Submitted:
27 May 2025
Posted:
29 May 2025
Read the latest preprint version here
Abstract
Keywords:
Introduction
2. The mRNA–LNP-induced Inflammatory Complications and their Root Cause
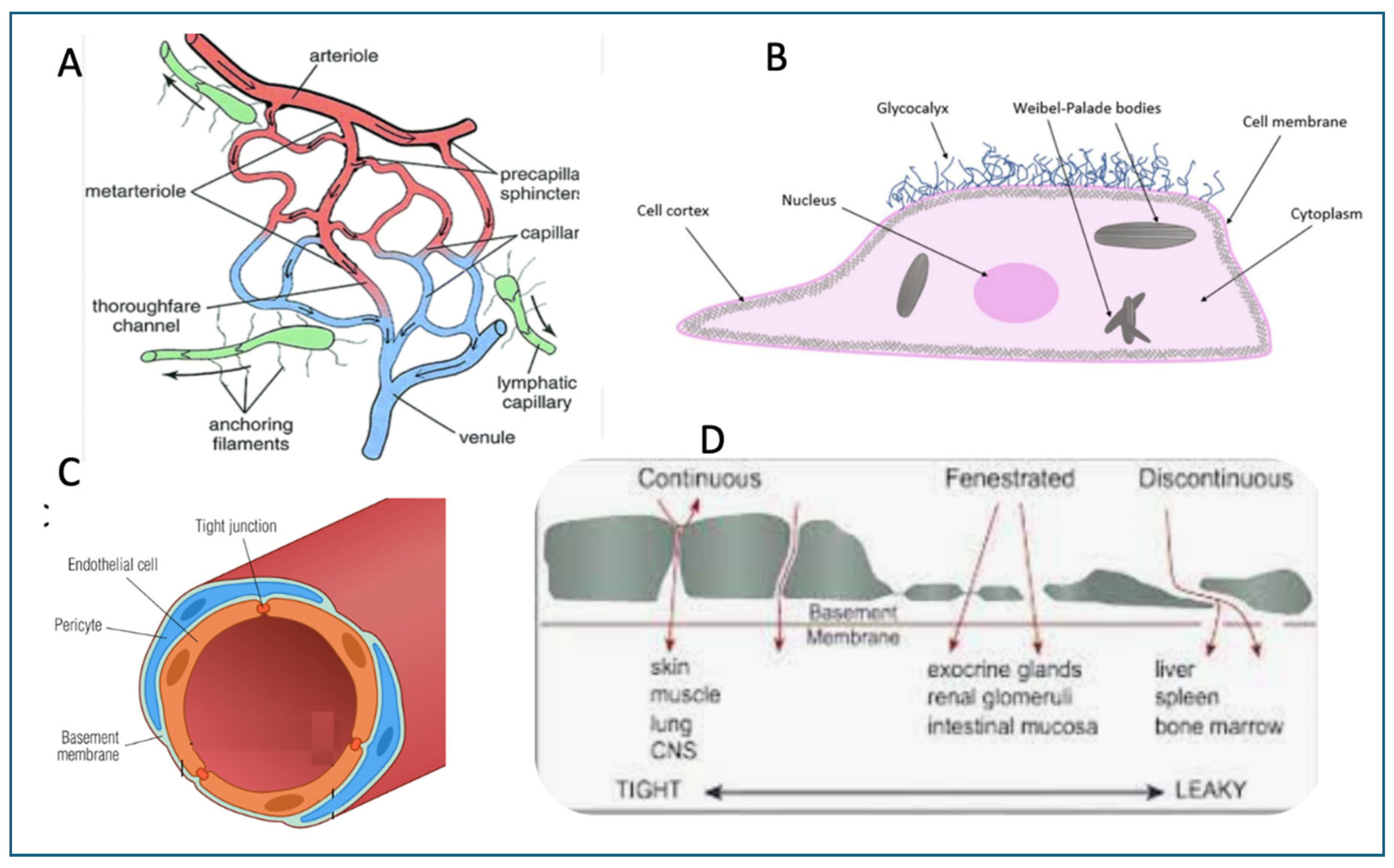
4. Endothelial Cells in the Frontline of Vaccine-Induced Inflammation
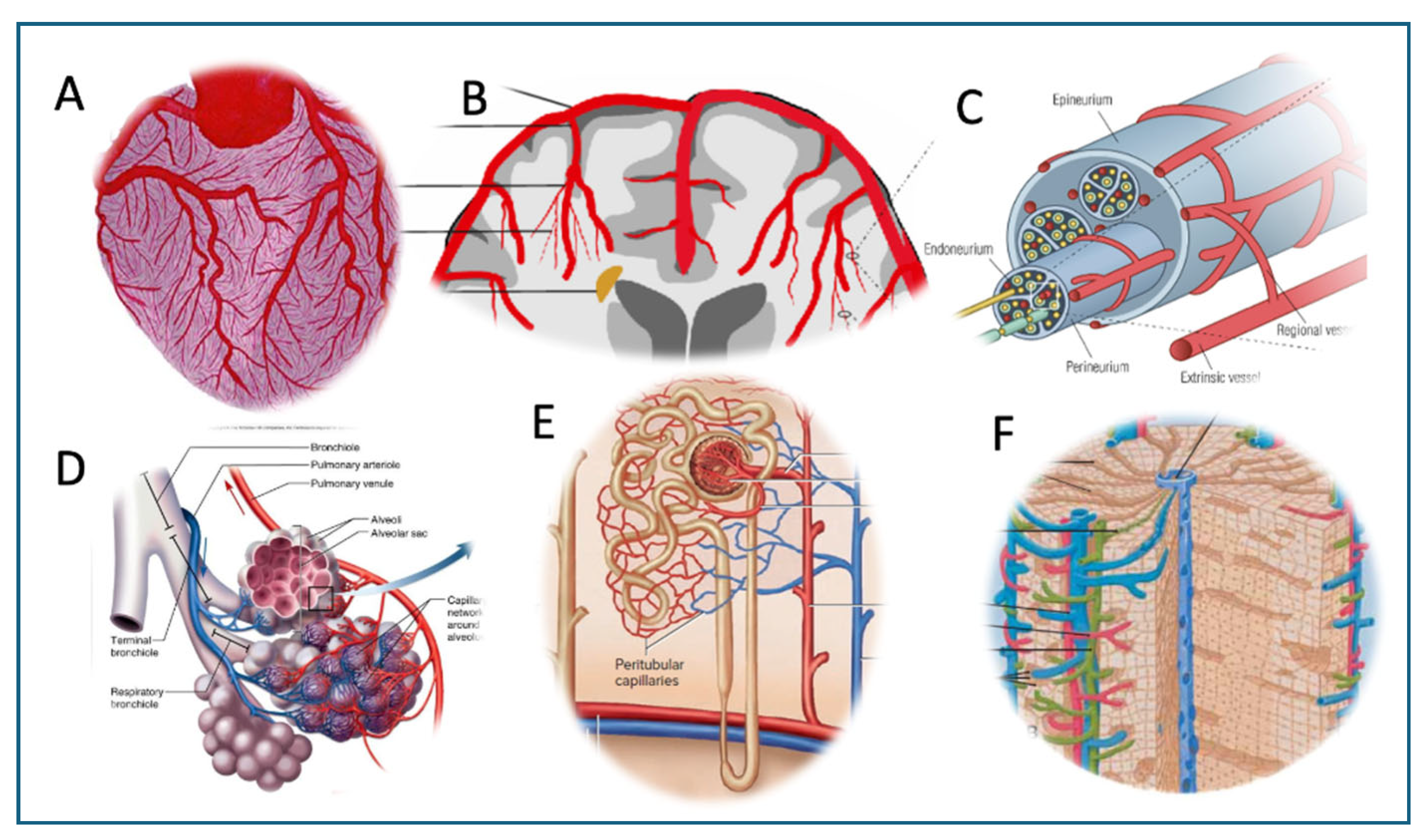
5. Distinctive Microcirculatory Architectures Across Organs and Their Impacts on Vaccine-Induced Inflammations
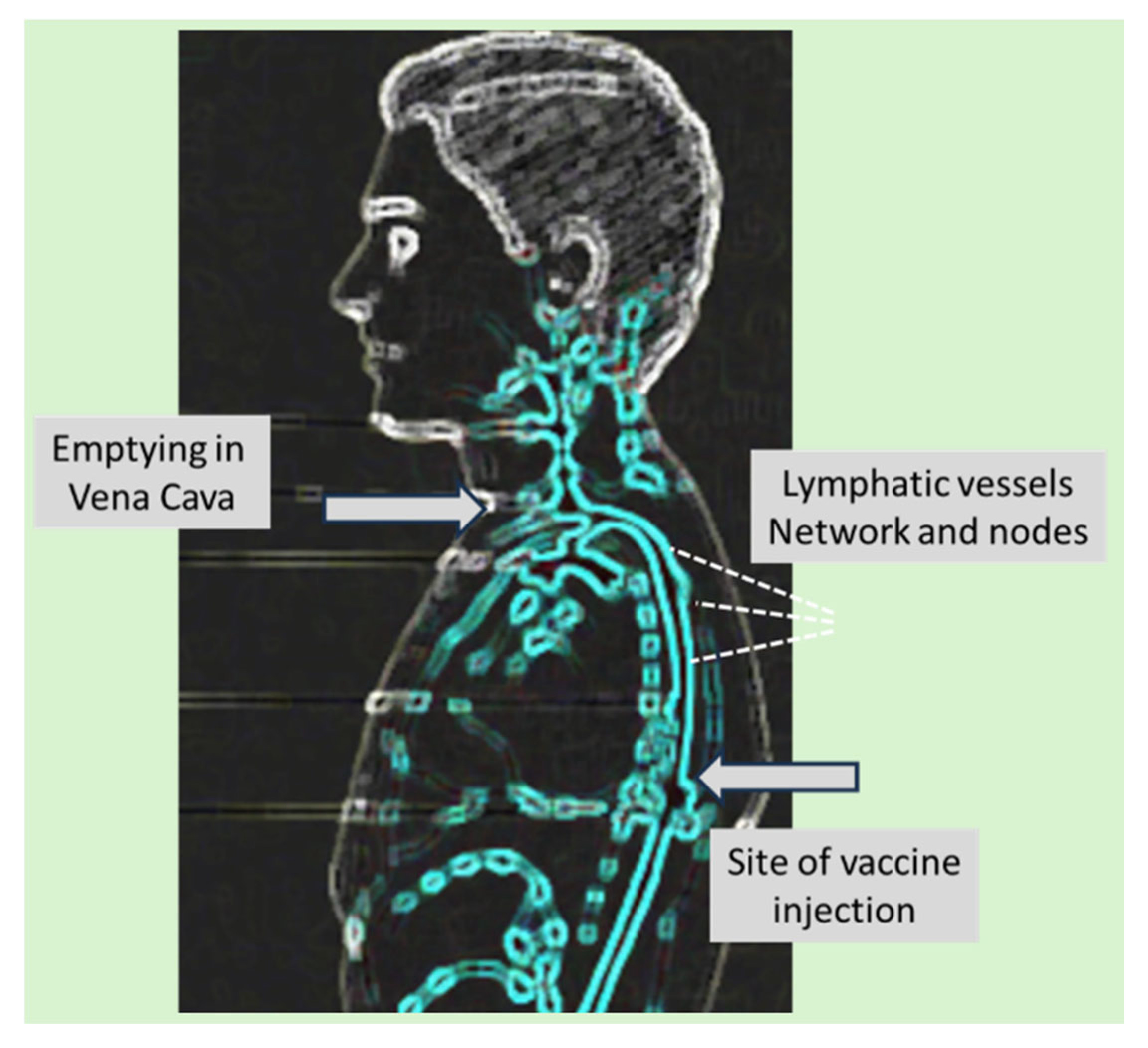
6. The Journey of mRNA-LNPs from the Deltoid Muscle to the Sites of Inflammations
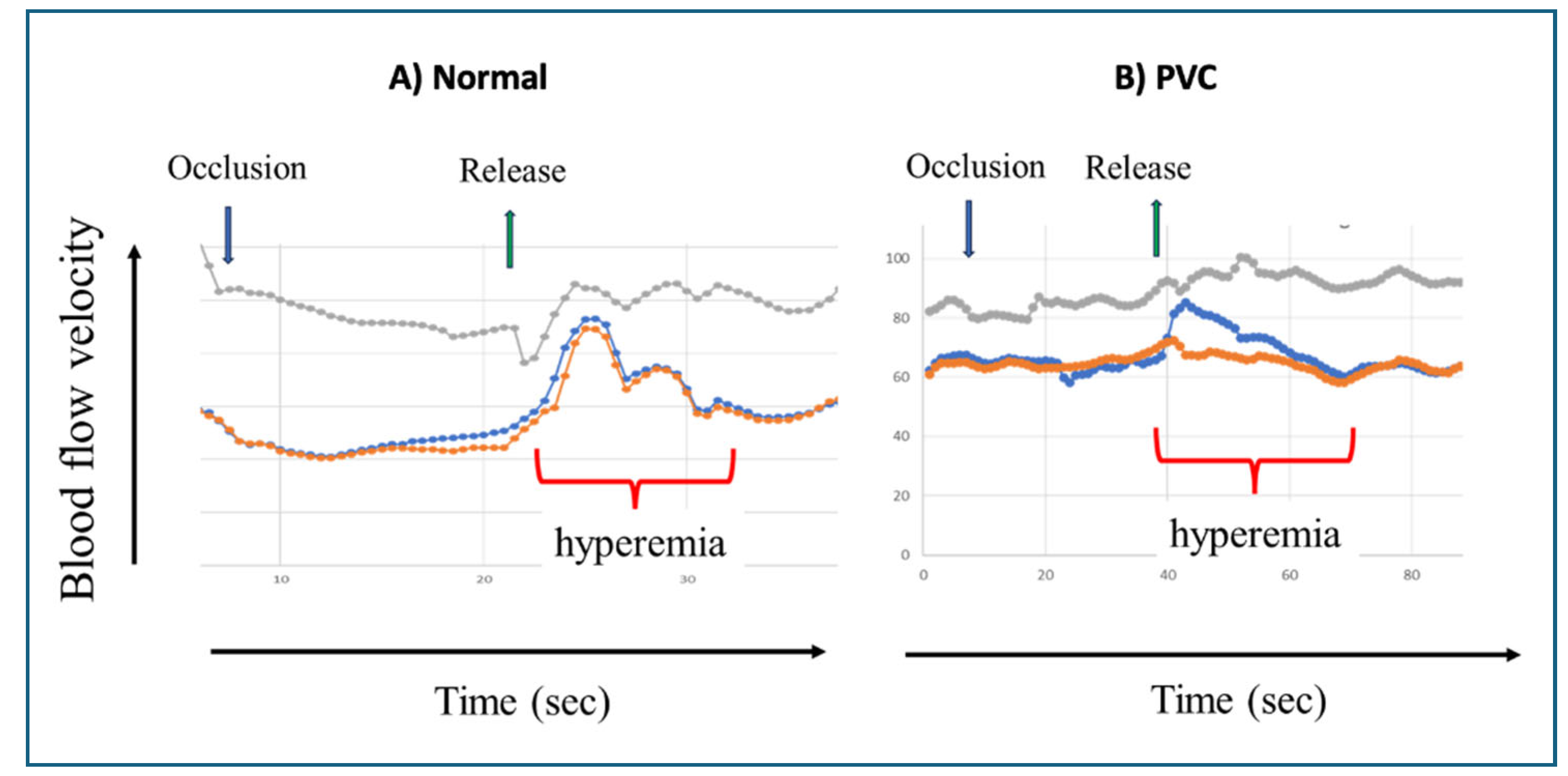
7. The Pathophysiology and Diagnosis of Vaccine-Induced Vasculitis
8. Systemic Biodistribution and Non-Target Organ Uptake of mRNA–LNPs
9. Adverse Impacts of mRNA-LNPs and the Spike Protein on Endothelial Cells
10. Causes and features of endothelitis
11. Outlook
Funding
References
- Szebeni, J. "Expanded Spectrum and Increased Incidence of Adverse Events Linked to Covid-19 Genetic Vaccines: New Concepts on Prophylactic Immuno-Gene Therapy, Iatrogenic Orphan Disease, and Platform-Inherent Challenges." Pharmaceutics 17, no. 4 (2025): 450.
- Krumholz, H. M., Y. Wu, M. Sawano, R. Shah, T. Zhou, A. S. Arun, P. Khosla, S. Kaleem, A. Vashist, B. Bhattacharjee, Q. Ding, Y. Lu, C. Caraballo, F. Warner, C. Huang, J. Herrin, D. Putrino, D. Hertz, B. Dressen, and A. Iwasaki. "Post-Vaccination Syndrome: A Descriptive Analysis of Reported Symptoms and Patient Experiences after Covid-19 Immunization." medRxiv posted , 2023. 10 November. [CrossRef]
- Palmer, M., S. Bhakdi, B. Hooker, M. Holland, M. DesBois, D. Rasnick, and C.A. Fitts. Mrna Vvaccine Toxicity, /: https, 2023. [Google Scholar]
- Costa, C. , and F. Moniati. "The Epidemiology of Covid-19 Vaccine-Induced Myocarditis." Adv Med 2024 (2024): 4470326.
- Novak, N., L. Tordesillas, and B. Cabanillas. "Adverse Rare Events to Vaccines for Covid-19: From Hypersensitivity Reactions to Thrombosis and Thrombocytopenia." Int Rev Immunol 41, no. 4 (2022): 438-47.
- Oueijan, R. I., O. R. Hill, P. D. Ahiawodzi, P. S. Fasinu, and D. K. Thompson. "Rare Heterogeneous Adverse Events Associated with Mrna-Based Covid-19 Vaccines: A Systematic Review." Medicines (Basel) 9, no. 8 (2022).
- Padilla-Flores, T., A. Sampieri, and L. Vaca. "Incidence and Management of the Main Serious Adverse Events Reported after Covid-19 Vaccination." Pharmacol Res Perspect 12, no. 3 (2024): e1224.
- Worldwide Safety. "Cumulative Analysis of Post-Authorization Adverse Event Reports of Pf-07302048 (Bnt162b2) Received through 28-Feb-2021." https://phmpt.org/wp-content/uploads/2021/11/5.3.6-postmarketing-experience.pdf?fbclid=IwAR2tWI7DKw0cc2lj8 (2021).
- Szebeni, J. "The Unique Features and Collateral Immune Effects of Mrna-Based Covid-19 Vaccines: Potential Plausible Causes of Adverse Events and Complications." In Preprints: Preprints, 2025.
- Kariko, K., M. Buckstein, H. Ni, and D. Weissman. "Suppression of Rna Recognition by Toll-Like Receptors: The Impact of Nucleoside Modification and the Evolutionary Origin of Rna." Immunity 23, no. 2 (2005): 165-75.
- Kariko, K., H. Muramatsu, F. A. Welsh, J. Ludwig, H. Kato, S. Akira, and D. Weissman. "Incorporation of Pseudouridine into Mrna Yields Superior Nonimmunogenic Vector with Increased Translational Capacity and Biological Stability." Mol Ther 16, no. 11 (2008): 1833-40.
- Anderson, B. R., H. Muramatsu, S. R. Nallagatla, P. C. Bevilacqua, L. H. Sansing, D. Weissman, and K. Kariko. "Incorporation of Pseudouridine into Mrna Enhances Translation by Diminishing Pkr Activation." Nucleic Acids Res 38, no. 17 (2010): 5884-92.
- Anderson, B. R., H. Muramatsu, B. K. Jha, R. H. Silverman, D. Weissman, and K. Kariko. "Nucleoside Modifications in Rna Limit Activation of 2'-5'-Oligoadenylate Synthetase and Increase Resistance to Cleavage by Rnase L." Nucleic Acids Res 39, no. 21 (2011): 9329-38.
- Kariko, K., H. Muramatsu, J. Ludwig, and D. Weissman. "Generating the Optimal Mrna for Therapy: Hplc Purification Eliminates Immune Activation and Improves Translation of Nucleoside-Modified, Protein-Encoding Mrna." Nucleic Acids Res 39, no. 21 (2011): e142.
- Rehwinkel, J. , and M. U. Gack. "Rig-I-Like Receptors: Their Regulation and Roles in Rna Sensing." Nat Rev Immunol 20, no. 9 (2020): 537-51.
- Lv, Y. , and Y. Chang. "Cytomegalovirus Proctitis Developed after Covid-19 Vaccine: A Case Report and Literature Review." Vaccines (Basel) 10, no. 9 (2022).
- Chakravorty, S., A. B. Cochrane, M. A. Psotka, A. Regmi, L. Marinak, A. Thatcher, O. A. Shlobin, A. W. Brown, C. S. King, K. Ahmad, V. Khangoora, A. Singhal, S. D. Nathan, and S. Aryal. "Cmv Infection Following Mrna Sars-Cov-2 Vaccination in Solid Organ Transplant Recipients." Transplant Direct 8, no. 7 (2022): e1344.
- Herzum, A., I. Trave, F. D'Agostino, M. Burlando, E. Cozzani, and A. Parodi. "Epstein-Barr Virus Reactivation after Covid-19 Vaccination in a Young Immunocompetent Man: A Case Report." Clin Exp Vaccine Res 11, no. 2 (2022): 222-25.
- Navarro-Bielsa, A., T. Gracia-Cazana, B. Aldea-Manrique, I. Abadias-Granado, A. Ballano, I. Bernad, and Y. Gilaberte. "Covid-19 Infection and Vaccines: Potential Triggers of Herpesviridae Reactivation." An Bras Dermatol 98, no. 3 (2023): 347-54.
- Shafiee, A., M. J. Amini, R. Arabzadeh Bahri, K. Jafarabady, S. A. Salehi, H. Hajishah, and S. H. Mozhgani. "Herpesviruses Reactivation Following Covid-19 Vaccination: A Systematic Review and Meta-Analysis." Eur J Med Res 28, no. 1 (2023): 278.
- Maple, P. A. C. "Covid-19, Sars-Cov-2 Vaccination, and Human Herpesviruses Infections." Vaccines (Basel) 11, no. 2 (2023).
- Martinez-Reviejo, R., S. Tejada, G. A. R. Adebanjo, C. Chello, M. C. Machado, F. R. Parisella, M. Campins, A. Tammaro, and J. Rello. "Varicella-Zoster Virus Reactivation Following Severe Acute Respiratory Syndrome Coronavirus 2 Vaccination or Infection: New Insights." Eur J Intern Med 104 (2022): 73-79.
- Elbaz, M., T. Hoffman, D. Yahav, S. Dovrat, N. Ghanem-Zoubi, A. Atamna, D. Grupel, S. Reisfeld, M. Hershman-Sarafov, P. Ciobotaro, R. Najjar-Debbiny, T. Brosh-Nissimov, B. Chazan, O. Yossepowitch, Y. Wiener-Well, O. Halutz, S. Reich, R. Ben-Ami, and Y. Paran. "Varicella-Zoster Virus-Induced Neurologic Disease after Covid-19 Vaccination: A Multicenter Observational Cohort Study." Open Forum Infect Dis 11, no. 6 (2024): ofae287.
- Attwell, D., A. Mishra, C. N. Hall, F. M. O'Farrell, and T. Dalkara. "What Is a Pericyte?" J Cereb Blood Flow Metab 36, no. 2 (2016): 451-5.
- Ishiko, S., A. Koller, W. Deng, A. Huang, and D. Sun. "Liposomal Nanocarriers of Preassembled Glycocalyx Restore Normal Venular Permeability and Shear Stress Sensitivity in Sepsis: Assessed Quantitatively with a Novel Microchamber System." Am J Physiol Heart Circ Physiol 327, no. 2 (2024): H390-H98.
- Takacs, J., D. Deak, and A. Koller. "Higher Level of Physical Activity Reduces Mental and Neurological Symptoms During and Two Years after Covid-19 Infection in Young Women." Sci Rep 14, no. 1 (2024): 6927.
- Nemeth, Z., A. Cziraki, S. Szabados, I. Horvath, and A. Koller. "Pericardial Fluid of Cardiac Patients Elicits Arterial Constriction: Role of Endothelin-1." Can J Physiol Pharmacol 93, no. 9 (2015): 779-85.
- Nemeth, Z., A. Cziraki, S. Szabados, B. Biri, S. Keki, and A. Koller. "Elevated Levels of Asymmetric Dimethylarginine (Adma) in the Pericardial Fluid of Cardiac Patients Correlate with Cardiac Hypertrophy." PLoS One 10, no. 8 (2015): e0135498.
- Cziraki, A., Z. Lenkey, E. Sulyok, I. Szokodi, and A. Koller. "L-Arginine-Nitric Oxide-Asymmetric Dimethylarginine Pathway and the Coronary Circulation: Translation of Basic Science Results to Clinical Practice." Front Pharmacol 11 (2020): 569914.
- Gill, J. R., R. Tashjian, and E. Duncanson. "Autopsy Histopathologic Cardiac Findings in 2 Adolescents Following the Second Covid-19 Vaccine Dose." Arch Pathol Lab Med 146, no. 8 (2022): 925-29.
- Verma, A. K., K. J. Lavine, and C. Y. Lin. "Myocarditis after Covid-19 Mrna Vaccination." N Engl J Med 385, no. 14 (2021): 1332-34.
- Rodriguez, E. R. , and C. D. Tan. "Structure and Anatomy of the Human Pericardium." Prog Cardiovasc Dis 59, no. 4 (2017): 327-40.
- Rinaldi, V., G. Bellucci, M. C. Buscarinu, R. Renie, A. Marrone, M. Nasello, V. Zancan, R. Nistri, R. Palumbo, A. Salerno, M. Salvetti, and G. Ristori. "Cns Inflammatory Demyelinating Events after Covid-19 Vaccines: A Case Series and Systematic Review." Front Neurol 13 (2022): 1018785.
- Patone, M., L. Handunnetthi, D. Saatci, J. Pan, S. V. Katikireddi, S. Razvi, D. Hunt, X. W. Mei, S. Dixon, F. Zaccardi, K. Khunti, P. Watkinson, C. A. C. Coupland, J. Doidge, D. A. Harrison, R. Ravanan, A. Sheikh, C. Robertson, and J. Hippisley-Cox. "Neurological Complications after First Dose of Covid-19 Vaccines and Sars-Cov-2 Infection." Nat Med 27, no. 12 (2021): 2144-53.
- Khayat-Khoei, M., S. Bhattacharyya, J. Katz, D. Harrison, S. Tauhid, P. Bruso, M. K. Houtchens, K. R. Edwards, and R. Bakshi. "Covid-19 Mrna Vaccination Leading to Cns Inflammation: A Case Series." J Neurol 269, no. 3 (2022): 1093-106.
- Yoshikawa, T., K. Tomomatsu, E. Okazaki, T. Takeuchi, Y. Horio, Y. Kondo, T. Oguma, and K. Asano. "Covid-19 Vaccine-Associated Organizing Pneumonia." Respirol Case Rep 10, no. 5 (2022): e0944.
- Park, J. Y., J. H. Kim, S. Park, Y. I. Hwang, H. I. Kim, S. H. Jang, K. S. Jung, Y. K. Kim, H. A. Kim, and I. J. Lee. "Clinical Characteristics of Patients with Covid-19 Vaccine-Related Pneumonitis: A Case Series and Literature Review." Korean J Intern Med 37, no. 5 (2022): 989-1001.
- Kervella, D., L. Jacquemont, A. Chapelet-Debout, C. Deltombe, and S. Ville. "Minimal Change Disease Relapse Following Sars-Cov-2 Mrna Vaccine." Kidney Int 100, no. 2 (2021): 457-58.
- Zhang, J., J. Cao, and Q. Ye. "Renal Side Effects of Covid-19 Vaccination." Vaccines (Basel) 10, no. 11 (2022).
- Rocco, A., C. Sgamato, D. Compare, and G. Nardone. "Autoimmune Hepatitis Following Sars-Cov-2 Vaccine: May Not Be a Casuality." J Hepatol 75, no. 3 (2021): 728-29.
- Schinas, G., E. Polyzou, V. Dimakopoulou, S. Tsoupra, C. Gogos, and K. Akinosoglou. "Immune-Mediated Liver Injury Following Covid-19 Vaccination." World J Virol 12, no. 2 (2023): 100-08.
- Sergi, C. M. "Covid-19 Vaccination-Related Autoimmune Hepatitis-a Perspective." Front Pharmacol 14 (2023): 1190367.
- Chen, C., D. Xie, and J. Xiao. "Real-World Evidence of Autoimmune Hepatitis Following Covid-19 Vaccination: A Population-Based Pharmacovigilance Analysis." Front Pharmacol 14 (2023): 1100617.
- Kim, J. H., H. B. Chae, S. Woo, M. S. Song, H. J. Kim, and C. G. Woo. "Clinicopathological Characteristics of Autoimmune-Like Hepatitis Induced by Covid-19 Mrna Vaccine (Pfizer-Biontech, Bnt162b2): A Case Report and Literature Review." Int J Surg Pathol 31, no. 6 (2023): 1156-62.
- Liang, F., G. Lindgren, A. Lin, E. A. Thompson, S. Ols, J. Rohss, S. John, K. Hassett, O. Yuzhakov, K. Bahl, L. A. Brito, H. Salter, G. Ciaramella, and K. Lore. "Efficient Targeting and Activation of Antigen-Presenting Cells in Vivo after Modified Mrna Vaccine Administration in Rhesus Macaques." Mol Ther 25, no. 12 (2017): 2635-47.
- Bettini, E. , and M. Locci. "Sars-Cov-2 Mrna Vaccines: Immunological Mechanism and Beyond." Vaccines (Basel) 9, no. 2 (2021).
- Szabo, G. T., A. J. Mahiny, and I. Vlatkovic. "Covid-19 Mrna Vaccines: Platforms and Current Developments." Mol Ther 30, no. 5 (2022): 1850-68.
- Buckley, M., M. Arainga, L. Maiorino, I. S. Pires, B. J. Kim, K. K. Michaels, J. Dye, K. Qureshi, Y. J. Zhang, H. Mak, J. M. Steichen, W. R. Schief, F. Villinger, and D. J. Irvine. "Visualizing Lipid Nanoparticle Trafficking for Mrna Vaccine Delivery in Non-Human Primates." Mol Ther 33, no. 3 (2025): 1105-17.
- Ndeupen, S., Z. Qin, S. Jacobsen, A. Bouteau, H. Estanbouli, and B. Z. Igyarto. "The Mrna-Lnp Platform's Lipid Nanoparticle Component Used in Preclinical Vaccine Studies Is Highly Inflammatory." iScience 24, no. 12 (2021): 103479.
- Krauson, A. J., F. V. C. Casimero, Z. Siddiquee, and J. R. Stone. "Duration of Sars-Cov-2 Mrna Vaccine Persistence and Factors Associated with Cardiac Involvement in Recently Vaccinated Patients." NPJ Vaccines 8, no. 1 (2023): 141.
- Mizuno, R., G. Dornyei, A. Koller, and G. Kaley. "Myogenic Responses of Isolated Lymphatics: Modulation by Endothelium." Microcirculation 4, no. 4 (1997): 413-20.
- Koller, A., R. Mizuno, and G. Kaley. "Flow Reduces the Amplitude and Increases the Frequency of Lymphatic Vasomotion: Role of Endothelial Prostanoids." Am J Physiol 277, no. 6 (1999): R1683-9.
- Breslin, J. W. "Mechanical Forces and Lymphatic Transport." Microvasc Res 96 (2014): 46-54.
- von der Weid, P. Y. "Lymphatic Vessel Pumping." Adv Exp Med Biol 1124 (2019): 357-77.
- Alazraki, N., E. C. Glass, F. Castronovo, R. A. Olmos, D. Podoloff, and Medicine Society of Nuclear. "Procedure Guideline for Lymphoscintigraphy and the Use of Intraoperative Gamma Probe for Sentinel Lymph Node Localization in Melanoma of Intermediate Thickness 1.0." J Nucl Med 43, no. 10 (2002): 1414-8.
- Dixon, J. B., S. T. Greiner, A. A. Gashev, G. L. Cote, J. E. Moore, and D. C. Zawieja. "Lymph Flow, Shear Stress, and Lymphocyte Velocity in Rat Mesenteric Prenodal Lymphatics." Microcirculation 13, no. 7 (2006): 597-610.
- Mizuno, R., A. Koller, and G. Kaley. "Regulation of the Vasomotor Activity of Lymph Microvessels by Nitric Oxide and Prostaglandins." Am J Physiol 274, no. 3 (1998): R790-6.
- Borresen, B., A. E. Hansen, F. P. Fliedner, J. R. Henriksen, D. R. Elema, M. Brandt-Larsen, L. K. Kristensen, A. T. Kristensen, T. L. Andresen, and A. Kjaer. "Noninvasive Molecular Imaging of the Enhanced Permeability and Retention Effect by (64)Cu-Liposomes: In Vivo Correlations with (68)Ga-Rgd, Fluid Pressure, Diffusivity and (18)F-Fdg." Int J Nanomedicine 15 (2020): 8571-81.
- Ltd, Pfizer Australia Pty. "Nonclinical Evaluation Report: Bnt162b2 [Mrna] Covid-19 Vaccine (Comirnatytm)." https://www.tga.gov.au/sites/default/files/foi-2389-06.pdf https://t.co/Zrhakh7Xgv (2021).
- Vegh, A., A. Csorba, A. Koller, B. Mohammadpour, P. Killik, L. Istvan, M. Magyar, T. Fenesi, and Z. Z. Nagy. "Presence of Sars-Cov-2 on the Conjunctival Mucosa in Patients Hospitalized Due to Covid-19: Pathophysiological Considerations and Therapeutic Implications." Physiol Int 109, no. 4 (2022): 475-85.
- Pardi, N., M. J. Hogan, F. W. Porter, and D. Weissman. "Mrna Vaccines - a New Era in Vaccinology." Nat Rev Drug Discov 17, no. 4 (2018): 261-79.
- Sabnis, S., E. S. Kumarasinghe, T. Salerno, C. Mihai, T. Ketova, J. J. Senn, A. Lynn, A. Bulychev, I. McFadyen, J. Chan, O. Almarsson, M. G. Stanton, and K. E. Benenato. "A Novel Amino Lipid Series for Mrna Delivery: Improved Endosomal Escape and Sustained Pharmacology and Safety in Non-Human Primates." Mol Ther 26, no. 6 (2018): 1509-19.
- Hou, X., T. Zaks, R. Langer, and Y. Dong. "Lipid Nanoparticles for Mrna Delivery." Nat Rev Mater 6, no. 12 (2021): 1078-94.
- Li, S., Y. Hu, A. Li, J. Lin, K. Hsieh, Z. Schneiderman, P. Zhang, Y. Zhu, C. Qiu, E. Kokkoli, T. H. Wang, and H. Q. Mao. "Payload Distribution and Capacity of Mrna Lipid Nanoparticles." Nat Commun 13, no. 1 (2022): 5561.
- Szebeni, J., G. Storm, J. Y. Ljubimova, M. Castells, E. J. Phillips, K. Turjeman, Y. Barenholz, D. J. A. Crommelin, and M. A. Dobrovolskaia. "Applying Lessons Learned from Nanomedicines to Understand Rare Hypersensitivity Reactions to Mrna-Based Sars-Cov-2 Vaccines." Nat Nanotechnol 17, no. 4 (2022): 337-46.
- Jaffe, E. A. "Cell Biology of Endothelial Cells." Hum Pathol 18, no. 3 (1987): 234-9.
- Rice, C. M. , and N. J. Scolding. "The Diagnosis of Primary Central Nervous System Vasculitis." Pract Neurol 20, no. 2 (2020): 109-14.
- Gupta, N., S. B. Hiremath, R. I. Aviv, and N. Wilson. "Childhood Cerebral Vasculitis : A Multidisciplinary Approach." Clin Neuroradiol 33, no. 1 (2023): 5-20.
- Ota, N., M. Itani, T. Aoki, A. Sakurai, T. Fujisawa, Y. Okada, K. Noda, Y. Arakawa, S. Tokuda, and R. Tanikawa. "Expression of Sars-Cov-2 Spike Protein in Cerebral Arteries: Implications for Hemorrhagic Stroke Post-Mrna Vaccination." J Clin Neurosci 136 (2025): 111223.
- Felgner, P. L., T. R. Gadek, M. Holm, R. Roman, H. W. Chan, M. Wenz, J. P. Northrop, G. M. Ringold, and M. Danielsen. "Lipofection: A Highly Efficient, Lipid-Mediated DNA-Transfection Procedure." Proc Natl Acad Sci U S A 84, no. 21 (1987): 7413-7.
- Horejs, C. "From Lipids to Lipid Nanoparticles to Mrna Vaccines." Nat Rev Mater 6, no. 12 (2021): 1075-76.
- HOLLAND, JOHN W. (Australia), PIETER R. (Canada) CULLIS, and THOMAS D. (Canada) MADDEN. "Bilayer Stabilizing Components and Their Use in Forming Programmable Fusogenic Liposomes." edited by THE UNIVERSITY OF BRITISH COLUMBIA, 2007-12-04.
- Chen, S., Y. Y. C. Tam, P. J. C. Lin, M. M. H. Sung, Y. K. Tam, and P. R. Cullis. "Influence of Particle Size on the in Vivo Potency of Lipid Nanoparticle Formulations of Sirna." J Control Release 235 (2016): 236-44.
- Cullis, P. R. , and M. J. Hope. "Lipid Nanoparticle Systems for Enabling Gene Therapies." Mol Ther 25, no. 7 (2017): 1467-75.
- Kulkarni, J. A., M. M. Darjuan, J. E. Mercer, S. Chen, R. van der Meel, J. L. Thewalt, Y. Y. C. Tam, and P. R. Cullis. "On the Formation and Morphology of Lipid Nanoparticles Containing Ionizable Cationic Lipids and Sirna." ACS Nano 12, no. 5 (2018): 4787-95.
- Akinc, A., M. A. Maier, M. Manoharan, K. Fitzgerald, M. Jayaraman, S. Barros, S. Ansell, X. Du, M. J. Hope, T. D. Madden, B. L. Mui, S. C. Semple, Y. K. Tam, M. Ciufolini, D. Witzigmann, J. A. Kulkarni, R. van der Meel, and P. R. Cullis. "The Onpattro Story and the Clinical Translation of Nanomedicines Containing Nucleic Acid-Based Drugs." Nat Nanotechnol 14, no. 12 (2019): 1084-87.
- Pateev, I., K. Seregina, R. Ivanov, and V. Reshetnikov. "Biodistribution of Rna Vaccines and of Their Products: Evidence from Human and Animal Studies." Biomedicines 12, no. 1 (2023).
- Dalby, B., S. Cates, A. Harris, E. C. Ohki, M. L. Tilkins, P. J. Price, and V. C. Ciccarone. "Advanced Transfection with Lipofectamine 2000 Reagent: Primary Neurons, Sirna, and High-Throughput Applications." Methods 33, no. 2 (2004): 95-103.
- Ferraresso, F., K. Badior, M. Seadler, Y. Zhang, A. Wietrzny, M. F. Cau, A. Haugen, G. G. Rodriguez, M. R. Dyer, P. R. Cullis, E. Jan, and C. J. Kastrup. "Protein Is Expressed in All Major Organs after Intravenous Infusion of Mrna-Lipid Nanoparticles in Swine." Mol Ther Methods Clin Dev 32, no. 3 (2024): 101314.
- Pardi, N., S. Tuyishime, H. Muramatsu, K. Kariko, B. L. Mui, Y. K. Tam, T. D. Madden, M. J. Hope, and D. Weissman. "Expression Kinetics of Nucleoside-Modified Mrna Delivered in Lipid Nanoparticles to Mice by Various Routes." J Control Release 217 (2015): 345-51.
- László Dézsi1, 2, **, Gábor Kökény2**, Gábor Szénási2, Csaba Révész1,2, Tamás Mészáros1,2,3,4, Balint A. Barta4, ????, Reka Facsko1,2,4, Anna Szilasi5, Tamás Bakos1, Gergely T. Kozma1,3, Attila B. Dobos4, Béla Merkely4, Tamás Radovits4, János Szebeni1,2,3,*. "Acute Anaphylactic and Multiorgan Inflammatory Effects of Mrna Vaccines in Pigs: Pcr Evidence of Spike Protein Mrna Transfection and Paralleling Inflammatory Cytokine Upregulation." Preprint (2025).
- Sago, C. D., M. P. Lokugamage, K. Paunovska, D. A. Vanover, C. M. Monaco, N. N. Shah, M. Gamboa Castro, S. E. Anderson, T. G. Rudoltz, G. N. Lando, P. Munnilal Tiwari, J. L. Kirschman, N. Willett, Y. C. Jang, P. J. Santangelo, A. V. Bryksin, and J. E. Dahlman. "High-Throughput in Vivo Screen of Functional Mrna Delivery Identifies Nanoparticles for Endothelial Cell Gene Editing." Proc Natl Acad Sci U S A 115, no. 42 (2018): E9944-E52.
- Francia, V., R. M. Schiffelers, P. R. Cullis, and D. Witzigmann. "The Biomolecular Corona of Lipid Nanoparticles for Gene Therapy." Bioconjug Chem 31, no. 9 (2020): 2046-59.
- Cheng, M. H. Y., J. Leung, Y. Zhang, C. Strong, G. Basha, A. Momeni, Y. Chen, E. Jan, A. Abdolahzadeh, X. Wang, J. A. Kulkarni, D. Witzigmann, and P. R. Cullis. "Induction of Bleb Structures in Lipid Nanoparticle Formulations of Mrna Leads to Improved Transfection Potency." Adv Mater 35, no. 31 (2023): e2303370.
- Liu, G. W., E. B. Guzman, N. Menon, and R. S. Langer. "Lipid Nanoparticles for Nucleic Acid Delivery to Endothelial Cells." Pharm Res 40, no. 1 (2023): 3-25.
- Cullis, P. R. , and P. L. Felgner. "The 60-Year Evolution of Lipid Nanoparticles for Nucleic Acid Delivery." Nat Rev Drug Discov (2024).
- Yazdi, M., J. Pohmerer, M. Hasanzadeh Kafshgari, J. Seidl, M. Grau, M. Hohn, V. Vetter, C. C. Hoch, B. Wollenberg, G. Multhoff, A. Bashiri Dezfouli, and E. Wagner. "In Vivo Endothelial Cell Gene Silencing by Sirna-Lnps Tuned with Lipoamino Bundle Chemical and Ligand Targeting." Small 20, no. 42 (2024): e2400643.
- Petersen, D. M. S., R. M. Weiss, K. A. Hajj, S. S. Yerneni, N. Chaudhary, A. N. Newby, M. L. Arral, and K. A. Whitehead. "Branched-Tail Lipid Nanoparticles for Intravenous Mrna Delivery to Lung Immune, Endothelial, and Alveolar Cells in Mice." Adv Healthc Mater 13, no. 22 (2024): e2400225.
- Papp, T. E., J. Zeng, H. Shahnawaz, A. Akyianu, L. Breda, A. Yadegari, J. Steward, R. Shi, Q. Li, B. L. Mui, Y. K. Tam, D. Weissman, S. Rivella, V. Shuvaev, V. R. Muzykantov, and H. Parhiz. "Cd47 Peptide-Cloaked Lipid Nanoparticles Promote Cell-Specific Mrna Delivery." Mol Ther (2025).
- Robles, J. P., M. Zamora, E. Adan-Castro, L. Siqueiros-Marquez, G. Martinez de la Escalera, and C. Clapp. "The Spike Protein of Sars-Cov-2 Induces Endothelial Inflammation through Integrin Alpha5beta1 and Nf-Kappab Signaling." J Biol Chem 298, no. 3 (2022): 101695.
- Yonker, L. M., Z. Swank, Y. C. Bartsch, M. D. Burns, A. Kane, B. P. Boribong, J. P. Davis, M. Loiselle, T. Novak, Y. Senussi, C. A. Cheng, E. Burgess, A. G. Edlow, J. Chou, A. Dionne, D. Balaguru, M. Lahoud-Rahme, M. Arditi, B. Julg, A. G. Randolph, G. Alter, A. Fasano, and D. R. Walt. "Circulating Spike Protein Detected in Post-Covid-19 Mrna Vaccine Myocarditis." Circulation 147, no. 11 (2023): 867-76.
- Avolio, E., M. Carrabba, R. Milligan, M. Kavanagh Williamson, A. P. Beltrami, K. Gupta, K. T. Elvers, M. Gamez, R. R. Foster, K. Gillespie, F. Hamilton, D. Arnold, I. Berger, A. D. Davidson, D. Hill, M. Caputo, and P. Madeddu. "The Sars-Cov-2 Spike Protein Disrupts Human Cardiac Pericytes Function through Cd147 Receptor-Mediated Signalling: A Potential Non-Infective Mechanism of Covid-19 Microvascular Disease." Clin Sci (Lond) 135, no. 24 (2021): 2667-89.
- Perico, L., M. Morigi, M. Galbusera, A. Pezzotta, S. Gastoldi, B. Imberti, A. Perna, P. Ruggenenti, R. Donadelli, A. Benigni, and G. Remuzzi. "Sars-Cov-2 Spike Protein 1 Activates Microvascular Endothelial Cells and Complement System Leading to Platelet Aggregation." Front Immunol 13 (2022): 827146.
- Perico, L., M. Morigi, A. Pezzotta, M. Locatelli, B. Imberti, D. Corna, D. Cerullo, A. Benigni, and G. Remuzzi. "Sars-Cov-2 Spike Protein Induces Lung Endothelial Cell Dysfunction and Thrombo-Inflammation Depending on the C3a/C3a Receptor Signalling." Sci Rep 13, no. 1 (2023): 11392.
- Huynh, T. V., L. Rethi, T. W. Lee, S. Higa, Y. H. Kao, and Y. J. Chen. "Spike Protein Impairs Mitochondrial Function in Human Cardiomyocytes: Mechanisms Underlying Cardiac Injury in Covid-19." Cells 12, no. 6 (2023).
- Schwartz, L., M. Aparicio-Alonso, M. Henry, M. Radman, R. Attal, and A. Bakkar. "Toxicity of the Spike Protein of Covid-19 Is a Redox Shift Phenomenon: A Novel Therapeutic Approach." Free Radic Biol Med 206 (2023): 106-10.
- Sahin, U., K. Kariko, and O. Tureci. "Mrna-Based Therapeutics--Developing a New Class of Drugs." Nat Rev Drug Discov 13, no. 10 (2014): 759-80.
- Forsyth, C. B., L. Zhang, A. Bhushan, B. Swanson, L. Zhang, J. I. Mamede, R. M. Voigt, M. Shaikh, P. A. Engen, and A. Keshavarzian. "The Sars-Cov-2 S1 Spike Protein Promotes Mapk and Nf-Kb Activation in Human Lung Cells and Inflammatory Cytokine Production in Human Lung and Intestinal Epithelial Cells." Microorganisms 10, no. 10 (2022).
- Niu, C., T. Liang, Y. Chen, S. Zhu, L. Zhou, N. Chen, L. Qian, Y. Wang, M. Li, X. Zhou, and J. Cui. "Sars-Cov-2 Spike Protein Induces the Cytokine Release Syndrome by Stimulating T Cells to Produce More Il-2." Front Immunol 15 (2024): 1444643.
- Pishesha, N., T. J. Harmand, and H. L. Ploegh. "A Guide to Antigen Processing and Presentation." Nat Rev Immunol 22, no. 12 (2022): 751-64.
- Palade, G. E. "A Small Particulate Component of the Cytoplasm." J Biophys Biochem Cytol 1, no. 1 (1955): 59-68.
- Embgenbroich, M. , and S. Burgdorf. "Current Concepts of Antigen Cross-Presentation." Front Immunol 9 (2018): 1643.
- Dezsi, L., T. Meszaros, G. Kozma, H. Velkei M, C. Z. Olah, M. Szabo, Z. Patko, T. Fulop, M. Hennies, M. Szebeni, B. A. Barta, B. Merkely, T. Radovits, and J. Szebeni. "A Naturally Hypersensitive Porcine Model May Help Understand the Mechanism of Covid-19 Mrna Vaccine-Induced Rare (Pseudo) Allergic Reactions: Complement Activation as a Possible Contributing Factor." Geroscience 44, no. 2 (2022): 597-618.
- Bakos, T., T. Meszaros, G. T. Kozma, P. Berenyi, R. Facsko, H. Farkas, L. Dezsi, C. Heirman, S. de Koker, R. Schiffelers, K. A. Glatter, T. Radovits, G. Szenasi, and J. Szebeni. "Mrna-Lnp Covid-19 Vaccine Lipids Induce Complement Activation and Production of Proinflammatory Cytokines: Mechanisms, Effects of Complement Inhibitors, and Relevance to Adverse Reactions." Int J Mol Sci 25, no. 7 (2024).
- Barta, B. A., T. Radovits, A. B. Dobos, G. Tibor Kozma, T. Meszaros, P. Berenyi, R. Facsko, T. Fulop, B. Merkely, and J. Szebeni. "Comirnaty-Induced Cardiopulmonary Distress and Other Symptoms of Complement-Mediated Pseudo-Anaphylaxis in a Hyperimmune Pig Model: Causal Role of Anti-Peg Antibodies." Vaccine X 19 (2024): 100497.
- Kozma, G. T., T. Meszaros, P. Berenyi, R. Facsko, Z. Patko, C. Z. Olah, A. Nagy, T. G. Fulop, K. A. Glatter, T. Radovits, B. Merkely, and J. Szebeni. "Role of Anti-Polyethylene Glycol (Peg) Antibodies in the Allergic Reactions to Peg-Containing Covid-19 Vaccines: Evidence for Immunogenicity of Peg." Vaccine 41, no. 31 (2023): 4561-70.
- Ali, Y. M., M. Ferrari, N. J. Lynch, S. Yaseen, T. Dudler, S. Gragerov, G. Demopulos, J. L. Heeney, and W. J. Schwaeble. "Lectin Pathway Mediates Complement Activation by Sars-Cov-2 Proteins." Front Immunol 12 (2021): 714511.
- Gibson, B. G., T. E. Cox, and K. J. Marchbank. "Contribution of Animal Models to the Mechanistic Understanding of Alternative Pathway and Amplification Loop (Ap/Al)-Driven Complement-Mediated Diseases." Immunol Rev 313, no. 1 (2023): 194-216.
- Brouwer, N., K. M. Dolman, R. van Zwieten, E. Nieuwenhuys, M. Hart, L. A. Aarden, D. Roos, and T. W. Kuijpers. "Mannan-Binding Lectin (Mbl)-Mediated Opsonization Is Enhanced by the Alternative Pathway Amplification Loop." Mol Immunol 43, no. 13 (2006): 2051-60.
- Risso, V., E. Lafont, and M. Le Gallo. "Therapeutic Approaches Targeting Cd95l/Cd95 Signaling in Cancer and Autoimmune Diseases." Cell Death Dis 13, no. 3 (2022): 248.
- Ramirez-Labrada, A., C. Pesini, L. Santiago, S. Hidalgo, A. Calvo-Perez, C. Onate, A. Andres-Tovar, M. Garzon-Tituana, I. Uranga-Murillo, M. A. Arias, E. M. Galvez, and J. Pardo. "All About (Nk Cell-Mediated) Death in Two Acts and an Unexpected Encore: Initiation, Execution and Activation of Adaptive Immunity." Front Immunol 13 (2022): 896228.
- Wiley, S. R., K. Schooley, P. J. Smolak, W. S. Din, C. P. Huang, J. K. Nicholl, G. R. Sutherland, T. D. Smith, C. Rauch, C. A. Smith, and et al. "Identification and Characterization of a New Member of the Tnf Family That Induces Apoptosis." Immunity 3, no. 6 (1995): 673-82.
- Schroder, K., P. J. Hertzog, T. Ravasi, and D. A. Hume. "Interferon-Gamma: An Overview of Signals, Mechanisms and Functions." J Leukoc Biol 75, no. 2 (2004): 163-89.
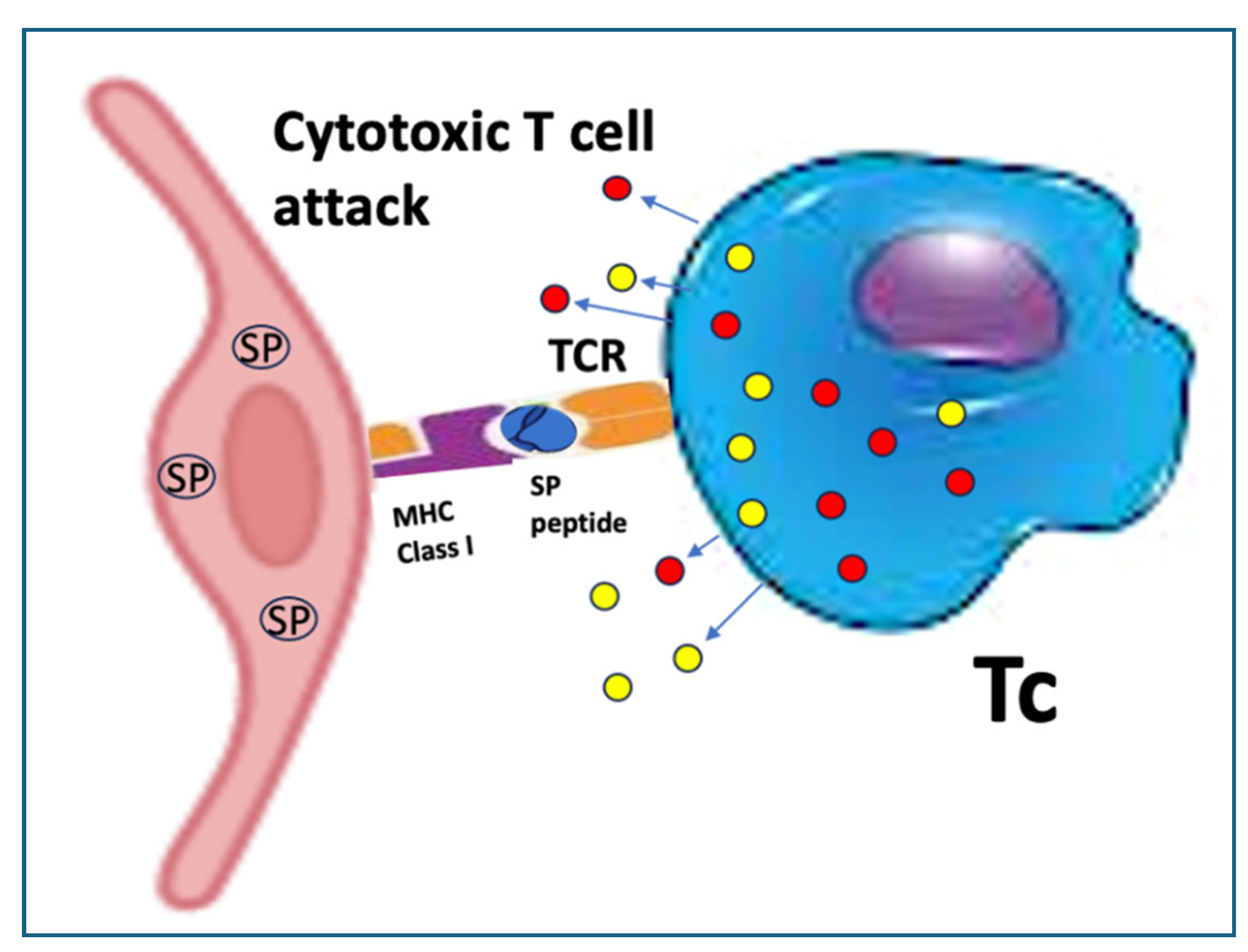
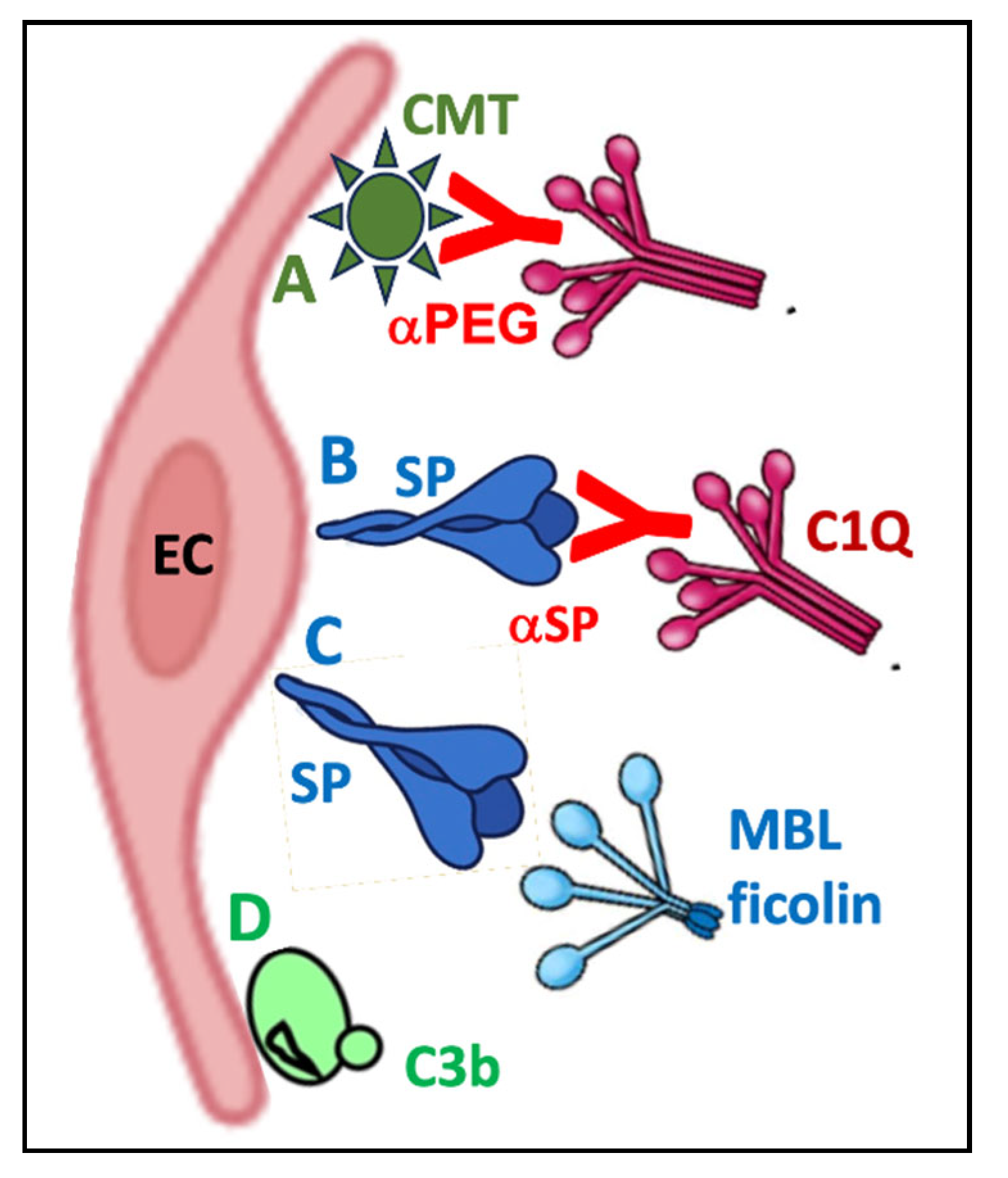
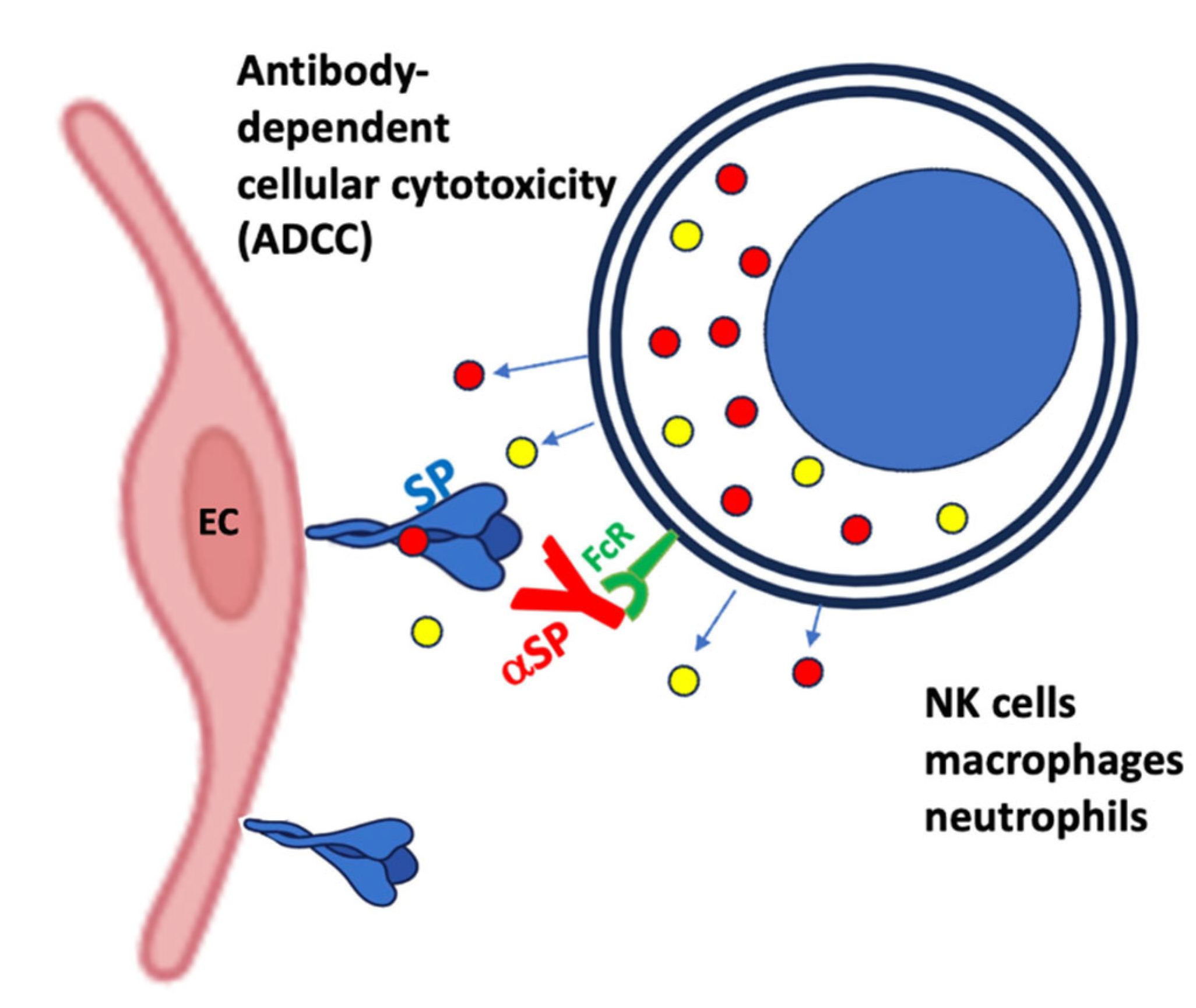
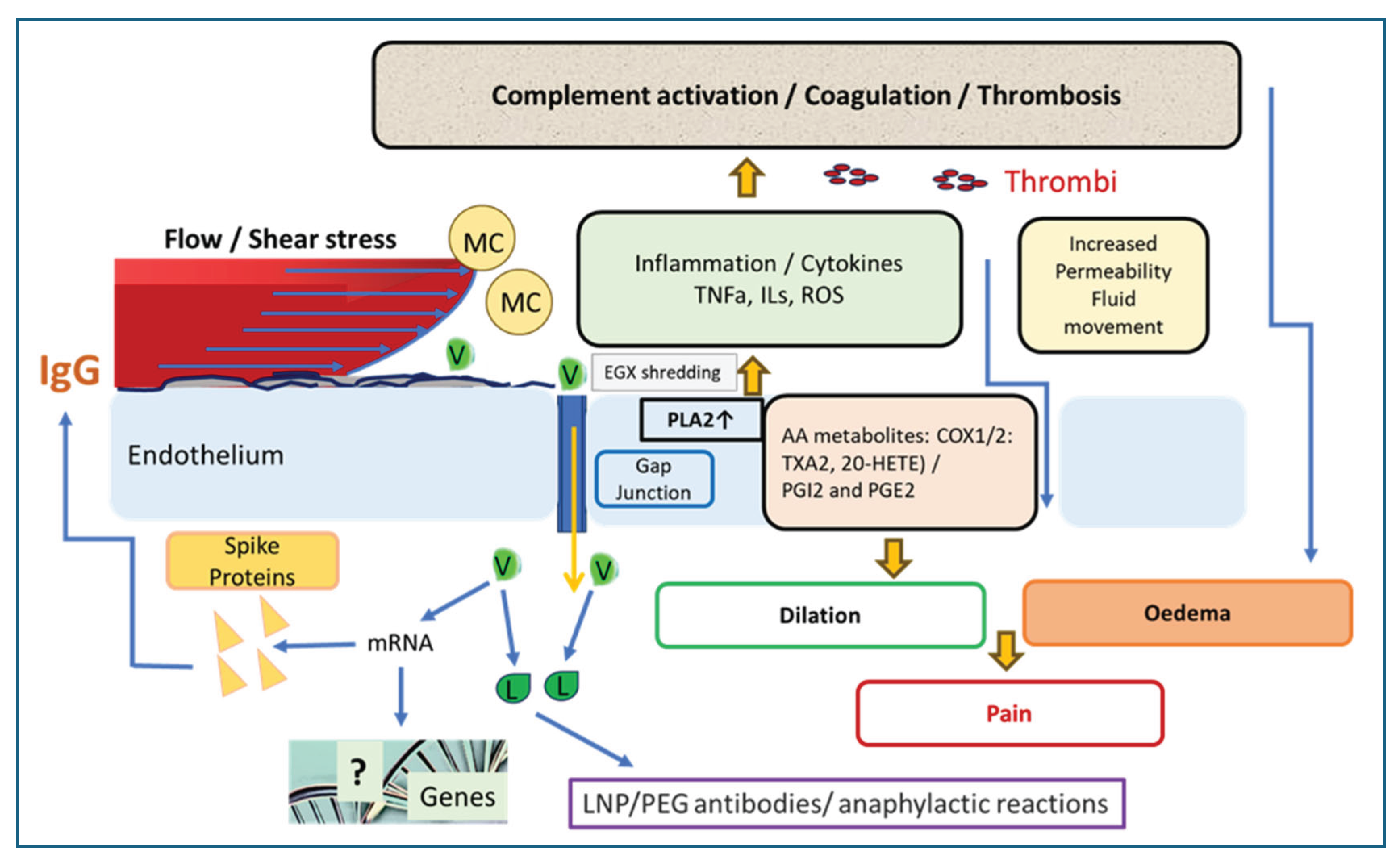
 |
| Strains | References |
| Cytomegalovirus (CMV) | [16, 17] |
| Epstein-Barr virus (EBV) | [18] |
| Herpes simplex virus (HSV) | [19-21] |
| Varicella Zoster virus (VZV), | [22, 23] |
| Vessel Size |
|
|
| Small |
|
|
| Medium |
|
|
| Large |
|
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




