Submitted:
07 January 2026
Posted:
09 January 2026
You are already at the latest version
Abstract
The rapid development of viticulture in subtropical regions represents a significant achievement in China’s table grape industry over the past two decades. However, insufficient winter chilling in these areas often leads to inadequate dormancy, which compromises nutrient translocation and storage in grapevine. Insufficient chilling accumulation results in asynchronous budbreak and reduced cane quality. In this study, ‘Shine Muscat’ grapevines were used to systematically evaluate how different defoliant agents affect budbreak characteristics from the perspective of nutrient translocation and storage. The results indicated applications of ethephon or urea alone, as well as their combinations with boric acid, yielded unstable effects, often causing primary bud necrosis, decreased flower formation rates, and phytotoxicity. In contrast, the combination of lime sulfur and boric acid exhibited a remarkable synergistic effect, significantly promoting dry matter and starch accumulation in the canes, while enhancing budbreak speed, uniformity, and flower cluster formation rate. Further experiments with varying concentrations of lime sulfur combined with 0.2% boric acid revealed 2% lime sulfur combined with 0.2% boric acid produced the most pronounced effects. This combination achieved the highest dormancy-breaking efficacy under conventional cultivation conditions and was used for the first time to produce a second crop in off-season cultivation. The dual effects of dormancy release and bud promotion offered by this approach provide a reliable solution for high-quality and efficient grape production in subtropical regions.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Location, Description of the Vineyard and Tested Chemicals
2.2. Treatments and Experimental Design
2.3. Data Collection and Measurement
2.3.1. Dry-Wet Weight Ratio Detection
2.3.2. Starch Content Analysis
2.3.3. Bud Necrosis Rate Statistics
2.3.4. Budbreak Rate and Speed Statistics
2.3.6. Fruiting Shoot Rate Statistics
2.3.7. Statistical Analysis
3. Results
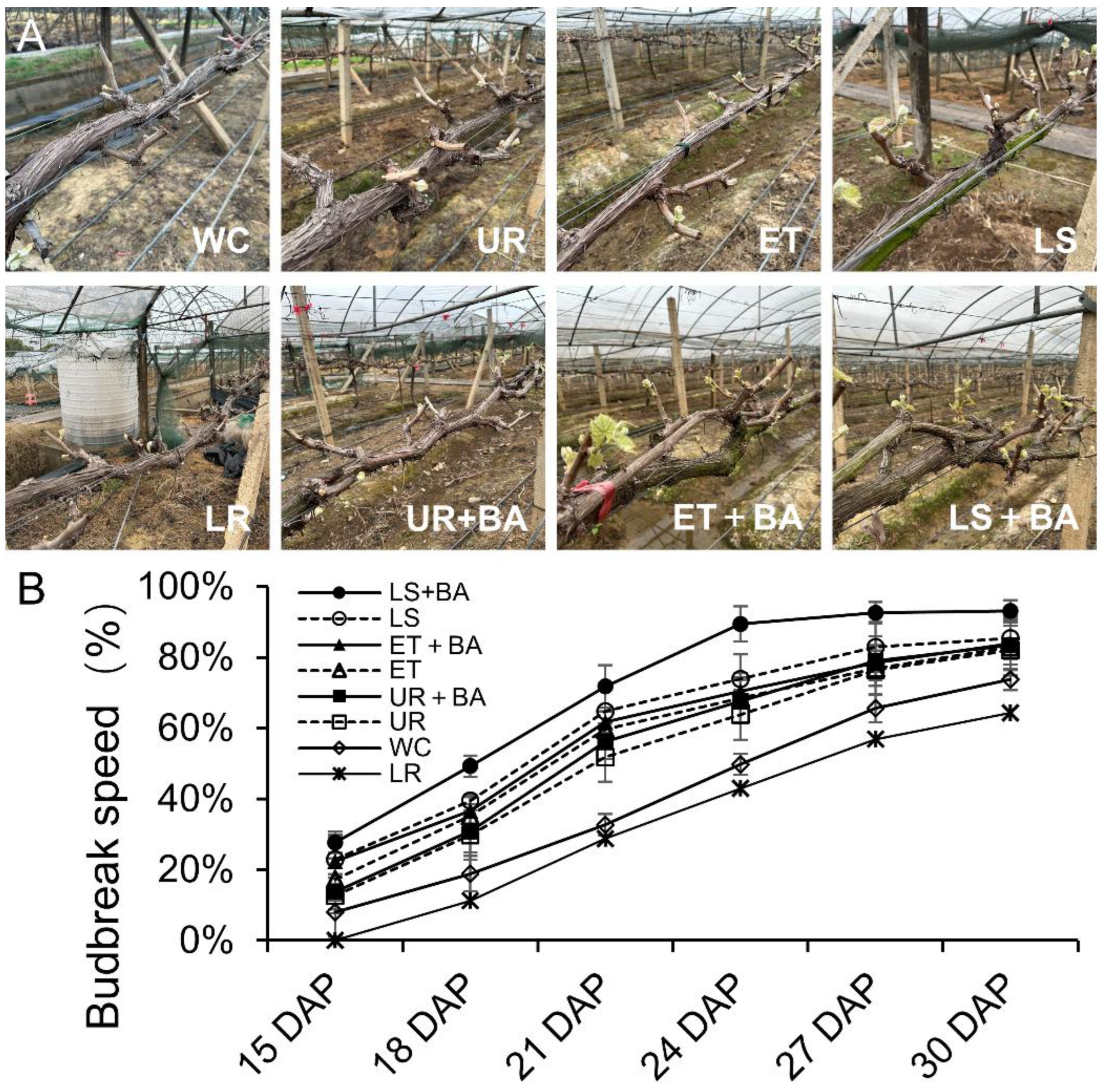
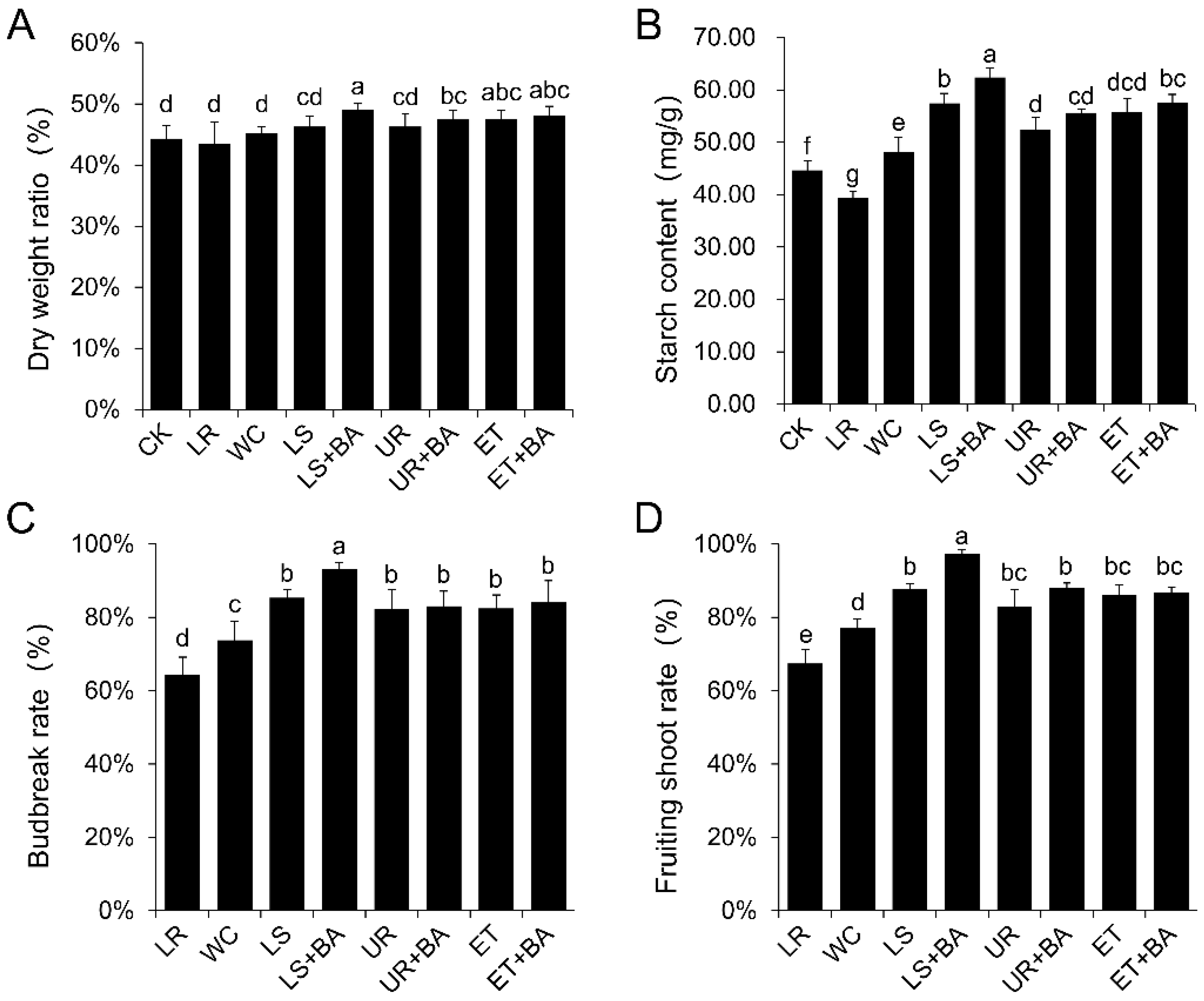
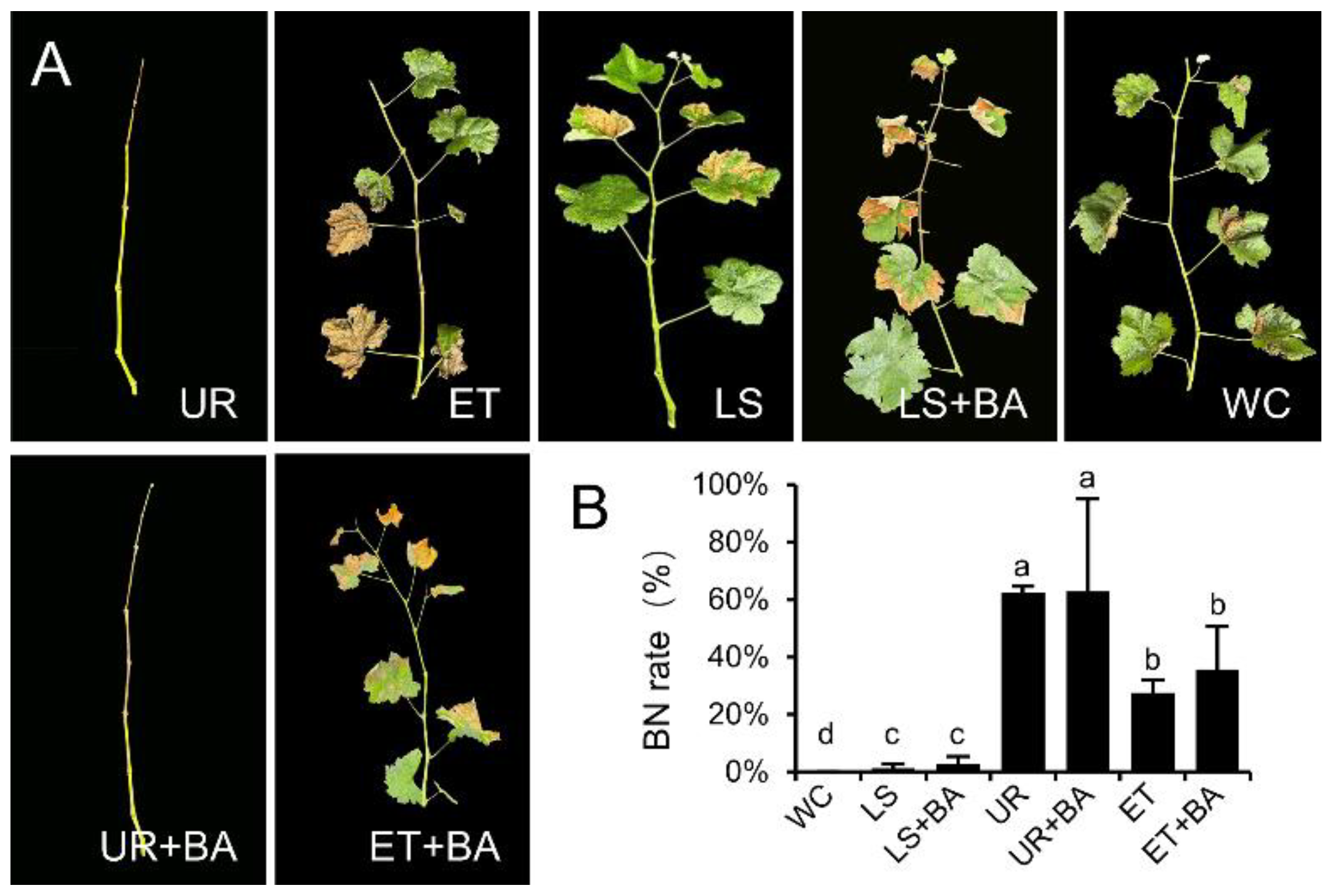
3.1. The Effects of Different Treatments on the First Crop
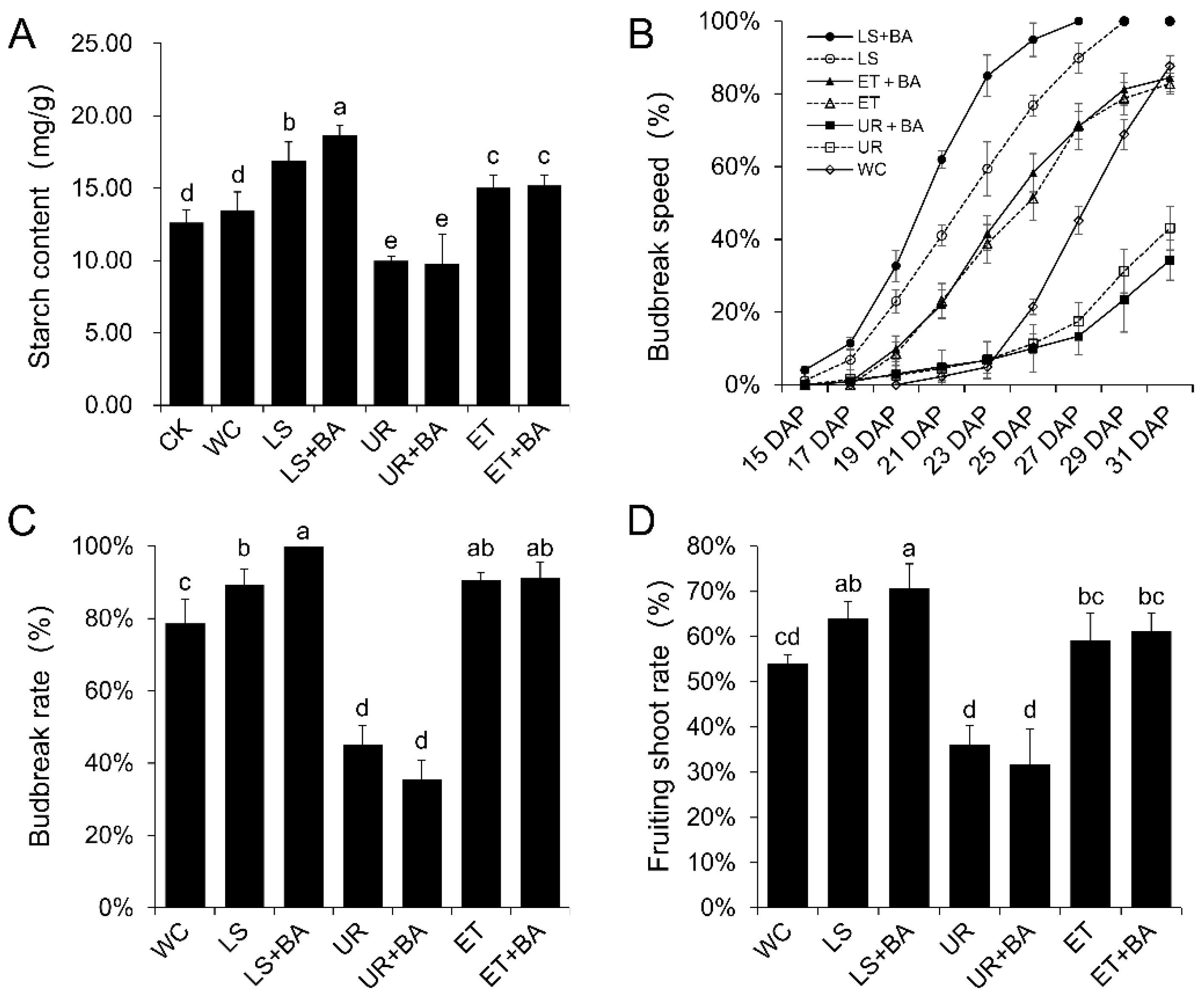
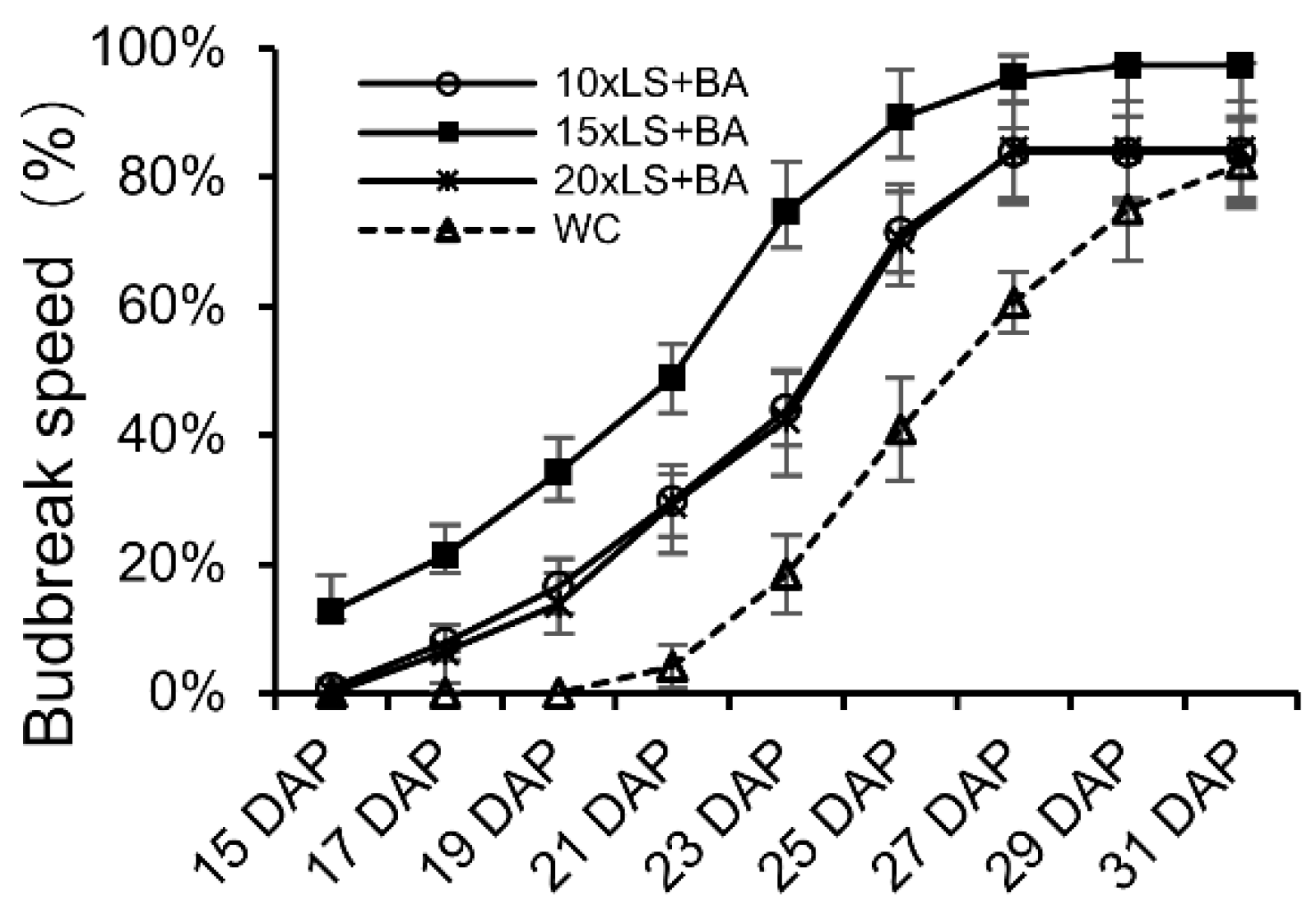
3.2. The Effects of Different Treatments on Grape Secondary Crop
3.3. Effects of Lime Sulfur at Different Concentrations Combined with Boric Acid Treatment
4. Discussion
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| BA | Boric Acid |
| ET | Ethephon |
| LR | Leaf Removal |
| LS | Lime Sulfur |
| UR | Urea |
| WC | Water Control |
References
- OIV Statistical Report on World Vitiviniculture. Paris: International Organisation of Vine and Wine 2018.
- Deng, Q.; Xia, H.; Lin, L.; Wang, J.; Yuan, L.; Li, K.; Zhang, J.; Lv, X.; Liang, D. SUNRED, a Natural Extract-Based Biostimulant, Application Stimulates Anthocyanin Production in the Skins of Grapes. Sci Rep 2019, 9, 2590. [Google Scholar] [CrossRef] [PubMed]
- Londo, J.P.; Johnson, L.M. Variation in the Chilling Requirement and Budburst Rate of Wild Vitis Species. Environmental and Experimental Botany 2014, 106, 138–147. [Google Scholar] [CrossRef]
- Lu, G.; Zhang, K.; Que, Y.; Li, Y. Grapevine Double Cropping: A Magic Technology. Frontiers in Plant Science 2023, 14. [Google Scholar] [CrossRef]
- Júnior, H.F. da R.; Pasa, M. da S.; Malgarim, M.B.; Pasa, E.H.; Paschoal, J.D.F. Bud-Break Promoters for the Improvement of the Budburst of Pecan Cultivars. Pesquisa Agropecuária Brasileira 2022, e02956–e02956. [Google Scholar] [CrossRef]
- Anderson, J.V.; Horvath, D.P.; Chao, W.S.; Foley, M.E. Bud Dormancy in Perennial Plants: A Mechanism for Survival. In Dormancy and Resistance in Harsh Environments; Lubzens, E., Cerda, J., Clark, M., Eds.; Springer: Berlin, Heidelberg, 2010; pp. 69–90. ISBN 978-3-642-12422-8. [Google Scholar]
- Or, E.; Vilozny, I.; Fennell, A.; Eyal, Y.; Ogrodovitch, A. Dormancy in Grape Buds: Isolation and Characterization of Catalase cDNA and Analysis of Its Expression Following Chemical Induction of Bud Dormancy Release. Plant Science 2002, 162, 121–130. [Google Scholar] [CrossRef]
- Sudawan, B.; Chang, C.-S.; Chao, H.; Ku, M.S.B.; Yen, Y. Hydrogen Cyanamide Breaks Grapevine Bud Dormancy in the Summer through Transient Activation of Gene Expression and Accumulation of Reactive Oxygen and Nitrogen Species. BMC Plant Biol 2016, 16, 202. [Google Scholar] [CrossRef]
- Ionescu, I.A.; López-Ortega, G.; Burow, M.; Bayo-Canha, A.; Junge, A.; Gericke, O.; Møller, B.L.; Sánchez-Pérez, R. Transcriptome and Metabolite Changes during Hydrogen Cyanamide-Induced Floral Bud Break in Sweet Cherry. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef]
- Walton, E.F.; Fowke, P.J. Effect of Hydrogen Cyanamide on Kiwifruit Shoot Flower Number and Position. Journal of Horticultural Science 1993, 68, 529–534. [Google Scholar] [CrossRef]
- Sudawan, B.; Chang, C.-S.; Chao, H.; Ku, M.S.B.; Yen, Y. Hydrogen Cyanamide Breaks Grapevine Bud Dormancy in the Summer through Transient Activation of Gene Expression and Accumulation of Reactive Oxygen and Nitrogen Species. BMC Plant Biol 2016, 16, 202. [Google Scholar] [CrossRef]
- Pérez, F.J.; Vergara, R.; Rubio, S. H2O2 Is Involved in the Dormancy-Breaking Effect of Hydrogen Cyanamide in Grapevine Buds. Plant Growth Regul 2008, 55, 149–155. [Google Scholar] [CrossRef]
- Pang, X.; Halaly, T.; Crane, O.; Keilin, T.; Keren-Keiserman, A.; Ogrodovitch, A.; Galbraith, D.; Or, E. Involvement of Calcium Signalling in Dormancy Release of Grape Buds. J Exp Bot 2007, 58, 3249–3262. [Google Scholar] [CrossRef] [PubMed]
- Khalil-Ur-Rehman, M.; Wang, W.; Zheng, H.; Faheem, M.; Iqbal, S.; Shen, Z.G.; Tao, J. Role of Hydrogen Cyanamide (HC) in Grape Bud Dormancy Release: Proteomic Approach. 3 Biotech 2020, 10, 229. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.; Huang, X.; Shen, Y.; Shen, T.; Zhang, H.; Lin, L.; Wang, J.; Deng, Q.; Lyu, X.; Xia, H. Hydrogen Cyanamide Induces Grape Bud Endodormancy Release through Carbohydrate Metabolism and Plant Hormone Signaling. BMC Genomics 2019, 20, 1034. [Google Scholar] [CrossRef]
- Bernasconi, L.; Carnovale, M.; Lonati, D.; Petrolini, V.M.; Schicchi, A.; Brolli, B.; Negrini, V.M.; Grazioli, C.; Maystrova, O.; Buscaglia, E.; et al. Hydrogen Cyanamide Exposure: A Case Series from Pavia Poison Control Centre. Occup Med (Lond) 2023, 73, 500–506. [Google Scholar] [CrossRef]
- Carranca, C.; Brunetto, G.; Tagliavini, M. Nitrogen Nutrition of Fruit Trees to Reconcile Productivity and Environmental Concerns. Plants 2018, 7, 4. [Google Scholar] [CrossRef]
- Tang, J.; Chen, Y.; Huang, C.; Li, C.; Feng, Y.; Wang, H.; Ding, C.; Li, N.; Wang, L.; Zeng, J.; et al. Uncovering the Complex Regulatory Network of Spring Bud Sprouting in Tea Plants: Insights from Metabolic, Hormonal, and Oxidative Stress Pathways. Front. Plant Sci. 2023, 14. [Google Scholar] [CrossRef]
- Hendgen, M.; Günther, S.; Schubert, S.; Löhnertz, O. Ethephon Activates the Transcription of Senescence-Associated Genes and Nitrogen Mobilization in Grapevine Leaves (Vitis Vinifera Cv. Riesling). Plants (Basel) 2021, 10, 333. [Google Scholar] [CrossRef] [PubMed]
- Guiboileau, A.; Sormani, R.; Meyer, C.; Masclaux-Daubresse, C. Senescence and death of plant organs: Nutrient recycling and developmental regulation. Comptes Rendus. Biologies 2010, 333, 382–391. [Google Scholar] [CrossRef]
- Buchanan-Wollaston, V.; Earl, S.; Harrison, E.; Mathas, E.; Navabpour, S.; Page, T.; Pink, D. The Molecular Analysis of Leaf Senescence – a Genomics Approach. Plant Biotechnology Journal 2003, 1, 3–22. [Google Scholar] [CrossRef]
- Lin, S.-Y.; Agehara, S. Foliar Application of Defoliants before Winter Chill Accumulation Advances Budbreak and Improves Fruit Earliness of Blackberry under Subtropical Climatic Conditions. 2021. [Google Scholar] [CrossRef]
- Tan, J.C.; Lin, S.-Y. Foliar Urea Sprays Induce Budbreak in Peach via Rapid Non-Structural Carbohydrate Metabolism – A Lower-Toxicity Alternative to Hydrogen Cyanamide. Scientia Horticulturae 2025, 349, 114267. [Google Scholar] [CrossRef]
- Bhadoria, P.; Nagar, M.; Bharihoke, V.; Bhadoria, A.S. Ethephon, an Organophosphorous, a Fruit and Vegetable Ripener: Has Potential Hepatotoxic Effects? Journal of Family Medicine and Primary Care 2018, 7, 179. [Google Scholar] [CrossRef]
- Gu, S.; Jacobs, S.D.; Mccarthy, B.S.; Gohil, H.L. Forcing Vine Regrowth and Shifting Fruit Ripening in a Warm Region to Enhance Fruit Quality in ‘Cabernet Sauvignon’ Grapevine (Vitis Vinifera L.). The Journal of Horticultural Science and Biotechnology 2012, 87, 287–292. [Google Scholar] [CrossRef]
- Xiu, L.I.; Juntuan, Z.; Zhaolong, S.; Yuxia, L.I.; Zhijun, L.I. Sex Differences in Morphological and Stoichiometric Characteristics of Populus Euphratica Oliv. Branches and Leaves. Chinese Journal of Ecology 2023, 42, 1586. [Google Scholar] [CrossRef]
- Clegg, K.M.; Clegg, K.M. The Application of the Anthrone Reagent to the Estimation of Starch in Cereals. J. Sci. Food Agric 1956, 7, 40–44. [Google Scholar] [CrossRef]
- The Science of Grapevines; 2025; ISBN 978-0-443-33006-3.
- Dahal, K.C.; Bhattarai, S.P.; Midmore, D.J.; Oag, D.; Sapkota, R.; Walsh, K.B. In Situ Sucrose Injection for Alteration of Carbohydrate Reserve Dynamics in Grapevine. Agronomy 2024, 14, 425. [Google Scholar] [CrossRef]
- Skinkis, P.A. Recognize the Symptoms and Causes of Stunted Growth in Vineyards. Available online: https://extension.oregonstate.edu/catalog/pub/em-8975-recognize-symptoms-causes-stunted-growth-vineyards (accessed on 20 December 2025).
- Hale, C.R.; Weaver, R.J. The Effect of Developmental Stage on Direction of Translocation of Photosynthate in Vitis Vinifera. Hilgardia 1962. [Google Scholar] [CrossRef]
- Brown, K.M. Ethylene and Abscission. Physiologia Plantarum 1997, 100, 567–576. [Google Scholar] [CrossRef]
- Zhang, M.; Luo, Y.; Meng, Q.; Han, W. Correction of Leaf Nutrient Resorption Efficiency on the Mass Basis. J Plant Ecol 2022, 15, 1125–1132. [Google Scholar] [CrossRef]
- Wang, W.-Q.; Wang, M.; Lin, P. Seasonal Changes in Element Contents in Mangrove Element Retranslocation during Leaf Senescene. Plant and Soil 2003, 252, 187–193. [Google Scholar] [CrossRef]
- Kowsalya, K.; Halka, J.; Anand, M.; Sahayarayan, J.J.; Rajkumar, R.; Arun, M. Unraveling the Multifaceted Role of Ethephon in Plant Physiology: From Seed Germination to Crop Maturation and Harvesting. J. Plant Biochem. Biotechnol. 2025, 34, 639–664. [Google Scholar] [CrossRef]
- Piekarniak, M.; Weston, L.A.; Gniazdowska, A.; Krasuska, U. Hydrogen Cyanide Acts as a Regulator of Reactive Oxygen Species Metabolism. Planta 2025, 262, 134. [Google Scholar] [CrossRef]
- Sudawan, B.; Chang, C.-S.; Chao, H.; Ku, M.S.B.; Yen, Y. Hydrogen Cyanamide Breaks Grapevine Bud Dormancy in the Summer through Transient Activation of Gene Expression and Accumulation of Reactive Oxygen and Nitrogen Species. BMC Plant Biol 2016, 16, 202. [Google Scholar] [CrossRef]
- Görlach, A.; Bertram, K.; Hudecova, S.; Krizanova, O. Calcium and ROS: A Mutual Interplay. Redox Biology 2015, 6, 260–271. [Google Scholar] [CrossRef] [PubMed]
- Matoh, T. Boron in Plant Cell Walls. Plant and Soil 193 59–70. [CrossRef]
- Kohli, S.K.; Kaur, H.; Khanna, K.; Handa, N.; Bhardwaj, R.; Rinklebe, J.; Ahmad, P. Boron in Plants: Uptake, Deficiency and Biological Potential. Plant Growth Regul 2023, 100, 267–282. [Google Scholar] [CrossRef]
- Tadayon, M.S.; Moafpourian, G. Effects of Exogenous Epi-Brassinolid, Zinc and Boron Foliar Nutrition on Fruit Development and Ripening of Grape (Vitis Vinifera L. Clv. ‘Khalili’). Scientia Horticulturae 2019, 244, 94–101. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



