1. Introduction
Folate receptor alpha (FRα) is a glycosylphosphatidylinositol-anchored cell surface protein that functions as a high-affinity, low-throughput folate transporter, facilitating folate uptake via receptor-mediated endocytosis to support one-carbon metabolism, DNA synthesis, and cellular proliferation [
1]. FRα is frequently overexpressed in a range of epithelial malignancies, including approximately 35–70% of breast cancers, 15–75% of lung cancers, 20–50% of endometrial cancers, and 75–90% of ovarian cancers [
2,
3,
4]. Among ovarian cancers, high-grade serous carcinoma demonstrates the highest frequency of FRα overexpression, reported in roughly 60–85% of cases [
2,
3,
4,
5,
6]. These estimates vary due to differences in detection methods, antibody clones, sample sizes, and scoring criteria across studies [
2,
3,
4,
5,
6].
Due to its selective expression in malignant tissues and limited distribution in normal adult tissues, FRα has emerged as a promising therapeutic target, with several FRα-directed agents currently under clinical investigation [
7,
8,
9,
10,
11,
12,
13].
Immunohistochemistry (IHC) is the standard method for evaluating FRα expression in clinical and research settings, guiding eligibility for FRα-targeted therapies [
7,
8,
9,
10,
11,
12,
13]. However, IHC scoring of FRα remains inherently semi-quantitative and susceptible to interobserver variability [
14,
15,
16,
17]. Most commonly adopted IHC scoring systems rely on the assessment of both the intensity and the extent of tumor cell membrane staining [
14,
15,
16,
17]. The scoring criteria used in clinical trials, and increasingly referenced in routine practice, classify tumors as “FRα-high” when ≥75% of viable tumor cells demonstrate at least moderate (2+) membranous staining [
14,
15,
16,
17]. While this threshold has shown clinical utility, particularly in patient selection for FRα-targeted treatments, its reproducibility across different observers and institutions remains insufficiently characterized [
14,
15,
16,
17].
In routine diagnostic practice, interpretation of FRα IHC can be influenced by a range of technical and subjective factors. These include variability in staining protocols, inconsistencies in the quality or presence of internal control tissue (typically represented by benign tubal epithelium), and differences in individual pathologist experience or interpretive thresholds [
14,
15,
16,
17]. Moreover, the distinction between membranous and cytoplasmic staining, and the identification of partial or heterogeneous staining patterns, may further complicate assessment, particularly in borderline or equivocal cases [
14,
15,
16,
17].
Given the clinical implications of FRα status for patient stratification and therapeutic decision-making, it is essential to establish the reliability and reproducibility of FRα IHC evaluation in real-world pathology settings. A robust understanding of interobserver agreement can help inform the standardization of scoring guidelines and support the integration of FRα testing into routine diagnostic workflows.
This study aims to evaluate interobserver variability in the assessment of FRα IHC in ovarian cancer among a group of 12 pathologists. Using a cohort of 37 preselected ovarian cancer cases with accompanying internal control tissue, we focus on three key parameters: (1) adequacy of internal control tissue, (2) FRα positivity versus negativity, and (3) estimation of the percentage of tumor cells exhibiting FRα staining. Through this analysis, we seek to quantify agreement across observers and identify areas where interpretive consistency may be improved.
2. Results
2.1. Internal Control Adequacy
The reference panel classified 36 out of 37 cases (97.3%) as having adequate internal controls, based on the presence of fallopian tube epithelium predominantly displaying moderate (2+) circumferential membranous staining, with negative stromal staining. One case was defined inadequate due to the absence of recognizable tubal epithelium or the presence of non-specific/background staining.
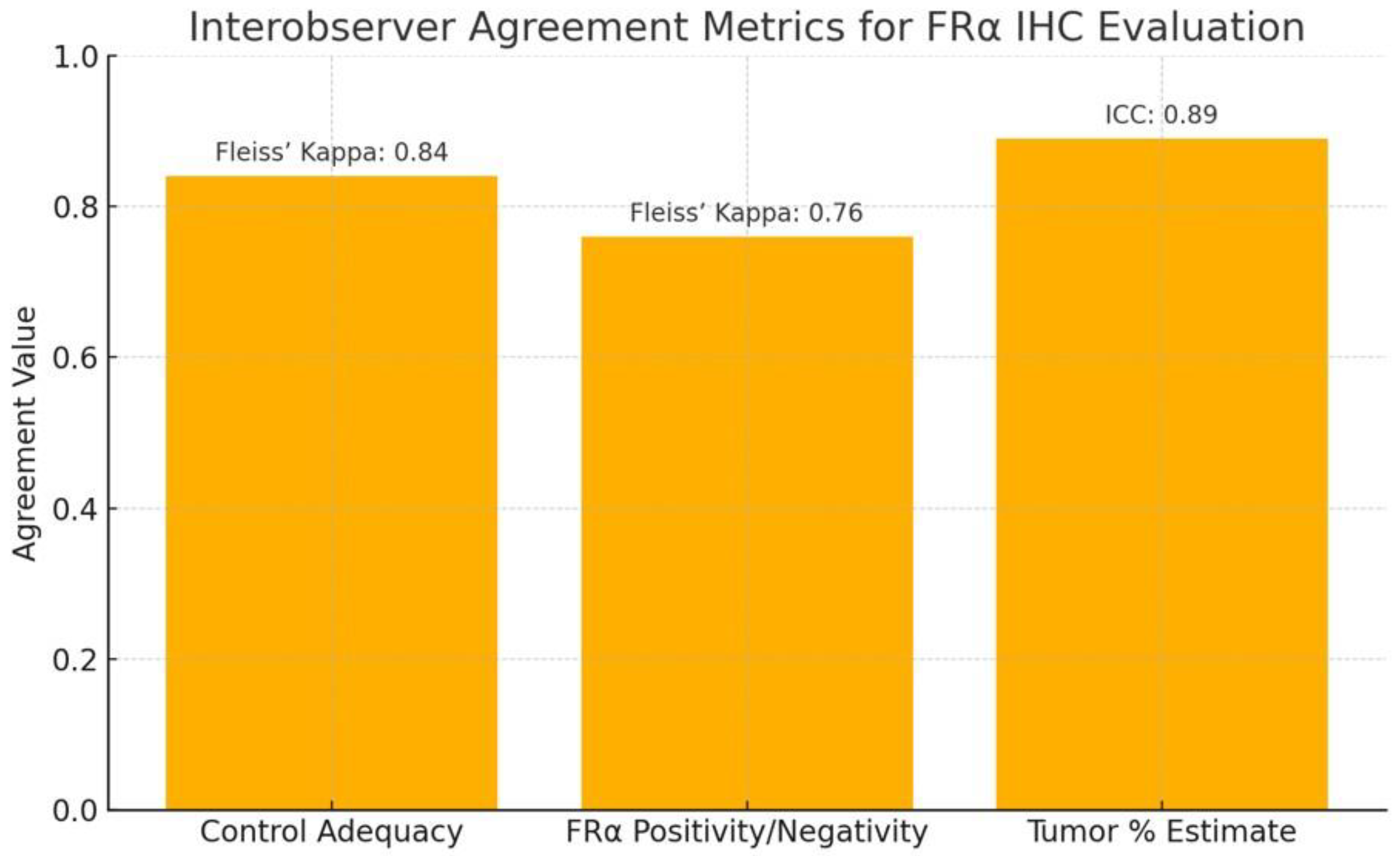
Observers demonstrated consistently high agreement in assessing internal control adequacy. In 35 of 37 cases (94.6%), all 12 observers unanimously classified the internal control as adequate. One case (2.7%) was unanimously judged inadequate due to the absence of recognizable tubal epithelium or the presence of non-specific/background staining. Another case (2.7%) received discordant evaluations, with one or more observers considering the control inadequate due to weak, incomplete, or absent staining; however, the reference panel had classified the control tissue in this case as adequate. The Fleiss’ kappa statistic for interobserver agreement on internal control adequacy was 0.84, indicating almost perfect agreement according to the Landis and Koch scale.
A graphical summary of agreement metrics across all evaluated parameters is shown in
Figure 1.
2.2. FRα Staining Intensity in Tumor Cells
Based on the reference panel assessment, 27 out of 37 cases (73%) demonstrated moderate (2+) and/or strong (3+) membranous FRα staining and were classified as positive for staining intensity. The remaining 10 cases (27%) were categorized as negative, showing absent (0) or weak (1+) staining.
Full agreement among all 12 observers regarding staining intensity (positive vs negative) was achieved in 31 cases (83.8%). The remaining 6 cases (16.2%) showed discordance among observers. Among these discordant cases, 4 were classified as positive by the reference panel but showed variability among observers, typically due to heterogeneous staining patterns or staining near the positivity threshold. In the other 2 cases, which were negative according to the reference panel, weak focal staining led some observers to incorrectly classify them as positive. The Fleiss’ kappa value for staining intensity assessment was 0.76, indicating substantial interobserver agreement.
2.3. Percentage of FRα-Positive Tumor Cells
According to the reference panel evaluation, the distribution of FRα expression across the 37 ovarian cancer cases encompassed the full spectrum of staining extent. Five cases (13.5%) exhibited absent or minimal staining, falling within the 0–5% range, and were classified as negative. Three cases (8.1%) showed weak and focal staining involving a limited subset of tumor cells, corresponding to the 6–15% range. Four cases (10.8%) demonstrated low-level but definite membranous staining and were placed in the 16–30% category, while another 3 cases (8.1%) exhibited more heterogeneous patterns and were assigned to the 31–45% range. Intermediate FRα expression, involving 46–64% of tumor cells, was observed in four cases (10.8%), while more extensive staining, ranging from 65% to 85% (borderline cases), was identified in 9 cases (24.3%). Finally, 9 additional cases (24.3%) displayed near-complete or complete membranous positivity, with 86–100% of tumor cells stained for FRα.
Quantitative estimation of the percentage of FRα-positive tumor cells showed excellent consistency among the 12 observers. The calculated intraclass correlation coefficient (ICC) for this continuous variable was 0.89, with a 95% confidence interval of 0.83 to 0.94, indicating a high level of agreement. The highest concordance was observed in tumors with either extensive and uniform staining (86–100% range) or minimal expression (0–5% range), where observer estimates typically clustered within ± 5–10% of the group median. Similarly, cases falling within the 65–85% interval (borderline cases) also showed strong agreement, although slightly greater variability was noted, likely due to more heterogeneous staining in some tumors. The greatest discrepancies were particularly evident among tumors exhibiting intermediate levels of FRα expression, with differences of up to 20 percentage points between observers. In fact, tumors within the 31–45% and 46–64% ranges exhibited greater interobserver variation, reflecting the interpretive challenges posed by patchy or focal staining patterns. Despite this variability, the overall agreement remained high, underscoring the reproducibility of FRα percentage estimation, particularly at the extremes of expression. An overview of interobserver agreement metrics across all evaluated parameters is provided in
Table 1.
2.4. Overall FRα Positivity and Analysis of Borderline and Discordant Cases
Based on the reference panel evaluation, tumors were considered positive for FRα expression when ≥75% of tumor cells demonstrated moderate (2+) or strong (3+) membranous staining, including circumferential, apical, and dot-like patterns [
18,
19]. Borderline cases were defined as tumors exhibiting 65–85% of neoplastic cells with 2+/3+ membranous staining, encompassing both circumferential, apical, and dot-like patterns [
18,
19]. Applying these criteria, the reference panel identified 9 borderline cases among the 37 analyzed tumors. These cases showed strong agreement among the observers although slightly greater variability was noted, likely due to more heterogeneous staining in some tumors.
In addition to borderline cases, discordant cases were also recognized among tumors exhibiting FRα-positive tumor cell percentages within the 31–45% and 46–64% intervals. These tumors demonstrated significant interobserver variability in the estimation of FRα-positive tumor cell percentages, with differences reaching up to 20 percentage points in some instances. In total, 4 discordant cases were identified based on their marked interpretive variability across observers. Representative examples of discordant cases are shown in
Figure 2.
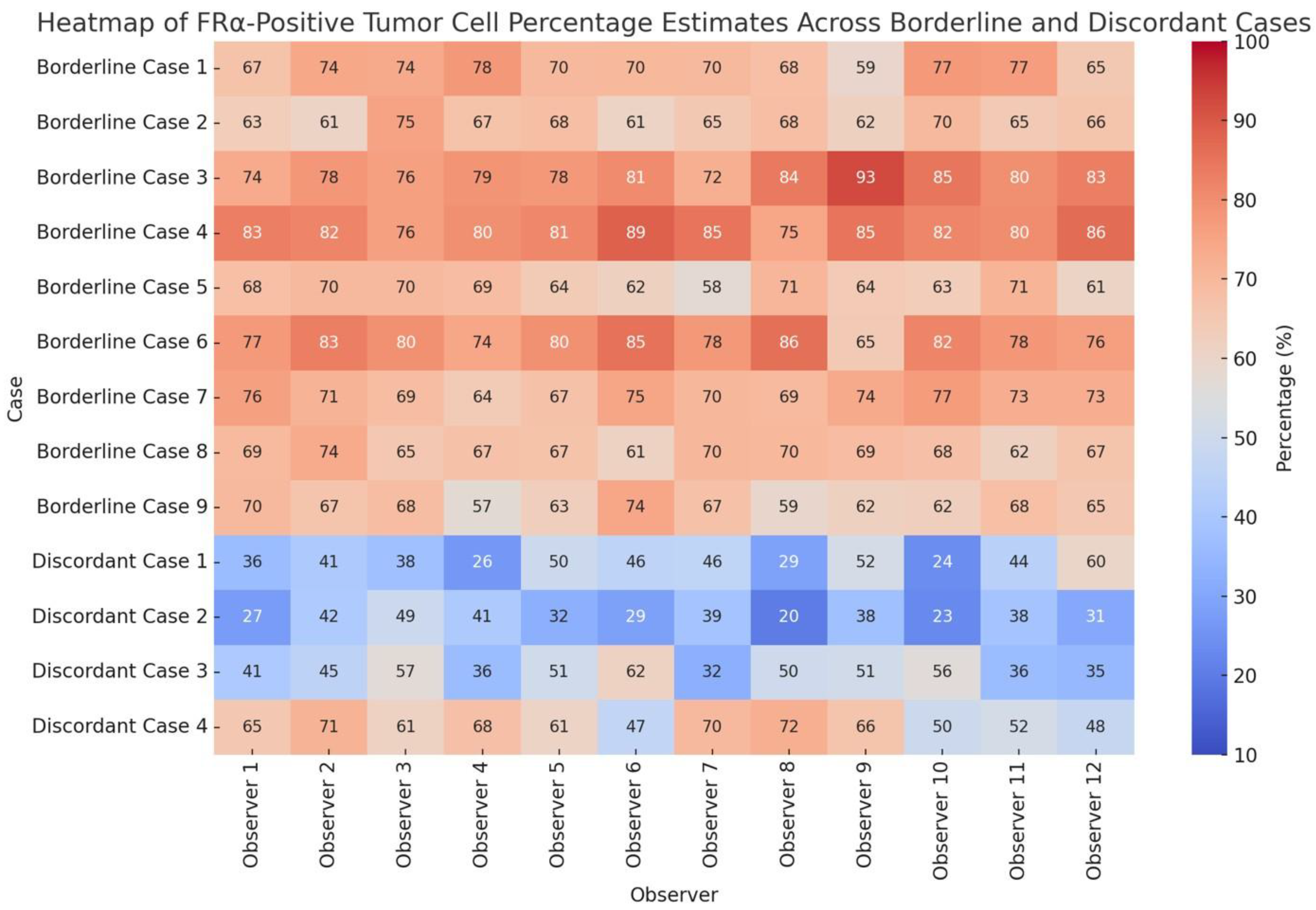
The group of borderline and discordant cases represented the principal source of interobserver disagreement, mainly affecting both intensity scoring and percentage estimation. Variability among observers for these cases was notably greater compared to tumors with either minimal (<5%) or extensive (>85%) staining. Distribution of observer estimates for all borderline and discordant cases is illustrated in
Figure 3, while
Figure 4 provides a detailed heatmap of individual observer ratings.
Despite these interpretive challenges, the majority of observers applied internal evaluation criteria consistently across the cohort. High-expression tumors, particularly those with homogeneous strong staining in more than 90% of tumor cells, and low-expression tumors with weak or absent staining involving less than 25% of tumor cells, showed minimal variability among observers.
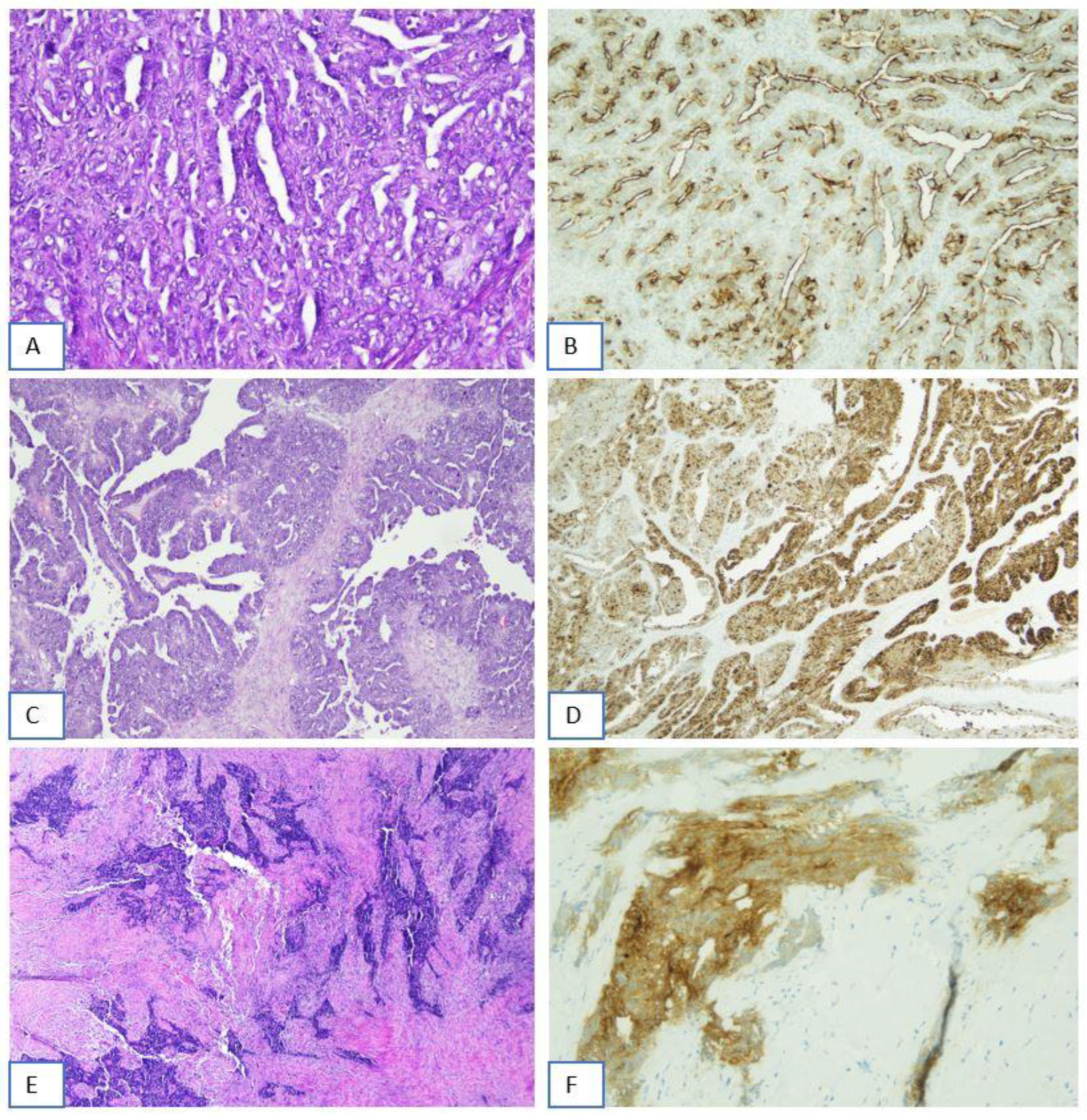
Figure 2.
Immunohistochemical determination of folate receptor 1 status: examples of discordant cases. (A,B) Ovarian High Grade Serous Carcinoma showing FOLR1 membrane positivity (score 2+/3+) in 70% of tumor cells. This case has been considered borderline (65-85% of cells with a score of 2+ or 3+ positivity) (A: H&E, 20x; B: FOLR1-2.1 antibody on Benckmark Ultra platform, LSAB-HRP, 10x). (C,D) Another example falling within the borderline cathegory: ovarian High Grade Serous Carcinoma showing FOLR1 membrane positivity (score 2+/3+) in 70% of tumor cells (C: H&E, 10x; D: Ventana FOLR1 RxDx Assay - FOLR1-2.1 antibody on (E,F) Peritoneal metastasis of ovarian High Grade Serous Carcinoma showing FOLR1 membrane positivity not adequately evaluable, due to the diffuse crushing or heat generated (cautery) artifacts (E: H&E, 4x; F:Ventana FOLR1 RxDx Assay - FOLR1-2.1 antibody on Benckmark Ultra platform, LSAB-HRP, 20x).
Figure 2.
Immunohistochemical determination of folate receptor 1 status: examples of discordant cases. (A,B) Ovarian High Grade Serous Carcinoma showing FOLR1 membrane positivity (score 2+/3+) in 70% of tumor cells. This case has been considered borderline (65-85% of cells with a score of 2+ or 3+ positivity) (A: H&E, 20x; B: FOLR1-2.1 antibody on Benckmark Ultra platform, LSAB-HRP, 10x). (C,D) Another example falling within the borderline cathegory: ovarian High Grade Serous Carcinoma showing FOLR1 membrane positivity (score 2+/3+) in 70% of tumor cells (C: H&E, 10x; D: Ventana FOLR1 RxDx Assay - FOLR1-2.1 antibody on (E,F) Peritoneal metastasis of ovarian High Grade Serous Carcinoma showing FOLR1 membrane positivity not adequately evaluable, due to the diffuse crushing or heat generated (cautery) artifacts (E: H&E, 4x; F:Ventana FOLR1 RxDx Assay - FOLR1-2.1 antibody on Benckmark Ultra platform, LSAB-HRP, 20x).
Figure 3.
Immunohistochemical determination of folate receptor 1 status: examples of discordant cases. Distribution of estimated FRα-positive tumor cell percentages across borderline and discordant cases. Borderline cases were defined as tumors with 65–85% of neoplastic cells exhibiting 2+/3+ membranous staining, while discordant cases included tumors within the 31–45% and 46–64% percentage ranges that showed significant interobserver variability. Each boxplot represents the range, median, and variability of observer estimates for a given case.
Figure 3.
Immunohistochemical determination of folate receptor 1 status: examples of discordant cases. Distribution of estimated FRα-positive tumor cell percentages across borderline and discordant cases. Borderline cases were defined as tumors with 65–85% of neoplastic cells exhibiting 2+/3+ membranous staining, while discordant cases included tumors within the 31–45% and 46–64% percentage ranges that showed significant interobserver variability. Each boxplot represents the range, median, and variability of observer estimates for a given case.
Figure 4.
Heatmap of individual observer estimates of FRα-positive tumor cell percentages across borderline and discordant cases. Borderline cases were defined based on a 65–85% staining threshold with 2+/3+ intensity, and discordant cases were selected among tumors showing marked variability in the 31–45% and 46–64% percentage intervals. Values highlight interobserver variability patterns.
Figure 4.
Heatmap of individual observer estimates of FRα-positive tumor cell percentages across borderline and discordant cases. Borderline cases were defined based on a 65–85% staining threshold with 2+/3+ intensity, and discordant cases were selected among tumors showing marked variability in the 31–45% and 46–64% percentage intervals. Values highlight interobserver variability patterns.
3. Discussion
This study assessed the interobserver agreement in the immunohistochemical (IHC) evaluation of folate receptor alpha (FRα) expression in ovarian cancer across a cohort of 12 pathologists reviewing 37 cases. To our knowledge, this is among the first studies to specifically quantify diagnostic concordance in FRα IHC interpretation using real-world pathology practice conditions. Our findings indicate that pathologists can evaluate FRα expression with a high degree of reliability, particularly when it comes to assessing internal control adequacy and estimating the percentage of FRα-positive tumor cells. However, some interpretive variability was observed in binary classification decisions, especially in borderline or equivocal cases, underscoring the need for standardization in clinical practice [
16,
17,
18].
The biological and clinical importance of FRα in ovarian cancer has been well established [
1,
2,
3,
4,
5,
6]. FRα is overexpressed in the majority of high-grade serous ovarian carcinomas and serves not only as a diagnostic and prognostic biomarker but also as a therapeutic target for antibody-drug conjugates, such as mirvetuximab soravtansine [
6,
7,
8,
9,
10,
11,
12,
13]. The efficacy of these targeted therapies is closely related to the level and distribution of FRα expression, necessitating accurate and reproducible IHC-based assessment [
6,
7,
8,
9,
10,
11,
12,
13]. Our results support the robustness of FRα as a biomarker, as evidenced by the strong interobserver agreement, particularly in estimating the percentage of tumor cells stained (ICC = 0.89). This finding is reassuring, as many current clinical trials and drug approvals rely on percentage thresholds (commonly ≥75%) to determine eligibility for FRα-targeted therapy [
7,
8,
9,
10,
11,
12,
13]. The evaluation of internal control tissue is a crucial first step in IHC interpretation, ensuring that staining was technically successful. Our study demonstrates excellent reproducibility in this area (Fleiss’ kappa = 0.84), likely attributable to the distinct morphology and reliable staining of fallopian tube epithelium. This supports the inclusion of tubal tissue on FRα IHC slides as an internal quality control measure in diagnostic protocols.
Agreement on the binary classification of FRα positivity or negativity was substantial (kappa = 0.76), though notably lower than agreement on control adequacy or percentage estimation. Discrepancies in binary classification were concentrated in tumors with intermediate staining (approximately 50–80% of cells positive) or with weak membranous staining. These are the same cases most likely to present interpretive challenges in clinical practice, where classification decisions can directly influence patient eligibility for FRα-targeted therapy. The reliance on semi-quantitative scoring systems, subjective intensity grading, and lack of digital standardization likely contribute to this variability. Notably, the cases that generated the most disagreement fell near the 75% cutoff point used in clinical trials such as MIRASOL [
19]. These findings suggest that interpretive ambiguity around such thresholds may lead to inconsistent classification in the absence of strict criteria or centralized review.
There is currently no universally accepted scoring system for FRα IHC in routine diagnostic practice, though clinical trials have converged on a simplified binary scheme based on the percentage of viable tumor cells showing ≥2+ membranous staining [
7,
8,
9,
10,
11,
12,
13,
14,
15].
Our findings reinforce the utility of this scheme but also emphasize that scoring performance may improve significantly with proper training and calibration of observers. Incorporating digital pathology tools or automated image analysis systems may further improve objectivity, especially in borderline cases. Alternatively, implementing a three-tier scoring system (e.g., FRα-high, FRα-intermediate, FRα-negative) could capture more nuance in expression patterns while mitigating the binary cutoff dilemma.
The strengths of this study include its multi-observer design, the use of a standardized FRα IHC protocol, and the inclusion of a well-characterized internal control. However, there are limitations. The study cohort was relatively small and enriched for high-grade serous carcinoma, which may limit generalizability to rarer histologic subtypes. Additionally, the variability in pathologist experience and lack of formal pre-review calibration could have contributed to some of the observed discrepancies, although this also makes the results reflective of real-world conditions. Finally, staining was assessed visually and manually; future studies using digital pathology and artificial intelligence may provide a more granular assessment of reproducibility and accuracy.
In conclusion, our results demonstrate a high level of interobserver agreement in FRα IHC interpretation among pathologists, particularly for percentage estimation and control tissue adequacy. While binary classification showed substantial concordance, cases near the threshold of positivity present interpretive challenges. These findings highlight the necessity of clear scoring criteria, suggest the incorporation of digital aids, and advocate for a more nuanced classification system to support clinical decision-making for FRα-targeted therapies. As FRα continues to gain relevance in ovarian cancer management, ensuring reproducibility in its assessment will be essential for its successful clinical implementation.
4. Materials and Methods
4.1. Case Selection and Immunohistochemistry
A total of 37 formalin-fixed, paraffin-embedded high-grade serous ovarian carcinoma cases were selected from the archives of our institution. All cases included accompanying fallopian tube epithelium on the same slide, serving as an internal control for FRα immunostaining. Immunohistochemistry for FRα was performed using the standardized VENTANA FOLR1 (FOLR1-2.1) RxDx Assay protocol, following manufacturer’s instructions and previously published recommendations [
18,
19]. Full-slide sections were evaluated for staining intensity and percentage of positive tumor cells.
4.2. Reference Evaluation
A panel of four experienced gynecological pathologists independently reviewed all 37 immunostained slides to establish a reference diagnosis. Their assessment included: i) adequacy of internal control tissue (fallopian tube epithelium); ii) FRα staining intensity in tumor cells (negative or positive); iii) percentage of tumor cells exhibiting membranous FRα staining (continuous variable). The consensus evaluation of the panel served as the diagnostic gold standard for subsequent interobserver comparison.
4.3. Observer Evaluation
Twelve pathologists, including general surgical and gynecological subspecialists with varying levels of experience, independently reviewed all 37 cases using digital or glass slide formats. Observers were blinded to both the reference panel evaluation and to each other’s assessments. A formal theoretical session (3 hours of lectures) was conducted; however, no training session or consensus discussion was held prior to the evaluation phase. Each observer was asked to record the same parameters evaluated by the reference panel: i) adequacy of internal control tissue (fallopian tube epithelium); ii) FRα staining intensity in tumor cells (negative or positive); iii) percentage of tumor cells exhibiting membranous FRα staining (continuous variable).
4.4. Assessment of Internal Control Adequacy
The internal control was assessed based on the presence and staining quality of fallopian tube epithelium on each slide. The internal control was considered adequate if fallopian tube epithelium was present on the slide and exhibited circumferential membranous staining of moderate intensity (2+), with or without intense apical staining (3+). The accompanying stromal tissue was required to show no staining (0) [
18,
19]. Adequacy of internal control tissue was independently assessed by all observers prior to tumor evaluation.
4.5. Assessment of FRα Staining Intensity in Tumor Cells
Only membranous staining, whether complete or incomplete, was considered for intensity scoring, in line with recommendations for FRα-targeted therapy eligibility [
18,
19]. Tumor staining intensity was categorized based on membranous immunoreactivity as negative (absent staining 0 or weak staining 1+) or positive (moderate staining 2+ or strong staining 3+) [
18,
19].
4.6. Assessment of the Percentage of FRα-Positive Tumor Cells
For each case, observers independently estimated the percentage of tumor cells exhibiting membranous FRα staining, regardless of intensity. Percentage estimates were recorded as continuous variables and grouped into predefined intervals (0–5%, 6–15%, 16–30%, 31–45%, 46–64%, 65–85%, and 86–100%).
4.7. Assessment of Overall FRα Positivity and Definition of Borderline Cases
Tumors were considered positive for FRα expression when ≥75% of tumor cells exhibited moderate (2+) or strong (3+) membranous staining, including apical or dot-like patterns, in accordance with established clinical thresholds for eligibility to FRα-targeted therapies [
18,
19].
Borderline cases were defined as tumors exhibiting moderate (2+) or strong (3+) membranous staining in 65–85% of tumor cells, corresponding to a range within ±10% of the established cutoff for FRα positivity, and thus positioned at the interface between positive and negative status [
18,
19].
Such borderline cases were identified because minor differences in the estimation of stained tumor cell percentage could lead to divergent clinical classifications regarding eligibility for FRα-targeted therapies [
18,
19].
Author Contributions
Conception and design: AS, GFZ, GF. Resources and methodology: AC, GF, LL, EGR. Collection and assembly of data: PC, ADL, MI. Data analysis and interpretation: AD, BPU, NDA. Writing—original draft preparation: AS, GA, AD. Writing—review and editing: AS, GA, Supervision: MV, GT, Project administration: GFZ. All authors have read and agreed to the final version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study complied with the Ethical Principles for Medical Research Involving Human Subjects according to the World Medical Association Declaration of Helsinki. The clinical information had been retrieved from the patients’ medical records and pathology reports. Patients’ initials or other personal identifiers did not appear in any image. Finally, all samples were anonymized before histology and immunohistochemistry. Therefore, no further ethical approval was necessary to perform the study.
Informed Consent Statement
The non-interventional, retrospective nature of our study did not require any informed consent, even if written informed consent had been obtained from each patient before surgical procedures.
Data Availability Statement
Manuscript data are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Webb, P.M.; Ibiebele, T.I.; Hughes, M.C.; Beesley, J.; van der Pols, J.C.; Chen, X.; et al. Folate and related micronutrients, folate-metabolising genes and risk of ovarian cancer. Eur J Clin Nutr 2011, 65, 1133–1140. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, T.; Muminovic, M.; Nano, O.; Vulfovich, M. Folate Receptor Alpha-A Novel Approach to Cancer Therapy. Int J Mol Sci 2024, 25, 1046. [Google Scholar] [CrossRef] [PubMed]
- Scaranti, M.; Cojocaru, E.; Banerjee, S.; Banerji, U. Exploiting the folate receptor α in oncology. Nat Rev Clin Oncol 2020, 17, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Goldman, I.D. Folate and thiamine transporters mediated by facilitative carriers (SLC19A1-3 and SLC46A1) and folate receptors. Mol Aspects Med 2013, 34, 373–385. [Google Scholar] [CrossRef] [PubMed]
- Kelemen, L.E. The role of folate receptor alpha in cancer development, progression and treatment: Cause, consequence or innocent bystander? Int J Cancer 2006, 119, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Bax, H.J.; Chauhan, J.; Stavraka, C.; Santaolalla, A.; Osborn, G.; Khiabany, A.; et al. Folate receptor alpha in ovarian cancer tissue and patient serum is associated with disease burden and treatment outcomes. Br J Cancer 2023, 128, 342–353. [Google Scholar] [CrossRef] [PubMed]
- Moore, K.N.; Angelergues, A.; Konecny, G.E.; García, Y.; Banerjee, S.; Lorusso, D.; et al. Mirvetuximab Soravtansine in FRα-Positive, Platinum-Resistant Ovarian Cancer. N Engl J Med 2023, 389, 2162–2174. [Google Scholar] [CrossRef] [PubMed]
- Kong, B.; Zheng, W. Mirvetuximab soravtansine: Current and future applications. J Hematol Oncol 2025, 18, 33. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, L.; Oaknin, A.; Matulonis, U.A.; Mantia-Smaldone, G.M.; Lim, P.C.; Castro, C.M.; et al. Safety and efficacy of mirvetuximab soravtansine, a folate receptor alpha (FRα)-targeting antibody-drug conjugate (ADC), in combination with bevacizumab in patients with platinum-resistant ovarian cancer. Gynecol Oncol 2023, 170, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Richardson, D.L.; Moore, K.N.; Vergote, I.; Gilbert, L.; Martin, L.P.; Mantia-Smaldone, G.M.; et al. Phase 1b study of mirvetuximab soravtansine, a folate receptor alpha (FRα)-targeting antibody-drug conjugate, in combination with carboplatin and bevacizumab in patients with platinum-sensitive ovarian cancer. Gynecol Oncol 2024, 185, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Coleman, R.L.; Lorusso, D.; Oaknin, A.; Cecere, S.C.; Denys, H.; Colombo, N.; et al. Mirvetuximab soravtansine in folate receptor alpha (FRα)-high platinum-resistant ovarian cancer: Final overall survival and post hoc sequence of therapy subgroup results from the SORAYA trial. Int J Gynecol Cancer 2024, 34, 1119–1125. [Google Scholar] [CrossRef] [PubMed]
- Alvarez Secord, A.; Lewin, S.N.; Murphy, C.G.; Cecere, S.C.; Barquín, A.; Gálvez-Montosa, F.; et al. The efficacy and safety of mirvetuximab soravtansine in FRα-positive, third-line and later, recurrent platinum-sensitive ovarian cancer: The single-arm phase II PICCOLO trial. Ann Oncol 2025, 36, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Van Gorp, T.; Moore, K.N.; Konecny, G.E.; Leary, A.; García-García, Y.; Banerjee, S.; et al. Patient-reported outcomes from the MIRASOL trial evaluating mirvetuximab soravtansine versus chemotherapy in patients with folate receptor α-positive, platinum-resistant ovarian cancer: A randomised, open-label, phase 3 trial. Lancet Oncol 2025, 26, 503–515. [Google Scholar] [CrossRef] [PubMed]
- Previs, R.A.; Strickland, K.C.; Wallen, Z.; Ko, H.; Green, M.; Cooper, M.; et al. Analysis of real world FRα testing in ovarian, fallopian tube, and primary peritoneal cancers. Gynecol Oncol 2025, 192, 102–110. [Google Scholar] [CrossRef] [PubMed]
- James, R.L.; Sisserson, T.; Cai, Z.; Dumas, M.E.; Inge, L.J.; Ranger-Moore, J.; Mason, A.; Sloss, C.M.; McArthur, K. Development of an FRα Companion Diagnostic Immunohistochemical Assay for Mirvetuximab Soravtansine. Arch Pathol Lab Med 2024, 148, 1226–1233. [Google Scholar] [CrossRef] [PubMed]
- Deutschman, E.; Fulton, R.; Sloss, C.M. Evaluation of Laboratory-Derived Immunohistochemical Assays for Folate Receptor α Expression in Epithelial Ovarian Cancer and Comparison With a Companion Diagnostic. Arch Pathol Lab Med 2025. [Google Scholar] [CrossRef] [PubMed]
- Lawson, B.C.; Marques-Piubelli, M.L.; Westin, S.N.; Malpica, A. Folate Receptor Immunohistochemical Staining and Gynecologic Tumors: Initial Experience With 216 Cases. Int J Gynecol Pathol 2025, 44, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Martin, L.P.; Konner, J.A.; Moore, K.N.; Seward, S.M.; Matulonis, U.A.; Perez, R.P.; Su, Y.; Berkenblit, A.; Ruiz-Soto, R.; Birrer, M.J. Characterization of folate receptor alpha (FRα) expression in archival tumor and biopsy samples from relapsed epithelial ovarian cancer patients: A phase I expansion study of the FRα-targeting antibody-drug conjugate mirvetuximab soravtansine. Gynecol Oncol 2017, 147, 402–407. [Google Scholar] [CrossRef] [PubMed]
- Dilawari, A.; Shah, M.; Ison, G.; Gittleman, H.; Fiero, M.H.; Shah, A.; Hamed, S.S.; Qiu, J.; Yu, J.; Manheng, W.; Ricks, T.K.; Pragani, R.; Arudchandran, A.; Patel, P.; Zaman, S.; Roy, A.; Kalavar, S.; Ghosh, S.; Pierce, W.F.; Rahman, N.A.; Tang, S.; Mixter, B.D.; Kluetz, P.G.; Pazdur, R.; Amiri-Kordestani, L. FDA Approval Summary: Mirvetuximab Soravtansine-Gynx for FRα-Positive, Platinum-Resistant Ovarian Cancer. Clin Cancer Res 2023, 29, 3835–3840. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).