3.1. Structural stability and electron-phonon properties of ZrO2
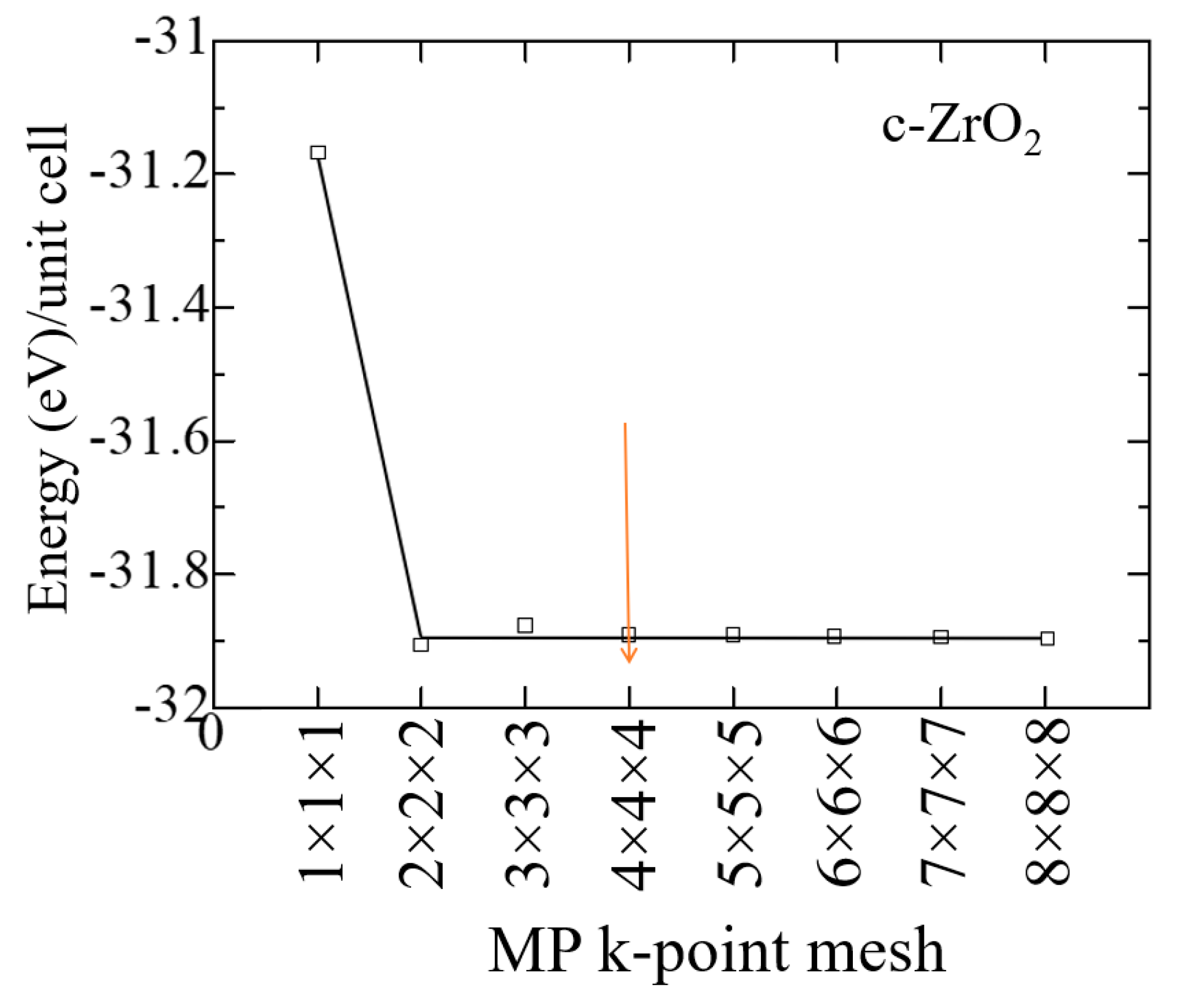
At the first stage of modeling, the structural-energy relaxation of pure ZrO2 phases was carried out using the VASP package. To find the optimal cutoff energy for the ENCUT plane wave basis functions and the corresponding number of k-points in the Brillouin zone, we tested the convergence of the total unit cell energy as a function of ENCUT and KPOINTS.
The results of the k-point convergence test for ZrO
2 cubic phases are shown in
Figure 3, 2 and performed to build a grid from k-point data with an initial value of ENCUT = 1.3*ENMAX. Based on the results obtained, it can be concluded that for a 4x4x4 k-point grid with the Monkhrost-Pack scheme, it is optimal for the geometric relaxation of ZrO2. However, when calculating the electronic structure of these compounds, the number of k-points was at least doubled in order to obtain a better density of states (DOS).
Figure 2.
The total energy of the c-ZrO2 unit cell as a function of the number of k-points under the condition ENCUT=1.3*ENMAX.
Figure 2.
The total energy of the c-ZrO2 unit cell as a function of the number of k-points under the condition ENCUT=1.3*ENMAX.
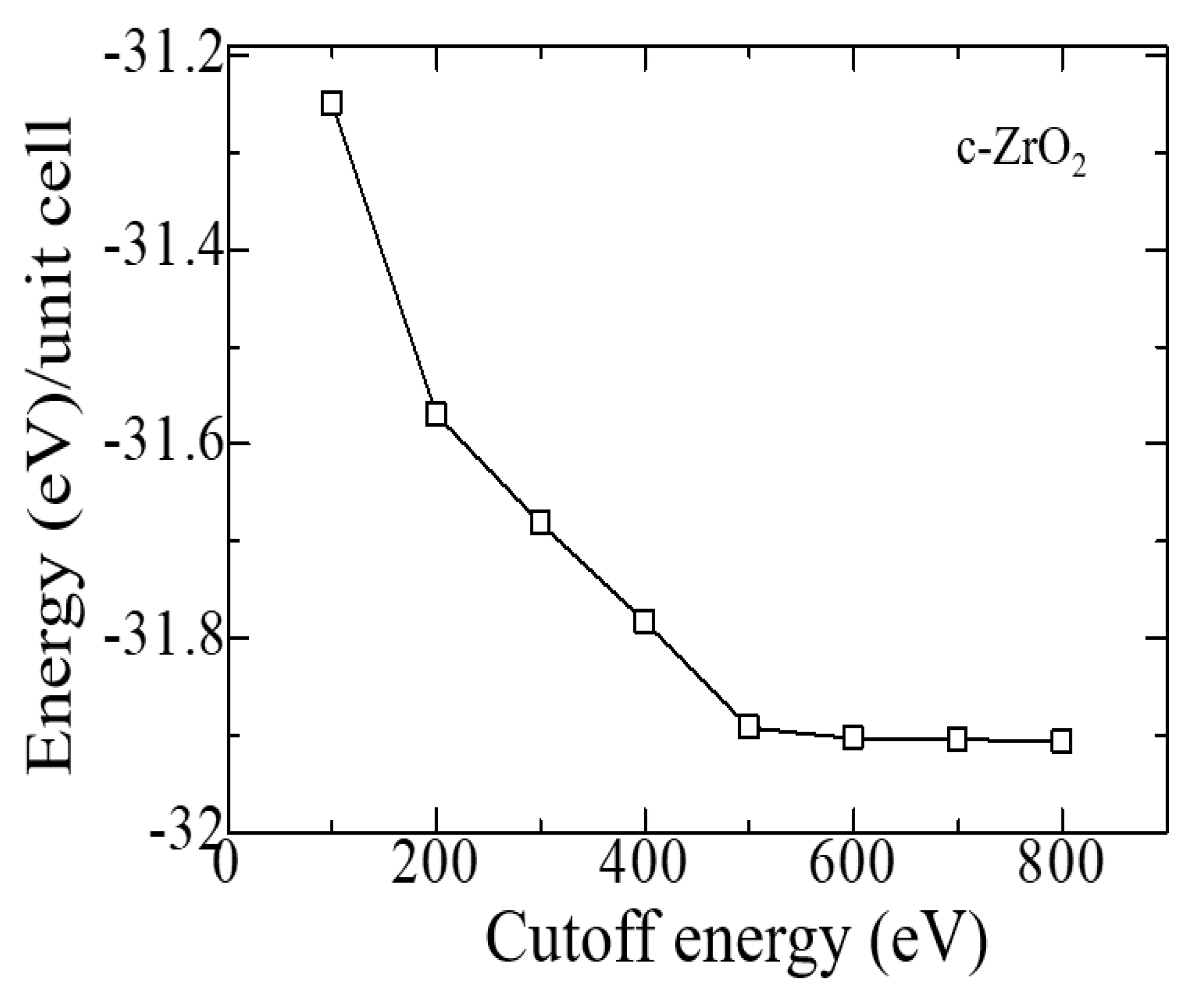
Similar tests were carried out to establish the cutoff energy, which shows that the choice of 600 eV is suitable for calculations, and a further increase in this energy increases the cost of the calculation without affecting its accuracy (
Figure 3). Therefore, all further calculations were carried out at ENCUT = 600 eV.
Similar convergence tests were also carried out for the tetragonal and monoclinic phases of ZrO
2 using the GGA potential. In
Table 1 compares the calculated values of the crystal lattice constants of the ZrO
2 phase, obtained from two exchange-correlation potentials, with the literature results.
According to the results given in
Table 1, it can be seen that during the transition from the high-temperature phase to the low-temperature phase, the lattice distortion leads to a displacement of O ions in the c direction by the value of dz, expressed in relative units. As a result of distortion in the tetragonal phase, all Zr-O bonds will become nonequivalent. According to
Table 3, the SCAN functionality describes the geometry much better than the standard GGA-PBE. However, it is also seen from the available data that GGA and SCAN almost identically describe the energy difference between the monoclinic and tetragonal phases of ZrO
2. Since the SCAN exchange-correlation functional describes the structural properties well, we decided to use this functional in the future when describing the geometry of other systems.
Table 2 compares the total energies calculated by the GGA method for systems of the monoclinic, tetragonal, and cubic phases of ZrO
2. It can be seen that among all systems, the most stable phase with the lowest energy is m-ZrO
2, that is, in fact, in terms of the field energy at low temperatures, the stable phase is monoclinic with the space group P21/c.
Table 2.
GGA- calculated total electronic energies of c-ZrO2, t-ZrO2, m-ZrO2.
Table 2.
GGA- calculated total electronic energies of c-ZrO2, t-ZrO2, m-ZrO2.
| System |
Energy |
ΔE |
| m-ZrO2
|
-115.179 |
0 |
| t-ZrO2
|
-114.754 |
0.425 |
| c-ZrO2
|
-114.346 |
0.833 |
Further, using the Phonopy code in the VASP package, the thermodynamic properties and phonon spectra of the ZrO
2 phase were calculated for a more detailed discussion and substantiation of the structural stability of the ZrO
2 monoclinic phase.
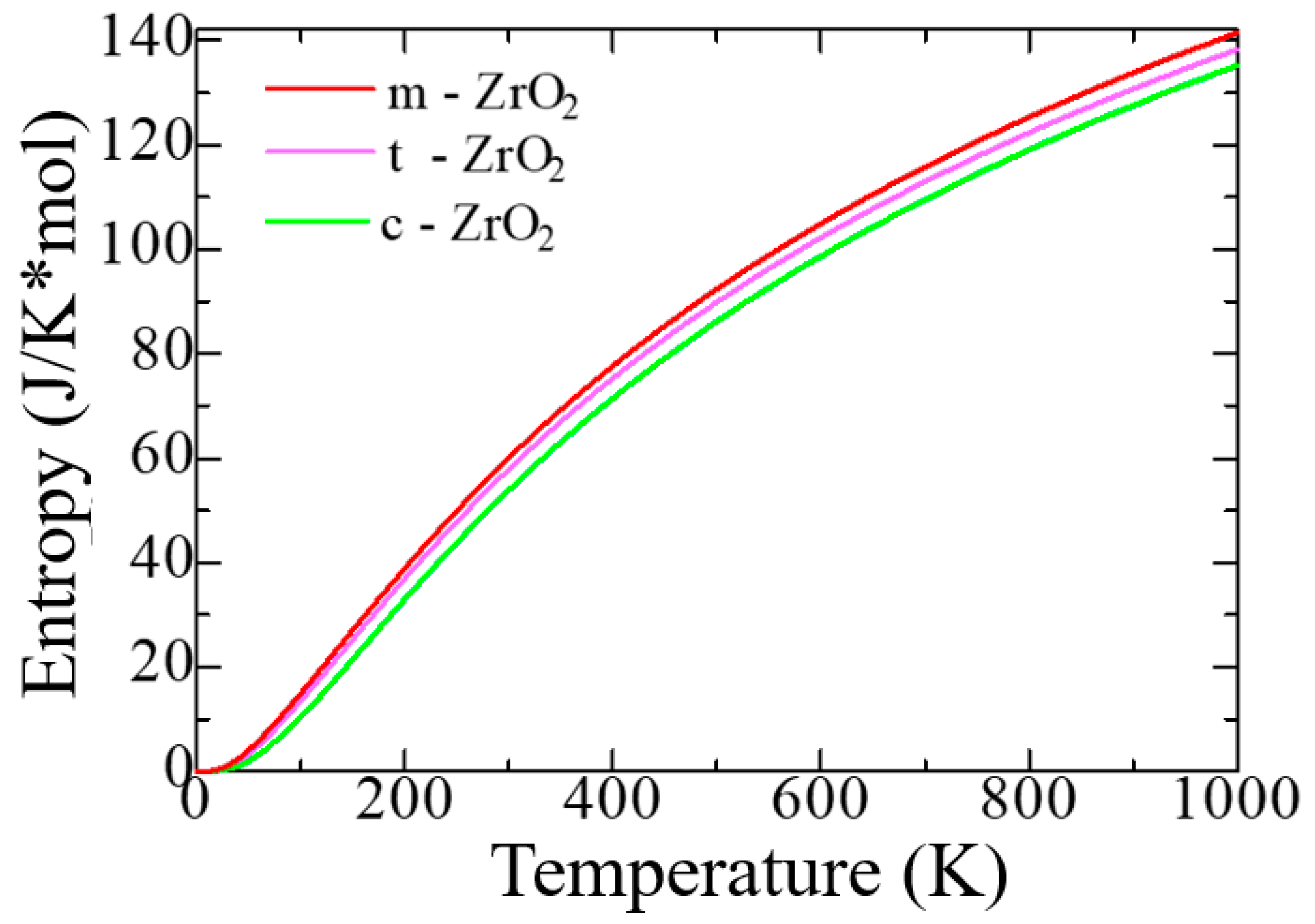
Figure 4 shows the change in the entropy of the unit cells of the ZrO
2 phase as a function of temperature.
According to
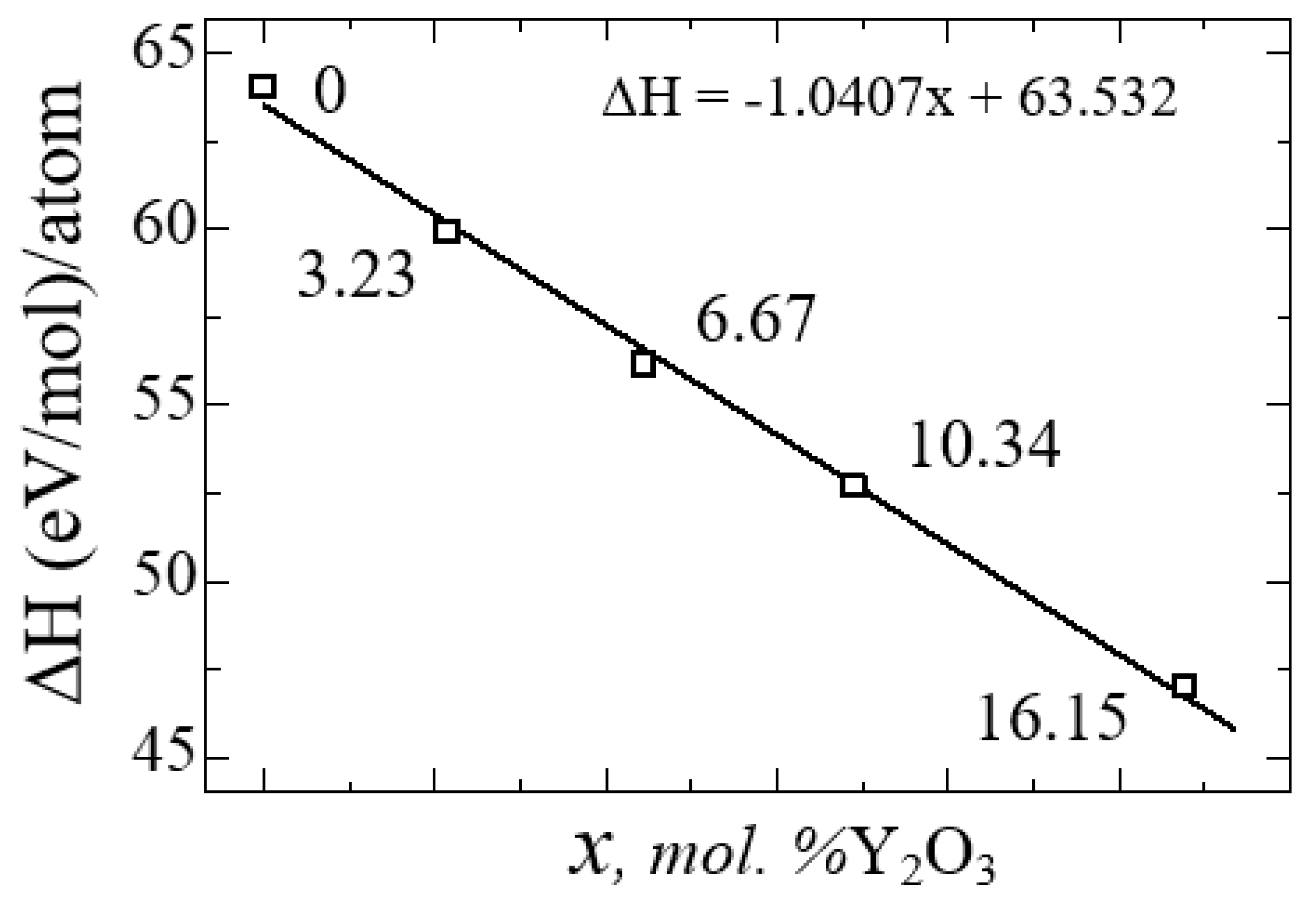
Figure 4, as the transition from monoclinic to tetragonal and cubic phases, the entropy of these compounds decreases, which corresponds to the criterion of inverse dependence of enthalpy or direct dependence of entropy and stability of solid systems [
53]. Thus, the monoclinic phase is the most stable with the highest entropy among other ZrO
2 phases. This pattern can be clearly observed after analyzing the pattern of phonon frequencies of the three phases of ZrO
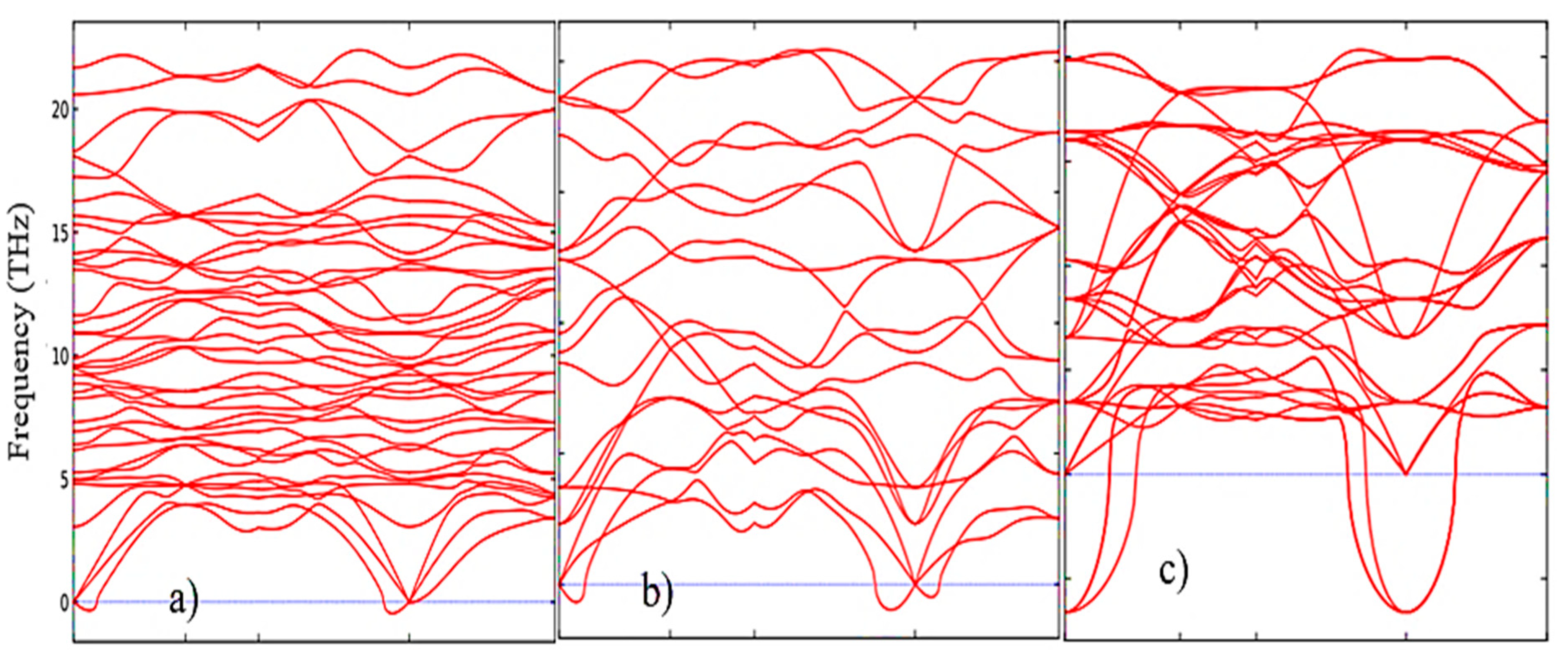
2 (
Figure 5a–c), from which it is clearly seen that the monoclinic phase has the smallest negative modes than the other two phases.
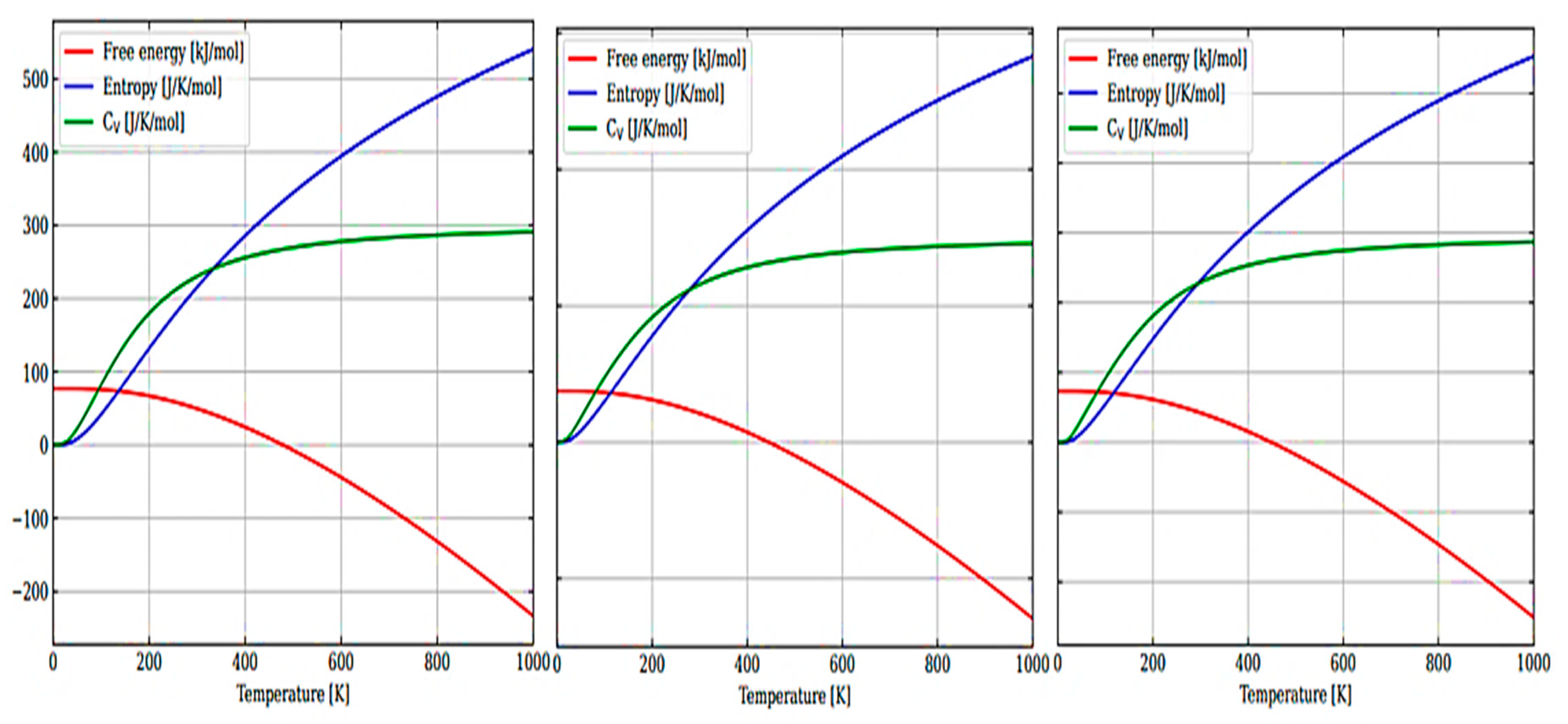
Figure 6a–c shows the temperature dependence of the free energy, entropy, and heat capacity of a 12-atom supercell for m-ZrO
2, t-ZrO
2, and m-ZrO
2.
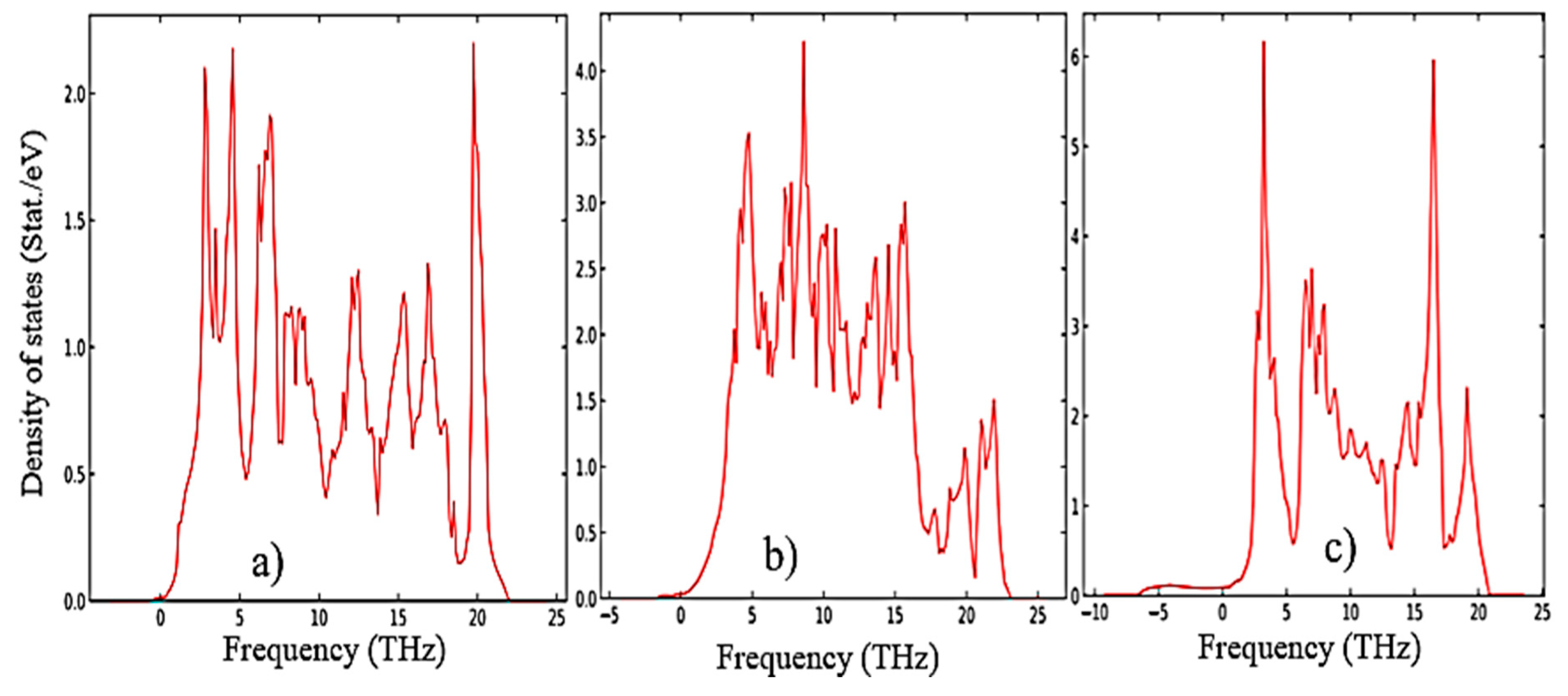
The results of calculations of the density of phonon states presented in
Figure 7a–c indicate that as the transition from monoclinic to tetragonal and cubic phases, the density of electronic states increases, and they also agree well with the results shown in
Figure 5 and confirm that the monoclinic phase is the most stable among the other phases of ZrO
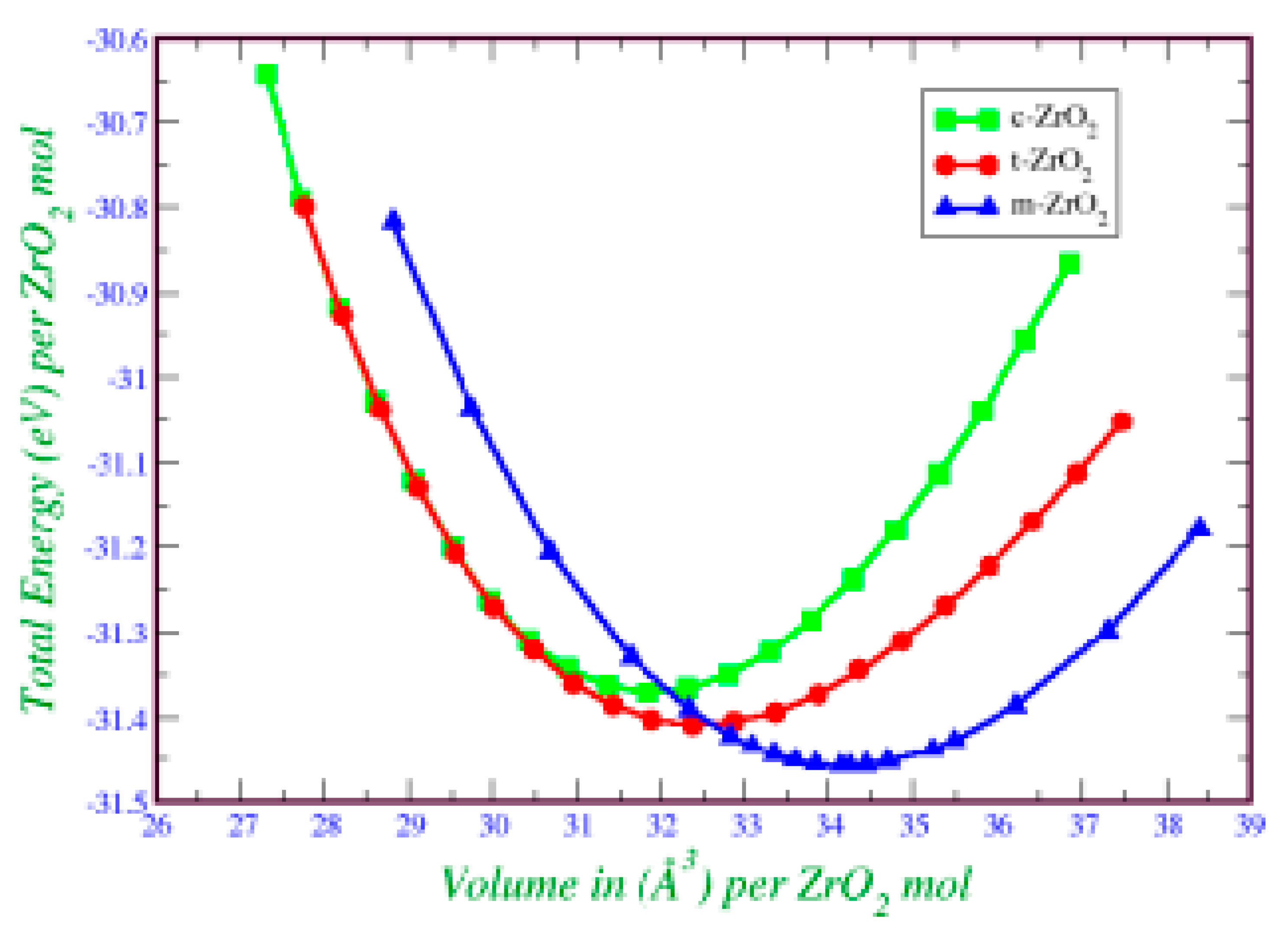
2. This is also confirmed by the result of the Energy/Volume diagram presented in
Figure 8. Therefore, for further stabilization by doping with Y
2O
3, it is reasonable to choose a monoclinic phase.
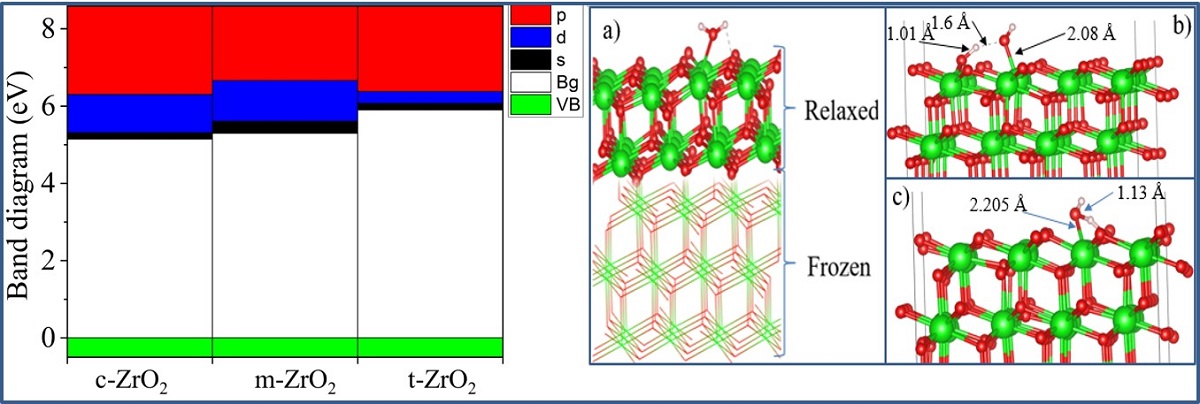
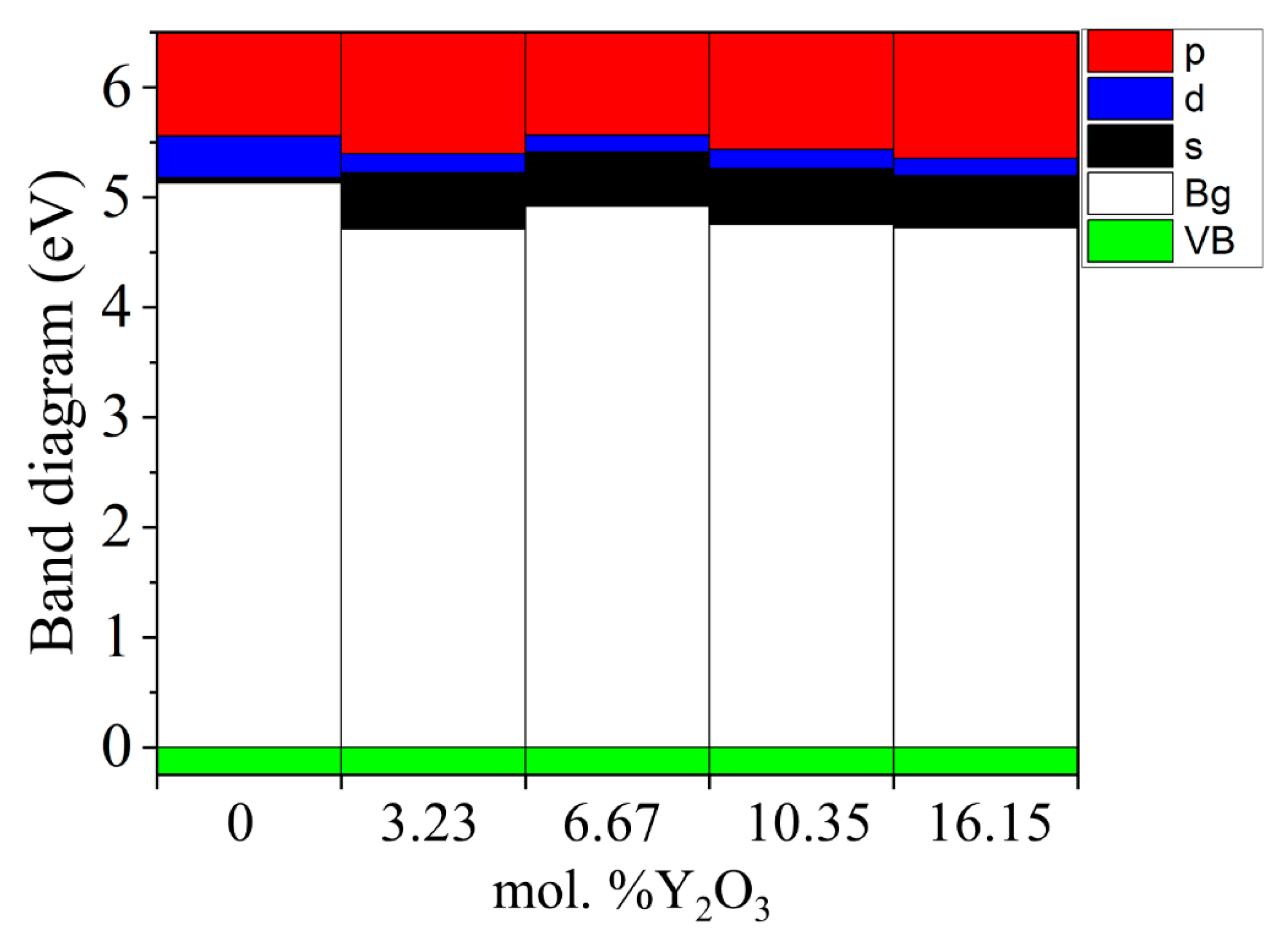
Next, using the well-optimized structures of the three phases of ZrO
2, we performed calculations to study their electronic properties. Using the GGA and SCAN functionals and the HSE06 hybrid functional, we found the band gaps of these systems (
Table 3), analyzed their orbital structure, and modeled the change in the position of the Fermi level in these systems.
Table 3.
Calculated and experimental band gap of c-ZrO2, t-ZrO2, m-ZrO2 in eV.
Table 3.
Calculated and experimental band gap of c-ZrO2, t-ZrO2, m-ZrO2 in eV.
| System |
This work |
Experiment [55] |
| GGA |
SCAN |
HSE06 |
VUV |
| m-ZrO2
|
3.9 |
3.8 |
5.288 |
5.78 |
| t-ZrO2
|
4.42 |
4.37 |
5.898 |
5.83 |
|
c-ZrO2
|
4.03 |
3.93 |
5.140 |
6.10 |
According to the results presented in
Table 3, the GGA and SCAN functionals showed a rather small band gap compared to the HSE06 hybrid functional [
56], which makes it possible to overcome the underestimation of the band gap. On the other hand, it is obvious that the standard SCAN and GGA functionals greatly underestimate the band gap. Given the suitability of HSE06 for estimating the band gap energy, we further used this hybrid functional to describe all the problems associated with the electronic properties of the systems under study.
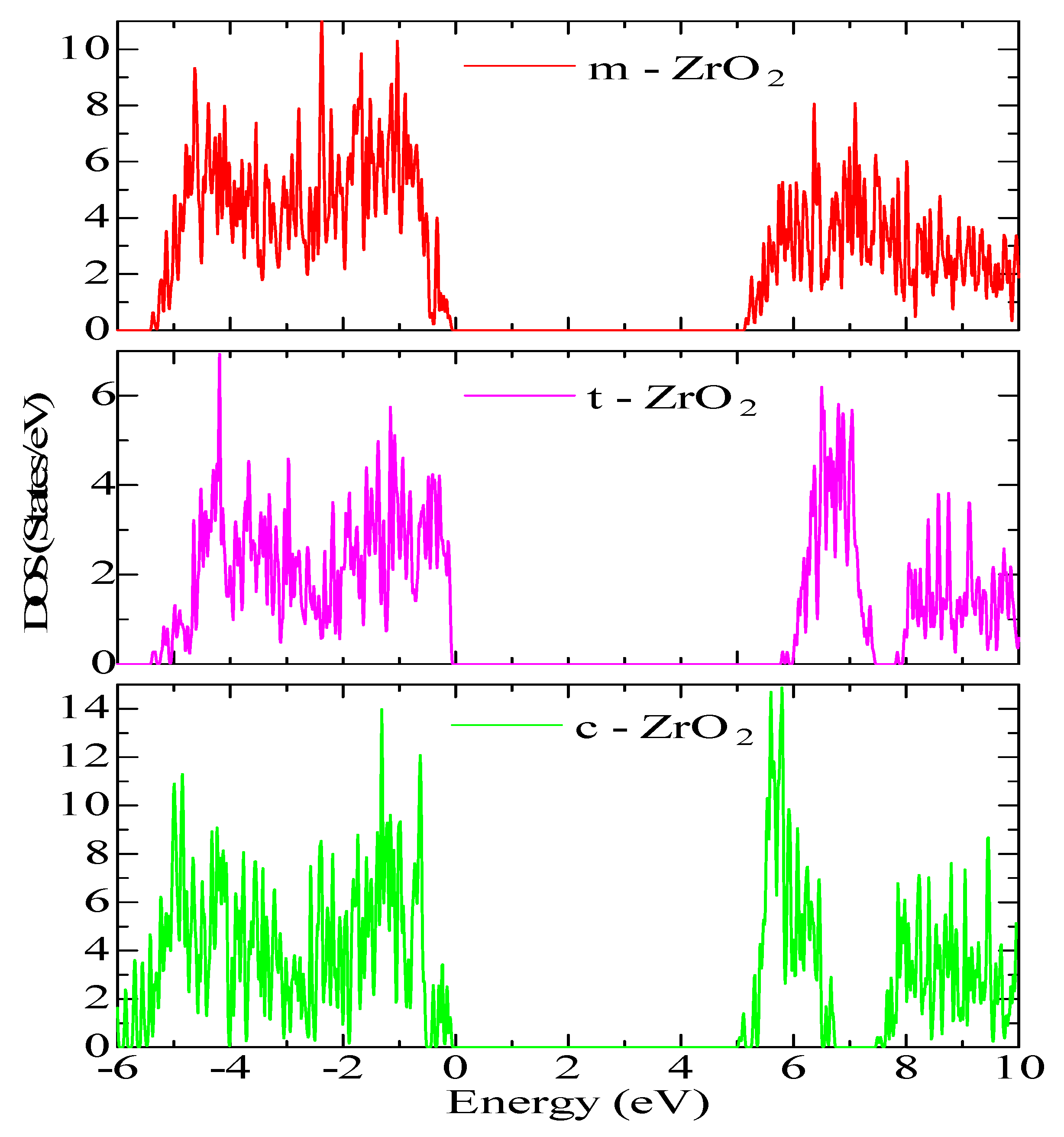
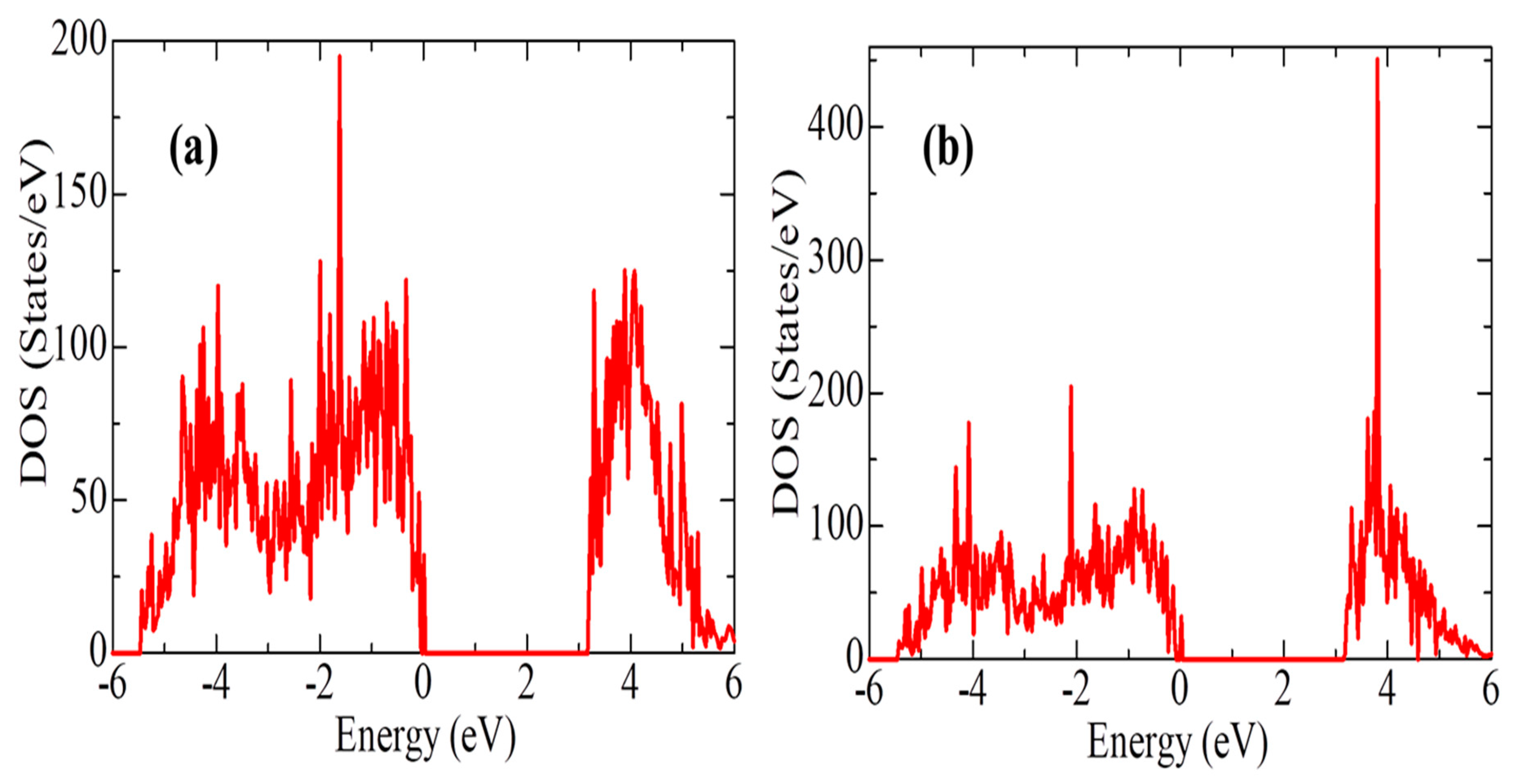
Next, for ZrO
2 structures relaxed using the SCAN functional, calculations were made of the density of available electronic states at the Fermi level (
Figure 9), which is crucial for interpreting the electronic properties of ZrO
2 and the transport characteristics of electronic devices based on this.
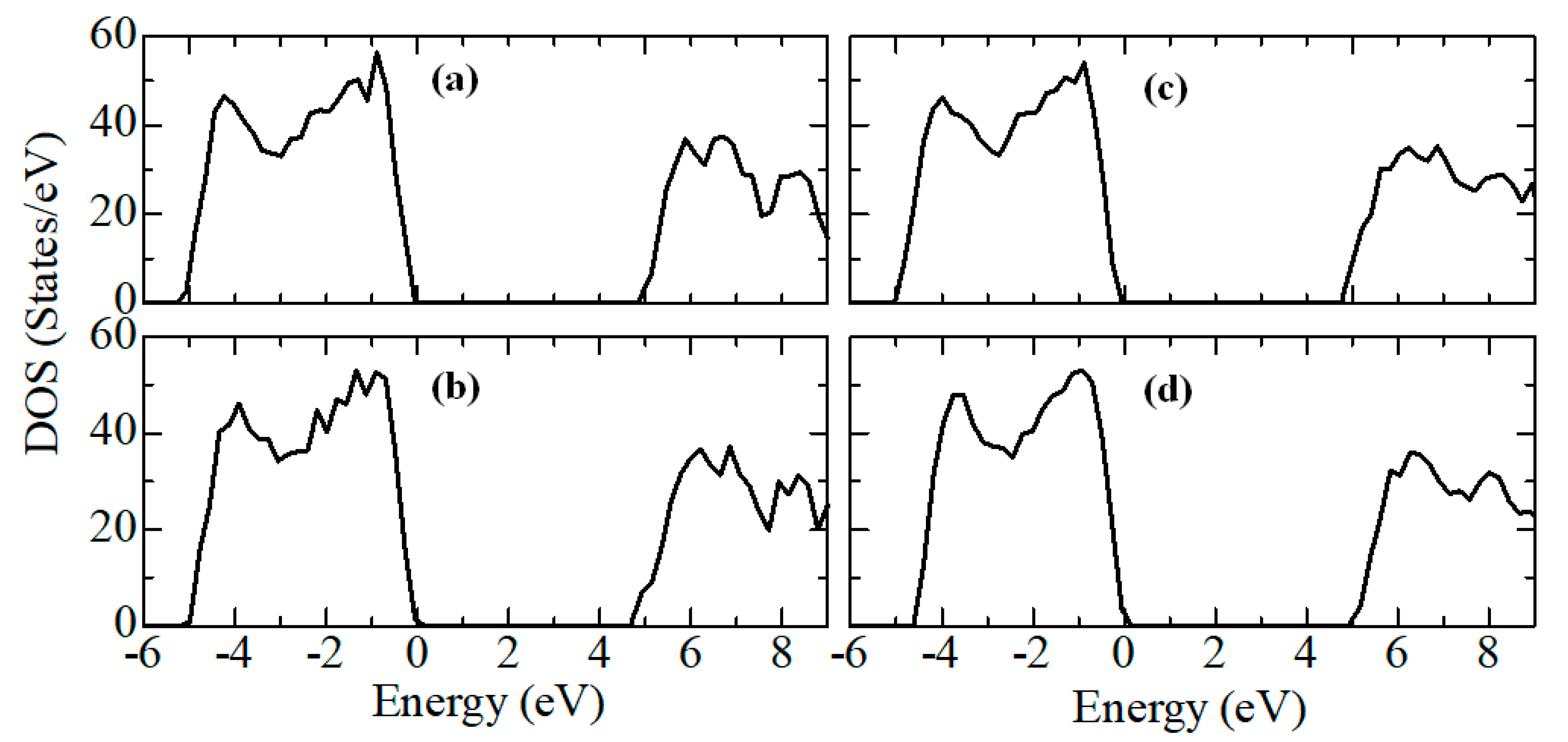
According to
Figure 9, the density of electronic states for c-ZrO
2 is somewhat overestimated compared to other phases. In addition, secondary energy gaps are observed in the energy diagram of the tetragonal and cubic phases. Also, this gap increases during the transition from the tetragonal to the cubic phase.
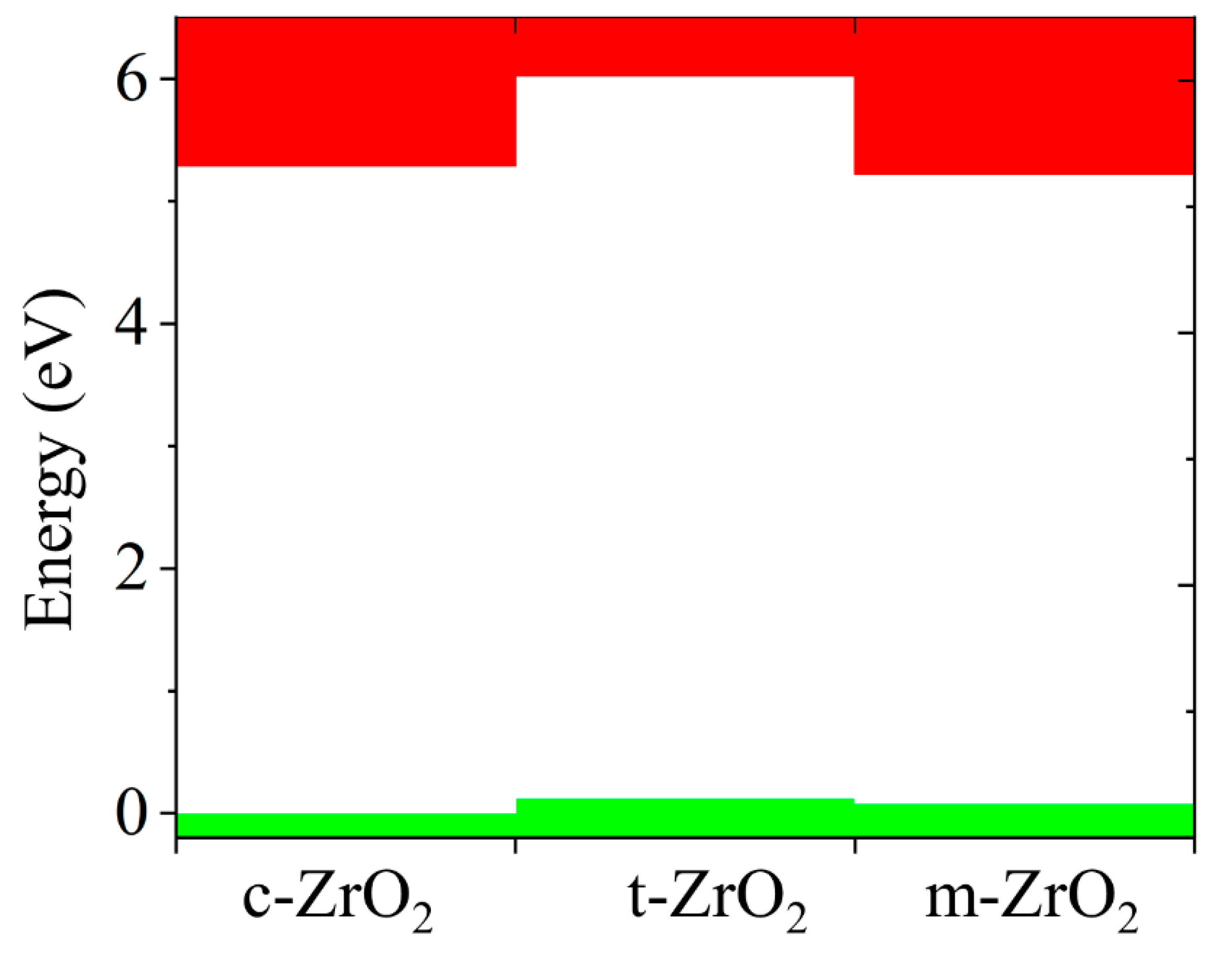
Next, the position of the Fermi level in ZrO
2 crystals and the shift of this level during their phase transformation were determined. As can be seen from
Figure 10, if we take the position of the Fermi level (maximum of the valence band) for the monoclinic phase as a reference point, then during the m-t phase transformation of ZrO
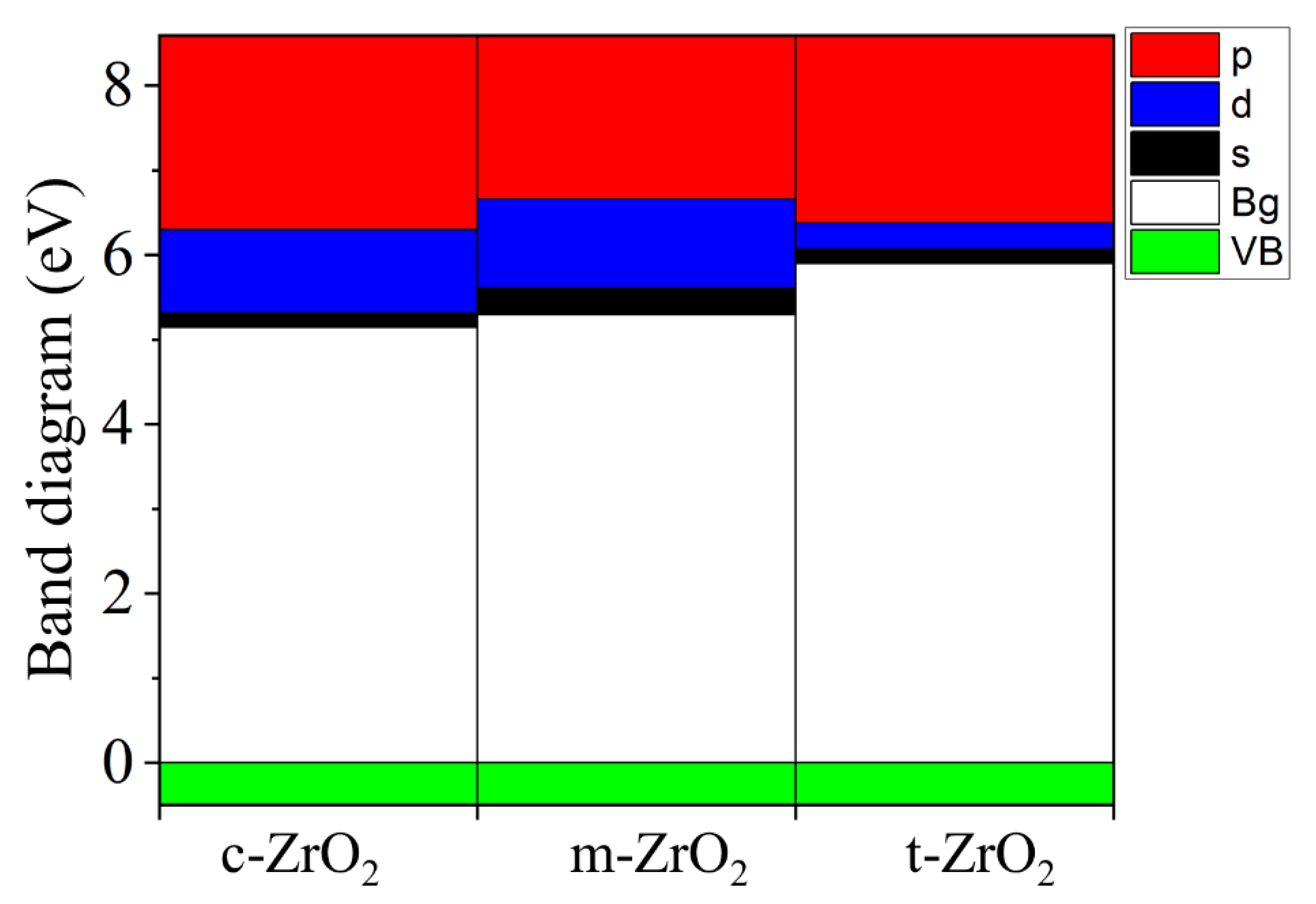
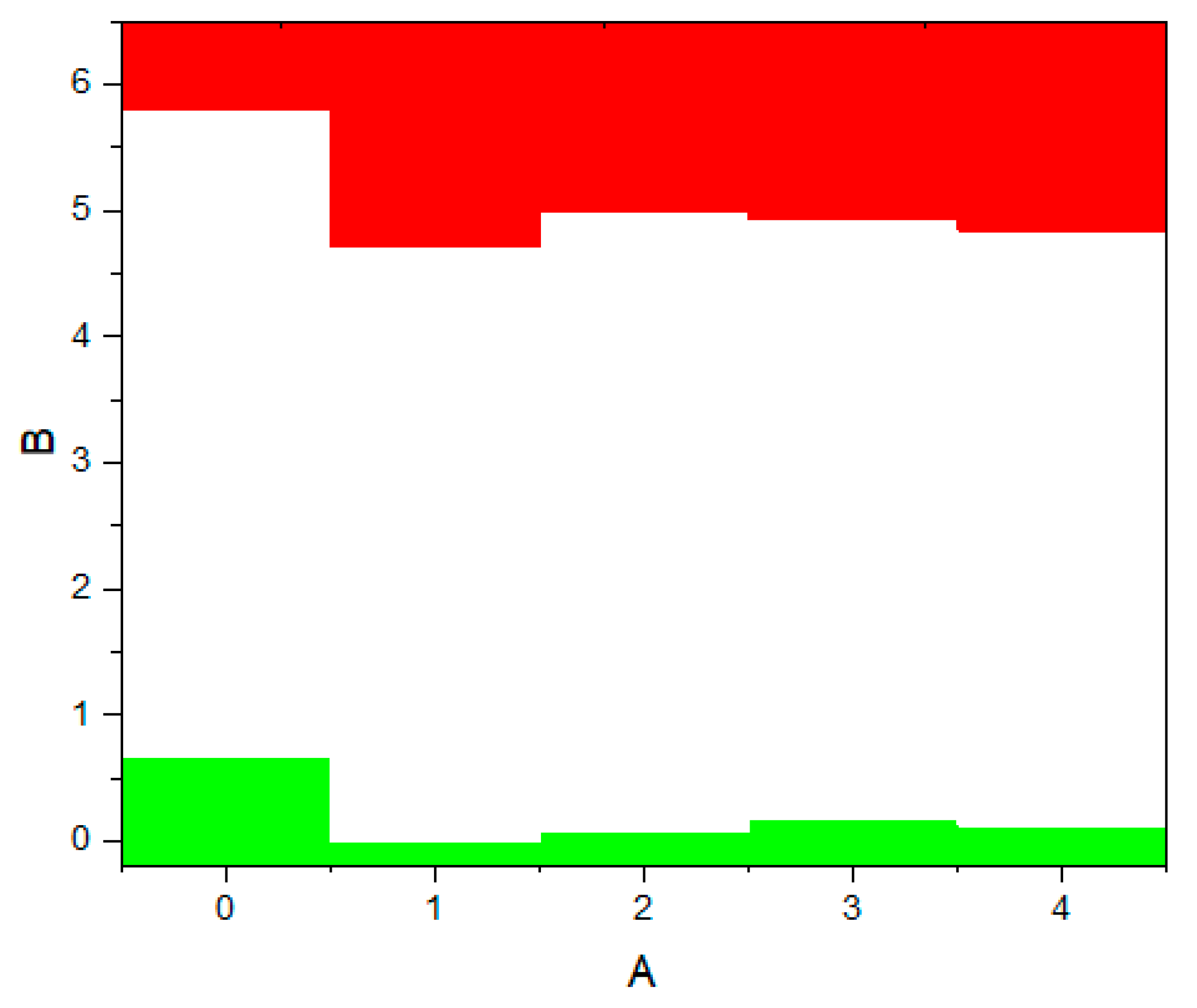
2, this level first shifts by 0.125 eV towards higher energies (towards the valence band), and then , in the t-c section, decrease by 0.08 eV. This is also observed in detail from the band stacking results for the orbital analysis, which are shown in
Figure 11 for the three phases of ZrO
2.
It can be seen that as the transition from the monoclinic to the tetragonal and cubic phases, the contribution of the p orbitals becomes more significant in CB, and the s orbitals make a small contribution, while the d state shows a different trend. It is assumed that this behavior may be associated with a change in the crystal field and covalence of ZrO2 during the phase transformation.