1. The Use of Fission Yeast to Study Eukaryotic Cytokinesis
Both mammalian cells and the fission yeast Schizosaccharomyces pombe use binary fission to divide medially. Fission yeast cells are encased in a cell-wall structure giving them their rod shape following growth by tip extension and divide equatorially. Therefore, the species is considered an excellent model organism for studying eukaryotic cytokinesis, where similar cellular processes occur.
Research using S. pombe has allowed the identification of many important conserved cell cycle regulators. As the mechanisms of assembly and constriction of the actin-myosin contractile ring (ACR) in S. pombe are very similar to those seen in mammalian cells, our current understanding of eukaryotic cytokinesis stems extensively from studies in fission yeast (Pollard and Wu, 2010).
During the 1970s, the work of Leland Hartwell and colleagues with the budding yeast Saccharomyces cerevisiae led to the discovery of a large number cell division cycle (CDC) mutants, and for the first-time eukaryotic genes required for cell division were characterized (Hartwell et al., 1970; Hartwell et al., 1973). Study of the cell division cycle continued in the distally related fission yeast S. pombe by Paul Nurse and colleagues, with the discovery of equivalent cdc gene mutants (Nurse, 1975; Nurse et al., 1976).
Later in the 1990s, two landmark reviews discussed aspects of the S. pombe cell cycle including the timing of events leading to cytokinesis, cell division, and mechanisms for determining the medial or equatorial division plane. At that time the cdc16 and cdc2 genes were thought to act as a molecular switch regulating S. pombe mitosis and cytokinesis (Chang and Nurse, 1993), and it was proposed that the division plane was determined by the position of the nucleus (Chang and Nurse, 1996). Subsequent research offered a deeper understanding of the S. pombe cell cycle regulation, including aspects of cytokinesis (reviewed in Nurse, 2020), and cell polarity (reviewed in Chang et al., 2019).
Establishment of cytokinesis is mediated by a cytokinetic ACR that leads to the final separation of the two daughter cells. In this short review, we describe
S. pombe cytokinesis starting from the medial positioning of the division plane and the assembly of the cytokinetic ACR (
Section 2), to the forces that generate tension and lead to constriction of the ACR and the role of ESCRT proteins in cytokinesis, to the final separation of the two daughter cells during septation (
Section 3).
2. Actin-myosin Contractile Ring (ACR) Assembly in Fission Yeast
2.1. Positioning of the Cell Division Plane
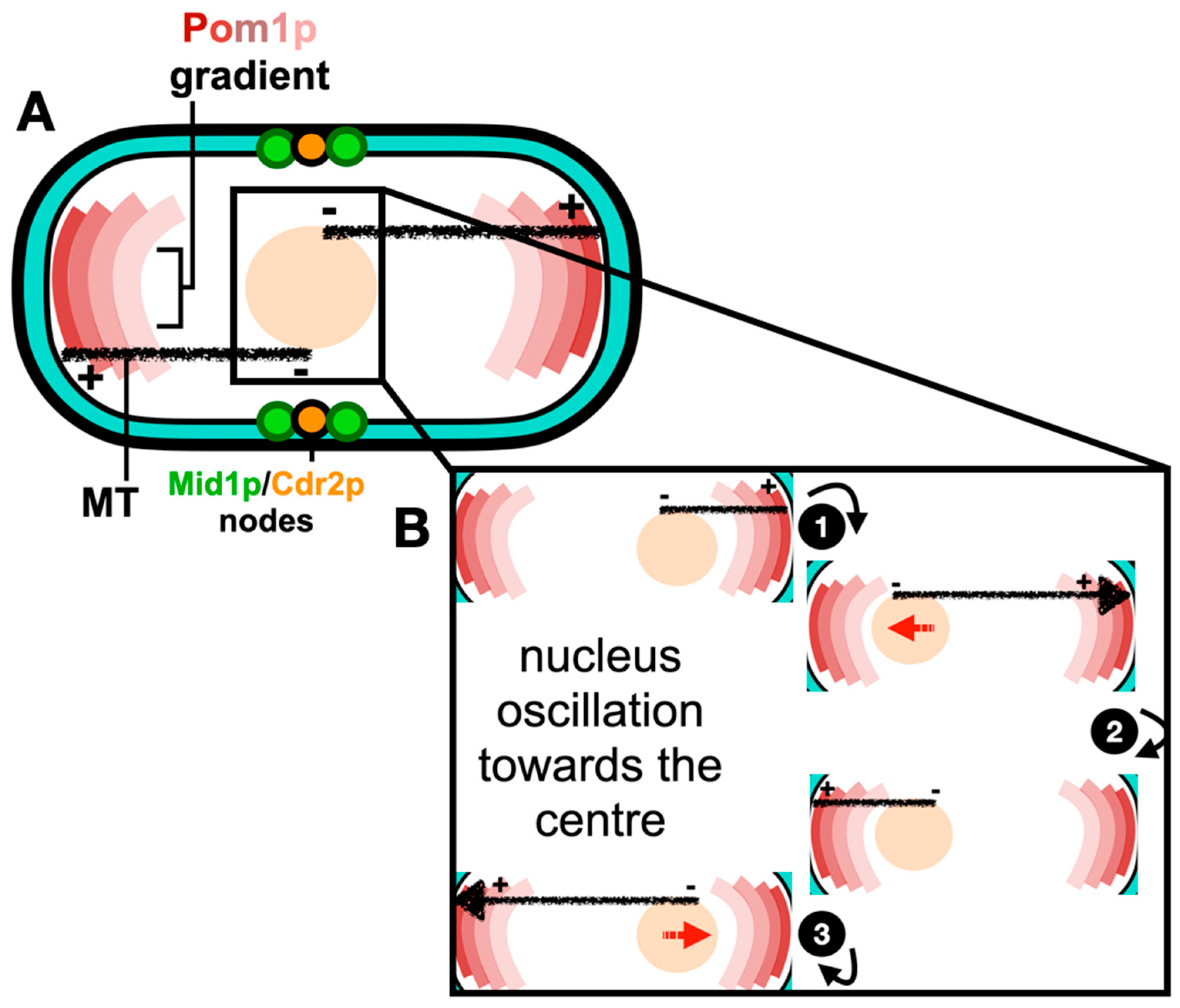
In S. pombe cellular growth occurs throughout a longer interphase period with this ceasing during the shorter mitosis and cytokinesis periods, after a certain cell length is achieved. During the cell cycle, “middle” and “end or tip” locations are specified by two spatial axes. The “middle” location is defined by the nucleus which is positioned at the cell centre by a microtubule-pushing mechanism where a force produced by the cytoplasmic microtubule bundles and is applied to the nucleus (Tran et al., 2001; Piel and Tran, 2009). Furthermore, this force is able to efficiently re-centre nuclei of cells exposed to nuclear displacement (Daga et al., 2005; Daga et al., 2006). The dynamic interplay between the nucleus and the microtubule cytoskeleton is illustrated and reviewed by Gallardo et al. (2019). Alternatively, the “end or tip” location is defined by formin-mediated actin assembly mechanism at cell tips (Martin et al., 2005), and polarity factors including the DYRK kinase Pom1p secreted in a gradient manner at cell poles. Additionally, Pom1p controls mitotic entry by phosphorylating the ACR scaffold protein Cdr2p at the cell “middle” through phosphorylating its membrane-binding C-terminal region (Gerganova et al., 2019).
Much evidence shows that the Anillin-like protein Mid1p localizes to the “middle” location and initiates ACR assembly (Paoletti and Chang, 2000; Almonacid
et al., 2011; Saha and Pollard, 2012, Rezig
et al., 2021). Mid1p has two membrane binding domains, the pleckstrin homology domain (PH) and the cryptic domain (C2) (Chatterjee
et al., 2019). However, it only binds the plasma membrane after it is activated and resealed from the nucleus (Bähler
et al., 1998; Almonacid
et al., 2011). The roles of Mid1p in positioning the ACR are now well understood in
S. pombe, and are reviewed in Rezig
et al. (2022), with the mechanism of medial positioning of the ACR schematically described in
Figure 1.
In contrast, comprehension of the organization of proteins that assemble the complex cytokinetic machinery during cytokinesis is still relatively rudimentary. In S. pombe, cytokinesis proteins are recruited to the cell centre pre-determining the future division plane, these organized as cortical spots, named “nodes” (Akamatsu et al., 2014; Laplante et al., 2016). The next Section will discuss the nature of these nodes including their constituent proteins and spatiotemporal organization.
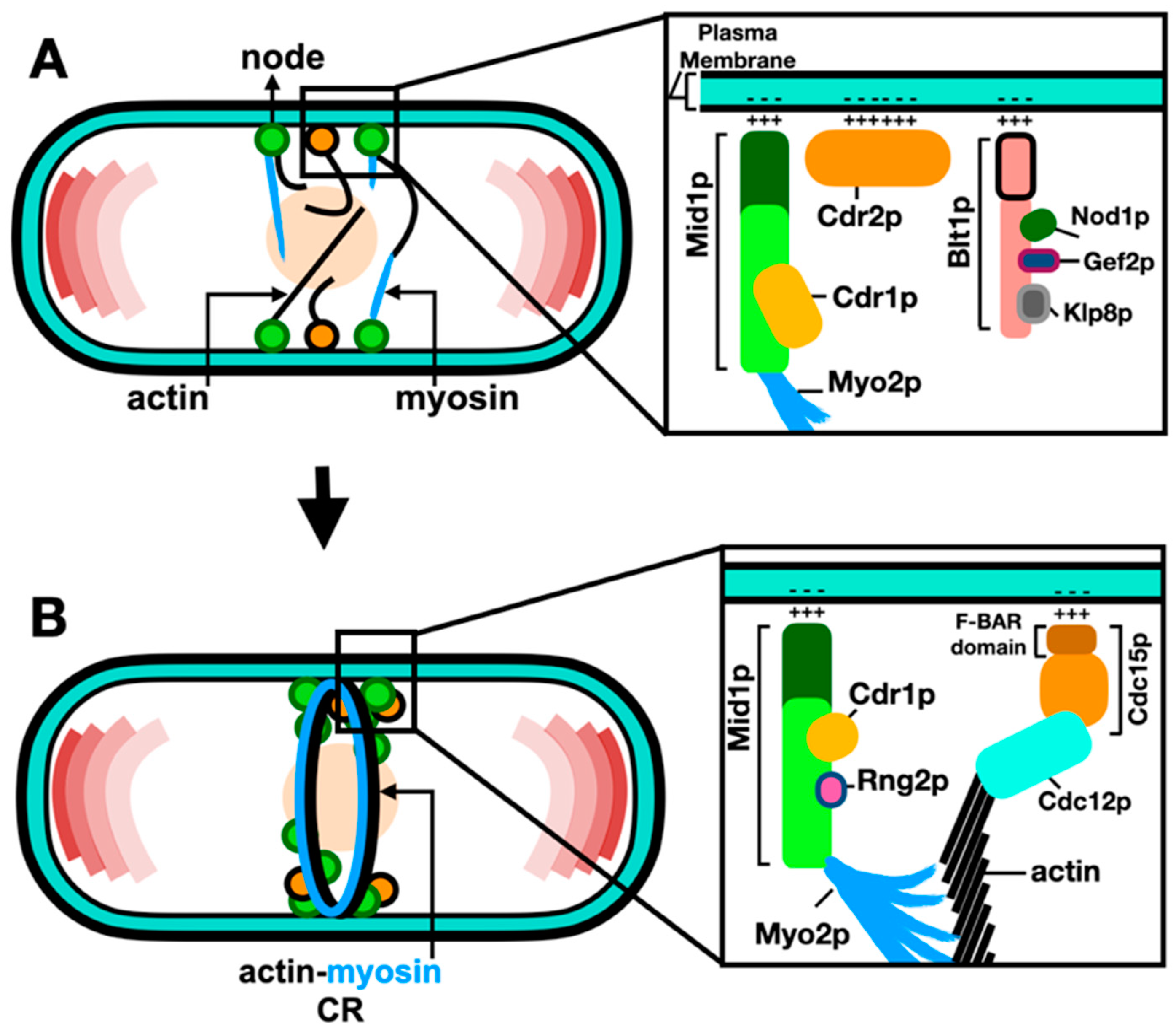
2.2. Molecular Organization of Nodes within the ACR
The current model for the ACR assembly includes two types of interphase nodes: type-1 “stationary” nodes containing Cdr1p, Cdr2p, Wee1p, Mid1p; and type-2 “anchoring” nodes containing Blt1p, Gef2p, Cdc15p, Rng2p, and Cdc12p (Zhu et al., 2013; Akamatsu et al., 2014). The “anchoring” nodes form the units that anchor the ends of myosin-II tails to the plasma membrane, with myosin-II heads extending into the cytoplasm. These nodes then merge into a ring-like structure, named the actin-myosin contractile ring (ACR) and, as its name implies, it is composed of actin filaments and myosin-II motors in addition to various classes of cytokinesis proteins (Malla et al., 2021).
Live cell imaging, high-speed fluorescence photo-activation localization microscopy (FPALM), and fluorescence resonance energy transfer (FRET) have shown to be excellent methods to dissect ACR nodes. Recent findings revealed that nodes are discrete units with stoichiometric ratios and specific distribution of constituent proteins (Laplante
et al., 2016, Akamatsu
et al., 2017, McDonald
et al., 2017; Malla
et al., 2021). Furthermore, the localization of the ACR constituents is thought to be arranged in several layers relative to the plasma membrane, starting with plasma membrane-binding proteins and the tail of myosin-II, to intermediate cytokinesis proteins, and farthest from the plasma membrane lays myosin motor domains, F-actin and its cross-linkers (McDonald
et al., 2017). Advances in laser scanning microscopy, such as Airy-scanning using very low laser power to acquire high-quality images, increased the resolution and signal-to-noise ratio and enabled the detection and measurement of even feint individual cytokinesis nodes (Sayyad and Pollard, 2022). Coalescence of nodes leads to the ACR assembly through the search capture pull and release (SCPR) mechanism, whereby Cdc12p nucleates actin filaments as Myo2p pulls actin filaments, thus producing the force required to pull the individual nodes into the ACR (Vavylonis
et al., 2008; Laplante
et al., 2016; Zimmermann
et al., 2017). Such assembly of the ACR from node precursors is schematically described in
Figure 2.
2.3. Anchorage of the ACR to the Plasma Membrane
After ACR assembly, Myo2p tails and Cdc15p localize to the plasma membrane, with the Myo2p heads, Myp2p and the bundle of actin filaments localizing 60 nm away from the plasma membrane (Swulius et al., 2018). It is suggested that this organization connects the bundle of actin filaments to the plasma membrane (Bellingham-Johnstun et al., 2021). Cdc15p next recruits Cdc12p to the ACR, and this interaction is thought to be essential for the ACR organization and stability (Snider et al., 2020).
The phospho-status of Cdc15p influences its ability to bind the plasma membrane, with phosphorylation of Cdc15p by Pom1p inhibiting its binding to the plasma membrane at cell tips (Bhattacharjee et al., 2020). Additionally, the p21-activated protein kinase (Pak1p), another polarity kinase, was found to regulate the function of Mid1p and Cdc15p (Magliozzi et al., 2020). Cdc15p has three regulatory components: an N-terminal Fre/Cip4 homology Bin/Maphiphysin/Rvs domain (F-BAR), a medial intrinsically disordered region (IDR), and a C-terminal Src homology 3 domain (SH3). While the F-BAR domain enables protein oligomerization and concentration on the plasma membrane to scaffold protein assemblies resulting in membrane deformation (Snider et al., 2021), it was recently found that phosphorylation of Cdc15p induces the separation of the Cdc15p IDR region resulting in an inhibition of Cdc15p phase separation, and the formation of condensate on the plasma membrane (Bhattacharjee et al., 2023).
Moshtohry and colleagues recently used laser ablation, a technique based on photodamage in which cellular structures could be degraded using focused pulsed laser (Khodjakov et al., 1997), to investigate the mechanical role of Cdc15p during S. pombe cytokinesis, they found that the ACR recoils after being severed, however, this recoil profile was greater and slower in the ablated ACR of Cdc15-depleted cells, suggesting that loss of Cdc15p decreases stiffness of the ACR material (Moshtohry et al., 2022). Furthermore, another F-BAR protein, Imp2p, was found to contribute to the stiffness of the ACR (Bellingham-Johnstun et al., 2021).
3. ACR Constriction is Coordinated with Septation in Fission Yeast
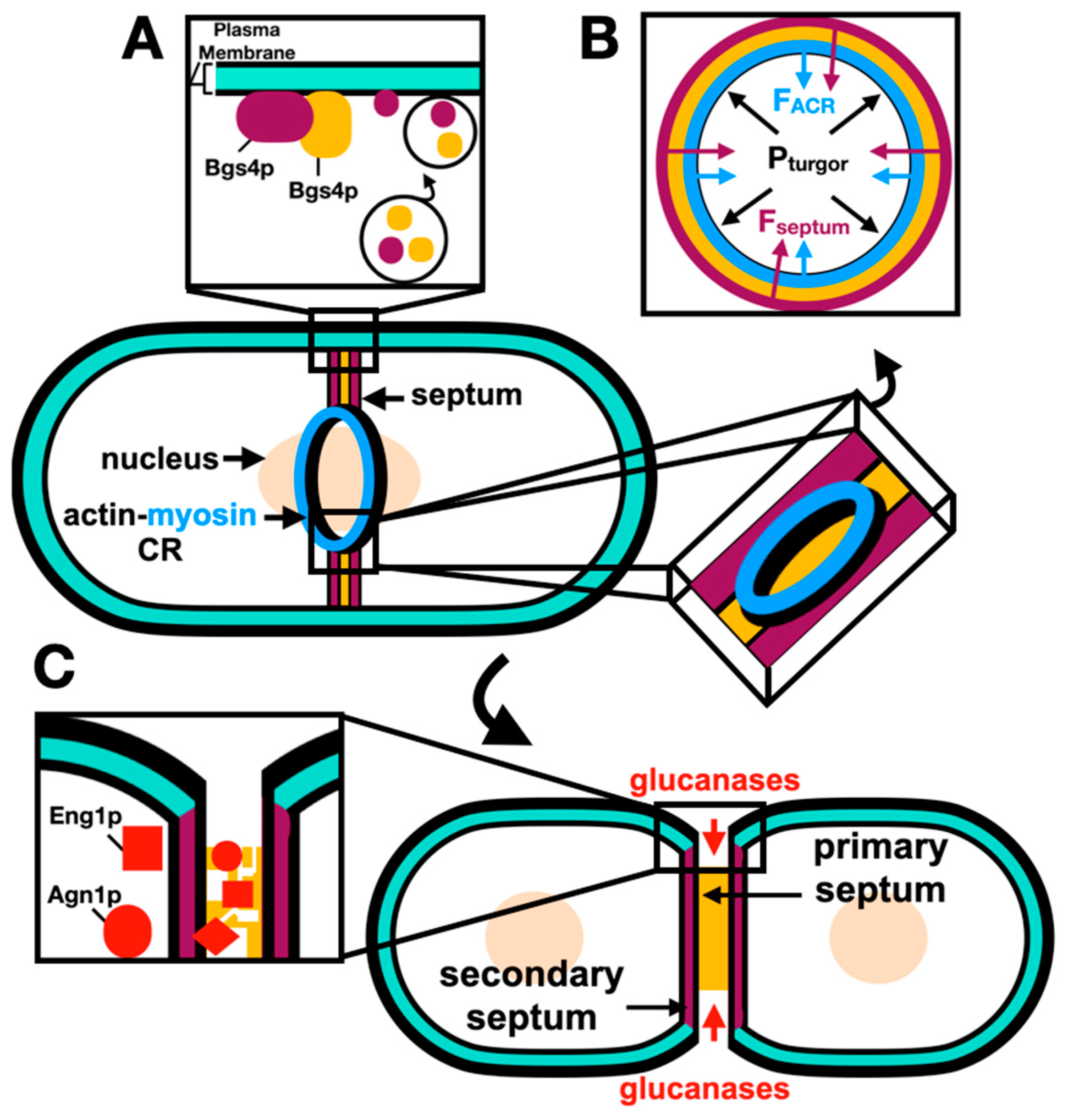
Unlike in mammalian cells where cytokinesis only implies the formation of a medial ACR, cytokinesis in S. pombe additionally requires the formation of a cell wall-like structure named the septum, physically separating the two daughter cells at the division site. After ACR assembly and constriction, the septation initiation network (SIN) mediates the synapses of the amphid defective (SAD) kinase, Cdr2p, dispersal from the cell cortex into the cytoplasm, and septation is coordinated with the ACR constriction (Rincon et al., 2017). The septum is composed of three layers: a middle primary septum layer that is later digested by the end of cytokinesis, and two flanking secondary septa layers which remain intact to form the new cell walls of separated daughter cells (reviewed by Pérez et al., 2018 and Hercyk et al., 2019).
S. pombe septation occurs in three stages. First, deposition of the septum cell-wall structure material is carried out through membrane trafficking events where secretory vesicles deliver the septum beta-glucan synthase 1, Bgs1p (Onwubiko et al., 2021). Localization of cell-wall building enzymes including Bgs1p depends on both Cdc42p and Cdc15p (Wei et al., 2016; Campbell et al., 2022). Second, during anaphase B the ACR constricts at a slow rate as the septum ingression is initiated; however, after the delivery of the alpha-glucan synthase 1, Ags1p, and Bgs4p, the rate of constriction and septum ingression is increased (Cortés et al., 2018). Third, after the ACR constricts, exocytosis leads to the delivery of glucanases and digestion of the primary septum leaving the two daughter cells each with a new cell wall (secondary septa) (Pérez et al., 2015).
Septation is coupled to ACR constriction, and the interaction between Bgs1p and the ACR is mediated by the paxillin Pxl1p (Cortés et al., 2015). Furthermore, Pxl1p accumulation during septum formation is thought to be mediated by an interaction with both the N-terminal F-BAR (Snider et al., 2020) and C-terminal SH3 domains of Cdc15p (Bhattacharjee et al., 2020). The mechanism of engagement of Pxl1p to both distal domains of Cdc15p is not well understood. However, a recent study demonstrated this interaction and found that Pxl1p binds to the Cdc15p F-BAR cytosolic domain (Snider et al., 2022).
Proctor and colleagues (2012) found that beta-glucan synthesis in important for the ACR to overcome the high turgor pressure during cytokinesis, suggesting that other pathways might coordinate with ACR constriction. Recent research used a fission yeast mutant cps1-191, defective in 1,3-beta-glucan-synthase septum synthesis, that arrests with a non-constricting ACR, to test if high turgor pressure restricted ACR constriction in such mutants (Chew et al., 2020). This showed that a decreased turgor pressure inhibits the ACR constriction in cps1-191 mutants, suggesting that the extracellular glycan network regulates the ACR constriction and septum ingression, and that subsequent remodeling of the extracellular components relieves this restriction and facilitates the ACR constriction.
At the last stage of cytokinesis, abscission, completed primary septum digestion splits the two daughter cells apart, this is thought to be driven by turgor pressure (Atilgan
et al., 2015). The primary septum is hydrolyzed by the digestive endo-(1,3)-beta-glucanase, Eng1p (Martin-Cuadrado
et al., 2003), and the endo-(1,3)-alpha-glucanase, Agn1p (Dekker
et al., 2004). Membrane trafficking events during abscission are tightly regulated to ensure complete abscission, where septins complexes are involved in the delivery of the digestive glucanases (Zheng
et al., 2018), interestingly, a recently characterized different Anillin homologue, Mid2p is required for this process (Tasto
et al., 2003). It will be interesting to discover how Mid2p regulate the later stages of cytokinesis. Septum synthesis, coordination with ACR constriction and the abscission mechanism are schematically described in
Figure 3.
4. Insights into the Role of ESCRT Machinery During Cytokinesis
4.1. The Role of ESCRTs in Mammalian Cytokinesis
The endosomal sorting complex required for transport (ESCRT) genes were first characterized in S. cerevisiae as vacuolar protein sorting (VPS) genes, as class E Vps mutants were found to have impaired sorting of vacuolar proteins (Raymond et al., 1990; Raymond et al., 1992; Bryant and Stevens, 1998). An additional ESCRT component, the AAA-type ATPase VPS4, was revealed to regulate membrane association of the VPS protein complex to control endosome function (Babst et al., 1998). Subsequently, the ESCRT eukaryotic membrane remodeling machinery was shown to play major roles in other important cellular processes including release of intralumenal vesicles (ILVs) during the multivesicular bodies (MVBs) formation (Liu et al., 2015), centrosome dynamics (Morita et al., 2010), and separation of daughter cells during cytokinesis (Morita et al., 2007). In this section, we will discuss dynamics of the ESCRT/VPS4 membrane remodeling during cytokinesis, as their role in other remodeling processes has been reviewed elsewhere (Bhutta et al., 2014a; Scourfield and Martin-Serrano, 2017; Stoten and Carlton, 2018).
The ESCRT machinery is composed of four complexes: ESCRT-0, ESCRT-I, ESCRT-II, and ESCRT-III, with the latter complex required for all ESCRT activities preforming the core function of membrane remodeling. ESCRT-III being the most evolutionary ancient ESCRT member, has a unique ability to catalyze fission of membrane necks from their luminal side. The ESCRT-III machinery is composed of 12 proteins named charged multivesicular body proteins (CHMPs) and plays a vital role during mammalian cell cytokinesis.
Numerous studies have dissected the behavior of ESCRTs during cytokinesis (reviewed by Nähse
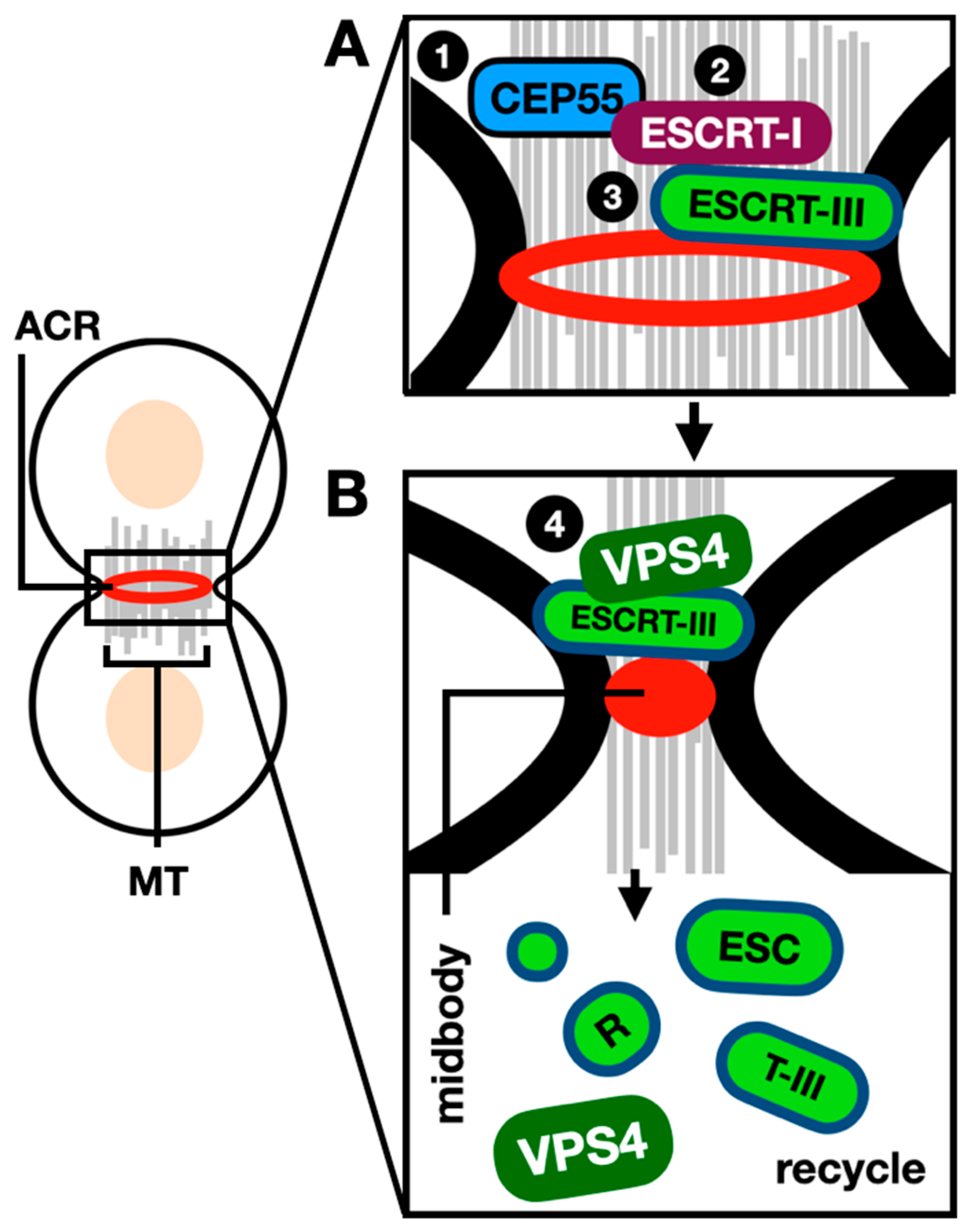
et al., 2017; McCullough and Sundquist, 2020). Briefly, during mammalian cell cytokinesis, ingression of the cleavage furrow creates a thin intracellular bridge connecting the two daughter cells named the midbody. ESCRT-I protein tumor-susceptibility gene 101 (TSG101) and ESCRT-I adaptor apoptosis-linked gene 2-interacting protein X (ALIX) interact with the centrosomal protein of 55 kDa (CEP55) at the midbody (Morita
et al., 2007). The ESCRT-III protein CHMP4B is then recruited to the site of constriction on both sites of the midbody center, polymerization of ESCRT-III into a spiral on the side of the microtubule bridge occurs, and VPS4 next catalyzes rearrangement of the spiral (Elia
et al., 2011; Elia
et al., 2012). VPS4 is the only nucleotide hydrolase in the ESCRT machinery (Babst
et al., 1998), and it functions by pulling ESCRT-III subunits out of the filament to unfold their polypeptide chain through a central pore; this is achieved via cycles of binding ATP, hydrolysis, and ADP release (Yang
et al., 2015; Monroe
et al., 2017). In earlier studies VPS4 was detected at the site of division only during the last stages of abscission (Elia
et al., 2011). However, more recently, VPS4 was also detected during the early stages of abscission and found to be required for ESCRT-III turnover at the midbody (Mierzwa
et al., 2017). The role of the ESCRTs during mammalian cell cytokinesis is schematically described in
Figure 4.
4.2. The Role of ESCRTs in Fission Yeast Cytokinesis
It is established that ESCRT-III recruits VPS4 to remodel filaments driving the midbody constriction in mammalian cells (Elia et al, 2012; Mierzwa et al., 2017; Pfitzner et al., 2020; Pfitzner et al., 2021). However, the exact mechanism of how ESCRT-III and VPS4 catalyze membrane fission during abscission in other model organisms including S. pombe remains unclear.
Sequencing the S. pombe genome revealed that the ESCRT proteins are conserved in this yeast species (Takegawa et al., 1995; Takegawa et al., 2003), and it was later confirmed that Class E Vps proteins regulate vesicle-mediated protein sorting (Iwaki et al., 2007). ESCRTs are also involved in the spindle pole body (SPB) dynamics, however, and there are many other unexplored functions of ESCRTs in S. pombe, including their role in sealing newly formed nuclear envelopes and during cytokinesis. ESCRTs demonstrate strong genetic interactions with SPB proteins and transmembrane nucleoporins, suggesting a role of ESCRTs in nuclear remodeling (Frost et al., 2012). An interesting study explored the ESCRT-mediated nuclear envelope closure in mammalian and S. pombe cells and showed that ESCRT-III mutants and vps4Δ cells display a defective SPB amplification and severe defects in nuclear integrity and morphology (Gu et al., 2017).
Emerging evidence suggest an important role of the ESCRT machinery during S. pombe cytokinesis. Septation is defective in individual S. pombe ESCRT mutants and vps4Δ cells remain attached during septation and fail to separate (Bhutta et al., 2014b). Furthermore, a genetic screen investigated genetic interactions between each of the S. pombe ESCRT genes and the anillin-encoding mid1 gene. Amongst all ESCRTs, only the vps4 gene showed genetic interactions as a mid1Δ vps4Δ double mutant demonstrated synthetic lethality (Rezig et al., 2021). Furthermore, the Vps4p protein has been suggested to interact directly or indirectly with the C-terminal domain of anillin/Mid1p, probably through the lipid-binding PH domain. But how is this coordination carried out if Mid1p leaves the ACR before the onset of abscission, when Vps4p is activated?
Until recently it was not understood how Mid1p leaves the site of division upon the ACR constriction. However, part of this has been explained when it was recently found that phosphorylation of Mid1p by Sid2p leads to Mid1p’s cortical dissociation from the plasma membrane attached ACR (Willet et al., 2019). It is possible that a direct or indirect coordination between the ATPase Vps4p and the SIN kinase Sid2p might regulate Mid1p’s disassociation, leading to its nuclear re-localization during the later stages of cytokinesis. Investigating the co-localization of Mdi1p, Vps4p and Sid2p in S. pombe could be informative in this regard. Additionally, it is known from time lapse imaging experiments that S. pombe cells lacking Sid2p phospho-sites cause Mid1p to maintain cortical attachment even at the onset of the ACR constriction (Willet et al., 2019). Therefore, similarly, tracking the localization of Mid1p in vps4Δ mutant cells throughout the cell cycle could lead to a clearer understanding of the ESCRT machinery dynamics in regulating S. pombe cytokinesis.
5. Concluding Remarks
Studying the function of gene products and their localization through the cell cycle in S. pombe has been a powerful way to understand eukaryotic cell division, especially with advanced live cell imaging, fluorescent macromolecules, and the integration of different research fields including cell molecular biology, cell mechanics and mathematical modelling. This enabled the generation of a model of S. pombe cytokinesis with a potential role of ESCRT proteins as described in the last section of this review. In summary, a ring first forms at the equator of the cell, and while the new cell-wall material is deposited through membrane trafficking, the ring constricts through a complex network of forces balanced by coordination between ring constriction and septum synthesis. Digestive enzymes then cut the remaining septum material connecting the two daughter cells leading to their final separation. During mammalian cytokinesis, ESCRTs and VPS4 localize to the abscission site implying their important role in this process, however similar localization experiments are necessary to confirm if this is the case during S. pombe abscission. This review addresses the dynamic nature of S. pombe cytokinesis and presents a visualized picture of current understanding of the proteins and processes that control this important event. It is anticipated that these mechanisms will be conserved across all eukaryotes and so will be informative about human cells and disease conditions, where they are defective.
Disclosure statement
No potential conflict of interest was reported by the author(s).
References
- Akamatsu M, Berro J, Pu KM, et al. Cytokinetic nodes in fission yeast arise from two distinct types of nodes that merge during interphase. J. Cell. Biol. 2014, 204, 977–88. [Google Scholar] [CrossRef] [PubMed]
- Akamatsu M, Lin Y, Bewersdorf J, et al. Analysis of interphase node proteins in fission yeast by quantitative and superresolution fluorescence microscopy. Mol. Biol. Cell. 2017, 28, 3203–3214. [Google Scholar] [CrossRef] [PubMed]
- Almonacid M, Celton-Morizur S, Jakubowski JL, et al. Temporal control of contractile ring assembly by Plo1 regulation of Myosin II recruitment by Mid1/Anillin. Curr. Biol. 2011, 21, 473–479. [Google Scholar] [CrossRef] [PubMed]
- Atilgan E, Magidson V, Khodjakov A, et al. Morphogenesis of the fission yeast cell through cell wall expansion. Curr. Biol 2015, 25, 2150–2157. [CrossRef]
- Babst M, Wendland B, Estepa EJ, et al. The Vps4p AAA ATPase regulates membrane association of a Vps protein complex required for normal endosome function. EMBO J. 1998, 17, 2982–2993. [Google Scholar] [CrossRef]
- Bähler J, Steever AB, Wheatley S, et al. Role of polo kinase and Mid1p in determining the site of cell division in fission yeast. J. Cell. Biol. 1998, 143, 1603–16. [Google Scholar] [CrossRef]
- Bellingham-Johnstun K, Anders EC, Ravi J, et al. Molecular organization of cytokinesis node predicts the constriction rate of the contractile ring. J. Cell. Biol. 2021, 220, e202008032. [Google Scholar]
- Bhattacharjee R, Hall AR, Mangione MC, et al. Multiple polarity kinases inhibit phase separation of F-BAR protein Cdc15 and antagonize cytokinetic ring assembly in fission yeast. Elife. 2023, 7, e83062. [Google Scholar]
- Bhattacharjee R, Mangione MC, Wos M, et al. DYRK kinase Pom1 drives F-BAR protein Cdc15 from the membrane to promote medial division. Mol. Biol. Cell. 2020, 31, 917–929. [Google Scholar] [CrossRef]
- Bhutta MS, McInerny CJ, Gould GW. ESCRT function in cytokinesis: location, dynamics and regulation by mitotic kinases. Int. J. Mol. Sci. 2014, 15, 21723–39. [Google Scholar] [CrossRef] [PubMed]
- Bhutta MS, Roy B, Gould GW, et al. A complex network of interactions between mitotic kinases, phosphatases and ESCRT proteins regulates septation and membrane trafficking in S. pombe. PLoS. One. 2014, 9, e111789. [Google Scholar]
- Bryant NJ, Stevens TH. Vacuole biogenesis in Saccharomyces cerevisiae: protein transport pathways to the yeast vacuole. Microbiol. Mol. Biol. Rev. 1998, 62, 230–47. [Google Scholar] [CrossRef] [PubMed]
- Campbell BF, Hercyk BS, Williams AR, et al. Cdc42 GTPase activating proteins Rga4 and Rga6 coordinate septum synthesis and membrane trafficking at the division plane during cytokinesis. Traffic. 2022, 23, 478–495. [Google Scholar] [CrossRef] [PubMed]
- Chang, F. Forces that shape fission yeast cells. Mol. Biol. Cell. 2017, 28, 1819–1824. [Google Scholar] [CrossRef]
- Chang F, Nurse P. Finishing the cell cycle: control of mitosis and cytokinesis in fission yeast. Trends. Genet. 1993, 9, 333–5. [Google Scholar] [CrossRef] [PubMed]
- Chang F, Nurse P. How fission yeast fission in the middle. Cell. 1996, 84, 191–4. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee M, Pollard TD. The functionally important N-terminal half of fission yeast Mid1p Anillin Is intrinsically disordered and undergoes phase separation. Biochemistry. 2019, 58, 3031–3041. [Google Scholar] [CrossRef] [PubMed]
- Chew TG, Lim TC, Osaki Y, et al. Inhibition of cell membrane ingression at the division site by cell walls in fission yeast. Mol. Biol. Cell. 2020, 31, 2306–2314. [Google Scholar] [CrossRef]
- Cortés JC, Pujol N, Sato M, et al. Cooperation between Paxillin-like protein Pxl1 and glucan synthase Bgs1 is essential for actomyosin ring stability and septum formation in fission yeast. PLoS. Genet. 2015, 11, e1005358. [Google Scholar]
- Cortés JC, Ramos M, Konomi M, et al. Specific detection of fission yeast primary septum reveals septum and cleavage furrow ingression during early anaphase independent of mitosis completion. PLoS. Genet. 2018, 14, e1007388. [Google Scholar]
- Daga RR, Chang F. Dynamic positioning of the fission yeast cell division plane. Proc. Natl. Acad. Sci. USA. 2005, 102, 8228–8232. [Google Scholar] [CrossRef]
- Daga RR, Yonetani A, Chang F. Asymmetric microtubule pushing forces in nuclear centering. Curr. Biol. 2006, 16, 1544–1550. [Google Scholar] [CrossRef]
- Dekker N, Speijer D, Grun CH, et al. Role of the alpha-glucanase Agn1p in fission-yeast cell separation. Mol. Biol. Cell. 2004, 15, 3903–3914. [Google Scholar] [CrossRef]
- Elia N, Sougrat R, Spurlin TA, et al. Dynamics of endosomal sorting complex required for transport (ESCRT) machinery during cytokinesis and its role in abscission. Proc. Natl. Acad. Sci. USA. 2011, 108, 4846–4851. [Google Scholar] [CrossRef] [PubMed]
- Elia N, Fabrikant G, Kozlov MM, et al. Computational model of cytokinetic abscission driven by ESCRT-III polymerization and remodeling. Biophys. J. 2012, 102, 2309–20. [Google Scholar] [CrossRef]
- Frost A, Elgort MG, Brandman O, et al. Functional repurposing revealed by comparing S. pombe and S. cerevisiae genetic interactions. Cell. 2012, 149, 1339–52. [Google Scholar] [CrossRef] [PubMed]
- Gallardo P, Barrales RR, Daga RR, et al. Nuclear mechanics in the fission yeast. Cells. 2019, 8, 1285. [Google Scholar] [CrossRef] [PubMed]
- Gerganova V, Floderer C, Archetti A, et al. Multi-phosphorylation reaction and clustering tune Pom1 gradient mid-cell levels according to cell size. Elife. 2019, 3, e45983. [Google Scholar]
- Gu M, LaJoie D, Chen OS, et al. LEM2 recruits CHMP7 for ESCRT-mediated nuclear envelope closure in fission yeast and human cells. Proc. Natl. Acad. Sci. USA. 2017, 114, E2166–E2175. [Google Scholar]
- Hartwell LH, Culotti J, Reid B. Genetic control of the cell-division cycle in yeast. I. Detection of mutants. Proc. Natl. Acad. Sci. USA. 1970, 66, 352–359. [Google Scholar] [CrossRef]
- Hartwell LH, Mortimer RK, Culotti J, et al. Genetic control of the cell division cycle in yeast: V. Genetic analysis of cdc mutants. Genetics. 1973, 74, 267–286. [Google Scholar] [CrossRef]
- Hercyk BS, Onwubiko UN, Das ME. Coordinating septum formation and the actomyosin ring during cytokinesis in Schizosaccharomyces pombe. Mol. Microbiol. 2019, 112, 1645–1657. [Google Scholar] [CrossRef] [PubMed]
- Iwaki T, Onishi M, Ikeuchi M, et al. Essential roles of class E Vps proteins for sorting into multivesicular bodies in Schizosaccharomyces pombe. Microbiology (Reading). 2007, 153(Pt 8), 2753–2764. [Google Scholar]
- Khodjakov A, Cole RW, Rieder CL. A synergy of technologies: combining laser microsurgery with green fluorescent protein tagging. Cell. Motil. Cytoskeleton. 1997, 38, 311–7. [Google Scholar] [CrossRef]
- Laplante C, Huang F, Tebbs IR, et al. Molecular organization of cytokinesis nodes and contractile rings by super- resolution fluorescence microscopy of live fission yeast. Proc. Natl. Acad. Sci. 2016, 113, E5876–E5885. [Google Scholar]
- Magliozzi JO, Sears J, Cressey L, et al. Fission yeast Pak1 phosphorylates anillin-like Mid1 for spatial control of cytokinesis. J. Cell. Biol. 2020, 219, e201908017. [Google Scholar]
- Malla M, Pollard TD, Chen Q. Counting actin in contractile rings reveals novel contributions of cofilin and type II myosins to fission yeast cytokinesis. Mol. Biol. Cell. 2022, 33, ar51. [Google Scholar]
- Martin SG, McDonald WH, Yates JR 3rd, et al. Tea4p links microtubule plus ends with the formin for3p in the establishment of cell polarity. Dev. Cell. 2005, 8, 479–91. [Google Scholar] [CrossRef] [PubMed]
- Martin-Cuadrado AB, Duenas E, Sipiczki M, et al. The endo-beta-1,3-glucanase eng1p is required for dissolution of the primary septum during cell separation in Schizosaccharomyces pombe. J. Cell. Sci 2003, 116 (Pt 9), 1689–1698. [CrossRef]
- McCullough J, Sundquist WI. Membrane Remodeling: ESCRT-III Filaments as Molecular Garrotes. Curr. Biol. 2020, 30, R1425–R1428. [Google Scholar] [CrossRef]
- McDonald NA, Lind AL, Smith SE, et al. Nanoscale architecture of the Schizosaccharomyces pombe contractile ring. Elife 2017, 15, e28865.
- Mierzwa BE, Chiaruttini N, Redondo-Morata L, et al. Dynamic subunit turnover in ESCRT-III assemblies is regulated by Vps4 to mediate membrane remodelling during cytokinesis. Nat. Cell. Biol. 2017, 19, 787–798. [Google Scholar] [CrossRef] [PubMed]
- Moshtohry M, Bellingham-Johnstun K, Elting MW, et al. Laser ablation reveals the impact of Cdc15p on the stiffness of the contractile ring. Mol. Biol. Cell. 2022, 33, br9. [Google Scholar]
- Morita E, Sandrin V, Chung HY, et al. Human ESCRT and ALIX proteins interact with proteins of the midbody and function in cytokinesis. EMBO. J. 2007, 26, 4215–4227. [Google Scholar] [CrossRef] [PubMed]
- Nähse V, Christ L, Stenmark H, et al. The Abscission Checkpoint: Making It to the Final Cut. Trends. Cell. Biol. 2017, 27, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Nurse, P. Genetic control of cell size at cell division in yeast. Nature. 1975, 256, 547–551. [Google Scholar] [CrossRef] [PubMed]
- Nurse, P. Fission yeast cell cycle mutants and the logic of eukaryotic cell cycle control. Mol. Biol. Cell. 2020, 31, 2871–2873. [Google Scholar] [CrossRef] [PubMed]
- Nurse P, Thuriaux P, Nasmyth K. Genetic control of the cell division cycle in the fission yeast Schizosaccharomyces pombe. Mol. Gen. Genet. 1976, 146, 167–78. [Google Scholar] [CrossRef]
- Onwubiko UN, Rich-Robinson J, Mustaf RA, et al. Cdc42 promotes Bgs1 recruitment for septum synthesis and glucanase localization for cell separation during cytokinesis in fission yeast. Small GTPases. 2021, 12, 257–264. [Google Scholar] [CrossRef]
- Paoletti A, Chang F. Analysis of mid1p, a protein required for placement of the cell division site, reveals a link between the nucleus and the cell surface in fission yeast. Mol. Biol. cell. 2000, 11, 2757–2773. [Google Scholar] [CrossRef]
- Pérez P, Cortés JCG, Cansado J, et al. Fission yeast cell wall biosynthesis and cell integrity signalling. Cell. Surf. 2018, 4, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Pérez P, Portales E, Santos B. Rho4 interaction with exocyst and septins regulates cell separation in fission yeast. Microbiology (Reading). 2015, 161(Pt 5), 948–959. [Google Scholar]
- Pfitzner A-K, Mercier V, Jiang X, et al. An ESCRT-III Polymerization Sequence Drives Membrane Deformation and Fission. Cell. 2020, 182, 1140–1155. [Google Scholar] [CrossRef]
- Pfitzner A-K, Moser von Filseck J, Roux A. Principles of membrane remodeling by dynamic ESCRT-III polymers. Trends. Cell. Biol. 2021, 31, 856–868. [Google Scholar] [CrossRef] [PubMed]
- Piel M, Tran PT. Cell shape and cell division in fission yeast. Curr. Biol. 2009, 19, R823–R827. [Google Scholar] [CrossRef] [PubMed]
- Proctor SA, Minc N, Boudaoud A, et al. Contributions of turgor pressure, the contractile ring, and septum assembly to forces in cytokinesis in fission yeast. Curr. Biol. 2012, 22, 1601–1608. [Google Scholar] [CrossRef]
- Pollard TD, Wu JQ. Understanding cytokinesis: lessons from fission yeast. Nat. Rev. Mol. Cell. Biol. 2010, 11, 149–55. [Google Scholar] [CrossRef]
- Raymond CK, O'Hara PJ, Eichinger G, et al. Molecular analysis of the yeast VPS3 gene and the role of its product in vacuolar protein sorting and vacuolar segregation during the cell cycle. J. Cell. Biol. 1990, 111, 877–92. [Google Scholar] [CrossRef]
- Raymond CK, Howald-Stevenson I, Vater CA, et al. Morphological classification of the yeast vacuolar protein sorting mutants: evidence for a prevacuolar compartment in class E vps mutants. Mol. Biol. Cell. 1992, 3, 1389–402. [Google Scholar] [CrossRef]
- Rezig IM, Yaduma WG, Gould GW, et al. Anillin/Mid1p interacts with the ESCRT-associated protein Vps4p and mitotic kinases to regulate cytokinesis in fission yeast. Cell Cycle. 2021, 20, 1845–1860. [Google Scholar] [CrossRef]
- Rezig IM, Yaduma WG, Gould GW, et al. The role of anillin/Mid1p during medial division and cytokinesis: from fission yeast to cancer cells. Cell Cycle. 2022, 22, 633–644. [Google Scholar]
- Rincon SA, Estravis M, Dingli F, et al. SIN-dependent dissociation of the SAD kinase Cdr2 from the cell cortex resets the division plane. Curr. Biol. 2017, 27, 534–542. [Google Scholar] [CrossRef]
- Saha S, Pollard TD. Anillin-related protein Mid1p coordinates the assembly of the cytokinetic contractile ring in fission yeast. Mol. Biol. Cell. 2012, 23, 3982–3992. [Google Scholar] [CrossRef]
- Sayyad WA, Pollard TD. The number of cytokinesis nodes in mitotic fission yeast scales with cell size. Elife. 2022, 12, e76249. [Google Scholar]
- Scourfield EJ, Martin-Serrano J. Growing functions of the ESCRT machinery in cell biology and viral replication. Biochem. Soc. Trans. 2017, 45, 613–34. [Google Scholar] [CrossRef]
- Snider CE, Bhattacharjee R, Igarashi MG, et al. Fission yeast paxillin contains two Cdc15 binding motifs for robust recruitment to the cytokinetic ring. Mol. Biol. Cell. 2022, 33, br4. [Google Scholar]
- Snider CE, Chandra M, McDonald NA, et al. Opposite surfaces of the Cdc15 f-bar domain create a membrane platform that coordinates cytoskeletal and signaling components for cytokinesis. Cell. Rep. 2020, 33, 108526. [Google Scholar] [CrossRef] [PubMed]
- Snider CE, Wan Mohamad Noor WNI, Nguyen NTH, et al. The state of F-BAR domains as membrane-bound oligomeric platforms. Trends. Cell. Biol. 2021, 31, 644–655. [Google Scholar] [CrossRef]
- Sohrmann M, Fankhauser C, Brodbeck C, et al. The dmf1/mid1 gene is essential for correct positioning of the division septum in fission yeast. Genes. Dev. 1996, 10, 2707–2719. [Google Scholar] [CrossRef] [PubMed]
- Stoten CL, Carlton JG. ESCRT-dependent control of membrane remodelling during cell division. Semin. Cell. Dev. Biol. 2018, 74, 50–65. [Google Scholar] [CrossRef] [PubMed]
- Swulius MT, Nguyen LT, Ladinsky MS, et al. Structure of the fission yeast actomyosin ring during constriction. Proc. Natl. Acad. Sci. USA. 2018, 115, e1455–E1464. [Google Scholar]
- Takegawa K, DeWald DB, Emr SD. Schizosaccharomyces pombe Vps34p, a phosphatidylinositol-specific PI 3-kinase essential for normal cell growth and vacuole morphology. J. Cell. Sci 1995, 108 (Pt 12), 3745–56. [CrossRef]
- Takegawa K, Iwaki T, Fujita Y, et al. Vesicle-mediated protein transport pathways to the vacuole in Schizosaccharomyces pombe. Cell. Struct. Funct. 2003, 28, 399–417. [Google Scholar] [CrossRef]
- Tasto JJ, Morrell JL, Gould KL. An anillin homologue, Mid2p, acts during fission yeast cytokinesis to organize the septin ring and promote cell separation. J. Cell. Biol, 2003, 160, 1093–103. [Google Scholar] [CrossRef]
- Tran PT, Marsh L, Doye V, et al. A mechanism for nuclear positioning in fission yeast based upon microtubule pushing. J. Cell. Biol 2001, 153, 397–411. [CrossRef]
- Vavylonis D, Wu JQ, Hao S, et al. Assembly mechanism of the contractile ring for cytokinesis by fission yeast. Science. 2008, 319, 97–100. [Google Scholar] [CrossRef]
- Wei B, Hercyk BS, Mattson N, et al. Unique spatiotemporal activation pattern of Cdc42 by Gef1 and Scd1 promotes different events during cytokinesis. Mol. Biol. Cell. 2016, 27, 1235–45. [Google Scholar] [CrossRef]
- Willet AH, DeWitt AK, Beckley JR, et al. NDR kinase Sid2 drives Anillin-like Mid1 from the membrane to promote cytokinesis and medial division site placement. Curr. Biol. 2019, 29, 1055–1063. [Google Scholar] [CrossRef]
- Wu JQ, Sirotkin V, Kovar DR, et al. Assembly of the cytokinetic contractile ring from a broad band of nodes in fission yeast. J. Cell. Biol. 2006, 174, 391–402. [Google Scholar] [CrossRef]
- Yang B, Stjepanovic G, Shen Q, et al. Vps4 disassembles an ESCRT-III filament by global unfolding and processive translocation. Nat. Struct. Mol. Biol. 2015, 22, 492–498. [Google Scholar] [CrossRef]
- Zimmermann D, Homa KE, Hocky GM, et al. Mechanoregulated inhibition of formin facilitates contractile actomyosin ring assembly. Nat. Commun. 2017, 8, 703. [Google Scholar] [CrossRef] [PubMed]
- Zheng S, Dong F, Rasul F, et al. Septins regulate the equatorial dynamics of the septation initiation network kinase Sid2p and glucan synthases to ensure proper cytokinesis. FEBS. J. 2018, 285, 2468–2480. [Google Scholar] [CrossRef]
- Zhu YH, Ye Y, Wu Z, et al. Cooperation between Rho-GEF Gef2 and its binding partner Nod1 in the regulation of fission yeast cytokinesis. Mol. Biol. Cell. 2013, 24, 3187–3204. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).