Submitted:
12 March 2026
Posted:
13 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Regents
Plant Material
Experimental Design
Analytical Analysis
The Quantitative Determination of Lactic acid Bacteria
Total Soluble Solids Content Evaluation
Ascorbic Acid Evaluation
Total Sugar Content Evaluation
Total Phenolic Content Evaluation
Antioxidant Activity Evaluation
Statistical Analysis
3. Results
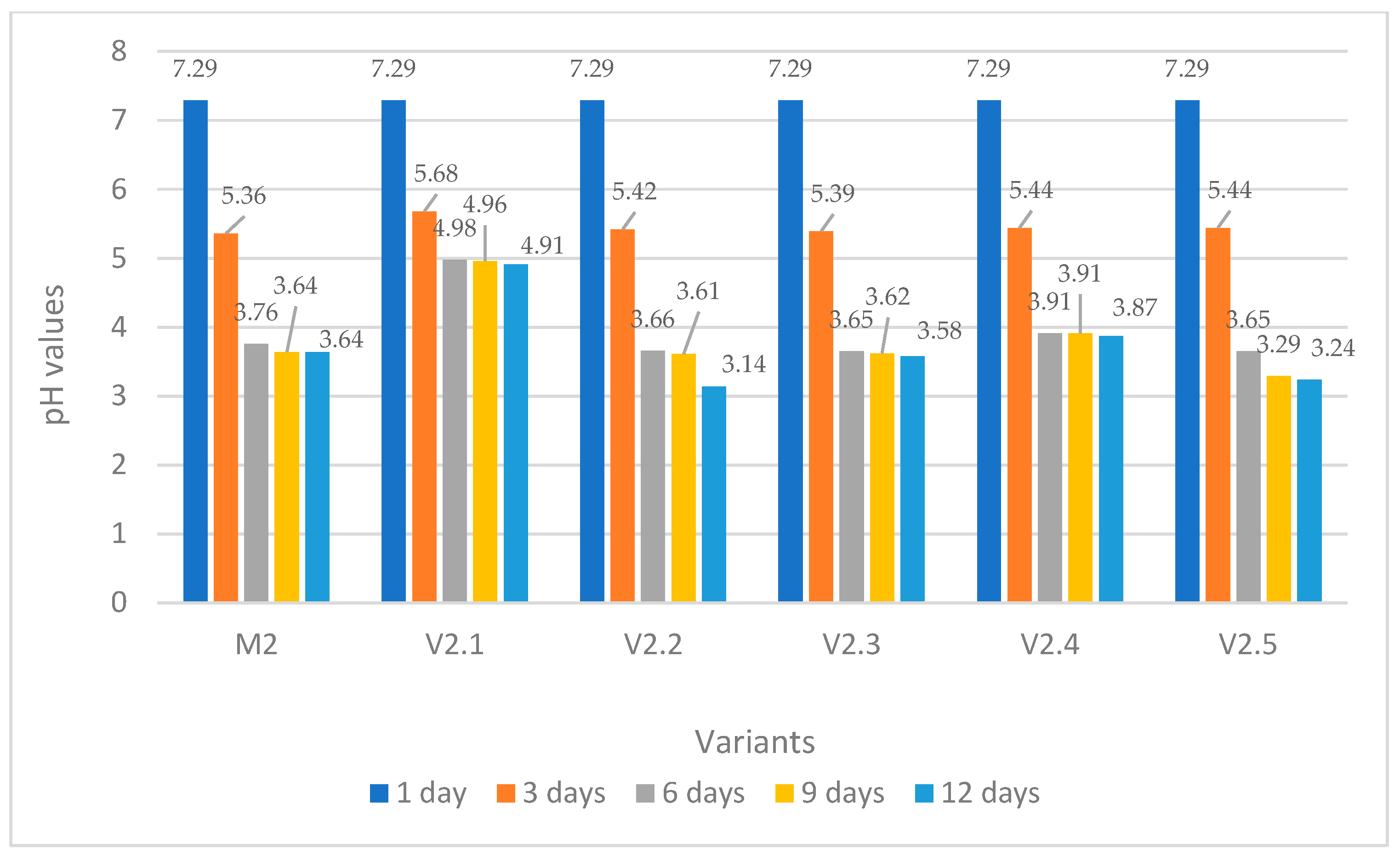
pH Evolution
Evolution of the Number of Lactic Acid Bacteria
Total Sugar Content
Ascorbic Acid Content
Total Phenolic Content
Antioxidant Activity
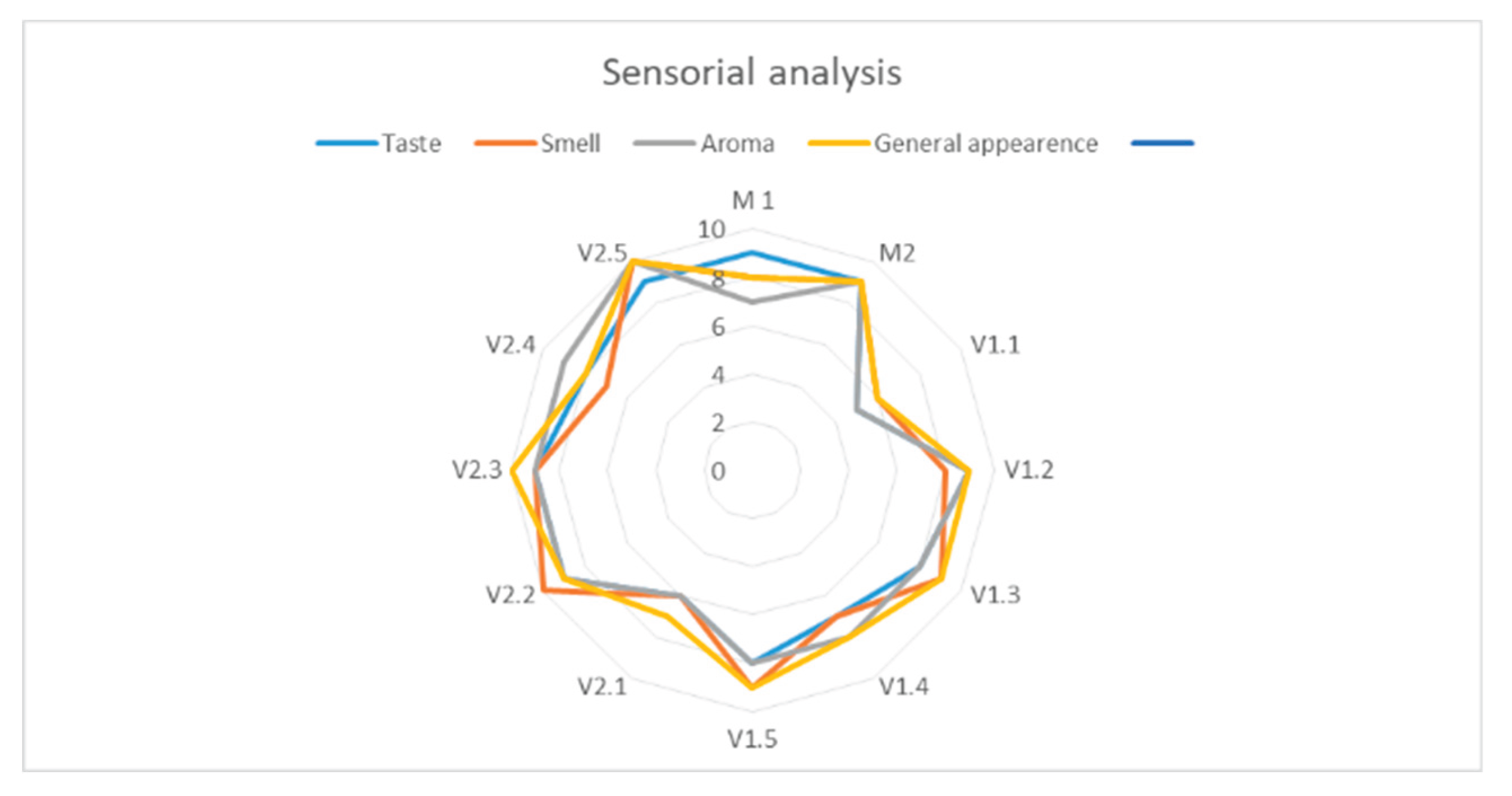
Sensory Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kapusta-Duch, J., Kusznierewicz, B., Leszczyńska, T., & Borczak, B. 2017. Effect of package type on selected parameters of nutritional quality of chill-stored white sauerkraut. Polish Journal of Food and Nutrition Sciences, 67(2), 137-144.
- Siddeeg, A., Afzaal, M., Saeed, F., Ali, R., Shah, Y. A., Shehzadi, U., ... & Al-Farga, A. 2022. Recent updates and perspectives of fermented healthy super food sauerkraut: a review. International Journal of Food Properties, 25(1), 2320-2331.
- Hallmann, E., Kazimierczak, R., Marszałek, K., Drela, N., Kiernozek, E., Toomik, P., ... & Rembiałkowska, E. 2017. The nutritive value of organic and conventional white cabbage (Brassica oleracea L. var. capitata) and anti-apoptotic activity in gastric adenocarcinoma cells of sauerkraut juice produced therof. Journal of agricultural and food chemistry, 65(37), 8171-8183.
- Premakumar, K., Sahana, S., & Sabrana, M. A. S. F. 2021. Effects of salt concentration on storage ability of sauerkraut. International Journal of Research, 7(2), 11-16.
- Mennes, E.M.1994. Make your own sauerkraut. Cooperative Extension Publication. UW-Extension. B2087, pp. 1-7.
- Trail, A. C., Fleming, H. P., Young, C. T., & McFeeters, R. F. 1996. Chemical and sensory characterization of commercial sauerkraut 1. Journal of food quality, 19(1), 15-30.
- Bonnesen, C., Eggleston, I. M., & Hayes, J. D. 2001. Dietary indoles and isothiocyanates that are generated from cruciferous vegetables can both stimulate apoptosis and confer protection against DNA damage in human colon cell lines. Cancer research, 61(16), 6120-6130.
- Kristek, S., Bešlo, D., Pavlović, H., & Kristek, A. 2004. Effect of starter cultures L. mesenteroides and L. lactis ssp. Lactis on sauerkraut fermentation and quality. Czech journal of food sciences, 22(4), 125.
- Delanoe, R., & Emard, L. O. 1971. Experimental manufacture of sauerkraut in Quebec. Quebec Laitier et Alimentaire, 30, 11-14.
- Gangopadhyay, H., & Mukherjee, S. 1971. Effect of different salt concentrations on the microflora and physicochemical changes in sauerkraut fermentation. Journal of Food Science and Technology, 8(3), 127-131.
- Holzapfel, W., Schillinger, U., & Buckenhüskes, H. J. 2003. 14 Sauerkraut. Handbook of fermented functional foods, 343.
- SS, Yuwono, Rahayu AP, and Blanc PJ. 2020. Effect of Lactobacillus plantarum and Leuconostoc mesenteroides starter cultures in lower salt concentration fermentation on the sauerkraut quality. Food Research, 4(4), 1038-1044.
- In Lactic acid bacteria: biodiversity and taxonomy; Holzapfel, W. H., & Wood, B. J. (Eds.). 2014. Lactic acid bacteria: biodiversity and taxonomy. John Wiley & Sons.
- Zabat, M. A., Sano, W. H., Wurster, J. I., Cabral, D. J., & Belenky, P. 2018. Microbial community analysis of sauerkraut fermentation reveals a stable and rapidly established community. Foods, 7(5), 77.
- Orgeron II, R. P., Corbin, A., & Scott, B. 2016. Sauerkraut: A probiotic superfood. Functional Foods in Health and Disease-Online ISSN: 2160-3855; Print ISSN: 2378-7007, 6(8), 536-543.
- Oshiro, M., Tanaka, M., Zendo, T., & Nakayama, J. 2020. Impact of pH on succession of sourdough lactic acid bacteria communities and their fermentation properties. Bioscience of microbiota, food and health, 39(3), 152-159.
- Xu, D., Ding, W., Ke, W., Li, F., Zhang, P., & Guo, X. 2019. Modulation of metabolome and bacterial community in whole crop corn silage by inoculating homofermentative Lactobacillus plantarum and heterofermentative Lactobacillus buchneri. Frontiers in Microbiology, 9, 3299.
- Weinberg, Z. G., & Muck, R. E. 1996. New trends and opportunities in the development and use of inoculants for silage. FEMS Microbiology Reviews, 19(1), 53-68.
- Dunière, L., Sindou, J., Chaucheyras-Durand, F., Chevallier, I., & Thévenot-Sergentet, D. 2013. Silage processing and strategies to prevent persistence of undesirable microorganisms. Animal Feed Science and Technology, 182(1-4), 1-15.
- Ávila, C. L. S., & Carvalho, B. F. 2020. Silage fermentation—updates focusing on the performance of microorganisms. Journal of applied microbiology, 128(4), 966-984.
- Tlais, A. Z. A., Lemos Junior, W. J. F., Filannino, P., Campanaro, S., Gobbetti, M., & Di Cagno, R. 2022. How microbiome composition correlates with biochemical changes during sauerkraut fermentation: A focus on neglected bacterial players and functionalities. Microbiology spectrum, 10(4), e00168-22.
- Fleming, H.P., Kyung, K.H. and Breidt, F. 1995.Vegetable fermentation in Biotechnology, 2nd ed,Vol.9 (eds. H. J. Rehm. and G. Reed), VCH, NewYork, pp. 629-631.
- Wood, B. J. 2012. Microbiology of fermented foods. Springer Science & Business Media.
- Nour V., Ionica, M. E., Trandafir I. 2015. Bioactive compounds, antioxidant activity and color of hydroponic tomato fruits at different stages of ripening. Notulae Botanicae Horti Agrobotanici Cluj-Napoca. 43, 2, 404–412.
- Chow, P. S., & Landhäusser, S. M. 2004. A method for routine measurements of total sugar and starch content in woody plant tissues. Tree physiology, 24(10), 1129-1136.
- Singleton VL, Rossi JA. 1965. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. American Journal of Enology and Viticulture 16:144-158.
- Sarkar, A., Rahman, S., Roy, M., Alam, M., Hossain, M. A., & Ahmed, T. 2021. Impact of blanching pretreatment on physicochemical properties, and drying characteristics of cabbage (Brassica oleracea). Food Research, 5(2), 393-400.
- Thakur, P. K., Panja, P., Kabir, J., & Dhua, R. S. 2020. Studies on shelf life of sauerkraut. Journal of Crop and Weed, 16(2), 204-209.
- Zubaidah, E., Arum, M. S., Widyaningsih, T. D., & Rahayu, A. P. 2020. Sauerkraut with the addition of Lactobacillus casei: Effects of salt and sugar concentrations on fermentation and antioxidant activity. Current Nutrition & Food Science, 16(8), 1265-1269.
- Chun, O. K., Smith, N., Sakagawa, A., & Lee, C. Y. 2004. Antioxidant properties of raw and processed cabbages. International journal of food sciences and nutrition, 55(3), 191-199.
- Özer, C., & Yıldırım, H. K. 2019. Some special properties of fermented products with cabbage origin: pickled cabbage, sauerkraut and kimchi. Turkish Journal of Agriculture-Food Science and Technology, 7(3), 490-497.
- Zhou, B., Feng, X., Huang, W., Liu, Q., Ibrahim, S. A., & Liu, Y. 2023. Effects of light intensity on the biosynthesis of glucosinolate in Chinese cabbage plantlets. Scientia Horticulturae, 316, 112036.
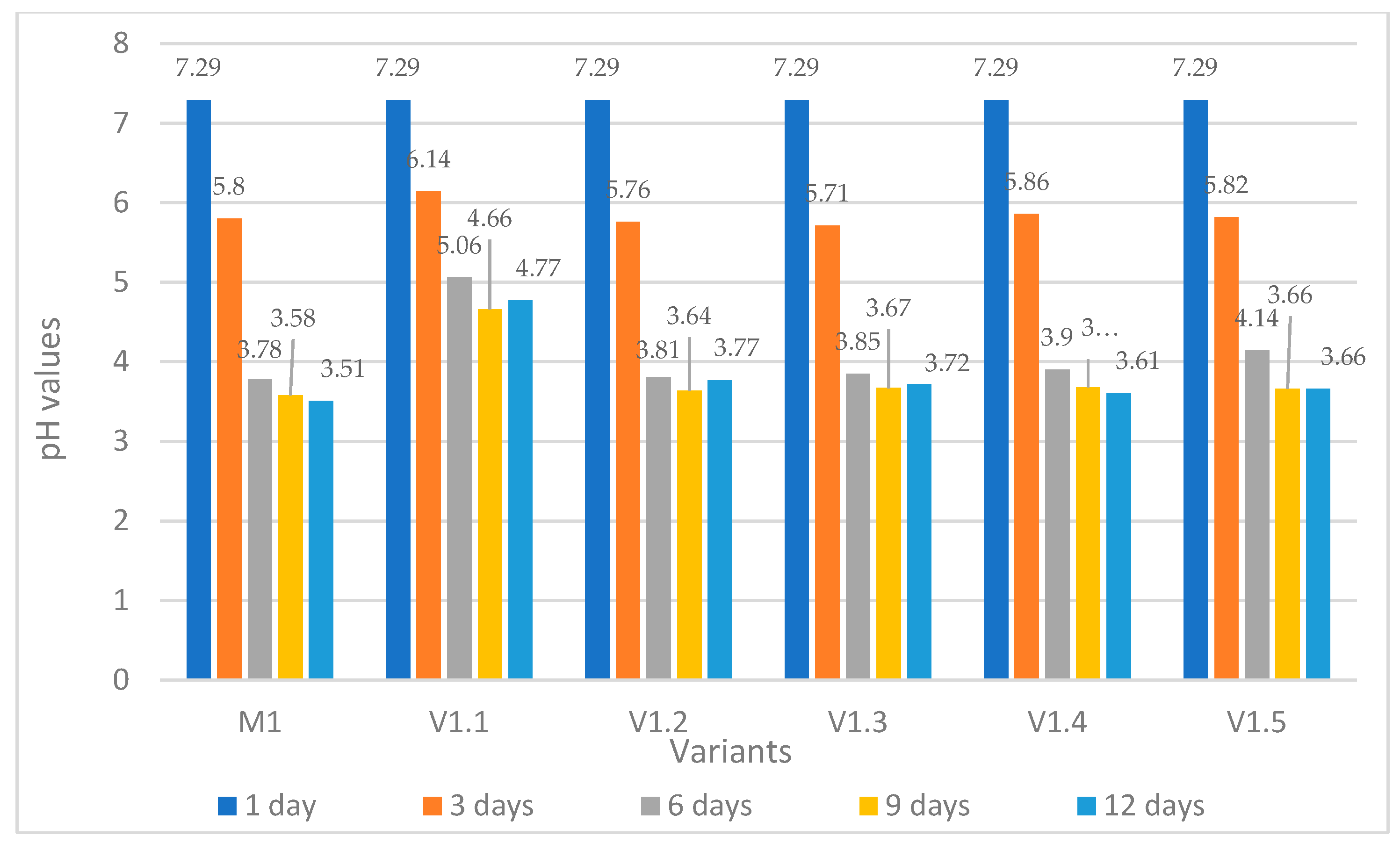
| Variant |
NTBL (UFC log) after 6 days from the start of fermentation |
NTBL (UFC log) 21 days after fermentation start |
| M1 | 3,60±0,16eA | 6,47±0,30eB |
| M2 | 3,04±0,13cA | 6,30±0,29deB |
| V1.1 | 1,99±0,08aA | 4,06±0,19aB |
| V1.2 | 2,54±0,11bA | 5,30±0,24bB |
| V1.3 | 3,00±0,12cA | 6,30±0,30deB |
| V1.4 | 3,36±0,15dA | 6,39±0,29eB |
| V1.5 | 3,61±0,17eA | 6,50±0,31eB |
| V2.1 | 1,98±0,07aA | 4,30±0,19aB |
| V2.2 | 3,36±0,14dA | 5,91±0,27cdB |
| V2.3 | 3,06±0,13cA | 5,60±0,25bcB |
| V2.4 | 3,30±0,14dA | 5,90±0,28cdB |
| V2.5 | 3,69±0,18eA | 5,87±0,27cdB |
| Variant | Fresh cabbage | Sauerkraut immediately after fermentation |
After 6 months of storage |
||||
| M 1 | 5,5±0,23aB | 6,5±0,31abcC | 4,5±0,19bcdA | ||||
| M2 | 5,5±0,23aB | 6,0±0,29aC | 4,0±0,17aA | ||||
| V1.1 | 5,5±0,23aB | 6,6±0,35bcC | 4,6±0,24cdA | ||||
| V1.2 | 5,5±0,23aB | 6,8±0,36cC | 4,8±0,22dA | ||||
| V1.3 | 5,5±0,23aB | 6,4±0,33abcC | 4,4±0,21bcA | ||||
| V1.4 | 5,5±0,23aB | 6,4±0,31abcC | 4,4±0,23bcA | ||||
| V1.5 | 5,5±0,23aB | 6,2±0,31abC | 4,2±0,22abA | ||||
| V2.1 | 5,5±0,23aB | 6,5±0,30abcC | 4,5±0,23bcdA | ||||
| V2.2 | 5,5±0,23aB | 6,2±0,32abC | 4,2±0,20abA | ||||
| V2.3 | 5,5±0,23aB | 6,5±0,33abcC | 4,5±0,23bcdA | ||||
| V2.4 | 5,5±0,23aB | 6,5±0,31abcC | 4,5±0,22bcdA | ||||
| V2.5 | 5,5±0,23aB | 6,0±0,27aC | 4,0±0,18aA | ||||
| Variant | Fresh cabbage | Sauerkraut immediately after fermentation |
After 6 months of storage |
| M 1 | 3,34±0,14aB | 0,29±0,01bA | - |
| M2 | 3,34±0,14aB | 0,12±0,006aA | - |
| V1.1 | 3,34±0,14aA | - | - |
| V1.2 | 3,34±0,14a | - | - |
| V1.3 | 3,34±0,14a | - | - |
| V1.4 | 3,34±0,14a | - | - |
| V1.5 | 3,34±0,14a | - | - |
| V2.1 | 3,34±0,14a | - | - |
| V2.2 | 3,34±0,14a | - | - |
| V2.3 | 3,34±0,14a | - | - |
| V2.4 | 3,34±0,14a | - | - |
| V2.5 | 3,34±0,14a | - | - |
| Variant | Fresh cabbage | Sauerkraut immediately after fermentation |
After 6 months of storage |
| M 1 | 20,56±1,07aB | 28,8±1,50deC | 17, 6±0,81abA |
| M2 | 20,56±1,07aB | 28,8±1,39deC | 18,2±0,88abA |
| V1.1 | 20,56±1,07aB | 25,28±1,23bcC | 19,28±0,94bcA |
| V1.2 | 20,56±1,07aB | 22±0,98aC | 17,4±0,85aA |
| V1.3 | 20,56±1,07aB | 23,52±1,15abC | 16,78±0,80aA |
| V1.4 | 20,56±1,07aB | 34,08±1,64ghC | 25,13±1,17eA |
| V1.5 | 20,56±1,07aB | 35,84±1,72hiC | 27,43±1,31fA |
| V2.1 | 20,56±1,07aB | 25,28±1,27bcC | 20,16±0,96cA |
| V2.2 | 20,56±1,07aB | 37,6±1,84iC | 29,62±1,46gA |
| V2.3 | 20,56±1,07aB | 32,32±1,51fgC | 26,98±1,32fA |
| V2.4 | 20,56±1,07aB | 27,04±1,33cdC | 18,06±0,87abA |
| V2.5 | 20,56±1,07aB | 30,56±1,49efC | 23,71±1,11eAB |
| Variant | Fresh cabbage | Sauerkraut immediately after fermentation |
After 6 months of storage |
| M 1 | 18,69±0,90aA | 22,71±1,10aB | 20,64±1,00abA |
| M2 | 18,69±0,90aA | 24,57±1,19abC | 21,19±1,03abcB |
| V1.1 | 18,69±0,90aA | 29,62±1,51bC | 25,79±1,22dB |
| V1.2 | 18,69±0,90aA | 27,66±1,36bC | 22,47±1,06cB |
| V1.3 | 18,69±0,90aA | 25,04±1,22bC | 21,52±0,97bcB |
| V1.4 | 18,69±0,90aA | 24,39±1,17abB | 20,26±0,93abA |
| V1.5 | 18,69±0,90aA | 23,83±1,15abB | 19,61±0,91aA |
| V2.1 | 18,69±0,90aA | 24,39±1,18abC | 21,37±1,01bcB |
| V2.2 | 18,69±0,90aA | 24,95±1,26bC | 21,39±1,03bcB |
| V2.3 | 18,69±0,90aA | 25,32±1,31bC | 22,51±1,16cB |
| V2.4 | 18,69±0,90aA | 24,01±1,24abC | 20,83±0,99abcB |
| V2.5 | 18,69±0,90aA | 23,64±1,15abB | 19,94±0,87abA |
| Variant | Fresh cabbage | Sauerkraut immediately after fermentation |
After 6 months of storage |
| M 1 | 0,89±0,03aB | 0,93±0,04aB | 0,77±0,02aA |
| M2 | 0,89±0,03aA | 0,98±0,05aB | 0,81±0,04aA |
| V1.1 | 0,89±0,03aA | 1,64±0,07cdeC | 1,12±0,05cB |
| V1.2 | 0,89±0,03aA | 1,84±0,09fC | 1,67±0,06eB |
| V1.3 | 0,89±0,03aA | 1,80±0,09fC | 1,59±0,07eB |
| V1.4 | 0,89±0,03aA | 1,74±0,07efC | 1,23±0,05dB |
| V1.5 | 0,89±0,03aA | 1,96±0,08gC | 1,79±0,09fB |
| V2.1 | 0,89±0,03aA | 0,98±0,03aC | 0,82±0,03aB |
| V2.2 | 0,89±0,03aA | 1,66±0,06deC | 1,11±0,04cB |
| V2.3 | 0,89±0,03aA | 1,55±0,07cdC | 1,05±0,05bcB |
| V2.4 | 0,89±0,03aA | 1,32±0,05bC | 0,97±0,03bB |
| V2.5 | 0,89±0,03aA | 1,54±0,06cC | 1,09±0,05cB |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




