Submitted:
13 November 2025
Posted:
14 November 2025
You are already at the latest version
Abstract
Cyanobacteria such as Synechocystis sp. PCC 6803 are promising chassis for sustainable bioproduction. During nitrogen starvation, Synechocystis redirects fixed carbon from biomass growth toward glycogen accumulation as a carbon and energy reserve. Inhibiting glycogen synthesis results in the excretion of excess carbon as organic acids, predominantly pyruvate and 2-oxoglutarate. Efficiently rerouting this carbon toward the formation of value-added products such as the plastic alternative polyhydroxybutyrate requires a deeper understanding of carbon partitioning and overflow metabolism. To investigate this, we quantified intra- and extracellular metabolites in Synechocystis wild-type and mutant strains with altered glycogen metabolism (Δpgm, ΔglgC, ΔglgA1, ΔglgA2), nitrogen signaling (ΔglnB), and carbon allocation (ΔpirC), including the double mutant ΔglgCΔpirC. Metabolites were analyzed after two days of nitrogen-replete or -depleted growth using enzymatic glycogen quantification and liquid chromatography–mass spectrometry. Excretion was primarily triggered by inhibition of glycogen synthesis but modulated by other changes in carbon flow, such as pirC deletion. Besides pyruvate and 2-oxoglutarate, small amounts of glutamate, succinate, and malate were excreted. Our findings suggest that, rather than a passive consequence of metabolite accumulation, excretion is a selective, threshold-dependent process that limits intracellular metabolite buildup, revealing an additional layer of metabolic control relevant to cyanobacterial bioengineering.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cultivation of Synechocystis sp. PCC 6803
2.2. Glycogen Quantification
2.3. Quantification of Intracellular Metabolites
2.4. Quantification of Extracellular Metabolites
3. Results
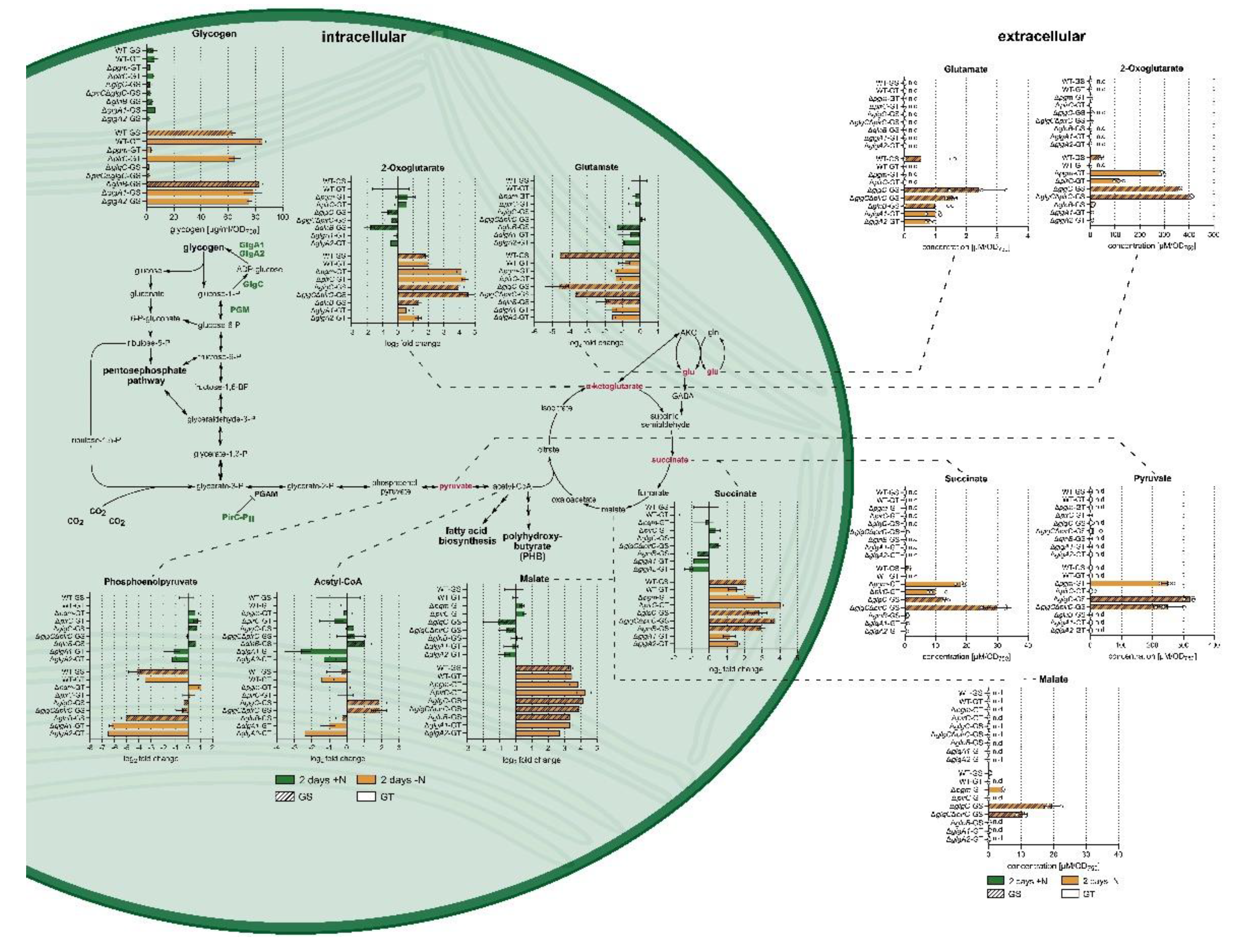
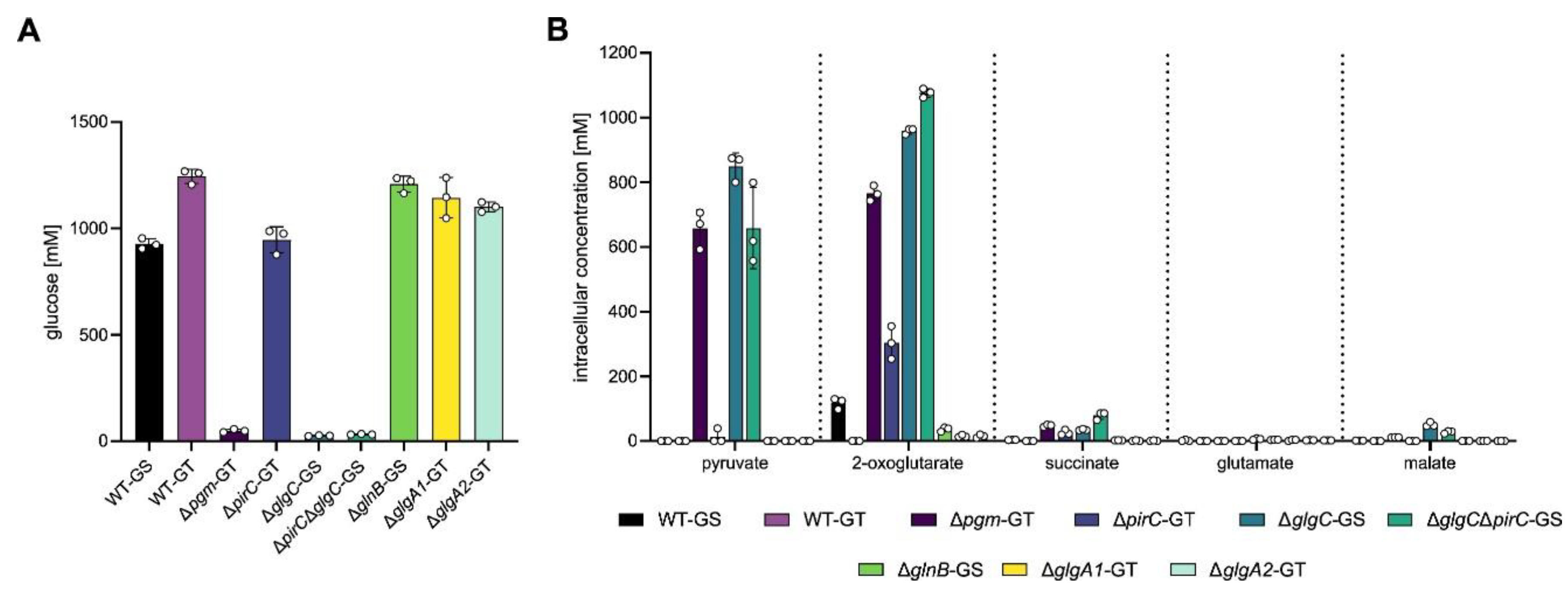
3.1. Extracellular Metabolite Levels Do Not Simply Mirror Intracellular Metabolite Levels
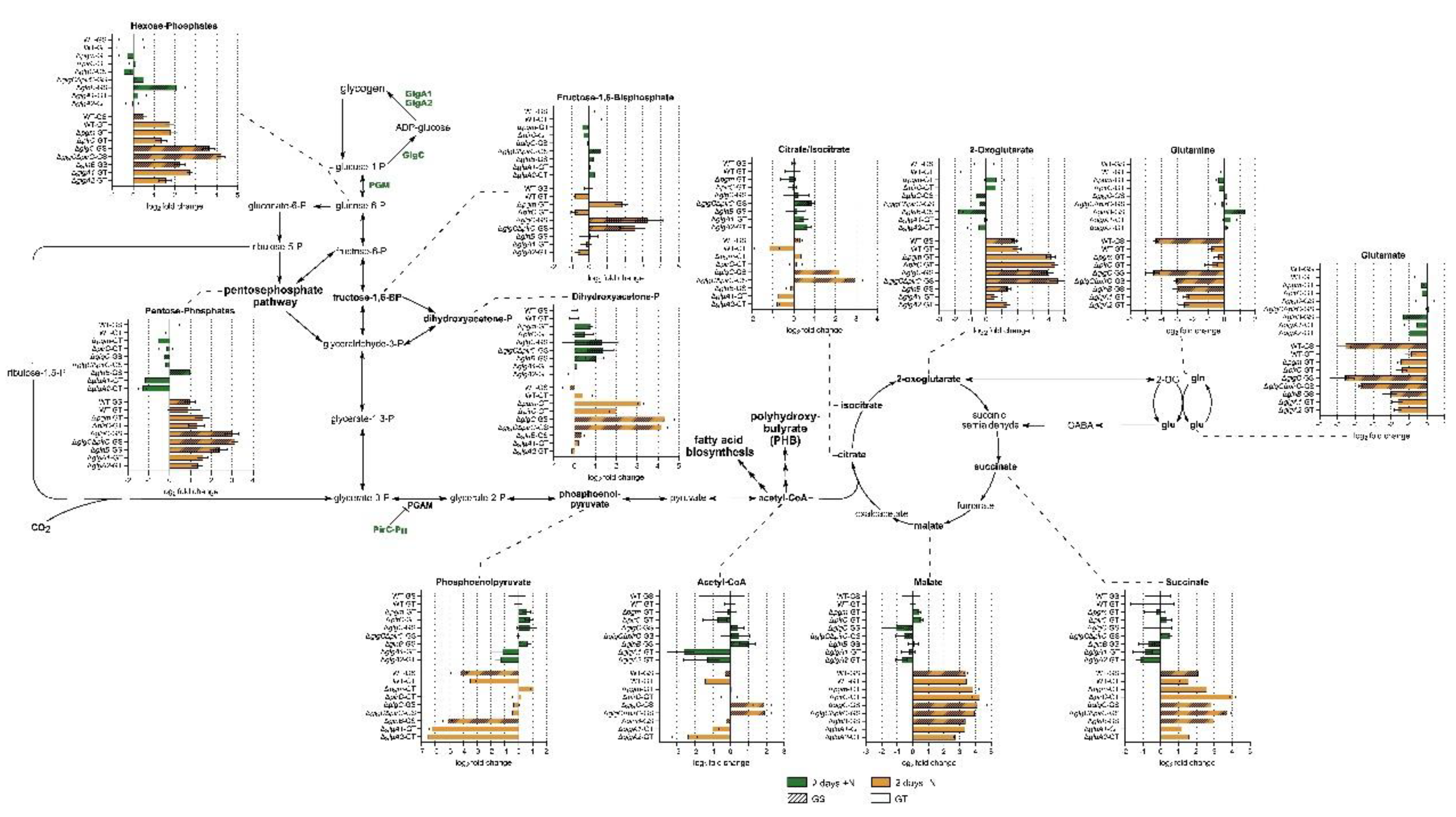
3.2. Intracellular Central Carbon and Nitrogen Metabolism
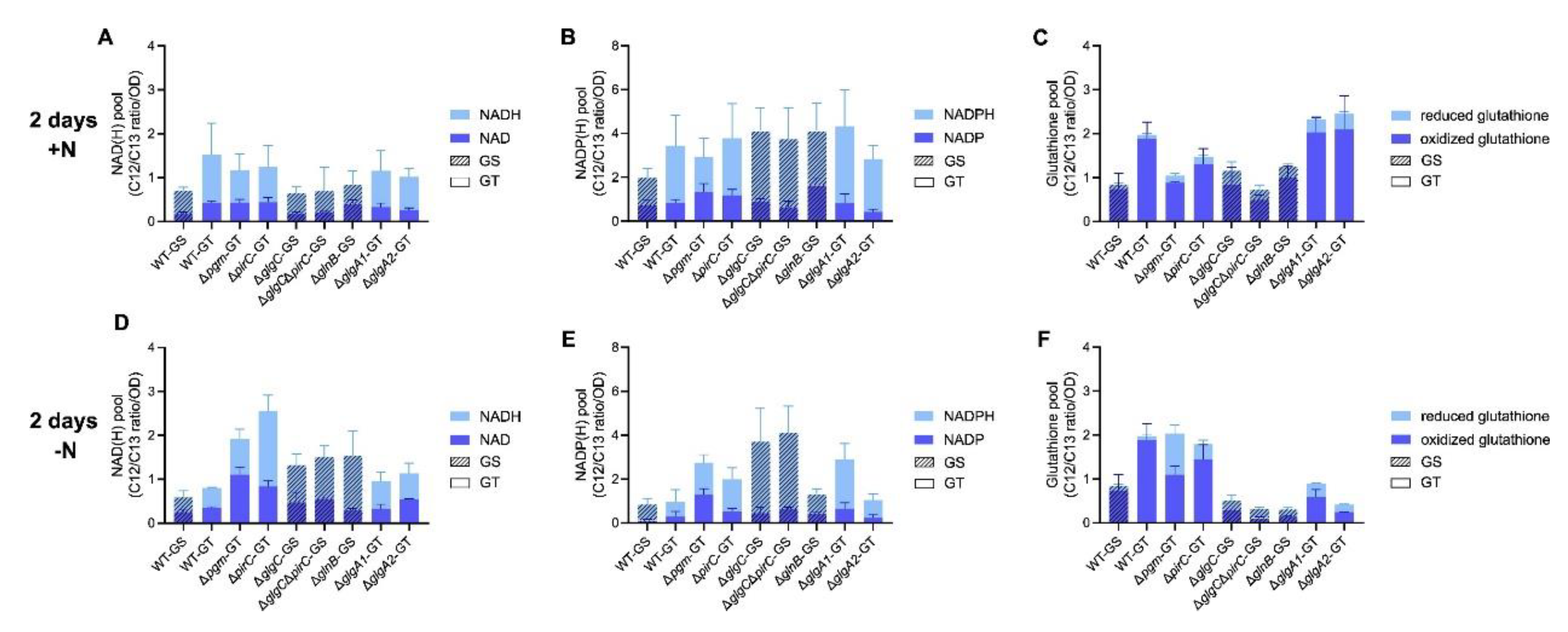
3.3. Intracellular Energy and Redox Balance
3.4. Metabolite Excretion Represents a Major Alternative Carbon Sink
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 2-OG | 2-oxoglutarate |
| AICAR | 5-amino-1-(5-phospho-D-ribosyl)imidazole-4-carboxamide |
| AMG | amyloglucosidase |
| DHAP | dihydroxyacetone-phosphate |
| GlgA | glycogen synthase |
| GlgC | glucose-1-phosphate adenylyltransferase |
| GO | glucose oxidase |
| GS | glucose-sensitive |
| GS/GOGAT | glutamine synthetase/glutamate synthase cycle |
| GT | glucose-tolerant |
| HPLC-MS | high-performance liquid chromatography-mass spectrometry |
| LC-MS/MS | liquid chromatography-tandem mass spectrometry |
| MG | methylglyoxal |
| OD | optical density |
| ODD | o-dianisidine dihydrochloride |
| PEP | phosphoenolpyruvate |
| PGAM | phosphoglycerate mutase |
| PGM | phosphoglucomutase |
| PHB | polyhydroxybutyrate |
| PirC | PII-interacting regulator of carbon metabolism |
| PX | peroxidase |
| SD | standard deviation |
| Synechocystis | Synechocystis sp. PCC 6803 |
| TCA | tricarboxylic acid |
| WT | wild type |
| WT-GS | glucose-sensitive wild type |
| WT-GT | glucose-tolerant wild type |
References
- Blankenship, R.E. How Cyanobacteria went green. Science 2017, 355, 1372–1373. [Google Scholar] [CrossRef]
- Mulkidjanian, A.Y.; Koonin, E.V.; Makarova, K.S.; Mekhedov, S.L.; Sorokin, A.; Wolf, Y.I.; Dufresne, A.; Partensky, F.; Burd, H.; Kaznadzey, D.; et al. The cyanobacterial genome core and the origin of photosynthesis. Proceedings of the National Academy of Sciences 2006, 103, 13126–13131. [Google Scholar] [CrossRef]
- Jaiswal, D.; Sahasrabuddhe, D.; Wangikar, P.P. Cyanobacteria as cell factories: the roles of host and pathway engineering and translational research. Current Opinion in Biotechnology 2022, 73, 314–322. [Google Scholar] [CrossRef]
- Toepel, J.; Karande, R.; Klähn, S.; Bühler, B. Cyanobacteria as whole-cell factories: current status and future prospectives. Current Opinion in Biotechnology 2023, 80, 102892. [Google Scholar] [CrossRef] [PubMed]
- Koch, M.; Bruckmoser, J.; Scholl, J.; Hauf, W.; Rieger, B.; Forchhammer, K. Maximizing PHB content in Synechocystis sp. PCC 6803: a new metabolic engineering strategy based on the regulator PirC. Microbial Cell Factories 2020, 19, 231. [Google Scholar] [CrossRef]
- Koch, M.; Doello, S.; Gutekunst, K.; Forchhammer, K. PHB is Produced from Glycogen Turn-over during Nitrogen Starvation in Synechocystis sp. PCC 6803. International Journal of Molecular Sciences 2019, 20. [Google Scholar] [CrossRef]
- Anfelt, J.; Kaczmarzyk, D.; Shabestary, K.; Renberg, B.; Rockberg, J.; Nielsen, J.; Uhlén, M.; Hudson, E.P. Genetic and nutrient modulation of acetyl-CoA levels in Synechocystis for n-butanol production. Microbial Cell Factories 2015, 14, 167. [Google Scholar] [CrossRef]
- van der Woude, A.D.; Angermayr, S.A.; Puthan Veetil, V.; Osnato, A.; Hellingwerf, K.J. Carbon sink removal: Increased photosynthetic production of lactic acid by Synechocystis sp. PCC6803 in a glycogen storage mutant. Journal of Biotechnology 2014, 184, 100–102. [Google Scholar] [CrossRef]
- Huang, C.-H.; Shen, C.R.; Li, H.; Sung, L.-Y.; Wu, M.-Y.; Hu, Y.-C. CRISPR interference (CRISPRi) for gene regulation and succinate production in cyanobacterium S. elongatus PCC 7942. Microbial Cell Factories 2016, 15, 196. [Google Scholar] [CrossRef] [PubMed]
- Klotz, A.; Georg, J.; Bučinská, L.; Watanabe, S.; Reimann, V.; Januszewski, W.; Sobotka, R.; Jendrossek, D.; Hess, Wolfgang R.; Forchhammer, K. Awakening of a Dormant Cyanobacterium from Nitrogen Chlorosis Reveals a Genetically Determined Program. Current Biology 2016, 26, 2862–2872. [CrossRef] [PubMed]
- Elmorjani, K.; Herdman, M. Metabolic Control of Phycocyanin Degradation in the Cyanobacterium Synechocystis PCC 6803: a Glucose Effect. Microbiology 1987, 133, 1685–1694. [Google Scholar] [CrossRef]
- Forchhammer, K.; Schwarz, R. Nitrogen chlorosis in unicellular cyanobacteria – a developmental program for surviving nitrogen deprivation. Environmental Microbiology 2019, 21, 1173–1184. [Google Scholar] [CrossRef]
- Krasikov, V.; Aguirre von Wobeser, E.; Dekker, H.L.; Huisman, J.; Matthijs, H.C.P. Time-series resolution of gradual nitrogen starvation and its impact on photosynthesis in the cyanobacterium Synechocystis PCC 6803. Physiologia Plantarum 2012, 145, 426–439. [Google Scholar] [CrossRef] [PubMed]
- Görl, M.; Sauer, J.; Baier, T.; Forchhammer, K. Nitrogen-starvation-induced chlorosis in Synechococcus PCC 7942: adaptation to long-term survival. Microbiology 1998, 144, 2449–2458. [Google Scholar] [CrossRef] [PubMed]
- Neumann, N.; Doello, S.; Forchhammer, K. Recovery of Unicellular Cyanobacteria from Nitrogen Chlorosis: A Model for Resuscitation of Dormant Bacteria. Microbial Physiology 2021, 31, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Doello, S.; Neumann, N.; Forchhammer, K. Regulatory phosphorylation event of phosphoglucomutase 1 tunes its activity to regulate glycogen metabolism. The FEBS Journal 2022, 289, 6005–6020. [Google Scholar] [CrossRef]
- Gründel, M.; Scheunemann, R.; Lockau, W.; Zilliges, Y. Impaired glycogen synthesis causes metabolic overflow reactions and affects stress responses in the cyanobacterium Synechocystis sp. PCC 6803. Microbiology 2012, 158, 3032–3043. [Google Scholar] [CrossRef]
- Kato, Y.; Hidese, R.; Matsuda, M.; Ohbayashi, R.; Ashida, H.; Kondo, A.; Hasunuma, T. Glycogen deficiency enhances carbon partitioning into glutamate for an alternative extracellular metabolic sink in cyanobacteria. Communications Biology 2024, 7, 233. [Google Scholar] [CrossRef]
- Carrieri, D.; Paddock, T.; Maness, P.-C.; Seibert, M.; Yu, J. Photo-catalytic conversion of carbon dioxide to organic acids by a recombinant cyanobacterium incapable of glycogen storage. Energy & Environmental Science 2012, 5, 9457–9461. [Google Scholar] [CrossRef]
- Russell, J.B. The energy spilling reactions of bacteria and other organisms. Journal of molecular microbiology and biotechnology 2007, 13, 1–11. [Google Scholar] [CrossRef]
- Paczia, N.; Nilgen, A.; Lehmann, T.; Gätgens, J.; Wiechert, W.; Noack, S. Extensive exometabolome analysis reveals extended overflow metabolism in various microorganisms. Microbial cell factories 2012, 11, 122. [Google Scholar] [CrossRef] [PubMed]
- Cano, M.; Holland, S.C.; Artier, J.; Burnap, R.L.; Ghirardi, M.; Morgan, J.A.; Yu, J. Glycogen Synthesis and Metabolite Overflow Contribute to Energy Balancing in Cyanobacteria. Cell Reports 2018, 23, 667–672. [Google Scholar] [CrossRef]
- Orthwein, T.; Scholl, J.; Spät, P.; Lucius, S.; Koch, M.; Macek, B.; Hagemann, M.; Forchhammer, K. The novel PII-interactor PirC identifies phosphoglycerate mutase as key control point of carbon storage metabolism in cyanobacteria. Proceedings of the National Academy of Sciences 2021, 118, e2019988118. [Google Scholar] [CrossRef]
- Orthwein, T.; Alford Janette, T.; Becker Nathalie, S.; Fink, P.; Forchhammer, K. Structural elements of cyanobacterial co-factor-independent phosphoglycerate mutase that mediate regulation by PirC. mBio 2025, 16, e03378–03324. [Google Scholar] [CrossRef]
- Forchhammer, K. Global carbon/nitrogen control by PII signal transduction in cyanobacteria: from signals to targets. FEMS Microbiology Reviews 2004, 28, 319–333. [Google Scholar] [CrossRef]
- Rippka, R.; Deruelles, J.; Waterbury, J.B.; Herdman, M.; Stanier, R.Y. Generic Assignments, Strain Histories and Properties of Pure Cultures of Cyanobacteria. Microbiology 1979, 111, 1–61. [Google Scholar] [CrossRef]
- Schlebusch, M.; Forchhammer, K. Requirement of the Nitrogen Starvation-Induced Protein Sll0783 for Polyhydroxybutyrate Accumulation in <i>Synechocystis</i> sp. Strain PCC 6803. Applied and Environmental Microbiology 2010, 76, 6101–6107. [Google Scholar] [CrossRef]
- Vidal, R.; Venegas-Calerón, M. Simple, fast and accurate method for the determination of glycogen in the model unicellular cyanobacterium Synechocystis sp. PCC 6803. Journal of Microbiological Methods 2019, 164, 105686. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Martínez, P.; Roldán, M.; Díaz-Troya, S.; Florencio, F.J. Stress response requires an efficient connection between glycogen and central carbon metabolism by phosphoglucomutases in cyanobacteria. Journal of Experimental Botany 2022, 74, 1532–1550. [Google Scholar] [CrossRef]
- Doello, S.; Sauerwein, J.; von Manteuffel, N.; Burkhardt, M.; Neumann, N.; Appel, J.; Rapp, J.; Just, P.; Link, H.; Gutekunst, K.; et al. Metabolite-level regulation of enzymatic activity controls awakening of cyanobacteria from metabolic dormancy. Current Biology 2025, 35, 77–86.e74. [Google Scholar] [CrossRef] [PubMed]
- Guder, J.C.; Schramm, T.; Sander, T.; Link, H. Time-optimized isotope ratio LC–MS/MS for high-throughput quantification of primary metabolites. Analytical Chemistry 2017, 89, 1624–1631. [Google Scholar] [CrossRef]
- Akiyama, M.; Osanai, T. Regulation of organic acid and hydrogen production by NADH/NAD+ ratio in Synechocystis sp. PCC 6803. Frontiers in Microbiology 2024. Volume 14 - 2023. [Google Scholar] [CrossRef]
- Mills, Lauren A.; McCormick, Alistair J.; Lea-Smith, David J. Current knowledge and recent advances in understanding metabolism of the model cyanobacterium Synechocystis sp. PCC 6803. Bioscience Reports 2020, 40. [CrossRef]
- SUGINAKA, K.; YAMAMOTO, K.; ASHIIDA, H.; KONO, Y.; SAW, Y.; SHIBATA, H. Cysteine Uptake for Accumulation of Glutathione by the Cyanobacterium Synechocystis strain PCC 6803. Bioscience, Biotechnology, and Biochemistry 1998, 62, 424–428. [Google Scholar] [CrossRef] [PubMed]
- Narainsamy, K.; Farci, S.; Braun, E.; Junot, C.; Cassier-Chauvat, C.; Chauvat, F. Oxidative-stress detoxification and signalling in cyanobacteria: the crucial glutathione synthesis pathway supports the production of ergothioneine and ophthalmate. Molecular Microbiology 2016, 100, 15–24. [Google Scholar] [CrossRef]
- Benson, P.J.; Purcell-Meyerink, D.; Hocart, C.H.; Truong, T.T.; James, G.O.; Rourke, L.; Djordjevic, M.A.; Blackburn, S.I.; Price, G.D. Factors Altering Pyruvate Excretion in a Glycogen Storage Mutant of the Cyanobacterium, Synechococcus PCC7942. Frontiers in Microbiology 2016. Volume 7 - 2016. [Google Scholar] [CrossRef]
- Carrieri, D.; Lombardi, T.; Paddock, T.; Cano, M.; Goodney, G.A.; Nag, A.; Old, W.; Maness, P.-C.; Seibert, M.; Ghirardi, M.; et al. Transcriptome and proteome analysis of nitrogen starvation responses in Synechocystis 6803 ΔglgC, a mutant incapable of glycogen storage. Algal Research 2017, 21, 64–75. [Google Scholar] [CrossRef]
- Muro-Pastor, M.I.; Reyes, J.C.; Florencio, F.J. Cyanobacteria Perceive Nitrogen Status by Sensing Intracellular 2-Oxoglutarate Levels*. Journal of Biological Chemistry 2001, 276, 38320–38328. [Google Scholar] [CrossRef]
- Lucius, S.; Hagemann, M. The primary carbon metabolism in cyanobacteria and its regulation. Frontiers in Plant Science 2024. Volume 15 - 2024. [Google Scholar] [CrossRef]
- Angermayr, S.A.; van der Woude, A.D.; Correddu, D.; Vreugdenhil, A.; Verrone, V.; Hellingwerf, K.J. Exploring metabolic engineering design principles for the photosynthetic production of lactic acid by Synechocystis sp. PCC6803. Biotechnology for Biofuels 2014, 7, 99. [Google Scholar] [CrossRef]
- Kammerscheit, X.; Hecker, A.; Rouhier, N.; Chauvat, F.; Cassier-Chauvat, C. Methylglyoxal Detoxification Revisited: Role of Glutathione Transferase in Model Cyanobacterium <i>Synechocystis</i> sp. Strain PCC 6803. mBio 2020, 11. [Google Scholar] [CrossRef]
- Osanai, T.; Oikawa, A.; Shirai, T.; Kuwahara, A.; Iijima, H.; Tanaka, K.; Ikeuchi, M.; Kondo, A.; Saito, K.; Hirai, M.Y. Capillary electrophoresis–mass spectrometry reveals the distribution of carbon metabolites during nitrogen starvation in Synechocystis sp. PCC 6803. Environmental Microbiology 2014, 16, 512–524. [Google Scholar] [CrossRef]
- Hauf, W.; Schlebusch, M.; Hüge, J.; Kopka, J.; Hagemann, M.; Forchhammer, K. Metabolic Changes in Synechocystis PCC6803 upon Nitrogen-Starvation: Excess NADPH Sustains Polyhydroxybutyrate Accumulation. Metabolites 2013, 3, 101–118. [Google Scholar] [CrossRef]
- Gerdes, S.Y.; Kurnasov, O.V.; Shatalin, K.; Polanuyer, B.; Sloutsky, R.; Vonstein, V.; Overbeek, R.; Osterman, A.L. Comparative Genomics of NAD Biosynthesis in Cyanobacteria. Journal of Bacteriology 2006, 188, 3012–3023. [Google Scholar] [CrossRef]
- Pinson, B.; Ceschin, J.; Saint-Marc, C.; Daignan-Fornier, B. Dual control of NAD+ synthesis by purine metabolites in yeast. eLife 2019, 8, e43808. [Google Scholar] [CrossRef]
- Nelson, C.; Giraldo-Silva, A.; Garcia-Pichel, F. A symbiotic nutrient exchange within the cyanosphere microbiome of the biocrust cyanobacterium, Microcoleus vaginatus. The ISME Journal 2020, 15, 282–292. [Google Scholar] [CrossRef] [PubMed]
- Nelson, C.; Dadi, P.; Shah, D.D.; Garcia-Pichel, F. Spatial organization of a soil cyanobacterium and its cyanosphere through GABA/Glu signaling to optimize mutualistic nitrogen fixation. The ISME Journal 2024, 18. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Schreiber, K.; Appel, J.; Makowka, A.; Fähnrich, B.; Roettger, M.; Hajirezaei, M.R.; Sönnichsen, F.D.; Schönheit, P.; Martin, W.F.; et al. The Entner–Doudoroff pathway is an overlooked glycolytic route in cyanobacteria and plants. Proceedings of the National Academy of Sciences 2016, 113, 5441–5446. [Google Scholar] [CrossRef]
- Kaewbai-ngam, J.; Sukkasam, N.; Phoraksa, O.; Incharoensakdi, A.; Monshupanee, T. Production of glycogen, PHB, biohydrogen, NAD(P)H, and proteins in Synechocystis sp. PCC 6803 disrupted in metabolically linked biosynthetic pathway(s). Journal of Applied Phycology 2022, 34, 1983–1995. [Google Scholar] [CrossRef]
- Hisbergues, M.; Jeanjean, R.; Joset, F.; Tandeau de Marsac, N.; Bédu, S. Protein PII regulates both inorganic carbon and nitrate uptake and is modified by a redox signal in Synechocystis PCC 6803. FEBS Letters 1999, 463, 216–220. [Google Scholar] [CrossRef]
| ATP | ADP | AMP | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| +N | -N | +N | -N | +N | -N | |||||||
| WT-GS | 0.31 | ± 0.15 | 0.06 | ± 0.03 | 3.53 | ± 1.68 | 1.84 | ± 0.71 | 1.37 | ± 0.39 | 1.15 | ± 0.28 |
| WT-GT | 0.39 | ± 0.10 | 0.15 | ± 0.07 | 3.44 | ± 0.90 | 1.18 | ± 0.94 | 0.74 | ± 0.06 | 0.50 | ± 0.29 |
| Δpgm-GT | 0.44 | ± 0.19 | 0.38 | ± 0.05 | 4.09 | ± 0.73 | 5.63 | ± 0.94 | 1.24 | ± 0.08 | 7.35 | ± 0.60 |
| ΔpirC-GT | 0.45 | ± 0.21 | 0.51 | ± 0.20 | 3.66 | ± 1.53 | 2.77 | ± 1.12 | 1.03 | ± 0.18 | 0.78 | ± 0.08 |
| ΔglgC-GS | 0.47 | ± 0.12 | 0.13 | ± 0.10 | 3.81 | ± 0.65 | 2.66 | ± 1.18 | 0.60 | ± 0.28 | 2.15 | ± 0.88 |
| ΔglgCΔpirC-GS | 0.25 | ± 0.02 | 0.28 | ± 0.07 | 3.29 | ± 1.66 | 6.14 | ± 2.15 | 1.10 | ± 0.64 | 3.32 | ± 0.50 |
| ΔglnB-GS | 0.99 | ± 0.12 | 0.14 | ± 0.02 | 6.04 | ± 1.15 | 4.58 | ± 0.53 | 0.88 | ± 0.24 | 2.90 | ± 0.22 |
| ΔglgA1-GT | 0.29 | ± 0.21 | 0.16 | ± 0.07 | 3.18 | ± 1.22 | 4.72 | ± 1.71 | 1.14 | ± 0.42 | 2.92 | ± 0.85 |
| ΔglgA2-GT | 0.19 | ± 0.05 | 0.05 | ± 0.02 | 2.18 | ± 0.21 | 1.59 | ± 0.95 | 0.77 | ± 0.29 | 2.60 | ± 0.65 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




