Submitted:
19 August 2025
Posted:
20 August 2025
You are already at the latest version
Abstract
Keywords:
1. Role of Phosphorus in Crop Production
- Phosphorus is an essential plant nutrient required for energy transfers, photosynthesis, and cell division
- Early-season deficiencies of P can reduce final crop yield
- Phosphorus deficiency symptoms are often subtle, but can include dark green or purple coloration of leaves and stems, stunting, reduced tillering, delayed maturity, and reduced yield
- Phosphorus removed from the soil in the harvested crop should be replaced to ensure long-term sustainability
1.1. Functions of P in the Plant
1.2. Accumulation of P by the Plant
1.3. Phosphorus Deficiency Symptoms
1.4. Summary
2. Constraints to Phosphorus Availability for Crop Uptake
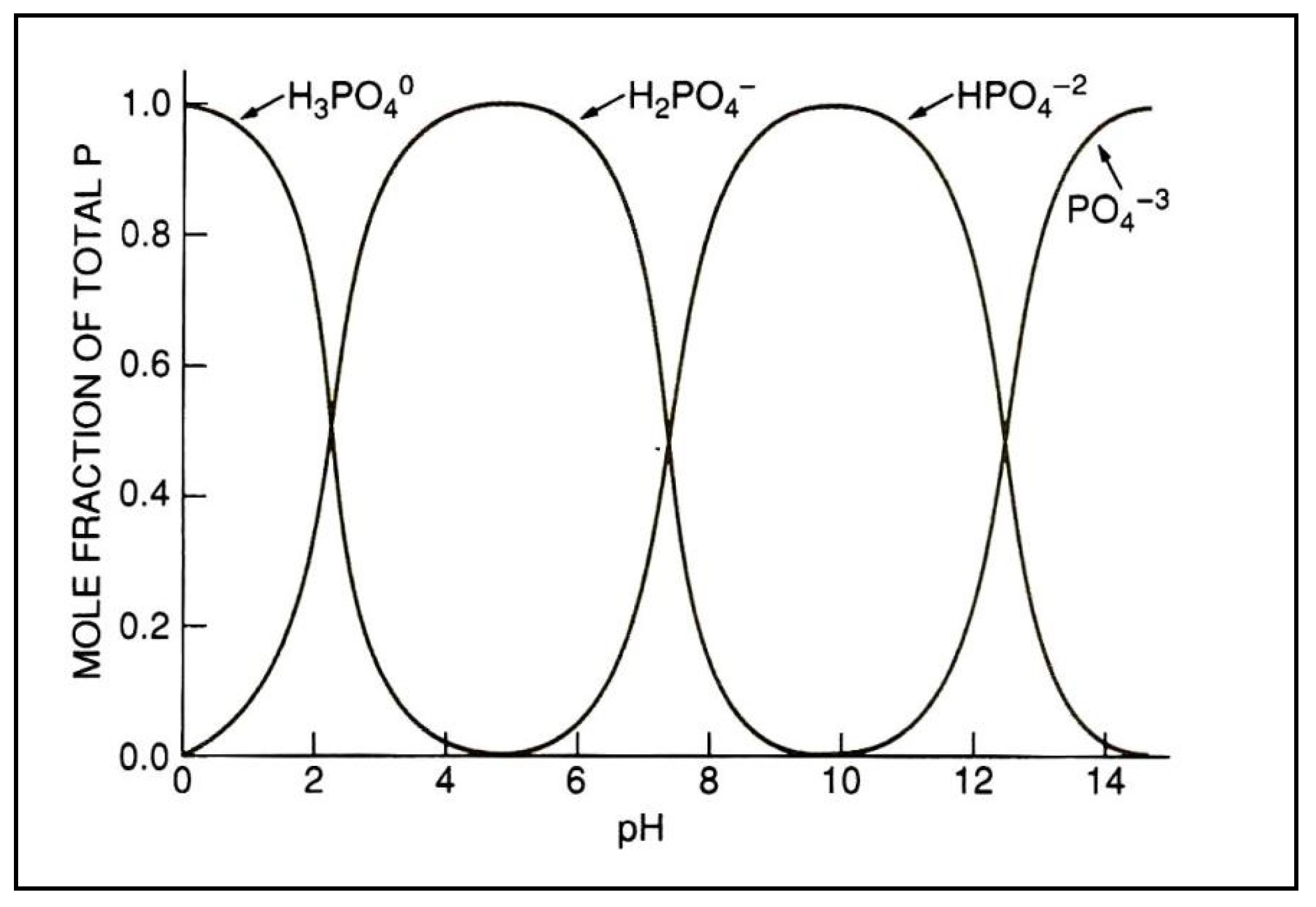
- Plants take up P from the soil solution as the inorganic orthophosphate ion Pi.
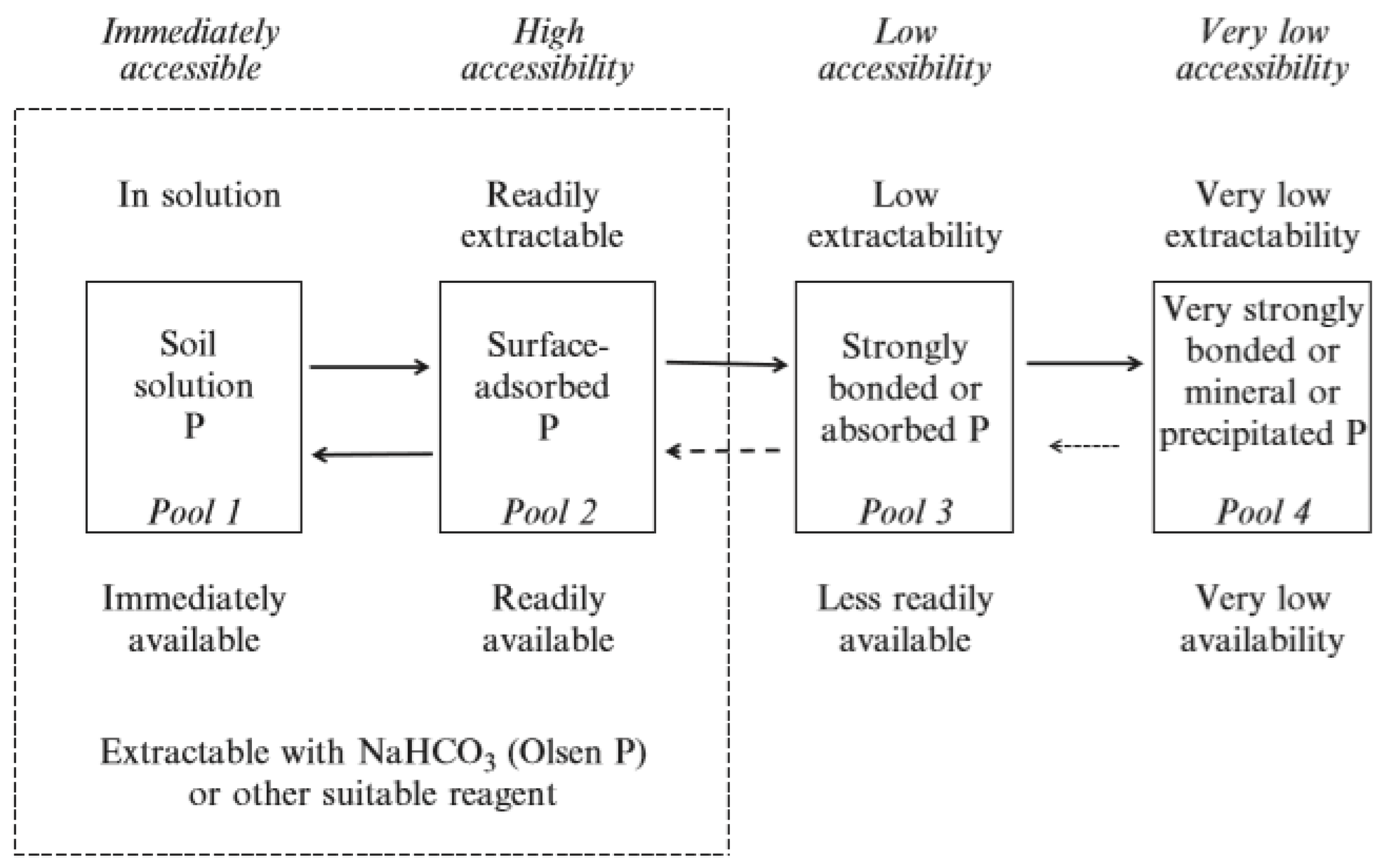
- Phosphorus concentration in the soil solution is very low and must be replenished from other soil pools to meet plant demand
- Uptake will be affected by the concentration of Pi at the root surface and the speed at which the concentration can be replenished
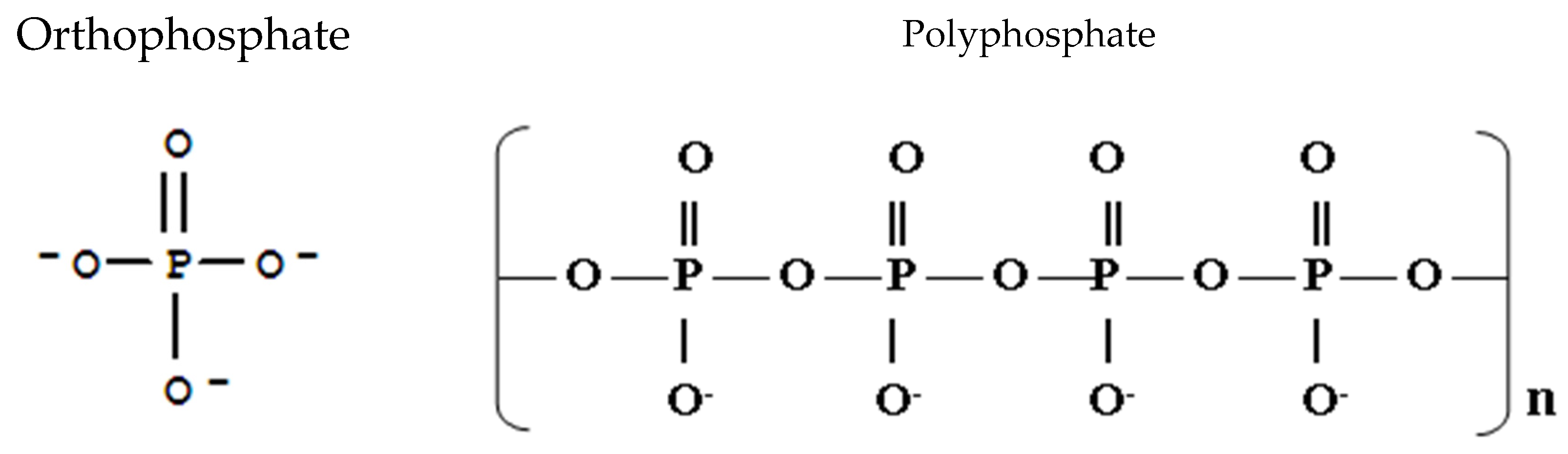
- Phosphorus fertilizer will undergo a series of adsorption and precipitation reactions that move it from solution into less soluble, labile and non-labile pools of P in the soil. These reactions are reversible and respond to the concentration gradient
- Phosphorus use efficiency can be measured through different methods that consider the short- and long-term use in the cropping system
2.1. Uptake of P from the Soil Solution
2.2. Reactions of P in the Soil
2.3. Phosphorus Use Efficiency
2.4. Summary
3. Traditional Fertilizer Formulations
- Traditional phosphate fertilizers are formulated to provide available phosphorus to the plant as required for crop growth
- Phosphorus availability will be affected by the solubility of the fertilizer source and its reactions in the soil
- Rock phosphate is the source material for most commercial phosphate fertilizers
- Presence of ammonium in the fertilizer can increase its uptake by plants
- Fluid forms of fertilizer may be more available than solid granules on dry, calcareous soils
3.1. Phosphorus Fertilizer Forms and Reactions
3.2. Phosphate Rock
3.3. Commercial Phosphate Fertilizers
3.3.1. Dry Granular Phosphate Fertilizers
3.3.2. Fluid Fertilizers
3.4. Summary
4. Microbial Products
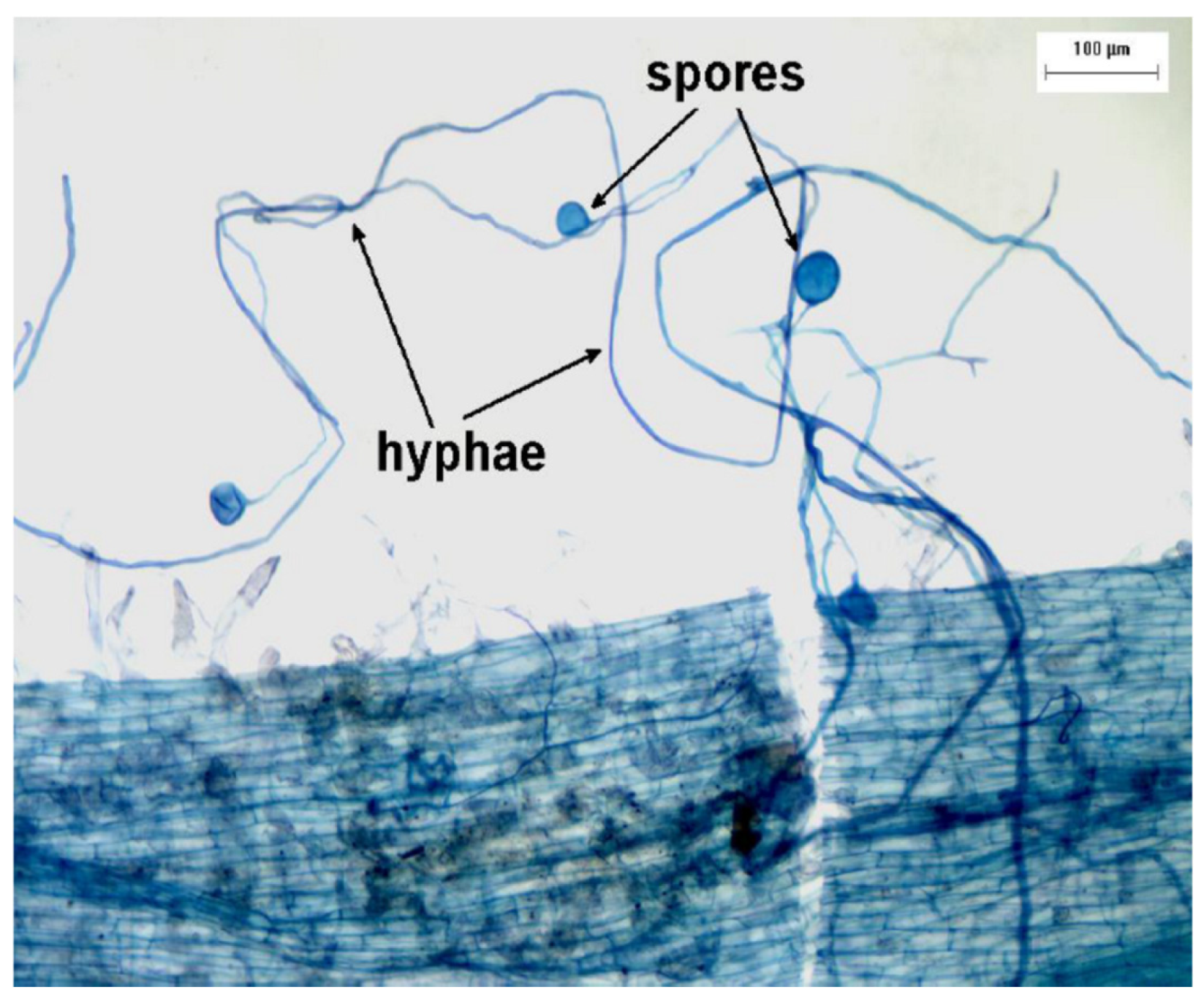
- Soil microorganisms play an important role in phosphorus interaction in the soil
- Soil microorganisms can mobilize soil P and increase its availability for plant uptake
- P solubilizing microorganisms are more often beneficial in pot studies than under field conditions
- Mycorrhizal associations are very important in natural ecosystems and for specific highly-dependant crop species
- Mycorrhizal inoculants have mixed results under field conditions
- Managing production systems to encourage the development of the natural mycorrhizal population may benefit crop types that depend on mycorrhizal colonization
- Increasing removal of soil P without replenishment can lead to loss of soil fertility over time
4.1. Microorganism and Phosphorus Availability
4.2. Phosphorus-Solubilizing Microorganisms
4.3. Mycorrhizal Inoculants
4.4. Summary
5. Humic Acids and Related Products
- Humic substances play an important role in the physical and chemical quality of soils
- Application of high rates of humic substances can act as a soil conditioner
- High rates of humic substances can increase P solution concentration and mobility, particularly in pot studies and on soils that are low in organic matter
- Low rates of humic acids have had mixed results, especially under field conditions.
- Novel fertilizer formulations, including humic substance coating or humic-metal-phosphate complexed fertilizers, also have not consistently shown an advantage over standard soluble P fertilizers
- Inconsistent benefits from humic acids may indicate that low rates used are insufficient to be effective, or that the native content of humic acids present in the soil makes additional applications unnecessary.
5.1. Characteristics of Humic Substances
5.2. Effects of Humic Acids on Soil P Reactions
5.3. Effects of Humic Substances on Plant-P Interactions
5.4. Impact of Humic Acids on Crop Yield
5.5. Humic Substance Coating or Co-Formulation with P Fertilizers
5.6. Summary
6. Fertilizer Coatings
- Coated soluble P fertilizers show some promise in improving phosphorus use efficiency.
- Coatings can effectively slow the release of soluble P fertilizers into the soil solution.
- Matching the release of the P to crop uptake can provide available P to the growing plant while limiting the conversion of soluble P to less available forms.
- Coated products can reduce the risk of seeding damage from soluble P fertilizers by lowering the fertilizer concentration in contact with the germinating seeding.
- Release pattern from the fertilizer must ensure that sufficient P is available to the crop early in the growing season to optimize crop growth.
- The economic benefit will depend on the yield differential and cost of the product use relative to alternative 4R fertilizer management options.
6.1. How Will Coated Fertilizer Products Affect P Efficiency?
6.2. Reaction of Coated Phosphorus Fertilizers
6.3. Impacts of Coated Products on Crop Yield
6.4. Impacts of Coated Products on Seedling Toxicity
6.5. Summary
7. Chemical Additives
- Additives that modify pH in the fertilizer reaction zone can improve mobility and availability of P fertilizer.
- Additives designed to increase pH around the fertilizer zone include carbonates, oxides, hydroxides, oxysulfates and silicates and can improve P availability on acid soils.
- Elemental sulphur, sulphate and ammonium can decrease pH around the fertilizer zone and can improve availability on high pH soils.
- Ammonium ions may also enhance plant rooting, which can also benefit P uptake by crops.
- Slow oxidation of elemental sulphur may reduce its effectiveness for enhancing P availability.
- The performance of substances that sequester ions has been highly variable, and they seem to be unreliable as a method of increasing P availability.
7.1. How Do Chemical Additives Increase Phosphorus Use Efficiency?
7.2. Additives to influence soil pH
7.3. Additives that Sequester Ions
7.4. Summary
8. Modified Structure Phosphorus Products
- The high surface area of nanoparticles may increase the mobility and availability of sparingly soluble phosphate sources, but effectiveness does not appear to exceed that of standard soluble fertilizer sources.
- Recovery of P from waste streams in a form that can be used agronomically can have the dual benefit of removing P from the waste stream and recycling it as a beneficial nutrient.
- Recycled materials such as layered double hydroxides and graphene oxides may act as slow- release P sources, and perform similarly to soluble P fertilizers, especially on acid soils.
- The low loading of P onto layered double hydroxides and graphene oxides increases the cost of transport and application.
- Struvite has low solubility relative to traditional P fertilizers, which will reduce its availability early in the season and recovery over the short term, but reduce the risk of seedling damage or nutrient leaching.
- Blending struvite or other slowly available sources with a soluble P source may enhance early-season availability and provide slowly available P over time.
- A large proportion of the P in struvite may still be unavailable several years after application.
- Well-designed field trials are needed to more adequately assess the effectiveness of modified structure products under a range of realistic field conditions.
8.1. What are Modified Structure Phosphorus Products
8.2. Nanoparticles
8.3. Layered Double Hydroxides
8.4. Graphene and Graphene Oxide
8.5. Metal-Organic Frameworks
8.6. Struvite
8.7. Summary
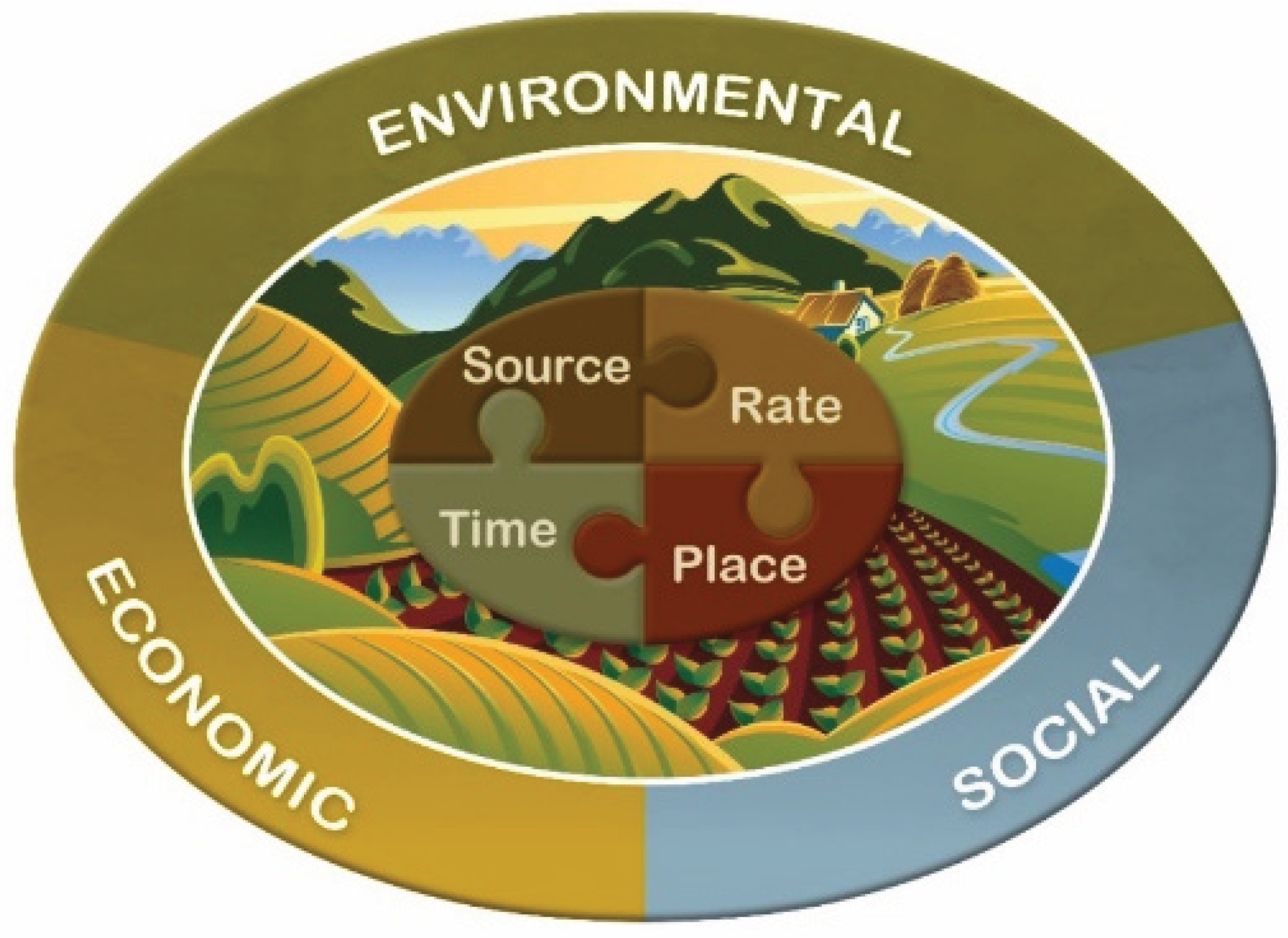
9. Enhanced Efficiency Products in a Sustainable Management System
- The 4Rs of source, rate, time and place interact and must fit with one another and with other agronomic management practices, and with economic, environmental and social goals.
- P fertilizer source should provide adequate plant-available P during early growth and throughout the growing season.
- Rate, placement and timing of P application can be selected to optimize the efficiency of different novel fertilizer sources.
- Phosphorus supply should be balanced with phosphorus removal over the long term to avoid excess depletion or accumulation.
- Efficient methods of P fertilizer management will improve agronomic, economic and environmental sustainability.
Integrating the 4Rs with Sustainable Phosphorus Management
10. Summary and Need for Future Research
- Phosphorus fertilizer will undergo a series of reversible, concentration-dependant adsorption and precipitation reactions that move it from solution into less soluble, labile and non-labile pools of P
- Phosphorus use efficiency can be measured through different methods that consider the short- and long-term use in the cropping system
- Phosphorus availability will be affected by the solubility of the fertilizer source and its reactions in the soil.
- In the long-term, P removal should be balanced with P addition to avoid nutrient depletion.
- Enhancing the removal of P through microbial inoculation will not replace P removed in the harvested crop.
- High rates of humic substances can increase P solution concentration and mobility, but low rates do not reliably improve P use efficiency.
- Coated products can match the release of the P to crop uptake to provide available P to the growing plant while limiting the conversion of soluble P to less available forms.
- Additives that modify pH in the fertilizer reaction zone can improve mobility and availability of P fertilizer.
- The performance of substances that sequester ions has been highly variable, and they seem to be unreliable as a method of increasing P availability.
- The high surface area of nanoparticles may increase the mobility and availability of sparingly soluble phosphate sources, but effectiveness does not appear to exceed that of standard soluble fertilizer sources.
- Graphene oxides, layered double hydroxides, and struvite can provide slowly available P but may not supply adequate P early in the growing season.
- Recovery of P from waste streams as a fertilizer can reduce the risk of environmental pollution and recycle P as a beneficial nutrient.
- Effectiveness in laboratory or pot studies is often greater than under field conditions. Well-designed field trials are needed to adequately assess the effectiveness of novel fertilizer products under a range of realistic field conditions.
- The economic benefit of any enhanced efficiency fertilizer will depend on the yield differential and cost of the product use relative to alternative 4R fertilizer management options.
Summary and Need for Future Research
References
- Ackerman JN, Zvomuya F, Cicek N, Flaten D (2013) Evaluation of manure-derived struvite as a phosphorus source for canola. Can J Plant Sci 93 (3):419-424. [CrossRef]
- Adnan M, Fahad S, Zamin M, Shah S, Mian IA, Danish S, Zafar-Ul-Hye M, Battaglia ML, Naz RMM, Saeed B, Saud S, Ahmad I, Yue Z, Brtnicky M, Holatko J, Datta R (2020) Coupling phosphate-solubilizing bacteria with phosphorus supplements improve maize phosphorus acquisition and growth under lime induced salinity stress. Plants (Basel) 9 (7). [CrossRef]
- Adriano DC, Murphy LS (1970) Effects of ammonium polyphosphates on yield and chemical composition of irrigated corn. Agron J 62 (5):561-567. [CrossRef]
- Ajiboye B, Akinremi OO, Hu Y, Flaten DN (2007) Phosphorus speciation of sequential extracts of organic amendments using nuclear magnetic resonance and x-ray absorption near-edge structure spectroscopies. J Environ Qual 36 (6):1563-1576. [CrossRef]
- Ajiboye B, Akinremi OO, Hu Y, Jürgensen A (2008) XANES speciation of phosphorus in organically amended and fertilized Vertisol and Mollisol. Soil Sci Soc Am J 72 (5):1256-1262. [CrossRef]
- Akinremi O, Cho C (1991a) Phosphate and accompanying cation transport in a calcareous cation-exchange resin system. Soil Sci Soc Am J 55 (4):959-964. [CrossRef]
- Akinremi O, Cho C (1991b) Phosphate transport in calcium-saturated systems: II. Experimental results in a model system. Soil Sci Soc Am J 55 (5):1282-1287. [CrossRef]
- Akinremi O, Cho C (1993) Phosphorus diffusion retardation in a calcareous system by coapplication of potassium chloride. Soil Sci Soc Am J 57 (3):845-850. [CrossRef]
- Akinremi O, Janzen H, Lemke R, Larney F (2000) Response of canola, wheat and green beans to leonardite additions. Can J Soil Sci 80 (3):437-443. [CrossRef]
- Allen SE, Mays DA (1971) Sulfur-coated fertilizers for controlled release. Agronomic evaluation. J Agric Food Chem 19 (5):809-812. [CrossRef]
- Allred SE, Ohlrogge AJ (1964) Principles of nutrient uptake from fertilizer bands. VI. Germination and emergence of corn as affected by ammonia and ammonium phosphate. Agron J 56 (3):309-313. [CrossRef]
- Alori ET, Glick BR, Babalola OO (2017) Microbial phosphorus solubilization and its potenital for use in sustainable agriculture Frontiers in Microbiology 8. [CrossRef]
- Alvarez R, Evans LA, Milham PJ, Wilson MA (2004) Effects of humic material on the precipitation of calcium phosphate. Geoderma 118 (3):245-260. [CrossRef]
- Amy C, Avice J-C, Laval K, Bressan M (2022a) Are native phosphate-solubilizing bacteria a relevant alternative to mineral fertilizations for crops? Part II: PSB inoculation enables a halving of P input and improves the microbial community in the rapeseed rhizosphere. Rhizosphere 21:100480. [CrossRef]
- Amy C, Avice J-C, Laval K, Bressan M (2022b) Are native phosphate solubilizing bacteria a relevant alternative to mineral fertilizations for crops? Part I. when rhizobacteria meet plant P requirements. Rhizosphere 21:100476. [CrossRef]
- Andelkovic IB, Kabiri S, Tavakkoli E, Kirby JK, McLaughlin MJ, Losic D (2018) Graphene oxide-Fe(III) composite containing phosphate—A novel slow release fertilizer for improved agriculture management. J Cleaner Prod 185:97-104. [CrossRef]
- Antelo J, Arce F, Avena M, Fiol S, López R, Macías F (2007) Adsorption of a soil humic acid at the surface of goethite and its competitive interaction with phosphate. Geoderma 138 (1):12-19. [CrossRef]
- Arcand MM, Lynch DH, Voroney RP, van Straaten P (2010) Residues from a buckwheat (Fagopyrum esculentum) green manure crop grown with phosphate rock influence bioavailability of soil phosphorus. Can J Soil Sci 90 (2):257-266. [CrossRef]
- Asea P, Kucey R, Stewart J (1988) Inorganic phosphate solubilization by two Penicillium species in solution culture and soil. Soil Biol Biochem 20 (4):459-464. [CrossRef]
- Bagshaw R, Vaidyanathan LV, Nye PH (1972) The supply of nutrient ions by diffusion to plant roots in soil—V. Direct determination of labile phosphate concentration gradients in a sandy soil induced by plant uptake. Plant Soil 37 (3):617-626. [CrossRef]
- Baigorri R, Urrutia O, Erro J, Mandado M, Pérez-Juste I, Garcia-Mina JM (2013) Structural characterization of anion–calcium–humate complexes in phosphate-based fertilizers. ChemSusChem 6 (7):1245-1251. [CrossRef]
- Barber S (1995) Soil Nutrient Availability. A Mechanistic Approach. 2nd edn. Wiley, New York.
- Barber SA (1980) Soil-plant interactions in the phosphorus nutrition of plants. In: Khasawneh FE, Sample EC, Kamprath EJ (eds) The Role of Phosphorus in Agriculture. ASA, CSSA, and SSSA Books. ASA, CSSA, and SSSA Madison, WI, pp 591-615. [CrossRef]
- Barber SA, Ernani PR (1991) Predicted soil phosphorus uptake as affected by banding potassium chloride with phosphorus. Soil Sci Soc Am J 55 (2):534-538. [CrossRef]
- Barber SA, Walker JM, Vasey EH (1963) Mechanisms for the movement of plant nutrients from the soil and fertilizer to the plant root. J Agric Food Chem 11 (3):204-207.
- Bargaz A, Elhaissoufi W, Khourchi S, Benmrid B, Borden KA, Rchiad Z (2021) Benefits of phosphate solubilizing bacteria on belowground crop performance for improved crop acquisition of phosphorus. Microbiological Research 252:126842. [CrossRef]
- Beaton J, Read D (1963) Effects of temperature and moisture on phosphorus uptake from a calcareous Saskatchewan soil treated with several pelleted sources of phosphorus Soil Sci Soc Am J 27 (1):61-65.
- Beckie H (1997) Response of alfalfa to phosphorus (P) fertilization and PB-50 (Provide). Saskatchewan Agricultural Development Fund, Saskatoon, SK.
- Beckie HJ, Schlechte D, Moulin AP, Gleddie SC, Pulkinen DA (1998) Response of alfalfa to inoculation with Penicillium bilaii (Provide). Can J Plant Sci 78 (1):91-102. [CrossRef]
- Bejarano Herrera WF, Rodrigues M, Bettoni Teles AP, Barth G, Pavinato PS (2016) Crop yields and soil phosphorus lability under soluble and humic-complexed phosphate fertilizers. Agron J 108 (4):1692-1702. [CrossRef]
- Bekele T, Cino B, Ehlert P, Van der Maas A, Van Diest A (1983) An evaluation of plant-borne factors promoting the solubilization of alkaline rock phosphates. Plant Soil 75 (3):361-378. [CrossRef]
- Benício LPF, Constantino VRL, Pinto FG, Vergütz L, Tronto J, da Costa LM (2017) Layered double hydroxides: new technology in phosphate fertilizers based on nanostructured materials. ACS Sustainable Chemistry & Engineering 5 (1):399-409. [CrossRef]
- Benjannet R, Nyiraneza J, Khiari L, Cambouris A, Fuller K, Hann S, Ziadi N (2020) Potato response to struvite compared with conventional phosphorus fertilizer in Eastern Canada. Agron J 112 (2):1360-1376. [CrossRef]
- Bertrand I, Holloway RE, Armstrong RD, McLaughlin MJ (2003) Chemical characteristics of phosphorus in alkaline soils from southern Australia. Aust J Soil Res 41 (1):61-76. [CrossRef]
- Bertrand I, McLaughlin MJ, Holloway RE, Armstrong RD, McBeath T (2006) Changes in P bioavailability induced by the application of liquid and powder sources of P, N and Zn fertilizers in alkaline soils. Nutr Cycling Agroecosyst 74 (1):27-40.
- Bittman S, Kowalenko CG, Hunt DE, Forge TA, Wu X (2006) Starter phosphorus and broadcast nutrients on corn with contrasting colonization by mycorrhizae. Agron J 98 (2):394-401.
- Borggaard OK, Raben-Lange B, Gimsing AL, Strobel BW (2005) Influence of humic substances on phosphate adsorption by aluminium and iron oxides. Geoderma 127 (3):270-279. [CrossRef]
- Bouldin DR, Sample EC (1959) Laboratory and greenhouse studies with monocalcium, monoammonium, and diammonium phosphates1. Soil Sci Soc Am J 23 (5):338-342. [CrossRef]
- Cahill S, Gehl RJ, Osmond D, Hardy D (2013) Evaluation of an organic copolymer fertilizer additive on phosphorus starter fertilizer response by corn Crop Manag 12 (1):CM-2013-0322-2001-RS. [CrossRef]
- Canellas LP, Olivares FL (2014) Physiological responses to humic substances as plant growth promoter. Chemical and Biological Technologies in Agriculture 1 (1):3. [CrossRef]
- Canellas LP, Olivares FL, Okorokova-Façanha AL, Façanha AR (2002) Humic acids isolated from earthworm compost enhance root elongation, lateral root emergence, and plasma membrane H+-ATPase activity in maize roots. Plant Physiol 130 (4):1951-1957.
- Chagas W, Guelfi D, Caputo ALC, Dominghetti A, Faquin V, Lopes RM, Chagas RMR (2016a) Agronomic efficiency of triple superphosphate coated by polymers in the initial growth of the coffee. 11:427-435.
- Chagas W, Guelfi D, Emrich E, Silva A, Faquin V (2016b) Agronomic efficiency of polymer-coated triple superphosphate in onion cultivated in contrasting texture soils. REVISTA CIÊNCIA AGRONÔMICA 47:439-446. [CrossRef]
- Chang C, Racz G (1977) Effects of temperature and phosphate concentration on rate of sodium pyrophosphate and sodium tripolyphosphate hydrolysis in soil. Can J Soil Sci 57 (3):271-278.
- Channab B-E, Tayi F, Aqlil M, Akil A, Essamlali Y, Chakir A, Zahouily M (2024) Graphene oxide, starch, and kraft lignin bio-nanocomposite controlled-release phosphorus fertilizer: Effect on P management and maize growth. Int J Biol Macromol 282:137190. [CrossRef]
- Chen M, Li Y, Wang C, Walter MT (2022) An investigation of the effects of humic acid on soil erosion and loss of phosphorus from soil to runoff: Experiment and modeling. Geoderma 427:116121. [CrossRef]
- Chen Q, Li Z, Qu Z, Zhou H, Qi Y, Liu Z, Zhang M (2020) Maize yield and root morphological characteristics affected by controlled-release diammonium phosphate and Paecilomyces variotii extracts. Field Crop Res 255:107862. [CrossRef]
- Chen Q, Qu Z, Li Z, Zhang Z, Ma G, Liu Z, Wang Y, Wu L, Fang F, Wei Z, Zhang M (2021) Coated diammonium phosphate combined with humic acid improves soil phosphorus availability and photosynthesis and the yield of maize. Frontiers in Plant Science 12:1-14. [CrossRef]
- Chien S, Edmeades D, McBride R, Sahrawat K (2014) Review of maleic–itaconic acid copolymer purported as urease inhibitor and phosphorus enhancer in soils. Agron J 106 (2):423-430.
- Chien S, Rehm G (2016) Theoretical equilibrium considerations explain the failure of the maleic-itaconic copolymer to increase efficiency of fertiliser phosphorus applied to soils. Soil Res 54 (1):120-124.
- Chien SH, Menon RG (1995) Factors affecting the agronomic effectiveness of phosphate rock for direct application. Fert Res 41 (3):227-234. [CrossRef]
- Chien SH, Prochnow LI, Tu S, Snyder CS (2011) Agronomic and environmental aspects of phosphate fertilizers varying in source and solubility: an update review. Nutr Cycling Agroecosyst 89 (2):229-255. [CrossRef]
- Choudhary M, Bailey L, Peck T (1996) Effect of rock phosphate and superphosphate on crop yield and soil phosphorus test in long-term fertility plots. Commun Soil Sci Plant Anal 27 (18-20):3085-3099. [CrossRef]
- Choudhary M, Bailey L, Peck T, Paul L (1994) Long-term comparison of rock phosphate with superphosphate on crop yield in two cereal-legume rotations. Can J Plant Sci 74 (2):303-310.
- Claassen N, Barber SA (1976) Simulation model for nutrient uptake from soil by a growing plant root system. Agron J 68 (6):961-964. [CrossRef]
- Close DC, Beadle CL (2003) The Ecophysiology of Foliar Anthocyanin. The Botanical Review 69 (2):149-161.
- Cruz D, Bortoletto-Santos R, Guimarães G, Polito W, Ribeiro C (2017) Role of polymeric coating on the phosphate availability as fertilizer: insight from phosphate release in soil castor polyurethane coatings. J Agric Food Chem 65. [CrossRef]
- Dahnke WC, Attoe OJ, Engelbert LE, Groskopp MD (1963) Controlling release of fertilizer constituents by means of coatings and capsules. Agron J 55 (3):242-244. [CrossRef]
- Dai M, Hamel C, Bainard LD, Arnaud MS, Grant CA, Lupwayi NZ, Malhi SS, Lemke R (2014) Negative and positive contributions of arbuscular mycorrhizal fungal taxa to wheat production and nutrient uptake efficiency in organic and conventional systems in the Canadian prairie. Soil Biol Biochem 74:156-166. [CrossRef]
- de Ávila MOT, Moreira SG, Lima FRD, Pimentel GV, Macedo JR, Nunes MR, Gomes LBW, Morais EG (2024) Effect of coating phosphorus with humic acids and micronutrients on yield of soybean and maize in succession. Journal of Agriculture and Food Research 18:101318. [CrossRef]
- de Melo BAG, Motta FL, Santana MHA (2016) Humic acids: Structural properties and multiple functionalities for novel technological developments. Materials Science and Engineering: C 62:967-974. [CrossRef]
- de Oliveira-Paiva CA, Bini D, de Sousa SM, Ribeiro VP, Dos Santos FC, de Paula Lana UG, de Souza FF, Gomes EA, Marriel IE (2024) Inoculation with Bacillus megaterium CNPMS B119 and Bacillus subtilis CNPMS B2084 improve P-acquisition and maize yield in Brazil. Frontiers in Microbiology 15. [CrossRef]
- Degryse F, Ajiboye B, Armstrong RD, McLaughlin MJ (2013) Sequestration of phosphorus-binding cations by complexing compounds is not a viable mechanism to increase phosphorus efficiency. Soil Sci Soc Am J 77 (6):2050-2059. [CrossRef]
- Degryse F, Ajiboye B, Baird R, da Silva RC, McLaughlin MJ (2016) Oxidation of elemental sulfur in granular fertilizers depends on the soil-exposed surface area. Soil Sci Soc Am J 80 (2):294-305.
- Degryse F, Baird R, Da Silva RC, McLaughlin MJ (2017) Dissolution rate and agronomic effectiveness of struvite fertilizers–effect of soil pH, granulation and base excess. Plant Soil 410 (1-2):139-152.
- Delgado A, Madrid A, Kassem S, Andreu L, Del Campillo MDC (2002) Phosphorus fertilizer recovery from calcareous soils amended with humic and fulvic acids. Plant Soil 245 (2):277-286.
- DeRosa MC, Monreal C, Schnitzer M, Walsh R, Sultan Y (2010) Nanotechnology in fertilizers. Nature Nanotechnology 5 (2):91-91. [CrossRef]
- Dick R, Tabatabai M (1986) Hydrolysis of polyphosphates in soils. Soil Sci 142 (3):132-140.
- do Nascimento CAC, Pagliari PH, Faria LdA, Vitti GC (2018) Phosphorus mobility and behavior in soils treated with calcium, ammonium, and magnesium phosphates. Soil Sci Soc Am J 82 (3):622-631. [CrossRef]
- Doydora S, Hesterberg D, Klysubun W (2017) Phosphate solubilization from poorly crystalline iron and aluminum hydroxides by AVAIL copolymer. Soil Sci Soc Am J 81 (1):20-28. [CrossRef]
- Du Z-Y, Wang Q-H, Liu F-C, Ma H-L, Ma B-Y, Malhi SS (2013) Movement of phosphorus in a calcareous soil as affected by humic acid. Pedosphere 23 (2):229-235. [CrossRef]
- Dudenhoeffer C, Nelson K, Motavalli P, Burdick B, Dunn D, Goyne K (2013) Utility of phosphorus enhancers and strip-tilolage for corn production J Agric Sci 5. [CrossRef]
- Dudenhoeffer C, Nelson K, Motavalli P, Dunn D, Stevens W, Goyne K, Nathan M, Scharf P (2012) Corn production as affected by phosphorus enhancers, phosphorus source and lime J Agric Sci 4. [CrossRef]
- Ekin Z (2010) Performance of phosphate solubilizing bacteria for improving growth and yield of sunflower(Helianthus annuus L.) in the presence of phosphorus fertilizer. African Journal of Biotechnology 925:3794-3800.
- El Mazlouzi M, Morel C, Chesseron C, Robert T, Mollier A (2020a) Contribution of External and Internal Phosphorus Sources to Grain P Loading in Durum Wheat (Triticum durum L.) Grown Under Contrasting P Levels. Frontiers in Plant Science 11. [CrossRef]
- El Mazlouzi M, Morel C, Robert T, Yan B, Mollier A (2020b) Phosphorus uptake and partitioning in two durum wheat cultivars with contrasting biomass allocation as affected by different P supply during grain filling. Plant and Soil 449 (1):179-192. [CrossRef]
- El-Leboudi AE-S, El-Sebaay A-ES, Abd-Elrahman SH, El-Etr WM, Saad HY (2019) Effect of silicon and phosphorus additions and their interactions on wheat plants grown on a clay soil Asian Soil Research Journal 2 (1):1-10. [CrossRef]
- Elliott AJ, Daniell TJ, Cameron DD, Field KJ (2021) A commercial arbuscular mycorrhizal inoculum increases root colonization across wheat cultivars but does not increase assimilation of mycorrhiza-acquired nutrients. Plants, people, planet 3 (5):588-599. [CrossRef]
- Elliott DE, Reuter DJ, Reddy GD, Abbott RJ (1997) Phosphorus nutrition of spring wheat (Triticum aestivum L.). 2. Distribution of phosphorus in glasshouse-grown wheat and the diagnosis of phosphorus deficiency by plant analysis. Aust J Agr Res 48 (6):869-881.
- Ellis R, Quader M, Truog E (1955) Rock phosphate availability as influenced by soil pH 1. Soil Sci Soc Am J 19 (4):484-487.
- Eltohamy KM, Li J, Gouda M, Menezes-Blackburn D, Milham PJ, Khan S, Li F, Liu C, Xu J, Liang X (2023) Nano and fine colloids suspended in the soil solution regulate phosphorus desorption and lability in organic fertiliser-amended soils. Sci Total Environ 858:160195. [CrossRef]
- Entz M, Guilford R, Gulden R (2001) Crop yield and soil nutrient status on 14 organic farms in the eastern portion of the northern Great Plains. Can J Plant Sci 81 (2):351-354. [CrossRef]
- Ernani P, Barber S (1991) Predicted soil phosphorus uptake as affected by banding potassium chloride with phosphorus. Soil Science Society of America journal (USA). [CrossRef]
- Erro J, Urrutia O, Baigorri R, Aparicio-Tejo P, Irigoyen I, Storino F, Mandado M, Yvin JC, Garcia-Mina JM (2012) Organic complexed superphosphates (CSP): Physicochemical characterization and agronomical properties. J Agric Food Chem 60 (8):2008-2017. [CrossRef]
- Etesami H, Schaller J (2023) Improving phosphorus availability to rice through silicon management in paddy soils: A review of the role of silicate-solubilizing bacteria. Rhizosphere 27:100749. [CrossRef]
- Everaert M, da Silva RC, Degryse F, McLaughlin MJ, Smolders E (2018a) Limited dissolved phosphorus runoff losses from layered double hydroxide and struvite fertilizers in a rainfall simulation study. J Environ Qual 47 (2):371-377. [CrossRef]
- Everaert M, Degryse F, McLaughlin MJ, De Vos D, Smolders E (2017) Agronomic effectiveness of granulated and powdered P-exchanged Mg–Al LDH relative to struvite and MAP. J Agric Food Chem 65 (32):6736-6744. [CrossRef]
- Everaert M, Degryse F, McLaughlin MJ, Smolders S, Andelkovic I, Baird R, Smolders E (2022) Enhancing the phosphorus content of layered double hydroxide fertilizers by intercalating polymeric phosphate instead of orthophosphate: A feasibility study. J Colloid Interface Sci 628:519-529. [CrossRef]
- Everaert M, Slenders K, Dox K, Smolders S, De Vos D, Smolders E (2018b) The isotopic exchangeability of phosphate in Mg-Al layered double hydroxides. J Colloid Interface Sci 520:25-32. [CrossRef]
- Everaert M, Warrinnier R, Baken S, Gustafsson J-P, De Vos D, Smolders E (2016) Phosphate-exchanged Mg–Al ayered double hydroxides: a new slow release phosphate fertilizer ACS Sustainable Chemistry & Engineering 4 (8):4280-4287.
- Farden KD, Knight JD Strategies for improving soil fertility in mature alfalfa stands In: Saskatchewan Soils and Crops Conference, Saskatoon, SK, 2005. p 6.
- Ferron LME, Koopmans GF, Rommelse H, Van Groenigen JW, Regelink IC (2024) A better understanding of the effectiveness of placed phosphorus fertilisation with struvite for silage maize: A pot experiment. Geoderma 448:116939. [CrossRef]
- Fertahi S, Bertrand I, Ilsouk M, Oukarroum A, Amjoud MB, Zeroual Y, Barakat A (2020) New generation of controlled release phosphorus fertilizers based on biological macromolecules: Effect of formulation properties on phosphorus release. Int J Biol Macromol 143:153-162. [CrossRef]
- Fixen P, Brentrup F, Bruulsema T, Garcia F, Norton R, Zingore S (2015) Nutrient/fertilizer use efficiency: measurement, current situation and trends. In: Drechsel P, Heffer P, Magen H, Mikkelsen R, Wichelns D (eds) Managing water and fertilizer for sustainable agricultural intensification, vol 8. International Fertilizer Industry Association (IFA), International Water Management Institute (IWMI), International Plant Nutrition Institute (IPNI), and International Potash Institute (IPI). , Paris, France, .
- Fixen P, Ludwick A, Olsen S (1983) Phosphorus and potassium fertilization of irrigated alfalfa on calcareous soils: II. Soil phosphorus solubility relationships 1. Soil Sci Soc Am J 47 (1):112-117.
- Follett RH, Murphy LS, Donahue RL (1981) Fertilizers and Soil Amendments. Prentice-Hall, Inc., Englewood Cliffs, New Jersey.
- Francioni M, Palmieri M, Fiorentini M, Deligios PA, Monaci E, Vischetti C, Rossa ÜB, Trozzo L, Bianchini M, Rivosecchi C, Ledda L, Orsini R, Santilocchi R, D’Ottavio P (2024) Scarcity of P-fertilisers: Humic-complexed phosphate as an adaptive solution for wheat and maize under rainfed conditions. European Journal of Agronomy 156:127143. [CrossRef]
- Freiberg Y, Bar-Yosef B, Fine P, Borisover M, Levkovitch I, Baram S (2024) Does dissolved humic acid suppress orthophosphate adsorption to soils? It depends on the type of clay in the soil. Applied Clay Science 256. [CrossRef]
- Fried M (1953) The feeding power of plants for phosphates 1. Soil Sci Soc Am J 17 (4):357-359.
- Gan Y, Hanson KG, Zentner RP, Selles F, McDonald CL (2005) Response of lentil to microbial inoculation and low rates of fertilization in the semiarid Canadian prairies. Can J Plant Sci 85 (4):847-855. [CrossRef]
- Gao S, Zhang S, Yuan L, Li Y, Zhao L, Wen Y, Xu J, Hu S, Zhao B (2023) Effects of humic acid–enhanced phosphate fertilizer on wheat yield, phosphorus uptake, and soil available phosphorus content. Crop Sci 63 (2):956-966. [CrossRef]
- García MC, Díez JA, Vallejo A, García L, Cartagena MC (1997) Effect of applying soluble and coated phosphate fertilizers on phosphate availablity in calcareous soils and on P absorption by a rye-grass crop. J Agric Food Chem 45 (5):1931-1936. [CrossRef]
- Gavito ME, Miller MH (1998) Changes in mycorrhiza development in maize indeed by crop management practices. Plant Soil 198 (2):185-192.
- Giroto AS, Guimarães GGF, Foschini M, Ribeiro C (2017) Role of slow-release nanocomposite fertilizers on nitrogen and phosphate availability in soil. Scientific Reports 7. [CrossRef]
- Glass ADM, Beaton JD, Bomke A (1980) Role of P in plant nutrition. Proceedings of the Western Canada Phosphate Symposium:357-368.
- Goh T, Karamanos R, Lee J (2013) Effects of phosphorus form on short-term solubility and availability in soils. Commun Soil Sci Plant Anal 44 (1-4):136-144.
- Goos R, Johnson B, Stack R (1994) Penicillium bilaji and phosphorus fertilization effects on the growth, development, yield and common root rot severity of spring wheat. Fert Res 39 (2):97-103.
- Gordon B, Tindall T (2006) Fluid P performance improved with polymers. Fluid J 14:12-13.
- Grant C, Bittman S, Montreal M, Plenchette C, Morel C (2005) Soil and fertilizer phosphorus: Effects on plant P supply and mycorrhizal development. Can J Plant Sci 85 (1):3-14. [CrossRef]
- Grant C, Clayton G, Monreal M, Lupwayi N, Turkington K, McLaren D (2006) Improving phosphorus nutrition in wheat. Brandon Research Centre. Agriculture and AgriFood Canda, Brandon, MB.
- Grant C, Tenuta M, Flaten D, Gowalko E (2008) Impact of cropping sequence and tillage system on response to P fertilization in durum wheat and soybean. Brandon, MB.
- Grant CA (2002) Effect of coated and on-coated monoammonium phosphate fertilizer on phosphorus uptake, crop yield and nutrient profile of wheat and canola Agriculture and Agri-Food Canada, Brandon, MB.
- Grant CA (2011) Impact of traditional and enhanced efficiency phosphorus fertilizers on canola emergence, yield, maturity and quality. Agriculture and Agri-Food Canada, Brandon, MB.
- Grant CA, Bailey LD, Harapiak JT, Flore NA (2002) Effect of phosphate source, rate and cadmium content and use of Penicillium bilaii on phosphorus, zinc and cadmium concentration in durum wheat grain. J Sci Food Agric 82 (3):301-308.
- Grant CA, Dribnenki JCP, Bailey LD (2000) Cadmium and zinc concentrations and ratios in seed and tissue of solin (cv Linola™ 947) and flax (cvs McGregor and Vimy) as affected by nitrogen and phosphorus fertiliser and provide (Penicillium bilaji. J Sci Food Agric 80 (12):1735-1743.
- Grant CA, Flaten DN, Tomasiewicz DJ, Sheppard SC (2001) The importance of early season phosphorus nutrition. Can J Plant Sci 81 (2):211-224.
- Grant CA, Monreal MA, Irvine RB, Mohr RM, McLaren DL, Khakbazan M (2009) Crop response to current and previous season applications of phosphorus as affected by crop sequence and tillage. Can J Plant Sci 89 (1):49-66. [CrossRef]
- Grant CA, Relf-Eckstein J (2009) Impact of traditional and enhanced efficiency phosphorus fertilizers on canola emergence, yield, maturity and quality. Report submitted to Canola council of Canada, Agrium Fertilizers, and Simplot Fertilizers. Agriculture and Agri-Food Canada, Brandon, MB.
- Grenkow LA (2013) Effect of seed-placed phosphorus and sulphur fertilizers on canola plant stand, early season biomass and seed yield., University of Manitoba, Winnipeg, MB.
- Grenkow LA, Flaten D, Grant C, Heard J (2013) Seed-placed phosphorus and sulphur fertilizers: Effect on canola plant stand and yield. Paper presented at the Saskatchewan Soils and Crops Conference, Saskatoon, .
- Guelfi D, Nunes APP, Sarkis LF, Oliveira DP (2022) Innovative phosphate fertilizer technologies to improve phosphorus use efficiency in agriculture. Sustainability 14 (21):14266. [CrossRef]
- Gulden RH, Vessey JK (2000) Penicillium bilaii inoculation increases root-hair production in field pea. Can J Plant Sci 80 (4):801-804.
- Guo H, White JC, Wang Z, Xing B (2018) Nano-enabled fertilizers to control the release and use efficiency of nutrients. Current Opinion in Environmental Science & Health 6:77-83.
- Guppy C, Menzies N, Moody P, Blamey FPC (2005) Competitive sorption reactions between phosphorus and organic matter in soil: A review. Aust J Soil Res 43. [CrossRef]
- Hamel C (2004) Impact of arbuscular mycorrhizal fungi on N and P cycling in the root zone. Can J Soil Sci 84 (4):383-395. [CrossRef]
- Hamel C, Ellouze W, Bainard L, Navarro-Borrell A, Rivera R, Gan Y (2014) Bases for the management of the AM symbiosis in cropping systems of the prairie Paper presented at the Saskatchewan Soils and Crops.
- Hamel C, Strullu D-G (2006) Arbuscular mycorrhizal fungi in field crop production: potential and new direction. Can J Plant Sci 86 (4):941-950.
- Hartz TK, Bottoms TG (2010) Humic substances generally ineffective in improving vegetable crop nutrient uptake or productivity. HortScience horts 45 (6):906-910. [CrossRef]
- Havlin JL, Tisdale SL, Nelson WL, Beaton JD (2014) Soil Fertility and Fertilizers: An Introduction to Nutrient Management. 8th edn. Pearson, Inc., Upper Saddle River, NJ, USA.
- Haynes RJ, Zhou Y-F (2018) Competitive and noncompetitive adsorption of silicate and phosphate by two acid Si-deficient soils and their effects on P and Si extractability. Soil Sci Plant Nutr 64 (4):535-541. [CrossRef]
- Hedley M, McLaughlin M (2005) Reactions of phosphate fertilizers and by-products in soils. In: Sims JT, Sharpley AN (eds) Phosphorus: Agriculture and the Environment. vol 46. American Society of Agronomy, Crop Science Society of America, Soil Science Society of America, Madison, WI, pp 181-252.
- Hegab R, Kotp YH, Eissa D (2018) Using nanotechnology for enhancing phosphorus fertilizer use efficiency of peanut bean grown in sandy soils. Journal of Advanced Pharmacy Education and Research 8:59-67. [CrossRef]
- Hertzberger AJ, Cusick RD, Margenot AJ (2020) A review and meta-analysis of the agricultural potential of struvite as a phosphorus fertilizer. Soil Sci Soc Am J 84 (3):653-671.
- Hettiarachchi GM, Lombi E, McLaughlin MJ, Chittleborough D, Self P (2006) Density changes around phosphorus granules and fluid bands in a calcareous soil. Soil Sci Soc Am J 70 (3):960-966. [CrossRef]
- Hinsinger P (2001) Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: A review. Plant Soil 237 (2):173-195. [CrossRef]
- Hinsinger P, Gilkes R (1995) Root-induced dissolution of phosphate rock in the rhizosphere of lupins grown in alkaline soil. Soil Res 33 (3):477-489. [CrossRef]
- Holloway RE, Bertrand I, Frischke AJ, Brace DM, McLaughlin MJ, Shepperd W (2001) Improving fertiliser efficiency on calcareous and alkaline soils with fluid sources of P, N and Zn. Plant Soil 236 (2):209-219.
- Holzapfel C (2014) Field Pea, Lentil and Soybean Response to Rhizobial and Mycorrhizal Inoculation (Project #201300392) Agricultural Demonstration of Practices and Technologies (ADOPT) Program Indian Head Agricultural Research Foundation, Indian Head, Sk.
- Hopkins B, Ellsworth J (2005) Phosphorus availability with alkaline/calcareous soil. Paper presented at the Western Nutrient Management Conference, Salt Lake City, .
- Hopkins B, Stark J (2003) Humic acid effects on potato response to phosphorus Paper presented at the Presented at the Idaho Potato Conference Pocatello, .
- Hopkins BG (2015) Phosphorus. In: Barker AV, Pilbeam DJ (eds) Handbook of Plant Nutrition. second edn. CRC press, Boca Ratan, FL, pp 65 -126.
- Hopkins BG, Fernelius KJ, Hansen NC, Eggett DL (2018) AVAIL phosphorus enhancer: meta-analysis of 503 field evaluations Agron J 110 (1):389-398. [CrossRef]
- Hoppo SD, Elliott DE, Reuter DJ (1999) Plant tests for diagnosing phosphorus deficiency in barley (Hordeum vulgare L.). Aust J Exp Agr 39 (7):857-872.
- Huygens D, Saveyn HGM (2018) Agronomic efficiency of selected phosphorus fertilisers derived from secondary raw materials for European agriculture. A meta-analysis. Agron Sustain Dev 38 (5):52. [CrossRef]
- IPNI (2016 ) 4R Plant Nutrition Manual: A Manual for Improving the Management of Plant Nutrition. In: Bruulsema TW, Fixen PE, Sulewski GD (eds). International Plant Nutrition Institute, Peachtree Corners, GA, USA.
- Izhar Shafi M, Adnan M, Fahad S, Wahid F, Khan A, Yue Z, Danish S, Zafar-ul-Hye M, Brtnicky M, Datta R (2020) Application of single superphosphate with humic acid improves the growth, yield and phosphorus uptake of wheat (Triticum aestivum L.) in calcareous osil. Agronomy 10 (9):1224.
- Jakobsen I, Abbott LK, Robson AD (1992) External hyphae of vesicular-arbuscular mycorrhizal fungi associated with Trifolium subterraneum L. 2. Hyphal transport of 32P over defined distances. New Phytol 120 (4):509-516.
- Jing J, Zhang F, Rengel Z, Shen J (2012) Localized fertilization with P plus N elicits an ammonium-dependent enhancement of maize root growth and nutrient uptake. Field Crop Res 133:176-185. [CrossRef]
- Jing J, Zhang S, Yuan L, Li Y, Lin Z, Xiong Q, Zhao B (2020) Combining humic acid with phosphate fertilizer affects humic acid structure and its stimulating efficacy on the growth and nutrient uptake of maize seedlings. Scientific Reports 10 (1):17502. [CrossRef]
- Jing J, Zhang S, Yuan L, Li Y, Zhang Y, Ye X, Zhang L, Xiong Q, Wang Y, Zhao B (2023) Effects of incorporating different proportions of humic acid into phosphate fertilizers on phosphorus migration and transformation in soil. Agronomy 13 (6):1576. [CrossRef]
- Johnston A, Richards I (2003) Effectiveness of different precipitated phosphates as phosphorus sources for plants. Soil Use Manage 19 (1):45-49. [CrossRef]
- Johnston AE, Poulton PR, Fixen PE, Curtin D (2014) Phosphorus: its efficient use in agriculture. In: Advances in agronomy, vol 123. Elsevier, pp 177-228.
- Jones CA, Jacobsen JS, Mugaas A (2007) Effect of low-rate commercial humic acid on phosphorus availability, micronutrient uptake, and spring wheat yield. Commun Soil Sci Plant Anal 38 (7-8):921-933.
- Joudeh N, Linke D (2022) Nanoparticle classification, physicochemical properties, characterization, and applications: a comprehensive review for biologists. Journal of Nanobiotechnology 20 (1):262. [CrossRef]
- Kabiri S, Andelkovic IB, da Silva RC, Degryse F, Baird R, Tavakkoli E, Losic D, McLaughlin MJ (2020) Engineered phosphate fertilizers with dual-release properties. Industrial & Engineering Chemistry Research 59 (13):5512-5524. [CrossRef]
- Kabiri S, Degryse F, Tran DN, da Silva RC, McLaughlin MJ, Losic D (2017) Graphene oxide: a new carrier for slow release of plant micronutrients. ACS Applied Materials & Interfaces 9 (49):43325-43335. [CrossRef]
- Kar G, Peak D, Schoenau JJ (2012) Spatial distribution and chemical speciation of soil phosphorus in a band application. Soil Sci Soc Am J 76 (6):2297-2306. [CrossRef]
- Karamanos R, Puurveen D (2011) Evaluation of a polymer treatment as enhancer of phosphorus fertilizer efficiency in wheat. Can J Soil Sci 91 (1):123-125. [CrossRef]
- Karamanos RE, Flore NA, Harapiak JT (2010) Re-visiting use of Penicillium bilaii with phosphorus fertilization of hard red spring wheat. Can J Plant Sci 90 (3):265-277. [CrossRef]
- Katanda Y, Zvomuya F, Flaten D, Cicek N (2016) Hog-manure-recovered struvite: Effects on canola and wheat biomass yield and phosphorus use efficiencies. Soil Sci Soc Am J 80 (1):135-146.
- Katanda Y, Zvomuya F, Flaten D, Cicek N, Amarakoon I (2019) Effects of seed-placed hog manure-recovered struvite on canola seedling emergence. Agron J 111:1-7.
- Kaya C, Şenbayram M, Akram NA, Ashraf M, Alyemeni MN, Ahmad P (2020) Sulfur-enriched leonardite and humic acid soil amendments enhance tolerance to drought and phosphorus deficiency stress in maize (Zea mays L.). Scientific Reports 10 (1):6432. [CrossRef]
- Khan A, Baloch MS, Ullah N, Abidin SZU, Bhatti MZ, Khan R, Khan AA, Ismail H, Saeed A, Gul H (2023) Comparative efficacy of phosphate solubilizing bacteria and synthetic phosphate fertilizers on the growth of wheat. Plant Science Today 10 (3):37-47. [CrossRef]
- Khasawneh FE, Hashimoto I, Sample EC (1979) Reactions of Ammonium Ortho- and Polyphosphate Fertilizers in Soil: II. Hydrolysis and Reactions with Soil1. Soil Sci Soc Am J 43 (1):52-58. [CrossRef]
- Khourchi S, Elhaissoufi W, Ibnyasser A, Haddine M, Ghani R, Zeroual Y, Delaplace P, Bargaz A (2023) Integrated use of polyphosphate and P-solubilizing bacteria enhanced P use efficiency and growth performance of durum wheat. Frontiers in Microbiology 14. [CrossRef]
- Knight JD (2011) Soil Activity of P Solubilizing Microorganisms. Report to Saskatchewan Agricultural Development Fund. University of Saskatchewan, Saskatoon, SK.
- Konesky D, Siddiqi M, Glass A, Hsiao A (1989) Wild oat and barley interactions: varietal differences in competitiveness in relation to phosphorus supply. Can J Bot 67 (11):3366-3371. [CrossRef]
- Kong L, Tian Y, Wang Y, Li N, Liu Y, Pang Z, Huang X, Li M, Zhang J, Zuo W (2019) Periclase-induced generation of flowerlike clay-based layered double hydroxides: A highly efficient phosphate scavenger and solid-phase fertilizer. Chemical Engineering Journal 359:902-913. [CrossRef]
- Kopittke P, Lombi E, Wang P, Schjørring J, Husted S (2019) Nanomaterials as fertilizers for improving plant mineral nutrition and environmental outcomes. Environmental Science: Nano 6. [CrossRef]
- Kottegoda N, Sandaruwan C, Priyadarshana G, Siriwardhana A, Rathnayake UA, Berugoda Arachchige DM, Kumarasinghe AR, Dahanayake D, Karunaratne V, Amaratunga GAJ (2017) Urea-hydroxyapatite nanohybrids for slow release of nitrogen. ACS Nano 11 (2):1214-1221. [CrossRef]
- Krey T, Vassilev N, Baum C, Eichler-Löbermann B (2013) Effects of long-term phosphorus application and plant-growth promoting rhizobacteria on maize phosphorus nutrition under field conditions. Eur J Soil Biol 55:124-130. [CrossRef]
- Kroeker MP (2005) Agronomic evaluation of a homogeneous nitrogen-phosphorus-sulphur fertilizer in southern Manitoba University of Manitoba, Winnipeg, MB.
- Kucey R, Bole J (1984) Availability of phosphorus from 17 rock phosphates in moderately and weakly acidic soils as determined by 32p dilution, A value, and total P uptake methods. Soil Sci 138 (2):180.
- Kucey RMN (1988) Effect of Penicillium bilaji on the solubility and uptake of P and micronutrients from soil by wheat. Can J Soil Sci 68:261-270. [CrossRef]
- Kucey RMN, Leggett ME (1989) Increased yields and phosphorus uptake by Westar canola (Brassica napus L.) inoculated with a phosphate-solubilizing isolate of Penicillium bilaji. Can J Soil Sci 69:425-432. [CrossRef]
- Kumaragamage D, Akinremi OO, Cho CM, Goh TB (2004) Phosphorus diffusion from monocalcium phosphate co-applied with salts in a calcareous soil. Can J Soil Sci 84 (4):447-458. [CrossRef]
- Kusi NYO, Stevens WB, Sintim HY, Garcia y Garcia A, Mesbah AO (2021) Phosphorus fertilization and enhanced efficiency products effects on sugarbeet. Ind Crops Prod 171:113887. [CrossRef]
- Lawrencia D, Wong SK, Low DYS, Goh BH, Goh JK, Ruktanonchai UR, Soottitantawat A, Lee LH, Tang SY (2021) Controlled release fertilizers: a review on coating materials and mechanisms of release Plants 10 (2):238. [CrossRef]
- Leggett ME, Newlands NK, Greenshields DL, West L, Inman S, Koivunen ME (2014) Maize yield response to a phosphorus-solubilizing microbial inoculant in field trials. J Agric Sci 153:1464—1478. [CrossRef]
- Leon P, Nakayama Y, Margenot AJ (2024) Field-scale evaluation of struvite phosphorus and nitrogen leaching relative to monoammonium phosphate. J Environ Qual 53 (1):23-34. [CrossRef]
- Leventoglu H, Erdal I (2014) Effect of high humic substance levels on growth and nutrient concentration of corn under calcareous conditions. J Plant Nutr 37:2074—2084. [CrossRef]
- Lewis ET, Racz G (1969) Phosphorus movement in some calcareous and noncalcareous Manitoba soils. Can J Soil Sci 49 (3):305-312. [CrossRef]
- Lindsay WL, Moreno EC (1960) Phosphate phase equilibria in soils1. Soil Sci Soc Am J 24 (3):177-182. [CrossRef]
- Liu R, Lal R (2014) Synthetic apatite nanoparticles as a phosphorus fertilizer for soybean (Glycine max). Scientific reports 4 (1):5686.
- Liu X, Zhi Y, Tian Y, Yuan Y, Zhong H (2024) Phosphate-adsorbed by concrete-based layered double hydroxide: a slow-release phosphate fertilizer. Pol J Environ Stud 33 (5):5191-5198. [CrossRef]
- Lombi E, McLaughlin MJ, Johnston C, Armstrong RD, Holloway RE (2004) Mobility and lability of phosphorus from granular and fluid monoammonium phosphate differs in a calcareous soil. Soil Sci Soc Am J 68 (2):682-689. [CrossRef]
- Lombi E, McLaughlin MJ, Johnston C, Armstrong RD, Holloway RE (2005) Mobility, solubility and lability of fluid and granular forms of P fertiliser in calcareous and non-calcareous soils under laboratory conditions. Plant Soil 269 (1-2):25-34. [CrossRef]
- Lombi E, Scheckel KG, Armstrong RD, Forrester S, Cutler JN, Paterson D (2006) Speciation and Distribution of Phosphorus in a Fertilized Soil. Soil Sci Soc Am J 70 (6):2038-2048. [CrossRef]
- Lyons G, Genc Y (2016) Commercial humates in agriculture: real substance or smoke and mirrors? Agronomy 6 (4):50. [CrossRef]
- Mabagala FS, Mng’ong’o ME (2022) On the tropical soils; The influence of organic matter (OM) on phosphate bioavailability. Saudi Journal of Biological Sciences 29 (5):3635-3641. [CrossRef]
- Majeed Z, Ramli NK, Mansor N, Man Z (2015) A comprehensive review on biodegradable polymers and their blends used in controlled-release fertilizer processes. Reviews in Chemical Engineering 31 (1):69-95. [CrossRef]
- Malhi S, Haderlein L, Pauly D, Johnston A (2002) Improving fertilizer phosphorus use efficiency. Development 85 (2):18-23.
- Malhi SS, Brandt SA, Vera CL, Leach D (2014) Potential of rock phosphate and other organic/inorganic amendments in preventing P deficiency in barley on a P-deficient soil in northeastern Saskatchewan. Paper presented at the Saskatchewan Soils and Crop Conference, Saskatoon, SK, .
- Malhi SS, Johnston AM, Schoenau JJ, Wang ZH, Vera CL (2006) Seasonal biomass accumulation and nutrient uptake of wheat, barley and oat on a Black Chernozem soil in Saskatchewan. Can J Plant Sci 86 (4):1005-1014. [CrossRef]
- Malhi SS, Johnston AM, Schoenau JJ, Wang ZH, Vera CL (2007b) Seasonal biomass accumulation and nutrient uptake of pea and lentil on a Black Chernozem soil in Saskatchewan. J Plant Nutr 30 (5):721-737. [CrossRef]
- Malhotra H, Vandana, Sharma S, Pandey R (2018) Phosphorus Nutrition: Plant Growth in Response to Deficiency and Excess. Plant Nutrients and Abiotic Stress Tolerance. Springer, Singapore. [CrossRef]
- Mastronardi E, Tsae P, Zhang X, Monreal C, DeRosa MC (2015) Strategic role of nanotechnology in fertilizers: potential and limitations. Nanotechnologies in food and agriculture:25-67. [CrossRef]
- McBeath TM, Armstrong RD, Lombi E, McLaughlin MJ, Holloway RE (2005) Responsiveness of wheat (Triticum aestivum) to liquid and granular phosphorus fertilisers in southern Australian soils. Aust J Soil Res 43 (2):203-212. [CrossRef]
- McBeath TM, McLaughlin MJ, Armstrong RD, Bell M, Bolland MDA, Conyers MK, Holloway RE, Mason SD (2007) Predicting the response of wheat (Triticum aestivum L.) to liquid and granular phosphorus fertilisers in Australian soils. Aust J Soil Res 45 (6):448-458. [CrossRef]
- McBeath TM, Smernik RJ, Lombi E, McLaughlin MJ (2006) Hydrolysis of Pyrophosphate in a Highly Calcareous Soil. Soil Sci Soc Am J 70 (3):856-862. [CrossRef]
- McGonigle TP, Hutton M, Greenley A, Karamanos R (2011) Role of mycorrhiza in a wheat–flax versus canola–flax rotation: A case study. Commun Soil Sci Plant Anal 42 (17):2134-2142. [CrossRef]
- McGonigle TP, Miller MH, Young D (1999) Mycorrhizae, crop growth, and crop phosphorus nutrition in maize-soybean rotations given various tillage treatments. Plant Soil 210 (1):33-42. [CrossRef]
- McGrath JM, Binford GD (2012) Corn response to starter fertilizer with and without AVAIL. Crop Manag 11 (1):0-0.
- McKnight MM, Qu Z, Copeland JK, Guttman DS, Walker VK (2020) A practical assessment of nano-phosphate on soybean (Glycine max) growth and microbiome establishment. Scientific Reports 10 (1):9151. [CrossRef]
- McLaughlin MJ, McBeath TM, Smernik R, Stacey SP, Ajiboye B, Guppy C (2011) The chemical nature of P accumulation in agricultural soils—implications for fertiliser management and design: an Australian perspective. Plant Soil 349 (1-2):69-87. [CrossRef]
- Mikhak A, Sohrabi A, Kassaee MZ, Feizian M (2017) Synthetic nanozeolite/nanohydroxyapatite as a phosphorus fertilizer for German chamomile (Matricariachamomilla L.). Ind Crops Prod 95:444-452. [CrossRef]
- Mikkelsen R (2005) Humic materials for agriculture. Better crops 89 (3):6-10.
- Miller M, Mamaril C, Blair G (1970) Ammonium effects on phosphorus absorption through pH changes and phosphorus precipitation at the soil-root interface. Agron J 62 (4):524-527. [CrossRef]
- Miller MH (2000) Arbuscular mycorrhizae and the phosphorus nutrition of maize: A review of Guelph studies. Can J Plant Sci 80 (1):47-52. [CrossRef]
- Miller MH, McGonigle TP, Addy HD (1995) Functional ecology of vesicular arbuscular mycorrhizas as influenced by phosphate fertilization and tillage in an agricultural ecosystem. CRC Cr Rev Biotechn 15:241-255. [CrossRef]
- Miller MH, Ohlrogge AJ (1958) Principles of nutrient uptake from fertilizer bands. I. Effect of placement of nitrogen fertilizer on the uptake of band-placed phosphorus at different soil phosphorus levels. Agron J 50:95-97. [CrossRef]
- Mills HA, Jones JB, Jr. (1996) Plant Analysis Handbook II. MicroMacro Publishing, Inc., Jefferson City, MO.
- Mitchell J (1946) The effect of phosphatic fertilizers on summer-fallow wheat crops in certain areas of Saskatchewan. Scientific Agriculture 26 (11):566-577.
- Mitchell J, Dehm J, Dion H (1952) The effect of small additions of elemental sulphur on the availability of phosphate fertilizers. Scientific Agriculture 32 (6):311-316.
- Mohamed GES, Marshall C (1979) The pattern of distribution of phosphorus and dry matter with time in spring wheat. Ann Bot 44 (6):721-730. [CrossRef]
- Mohr R, Irvine B, Grant C, Holzapfel C, Hogg T, Malhi S, Kirk A (2013) Response of canola to the application of phosphorus fertilizer and Penicillium bilaii (JumpStart®). Saskatchewan Canola Development Commission, Saskatoon, SK.
- Molin P, Rampim L, Fávero F, Do M, Lana C, Sarto M, Rosset J, Mattei D, Diel P, Molin R (2015) Response to simple superphosphate and top-phos fertilizer on wheat in an oxisoil. Tropical and Subtropical Agroecosystems 18:121-132.
- Monreal MA, Grant CA, Irvine RB, Mohr RM, McLaren DL, Khakbazan M (2011) Crop management effect on arbuscular mycorrhizae and root growth of flax. Can J Plant Sci 91 (2):315-324. [CrossRef]
- Montalvo D, McLaughlin MJ, Degryse F (2015) Efficacy of hydroxyapatite nanoparticles as phosphorus fertilizer in Andisols and Oxisols. Soil Sci Soc Am J 79 (2):551-558. [CrossRef]
- Morel C, Plenchette C (1994) Is the isotopically exchangeable phosphate of a loamy soil the plant-available P? Plant Soil 158 (2):287-297. [CrossRef]
- Morel C, Tunney H, Plenet D, Pellerin S (2000) Transfer of phosphate ions between soil and solution: Perspectives in soil testing. J Environ Qual 29 (1):50-59. [CrossRef]
- Nadeem M, Mollier A, Morel C, Vives A, Prud’homme L, Pellerin S (2011) Relative contribution of seed phosphorus reserves and exogenous phosphorus uptake to maize (Zea mays L.) nutrition during early growth stages. Plant Soil 346 (1):231-244. [CrossRef]
- Nelson NO, Janke RR (2007) Phosphorus sources and management in organic production systems. HortTechnology 17 (4):442-454.
- Nkebiwe PM, Stevens Lekfeldt JD, Symanczik S, Thonar C, Mäder P, Bar-Tal A, Halpern M, Biró B, Bradáčová K, Caniullan PC, Choudhary KK, Cozzolino V, Di Stasio E, Dobczinski S, Geistlinger J, Lüthi A, Gómez-Muñoz B, Kandeler E, Kolberg F, Kotroczó Z, Kulhánek M, Mercl F, Tamir G, Moradtalab N, Piccolo A, Maggio A, Nassal D, Szalai MZ, Juhos K, Fora CG, Florea AB, Poșta G, Lauer KF, Tóth B, Tlustoš P, Mpanga IK, Weber NF, Weinmann M, Yermiyahu U, Magid J, Müller T, Neumann G, Ludewig U, de Neergaard A (2024) Effectiveness of bio-effectors on maize, wheat and tomato performance and phosphorus acquisition from greenhouse to field scales in Europe and Israel: a meta-analysis. Frontiers in Plant Science 15. [CrossRef]
- Noor S, Yaseen M, Naveed M, Ahmad R (2017) Use of controlled release phosphatic fertilizer to improve growth, yield and phosphorus use efficiency of wheat crop. Pak J Agri Sci 54 (4):541-547.
- Nunes APP, Santos CF, Guelfi D (2022) Interfaces between biodegradable organic matrices coating and MAP fertilizer for improve use efficiency. Science of The Total Environment 804:149896. [CrossRef]
- Nyborg M, Hennig AMF (1969) Field experiments with different placements of fertilizers for barley, flax and rapeseed. Can J Soil Sci 49:79-88. [CrossRef]
- Nyborg M, Solberg ED, Pauly DG (1998) Controlled release of phosphorus fertilizers by small, frequent additions in water solution. Can J Soil Sci 78:317-320. [CrossRef]
- Olatuyi S, Akinremi O, Flaten D, Crow G (2009a) Accompanying cations and anions affect the diffusive transport of phosphate in a model calcareous soil system. Can J Soil Sci 89 (2):179-188. [CrossRef]
- Olatuyi S, Akinremi O, Flaten D, Crow G (2009b) Solubility and transport of phosphate and the accompanying ions as influenced by sulphate salts in a model calcareous soil system. Can J Soil Sci 89 (5):589-601. [CrossRef]
- Ortas I (2012) The effect of mycorrhizal fungal inoculation on plant yield, nutrient uptake and inoculation effectiveness under long-term field conditions. Field Crop Res 125:35-48. [CrossRef]
- Owino-Gerroh C, Gascho GJ (2005) Effect of silicon on low pH soil phosphorus sorption and on uptake and growth of maize Commun Soil Sci Plant Anal 35:2369–2378. [CrossRef]
- Pauly DG, Malhi SS, Nyborg M (2002) Controlled-release P fertilizer concept evaluation using growth and P uptake of barley from three soils in greenhouse. Can J Soil Sci 82 (2):201-210. [CrossRef]
- Pellegrino E, Öpik M, Bonari E, Ercoli L (2015) Responses of wheat to arbuscular mycorrhizal fungi: a meta-analysis of field studies from 1975 to 2013. Soil Biol Biochem 84:210-217. [CrossRef]
- Penn CJ, Camberato JJ (2019) A Critical Review on Soil Chemical Processes that Control How Soil pH Affects Phosphorus Availability to Plants. Agriculture 9 (6):120. [CrossRef]
- Pierre W, Parker F (1927) Soil phosphorus studies: II. The concentration of organic and inorganic phosphorus in the soil solution and soil extracts and the availibility of the organic phosphorus to plants. Soil Sci 24 (2):119-128.
- Pierzynski GM, McDowell RW (2005) Chemistry, cycling, and potential movement of inorganic phosphorus in soils. Phosphorus: agriculture and the environment (phosphorusagric):53-86. [CrossRef]
- Pierzynski J, Hettiarachchi G (2018) Reactions of phosphorus fertilizers with and without a fertilizer enhancer in three acidic soils with high phosphorus-fixing capacity Soil Sci Soc Am J. [CrossRef]
- Plaxton W, Lambers H (2015) Phosphorus: back to the roots. Annual plant reviews 48:3-15.
- Plenchette C, Fortin J, Furlan V (1983) Growth responses of several plant species to mycorrhizae in a soil of moderate P-fertility: I. Mycorrhizal dependency under field conditions. Plant Soil 70:199-209. [CrossRef]
- Pons C, Müller C (2022) Impacts of drought atress and mycorrhizal inoculation on the performance of two spring wheat cultivars. Plants 11 (17):2187. [CrossRef]
- Powers KW, Brown SC, Krishna VB, Wasdo SC, Moudgil BM, Roberts SM (2006) Research strategies for safety evaluation of nanomaterials. Part VI. characterization of nanoscale particles for toxicological evaluation. Toxicological Sciences 90 (2):296-303. [CrossRef]
- Racz G, Savant N (1972) Pyrophosphate hydrolysis in soil as influenced by flooding and fixation Soil Sci Soc Am J 36 (4):678-682.
- Racz G, Soper R (1970) Solubility of phosphorus added to four Manitoba soils with different calcium and magnesium contents. Plant and Soil 32 (1):303-315. [CrossRef]
- Racz GJ, Soper R (1967) Reaction products of orthophosphates in soils containing varying amounts of calcium and magnesium. Can J Soil Sci 47 (3):223-230.
- Raven PH, Evert RF, Eichhorn SE (2005) Biology of Plants. Seventh edn. W. H. Freeman and Company, New York.
- Raymond NS, Gómez-Muñoz B, van der Bom FJT, Nybroe O, Jensen LS, Müller-Stöver DS, Oberson A, Richardson AE (2021) Phosphate-solubilising microorganisms for improved crop productivity: a critical assessment. New Phytol 229 (3):1268-1277. [CrossRef]
- Rech I, Withers P, Jones D, Pavinato P (2018) Solubility, diffusion and crop uptake of phosphorus in three different struvites. Sustainability 11:134. [CrossRef]
- Richardson AE (2001) Prospects for using soil microorganisms to improve the acquisition of phosphorus by plants. Funct Plant Biol 28 (9):897-906. [CrossRef]
- Richardson AE, Lynch JP, Ryan PR, Delhaize E, Smith FA, Smith SE, Harvey PR, Ryan MH, Veneklaas EJ, Lambers H, Oberson A, Culvenor RA, Simpson RJ (2011) Plant and microbial strategies to improve the phosphorus efficiency of agriculture. Plant Soil 349 (1):121-156. [CrossRef]
- Richardson AE, Simpson RJ (2011) Soil microorganisms mediating phosphorus availability update on microbial phosphorus. Plant Physiol 156 (3):989-996. [CrossRef]
- Riley D, Barber SA (1971) Effect of Ammonium and Nitrate Fertilization on Phosphorus Uptake as Related to Root-Induced pH Changes at the Root-Soil Interface. Soil Sci Soc Am J 35 (2):301-306. [CrossRef]
- Roberts TL, Johnston AE (2015) Phosphorus use efficiency and management in agriculture. Resources, Conservation and Recycling 105:275-281. [CrossRef]
- Rose MT, Patti AF, Little KR, Brown AL, Jackson WR, Cavagnaro TR (2014) Chapter Two—A meta-analysis and review of plant-growth response to humic substances: practical implications for agriculture. In: Sparks DL (ed) Advances in Agronomy, vol 124. Academic Press, pp 37-89. [CrossRef]
- Ryan MH, Angus JF (2003) Arbuscular mycorrhizae in wheat and field pea crops on a low P soil: Increased Zn-uptake but no increase in P-uptake or yield. Plant Soil 250 (2):225-239. [CrossRef]
- Ryan MH, Graham JH (2002) Is there a role for arbuscular mycorrhizal fungi in production agriculture? Plant Soil 244 (1-2):263-271. [CrossRef]
- Salomon MJ, Demarmels R, Watts-Williams SJ, McLaughlin MJ, Kafle A, Ketelsen C, Soupir A, Bücking H, Cavagnaro TR, van der Heijden MGA (2022) Global evaluation of commercial arbuscular mycorrhizal inoculants under greenhouse and field conditions. Appl Soil Ecol 169:104225. [CrossRef]
- Sample EC, Soper RJ, Racz GJ (1980) Reaction of phosphate fertilizers in soils. In: Khasawneh FE, Sample EC, Kamprath EJ (eds) The Role of Phosphorus in Agriculture. ASA, CSSA, and SSSA, Madision, , WI, pp 262-310. [CrossRef]
- Sattar B, Ahmad S, Daur I, Hussain MB, Ali M, Ul Haq T, Arif M, Bakhtawer M (2021) Bioactive-sulfur coated diammonium phosphate improves nitrogen and phosphorus use efficiency and maize (Zea mays L.) yield. Journal of Environmental and Agricultural Science, 23 (3&4):23-29.
- Schachtman DP, Reid RJ, Ayling SM (1998) Phosphorus uptake by plants: from soil to cell. Plant Physiol 116 (2):447-453. [CrossRef]
- Schaller J, Wu B, Amelung W, Hu Z, Stein M, Lehndorff E, Obst M (2022) Silicon as a potential limiting factor for phosphorus availability in paddy soils. Scientific Reports 12 (1):16329. [CrossRef]
- Schlechte D, Beckie H, Gleddie SC (1996) Response of alfalfa in the establishment year to inoculation with the phosphate-solubilizing fungus Penicillium bilaii (Provide). Paper presented at the Saskatchewan Soils alnd Crops Conference, Saskatoon, SK, .
- Schneider A, Morel C (2000) Relationship between the isotopically exchangeable and resin-extractable phosphate of deficient to heavily fertilized soil. Eur J Soil Sci 51 (4):709-715. [CrossRef]
- Schnitzer MI (1978) Chapter 1 Humic substances: chemistry and reactions. Developments in soil science 8:1-64.
- Schoenau JJ, Qian P, King T (2007) Strategies for improving the efficiency and crop safety of starter fertilizer phosphorus and potassium University of Saskatchewan, Saskatoon.
- Schroeder JI, Delhaize E, Frommer WB, Guerinot ML, Harrison MJ, Herrera-Estrella L, Horie T, Kochian LV, Munns R, Nishizawa NK (2013) Using membrane transporters to improve crops for sustainable food production. Nature 497 (7447):60. [CrossRef]
- Sharif M, Khattak RA, Sarir MS (2002) Effect of different levels of lignitic coal derived humic acid on growth of maize plants Commun Soil Sci Plant Anal 33 (19-20):3567-3580. [CrossRef]
- Sharma M, Pang J, Mickan BS, Ryan MH, Jenkins SN, Siddique KHM (2024) Wastewater-derived struvite has the potential to substitute for soluble phosphorus fertiliser for growth of chickpea and wheat. Journal of Soil Science and Plant Nutrition 24 (2):3011-3025. [CrossRef]
- Sharma SB, Sayyed RZ, Trivedi MH, Gobi TA (2013) Phosphate solubilizing microbes: sustainable approach for managing phosphorus deficiency in agricultural soils. SpringerPlus 2:1-14. [CrossRef]
- Shaviv A, Mikkelsen RL (1993) Controlled-release fertilizers to increase efficiency of nutrient use and minimize environmental degradation—A review. Fert Res 35 (1-2):1-12. [CrossRef]
- Shaviv A, Raban S, Zaidel E (2003) Modeling controlled nutrient release from polymer coated fertilizers: diffusion release from single granules. Environ Sci Technol 37 (10):2251-2256. [CrossRef]
- Singha Roy A, de Beer M, Pillai S, Sinha Ray S (2023) Application of layered double hydroxides as a slow-release phosphate source: a comparison of hydroponic and soil systems ACS Omega 8. [CrossRef]
- Smyth TJ, Sanchez PA (1980) Effects of lime, silicate, and phosphorus applications to an Oxisol on phosphorus sorption and ion retention Soil Sci Soc Am J 44 (3):500-505. [CrossRef]
- Sparvoli F, Cominelli E (2015) Seed biofortification and phytic acid reduction: a conflict of interest for the plant? Plants (Basel) 4 (4):728-755. [CrossRef]
- Strambeanu N, Demetrovici L, Dragos D, Lungu M (2015) Nanoparticles: definition, classification and general physical properties.3-8. [CrossRef]
- Sultenfuss J, Doyle W (1999) Functions of phosphorus in plants. Better Crops 83 (1):6-7.
- Syers J, Johnston A, Curtin D (2008) Efficiency of soil and fertilizer phosphorus use., FAO Fertilizer and Plant Nutrition Bulletin No. 18.(FAO: Rome).
- Symanczik S, Lipp C, Mäder P, Thonar C, Kundel D (2023) Limited effectiveness of selected bioeffectors combined with recycling phosphorus fertilizers for maize cultivation under Swiss farming conditions. Frontiers in Plant Science 14. [CrossRef]
- Talboys PJ, Heppell J, Roose T, Healey JR, Jones DL, Withers PJ (2016) Struvite: a slow-release fertiliser for sustainable phosphorus management? Plant Soil 401 (1-2):109-123. [CrossRef]
- Taskin M, Sahin O, Taskin H, Atakol O, Inal A, Gunes A (2018) Effect of synthetic nano-hydroxyapatite as an alternative phosphorus source on growth and phosphorus nutrition of lettuce ( Lactuca sativa L.) plant. J Plant Nutr 41:1-7. [CrossRef]
- Thiessen Martens JR, Entz MH, Schneider KD, Zvomuya F, Wilson HF (2022) Response of organic grain and forage crops to struvite application in an alkaline soil. Agron J 114 (1):795-810. [CrossRef]
- Usman M, Farooq M, Wakeel A, Nawaz A, Cheema SA, Rehman Hu, Ashraf I, Sanaullah M (2020) Nanotechnology in agriculture: Current status, challenges and future opportunities. Sci Total Environ 721:137778. [CrossRef]
- Vance CP, Uhde-Stone C, Allan DL (2003) Phosphorus acquisition and use: critical adaptations by plants for securing a nonrenewable resource. New Phytol 157 (3):423-447. [CrossRef]
- Vessey JK, Heisinger KG (2001) Effect of Penicillium bilaii inoculation and phosphorus fertilisation on root and shoot parameters of field-grown pea. Can J Plant Sci 81 (3):361-366. [CrossRef]
- Wakelin S, Vadakattu G, Harvey P, Ryder M (2007a) The effect of Penicillium fungi on plant growth and phosphorus mobilization in neutral to alkaline soils from southern Australia. Can J Microbiol 53:106-115. [CrossRef]
- Wakelin SA, Gupta VVSR, Harvey PR, Ryder MH (2007b) The effect of Penicillium fungi on plant growth and phosphorus mobilization in neutral to alkaline soils from southern Australia. Can J Microbiol 53 (1):106-115. [CrossRef]
- Wakelin SA, Warren RA, Harvey PR, Ryder MH (2004) Phosphate solubilization by Penicillium spp. closely associated with wheat roots. Biol Fertil Soils 40 (1):36-43. [CrossRef]
- Walley F, Germida J (2015) Asssessment of arbuscular mycorrhizal inoculants for flax production. Saskatchewan Agricultural Development Fund, Saskatoon, SK.
- Ward NC (2010) Impact of Avail® and Jumpstart® on yield and phosphorus response of coron and winter wheat in Kansas. . Kansas State University, Manhattan, KS.
- Weeks JJ, Hettiarachchi GM (2018) Can humic substances alter fertilizer reaction pathways in acid soils. Fluid J 26:5-7.
- Wu K, Du C, Ma F, Shen Y, Liang D, Zhou J (2019a) Degradation of metal-organic framework materials as controlled-release fertilizers in crop fields. Polymers (Basel) 11 (6). [CrossRef]
- Wu K, Du C, Ma F, Shen Y, Zhou J (2019b) Optimization of metal–organic (citric acid) frameworks for controlled release of nutrients. RSC Advances 9 (55):32270-32277. [CrossRef]
- Wu K, Xu X, Ma F, Du C (2022) Fe-based metal–organic frameworks for the controlled release of fertilizer nutrients. ACS Omega 7 (40):35970-35980. [CrossRef]
- Xavier LJC, Germida JJ (1997) Growth response of lentil and wheat to Glomus clarum NT4 over a range of P levels in a Saskatchewan soil containing indigenous AM fungi. Mycorrhiza 7 (1):3-8. [CrossRef]
- Xiong L (2019) Tailoring hydroxyapatite (HA) nanoparticles as a phosphorus (P) fertiliser in soils. University of Queensland, Queensland, Australia.
- Yang SY, Huang TK, Kuo HF, Chiou TJ (2017) Role of vacuoles in phosphorus storage and remobilization. J Exp Bot 68 (12):3045-3055. [CrossRef]
- Yaseen M, Aziz MZ, Manzoor A, Naveed M, Hamid Y, Noor S, Khalid MA (2017) Promoting growth, yield and phosphorus-use efficiency of crops in maize-wheat cropping system by using polymer-coated diammonium phosphate Commun Soil Sci Plant Anal 48 (6):646-655. [CrossRef]
- Zavaschi E, Ferraz-Almeida R, Fária LA, Otto R, Vitti AC, Vitti GC (2020) Application of superphosphate complexed with humic acid in an area of sugarcane. Revista Ciência Agronômica 51 (1):e20186463. [CrossRef]
- Zhang M, Nyborg M, Malhi SS, McKenzie RH, Solberg E (2000) Phosphorus release from coated monoammonium phosphate: Effect of coating thickness, temperature, elution medium, soil moisture and placement method. Can J Soil Sci 80 (1):127-134. [CrossRef]
- Zhang S, Lehmann A, Zheng W, You Z, Rillig MC (2019) Arbuscular mycorrhizal fungi increase grain yields: a meta-analysis. New Phytol 222 (1):543-555. [CrossRef]
- Zhou R, Zhou J, Jia L, Wei J, Yan J, Ji J, Wei Z (2023) Polyphosphate hydrolysis, sorption, and conversion in two different soils. Eur J Soil Sci 74 (1):e13341. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




