Submitted:
27 June 2024
Posted:
28 June 2024
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
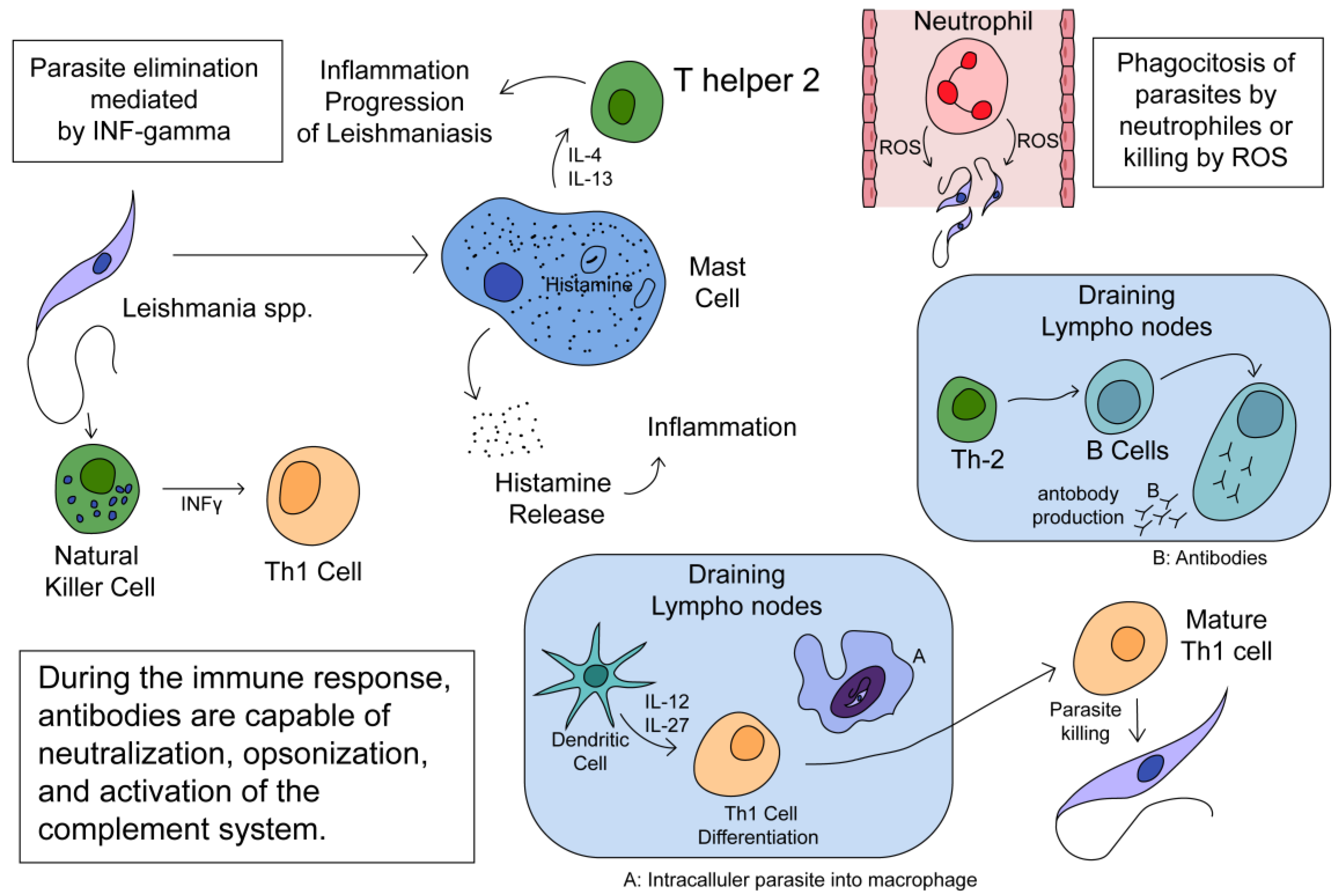
2. Immune Response to the Parasite
2.1. Immune Response in Cutaneous Leishmaniasis
2.2. Immune Response against Mucocutaneous Leishmaniasis (ML)
2.3. Immune Response in Visceral Leishmaniasis (VL)
2.4. Innate Immune Response
2.5. Inflammasome in Leishmaniasis
2.6. Leishmania Infection Induces the Production of Reactive Oxygen Species (ROS) and Nitric Oxide (NO) in Macrophages
2.7. Summary of the Immune Response to Leishmania spp.
3. Leishmaniasis Biomarkers
4. Investigation on the Effects of Treatment of Human Leishmaniasis
5. Vaccines against Leishmaniasis
| Type of vaccine | Reference |
|---|---|
| Killed parasite antigen | [63] |
| Oral immunization using live Lactococcus lactis co-expressing LACK and IL-12 | [64,65] |
| Live attenuated Centrin gene-deleted Leishmania vaccine | [66] |
| Growth-arrested Leishmania amastigotes used to develop live attenuated vaccines | [67] |
| Live attenuated L. major parasites with p27 gene deletion | [68] |
| Genetically modified live attenuated vaccines for VL | [69] |
| Recombinant protein derived from sandfly saliva against Leishmania infection | [70,71,72,73,74,75,76,77,78,79,80,81,82,83] |

- LEISHF3+ GLA-SE: A recombinant fusion protein delivered with strong Th1-inducing adjuvants.
- LeishDNAvax: A naked multi-epitope DNA vaccine.
- ChAd63-KH: An adenovirus-based vaccine.
- Live genetically attenuated vaccine: A live genetically attenuated vaccine.
6. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- von Stebut, E. Cutaneous Leishmania Infection: Progress in Pathogenesis Research and Experimental Therapy. Exp. Dermatol. 2007, 16, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Marín, M.; López, M.; Gallego-Yerga, L.; Álvarez, R.; Peláez, R. Experimental Structure Based Drug Design (SBDD) Applications for Anti-Leishmanial Drugs: A Paradigm Shift? Med. Res. Rev. 2023. [Google Scholar] [CrossRef] [PubMed]
- Pace, D. Leishmaniasis. J. Infect. 2014, 69 Suppl 1, S10–S18. [Google Scholar] [CrossRef]
- Bendelac, A.; Savage, P.B.; Teyton, L. The Biology of NKT Cells. Annu. Rev. Immunol. 2007, 25, 297–336. [Google Scholar] [CrossRef] [PubMed]
- Zamora-Chimal, J.; Hernández-Ruiz, J.; Becker, I. NKT Cells in Leishmaniasis. Immunobiology 2017, 222, 641–646. [Google Scholar] [CrossRef]
- Jain, K.; Jain, N.K. Vaccines for Visceral Leishmaniasis: A Review. J. Immunol. Methods 2015, 422, 1–12. [Google Scholar] [CrossRef]
- Veras, P.S.T.; Ramos, P.I.P.; de Menezes, J.P.B. In Search of Biomarkers for Pathogenesis and Control of Leishmaniasis by Global Analyses of Leishmania-Infected Macrophages. Front. Cell. Infect. Microbiol. 2018, 8, 326. [Google Scholar] [CrossRef]
- Seyed, N.; Taheri, T.; Rafati, S.; Taslimi, Y. Identification of Leishmania Vaccine Candidates: A Proteome-Wide Immunoinformatics Approach. Parasitol. Res. 2018. [Google Scholar] [CrossRef]
- Liese, J.; Schleicher, U.; Bogdan, C. The Innate Immune Response against Leishmania Parasites. Immunobiology 2008, 213, 377–387. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Coll, H.; Cardona-Castro, N.; Ramos, A.P.; Llanos-Cuentas, A. Innate Immune Response: Ally or Enemy in Cutaneous Leishmaniasis? Pathog. Dis. 2021, 79. [Google Scholar] [CrossRef]
- Feijó, D.; Tibúrcio, R.; Ampuero, M.; Brodskyn, C.; Tavares, N. Dendritic Cells and Leishmania Infection: Adding Layers of Complexity to a Complex Disease. J Immunol Res 2016, 2016, 3967436. [Google Scholar] [CrossRef]
- Carlsen, E.D.; Liang, Y.; Shelite, T.R.; Walker, D.H.; Melby, P.C.; Soong, L. Permissive and Protective Roles for Neutrophils in Leishmaniasis. Clin. Exp. Immunol. 2015, 182, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Volpedo, G.; Pacheco-Fernandez, T.; Bhattacharya, P.; Oljuskin, T.; Dey, R.; Gannavaram, S.; Satoskar, A.R.; Nakhasi, H.L. Determinants of Innate Immunity in Visceral Leishmaniasis and Their Implication in Vaccine Development. Front. Immunol. 2021, 12, 748325. [Google Scholar] [CrossRef] [PubMed]
- Montakhab-Yeganeh, H.; Shafiei, R.; Najm, M.; Masoori, L.; Aspatwar, A.; Badirzadeh, A. Immunogenic Properties of Empty pcDNA3 Plasmid against Zoonotic Cutaneous Leishmaniasis in Mice. PLoS One 2022, 17, e0263993. [Google Scholar] [CrossRef] [PubMed]
- Badirzadeh, A.; Heidari-Kharaji, M.; Fallah-Omrani, V.; Dabiri, H.; Araghi, A.; Salimi Chirani, A. Antileishmanial Activity of Urtica Dioica Extract against Zoonotic Cutaneous Leishmaniasis. PLoS Negl. Trop. Dis. 2020, 14, e0007843. [Google Scholar] [CrossRef] [PubMed]
- Gomes-Silva, A.; de Cássia Bittar, R.; Dos Santos Nogueira, R.; Amato, V.S.; da Silva Mattos, M.; Oliveira-Neto, M.P.; Coutinho, S.G.; Da-Cruz, A.M. Can Interferon-Gamma and Interleukin-10 Balance Be Associated with Severity of Human Leishmania (Viannia) Braziliensis Infection? Clin. Exp. Immunol. 2007, 149, 440–444. [Google Scholar] [CrossRef]
- Olivier, M.; Zamboni, D.S. Leishmania Viannia Guyanensis, LRV1 Virus and Extracellular Vesicles: A Dangerous Trio Influencing the Faith of Immune Response during Muco-Cutaneous Leishmaniasis. Curr. Opin. Immunol. 2020, 66, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Kupani, M.; Pandey, R.K.; Mehrotra, S. Neutrophils and Visceral Leishmaniasis: Impact on Innate Immune Response and Cross-Talks with Macrophages and Dendritic Cells. J. Cell. Physiol. 2021, 236, 2255–2267. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, P.; Ali, N. Involvement and Interactions of Different Immune Cells and Their Cytokines in Human Visceral Leishmaniasis. Rev. Soc. Bras. Med. Trop. 2013, 46, 128–134. [Google Scholar] [CrossRef]
- Dayakar, A.; Chandrasekaran, S.; Kuchipudi, S.V.; Kalangi, S.K. Cytokines: Key Determinants of Resistance or Disease Progression in Visceral Leishmaniasis: Opportunities for Novel Diagnostics and Immunotherapy. Front. Immunol. 2019, 10, 670. [Google Scholar] [CrossRef]
- Regli, I.B.; Passelli, K.; Hurrell, B.P.; Tacchini-Cottier, F. Survival Mechanisms Used by Some Leishmania Species to Escape Neutrophil Killing. Front. Immunol. 2017, 8, 1558. [Google Scholar] [CrossRef]
- Salei, N.; Hellberg, L.; Köhl, J.; Laskay, T. Enhanced Survival of Leishmania Major in Neutrophil Granulocytes in the Presence of Apoptotic Cells. PLoS One 2017, 12, e0171850. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.; Mandal, A.; Ansari, M.Y.; Kumar, A.; Abhishek, K.; Ghosh, A.K.; Kumar, A.; Kumar, V.; Das, S.; Das, P. Leishmania Donovani Inhibitor of Serine Peptidases 2 Mediated Inhibition of Lectin Pathway and Upregulation of C5aR Signaling Promote Parasite Survival inside Host. Front. Immunol. 2018, 9, 63. [Google Scholar] [CrossRef] [PubMed]
- Hermoso, T.; Fishelson, Z.; Becker, S.I.; Hirschberg, K.; Jaffe, C.L. Leishmanial Protein Kinases Phosphorylate Components of the Complement System. EMBO J. 1991, 10, 4061–4067. [Google Scholar] [CrossRef]
- Francesquini, F.C.; Silveira, F.T.; Passero, L.F.D.; Tomokane, T.Y.; Carvalho, A.K.; Corbett, C.E.P.; Laurenti, M.D. Salivary Gland Homogenates from Wild-Caught Sand Flies Lutzomyia Flaviscutellata and Lutzomyia (Psychodopygus) Complexus Showed Inhibitory Effects on Leishmania (Leishmania) Amazonensis and Leishmania (Viannia) Braziliensis Infection in BALB/c Mice. Int. J. Exp. Pathol. 2014, 95, 418–426. [Google Scholar] [CrossRef]
- Lestinova, T.; Vlkova, M.; Votypka, J.; Volf, P.; Rohousova, I. Phlebotomus Papatasi Exposure Cross-Protects Mice against Leishmania Major Co-Inoculated with Phlebotomus Duboscqi Salivary Gland Homogenate. Acta Trop. 2015, 144, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Thiakaki, M.; Rohousova, I.; Volfova, V.; Volf, P.; Chang, K.-P.; Soteriadou, K. Sand Fly Specificity of Saliva-Mediated Protective Immunity in Leishmania Amazonensis-BALB/c Mouse Model. Microbes Infect. 2005, 7, 760–766. [Google Scholar] [CrossRef]
- Abdeladhim, M.; Kamhawi, S.; Valenzuela, J.G. What’s behind a Sand Fly Bite? The Profound Effect of Sand Fly Saliva on Host Hemostasis, Inflammation and Immunity. Infect. Genet. Evol. 2014, 28, 691–703. [Google Scholar] [CrossRef]
- Pacheco-Fernandez, T.; Volpedo, G.; Verma, C.; Satoskar, A.R. Understanding the Immune Responses Involved in Mediating Protection or Immunopathology during Leishmaniasis. Biochem. Soc. Trans. 2021, 49, 297–311. [Google Scholar] [CrossRef]
- Charmoy, M.; Auderset, F.; Allenbach, C.; Tacchini-Cottier, F. The Prominent Role of Neutrophils during the Initial Phase of Infection by Leishmania Parasites. J. Biomed. Biotechnol. 2010, 2010, 719361. [Google Scholar] [CrossRef]
- Passelli, K.; Billion, O.; Tacchini-Cottier, F. The Impact of Neutrophil Recruitment to the Skin on the Pathology Induced by Leishmania Infection. Front. Immunol. 2021, 12, 649348. [Google Scholar] [CrossRef]
- Mohamed, A.M.; Taye, T.; Akuffo, H.O. Mechanisms of Resistance to Leishmania Aethiopica. I. Interferon-Gamma in Combination with a Cytokine (not Tumor Necrosis Factor-Alpha) Is Required, but Cannot Act Alone in the Inhibition of Intracellular Forms of L. Aethiopica in THP1 Cells. Eur. J. Immunol. 1992, 22, 2331–2337. [Google Scholar] [CrossRef] [PubMed]
- Siqueira-Neto, J.L.; Moon, S.; Jang, J.; Yang, G.; Lee, C.; Moon, H.K.; Chatelain, E.; Genovesio, A.; Cechetto, J.; Freitas-Junior, L.H. An Image-Based High-Content Screening Assay for Compounds Targeting Intracellular Leishmania Donovani Amastigotes in Human Macrophages. PLoS Negl. Trop. Dis. 2012, 6, e1671. [Google Scholar] [CrossRef] [PubMed]
- Corradin, S.B.; Buchmüller-Rouiller, Y.; Mauël, J. Phagocytosis Enhances Murine Macrophage Activation by Interferon-Gamma and Tumor Necrosis Factor-Alpha. Eur. J. Immunol. 1991, 21, 2553–2558. [Google Scholar] [CrossRef] [PubMed]
- Argueta-Donohué, J.; Wilkins-Rodríguez, A.A.; Aguirre-García, M.; Gutiérrez-Kobeh, L. Differential Phagocytosis of Leishmania Mexicana Promastigotes and Amastigotes by Monocyte-Derived Dendritic Cells. Microbiol. Immunol. 2016, 60, 369–381. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Fernández, I.; Argueta-Donohué, J.; Wilkins-Rodríguez, A.A.; Gutiérrez-Kobeh, L. Effect of Two Different Isolates of Leishmania Mexicana in the Production of Cytokines and Phagocytosis by Murine Dendritic Cells. J. Parasitol. 2019, 105, 359–370. [Google Scholar] [CrossRef] [PubMed]
- Kautz-Neu, K.; Schwonberg, K.; Fischer, M.R.; Schermann, A.I.; von Stebut, E. Dendritic Cells in Leishmania Major Infections: Mechanisms of Parasite Uptake, Cell Activation and Evidence for Physiological Relevance. Med. Microbiol. Immunol. 2012, 201, 581–592. [Google Scholar] [CrossRef]
- Filardy, A.A.; Costa-da-Silva, A.C.; Koeller, C.M.; Guimarães-Pinto, K.; Ribeiro-Gomes, F.L.; Lopes, M.F.; Heise, N.; Freire-de-Lima, C.G.; Nunes, M.P.; DosReis, G.A. Infection with Leishmania Major Induces a Cellular Stress Response in Macrophages. PLoS One 2014, 9, e85715. [Google Scholar] [CrossRef] [PubMed]
- Gurung, P.; Karki, R.; Vogel, P.; Watanabe, M.; Bix, M.; Lamkanfi, M.; Kanneganti, T.-D. An NLRP3 Inflammasome-Triggered Th2-Biased Adaptive Immune Response Promotes Leishmaniasis. J. Clin. Invest. 2015, 125, 1329–1338. [Google Scholar] [CrossRef]
- Paget, C.; Doz-Deblauwe, E.; Winter, N.; Briard, B. Specific NLRP3 Inflammasome Assembling and Regulation in Neutrophils: Relevance in Inflammatory and Infectious Diseases. Cells 2022, 11. [Google Scholar] [CrossRef]
- Meyers, A.K.; Zhu, X. The NLRP3 Inflammasome: Metabolic Regulation and Contribution to Inflammaging. Cells 2020, 9. [Google Scholar] [CrossRef]
- Raneros, A.B.; Bernet, C.R.; Flórez, A.B.; Suarez-Alvarez, B. An Epigenetic Insight into NLRP3 Inflammasome Activation in Inflammation-Related Processes. Biomedicines 2021, 9. [Google Scholar] [CrossRef] [PubMed]
- Wanasen, N.; Soong, L. L-Arginine Metabolism and Its Impact on Host Immunity against Leishmania Infection. Immunol. Res. 2008, 41, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Gandarilla, J.A.; Osorio-Trujillo, C.; Hernández-Ramírez, V.I. Leishmanicidal Activity of NO and H2O2 Alone or in Combination against Leishmania Mexicana. Parasitol. Res. 2013. [Google Scholar]
- Bedard, K.; Krause, K.-H. The NOX Family of ROS-Generating NADPH Oxidases: Physiology and Pathophysiology. Physiol. Rev. 2007, 87, 245–313. [Google Scholar] [CrossRef] [PubMed]
- Nathan, C.; Cunningham-Bussel, A. Beyond Oxidative Stress: An Immunologist’s Guide to Reactive Oxygen Species. Nat. Rev. Immunol. 2013, 13, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Bogdan, C. Nitric Oxide and the Immune Response. Nat. Immunol. 2001, 2, 907–916. [Google Scholar] [CrossRef] [PubMed]
- Antoine, J.C.; Prina, E.; Jouanne, C.; Bongrand, P. Parasitophorous Vacuoles of Leishmania Amazonensis-Infected Macrophages Maintain an Acidic pH. Infect. Immun. 1990, 58, 779–787. [Google Scholar] [CrossRef] [PubMed]
- Nandan, D.; Reiner, N.E. Attenuation of Gamma Interferon-Induced Tyrosine Phosphorylation in Mononuclear Phagocytes Infected with Leishmania Donovani: Selective Inhibition of Signaling through Janus Kinases and Stat1. Infect. Immun. 1995, 63, 4495–4500. [Google Scholar] [CrossRef] [PubMed]
- Green, S.J.; Meltzer, M.S.; Hibbs, J.B., Jr; Nacy, C.A. Activated Macrophages Destroy Intracellular Leishmania Major Amastigotes by an L-Arginine-Dependent Killing Mechanism. J. Immunol. 1990, 144, 278–283. [Google Scholar] [CrossRef]
- Assreuy, J.; Cunha, F.Q.; Epperlein, M.; Noronha-Dutra, A.; O’Donnell, C.A.; Liew, F.Y.; Moncada, S. Production of Nitric Oxide and Superoxide by Activated Macrophages and Killing of Leishmania Major. Eur. J. Immunol. 1994, 24, 672–676. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Gandarilla, J.A.; Osorio-Trujillo, C.; Hernández-Ramírez, V.I.; Salaiza-Suazo, N. Human Monocytes Infected with Leishmania Mexicana Are Primed for IL-12p70 Production. Parasite Immunology 2011, 33, 643–651. [Google Scholar]
- Vieira, L.Q.; Goldschmidt, M.; Nashleanas, M.; Pfeffer, K.; Mak, T. Association of LMP-7 with MHC Class I Molecules Accounts for a Maturation-Dependent Increase in Their Peptide Presentation in Leishmania Amazonensis-Infected Cells. European Journal of Immunology 1996, 26, 1001–1010. [Google Scholar]
- Mohapatra, S. Drug Resistance in Leishmaniasis: Newer Developments. Trop. Parasitol. 2014, 4, 4–9. [Google Scholar] [PubMed]
- Mukbel, R.M.; Patten, C., Jr; Gibson, K.; Ghosh, M.; Petersen, C.; Jones, D.E. Macrophage Killing of Leishmania Amazonensis Amastigotes Requires Both Nitric Oxide and Superoxide. Am. J. Trop. Med. Hyg. 2007, 76, 669–675. [Google Scholar] [CrossRef] [PubMed]
- Farias, L.H.S.; Rodrigues, A.P.D.; Silveira, F.T. New Approaches on Leishmania Therapy and Prevention Based on Understanding of Leishmania Interactions within the Mammalian Host. Veterinary Parasitology 2018, 258, 54–64. [Google Scholar]
- Conde, L.; Maciel, G.; de Assis, G.M.; Freire-de-Lima, L.; Nico, D.; Vale, A.; Freire-de-Lima, C.G.; Morrot, A. Humoral Response in Leishmaniasis. Front. Cell. Infect. Microbiol. 2022, 12, 1063291. [Google Scholar] [CrossRef] [PubMed]
- Volpedo, G.; Pacheco-Fernandez, T.; Holcomb, E.A.; Cipriano, N.; Cox, B.; Satoskar, A.R. Mechanisms of Immunopathogenesis in Cutaneous Leishmaniasis And Post Kala-Azar Dermal Leishmaniasis (PKDL). Front. Cell. Infect. Microbiol. 2021, 11, 685296. [Google Scholar] [CrossRef]
- de Mesquita, T.G.R.; Junior, J.d.E.S.; da Silva, L.D.O.; Silva, G.A.V.; de Araújo, F.J.; Pinheiro, S.K.; Kerr, H.K.A.; da Silva, L.S.; de Souza, L.M.; de Almeida, S.A.; et al. Distinct Plasma Chemokines and Cytokines Signatures in Leishmania Guyanensis-Infected Patients with Cutaneous Leishmaniasis. Front. Immunol. 2022, 13, 974051. [Google Scholar] [CrossRef] [PubMed]
- Giunchetti, R.C.; Silveira, P.; Resende, L.A.; Leite, J.C.; Melo-Júnior, O.A.d.O.; Rodrigues-Alves, M.L.; Costa, L.M.; Lair, D.F.; Chaves, V.R.; Soares, I.D.S.; et al. Canine Visceral Leishmaniasis Biomarkers and Their Employment in Vaccines. Vet. Parasitol. 2019, 271, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Cuna, W.R.; Velasquez, R.; Riva, J.; Guachalla, I.; Rodríguez, C. Enhancement of a TH1 Immune Response in Amphotericin B-Treated Mucocutaneous Leishmaniasis. J. Biomed. Biotechnol. 2007, 2007, 96410. [Google Scholar] [CrossRef]
- Gannavaram, S.; Bhattacharya, P.; Siddiqui, A.; Ismail, N.; Madhavan, S.; Nakhasi, H.L. miR-21 Expression Determines the Early Vaccine Immunity Induced by LdCen-/- Immunization. Front. Immunol. 2019, 10, 2273. [Google Scholar] [CrossRef] [PubMed]
- Mendonça, S.C.F. Differences in Immune Responses against Leishmania Induced by Infection and by Immunization with Killed Parasite Antigen: Implications for Vaccine Discovery. Parasit. Vectors 2016, 9, 492. [Google Scholar] [CrossRef] [PubMed]
- Hugentobler, F.; Di Roberto, R.B.; Gillard, J.; Cousineau, B. Oral Immunization Using Live Lactococcus Lactis Co-Expressing LACK and IL-12 Protects BALB/c Mice against Leishmania Major Infection. Vaccine 2012, 30, 5726–5732. [Google Scholar] [CrossRef] [PubMed]
- Hugentobler, F.; Yam, K.K.; Gillard, J.; Mahbuba, R.; Olivier, M.; Cousineau, B. Immunization against Leishmania Major Infection Using LACK- and IL-12-Expressing Lactococcus Lactis Induces Delay in Footpad Swelling. PLoS One 2012, 7, e30945. [Google Scholar] [CrossRef]
- Volpedo, G.; Bhattacharya, P.; Gannavaram, S.; Pacheco-Fernandez, T.; Oljuskin, T.; Dey, R.; Satoskar, A.R.; Nakhasi, H.L. The History of Live Attenuated Centrin Gene-Deleted Leishmania Vaccine Candidates. Pathogens 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Selvapandiyan, A.; Dey, R.; Gannavaram, S.; Solanki, S.; Salotra, P.; Nakhasi, H.L. Generation of Growth Arrested Leishmania Amastigotes: A Tool to Develop Live Attenuated Vaccine Candidates against Visceral Leishmaniasis. Vaccine 2014, 32, 3895–3901. [Google Scholar] [CrossRef] [PubMed]
- Elikaee, S.; Mohebali, M.; Rezaei, S.; Eslami, H.; Khamesipour, A.; Keshavarz, H.; Eshraghian, M.R. Leishmania Major p27 Gene Knockout as a Novel Live Attenuated Vaccine Candidate: Protective Immunity and Efficacy Evaluation against Cutaneous and Visceral Leishmaniasis in BALB/c Mice. Vaccine 2019, 37, 3221–3228. [Google Scholar] [CrossRef]
- Moreira, P.O.L.; Nogueira, P.M.; Monte-Neto, R.L. Next-Generation Leishmanization: Revisiting Molecular Targets for Selecting Genetically Engineered Live-Attenuated Leishmania. Microorganisms 2023, 11. [Google Scholar] [CrossRef]
- Zahedifard, F.; Gholami, E.; Taheri, T.; Taslimi, Y.; Doustdari, F.; Seyed, N.; Torkashvand, F.; Meneses, C.; Papadopoulou, B.; Kamhawi, S.; et al. Enhanced Protective Efficacy of Nonpathogenic Recombinant Leishmania Tarentolae Expressing Cysteine Proteinases Combined with a Sand Fly Salivary Antigen. PLoS Negl. Trop. Dis. 2014, 8, e2751. [Google Scholar] [CrossRef]
- Valenzuela, J.G.; Belkaid, Y.; Garfield, M.K.; Mendez, S.; Kamhawi, S.; Rowton, E.D.; Sacks, D.L.; Ribeiro, J.M. Toward a Defined Anti-Leishmania Vaccine Targeting Vector Antigens: Characterization of a Protective Salivary Protein. J. Exp. Med. 2001, 194, 331–342. [Google Scholar] [CrossRef]
- Gomes, R.; Teixeira, C.; Teixeira, M.J.; Oliveira, F.; Menezes, M.J.; Silva, C.; de Oliveira, C.I.; Miranda, J.C.; Elnaiem, D.-E.; Kamhawi, S.; et al. Immunity to a Salivary Protein of a Sand Fly Vector Protects against the Fatal Outcome of Visceral Leishmaniasis in a Hamster Model. Proc. Natl. Acad. Sci. U. S. A. 2008, 105, 7845–7850. [Google Scholar] [CrossRef] [PubMed]
- Collin, N.; Gomes, R.; Teixeira, C.; Cheng, L.; Laughinghouse, A.; Ward, J.M.; Elnaiem, D.-E.; Fischer, L.; Valenzuela, J.G.; Kamhawi, S. Sand Fly Salivary Proteins Induce Strong Cellular Immunity in a Natural Reservoir of Visceral Leishmaniasis with Adverse Consequences for Leishmania. PLoS Pathog. 2009, 5, e1000441. [Google Scholar] [CrossRef] [PubMed]
- Tavares, N.M.; Silva, R.A.; Costa, D.J.; Pitombo, M.A.; Fukutani, K.F.; Miranda, J.C.; Valenzuela, J.G.; Barral, A.; de Oliveira, C.I.; Barral-Netto, M.; et al. Lutzomyia Longipalpis Saliva or Salivary Protein LJM19 Protects against Leishmania Braziliensis and the Saliva of Its Vector, Lutzomyia Intermedia. PLoS Negl. Trop. Dis. 2011, 5, e1169. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Oliveira, F.; Chang, B.W.; Collin, N.; Gomes, R.; Teixeira, C.; Reynoso, D.; my Pham, V.; Elnaiem, D.-E.; Kamhawi, S.; et al. Structure and Function of a “Yellow” Protein from Saliva of the Sand Fly Lutzomyia Longipalpis That Confers Protective Immunity against Leishmania Major Infection*. J. Biol. Chem. 2011, 286, 32383–32393. [Google Scholar] [CrossRef]
- Martin-Martin, I.; Chagas, A.C.; Guimaraes-Costa, A.B.; Amo, L.; Oliveira, F.; Moore, I.N.; DeSouza-Vieira, T.S.; Sanchez, E.E.; Suntravat, M.; Valenzuela, J.G.; et al. Immunity to LuloHya and Lundep, the Salivary Spreading Factors from Lutzomyia Longipalpis, Protects against Leishmania Major Infection. PLoS Pathog. 2018, 14, e1007006. [Google Scholar] [CrossRef] [PubMed]
- Lerner, E.A.; Ribeiro, J.M.; Nelson, R.J.; Lerner, M.R. Isolation of Maxadilan, a Potent Vasodilatory Peptide from the Salivary Glands of the Sand Fly Lutzomyia Longipalpis. J. Biol. Chem. 1991, 266, 11234–11236. [Google Scholar] [CrossRef] [PubMed]
- Gomes, R.; Oliveira, F.; Teixeira, C.; Meneses, C.; Gilmore, D.C.; Elnaiem, D.-E.; Kamhawi, S.; Valenzuela, J.G. Immunity to Sand Fly Salivary Protein LJM11 Modulates Host Response to Vector-Transmitted Leishmania Conferring Ulcer-Free Protection. J. Invest. Dermatol. 2012, 132, 2735–2743. [Google Scholar] [CrossRef]
- Oliveira, F.; Rowton, E.; Aslan, H.; Gomes, R.; Castrovinci, P.A.; Alvarenga, P.H.; Abdeladhim, M.; Teixeira, C.; Meneses, C.; Kleeman, L.T.; et al. A Sand Fly Salivary Protein Vaccine Shows Efficacy against Vector-Transmitted Cutaneous Leishmaniasis in Nonhuman Primates. Sci. Transl. Med. 2015, 7, 290ra90. [Google Scholar] [CrossRef]
- Cunha, J.M.; Abbehusen, M.; Suarez, M.; Valenzuela, J.; Teixeira, C.R.; Brodskyn, C.I. Immunization with LJM11 Salivary Protein Protects against Infection with Leishmania Braziliensis in the Presence of Lutzomyia Longipalpis Saliva. Acta Trop. 2018, 177, 164–170. [Google Scholar] [CrossRef]
- Gholami, E.; Oliveira, F.; Taheri, T.; Seyed, N.; Gharibzadeh, S.; Gholami, N.; Mizbani, A.; Zali, F.; Habibzadeh, S.; Bakhadj, D.O.; et al. DNA Plasmid Coding for Phlebotomus Sergenti Salivary Protein PsSP9, a Member of the SP15 Family of Proteins, Protects against Leishmania Tropica. PLoS Negl. Trop. Dis. 2019, 13, e0007067. [Google Scholar] [CrossRef]
- Chagas, A.C.; Oliveira, F.; Debrabant, A.; Valenzuela, J.G.; Ribeiro, J.M.C.; Calvo, E. Lundep, a Sand Fly Salivary Endonuclease Increases Leishmania Parasite Survival in Neutrophils and Inhibits XIIa Contact Activation in Human Plasma. PLoS Pathog. 2014, 10, e1003923. [Google Scholar] [CrossRef] [PubMed]
- Morris, R.V.; Shoemaker, C.B.; David, J.R.; Lanzaro, G.C.; Titus, R.G. Sandfly Maxadilan Exacerbates Infection with Leishmania Major and Vaccinating against It Protects against L. Major Infection. J. Immunol. 2001, 167, 5226–5230. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, P.; Dey, R.; Saxena, A.; Karmakar, S.; Ismail, N.; Gannavaram, S.; Dagur, P.K.; Satoskar, M.; Satoskar, S.; De Paoli, S.; et al. Essential Role of Neutrophils in the Protective Immune Response Induced by a Live Attenuated Leishmania Vaccine. J. Immunol. 2020, 205, 3333–3347. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, P.; Ismail, N.; Saxena, A.; Gannavaram, S.; Dey, R.; Oljuskin, T.; Akue, A.; Takeda, K.; Yu, J.; Karmakar, S.; et al. Neutrophil-Dendritic Cell Interaction Plays an Important Role in Live Attenuated Leishmania Vaccine Induced Immunity. PLoS Negl. Trop. Dis. 2022, 16, e0010224. [Google Scholar] [CrossRef]
- Gannavaram, S.; Dey, R.; Avishek, K.; Selvapandiyan, A.; Salotra, P.; Nakhasi, H.L. Biomarkers of Safety and Immune Protection for Genetically Modified Live Attenuated Leishmania Vaccines against Visceral Leishmaniasis - Discovery and Implications. Front. Immunol. 2014, 5, 241. [Google Scholar] [CrossRef] [PubMed]
- Fiuza, J.A.; Gannavaram, S.; Santiago, H. da C.; Selvapandiyan, A.; Souza, D.M.; Passos, L.S.A.; de Mendonça, L.Z.; Lemos-Giunchetti, D. da S.; Ricci, N.D.; Bartholomeu, D.C.; et al. Vaccination Using Live Attenuated Leishmania Donovani Centrin Deleted Parasites Induces Protection in Dogs against Leishmania Infantum. Vaccine 2015, 33, 280–288. [Google Scholar] [PubMed]
- Pandey, S.C.; Kumar, A.; Samant, M. Genetically Modified Live Attenuated Vaccine: A Potential Strategy to Combat Visceral Leishmaniasis. Parasite Immunol. 2020, 42, e12732. [Google Scholar] [CrossRef]
- Fernandes, A.P.; Coelho, E.A.F.; Machado-Coelho, G.L.L.; Grimaldi, G., Jr; Gazzinelli, R.T. Making an Anti-Amastigote Vaccine for Visceral Leishmaniasis: Rational, Update and Perspectives. Curr. Opin. Microbiol. 2012, 15, 476–485. [Google Scholar] [CrossRef]
- Ismail, N.; Karmakar, S.; Bhattacharya, P.; Sepahpour, T.; Takeda, K.; Hamano, S.; Matlashewski, G.; Satoskar, A.R.; Gannavaram, S.; Dey, R.; et al. Leishmania Major Centrin Gene-Deleted Parasites Generate Skin Resident Memory T-Cell Immune Response Analogous to Leishmanization. Front. Immunol. 2022, 13, 864031. [Google Scholar] [CrossRef]
- Seyed, N.; Rafati, S. Th1 Concomitant Immune Response Mediated by IFN-γ Protects against Sand Fly Delivered Leishmania Infection: Implications for Vaccine Design. Cytokine 2021, 147, 155247. [Google Scholar] [CrossRef]
- Zhang, W.-W.; Karmakar, S.; Gannavaram, S.; Dey, R.; Lypaczewski, P.; Ismail, N.; Siddiqui, A.; Simonyan, V.; Oliveira, F.; Coutinho-Abreu, I.V.; et al. A Second Generation Leishmanization Vaccine with a Markerless Attenuated Leishmania Major Strain Using CRISPR Gene Editing. Nat. Commun. 2020, 11, 3461. [Google Scholar] [CrossRef]
- Peters, N.C.; Kimblin, N.; Secundino, N.; Kamhawi, S.; Lawyer, P.; Sacks, D.L. Vector Transmission of Leishmania Abrogates Vaccine-Induced Protective Immunity. PLoS Pathog. 2009, 5, e1000484. [Google Scholar] [CrossRef] [PubMed]
- Seyed, N.; Peters, N.C.; Rafati, S. Translating Observations From Leishmanization Into Non-Living Vaccines: The Potential of Dendritic Cell-Based Vaccination Strategies Against Leishmania. Front. Immunol. 2018, 9, 1227. [Google Scholar] [CrossRef] [PubMed]
- Almeida, A.P.M.M.; Machado, L.F.M.; Doro, D.; Nascimento, F.C.; Damasceno, L.; Gazzinelli, R.T.; Fernandes, A.P.; Junqueira, C. New Vaccine Formulations Containing a Modified Version of the Amastigote 2 Antigen and the Non-Virulent Trypanosoma Cruzi CL-14 Strain Are Highly Antigenic and Protective against Leishmania Infantum Challenge. Front. Immunol. 2018, 9, 465. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, E.M.; Bacellar, O.; Brownell, C.; Regis, T.; Coffman, R.L.; Reed, S.G. Restoration of IFN-Gamma Production and Lymphocyte Proliferation in Visceral Leishmaniasis. J. Immunol. 1994, 152, 5949–5956. [Google Scholar] [CrossRef]
- Taylor, A.P.; Murray, H.W. Intracellular Antimicrobial Activity in the Absence of Interferon-Gamma: Effect of Interleukin-12 in Experimental Visceral Leishmaniasis in Interferon-Gamma Gene-Disrupted Mice. J. Exp. Med. 1997, 185, 1231–1239. [Google Scholar] [CrossRef]
- Nathan, C.F.; Hibbs, J.B., Jr. Role of Nitric Oxide Synthesis in Macrophage Antimicrobial Activity. Curr. Opin. Immunol. 1991, 3, 65–70. [Google Scholar] [CrossRef]
- Hart, P.H.; Whitty, G.A.; Piccoli, D.S.; Hamilton, J.A. Control by IFN-Gamma and PGE2 of TNF Alpha and IL-1 Production by Human Monocytes. Immunology 1989, 66, 376–383. [Google Scholar]
- de Waal Malefyt, R.; Haanen, J.; Spits, H.; Roncarolo, M.G.; te Velde, A.; Figdor, C.; Johnson, K.; Kastelein, R.; Yssel, H.; de Vries, J.E. Interleukin 10 (IL-10) and Viral IL-10 Strongly Reduce Antigen-Specific Human T Cell Proliferation by Diminishing the Antigen-Presenting Capacity of Monocytes via Downregulation of Class II Major Histocompatibility Complex Expression. J. Exp. Med. 1991, 174, 915–924. [Google Scholar] [CrossRef]
- Kelly, C.D.; Welte, K.; Murray, H.W. Antigen-Induced Human Interferon-Gamma Production. Differential Dependence on Interleukin 2 and Its Receptor. J. Immunol. 1987, 139, 2325–2328. [Google Scholar] [CrossRef]
- Smith, K.A. Interleukin-2: Inception, Impact, and Implications. Science 1988, 240, 1169–1176. [Google Scholar] [CrossRef] [PubMed]
- Titus, R.G.; Sherry, B.; Cerami, A. Tumor Necrosis Factor Plays a Protective Role in Experimental Murine Cutaneous Leishmaniasis. J. Exp. Med. 1989, 170, 2097–2104. [Google Scholar] [CrossRef] [PubMed]
- Manetti, R.; Parronchi, P.; Giudizi, M.G.; Piccinni, M.P.; Maggi, E.; Trinchieri, G.; Romagnani, S. Natural Killer Cell Stimulatory Factor (interleukin 12 [IL-12]) Induces T Helper Type 1 (Th1)-Specific Immune Responses and Inhibits the Development of IL-4-Producing Th Cells. J. Exp. Med. 1993, 177, 1199–1204. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T.R. Cytokine Secretion Patterns and Cross-Regulation of T Cell Subsets. Immunol. Res. 1991, 10, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Holaday, B.J.; Pompeu, M.M.; Jeronimo, S.; Texeira, M.J.; Sousa, A. de A.; Vasconcelos, A.W.; Pearson, R.D.; Abrams, J.S.; Locksley, R.M. Potential Role for Interleukin-10 in the Immunosuppression Associated with Kala Azar. J. Clin. Invest. 1993, 92, 2626–2632. [Google Scholar] [CrossRef]
- Corraliza, I.M.; Soler, G.; Eichmann, K.; Modolell, M. Arginase Induction by Suppressors of Nitric Oxide Synthesis (IL-4, IL-10 and PGE2) in Murine Bone-Marrow-Derived Macrophages. Biochem. Biophys. Res. Commun. 1995, 206, 667–673. [Google Scholar] [CrossRef] [PubMed]
- Omer, F.M.; Kurtzhals, J.A.; Riley, E.M. Maintaining the Immunological Balance in Parasitic Infections: A Role for TGF-Beta? Parasitol. Today 2000, 16, 18–23. [Google Scholar] [CrossRef]
- Iniesta, V.; Gómez-Nieto, L.C.; Corraliza, I. The Inhibition of Arginase by N(omega)-Hydroxy-L-Arginine Controls the Growth of Leishmania inside Macrophages. J. Exp. Med. 2001, 193, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Brombacher, F. The Role of Interleukin-13 in Infectious Diseases and Allergy. Bioessays 2000, 22, 646–656. [Google Scholar] [CrossRef]
- Tripathi, P.; Singh, V.; Naik, S. Immune Response to Leishmania: Paradox rather than Paradigm. FEMS Immunol. Med. Microbiol. 2007, 51, 229–242. [Google Scholar] [CrossRef]
- Gomez, M.A.; Contreras, I.; Hallé, M.; Tremblay, M.L.; McMaster, R.W.; Olivier, M. Leishmania GP63 Alters Host Signaling through Cleavage-Activated Protein Tyrosine Phosphatases. Sci. Signal. 2009, 2, ra58. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, S.; Ghosh, S.; Jhonson, P.L.; Bhattacharya, S.K.; Majumdar, S. Immunomodulatory Role of Interleukin-10 in Visceral Leishmaniasis: Defective Activation of Protein Kinase C-Mediated Signal Transduction Events. Infect. Immun. 2001, 69, 1499–1507. [Google Scholar] [CrossRef]
- Dos Santos, P.L.; de Oliveira, F.A.; Santos, M.L.B.; Cunha, L.C.S.; Lino, M.T.B.; de Oliveira, M.F.S.; Bomfim, M.O.M.; Silva, A.M.; de Moura, T.R.; de Jesus, A.R.; et al. The Severity of Visceral Leishmaniasis Correlates with Elevated Levels of Serum IL-6, IL-27 and sCD14. PLoS Negl. Trop. Dis. 2016, 10, e0004375. [Google Scholar] [CrossRef] [PubMed]
- Handman, E. Leishmaniasis: Current Status of Vaccine Development. Clin. Microbiol. Rev. 2001, 14, 229–243. [Google Scholar] [CrossRef]
- Babaloo, Z.; Kaye, P.M.; Eslami, M.B. Interleukin-13 in Iranian Patients with Visceral Leishmaniasis: Relationship to Other Th2 and Th1 Cytokines. Trans. R. Soc. Trop. Med. Hyg. 2001, 95, 85–88. [Google Scholar] [CrossRef] [PubMed]
- d’Ettorre, G.; Ceccarelli, G.; Carnevalini, M.; Forcina, G.; Zaffiri, L.; Massetti, A.P.; Mastroianni, C.M.; Vullo, V. Central Role of Interleukin-15 in Human Immunodeficiency Virus (HIV)-Infected Patients with Visceral Leishmaniasis. Acta Trop. 2006, 99, 83–87. [Google Scholar] [CrossRef]
- Munger, W.; DeJoy, S.Q.; Jeyaseelan, R., Sr; Torley, L.W.; Grabstein, K.H.; Eisenmann, J.; Paxton, R.; Cox, T.; Wick, M.M.; Kerwar, S.S. Studies Evaluating the Antitumor Activity and Toxicity of Interleukin-15, a New T Cell Growth Factor: Comparison with Interleukin-2. Cell. Immunol. 1995, 165, 289–293. [Google Scholar] [CrossRef]
- Bacellar, O.; Brodskyn, C.; Guerreiro, J.; Barral-Netto, M.; Costa, C.H.; Coffman, R.L.; Johnson, W.D.; Carvalho, E.M. Interleukin-12 Restores Interferon-Gamma Production and Cytotoxic Responses in Visceral Leishmaniasis. J. Infect. Dis. 1996, 173, 1515–1518. [Google Scholar] [CrossRef]
- Coutinho De Oliveira, B.; Duthie, M.S.; Alves Pereira, V.R. Vaccines for Leishmaniasis and the Implications of Their Development for American Tegumentary Leishmaniasis. Hum. Vaccin. Immunother. 2020, 16, 919–930. [Google Scholar] [CrossRef]
- Sacks, D.; Kamhawi, S. Molecular Aspects of Parasite-Vector and Vector-Host Interactions in Leishmaniasis. Annu. Rev. Microbiol. 2001, 55, 453–483. [Google Scholar] [CrossRef]
- Melikishvili, M. KEY IMMUNOLOGICAL CONSIDERATIONS FOR LEISHMANIASIS VACCINE DEVELOPMENT. 2023. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).