) Construct origin |
(†) Association
with PVM |
| SNAP29 |
Cytosolic, PM, EE, RE |
GFP-SNAP29 |
Lab |
- |
| Flag-SNAP29 |
[44] |
| Stx3 |
PM, Phagosomes |
Antibody |
Lab |
+ |
| Stx6 |
PM, EE, RE, LE, Golgi |
GFP-Stx6 |
Lab |
+ |
| 3xFlag-Stx6 |
[45] |
| Stx7 |
PM, EE, RE, LE, Lysosomes |
Stx7-GFP |
Lab |
++ |
| Stx7-Myc |
Gift by J. Brummel |
| Stx8 |
EE, RE, LE, Lysosomes |
GFP-Stx8 |
Lab |
+ |
| Stx17 |
Phagosomes, Autophagosomes |
GFP-Stx17 |
Lab |
+ |
| Flag-Stx17 |
[44] |
| Stx11 |
PM, EE, RE, LE,
Lysosomes, TGN |
GFP-Stx11 |
Lab |
- |
| Vti1b |
EE, LE, Lysosomes, Golgi |
antibody |
Lab |
+++ |
| Flag-Vti1b |
[44] |
| VAMP3 |
EE, RE, Phagosomes |
GFP-VAMP3 |
Lab |
- |
| VAMP7 |
PM, EE, LE, Lysosomes, Autophagosomes |
GFP-VAMP7 |
Lab
[46] |
+++ |
| VAMP8 |
PM, EE, RE, LE, Lysosomes, TGN |
VAMP8-GFP |
Lab |
+++ |
| VAMP8-mCherry |
[47] |