Submitted:
12 December 2024
Posted:
12 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. AMPs from Amphibians with Antioxidant Properties
Antioxidin Family
Pleurain Family
Cathelicidin Family
Spinosan Family
Jindongenin and Palustrin Families
Taipehensin Family
Brevinin Family
Nigroain Family
Andersonin Family
Odorranain Family
Hainanenin Family
FW Family
OM Family
OA Family
Temporin Family
Daiyunin and Pleskein Families
Jindongenin Family
Tryptophilins Family
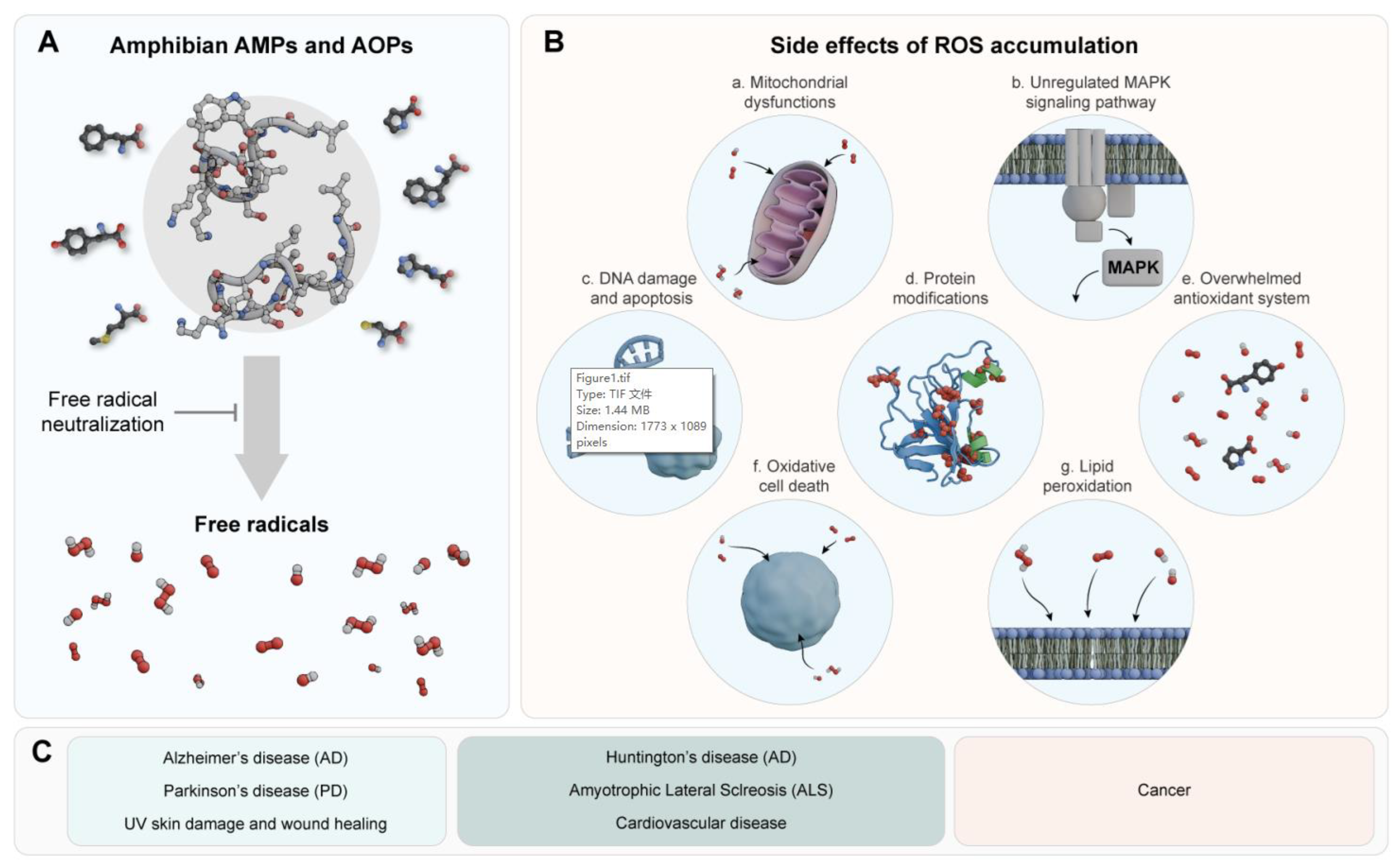
3. Amphibian AOPs and Oxidative Stress-Related Diseases
4. Concluding Remarks and Prospects
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Marenah, L.; Flatt, P.R.; Orr, D.F.; Shaw, C.; Abdel-Wahab, Y.H.A. Skin Secretions of Rana Saharica Frogs Reveal Antimicrobial Peptides Esculentins-1 and -1B and Brevinins-1E and -2EC with Novel Insulin Releasing Activity. Journal of Endocrinology 2006, 188, 1–9. [Google Scholar] [CrossRef]
- Arcanjo, D.D.R.; Vasconcelos, A.G.; Comerma-Steffensen, S.G.; Jesus, J.R.; Silva, L.P.; Pires, O.R.; Costa-Neto, C.M.; Oliveira, E.B.; Migliolo, L.; Franco, O.L.; et al. A Novel Vasoactive Proline-Rich Oligopeptide from the Skin Secretion of the Frog Brachycephalus Ephippium. PLoS One 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Song, Y.; Li, J.; Liu, H.; Xu, X.; Lai, R.; Zhang, K. A New Family of Antimicrobial Peptides from Skin Secretions of Rana Pleuraden. Peptides (N.Y.) 2007, 28, 2069–2074. [Google Scholar] [CrossRef] [PubMed]
- Feng, G.; Wu, J.; Yang, H.-L.; Mu, L. Discovery of Antioxidant Peptides from Amphibians: A Review. Protein Pept Lett 2021, 28, 1220–1229. [Google Scholar] [CrossRef]
- Kirk, J.T.O. Light and Photosynthesis in Aquatic Ecosystems; Cambridge University Press, 2010; ISBN 9780521151757.
- Clarke, B.T. THE NATURAL HISTORY OF AMPHIBIAN SKIN SECRETIONS, THEIR NORMAL FUNCTIONING AND POTENTIAL MEDICAL APPLICATIONS. Biological Reviews 1997, 72, 365–379. [Google Scholar] [CrossRef]
- Geihs, M.A.; Moreira, D.C.; López-Martínez, G.; Minari, M.; Ferreira-Cravo, M.; Carvajalino-Fernández, J.M.; Hermes-Lima, M. Commentary: Ultraviolet Radiation Triggers “Preparation for Oxidative Stress” Antioxidant Response in Animals: Similarities and Interplay with Other Stressors. Comp Biochem Physiol A Mol Integr Physiol 2020, 239. [Google Scholar] [CrossRef]
- Yang, H.; Wang, X.; Liu, X.; Wu, J.; Liu, C.; Gong, W.; Zhao, Z.; Hong, J.; Lin, D.; Wang, Y.; et al. Antioxidant Peptidomics Reveals Novel Skin Antioxidant System. Molecular and Cellular Proteomics 2009, 8, 571–583. [Google Scholar] [CrossRef] [PubMed]
- Chevion, S.; Berry, E.M.; Kitrossky, N.; Kohen, R. Evaluation of Plasma Low Molecular Weight Antioxidant Capacity by Cyclic Voltammetry. Free Radic Biol Med 1997, 22, 411–421. [Google Scholar] [CrossRef]
- Shindo, Y.; Witt, E.; Packer, L. Antioxidant Defense Mechanisms in Murine Epidermis and Dermis and Their Responses to Ultraviolet Light. Journal of Investigative Dermatology 1993, 100, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.; Wang, Y.; Liu, N.; Yang, M.; Hu, Y.; Li, X.; Fu, Y.; Luo, M.; Sun, J.; Yang, X. Potential Skin Protective Effects after UVB Irradiation Afforded by an Antioxidant Peptide from Odorrana Andersonii. Biomedicine and Pharmacotherapy 2019, 120. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, K.; Jia, X.; Fu, C.; Yu, H.; Wang, Y. Antioxidant Peptides, the Guardian of Life from Oxidative Stress. Med Res Rev 2024, 44, 275–364. [Google Scholar] [CrossRef]
- Xie, H.; Hou, S.; Jiang, J.; Sekutowicz, M.; Kelly, J.; Bacskai, B.J. Rapid Cell Death Is Preceded by Amyloid Plaque-Mediated Oxidative Stress. Proc Natl Acad Sci U S A 2013, 110, 7904–7909. [Google Scholar] [CrossRef]
- van der Vliet, A.; Janssen-Heininger, Y.M.W. Hydrogen Peroxide as a Damage Signal in Tissue Injury and Inflammation: Murderer, Mediator, or Messenger? J Cell Biochem 2014, 115, 427–435. [Google Scholar] [CrossRef]
- Di Meo, S.; Reed, T.T.; Venditti, P.; Victor, V.M. Role of ROS and RNS Sources in Physiological and Pathological Conditions. Oxid Med Cell Longev 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Nieborowska-Skorska, M.; Kopinski, P.K.; Ray, R.; Hoser, G.; Ngaba, D.; Flis, S.; Cramer, K.; Reddy, M.M.; Koptyra, M.; Penserga, T.; et al. Rac2-MRC-CIII–Generated ROS Cause Genomic Instability in Chronic Myeloid Leukemia Stem Cells and Primitive Progenitors. Blood 2012, 119, 4253–4263. [Google Scholar] [CrossRef] [PubMed]
- Radisky, D.C.; Levy, D.D.; Littlepage, L.E.; Liu, H.; Nelson, C.M.; Fata, J.E.; Leake, D.; Godden, E.L.; Albertson, D.G.; Nieto, M.A.; et al. Rac1b and Reactive Oxygen Species Mediate MMP-3-Induced EMT and Genomic Instability. Nature 2005, 436, 123–127. [Google Scholar] [CrossRef]
- Deng, L.; Du, C.; Song, P.; Chen, T.; Rui, S.; Armstrong, D.G.; Deng, W. The Role of Oxidative Stress and Antioxidants in Diabetic Wound Healing. Oxid Med Cell Longev 2021, 2021. [Google Scholar] [CrossRef] [PubMed]
- Senoner, T.; Dichtl, W. Oxidative Stress in Cardiovascular Diseases: Still a Therapeutic Target? Nutrients 2019, 11. [Google Scholar] [CrossRef]
- Bourdon, E.; Blache, D. The Importance of Proteins in Defense Against Oxidation. Antioxid Redox Signal 2001, 3, 293–311. [Google Scholar] [CrossRef] [PubMed]
- Hardas, S.S.; Sultana, R.; Clark, A.M.; Beckett, T.L.; Szweda, L.I.; Paul Murphy, M.; Butterfield, D.A. Oxidative Modification of Lipoic Acid by HNE in Alzheimer Disease Brain. Redox Biol 2013, 1, 80–85. [Google Scholar] [CrossRef]
- Hou, L.; Zhou, X.; Zhang, C.; Wang, K.; Liu, X.; Che, Y.; Sun, F.; Li, H.; Wang, Q.; Zhang, D.; et al. NADPH Oxidase-Derived H2O2 Mediates the Regulatory Effects of Microglia on Astrogliosis in Experimental Models of Parkinson’s Disease. Redox Biol 2017, 12, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Berggren, K.L.; Chen, J.; Fox, J.; Miller, J.; Dodds, L.; Dugas, B.; Vargas, L.; Lothian, A.; McAllum, E.; Volitakis, I.; et al. Neonatal Iron Supplementation Potentiates Oxidative Stress, Energetic Dysfunction and Neurodegeneration in the R6/2 Mouse Model of Huntington’s Disease. Redox Biol 2015, 4, 363–374. [Google Scholar] [CrossRef]
- Martins, D.; English, A.M. SOD1 Oxidation and Formation of Soluble Aggregates in Yeast: Relevance to Sporadic ALS Development. Redox Biol 2014, 2, 632–639. [Google Scholar] [CrossRef]
- Dimri, G.P.; Leet, X.; Basile, G.; Acosta, M.; Scorrt, G.; Roskelley, C.; Medrano, E.E.; Linskensi, M.; Rubeljii, I.; Pereira-Smithii, O.; et al. A Biomarker That Identifies Senescent Human Cells in Culture and in Aging Skin in Vivo (Replicative Senescence/Tumor Suppression/18-Galactosidase) Communicated by Arthur; 1995; Vol. 92;
- Millis,’ Marianhoyle, A.J.T.; Mccue, H.M. Differential Expression of Metalloproteinase and Tissue Inhibitor of Metalloproteinase Genes in Aged Human Fibroblasts’, 1992.
- Gu, Y.; Han, J.; Jiang, C.; Zhang, Y. Biomarkers, Oxidative Stress and Autophagy in Skin Aging. Ageing Res Rev 2020, 59, 101036. [Google Scholar] [CrossRef]
- Mookherjee, N.; Anderson, M.A.; Haagsman, H.P.; Davidson, D.J. Antimicrobial Host Defence Peptides: Functions and Clinical Potential. Nat Rev Drug Discov 2020, 19, 311–332. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Dou, X.; Song, J.; Lyu, Y.; Zhu, X.; Xu, L.; Li, W.; Shan, A. Antimicrobial Peptides: Promising Alternatives in the Post Feeding Antibiotic Era. Med Res Rev 2019, 39, 831–859. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Lai, R. The Chemistry and Biological Activities of Peptides from Amphibian Skin Secretions. Chem Rev 2015, 115, 1760–1846. [Google Scholar] [CrossRef]
- Zhang, X.; Feng, C.; Wang, S.; Wang, Y.; Fu, Z.; Zhang, Y.; Sun, H.; Xie, C.; Fu, Y.; Tao, J.; et al. A Novel Amphibian-Derived Peptide Alleviated Ultraviolet B-Induced Photodamage in Mice. Biomedicine and Pharmacotherapy 2021, 136, 111258. [Google Scholar] [CrossRef]
- Guo, C.; Hu, Y.; Li, J.; Liu, Y.; Li, S.; Yan, K.; Wang, X.; Liu, J.; Wang, H. Identification of Multiple Peptides with Antioxidant and Antimicrobial Activities from Skin and Its Secretions of Hylarana Taipehensis, Amolops Lifanensis, and Amolops Granulosus. Biochimie 2014, 105, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Zhai, L.; Wang, H.; Che, Q.; Wang, D.; Feng, F.; Zhao, Z.; Yu, H. Novel Families of Antimicrobial Peptides with Multiple Functions from Skin of Xizang Plateau Frog, Nanorana Parkeri. Biochimie 2010, 92, 475–481. [Google Scholar] [CrossRef]
- Chen, Z.; Yang, X.; Liu, Z.; Zeng, L.; Lee, W.; Zhang, Y. Two Novel Families of Antimicrobial Peptides from Skin Secretions of the Chinese Torrent Frog, Amolops Jingdongensis. Biochimie 2012, 94, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Feng, G.; Wei, L.; Che, H.; Shen, Y.; Yang, J.; Mi, K.; Liu, J.; Wu, J.; Yang, H.; Mu, L. A Frog Peptide Ameliorates Skin Photoaging Through Scavenging Reactive Oxygen Species. Front Pharmacol 2022, 12. [Google Scholar] [CrossRef]
- Sousa, N.A.; Oliveira, G.A.L.; de Oliveira, A.P.; Lopes, A.L.F.; Iles, B.; Nogueira, K.M.; Araújo, T.S.L.; Souza, L.K.M.; Araújo, A.R.; Ramos-Jesus, J.; et al. Novel Ocellatin Peptides Mitigate LPS-Induced ROS Formation and NF-KB Activation in Microglia and Hippocampal Neurons. Sci Rep 2020, 10, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Rong, M.; Liu, J.; Liao, Q.; Lin, Z.; Wen, B.; Ren, Y.; Lai, R. The Defensive System of Tree Frog Skin Identified by Peptidomics and RNA Sequencing Analysis. Amino Acids 2019, 51, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, L.M.; Segundo, M.A.; Reis, S.; Lima, J.L.F.C. Methodological Aspects about in Vitro Evaluation of Antioxidant Properties. Anal Chim Acta 2008, 613, 1–19. [Google Scholar] [CrossRef]
- Jung, H.A.; Jeong, D.-M.; Chung, H.Y.; Lim, H.A.; Kim, J.Y.; Yoon, N.Y.; Choi, J.S. Re-Evaluation of the Antioxidant Prenylated Flavonoids from the Roots of Sophora Flavescens. Biol Pharm Bull 2008, 31, 908–915. [Google Scholar] [CrossRef]
- Xu, X.; Yang, H.; Ma, D.; Wu, J.; Wang, Y.; Song, Y.; Wang, X.; Lu, Y.; Yang, J.; Lai, R. Toward an Understanding of the Molecular Mechanism for Successful Blood Feeding by Coupling Proteomics Analysis with Pharmacological Testing of Horsefly Salivary Glands. Molecular and Cellular Proteomics 2008, 7, 582–590. [Google Scholar] [CrossRef] [PubMed]
- Peskin, A. V.; Winterbourn, C.C. Assay of Superoxide Dismutase Activity in a Plate Assay Using WST-1. Free Radic Biol Med 2017, 103, 188–191. [Google Scholar] [CrossRef]
- Radak, Z.; Zhao, Z.; Goto, S.; Koltai, E. Age-Associated Neurodegeneration and Oxidative Damage to Lipids, Proteins and DNA. Mol Aspects Med 2011, 32, 305–315. [Google Scholar] [CrossRef]
- Pisoschi, A.M.; Pop, A.; Cimpeanu, C.; Predoi, G. Antioxidant Capacity Determination in Plants and Plant-Derived Products: A Review. Oxid Med Cell Longev 2016, 2016. [Google Scholar] [CrossRef]
- Yu, H.; Qiao, X.; Gao, J.; Wang, C.; Cai, S.; Feng, L.; Wang, H.; Wang, Y.-P. Send Orders for Reprints to Reprints@benthamscience.Ae Identification and Characterization of Novel Antioxidant Peptides Involved in Redox Homeostasis of Frog: Limnonectes Fragilis; 2015; Vol. 22.
- Qian, Z.J.; Jung, W.K.; Kim, S.K. Free Radical Scavenging Activity of a Novel Antioxidative Peptide Purified from Hydrolysate of Bullfrog Skin, Rana Catesbeiana Shaw. Bioresour Technol 2008, 99, 1690–1698. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, E.A.; Plácido, A.; Moreira, D.C.; Albuquerque, L.; Dematei, A.; Silva-Carvalho, A.; Cabral, W.F.; Báo, S.N.; Saldanha-Araújo, F.; Kuckelhaus, S.A.S.; et al. The Peptide Secreted at the Water to Land Transition in a Model Amphibian Has Antioxidant Effects. Proceedings of the Royal Society B: Biological Sciences 2021, 288. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, E.A.; Oliveira, A.; Plácido, A.; Socodato, R.; Portugal, C.C.; Mafud, A.C.; Ombredane, A.S.; Moreira, D.C.; Vale, N.; Bessa, L.J.; et al. Structure and Function of a Novel Antioxidant Peptide from the Skin of Tropical Frogs. Free Radic Biol Med 2018, 115, 68–79. [Google Scholar] [CrossRef] [PubMed]
- Åkerström, B.; Maghzal, G.J.; Winterbourn, C.C.; Kettle, A.J. The Lipocalin A1-Microglobulin Has Radical Scavenging Activity. Journal of Biological Chemistry 2007, 282, 31493–31503. [Google Scholar] [CrossRef] [PubMed]
- Wen, C.; Zhang, J.; Zhang, H.; Duan, Y.; Ma, H. Plant Protein-Derived Antioxidant Peptides: Isolation, Identification, Mechanism of Action and Application in Food Systems: A Review. Trends Food Sci Technol 2020, 105, 308–322. [Google Scholar] [CrossRef]
- Von Heijne, G. MEMBRANE PROTEINS: From Sequence to Structure, PROTEINS, 1994.
- Mosmann, T. Rapid Colorimetric Assay for Cellular Growth and Survival: Application to Proliferation and Cytotoxicity Assays. J Immunol Methods 1983, 65, 55–63. [Google Scholar] [CrossRef] [PubMed]
- López-García, G.; Dublan-García, O.; Arizmendi-Cotero, D.; Gómez Oliván, L.M. Antioxidant and Antimicrobial Peptides Derived from Food Proteins. Molecules 2022, 27, 1343. [Google Scholar] [CrossRef]
- Zhang, S.; Guo, H.; Shi, F.; Wang, H.; Li, L.; Jiao, X.; Wang, Y.; Yu, H. Hainanenins: A Novel Family of Antimicrobial Peptides with Strong Activity from Hainan Cascade-Frog, Amolops Hainanensis. Peptides (N.Y.) 2012, 33, 251–257. [Google Scholar] [CrossRef]
- Yang, X.; Wang, Y.; Zhang, Y.; Lee, W.H.; Zhang, Y. Rich Diversity and Potency of Skin Antioxidant Peptides Revealed a Novel Molecular Basis for High-Altitude Adaptation of Amphibians. Sci Rep 2016, 6, 1–11. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a Free Radical Method to Evaluate Antioxidant Activity; 1995; Vol. 28.
- He, W.; Feng, F.; Huang, Y.; Guo, H.; Zhang, S.; Li, Z.; Liu, J.; Wang, Y.; Yu, H. Host Defense Peptides in Skin Secretions of Odorrana Tiannanensis: Proof for Other Survival Strategy of the Frog than Merely Anti-Microbial. Biochimie 2012, 94, 649–655. [Google Scholar] [CrossRef]
- Yang, X.; Lee, W.H.; Zhang, Y. Extremely Abundant Antimicrobial Peptides Existed in the Skins of Nine Kinds of Chinese Odorous Frogs. J Proteome Res 2012, 11, 306–319. [Google Scholar] [CrossRef]
- Chen, T.; Orr, D.F.; O’Rourke, M.; McLynn, C.; Bjourson, A.J.; McClean, S.; Hirst, D.; Rao, P.; Shaw, C. Pachymedusa Dacnicolor Tryptophyllin-1: Structural Characterization, Pharmacological Activity and Cloning of Precursor CDNA. Regul Pept 2004, 117, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Ellis-Steinborner, S.T.; Scanlon, D.; Musgrave, I.F.; Tran, T.T.N.; Hack, S.; Wang, T.; Abell, A.D.; Tyler, M.J.; Bowie, J.H. An Unusual Kynurenine-Containing Opioid Tetrapeptide from the Skin Gland Secretion of the Australian Red Tree Frog Litoria Rubella. Sequence Determination by Electrospray Mass Spectrometry. Rapid Communications in Mass Spectrometry 2011, 25, 1735–1740. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Chen, T.; Zhou, M.; Wang, L.; Shaw, C. PsT-1: A New Tryptophyllin Peptide from the Skin Secretion of Waxy Monkey Leaf Frog, Phyllomedusa Sauvagei. Regul Pept 2013, 184, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Zasloff, M. Magainins, a Class of Antimicrobial Peptides from Xenopus Skin: Isolation, Characterization of Two Active Forms, and Partial CDNA Sequence of a Precursor (Vertebrate Peptide Antibiotics); 1987; Vol. 84.
- Liu, C.; Hong, J.; Yang, H.; Wu, J.; Ma, D.; Li, D.; Lin, D.; Lai, R. Frog Skins Keep Redox Homeostasis by Antioxidant Peptides with Rapid Radical Scavenging Ability. Free Radic Biol Med 2010, 48, 1173–1181. [Google Scholar] [CrossRef] [PubMed]
- Qin, D.; Lee, W.H.; Gao, Z.; Zhang, W.; Peng, M.; Sun, T.; Gao, Y. Protective Effects of Antioxidin-RL from Odorrana Livida against Ultraviolet B-Irradiated Skin Photoaging. Peptides (N.Y.) 2018, 101, 124–134. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhou, Y.; Yang, G.Y.; Li, S. Biomimetic Peptides Protect Cells from Oxidative Stress. Am J Transl Res 2017, 9, 5518–5527. [Google Scholar]
- Song, Y.; Ji, S.; Liu, W.; Yu, X.; Meng, Q.; Lai, R. Different Expression Profiles of Bioactive Peptides in Pelophylax Nigromaculatus from Distinct Regions. Biosci Biotechnol Biochem 2013, 77, 1075–1079. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, N.; Dickman, M.B.; Becker, D.F. Proline Modulates the Intracellular Redox Environment and Protects Mammalian Cells against Oxidative Stress. Free Radic Biol Med 2008, 44, 671–681. [Google Scholar] [CrossRef] [PubMed]
- Kościuczuk, E.M.; Lisowski, P.; Jarczak, J.; Strzałkowska, N.; Jóźwik, A.; Horbańczuk, J.; Krzyżewski, J.; Zwierzchowski, L.; Bagnicka, E. Cathelicidins: Family of Antimicrobial Peptides. A Review. Mol Biol Rep 2012, 39, 10957–10970. [Google Scholar] [CrossRef]
- Feng, G.; Wei, L.; Che, H.; Shen, Y.; Mi, K.; Bian, H.; Yang, H.; Wu, J.; Mu, L. Cathelicidin-NV from Nanorana Ventripunctata Effectively Protects HaCaT Cells, Ameliorating Ultraviolet B-Induced Skin Photoaging. Peptides (N.Y.) 2022, 150. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Wang, Y.; Wu, C.; Li, X.; Fu, Z.; Yang, M.; Bian, W.; Wang, S.; Song, Y.; Tang, J.; et al. Cathelicidin-OA1, a Novel Antioxidant Peptide Identified from an Amphibian, Accelerates Skin Wound Healing. Sci Rep 2018, 8. [Google Scholar] [CrossRef]
- Mu, L.; Tang, J.; Liu, H.; Shen, C.; Rong, M.; Zhang, Z.; Lai, R. A Potential Wound-Healing-Promoting Peptide from Salamander Skin. FASEB Journal 2014, 28, 3919–3929. [Google Scholar] [CrossRef]
- Lu, H.; Chai, J.; Xu, Z.; Wu, J.; He, S.; Liao, H.; Huang, P.; Huang, X.; Chen, X.; Jiang, H.; et al. Cath-KP, a Novel Peptide Derived from Frog Skin, Prevents Oxidative Stress Damage in a Parkinson’s Disease Model. Zool Res 2024, 45, 108–124. [Google Scholar] [CrossRef] [PubMed]
- Dong, B.J.; Zhan, Z.G.; Zheng, R.Q.; Chen, W.; Min, J.J. CDNA Cloning and Functional Characterisation of Four Antimicrobial Peptides from Paa Spinosa. Zeitschrift fur Naturforschung - Section C Journal of Biosciences 2015, 70, 251–256. [Google Scholar] [CrossRef]
- Chai, J.; Liu, J.; Tian, M.; Liao, H.; Wu, J.; Xie, J.; Lai, S.; Mo, G.; Chen, X.; Xu, X. Multiple Mechanistic Action of Brevinin-1FL Peptide against Oxidative Stress Effects in an Acute Inflammatory Model of Carrageenan-Induced Damage. Oxid Med Cell Longev 2022, 2022. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ren, S.; Guo, C.; Zhang, W.; Zhang, X.; Zhang, B.; Li, S.; Ren, J.; Hu, Y.; Wang, H. Identification and Functional Analyses of Novel Antioxidant Peptides and Antimicrobial Peptides from Skin Secretions of Four East Asian Frog Species. Acta Biochim Biophys Sin (Shanghai) 2017, 49, 550–559. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Guo, X.; Yi, T.; Zhu, Y.; Ren, X.; Guo, R.; Dai, Y.; Liang, S. Frog Skin Derived Peptides With Potential Protective Effects on Ultraviolet B–Induced Cutaneous Photodamage. Front Immunol 2021, 12. [Google Scholar] [CrossRef]
- Li, X.; Wang, Y.; Zou, Z.; Yang, M.; Wu, C.; Su, Y.; Tang, J.; Yang, X. OM-LV20, a Novel Peptide from Odorous Frog Skin, Accelerates Wound Healing in Vitro and in Vivo. Chem Biol Drug Des 2018, 91, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Cao, X.; Fu, Z.; Wang, S.; Li, X.; Liu, N.; Feng, Z.; Yang, M.; Tang, J.; Yang, X. Identification and Characterization of a Novel Gene-Encoded Antioxidant Peptide Obtained from Amphibian Skin Secretions. Nat Prod Res 2020, 34, 754–758. [Google Scholar] [CrossRef]
- Bian, W.; Meng, B.; Li, X.; Wang, S.; Cao, X.; Liu, N.; Yang, M.; Tang, J.; Wang, Y.; Yang, X. OA-GL21, a Novel Bioactive Peptide from Odorrana Andersonii, Accelerated the Healing of Skin Wounds. Biosci Rep 2018, 38. [Google Scholar] [CrossRef] [PubMed]
- Ge, L.; Lyu, P.; Zhou, M.; Zhang, H.; Wan, Y.; Li, B.; Li, R.; Wang, L.; Chen, T.; Shaw, C. AcT-2: A Novel Myotropic and Antimicrobial Type 2 Tryptophyllin from the Skin Secretion of the Central American Red-Eyed Leaf Frog, Agalychnis Callidryas. The Scientific World Journal 2014, 2014. [Google Scholar] [CrossRef]
- Plácido, A.; do Pais do Amaral, C.; Teixeira, C.; Nogueira, A.; Brango-Vanegas, J.; Alves Barbosa, E.; C. Moreira, D.; Silva-Carvalho, A.; da Silva, M. da G.; do Nascimento Dias, J.; et al. Neuroprotective Effects on Microglia and Insights into the Structure–Activity Relationship of an Antioxidant Peptide Isolated from Pelophylax Perezi. J Cell Mol Med 2022, 26, 2793–2807. [Google Scholar] [CrossRef]
- Tossi, A.; Sandri, L.; Giangaspero, A. Amphipathic,-Helical Antimicrobial Peptides; 2000; Vol. 55.
- Chaudhary, M.R.; Chaudhary, S.; Sharma, Y.; Singh, T.A.; Mishra, A.K.; Sharma, S.; Mehdi, M.M. Aging, Oxidative Stress and Degenerative Diseases: Mechanisms, Complications and Emerging Therapeutic Strategies. Biogerontology 2023, 24, 609–662. [Google Scholar] [CrossRef] [PubMed]
- Schieber, M.; Chandel, N.S. ROS Function in Redox Signaling and Oxidative Stress. Current Biology 2014, 24. [Google Scholar] [CrossRef] [PubMed]
- Kowalczyk, P.; Sulejczak, D.; Kleczkowska, P.; Bukowska-Ośko, I.; Kucia, M.; Popiel, M.; Wietrak, E.; Kramkowski, K.; Wrzosek, K.; Kaczyńska, K. Mitochondrial Oxidative Stress—a Causative Factor and Therapeutic Target in Many Diseases. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef] [PubMed]
- Bai, R.; Guo, J.; Ye, X.Y.; Xie, Y.; Xie, T. Oxidative Stress: The Core Pathogenesis and Mechanism of Alzheimer’s Disease. Ageing Res Rev 2022, 77. [Google Scholar] [CrossRef]
- Arfin, S.; Jha, N.K.; Jha, S.K.; Kesari, K.K.; Ruokolainen, J.; Roychoudhury, S.; Rathi, B.; Kumar, D. Oxidative Stress in Cancer Cell Metabolism. Antioxidants 2021, 10. [Google Scholar] [CrossRef]
- Dhapola, R.; Beura, S.K.; Sharma, P.; Singh, S.K.; HariKrishnaReddy, D. Oxidative Stress in Alzheimer’s Disease: Current Knowledge of Signaling Pathways and Therapeutics. Mol Biol Rep 2024, 51. [Google Scholar] [CrossRef] [PubMed]
- Gao, C.; Jiang, J.; Tan, Y.; Chen, S. Microglia in Neurodegenerative Diseases: Mechanism and Potential Therapeutic Targets. Signal Transduct Target Ther 2023, 8. [Google Scholar] [CrossRef] [PubMed]
- Dash, U.C.; Bhol, N.K.; Swain, S.K.; Samal, R.R.; Nayak, P.K.; Raina, V.; Panda, S.K.; Kerry, R.G.; Duttaroy, A.K.; Jena, A.B. Oxidative Stress and Inflammation in the Pathogenesis of Neurological Disorders: Mechanisms and Implications. Acta Pharm Sin B 2024. [CrossRef]
- Marucci, G.; Buccioni, M.; Ben, D.D.; Lambertucci, C.; Volpini, R.; Amenta, F. Efficacy of Acetylcholinesterase Inhibitors in Alzheimer’s Disease. Neuropharmacology 2021, 190. [Google Scholar] [CrossRef] [PubMed]
- Spinelli, R.; Aimaretti, F.M.; López, J.A.; Siano, A.S. Amphibian Skin Extracts as Source of Bioactive Multi-Target Agents against Different Pathways of Alzheimer’s Disease. Nat Prod Res 2021, 35, 686–689. [Google Scholar] [CrossRef]
- Spinelli, R.; Humpola, M.V.; Sanchís, I.; de los Angeles Méndez, E.; Siano, A. Biological Characterization of Natural Peptide BcI-1003 from Boana Cordobae (Anura): Role in Alzheimer’s Disease and Microbial Infections. Int J Pept Res Ther 2023, 29. [Google Scholar] [CrossRef]
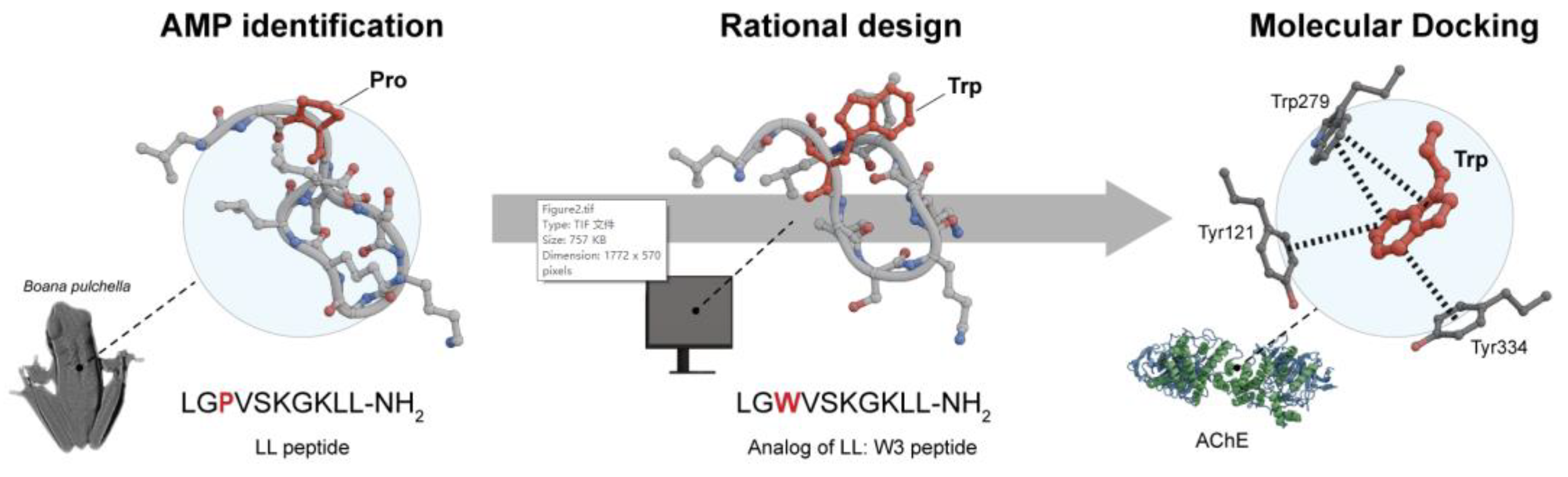
- Sanchis, I.; Spinelli, R.; Aschemacher, N.; Humpola, M.V.; Siano, A. Acetylcholinesterase Inhibitory Activity of a Naturally Occurring Peptide Isolated from Boana Pulchella (Anura: Hylidae) and Its Analogs. Amino Acids 2020, 52, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Sanchis, I.; Spinelli, R.; Aschemacher, N.; Siano, A.S. Rational Design and Synthesis of Modified Natural Peptides from Boana Pulchella (Anura) as Acetylcholinesterase Inhibitors and Antioxidants. Amino Acids 2022, 54, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Jenner, P.; Olanow, C.W. Oxidative Stress and the Pathogenesis of Parkinson’s Disease. Neurology 1996, 47. [Google Scholar] [CrossRef]
- Dias, V.; Junn, E.; Mouradian, M.M. The Role of Oxidative Stress in Parkinson’s Disease. J Parkinsons Dis 2013, 3, 461–491. [Google Scholar] [CrossRef] [PubMed]
- Dorszewska, J.; Kowalska, M.; Prendecki, M.; Piekut, T.; Kozłowska, J.; Kozubski, W. Oxidative Stress Factors in Parkinson’s Disease. Neural Regen Res 2021, 16, 1383–1391. [Google Scholar] [CrossRef]
- Orfali, R.; Alwatban, A.Z.; Orfali, R.S.; Lau, L.; Chea, N.; Alotaibi, A.M.; Nam, Y.W.; Zhang, M. Oxidative Stress and Ion Channels in Neurodegenerative Diseases. Front Physiol 2024, 15. [Google Scholar] [CrossRef]
- Krueger, J.S.; Keshamouni, V.G.; Atanaskova, N.; Reddy, K.B. Temporal and Quantitative Regulation of Mitogen-Activated Protein Kinase (MAPK) Modulates Cell Motility and Invasion. Oncogene 2001, 20, 4209–4218. [Google Scholar] [CrossRef]
- Singh, R.; Letai, A.; Sarosiek, K. Regulation of Apoptosis in Health and Disease: The Balancing Act of BCL-2 Family Proteins. Nat Rev Mol Cell Biol 2019, 20, 175–193. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Wu, J.; Li, X.; Ye, Z.; Yang, H.; Mu, L. Amphibian-Derived Natural Anticancer Peptides and Proteins: Mechanism of Action, Application Strategies, and Prospects. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef]
- Glorieux, C.; Liu, S.; Trachootham, D.; Huang, P. Targeting ROS in Cancer: Rationale and Strategies. Nat Rev Drug Discov 2024, 23, 583–606. [Google Scholar] [CrossRef]
- Rezatabar, S.; Karimian, A.; Rameshknia, V.; Parsian, H.; Majidinia, M.; Kopi, T.A.; Bishayee, A.; Sadeghinia, A.; Yousefi, M.; Monirialamdari, M.; et al. RAS/MAPK Signaling Functions in Oxidative Stress, DNA Damage Response and Cancer Progression. J Cell Physiol 2019, 234, 14951–14965. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.D.; Dinkova-Kostova, A.T.; Tew, K.D. Oxidative Stress in Cancer. Cancer Cell 2020, 38, 167–197. [Google Scholar] [CrossRef] [PubMed]
- Najafi, M.; Goradel, N.H.; Farhood, B.; Salehi, E.; Solhjoo, S.; Toolee, H.; Kharazinejad, E.; Mortezaee, K. Tumor Microenvironment: Interactions and Therapy. J Cell Physiol 2019, 234, 5700–5721. [Google Scholar] [CrossRef]
- Acevedo-León, D.; Monzó-Beltrán, L.; Pérez-Sánchez, L.; Naranjo-Morillo, E.; Gómez-Abril, S.Á.; Estañ-Capell, N.; Bañuls, C.; Sáez, G. Oxidative Stress and DNA Damage Markers in Colorectal Cancer. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef]
- Mahalingaiah, P.K.S.; Singh, K.P. Chronic Oxidative Stress Increases Growth and Tumorigenic Potential of MCF-7 Breast Cancer Cells. PLoS One 2014, 9. [Google Scholar] [CrossRef]
- Luo, M.; Zhou, L.; Huang, Z.; Li, B.; Nice, E.C.; Xu, J.; Huang, C. Antioxidant Therapy in Cancer: Rationale and Progress. Antioxidants 2022, 11. [Google Scholar] [CrossRef]
- Schalka, S.; Silva, M.S.; Lopes, L.F.; de Freitas, L.M.; Baptista, M.S. The Skin Redoxome. Journal of the European Academy of Dermatology and Venereology 2022, 36, 181–195. [Google Scholar] [CrossRef] [PubMed]
- Emanuelli, M.; Sartini, D.; Molinelli, E.; Campagna, R.; Pozzi, V.; Salvolini, E.; Simonetti, O.; Campanati, A.; Offidani, A. The Double-Edged Sword of Oxidative Stress in Skin Damage and Melanoma: From Physiopathology to Therapeutical Approaches. Antioxidants 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Yang, T.; Yu, D.; Xiong, H.; Zhang, S. Current Insights and Future Perspectives of Ultraviolet Radiation (UV) Exposure: Friends and Foes to the Skin and beyond the Skin. Environ Int 2024, 185. [Google Scholar] [CrossRef]
- Wang, S.; Yang, M.; Yin, S.; Zhang, Y.; Zhang, Y.; Sun, H.; Shu, L.; Liu, Y.; Kang, Z.; Liu, N.; et al. A New Peptide Originated from Amphibian Skin Alleviates the Ultraviolet B-Induced Skin Photodamage. Biomedicine and Pharmacotherapy 2022, 150. [Google Scholar] [CrossRef]
- Xie, C.; Fan, Y.; Yin, S.; Li, Y.; Liu, N.; Liu, Y.; Shu, L.; Fu, Z.; Wang, Y.; Zhang, Y.; et al. Novel Amphibian-Derived Antioxidant Peptide Protects Skin against Ultraviolet Irradiation Damage. J Photochem Photobiol B 2021, 224. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.; Li, S.; Bian, W.; Yang, M.; Liu, N.; Hu, Y.; Li, X.; Wang, Y.; Li, Z.; Sun, J.; et al. Antioxidant Peptide AOP-P1 Derived from Odorous Frog Showed Protective Effects Against UVB-Induced Skin Damages. Int J Pept Res Ther 2020, 26, 557–565. [Google Scholar] [CrossRef]
- Yin, S.; Wang, Y.; Yang, X. Amphibian-Derived Wound Healing Peptides: Chemical Molecular Treasure Trove for Skin Wound Treatment. Front Pharmacol 2023, 14. [Google Scholar] [CrossRef]
- Lammi, C.; Aiello, G.; Boschin, G.; Arnoldi, A. Multifunctional Peptides for the Prevention of Cardiovascular Disease: A New Concept in the Area of Bioactive Food-Derived Peptides. J Funct Foods 2019, 55, 135–145. [Google Scholar] [CrossRef]
- Kim, M.J.; Chilakala, R.; Jo, H.G.; Lee, S.J.; Lee, D.S.; Cheong, S.H. Anti-Obesity and Anti-Hyperglycemic Effects of Meretrix Lusoria Protamex Hydrolysate in Ob/Ob Mice. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



