1. Introduction
Seed germination and early seedling establishment represent a critical phase in the plant life cycle because they concentrate largely irreversible physiological decisions—metabolic reactivation, seed-cover weakening, radicle protrusion, and initial tissue differentiation—that ultimately shape seedling vigour and the probability of successful establishment. These processes are governed by the interplay between the internal status of the seed (developmental maturity, reserve mobilization capacity, membrane integrity, and dormancy level) and external drivers such as temperature, water availability, oxygen supply, and the ionic composition of the surrounding medium [
1,
2,
3,
4].
Among environmental factors, temperature is a primary regulator of germination rate and uniformity and can generate contrasting responses even within horticultural species when other conditions are held constant [
5]. Therefore, seed performance is typically evaluated not only by final germination but also by the speed of emergence, which is closely related to subsequent establishment under practical conditions [
6]. In addition, variation among commercial lots and varieties can be amplified by differences in seed ageing and survival, affecting both viability and early performance even before visible emergence occurs [
7].
Although final germination percentage is widely used as a quality indicator, it is often insufficient to describe real seed lot performance: different treatments may reach similar final values but exhibit distinct temporal dynamics, translating into differences in synchrony, uniformity, and early vigour. For this reason, methodological frameworks emphasize quantifying the germination process as a time-dependent phenomenon rather than a single endpoint [
8]. Likewise, critical evaluations of single-value germination indices show that the shape and dispersion of the cumulative curve contain biologically meaningful information that should be captured through complementary metrics [
9]. This is consistent with the broader concept of seed vigour as the capacity to establish rapidly and uniformly, extending beyond mere viability [
10].
Early growth—particularly the development of the primary root—constitutes another key component of vigour because it integrates reserve utilization efficiency, cell expansion, and the ability to explore the medium soon after emergence. Foundational plant physiology frameworks highlight that early root elongation is a sensitive readout of growth regulation and resource acquisition potential [
11]. Because ionic and osmotic constraints can directly impair cell expansion and water relations, salinity tolerance mechanisms provide an explanatory framework for understanding how ionic stress conditions can penalize early establishment [
12]. In parallel, mineral nutrition theory underscores that ions are essential to sustain metabolism and growth, yet imbalanced ionic environments can generate osmotic stress or ion-specific toxicity, compromising germination and seedling development [
13,
14].
In vitro systems provide a controlled platform to study these processes by minimizing environmental noise and enabling standardized comparisons across genotypes and treatments. Defined culture media have been central to plant tissue culture and early development research, with the Murashige–Skoog formulation serving as a widely adopted reference for controlled growth [
15]. Comprehensive tissue culture manuals further emphasize the importance of medium composition and physical support matrices for reproducible development [
16]. At the same time, low-cost approaches—including alternative gelling agents and simplified support matrices—have been explored to facilitate large-scale or resource-limited experimentation [
17]. However, the interpretability of in vitro assays depends strongly on asepsis: contamination can rapidly alter local pH, oxygen availability, and nutrient dynamics, biasing treatment effects [
18], and microbial hazards remain a recurrent challenge in plant tissue and cell culture systems [
19].
Accordingly, robust surface-sterilization and establishment protocols are essential to ensure that observed differences reflect treatment and genotype rather than uncontrolled microbial growth. Recent work has demonstrated that optimization of in vitro sterilization and germination conditions benefits from systematic modeling and predictive approaches [
20]. From a design standpoint, factorial experiments are especially valuable because they allow separation of main effects (e.g., genotype and fertilization) and their interaction under randomized conditions [
21]. When multiple treatments are compared, classical multiple-comparison procedures remain standard for mean separation after ANOVA [
22], while rank-based nonparametric approaches provide alternatives when normality assumptions are not met [
23,
24]. Moreover, controlling error rates under multiple testing is critical to preserve inferential validity, particularly when several indices and response variables are analyzed simultaneously [
25].
Beyond univariate inference, early establishment is increasingly interpreted as a multivariate phenotype in which germination dynamics and growth traits covary. Principal component analysis (PCA) is a standard tool for reducing dimensionality and revealing latent axes that summarize complex trait relationships [
26,
27]. Practical frameworks for exploratory multivariate analysis in R provide coherent workflows for interpreting these structures and linking them to biological meaning [
28]. Subsequently, hierarchical clustering—particularly Ward’s minimum-variance criterion—supports the identification of treatment groups with homogeneous phenotypic profiles, strengthening interpretation at the “profile” level rather than single-variable comparisons [
29].
Radish (
Raphanus sativus L.) is frequently used as a rapid and informative model for germination and early growth studies due to its short emergence time and sensitivity to metabolic and environmental modulation, making it well suited for detecting genotype-dependent responses under controlled conditions [
30]. To ensure comparability and reproducibility, seed testing standards recommend rigorous sampling procedures and controlled handling, which are particularly relevant when working with commercial seed lots [
31]. In addition, reproducible statistical workflows benefit from validated software ecosystems: multivariate analyses are commonly implemented with dedicated R packages for PCA and related methods [
32], complemented by visualization tools for extracting and communicating multivariate results [
33], all within the R statistical computing environment [
34].
Mechanistic and semi-mechanistic modeling further strengthens interpretation of germination dynamics by explicitly linking rate processes to water relations and temperature constraints. Hydrothermal-time and related frameworks have been widely applied to quantify and model germination and dormancy responses under varying environmental conditions [
35]. In particular, water-relations analyses of germination rates provide a physiological basis for interpreting delays or inhibition under osmotic stress [
36]. Related work has formalized the joint role of time and temperature in germination processes [
37], and demonstrated that constant temperatures combined with defined osmotic potentials can systematically shift germination outcomes [
38]. Hydrothermal-time models have also been used to explain cardinal temperatures for germination and to predict responses across environmental gradients [
39]. Extensions of these models can characterize thermoinhibition processes [
40], while methodological studies emphasize that distribution-function choice can influence model performance and biological inference [
41]. More recently, alternative modeling strategies such as cure models have been proposed to better represent heterogeneous germination responses when a fraction of seeds fails to germinate within the observation window [
42].
Complementary to modeling, classical indices remain valuable for practical discrimination among treatments and seed lots when applied with appropriate caution. Indices combining speed and completeness, such as germination value, have long been used to integrate performance dimensions into single interpretable measures [
43,
44]. Moreover, frequent radicle-emergence scoring has been shown to improve the predictive value of time-based metrics for seed vigour, reinforcing the importance of high-resolution temporal data in early establishment studies [
45].
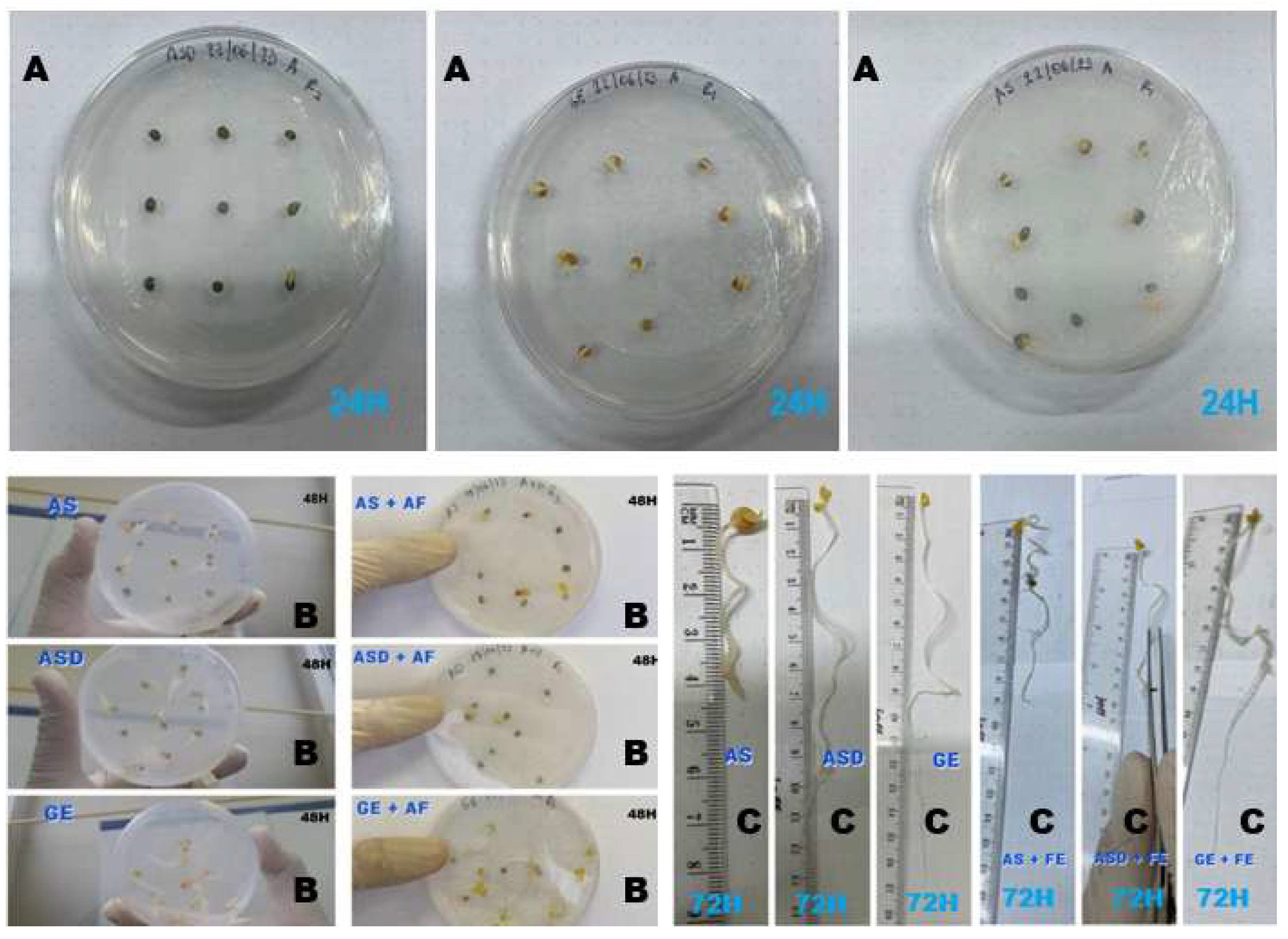
In this study, we investigate early phenotypic plasticity of radish under a simplified ionic contrast in vitro by comparing three commercial varieties (two domestic and one imported) acquired in Milagro, Ecuador. We implemented a completely randomized 3 × 2 factorial design (Variety × Fertilization) using two media: 1.5% agar without fertilizer (A) and 1.5% agar supplemented with a commercial NPK fertilizer (AF). Germination was recorded at 24, 48, and 72 h, and performance was characterized through germination dynamics and vigour-related indices alongside early root growth (primary root length at 72 h). Multivariate profiling (PCA and hierarchical clustering on components) was used to integrate response variables and classify treatments according to global phenotypic patterns.
Objective. To determine the effect of supplementing a 1.5% agar-based in vitro system with a commercial NPK fertilizer on germination dynamics, vigour indices, and early root growth of three commercial Raphanus sativus L. varieties, and to classify treatments by their overall phenotypic profile using multivariate analysis (PCA and HCPC).
4. Discussion
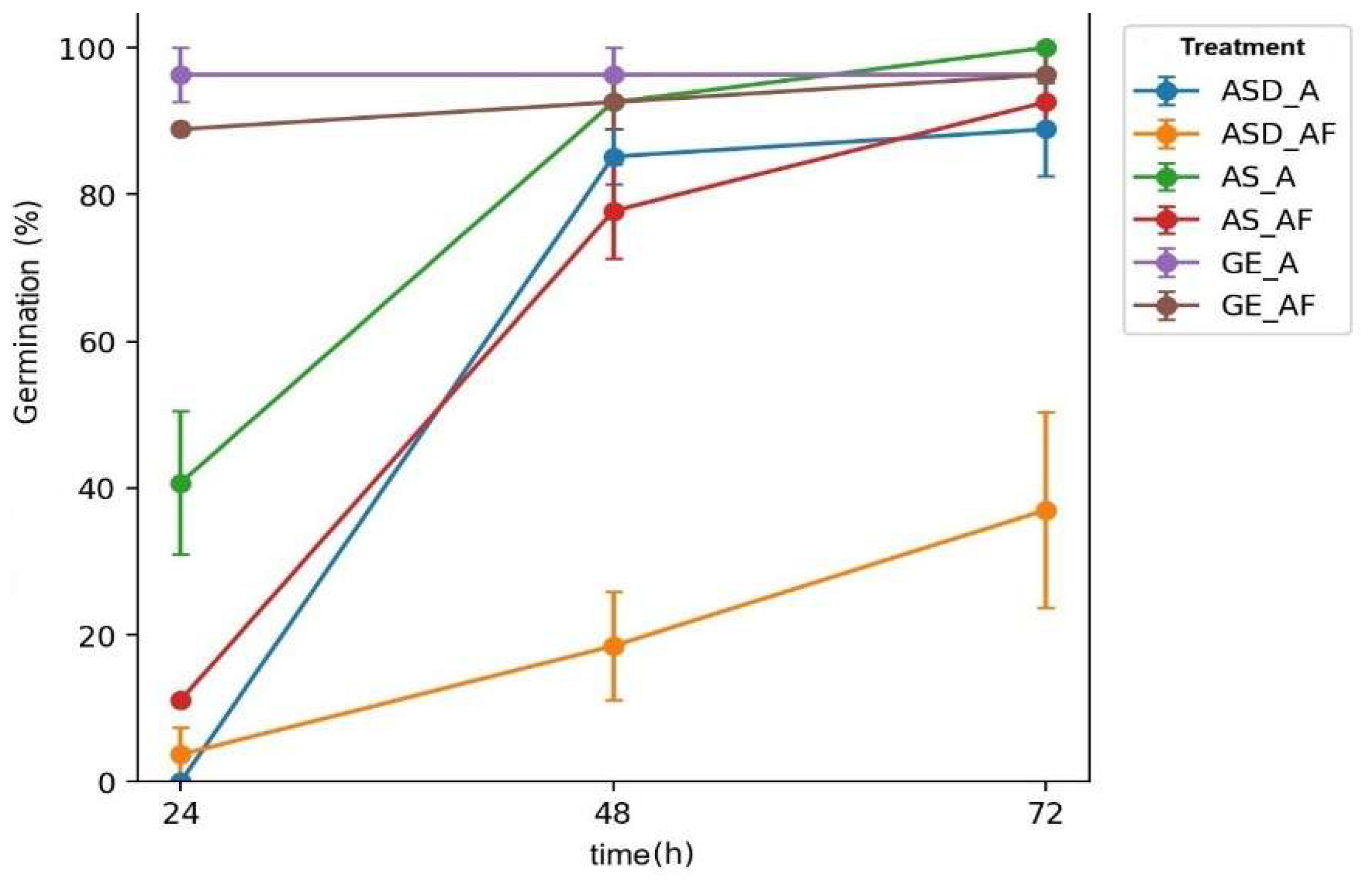
The strong Variety × Fertilization interaction observed in this experiment (notably the collapse of %G and IVG in ASD under AF, together with the marked inhibition of root elongation in ASD and GE) indicates that adding a commercial NPK blend to an agar-based in vitro system acts less as a “universal nutrient boost” and more as a genotype-specific ionic/osmotic filter, where small differences in seed physiology, membrane integrity, reserve mobilization, and ion-handling capacity are amplified under controlled conditions.
A plausible mechanistic interpretation is that AF increased ionic strength and altered the balance among N forms and counter-ions, shifting water relations during imbibition and early cell expansion; this would explain why final germination could remain high in AS and GE while germination timing (MGT) and vigor (IVG) deteriorated, and why radicle growth was disproportionately penalized in ASD and GE. In radish specifically, early growth is known to respond strongly to salinity/ionic stress, and management strategies that mitigate ion toxicity can partially restore seedling performance; for example, silicon supplementation has been reported to alleviate ammonium toxicity in radish, supporting the idea that ion-form/ion-balance effects (not only “nutrient quantity”) can drive root-growth inhibition and could be leveraged in follow-up assays to test mitigation options under AF-like conditions [
49].
Likewise, work in other in vitro crop systems shows that shifting NPK concentrations can redirect developmental outcomes and impose cultivar-dependent penalties, reinforcing that commercial fertilizers behave as complex inputs whose net effect depends on genotype and medium context rather than on “NPK dose” alone [
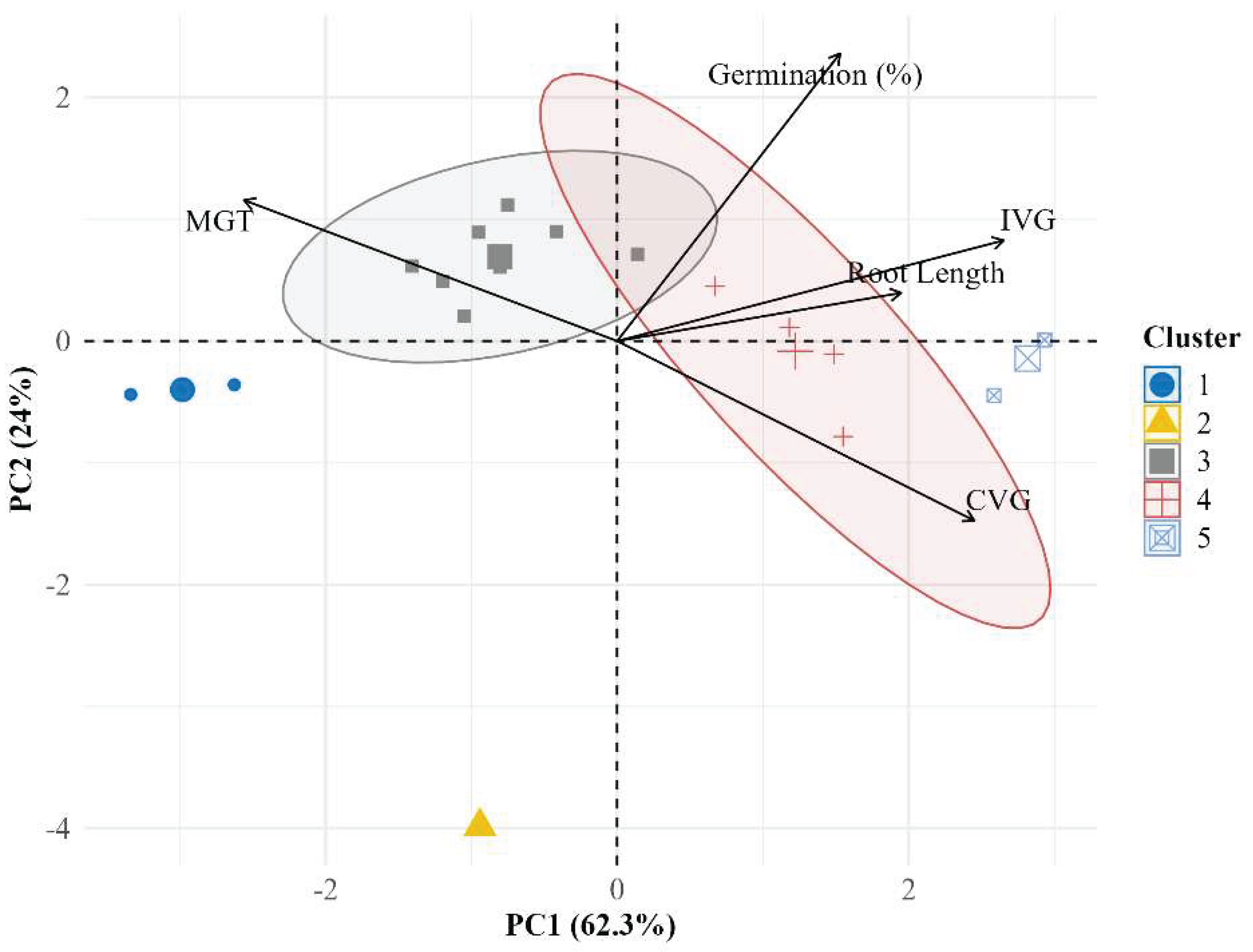
50]. From a phenotyping standpoint, the separation of treatments along an integrated vigour axis (PC1 dominated by IVG/MGT/root length) is consistent with the modern view that early establishment is a multivariate phenotype and should be interpreted as a coordinated trait network rather than isolated endpoints; this argues for expanding image-based quantification (root architecture, curvature, branching proxies) to capture subtle stress signatures that precede visible failure, aligning with broader plant-phenotyping roadmaps and the routine use of reproducible image-analysis platforms to standardize trait extraction across treatments and labs [
51,
52].
The fact that GE exhibited rapid establishment in the control yet lost more than half of its root length under AF also fits a stress-response pattern reported in microgreen/brassicaceous systems under combined thermal/ionic constraints highlighting the importance of jointly interpreting germination curves and morphometry instead of relying on %G alone [
53]. Statistically, the study’s multi-endpoint structure (several vigor indices + morphology + multivariate profiling) strengthens inference when multiplicity is explicitly controlled, and future expansions with finer temporal scoring or additional traits should consider procedures robust to dependency among tests (common in correlated vigor variables), such as FDR control under dependency and/or sequentially rejective adjustments to keep conclusions stable as the number of outcomes increases [
54,
55].
Biologically, the genotype dependence points to early metabolic regulation and signal integration as a likely driver: in vitro environments can shift the balance between nitrate signaling, dormancy release, and reactive nitrogen species pathways, and the literature shows that nitrate can act as a dormancy-relief signal, that nitrate-dependent regulation of dormancy/germination is genetically and physiologically structured, and that nitric oxide can break dormancy and modulate germination through interactions with hormone signaling and ROS; together, these mechanisms offer a coherent explanation for why one genotype (ASD) could be pushed into a low-germination/low-vigor state under AF while another (AS) remains comparatively stable [
61,
62,
63,
64]. At the same time, the pronounced root-length penalties under AF are consistent with the idea that early roots integrate nutrient-availability cues into architectural decisions and growth rates; nutrient deficiencies/excesses and imbalanced ion availability can reshape root system plasticity, and traits (“root phenes”) that improve soil exploration and nutrient acquisition in later stages may already be partially expressed as differential early elongation responses under in vitro ionic contrasts, suggesting that the observed patterns could be an early proxy for broader nutrient-use strategies among varieties [
65,
66,
67,
68].
Methodologically, these findings support treating commercial fertilizer–agar systems as a low-cost screening tool rather than as a replacement for defined media: classic culture-medium research emphasizes that mineral requirements and ion forms matter, and micropropagation literature shows that “commercially feasible” protocols often succeed by controlling specific medium components and interactions rather than by increasing generic fertilization; thus, a rational next step is to chemically characterize AF (EC, pH, major ion forms, N speciation) and run a small concentration series to identify thresholds where each genotype transitions from stimulation to inhibition [
56,
57]. In parallel, complementary pre-sowing strategies—such as priming and targeted seed treatments known to improve emergence and early growth under abiotic stress—could be tested as practical countermeasures to AF-induced inhibition, especially for ASD-like sensitive lots [
58,
59,
60].
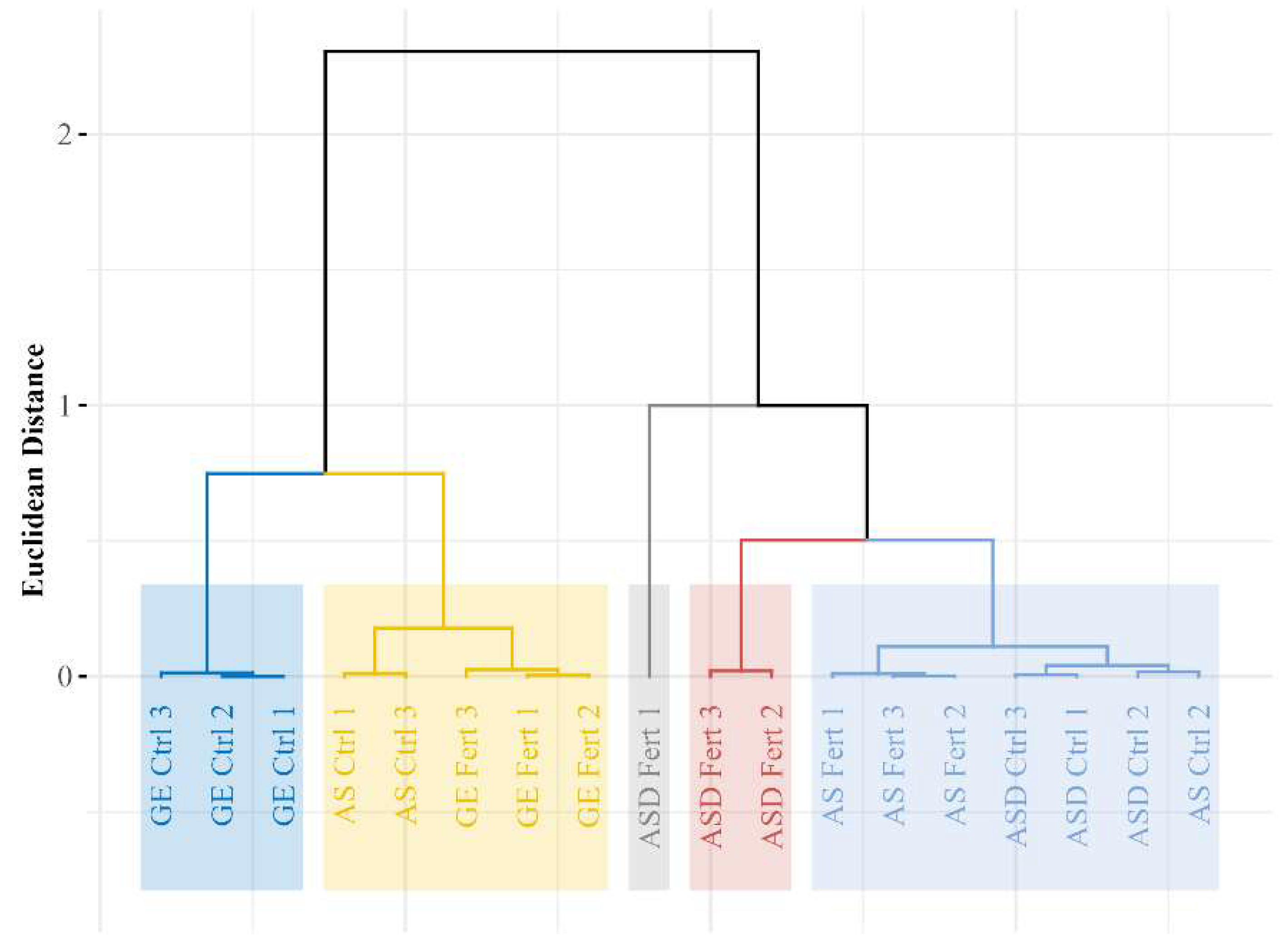
Finally, because PCA + hierarchical clustering are ultimately pattern-recognition frameworks, their interpretability improves when grounded in established multivariate ecology and clustering principles (distance choice, scaling, linkage effects, and cluster validity), so reporting robustness checks (e.g., sensitivity to scaling, alternative distance metrics, silhouette-like diagnostics) would further strengthen the generality of the five-group solution and its biological meaning for seed-lot screening applications [
69,
70].
Author Contributions
For research articles with several authors, a short paragraph specifying their individual contributions must be provided. The following statements should be used “Conceptualization, L. E. C. M., K R-P, F G-V, B. P-G., S. V-A., R. P-P., I. A. A. G., E. R-N. and C. E. D. E.; methodology, L. E. C. M., K. R-P., B. P-G., R. P-P., S. P. J. J., E. R-N. and C. E. D. E.; software, I. I-V., S. V-A and I. A. A. G.; validation, I. I-V., S V-A. and I. A. A. G.; formal analysis, L. E. C. M., F. G-V., I. I-V., S. V-A; investigation, L. E. C. M., K. R-P, F. G-V. B. P-G., S.V-A., and E. R-N.; resources, L. E. C. M., F. G-V., S. P. J. J. and C. E. D. E.; data curation, I. I-V., S. V-A.; writing—original draft preparation, L. E. C. M. and C. E. D. E.; writing—review and editing, L. E. C. M., K. R-P., B. P-G., I. I-V., S. V-A., R. P-P., S. P. J. J. and E. R-N.; visualization, L. E. C. M., K. R-P., B. P-G., R. P-P., I. A. A. G., E. R-N. and C. E. D. E.; supervision, L. E. C. M., E. R-N. and C. E. D. E.; project administration, C. E. D. E.