1. Introduction
Cannabinoids constitute a group of compounds that can exert different homeostatic and pharmacological effects through the -so called- Endocannabinoid System (ECS) a network of molecular signaling made up of a group of signaling molecules, known as endocannabinoids, a group of cannabinoid receptors (CB1 mostly represented in central nervous system, and CB2, predominantly distributed in peripheral and immune cells) and some enzymes that module the synthesis, or breakdown, of the signaling molecules and their transporters [
1,
2]. It has been described by many authors the importance of cannabinoids for the potential treatment of different inflammatory disorders in which the ECS is involved at different levels [
3,
4].
It is well known nowadays the importance of the ECS in the skin due to the presence of CB1 and CB2 receptors identified in different layers of the skin, which supports the potential therapeutic uses of dermal and transdermal administration of cannabinoids for the treatment of different, chronic or occasional, inflammatory skin disorders such as psoriasis, pruritus, urticaria or atopic dermatitis [
5,
6,
7,
8,
9,
10].
Cannabidiol (CBD) is the most abundant non-psychotropic cannabinoid found in the Cannabis plant, its high safety profile shows great potential and has opened a wide range of therapeutic uses increasingly described since 1963, when its structure was first reported [
11,
12]. Concerning the mechanism of action of CBD, it is known that its anti-inflammatory effect can be ECS receptor-dependent or non-ECS receptor-dependent. For the first mechanism, CBD exerts its activity via cannabinoid receptor CB2 [
11].
Many authors have also described a significant non-ECS receptor dependent activity for CBD. Carrier
et al. [
13] suggested that CBD has the ability to enhance adenosine activity (via A2A receptors) provoking a decrease of the inflammatory activity and a down regulation of over-reactive immune cells. Bisogno
et al. [
14] described the agonist effect in TRPV1 (transient potential vanilloid receptor type 1) due to the analogy of CBD with the capsaicin, exerting similar effects (without noxious effects). Russo
et al. [
15] consider that CBD can act as an agonist to the serotonin receptor (5-HT1A) reflecting their consequent activities and they also described CBD activity as an antagonist of GPR55 receptors decreasing IL-12 and TNF-alpha production [
16,
17]. It has been also found that CBD can act through ion channels as an agonist or inhibit enzymes such as Phospholipase A2, Cyclooxygenase 1 and 2 or Fatty Acid Amide Hydrolase (FAAH), responsible for the diminution of arachidonic acid levels [
18].
All of these actions provide a strong anti-inflammatory, antioxidant and the appropriate immunological response activity seeing itself reflected in a wide variety of potential therapeutic uses [
19]. Hence, the most widely reported effects of CBD in the literature are anti-inflammatory and antioxidant [
11,
12,
16,
17,
20,
21,
22], but there exist evidences of other effects related to the treatment of skin disorders [
1,
16,
20,
21,
25], arthritis [
11,
12,
16,
17,
22,
23], diabetes [
11,
16], neurodegenerative diseases [
2,
11,
16,
22,
24] or epilepsy [
11,
16,
22,
24]. CBD has been also reported as a very promising drug for the treatment of anxiety, depression and for sleep regulation [
11,
16,
19,
20,
21,
22,
23,
24], as well as a for the treatment of chronic pain [
17,
27] and also coadyuvant for different types of cancer [
11,
19,
22,
26].
Chemically, CBD is a terpene-phenol exhibiting affinity to oil phases due to its poor solubility in water. The high lipophilicity of CBD conditions its pharmacokinetic properties. Orally administered CBD shows a very low bioavailability (around 6%) because the hepatic first-pass metabolism is very high -over 95%- [
22,
28]. There have also been reports of variable pharmacokinetic profiles and complications in the dosage forms leading to unpredictable side effects and multiple interactions with other substances since CBD is metabolized acting as a competitive inhibitor of CYP-450 enzymes [
19,
23].
The physicochemical characteristics of CBD conditions its therapeutic efficacy by different routes of administration, which has made it necessary to find some strategies to protect or/and encapsulate this active substance with the aim to improve its pharmacological activity showing little side-effects. Many attempts have been made to design an optimal formulation with CBD for topical administration, due to its lipophilicity, and the inherent potential of therapeutic use of phytocannabinoids for the treatment of skin disorders [
29,
30]. A CBD solution using propylene glycol as a vehicle showed interesting transdermal absorption in vitro, including some essential oils and oleic acid as penetration enhancers [
31]. Dimethyl sulfoxide has been also proposed as a vehicle for CBD for in vitro experiments to confirm its efficacy for the treatment of allergic contact dermatitis [
32]. This solvent can act as penetration enhancer and solubilizer but cannot be used as a main vehicle in a topical formulation due to its poor organoleptic and potentially irritant properties. Other options like the use of CBD in a shampoo have demonstrated its efficacy for the treatment of scalp psoriasis and dermatitis [
33]. Some cosmeceuticals formulated in cream or gel have also been described as effective for the topical treatment of pruritus [
34]. Zielińska and co-workers [
21] recently published an interesting review on the latest examples of formulation of CBD in lipid systems for topical administration, mainly in the form of emulsions to include any additional technologies like lipid nanoparticles. Many examples of commercially-available CBD oils registered as cosmeceuticals, or medical devices, exist as well. In the light of these products, some studies have been made in the last few years to demonstrate their therapeutic efficacy in topical treatments [
35,
36]. Although mere oils could constitute an ideal vehicle for CBD due to its lipophilicity, this simple formulation does not permit the concomitant inclusion of other hydrophilic molecules with potential activities like antioxidant, anti-inflammatory, antibiotic, etc., let alone aqueous extracts from different plants. In those cases, the use of emulsions, liposomes and any other system that permits the inclusion of oils and aqueous phases is required.
The strategy of obtaining encapsulated oils has been proposed by many authors for the elaboration of pharmaceutical formulations intended for the treatment of a variety of health problems and diseases, providing more protection against oxidation when compared to un-encapsulated oils. Microemulsions have resulted to be an effective vehicle for the solubilization of different drugs as well as a protecting medium from degradation against light exposure, oxygen, etc., preventing hydrolysis and oxidation reactions. A prolonged release of the drug can increase bioavailability across different administration routes as well as prevent irritative, or toxic effects, of the active ingredient [
21,
37,
38].
Microemulsions are defined as a transparent and isotropic dispersion of an aqueous and an oil phase structured as a bi-continuous unique phase, or a discontinuous system, at a nanometric scale (10-100 nm droplet size), stabilized by an appropriate proportion of both phases with surfactants and co-surfactants [
39,
40]. Unlike emulsions, this system has the advantage of being thermodynamically stable and easier to elaborate, since a simple stirring to mix the components of the formulation will be enough to achieve the formulation, providing the right proportion of both phases and amphiphile components is obtained. Apart from its better stability and its simplicity of elaboration, even at industrial scale, microemulsions have important advantages, showing interesting rheological properties like low viscosity [
41], the possibility of sterilization, and their capacity of solubilizing both hydrophilic and lipophilic drugs [
42,
43]. Hence, microemulsions constitute a compelling strategy for the vehiculization of many different substances by different administration routes.
In our study, a preformulation study was performed using purified Milli®-Q water as hydrophilic phase in order to accurately define the optimum composition of the microemulsion. Subsequently, water was replaced by an aqueous extract obtained from ripe fruits of Sambucus ebulus L. The final formulation was characterized using the same protocol as described for the previous microemulsion to be sure that the physicochemical properties were not altered with the inclusion of the extract. The stability of the microemulsion system was also assessed in terms of physical and chemical properties under room temperature storage conditions.
Sambucus ebulus sp. is one of the many species of
Sambucus genre. Also known as “dwarf elder”, it is located all over Europe, South Asia and lots of territories of America. This herb grows approximately 1.5-2 meters high in humidity (near rivers) and luminous areas constituting in large and extensive groups of underground rhizomes [
44,
45].
The most reported pharmacological properties are the potential antioxidant and anti-inflammatory effect of their aerial parts: fruits, leaves and flower mainly [
46,
47,
48,
49], although some authors also describe a promising anti-inflammatory activity in its rhizomes [
50,
51]. Anti-inflammatory activity is not the only reported property of
Sambucus ebulus. Jabbari
et al. in 2017 [
44] described their potential effects for the treatment of metabolic disorders, antidepressant, neuroprotective and antioxidant activity, analgesic and would healing/knee osteoarthritis improving effects, as well as antimicrobial and antigiardial activity among others.
Caffeic acid (CAF), is an hydroxycinnamic-derived acid thoroughly described by several authors. This phenolic acid is one of the compounds that can be found naturally in the extracts obtained from
Sambucus ebulus and due to its antioxidant and anti-inflammatory effect mainly [
52] it becomes an interesting compound. CAF is a hydrophilic substance that can be found mainly in extracts obtained from mature fruits [
53]. In our study, CAF was selected as a model hydro-soluble and antioxidant compound to characterize the obtained extract and the microemulsion due to its abundance in the extracts and also because CAF could be easily analyzed by fluorescence as previously described in the literature [
54] using the same chromatographic conditions selected for the analysis of CBD.
The main aim of this work is focused on the development and characterization of a new microemulsion formulation obtained from a CBD oil, including the determination of the adequate components and its proportions to obtain a stable microemulsion, compatibility studies and release characteristics, with the intent to include the maximum proportion of CBD oil concomitantly with an aqueous S. ebulus extract to reinforce the potential anti-inflammatory activity for the treatment of topical disorders, including psoriasis, etc. what could expand the possibility of new therapeutical perspectives.
To our knowledge, the nearest significant approach to this strategy was published by Vanti and co-workers [
55], who described a gel based on an O/A microemulsion loaded with 1% cannabidiol. The microemulsion developed in the present study would allow the future inclusion of different hydrophilic compounds together to CBD in a thermodynamically stable system, without modifying significantly the rheological and release properties of the original oil. The idea of reinforcing the pharmacological activity of the CBD oil with hydrophilic compounds providing a potential synergic effect, will suppose an important improvement of the therapeutic use of CBD for the treatment of different skin disorders.
2. Materials and Methods
2.1. Chemicals and Reagents
Labrasol® (PEG-8 caprylic/capric glycerides) and Plurol-Oleique ® CC497 (Polyglyceryl-3 oleate) were selected as surfactant and co-surfactant to elaborate the microemulsion formulation and were kindly supplied by Gattefosé-Spain.
Cannabidiol, 99.9% crystals, as well as Beemine CDB oil Forte+® 20% were kindly supplied by Beemine Laboratories (Madrid-Spain). The commercially-available CBD oil contained CBD in dissolution (20% w:w) and an oil mixture of hemp (majority component) and sunflower seed oil as vehicle.
Sambucus ebulus L. ripe fruits were collected (40°10′15′′N 4°24′19′′O), when optimal ripening conditions were assured, and taxonomically characterized at the MAF herbarium (assigned number MAF 176062) located at the Faculty of Pharmacy of the Complutense University of Madrid.
Fruits were cleaned, frozen and stored at -20.0 ± 0.5 ºC until use. The extract was elaborated as follows: an accurate weighed amount of thawed fruits were digested in methanol for 24 hours and filtered. The resulting methanolic solution was dried using a Buchi® R-100 rotary evaporator and a Telstar LyoQuest® freeze dryer to ensure the total elimination of liquid solvents and stored at -20.0 ± 0.5 ºC until use. The dry extract was re-dissolved in purified water before use.
All reagents and chemicals were of analytical grade. Folin-Ciocalteu′s phenol reagent was purchased from Sigma-Aldrich- Spain. Methanol, Acetonitrile and Ethanol were purchased form Fischer Chemical Spain, and were supplied as HPLC reagent grade. Milli-Q® water was used for all the experiments.
2.2. Quantification of Cannabidiol and Caffeic acid by HPLC
A previously developed and validated RP-HPLC method according to the Q2 ICH guideline was used for the simultaneous quantification of CBD and Caffeic Acid (CAF). Briefly, a JASCO modular high-performance liquid chromatograph (Jasco International Co Ltd, Japan) equipped with a LG- 2080-04 quaternary low-gradient unit, a PU-2080 pump, a DG-2080-54 degasser, an AS-2050-plus autosampler was used. The equipment had two detectors to be able to collect an absorbance signal (UV-2070 plus UV/Vis detector) and fluorescence (FP-4025 Fluorescence detector).
A chromatographic C18 column (Mediterranea® Sea, 5 µm, 150x4.6 mm) (Teknokroma S. Coop., Spain) thermostated at 40ºC in a Jasco column oven 2065 Plus was selected as stationary phase. The mobile phase consisted of Acetonitrile: Methanol: Acid water solution (adjusted to pH 4.5 with glacial acetic acid) 30:52:18, pumped at a rate of 1.8 mL·min-1. The injection volume was fixed to 20 µL and the run time for data collecting was selected to 7 min. Detection was carried out spectrophotometrically for CBD at a wavelength of 228 nm, obtaining a repetitive peak at 4.8 min. The simultaneous analysis of CAF was carried out by spectrofluorimetry selecting 262 and 426 nm for excitation and emission wavelengths respectively. A repetitive peak for Caffeic Acid at 2.1 min was obtained.
For the developed method, linearity (R2 = 0.9999) was observed between the range of 11,2–500 μg/mL. Limit of detection (LOD) and limit of quantitation (LOQ) were estimated at 3.7 and 11.2 μg/mL, respectively for CBD and 5.9 and 18.1 µg/mL for CAF. Both values were calculated from the standard deviation of the linear response and the slope of the calibration line, as described in the ICH Q2(R2) Guideline. The method was accurate and precise inter-day and intra-day (RSD of 3.35 and 3.61% respectively), with accuracy values of 100.63% for CBD and 99.7% for CAF.
2.3. Design and Preparation of the Microemulsion System
The microemulsion was elaborated using the aqueous titration method as described in previous papers [
40]. Briefly, the 20% CBD oil constituting the oily phase, was combined with different proportions of Surfactant / Co-surfactant (S/CoS) mixtures (Labrasol® and Plurol-Oleique ® CC497 respectively, 1:1 gravimetric ratio) under stirring at 30ºC for 10 minutes. The obtained mixtures were titrated with purified water under magnetic stirring at the same temperature and the resulting microemulsion was thus allowed to stabilize for 20 minutes. The composition of different mixtures obtained are shown in
Table 1. A pseudo-ternary phase diagram was elaborated to determine the microemulsion area. When idoneal microemulsion area was determined, the experiment was repeated replacing Milli-Q® water by the aqueous fruit extract previously elaborated.
2.4. Physicochemical Characterization of Microemulsion
As previously specified, the main objective of this study is focused on the inclusion of a water phase while preserving the maximum proportion of CBD oil for the future incorporation of different hydrophilic compounds with potential therapeutic uses like antioxidant, anti-inflammatory, antibiotic, etc. in a microemulsion, preserving as much as possible the original properties of the CDB oil in terms of rheological behavior, spread-ability and drug release. Based on this premise, the microemulsion containing a proportion of S-CoS : O : W of [62 / 27 / 11] containing a CBD load of 5.4 % (w:w) was selected for further characterization studies.
2.4.1. Conductivity
The measurement of electrical conductivity is widely described as an adequate method to study structural changes in biphasic systems like emulsions and microemulsions [
41]. In our study, conductivity measurements were carried out as described in a previous paper [
40]. Briefly, a METROHM® 644-Conductimeter equipped with a CRISON® conductivity cell was used under continuous stirring at a constant temperature of 30ºC, while the titration of the microemulsion by different amounts of Milli-Q® water was performed.
2.4.2. Droplet Size Distribution
A MICROTAC INC® instrument (MBT, Madrid-Spain) equipped with a Zetatrac software was used for the measurement of the mean droplet size and the size distribution by laser diffraction. All the measures were conducted in triplicate from three samples of microemulsion and emulsion, using purified water as a blank in both cases. Semi-logarithmic plots of droplet size distribution were obtained for each formulation.
2.4.3. Rheological Properties
A DVNext Brookfield cone / plate rotational rheometer equipped with a CP42 cone spindle was used to determine the apparent viscosity as shear rate increased and decreased sequentially (0 - 61.4 – 0 s-1; 255 s; 25ºC). Upward and downward curves corresponding to Shear stress (SS) vs. Shear rate (SR) and SR vs. viscosity (mPa·s) were collected using the internal Rheocalc software of the equipment. The power law model of Ostwald-de-Waele was used to analyze the possible structural changes of the original CBD oil and once included in the microemulsion system as the oil phase [
56,
57]. Briefly, the SS (D·cm-2) vs. SR (s-1) rheograms were adjusted to the model defined by equation 1:
where K is the consistency coefficient and n denote the flow behavior index or Power Law index (being 1 in Newtonian fluids, and lower than 1 in pseudoplastic systems). The shear properties of the microemulsion can also be characterized by the Herschel-Bulkeley model, according to equation 2:
where SS
0 is the yield stress, defined as the minimum shear stress required for the formulation to initiate flow.
The thixotropy was evaluated through hysteresis (area between the ascending and descending curves) where the greater area meant a greater thixotropic effect.
2.4.4. Compatibility Studies
There exist several approaches to study the drug-excipients chemical compatibility for the development of new formulations, being differential scanning calorimetry (DSC) a rapid and easy technique in this field [
58], very useful to detect potential incompatibilities in the early stages of developing a new product. In our study, chemical compatibility of CBD with the other components of the formulations, 20% CBD oil and the designed microemulsion, was determined by DSC.
DSC experiments were conducted on CBD crystals and fluid formulations (CBD oil and microemulsion) using a TA Instruments DSC-Q200 (Waters-TA Instruments, Spain). Quantities ranging from 3 to 10 mg depending on the kind of sample, were placed in a closed aluminum crucible. The heating rate was 10ºC/min, programming temperatures from 35 to 535ºC. All the experiments were conducted under nitrogen atmosphere at a flow rate of 50 mL·min-1.
2.4.5. In Vitro Drug Release
The release characteristics of CBD from the developed microemulsion system was determined in vitro in comparison to the 20% CBD oil using flow-through cells. Data were mathematically studied using a consecutive reaction model simplified to a growth model as described in a previous paper [
59]. The release of Caffeic acid from the microemulsion containing the
S. ebulus extract as aqueous phase was also studied using the same methodology. Briefly, the diffusion equipment (PermeGear® ILC-07 automated system (Riegelsville PA, USA) consisted of seven in-line flow-through diffusion cells, made of Kel-F, in which the donor and receptor chambers, and the diffusion membrane (SpectraPor® - LE dialysis Cut off 10,000 Da), was placed over a support with a hole of 1 cm in diameter (diffusional area, 0.785 cm2). Tygon tubing was used for all the inlet and outlet connections to prevent drug adsorption. The cells were thermostatized at 37ºC, placed in a cell warmer connected to a Haake -DC10® circulating bath (Gebruder Haake, Karlsruhe, Germany). A peristaltic pump Ismatec® IPC-16 (Ismatec, Zurich, Switzerland) was used to provide a constant flow rate of 1 mL·min-1 for the receptor medium (Ethanol / water mixture 70:30 v:v). Samples were collected in glass receptor tubes from an Isco® Retriever IV fraction collector (Isco, Lincoln, NE, USA). An Indexing Controller (also available from PermeGear) was used to program the duration of each shuttle in the retriever, independently, so that 19 samples could be collected simultaneously from each cell.
The evolution of the CBD and CAF concentrations permeated to the receiver chamber (Crec) versus time (t) was studied by using the following equation proposed by Harrison
et al. for flow-through diffusion cells [
60]:
where V
rec is the volume of the receiver chamber, J the apparent intrinsic flux of the drug through the membrane, A the diffusional area and F
rec the flow rate of receptor fluid. The term dC
rec/dt was easily estimated from the concentration versus time raw data. Permeability coefficient (Kp expressed in mg·h
-1) was calculated from the J value divided by the concentration in the donor chamber at time zero according to the fickian diffusion model.
The release kinetic constants of the lipophillic CBD and the hydropillic CAF from the formulation were calculated from the cumulative permeated amounts of drug vs. time profiles using the following expression derived from the first-order consecutive reactions mathematical model [
61]:
where Q
rec is the cumulative amount of drug permeated to the receptor chamber at each time, Q
0 is the initial amount of drug into the donor compartment. K
1 (h
-1) is the kinetic constant expressing the release of the drug from the formulation and K
2 (h
-1) expresses the diffusion through the membrane to the receptor chamber. First-order kinetic was assumed for comparison between the two formulations tested as a concentration-dependent release process. This assumption is based on the fact that a finite-dose diffusion system is obtained and the assumption that the amount of drug applied is sufficient to sustain a constant rate of absorption over study duration and that a steady state is maintained is not valid.
Analysis of data was carried out using the GCFIT program (SIMFIT package 6.0.24. W.G. Bardsley, University of Manchester). Best fit was achieved using growth/survival mathematical models based on the function A [1 exp( K t)] for all the experiments. Goodness of fitting was determined by the analysis of residuals and Durbin-Watson test, as well as Shapiro–Wilks and Akaike AIC stats. All data were expressed as mean ± confidence interval. A p value < 0.05 was considered to be statistically significant using the t-test between the two means for the unpaired data. Data analysis was conducted with SPSS software, version 14.0 (SPSS Science, Chicago, Illinois).
2.5. Antioxidant Activity and Total Phenolics of the Microemulsion-Extract
Total Phenolics Content (TPC) and Total Antioxidant Capacity (TAC) of the microemulsion were determined by using Folin-Ciocalteau and CUPRAC (Cupric Reducing Antioxidant Capacity) methods respectively following the protocols described by Karadirek
et al. (2016) and Flores (2017) [
62,
63] to assess the integrity of the antioxidant compounds after the extraction process with methanol and the subsequent inclusion of the extract into a microemulsion system.
Briefly, TPC method is based on the reduction of phenolic compounds in the presence of the Folin-Ciocalteu reagent (Sigma-Aldrich Spain), provoking a colorimetric reaction that can be quantified spectrophotometrically at a wavelength of 760 nm. TPC results were expressed as equivalent micrograms of gallic acid. A calibration curve was previously set-up by analyzing different concentrations of gallic acid standard solutions as follows: 1.8 mL of Na2CO3 7.5% w:v aqueous solution, 0.6 mL of Folin-Ciocalteu reagent, and different amounts of a 1.25 mM gallic acid solution were added in each glass tube, completing the volume up to 4.5 mL with Milli-Q water. The standard solutions were incubated during 60 min in darkness at room temperature and the absorbance was measured, in the interval from 12.8 to 63.9 µg equivalents of gallic acid. Linearity, repeatability and accuracy were validated within the interval of gallic acid concentrations measured. Extract and microemulsion samples were prepared in the same way as the standard solutions at different concentrations to determine the average TPC as µg equivalents of gallic acid per µg of dry extract.
TAC was determined using CUPRAC reagent (neocuproine – Merk Spain), using a similar methodology as previously described for TPC method: 1 mL of CuCl2 10mM solution, 1 mL of 0.5 mM Neocuproine solution, 1 mL of 1 M ammonium acetate and different amounts of 1 mM gallic acid solution were added in each glass tube completing the volume up to 4 mL with Milli-Q water. Samples were incubated and quantified spectrophotometrically at a wavelength of 450 nm. Equivalent amounts of gallic acid covering from 3.4 to 17 µg were found to have good linearity, accuracy and repeatability. Extract and microemulsion samples were prepared in the same way as the standard solutions at different concentrations to determine the average TAC as µg equivalents of gallic acid per µg of dry extract.
3. Results and Discussion
This section may be divided by subheadings. It should provide a concise and precise description of the experimental results, their interpretation, as well as the experimental conclusions that can be drawn.
3.1. Elaboration of the Pseudo-Ternary Phase Diagram And Design Of The Microemulsion System
For the design of the formulation as a microemulsion system, Labrasol® (PEG-8 caprylic/capric glycerides) and Plurol-Oleique® CC497 (Polyglyceryl-3 oleate) were selected as surfactant and co-surfactant respectively, due to their biocompatibility and to their effectiveness for the formation of a microemulsion in a 1:1 gravimetric ratio, as determined in previous studies [
40]. These excipients are also widely used in formulations intended for topical administration in cosmetic or pharmaceutical use.
From our previous experience, different proportions of water were incorporated by titration in a system where the oily phase consisted of a commercially-available CBD oil for topical use containing CBD at a dose of 20% w:v with hemp oil as main vehicle. As previously described, each mixture was titrated carefully under continuous stirring at a temperature of 30ºC to achieve equilibrium of liquid phases taking care not to incorporate air bubbles in the formulation. The obtained preparation was then visually assessed for transparency and further titrated over the entire phase region.
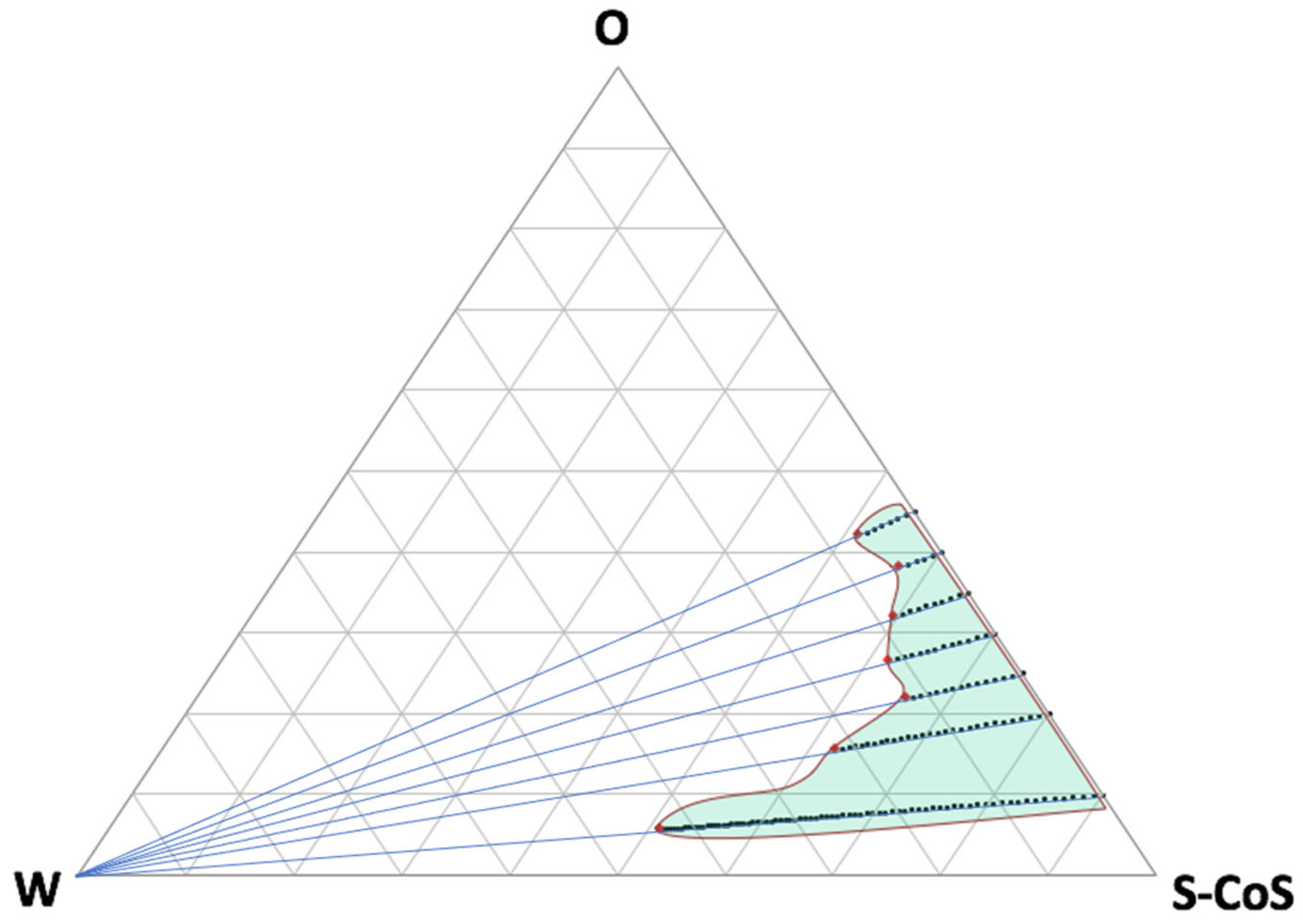
Figure 1 shows the resulting pseudo-ternary phase diagram from all the mixtures described in
Table 1 and the titration lines.
As can be seen, from the resulting microemulsion area, a lot of compositions were obtained including big proportions of water or oily phase, what can be modulated in accordance to the requirements of the future formulation. Further titration with water led to the formation of an emulsion (white area on the left side of the diagram). The confirmation of the microemulsion area was based, not only on the transparency and external appearance of the formulation, but also on the droplet size distribution by light scattering and conductivity results.
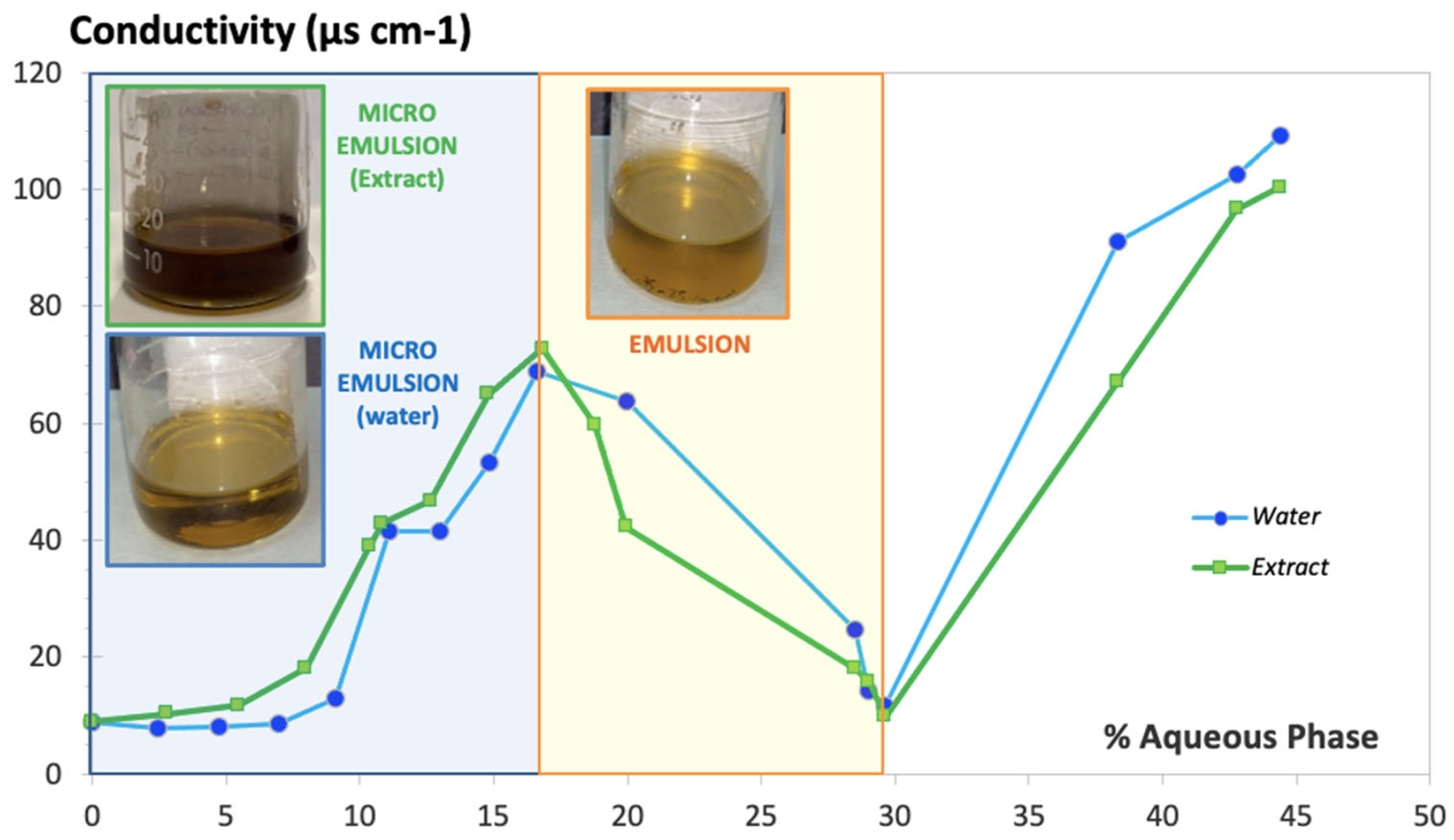
Figure 2 shows the conductivity values obtained (expressed in µS·cm
-1) along the titration process with water over the oily phase mixed with S-CoS, starting with a composition S-CoS / O / W of 70 / 30 / 0 % (w/w), having a total initial weight of 20 g (blue line). Identical measurement of conductivity was carried out with mixtures where the hydrophilic phase was substituted by the aqueous extract of
S. ebulus previously described (green line).
The resulting conductivity vs. water or aqueous extract content plot showed a clearly different zone in both cases (light blue
background on the left side of
Figure 2) in which an increase of conductivity was obtained up to 4 mL of hydrophilic phase added to the oily phase containing the surfactant/cosurfactant, showing a clear behavior of a microemulsion system. Conductivity values decreased dramatically when the volume of hydrophilic phase added during the titration ranged from 4 to 8.5 mL, (light yellow background zone of the plot) as a result of the formation of a thicker and opaque system, denoting the conformation of an emulsion. Volumes of water bigger than 8.5 mL (white zone of the plot) resulted in a new increase of conductivity associated with the breakage of the emulsion, leading to two separate phases. It was observed that the physical behavior of the systems including water or the aqueous extract were practically identical.
Droplet size distribution measured by laser scattering was very useful to confirm the microemulsion area of the pseudo-ternary phase diagram.
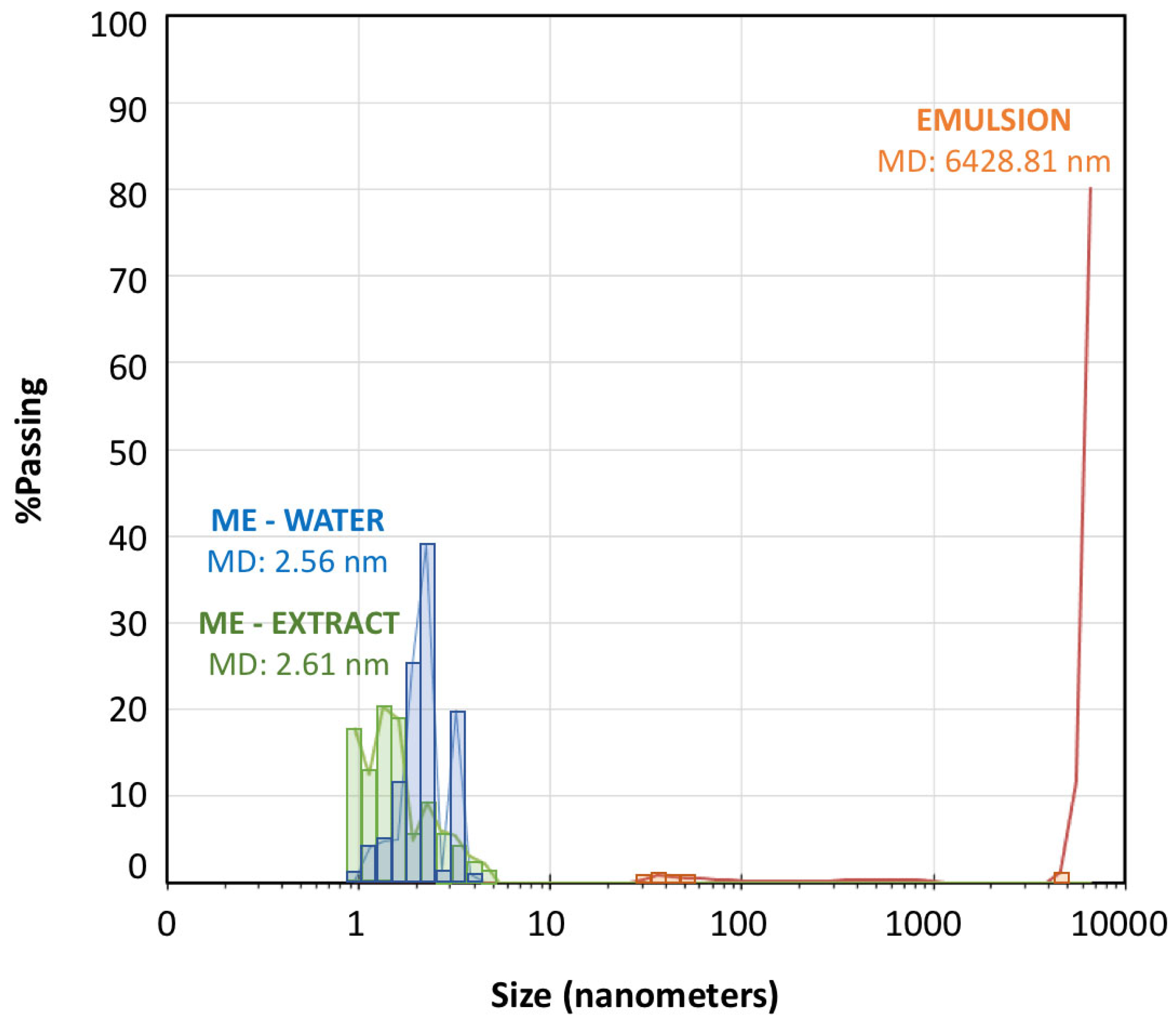
Figure 3 shows the droplet size distribution for the microemulsion-water (titration point S-CoS / O / W: 62 / 27 / 11 % represented by blue bars) and for a subsequent emulsion obtained from the titration process (point S-CoS / O / W: 56 / 24 / 20 % represented by orange bars). The measurement was repeated under the same proportions for a microemulsion containing the vegetal aqueous extract as hydrophilic phase (green bars). The mean diameter volume/surface area (Sauter diameter) was determined for each system.
As can be seen, both microemulsions prepared with water of with the S. ebulus extract showed droplets ranging from 0.9 to 6 nanometers with very similar mean diameter values. Increasing the water content led to an emulsion showing a minority group of droplets higher than 30 nm and a vast group of droplets higher than 5000 nm, what provided a mean diameter higher than 6000 nanometers. It was evidenced the formation of an emulsion when the proportion of water phase was increased, but also, it could be demonstrated that the physical structures of both microemulsions (with water or with the hydrophilic extract was practically identical. These data evidenced that the possible precipitation of any compounds present in solution in the plant extract by a possible migration to the oil phase was negligible.
3.2. Rheological Properties
The rheological behavior of microemulsions has been widely studied. It has been described that the kind of structure conforming the system, bicontinuous or discontinuous, conditions the rheological characteristics of the microemulsion, as well as the interaction between the different aggregates. In general lines, it is widely accepted that microemulsions exhibit a Newtonian behavior (constant viscosity value) at low to medium shear rates but this behavior is modified at high shear rates, especially for bicontinuous systems, due to a probable fragmentation of the structure, leading to shear thinning or pseudoplastic behavior [
64]. It has been also described that the proportion of oil phase can modify the rheological behavior of microemulsions [
65].
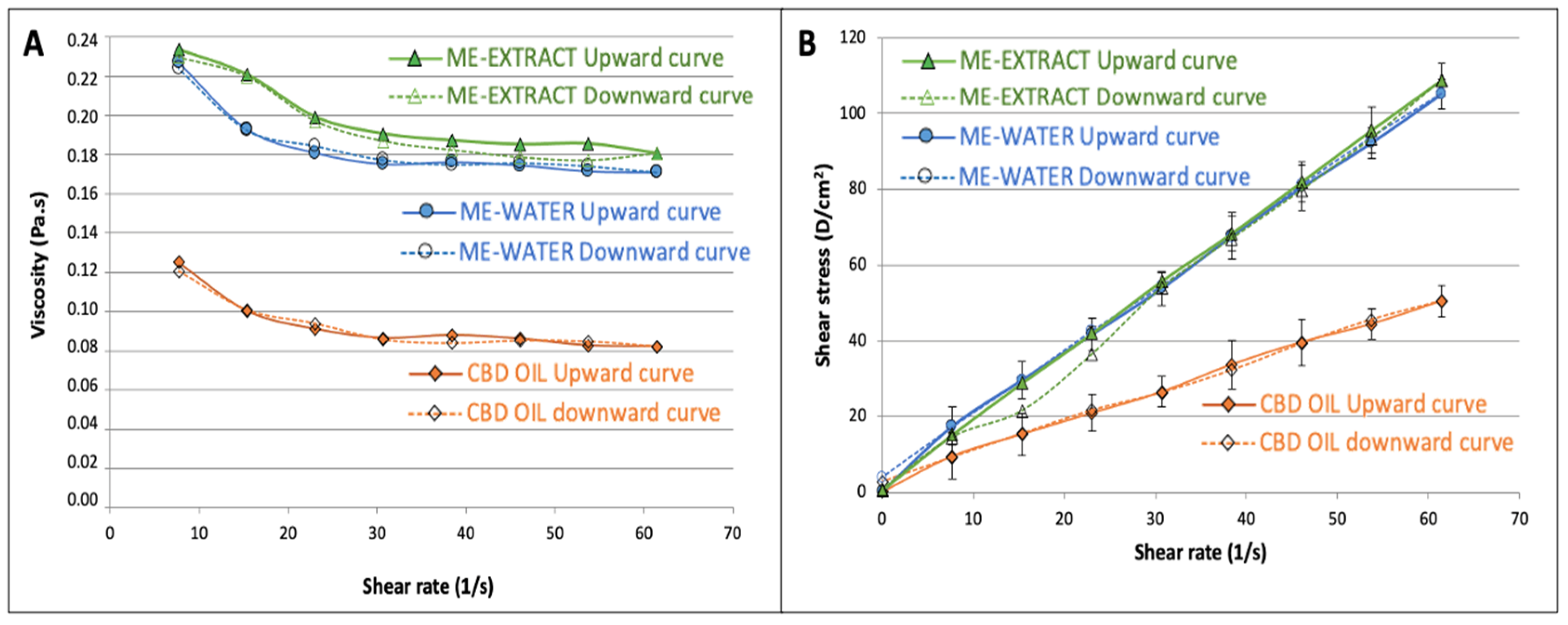
Our data revealed a Newtonian behavior for the CBD oil as well as for our selected microemulsions (61:27:11). SS vs. SR plots show good linearity (see
Figure 4b) and a constant viscosity could be calculated for both systems. The average value of viscosity obtained at shear rates ranging from 23 to 61 s
-1 was used as a reference viscosity since it was observed that this value did not differ significantly from the viscosity obtained from the linear regression of SS vs. SR rheograms. The results from the rheological measurements were mathematically adjusted to the Power Law model of Ostwald-de-Waele (equation 1) using logarithmic plots, and the resulting coefficients for the CBD oil, and for the microemulsions, are shown in
Table 2.
Although all formulations were very fluid showing good spread-ability, it can be observed that viscosity value was approximately double for the microemulsion-water in comparison to the value obtained for the CBD oil, probably due to the presence of a big proportion of surfactant/cosurfactant. Apparent viscosity was slightly superior for the microemulsion- extract in comparison to the formulation with water, due to the higher consistency of the extract in the water phase. Nevertheless, consistency coefficient did not show statistically significant differences between the CBD oil and the two microemulsions studied, due to the high variability found.
Comparing flow behavior index, it could be observed that all exhibited a behavior near Newtonian. Nevertheless, it was found that the n values obtained for both microemulsions were statistically lower than 1, which denotes a slight pseudoplastic behavior. The viscosity vs. shear rate diagrams (
Figure 4a) showed a transition of Newtonian to non-Newtonian fluid behavior: both microemulsions behaved as a shear-thinning fluid at higher shear rates, which coincides with the behavior found by other authors on microemulsions [
65]. Adjusting the Herschel-Bulkeley model to our data, it was found that the yield stress value did not differ from zero (
p=0.09) for both microemulsions.
The area under the curve of the upward and downward curves corresponding to the viscosity vs. shear rate to define the hysteresis, was used to estimate the thixotropy of the three formulations. It was found that the microemulsions showed significantly higher (3 times higher hysteresis, see table 2) thixotropic behavior, which indicates that the microemulsions needs a bigger effort to reorganize its structure when it is exposed to higher shear rates. The oil does not have such a complicated structure and it is evidenced from the differences found in this parameter. Nevertheless, the thixotropy is also very low in the microemulsions, showing a great recuperative ability at the studied shear rates.
In order to study the physical stability of the microemulsion including the S. ebulus extract as water phase, its rheological characteristics were measured at 12 moths in which the formulation was stored under the standard conditions of darkness, and a temperature of 25,0 ± 0,5 ºC. Mean apparent viscosity was 0.188 ± 0.001 Pa·s. No statistically significant differences were found for this parameter and for the other determined rheological values (n= 0.93 ± 0.11, hysteresis= 0.251 ± 0.135 Pa). These data confirmed that no structural changes took place in the microemulsion system along the storage period, what confirmed the good stability of the formulation. No signs of separation of phases or precipitation were detected.
3.3. Compatibility Studies by DSC
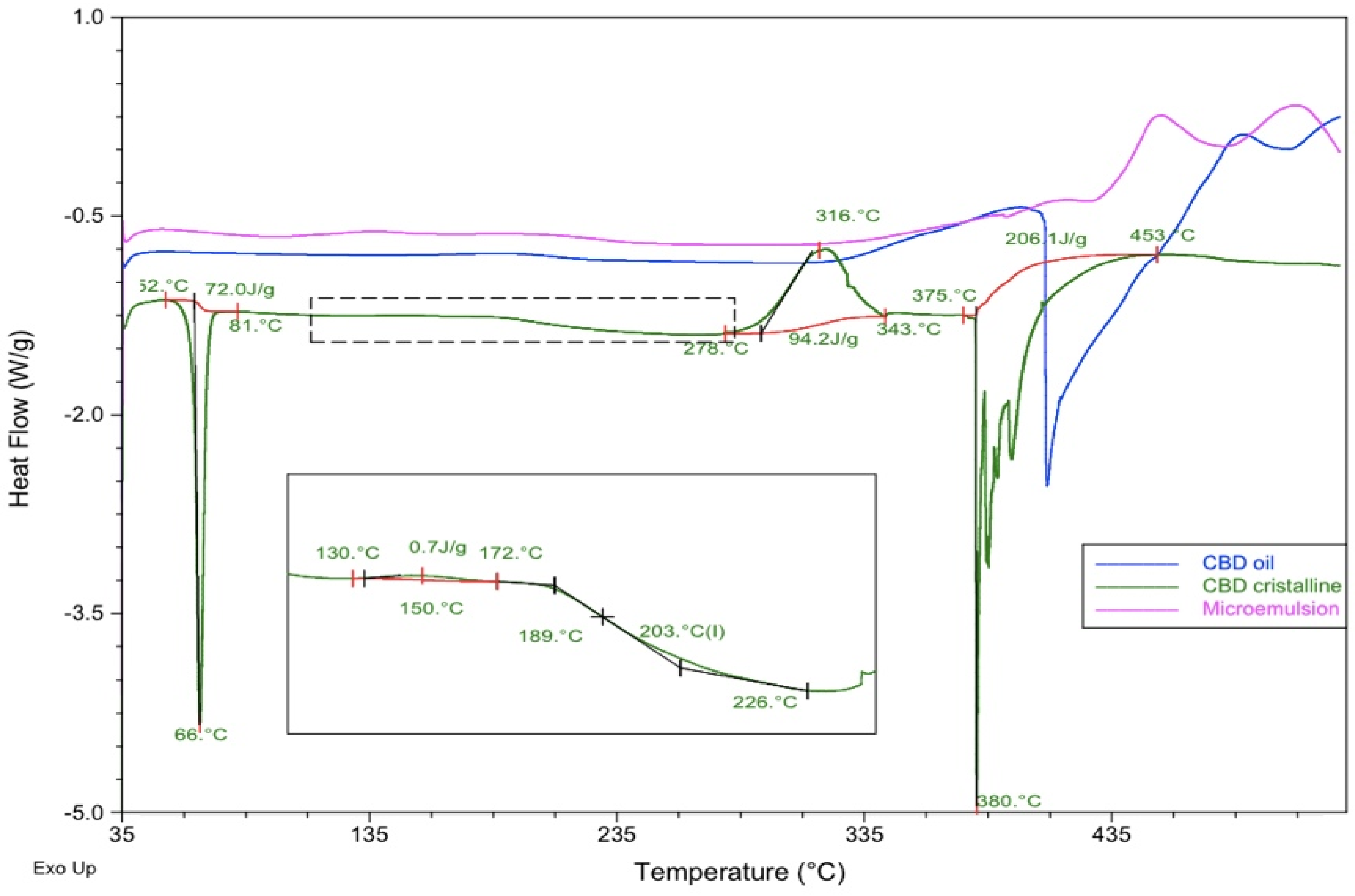
DSC experiments were conducted as previously described, for three samples: pure CBD in crystalline original form, a commercial CBD oil, and a microemulsion containing the CBD oil as oily phase, surfactant-cosurfactant 1:1 mixture and water at proportions of [27:31:31:11] (w:w). Thermograms were obtained from 35 to 535ºC at a heating rate of 10ºC/min to ensure that all possible melting or decomposition processes could be analyzed.
Table 3 and
Figure 5 describe the main thermal processes found.
As can be seen, CBD crystals presented an endothermic process that corresponds to the melting at 66ºC within a range of temperatures of 52 – 81ºC (T
onset and T
enset) and an enthalpy value of 72.0 J/g. The DSC profile showed an endothermic event indicating the decomposition of the DSC at a peak temperature of 380ºC ranging from 375 to 453ºC (T
onset and T
enset) and an ∆H= 206.1 J/g. It was also found a slight exothermic event at 150ºC and a significant exothermic peak at 316ºC. A possible glass transition was observed from 189 to 226ºC. These details introduce some differences in comparison to the DSC profile obtained for CBD by other authors [
66].
The DSC line corresponding to the commercial oil containing 20% CBD in dissolution (blue line in
Figure 5), showed a slight exothermic process from 126 to 234 ºC (T
peak = 192ªC) and a majoritarian endothermic event indicating the decomposition of the components at 409ºC within range of temperatures from 398 to 489ºC.
The microemulsion profile showed a different behavior, with a slight endothermic process at 393ºC with ∆H= 1.8 J/g, and a majoritarian exothermic event at 451ºC ranging from 427º to 480ºC (Tonset and Tenset) and a resulting enthalpy of 55.5 J/g. This thermal profile evidenced that the microemulsion was more stable than the commercial oil, both having the CBD in dissolution.
In the light of these findings, no incompatibilities were detected between CBD and the components of the CBD oil and the microemulsion. Although these results are promising at this point of a pre-formulation study, they must be considered cautiously. Further techniques should be used to support the compatibility of the drug, like Isothermal Stress Testing-Fourier Transform Infrared Spectroscopy, or quantitative analysis after storage under stressed conditions, [
58,
67] once a complete formulation is designed.
3.4. In Vitro Drug Release
The in vitro release studies were performed to characterize the release of CBD from the commercially available CBD oil, and to elucidate whether or not the creation of a microemulsion system could modify significantly the retention or release of the CBD in comparison to the original CBD oil. The release of CBD from the oil phase was studied concomitantly with the release of CAF from the aqueous phase as a model hydrophilic antioxidant compound, found in the S. ebulus extract used in the microemulsion.
Apparent steady-state intrinsic flux (J
app in µg/cm
2·h) was calculated for each experiment by non-linear regression using a bi-exponential model from the flux calculated at each time interval from equation 3 and following the mathematical approach proposed by many authors for the evolution of the intrinsic flux along time experiment and flow-through diffusion cells [
68,
69]. Permeation coefficient (Kp in cm·h
-1) was calculated from the J
app value divided by the donor concentration at time zero, according to the Fickian diffusional model.
In order to calculate, not only the global release process by mean of the Kp value, but also the release of CBD from the formulation, the parameter K1 expressing the release kinetic from the formulation expressed in h
-1 was determined by non-linear curve fitting of the cumulative amounts permeated vs. time profile, according to the equation 4 derived from the first-order consecutive reactions mathematical approach [
61]. SIMFIT statistical package was used for the nonlinear curve fitting by weighed least squares selecting the model described as monomolecular growth-GCFIT model. Information on goodness of fitting was also provided by the software to determine the reliability of the constants obtained.
A previous experiment was carried out using a CBD solution with a concentration of 1 mg·mL-1 in an ethanol:water [70:30] (v:v) mixture, and selecting the same medium in the receptor chamber to ensure that the solubility is exactly the same at both sides of the diffusion membrane. This experiment permitted us to characterize the diffusional parameters of the CBD previously dissolved, without the interference of a formulation that could condition the release from its structure or modify the solubility and hence the diffusion process. In our study, the flow rate of the receptor medium (Frec of equation 3) was 1 mL·min-1 and the temperature of the cell warmer was fixed to 37ºC. The receptor fluid was collected every hour for the first 10 hours and finally, every 4 hours up to 24 hours. The amount of CBD permeated at each time was analyzed by the previously described HPLC method. All experiments were conducted in sextuplicate.
Once the diffusional properties of CBD in solution were determined, in vitro release experiments were carried out for the commercial CBD oil and for the microemulsions under the same conditions as described for the previous experience with the CBD solution.
Table 4 describes the main diffusional and release parameters for CBD dissolved in the commercial oil, and from the microemulsion with water. The same table also includes the values related the microemulsion containing the
S. ebulus extract as aqueous phase, analyzing simultaneously the release parameters of CBD from the oil phase and CAF from the extract.
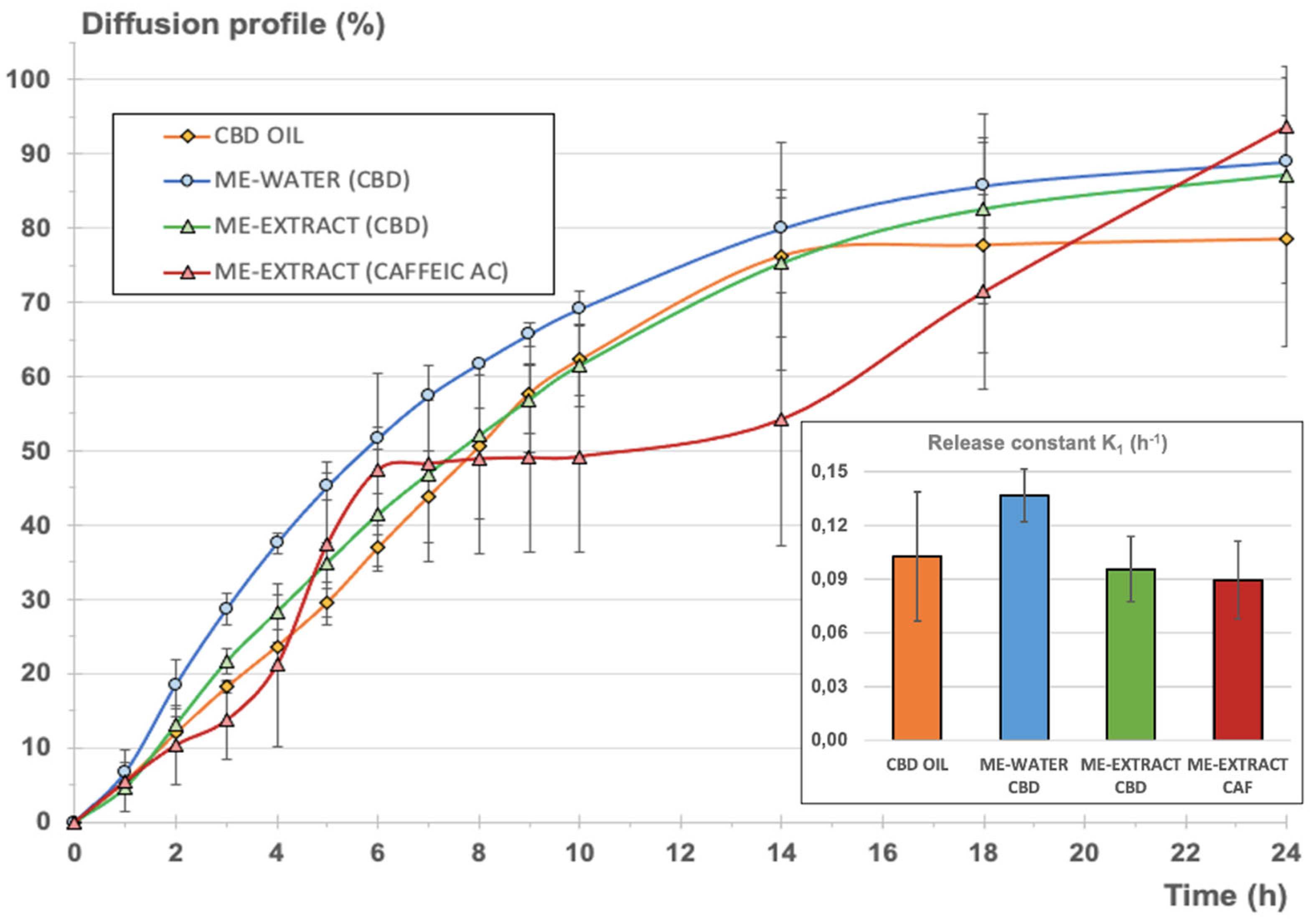
Figure 6 shows the comparative release profiles for the two systems analyzed, expressed as cumulative amount permeated vs. time in percentage of the total amount of CBD disposed in the donor chamber for each experiment for normalization. For the second microemulsion including the vegetal extract, the release profiles of CBD and CAF were simultaneously determined.
As can be seen, steady-state intrinsic flux values were very different in each case, as expected, considering the different initial CBD concentrations in the donor chamber. Nevertheless, permeability coefficients for CBD from the oil and the microemulsions were very similar, what denotes that the full CBD release and diffusion process was similar to the oil and the microemulsions. It was also observed that the estimated time necessary to reach the steady state was almost double for the microemulsions in comparison to the value calculated for the oil. These results indicates that the microemulsion system does not suppose a barrier for the release of CBD but provides a slight delay to get to the steady state were the maximum apparent intrinsic flux is reached.
Comparing the permeability coefficient Kp for CBD and CAF from the emulsion containing the extract, it can be observed that global diffusion was 1 magnitude order (more than 20 times bigger) for CBD.
Table 4 also shows the main release parameters calculated from the cumulative amounts permeated vs. time profile adjusting the monomolecular growth fitting exponential model to the experimental data. Goodness of fitting was good enough in all cases. Durbin-Watson statistic provided values below 2.5, denoting good correlation except for the microemulsion with water, where the value obtained was a bit higher. Nevertheless, the rest of the fitting parameters confirmed the good quality of the fitting. Shapiro-Wilks values were greater than 0.05, confirming the normality of the data. It could be concluded that the calculated constants from the model were consistent and reliable in all cases. Higher variability was observed for the release model calculated for CAF but the goodness of the curve-fitting was also acceptable.
The normalized diffusion profile expressed as cumulative percentage of each compound (CBD or CAF) diffused with respect to the initial amount in the donor chamber (see
Figure 6), reveals that the inclusion of the CBD oil in a microemulsion with a hydrophilic phase (blue and green lines), was similar to the normalized profile corresponding to the original CBD oil (orange profile). To refine this, K
1 release constants were determined from the cumulative amount of CBD diffused vs. time profiles according to equation 4, assuming first-order consecutive kinetics for the comparison of all formulations. This kinetic can be assumed, considering that a diffusional process through a membrane starting from a drug in dissolution is taking place. Besides, the release of a certain drug from a microemulsion is likely to adopt a pseudo-zero-order kinetic, but conditioned to the volume of the dispersed phase, the partitioning of the drug between the interphases, and the diffusion rate of the drug [
70].
Comparing the calculated K
1 values for CBD release from the oil and the microemulsions, it could be concluded that no statistically significant differences were found. The inclusion of the oil in a stable structure with a big proportion of surfactant/cosurfactant mixture and a aqueous phase did not provoke a significant modification of the release of the CBD, with the advantage that hydrophilic compounds can be included in this stable system without provoking a significant retention of CBD or other non-desirable phenomena like the insolubilization of the hydrophilic or lipophilic compounds or the breakdown of the structure, as can be seen in
Figure 6 in the results of the microemulsion elaborated with the aqueous vegetal extract.
Comparing the resulting release parameters for CBD and CAF, it could be observed that the release constants (K
1) were very similar and no statistically significant differences were found (
p=0.05). A pseudo-plateau was observed from the mean data between 6 and 14 hours (red profile in
Figure 6), but this observation was considered to be negligible due to the higher variability observed on the CAF profiles. It can be concluded that the microemulsion did not suppose a barrier for the release of CBD, in comparison to the oil, and CAF, showing similar release rate values, also considering the different nature of both compounds and the different initial doses in the microemulsion. Subsequent Ex vivo and In vivo pharmacokinetic studies are necessary to characterize the final formulation.
3.5. Antioxidant Activity and Total Phenolics of the Microemulsion-Extract
Total Phenolics Content (TPC) and Total Antioxidant Capacity (TAC) of the microemulsion containing the
S. ebulus extract as aqueous phase were determined as described under 2.5. Both studies were conducted at time zero, from the just-prepared microemulsion and from the microemulsion stored during 12 months under room temperature storage conditions in sealed containers in the absence of light, to elucidate whether or not the antioxidant activity of the extract was preserved when it was formulated in the microemulsion.
Table 5 shows the TPC and TAC resulting mean values for each situation.
No statistically significant variability of the total phenolics content and the total antioxidant capacity after 12 months, what confirmed that the integrity of the antioxidant compounds after the extraction process with methanol and the subsequent inclusion of the extract into a microemulsion system was preserved under the previously described storage conditions. As previously mentioned, no insolubilization phenomena were observed up to 12 months under standard storage conditions and no breakdown of the structure turbidity were observed.
4. Conclusions
It is widely described that CBD and any other cannabinoids can play an important role for the treatment of different skin disorders, due to the importance of the endocannabinoid system in the skin [
1,
32,
71]. The lipophilicity of CBD makes it very easy to formulate an oil or a semisolid oily preparation for topical use but no hydrophilic compounds could be included. The formulation of an emulsion could be an option to create a cream but there exist inherent advantages of microemulsions, being easier to elaborate at industrial scale, thermodynamically stable and exhibiting lower viscosity and Newtonian flow, while emulsions are thermodynamically unstable and show higher viscosity [
72].
In our study, microemulsions from the commercial CBD oil has been successfully developed and characterized in terms of pseudo-ternary phase diagram to find the adequate microemulsion composition, based on additional confirmatory data of conductivity, droplet size and rheological characteristics. It was also found that the viscosity of the designed microemulsion was suitable for topical administration, showing a slight pseudoplastic behavior. The inclusion of a vegetal extract as aqueous phase did not significantly modify the rheological properties, in comparison to the original CBD oil, maintaining a CBD load bigger than 5% w:w. The physical integrity and stability of the microemulsion system was also evidenced as the rheological parameters were not significantly altered after 12 moths’ storage.
A lot of strategies for the vehiculization of CBD based on Nano-emulsions (thermodynamically unstable), and other lipid-based formulations were described in an exhaustive and very interesting recently published review [
73], were only one microemulsion structured in a gel, loaded with 1% CBD was described. The microemulsions developed in the present study provide a wide variety of proportions, with a significantly bigger load of CBD. As can be seen, the formulation used as a reference for physicochemical characterization in our study contained more than 5% of CBD, and other compositions contained up to 8.4% of CBD. It must be also taken into consideration that there exists the possibility of working with a CBD oil containing 30% of CBD, instead of 20%, what could give us the option of producing a microemulsion with a final load of CBD bigger than 12%, for the last composition described in table 1. This versatility and the load ability can suppose a significant advantage to increase transdermal penetration, in comparison to other reported formulations.
It was also studied the release or retention properties of CBD from the microemulsions as a comparison to the original oil. The creation of the bi-continuous system did not significantly affect the release of the CBD. The release of Caffeic acid as a model hydrophilic compound was found to be similar than the observed for CBD. It can be concluded that the developed microemulsion was able to include CBD in combination to different hydro-soluble compounds with a potential antioxidant and anti-inflammatory effect, without creating a barrier that could difficult their release from the topical formulation.
It was demonstrated that the microemulsion containing the vegetal extract of S. ebulus ripe fruits showed good antioxidant capacity that was preserved after 12 moths’ storage in terms of Total Phenolic Compounds and Total Antioxidant Capacity.
This strategy appears as a promising galenical alternative due to its inherent advantages as a drug carrier for both hydrophilic and lipophilic substances, stability and simplicity compared to other kinds of formulations. The microemulsion here designed and characterized acquires unique potential pharmacological properties for future therapeutic uses.