1. Introduction
Terpenes are a large family of chemicals with the formula (C
5H
8)
n that are often produced by plants where they act in their photoautotrophic metabolism, protection against pathogens and/or attraction of pollinators. Many high-value terpenes are exploited by cosmetic, food, pharmaceutical and/or biofuel industries, including bisabolene, farnesene, limonene and pinene, the focus of the present study [
1,
2].
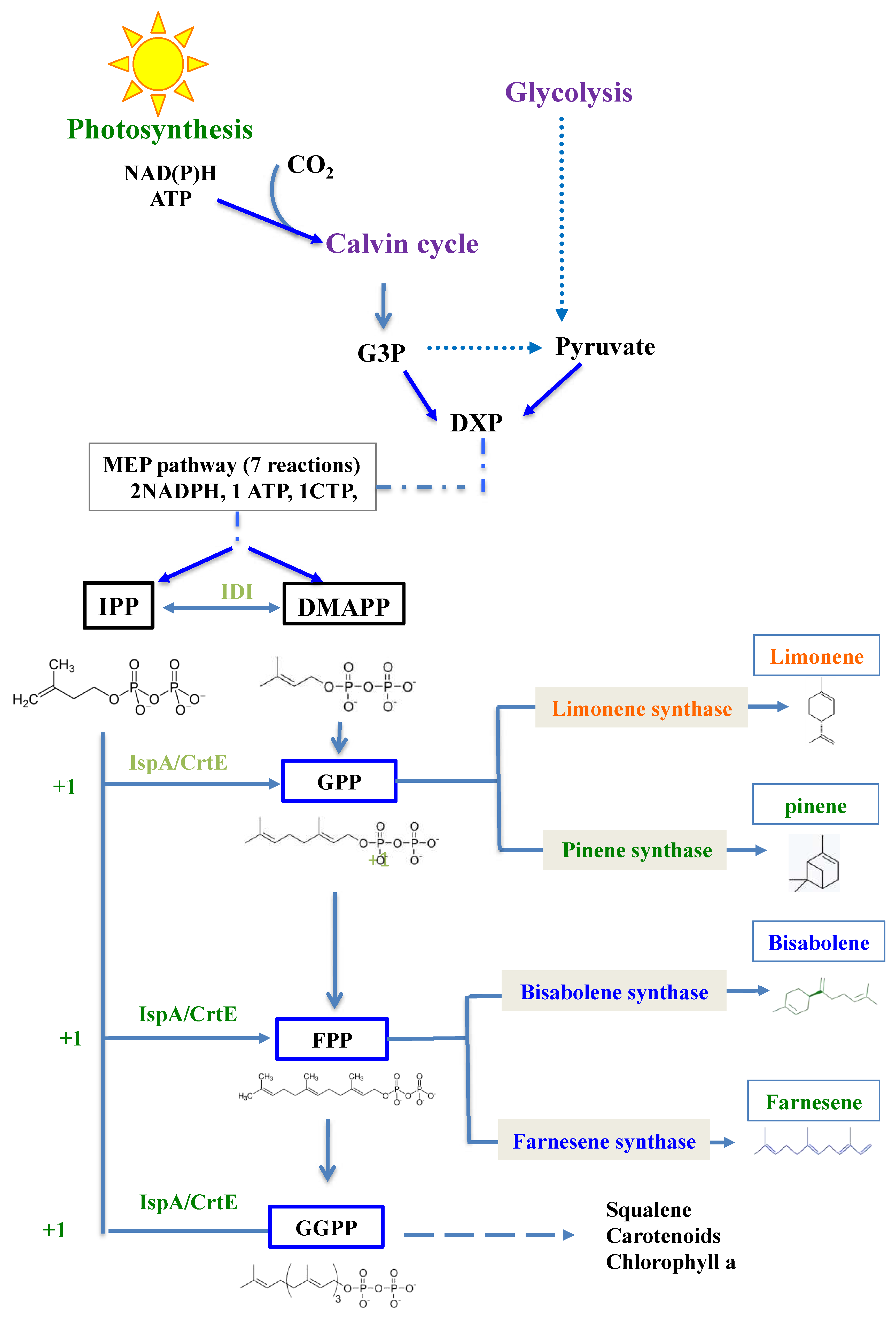
All terpenes are derived from the five-carbons (C5) building blocks, isopentenyl pyrophosphate (IPP) and dimethylallyl pyrophosphate (DMAPP) that are condensed to form the geranyl pyrophosphate (GPP, C10) precursor of monoterpenes (C
10H
16) such as limonene and pinene. The subsequent addition of one IPP unit on GPP forms the farnesyl pyrophosphate (FPP, C15) precursor of sesquiterpenes (C
15H
24), such as bisabolene and farnesene (
Figure 1).
Instead of plants that must be preserved for food production, cyanobacteria, the robust photosynthetic prokaryotes that colonize our planet, can be used for the sustainable production of terpenes from the plentiful natural resources: solar energy, water, CO
2 and minerals [
1]. Cyanobacteria possess the methylerythritol-4-phosphate (MEP) pathway that produces GPP and FPP (
Figure 1), which can be transformed into terpenes. For this purpose, synthetic genes encoding terpene synthases need to be adapted to the cyanobacterial codon usage, and subsequently introduced and expressed in cyanobacteria [
1,
2].
The model (unicellular) cyanobacteria
Synechococcus PCC 7002 (hereafter S.7002) and
Synechococcus PCC 7942 (S.7942) are attractive for this purpose, in having good genetic tools and interesting physiological difference (for review see [
3]) which make their comparative analysis very interesting. The salt-tolerant marine model S.7002 (genome size 3.41 Mb) is able to grow on nitrate (the classical nitrogen source for cyanobacteria) or urea (cheaper than nitrate) [
4], but it requires vitamin B12 for growing [
5]. In contrast, the salt-sensitive freshwater model S.7942 (genome size 2.74 Mb) does not require vitamin B12, but is unable to grow on urea [
6]. S.7002 and S.7942 have other key differences in their DNA repair [
7] and glutathione-dependent systems that play a prominent role in tolerance to stresses [
3]).
Two proof-of-concept studies showed that S.7002 can be engineered for the photosynthetic production of bisabolene and limonene [
8] or pinene [
9], but not farnesene as we presently report. Concerning S.7942, several laboratories reported that S.7942 can be engineered for the production of farnesene [
10,
11,
12] or limonene [
13,
14], but neither bisabolene nor pinene as we report here. The limited number of previous studies and their multiple experimental differences (nature of the terpene synthase genes and their promoters, and of the chromosome/plasmid vectors propagating them) make it is difficult to know whether S.7002 and/or S.7942 are suitable chassis for the photoproduction of chemically-diverse terpenes or only a few of them.
In this study, we have employed the same genetic strategy to engineer S.7002 and S.7942 for the photoproduction of four chemically-different high-value terpenes: two monoterpenes, limonene (cyclic molecule) and pinene (bicyclic), and two sesquiterpenes, bisabolene (cyclic) and farnesene (linear), in order to compare the potential of these two model cyanobacteria for the production of various terpenes. Each terpene synthase gene was expressed from the same strong promoter propagated by the same autonomously replicating plasmid vector in both S.7002 and S.7942. We showed for the first time that S.7002 and S.7942 can produce farnesene and bisabolene, respectively. Both S.7002 and S.7942 produced farnesene and pinene with the best and lowest efficiencies, respectively. Furthermore, S.7002 produced limonene better than bisabolene, whereas S.7942 produced bisabolene better than limonene. We also showed for the first time, that S.7002 cultivated on urea (a frequent pollutant) produce terpenes more efficiently than cultures growing on either nitrate or ammonium (standard, but more expensive, nitrogen sources for cyanobacteria). Finally, we showed in both S.7002 and S.7942 that increasing the copy number of the terpene synthase-encoding gene increases the level of terpene production, suggesting that terpene production is more limited by the activity of terpene synthases rather than the abondance of terpene precursors.
2. Materials and Methods
2.1. Bacterial Strains and Growth Conditions
E. coli strains used for gene manipulations (TOP10 and NEB10 beta,
Table S1) or conjugative transfer of pC-derived replicative plasmids (
Table S1) to S.7942 or S.7002 (CM404 [
15]) were grown at 37°C (TOP10 and NEB10 beta) or 30°C (CM404) on LB medium containing appropriate antibiotics: ampicillin (Ap) 100 μg.mL
-1, kanamycin (Km) 50 μg.mL
-1, streptomycin (Sm) 25 μg.mL
-1 or spectinomycin (Sp) 75 μg.mL
-1.
S.7942 and S.7002 were grown at 30°C, under continuous white light (2500 lux; 31.25 μE m
-2 s
-1) and agitation (140 rpm, Infors rotary shaker) in liquid mineral medium: MM,
i.e. BG11 [
16] enriched with 3.78 mM Na
2CO
3 [
17] for S.7942, or A+ containing B12 vitamin (4 μg.L
-1) for S.7002 [
18]. The terpene-producing strains were grown in the presence of the selective antibiotics: Km 50 μg.mL
-1 for both S.7002 and S.7942, and/or Sm 50 μg.mL
-1 and Sp 50 μg.mL
-1 for S.7002 and 10 times less for S.7942. For some experiments carried out with S.7002, the standard nitrogen source (NaNO
3, 11.8 mM) was replaced by urea or ammonium. Nitrate-grown cells were washed twice and resuspended in nitrogen free media that were supplemented with either urea or nitrate, as indicated. Growth in liquid cultures was monitored by regular measurements of optical density at 750 nm (OD
750) using a spectrophotometer (Jenway 6700).
2.2. Genetic Manipulations
The pC-derived replicative plasmids for high-level expression of terpene synthase genes (
Table S1) were introduced in S.7942 and S.7002 by conjugation, as described [
19,
20], using a 72h co-incubation of
E. coli and cyanobacterial cells. The DNA cassettes for high-level expression of terpene synthase genes were introduced in neutral sites of the chromosome of S.7942 or S.7002 by transformation, as described [
21,
22]. Cells were then plated on MM containing Km 50 μg.mL
-1 or both Sp 5 μg.mL
-1 and Sm 5 μg.mL
-1 (S.7942), or A+ medium containing 4 μg.L
-1 B12 vitamin, 3 g.L
-1 sodium thiosulfate, Km 50 μg.mL
-1 or both Sp 50 μg.mL
-1 and Sm 50 μg.mL
-1 (S.7002), which were solidified with 1% (S.7942) or 1.5% (S.7002) Bacto Agar (Difco).
The presence of the terpene-synthase-encoding DNA cassette propagated in pC-derived plasmids or a neutral chromosomal site was verified by PCR and DNA sequencing (Mix2Seq Kit, Eurofins Genomics) using appropriate oligonucleotide primers (
Table S2).
2.3. Terpene Collection and Quantification by Gas Chromatography–Mass Spectrometry
S.7002 and S.7942 strains engineered for terpene production were photoautotrophically grown in the presence of selective antibiotics, in 250-mL erlenmeyers containing 50 mL cell suspensions overlaid with 20% (vol/vol) dodecane (analytical grade, Sigma-Aldrich) to trap terpenes [
1,
20,
23]. At time intervals, 300 μL aliquots of the dodecane overlay were collected. 1 μL aliquots of these dodecane samples were injected in a split mode 10:1 (limonene) or 5:1 (E-α-bisabolene, farnesene and pinene) into a GC-MS apparatus (Trace1300 (GC) + ISQ LT (MS), ThermoScientific). The quantification of the production of terpenes was performed as we previously described [
20,
23].
3. Results and Discussion
3.1. Construction of the Synechococcus PCC 7002 and the Synechococcus PCC 7942 Strains Expressing the Studied Terpene Synthase Genes from the STRONG LAMBDA-PHAGE PR PROMOTER and Propagated in Either or Both a Neutral Chromosomal Site or a Replicative Plasmid
In this study, we tested and compared the ability of the unicellular model cyanobacteria S.7002 and S.7942) to produce the four chemically-different high-value terpenes: bisabolene, farnesene, limonene and pinene (
Figure 1). For this purpose, we used the following terpene synthases-encoding genes that worked well in cyanobacteria [
1], namely: (i) the Abies grandis E-α-bisabolene synthase (bs), (ii) the Picea abies α-farnesene synthase (fs), (iii) the Mentha spicata 4S-limonene synthase gene (ls) and (iv) the Pinus taeda (+)-α-pinene synthase gene (ps). Each codon-adapted terpene synthase gene was cloned downstream of the strong λpR-promoter and associated λcro ribosome-binding site of the autonomously replicating RSF1010-derived [
15,
19] pC plasmid vector [
24], yielding the plasmids pCBS, pCFS, pCLS and pCPS [
20,
23]. These plasmids were introduced by conjugation in S.7002 and S.7942 [
15,
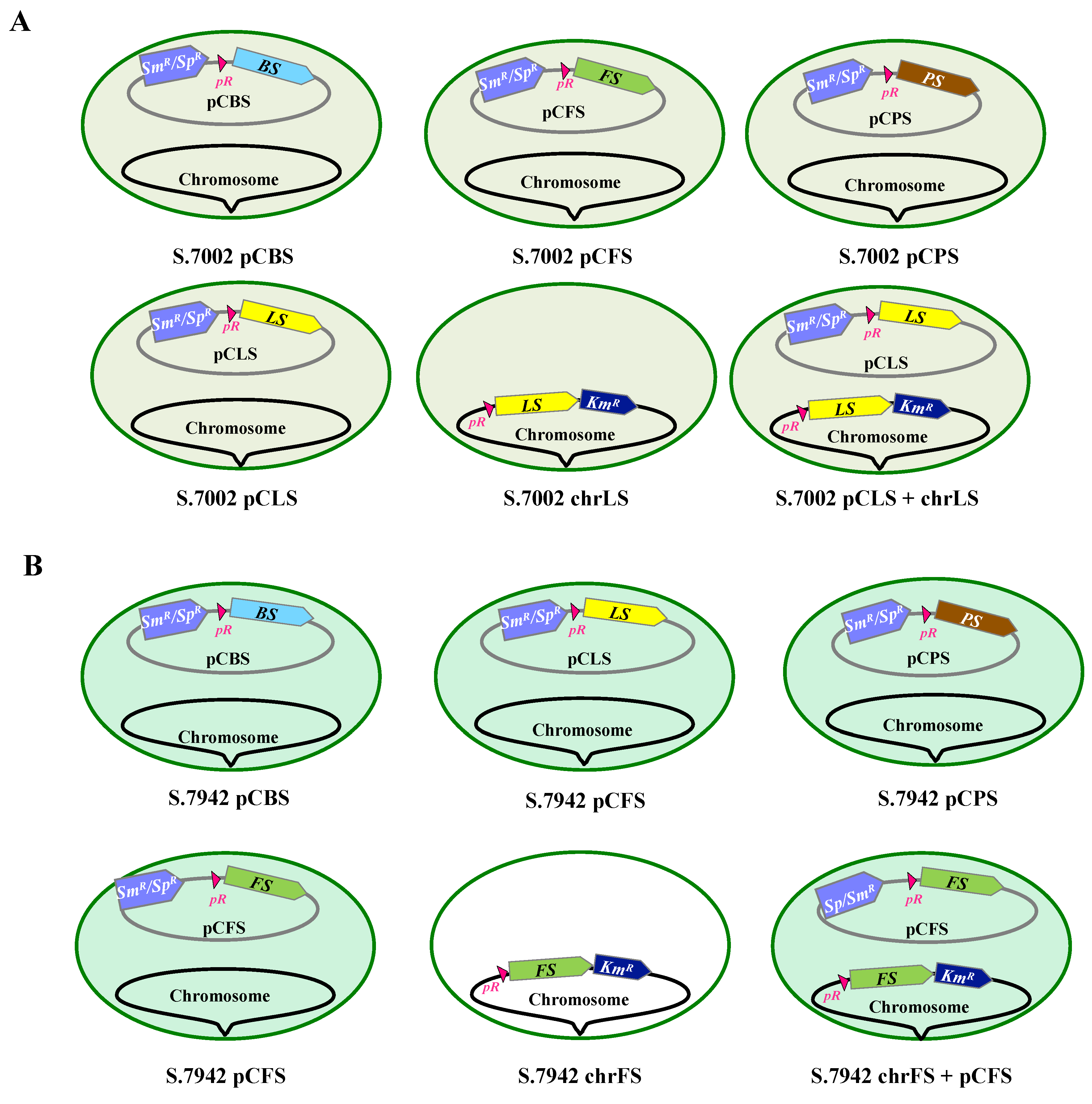
23], yielding the strains represented in
Figure 2. In each case, two independent Sm
R/Sp
R clones were selected and analyzed by PCR and DNA sequencing (
Figure S1) to verify that pCBS, pCFS, pCLS and pCPS plasmids replicate stably in S.7002 and S.7942, without affecting their photoautotrophic growth (
Figure S2).
The pR-LS and pR-FS recombinant genes were also cloned in the NS
7002 and NSI
7942 neutral chromosomal sites of respectively S.7002 and S.7942, to compare the level of terpene production driven by either or both the plasmid and the chromosome (
Figure 2).
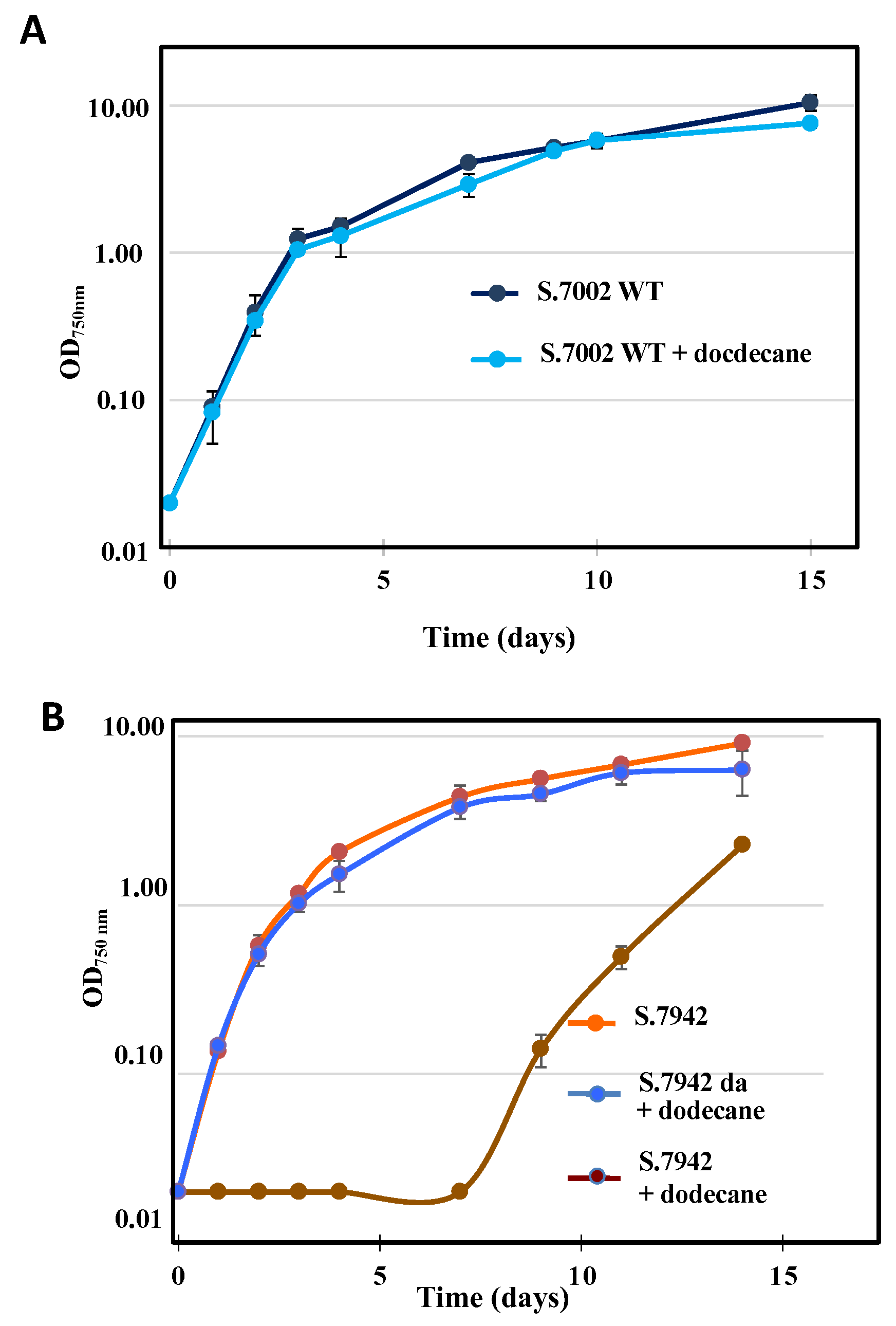
3.2. Unlike Synechococcus PCC 7002, the Growth of Synechococcus PCC 7942 is Affected by the Dodecane Overlay used to Trap Terpenes, but Dodecane-Adapted Strains Grow Healthy under Dodecane
As terpenes are volatile chemicals, cyanobacteria engineered for terpene production are routinely grown under a dodecane overlay to trap terpene, which are subsequently quantified by gaz chromatography coupled to mass spectrometry [
1]. The growth of S.7002 was not affected by a dodecane overlay (
Figure 3), in agreement with previous studies on S.7002 [
8,
9]. In contrast, the growth of S.7942 was strongly delayed by a dodecane overlay (
Figure 3), but S.7942 strains precultivated under dodecane grew subsequently well under dodecane (
Figure 3), a finding not discussed by previous workers [
10,
11,
12]. Consequently, we used dodecane-adapted strains to analyze terpene production by S.7942.
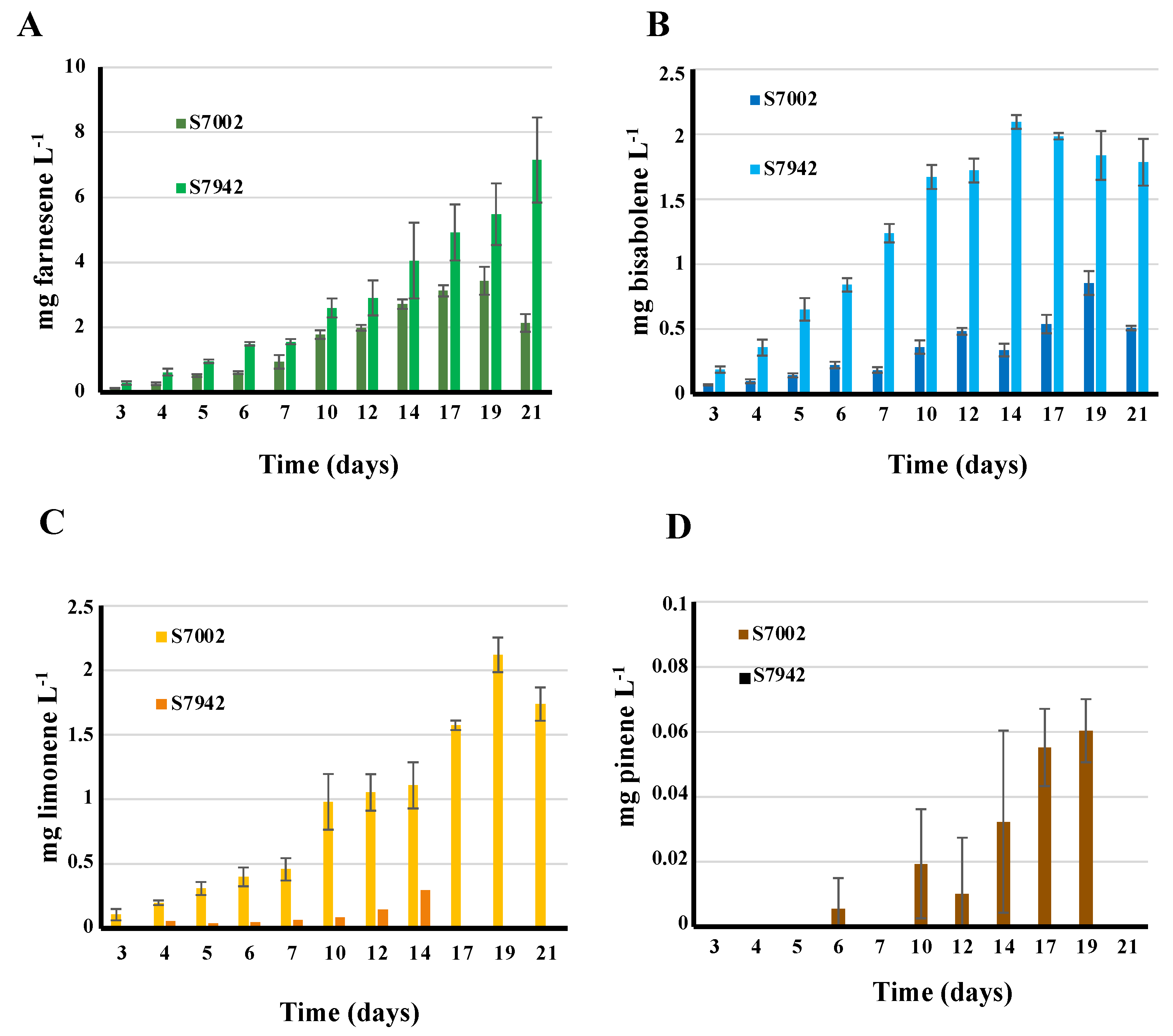
3.3. First Report of Farnese Production by Synechococcus PCC 7002: the Production is Lower than that of Synechococcus PCC 7942
The production of farnesene during the photoautotrophic growth of the S.7002 and S.7942 strains propagating the pCFS plasmid, or the empty pC plasmid vector used as negative control, were measured over 21-days periods of photoautotrophic growths. GC-MS analysis of dodecane overlay samples from the pCFS propagating strains showed a peak with a similar retention time and ion chromatogram than a pure standard of α-farnesene, which was not observed in the negative-control pC strains (
Figures S3A and S4A). These data showed that both S.7002 and S.7942 can photosynthetically produce farnesene (
Figure 4A), a novel finding in the case of S.7002. However, lower level of farnesene was produced by S.7002 (∼ 2 mg.L
-1 after 21 days,
Figure 4), as compared to S.7942 (∼ 8 mg.L
-1 after 21 days,
Figure 4A).
The level of farnesene production by S.7942 (∼1.5 mg.L
-1 after 7 days,
Figure 4A) was roughly similar to what was reported by an other group (∼3 mg.L
-1 after 8 days, no subsequent measurements) [
10,
12]) who cloned into a neutral chromosomal site (we used a replicative plasmid) a codon-adapted α-farnesene synthase gene from Malus domestica Borkh or Picea abies (we used only the Picea abies enzyme) expressed from the strong pTrc promoter (we used the strong lambda-phage pR promoter). However, the multiple experimental differences between these reports and our own study make their comparison difficult. This explains why we employed the same genetic engineering strategy to explore the capability of various model cyanobacteria to photosynthetically produce various high-value terpenes.
The level of farnesene production driven by the pCFS plasmid in S.7942 and S.7002 could be really compared to the level driven by the same pCFS plasmid we previously reported in Synechocystis PCC 6803 [
20], another unicellular model cyanobacterium phylogenetically-distant to both S.7942 and S7002 [
3]. Intestingly, the pCFS-driven farnesene production in S.7942 and S.7002 (∼8 and ∼2 mg.L
-1 after 21 days, respectively,
Figure 4A) was lower than what we observed in S.6803 harboring the same pCFS plasmid (∼30 mg.L
-1 after 21 days, [
20],
Figure S5A). These findings indicate that S.7942 and S.7002 are less suitable cyanobacterial factories than Synechocystis PCC 6803 (hereafter S.6803) for the photoproduction of farnesene.
3.4. First report of Bisabolene Production by Synechococcus PCC 7942: the Production is Similar to that of Synechococcus PCC 7002
The photosynthetic production of bisabolene by the S.7002 and S.7942 strains propagating the pCBS plasmid, or the empty pC plasmid, was measured over a 21-days period of cultures (
Figure 4B). GC-MS analysis of dodecane overlay samples from the pCBS strains showed a peak with a similar retention time (
Figures S3A and S4A) and ion chromatogram than a pure standard of E-α-bisabolene, which was not observed in the negative-control pC strain (
Figures S3A and S4A). Both S.7002 and S.7942 produced similar levels of bisabolene (∼0.5 and ∼ 2.0 mg.L
-1 after 21 days, respectively
Figure 4B).
The production of bisabolene by S.7002 (∼0.1 mg.L
-1 after 4 days; ∼0.5 mg.L
-1 after 21 days) is lower than the previously reported level (∼0.5 mg.L
-1 after 6 days, no subsequent measurement [
8]) observed after cloning the Abies grandis (E)-α-bisabolene synthase gene expressed from the cpc phycocyanin promoter (we used the lambda-phage pR promoter) into a neutral chromosomal site (we used a replicative plasmid). The important differences between the previous report and our study, combined to the use of different growth conditions, make their comparison difficult.
The presently-observed levels of bisabolene production driven by the pCBS plasmid in S.7002 and S.7942 (∼0.5 and ∼ 2.0 mg.L
-1 after 21 days, respectively
Figure 4B), are lower than the value we previously reported for S.6803 harboring the same pCBS plasmid (10-14 mg.L
-1 after 21 days, [
20,
25]). Together with the above-mentionned findings, these data (
Figure S5B) indicate that S.7942 and S.7002 are less efficient cyanobacterial factories than S.6803 for the photoproduction of the sesquiterpenes farnesene and bisabolene.
3.5. The Synechococcus PCC 7002 and Synechococcus PCC 7942 Strains Harboring the pCLS Plasmid Produce Limonene in this Order
The photosynthetic production of limonene by the S.7002 and S.7942 strains propagating the pCLS plasmid, or the empty pC plasmid, was monitored during 21 days (
Figure 4C). Dodecane overlay samples from the pCLS strains, not the pC strains, showed a GC-MS peak typical of a pure S-(-)-limonene standard (
Figures S3B and S4B). Interestingly, higher level of limonene was produced by S.7002 ((∼ 1.0 mg.L
-1 after 14 days and ∼ 1.7 mg.L
-1 after 21 days,
Figure 4C) as compared to S.7942 (∼ 0.3 mg.L
-1 after 14 days,
Figure 4C).
The presently-observed production of limonene by S.7002 (∼ 0.3 mg.L
-1 after 4 days,
Figure 4C) seems lower than the previously reported level (∼4.0 mg.L
-1 after 4 days, but no subsequent measurement [
8]) observed after cloning into a neutral chromosomal site (we used a replicative plasmid) the Mentha spicata limonene synthase gene expressed from the cpc phycocyanin promoter (we used the lambda-phage pR promoter).
Similarly, the presently-observed production of limonene by S.7942 (∼0.06 mg.L
-1 after 7 days,
Figure 4C) seems lower than the level (∼17 mg.L
-1 after 7 days, but no subsequent measurement [
14]) observed after cloning into a neutral chromosomal site (we used a replicative plasmid) the Mentha spicata limonene synthase gene expressed from the psbA promoter (we used the lambda-phage pR promoter).
The multiple experimental differences between the previous reports and our study make it difficult to ascribe the difference in limonene production to one or several parameters: growth conditions, nature of the promoter expressing the limonene synthase gene and/or of the cloning vehicle propagating it.
The presently-observed level of limonene production driven by the pCLS plasmid in S.7002 (∼ 1.7 mg.L
-1 after 21 days,
Figure 4C) is slightly higher than the value we previously observed in S.6803 (∼0.7 mg.L
-1 after 21 days [
20,
25]. These findings (
Figure S5C) indicate that S.7002 is as a cyanobacterial cell factory as effective as S.6803 for the production of the monoterpene limonene, unlike what we observed in the case of the two sesquiterpenes, bisabolene and farnesene (
Figures S5B and S5C).
3.6. The Synechococcus PCC 7002 and Synechococcus PCC 7942 Strains Harboring the pCPS Plasmid Produce Pinene Inefficiently
The photosynthetic production of pinene by the S.7002 and S.7942 strains propagating the pCPS plasmid, or the empty pC plasmid, was measured during 21-days (
Figure 4D). GC-MS analysis of dodecane overlay samples from the pCPS strain of S.7002 showed a peak typical of a pure standard of α-pinene, which was not observed in the pC strain (
Figure S3C). In contrast, no pinene production was detected in S.7942 harboring pCPS (
Figure 4D).
The level of pinene production by S.7002 (∼60 μg.L
-1 after 21 days,
Figure 4D) is similar to what we previously reported for S.6803 harboring the same pCPS plasmid (∼80 μg.L
-1 after 21 days; [
20],
Figure S5D).
The presently observed production of pinene by S.7002 (∼60 μg.L
-1 after 21 days) is lower than the previously reported level (1.5 mg.L
-1 after 6 days, [
9]) observed after cloning into the endogenous pAQ1 plasmid (we used the non-cyanobacterial pC vector derived from the broad-host-range plasmid RSF1010 [
20]) a Abies grandis gene encoding a 6xhistidine-tagged pinene synthase (we used the native Pinus taeda enzyme) expressed from the cpc promoter (we used the lambda-phage pR promoter). As both the pAQ1 and pC vectors on one hand, and the cpc and pR promoters on the other hand, function well in cyanobacteria [
3] we assume that the 6xhistidine-tagged pinene synthase from Abies grandis is more efficient than the native Pinus taeda enzyme we have used in this study (
Figure 4D), and our previous analysis of pinene production in S.6803 [
20],
Figure S5D).
3.7. Comparison of Limonene Production Driven in Synechococcus PCC 7002 by the Recombinant LS Gene Propagated by Either or Both the pCFS Replicative Plasmid and the NS7002 Neutral Chromosomal Site
As the activity of an enzyme can be increased by increasing the copy number of the gene encoding it, we attempted to increase the limonene production of the S.7002 strain harboring pCLS by cloning an extra copy of the same pR-LS recombinant gene (LS expressed from the strong pR promoter) in the NS
7002 neutral chromosomal site (region in between A0935-A0936 [
26]) often used for gene cloning in S.7002 [
3]. Therefore, a pR-LS-Km
R DNA cassette was assembled by Gibson [
27] (
Figure S6) in between the two 500-bp DNA regions surrounding NS
7002 to serve as platform of homology for DNA recombination promoting targeted integration of the pR-LS-Km
R cartridge into the chromosome. The resulting pTwist_NS-S7002p
RLSKm
R plasmid (
Table S1) was transformed to S.7002 pCLS cells where the pR-LS-Km
R DNA cassette was integrated in all chromosome copies (
Figure S7). The resulting strain expressing the pR-LS gene from both the pCLS plasmid and the chromosome was designated as pCLS+chrLS (
Figure 2).
In parallel, the pTwist_NS-S7002p
RLSKm
R plasmid was transformed to S.7002 wild-type cells where the pR-LS-Km
R DNA cassette was integrated in all chromosome copies (
Figure S7). The resulting strain named chrLS served to measure the level of limonene production (
Figure 2) driven by the chromosomal pR-LS gene.
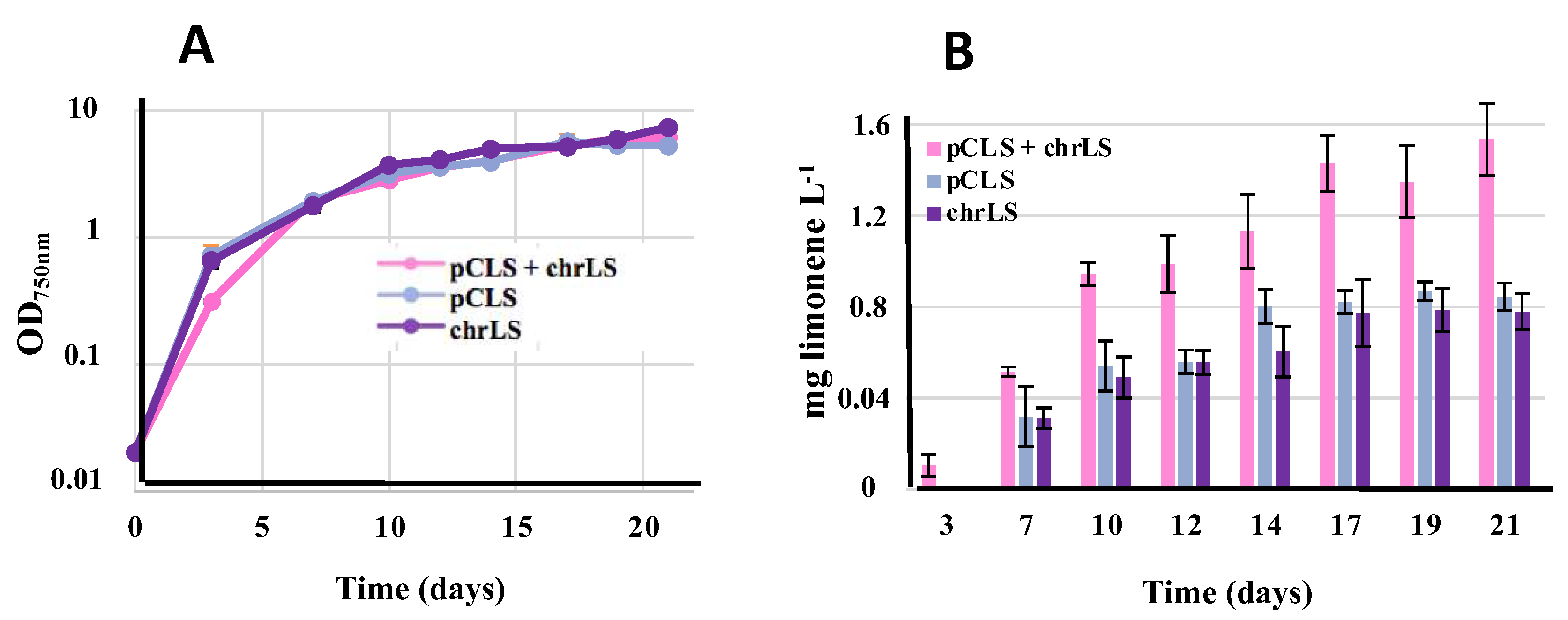
All three strains pCLS, chrLS and pCLS+chrLS grew well under standard photoautotrophic conditions in the presence of the dodecane overlay (
Figure 5). Interestingly, the level of limonene produced by the pCLS+chrLS strain was roughly equivalent to the sum of limonene produced by the strains pCLS and chrLS, which were similarly active (
Figure 5). These results indicate that the production of terpenes by an engineered S.7002 strain is probably more limited by the activity of the employed terpene synthase than the abundance of terpene precursors, as we recently discussed in the case of S.6803 [
20].
3.8. Comparison of Farnesene Production Driven in Synechococcus PCC 7942 by the Recombinant FS Gene Propagated by Either or Both the pCFS Replicative Plasmid and the NSI7942 Neutral Chromosomal Site
We also attempted to increase farnesene production in the pCFS S.7942 strain by cloning an extra copy of the pR-FS recombinant gene (FS expressed from the pR promoter) in the NSI
7942 neutral chromosomal site (GenBank accession U30252, [
28]) often used for gene cloning in S.7942 [
3]. Therefore, a pR-FS-Km
R DNA cassette was assembled by Gibson [
27] (
Figure S6) in between by the two 300-bp DNA regions surrounding NSI
7942 to mediate the targeted integration of the pR-FS-Km
R cartridge in NSI
7942. The resulting pTwist_NSI-S7942 p
RFSKm
R plasmid (
Table S1) was transformed to S.7942 pCFS cells where the pR-FS-Km
R DNA cassette was integrated in the NSI
7942 site of all chromosome copies (
Figure S8). The resulting strain expressing the pR-FS gene from both the pCFS plasmid and the chromosome was designated as pCFS+chrFS (
Figure 2).
In parallel, the pTwist_NSI-S7942 p
RFSKm
R plasmid was transformed to S.7942 wild-type cells where the pR-FS-Km
R DNA cassette was integrated in the NSI site of all chromosome copies (
Figure S8). The resulting strain named chrFS (
Figure 2) served to measure the level of farnesene production driven by the chromosomal pR-FS gene (
Figure 6).
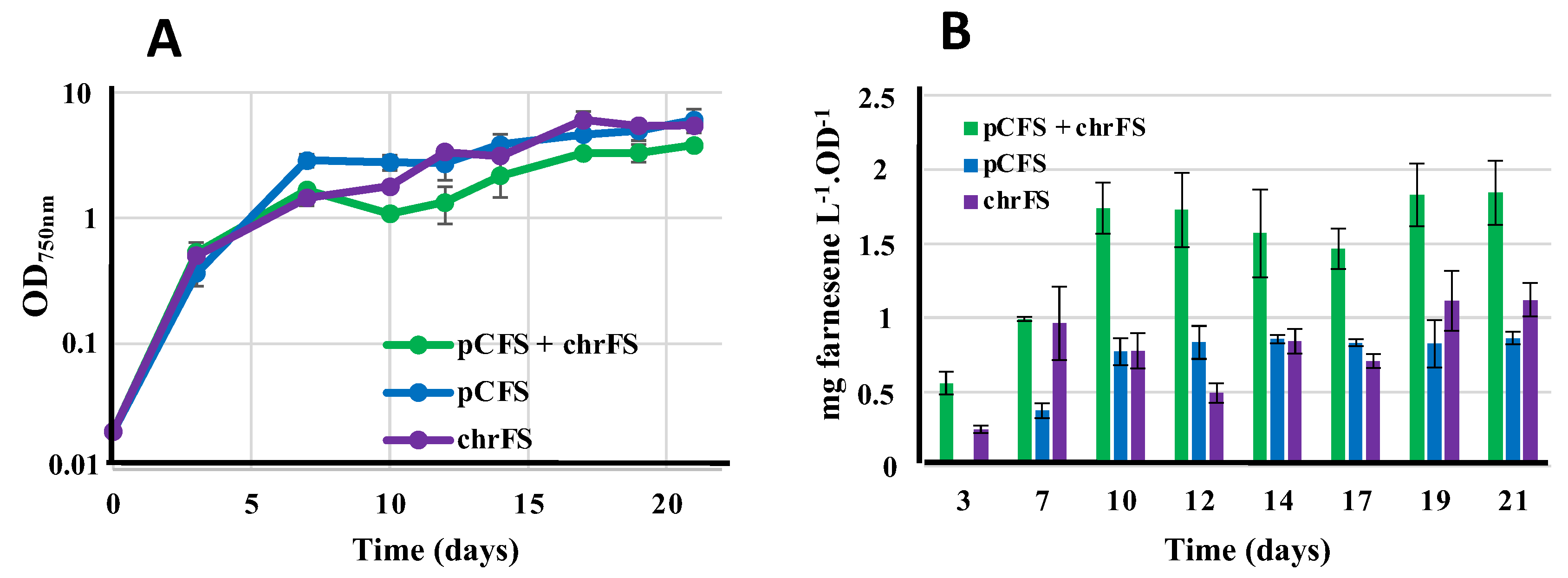
The two pCFS and chrFS grew slightly better than the pCFS+chrFS strain under standard photoautotrophic conditions, irrespectively of the presence of the terpene-trapping dodecane overlay (
Figure 6A). Interestingly, the level of farnesene produced by the pCFS+chrFS strain was roughly equivalent to the sum of farnesene produced by the strains pFCS and chrFS, which were similarly active (
Figure 6B). These results indicate that the production of terpenes by an engineered S.7942 strain is probably more limited by the terpene-synthase activity than the terpene-precursors abundance, as we recently discussed in the case of S.6803 [
20]. This assumption is consistent with the finding that terpene synthases are slow enzymes, and that selecting a terpene synthase with high Kcat and low Km is pivotal in terpenoids biosynthesis [
29].
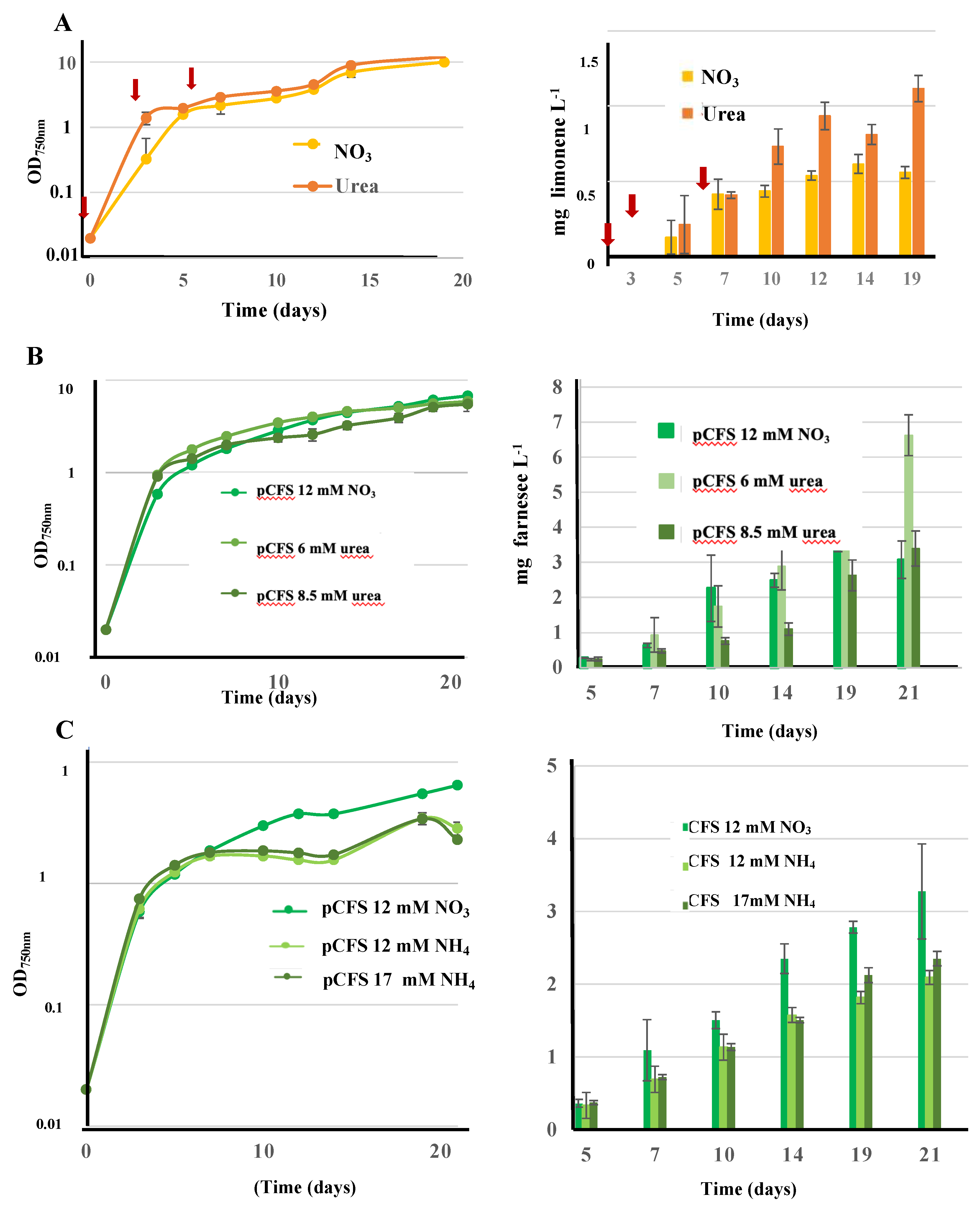
3.9. Influence of Growth Conditions on Terpene Production: First Report that Synechococcus PCC 7002 Cultivated on Urea Produces Terpenes More Efficiently than Cultures Growing on Nitrate or Ammonium in This Order
In the view of reducing the costs of future industrial production of terpenes, eventually coupled with wastewater treatment, we have tested the influence of urea (frequently present in natural waters [
4]) on cell growth and the production of the monoterpene limonene and the sesquiterpene farnesene. For this purpose, we have used S.7002 because it grows well on all three nitrogen sources [
5], ammonium, nitrate (the standard nitrogen source for cyanobacteria [
6]) and urea (cheaper than nitrate), unlike S.7942 that cannot grow on urea because it has no urea catabolic enzymes [
4]. In these assays, S.7002 strains harboring the pFS or pCLS plasmids were cultivated in the same standard concentration of nitrogen (12 mM) supplied as either nitrate (NaNO
3, 12 mM), ammonium (NH
4Cl, 12 mM) or urea (CO(NH
2)
2, 6 mM). In the case of urea (and nitrate as a control) we tested both protocols where nitrogen is supplied either all at once at the beginning of the culture, or provided gradually as successive doses during cultivation as was required by the other cyanobacteria Cyanothece PCC 7425 and Synechocystis PCC 6803 [
20,
23]. The results (
Figure 7) showed, for the first time, that S.7002 growing on urea as the sole nitrogen source can produce terpene, even better than cells cultivated on nitrate or ammonium (in this order). Interestingly, Cyanothece PCC 7425 and S.6803 produced terpene less-efficiently (C.7425) or similarly (S.6803) when cultivated on urea as compared to nitrate [
20,
23].
4. Conclusions
We have performed the first comparative analysis of the ability of two physiologically-diverse model unicellular cyanobacteria
Synechococcus PCC 7002 (marine host) and
Synechococcus PCC 7942 (fresh-water host), to photosynthetically produce chemically-diverse terpenes of high-value (bisabolene, farnesene, limonene and pinene). For this purpose, each terpene synthase gene expressed from the same strong promoter was propagated by the same RSF1010-derived replicative plasmid vector. We showed, for the first time, that S.7002 and S.7942 can be engineered for the photoproduction of farnesene and bisabolene, respectively. S.7942 and S.7002 (in this order) produced farnesene more efficiently than the other tested terpenes (especially pinene the weakest produced terpene) similarly to what we previously observed in the other model cyanobacterium
Synechocystis PCC 6803 [
20]. Furthermore, S.7002 produced limonene better than bisabolene, whereas S.7942 produced bisabolene better than limonene. We showed for the first time that S.7002 can produce terpenes when growing on urea (a frequent pollutant) as the sole nitrogen source, similarly to what we observed for
Synechocystis PCC 6803 [
20]. In the case of S.7002, higher levels of terpenes were produced by cultures growing on urea, as compared to nitrate or ammonium, the standard or frequent nitrogen source for cyanobacteria, respectively. Also interestingly, higher levels of terpenes were produced by both S.7002 and S.7942 expressing the terpene synthase gene from both a RSF1010-derived replicating pC plasmid and a neutral chromosomal site, as we observed in
Synechocystis PCC 6803 [
20]. These results suggest that the production of terpene
s in cyanobacteria is more limited by the activity of terpene synthases than the abondance of terpene precursors, similarly to what we discussed in the case of
Synechocystis PCC 6803 [
20]. Finally, we compared the levels of terpene production by S.7002 and S.7942 with what we previously observed for
Synechocystis PCC 6803 that was engineered the same way than S.7002 and S.7942 [
20]. We found that S.6803 produces higher levels of the sesquiterpenes farnesene and bisabolene than S7942 and S.7002 (in this order), suggesting that S.6803 could be a better cyanobacterial chassis for future industrial photoproduction of these chemicals.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on Preprints.org. Figures S1–S8: title; Tables S1 and S2.
Author Contributions
Conceptualization, F.C. and C.C.-C.; methodology, F.C., C.C.-C. and A.G.; validation, C.C., V-B.G., M.V., E.D-S. and A.G.; formal analysis, C.C., V-B.G., M.V. and E.D-S.; investigation, C.C., V-B.G., M.V., E.D-S., C.C.-C. and F.C.; resources, F.C. and C.C.-C.; writing—original draft preparation, F.C., C.C.-C., C.C., V-B.G. and M.V.; writing—review and editing, F.C. and C.C.-C.; supervision, F.C. and C.C.-C.; project administration, F.C. and C.C.-C.; funding acquisition, F.C. and C.C.-C. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded in part by the CEA program Focus ECC and the ANR CalvinDesign project and the ABACUS project, grant agreement No 745668, funded by the BBI-JU under the European Union’s Horizon 2020 R&I program.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
We thank Frédéric Taran for access to GC-MS.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lin, P.C.; Pakrasi, H.B. Engineering cyanobacteria for production of terpenoids. Planta 2019, 249, 145–154. [CrossRef]
- Rautela, A.; Kumar, S. Engineering plant family TPS into cyanobacterial host for terpenoids production. Plant Cell Rep. 2022, 41, 1791–1803. [CrossRef]
- Cassier-Chauvat, C.; Blanc-Garin, V.; Chauvat, F. Genetic, Genomics, and Responses to Stresses in Cyanobacteria: Biotechnological Implications. Genes (Basel). 2021, 12, 500. [CrossRef]
- Veaudor, T.; Cassier-Chauvat, C.; Chauvat, F. Genomics of Urea Transport and Catabolism in Cyanobacteria: Biotechnological Implications. Front. Microbiol. 2019, 10, 2052. [CrossRef]
- Ludwig, M.; Bryant, D.A. Acclimation of the global transcriptome of the cyanobacterium Synechococcus sp. strain PCC 7002 to nutrient limitations and different nitrogen sources. Front. Microbiol. 2012, 11. [CrossRef]
- Veaudor, T.; Blanc-Garin, V.; Chenebault, C.; Diaz-Santos, E.; Sassi, J.F.; Cassier-Chauvat, C.; Chauvat, F. Recent advances in the photoautotrophic metabolism of cyanobacteria: Biotechnological implications. Life 2020, 10. [CrossRef]
- Cassier-Chauvat, C.; Veaudor, T.; Chauvat, F. Comparative genomics of DNA recombination and repair in cyanobacteria: Biotechnological implications. Front. Microbiol. 2016, 7. [CrossRef]
- Davies, F.K.; Work, V.H.; Beliaev, A.S.; Posewitz, M.C. Engineering Limonene and Bisabolene Production in Wild Type and a Glycogen-Deficient Mutant of Synechococcus sp. PCC 7002. Front. Bioeng. Biotechnol. 2014, 2. [CrossRef]
- Yang, R.; Zhu, L.; Li, T.; Zhu, L.; Ye, Z.; Zhang, D. Photosynthetic Conversion of CO 2 Into Pinene Using Engineered Synechococcus sp. PCC 7002. Front. Bioeng. Biotechnol. 2021, 9. [CrossRef]
- Lee, H.J.; Lee, J.; Lee, S.-M.; Um, Y.; Kim, Y.; Sim, S.J.; Choi, J.-I.; Woo, H.M. Direct Conversion of CO2 to α-Farnesene Using Metabolically Engineered Synechococcus elongatus PCC 7942. J. Agric. Food Chem. 2017, 60, 10424–28. [CrossRef]
- Pattharaprachayakul, N.; Lee, H.J.; Incharoensakdi, A.; Woo, H.M. Evolutionary Engineering of Cyanobacteria to Enhance the Production of α-Farnesene from CO2. J. Agric. Food Chem. 2019, 67, 13658–13664. [CrossRef]
- Lee, H.J.; Choi, J. Il; Woo, H.M. Biocontainment of Engineered Synechococcus elongatus PCC 7942 for Photosynthetic Production of α-Farnesene from CO2. J. Agric. Food Chem. 2021, 69, 698−703. [CrossRef]
- Wang, X.; Liu, W.; Xin, C.; Zheng, Y.; Cheng, Y.; Sun, S.; Li, R.; Zhu, X.-G.; Dai, S.Y.; Rentzepis, P.M.; et al. Enhanced limonene production in cyanobacteria reveals photosynthesis limitations. Proc. Natl. Acad. Sci. 2016, 113, 14225–30. [CrossRef]
- Shinde, S.; Singapuri, S.; Jiang, Z.; Long, B.; Wilcox, D.; Klatt, C.; Jones, J.A.; Yuan, J.S.; Wang, X. Thermodynamics contributes to high limonene productivity in cyanobacteria. Metab. Eng. Commun. 2022, 14. [CrossRef]
- Mermet-Bouvier, P.; Chauvat, F. A conditional expression vector for the cyanobacteria Synechocystis sp. strains PCC6803 and PCC6714 or Synechococcus sp. strains PCC7942 and PCC6301. Curr. Microbiol. 1994, 28, 145–148. [CrossRef]
- Stanier, R.Y.; Kunisawa, R.; Mandel, M.; Cohen-Bazire, G. Purification and properties of unicellular blue-green algae (order Chroococcales). Bacteriol. Rev. 1971, 35, 171–205. [CrossRef]
- Domain, F.; Houot, L.; Chauvat, F.; Cassier-Chauvat, C. Function and regulation of the cyanobacterial genes lexA, recA and ruvB: LexA is critical to the survival of cells facing inorganic carbon starvation. Mol. Microbiol. 2004, 53, 65–80. [CrossRef]
- Stevens, S.E.; Pat Paterson, C.O.; Myers, J. The production of hydrogen peroxide by blue-green algae: a survey. J. Phycol. 1973, 9, 427–430. [CrossRef]
- Mermet-Bouvier, P.; Cassier-Chauvat, C.; Marraccini, P.; Chauvat, F. Transfer and replication of RSF1010-derived plasmids in several cyanobacteria of the genera Synechocystis and Synechococcus. Curr. Microbiol. 1993, 27, 323–327. [CrossRef]
- Blanc-Garin, V.; Chenebault, C.; Diaz-Santos, E.; Vincent, M.; Sassi, J.F.; Cassier-Chauvat, C.; Chauvat, F. Exploring the potential of the model cyanobacterium Synechocystis PCC 6803 for the photosynthetic production of various high-value terpenes. Biotechnol. Biofuels 2022, 15. [CrossRef]
- Chauvat, F.; Astier, C.; Vedel, F.; Joset-Espardellier, F. Transformation in the cyanobacterium Synechococcus R2: Improvement of efficiency; Role of the pUH24 plasmid. Mol. Gen. Genet. 1983, 191, 39–45. [CrossRef]
- Labarre, J.; Chauvat, F.; Thuriaux, P. Insertional mutagenesis by random cloning of antibiotic resistance genes into the genome of the cyanobacterium Synechocystis strain PCC 6803. J. Bacteriol. 1989, 171, 3449–3457. [CrossRef]
- Chenebault, C.; Diaz-Santos, E.; Kammerscheit, X.; Görgen, S.; Ilioaia, C.; Streckaite, S.; Gall, A.; Robert, B.; Marcon, E.; Buisson, D.-A.; et al. A Genetic Toolbox for the New Model Cyanobacterium Cyanothece PCC 7425: A Case Study for the Photosynthetic Production of Limonene. Front. Microbiol. 2020, 11, 586–601. [CrossRef]
- Veaudor, T.; Cassier-Chauvat, C.; Chauvat, F. Overproduction of the cyanobacterial hydrogenase and selection of a mutant thriving on urea, as a possible step towards the future production of hydrogen coupled with water treatment. PLoS One 2018, 13, e0198836. [CrossRef]
- Blanc-Garin, V.; Veaudor, T.; Sétif, P.; Gontero, B.; Lemaire, S.D.; Chauvat, F.; Cassier-Chauvat, C. First in vivo analysis of the regulatory protein CP12 of the model cyanobacterium Synechocystis PCC 6803: Biotechnological implications. Front. Plant Sci. 2022. [CrossRef]
- Vogel, A.I.M.; Lale, R.; Hohmann-Marriott, M.F. Streamlining recombination-mediated genetic engineering by validating three neutral integration sites in Synechococcus sp. PCC 7002. J. Biol. Eng. 2017, 11. [CrossRef]
- Gibson, D..; Young, L.; Chuang, R.-Y.; Venter, J.C.; Hutchinson, C.A.; Smith, H.. Enzymatic assembly of DNA molecules up to several hundred kilobases. Nat. Methods 2009, 6, 343–345. [CrossRef]
- Bustos, S.A.; Golden, S.S. Light-regulated expression of the psbD gene family in Synecbococcus sp. strain PCC 7942: evidence for the role of duplicated psbD genes in cyanobacteria. Mol. Gen. Genet. 1992. [CrossRef]
- Betterle, N.; Melis, A. Photosynthetic generation of heterologous terpenoids in cyanobacteria. Biotechnol. Bioeng. 2019, 116, 2041–2051. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).