Submitted:
30 December 2025
Posted:
31 December 2025
You are already at the latest version
Abstract
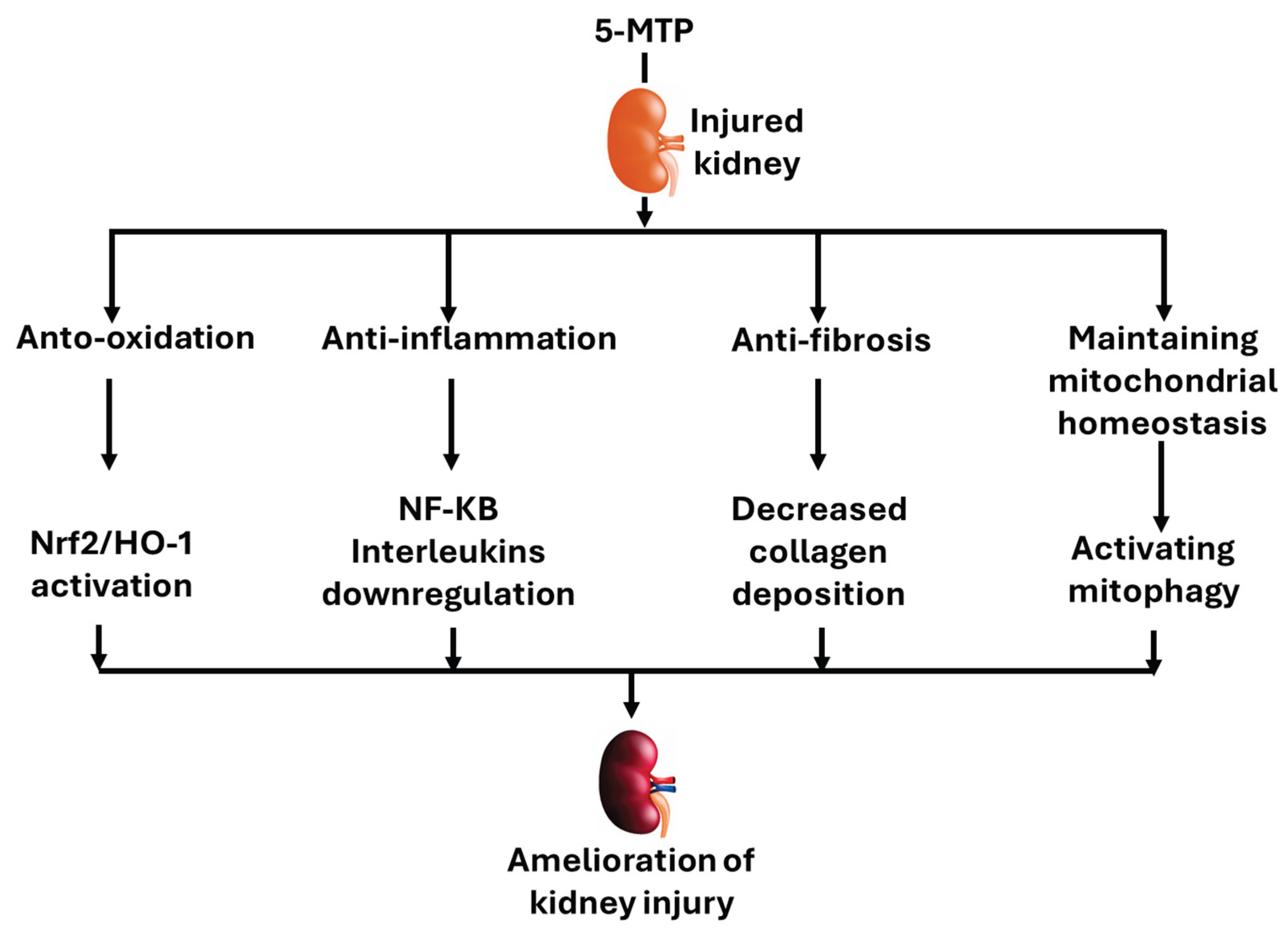
Kidney disease, be it acute or chronic, has a complex pathology and is a significant human health problem. Increasing interest has been focused on exploring therapeutic targets that can be used to safeguard kidney function under a variety of detrimental conditions. In this article, we review the protective effects of 5-methoxytryptophan (5-MTP), a tryptophan metabolite, on kidney injury. Published studies indicate that serum 5-MTP is increased in patients with chronic kidney disease (CKD), suggesting that 5-MTP is a biomarker for CKD and has therapeutic values. Indeed, rodent models of kidney injury induced by folic acid, lipopolysaccharide (LPS), unilateral ureteral obstruction (UUO), and ischemia/reperfusion all demonstrate that exogenous 5-MTP exhibits nephroprotective effects. The underlying mechanisms involve anti-oxidative damage via activating antioxidant systems such as Nrf2/heme oxygenase-1, anti-inflammation, anti-fibrosis, and enhanced mitophagy. To further explore the underlying mechanisms and the potential of 5-MTP as a kidney therapeutic compound, future studies need to include more rodent models of kidney injury induced by a variety of insults. Moreover, how to boost endogenous 5-MTP content and its potential synergistic effects with other therapeutic approaches aiming to combat kidney diseases also remain to be explored.
Keywords:
1. Introduction
1.1. Acute Kidney Injury
1.2. Chronic Kidney Disease
1.3. Diabetic Kidney Disease
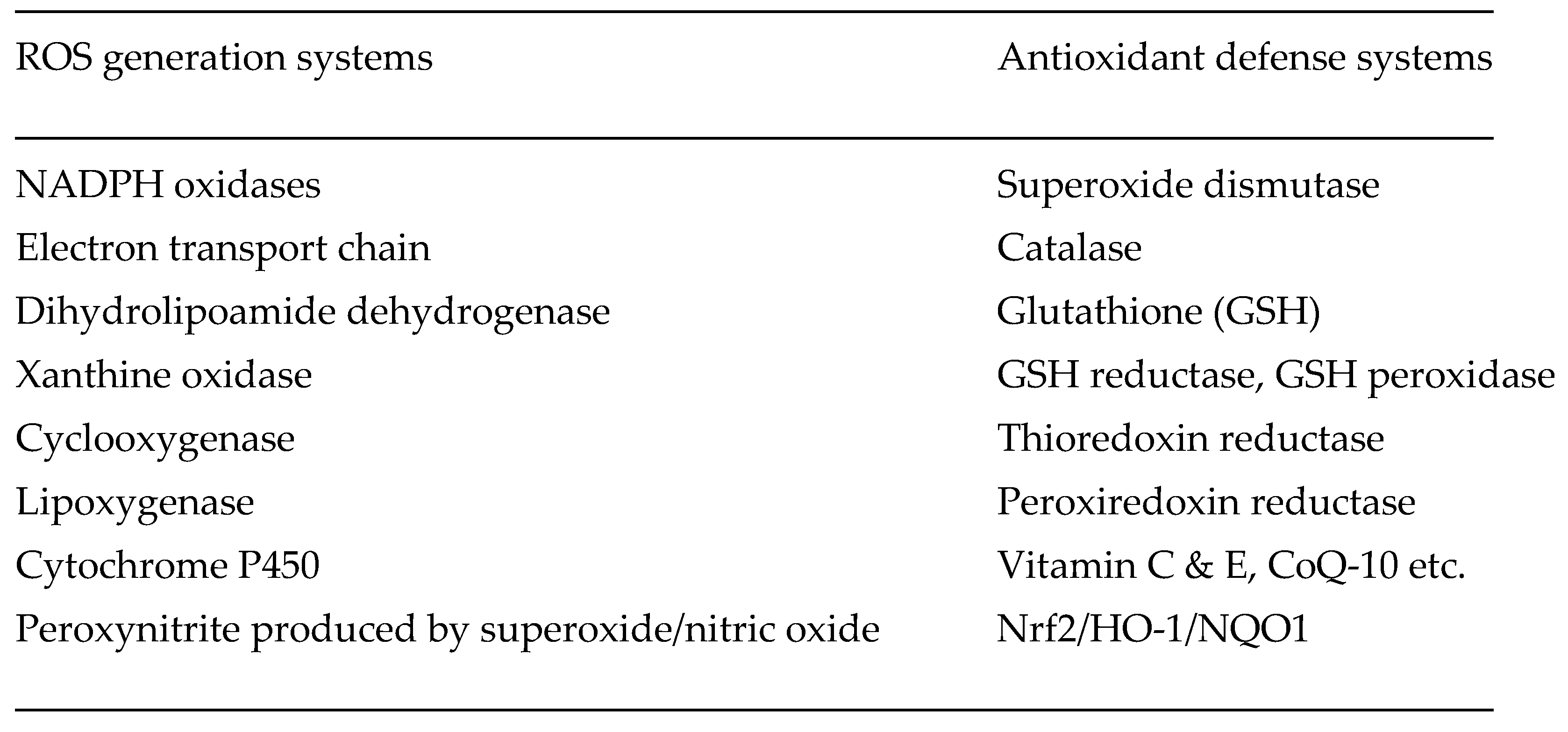
1.4. Oxidative Stress
1.5. Inflammation
1.6. Fibrosis
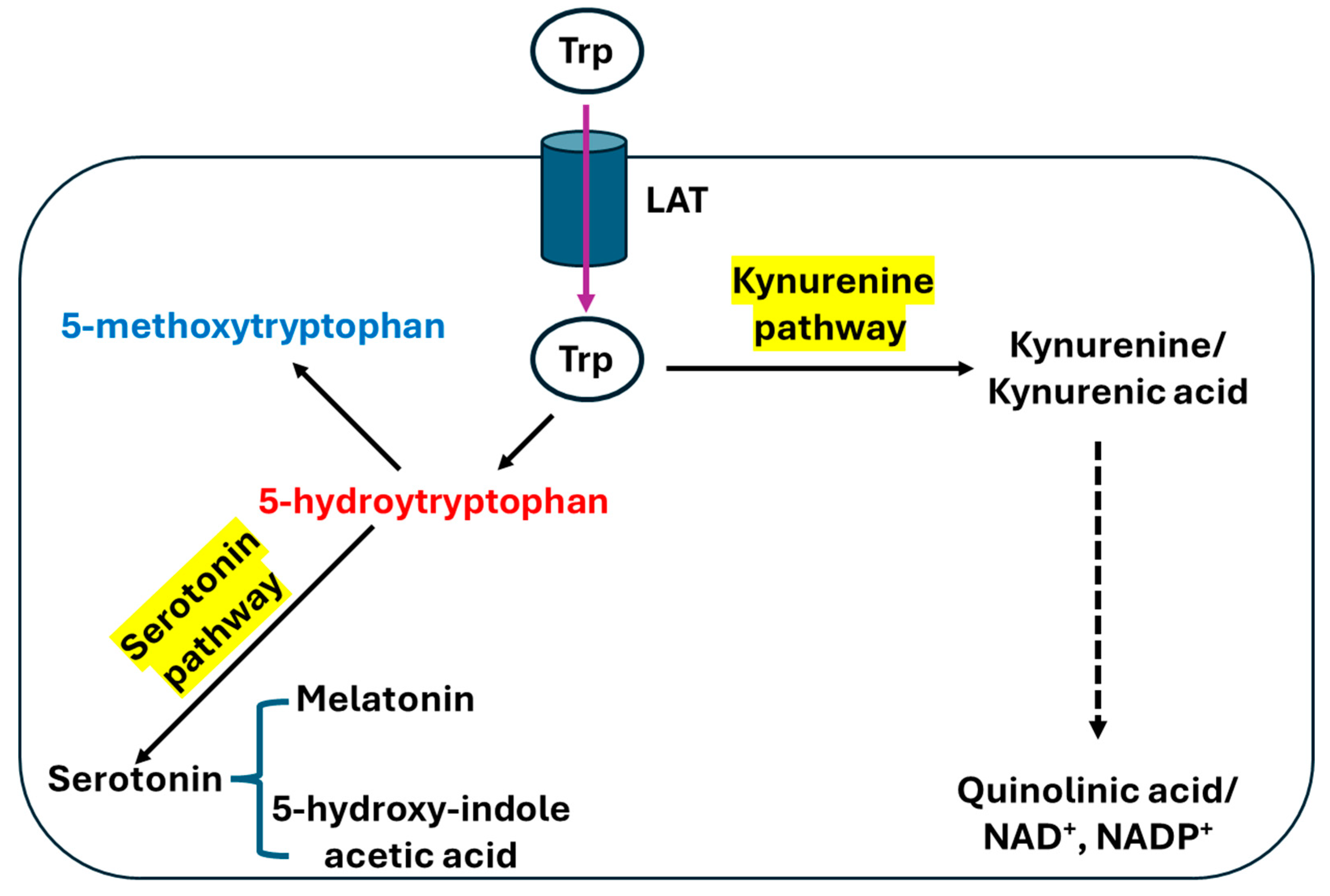
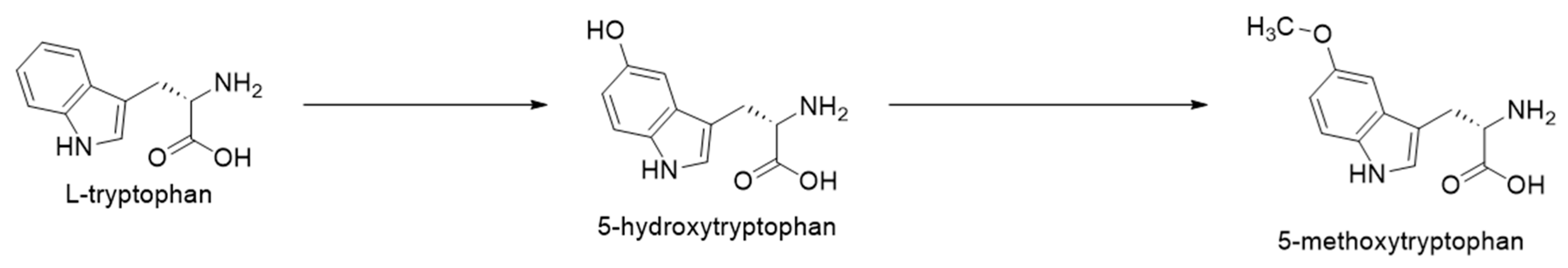
2. Tryptophan and Biosynthesis of 5-MTP
3. Potential Biological Functions of 5-MTP
3.1. 5-MTP and Chronic Kidney Disease (CKD)
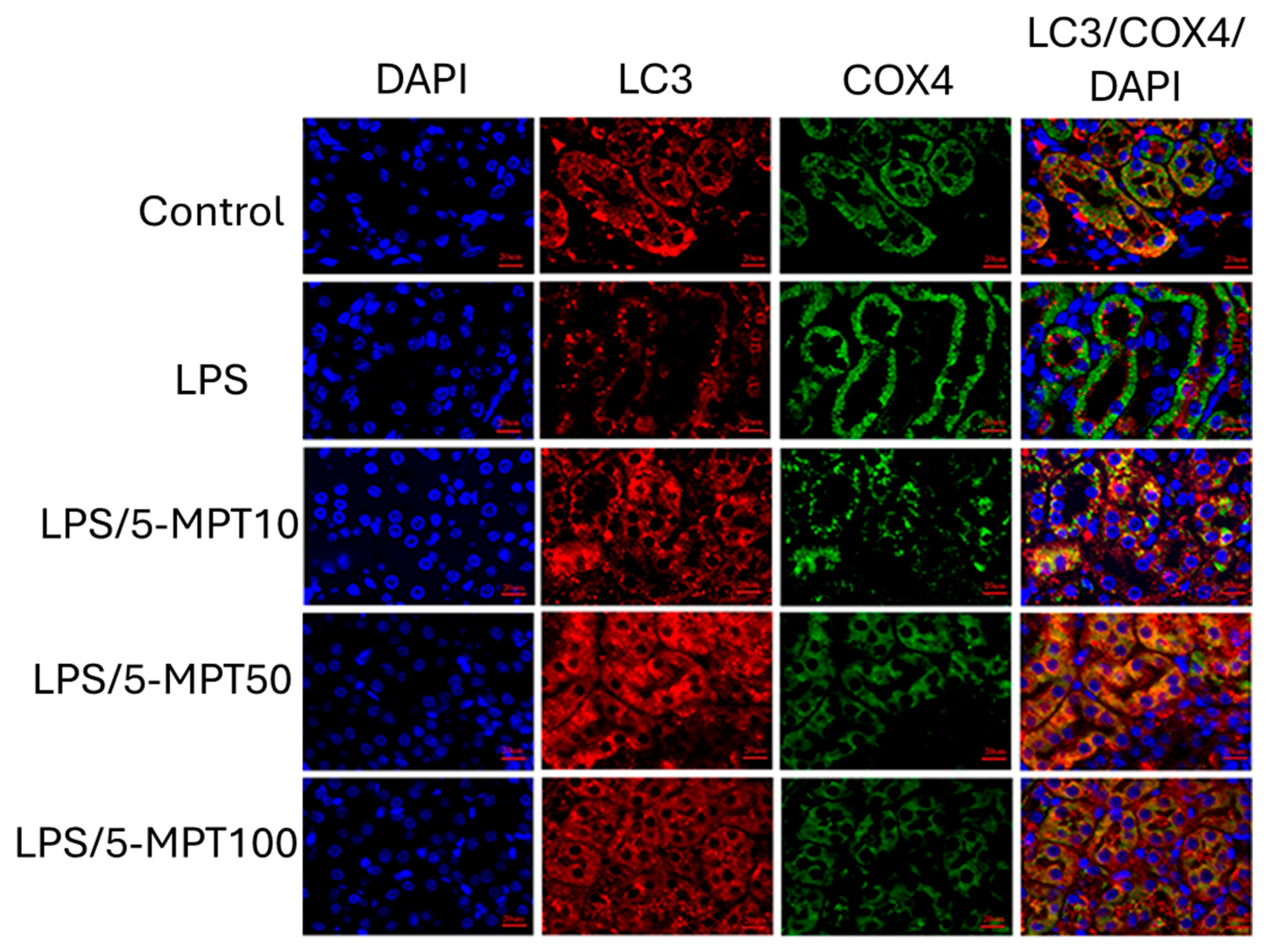
3.2. Effect of 5-MTP on Lipopolysaccharide (LPS)-Induced Acute Kidney Injury
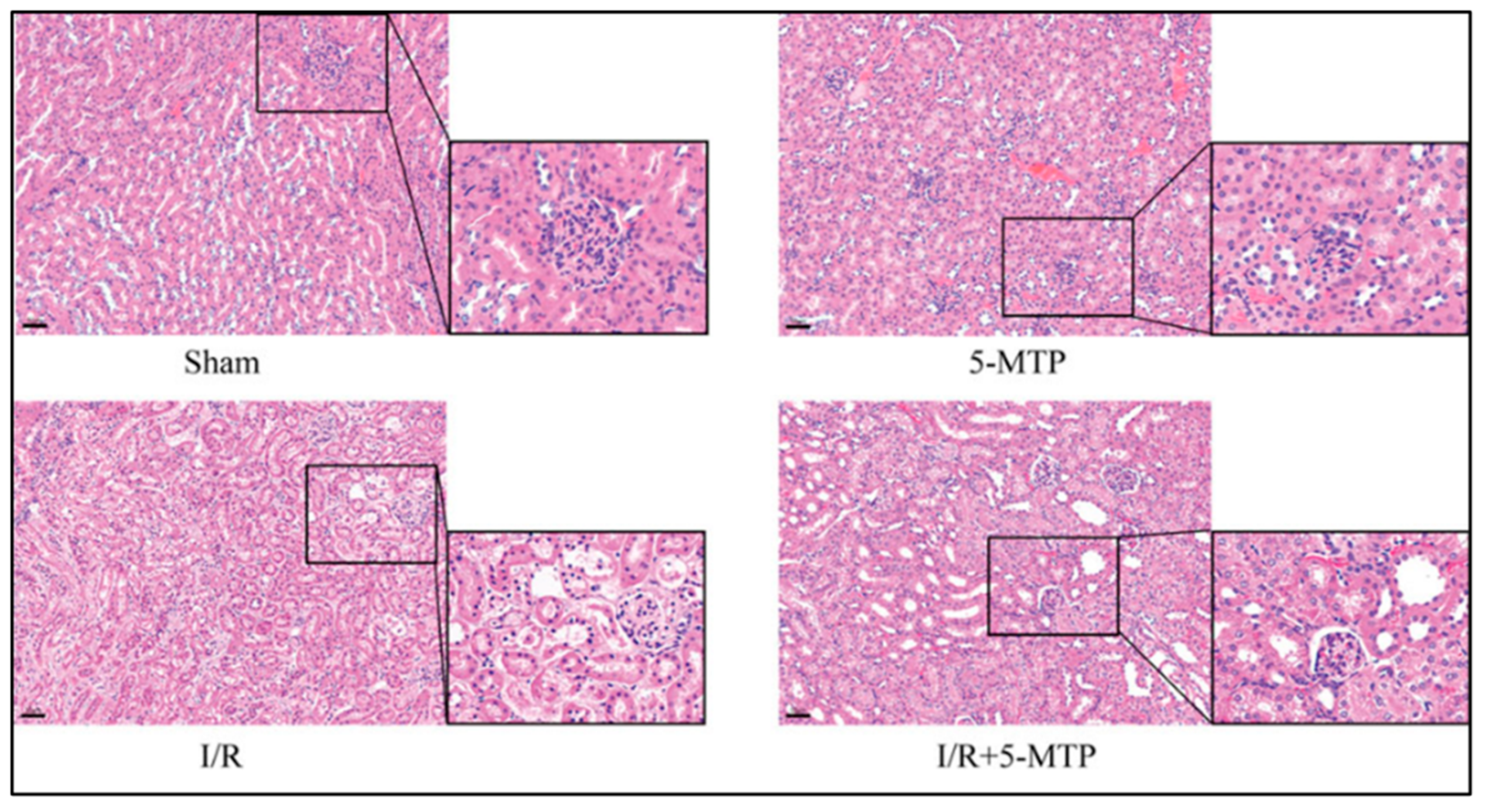
3.3. Effect of 5-MTP on Renal Ischemia/Reperfusion Injury
3.4. Effects of 5-MTP on Renal Tissue Inflammation and Fibrosis
3.5. Effects of 5-MTP on CKD-Induced Cerebrovascular Injury
3.6. Predictive Value of 5-MTP on Clinical Outcomes in Patients Having Septic AKI
3.7. Does Microbial 5-MTP Have a Nephroprotective Effect?
3.8. Pharmacological Boosting of 5-MTP Levels for Renoprotection
4. Future Perspectives
5. Summary
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Reed, S. Essential physiological biochemistry: An organ-based approach; Willey-Baclkwell: Noida, India, 2009. [Google Scholar]
- Lieberman, M.; Peet, A. Marks' Basic Medical Biochemistry: A clinical Approach, 6th ed.; Wolters Kluwer/Lippincott Williams & Wilkins: Philadelphia, 2023. [Google Scholar]
- Duann, P.; Lin, P.H. Mitochondria Damage and Kidney Disease. Adv. Exp. Med. Biol. 2017, 982, 529–551. [Google Scholar]
- Kamt, S.F.; Liu, J.; Yan, L.-J. Renal-Protective Roles of Lipoic Acid in Kidney Disease. Nutrients 2023, 15, 1732. [Google Scholar] [CrossRef] [PubMed]
- Marquez-Exposito, L.; Tejedor-Santamaria, L.; Santos-Sanchez, L.; Valentijn, F.A.; Cantero-Navarro, E.; Rayego-Mateos, S.; Rodrigues-Diez, R.R.; Tejera-Muñoz, A.; Marchant, V.; Sanz, A.B.; et al. Acute Kidney Injury is Aggravated in Aged Mice by the Exacerbation of Proinflammatory Processes. Front. Pharmacol. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Bai, J.; Zhou, X.; Tang, J.; Jiang, C.; Tolbert, E.; Bayliss, G.; Gong, R.; Zhao, T.C.; Zhuang, S. P2X7 receptor inhibition protects against ischemic acute kidney injury in mice. Am. J. Physiol. Physiol. 2015, 308, C463–C472. [Google Scholar] [CrossRef] [PubMed]
- Rosner, M.H.; Okusa, M.D. Acute Kidney Injury Associated with Cardiac Surgery. Clin. J. Am. Soc. Nephrol. 2006, 1, 19–32. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, J.; Liu, Z.; Shu, S.; Fu, Y.; Liu, Y.; Cai, J.; Tang, C.; Liu, Y.; Yin, X.; et al. The PINK1/PARK2/optineurin pathway of mitophagy is activated for protection in septic acute kidney injury. Redox Biol. 2021, 38, 101767. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, J.; Chen, S.; Peng, J.; Luo, X.; Wang, L.; Liao, R.; Zhao, Y.; Zhang, S.; Su, B. Genetic and Pharmacological Inhibition of NOX4 Protects Against Rhabdomyolysis-Induced Acute Kidney Injury Through Suppression of Endoplasmic Reticulum Stress. Antioxidants 2025, 14, 1162. [Google Scholar] [CrossRef]
- Iskander, A.; Yan, L.-J. Cisplatin-Induced Kidney Toxicity: Potential Roles of Major NAD+-Dependent Enzymes and Plant-Derived Natural Products. Biomolecules 2022, 12, 1078. [Google Scholar] [CrossRef]
- Fiorentino, M.; Grandaliano, G.; Gesualdo, L.; Castellano, G. Acute Kidney Injury to Chronic Kidney Disease Transition. Contrib Nephrol 2018, 193, 45–54. [Google Scholar]
- Ameer, O.Z. Hypertension in chronic kidney disease: What lies behind the scene. Front. Pharmacol. 2022, 13. [Google Scholar] [CrossRef]
- Robles-Osorio, M.L.; Sabath-Silva, E.; Sabath, E. Arsenic-mediated nephrotoxicity. Ren. Fail. 2015, 37, 542–547. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.-J.; Allen, D.C. Cadmium-Induced Kidney Injury: Oxidative Damage as a Unifying Mechanism. Biomolecules 2021, 11, 1575. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.-J. The Nicotinamide/Streptozotocin Rodent Model of Type 2 Diabetes: Renal Pathophysiology and Redox Imbalance Features. Biomolecules 2022, 12, 1225. [Google Scholar] [CrossRef] [PubMed]
- Pedraza-Chaverri, J.; Eugenio-Pérez, D.; Medina-Fernández, L.Y.; Saldivar-Anaya, J.A.; Molina-Jijón, E. Role of Dietary Antioxidant Agents in Chronic Kidney Disease. In Free Radicals and Diseases; Ahmad, R., Ed.; IntechOpen: London, UK, 2016. [Google Scholar]
- Hsu, C.-Y. Linking the population epidemiology of acute renal failure, chronic kidney disease and end-stage renal disease. Curr. Opin. Nephrol. Hypertens. 2007, 16, 221–226. [Google Scholar] [CrossRef]
- Ammirati, A.L. Chronic Kidney Disease. Revista da Associação Médica Brasileira 2020, 66. [Google Scholar] [CrossRef]
- Diep, T.N.; Liu, H.; Yan, L.-J. Beneficial Effects of Butyrate on Kidney Disease. Nutrients 2025, 17, 772. [Google Scholar] [CrossRef]
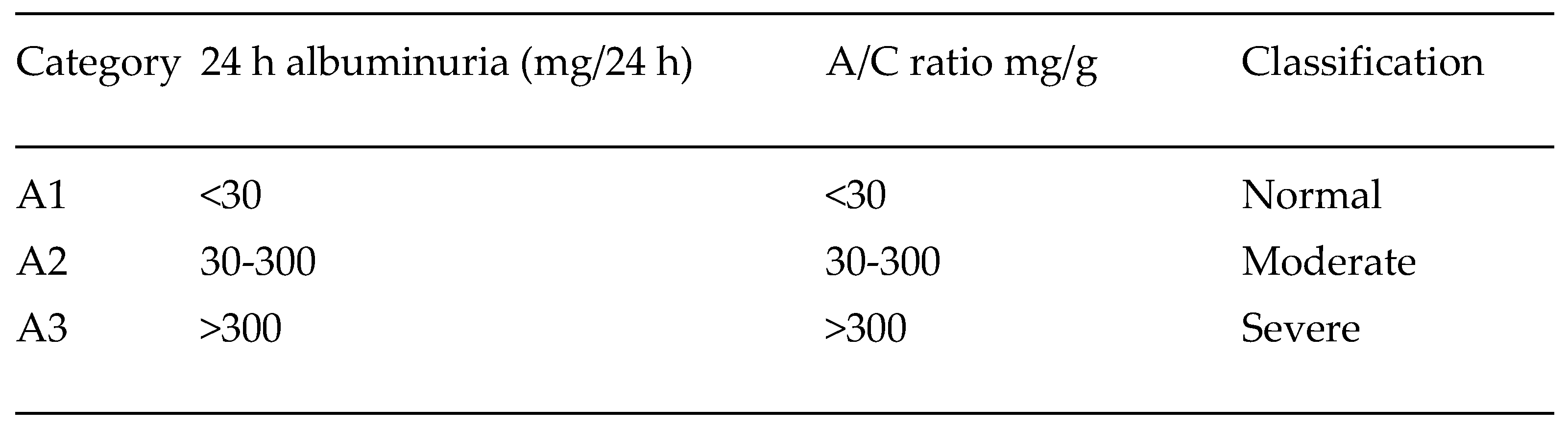
- Beernink, J.M.; van Mil, D.; Laverman, G.D.; Heerspink, H.J.L.; Gansevoort, R.T. Developments in albuminuria testing: A key biomarker for detection, prognosis and surveillance of kidney and cardiovascular disease—A practical update for clinicians. Diabetes, Obes. Metab. 2025, 27, 15–33. [Google Scholar] [CrossRef]
- Yan, L.J. NADH/NAD(+) Redox Imbalance and Diabetic Kidney Disease. Biomolecules 2021, 11. [Google Scholar] [CrossRef]
- Yan, L.J. Analysis of oxidative modification of proteins. Curr Protoc Protein Sci 2009, Chapter 14, Unit14 4. [Google Scholar]
- Kuroda, J.; Sadoshima, J. NADPH Oxidase and Cardiac Failure. J. Cardiovasc. Transl. Res. 2010, 3, 314–320. [Google Scholar] [CrossRef]
- Wu, J.; Jin, Z.; Yan, L.-J. Redox imbalance and mitochondrial abnormalities in the diabetic lung. Redox Biol. 2017, 11, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Song, J.; Yan, L.-J. Chronic Inhibition of Mitochondrial Dihydrolipoamide Dehydrogenase (DLDH) as an Approach to Managing Diabetic Oxidative Stress. Antioxidants 2019, 8, 32. [Google Scholar] [CrossRef] [PubMed]
- Andrés, C.M.C.; de la Lastra, J.M.P.; Juan, C.A.; Plou, F.J.; Pérez-Lebeña, E. Hypochlorous Acid Chemistry in Mammalian Cells—Influence on Infection and Role in Various Pathologies. Int. J. Mol. Sci. 2022, 23, 10735. [Google Scholar] [CrossRef]
- Nishino, T.; Okamoto, K.; Eger, B.T.; Pai, E.F.; Nishino, T. Mammalian xanthine oxidoreductase – mechanism of transition from xanthine dehydrogenase to xanthine oxidase. FEBS J. 2008, 275, 3278–3289. [Google Scholar] [CrossRef] [PubMed]
- Ames, B.N.; Shigenaga, M.K. Oxidants Are a Major Contributor to Aginga. Ann. New York Acad. Sci. 1992, 663, 85–96. [Google Scholar] [CrossRef]
- Miwa, S.; Muller, F.L.; Beckman, K.B. The basics of oxidative biochemistry. In Oxidative stress in aging; Miwa, S., Beckman, K.B., Muller, F.L., Eds.; Humana Press: Totowa, NJ, USA, 2008; pp. 11–35. [Google Scholar]
- Yan, L.-J.; Lodge, J.K.; Traber, M.G.; Packer, L. Apolipoprotein B Carbonyl Formation Is Enhanced by Lipid Peroxidation during Copper-Mediated Oxidation of Human Low-Density Lipoproteins. Arch. Biochem. Biophys. 1997, 339, 165–171. [Google Scholar] [CrossRef]
- Yan, L.J.; Lodge, J.K.; Traber, M.G.; Matsugo, S.; Packer, L. Comparison between copper-mediated and hypochlorite-mediated modifications of human low density lipoproteins evaluated by protein carbonyl formation. J. Lipid Res. 1997, 38, 992–1001. [Google Scholar] [CrossRef]
- Yegin, S.Ç.; Dede, S.; Mis, L.; Yur, F. Effects of Zinc Supplementation on DNA Damage in Rats with Experimental Kidney Deficiency. Biol. Trace Element Res. 2016, 176, 338–341. [Google Scholar] [CrossRef]
- Baek, J.H.; Yalamanoglu, A.; Brown, R.P.; Saylor, D.M.; A Malinauskas, R.; Buehler, P.W. Renal Toxicodynamic Effects of Extracellular Hemoglobin After Acute Exposure. Toxicol. Sci. 2018, 166, 180–191. [Google Scholar] [CrossRef]
- Yan, L.-J.; Rajasekaran, N.S.; Sathyanarayanan, S.; Benjamin, I.J. Mouse HSF1 Disruption Perturbs Redox State and Increases Mitochondrial Oxidative Stress in Kidney. Antioxidants Redox Signal. 2005, 7, 465–471. [Google Scholar] [CrossRef]
- Yan, W.; Xu, Y.; Yuan, Y.; Tian, L.; Wang, Q.; Xie, Y.; Shao, X.; Zhang, M.; Ni, Z.; Mou, S. Renoprotective mechanisms of Astragaloside IV in cisplatin-induced acute kidney injury. Free. Radic. Res. 2017, 51, 669–683. [Google Scholar] [CrossRef] [PubMed]
- Sedaghat, Z.; Kadkhodaee, M.; Seifi, B.; Salehi, E.; Najafi, A.; Dargahi, L. Remote per-conditioning reduces oxidative stress, downregulates cyclo-oxygenase-2 expression and attenuates ischaemia-reperfusion-induced acute kidney injury. Clin. Exp. Pharmacol. Physiol. 2013, 40, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Menini, S.; Iacobini, C.; Vitale, M.; Pugliese, G. The Inflammasome in Chronic Complications of Diabetes and Related Metabolic Disorders. Cells 2020, 9, 1812. [Google Scholar] [CrossRef] [PubMed]
- Black, L.M.; Lever, J.M.; Agarwal, A. Renal Inflammation and Fibrosis: A Double-edged Sword. J. Histochem. Cytochem. 2019, 67, 663–681. [Google Scholar] [CrossRef]
- Luo, Y.; Long, M.; Wu, X.; Zeng, L. Targeting the NLRP3 inflammasome in kidney disease: molecular mechanisms, pathogenic roles, and emerging small-molecule therapeutics. Front. Immunol. 2025, 16, 1703560. [Google Scholar] [CrossRef]
- Liu, H.; Xiang, X.; Shi, C.; Guo, J.; Ran, T.; Lin, J.; Dong, F.; Yang, J.; Miao, H. Oxidative stress and inflammation in renal fibrosis: Novel molecular mechanisms and therapeutic targets. Chem. Interactions 2025, 421, 111784. [Google Scholar] [CrossRef]
- Fu, Y.; Xiang, Y.; Wu, W.; Cai, J.; Tang, C.; Dong, Z. Persistent Activation of Autophagy After Cisplatin Nephrotoxicity Promotes Renal Fibrosis and Chronic Kidney Disease. Front. Pharmacol. 2022, 13, 918732. [Google Scholar] [CrossRef]
- Jiao, H.; Zhang, M.; Chen, L.; Zhang, Z. Traditional Chinese Medicine targeting the TGF-β/Smad signaling pathway as a potential therapeutic strategy for renal fibrosis. Front. Pharmacol. 2025, 16, 1513329. [Google Scholar] [CrossRef]
- Zeng, Z.; Lao, B.; Liu, K.; Cao, Y.; Liao, Y.; Zhou, J. Review of the Delay of Chronic Kidney Disease Progression by Wuling San on Improving Renal Fibrosis. Basic Clin. Pharmacol. Toxicol. 2025, 137, e70071. [Google Scholar] [CrossRef]
- Fu, Y.; Tang, C.; Cai, J.; Chen, G.; Zhang, D.; Dong, Z. Rodent models of AKI-CKD transition. Am. J. Physiol. Physiol. 2018, 315, F1098–F1106. [Google Scholar] [CrossRef]
- Nogueira, A.; Pires, M.J.; Oliveira, P.A. Pathophysiological Mechanisms of Renal Fibrosis: A Review of Animal Models and Therapeutic Strategies. Vivo 2017, 31, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, A.; Ikeda, Y.; Yoshikawa, S.; Taniguchi, K.; Sawamura, H.; Morikawa, S.; Nakashima, M.; Asai, T.; Matsuda, S. The Tryptophan and Kynurenine Pathway Involved in the Development of Immune-Related Diseases. Int. J. Mol. Sci. 2023, 24, 5742. [Google Scholar] [CrossRef] [PubMed]
- Ramprasath, T.; Han, Y.-M.; Zhang, D.; Yu, C.-J.; Zou, M.-H. Tryptophan Catabolism and Inflammation: A Novel Therapeutic Target For Aortic Diseases. Front. Immunol. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Gagliardi, F.; De Domenico, P.; Snider, S.; Roncelli, F.; Comai, S.; Mortini, P. Immunomodulatory mechanisms driving tumor escape in glioblastoma: The central role of IDO and tryptophan metabolism in local and systemic immunotolerance. Crit. Rev. Oncol. 2025, 209, 104657. [Google Scholar] [CrossRef]
- Umeda, R.; Ito, Y.; Minatoguchi, S.; Koide, S.; Takahashi, K.; Hayashi, H.; Hasegawa, M.; Yuzawa, Y.; Yamamoto, Y.; Saito, K.; et al. Investigation of the Impact of Tryptophan-Metabolizing Enzymes and Kynurenic Acid on Antibody-Mediated Glomerulonephritis. FASEB J. 2025, 39, e71076. [Google Scholar] [CrossRef]
- Liu, L.; Su, X.; Quinn, W.J., 3rd; Hui, S.; Krukenberg, K.; Frederick, D.W.; Redpath, P.; Zhan, L.; Chellappa, K.; White, E. Quantitative Analysis of NAD Synthesis-Breakdown Fluxes. Cell Metab 2018, 27, 1067–1080 e5. [Google Scholar] [CrossRef]
- Satyanarayana, U.; Chakrapani, U. Biochemistry; Elsevier: Amsterdam, Netherlands, 2020. [Google Scholar]
- Fang, L.; Chen, H.; Kong, R.; Que, J. Endogenous tryptophan metabolite 5-Methoxytryptophan inhibits pulmonary fibrosis by downregulating the TGF-β/SMAD3 and PI3K/AKT signaling pathway. Life Sci. 2020, 260, 118399. [Google Scholar] [CrossRef]
- Morton, D.J. Production of Methoxyindoles In Vitro From Methoxytryptophan by Rat Pineal Gland. J. Pineal Res. 1987, 4, 7–11. [Google Scholar] [CrossRef]
- Su, Y.-C.; Wang, C.-C.; Weng, J.-H.; Yeh, S.-A.; Chen, P.-J.; Hwang, T.-Z.; Chen, H.-C. 5-Methoxytryptophan Sensitizing Head and Neck Squamous Carcinoma Cell to Cisplatitn Through Inhibiting Signal Transducer and Activator of Transcription 3 (STAT3). Front. Oncol. 2022, 12, 834941. [Google Scholar] [CrossRef]
- Yan, L.-J.; Wang, Y. Roles of Dihydrolipoamide Dehydrogenase in Health and Disease. Antioxidants Redox Signal. 2023, 39, 794–806. [Google Scholar] [CrossRef]
- Sumien, N.; Huang, R.; Chen, Z.; Vann, P.H.; Wong, J.M.; Li, W.; Yang, S.; Forster, M.J.; Yan, L.-J. Effects of dietary 5-methoxyindole-2-carboxylic acid on brain functional recovery after ischemic stroke. Behav. Brain Res. 2020, 378, 112278–112278. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Jin, Z.; Yang, X.; Yan, L.-J. Post-ischemic administration of 5-methoxyindole-2-carboxylic acid at the onset of reperfusion affords neuroprotection against stroke injury by preserving mitochondrial function and attenuating oxidative stress. Biochem. Biophys. Res. Commun. 2018, 497, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Li, R.; Li, W.; Ren, M.; Thangthaeng, N.; Sumien, N.; Liu, R.; Yang, S.; Simpkins, J.W.; Forster, M.J.; et al. Administration of 5-methoxyindole-2-carboxylic acid that potentially targets mitochondrial dihydrolipoamide dehydrogenase confers cerebral preconditioning against ischemic stroke injury. Free. Radic. Biol. Med. 2017, 113, 244–254. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.-H.; Kuo, C.-C.; Yan, J.-L.; Chen, H.-L.; Lin, W.-C.; Wang, K.-H.; Tsai, K.K.-C.; Guvén, H.; Flaberg, E.; Szekely, L.; et al. Control of cyclooxygenase-2 expression and tumorigenesis by endogenous 5-methoxytryptophan. Proc. Natl. Acad. Sci. 2012, 109, 13231–13236. [Google Scholar] [CrossRef]
- Wu, K.K.; Cheng, H.-H.; Chang, T.-C. 5-methoxyindole metabolites of L-tryptophan: control of COX-2 expression, inflammation and tumorigenesis. J. Biomed. Sci. 2014, 21, 17–17. [Google Scholar] [CrossRef]
- Deng, W.; Saunders, M.A.; Gilroy, D.W.; He, X.; Yeh, H.; Zhu, Y.; Shtivelband, M.I.; Ruan, K.; Wu, K.K. Purification and characterization of a cyclooxygenase-2 and angiogenesis suppressing factor produced by human fibroblasts. FASEB J. 2002, 16, 1286–1288. [Google Scholar] [CrossRef]
- Wu, K.K. Cytoguardin: A Tryptophan Metabolite against Cancer Growth and Metastasis. Int. J. Mol. Sci. 2021, 22, 4490. [Google Scholar] [CrossRef]
- Wang, Y.-F.; Hsu, Y.-J.; Wu, H.-F.; Lee, G.-L.; Yang, Y.-S.; Wu, J.-Y.; Yet, S.-F.; Wu, K.K.; Kuo, C.-C. Endothelium-Derived 5-Methoxytryptophan Is a Circulating Anti-Inflammatory Molecule That Blocks Systemic Inflammation. Circ. Res. 2016, 119, 222–236. [Google Scholar] [CrossRef]
- Kuo, T.-H.; Wu, P.-H.; Liu, P.-Y.; Chuang, Y.-S.; Tai, C.-J.; Kuo, M.-C.; Chiu, Y.-W.; Lin, Y.-T. Identification of Gut Microbiome Signatures Associated with Serotonin Pathway in Tryptophan Metabolism of Patients Undergoing Hemodialysis. Int. J. Mol. Sci. 2025, 26, 10463. [Google Scholar] [CrossRef]
- Lee, C.-M.; Chien, T.-C.R.; Wang, J.-S.; Chen, Y.-W.; Chen, C.-Y.; Kuo, C.-C.; Chiang, L.-T.; Wu, K.K.; Hsu, W.-T. 5-Methoxytryptophan attenuates oxidative stress-induced downregulation of PINK1 and mitigates mitochondrial damage and apoptosis in cardiac myocytes. Free. Radic. Biol. Med. 2025, 232, 398–411. [Google Scholar] [CrossRef]
- Lai, X.; Wan, X.; Bao, X.; Yang, Q.; Zhang, W.; Tan, Y.; Cai, X.; Wang, Z.; Li, Y.; Liu, C.; et al. 5-Methoxytryptophan attenuates hypobaric hypoxia induced acute lung injury by alleviating lipid peroxidation via targeting peroxiredoxin 6. Redox Biol. 2025, 88, 103922. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.K.; Kuo, C.-C.; Yet, S.-F.; Lee, C.-M.; Liou, J.-Y. 5-methoxytryptophan: an arsenal against vascular injury and inflammation. J. Biomed. Sci. 2020, 27, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.K. Control of Mesenchymal Stromal Cell Senescence by Tryptophan Metabolites. Int. J. Mol. Sci. 2021, 22, 697. [Google Scholar] [CrossRef] [PubMed]
- Ho, Y.-C.; Wu, M.-L.; Su, C.-H.; Chen, C.-H.; Ho, H.-H.; Lee, G.-L.; Lin, W.-S.; Lin, W.-Y.; Hsu, Y.-J.; Kuo, C.-C.; et al. A Novel Protective Function of 5-Methoxytryptophan in Vascular Injury. Sci. Rep. 2016, 6, 25374. [Google Scholar] [CrossRef]
- Chen, D.-Q.; Cao, G.; Chen, H.; Liu, D.; Su, W.; Yu, X.-Y.; Vaziri, N.D.; Liu, X.-H.; Bai, X.; Zhang, L.; et al. Gene and protein expressions and metabolomics exhibit activated redox signaling and wnt/β-catenin pathway are associated with metabolite dysfunction in patients with chronic kidney disease. Redox Biol. 2017, 12, 505–521. [Google Scholar] [CrossRef]
- Chen, D.-Q.; Cao, G.; Chen, H.; Argyopoulos, C.P.; Yu, H.; Su, W.; Chen, L.; Samuels, D.C.; Zhuang, S.; Bayliss, G.P.; et al. Identification of serum metabolites associating with chronic kidney disease progression and anti-fibrotic effect of 5-methoxytryptophan. Nat. Commun. 2019, 10, 1–15. [Google Scholar] [CrossRef]
- Mogos, M.; Socaciu, C.; Socaciu, A.I.; Vlad, A.; Gadalean, F.; Bob, F.; Milas, O.; Cretu, O.M.; Suteanu-Simulescu, A.; Glavan, M.; et al. Metabolomic Investigation of Blood and Urinary Amino Acids and Derivatives in Patients with Type 2 Diabetes Mellitus and Early Diabetic Kidney Disease. Biomedicines 2023, 11, 1527. [Google Scholar] [CrossRef]
- Peerapornratana, S.; Manrique-Caballero, C.L.; Gómez, H.; Kellum, J.A. Acute kidney injury from sepsis: current concepts, epidemiology, pathophysiology, prevention and treatment. Kidney Int. 2019, 96, 1083–1099. [Google Scholar] [CrossRef]
- Singh, A.P.; Muthuraman, A.; Jaggi, A.S.; Singh, N.; Grover, K.; Dhawan, R. Animal models of acute renal failure. Pharmacol. Rep. 2012, 64, 31–44. [Google Scholar] [CrossRef]
- Doi, K.; Leelahavanichkul, A.; Yuen, P.S.; Star, R.A. Animal models of sepsis and sepsis-induced kidney injury. J. Clin. Investig. 2009, 119, 2868–2878. [Google Scholar] [CrossRef]
- Gonçalves, G.M.; Zamboni, D.S.; Câmara, N.O.S. THE ROLE OF INNATE IMMUNITY IN SEPTIC ACUTE KIDNEY INJURIES. Shock 2010, 34, 22–26. [Google Scholar] [CrossRef]
- Sun, X.; Wang, H.; Liu, Y.; Yang, Y.; Wang, Y.; Liu, Y.; Ai, S.; Shan, Z.; Luo, P. 5-Methoxytryptophan Alleviates Lipopolysaccharide-Induced Acute Kidney Injury by Regulating Nrf2-Mediated Mitophagy. J. Inflamm. Res. 2024, ume 17, 9857–9873. [Google Scholar] [CrossRef] [PubMed]
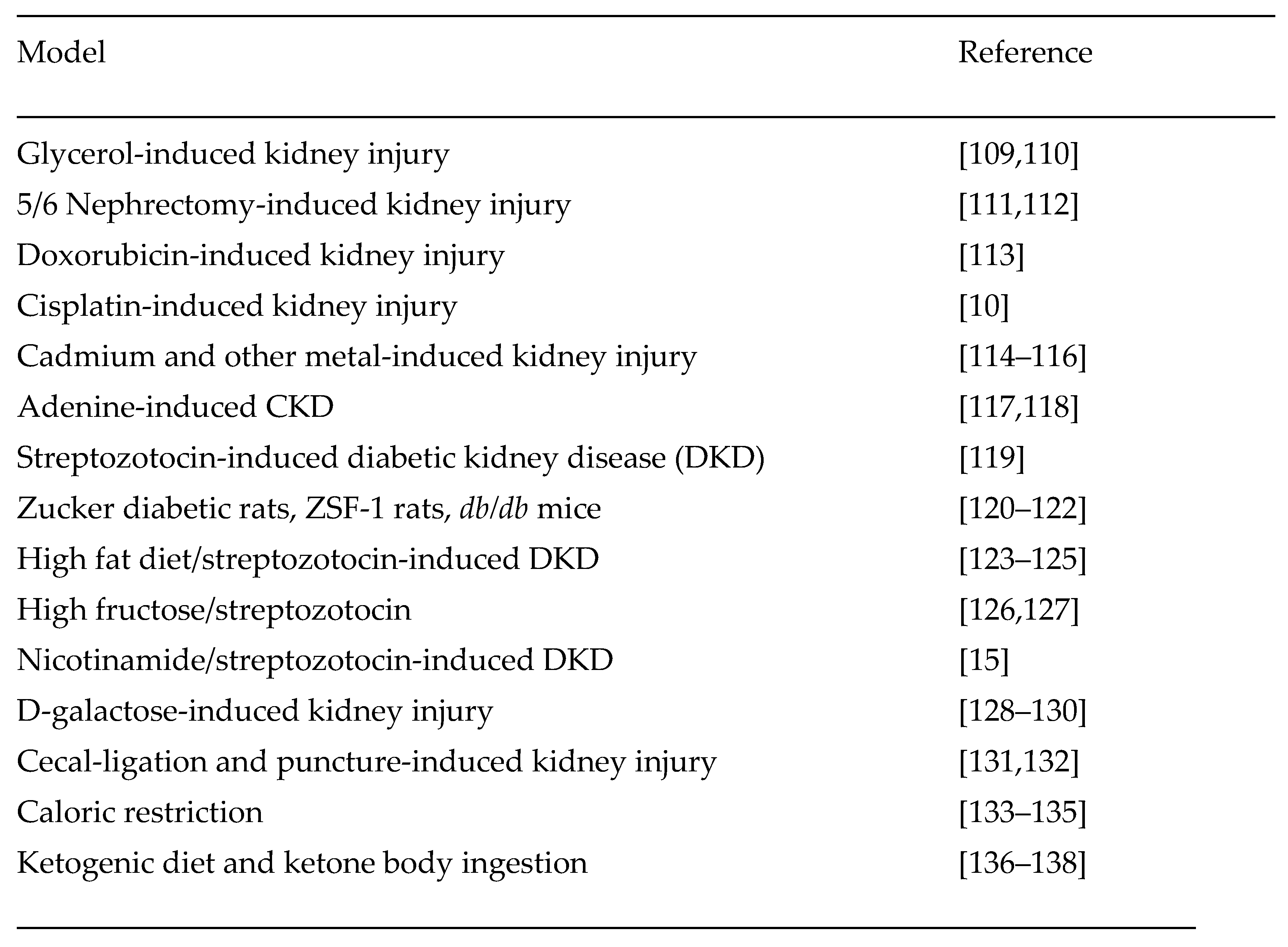
- Liu, H.; Diep, T.N.; Yan, L.-J. Mitigating Oxidative Stress and Inflammation: The Protective Role of β-lapachone in Kidney Disease. In Inflammation: From Chemical Mediators and Pathophysiology to Practice and Treatment; Wang, Y.-X., Ed.; Springer Nature Switzerland: Cham, 2025; pp. 1–18. [Google Scholar]
- Smith, T.; Zaidi, A.; Brown, C.V.M.; Pino-Chavez, G.; Bowen, T.; Meran, S.; Fraser, D.; Chavez, R.; Khalid, U. Robust Rat and Mouse Models of Bilateral Renal Ischemia Reperfusion Injury. Vivo 2024, 38, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Xiang, Y.; Wei, Q.; Ilatovskaya, D.; Dong, Z. Rodent models of AKI and AKI-CKD transition: an update in 2024. Am. J. Physiol. Physiol. 2024, 326, F563–F583. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, S.; Lu, B.; Daubert, R.A.; Chavin, K.D.; Wang, L.; Schnellmann, R.G. Suramin promotes recovery from renal ischemia/reperfusion injury in mice. Kidney Int. 2009, 75, 304–311. [Google Scholar] [CrossRef]
- Zhu, Y.B.; Zhang, Y.P.; Zhang, J.; Zhang, Y.B. Evaluation of Vitamin C Supplementation on Kidney Function and Vascular Reactivity Following Renal Ischemic Injury in Mice. Kidney Blood Press Res 2016, 41, 460–70. [Google Scholar] [CrossRef]
- Wu, L.; Li, Q.; Liu, S.; An, X.; Huang, Z.; Zhang, B.; Yuan, Y.; Xing, C. Protective effect of hyperoside against renal ischemia-reperfusion injury via modulating mitochondrial fission, oxidative stress, and apoptosis. Free. Radic. Res. 2019, 53, 727–736. [Google Scholar] [CrossRef]
- Li, S.; Yang, H.; Zhang, B.; Li, L.; Li, X. 5-methoxytryptophan ameliorates renal ischemia/reperfusion injury by alleviating endoplasmic reticulum stress-mediated apoptosis through the Nrf2/HO-1 pathway. Front. Pharmacol. 2025, 16, 1506482. [Google Scholar] [CrossRef]
- Li, X.; Ma, T.-K.; Wang, P.; Shi, H.; Hai, S.; Qin, Y.; Zou, Y.; Zhu, W.-T.; Li, H.-M.; Li, Y.-N.; et al. HOXD10 attenuates renal fibrosis by inhibiting NOX4-induced ferroptosis. Cell Death Dis. 2024, 15, 1–14. [Google Scholar] [CrossRef]
- Su, C.-T.; Jao, T.-M.; Urban, Z.; Huang, Y.-J.; See, D.H.W.; Tsai, Y.-C.; Lin, W.-C.; Huang, J.-W. LTBP4 affects renal fibrosis by influencing angiogenesis and altering mitochondrial structure. Cell Death Dis. 2021, 12, 1–11. [Google Scholar] [CrossRef]
- Miguel, V.; Ramos, R.; García-Bermejo, L.; Rodríguez-Puyol, D.; Lamas, S. The program of renal fibrogenesis is controlled by microRNAs regulating oxidative metabolism. Redox Biol. 2021, 40, 101851. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.-Y.; Lee, G.-L.; Chueh, Y.-F.; Kuo, C.-C.; Hsu, Y.-J.; Wu, K.K. 5-Methoxytryptophan Protects against Toll-Like Receptor 2-Mediated Renal Tissue Inflammation and Fibrosis in a Murine Unilateral Ureteral Obstruction Model. J. Innate Immun. 2025, 17, 78–94. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Fu, L.; Li, M.; Xie, K.; Li, X.; Zhou, X.-J.; Yang, L.; Zhang, L.; Xue, C.; Mao, Z. Decreased brain-derived neurotrophic factor expression in chronic kidney disease: integrated clinical and experimental evidence. Front. Mol. Biosci. 2025, 12, 1627534. [Google Scholar] [CrossRef] [PubMed]
- Ito, H.; Mori, T. The brain-kidney axis in cerebral small vessel disease: chronic kidney disease as an active mediator, not just a confounder. Hypertens. Res. 2025, 48, 3309–3310. [Google Scholar] [CrossRef]
- Heo, C.M.; Yi, J.; Lee, D.A.; Park, K.M.; Lee, Y.J.; Park, S.; Kim, Y.W.; Ko, J.; Djojo, A.Y.; Park, B.S. Analysis of functional brain connectivity in patient with end-stage kidney disease undergoing peritoneal dialysis using functional near infrared spectroscopy. PLOS ONE 2025, 20, e0323319. [Google Scholar] [CrossRef]
- Xu, C.; Miao, H.; Chen, Y.; Liao, L. Crosstalk of kidney and brain in diabetes-related cognitive impairment and therapeutic strategies. Front. Endocrinol. 2025, 16, 1562518. [Google Scholar] [CrossRef]
- Xie, Z.; Tong, S.; Chu, X.; Feng, T.; Geng, M. Chronic Kidney Disease and Cognitive Impairment: The Kidney-Brain Axis. Kidney Dis. 2022, 8, 275–285. [Google Scholar] [CrossRef]
- Yan, Q.; Liu, M.; Xie, Y.; Lin, Y.; Fu, P.; Pu, Y.; Wang, B. Kidney-brain axis in the pathogenesis of cognitive impairment. Neurobiol. Dis. 2024, 200, 106626. [Google Scholar] [CrossRef]
- Zhou, X.; Sun, Y.; Yang, G. 5-Methoxytryptophan improves cerebrovascular injury induced by chronic kidney disease through NF-κB pathway. Vitr. Cell. Dev. Biol.-Anim. 2025, 61, 774–788. [Google Scholar] [CrossRef]
- Sun, X.; Liu, Y.; Liu, Y.; Ai, S.; Luo, P. Predictive Value of 5-Methoxytryptophan on Clinical Outcome in Patients with Sepsis Associated Acute Kidney Injury. J. Inflamm. Res. 2025, ume 18, 6865–6875. [Google Scholar] [CrossRef]
- Jia, Y.; Shi, Y.; Wang, J.; Liu, H.; Wang, H.; Huang, Y.; Liu, Y.; Chen, P.; Peng, J. Astragalin attenuates caerulein-induced acute pancreatitis by targeting the NLRP3 signaling pathway and gut microbiota. Bioresour. Bioprocess. 2025, 12, 139. [Google Scholar] [CrossRef]
- Pan, H.; Yang, S.; Kulyar, M.F.; Ma, H.; Li, K.; Zhang, L.; Mo, Q.; Li, J. Lactobacillus fermentum 016 Alleviates Mice Colitis by Modulating Oxidative Stress, Gut Microbiota, and Microbial Metabolism. Nutrients 2025, 17. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Lu, H.; Li, Y.; Xu, Q.; Ma, Y.; Lou, A.; Cui, W.; Song, W.; Qu, P.; Chen, Z.; et al. ACE2 shedding exacerbates sepsis-induced gut leak via loss of microbial metabolite 5-methoxytryptophan. Microbiome 2025, 13, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Wang, X.; Zhang, Y.; Xie, Y.; Cui, R.; Xia, J.; Sun, Z. Renal Metabolomics Study and Critical Pathway Validation of Shenkang Injection in the Treatment of Chronic Renal Failure. Biol. Pharm. Bull. 2024, 47, 499–508. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, R.M. Methionine Restriction and Life-Span Extension. Methods Mol Biol 2019, 1866, 263–266. [Google Scholar]
- Green, C.L.; Trautman, M.E.; Chaiyakul, K.; Jain, R.; Alam, Y.H.; Babygirija, R.; Pak, H.H.; Sonsalla, M.M.; Calubag, M.F.; Yeh, C.-Y.; et al. Dietary restriction of isoleucine increases healthspan and lifespan of genetically heterogeneous mice. Cell Metab. 2023, 35, 1976–1995.e6. [Google Scholar] [CrossRef]
- Huang, X.; Zhang, F.; Yang, Y.; Liu, J.; Tan, X.; Zhou, P.; Tang, X.; Hu, J.; Chen, L.; Yuan, M.; et al. Curcumin-copper complex nanoparticles as antioxidant nanozymes for acute kidney injury alleviation. Mater. Today Bio 2025, 32, 101794. [Google Scholar] [CrossRef]
- Hu, K.; Wang, P.; Zhao, Y.; Zhang, T.; Zhao, L.; Su, Y.; Wen, H.; Liu, C.; Zou, G.; Wei, L.; et al. Hypoxia-responsive modules via tunable hydrophobicity reversal enhance renal-targeted release of CD36-interfering nanoparticles to ameliorate acute kidney injury. J. Control. Release 2025, 389, 114453. [Google Scholar] [CrossRef]
- Verhoven, B.; Tong, Y.; Chlebeck, P.; Zhong, W.; Zeng, W.; Jennings, H.; Miller, B.; Heise, G.; Levitsky, M.; Xie, R.; et al. Attenuating Ischemia and Reperfusion Injury Using NAD+-Loaded Nanoparticles in Mouse Kidneys. Transplant. Direct 2025, 12, e1890. [Google Scholar] [CrossRef]
- Wu, J.; Jin, Z.; Zheng, H.; Yan, L.-J. Sources and implications of NADH/NAD+ redox imbalance in diabetes and its complications. Diabetes, Metab. Syndr. Obes. 2016, ume 9, 145–153. [Google Scholar] [CrossRef]
- Alaygut, D.; Ozturk, I.; Ulu, S.; Gungor, O. NETosis and kidney disease: what do we know? Int. Urol. Nephrol. 2023, 55, 1985–1994. [Google Scholar] [CrossRef]
- Hammouri, D.; Weis, T.; Siskind, L.J. Macrophage Plasticity and Functional Dynamics in Acute Kidney Injury and Its Progression to Chronic Kidney Disease. Semin. Nephrol. 2025, 151672. [Google Scholar] [CrossRef] [PubMed]
- Gorji, A.V.; Fakhredini, F.; Soltanpour, F.; Mansouri, E. Investigating the Effect of Secretome Secreted from Adult Rat Kidney-Derived Stem Cells on the Expression of miR375, miR494 and Tissue Changes in Glycerol-Induced Acute Kidney Injury. Adv. Biomed. Res. 2025, 14, 48. [Google Scholar] [CrossRef]
- Abugomaa, A.; Elbadawy, M. Olive leaf extract modulates glycerol-induced kidney and liver damage in rats. Environ. Sci. Pollut. Res. 2020, 27, 22100–22111. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Xia, J.; Shi, C.; Zhang, K.; Qu, Y.; He, G.; Fu, Z.; Deng, L.; Liu, R.; Wang, X.; et al. Direct pharmacological targeting of Piezo1 by Paeoniflorin: a novel therapeutic approach for renal fibrosis. J. Adv. Res. 2025. [Google Scholar] [CrossRef]
- Zhang, X.; Li, S.; Li, S.; Liu, B.; Ding, G.; Wu, M.; Zhang, Y.; Huang, S.; Gong, W.; Jia, Z.; et al. Endothelial Lon protease 1 facilitates the redox balance to prevent glomerulosclerosis by acting on superoxide dismutase 2 ubiquitination. Redox Biol. 2025, 88, 103929. [Google Scholar] [CrossRef]
- Zhang, Q.; Wu, G.; Guo, S.; Liu, Y.; Liu, Z. Effects of tristetraprolin on doxorubicin (adriamycin)-induced experimental kidney injury through inhibiting IL-13/STATE signal pathway. 2020, 12, 1203–1221. [Google Scholar]
- Kimura, A.; Ishida, Y.; Hayashi, T.; Wada, T.; Yokoyama, H.; Sugaya, T.; Mukaida, N.; Kondo, T. Interferon-γ Plays Protective Roles in Sodium Arsenite-Induced Renal Injury by Up-Regulating Intrarenal Multidrug Resistance-Associated Protein 1 Expression. Am. J. Pathol. 2006, 169, 1118–1128. [Google Scholar] [CrossRef]
- Riaz, M.A.; Nisa, Z.U.; Mehmood, A.; Anjum, M.S.; Shahzad, K. Metal-induced nephrotoxicity to diabetic and non-diabetic Wistar rats. Environ. Sci. Pollut. Res. 2019, 26, 31111–31118. [Google Scholar] [CrossRef]
- Yan, L.-J.; Allen, D.C. Cadmium-Induced Kidney Injury: Oxidative Damage as a Unifying Mechanism. Biomolecules 2021, 11, 1575. [Google Scholar] [CrossRef]
- Diwan, V.; Brown, L.; Gobe, G.C. Adenine-induced chronic kidney disease in rats. Nephrology 2017, 23, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Ho, H.J.; Kikuchi, K.; Oikawa, D.; Watanabe, S.; Kanemitsu, Y.; Saigusa, D.; Kujirai, R.; Ikeda-Ohtsubo, W.; Ichijo, M.; Akiyama, Y.; et al. SGLT-1-specific inhibition ameliorates renal failure and alters the gut microbial community in mice with adenine-induced renal failure. Physiol Rep 2021, 9, e15092. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wang, Y.; Wang, Y.; Yan, L.-J. Rodent Models of Streptozotocin-Induced Diabetes as Suitable Paradigms for Studying Diabetic Kidney Disease. Free Radicals and Antioxidants 2024, 14, 32–33. [Google Scholar] [CrossRef]
- Ikeda, Y.; Enomoto, H.; Tajima, S.; Izawa-Ishizawa, Y.; Kihira, Y.; Ishizawa, K.; Tomita, S.; Tsuchiya, K.; Tamaki, T. Dietary iron restriction inhibits progression of diabetic nephropathy in db/db mice. Am J Physiol Renal Physiol 2013, 304, F1028–36. [Google Scholar] [CrossRef]
- Agil, A.; Chayah, M.; Visiedo, L.; Navarro-Alarcon, M.; Ferrer, J.M.R.; Tassi, M.; Reiter, R.J.; Fernández-Vázquez, G. Melatonin Improves Mitochondrial Dynamics and Function in the Kidney of Zücker Diabetic Fatty Rats. J. Clin. Med. 2020, 9, 2916. [Google Scholar] [CrossRef]
- Li, C.-Y.; Ma, W.-X.; Yan, L.-J. 5-Methoxyindole-2-Carboylic Acid (MICA) Fails to Retard Development and Progression of Type II Diabetes in ZSF1 Diabetic Rats. React. Oxyg. Species 2020, 9, 144–147–144–147. [Google Scholar] [CrossRef]
- Zhai, L.; Gu, J.; Yang, D.; Hu, W.; Wang, W.; Ye, S. Metformin ameliorates podocyte damage by restoring renal tissue nephrin expression in type 2 diabetic rats. J. Diabetes 2016, 9, 510–517. [Google Scholar] [CrossRef]
- Skovsø, S. Modeling type 2 diabetes in rats using high fat diet and streptozotocin. J. Diabetes Investig. 2014, 5, 349–358. [Google Scholar] [CrossRef]
- Glastras, S.J.; Chen, H.; Teh, R.; McGrath, R.T.; Chen, J.; A Pollock, C.; Wong, M.G.; Saad, S. Mouse Models of Diabetes, Obesity and Related Kidney Disease. PLOS ONE 2016, 11, e0162131–e0162131. [Google Scholar] [CrossRef]
- Wilson, R.D.; Islam, M.S. Fructose-fed streptozotocin-injected rat: an alternative model for type 2 diabetes. Pharmacol Rep 2012, 64, 129–39. [Google Scholar] [CrossRef]
- Pan, X.; Olatunji, O.J.; Basit, A.; Sripetthong, S.; Nalinbenjapun, S.; Ovatlarnporn, C. Insights into the phytochemical profiling, antidiabetic and antioxidant potentials of Lepionurus sylvestris Blume extract in fructose/streptozotocin-induced diabetic rats. Front. Pharmacol. 2024, 15, 1424346. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Ling, X.; Meng, P.; Liang, Y.; Shen, K.; Wu, Q.; Zhang, Y.; Chen, Q.; Chen, S.; Liu, Y.; et al. Cannabinoid receptor 2 plays a central role in renal tubular mitochondrial dysfunction and kidney ageing. J. Cell. Mol. Med. 2021, 25, 8957–8972. [Google Scholar] [CrossRef] [PubMed]
- Azman, K.F.; Zakaria, R. d-Galactose-induced accelerated aging model: an overview. Biogerontology 2019, 20, 763–782. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Wu, P.; Li, Q.; Feng, Q.; Lin, X.; Luo, Y.; Wang, Y.; Wu, M.; Cai, F.; Zhang, J.; et al. NUAK1 Promotes Diabetic Kidney Disease by Accelerating Renal Tubular Senescence via the ROS/P53 Axis. Diabetes 2025, 74, 2405–2417. [Google Scholar] [CrossRef]
- Wang, Q.-L.; Xing, W.; Yu, C.; Gao, M.; Deng, L.-T. ROCK1 regulates sepsis-induced acute kidney injury via TLR2-mediated endoplasmic reticulum stress/pyroptosis axis. Mol. Immunol. 2021, 138, 99–109. [Google Scholar] [CrossRef]
- Arslan, M.; Küçük, A.; Bozok, Ü.G.; Ergörün, A.I.; Sezen, Ş.C.; Yavuz, A.; Kavutçu, M. The role of pomegranate seed oil on kidney and lung tissues in the treatment of sepsis: animal pre-clinical research. J. Infect. Dev. Ctries. 2023, 17, 1791–1797. [Google Scholar] [CrossRef]
- Wang, X.-H.; Ao, Q.-G.; Cheng, Q.-L. Caloric restriction inhibits renal artery ageing by reducing endothelin-1 expression. Ann. Transl. Med. 2021, 9, 979–979. [Google Scholar] [CrossRef]
- Liu, J.-R.; Cai, G.-Y.; Ning, Y.-C.; Wang, J.-C.; Lv, Y.; Guo, Y.-N.; Fu, B.; Hong, Q.; Sun, X.-F.; Chen, X.-M. Caloric restriction alleviates aging-related fibrosis of kidney through downregulation of miR-21 in extracellular vesicles. Aging 2020, 12, 18052–18072. [Google Scholar] [CrossRef]
- Serna, J.D.C.; Amaral, A.G.; da Silva, C.C.C.; Munhoz, A.C.; Vilas-Boas, E.A.; Menezes-Filho, S.L.; Kowaltowski, A.J. Regulation of kidney mitochondrial function by caloric restriction. Am. J. Physiol. Physiol. 2022, 323, F92–F106. [Google Scholar] [CrossRef]
- Athinarayanan, S.J.; Roberts, C.G.P.; Vangala, C.; Shetty, G.K.; McKenzie, A.L.; Weimbs, T.; Volek, J.S. The case for a ketogenic diet in the management of kidney disease. BMJ Open Diabetes Res. Care 2024, 12, e004101. [Google Scholar] [CrossRef]
- Mikami, D.; Kobayashi, M.; Uwada, J.; Yazawa, T.; Kamiyama, K.; Nishimori, K.; Nishikawa, Y.; Morikawa, Y.; Yokoi, S.; Takahashi, N.; et al. β-Hydroxybutyrate, a ketone body, reduces the cytotoxic effect of cisplatin via activation of HDAC5 in human renal cortical epithelial cells. Life Sci. 2019, 222, 125–132. [Google Scholar] [CrossRef]
- Liu, H.; Yan, L.-J. The Role of Ketone Bodies in Various Animal Models of Kidney Disease. Endocrines 2023, 4, 236–249. [Google Scholar] [CrossRef]
- Yan, L. Folic acid-induced animal model of kidney disease. Anim. Model. Exp. Med. 2021, 4, 329–342. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





