Submitted:
09 February 2026
Posted:
10 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
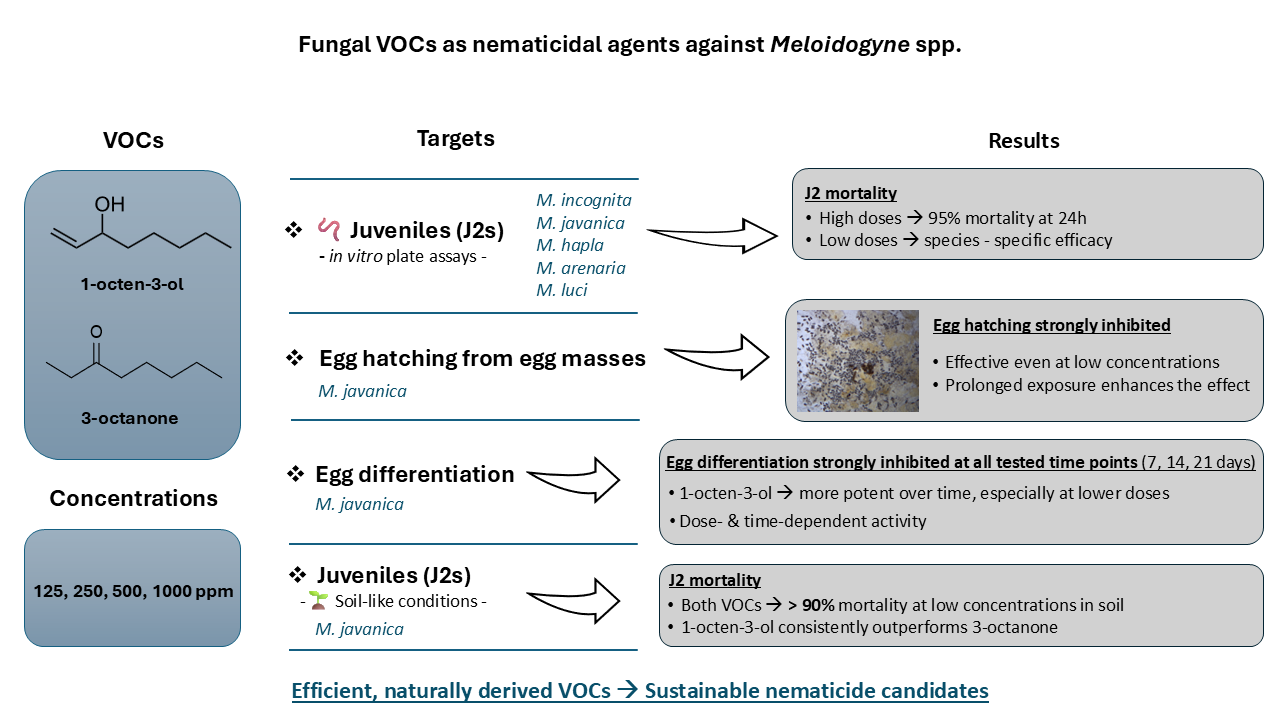
2. Results
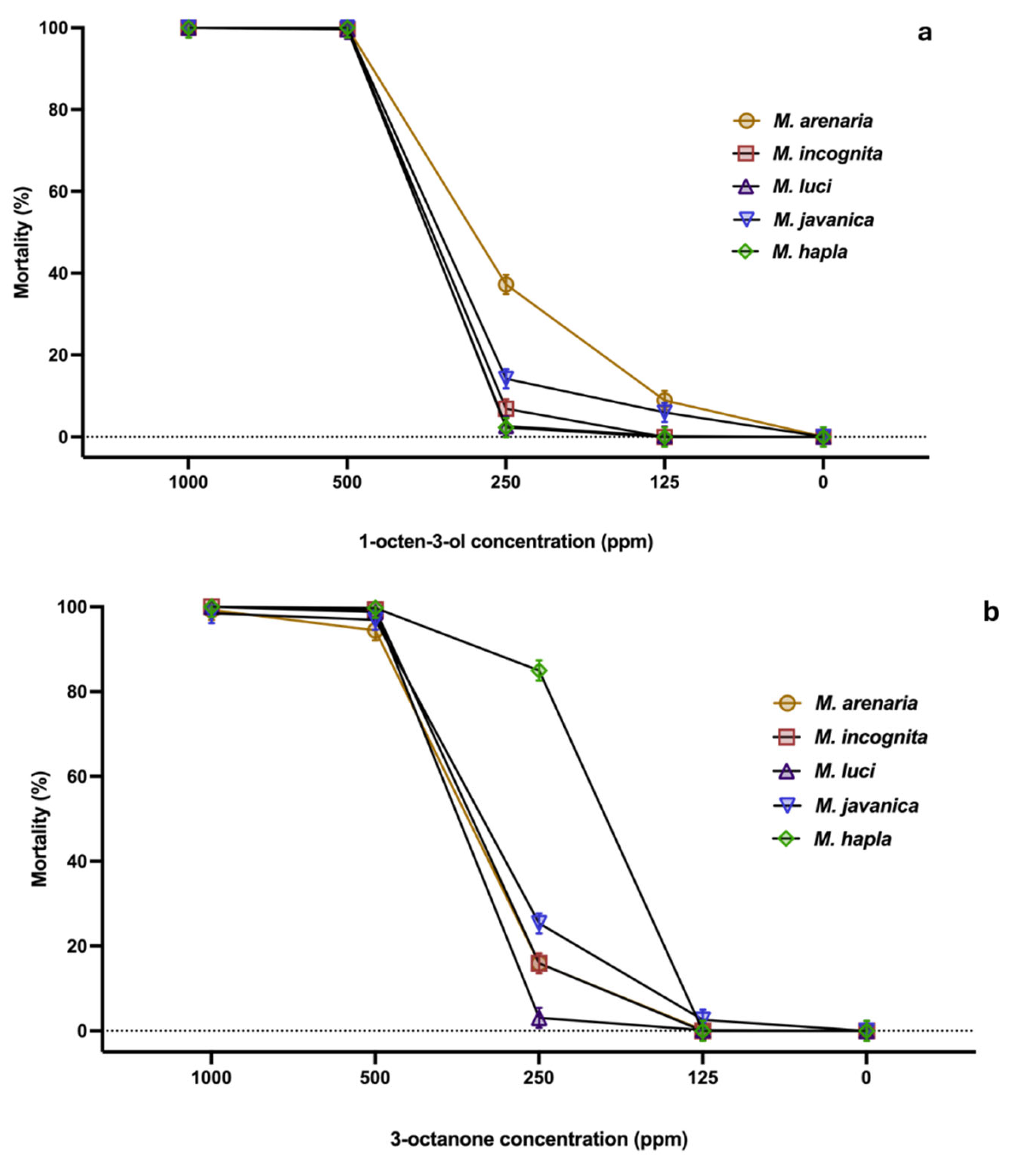
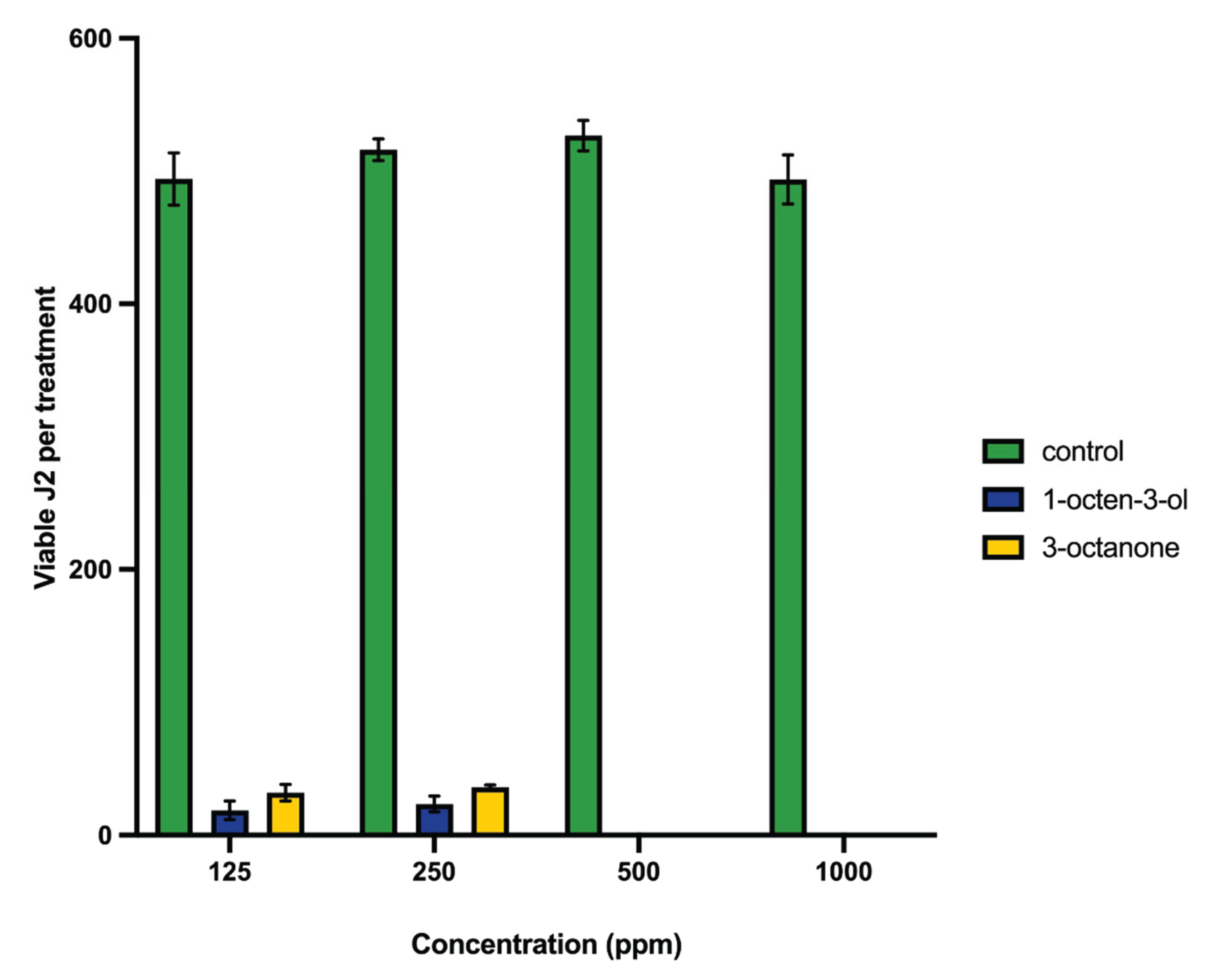
2.1. Nematode Mortality Bioassays
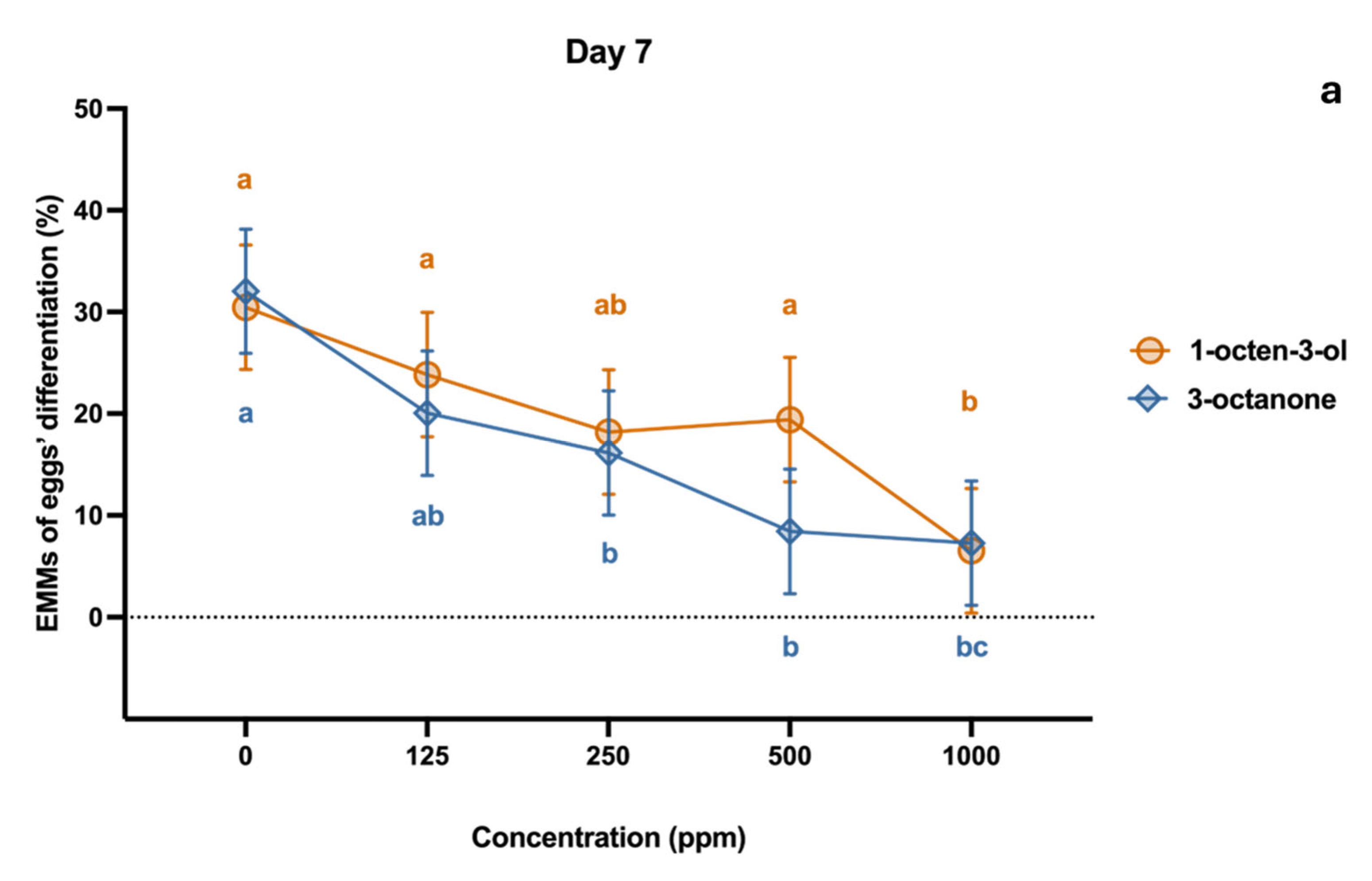
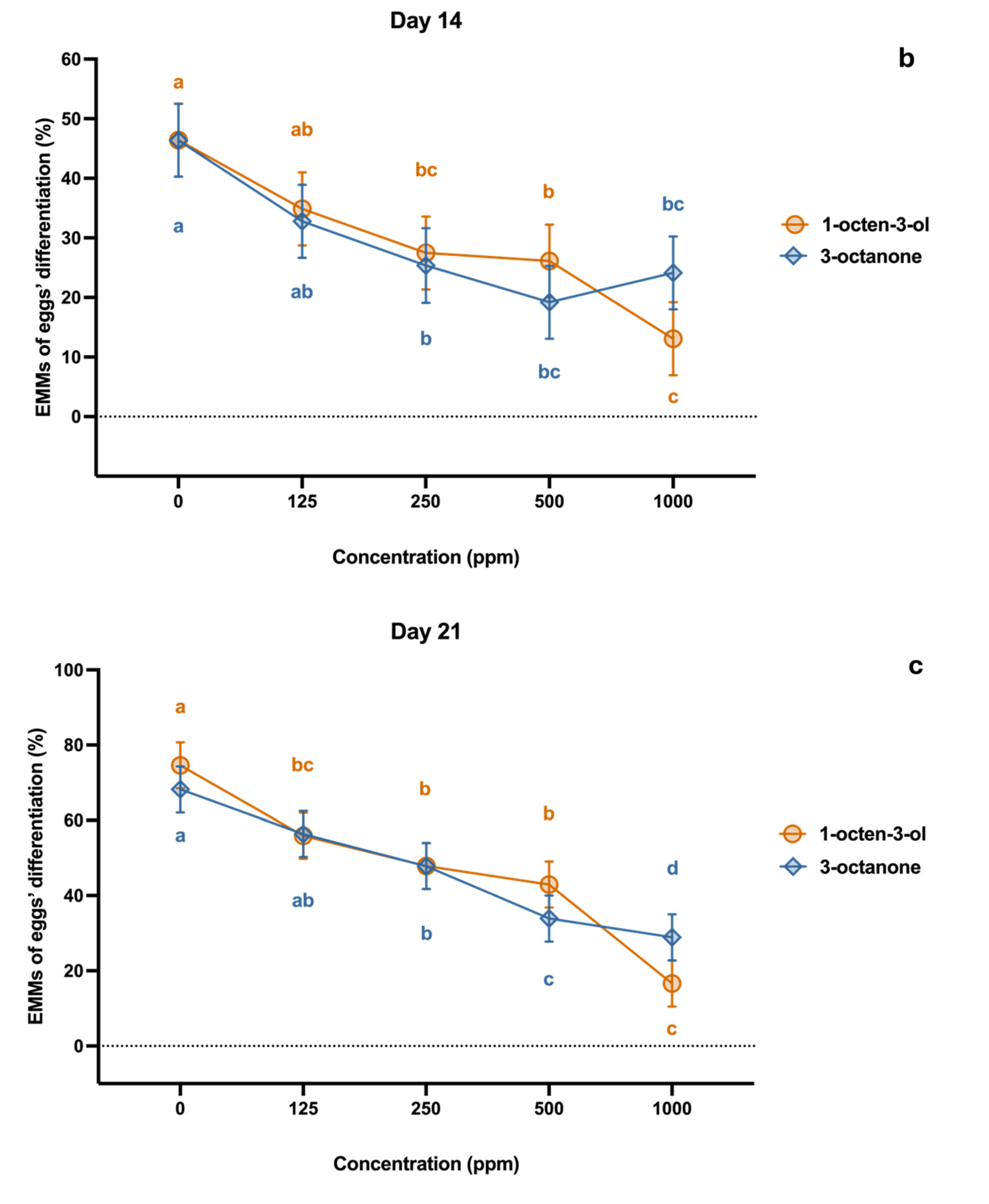
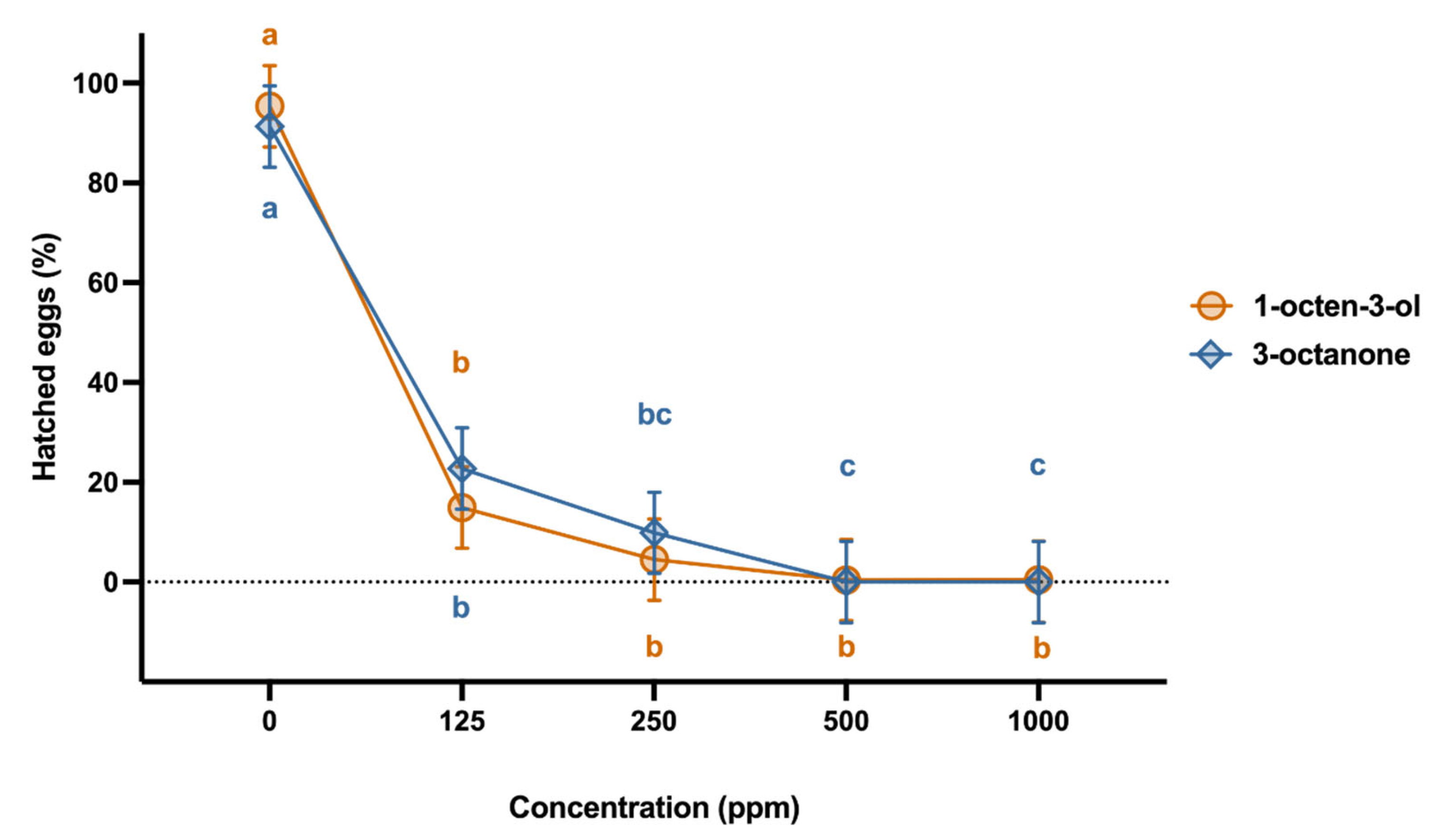
2.2. Effect on Egg Development
2.3. Effect on Egg Hatching from Egg Masses
2.4. Soil Survival Assays
3. Discussion
4. Materials and Methods
4.1. Nematode Cultures
4.2. Nematode Mortality Bioassays
4.3. Effect on Egg Development
4.4. Effect on Egg Hatching from Egg Masses
4.5. Soil Assays
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Li, J.; Zou, C.; Xu, J.; Ji, X.; Niu, X.; Yang, J.; Zhang, K.Q. Molecular mechanisms of nematode-nematophagous microbe interactions: Basis for biological control of plant-parasitic nematodes. Annu. Rev. Phytopathol. 2015, 53, 67–95. [Google Scholar] [CrossRef]
- Kayani, M.Z.; Mukhtar, T.; Hussain, M.A. Effects of southern root-knot nematode population densities and plant age on growth and yield parameters of cucumber. Crop Prot. 2017, 92, 207–213. [Google Scholar] [CrossRef]
- Sikora, R.; Molendijk, L.P.G.; Desaeger, J. Integrated nematode management and crop health: Future challenges and opportunities. In Integrated Nematode Management: State-of-the-Art and Visions for the Future; Sikora, R., Desaeger, J., Molendijk, L.P.G., Eds.; CAB International: Wallingford, UK, 2022; pp. 3–10. [Google Scholar] [CrossRef]
- Jones, J.T.; Haegeman, A.; Danchin, E.G.; Gaur, H.S.; Helder, J.; Jones, M.G.; Kikuchi, T.; Manzanilla-López, R.; Palomares-Rius, J.E.; Wesemael, W.M.; Perry, R.N. Top 10 plant-parasitic nematodes in molecular plant pathology. Mol. Plant Pathol. 2013, 14, 946–961. [Google Scholar] [CrossRef] [PubMed]
- Trudgill, D.L.; Blok, V.C. Apomictic, polyphagous root-knot nematodes: Exceptionally successful and damaging biotrophic root pathogens. Annu. Rev. Phytopathol. 2001, 39, 53–77. [Google Scholar] [CrossRef]
- Bebber, D.P.; Holmes, T.; Gurr, S.J. The global spread of crop pests and pathogens. Glob. Ecol. Biogeogr. 2014, 23, 1398–1407. [Google Scholar] [CrossRef]
- Onkendi, E.M.; Kariuki, G.M.; Marais, M.; Moleleki, L.N. The threat of root-knot nematodes (Meloidogyne spp.) in Africa: A review. Plant Pathol. 2014, 63, 727–737. [Google Scholar] [CrossRef]
- Daramola, F.; Popoola, J.; Eni, A.; Sulaiman, O. Characterization of root-knot nematodes (Meloidogyne spp.) associated with Abelmoschus esculentus, Celosia argentea and Corchorus olitorius. Asian J. Biol. Sci. 2014, 8, 42–50. [Google Scholar] [CrossRef]
- Ralmi, N.H.A.; Khandaker, M.M.; Mat, N. Occurrence and control of root-knot nematode in crops: A review. Aust. J. Crop Sci. 2016, 10, 1649–1654. [Google Scholar] [CrossRef]
- Ntinokas, D.; Roussis, I.; Mavroeidis, A.; Stavropoulos, P.; Folina, A.; Kakabouki, I.; Tzortzakakis, E.A.; Bilalis, D.; Giannakou, I.O. Virulence of five root-knot nematodes (Meloidogyne spp.) on nine industrial hemp (Cannabis sativa L.) varieties and nematicidal potential of hemp seed extracts against Meloidogyne javanica. Plants 2025, 14, 227. [Google Scholar] [CrossRef]
- Ye, L.; Wang, J.Y.; Liu, X.F. Nematicidal activity of volatile organic compounds produced by Bacillus altitudinis AMCC 1040 against Meloidogyne incognita. Arch. Microbiol. 2022, 204, 521. [Google Scholar] [CrossRef]
- Carneiro, R.M.D.G.; Gomes, A.C.M.M.; Almeida, M.R.A.; Correa, V.R.; Karssen, G.; Castagnone-Sereno, P.; Mohammad Deimi, A. Meloidogyne luci n. sp. (Nematoda: Meloidogynidae), a root-knot nematode parasitising different crops in Brazil, Chile and Iran. Nematology 2014, 16, 289–301. [Google Scholar] [CrossRef]
- Gerič Stare, B.; Strajnar, P.; Susič, N.; Urek, G.; Širca, S. Reported populations of Meloidogyne ethiopica in Europe identified as Meloidogyne luci. Plant Dis. 2017, 101, 1627–1632. [Google Scholar] [CrossRef] [PubMed]
- Bačić, J.; Pavlović, M.; Kušić-Tišma, J.; Širca, S.; Theuerschuh, M.; Gerič Stare, B. First report of the root-knot nematode Meloidogyne luci on tomato in Serbia. Plant Dis. 2023, 107, 8. [Google Scholar] [CrossRef]
- Francis, C.A.; Clegg, M.D. Crop rotations in sustainable production systems. In Sustainable Agricultural Systems; CRC Press: Boca Raton, FL, USA, 2020; pp. 107–122. [Google Scholar] [CrossRef]
- Sasanelli, N.; Konrat, A.; Migunova, V.; Toderas, I.; Iurcu-Straistaru, E.; Rusu, S.; Bivol, A.; Andoni, C.; Veronico, P. Review on control methods against plant parasitic nematodes applied in Southern Member States (C Zone) of the European Union. Agriculture 2021, 11, 602. [Google Scholar] [CrossRef]
- Martin, F.N. Development of alternative strategies for management of soilborne pathogens currently controlled with methyl bromide. Annu. Rev. Phytopathol. 2003, 41, 325–350. [Google Scholar] [CrossRef]
- Villaverde, J.J.; Sevilla-Morán, B.; Sandín-España, P.; López-Goti, C.; Alonso-Prados, J.L. Biopesticides in the framework of the European pesticide regulation (EC) No. 1107/2009. Pest Manag. Sci. 2014, 70, 2–5. [Google Scholar] [CrossRef]
- Couceiro, J.C.; Wood, M.J.; Papadopoulos, A.; Silva, J.J.; Vontas, J.; Dimopoulos, G. Development of mycoinsecticides: Advances in formulation, regulatory challenges and market trends for entomopathogenic fungi. J. Fungi 2026, 12, 7. [Google Scholar] [CrossRef]
- Meher, H.C.; Gajbhiye, V.T.; Chawla, G.; Singh, G. Virulence development and genetic polymorphism in Meloidogyne incognita after prolonged exposure to sublethal concentrations of nematicides and continuous growing of resistant tomato cultivars. Pest Manag. Sci. 2009, 65, 1201–1207. [Google Scholar] [CrossRef]
- Chen, J.; Li, Q.X.; Song, B. Chemical nematicides: Recent research progress and outlook. J. Agric. Food Chem. 2020, 68, 12175–12188. [Google Scholar] [CrossRef]
- Sharon, E.; Bar-Eyal, M.; Chet, I.; Herrera-Estrella, A.; Kleifeld, O.; Spiegel, Y. Biological control of the root-knot nematode Meloidogyne javanica by Trichoderma harzianum. Phytopathology 2001, 91, 687–693. [Google Scholar] [CrossRef]
- Siddiqui, I.A.; Shaukat, S.S.; Khan, A. Differential impact of some Aspergillus species on Meloidogyne javanica biocontrol by Pseudomonas fluorescens strain CHA0. Lett. Appl. Microbiol. 2004, 39, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Xie, G.L.; Soad, A.; Coosemans, J. Suppression of Meloidogyne javanica by antagonistic and plant growth-promoting rhizobacteria. J. Zhejiang Univ. Sci. B 2005, 6, 496–501. [Google Scholar] [CrossRef]
- Caboni, P.; Saba, M.; Tocco, G.; Casu, L.; Murgia, A.; Maxia, A.; Menkissoglu-Spiroudi, U.; Ntalli, N. Nematicidal activity of mint aqueous extracts against the root-knot nematode Meloidogyne incognita. J. Agric. Food Chem. 2013, 61, 9784–9788. [Google Scholar] [CrossRef]
- Eloh, K.; Kpegba, K.; Sasanelli, N.; Koumaglo, H.K.; Caboni, P. Nematicidal activity of some essential plant oils from tropical West Africa. Int. J. Pest Manag. 2019, 66, 131–141. [Google Scholar] [CrossRef]
- Leoni, C.; Piancone, E.; Sasanelli, N.; Bruno, G.L.; Manzari, C.; Pesole, G.; Ceci, L.R.; Volpicella, M. Plant health and rhizosphere microbiome: Effects of the bionematicide Aphanocladium album in tomato plants infested by Meloidogyne javanica. Microorganisms 2020, 8, 1922. [Google Scholar] [CrossRef]
- Nasiou, E.; Giannakou, I.O. Nematicidal potential of thymol against Meloidogyne javanica (Treub) Chitwood. Plants 2023, 12, 1851. [Google Scholar] [CrossRef]
- Bui, H.X.; Desaeger, J.A. Volatile compounds as potential bio-fumigants against plant-parasitic nematodes—A mini review. J. Nematol. 2021, 53, e2021-14. [Google Scholar] [CrossRef]
- De Clerck, C.; Genva, M.; Jijakli, M.H.; Fauconnier, M.L. Use of essential oils and volatile compounds as biological control agents. Foods 2021, 10, 1062. [Google Scholar] [CrossRef] [PubMed]
- Thomas, G.; Withall, D.; Birkett, M. Harnessing microbial volatiles to replace pesticides and fertilizers. Microb. Biotechnol. 2020, 13, 1366–1376. [Google Scholar] [CrossRef] [PubMed]
- Kang, B.R.; Anderson, A.J.; Kim, Y.C. Hydrogen cyanide produced by Pseudomonas chlororaphis O6 exhibits nematicidal activity against Meloidogyne hapla. Plant Pathol. J. 2018, 34, 35–43. [Google Scholar] [CrossRef]
- Yan, D.; Cao, A.; Wang, Q.; Li, Y.; Guo, M.; Guo, X. Dimethyl disulfide (DMDS) as an effective soil fumigant against nematodes in China. PLoS ONE 2019, 14, e0224456. [Google Scholar] [CrossRef]
- Sarri, K.; Mourouzidou, S.; Ntalli, N.; Monokrousos, N. Recent advances and developments in the nematicidal activity of essential oils and their components against root-knot nematodes. Agronomy 2024, 14, 213. [Google Scholar] [CrossRef]
- Wood, M.J.; Kortsinoglou, A.M.; Khoja, S.; Kouvelis, V.N.; Myrta, A.; Midthassel, A.; Loveridge, E.J.; Butt, T.M. Metarhizium brunneum (Hypocreales: Clavicipitaceae) and its derived volatile organic compounds as biostimulants of commercially valuable angiosperms and gymnosperms. J. Fungi 2022, 8, 1052. [Google Scholar] [CrossRef] [PubMed]
- Wood, M.J.; Kortsinoglou, A.M.; Bull, J.C.; Eastwood, D.C.; Kouvelis, V.N.; Bourdon, P.A.; Loveridge, E.J.; Mathias, S.; Meyrick, A.; Midthassel, A.; Myrta, A.; Butt, T. Evaluation of Metarhizium brunneum- and Metarhizium-derived VOCs as dual-active biostimulants and pest repellents in a wireworm-infested potato field. J. Fungi 2023, 9, 599. [Google Scholar] [CrossRef]
- Kishimoto, K.; Matsui, K.; Ozawa, R.; Takabayashi, J. Volatile 1-octen-3-ol induces a defensive response in Arabidopsis thaliana. J. Gen. Plant Pathol. 2007, 73, 35–37. [Google Scholar] [CrossRef]
- Khoja, S.; Eltayef, K.M.; Baxter, I.; Bull, J.C.; Loveridge, E.J.; Butt, T. Fungal volatile organic compounds show promise as potent molluscicides. Pest Manag. Sci. 2019, 75, 3392–3404. [Google Scholar] [CrossRef]
- Bourdon, P.A.; Zottele, M.; Baxter, I.; Myrta, A.; Midthassel, A.; Wechselberger, K.F.; Butt, T.M. Fumigation of three major soil pests (Agriotes lineatus, Diabrotica virgifera virgifera, Phyllopertha horticola) with 3-octanone and 1-octen-3-ol enantiomers. Biocontrol Sci. Technol. 2022, 32, 863–876. [Google Scholar] [CrossRef]
- Hummadi, E.H.; Dearden, A.; Generalovic, T.; Clunie, B.; Harrott, A.; Cetin, Y.; Demirbek, M.; Khoja, S.; Eastwood, D.; Dudley, E.; Hazir, S.; Touray, M.; Ulug, D.; Gulsen, S.H.; Cimen, H.; Butt, T. Volatile organic compounds of Metarhizium brunneum influence the efficacy of entomopathogenic nematodes in insect control. Biol. Control 2021, 155, 104527. [Google Scholar] [CrossRef] [PubMed]
- Khoja, S.; Eltayef, K.M.; Baxter, I.; Myrta, A.; Bull, J.C.; Butt, T. Volatiles of the entomopathogenic fungus Metarhizium brunneum attract and kill plant parasitic nematodes. Biol. Control 2021, 152, 104472. [Google Scholar] [CrossRef]
- Kline, D.L.; Allan, S.A.; Bernier, U.R.; Welch, C.H. Evaluation of the enantiomers of 1-octen-3-ol and 1-octyn-3-ol as attractants for mosquitoes associated with a freshwater swamp in Florida, USA. Med. Vet. Entomol. 2007, 21, 323–331. [Google Scholar] [CrossRef]
- Xu, P.; Zhu, F.; Buss, G.K.; Leal, W.S. 1-Octen-3-ol—the attractant that repels. F1000Research 2015, 4, 156. [Google Scholar] [CrossRef]
- Chitarra, G.S.; Abee, T.; Rombouts, F.M.; Posthumus, M.A.; Dijksterhuis, J. Germination of Penicillium paneum conidia is regulated by 1-octen-3-ol, a volatile self-inhibitor. Appl. Environ. Microbiol. 2004, 70, 2823–2829. [Google Scholar] [CrossRef] [PubMed]
- Veronico, P.; Sasanelli, N.; Troccoli, A.; Myrta, A.; Midthassel, A.; Butt, T. Evaluation of fungal volatile organic compounds for control of the plant-parasitic nematode Meloidogyne incognita. Plants 2023, 12, 1935. [Google Scholar] [CrossRef]
- Gourd, T.R.; Schmitt, D.P.; Barker, K.R. Differential sensitivity of Meloidogyne spp. and Heterodera glycines to selected nematicides. J. Nematol. 1993, 25, 746–751. [Google Scholar]
- Watson, T.T. Sensitivity of Meloidogyne enterolobii and Meloidogyne incognita to fluorinated nematicides. Pest Manag. Sci. 2022, 78, 1398–1406. [Google Scholar] [CrossRef]
- Jang, J.; Đăng, Q.; Choi, G.; Park, H.; Kim, J. Control of root-knot nematodes using Waltheria indica producing 4-quinolone alkaloids. Pest Manag. Sci. 2019, 75, 2264–2270. [Google Scholar] [CrossRef]
- Oka, Y. Nematicidal activity of fluensulfone against some migratory nematodes under laboratory conditions. Pest Manag. Sci. 2014, 70, 1850–1858. [Google Scholar] [CrossRef]
- Eisenback, J.D.; Hirschmann, H.; Sasser, J.N.; Triantaphyllou, A.C. A guide to the four most common species of root-knot nematodes (Meloidogyne spp.), with a pictorial key; North Carolina State University Graphics: Raleigh, NC, USA, 1981. [Google Scholar]
- Adam, M.A.M.; Phillips, M.S.; Blok, V.C. Molecular diagnostic key for identification of single juveniles of seven common and economically important species of root-knot nematode (Meloidogyne spp.). Plant Pathol. 2007, 56, 190–197. [Google Scholar] [CrossRef]
- Castagnone-Sereno, P.; Danchin, E.G.; Perfus-Barbeoch, L.; Abad, P. Diversity and evolution of root-knot nematodes, genus Meloidogyne: New insights from the genomic era. Annu. Rev. Phytopathol. 2013, 51, 203–220. [Google Scholar] [CrossRef] [PubMed]
- Terra, W.C.; Campos, V.P.; Martins, S.J.; Costa, L.S.A.S.; da Silva, J.C.P.; Barros, A.F.; Lopez, L.E.; Santos, T.C.N.; Smant, G.; Oliveira, D.F. Volatile organic molecules from Fusarium oxysporum strain 21 with nematicidal activity against Meloidogyne incognita. Crop Prot. 2018, 106, 125–131. [Google Scholar] [CrossRef]
- Huang, D.; Yu, C.; Shao, Z.; Cai, M.; Li, G.; Zheng, L.; Yu, Z.; Zhang, J. Identification and characterization of nematicidal volatile organic compounds from deep-sea Virgibacillus dokdonensis MCCC 1A00493. Molecules 2020, 25, 744. [Google Scholar] [CrossRef]
- Mei, X.; Wang, X.; Li, G. Pathogenicity and volatile nematicidal metabolites from Duddingtonia flagrans against Meloidogyne incognita. Microorganisms 2021, 9, 2268. [Google Scholar] [CrossRef] [PubMed]
- Freire, E.S.; Campos, V.P.; Pinho, R.S.; Oliveira, D.F.; Faria, M.R.; Pohlit, A.M.; Noberto, N.P.; Rezende, E.L.; Pfenning, L.H.; Silva, J.R. Volatile substances produced by Fusarium oxysporum from coffee rhizosphere and other microbes affect Meloidogyne incognita and Arthrobotrys conoides. J. Nematol. 2012, 44, 321–328. [Google Scholar]
- Eisenback, J.D.; Hunt, D.J. General morphology. In Root-knot Nematodes; Perry, R.N., Moens, M., Starr, F.J., Eds.; CAB International: Wallingford, UK, 2009; p. 23. [Google Scholar]
- Chen, W.; Wang, J.; Huang, D.; Cheng, W.; Shao, Z.; Cai, M.; Zheng, L.; Yu, Z.; Zhang, J. Volatile organic compounds from Bacillus aryabhattai MCCC 1K02966 with multiple modes against Meloidogyne incognita. Molecules 2022, 27, 103. [Google Scholar] [CrossRef]
- Deng, X.; Wang, X.; Li, G. Nematicidal effects of volatile organic compounds from microorganisms and plants on plant-parasitic nematodes. Microorganisms 2022, 10, 1201. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.-Q.; Mo, M.-H.; Zhou, J.-P.; Zou, C.-S.; Zhang, K.-Q. Evaluation and identification of potential organic nematicidal volatiles from soil bacteria. Soil Biol. Biochem. 2007, 39, 2567–2575. [Google Scholar] [CrossRef]
- Liarzi, O.; Bucki, P.; Braun Miyara, S.; Ezra, D. Bioactive volatiles from an endophytic Daldinia cf. concentrica isolate affect the viability of the plant parasitic nematode Meloidogyne javanica. PLoS ONE 2016, 11, e0168437. [Google Scholar] [CrossRef]
- Terra, W.C.; Campos, V.P.; Pedroso, M.P.; da Costa, A.L.; Freire, E.S.; de Pinto, I.P.; da Silva, J.C.P.; Lopez, L.E.; Santos, T.C.N. Volatile molecules of Fusarium oxysporum strain 21 are retained in water and control Meloidogyne incognita. Biol. Control 2017, 112, 34–40. [Google Scholar] [CrossRef]
- Inamdar, A.A.; Hossain, M.M.; Bernstein, A.I.; Miller, G.W.; Richardson, J.R.; Bennett, J.W. Fungal-derived semiochemical 1-octen-3-ol disrupts dopamine packaging and causes neurodegeneration. Proc. Natl. Acad. Sci. USA 2013, 110, 19561–19566. [Google Scholar] [CrossRef]
- Hussey, R.S.; Barker, K.R. A comparison of methods of collecting inocula of Meloidogyne spp., including a new technique. Plant Dis. Rep. 1973, 57, 1025–1028. [Google Scholar]
| Source | Type III Sum of Squares | df | Mean Square | F | Significance |
| Corrected Model | 1095061 | 49 | 22348.19 | 1575.517 | <0.001 |
| Intercept | 979282.6 | 1 | 979282.6 | 69038.09 | <0.001 |
| Compound | 663.46 | 1 | 663.46 | 46.773 | <0.001 |
| Species | 4043.962 | 4 | 1010.99 | 71.273 | <0.001 |
| Concentration | 1037025 | 4 | 259256.2 | 18277.21 | <0.001 |
| Compound x Species | 7537.917 | 4 | 1884.479 | 132.853 | <0.001 |
| Compound x Concentration | 6316.356 | 4 | 1579.089 | 111.324 | <0.001 |
| Species x Concentration | 15836.88 | 16 | 989.805 | 69.78 | <0.001 |
| Compound x Species x Concentration | 23638.1 | 16 | 1477.381 | 104.153 | <0.001 |
| Error | 6383.102 | 450 | 14.185 | ||
| Total | 2080727 | 500 | |||
| Corrected Total | 1101444 | 499 |
| Source | Type III Sum of Squares | df | Mean Square | F | Significance | |
| Corrected Model | 177800.038 | 29 | 6131.036 | 31.631 | <0.001 | |
| Intercept | 602241.240 | 1 | 602241.240 | 3107.101 | <0.001 | |
| Compound | 199.341 | 1 | 199.341 | 1.028 | 0.311 | |
| Day | 85883.546 | 3 | 42941.773 | 221.546 | <0.001 | |
| Concentration | 77365.078 | 4 | 19341.269 | 99.786 | <0.001 | |
| Compound × Day | 234.110 | 2 | 117.055 | 0.604 | 0.547 | |
| Compound × Concentration | 4388.443 | 4 | 1097.111 | 5.660 | <0.001 | |
| Day × Concentration | 8636.658 | 8 | 1079.582 | 5.570 | <0.001 | |
| Compound × Day × Concentration | 1121.377 | 8 | 140.172 | 0.723 | 0.671 | |
| Error | 110287.779 | 569 | 193.827 | |||
| Total | 890782.409 | 599 | ||||
| Corrected Total | 288087.817 | 598 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





