1. Introduction
Carbon quantum dots (CQDs) have emerged as an important class of zero-dimensional carbon-based nanomaterials owing to their tunable photoluminescence, excellent chemical stability, and low toxicity [
1,
2,
3]. Their distinctive optical characteristics, including size-dependent emission and surface-state-mediated fluorescence, have enabled a wide range of applications in bioimaging, chemical sensing, photocatalysis, and optoelectronic devices [
4,
5,
6,
7]. However, most reported CQDs are hydrophilic in nature [
8,
9,
10], which limits their compatibility with nonpolar environments and restricts their integration into hydrophobic matrices or organic optoelectronic systems. In addition, extending the emission of CQDs toward the low-energy visible and red spectral regions remains a significant challenge. Typically, pristine CQDs exhibit dominant blue emission arising from surface defect states [
11,
12], while red or white-light emission is relatively rare and usually requires complex surface passivation [
13,
14,
15] or hybridization strategies [
16,
17,
18].
To overcome these limitations, hybrid nanostructures that combine CQDs with luminescent organic dyes have recently attracted considerable attention. In such systems, CQDs can function as broadband surface-state emitters or energy donors, whereas organic dyes contribute strong and well-defined molecular fluorescence in the visible region. Among the available dyes, Rhodamine B (RhB) has emerged as a promising candidate due to its high molar absorption coefficient, excellent photostability, and intense emission in the orange–red region of the spectrum [
19,
20,
21].
The spectral overlap between the emission of CQDs and the absorption band of RhB facilitates efficient interfacial photophysical processes such as Förster resonance energy transfer (FRET) or photoinduced charge transfer, which can broaden the overall emission bandwidth and improve light-harvesting efficiency [
22,
23,
24]. Through these interactions, the excited-state energy generated within CQDs can be partially transferred to RhB molecules, thereby modifying exciton recombination pathways and extending the emission toward longer wavelengths. As a result, CQD–RhB hybrid systems can exhibit dual-emission behavior and broadened photoluminescence spectra, which are highly desirable for achieving tunable visible-light emission and potentially near white-light generation under ultraviolet excitation [
11,
12].
Although organic dyes such as RhB provide strong visible-light emission [
25,
26], the direct incorporation of dye molecules into CQD systems to form stable and reproducible composites without fluorescence quenching remains insufficiently explored. In particular, systematic investigations on how the concentration of RhB influences the photophysical properties, quantum yield, and emission tunability of hydrophobic CQDs are still limited.
In this work, these challenges are addressed by synthesizing hydrophobic carbon quantum dots (hbCQDs) and fabricating hbCQDs/RhB hybrid composites with controlled dye loading. The study aims to elucidate the structural and optical interactions between CQDs and RhB, evaluate the contribution of molecular-state emission to surface-state fluorescence, and identify optimized compositions that enhance quantum yield and broaden visible-light emission. These findings provide valuable insights for designing CQD-based hybrid luminescent materials and advance their potential applications in white-light optoelectronic devices and hydrophobic media-compatible photonic systems.
2. Experimental Section
2.1. Materials
Melamine (MA) (C3N3(NH2)3), dithiosalicylic acid (C14H10O4S2), and Rhodamine B (RhB) (C28H31ClN2O3) were procured from Shanghai Adamas Reagent Co., Ltd. Acetic acid (CH3COOH) was supplied by Guangdong Guanghua Sci-Tech Co., Ltd.. All reagents were of analytical grade and used as received without further purification. Deionized water was produced using a Milli-Q water purification system (Millipore) and was employed consistently throughout all experimental procedures.
2.2. Synthesis of Hydrophobic Powder CQDs
The hydrophobic carbon quantum dots were synthesized via a solvothermal route [
27,
28] following the procedure schematically illustrated in
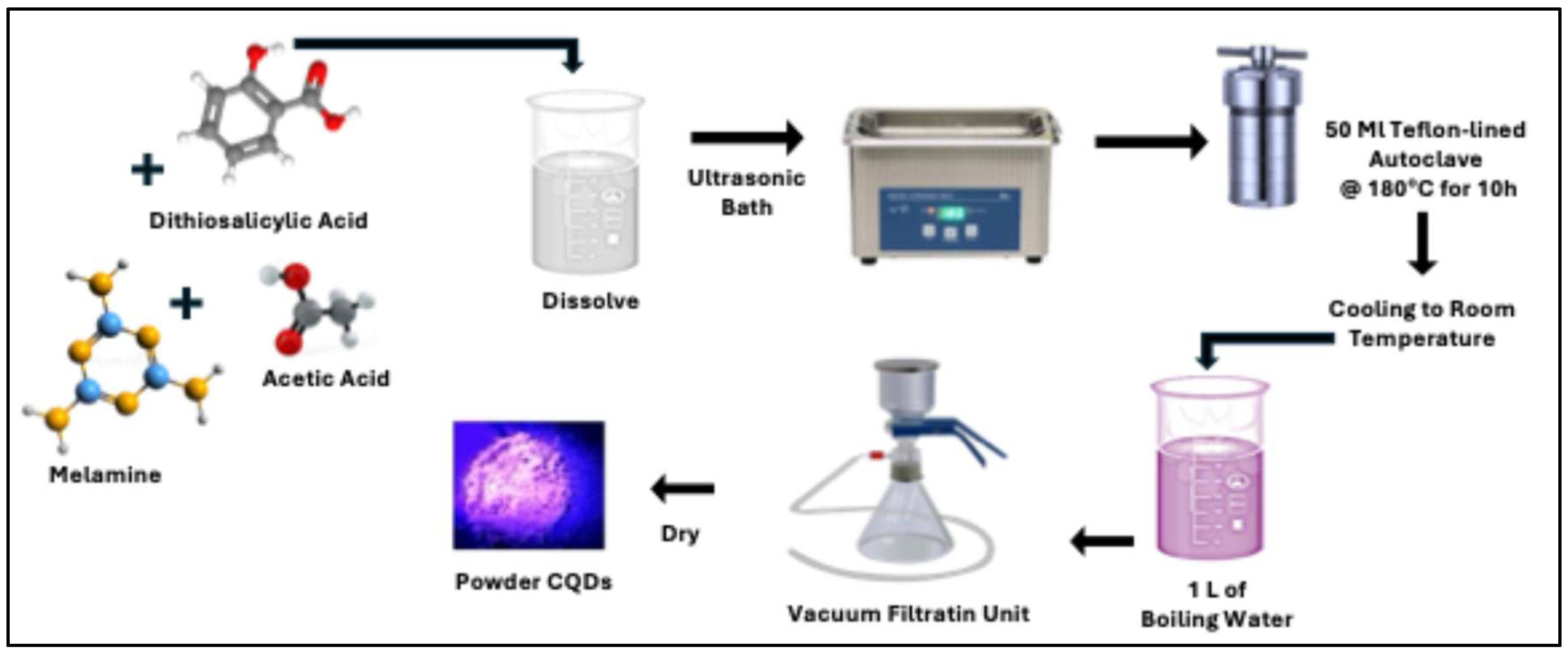
Figure 1. Initially, melamine (MA, 0.00079 mol) and thiosalicylic acid (0.0064 mol) were dissolved in 20 mL of acetic acid under ultrasonic irradiation until a homogeneous and transparent solution was obtained, ensuring complete precursor dispersion and molecular-level mixing. Subsequently, the resulting solution was transferred into a 50 mL Teflon-lined autoclave reactor and heated in an oven at 180 °C for 10 h to promote carbonization and nucleation of CQDs under autogenous pressure. After completion of the reaction and natural cooling to room temperature, the as-prepared CQD solution was rapidly injected into 1 L of boiling deionized water (100 °C). This step induced flocculation of the hydrophobic CQDs, facilitating the removal of unreacted precursors and residual solvent through phase separation. Aggregated CQDs were thereby formed and isolated. The precipitated CQDs were collected by vacuum filtration and thoroughly washed. Finally, the purified CQD powder was dried in an oven at 70 °C until constant weight was achieved, confirming the effectiveness and reproducibility of the water-washing purification protocol.
2.3. Synthesis of Powder CQDs/Rhodamine B Hybrid Composite
To broaden the emission profile of the CQDs toward the low-energy (red) region—an area of considerable interest due to its limited availability and typically low production yield—a CQDs/RhB hybrid system was developed. The hybrid materials were synthesized following the same solvothermal protocol used for the preparation of hydrophobic CQDs, with the incorporation of Rhodamine B (RhB) as a luminescent modifier. Specifically, controlled volumes of RhB (0.2 and 0.4 mL) were introduced into the precursor solution prior to the solvothermal treatment. Two hybrid samples were thus fabricated with different RhB loadings to systematically investigate the influence of dye concentration on the optical properties and emission tunability of the resulting CQDs/RhB nanocomposites.
3. Instrumentation
X-ray diffraction (XRD) analysis was conducted using a Rigaku Ultima IV diffractometer. High-resolution transmission electron microscopy (HR-TEM) characterization was performed on a JEOL ARM-200F operating at 200 kV and equipped with dual Cs aberration correctors. Micrographs were captured using an Orius camera, and subsequent image analysis and data processing were carried out with Gatan software. Fourier-transform infrared (FTIR) spectra were acquired using a PerkinElmer FTIR spectrometer. Samples were mounted on a thin-film holder with the film surface oriented toward the incident IR beam, and spectra were recorded across the 400–4000 cm⁻¹ wavenumber range. X-ray photoelectron spectroscopy (XPS) measurements were performed using an XPS system, and spectral deconvolution and analysis were completed with CasaXPS. For XPS characterization, CQD dispersions were drop-cast onto 10 mm × 10 mm indium tin oxide (ITO) substrates and oven-dried prior to measurement. Optical absorption spectra were obtained using a Jasco V-770 UV–Vis–NIR spectrophotometer. Photoluminescence (PL) measurements were conducted with a Jasco fluorescence spectrometer. The quantum yield (QY) of the CQD samples was determined using a 100 mm integrating sphere coupled to a Jasco ILF-835 system.
4. Results and Discussions
4.1. Structural Characterization
4.1.1. X-Ray Diffraction Analysis (XRD)
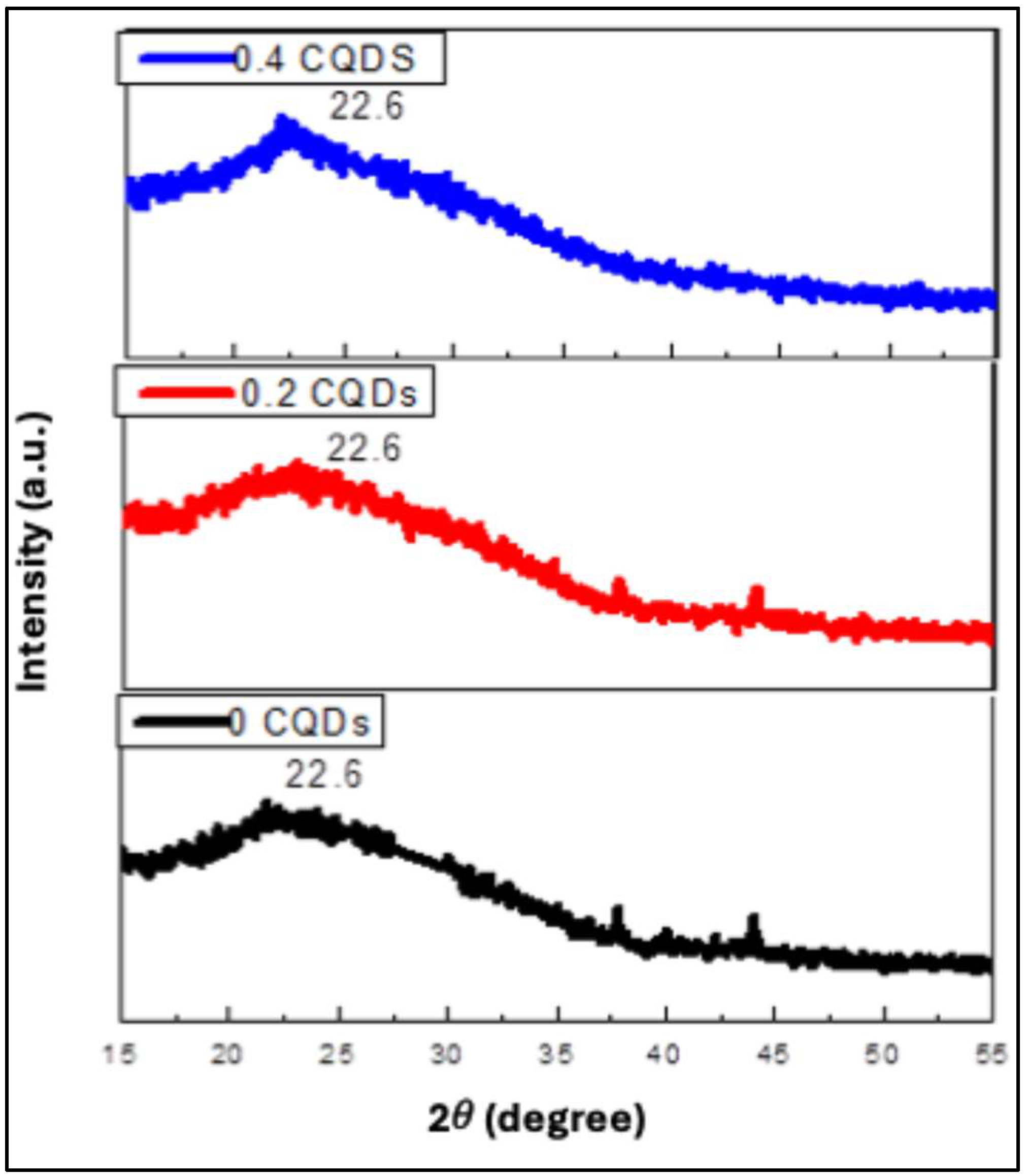
The XRD patterns of pure hydrophobic CQDs (hbCQDs) and hbCQDs/RhB composites are presented in
Figure 2. The diffraction profile of pristine hbCQDs exhibits a broad peak centered at approximately 22.65°, corresponding to an interlayer spacing (d-spacing) of 0.39 nm, which is indexed to the (002) plane of graphitic carbon. This broadened feature indicates a low degree of crystallinity and the presence of turbostratic or amorphous carbon domains. Additionally, a weak diffraction peak is observed around 41°, with a calculated interlayer spacing of 0.21 nm, attributed to the (100) in-plane lattice reflection. These structural features collectively confirm the formation of carbon quantum dots with predominantly disordered graphitic structures. For the hbCQDs/Rhodamine B composites, the characteristic (002) and (100) diffraction peaks are retained at low RhB loading, indicating that the fundamental carbon framework remains largely unaffected. However, at higher RhB content, the (100) reflection becomes indistinguishable, suggesting increased structural disorder or partial surface coverage by RhB molecules, which may disrupt the in-plane graphitic ordering of the CQDs.
4.1.2. Transmission Electron Microscopy Analysis (TEM)
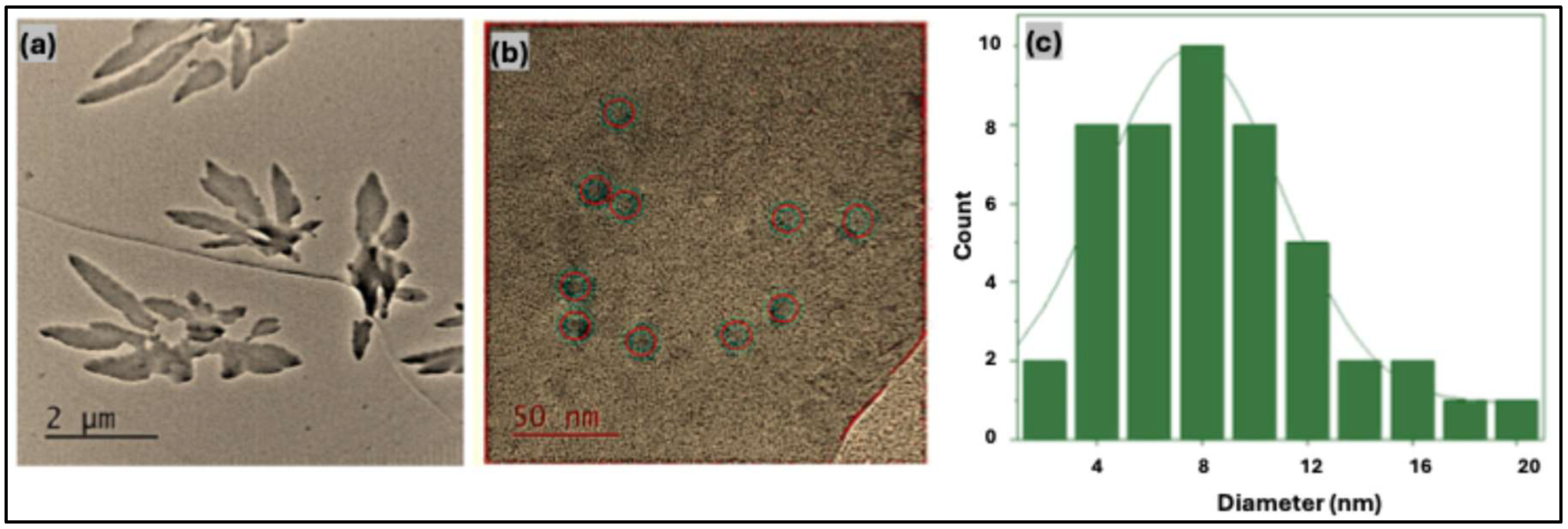
Transmission electron microscopy (TEM) was employed to examine the morphology, dispersion behavior, and particle size distribution of the carbon quantum dots. The low-magnification TEM image (scale bar: 2 μm) in
Figure 3(a) illustrates the aggregation behavior of hbCQDs in aqueous media. The particles exhibit a pronounced tendency to form irregular, randomly shaped clusters, reflecting their hydrophobic surface characteristics and limited colloidal stability in water. High-magnification TEM images (
Figure 3(b)) reveal that the hbCQDs are quasi-spherical and partially crystalline in nature. Lattice fringes observed in certain regions indicate localized graphitic ordering within the carbon core. Statistical analysis of particle diameters, derived from multiple TEM micrographs, shows a size distribution ranging from 2 to 20 nm, with an average particle size of approximately 8 nm, as summarized in the histogram presented in
Figure 3(c).
4.1.3. Fourier-Transform Infrared Analysis (FTIR)
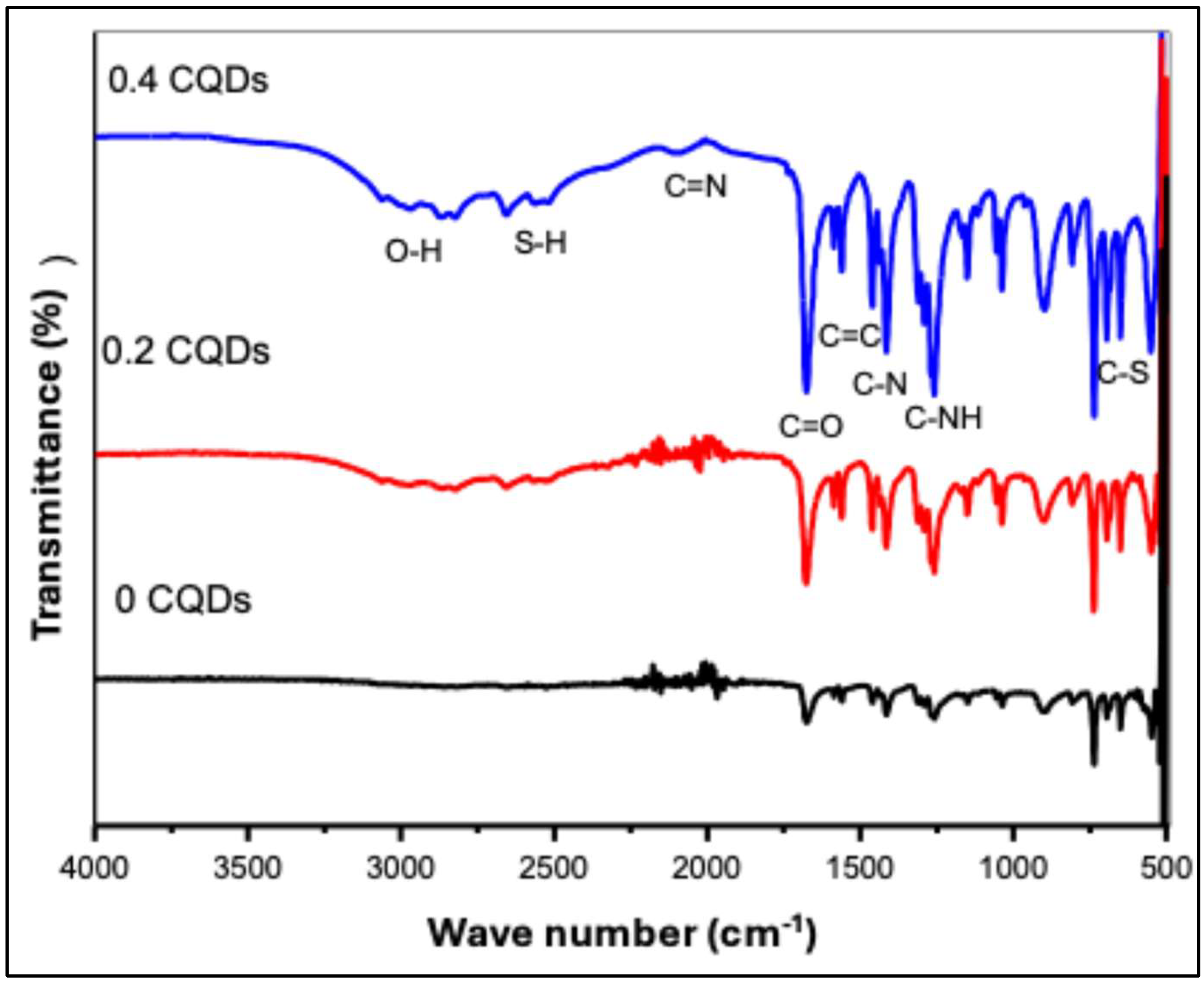
Fourier-transform infrared (FTIR) spectroscopy was conducted to identify the surface functional groups of the hydrophobic CQDs. The FTIR spectra of pristine hbCQDs and hbCQDs/RhB composites are presented in
Figure 4. The spectrum of pure hbCQDs exhibits relatively weak absorption features, indicating a surface with limited oxygen-containing functionalities. The broad region between 4000 and 2000 cm⁻¹ shows minimal prominent peaks, confirming that the structure is predominantly carbonaceous with low surface polarity. This observation is consistent with the hydrophobic nature of the synthesized CQDs [
29]. Upon incorporation of a low amount (0.2 mL) of Rhodamine B (RhB), new low-intensity functional group signals emerge. A noticeable O–H stretching vibration appears around 2852 cm⁻¹, indicating the introduction of hydrophilic groups associated with RhB molecules. With further increase of RhB content to 0.4 mL, the O–H stretching band becomes significantly more intense, suggesting enhanced surface functionalization. Consequently, the surface character transitions from hydrophobic to hydrophilic, enabling improved water dispersibility of the composite [
30,
31]. All samples display characteristic absorption bands corresponding to C≡N stretching (2034 cm⁻¹), S–H stretching (2650 cm⁻¹), and amide carbonyl (C=O) stretching at 1682 cm⁻¹. Additional peaks are observed at 1469 cm⁻¹ (C=C), 1415 cm⁻¹ (C–N), 1264 cm⁻¹ (aromatic C–NH), 1155 cm⁻¹ (C–O), and 690 cm⁻¹ (C–S). A weak band near 491 cm⁻¹ is attributed to S–S vibrations, likely arising from aggregated RhB molecular structures. Moreover, the asymmetric stretching mode of –N=C=S at 2150 cm⁻¹ confirms the presence of isothiocyanate groups derived from RhB [
32]. For the composite with higher RhB loading, the spectrum clearly exhibits the characteristic absorption features of RhB in the 1600–1000 cm⁻¹ region, reflecting the increased contribution of dye-related functional groups to the overall chemical structure.
4.1.4. X-Ray Photoelectron Spectroscopy Analysis (XPS)
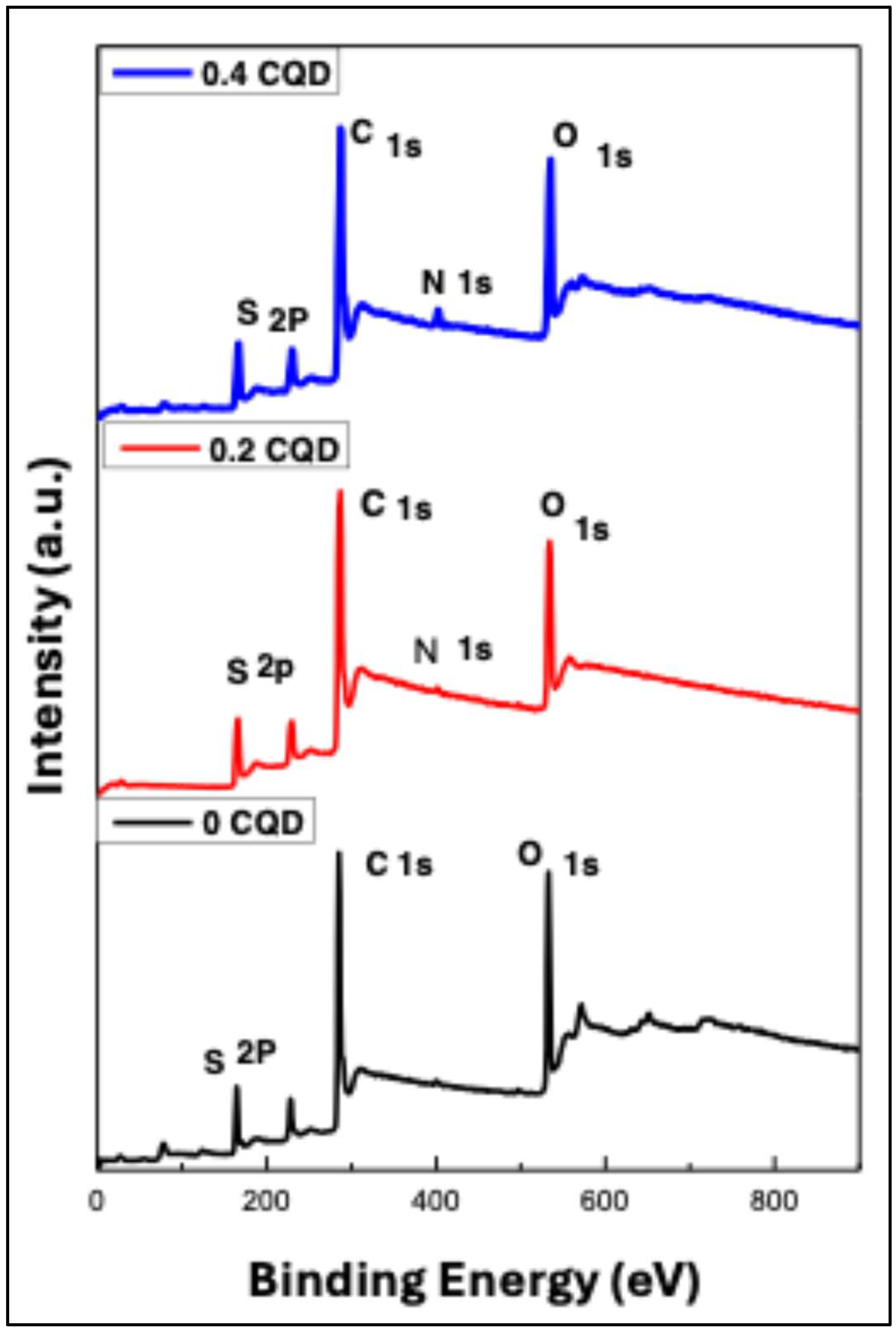
X-ray photoelectron spectroscopy (XPS) was employed to investigate the elemental composition and chemical bonding of the hbCQDs, as illustrated in
Figure 5. The survey spectrum reveals four prominent peaks at binding energies of 531 eV [133], 400 eV [133], 285 eV, and 164 eV [134], corresponding to the O 1s, N 1s, C 1s, and S 2p core levels, respectively. These results confirm the presence of oxygen, nitrogen, carbon, and sulfur in the hbCQDs and provide insight into the surface chemical environment of the synthesized nanomaterials.
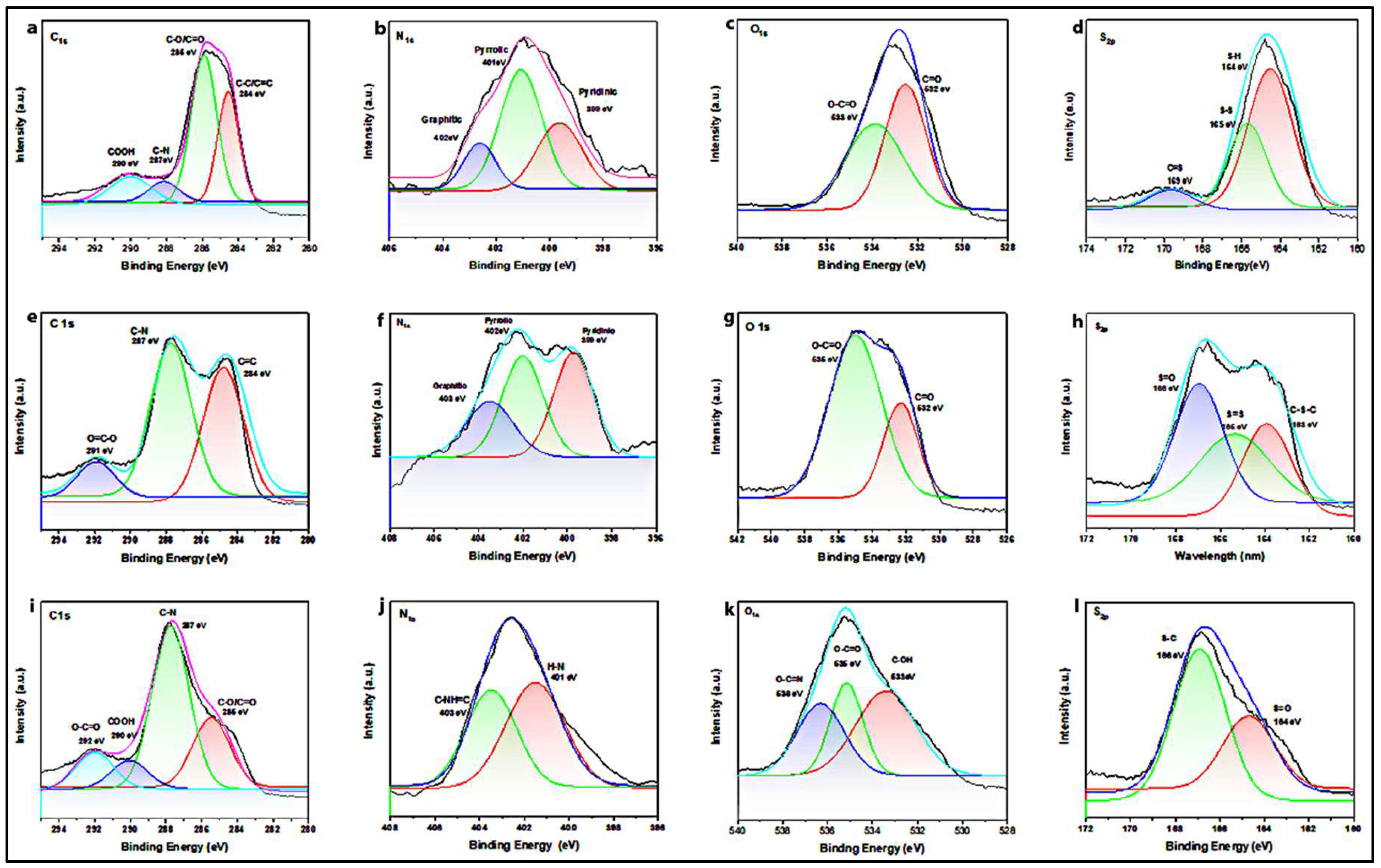
The high-resolution XPS spectra of pure hbCQDs were deconvoluted to analyze the chemical states of the constituent elements, as presented in
Figure 6.
Figure 6(a) shows the C 1s spectrum of pure hbCQDs, revealing characteristic peaks corresponding to C=C (284.6 eV), C=O/C–O (285.9 eV), and C–N (287.5 eV) bonds. The hbCQDs/RhB composites exhibit similar C 1s profiles; however, a slight positive shift in the C–N binding energy is observed with increasing RhB concentration, suggesting the formation of additional amide linkages within the hybrid structure (
Figure 6(i)).
The O 1s spectra, depicted in
Figure 6(b), indicate the presence of C=O groups at 532.2 eV in samples with lower RhB content. At higher RhB loading, a new peak emerges at 533.0 eV, corresponding to C–O/C–OH functionalities, reflecting enhanced surface oxidation and functionalization.
Figure 6(c) presents the N 1s spectra, showing contributions from pyrrolic nitrogen (399.6 eV) and pyridinic nitrogen (398.17 eV). With increasing RhB content, the pyridinic nitrogen peak becomes more prominent, indicating the expansion of conjugated aromatic domains within the carbon framework (
Figure 6(j)).
Finally, the S 2p spectra, shown in
Figure 6(d), confirm the presence of multiple sulfur species, including thiol S–H (164.5 eV), thiophene C–S–C (163.3 eV), disulfide S=S (165.1 eV), and sulfoxide S=O (169.0 eV). With higher RhB concentrations, partial oxidation of S–OH groups occurs, leading to the formation of SOₓ species, which reflects the chemical interaction between CQDs and the RhB molecules. This comprehensive XPS analysis demonstrates that RhB incorporation modifies the surface chemistry of hbCQDs, promoting functional group formation and enhanced aromaticity within the composites.
4.2. Optical Characterization
4.2.1. UV-Vis Absorption Spectroscopy Analysis
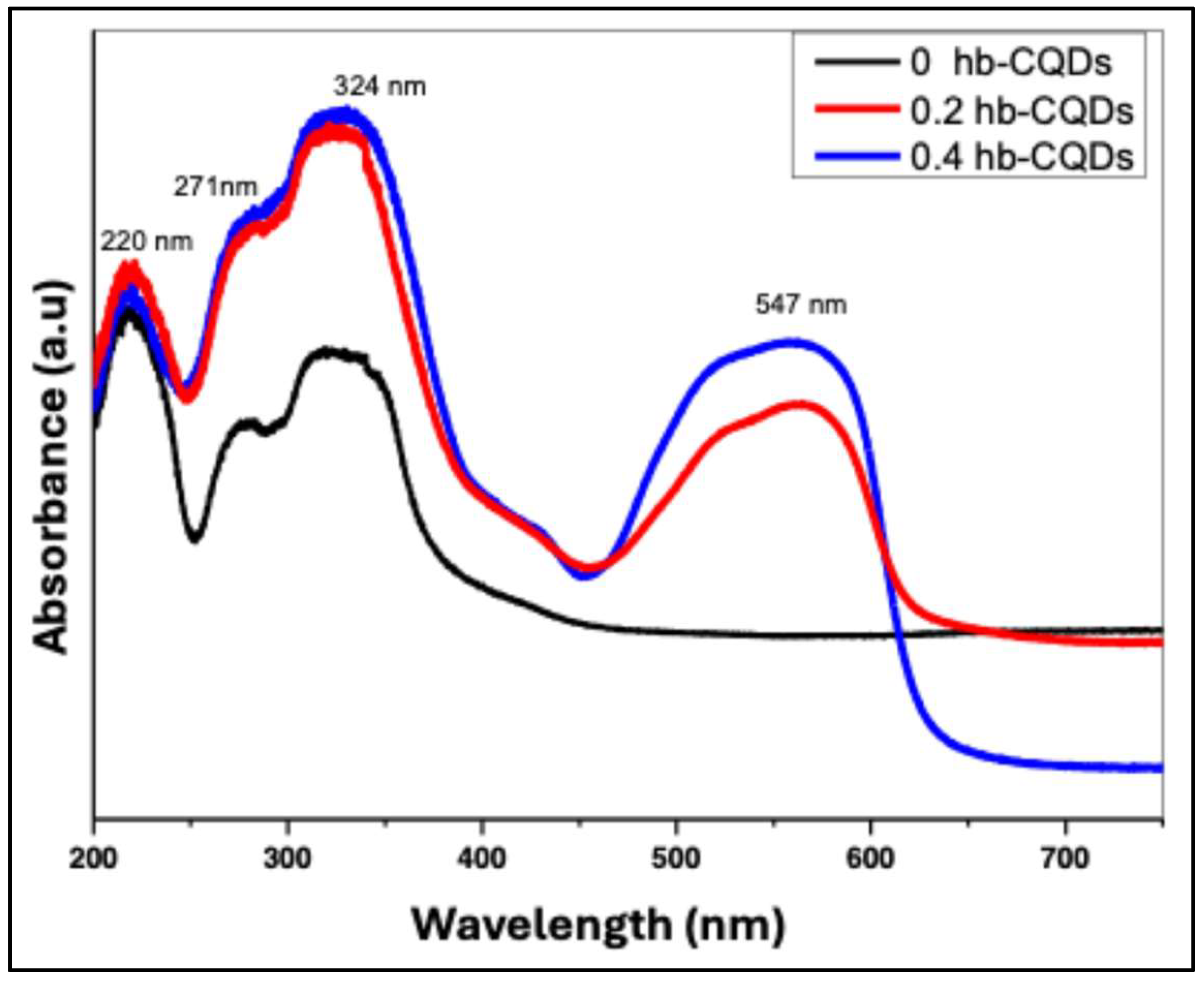
The UV–Vis absorption spectra of the samples can be divided into two distinct regions: a high-energy region spanning 200–400 nm and a low-energy region at wavelengths above 400 nm.
Figure 7 presents the absorption profiles of pure hbCQDs and hbCQDs/RhB composites. In the high-energy region, all three samples exhibit similar spectral features with minimal variation. Three prominent absorption peaks are observed, extending into the visible range. The first two peaks, located at 220 nm and 271 nm, are attributed to π–π* transitions of the C=C bonds within the graphitic carbon core of the hbCQDs [
31]. A third peak, appearing around 342 nm, is assigned to n–π* transitions arising from surface states, including C=N/C=O, C–O, and C–S functional groups. These features reflect the electronic structure and surface chemistry of the hbCQDs. In the low-energy region above 400 nm, the hbCQDs/RhB composites display a distinct absorption band at 547 nm, corresponding closely to the characteristic absorption of Rhodamine B [
33]. This peak confirms successful incorporation of RhB into the CQD matrix and indicates potential for extending photoluminescence into the visible range.
4.2.2. Photoluminescence (PL) Spectroscopy Analysis
4.2.1.1. Pure Hydrophobic CQDs Without RhB
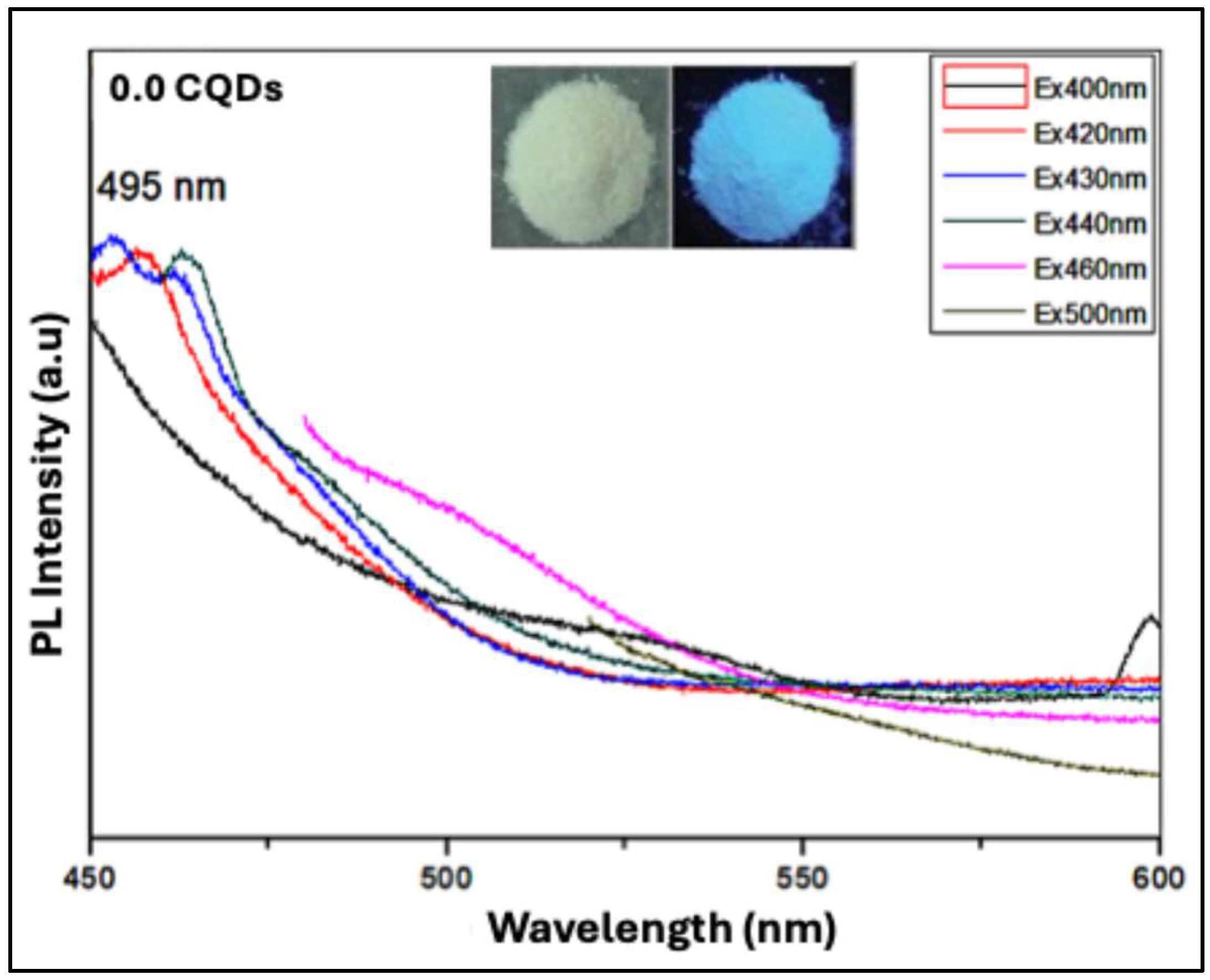
Figure 8 illustrates the photoluminescence (PL) behavior of pure hbCQDs (0% RhB) under varying excitation wavelengths. Across this excitation range, the CQDs exhibit a characteristic blue emission, with the maximum emission peak centered at approximately 453 nm. The inset displays photographs of the powdered sample under daylight and upon 365 nm UV illumination, clearly demonstrating a vivid blue emission, which confirms the primary luminescent behavior of the CQDs. This emission originates predominantly from surface functional group states rather than the carbon core, with a measured quantum yield (QY) of 6.2%.
4.2.1.2. Hydrophobic CQDs Composite with RhB of 0.2 mL
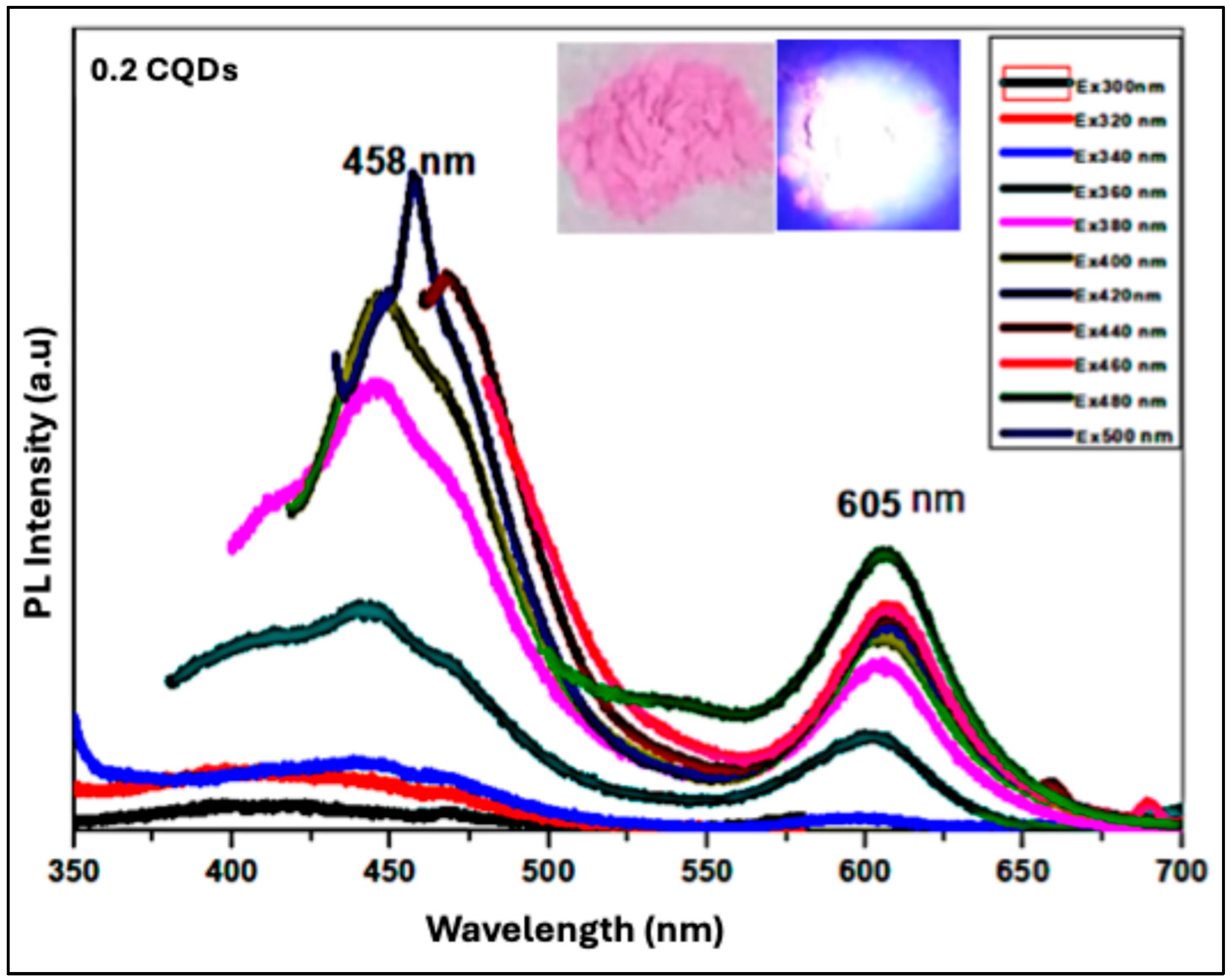
Figure 9 presents the photoluminescence (PL) spectrum of the hydrophobic CQDs/RhB composite prepared with 0.2 mL RhB. The spectrum exhibits two distinct emission peaks: a higher-energy peak originating from carbon surface states, and a lower-energy peak attributed to molecular states derived from organic fluorophores used as precursors in CQD synthesis. These fluorophores may be covalently bonded to the carbon core or coupled to edge carbon atoms or functional groups, giving rise to n–π* and π–π* transitions. The surface-state emission shows excitation-dependent behavior, with maximum excitation/emission at 458 nm (blue), whereas the molecular-state emission is excitation-independent, with a peak at 605 nm (orange). The inset of
Figure 9 shows a photograph of the 0.2 mL RhB–CQD composite powder. Under ambient light, the powder appears bright pink (left), while under 365 nm UV illumination, it emits a strong white light (right), resulting from the combination of the blue and orange emissions from the composite. This composite demonstrates optimal properties, with a high quantum yield of 13% and a production yield of 82%, making it a promising candidate for future white-light optoelectronic applications.
4.2.1.3. Hydrophobic CQDS Composite with RhB of 0.4 mL
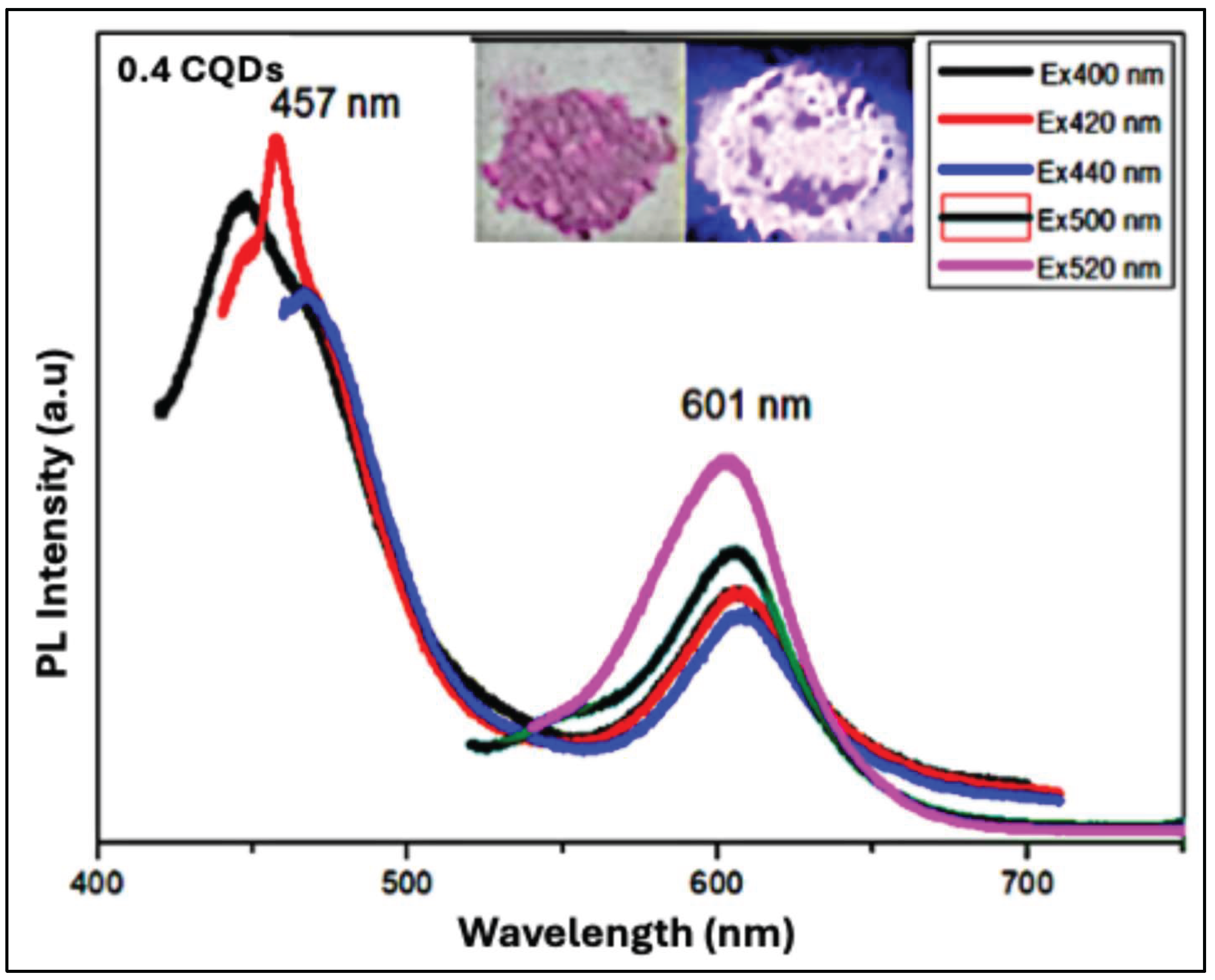
Figure 10 presents the photoluminescence (PL) spectra of the hydrophobic hbCQDs/RhB composite prepared with 0.4 mL RhB. The spectrum displays two emission peaks: a higher-energy peak arising from carbon surface states and a lower-energy peak associated with molecular states from the incorporated RhB. Compared to the 0.2 mL RhB–hbCQDs sample, the positions of both peaks show slight shifts, reflecting the higher RhB loading. The quantum yield of this sample is reduced to below 9%, indicating decreased emission efficiency at higher dye content. The inset of
Figure 10 shows a photograph of the composite powder. Under ambient light (left), the powder appears purple, while under 365 nm UV illumination (right), it emits a weak orange fluorescence. This observation demonstrates that increasing RhB content alters the emission profile and diminishes the overall photoluminescence intensity.
5. Conclusion and Future Work
In this study, hydrophobic carbon quantum dots (hbCQDs) were successfully synthesized via a solvothermal method and subsequently hybridized with Rhodamine B (RhB) to achieve tunable photoluminescence. The pure hbCQDs exhibited blue emission at 453 nm with a quantum yield (QY) of 6.2%, while the incorporation of 0.2 mL RhB enabled dual-emission, combining surface-state blue emission and molecular-state orange emission, resulting in white-light emission with a QY of 13% and a production yield of 82%. Higher RhB loading (0.4 mL) caused slight peak shifts and reduced QY (<9%), demonstrating the critical influence of RhB concentration on optical performance. Structural and chemical analyses (TEM, XRD, FTIR, XPS) confirmed the quasi-spherical morphology, partially crystalline carbon core, and the presence of functional groups (C–N, C=O/C–O, S–H) that mediate photoluminescence. Overall, these results indicate that controlled RhB hybridization offers a simple and reproducible strategy to extend the emission of hbCQDs into the visible spectrum, making them suitable candidates for optoelectronic applications, including white-light-emitting devices and hydrophobic media-compatible photonic systems.
Future investigations should focus on optimizing the emission balance between surface-state and molecular-state contributions to maximize white-light efficiency. The stability of hbCQDs/RhB composites under prolonged UV exposure, varying temperatures, and different solvent environments should be systematically evaluated. Furthermore, exploring other organic dyes or co-dopants could expand the emission range and enhance quantum yield. Integration of these composites into device architectures, such as LEDs or flexible optoelectronic systems, will be essential to translate the material’s photophysical properties into practical applications. Additionally, understanding the precise mechanism of energy transfer between CQDs and organic fluorophores at the molecular level would provide insights for designing next-generation hybrid luminescent nanomaterials.
Funding
This research received no external funding.
Data Availability Statement
The authors confirm that the data supporting the findings of this study are available and can be accessed within the article.
Acknowledgments
The authors acknowledge with thanks the University of Jeddah for its research facilities.
Conflicts of Interest
The authors declare no conflict of interest.
References
- U. Woggon and P. Alivisatos, "Optical Properties of Semiconductor Quantum Dots ," Physics Today, vol. 51, no. 3, p. 94–94, 1998. [CrossRef]
- P. S. Karthik, A. L. Himaja, and S. P. Singh, "Carbon-allotropes: Synthesis methods, applications and future perspectives," Carbon Letters, vol. 15, no. 4, p. 219–237, 2014. [CrossRef]
- E. Liu et al., "Highly Emissive Carbon Dots in Solid State and Their Applications in Light-Emitting Devices and Visible Light Communication," ACS Sustain Chem Eng , vol. 7, no. 10, p. 9301–9308, 2019. [CrossRef]
- W. dong Sheng et al., "Electronic and optical properties of semiconductor and graphene quantum dots," Front Physics (Beijing), vol. 7, no. 3, pp. 328–352, 2012. [CrossRef]
- J. Sharma and P. Y. Dave, "photostabilityCarbon Dots : Zero Dimensional Fluorescent Material,," Journal of Nanomaterials & Molecular Nanotechnology, vol. 10, no. February, p. 1–10, 2020.
- M. Pourmadadi et al., "Properties and application of carbon quantum dots (CQDs) in biosensors for disease detection: a comprehensive review," J Drug Deliv Sci Technol, p. 104156, 2023.
- "Color Emission Carbon Dots with Quench-ResixAstant Solid-State Fluorescence for Light-Emitting Diodes," ACS Sustain Chem Eng, vol. 9, no. 10, p. 3901–3908, 2021. [CrossRef]
- Alaa Y. Mahmoud and Walaa Al-masri, "Synthesis, optical tuning, and fluorescent sensing of Fe3+ with N-doped carbon quantum dots," Microchemical Journal, vol. 224, no. 5, pp. 1-10, 2026. [CrossRef]
- Magesh Vasanth , Sundramoorthy Ashok K. , Ganapathy Dhanraj, "Recent Advances on Synthesis and Potential Applications of Carbon Quantum Dots," Frontiers in Materials, vol. 9, no. 906838, pp. 1-27, 2022. [CrossRef]
- Y. Wang, Q. Zhuang, and Y. Ni, "Facile Microwave-Assisted Solid-Phase Synthesis of Highly Fluorescent Nitrogen-Sulfur-Codoped Carbon Quantum Dots for Cellular Imaging Applications," Chemistry- A European Journal, vol. 21, no. 37, p. 13004–13011, 2015. [CrossRef]
- Junli Wang, Yongzhen Yang, Xuguang Liu, "Solid-state fluorescent carbon dots: quenching resistance strategies, high quantum efficiency control, multicolor tuning, and applications," Materials Advances, vol. 1, no. 9, pp. 3122-3142, 2020. [CrossRef]
- Hyo Jeong Yoo, Byeong Eun Kwak, Do Hyun Kim, "The Self-Quenching Origin of Carbon Dots and the Guideline for Its Solid-State Luminescence," The Journal of Physical Chemistry C , vol. 123, no. 44, pp. 1-14, 2019. [CrossRef]
- K. Dimos, "Carbon Quantum Dots: Surface Passivation and Functionalization," Current Organic Chemistry , vol. 20, no. 6, pp. 682 - 695, 2006. [CrossRef]
- Kokilavani S., Gurpreet Singh Selopal , "Advanced Surface Engineering and Passivation Strategies of Quantum Dots for Breaking Efficiency Barrier of Clean Energy Technologies: A Comprehensive Review," Advanced Fubctional Materials, vol. 36, no. 4, pp. 1-41, 2026. [CrossRef]
- Hui Ding ORCID logo ; Xue-Hua Li; Xiao-Bo Chen ORCID logo; Ji-Shi Wei; Xiao-Bing Li; Huan-Ming Xiong, "Surface states of carbon dots and their influences on luminescence," Journal of Applied Physics, vol. 127, no. 23, pp. 1-21, 2020. [CrossRef]
- I. Denmark, S. Macchi, F. Watanabe, T. Viswanathan, and N. Siraj, "Effect of KOH on the Energy Storage Performance of Molasses-Based Phosphorus and Nitrogen Co-Doped Carbon," Electrochem, vol. 2, no. 1, p. 29–41, 2021. [CrossRef]
- Chenhao Li, Yuyue Yang, Fanglong Yuan, "Unprecedented Matrix-Induced Emission Enhancement Enables Bright and Efficient Carbon Quantum Dot-Based Electroluminescent Light-Emitting Diodes," Advanced functional Materials, vol. 22, no. 3, pp. 1-11, 2025. [CrossRef]
- Arif Kamal, Seongin Hong, Heongkyu Ju , "Carbon Quantum Dots: Synthesis, Characteristics, and Quenching as Biocompatible Fluorescent Probes," Biosensors (Basel), vol. 15, no. 2, pp. 1-19, 2025. [CrossRef]
- L. Cui, X. Ren, M. Sun, H. Liu, and L. Xia, "Carbon dots: Synthesis, properties and applications," Nanomaterials, vol. 11, no. 12, pp. 1-38, 2021. [CrossRef]
- H. Yamashita, A. Tanaka, M. Nishimura, K. Koyano, T. Tatsumi, M. Anpo, "Photochemical properties of Rhodamine-B dye molecules included within mesoporous molecular sieves," Studies in Surface Science and Catalysis, vol. 117, no. 1, pp. 551-558, 1998. [CrossRef]
- Mansoureh Tavan, Zeynab Yousefian, Ziba Bakhtiar, Moones Rahmandoust, Mohammad Hossein Mirjalili, "Carbon quantum dots: Multifunctional fluorescent nanomaterials for sustainable advances in biomedicine and agriculture," Industrial Crops and Products, vol. 231, no. 121207, pp. 1-29, 2025. [CrossRef]
- Lavanya Thyda, Joel K Joseph, Naresh Koppula, Suneetha Sana, Thangaraju Kuppusamy, "Carbon Quantum Dots/ZnO Hybrid Nanostructured Thin Films and UV Photodetector Performances," Physica Status Solidi (a), vol. 222, no. 18, pp. 1-13, 2025. [CrossRef]
- Boyang Wang, Junwei Wang, Siyu Lu, "Carbon Dots: Small Materials With Big Impacts on Optoelectronic Devices," Aggregate, vol. 6, no. 12, pp. 1-9, 2025. [CrossRef]
- RaghvendraKumar Mishra, Iva Chianella, Jayati Sarkar, Hamed YazdaniNezhad, and Saurav Goel, "NanostructuredZnO-CQDHybridHeterostructure Nanocomposites:SynergisticEngineeringforSustainable Design,FunctionalProperties,andHigh-Performance Applications," ChemNanoMat , vol. 10, no. 5, pp. 1-36, 2024. [CrossRef]
- C. Ravi Dhas, R. Venkatesh, K. Jothivenkatachalam, A. Nithya, B. Suji Benjamin, A. Moses Ezhil Raj, K. Jeyadheepan, C. Sanjeeviraja, "Visible light driven photocatalytic degradation of Rhodamine B and Direct Red using cobalt oxide nanoparticles," Ceramics International, vol. 41, no. 8, pp. 9301-9313, 2015. [CrossRef]
- T. Karstens and K. Kobs, "Rhodamine B and rhodamine 101 as reference substances for fluorescence quantum yield measurements," The Journal of Physical Chemistry , vol. 84, no. 14, pp. 1-10, 1980. [CrossRef]
- Magesh Vasanth , Sundramoorthy Ashok K. , Ganapathy Dhanraj , "Recent Advances on Synthesis and Potential Applications of Carbon Quantum Dots," Frontiers in Materials, vol. 9, no. 906838, pp. 1-27, 2022. [CrossRef]
- Salah Elkun, M. Ghali, T. Sharshar & M. M. Mosaad, "Green synthesis of fluorescent N-doped carbon quantum dots from castor seeds and their applications in cell imaging and pH sensing," Scientific Reports, vol. 14, no. 27927, pp. 1-14, 2024. [CrossRef]
- V. Gude, A. Das, T. Chatterjee, and P. K. Mandal, "Molecular origin of photoluminescence of carbon dots: Aggregation-induced orange-red emission,," Physical Chemistry Chemical Physics, vol. 18, no. 40, p. 28274–28280, 2016. [CrossRef]
- P. Zhao and L. Zhu, "Dispersibility of carbon dots in aqueous and/or organic solvents," Chemical Communications,, vol. 54, no. 43, p. 5401–5406, 2018. [CrossRef]
- S. Han, X. Chen, Y. Hu, and L. Han, "Solid-state N,P-doped carbon dots conquer aggregation-caused fluorescence quenching and couple with europium metal-organic frameworks toward white light-emitting diodes," Dyes and Pigments, vol. 187, no. 12, pp. 1-13, 2021. [CrossRef]
- Diac et al., "Covalent conjugation of carbon dots with Rhodamine B and assessment of their photophysical properties," RSC Adv, vol. 5, no. 95, p. 77662–77669, 2015. [CrossRef]
- Sun et al., "Synthesis of Nitrogen and Sulfur Co-doped Carbon Dots from Garlic for Selective Detection of Fe3+," Nanoscale Res Lett, vol. 11, no. 1, pp. 1–9, 2016. [CrossRef]
Figure 1.
Schematic diagram for the synthesis of carbon quantum dots by Solvothermal method.
Figure 1.
Schematic diagram for the synthesis of carbon quantum dots by Solvothermal method.
Figure 2.
X-ray diffraction (XRD) patterns of hydrophobic carbon quantum dots (hbCQDs) and hbCQDs/Rhodamine B composite solid powders prepared with varying RhB loadings.
Figure 2.
X-ray diffraction (XRD) patterns of hydrophobic carbon quantum dots (hbCQDs) and hbCQDs/Rhodamine B composite solid powders prepared with varying RhB loadings.
Figure 3.
Transmission electron microscopy (TEM) images of hydrophobic carbon quantum dots (hbCQDs): (a) low-magnification image (scale bar: 2 μm) showing particle aggregation behavior; (b) high-magnification image (scale bar: 50 nm) illustrating quasi-spherical morphology and structural features; and (c) particle size distribution histogram obtained from TEM analysis using ImageJ software.
Figure 3.
Transmission electron microscopy (TEM) images of hydrophobic carbon quantum dots (hbCQDs): (a) low-magnification image (scale bar: 2 μm) showing particle aggregation behavior; (b) high-magnification image (scale bar: 50 nm) illustrating quasi-spherical morphology and structural features; and (c) particle size distribution histogram obtained from TEM analysis using ImageJ software.
Figure 4.
FTIR spectra of hydrophobic carbon quantum dots (hbCQDs) and hbCQDs/Rhodamine B composites: pure hbCQDs (black), 0.2 mL RhB–hbCQDs (red), and 0.4 mL RhB–hbCQDs (blue), highlighting the evolution of surface functional groups with increasing RhB content.
Figure 4.
FTIR spectra of hydrophobic carbon quantum dots (hbCQDs) and hbCQDs/Rhodamine B composites: pure hbCQDs (black), 0.2 mL RhB–hbCQDs (red), and 0.4 mL RhB–hbCQDs (blue), highlighting the evolution of surface functional groups with increasing RhB content.
Figure 5.
X-ray photoelectron spectroscopy (XPS) survey spectra of hydrophobic carbon quantum dots (hbCQDs) and hbCQDs/Rhodamine B composites: pure hbCQDs (black), 0.2 mL RhB–hbCQDs (red), and 0.4 mL RhB–hbCQDs (blue), illustrating the elemental composition and chemical bonding variations with increasing RhB content.
Figure 5.
X-ray photoelectron spectroscopy (XPS) survey spectra of hydrophobic carbon quantum dots (hbCQDs) and hbCQDs/Rhodamine B composites: pure hbCQDs (black), 0.2 mL RhB–hbCQDs (red), and 0.4 mL RhB–hbCQDs (blue), illustrating the elemental composition and chemical bonding variations with increasing RhB content.
Figure 6.
High-resolution XPS spectra of hbCQDs and hbCQDs/RhB composites. (a–d) Deconvoluted C 1s, N 1s, O 1s, and S 2p spectra of pure hbCQDs showing characteristic functional groups. (e–h) Spectra of hbCQDs hybridized with 0.2 mL RhB, indicating slight binding energy shifts due to dye interaction. (i–l) Spectra of hbCQDs with 0.4 mL RhB, showing changes in peak intensity that confirm successful RhB incorporation and surface modification.
Figure 6.
High-resolution XPS spectra of hbCQDs and hbCQDs/RhB composites. (a–d) Deconvoluted C 1s, N 1s, O 1s, and S 2p spectra of pure hbCQDs showing characteristic functional groups. (e–h) Spectra of hbCQDs hybridized with 0.2 mL RhB, indicating slight binding energy shifts due to dye interaction. (i–l) Spectra of hbCQDs with 0.4 mL RhB, showing changes in peak intensity that confirm successful RhB incorporation and surface modification.
Figure 7.
UV–Vis absorption spectra of hydrophobic carbon quantum dots (hbCQDs) and hbCQDs/Rhodamine B composites: pure hbCQDs (black), 0.2 mL RhB–hbCQDs (red), and 0.4 mL RhB–hbCQDs (blue), highlighting the influence of increasing RhB content on the optical absorption characteristics.
Figure 7.
UV–Vis absorption spectra of hydrophobic carbon quantum dots (hbCQDs) and hbCQDs/Rhodamine B composites: pure hbCQDs (black), 0.2 mL RhB–hbCQDs (red), and 0.4 mL RhB–hbCQDs (blue), highlighting the influence of increasing RhB content on the optical absorption characteristics.
Figure 8.
Photoluminescence (PL) spectra of pure hbCQDs (0% RhB). The inset shows comparative photographs of the sample under natural daylight and 365 nm UV illumination, highlighting the characteristic blue emission of the CQDs.
Figure 8.
Photoluminescence (PL) spectra of pure hbCQDs (0% RhB). The inset shows comparative photographs of the sample under natural daylight and 365 nm UV illumination, highlighting the characteristic blue emission of the CQDs.
Figure 9.
Photoluminescence (PL) spectra of hbCQDs/RhB composite with 0.2 mL RhB. The inset shows comparative photographs of the powder under natural daylight and 365 nm UV illumination, highlighting the color change from bright pink to white emission.
Figure 9.
Photoluminescence (PL) spectra of hbCQDs/RhB composite with 0.2 mL RhB. The inset shows comparative photographs of the powder under natural daylight and 365 nm UV illumination, highlighting the color change from bright pink to white emission.
Figure 10.
Photoluminescence (PL) spectra of hbCQDs/RhB composite with 0.4 mL RhB. The inset shows comparative photographs of the powder under natural daylight and 365 nm UV illumination, highlighting the color change from purple under ambient light to weak orange emission under UV excitation.
Figure 10.
Photoluminescence (PL) spectra of hbCQDs/RhB composite with 0.4 mL RhB. The inset shows comparative photographs of the powder under natural daylight and 365 nm UV illumination, highlighting the color change from purple under ambient light to weak orange emission under UV excitation.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).