1. Introduction
Cardiovascular diseases (CVDs) are a leading cause of morbidity and mortality worldwide, accounting for approximately 17.9 million deaths annually, which is about 32% of all global deaths according to the World Health Organization [
1]. Electrocardiograms (ECGs) are a primary diagnostic tool for various CVDs, including arrhythmias, myocardial infarctions, and other cardiac abnormalities. The high diagnostic accuracy of ECGs in detecting myocardial ischemia was demonstrated, reinforcing the critical role of ECGs in early detection and treatment planning for cardiovascular conditions [
2]. Moreover, studies such as those by Mincholé and Rodríguez have highlighted the predictive power of ECG signals in identifying patients at risk of future cardiac events, further underscoring the importance of ECG-based diagnostics [
3].
Remote monitoring of ECG signals has become increasingly important, especially with the rise of chronic heart disease and the need for continuous, real-time monitoring. A report by the American Heart Association (AHA) in 2019 emphasized that remote monitoring can significantly improve patient outcomes by enabling early detection of potential issues, thereby allowing for timely medical interventions [
4]. It was found that remote monitoring of heart failure patients reduced hospitalizations and improved survival rates. These findings highlight the critical role of remote ECG monitoring in managing cardiovascular health, especially in at-risk populations [
5].
Internet of Medical Things (IoMT) is a term for medical objects that apply IoT technology to the medical field and can transmit medical information and bio-signal data in real time through network communication. IoMT device includes an integrated circuit for data collection, a signal processing device for data processing, and a network module through data transmission. So, then this system can make a minimal human intervention and effectively monitor things that were not monitored before. Digital health, particularly involving the IoMT devices, is revolutionizing health management in daily life. Lupton reported that it was discussed how IoMT devices, such as wearable ECG monitors, provide continuous health data that can be analyzed to offer personalized health insights and interventions [
6]. The advantages of using IoMT devices include improved patient adherence to treatment plans, real-time health monitoring, and the ability to collect large datasets for advanced analytics, which can lead to more accurate predictive models and personalized healthcare strategies. Also, it was demonstrated that digital health technologies not only improve health outcomes but also enhance the quality of life by allowing individuals to manage their health proactively and conveniently [
7].
This study aimed to verify the effectiveness of remote biological signal monitoring technology using the IoMT device.
2. Materials and Methods
2.1. Participants
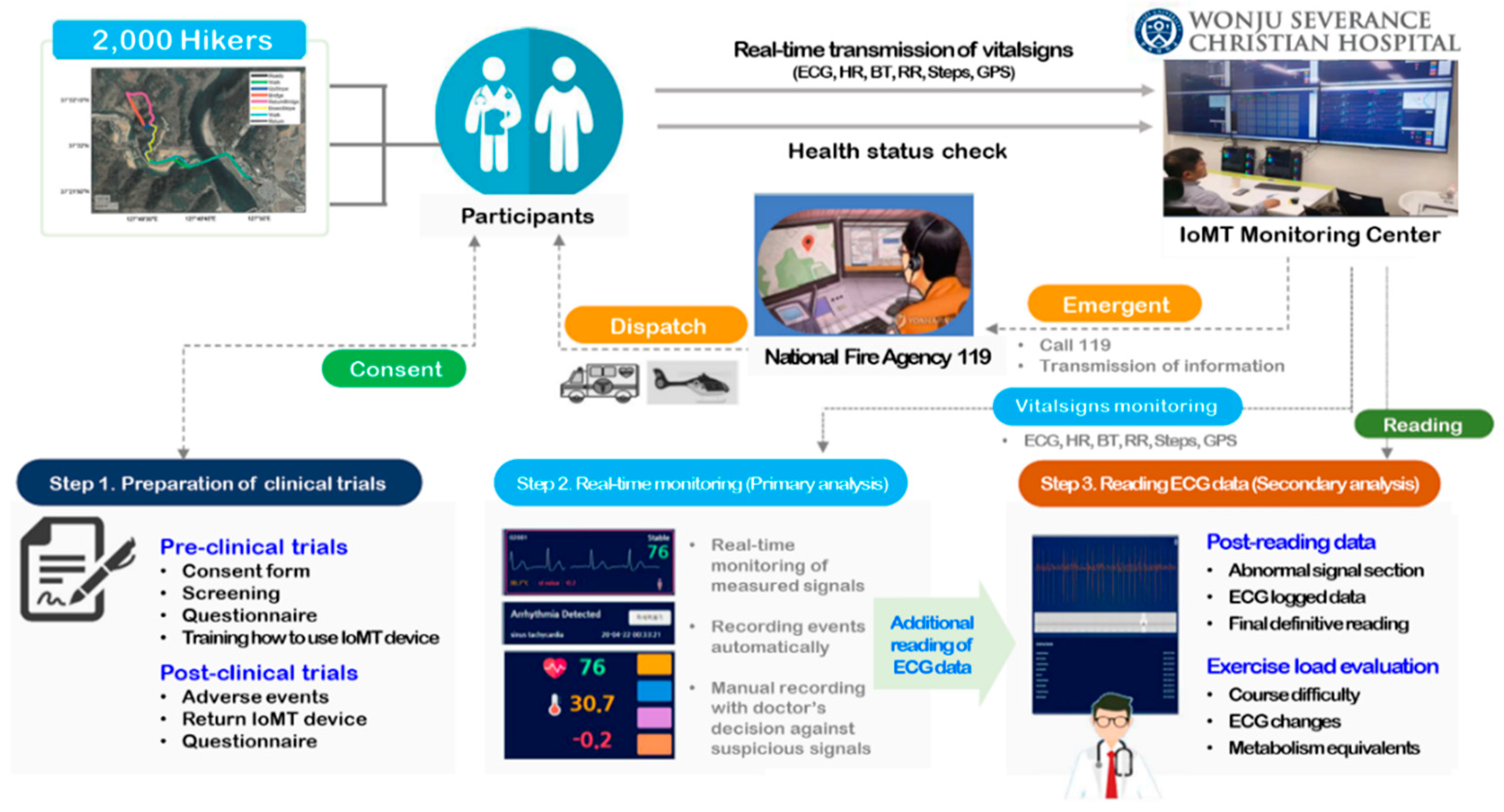
In this study, a clinical trial was conducted to verify the effectiveness of the remote monitoring diagnosis technology using the IoMT device. The clinical trial was conducted a total of 30 times in about 8 months and a total of 2,000 hikers participated. Each hiker filled out a survey about the clinical trial consent and existing health information in advance then attached a wearable ECG monitoring patch to their chest and climbed the mountain. This study was carried out with approval from the Research Ethics Committee of Yonsei University Wonju Severance Christian Hospital (IRB approval number: CR319186) and registered with the Clinical Research Information Service (CRIS) operated by the Korea Disease Control and Prevention Agency (CRIS trial registration number: KCT0005795).
2.2. ECG Monitoring Device
HiCardi
® (MEZOO Co. Ltd., Wonju, Gangwon State, Republic of Korea), an IoMT device used in this study, is a patch-type electrocardiograph attached to the hiker's chest. This wearable device could monitor and record various parameters related to heart signals such as a single-lead ECG, heart rate, respiration, skin surface temperature, and activity. The data from the wearable patch were transferred through Bluetooth low energy to a mobile gateway, which was implemented as a smartphone application. The mobile gateway transmitted the real-time data to the core, analytics, and service platform [
8].
2.3. Role of a Physician at the IoMT Monitoring Center
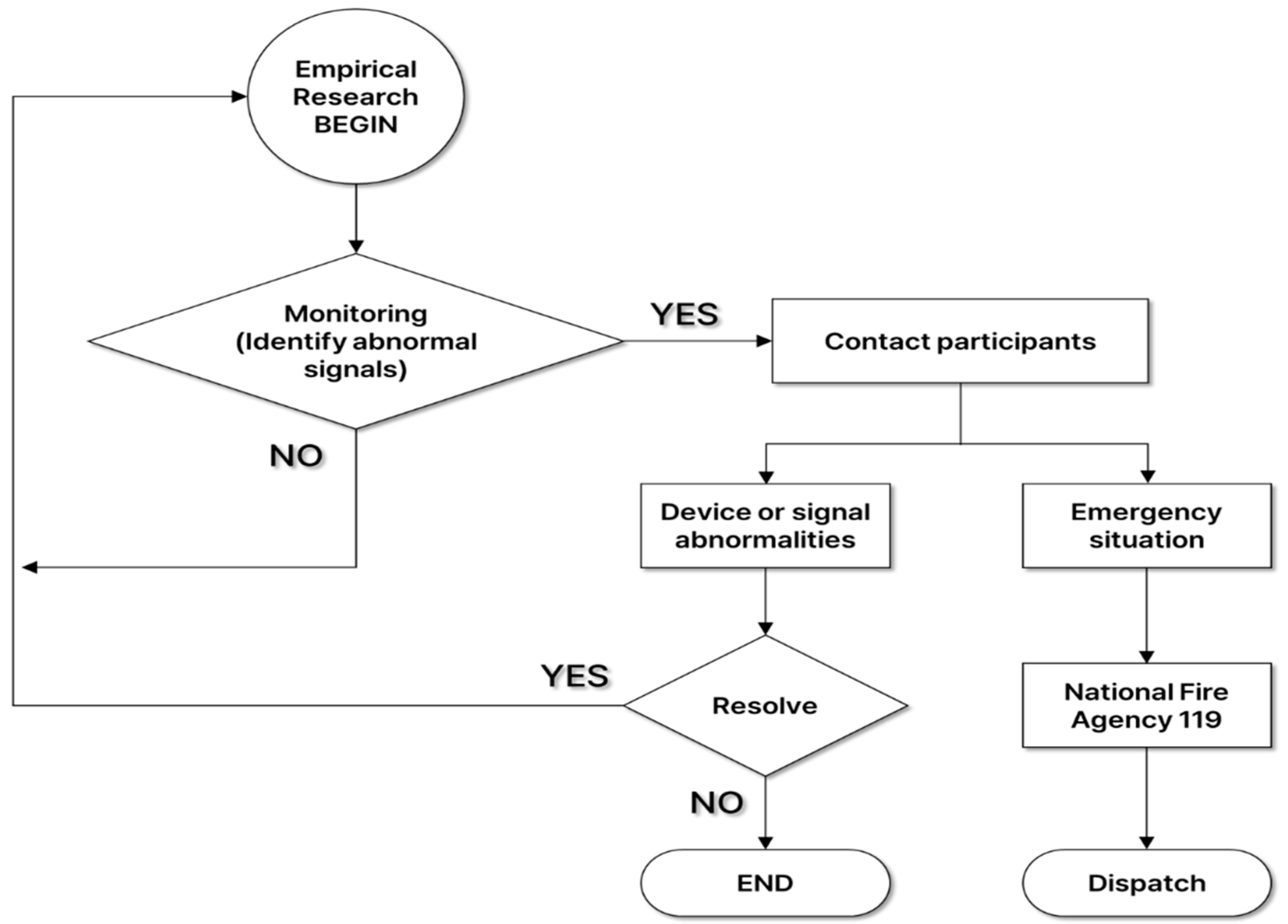
Wonju Severance Christian Hospital operates an IoMT monitoring center for digital health research, including remote monitoring and non-face-to-face collaboration. At the IoMT monitoring center, a physician, who was an emergency medicine specialist, collected health status information such as basic information, vital signs (blood pressure and body temperature), medical history, and medications surveyed by participants at the empirical research research site. After that, he monitored the hikers' biosignals transmitted in real-time to check whether abnormal ECG was observed. If an abnormal signal was detected during ECG biosignal monitoring and disease was suspected, the physician notified the clinical research coordinators (CRCs) at the empirical research site, and then the CRC advised the participant to visit a hospital. Also, if problems such as noise in the device or disconnection due to poor attachment occurred, the problem was resolved through on-site response. In the event of an emergency, the condition of the participant was checked according to the emergency response protocol, and the patient's condition and location information was delivered to the National Fire Agency 119 to prepare for rapid emergency treatment through emergency dispatch. (
Figure 1)
2.4. Empirical Research Scenarios
Figure 2 shows the flowchart of IoMT monitoring service. Before starting the clinical trial, the CRCs explained the purpose of the clinical trial. Those who agreed to participate in the clinical trial completed a survey, physical information, and health status. When selected as participants, they were provided with an IoMT medical device, installed an application linked to the device, and instructed on how to use it and precautions. Participants attach the IoMT medical device linked to the application to a designated location and begin measuring biometric information. The measured biometric information is transmitted to the IoMT monitoring center in real-time. An emergency medical specialist monitored remotely the participants' vital signs at the center. Participants will wear the IoMT medical device and take the clinical test while hiking. However, in the event of an emergency, the test subject's emergency situation will be responded to by contacting the National Fire Agency (119) according to established protocols. Once monitoring of the IoMT medical device is completed, biosignal changes are observed and tracked by exercise load. The emergency medicine specialist identified the presence or absence of any adverse reactions that occurred while hiking, and the participants completed the clinical trial by returning the IoMT medical devices after completing a satisfaction survey regarding their health status and service model.
2.5. Follow-up Management of Participants Who Were Recommended to Visit a Hospital
Follow-up observations were conducted through telephone surveys on participants who were recommended to receive hospital treatment after abnormal signals were discovered at the demonstration site. A survey on telephone calls was conducted regarding whether or not the patient visited the hospital, the date of the hospital visit, the method of diagnosis, and the results of the diagnosis or treatment.
2.6. Satisfaction Survey
At the end of the empirical research, participants completed a satisfaction survey to evaluate their experiences. The survey included questions to assess various aspects of their participation and the usefulness of the provided health information and devices. Participants were asked about their previous experiences in other clinical studies and whether the health information provided (ECG, heart rate, respiration, body temperature) was helpful. They were also asked if they found the IoMT device to be helpful for health management. Satisfaction with the IoMT device was measured using a five-point Likert scale ranging from "Very Satisfied" to "Very Dissatisfied". Specific aspects evaluated included discomfort experienced after attaching the device (e.g., detachment, itchiness, restriction of movement), trust in the provided health information, and willingness to use the attached IoMT device in the future. Additionally, satisfaction with the HiCardi mobile application was assessed, particularly the ease of accessing health information through the application, using a five-point Likert scale. Participants also rated their overall satisfaction with the study and were invited to provide comments on potential improvements. The responses were collected and analyzed to gauge overall satisfaction and identify areas for improvement. Feedback from participants provided valuable insights into the user experience and the effectiveness of the health information and devices used in the study.
3. Results
3.1. General Characteristics
Table 1 presents the demographic characteristics of the participants. A total of 2,000 people participated, including 811 men and 1,189 women. The average age of the participants was 49.36±14.65 years old. Also, males had a mean age of 49.35 years, while females had a mean age of 49.37 years. The age distribution shows that the highest proportion of hikers falls into their 50s (28.05%), followed by those in their 60s (21.40%) and 40s (17.65%). Among men, the 50s (23.18%) and 60s (23.05%) age groups are more prominent, whereas women show higher proportions in the 50s (31.37%) and 60s (20.26%). Notably, 4.65% of the hikers are in their 70s or older, with a higher proportion of men (7.15%) compared to women (2.94%). In terms of physical characteristics, there are clear gender differences. The average height of all hikers is 164.16 cm and the average weight is 64.26 kg. Men have an average height of 171.35 cm and an average weight of 73.04 kg, while women average 159.26 cm in height and 58.26 kg in weight. Blood pressure measurements show a mean systolic blood pressure (SBP) of 130.89 mmHg and a mean diastolic blood pressure (DBP) of 85.61 mmHg. Men have higher average SBP (136.85 mmHg) and DBP (88.79 mmHg) compared to women, who have average values of 126.82 mmHg and 83.44 mmHg, respectively. The mean pulse rate (PR) is 74.87 bpm, with only slight differences between men (76.05 bpm) and women (74.07 bpm). The average body temperature is 36.72℃. Regarding health status, 66.25% of hikers reported having no chronic diseases, with 63.87% of men and 67.87% of women reporting the same. The prevalence of chronic diseases among hikers includes one disease in 21.10%, two diseases in 9.35%, and more than three diseases in 3.30%. For family history, 45.30% of hikers reported having a family history of health issues, with a higher proportion among women (51.22%) compared to men (36.62%).
3.2. Detection of Abnormal Signals and Recommendation to a Hospital through Participant ECG Monitoring
Table 2 summarizes the results of clinical trials, focusing on the number of participants, the detection of abnormal ECG signals, recommendations to visit the hospital, and detailed cardiovascular disease (CD) findings. A total of 2,000 participants were involved across 30 trials. Among these participants, 318 (15.90%) were monitored for abnormal signals on their ECGs. Also, 296 (93.08% of the patients who received abnormal signals) were recommended to visit the hospital, with 182 (9.10%) showing specific details of cardiovascular diseases. The abnormalities detected included atrial fibrillation (AF), sinus arrhythmia (SA), premature atrial contraction (PAC), bundle branch block (BBB), atrioventricular (AV) block, premature ventricular contraction (PVC), ventricular fibrillation (VF), tachycardia, bradycardia, ST depression, chest pain, palpitations, and other abnormal rhythms. Notably, trial 13 had the highest number of abnormalities with 40 cases (27.78% of its participants), including PVC, arrhythmia, and tachycardia, among others. The detailed cardiovascular disease findings indicated the presence of various types of arrhythmias and other heart conditions, with PVC being the most frequently observed abnormality.
3.3. Observations of Participants Who Were Recommended to Visit a Hospital
The 182 people who participated in the empirical research and received recommendations for cardiovascular disease-related treatment through abnormal signal monitoring were followed up through phone calls to determine whether they visited the hospital, the timing of hospital visits, the diagnosis results, and diagnosis details. 139 (76.37%) answered the phone, and 30 (21.58% of those who answered the phone) visited the hospital and received treatment.
Table 3 presents the results of observations among the 30 participants who received treatment after visiting the hospital. Based on the date each participant participated in the empirical research, 24 (80.00%) visited the hospital and underwent examination within one month. A total of 30 patients were analyzed, with the majority (56.67%) being diagnosed using only an electrocardiogram (EKG). A combined approach of EKG and ultrasound was utilized for 20% of the patients, while ultrasound alone accounted for 16.67%. Additionally, a small percentage were diagnosed using EKG and X-ray (3.33%) or general treatment methods (3.33%). Regarding the timing of hospital visits, 80% of the patients sought medical attention within one month of experiencing symptoms, whereas 20% delayed their visits beyond one month. The diagnosis results were meaningful as cardiovascular disease was discovered in seven people. In detail, there were included progress observation (6.67%), stent procedures (3.33%), arrhythmia (3.33%), combined atrial fibrillation and arrhythmia (3.33%), heart medication prescriptions (3.33%), and panic disorder (3.33%).
3.4. Satisfaction Survey after Completion of Empirical Research
Table 4 indicates the results of the participants' satisfaction survey after the completion of empirical research. 1,883 (94.15%) had not previously participated in clinical studies. A vast majority (96.55%) found the health information provided by the device, including ECG, heart rate, respiration, and body temperature, to be helpful. Most participants (97.95%) believed that the device would be beneficial for health management. In terms of comfort, 64.35% reported feeling 'Very Satisfied' or 'Satisfied', with an average satisfaction score of 2.02±1.35. Some participants did experience discomfort related to detachment, itchiness, or restriction of movement. Trust in the health information provided by the devices was also strong, with 1,510 participants (75.50%) indicating 'Very Satisfied' or 'Satisfied' and an average satisfaction score of 3.87±1.20. Interest in the future use of the HiCardi device was moderate, with 48.20% of all showing 'Very Satisfied' or 'Satisfied' responses and an average satisfaction score of 3.53±1.13. Regarding the ease of accessing health information through the mobile application, 75.95% of participants were 'Very Satisfied' or 'Satisfied', with an average satisfaction score of 3.88±1.15. Overall satisfaction with the empirical research was high, with 78.65% of participants expressing 'Very Satisfied' or 'Satisfied' sentiments and an average satisfaction score of 3.98±1.17.
4. Discussion
This study conducted a clinical trial on 2,000 ordinary hikers to verify the effectiveness of the remote monitoring technology using the IoMT devices and system. As a result, 7 of 2,000 hikers in real-time using the IoMT devices and system were real-diagnosed with heart diseases. In the case of some heart disease diagnoses, many cases are difficult to detect in the existing monitoring system, and monitoring for more than a week is required in some cases. In this study, it was meaningful to test that the remote IoMT monitoring system and device in daily activities can be effective in detecting heart disease. Similarly, in the study by Paddy M. Barrett et al [
9], comparing 24-hour Holter monitors with 14-day adhesive patch monitors in 146 patients, the patch monitor detected more arrhythmic events. Furthermore, in the study by Warren M. Smith et al [
10], using a P-wave-centric ECG patch monitor, 46% of the 50 patients detected rhythm changes requiring management alterations, compared to 12% with the Holter monitor. These studies suggest that the IoMT device may offer higher diagnostic rates and convenience compared to traditional Holter monitors.
In this study, approximately 15 types of heart disease-related abnormal signals were observed using the IoMT device, including PVCs and atrial fibrillation. Among these, PVCs were the most frequently observed, with 79 instances. Examining other studies related to remote ECG monitoring, Markus Lueken et al [
11] focused on the automated signal quality assessment of single-lead ECG recordings for the early detection of various arrhythmias, including PVCs. This study reported that PVCs were observed in approximately 75% of participants during 24 to 48-hour ambulatory ECG monitoring, demonstrating a high prevalence of these abnormal signals in the monitored population. Similarly, in the study by B. Venkataramanaiah et al [
12], the prevalence and characteristics of premature ventricular contractions (PVCs) were examined using remote ECG monitoring systems. This study emphasized the importance of detecting PVCs due to their potential to develop into more serious arrhythmias. The results indicated that PVCs were observed in 67.7% of the monitored population via 24-hour Holter monitoring. Among these, 60.0% had a PVC burden of less than 5%, while 7.7% had a PVC burden of 5% or more. As shown in our study and others, the occurrence rate of PVCs among abnormal ECG signals is relatively high in both IoMT devices and Holter monitoring systems. This is because PVCs can be precursors to various serious arrhythmias [
13].
Additionally, our study found that 7 out of 30 patients (23%) who visited the hospital were diagnosed with actual heart disease using the IoMT device. This indicates that remote monitoring can have clinical effectiveness in preventative healthcare. A similar study by Marco V. Perez et al [
14] involved 419,297 participants wearing Apple Watches, where those with detected irregular pulses acquised ECG patches for 7 days of monitoring. Among the 2,161 participants who received irregular pulse alarms, 450 returned ECG patches, and 34% of these showed atrial fibrillation. The positive predictive value of the irregular pulse alarms was 84%. The Fitbit Heart Study by Steven A. Lubitz et al [
15] followed a similar approach, analyzing 455,699 participants. Those with detected irregular heart rhythms were provided with an ECG patch for one week. The Fitbit PPG-based algorithm detected irregular heart rhythms and used ECG patches to confirm atrial fibrillation. Results indicated a high positive predictive value of 98%, demonstrating the effectiveness of consumer wearable technology in identifying AF episodes. Compared to these two studies, our study's diagnostic accuracy appears relatively lower. However, this could be attributed to factors such as sweat, heavy breathing, or movement during hiking causing poor contact with the patch, and geographic characteristics of being in the mountains potentially affecting the transmission of ECG signals. While our study's accuracy of diagnosis is relatively lower, this can be attributed to the limitation of a small number of confirmed follow-up calls.
Moreover, a small number of confirmed follow-up calls is a limitation of our study. Many other studies also face low compliance with follow-up calls. For example, in a study by Dorothy M. Mwachiro et al [
16], nurses conducted follow-up calls to evaluate their effectiveness in reducing 30-day readmissions post-discharge in neurosurgery patients. However, only 45% of the 83 patients received follow-up calls, impacting the reliability of the study's assessment of readmission rates and post-discharge care. Another study by Phil Edwards [
17] investigated the challenges of long-term follow-up data collection in non-commercial, academically-led breast cancer clinical trials. Based on the UK perspective, the study highlighted difficulties in patient compliance and data collection in long-term follow-up studies. Low response rates made data collection challenging, affecting the reliability of the study outcomes. The researchers suggested that maintaining continuous contact with patients plays a crucial role in securing long-term follow-up data. Comparing these studies, our research also faced challenges with follow-up calls, which may have limited the study's results. If a higher response rate had been achieved, the diagnostic accuracy might have exceeded 23%. This highlights the difficulty of data collection through follow-up calls and indicates the need for new approaches to address these challenges in future research.
5. Conclusions
We demonstrated the effectiveness of remote cardiovascular monitoring during daily activities using IoMT devices by diagnosing actual heart disease with 23% accuracy through monitoring abnormal ECG signals collected from 2,000 general hikers using the patch-type electrocardiograph, HiCardi. Furthermore, to validate the practical effectiveness of the data collected in this study, we plan to conduct an in-depth kinetic study through the metabolic equivalents (METs) analysis from a kinematic perspective as a further study. This is significant as a more convenient method for monitoring and diagnosing cardiovascular diseases compared to treadmill tests conducted in hospitals. It is also expected to contribute to preventative healthcare by enabling immediate actions such as hospital visits or medication intake.
Author Contributions
Conceptualization, H.Y.; methodology, K.-H.L. and H.Y.; software, S.-P.C. and J.-H.P.; formal analysis, J.-H.L.; investigation, J.-H.L.; data curation, K.-H.L., I.-H.P. and H.Y.; writing—original draft preparation, H.-Y.L. and Y.-J.K.; writing—review and editing, H.-Y.L..; supervision, H.Y.; project administration, H.-Y.L. and J.-H.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the Korea Medical Device Development Fund grant funded by the Korea government (the Ministry of Science and ICT, the Ministry of Trade, Industry and Energy, the Ministry of Health & Welfare, Republic of Korea, the Ministry of Food and Drug Safety) (project number: RS-2020-KD000030). It was also supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (No. HR21C0885).
Institutional Review Board Statement
This study was carried out with approval from the Research Ethics Committee of Yonsei University Wonju Severance Christian Hospital (IRB approval number: CR319186) and registered with the Clinical Research Information Service (CRIS) operated by the Korea Disease Control and Prevention Agency (CRIS trial registration number: KCT0005795).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Acknowledgments
In this section, you can acknowledge any support given which is not covered by the author contribution or funding sections. This may include administrative and technical support, or donations in kind (e.g., materials used for experiments).
Conflicts of Interest
The authors declare no conflicts of interest.
References
- World Health Organization (2021). Cardiovascular diseases (CVDs). Available at: https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds).
- Zarama, V.; Arango-Granados, M.C.; Manzano-Nunez, R.; Sheppard, J. P.; Roberts, N.; Plüddemann, A. The diagnostic accuracy of cardiac ultrasound for acute myocardial ischemia in the emergency department: a systematic review and meta-analysis. Scand. J. Trauma. Resusc. Emerg. Med. 2024, 32, 19. [Google Scholar] [CrossRef]
- Mincholé, A.; Rodriguez, B. Artificial intelligence for the electrocardiogram. Nat. Med. 2019, 25, 22–23. [Google Scholar] [CrossRef] [PubMed]
- American Heart Association. (2019). Using remote patient monitoring technologies for better cardiovascular disease outcomes: guidance. American Heart Association.
- Simovic, S.; Providencia, R.; Barra, S.; Kircanski, B.; Guerra, J.M.; Conte, G.; Duncker, D.; Marijon, E.; Anic, A.; Boveda, S. The use of remote monitoring of cardiac implantable devices during the COVID-19 pandemic: an EHRA physician survey. Europace. 2022, 24, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Lupton, D. How does health feel? Towards research on the affective atmospheres of digital health. Digit. Health. 2017, 3, 2055207617701276. [Google Scholar] [CrossRef] [PubMed]
- Awotunde, J. B., Ajagbe, S. A., & Florez, H. (2022, October). Internet of things with wearable devices and artificial intelligence for elderly uninterrupted healthcare monitoring systems. In International Conference on Applied Informatics (pp. 278-291). Cham: Springer International Publishing.
- Lee, H.Y.; Lee, K.H.; Lee, K.H.; Erdenbayar, U.; Hwang, S.; Lee, E. Y.; Lee, J. H.; Kim, H. J.; Park, S. B.; Park, J. W.; Chung, T. Y. , Kim, T. H.; Youk, H. Internet of medical things-based real-time digital health service for precision medicine: Empirical studies using MEDBIZ platform. Digit. Health. 2023, 9, 20552076221149659. [Google Scholar] [PubMed]
- Barrett, P. M.; Komatireddy, R.; Haaser, S.; Topol, S.; Sheard, J.; Encinas, J.; Fought, A. J.; Topol, E. J. Comparison of 24-hour Holter Monitoring with 14-day Novel Adhesive Patch Electrocardiographic Monitoring. Am. J. Med. 2014, 127, 95.e11–95.e17. [Google Scholar] [CrossRef] [PubMed]
- Smith, W. M.; Riddell, F.; Madon, M.; Gleva, M. J. Comparison of diagnostic value using a small, single channel, P-wave centric sternal ECG monitoring patch with a standard 3-lead Holter system over 24 hours. Am. Heart. J. 2017, 185, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Lueken, M.; Gramlich, M.; Leonhardt, S.; Marx, N.; Zink, M. D. Automated signal quality assessment of single-lead ecg recordings for early detection of silent atrial fibrillation. Sens. 2023, 23, 5618. [Google Scholar] [CrossRef] [PubMed]
- Venkataramanaiah, B.; Kamala, J. ECG signal processing and KNN classifier-based abnormality detection by VH-doctor for remote cardiac healthcare monitoring. Soft Comput. 2020, 24, 17457–17466. [Google Scholar] [CrossRef]
- Suba, S.; Fleischmann, K. E.; Schell-Chaple, H.; Prasad, P.; Marcus, G. M.; Hu, X.; Pelter, M. M. Diagnostic and prognostic significance of premature ventricular complexes in community and hospital-based participants: a scoping review. PLoS One. 2021, 16, e0261712. [Google Scholar] [CrossRef] [PubMed]
- Perez, M. V.; Mahaffey, K. W.; Hedlin, H.; Rumsfeld, J. S.; Garcia, A.; Ferris, T.; Balasubramanian, V.; Russo, A. M.; Rajmane, A.; Cheung, L.; Hung, G.; Lee, J.; Kowey, P.; Talati, N.; Gummidipundi, D. N. S. E.; Beatty, A.; Hills, M. T. H.; Desai, S.; Granger, C. B.; Desai, M.; Turakhia, M. P. Large-scale assessment of a smartwatch to identify atrial fibrillation. N. Engl. J. Med. 2019, 381, 1909–1917. [Google Scholar] [CrossRef] [PubMed]
- Lubitz, S. A.; Faranesh, A. Z.; Atlas, S. J; McManus, D. D.; Singer, D. E.; Pagoto, S.; Pantelopoulos, A.; Foulkes, A. S. Rationale and design of a large population study to validate software for the assessment of atrial fibrillation from data acquired by a consumer tracker or smartwatch: the Fitbit heart study. Am. Heart J. 2021, 238, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Mwachiro, D. M.; Baron-Lee, J.; Kates, F. R. Impact of post-discharge follow-up calls on 30-day hospital Readmissions in Neurosurgery. Glob. J. Qual. Saf. Healthc. 2019, 2, 46–52. [Google Scholar] [CrossRef]
- Edwards, P. Questionnaires in clinical trials: guidelines for optimal design and administration. Trials. 2010, 11, 1–8. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).