Submitted:
12 December 2023
Posted:
12 December 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
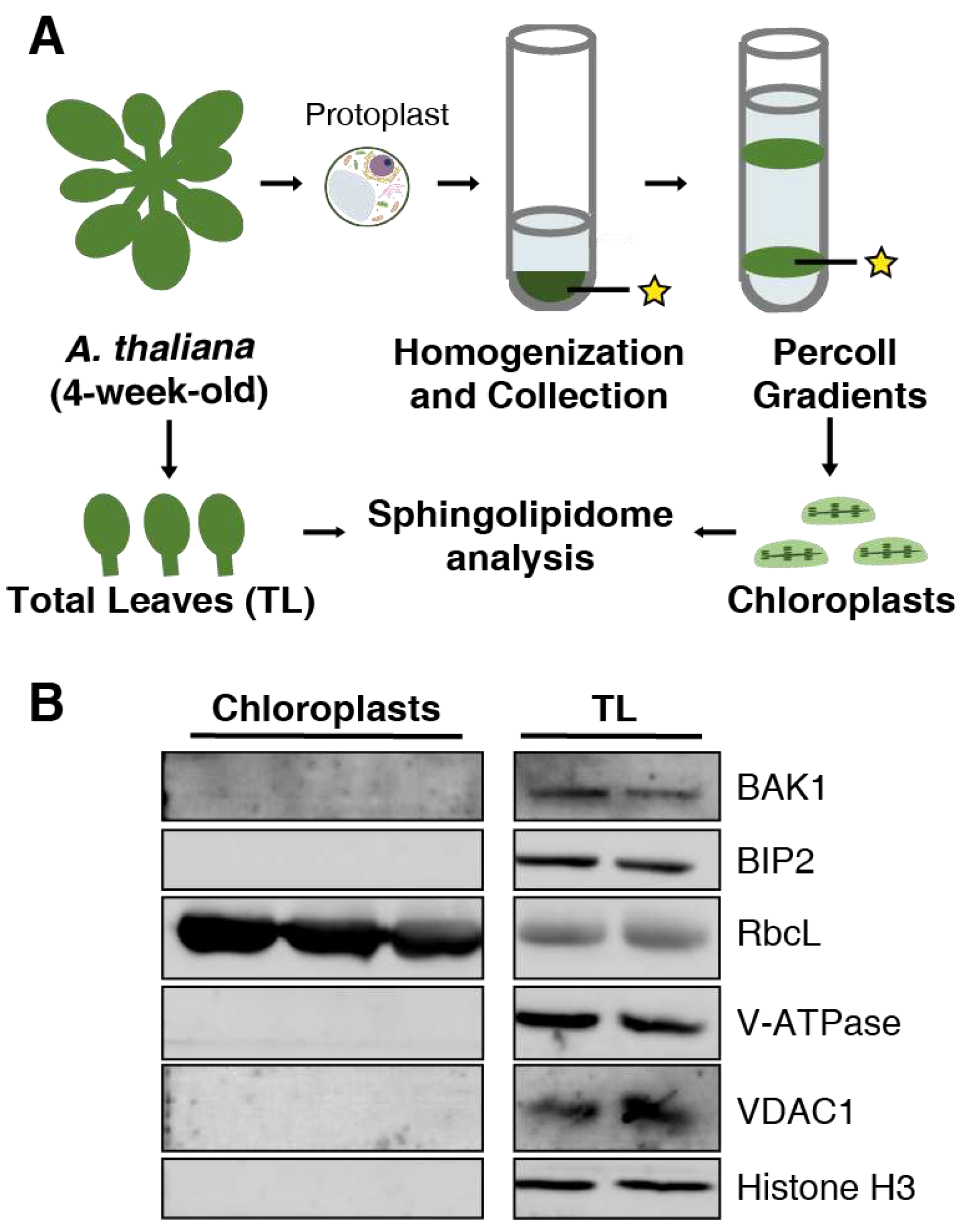
2.1. Purification of chloroplast fractions
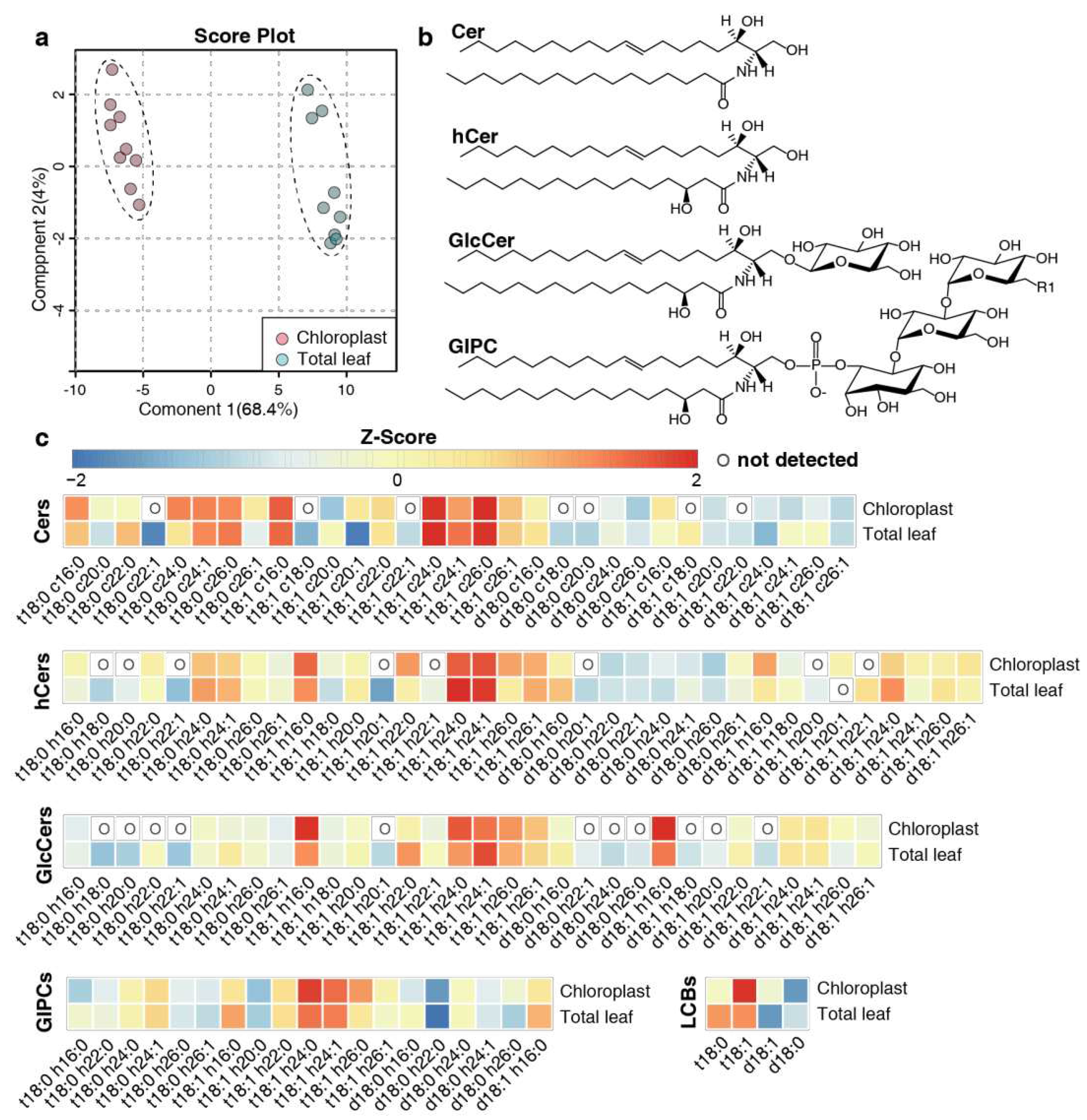
2.2. Overall assessment of sphingolipid profiles
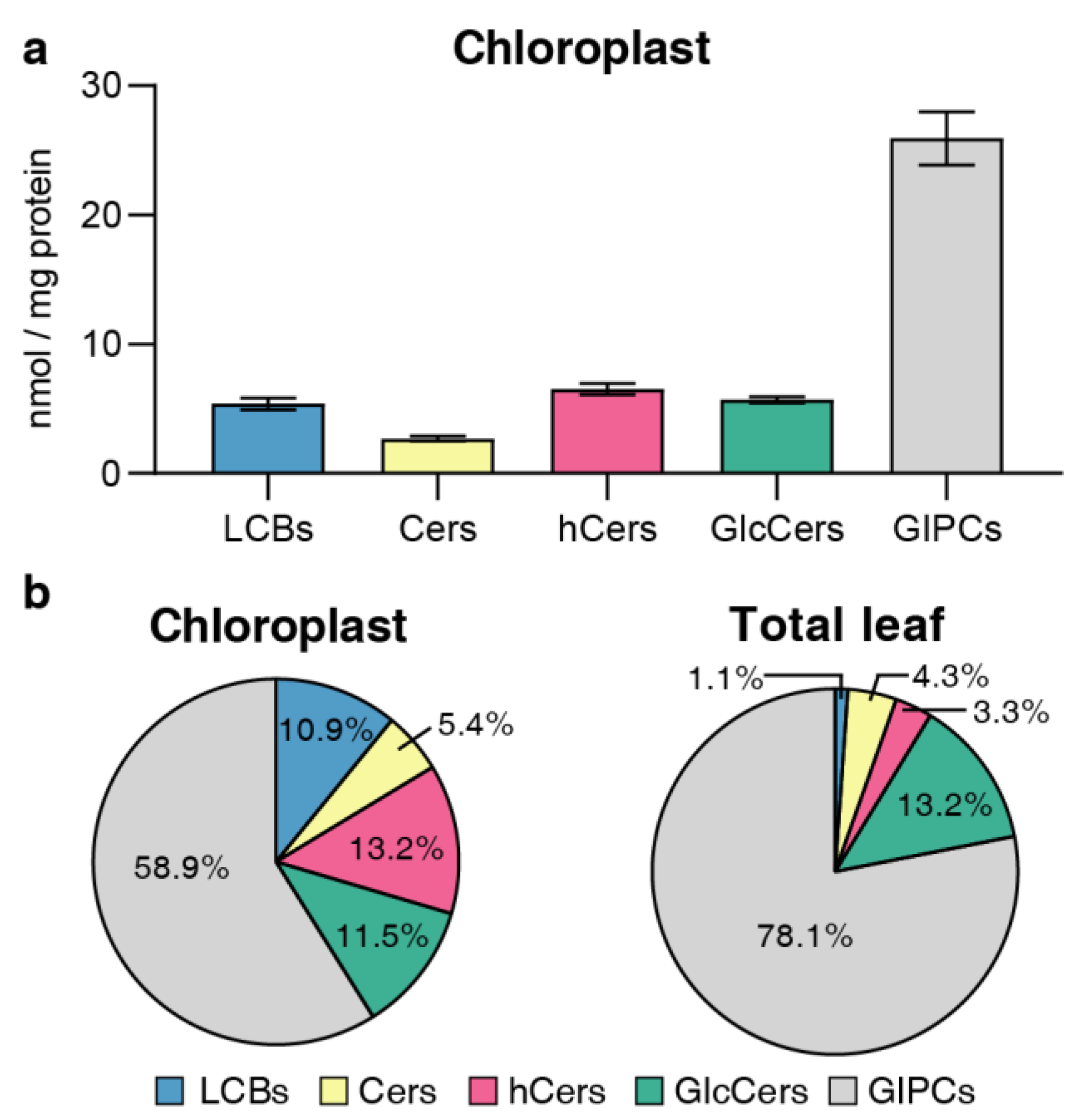
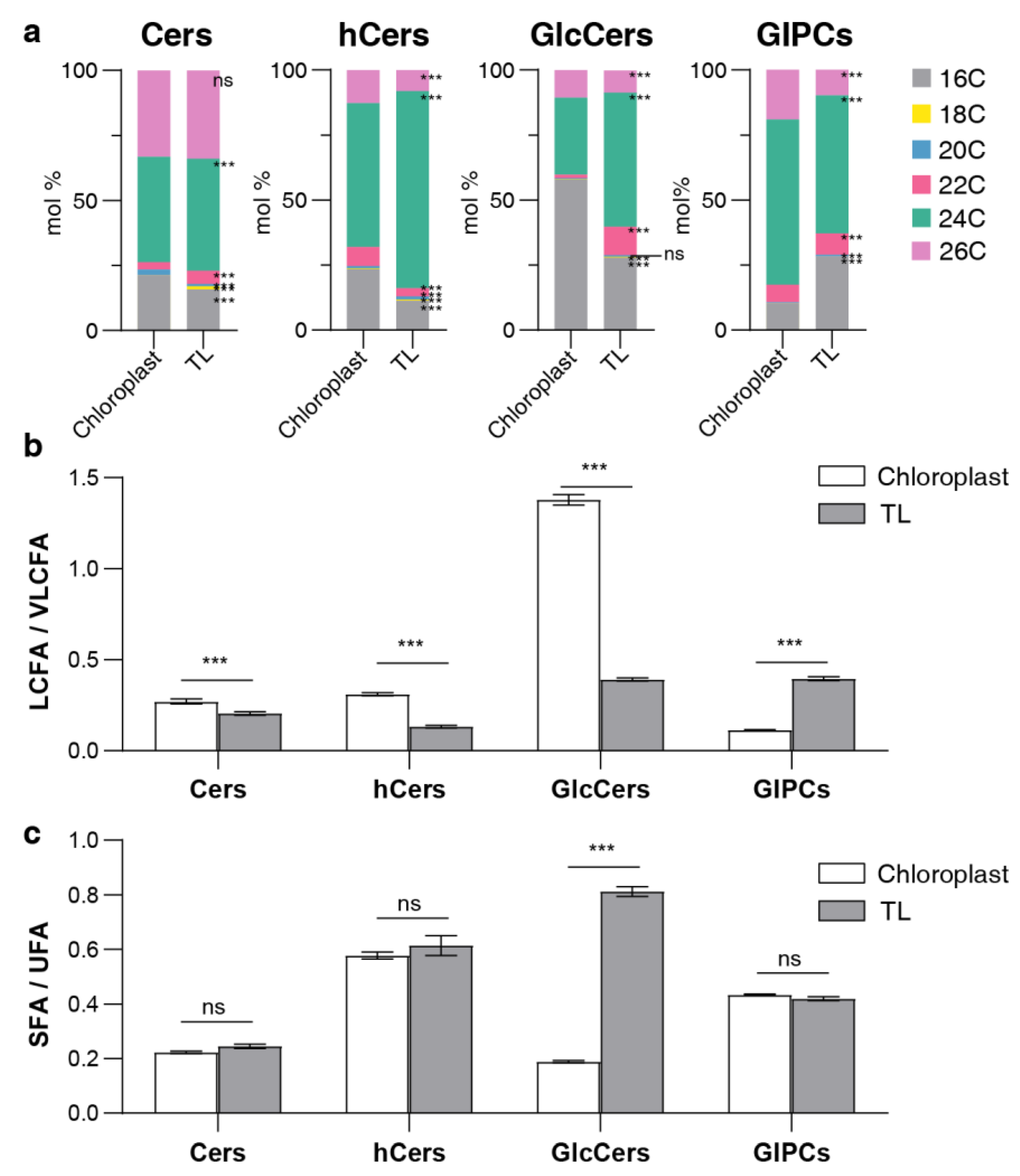
2.3. Distribution of sphingolipid classes
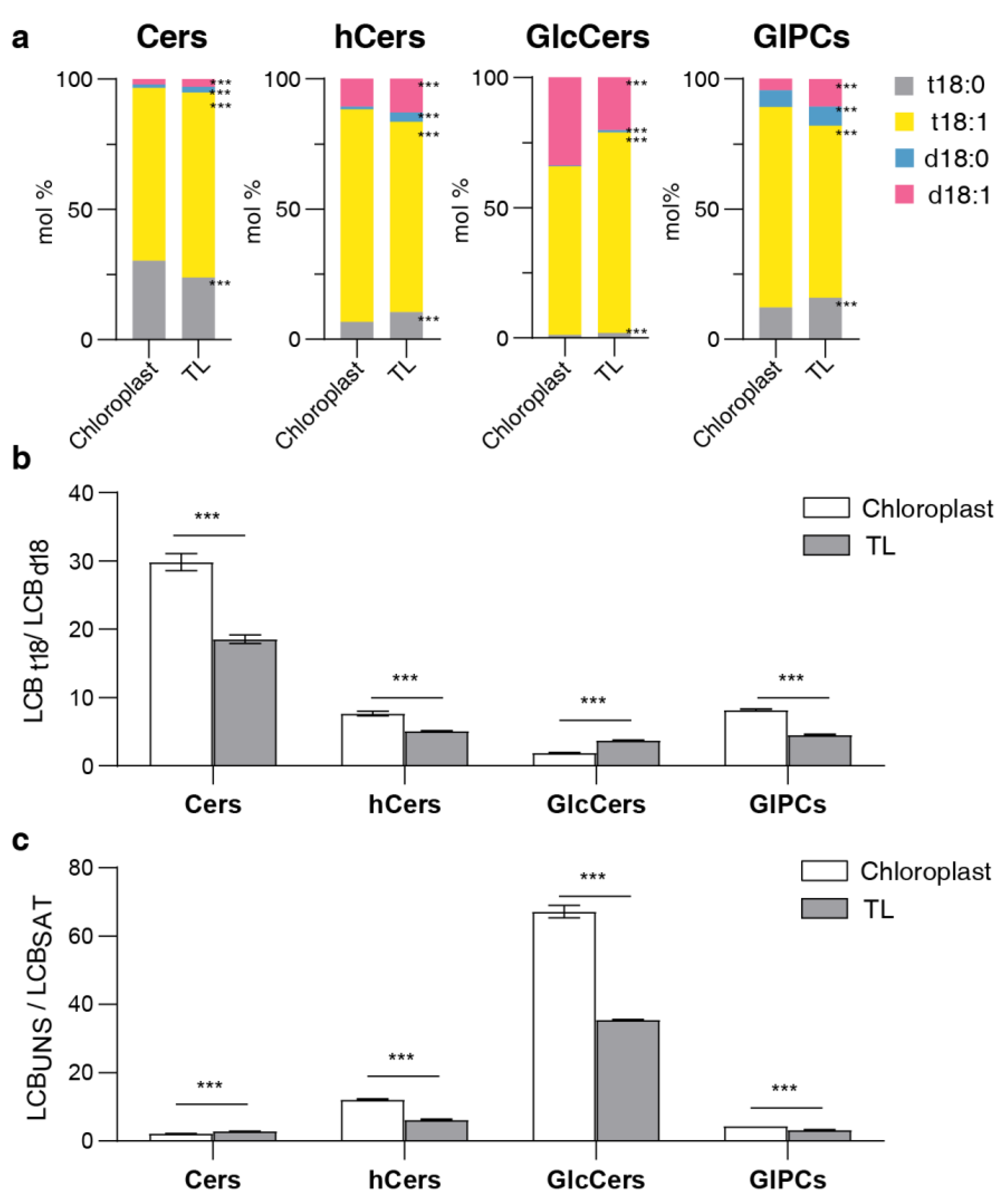
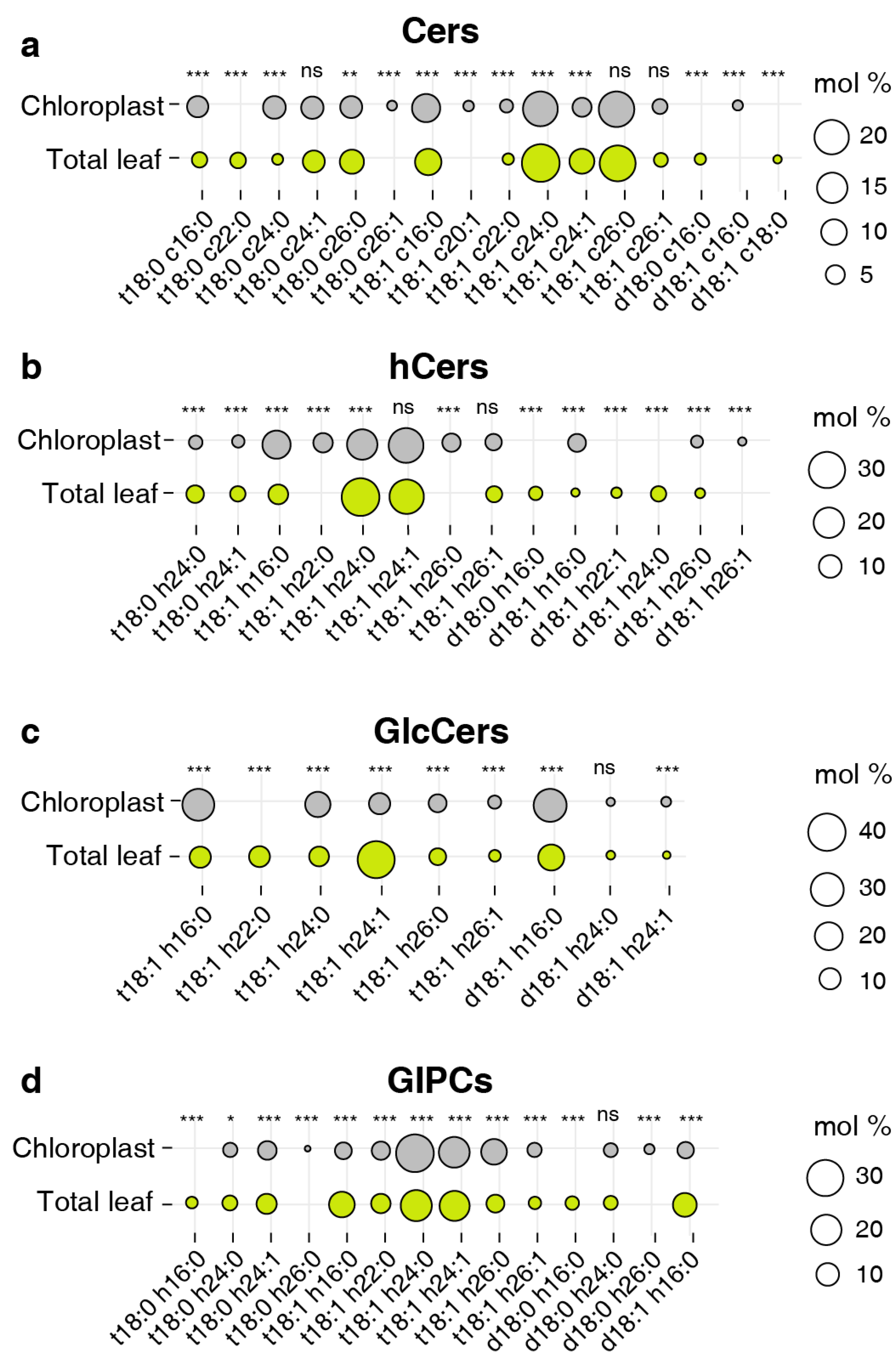
2.4. LCB and FA profiling of sphingolipids
2.5. Predominant sphingolipid species
3. Discussion
4. Materials and Methods
4.1. Plants and growth conditions
4.2. Chloroplast isolation
4.3. Protein quantification and immunoblotting assays
4.4. Sphingolipid analysis
4.5. Data analysis
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Heaver, S.L.; Johnson, E.L.; Ley, R.E. Sphingolipids in host-microbial interactions. Curr. Opin. Microbiol, 2018, 43, 92-99. [CrossRef]
- Sperling, P.; Heinz, E. Plant sphingolipids: structural diversity, biosynthesis, first genes and functions. Biochim Biophys Acta, 2003, 1632, 1-15. [CrossRef]
- Grison, M.S.; Brocard, L.; Fouillen, L.; Nicolas, W.; Wewer, V.; Dörmann, P.; Nacir, H.; Benitez-Alfonso, Y.; Claverol, S.; Germain, V.; et al. Specific membrane lipid composition is important for plasmodesmata function in Arabidopsis. Plant Cell, 2015, 27, 1228-1250. [CrossRef]
- Simons, K.; van Meer, G. Lipid sorting in epithelial cells. Biochemistry, 1988, 27, 6197-6202. [CrossRef]
- Abbas, H.K.; Tanaka, T.; Duke, S.O.; Porter, J.K.; Wray, E.M.; Hodges, L.; Sessions, A.E.; Wang, E.; Merrill, A.H.; Jr; et al. Fumonisin-and AAL-toxin-induced disruption of sphingolipid metabolism with accumulation of free sphingoid bases. Plant Physiology (Rockville) , 1994, 106, 1085-1093. [CrossRef]
- Ng, C.K.; Carr, K.; Mcainsh, M.R.; Powell, B.; Hetherington, A.M. Drought-induced guard cell signal transduction involves sphingosine-1-phosphate. Nature, 2001, 410, 596-599. [CrossRef]
- Msanne, J.; Chen, M.; Luttgeharm, K.D.; Bradley, A.M.; Mays, E.S.; Paper, J.M.; Boyle, D.L.; Cahoon, R.E.; Schrick, K.; Cahoon, E.B. Glucosylceramides are critical for cell-type differentiation and organogenesis, but not for cell viability in Arabidopsis. Plant J., 2015, 84, 188-201. [CrossRef]
- Liao, P.; Huang, J.; Tong, P.; Nie, W.; Yan, X.; Feng, Y.; Peng, H.; Peng, X.; Li, S. Characterization and expression analysis of inositolphosphorylceramide synthase family genes in rice (Oryza sativa L.). Genes Genom. 2017, 39, 485-492. [CrossRef]
- Liu, N.J.; Zhang, T.; Liu, Z.H.; Chen, X.; Guo, H.S.; Ju, B.H.; Zhang, Y.Y.; Li, G.Z.; Zhou, Q.H.; Qin, Y.M.; et al. Phytosphinganine Affects Plasmodesmata Permeability via Facilitating PDLP5-Stimulated Callose Accumulation in Arabidopsis. Mol. Plant., 2020, 13, 128-143. [CrossRef]
- Moore, W.M.; Chan, C.; Ishikawa, T.; Rennie, E.A.; Wipf, H.M.; Benites, V.; Kawai-Yamada, M.; Mortimer, J.C.; Scheller, H.V. Reprogramming sphingolipid glycosylation is required for endosymbiont persistence in Medicago truncatula. Curr. Biol., 2021, 31, 2374-2385. [CrossRef]
- Sperling, P.; Heinz, E. Plant sphingolipids: structural diversity, biosynthesis, first genes and functions. Biochim. Biophys. Acta Mol. Cell Biol. Lipids, 2003, 1632, 1-15. [CrossRef]
- Markham, J.E.; Li, J.; Cahoon, E.B.; Jaworski, J.G. Separation and identification of major plant sphingolipid classes from leaves. J. Biol. Chem., 2006, 281, 22684-22694. [CrossRef]
- Liu, N.J.; Hou, L.P.; Bao, J.J.; Wang, L.J.; Chen, X.Y. Sphingolipid metabolism, transport, and functions in plants: Recent progress and future perspectives. Plant Commun., 2021, 2, 100214. [CrossRef]
- Luttgeharm, K.D.; Kimberlin, A.N.; Cahoon, E.B. Plant Sphingolipid Metabolism and Function; Nakamura, Y., Libeisson, Y., Eds., 2016; Volume 86, 249-286. ISBN 0306-0225(print). [CrossRef]
- Markham, J.E.; Jaworski, J.G. Rapid measurement of sphingolipids from Arabidopsis thaliana by reversed-phase high-performance liquid chromatography coupled to electrospray ionization tandem mass spectrometry. Rapid Commun. Mass Spectrom., 2007, 21, 1304-1314. [CrossRef]
- Huby, E.; Napier, J.A.; Baillieul, F.; Michaelson, L.V.; Dhondt-Cordelier, S. Sphingolipids: towards an integrated view of metabolism during the plant stress response. New Phytol., 2020, 225, 659-670. [CrossRef]
- Zeng, H.; Yao, N. Sphingolipids in plant immunity. Phytopathol. Res., 2022, 4,. [CrossRef]
- Sperling, P.; Franke, S.; Lüthje, S.; Heinz, E. Are glucocerebrosides the predominant sphingolipids in plant plasma membranes? Plant Physiol. Biochem., 2005, 43, 1031-1038. [CrossRef]
- Tarazona, P.; Feussner, K.; Feussner, I. An enhanced plant lipidomics method based on multiplexed liquid chromatography-mass spectrometry reveals additional insights into cold- and drought-induced membrane remodeling. Plant J., 2015, 84, 621-633. [CrossRef]
- Melser, S.; Batailler, B.; Peypelut, M.; Poujol, C.; Bellec, Y.; Wattelet-Boyer, V.; Maneta-Peyret, L.; Faure, J.D.; Moreau, P. Glucosylceramide biosynthesis is involved in Golgi morphology and protein secretion in plant cells. Traffic, 2010, 11, 479-490. [CrossRef]
- Fouillen, L.; Maneta-Peyret, L.; Moreau, P. ER Membrane Lipid Composition and Metabolism: Lipidomic Analysis. Methods Mol Biol, 2018, 1691, 125-137. [CrossRef]
- Warnecke, D.; Heinz, E. Recently discovered functions of glucosylceramides in plants and fungi. Cell. Mol. Life Sci., 2003, 60, 919-941. [CrossRef]
- Wang, L.Y.; Li, J.; Gong, B.; Wang, R.H.; Chen, Y.L.; Yin, J.; Yang, C.; Lin, J.T.; Liu, H.Z.; Yang, Y.; et al. Orosomucoid proteins limit endoplasmic reticulum stress in plants. The New phytologist, 2023, 240, 1134-1148. [CrossRef]
- Carmona-Salazar, L.; Cahoon, R.E.; Gasca-Pineda, J.; Gonzalez-Solis, A.; Vera-Estrella, R.; Trevino, V.; Cahoon, E.B.; Gavilanes-Ruiz, M. Plasma and vacuolar membrane sphingolipidomes: composition and insights on the role of main molecular species. Plant Physiol., 2021, 186, 624-639. [CrossRef]
- Liu, Y.; Senkler, J.; Herrfurth, C.; Braun, H.; Feussner, I. Defining the lipidome of Arabidopsis leaf mitochondria: Specific lipid complement and biosynthesis capacity. Plant Physiol., 2023. [CrossRef]
- Hölzl, G.; Dörmann, P. Chloroplast Lipids and Their Biosynthesis. Annu. Rev. Plant Biol., 2019, 70, 51-81. [CrossRef]
- Wang, Z.; Benning, C. Chloroplast lipid synthesis and lipid trafficking through ER-plastid membrane contact sites. Biochem. Soc. Trans., 2012, 40, 457-463. [CrossRef]
- Chen, M.; Markham, J.E.; Cahoon, E.B. Sphingolipid Δ8 unsaturation is important for glucosylceramide biosynthesis and low-temperature performance in Arabidopsis. The Plant Journal, 2012, 69, 769-781. [CrossRef]
- Lenarčič, T.; Albert, I.; Böhm, H.; Hodnik, V.; Pirc, K.; Zavec, A.B.; Podobnik, M.; Pahovnik, D.; ~agar, E.; Pruitt, R.; et al. Eudicot plant-specific sphingolipids determine host selectivity of microbial NLP cytolysins. Science, 2017, 358, 1431-1434. [CrossRef]
- Jiang, Z.; Zhou, X.; Tao, M.; Yuan, F.; Liu, L.; Wu, F.; Wu, X.; Xiang, Y.; Niu, Y.; Liu, F.; et al. Plant cell-surface GIPC sphingolipids sense salt to trigger Ca(2+) influx. Nature, 2019, 572, 341-346. [CrossRef]
- Nagano, M.; Ishikawa, T.; Ogawa, Y.; Iwabuchi, M.; Nakasone, A.; Shimamoto, K.; Uchimiya, H.; Kawai-Yamada, M. Arabidopsis Bax inhibitor-1 promotes sphingolipid synthesis during cold stress by interacting with ceramide-modifying enzymes. Planta, 2014, 240, 77-89. [CrossRef]
- Ohlrogge, J.; Pollard, M.; Bao, X.; Focke, M.; Girke, T.; Ruuska, S.; Mekhedov, S.; Benning, C. Fatty acid synthesis: from CO2 to functional genomics. Biochem. Soc. Trans., 2000, 28, 567-573. [CrossRef]
- Rawsthorne, S. Carbon flux and fatty acid synthesis in plants. Prog. Lipid Res., 2002, 41, 182-196. [CrossRef]
- Luttgeharm, K.D.; Kimberlin, A.N.; Cahoon, E.B. Plant Sphingolipid Metabolism and Function. Subcell Biochem, 2016, 86, 249-286. [CrossRef]
- Wang, Z.; Benning, C. Chloroplast lipid synthesis and lipid trafficking through ER-plastid membrane contact sites. Biochem. Soc. Trans., 2012, 40, 457-463. [CrossRef]
- Berkey, R.; Bendigeri, D.; Xiao, S. Sphingolipids and plant defense/disease: the "death" connection and beyond. Front. Plant Sci., 2012, 3, 68. [CrossRef]
- Haslam, T.M.; Feussner, I. Diversity in sphingolipid metabolism across land plants. J. Exp. Bot., 2022, 73, 2785-2798. [CrossRef]
- Cha, H.J.; He, C.; Zhao, H.; Dong, Y.; An, I.S.; An, S. Intercellular and intracellular functions of ceramides and their metabolites in skin (Review). Int. J. Mol. Med., 2016, 38, 16-22. [CrossRef]
- Melser, S.; Molino, D.; Batailler, B.; Peypelut, M.; Laloi, M.; Wattelet-Boyer, V.; Bellec, Y.; Faure, J.D.; Moreau, P. Links between lipid homeostasis, organelle morphodynamics and protein trafficking in eukaryotic and plant secretory pathways. Plant Cell Reports, 2011, 30, 177-193. [CrossRef]
- Gronnier, J.; Germain, V.; Gouguet, P.; Cacas, J.L.; Mongrand, S. GIPC: Glycosyl Inositol Phospho Ceramides, the major sphingolipids on earth. Plant Signal. Behav., 2016, 11. [CrossRef]
- Wattelet-Boyer, V.; Brocard, L.; Jonsson, K.; Esnay, N.; Joubès, J.; Domergue, F.; Mongrand, S.; Raikhel, N.; Bhalerao, R.P.; Moreau, P.; et al. Enrichment of hydroxylated C24- and C26-acyl-chain sphingolipids mediates PIN2 apical sorting at trans-Golgi network subdomains. Nat. Commun., 2016, 7. [CrossRef]
- Lv, B.; Wei, K.; Hu, K.; Tian, T.; Zhang, F.; Yu, Z.; Zhang, D.; Su, Y.; Sang, Y.; Zhang, X.; et al. MPK14-mediated auxin signaling controls lateral root development via ERF13-regulated very-long-chain fatty acid biosynthesis. Mol. Plant., 2021, 14, 285-297. [CrossRef]
- Akolová, B.; Hudská, K.; Pullmannová, P.; Kováčik, A.; Palát, K.; Roh, J.; Fleddermann, J.; Estrela-Lopis, I.; Vávrová, K. Different phase behavior and packing of ceramides with long (C16) and very long (C24) acyls in model membranes: infrared spectroscopy using deuterated lipids. J. Phys. Chem. B, 2014, 118, 10460-10470. [CrossRef]
- Ternes, P.; Franke, S.; Zähringer, U.; Sperling, P.; Heinz, E. Identification and characterization of a sphingolipid delta 4-desaturase family. J. Biol. Chem., 2002, 277, 25512-25518. [CrossRef]
- Flores-Pérez, Ú.; Jarvis, P. Isolation and Suborganellar Fractionation of Arabidopsis Chloroplasts. Methods Mol Biol, 2017, 1511, 45-60. [CrossRef]
- Markham, J.E.; Jaworski, J.G. Rapid measurement of sphingolipids from Arabidopsis thaliana by reversed-phase high-performance liquid chromatography coupled to electrospray ionization tandem mass spectrometry. Rapid Commun. Mass Spectrom., 2007, 21, 1304-1314. [CrossRef]
- Zeng, H.Y.; Liu, Y.; Chen, D.K.; Bao, H.N.; Huang, L.Q.; Yin, J.; Chen, Y.L.; Xiao, S.; Yao, N. The immune components ENHANCED DISEASE SUSCEPTIBILITY 1 and PHYTOALEXIN DEFICIENT 4 are required for cell death caused by overaccumulation of ceramides in Arabidopsis. Plant J., 2021, 107, 1447-1465. [CrossRef]
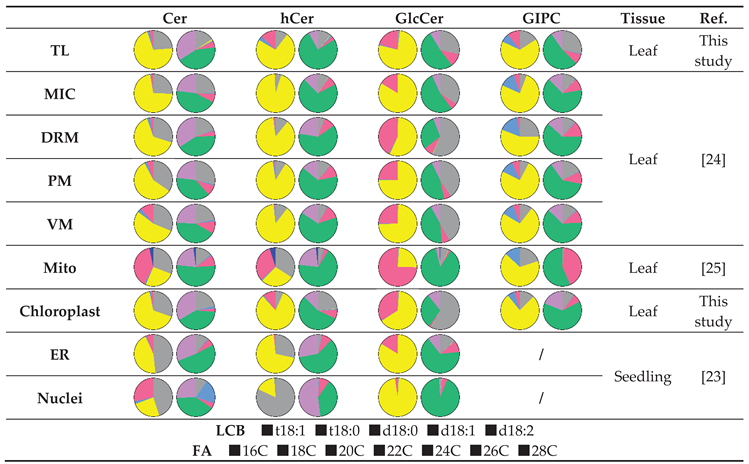 |
| Cer | hCer | GlcCer | GIPC | Tissue | Ref. | |
| TL | t18:1 c24:0 t18:1 c26:0 |
t18:1 h24:0 t18:1 h24:1 |
t18:1 h24:1 | t18:1 h24:0 t18:1 h24:1 |
Leaf | This study |
| MIC | t18:1 c24:0 t18:1 c26:0 t18:1 c16:0 t18:1 c24:1 |
t18:1 h24:1 t18:1 h24:0 |
t18:1 h24:1 | t18:1 h24:0 t18:1 h24:1 |
Leaf | [24] |
| DRM | t18:1 c24:0 t18:1 c26:0 |
t18:1 h24:0 t18:1 h26:0 t18:1 h24:1 |
d18:1 h16:0 |
t18:1 h24:0 t18:0 h24:0 |
||
| PM | t18:1 c24:0 t18:1 c26:0 t18:0 c16:0 |
t18:1 h24:1 t18:1 h24:0 |
t18:1 h24:1 d18:1 h16:0 t18:1 h16:0 |
t18:1 h24:1 t18:1 h24:0 |
||
| VM | t18:1 c24:0 t18:1 c26:0 t18:0 c24:0 |
t18:1 h24:1 t18:1 h24:0 |
t18:1 h24:1 d18:1 h16:0 t18:1 h16:0 |
t18:1 h24:1 t18:1 h24:0 |
||
| Mito | d18:1 c24:0 t18:1 c26:0 |
t18:0 h24:0 d18:1 h24:0 |
d18:1 h24:1 | t18:1 h24:1 t18:1 h24:0 |
Leaf | [25] |
| Chloroplast | t18:1 c24:0 t18:1 c26:0 |
t18:1 h24:1 t18:1 h24:0 t18:1 h16:0 |
d18:1 h16:0 t18:1 h16:0 |
t18:1 h24:0 t18:1 h24:1 |
Leaf | This study |
| ER | t18:0 c24:0 t18:1 c24:0 t18:1 c26:0 |
t18:0 h24:0 t18:1 h24:0 t18:0 h26:1 |
t18:1 h24:0 t18:1 h24:1 |
/ | Seedling | [23] |
| Nuclei | t18:0 c24:0 d18:1 c20:0 |
t18:0 h26:1 t18:0 h24:0 |
t18:1 h24:1 | / |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




