1. Introduction
Atrial fibrillation (AF) is the most prevalent cardiac arrhythmia globally, particularly among the elderly. [
1] Furthermore, AF increases the risk of stroke, thromboembolic events, heart failure, and mortality. [
2] A challenge with AF is that it often presents with no symptoms or very vague ones, leading to late detection. [
2]
But what exactly is AF and how does it occur? AF is a supraventricular tachyarrhythmia with uncoordinated atrial electrical activation and consequently ineffective atrial contraction and the electrocardiographic characteristics of AF are well defined. [
3] If the atrioventricular (AV) conduction system is intact, this will usually cause a fast and irregular heart rate (irregular tachycardia). This arrhythmia however can also result in slow heart rhythm (irregular bradycardia). The genesis of AF requires a vulnerable atrial substrate as well as a trigger, which usually originated from pulmonary veins. [
4] The vulnerable substrate is characterised by structural, architectural and electrophysiological abnormalities and can have a large spectrum of origins. The probably most common trigger for AF is ectopic activity localised in the pulmonary veins, which have unique electrical properties and complex fibre architecture. [
5] Another important trigger for atrial fibrillation is inflammation [
6] and that is the reason why AF is not uncommon after cardiac surgery.
AF is not just an important issue in internal medicine. In cardiac surgery, postoperative atrial fibrillation (POAF) is the most common complication following surgical procedures, with a frequency of 20 to 30 %. [
7] Previously, POAF was believed to be a transitory, self-resolving condition. However, modern understanding associates it with complications such as ischaemic stroke, heart failure, prolonged hospitalisation and death. [
8,
9] Similar to common AF, POAF often goes unnoticed and tends to recur. [
10,
11] Therefore, there are many different considerations and approaches on how to avoid these complications. For example, the left atrial appendage can be amputated during surgery to prevent thrombus development even before POAF occurs. [
12] Nevertheless, prompt and efficient detection is crucial to manage POAF effectively and prevent potential serious complications. However, intensive monitoring is still expensive and time-consuming, and continuous monitoring over longer time is not practical.
As science advances, so does technology, particularly in the area of e-health and wearable technologies such as fitness trackers and smartwatches. These wearables are becoming increasingly popular and consumers have a growing variety of options available to them. One of the most important methods of measuring used by smartwatches is photoplethysmography (PPG) for pulse rate measurement. A measuring technique that has been known for many decades and that is still being researched. One of the latest research subjects is the detection of atrial fibrillation by wearables using PPG.
The first versions of these devices have already been extensively evaluated, but newer and subsequent models still need to be further analysed. One such device that has not been thoroughly examined is the Scanwatch by Withings. While some studies have assessed its ability to detect AF, [
13,
14] to our knowledge, none have explored its use of a PPG sensor for this purpose. Therefore, we undertook a study to assess the reliability and feasibility of the Scanwatch in detecting POAF.
2. Methods
2.1. Study Design and Objective
We conducted a comprehensive prospective observational study at the Leipzig Heart Centre, aiming to evaluate the effectiveness and reliability of a commercially available smartwatch in detecting atrial fibrillation in patients who had recently undergone cardiac surgery. The study was meticulously designed to include a diverse cohort of participants, each of whom provided informed consent prior to their involvement. This research was carefully reviewed and received formal approval from the Ethics Committee of the University of Leipzig, ensuring that all ethical considerations were addressed. We affirm that every aspect of the study, from the initial recruitment of participants to the final data analysis, was performed in strict accordance with the established guidelines and regulations governing clinical research. These rigorous procedures were implemented to maintain the highest standards of scientific integrity and to ensure the accuracy and validity of our findings.
2.2. Participants
Eligible participants for this study were individuals aged 18 years and older who had recently undergone cardiac surgery. To be considered for inclusion, participants needed to have an electrocardiogram (ECG) indicating sinus rhythm prior to their surgery, ensuring that their heart rhythm was normal at the baseline. Additionally, they were required to be continuously monitored by telemetry throughout the study period to accurately capture any changes in heart rhythm that might occur postoperatively. We implemented strict exclusion criteria to maintain the integrity of the study. Patients with a documented history of atrial fibrillation were excluded, as were those who had implanted cardiac devices such as pacemakers or defibrillators, which could interfere with the detection of atrial fibrillation by the smartwatch. Furthermore, individuals with cognitive impairments were also excluded to ensure that participants could fully understand and comply with the study procedures. This careful selection process was designed to create a well-defined participant pool, enabling us to accurately assess the smartwatch's ability to detect new-onset atrial fibrillation in a postoperative setting.
2.3. AF Detection Methodology: Smartwatch versus Telemetry
Smartwatch: After being informed and providing consent, patients were equipped with a Scanwatch provided by Withings. Patients had the flexibility to wear the watch on either arm, typically choosing an arm without obstructions or based on personal comfort. They were advised to ensure continuous skin contact for accurate readings. If patients had to leave the ward, they could remove the watch since telemetry monitoring wasn't active during such instances. Patients were instructed to wear the watch for 24 hours without interruption if possible. We decided on this time period because some patients were only monitored by telemetry for 24 hours after being discharged from the Intensive Care Unit (ICU) or Intermediate Care (IMC). The Scanwatch recorded the heart rate every 10 minutes using a PPG sensor and scanned the data for AF indicators. PPG is a non-invasive optical measurement technique widely used in medical and wearable technologies to assess various physiological parameters. The method works by emitting light of a specific wavelength, usually in the visible or near-infrared spectrum, into the tissues, typically the skin. As the light penetrates the tissue, it interacts with various components, primarily blood. A photosensor, positioned either adjacent to or in the same device as the light source, detects the light that is either reflected off or transmitted through the tissue.
The information captured by the photosensor is processed to generate a photoplethysmogram, a signal that displays a characteristic waveform. [
15] This waveform fluctuates in response to the changes in blood volume within the tissue during each cardiac cycle. During systole, when the heart contracts and blood is pumped through the arteries, the tissue's blood volume increases, leading to a peak in the PPG waveform. Conversely, during diastole, when the heart relaxes and the blood volume decreases, the waveform reaches a trough. The resulting pattern can be analysed to draw conclusions about the peripheral pulse, which provides insights into cardiovascular health. Photoplethysmography is particularly valuable due to its simplicity, low cost, and ability to provide real-time monitoring. This technique is now widely integrated into wearable devices, such as smartwatches and fitness trackers, to continuously monitor heart rate, blood oxygen saturation, and other vital signs. It can also be used to detect irregularities in heart rhythms, such as atrial fibrillation, by analysing the variability and consistency of the pulse wave over time. Because of its convenience and effectiveness, PPG has become a vital tool in both clinical settings and remote health monitoring, offering significant benefits for early detection and management of cardiovascular conditions. This data was then synced and stored using Withings' Health Mate App. The smartwatch's PPG-recordings and analyses were not visible on the smartwatch itself or in the Health Mate App. The app only gave information about the patients' heart rate while wearing the smartwatch. As soon as the app registered 10 suspicious measurements within 24 hours, a notification appeared on the connected smartphone.
Figure 1 shows the appearance of a Withings Scanwatch we used.
Telemetry: Telemetry monitoring utilized a 5-lead ECG. Five electrodes were appropriately positioned on the patient's torso. The telemetry system captured data from various leads, including leads I-III, aVL, aVR, aVF, and a chest wall lead V. Notably, the electrode for the chest wall lead V was slightly repositioned to the left due to the dressing of the sternotomy wound. Typically, leads II and V were monitored with heart rate alarm thresholds set to <50/min and >135/min. However, medical staff had the discretion to adjust these limits. In comparison to the smartwatch, monitoring via telemetry was performed continuously. Any occurrences of tachyarrhythmia or AF were documented by the attending physicians or nurses, and we sourced our data from these records. The physicians themselves decided whether they wanted or not to confirm the diagnosis of atrial fibrillation with a 12-lead ECG or long-term ECG.
2.4. Technical data of PPG in smartwatches
In the following, we would like to provide an overview of the technical features of the scan watch used in the study and probably the most popular smartwatch on the market - the Apple Watch. The Scanwatch's PPG system utilizes both red and infrared LEDs, along with photodiodes, to detect the amount of light that is reflected back from the skin. When the heart beats, the volume of blood in the vessels increases, absorbing more light and reducing the amount that is reflected. Between heartbeats, the blood volume decreases, allowing more light to be reflected. The photodiodes capture these variations in light reflectance, and the data is processed through algorithms to determine the heart rate. [
15] The use of red and infrared LEDs offers several advantages. These wavelengths penetrate deeper into the skin, making the Scanwatch effective for users with a wide range of skin tones and under various conditions, including during physical activity. The infrared component, in particular, is less prone to interference from ambient light, ensuring more consistent readings. The PPG technology in the Scanwatch is complemented by advanced algorithms that analyze the captured data. These algorithms filter out noise and motion artifacts, which is crucial for maintaining accuracy during activities or in environments with variable lighting. This ensures that the heart rate readings are reliable and reflective of the user's actual physiological state. Beyond heart rate monitoring, the Scanwatch's PPG sensor plays a key role in detecting irregular heart rhythms, such as atrial fibrillation. By continuously monitoring the heart's rhythm, the device can alert users to potential irregularities, prompting further medical evaluation if necessary. [
16] The Scanwatch also incorporates peripheral oxygen saturation (SpO2) measurement, using the same PPG sensors to assess blood oxygen levels. This function is particularly useful for identifying potential issues related to respiratory function and overall cardiovascular health. The design of the Scanwatch's PPG system reflects a balance between technological sophistication and user convenience. Its non-invasive nature, combined with the ability to provide continuous monitoring, makes it a powerful tool for health tracking in daily life. Whether for fitness enthusiasts looking to optimize their training or individuals monitoring their cardiovascular health, the Scanwatch's PPG function offers a comprehensive and user-friendly approach to health monitoring. In conclusion, the PPG technology in the Withings Scanwatch is a sophisticated system that leverages light-based sensors and advanced data analysis to provide accurate and reliable heart rate monitoring. Its integration into a stylish and user-friendly watch design makes it accessible for everyday use, offering valuable health insights for a broad range of users.
PPG in the Apple Watch is a sophisticated optical technology used for heart rate monitoring. This method employs light to measure the blood volume changes in the microvascular bed of tissue. In the case of the Apple Watch, this process involves the use of green LEDs and light-sensitive photodiodes. The green LEDs emit light onto the skin, which penetrates through to the blood vessels. Blood, being red, absorbs green light. Therefore, when the heart beats and more blood flows through the vessels, more green light is absorbed. Between heartbeats, less green light is absorbed. The photodiodes in the Apple Watch then measure the amount of light that is reflected back to the sensor. By analyzing these changes in light absorption, the Apple Watch can determine the heart rate. One of the key advantages of PPG in the Apple Watch is its non-invasive nature. The technology does not require any physical contact with blood, making it convenient for continuous monitoring. Additionally, the use of green LEDs, which are particularly efficient in detecting blood flow, enhances the accuracy of the measurements. [
17] Moreover, the PPG technology in the Apple Watch is complemented by advanced algorithms. These algorithms are designed to filter out noise and artifacts, such as those caused by movement or changes in environmental light, ensuring that the heart rate data remains accurate and reliable. This is particularly important during physical activities, where motion can introduce significant interference. The Apple Watch also uses infrared LEDs for heart rate monitoring in the background, as this consumes less power and is sufficient for regular, continuous tracking throughout the day. The switch between green and infrared LEDs allows the device to balance performance and battery life. [
18] In summary, the PPG technology in the Apple Watch is a blend of optical science and advanced algorithms, offering a non-invasive, adaptive, and efficient means of tracking heart rate. It exemplifies how wearable technology can leverage light-based sensors to provide valuable health insights in everyday life.
2.5. Statistical analysis
Continuous data were presented in this study as mean values accompanied by their corresponding standard deviations (SD) to provide a clear indication of the central tendency and the spread or variability within the data set. For categorical or binary data, results were expressed as absolute numbers along with their relative frequencies, reported in percentages, to offer an intuitive understanding of the proportion of events or characteristics within the sample. To compare groups, appropriate statistical tests were applied based on the type of data. For continuous variables, group differences were analysed using the t-test, a widely used parametric test that assesses whether the means of two groups differ significantly from each other. In cases of categorical data, the Chi-Square Test was employed to evaluate the association between groups by comparing the observed frequencies to those expected under the null hypothesis. Both tests were two-sided to account for the possibility of differences in either direction and a p-value of less than 0.05 was considered the threshold for statistical significance, indicating that the results were unlikely to have occurred by chance. For time-to-event analyses, such as examining the occurrence of atrial fibrillation over the study period, nonparametric estimates were calculated using the Kaplan-Meier method. This approach allows for the estimation of survival functions, enabling the visualization of time-related events, while the log-rank (Mantel-Cox) test was utilized to compare the survival curves between groups, determining whether there was a statistically significant difference in the event times. All statistical analyses were conducted using SPSS software, specifically Version 27, which provided a robust platform for executing both descriptive and inferential statistics. The use of SPSS ensured that data handling and analysis were performed with precision, allowing for accurate interpretation of the findings and adherence to rigorous statistical standards.
3. Results
Between November 2021 and January 2023, we enrolled 264 patients for our study. However, 4 participants were later excluded, leaving us with 260 datasets that underwent statistical analysis. The reasons for exclusion were the patient's decision not to wear the watch or a known but undocumented atrial fibrillation before the surgery. Out of these 260 patients, telemetry identified 29 with POAF while they wore the Scanwatch. From this subset, the Scanwatch detected "signs of AF" in 20 patients. Interestingly, 3 patients displayed "signs of AF" on the Scanwatch, though telemetry did not confirm this diagnosis. Consequently, the Scanwatch demonstrated a sensitivity of 69% and a specificity of 98.7%. Given the AF prevalence of 9% in our study population, we determined a positive predictive value (PPV) of 87.0% and a negative predictive value (NPV) of 96.2%. All these main results are also summarized in
Table 1.
Table 2 displays the baseline patients’ characteristics. Information for this table was driven from admission records, surgical reports, and discharge documentation. It reveals a significant age difference between patients diagnosed with AF and those without. Moreover, AF patients typically had higher CHA2DS2-VASc scores—a metric which considers factors like heart failure, hypertension, age, diabetes, prior strokes, vascular disease, and gender—and last but not least, were more likely to have multiple surgical indications.
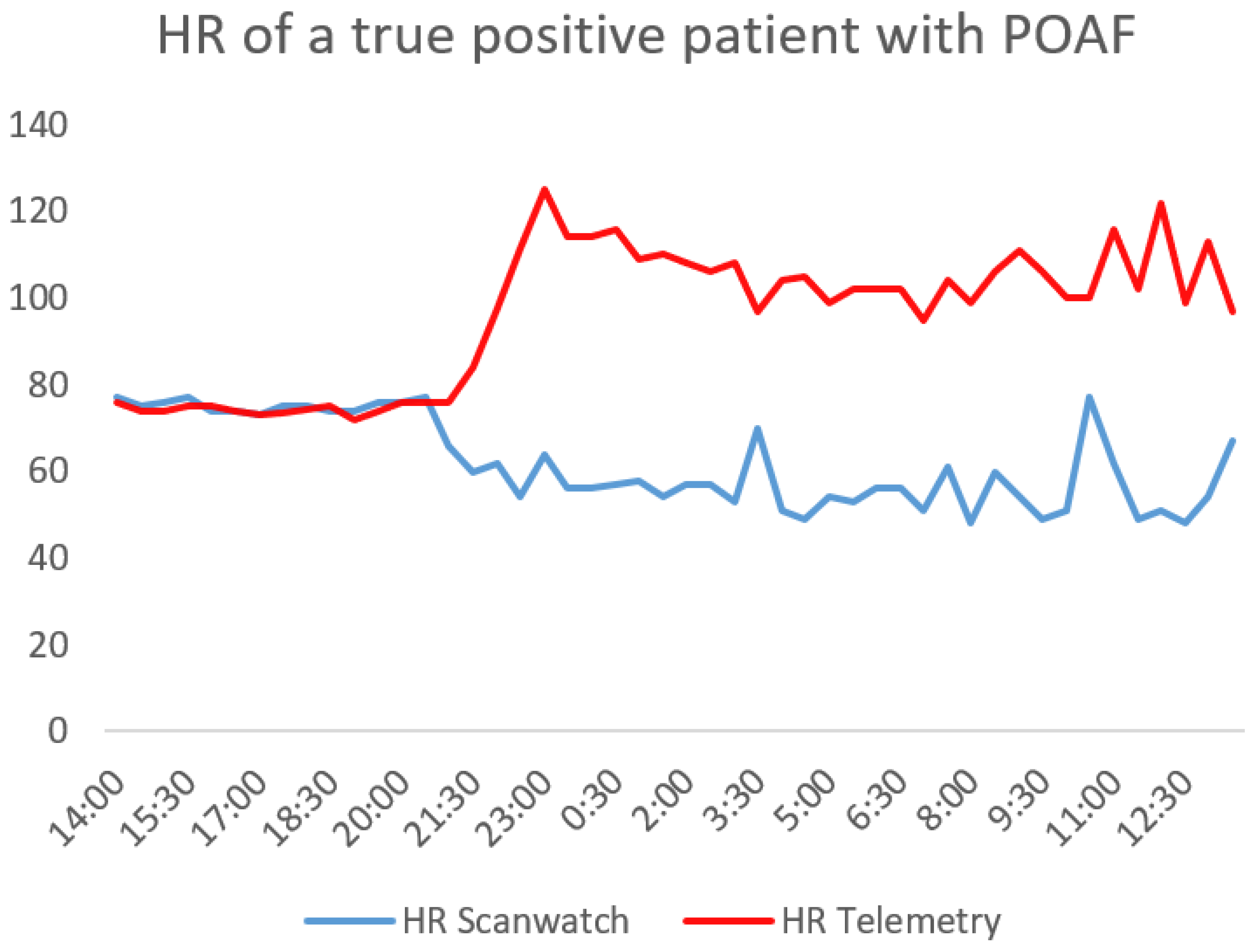
Five patients underwent aortic replacement. On average, patients spent about 1.2 days in either the ICU or IMC post-operatively and were equipped with the Scanwatch after 1.45 days. We also compared the heart rate of a true positive patient as an example.
Figure 2 shows the measured HR during the 24 hours of monitoring and
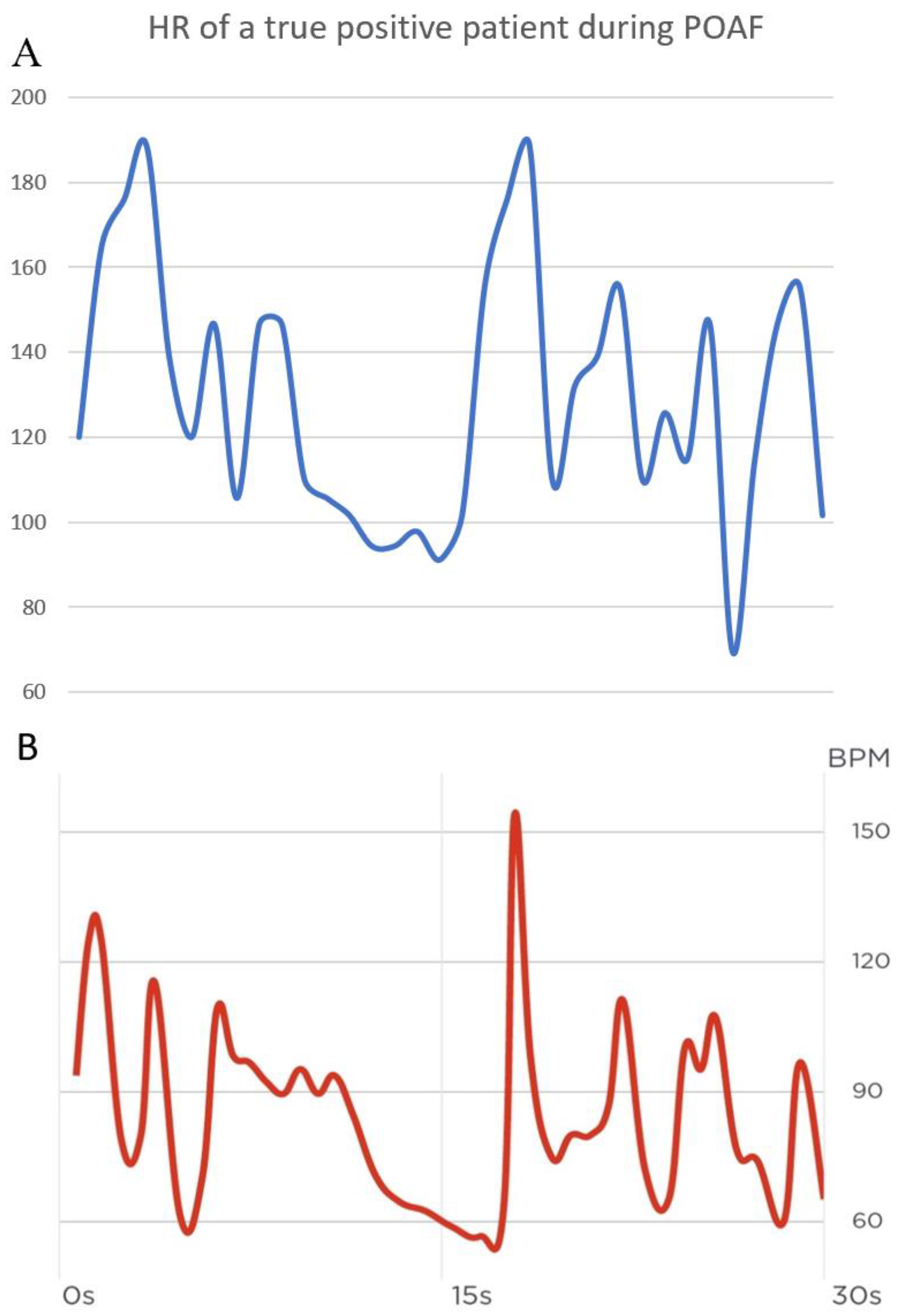
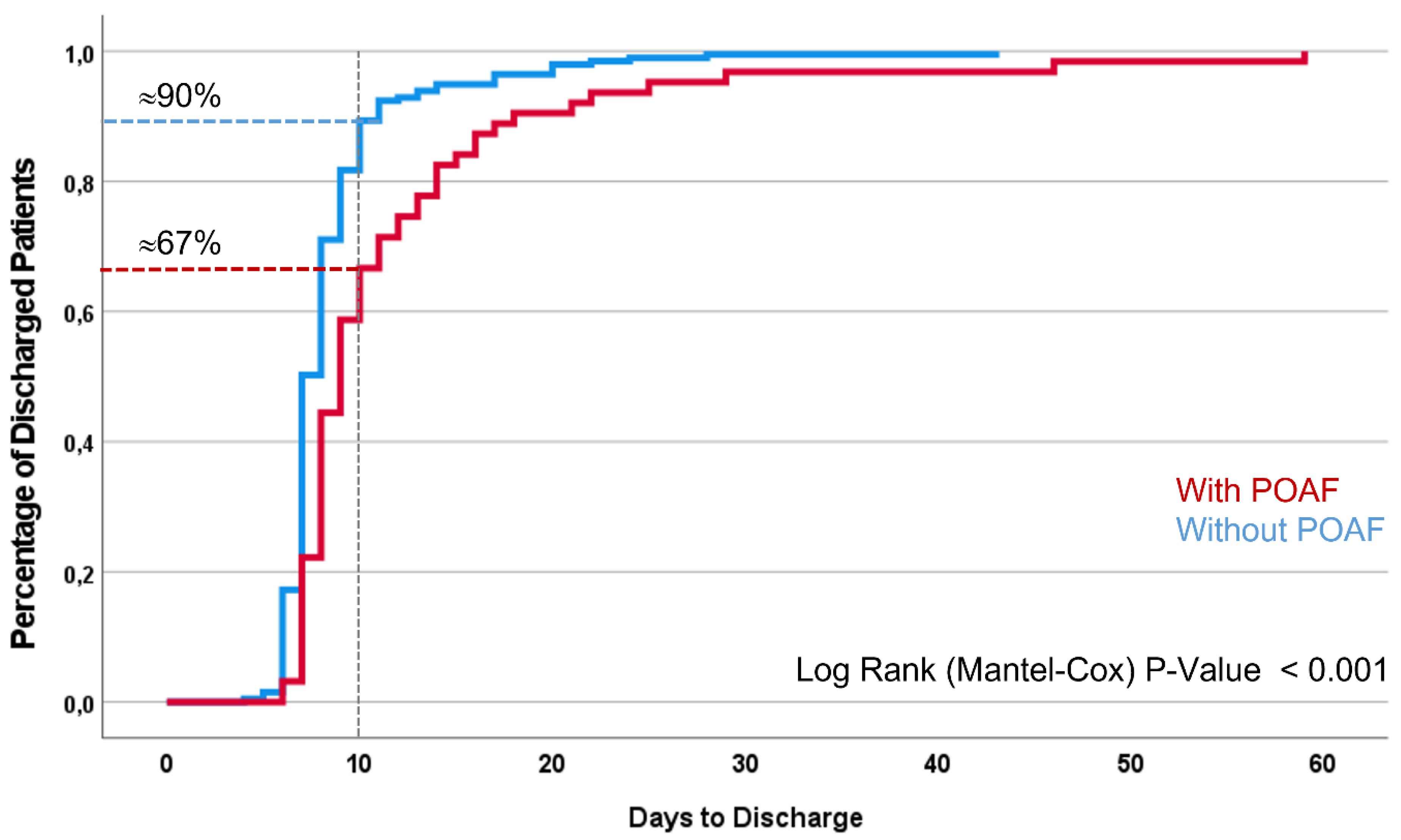
Figure 3 shows the measured heart rate during 30 seconds of AF. Throughout their hospital stay, 63 patients exhibited POAF. The average post-surgical hospitalization duration was 9.3 days. Notably, a significant difference in the duration of hospital stay was observed between the two patient groups, as depicted in
Figure 4. Patients who did not develop POAF tended to have considerably shorter hospital stays. By the 10-day mark, nearly 90% of these patients had been discharged, reflecting a quicker recovery and return to normal health. In contrast, fewer than 70% of patients who experienced POAF were discharged by the same time point, indicating that those with POAF generally required longer hospitalization. This extended stay may be attributable to the need for additional monitoring, treatment, and management of complications associated with POAF, further highlighting the clinical impact of this condition on postoperative outcomes.
4. Discussion
Our study demonstrates that using a smartwatch to detect POAF is both practical and reliable. To the best of our knowledge, this is the first study to specifically investigate the detection of atrial fibrillation using the PPG sensor in the Withings Scanwatch. Prior to our research, the only available data on AF detection with this device came directly from studies conducted by Withings itself. [
19] Withings reported a sensitivity of 98% and a specificity of 93% in their investigation. However, we were unable to locate specific details regarding the performance metrics or the underlying study data. Additionally, exact information on the algorithm used to evaluate the PPG signal has not yet been made publicly available. [
20] While the sensitivity and specificity figures reported by Withings are higher than those observed in our study, the two investigations are not entirely comparable. One key difference is that our study focused on a specific patient group, whereas in the Withings study, about 20% of the recordings were considered inconclusive and were subsequently excluded. Another factor that may account for the difference in outcomes is that our study was designed to reflect real-life clinical scenarios rather than being conducted under tightly controlled conditions. For example, we provided participants with detailed instructions on how to use the smartwatch but did not supervise or influence how they wore it or for how long. This approach likely makes our results more representative of real-world usage and outcomes.
There are some wearables available with higher accuracy compared to the Scanwatch. [
21] However, there is not prospective head-to-head comparison. With a sensitivity of 69%, episodes of atrial fibrillation can obviously be missed by the Scanwatch. This in turn can lead to serious complications. Therefore, advanced algorithms, modelling, such as deep learning, or increasing the PPG sampling frequency could help to improve detection accuracy and should be the aim of future studies. But it's crucial to understand that the Scanwatch PPG isn't designed for definitive AF diagnosis but rather as a screening tool. Any positive screening result from the smartwatch should always be followed by further verification. This can be achieved either through a single-lead ECG provided by the watch itself or by seeking confirmation from a medical professional using a 12-lead ECG or extended ECG monitoring. This step is essential to accurately confirm or rule out the diagnosis of AF. This approach is also strongly supported by the current European Society of Cardiology (ESC) guidelines for the diagnosis and management of atrial fibrillation, which emphasize the need for thorough validation following initial AF detection. [
3]
The Scanwatch's appeal extends beyond its AF screening capabilities, which are notable for their high specificity and reasonable sensitivity. It also has a longer battery life compared to many of its competitors and features a user-friendly design that operates solely through a single crown. This simplicity is particularly advantageous for older individuals who may find technology-intensive wearables challenging to use, yet are at a higher risk for AF. The straightforward operation of the Scanwatch ensures ease of use while providing valuable health monitoring for this vulnerable demographic. Some research even suggests that given a choice, patients lean towards the Scanwatch over other smartwatches [
13] or wearable devices [
14].
It is quite likely that, in the near future, smartwatches and other wearable devices will become instrumental in screening for AF, both after cardiac surgery and in the everyday lives of individuals at increased risk for the condition. While the hospital setting, we tested may not perfectly reflect daily life, it is conceivable that smartwatches and wearables could enable earlier detection of AF. These devices might help in preventing complications associated with POAF, such as strokes and premature death [
8]. This becomes increasingly important because POAF can recur after its initial resolution or may develop after discharge. [
10,
11] The viability and cost benefits of extended POAF screening should be the subject of upcoming research. Nevertheless, existing studies indicate that systematic AF screening for the elderly is effective [
22], and smartwatch-based methods are cost-efficient. [
23] Given these advantages, health insurance providers and hospitals might contemplate providing high-risk patients with smartwatches for AF screening, aiming to curtail both complications and expenses.
It is important to emphasize that wearables should not and cannot replace telemetry as the gold standard. Telemetry not only excels in detecting AF but is also superior in identifying other potentially fatal cardiac arrhythmias. To illustrate, based on data from our study sample depicted in Fig. 1, the Scanwatch tends to underestimate heart rates post-AF onset. That said, it does detect AF, registering irregular and elevated pulses at multiple instances, as depicted in a comparative view with telemetry data in Fig. 2. One possible explanation would be that the PPG was not able to detect all beats in each of the measurements and therefore estimated the heart rate incorrectly. However, we cannot answer this question conclusively without the exact PPG data and the algorithm used to evaluate the data. Still, devices like the Scanwatch gain relevance when patients regain mobility. Undoubtedly, a significant advantage of the Scanwatch over telemetry is its enhanced comfort for the patients and users.
Utilizing the Scanwatch, one can also envision the application of pharmacological cardioversions through the "pill in the pocket" method. Here, upon detecting and confirming an AF episode using wearable or standard ECG, antiarrhythmic drugs like flecainide or propafenone are used to restore sinus rhythm. This treatment strategy is already endorsed in the ESC guidelines, though it excludes patients who recently experienced acute coronary syndrome or those with severe heart failure. [
3]
Our study found that AF was more common in the early days following surgery, particularly among older patients with higher CHA2DS2-VASc scores and those undergoing multiple surgeries. While specific diagnoses did not show statistical significance - likely due to the characteristics of our study population and the focus on POAF that developed shortly after cardiac surgery - our results confirm that older age and multimorbidity are significant risk factors for POAF. Therefore, patients with these characteristics could benefit from more intensive and extended AF screening. Additionally, our findings revealed that patients with POAF experienced a notably longer time to discharge, as illustrated in
Figure 4. Although these results are consistent with existing knowledge, they serve to reinforce and validate previous findings. [
8] Despite sharing the same fundamental principle—using light to measure blood volume changes in the skin—the Withings Scanwatch implement PPG differently than the Apple Watch, optimizing it for unique use cases and user preferences. The Apple Watch primarily uses green LEDs for PPG. Green light is efficiently absorbed by haemoglobin, making it highly effective for measuring heart rate during various activities. [
24] Additionally, the Apple Watch utilizes infrared LEDs for background heart rate monitoring, which consumes less power and is better suited for resting conditions. The combination of green and infrared LEDs allows for flexibility in adapting to different scenarios. In contrast, the Withings Scanwatch employs red and infrared LEDs. Red light penetrates deeper into the skin compared to green light, making it advantageous for measuring metrics like blood oxygen saturation and ensuring accuracy across a wider range of skin tones. The use of red light also enhances the device’s ability to monitor users with tattoos or darker skin, areas where green light might face challenges due to lower penetration. Both devices incorporate advanced algorithms to process the reflected light data and filter out noise caused by movement, ambient light, and other interferences. The Apple Watch excels in real-time tracking during physical activities, leveraging its green-light-based system to provide rapid updates on heart rate during high-intensity workouts. Its algorithms are designed to handle the dynamic conditions of exercise environments. The Scanwatch, however, focuses on deeper physiological monitoring, such as detecting AF and sleep apnoea. Its infrared and red-light system is particularly suited for capturing detailed health metrics in static or resting states, as well as continuous monitoring during sleep. While both devices offer reliable heart rate data, the Scanwatch’s design is more specialized for long-term health analysis, whereas the Apple Watch leans towards fitness and wellness monitoring. The choice of LEDs also impacts power consumption. The Apple Watch, with its frequent use of green LEDs during workouts, consumes more energy, contributing to shorter battery life. The Scanwatch’s reliance on infrared and red LEDs, which are more energy-efficient, enables longer battery life, often lasting up to 30 days on a single charge—significantly outperforming the Apple Watch.
Limitations
Our study has several limitations. One key limitation is the relatively small sample size of 32 patients who experienced AF in the early days following surgery. Additionally, we did not personally review all the telemetry data, which raises concerns about the accuracy of the rhythm evaluations performed by medical staff and the potential for missing brief AF episodes. Particularly, episodes of normofrequent AF might not have been captured by the telemetry. Lastly, the functionality of the Scanwatch itself presents a limitation. The device measures pulse using a PPG sensor at fixed 10-minute intervals, which cannot be adjusted. Consequently, short AF episodes may have gone undetected by the device.