1. Introduction
Light-based technologies are increasingly used in clinical dentistry to enhance the effectiveness of various treatment procedures. In in-office tooth whitening, light activation systems are commonly employed to accelerate the activity of hydrogen peroxide–based bleaching agents. These light-assisted systems may influence reaction kinetics, heat generation, and the interaction between the whitening gel and dental tissues. Therefore, understanding the clinical performance of different light activation devices is important for optimizing treatment outcomes and ensuring patient safety [
1,
2,
3].
Hydrogen peroxide remains the most widely used oxidizing agent for in-office whitening procedures due to its ability to diffuse through enamel and dentin and degrade chromogenic molecules via reactive oxygen species [
1,
2,
3]. High-concentration formulations (25–40%) allow for rapid clinical effects but require controlled application conditions to balance efficacy with biological safety. From a biomedical perspective, the manner in which light energy is delivered to activate peroxide gels may influence reaction kinetics, heat generation, and tissue interaction. The interaction between peroxide-based whitening agents and light activation systems remains an area of ongoing clinical and biomedical interest [
4,
5,
6].
Light activation systems used in clinical whitening protocols primarily include light-emitting diode (LED) and laser-based devices. LED systems deliver non-coherent light across a defined spectral range, while diode laser systems emit coherent, monochromatic radiation capable of producing localized photothermal effects. These distinct physical properties may result in different activation efficiencies, treatment durations, and thermal profiles, positioning LED and laser technologies as functionally different medical devices rather than interchangeable light sources [
7,
8,
9].
Accurate assessment of treatment outcomes remains essential in evaluating light-based clinical interventions. Visual shade guides, although commonly used, are inherently subjective and susceptible to operator bias and transient optical changes such as enamel dehydration. Digital spectrophotometry offers an objective and reproducible method for quantifying color changes through CIE L*a*b* parameters, enabling precise evaluation of treatment effects immediately following intervention [
10,
11,
12].
Despite the widespread clinical adoption of both LED- and laser-assisted whitening systems, direct randomized comparisons using objective measurement techniques remain limited. Existing studies often differ in protocol design, outcome assessment, and reporting standards, which complicates meaningful comparison of device performance. Consequently, pilot randomized clinical trials employing standardized protocols and objective endpoints are necessary to generate reliable comparative data [
13,
14,
15].
The present study was designed as a pilot randomized clinical trial to compare the short-term clinical effectiveness of LED- and diode laser–assisted in-office whitening systems when applied as light-based medical device interventions. Treatment outcomes were objectively assessed using digital spectrophotometry, with the aim of providing reproducible comparative data to inform future biomedical research and clinical protocol development. The aim of this pilot randomized clinical trial was to compare the short-term clinical effectiveness of two commercially available light-activated in-office whitening systems applied according to their respective manufacturer-recommended protocols.
2. Materials and Methods
2.1. Ethical Approval
This study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki and received approval from the Research Ethics Committee of Grigore T. Popa University of Medicine and Pharmacy, Iași, Romania (approval no. 643/28.09.2025). All participants received detailed information regarding the study objectives, procedures, and potential risks, and written informed consent was obtained prior to enrollment. The study was registered in the ISRCTN clinical trial registry (ISRCTN62124700). The registration was performed retrospectively after the initiation of participant recruitment.
2.2. Patient Selection
A total of 12 healthy adult participants (aged 18–45 years) presenting clinically perceptible tooth discoloration (extrinsic or mixed-type) with a baseline shade of VITA A3 or darker were enrolled in this pilot randomized clinical trial. Participants were recruited between October and December 2025. All participants underwent professional prophylaxis prior to baseline color assessment to eliminate superficial stains.
2.2.1. Inclusion Criteria
2.2.2. Exclusion Criteria
The exclusion criteria were established to minimize confounding variables and to ensure participant safety.
Pregnancy or breastfeeding were excluded due to ethical considerations and the absence of sufficient safety data on bleaching agents in these populations.
Severe dental hypersensitivity could be exacerbated by hydrogen peroxide, influencing both patient comfort and outcomes.
Presence of active carious or periodontal lesions
Decompensated systemic diseases were excluded due to possible complications in healing or treatment response, while known allergies to bleaching ingredients represent a direct contraindication.
Oral mucosal lesions might interfere with bleaching agent application and increase adverse event risk.
Known allergies to bleaching agent ingredients
Participant flow throughout the study is summarized in
Figure 1.
2.3. Sample Size and Randomization
This study was designed as an exploratory pilot randomized clinical trial with two parallel groups and a 1:1 allocation ratio. A formal a priori sample size calculation was not performed, as the study was designed as a pilot investigation aimed at generating preliminary effect size estimates and assessing feasibility.
Participants were randomly allocated in a 1:1 ratio to two parallel intervention groups (n = 6 per group) using a computer-generated random allocation sequence in Microsoft Excel (RAND function) by an investigator not involved in outcome assessment. Each participant was assigned a numeric study code, and all personal identifiers were removed prior to data analysis to minimize potential bias.
The study design followed CONSORT recommendations for randomized clinical trials [
16,
17].
2.4. Allocation Concealment
Allocation concealment was achieved using sealed, opaque envelopes containing the assigned intervention protocol. Envelopes were opened only at the time of intervention. All procedures were performed by the same clinician under standardized clinical conditions, including isolation and photoprotection.
2.5. Intervention Groups
Participants received a single in-office whitening session according to group allocation. In all participants, whitening treatment was standardized and performed on the maxillary and mandibular anterior teeth (canine to canine). All whitening procedures were performed by the same trained operator to ensure procedural consistency.
Each whitening system was applied according to the manufacturer’s recommended clinical protocol in order to reproduce real-world treatment condition.
The total duration of each clinical whitening session was approximately 60–90 min, including baseline color assessment, the whitening procedure, and immediate post-treatment spectrophotometric evaluation.
Group A – Diode Laser–Activated System (Biolase Epic Inc., Irvine, CA, USA)
- ○
Whitening agent: 35% Hydrogen Peroxide Gel (Biolase Inc., Irvine, CA, USA)
- ○
Activation: Biolase Epic diode laser (7W, pulsed mode)
- ○
Protocol: Three cycles of 30 seconds per cycle , in one session
Group B – LED-Activated System (Philips Zoom WhiteSpeed LED lamp - Philips Oral Healthcare, Ontario, CA, USA):
- ○
Whitening agent: Zoom 25% Hydrogen Peroxide Gel (Philips Oral Healthcare, Ontario, CA, USA)
- ○
Activation: Philips Zoom WhiteSpeed LED lamp (400–505 nm)
- ○
Protocol: 3 cycles of 15 minutes each, with gel reapplied between cycles
2.6. Blinding
Tooth color assessment was performed by a second investigator (Co-investigator 2), a licensed dentist experienced in aesthetic dentistry, using a digital spectrophotometer (VITA Easyshade V, VITA Zahnfabrik, Bad Säckingen, Germany). Both investigators were calibrated prior to the study using standardized shade tabs to ensure measurement consistency.
The study followed a single-blind design. Due to the nature of the interventions, the treating clinician was aware of group allocation; however, colorimetric assessment was performed by the second calibrated investigator who was blinded to treatment assignment.
2.7. Whitening Systems Compared
The study compared two commercially available light-based medical device systems:
Diode laser system (Biolase Epic diode laser -Biolase Inc., Irvine, CA, USA; 940–980 nm)
LED-based system (Philips Zoom WhiteSpeed LED lamp-Philips Oral Healthcare, Ontario, CA, USA; 400–505 nm)
Each whitening system was applied strictly according to the manufacturer’s recommended clinical protocol in order to reproduce real-world treatment conditions. Material composition and manufacturer details are summarized in
Table 1.
2.8. Treatment Protocol
2.8.1. Patient Preparation
All patients underwent standardized oral hygiene procedures, including ultrasonic scaling (Woodpecker UDS-E ultrasonic scaler- Guilin Woodpecker Medical Instrument Co., Ltd., Guilin, China) according to the manufacturer’s instructions with sterile universal tips, followed by polishing with sterile prophylaxis brushes and non-fluoridated prophy paste. Identical equipment and materials were used for all participants to ensure consistency.
2.8.2. LED-Activated Whitening Protocol (Philips Zoom WhiteSpeed LED lamp-Philips Oral Healthcare, Ontario, CA, USA)
The initial tooth shade was assessed using a calibrated digital spectrophotometer (VITA Easyshade V, VITA Zahnfabrik, Bad Säckingen, Germany). For patient safety and comfort during the procedure, protective eyewear was provided. Cheeks were retracted using cheek retractors, and a lip and cheek retractor was placed to ensure optimal access to the dental arches. A light-cured gingival barrier (Liquidam, Philips Oral Healthcare, Ontario, CA, USA) was applied along the gingival margins to protect the soft tissues. The barrier was applied in an approximately 2-mm-wide layer and light-cured according to the manufacturer’s instructions to ensure complete isolation (“no pink rule”).
The whitening gel used in the LED-activated protocol (Philips Zoom) contains 25% hydrogen peroxide combined with light-absorbing chromophore components designed to enhance interaction with the LED light source during photo-activation, according to the manufacturer’s protocol.
A hydrogen peroxide whitening gel (Zoom Whitening Gel, 25% hydrogen peroxide; Philips Oral Healthcare, Ontario, CA, USA) was applied in a uniform layer approximately 1–2 mm thick on the buccal surfaces of the maxillary and mandibular anterior teeth involved in the treatment (canine to canine).
The whitening gel was activated by positioning the Zoom LED lamp approximately 2–3 cm away from the tooth surface. Three exposure cycles of 15 minutes each were performed. The gel was reapplied at the beginning of each new cycle. After each activation cycle, the whitening gel was removed using high-volume suction and gentle water irrigation, in accordance with manufacturer instructions.
After completion of the final activation cycle, the whitening gel was carefully removed using high-volume suction and sterile gauze. The gingival barrier was then removed, and the patient was instructed to rinse thoroughly.
At the end of the session, a desensitizing gel containing amorphous calcium phosphate (Relief ACP, Philips Oral Healthcare, Ontario, CA, USA) was applied on the treated tooth surfaces according to the manufacturer’s recommendations. The patient received information about the potential for transient tooth sensitivity following the procedure.
2.8.3. Diode Laser–Activated Whitening Protocol (Biolase Epic diode laser -Biolase Inc., Irvine, CA, USA)
The diode laser whitening procedure was performed using a 940-nm diode laser system (Biolase Epic diode laser Biolase Inc., Irvine, CA, USA) in combination with a hydrogen peroxide whitening gel (LaserWhite 20, Biolase Inc., Irvine, CA, USA).
Prior to treatment, professional prophylaxis was performed using pumice to remove superficial plaque and extrinsic stains. Soft tissue isolation was achieved using a cheek and lip retractor and a light-cured gingival barrier (Liquidam, Biolase Inc., Irvine, CA, USA), applied along the gingival margins (approximately 2 mm thickness, extending at least one tooth beyond the treatment area) and polymerized according to the manufacturer’s instructions (“no pink rule”).
Both the patient and operator wore protective goggles (Biolase) to ensure safety during the procedure.
The LaserWhite20 whitening system (BIOLASE Inc., Irvine, CA, USA) consists of a dual-syringe system containing a 45% hydrogen peroxide base gel and a proprietary activator. After mixing according to manufacturer instructions, the final hydrogen peroxide working concentration is 35%. The whitening gel was applied in a uniform layer approximately 1–1.5 mm thick on the buccal surfaces of the maxillary and mandibular anterior teeth involved in the treatment (canine to canine).
Laser activation was performed using the manufacturer’s preset whitening mode with a power setting of 7W in continuous mode. The whitening handpiece was positioned in close proximity to the gel surface without direct contact.
The laser irradiation was performed sequentially across the dental quadrants, with 30 seconds pre-set time of irradiation per quadrant. Three cycles were performed for each patient in a single session. Following the activation cycles, the gel was allowed to remain on the tooth surfaces for several minutes before being removed.
After completion of the final activation cycle, the whitening gel was carefully removed using high-volume suction and sterile gauze. The gingival barrier was then removed, and the patient was instructed to rinse thoroughly. The patient was informed about the possibility of postoperative tooth hypersensitivity.
At the end of the session, a desensitizing gel included in the kit (Laser White 20, containing 3% potassium nitrate) was applied on the treated tooth surfaces according to the manufacturer’s recommendations.
2.9. Color Measurement Protocol
For colorimetric analysis, measurements were standardized by evaluating only the maxillary central incisor in each participant. The central incisor was selected due to its high esthetic relevance and central position in the smile line. Measurements were obtained using a calibrated digital spectrophotometer (VITA Easyshade V, VITA Zahnfabrik, Bad Säckingen, Germany), at two time points: baseline (T0) and immediately after treatment (T1).
Measurements were performed on the middle third of the labial surface of the maxillary central incisor in each participant. The probe tip of the spectrophotometer was positioned perpendicular to the tooth surface under standardized ambient lighting conditions.
A positioning jig was not used; however, probe placement was standardized according to the manufacturer’s recommendations to minimize variability.
Colorimetric outcomes were assessed by measuring changes in tooth color (ΔE). For each participant, ΔE values were calculated from the differences in CIE L*a*b* coordinates obtained before and after intervention [
8,
12].
Color differences (ΔE) were calculated using the following formula:
where L* represents lightness, a* represents the red–green axis, and b* represents the yellow–blue axis.
3. Results
A total of 12 participants were enrolled and randomized into two intervention groups (n = 6 per group). No participants were lost to follow-up or excluded from the analysis, and all enrolled participants completed the study protocol.
3.1. Colorimetric outcomes
Colorimetric outcomes were evaluated based on ΔE values calculated from spectrophotometric measurements performed at baseline (T0) and immediately after treatment (T1). Measurements were standardized by evaluating the maxillary central incisor in each participant.The calculated ΔE values were used to quantify the magnitude of tooth color change following the whitening procedures. Descriptive statistics for ΔE values in both study groups are presented in
Table 2.
3.2. Descriptive Outcomes
As shown, the Zoom group presented a slightly higher mean ΔE value compared to the Biolase group. Descriptive statistics for ΔE values are summarized in
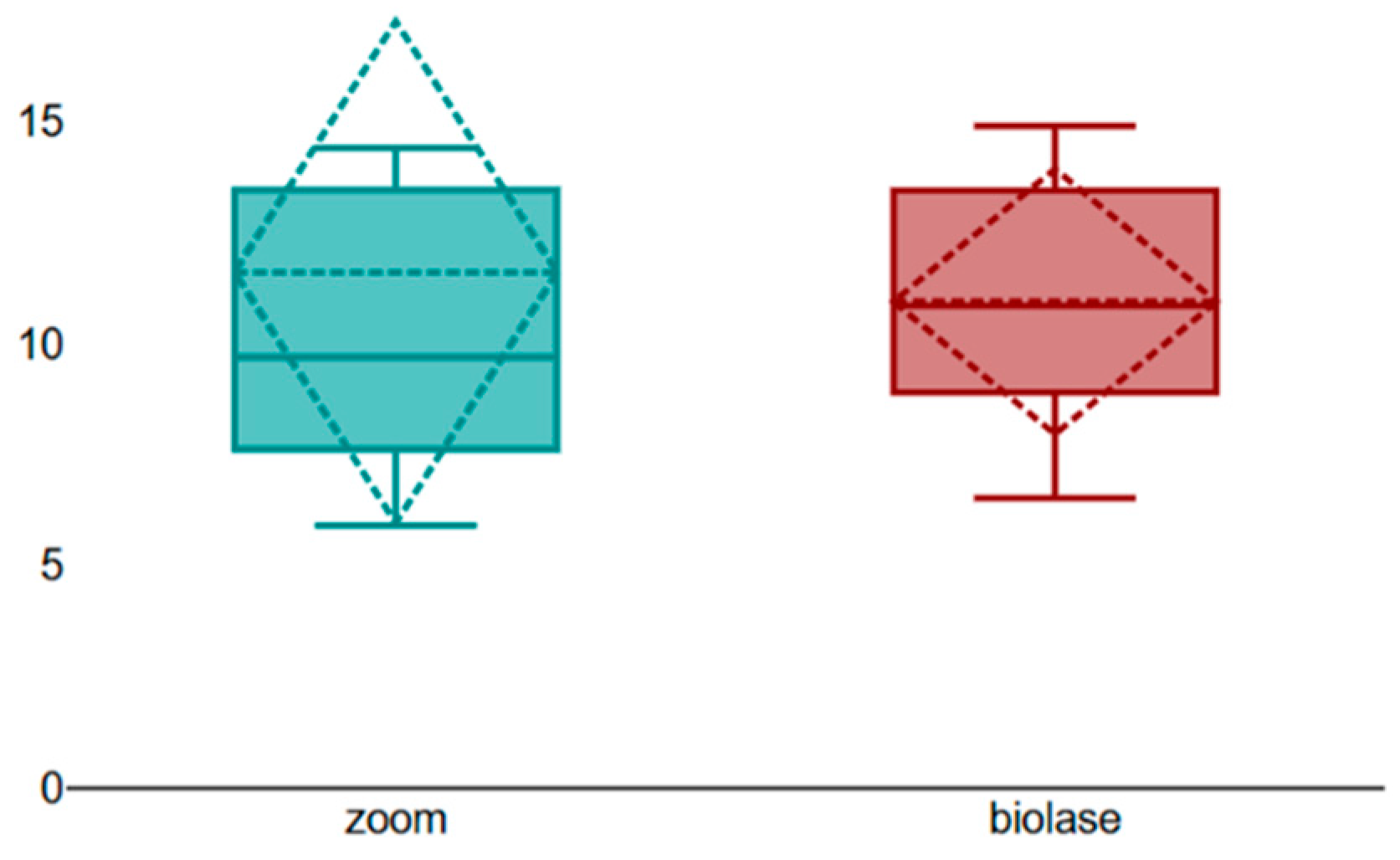
Table 2. In the diode laser–assisted group (Biolase), the mean ΔE value was 10.96 ± 3.27, with values ranging from 6.53 to 14.91. In the LED-assisted group (Zoom), the mean ΔE value was 11.62 ± 5.93, with a range between 5.92 and 22.71. In both groups, ΔE values exceeded the commonly accepted perceptibility threshold (ΔE > 3.3).
3.3. Comparative Statistical Analysis
Group comparisons were performed using the Mann–Whitney U test due to the small sample size and non-normal data distribution. The statistical results are presented in
Table 3. No statistically significant difference in ΔE values was observed between the diode laser–assisted and LED-assisted groups (U = 16, Z = −0.32, p = 0.818). The calculated effect size (r = 0.09) indicated a small difference between groups.
3.4. Visual Representation of ΔE Value Distribution
The distribution of ΔE values for both intervention groups is illustrated in
Figure 2. The box plot demonstrates overlapping interquartile ranges and similar median values between the diode laser–assisted and LED-assisted groups. Variability was observed in both groups, with a wider range of ΔE values noted in the LED-assisted group.
3.5. ΔE Threshold for Perceptible Colorimetric Change
A ΔE value greater than 3.3 is widely used as a reference threshold for perceptible colorimetric change [
20,
21]. In the present study, all participants in both intervention groups exhibited ΔE values exceeding this threshold following treatment. Specifically, 6 out of 6 participants (100%) in both the diode laser–assisted and LED-assisted groups demonstrated ΔE values above 3.3.
Mean ΔE values for the diode laser–assisted group and the LED-assisted group were 10.96 ± 3.27 and 11.62 ± 5.93, respectively. As previously reported, no statistically significant difference was observed between groups. The proportion of cases exceeding the perceptibility threshold is summarized in
Table 4.
Both whitening systems produced clinically perceptible color changes. No statistically significant difference was observed between the two protocols. No adverse events or clinically relevant complications were recorded during or immediately after the whitening procedures in either treatment group
4. Discussion
This pilot randomized clinical trial evaluated the immediate objective whitening outcomes of diode laser–activated and LED-activated in-office whitening systems when applied as light-based medical device interventions and assessed using objective digital spectrophotometry. Although the LED-activated protocol demonstrated a slightly higher mean ΔE value compared to the laser-assisted system, the observed difference was not statistically significant (p = 0.818). These findings indicate that both activation modalities were capable of producing immediate measurable color changes under standardized exposure conditions. However, the absence of statistically significant differences should not be interpreted as evidence of equivalence between activation modalities.
Given the limited sample size (n = 6 per group), the present study was underpowered for definitive comparative inference. As an exploratory pilot investigation, its primary objective was to generate preliminary effect size estimates and assess feasibility rather than to establish superiority or equivalence. Larger, adequately powered randomized trials are required to confirm these findings and allow robust comparative conclusions.
The lack of significant differences between the two systems is biologically plausible when considering the fundamental mechanisms of peroxide-based chromogen oxidation. Hydrogen peroxide acts primarily through the generation of reactive oxygen species that oxidize organic chromophores within dental hard tissues. Light activation—whether delivered via coherent laser radiation or non-coherent LED emission—is intended to accelerate peroxide decomposition and reaction kinetics. Once key parameters such as gel concentration, exposure time, and application protocol are standardized, additional differences in light delivery characteristics may not proportionally influence immediate measurable color change [
4,
5,
6,
9].
The whitening protocols evaluated in this study correspond to manufacturer-recommended clinical workflows, which may involve different peroxide concentrations and activation times depending on the specific light-based device and product formulation.
From a biomedical device perspective, diode laser and LED systems exhibit distinct physical and optical properties, including wavelength specificity, coherence, and energy distribution. Nevertheless , these technical differences do not necessarily translate into superior short-term outcomes when evaluated immediately after treatment using objective colorimetric methods. The present findings support the concept that device-related optical characteristics may influence operational parameters, such as treatment efficiency or energy delivery dynamics, rather than the magnitude of immediate colorimetric change [
7,
14,
21].
Tooth color changes were assessed using digital spectrophotometry, which provides standardized quantitative measurements of color change through
CIE Lab* color system. Objective quantification through ΔE values reduces operator-dependent variability and enhances methodological transparency. This approach allows objective comparison between different whitening protocols while minimizing operator-dependent variability [
12,
15].
The novelty of the present study lies in the device-oriented, biomedical evaluation of light-assisted tooth whitening using objective colorimetric outcomes, rather than subjective visual assessment or purely procedural comparisons.
Although no statistically significant differences were detected between the two protocols, the limited statistical power precludes definitive comparative conclusions. Larger randomized trials are required to determine whether clinically meaningful differences exist between activation modalities.
Although laser-assisted whitening systems are frequently promoted as offering enhanced performance, existing clinical evidence remains inconsistent when standardized protocols and objective measurement techniques are employed. The present results are consistent with previous comparative investigations reporting similar short-term colorimetric outcomes between laser- and LED-activated systems. These observations underscore the importance of protocol standardization and objective assessment in distinguishing true device-related effects from transient optical phenomena such as enamel dehydration.
Several limitations must be acknowledged. The limited sample size reflects the pilot nature of the trial and restricts statistical power. Consequently, the study was not designed or powered to establish superiority or equivalence between treatment modalities, but rather to generate preliminary data and effect size estimates for future investigations. Colorimetric outcomes were assessed exclusively immediately after treatment. Therefore, long-term color stability, relapse phenomena, and sustained biological effects could not be evaluated. Given that post-treatment color rebound is clinically relevant, future trials should incorporate longitudinal follow-up as a primary endpoint. Furthermore, colorimetric assessment was limited to a single maxillary central incisor, which may not fully represent potential intra-arch variability in whitening response.
Additionally, a customized positioning jig was not used during spectrophotometric assessment. Although measurements were standardized and performed by a calibrated operator, minor variability related to probe positioning cannot be excluded.
Thermal changes associated with light activation were not directly monitored. Although no adverse events were recorded, potential intrapulpal temperature increase remains a relevant safety consideration in light-assisted bleaching procedures and should be systematically evaluated in future research.
Another limitation is the absence of patient-reported outcomes, including tooth sensitivity and subjective aesthetic perception. Although objective ΔE measurements provide quantitative evidence of color change, whitening treatments are ultimately evaluated from both clinical and patient-centered perspectives. Tooth sensitivity and patient satisfaction represent important components of overall treatment evaluation. Therefore, incorporating validated patient-reported outcome measures in future studies would provide a more comprehensive evaluation of the clinical impact of light-assisted bleaching protocols.
Despite these limitations, this pilot study provides preliminary data supporting the feasibility of objective, device-oriented comparison of light-based whitening technologies.
The present study compared two complete clinical whitening systems rather than isolating peroxide concentration as a single variable. Therefore, the findings reflect real-world device performance under manufacturer-recommended conditions.
The findings suggest that both diode laser and LED activation can be incorporated into standardized protocols without compromising immediate colorimetric outcomes. Further investigations are warranted to clarify the long-term biomedical implications of light-assisted whitening systems and to establish standardized evaluation frameworks applicable across different light-based medical device applications.
5. Conclusions
Within the limitations of this exploratory pilot randomized clinical trial, both light-assisted in-office whitening protocols produced measurable immediate color changes as assessed by objective digital spectrophotometry.
No statistically significant difference was observed between the two systems; however, due to the limited sample size, the study was underpowered for definitive comparative conclusions.
These preliminary findings suggest that both light-assisted whitening protocols were capable of producing short-term whitening effects under the clinical conditions evaluated, but larger, adequately powered trials incorporating longitudinal follow-up, patient-reported outcomes, and safety monitoring are necessary to establish their relative efficacy and long-term clinical performance.
Author Contributions
Conceptualization, C.A., T.P., C.B. and M.C.; methodology, C.A., T.P., C.B. and M.C.; software, T.P.; validation, C.A. and C.B.; formal analysis C.B.; investigation, C.A.; resources, M.C.; data curation, T.P.; writing—original draft preparation, C.A.; writing—review and editing, C.B.; visualization, T.P..; supervision, M.C.; project administration, C.A.; funding acquisition, M.C. All authors have reviewed and approved the final version of the manuscript and take full responsibility for its content.
Funding
This research was investigator-initiated and received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Research Ethics Committee of Grigore T. Popa University of Medicine and Pharmacy, Iași, Romania (approval no. 643, approved on 28 September 2025).
Informed Consent Statement
Written informed consent was obtained from all participants involved in the study.
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request. A minimal anonymized dataset has been provided during submission.
Clinical Trial Registration
The study was registered in the ISRCTN clinical trial registry (ISRCTN62124700). Registration was completed retrospectively.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| LED |
Light-emitting diode |
| Nm |
Nanometre |
| ΔE |
total color difference in the CIE L*a*b* color space |
| CIE L*a*b* |
Commission Internationale de l’Éclairage color space |
References
- Joiner, A. Whitening Toothpastes: A Review of the Literature. J. Dent. 2010, 38 (Suppl. 2), e17–e24. [Google Scholar] [CrossRef] [PubMed]
- Kwon, S.R.; Wertz, P.W. Review of the Mechanism of Tooth Whitening. J. Esthet. Restor. Dent. 2015, 27, 240–257. [Google Scholar] [CrossRef]
- Epple, M.; Meyer, F.; Enax, J. A Critical Review of Modern Concepts for Teeth Whitening. Dentistry Journal 2019, 7, 79. [Google Scholar] [CrossRef]
- Carey, C.M. Tooth Whitening: What We Now Know. J. Evid.-Based Dent. Pract. 2014, 14, 70–76. [Google Scholar] [CrossRef]
- Buchalla, W.; Attin, T. External Bleaching Therapy with Activation Systems. Dent. Mater. 2007, 23, 586–596. [Google Scholar] [CrossRef] [PubMed]
- Luk, K.; Tam, L.; Hubert, M. Effect of Light Energy on Peroxide Tooth Bleaching. J. Am. Dent. Assoc. 2004, 135, 194–201. [Google Scholar] [CrossRef]
- Jacques, S.L. Optical and Thermal Mechanisms in Laser–Tissue Interactions. Phys. Med. Biol. 2013, 58, R37–R61. [Google Scholar] [CrossRef]
- International Organization for Standardization (ISO). ISO/TR 28642:2016—Dentistry—Guidance on Colour Measurement; ISO. Geneva, Switzerland, 2016.
- Almeida, J.R.; Farias-Neto, A.M.; Almeida, R.R. Reliability of Spectrophotometers in Detecting Color Differences in Dental Materials: A Systematic Review. J. Esthet. Restor. Dent. 2021, 33, 182–190. [Google Scholar] [CrossRef]
- Möbius, D.; Braun, A.; Franzen, R. Evaluation of Tooth Color Change after a Bleaching Process with Different Lasers. Odontology 2024, 112, 872–883. [Google Scholar] [CrossRef]
- Lee, Y.K.; Yu, B. Measurement of Opalescence of Tooth Enamel. J. Dent. 2007, 35, 690–694. [Google Scholar] [CrossRef] [PubMed]
- International Organization for Standardization (ISO). ISO 11664-4:2019—Colorimetry—Part 4: CIE L*a*b* Colour Space; ISO. Geneva, Switzerland, 2019; 12.
- Marson, F.C.; Sensi, L.G.; Vieira, L.C.; Araújo, E. Clinical Evaluation of In-Office Dental Bleaching with and without Light Activation. Oper. Dent. 2008, 33, 15–22. [Google Scholar] [CrossRef]
- Gürgan, S.; et al. Different Light-Activated In-Office Bleaching Systems: A Clinical Evaluation. Lasers Med. Sci. 2010, 25, 817–822. [Google Scholar] [CrossRef]
- Kiryk, J.; et al. Effectiveness of Laser-Assisted Teeth Bleaching: A Systematic Review. Appl. Sci. 2024, 14, 3840. [Google Scholar] [CrossRef]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 Statement: Updated Guidelines for Reporting Randomized Trials. BMJ 2010, 340, c332. [Google Scholar] [CrossRef]
- Moher, D.; Hopewell, S.; Schulz, K.F.; et al. CONSORT 2010 Explanation and Elaboration. BMJ 2010, 340, c869. [Google Scholar] [CrossRef] [PubMed]
- Johnston, W.M.; Kao, E.C. Assessment of Appearance Match by Visual Observation and Clinical Colorimetry. J. Dent. Res. 1989, 68, 819–822. [Google Scholar] [CrossRef] [PubMed]
- Ghinea, R.; Pérez, M.M.; Herrera, L.J.; et al. Dental Color Difference Thresholds for Perceptibility and Acceptability. Dent. Mater. 2010, 26, 681–688. [Google Scholar] [CrossRef]
- Paravina, R.D.; Ghinea, R. Color Difference Thresholds in Dentistry. J. Esthet. Restor. Dent. 2015, 27 (Suppl. 1), S1–S9. [Google Scholar] [CrossRef] [PubMed]
- Tekce, A.U.; Yazici, A.R. Clinical Comparison of Diode Laser- and LED-Activated Tooth Bleaching: 9-Month Follow-Up. Lasers Med. Sci. 2022, 37, 3237–3247. [Google Scholar] [CrossRef]
- Ontiveros, J.C. In-Office Vital Bleaching with Adjunct Light. Dent. Clin. North Am. 2011, 55, 241–253. [Google Scholar] [CrossRef]
- Basting, R.T.; Amaral, F.L.; França, F.M.; Flório, F.M. Clinical Comparative Study of Bleaching Materials. Oper. Dent. 2012, 37, 464–473. [Google Scholar] [CrossRef]
- Baroudi, K.; Hassan, N.A. The Effect of Light-Activation Sources on Tooth Bleaching. Niger. Med. J. 2014, 55, 363–368. [Google Scholar] [CrossRef] [PubMed]
- He, L.B.; et al. Effects of Light on Bleaching and Tooth Sensitivity: A Systematic Review and Meta-Analysis. J. Dent. 2012, 40, 644–653. [Google Scholar] [CrossRef]
- Attin, T.; Paqué, F.; Ajam, F.; Lennon, A.M. Review of the Current Status of Tooth Whitening. Int. Endod. J. 2003, 36, 313–329. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.R.; Parrish, J.A. Selective Photothermolysis. Science 1983, 220, 524–527. [Google Scholar] [CrossRef]
- Hamblin, M.R. Mechanisms and Mitochondrial Redox Signaling in Photobiomodulation. Photochem. Photobiol. 2018, 94, 199–212. [Google Scholar] [CrossRef]
- Welch, A.J.; van Gemert, M.J.C. Optical–Thermal Response of Laser-Irradiated Tissue; Springer: New York, NY, USA.
- Zhang, Q.; Liu, Y.; Ding, M.; et al. On-Demand Free Radical Release by Laser Irradiation. Gels 2023, 9, 554. [Google Scholar] [CrossRef] [PubMed]
- Marto, C.M.; et al. Cytotoxic Effects of Zoom® Whitening Product. Materials 2020, 13, 1491. [Google Scholar] [CrossRef]
- Tunér, J.; Hode, L. Laser Phototherapy—Clinical Practice and Mechanisms. Photomed. Laser Surg. 2010, 28, 1–5. [Google Scholar]
- Joiner, A.; Luo, W. Tooth Colour and Whiteness: A Review. J. Dent. 2017, 67, 3–10. [Google Scholar] [CrossRef]
- Commission Internationale de l’Éclairage (CIE). Colorimetry—CIE L*a*b* Colour Space (CIE 1976); CIE: Vienna, Austria.
- Haywood, V.B. Current Status of Nightguard Vital Bleaching. Compend. Contin. Educ. Dent. 2000, 21, 28–34. [Google Scholar]
- Sulieman, M. An Overview of Bleaching Techniques. Dent. Update 2004, 31, 608–616. [Google Scholar] [CrossRef] [PubMed]
- Ontiveros, J.C.; Paravina, R.D. Color Change of Vital Teeth Exposed to Bleaching with and without Supplementary Light. J. Dent. 2009, 37, 840–847. [Google Scholar] [CrossRef] [PubMed]
- Kugel, G.; Aboushala, A.; Zhou, X.; Gerlach, R.W. Clinical Evaluation of Tooth Whitening Systems. Compend. Contin. Educ. Dent. 2006, 27, 54–63. [Google Scholar] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |