Submitted:
24 May 2024
Posted:
27 May 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
- L. fermentum U-21 (collection number VKPM V-12075, NCBI Genome assembly ASM286982v2);
- E. coli SG20250 (ΔlacU169 araD flbB relA clpB+), and its insertion derivative SG22100 clpB::kan- (kindly provided by S. Gottesman) [24];
- E. coli XL1-Blue (recA1 endA1 gyrA96 thi-1 hsdR17 supE44 relA1 lac [F’proAB lacIqZΔM15 Tn10 (Tetr)]) (Stratagene, USA);
- E. coli BL21-Gold (DE3) (F– ompT dcm+ TetR gal lon hsdSB(rB– mB–) λ(DE3 [lacI lacUV5-T7p07 ind1sam7 nin5]) used for the biosynthesis of luciferase LuxAB and NADH-FMN oxidoreductases LuxG (obtained from VKPM);
- E. coli TG1 (thi relA supE44 hsdR17 hsdM Δ(lacproAB) [F’traD36 proAB lacIqZ ΔM15]) for plasmid preparation (obtained from VKPM).
Cultivation Conditions
In vivo Luminescence Measurement
Biosynthesis, Isolation, and Purification of P. luminescens Luciferase and V. aquamarinus LuxG
Spent L. fermentum U-21 Culture Medium
Measurement of P. luminescens Luciferase Activity In Vitro
Phylogenetic and Molecular Evolutionary Analysis
Results
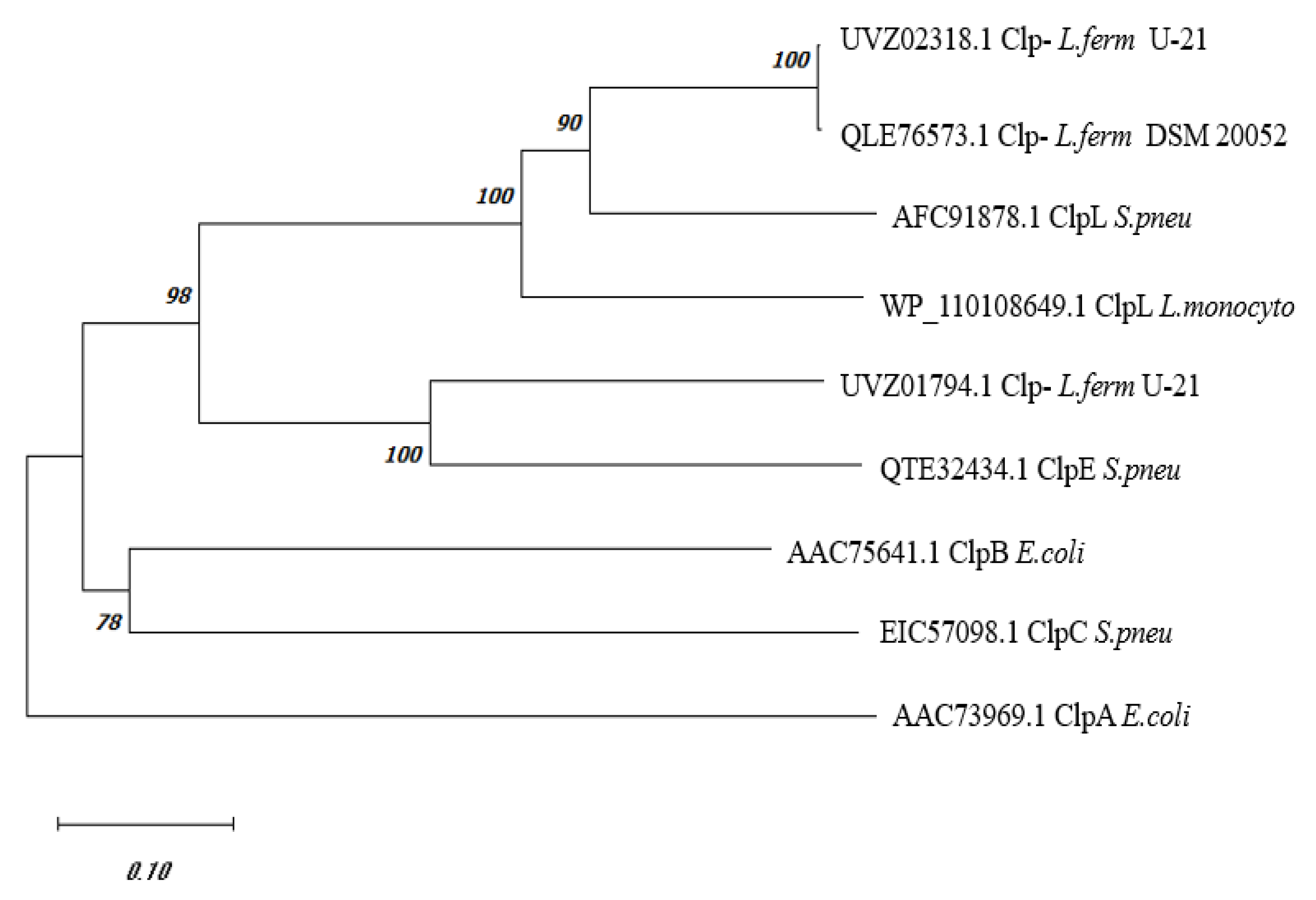
Phylogenetic Analysis of C0965_000195 L. fermentum U-21
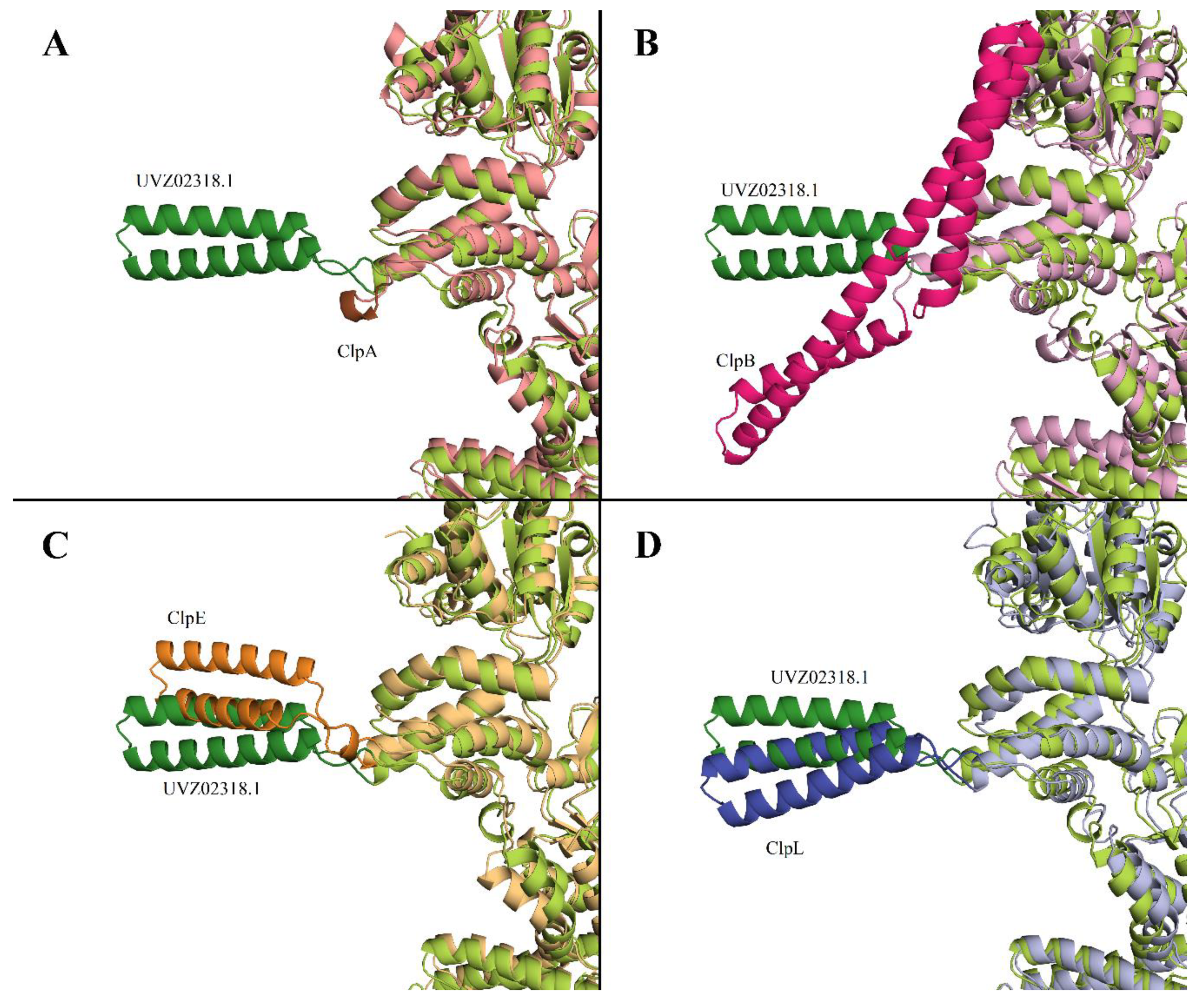
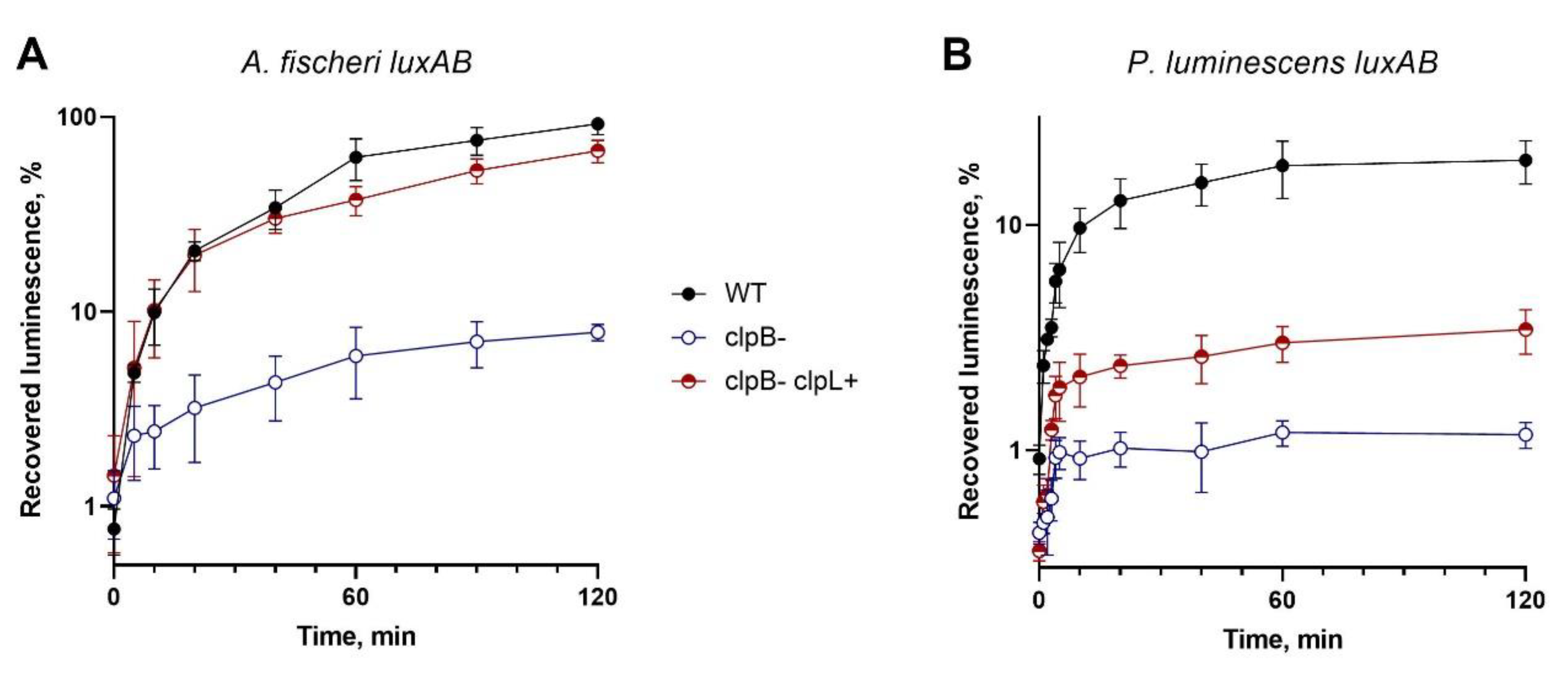
Gene Cloning and Expression, and Chaperone Activity Investigation of the L. fermentum U-21 ClpL Protein in E. coli Cells
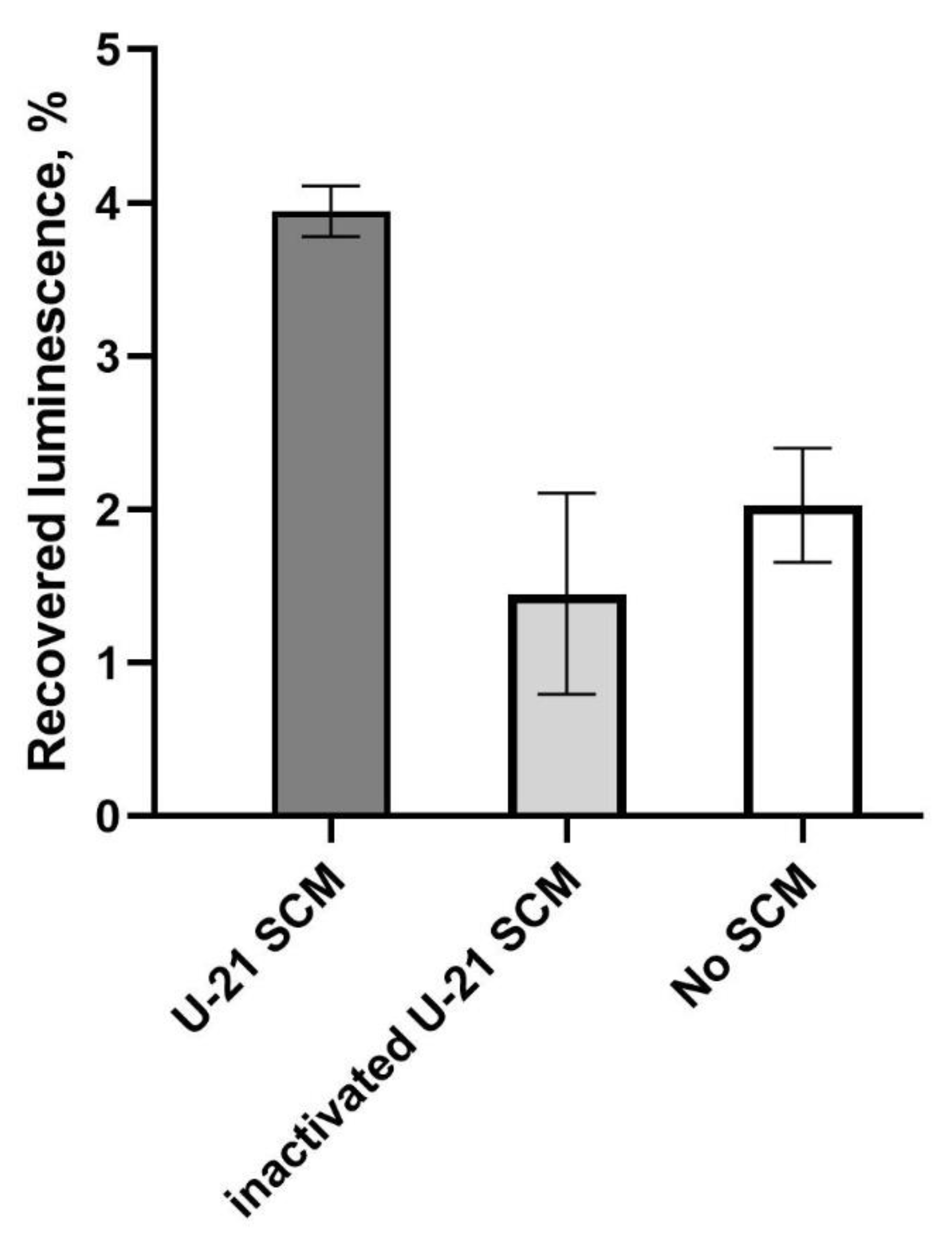
Investigation of the Chaperone Activity of L. fermentum U-21 SCM In Vitro
Discussion
Supplementary Materials
References
- Katayama, Y.; Gottesman, S.; Pumphrey, J.; Rudikoff, S.; Clark, W.P.; Maurizi, M.R. The Two-Component, ATP-Dependent Clp Protease of Escherichia Coli. Purification, Cloning, and Mutational Analysis of the ATP-Binding Component. Journal of Biological Chemistry 1988, 263, 15226–15236. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, M.; Wada, C.; Yoshioka, S.; Yura, T. Expression of ClpB, an Analog of the ATP-Dependent Protease Regulatory Subunit in Escherichia Coli, Is Controlled by a Heat Shock Sigma Factor (Sigma 32). J Bacteriol 1991, 173, 4247–4253. [Google Scholar] [CrossRef] [PubMed]
- Hoskins, J.R.; Sharma, S.; Sathyanarayana, B.K.; Wickner, S. Clp ATPases and Their Role in Protein Unfolding and Degradation. Adv Protein Chem 2001, 59, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Wawrzynow, A.; Wojtkowiak, D.; Marszalek, J.; Banecki, B.; Jonsen, M.; Graves, B.; Georgopoulos, C.; Zylicz, M. The ClpX Heat-Shock Protein of Escherichia Coli, the ATP-Dependent Substrate Specificity Component of the ClpP-ClpX Protease, Is a Novel Molecular Chaperone. EMBO J 1995, 14, 1867–1877. [Google Scholar] [CrossRef] [PubMed]
- Wickner, S.; Gottesman, S.; Skowyra, D.; Hoskins, J.; McKenney, K.; Maurizi, M.R. A Molecular Chaperone, ClpA, Functions like DnaK and DnaJ. Proc Natl Acad Sci U S A 1994, 91, 12218. [Google Scholar] [CrossRef] [PubMed]
- Weber-Ban, E.U.; Reid, B.G.; Miranker, A.D.; Horwich, A.L. Global Unfolding of a Substrate Protein by the Hsp100 Chaperone ClpA. Nature 1999, 401, 90–93. [Google Scholar] [CrossRef] [PubMed]
- Hoskins, J.R.; Singh, S.K.; Maurizi, M.R.; Wickner, S. Protein Binding and Unfolding by the Chaperone ClpA and Degradation by the Protease ClpAP. Proc Natl Acad Sci U S A 2000, 97, 8892–8897. [Google Scholar] [CrossRef] [PubMed]
- Gottesman, S.; Wickner, S.; Maurizi, M.R. Protein Quality Control: Triage by Chaperones and Proteases. Genes Dev 1997, 11, 815–823. [Google Scholar] [CrossRef] [PubMed]
- Motohashi, K.; Watanabe, Y.; Yohda, M.; Yoshida, M. Heat-Inactivated Proteins Are Rescued by the DnaK.J-GrpE Set and ClpB Chaperones. Proc Natl Acad Sci U S A 1999, 96, 7184–7189. [Google Scholar] [CrossRef] [PubMed]
- Zavilgelsky, G.B.; Kotova, V.Y.; Mazhul’, M.M.; Manukhov, I. V. Role of Hsp70 (DnaK-DnaJ-GrpE) and Hsp100 (ClpA and ClpB) Chaperones in Refolding and Increased Thermal Stability of Bacterial Luciferases in Escherichia Coli Cells. Biochemistry (Moscow) 2002, 67, 986–992. [Google Scholar] [CrossRef]
- Kim, G.; Lee, S.G.; Han, S.; Jung, J.; Jeong, H.S.; Hyun, J. kyung; Rhee, D.K.; Kim, H.M.; Lee, S. ClpL Is a Functionally Active Tetradecameric AAA+ Chaperone, Distinct from Hexameric/Dodecameric Ones. The FASEB Journal 2020, 34, 14353–14370. [Google Scholar] [CrossRef] [PubMed]
- Namy, O.I.; éle Mock, M.; és Fouet, A. Co-Existence of ClpB and ClpC in the Bacillaceae. FEMS Microbiol Lett 1999, 173, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Breton, J.; Legrand, R.; Akkermann, K.; Järv, A.; Harro, J.; Déchelotte, P.; Fetissov, S.O. Elevated Plasma Concentrations of Bacterial ClpB Protein in Patients with Eating Disorders. Int J Eat Disord 2016, 49, 805–808. [Google Scholar] [CrossRef] [PubMed]
- Dominique, M.; Breton, J.; Guérin, C.; Bole-Feysot, C.; Lambert, G.; Déchelotte, P.; Fetissov, S. Effects of Macronutrients on the In Vitro Production of ClpB, a Bacterial Mimetic Protein of α-MSH and Its Possible Role in Satiety Signaling. Nutrients 2019, Vol. 11, Page 2115 2019, 11, 2115. [Google Scholar] [CrossRef] [PubMed]
- Tennoune, N.; Chan, P.; Breton, J.; Legrand, R.; Chabane, Y.N.; Akkermann, K.; Järv, A.; Ouelaa, W.; Takagi, K.; Ghouzali, I.; et al. Bacterial ClpB Heat-Shock Protein, an Antigen-Mimetic of the Anorexigenic Peptide α-MSH, at the Origin of Eating Disorders. Translational Psychiatry 2014 4:10 2014, 4, e458–e458. [Google Scholar] [CrossRef] [PubMed]
- Soloveva, I. V.; Novikova, N.A.; Tochilina, A.G.; Belova, I. V.; Kashnikov, A.Y.; Sashina, T.A.; Zhirnov, V.A.; Molodtsova, S.B. The Probiotic Strain Lactobacillus Fermentum 39: Biochemical Properties, Genomic Features, and Antiviral Activity. Microbiology (Russian Federation) 2021, 90, 219–225. [Google Scholar] [CrossRef]
- Rodríguez-Sojo, M.J.; Ruiz-Malagón, A.J.; Rodríguez-Cabezas, M.E.; Gálvez, J.; Rodríguez-Nogales, A. Limosilactobacillus Fermentum CECT5716: Mechanisms and Therapeutic Insights. Nutrients 2021, 13, 1–22. [Google Scholar] [CrossRef] [PubMed]
- de Luna Freire, M.O.; Cruz Neto, J.P.R.; de Albuquerque Lemos, D.E.; de Albuquerque, T.M.R.; Garcia, E.F.; de Souza, E.L.; de Brito Alves, J.L. Limosilactobacillus Fermentum Strains as Novel Probiotic Candidates to Promote Host Health Benefits and Development of Biotherapeutics: A Comprehensive Review. Probiotics and Antimicrobial Proteins 2024 2024, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Marsova, M.; Abilev, S.; Poluektova, E.; Danilenko, V. A Bioluminescent Test System Reveals Valuable Antioxidant Properties of Lactobacillus Strains from Human Microbiota. World J Microbiol Biotechnol 2018, 34, 1–9. [Google Scholar] [CrossRef]
- Marsova, M.; Poluektova, E.; Odorskaya, M.; Ambaryan, A.; Revishchin, A.; Pavlova, G.; Danilenko, V. Protective Effects of Lactobacillus Fermentum U-21 against Paraquat-Induced Oxidative Stress in Caenorhabditis Elegans and Mouse Models. World J Microbiol Biotechnol 2020, 36, 1–10. [Google Scholar] [CrossRef]
- Danilenko, V.N.; Stavrovskaya, A. V.; Voronkov, D.N.; Gushchina, A.S.; Marsova, M. V.; Yamshchikova, N.G.; Ol’shansky, А.S.; Ivanov, M. V.; Illarioshkin, S.N. The Use of a Pharmabiotic Based on the Lactobacillus Fermentum U-21 Strain to Modulate the Neurodegenerative Process in an Experimental Model of Parkinson Disease. Annals of Clinical and Experimental Neurology 2020, 14, 62–69. [Google Scholar] [CrossRef]
- Stavrovskaya, A. V.; Danilenko, V.N.; Voronkov, D.N.; Gushchina, A.S.; Marsova, M. V.; Olshansky, A.S.; Yamshikova, N.G.; Illarioshkin, S.N. Pharmabiotic Based on Lactobacillus Fermentum Strain U-21 Modulates the Toxic Effect of 1-Methyl-4-Phenyl-1,2,3,6-Tetrahydropyridine as Parkinsonism Inducer in Mice. Hum Physiol 2021, 47, 891–900. [Google Scholar] [CrossRef]
- Poluektova, E.U.; Mavletova, D.A.; Odorskaya, M. V.; Marsova, M. V.; Klimina, K.M.; Koshenko, T.A.; Yunes, R.A.; Danilenko, V.N. Comparative Genomic, Transcriptomic, and Proteomic Analysis of the Limosilactobacillus Fermentum U-21 Strain Promising for the Creation of a Pharmabiotic. Russ J Genet 2022, 58, 1079–1090. [Google Scholar] [CrossRef]
- Gottesman, S.; Roche, E.; Zhou, Y.N.; Sauer, R.T. The ClpXP and ClpAP Proteases Degrade Proteins with Carboxy-Terminal Peptide Tails Added by the SsrA-Tagging System. Genes Dev 1998, 12, 1338–1347. [Google Scholar] [CrossRef] [PubMed]
- Bazhenov, S.; Novoyatlova, U.; Scheglova, E.; Fomin, V.; Khrulnova, S.; Melkina, O.; Chistyakov, V.; Manukhov, I. Influence of the LuxR Regulatory Gene Dosage and Expression Level on the Sensitivity of the Whole-Cell Biosensor to Acyl-Homoserine Lactone. Biosensors (Basel) 2021, 11, 166. [Google Scholar] [CrossRef]
- Zavil’gel’skiĭ, GB.; Kotova VIu; Manukhov IV. A Kinetic Method of Determining the Frequency of Homologous Recombination of Plasmids in Escherichia Coli Cells. Mol Biol (Mosk) 1994, 28, 1299–1307. [Google Scholar] [PubMed]
- Zavilgelsky GB; Zarubina AP; Manukhov IV Sequencing and Comparative Analysis of the Lux-Operon of Photorhabdus Luminescens Strain ZM1: ERIC Elements as Putative Recombination Spots. Mol Biol 2002, 36, 792–804. [CrossRef]
- Sambrook Joseph; Maccallum P; Russell David William. In Molecular Cloning A Laboratory Manual; Molecular Cloning A Laboratory Manual, 2001.
- Gibson, D.G.; Young, L.; Chuang, R.Y.; Venter, J.C.; Hutchison, C.A.; Smith, H.O. Enzymatic Assembly of DNA Molecules up to Several Hundred Kilobases. Nature Methods 2009 6:5 2009, 6, 343–345. [Google Scholar] [CrossRef]
- Nazarov, P.A.; Khrulnova, S.A.; Kessenikh, A.G.; Novoyatlova, U.S.; Kuznetsova, S.B.; Bazhenov, S. V.; Sorochkina, A.I.; Karakozova, M. V.; Manukhov, I. V. Observation of Cytotoxicity of Phosphonium Derivatives Is Explained: Metabolism Inhibition and Adhesion Alteration. Antibiotics 2023, Vol. 12, Page 720 2023, 12, 720. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Kumar, S. MEGA11: Molecular Evolutionary Genetics Analysis Version 11. Mol Biol Evol 2021, 38, 3022–3027. [Google Scholar] [CrossRef]
- Park, S.S.; Kwon, H.Y.; Tran, T.D.H.; Choi, M.H.; Jung, S.H.; Lee, S.; Briles, D.E.; Rhee, D.K. ClpL Is a Chaperone without Auxiliary Factors. FEBS Journal 2015, 282, 1352–1367. [Google Scholar] [CrossRef] [PubMed]
- Bohl, V.; Hollmann, N.M.; Melzer, T.; Katikaridis, P.; Meins, L.; Simon, B.; Flemming, D.; Sinning, I.; Hennig, J.; Mogk, A. The Listeria Monocytogenes Persistence Factor ClpL Is a Potent Stand-Alone Disaggregase. Elife 2024, 12. [Google Scholar] [CrossRef] [PubMed]
- Rotanova, T. V.; Andrianova, A.G.; Kudzhaev, A.M.; Li, M.; Botos, I.; Wlodawer, A.; Gustchina, A. New Insights into Structural and Functional Relationships between LonA Proteases and ClpB Chaperones. FEBS Open Bio 2019, 9, 1536. [Google Scholar] [CrossRef] [PubMed]
- Kotova, V.Y.; Manukhov, I. V.; Melkina, O.E.; Zavilgelsky, G.B. Mutation ClpA::Kan of the Gene for an Hsp100 Family Chaperone Impairs the DnaK-Dependent Refolding of Proteins in Escherichia Coli. Mol Biol 2008, 42, 906–910. [Google Scholar] [CrossRef]
- Zavilgelsky, G.B.; Kotova, V.Y.; Mazhul’, M.M.; Manukhov, I. V. The Effect of Clp Proteins on DnaK-Dependent Refolding of Bacterial Luciferases. Mol Biol 2004, 38, 427–433. [Google Scholar] [CrossRef]
- Huang, D.C.; Huang, X.F.; Novel, G.; Novel, M. Two Genes Present on a Transposon-like Structure in Lactococcus Lactis Are Involved in a Clp-Family Proteolytic Activity. Mol Microbiol 1993, 7, 957–965. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.Y.; Kim, S.W.; Choi, M.H.; Ogunniyi, A.D.; Paton, J.C.; Park, S.H.; Pyo, S.N.; Rhee, D.K. Effect of Heat Shock and Mutations in ClpL and ClpP on Virulence Gene Expression in Streptococcus Pneumoniae. Infect Immun 2003, 71, 3757–3765. [Google Scholar] [CrossRef] [PubMed]
- Tao, L.; Biswas, I. ClpL Is Required for Folding of CtsR in Streptococcus Mutans. J Bacteriol 2013, 195, 576. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.N.; Khan, R.H. Protein Misfolding and Related Human Diseases: A Comprehensive Review of Toxicity, Proteins Involved, and Current Therapeutic Strategies. Int J Biol Macromol 2022, 223, 143–160. [Google Scholar] [CrossRef]
- Lo, A.C.; Callaerts-Vegh, Z.; Nunes, A.F.; Rodrigues, C.M.P.; D’Hooge, R. Tauroursodeoxycholic Acid (TUDCA) Supplementation Prevents Cognitive Impairment and Amyloid Deposition in APP/PS1 Mice. Neurobiol Dis 2013, 50, 21–29. [Google Scholar] [CrossRef]
- Almeida, Z.L.; Brito, R.M.M. Amyloid Disassembly: What Can We Learn from Chaperones? Biomedicines 2022, Vol. 10, Page 3276 2022, 10, 3276. [Google Scholar] [CrossRef] [PubMed]
- Auburger, G.; Key, J.; Gispert, S. The Bacterial ClpXP-ClpB Family Is Enriched with RNA-Binding Protein Complexes. Cells 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Ojha, S.; Patil, N.; Jain, M.; Kole, C.; Kaushik, P. Probiotics for Neurodegenerative Diseases: A Systemic Review. Microorganisms 2023, Vol. 11, Page 1083 2023, 11, 1083. [Google Scholar] [CrossRef] [PubMed]
- Dhyani, P.; Goyal, C.; Dhull, S.B.; Chauhan, A.K.; Singh Saharan, B.; Harshita; Duhan, J.S.; Goksen, G. Psychobiotics for Mitigation of Neuro-Degenerative Diseases: Recent Advancements. In Mol Nutr Food Res; 2023. [Google Scholar] [CrossRef]
- Peña-Díaz, S.; García-Pardo, J.; Ventura, S. Development of Small Molecules Targeting α-Synuclein Aggregation: A Promising Strategy to Treat Parkinson’s Disease. Pharmaceutics 2023, Vol. 15, Page 839 2023, 15, 839. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.; Yu, L.; Li, Y.; Guo, H.; Zhai, Q.; Chen, W.; Tian, F. Meta-Analysis of Randomized Controlled Trials of the Effects of Probiotics in Parkinson’s Disease. Food Funct 2023, 14, 3406–3422. [Google Scholar] [CrossRef]
- Stavrovskaya, A. V; Voronkov, D.N.; Marsova, M. V; Olshansky, A.S.; Gushchina, A.S.; Danilenko, V.N.; Illarioshkin, S.N. Effects of the Pharmabiotic U-21 in a Combined Neuroinflammatory Model of Parkinson’s Disease in Rats. Bull Exp Biol Med 2024, 177, 193–199. [Google Scholar] [CrossRef]
| Name | Description | Source |
|---|---|---|
| p15Tc-lac | Expression vector. Ptac-MCS, lacI, ori-p15A, and Tcr | [25] |
| pF6 | pACYC184 vector with luxABE from A. fischeri under Plac promoter, Cmr | [26] |
| p15FisAB | p15Tc-lac with luxAB from A. fischeri under Ptac promoter, Tcr | This study |
| pXen7 | pUC18 with luxCDABE operon from P. luminescens under control of its own promoter, Apr | [27] |
| p15XenAB | p15Tc-lac with luxAB from P. luminescens under Ptac promoter, Tcr | This study |
| pUC19:clpL | pUC19 vector (Sigma-Aldrich Inc., USA) with clpL from L. fermentum U-21 under Plac promoter, Apr | This study |
| pABX-T7 | pET15b with luxAB from P. luminescens under T7 promoter, Apr | This study |
| pLuxG-T7 | pET15b with luxG from V. aquamarinus under T7 promoter, Apr | This study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




