Submitted:
09 March 2026
Posted:
09 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental Principles and Apparatus
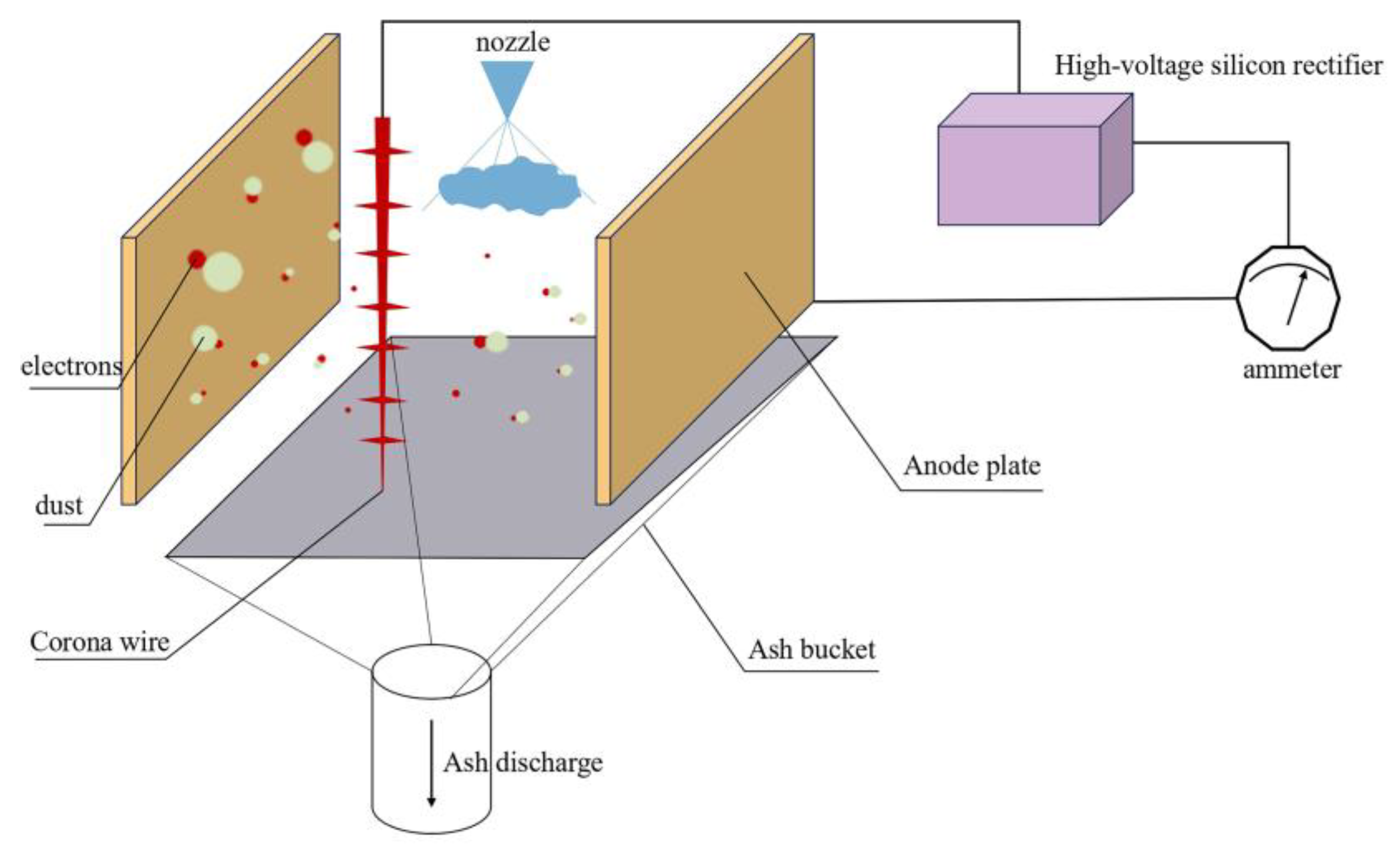
2.1. Experimental Principles
2.2. Experimental Apparatus
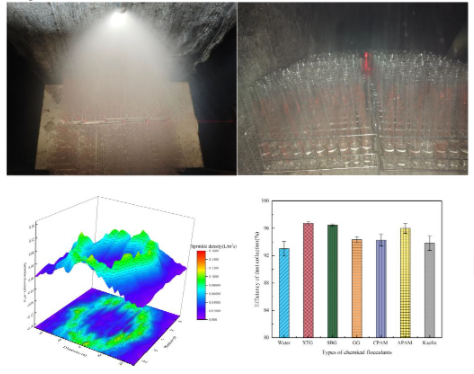
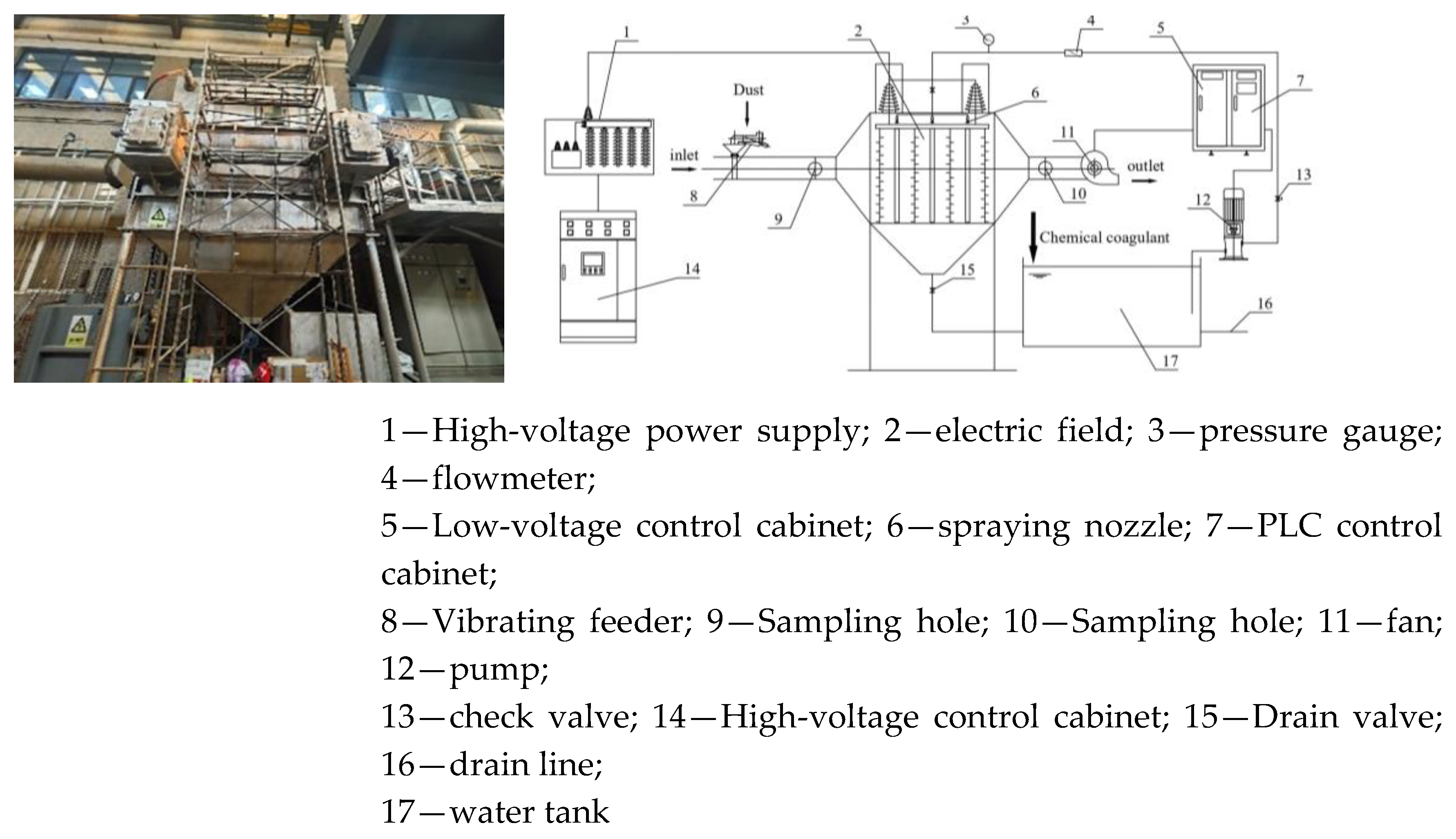
2.2.1. Integrated Wet Electrostatic Precipitator Test Platform
2.3. Detection Instruments and Analysis System
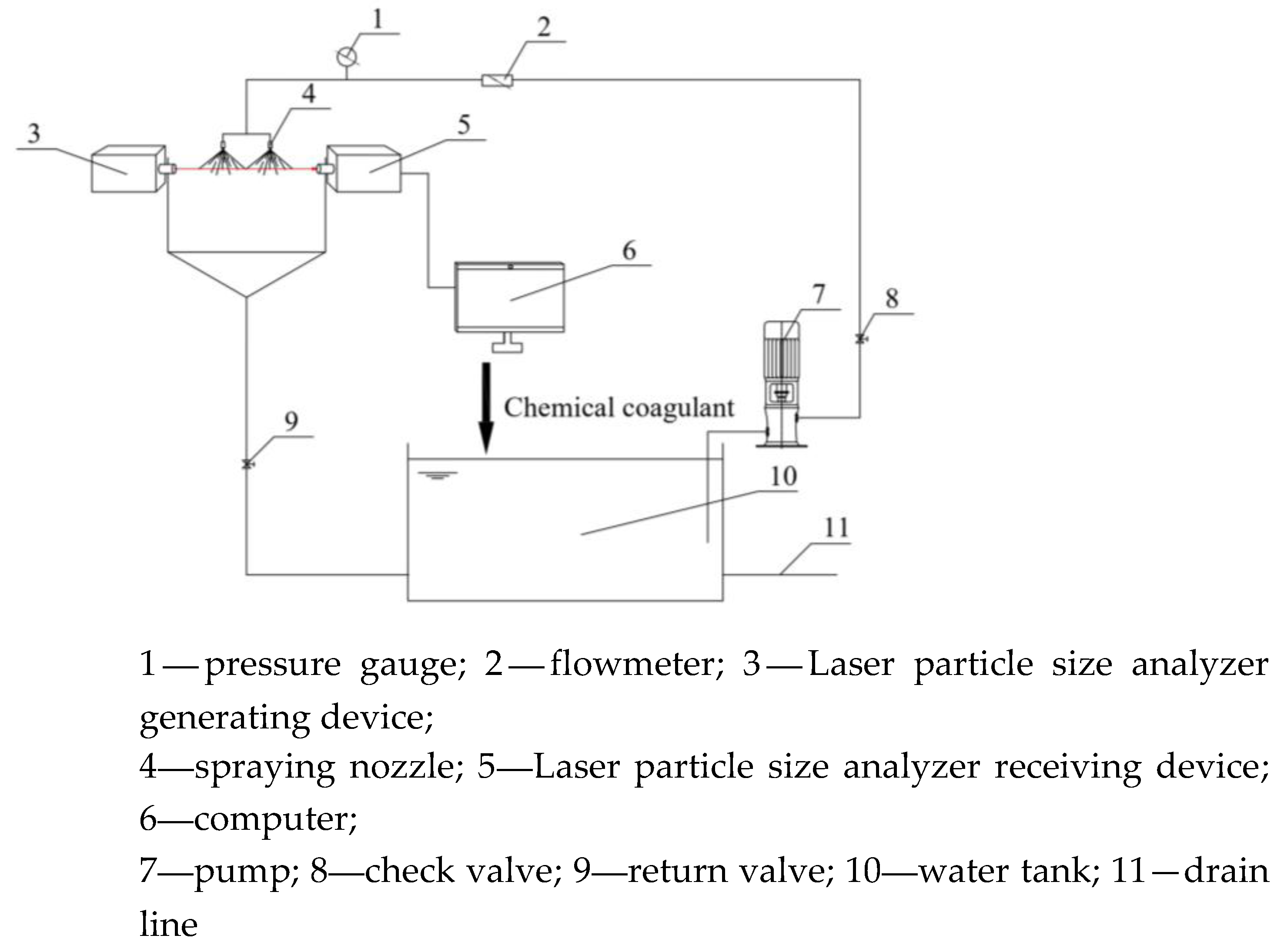
2.3.1. Spray Atomization Test Platform
2.3.2. Fine Particulate Dust Detection Platform
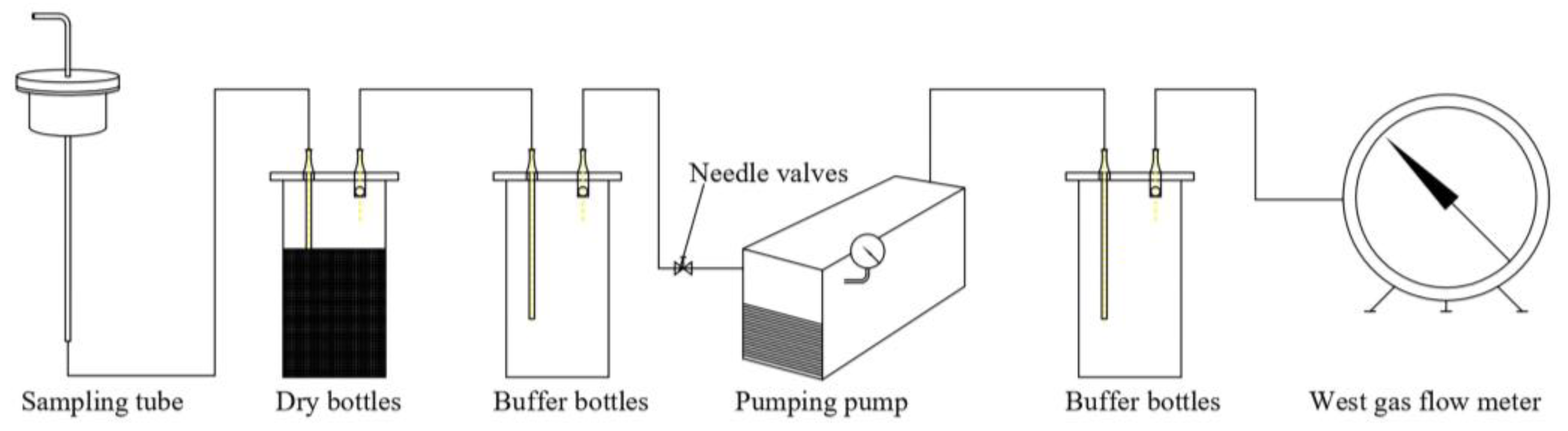
Dust Sampling System
Particle Size Analysis Apparatus
2.4. Experimental Reagents
| Species | Name | Type | Characteristics | Manufacturer |
|---|---|---|---|---|
| Chemical Agglomerant | Cationic Polyacrylamide (CPAM) | Cationic Synthetic Organic Polymer Flocculant | White granules, soluble in water at any ratio to form a uniform, transparent solution. | Tianjin Damao Chemical Reagent Factory |
| Anionic Polyacrylamide (APAM) | Anionic Synthetic Organic Polymer Flocculant | White granules, soluble in water at any ratio to form a uniform, transparent solution. | Tianjin Damao Chemical Reagent Factory | |
| Sesbania Gum (SBG) | Anionic Natural Organic Polymer Flocculant | White powder, dispersible in cold water at room temperature to form a highly viscous hydrocolloidal solution. | Xinrui Biotechnology Co., Ltd | |
| Guar Gum (GG) | Non-ionic Natural Organic Polymer Flocculant | Pale yellowish-brown powder, dispersible in hot or cold water to form a viscous solution; viscosity influenced by dissolution time, temperature, and pH. | Beijing Guarun Technology Co., Ltd | |
| Xanthan Gum (XTG) | Anionic Natural Organic Polymer Flocculant | Light yellow powder, disperses and emulsifies in water to form a stable hydrophilic viscous gel. | Tianjin Guangfu Fine Chemical Research Institute | |
| Kaolin | Natural Mineral Coagulant Aid | Solid powder with varied colors and soft luster, exhibiting good water absorption. | Tianjin Zhonglian Chemical Reagent Co., Ltd | |
| Surfactant | Dodecyl Dimethyl Benzyl Ammonium Chloride (DDBAC) | Cationic Surfactant | Pale yellow waxy solid or gelatinous mass, readily soluble in water, slightly soluble in ethanol; aqueous solution is weakly alkaline. | Macklin Biochemical Technology Co., Ltd |
| Octylphenol Ethoxylate (OP-10) | Non-ionic Surfactant | Pale yellow oily liquid, readily soluble in oils and other organic solvents, strongly lipophilic, poorly soluble in water; acts as a lipophilic emulsifier. | Wuxi Yatai United Chemical Co., Ltd | |
| Sodium Dodecyl Benzene Sulfonate (SDBS) | Anionic Surfactant | White or pale yellow powder or flakes, low volatility, readily soluble in water to form a semi-transparent solution; slightly toxic. | Tianjin Guangfu Fine Chemical Research Institute |
3. Results and Analysis
3.1. Particle Size and Morphology Analysis of Blast Furnace Dust
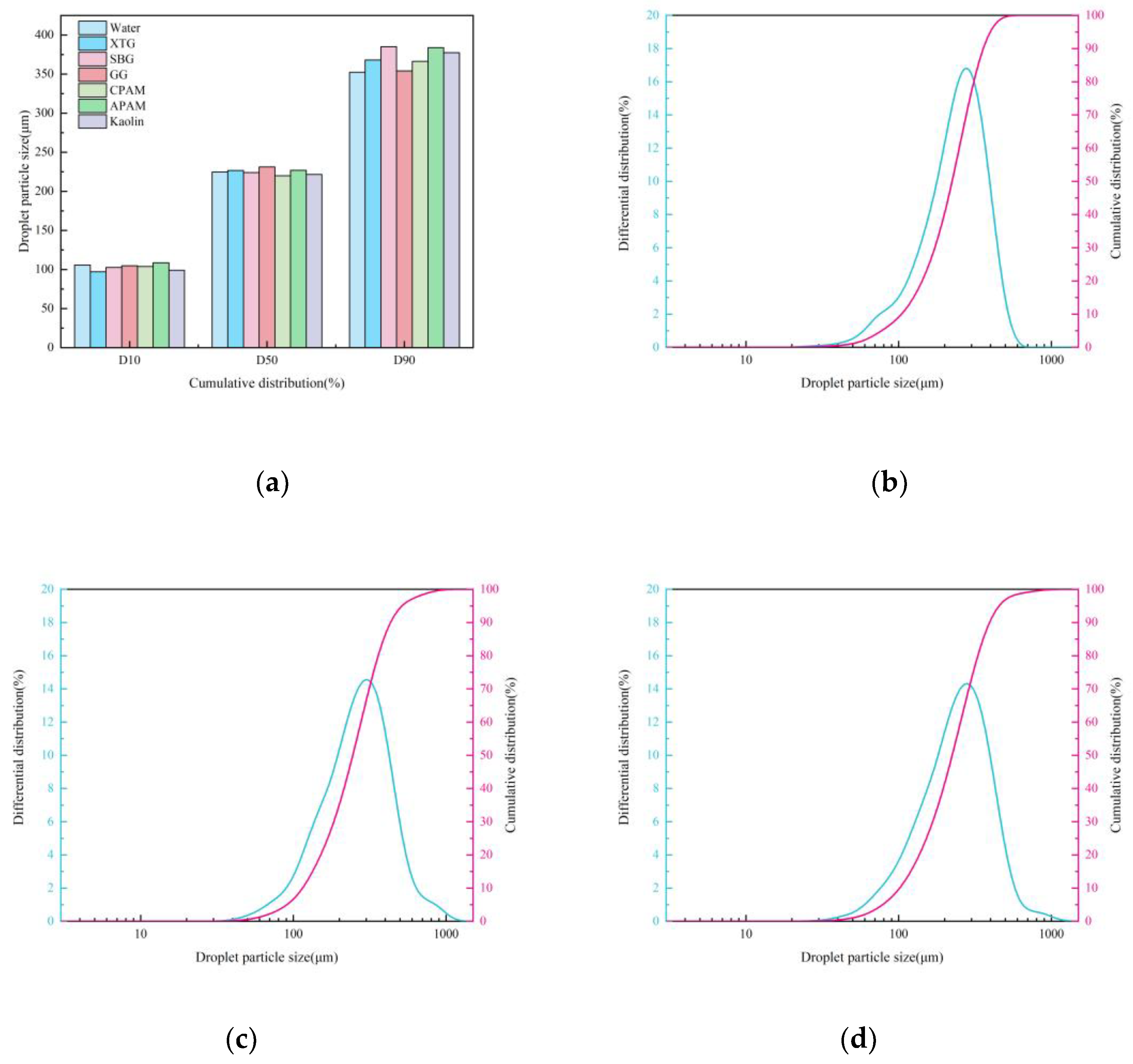
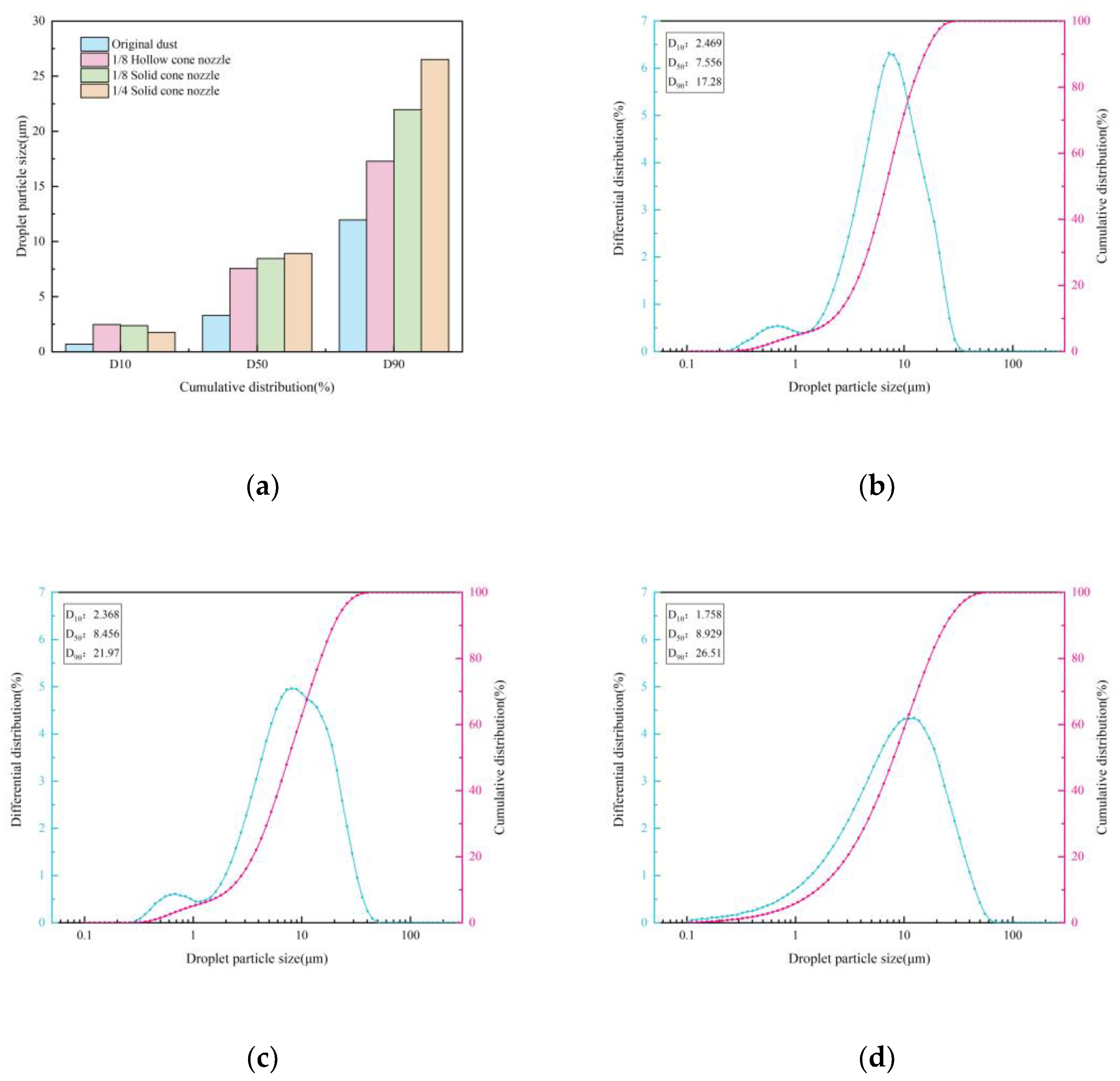
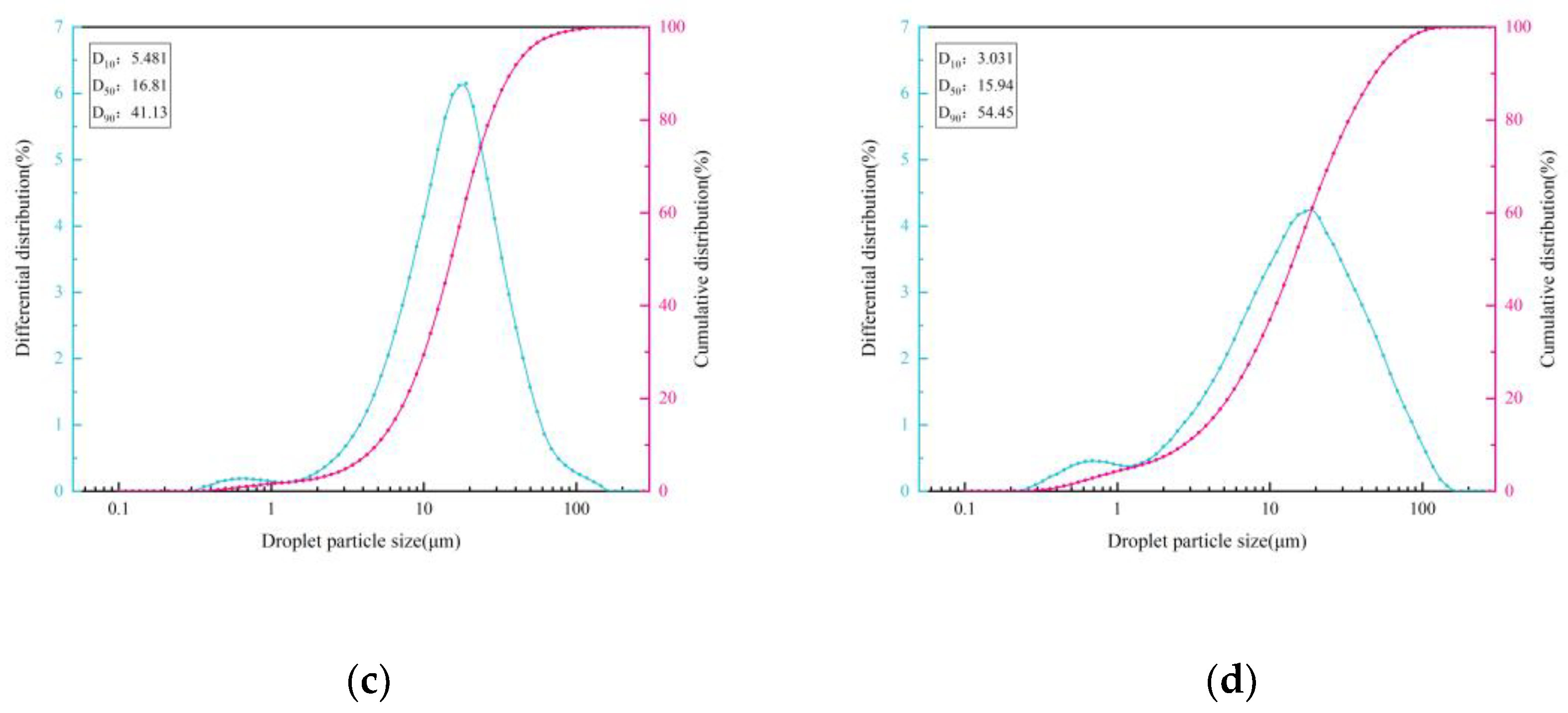
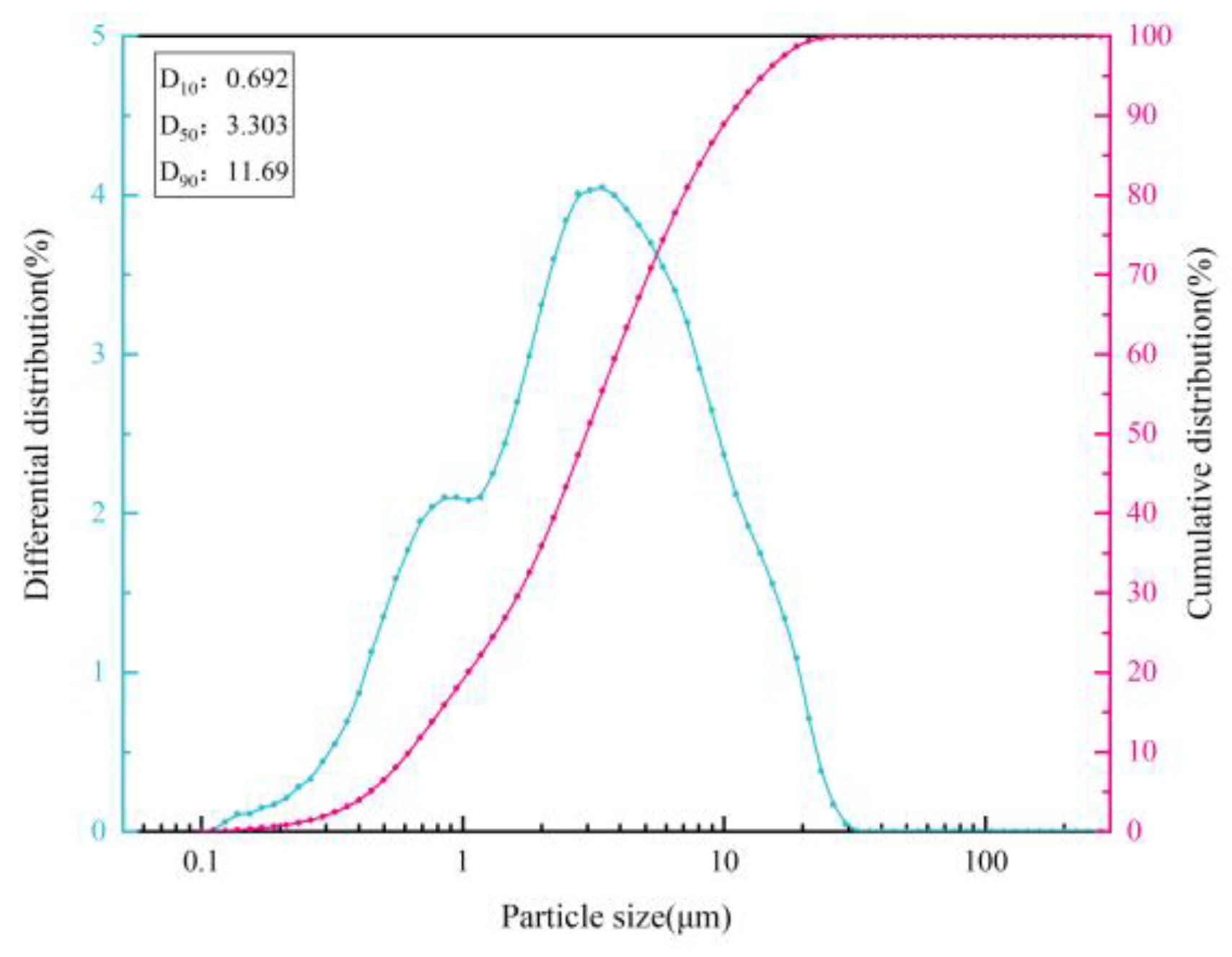
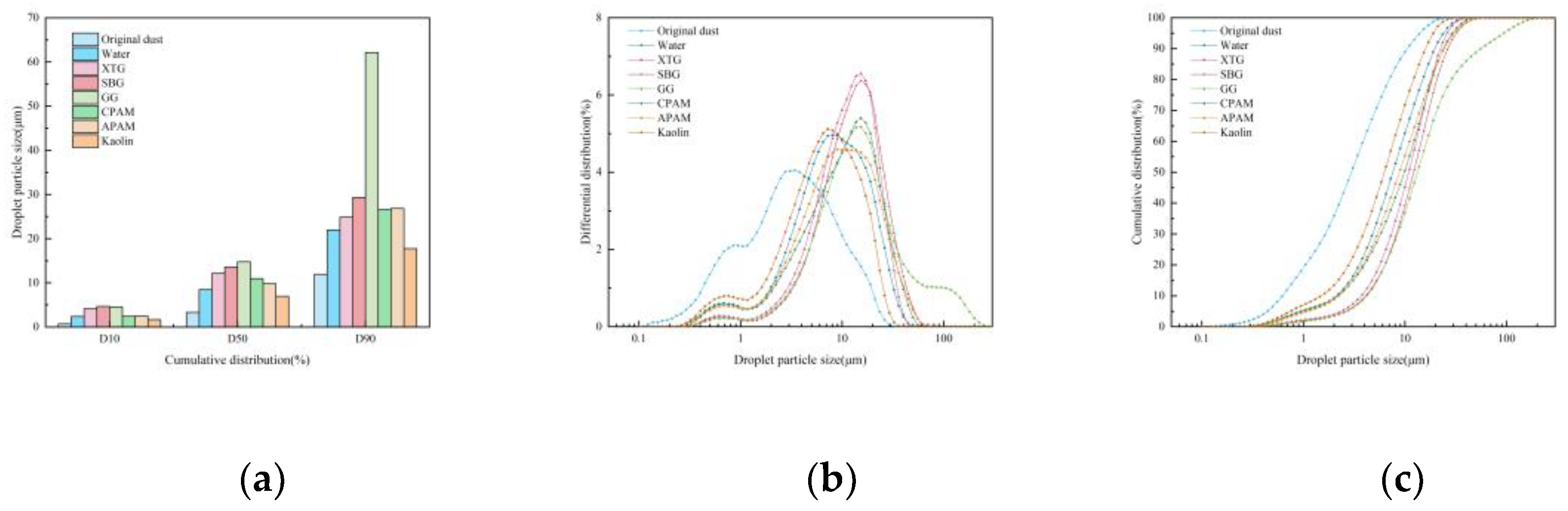
3.1.1. Blast Furnace Dust Particle Size Analysis
3.1.2. Blast Furnace Dust Morphology Analysis
3.2. Influence of Nozzle Type and Agglomeration Solution on Spray Performance
3.2.1. Influence of Nozzle Type
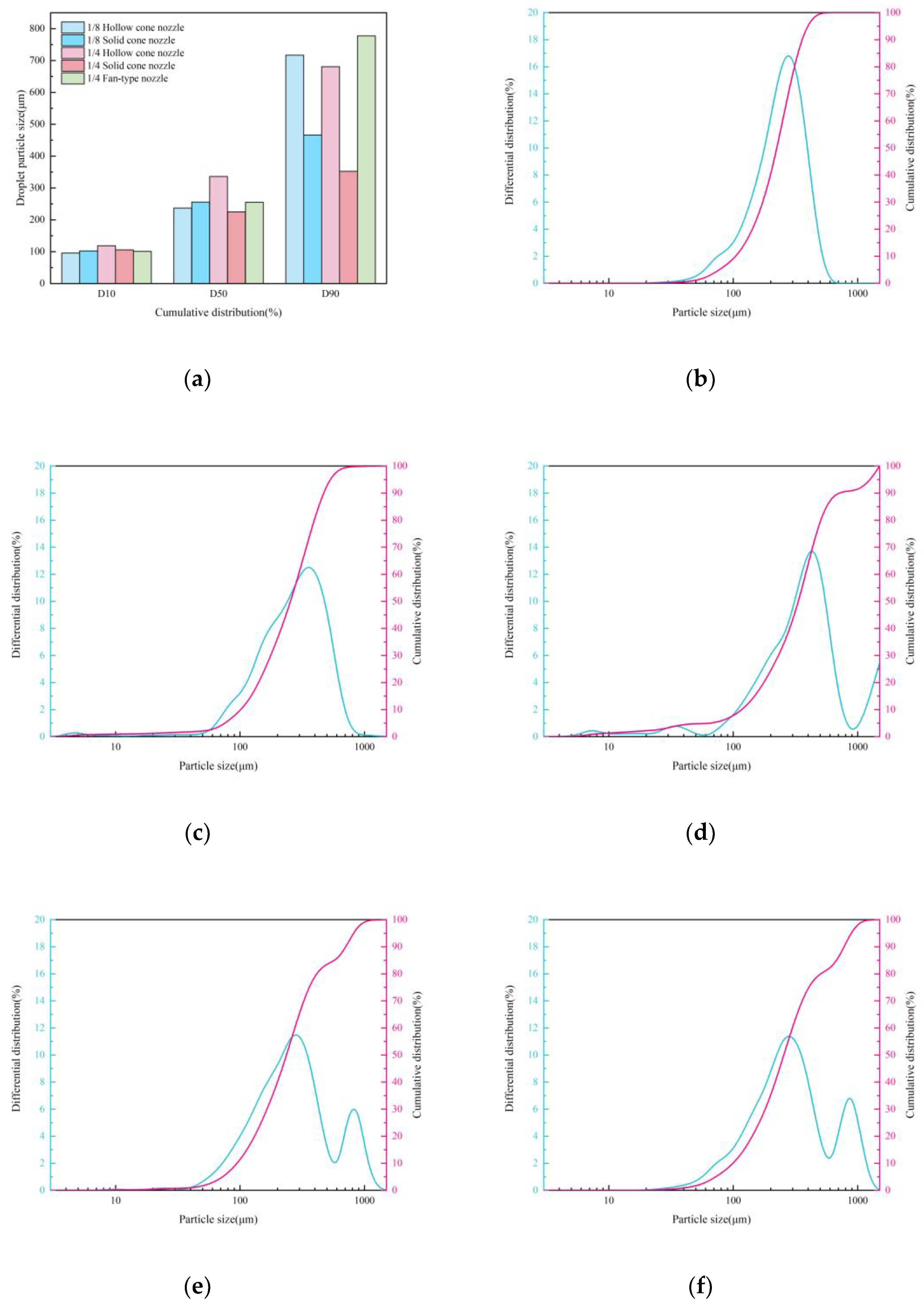
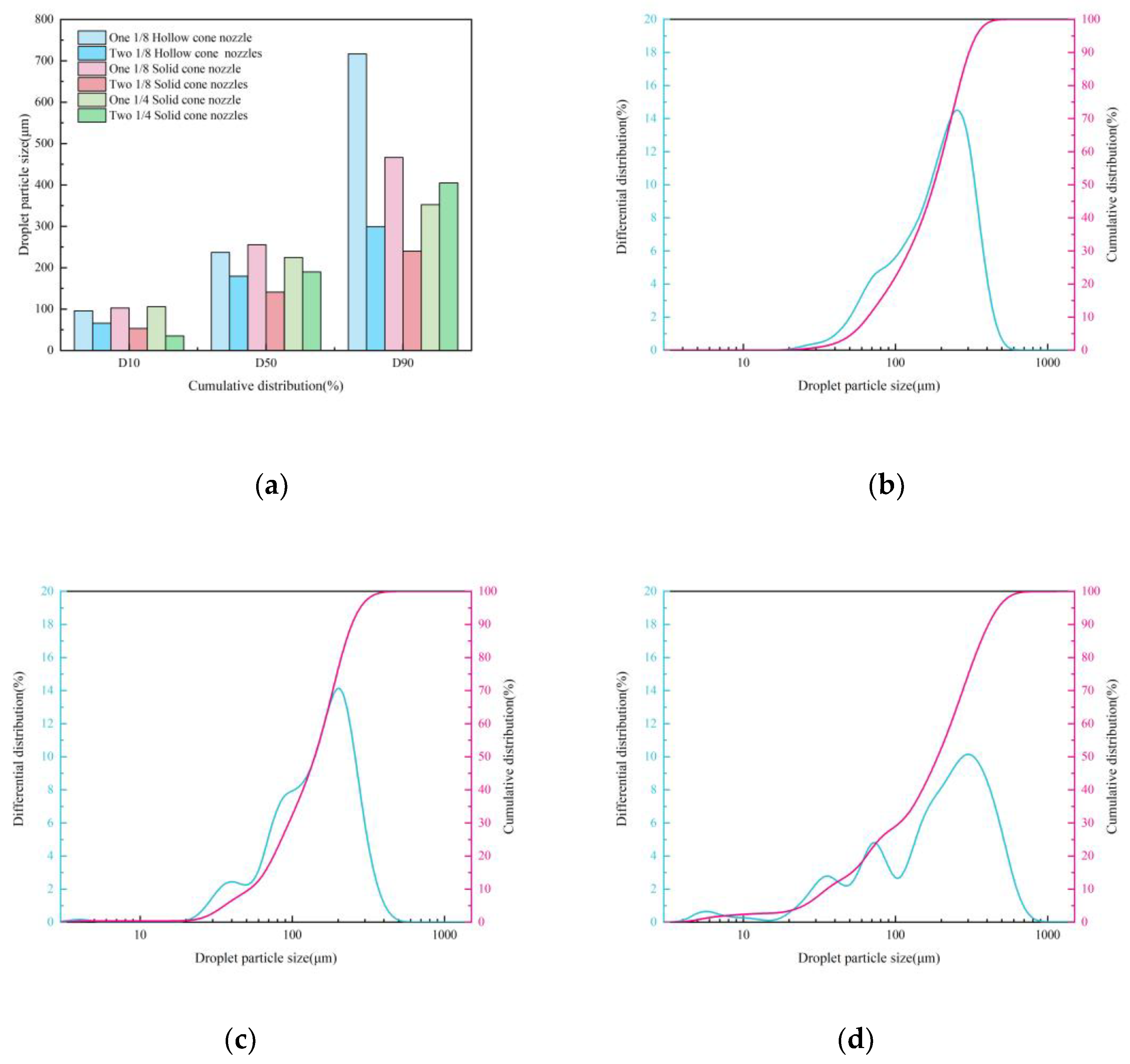
Effect of Nozzle Type on Droplets Size
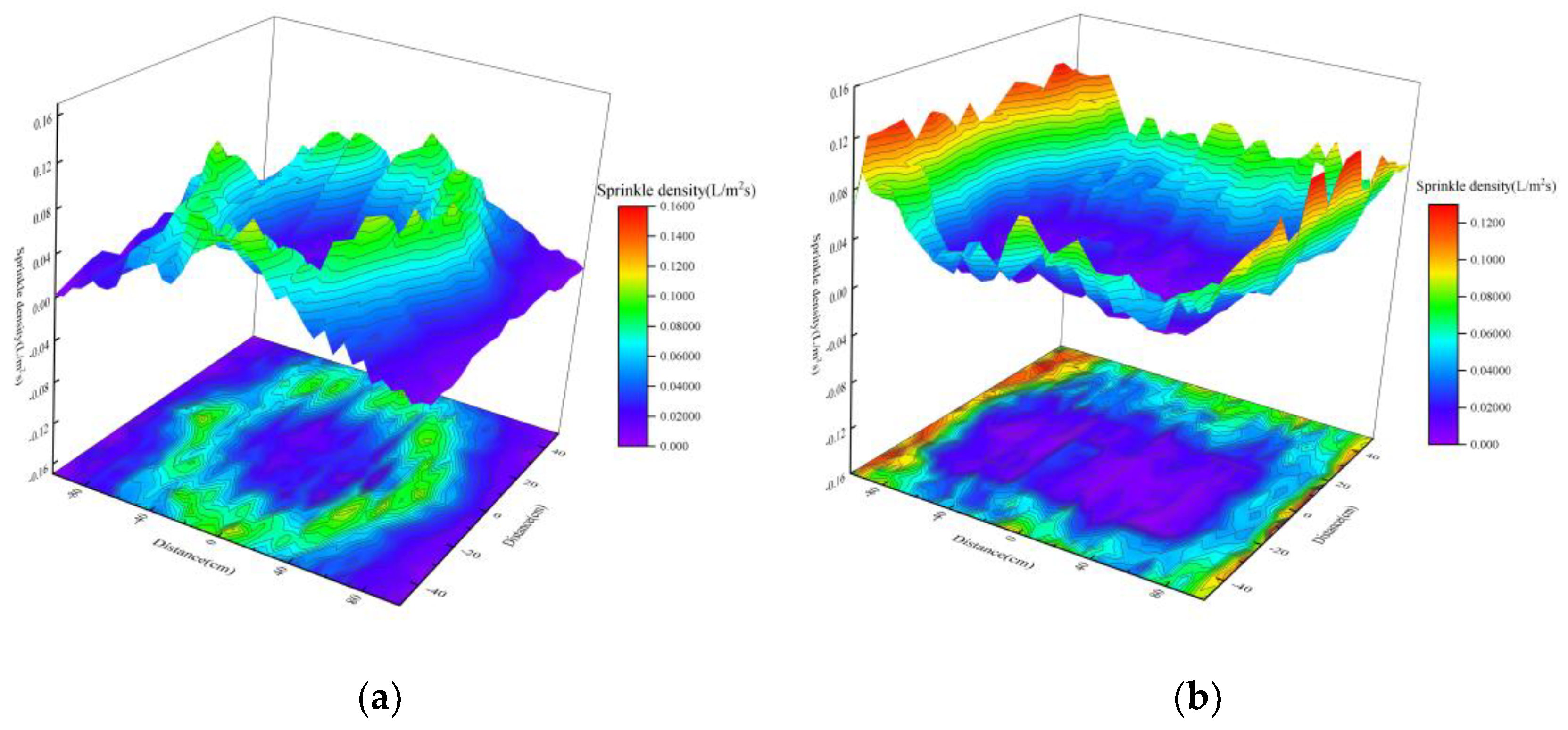
Effect of Nozzle Type on Spatial Distribution of Droplets

3.2.2. Effect of Nozzle Overlap on Atomization Performance
Effect of Nozzle Overlap on Droplets Size
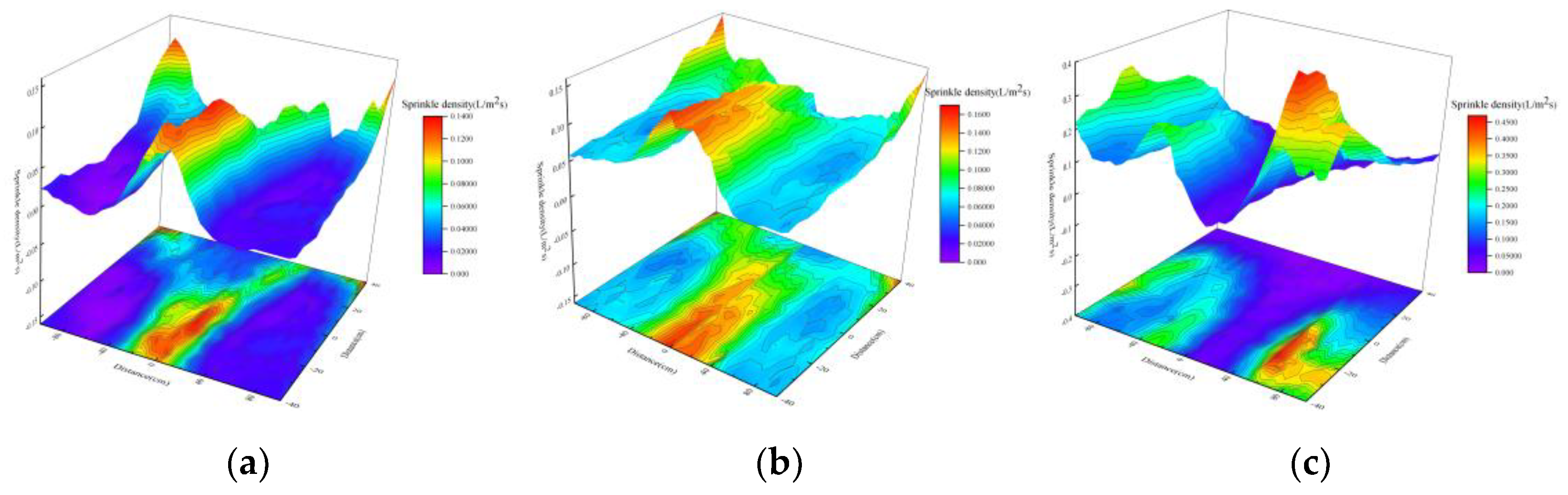
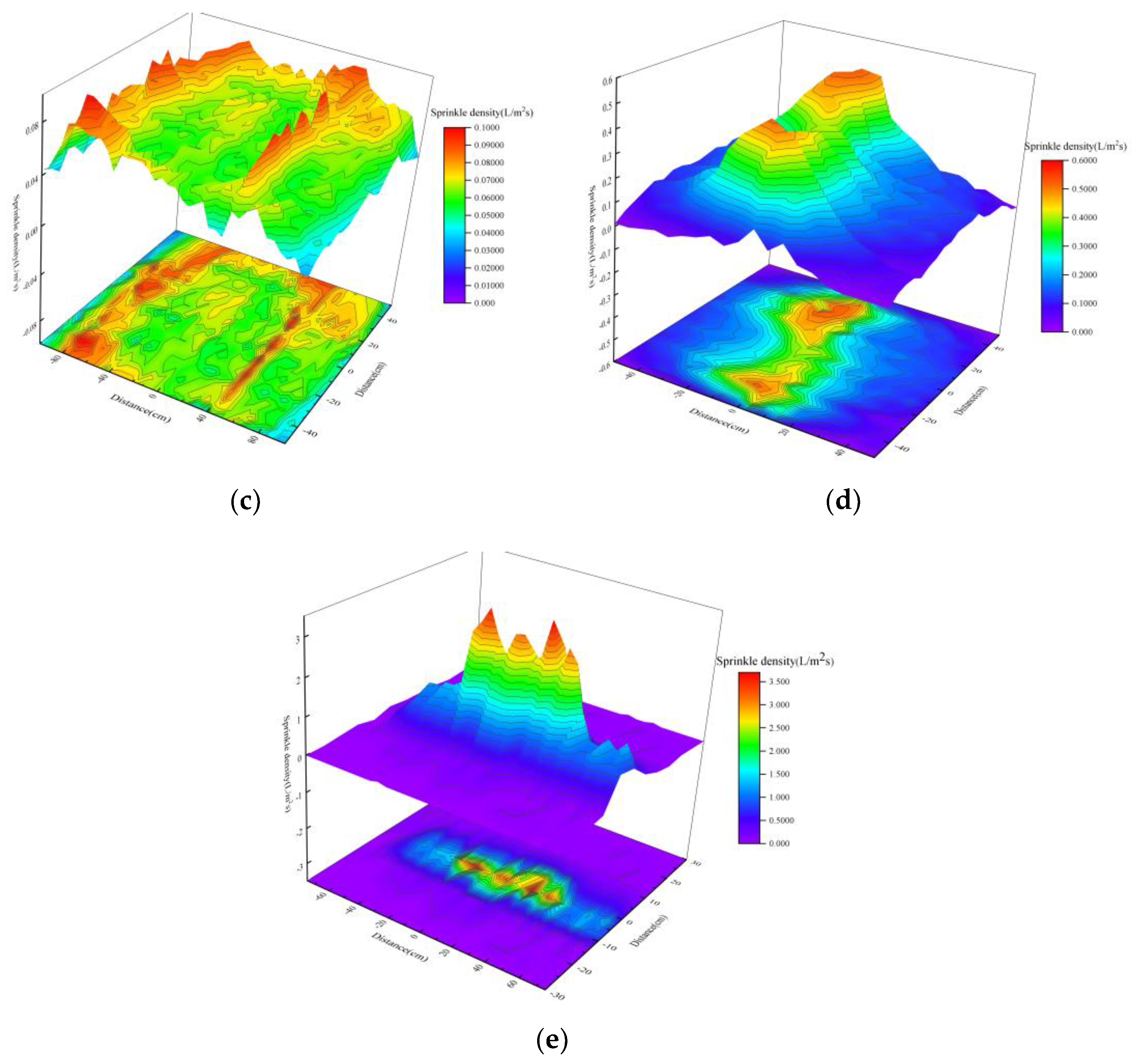
Effect of Nozzle Overlap on Spatial Distribution of Droplets


3.2.3. Effect of Chemical Agglomeration Solution
Effect of Chemical Agglomeration Solution on Droplets Size
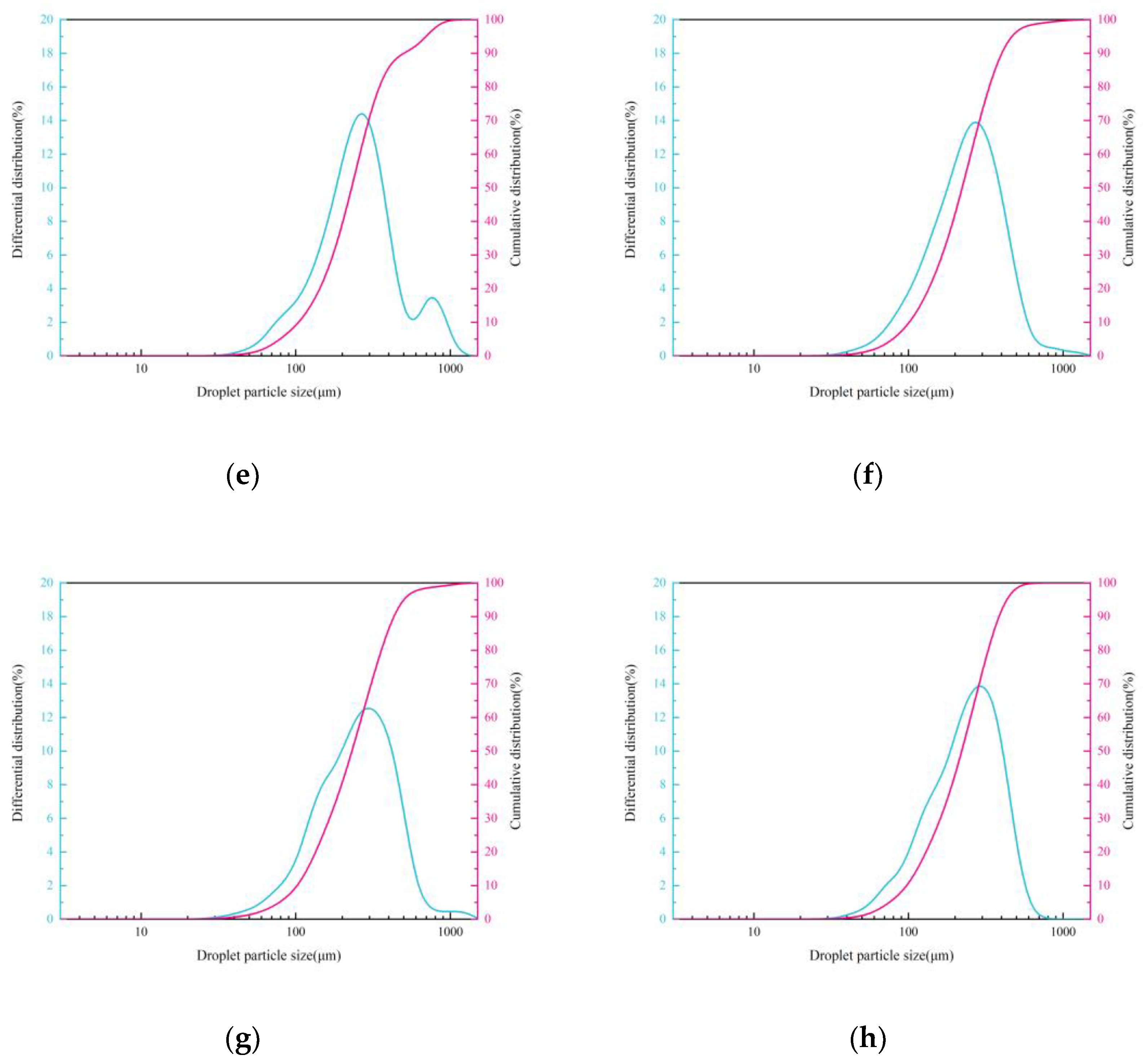
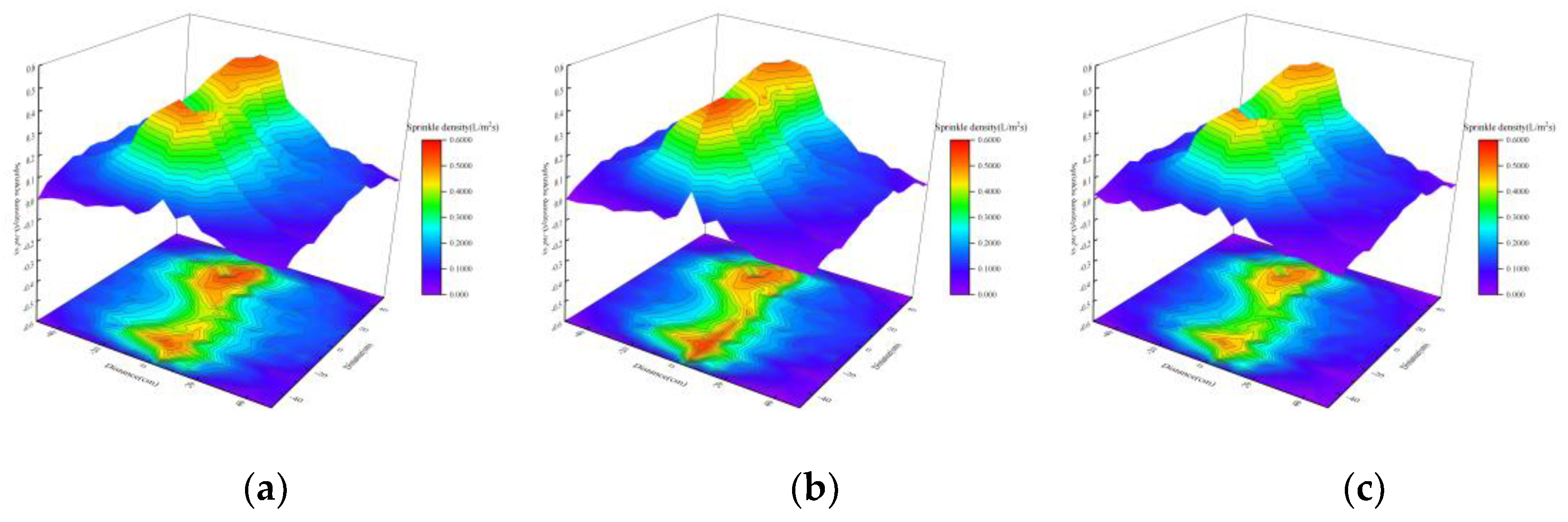
Effect of Chemical Agglomeration Solution on Spatial Distribution of Droplets
3.3. Influence of Chemical Agglomerants and Atomization Performance on Dust Agglomeration
3.3.1. Influence of Chemical Agglomerants
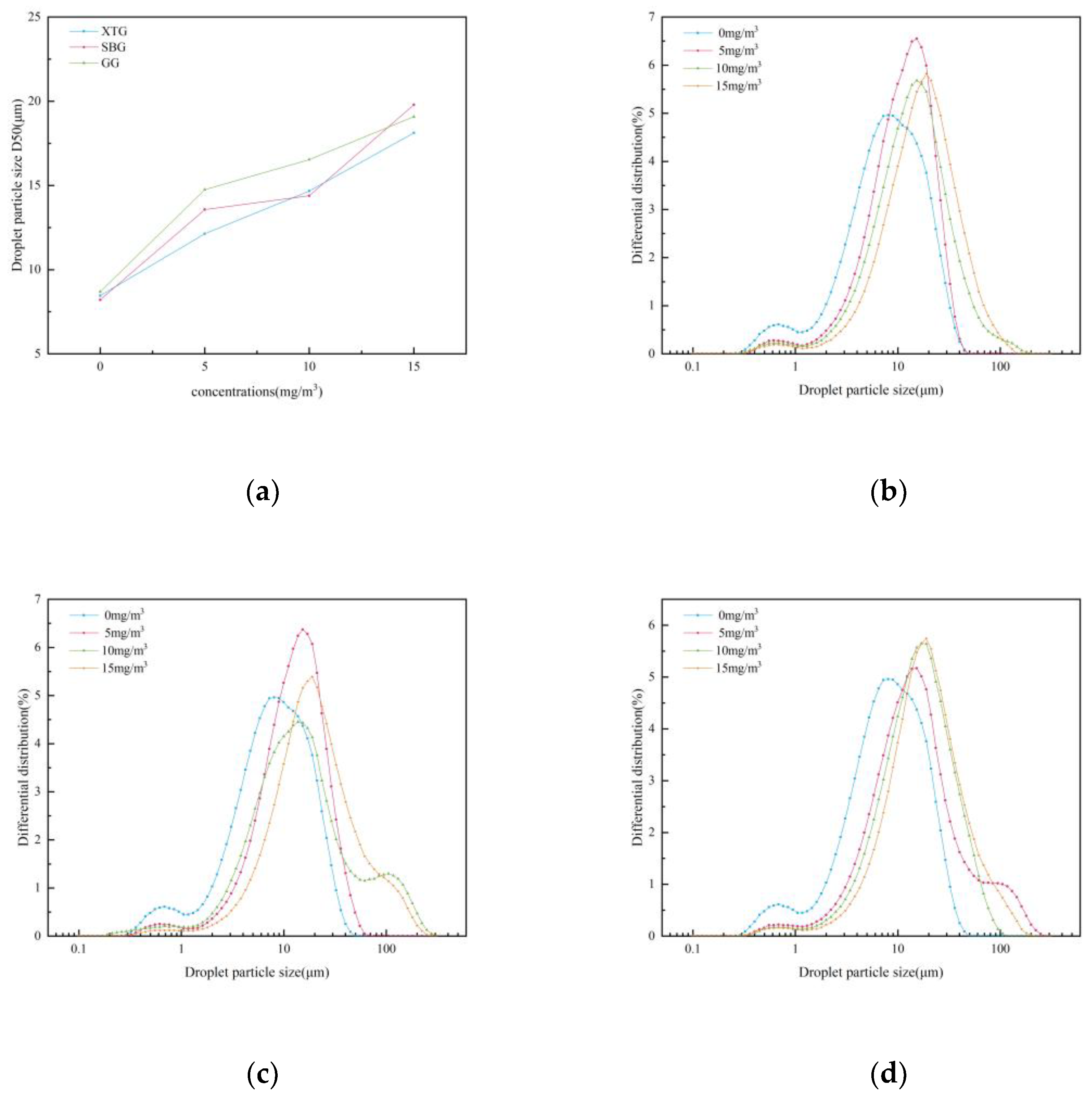
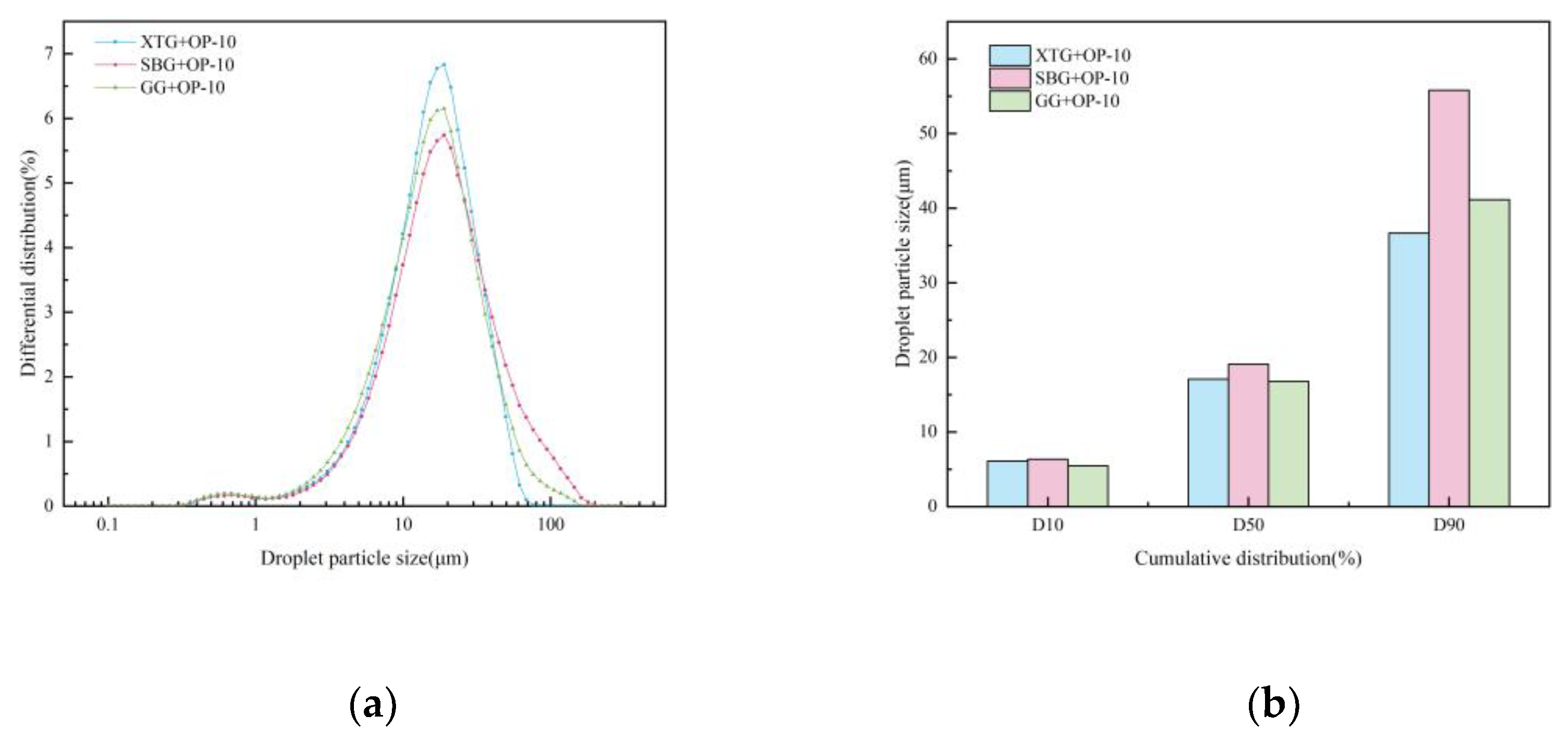
Influence of Chemical Agglomerant Type
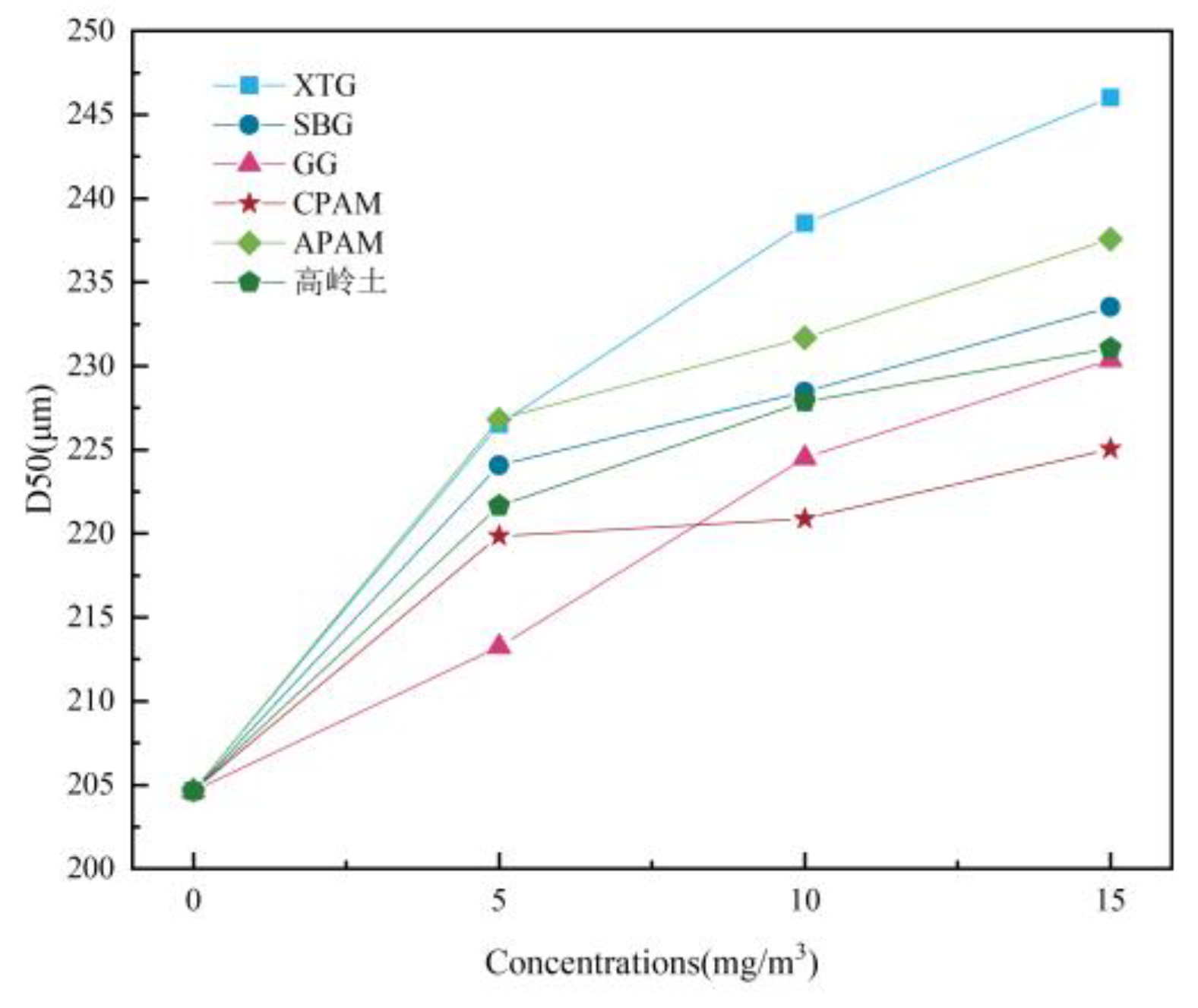
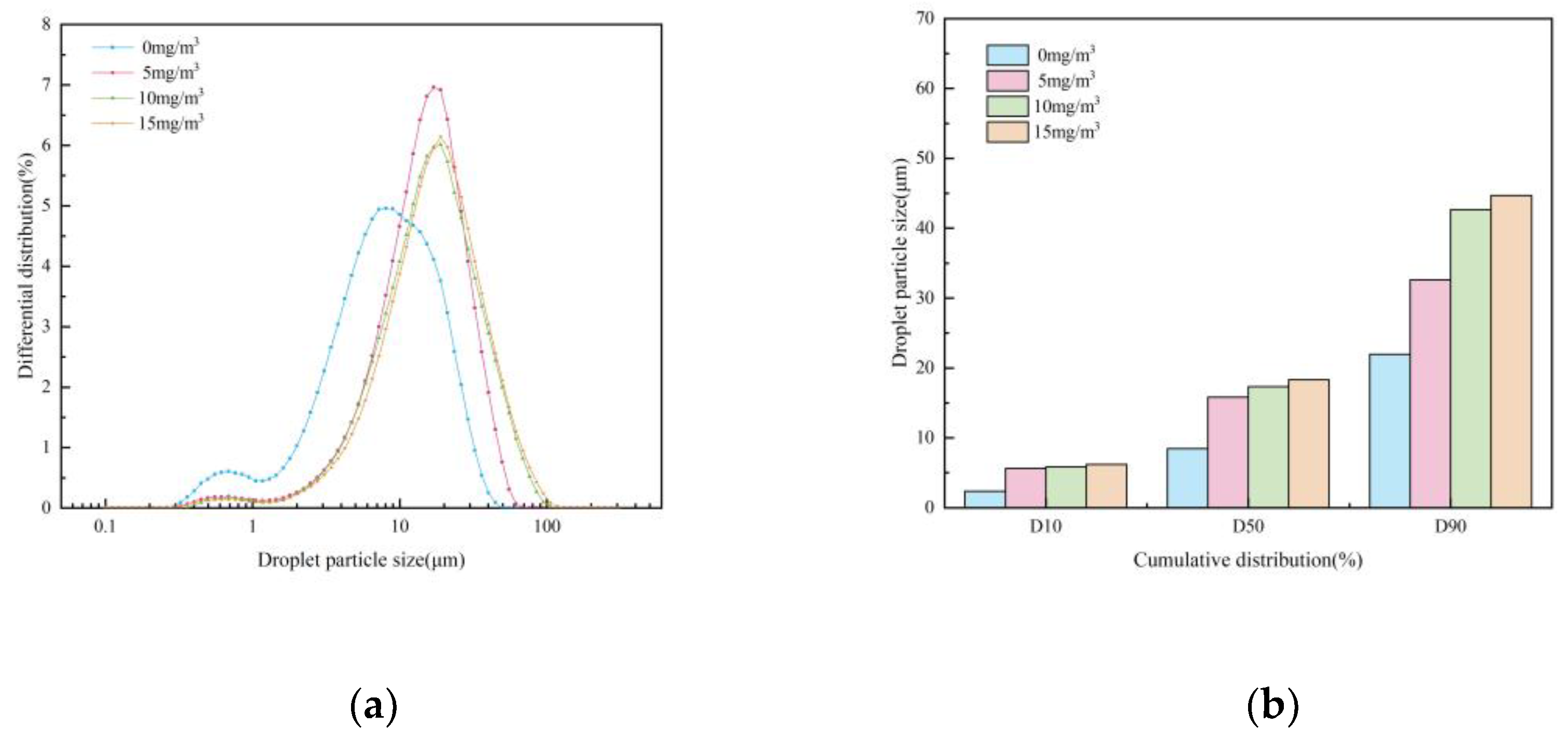
Influence of Chemical Agglomerant Concentration
3.3.2. Influence of Surfactants
Influence of Surfactant Type
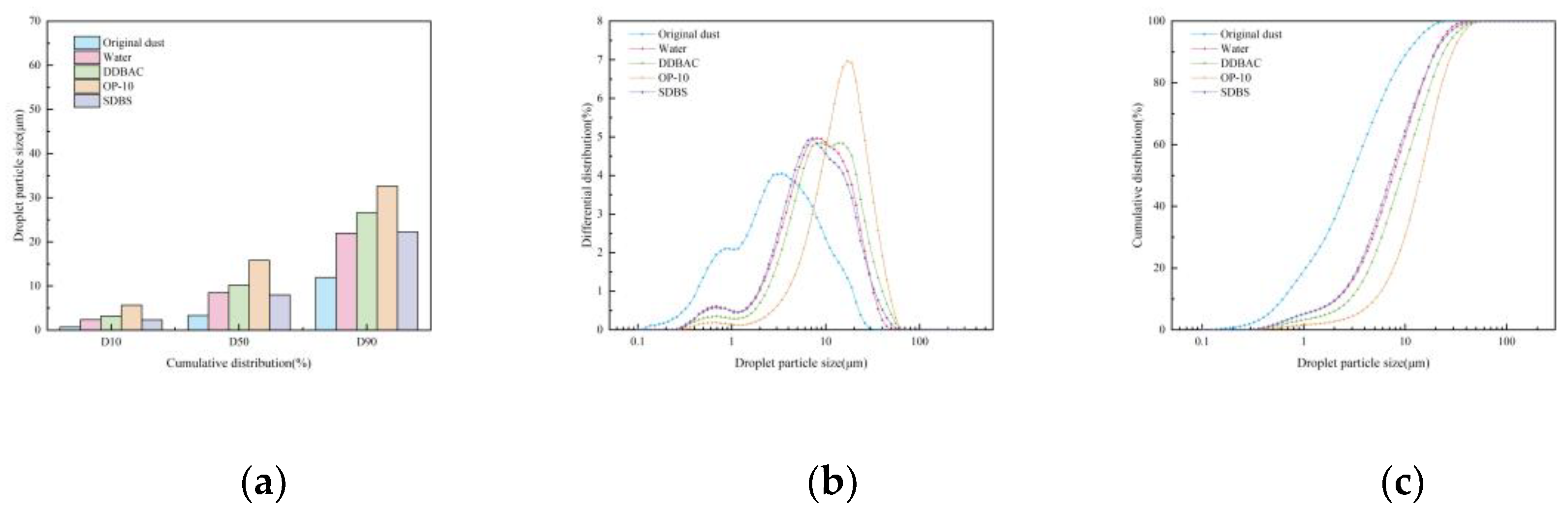
Influence of Surfactant Concentration
Influence of Synergistic Effects Between Agglomerants and Surfactants
3.3.3. Influence of Spray Performance
Effect of Nozzle Type
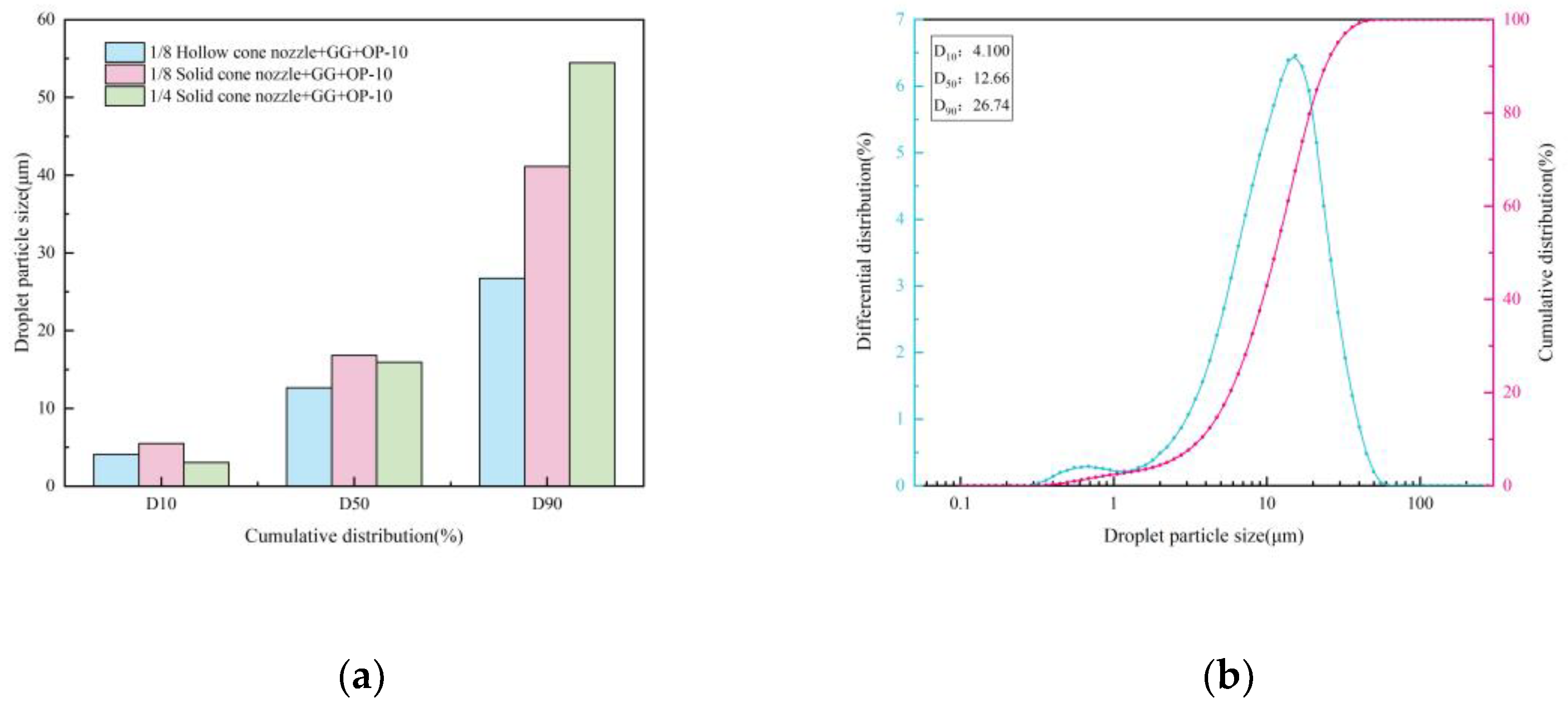
Effect of Synergistic Interaction Between Spray Liquid and Atomization on Dust Agglomeration
3.4. Analysis of Dust Collection Efficiency
3.4.1. Effect of Chemical Agglomerants
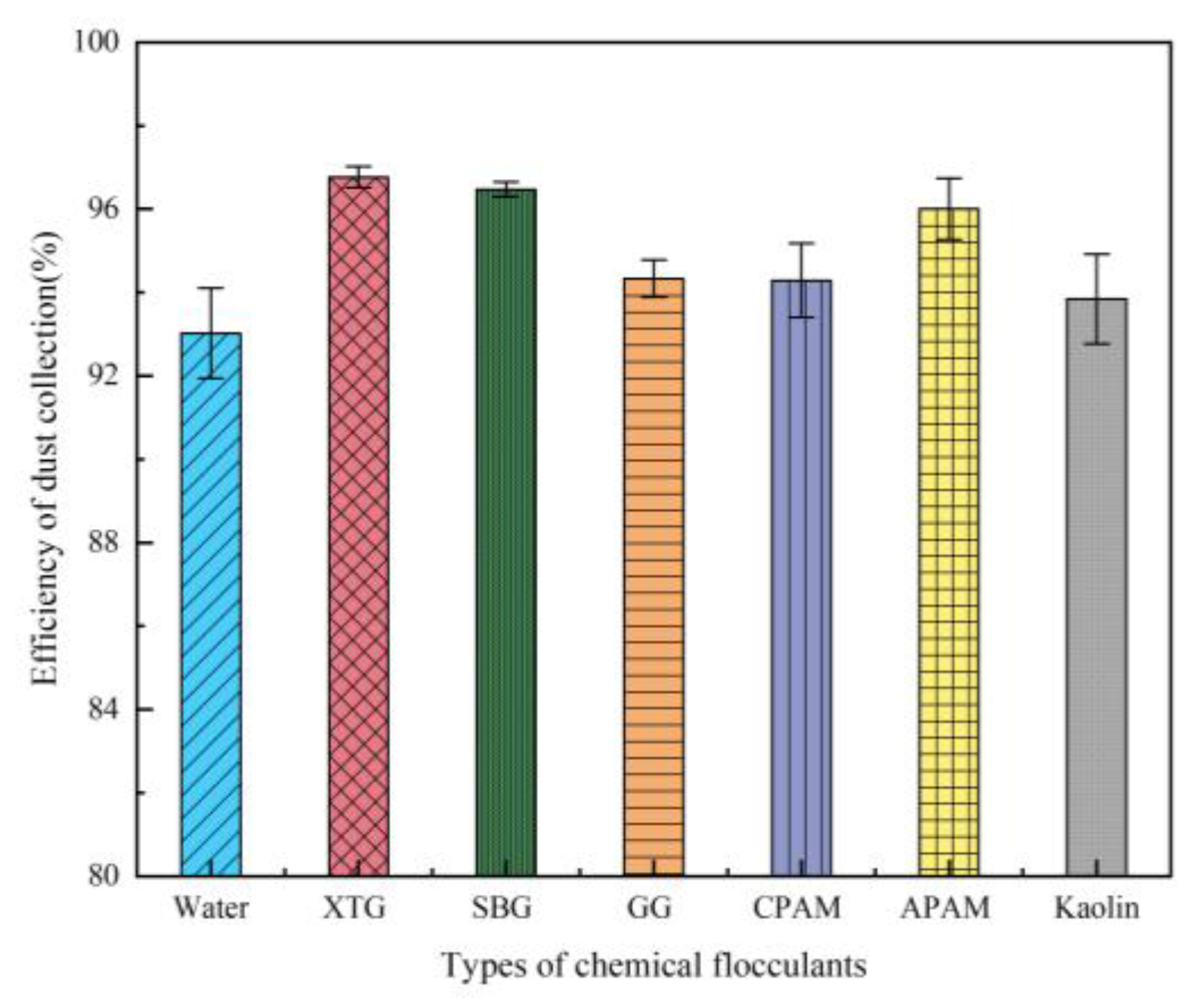
Effect of Chemical Agglomerant Type
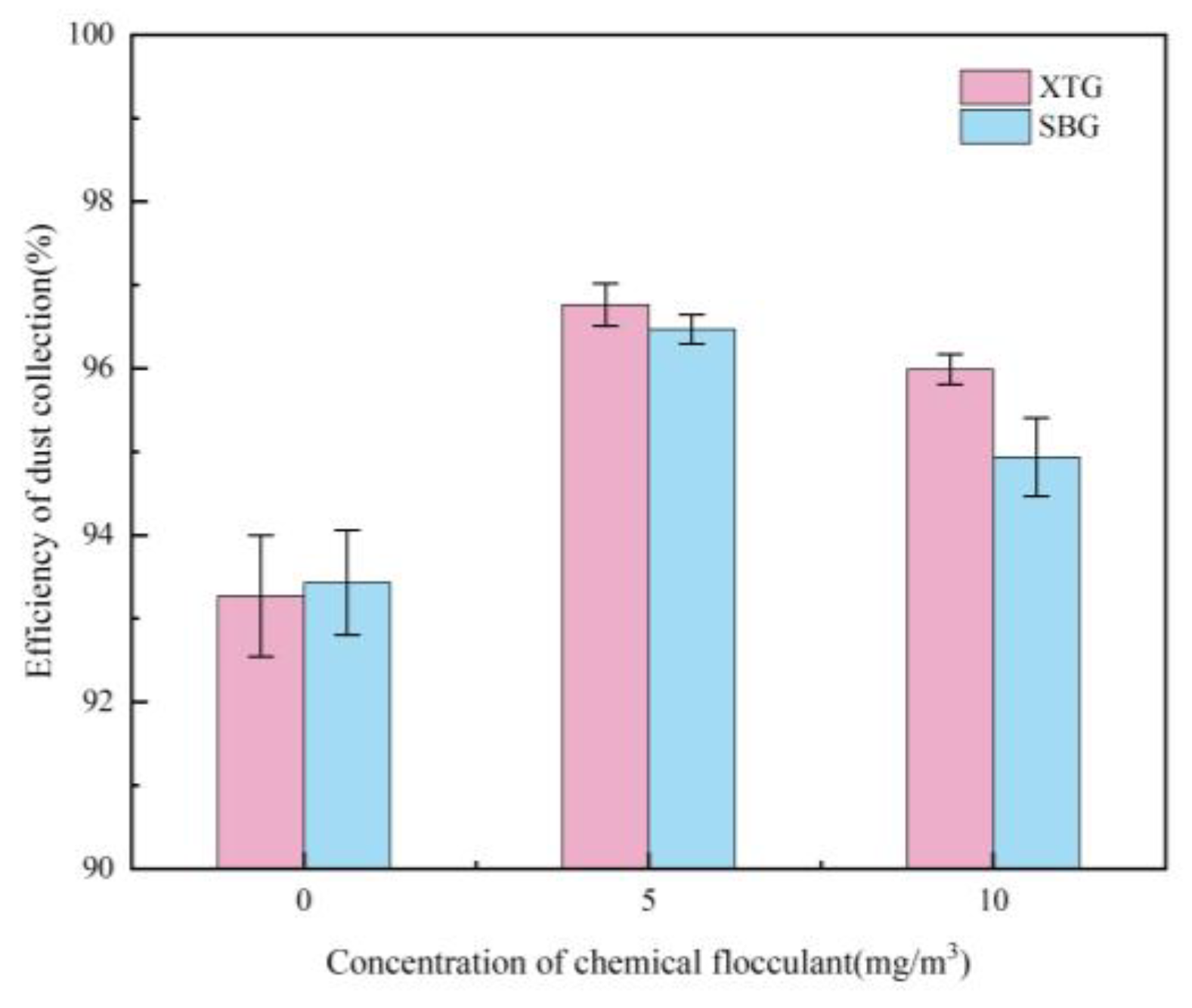
Effect of Chemical Agglomerant Concentration
3.4.2. Effect of Surfactants
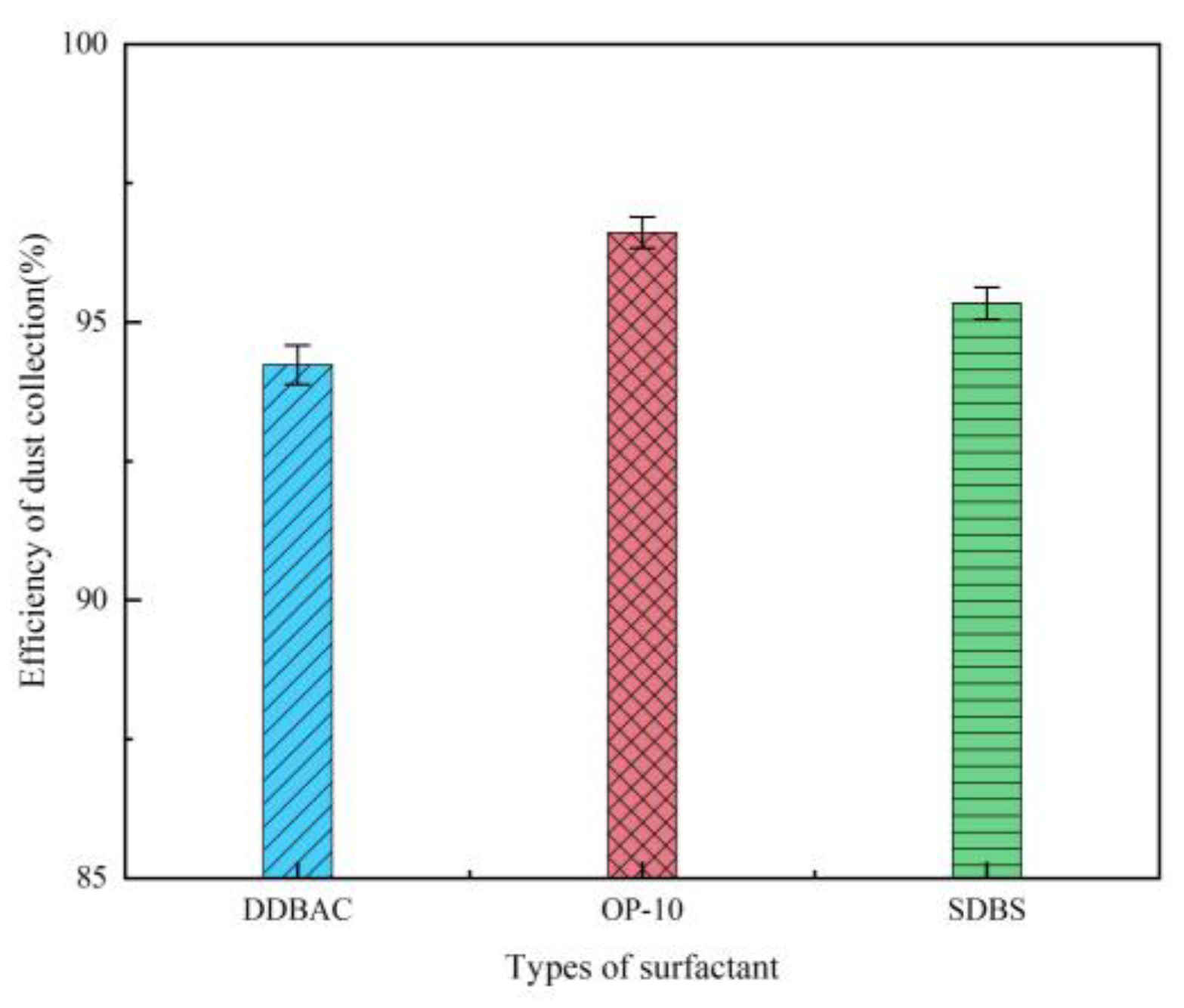
Effect of Surfactant Type
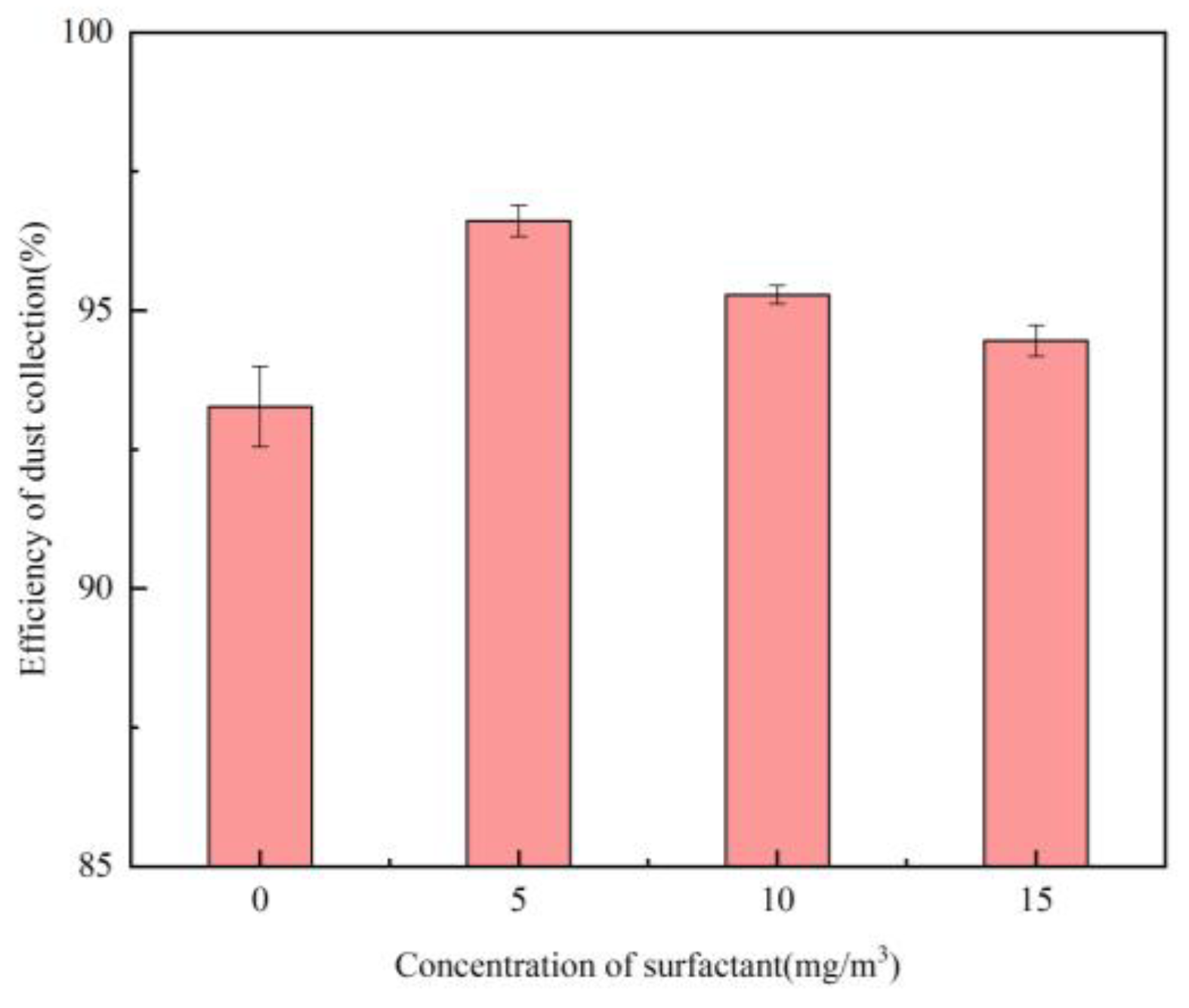
Effect of Surfactant Concentration
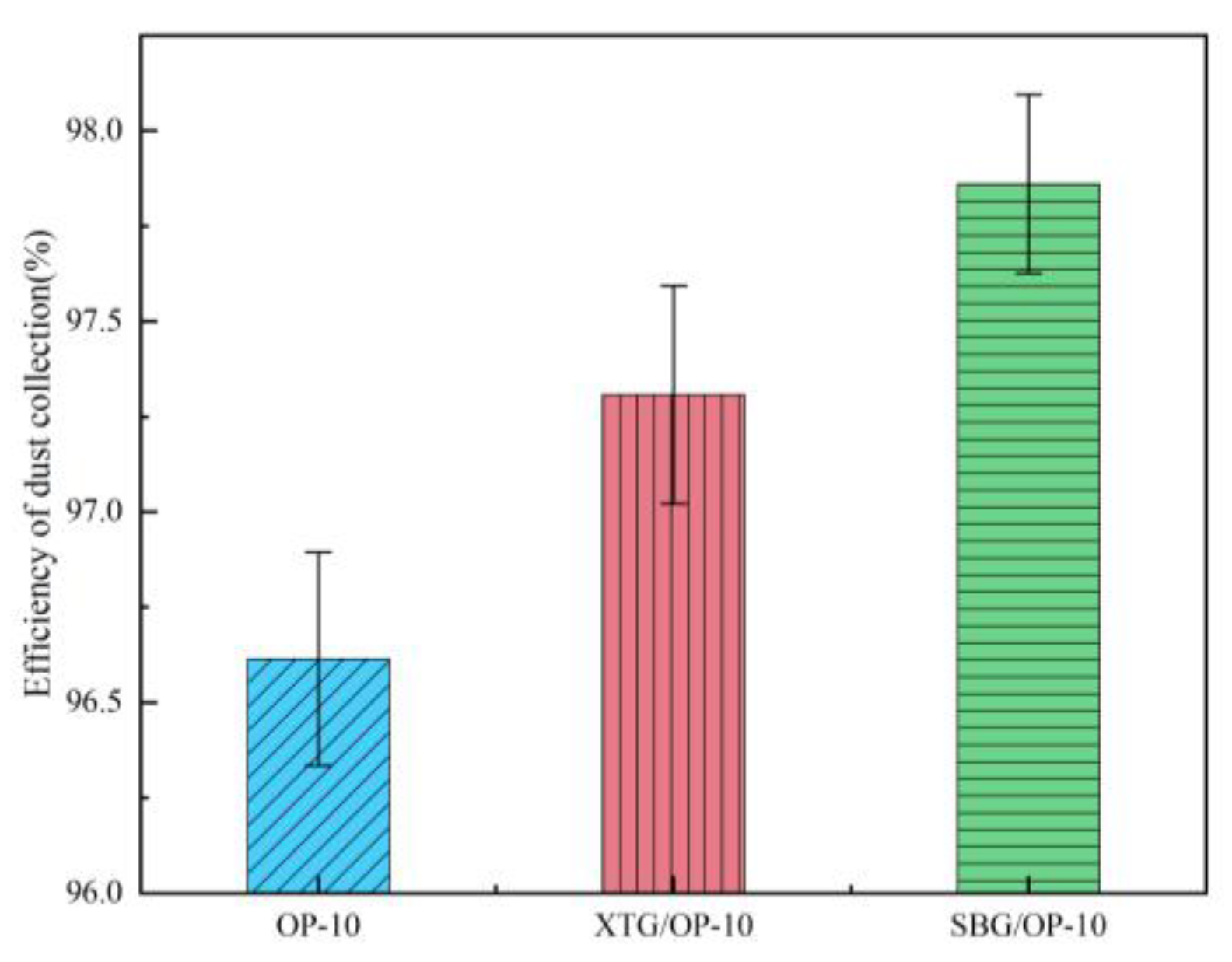
Effect of Synergistic Action Between Agglomerants and Surfactants
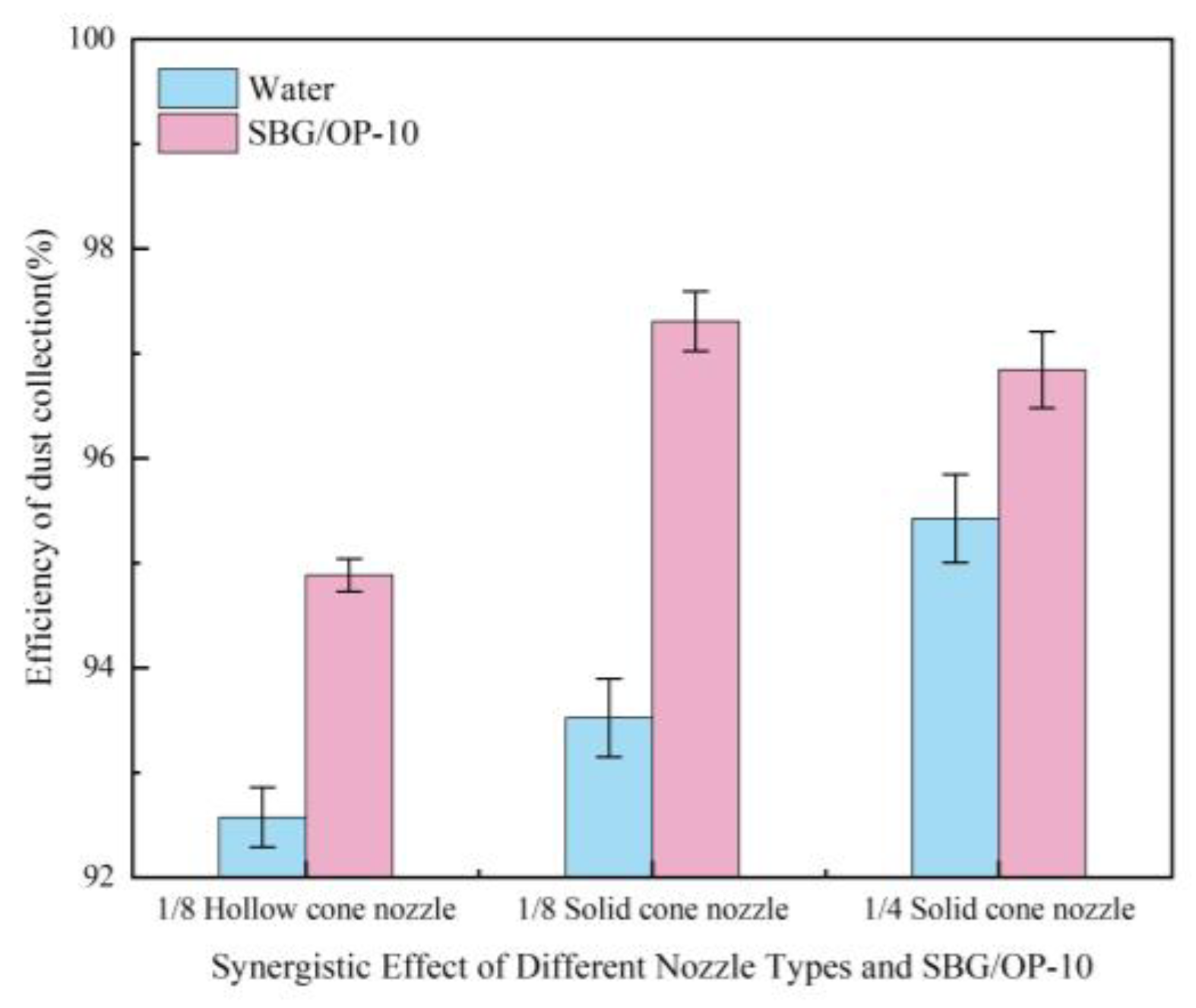
3.4.3. Effect of Spray Performance on Dust Collection Efficiency
4. Conclusions
- Under single-nozzle spraying conditions at a spray pressure of 0.5 MPa, the smallest droplets size was produced by the 1/4 solid cone nozzle, while the most uniform spatial droplets distribution was achieved with the 1/8 solid cone nozzle. After adding chemical agglomerants, among the six agglomerants tested, SBG resulted in the largest increase in droplets size, whereas GG led to the smallest increase. Under twin-nozzle overlapping conditions, droplets sizes for all three twin-nozzle configurations were smaller than those of single-nozzle spraying. The 1/8 solid cone twin-nozzle produced the finest droplets, while the 1/4 solid cone twin-nozzle exhibited relatively uniform spatial distribution.
- When six chemical agglomerants were added separately to the spray liquid at a concentration of 5 mg/m³, the agglomeration effectiveness ranked from highest to lowest as follows: GG, SBG, XTG, CPAM, APAM, and kaolin. With three surfactants added at 5 mg/m³, OP-10 showed the best agglomeration effect, followed by DDBAC, while SDBS performed the poorest. As the concentration of agglomerants or surfactants increased, the enhancement in agglomeration gradually weakened. The most effective combination was the synergy of SBG and OP-10, which yielded a median particle size of 19.08 μm.
- At the chemical agglomerant concentration of 5 mg/m³, XTG achieved the highest collection efficiency of 96.76%, followed by SBG and APAM, while GG, CPAM, and kaolin showed the lowest efficiencies. Among the three surfactants concentration of 5 mg/m³, OP-10 delivered the highest collection efficiency of 96.61%. Using the 1/4 solid cone nozzle alone resulted in a maximum efficiency of 95.43%. The highest dust collection efficiency was 97.86% under the combined conditions of adding 5 mg/m³ SBG/OP-10 and using 1/8 solid cone nozzle for atomization.
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Nomenclature
| d | Particle diameter |
| D10 | Diameter when the cumulative distribution of dust is 10% |
| d50 | Diameter when the cumulative distribution of dust is 50% |
| D90 | Diameter when the cumulative distribution of dust is 90% |
References
- China Energy Big Data Report (2025) [R]. China Energy Research Institute, 2025.
- Liang T, Ma S P. Brief Analysis of Nozzle Design for Spray Dust Suppression [J]. Mechanical Management and Development, 2003, (01): 13-14.
- Ma S P, Kou Z M. Design Study of Pressure Atomization Nozzle for Spray Dust Suppression [J]. Mining Machinery, 2006, (01): 67-68+5.
- Cheng W M, Zhou G, Zuo Q M, et al. Experimental Study on the Relationship between Nozzle Spray Pressure and Atomization Particle Size [J]. Journal of China Coal Society, 2010, 35(08): 1308-1313.
- Chen Y X. Spray Dust Suppression Technology Using Tapered Hollow Nozzles at Belt Conveyor Transfer Points [J]. Safety in Coal Mines, 2012, 43(04): 31-33.
- Ding Z J, Wu Y H, Xiao L C. Atomization Characteristics of Water Spray in Electrostatic Precipitators and Its Impact on Dust Removal Performance [J]. Environmental Engineering, 2013, 31(S1): 354-356.Vallet A, Tinet C. Characteristics of droplets from single and twin jet air induction nozzles: A preliminary investigation[J]. Crop Protection, 2013,4863-68.
- Chen C C, Li S, Wu X Y, et al. Analysis of droplets size uniformity and selection of spray parameters based on the biological optimum particle size theory[J]. Environmental research, 2021,204(Pt B):112076-112076. [CrossRef]
- Yuan F X, Gu C C, Yi K C, et al. Atomization Characteristics of a Hollow Cone Nozzle for Air-Assisted Variable-Rate Spraying[J]. Agriculture, 2023,13(10). [CrossRef]
- Narendra D, Varun K, Sivakumar D. High-energy droplets collisions in multi-interacting hollow cone sprays[J]. International Journal of Multiphase Flow. Volume 195, Issue. 2026. PP 105525-105525. [CrossRef]
- Durham M D, Schlager R J, Ebner T G, et al. Method and apparatus for decreased undesired particle emissions in gas streams: US, US5893943 A[P]. 1999.
- Baldrey K E. Advanced flue gas conditioning as a retrofit upgrade to enhance PM collection from coal-fired electric utility boilers [J]. Office of Scientific & Technical Information Technical Reports, 2000.
- Geng J F, Song S J, Bao J J, et al. Promoting removal of fine particulate matter in WFGD system by using wetting agent [J]. CIESC Journal, 2011, 62(04): 1084-1090.Li R, Li C, Zhuang J, et al. Mechanistic Influence of Chemical Agglomeration Agents on Removal of Inhalable Particles from Coal Combustion[J]. ACS omega, 2020,5(40): 25906-25912.
- Shan G, Chun X L. Study on Dust Turbulence-Chemical Agglomeration for Electrostatic Precipitation Technology[J]. E3S Web of Conferences, 2021,245: 3011. [CrossRef]
- Yang G Z, Zhao Y C, Xiong Z, et al. Study on Chemical Agglomeration Enhanced Dust Removal and Synergistic Zero Discharge of Desulfurization Wastewater in a 300 MW Coal-fired Power Plant [J]. Proceedings of the CSEE, 2021, 41(15): 5274-5283.
- Zhang J F. Promotion Effect of Chemical Agglomeration Technology on the Removal of Fine Particulate Matter and SO₃ Acid Mist by Electrostatic Precipitation [D]. Nanchang Hangkong University, 2022.
- Li T Y, Cui X Z, Ma F X, et al. Experimental Study on Enhancing Fine Particulate Matter Removal Based on Chemical Agglomeration Technology [J]. Coal Conversion: 1-12.
- Qin J J, Liu D, Gao X, et al. Study on Rheological Properties of Guar Gum Clean Gel [J]. Journal of Xiangtan University (Natural Science Edition), 2023, 45(03): 66-74.
- Zhu X F, Su X X, Yang X L, et al. Preparation and Application of Sepiolite/Xanthan Gum Composite Flocculant [J]. Applied Chemical Industry, 2009, 38(09): 1241-1244.
- Du X Y, Gao Z L, He L L. Quantifying the effect of non-ionic surfactant alkylphenol ethoxylates on the persistence of thiabendazole on fresh produce surface[J]. Journal of the science of food and agriculture. Volume 104, Issue 5. 2023. PP 2630-2640. [CrossRef]
- Huang H, Zhang L, Ren T. Analysis of Surface Properties of Polyalkylphenol Polyoxyethylene Ether Surfactants [J]. World Pesticides, 2014, 36(02): 50-54.


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




