Submitted:
17 February 2026
Posted:
05 March 2026
You are already at the latest version
Abstract
Keywords:
1. Emergent Engineering
- 1.
- Design according to well-understood principles that hold for components in isolation and in aggregate.
- 2.
- Use nearly fault-free components to achieve very high levels of combined precision.
- 3.
- Minimize error and system failure rates by eliminating uncertainty and reducing the degrees of freedom of the components.
- 4.
- Operate within linear regimes where collective dynamics are predictable and controllable.
- 5.
- Reduce noise and adaptability of components to prevent unexpected emergent behaviors.

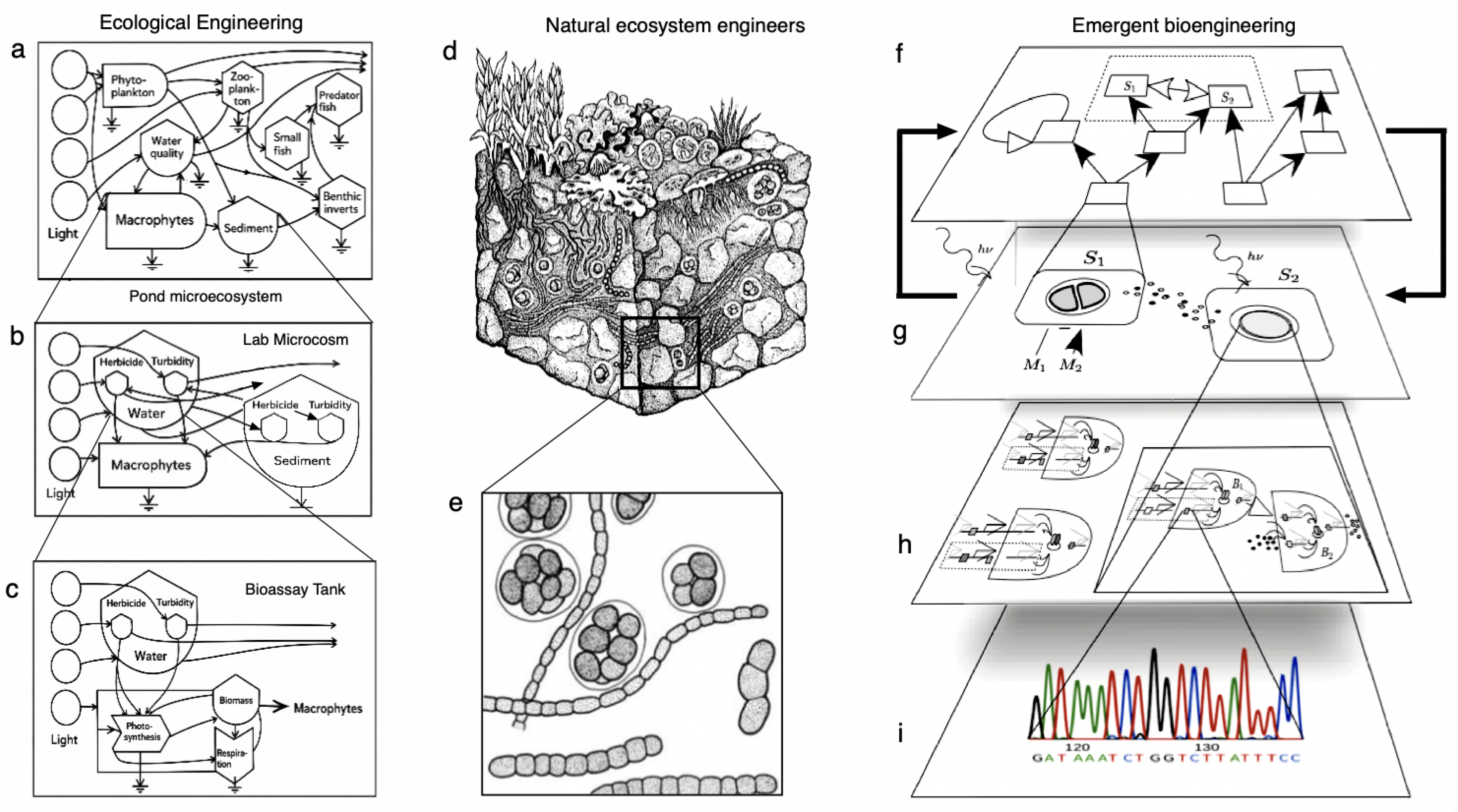
2. Ecology as a Design Language
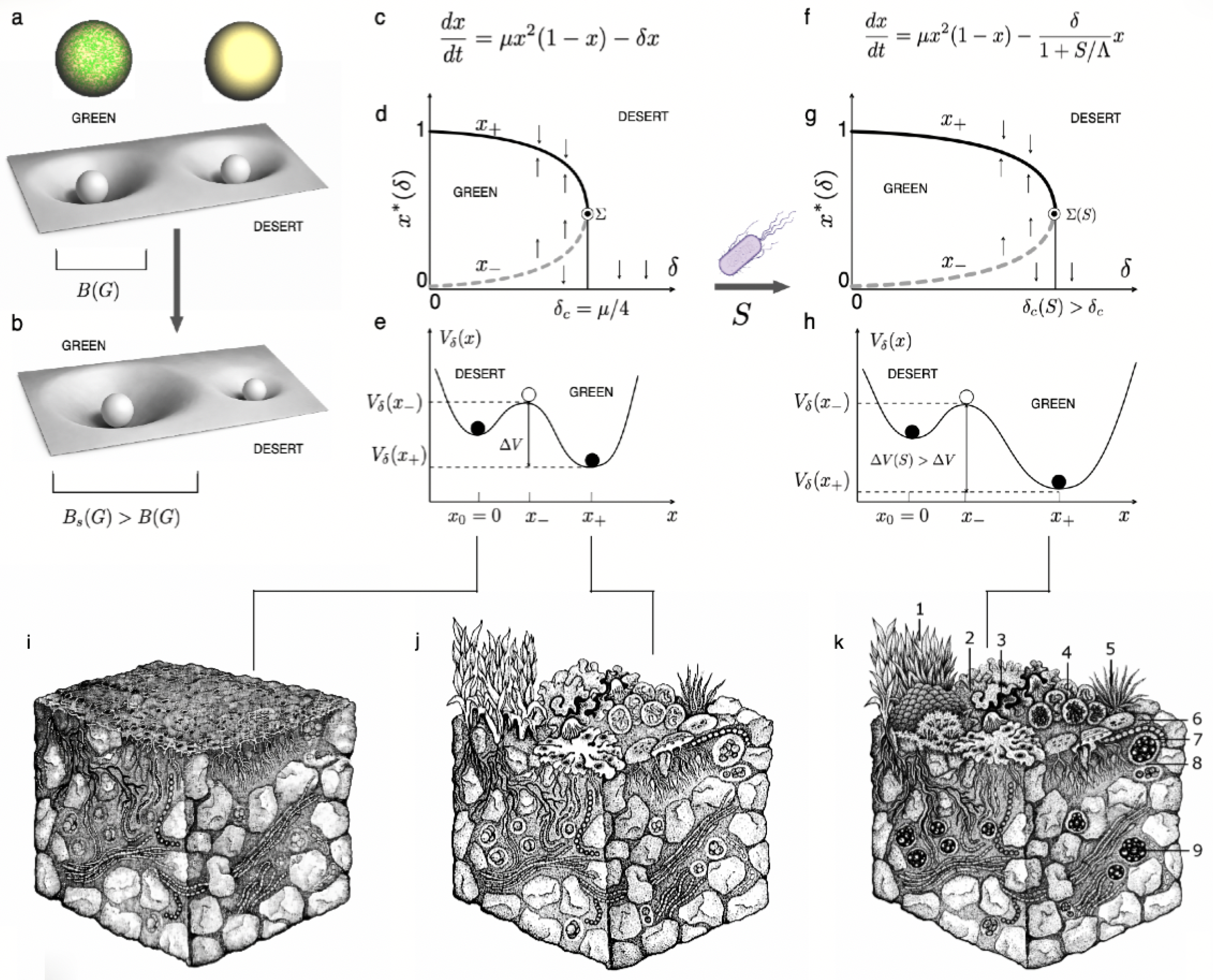
3. Biodiversity as Both Shield and Scaffold

4. Discussion
Acknowledgments
References
- Dowden, H.; Munro, J. Trends in clinical success rates and therapeutic focus. Nature Reviews Drug Discovery 2019, 18, 495–496. [Google Scholar] [CrossRef]
- Harrison, R.K. Phase II and phase III failures: 2013–2015. Nature Reviews Drug Discovery 2016, 15, 817–818. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Dua, M.; Singh, A.; Sethunathan, N.; Johri, A. Biotechnology and bioremediation: successes and limitations. Applied Microbiology and Biotechnology 2002, 59, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Cowan, G.; Pines, D.; Meltzer, D.E. Complexity: Metaphors, models, and reality. 1994. [Google Scholar]
- Levin, S.A. Ecosystems and the Biosphere as Complex Adaptive Systems. Ecosystems 1998, 1, 431–436. [Google Scholar] [CrossRef]
- Krakauer, D.C. Emergent Engineering: Reframing the Grand Challenge for the 21st Century. In Worlds Hidden in Plain Sight: The Evolving Idea of Complexity at the Santa Fe Institute; Krakauer, D.C., Ed.; SFI Press, 2019; chapter 37.** Raises the concept of Emergent Engineering for the first time. [Google Scholar]
- Rid, T. Rise of the machines: A cybernetic history; WW Norton & Company, 2016. [Google Scholar]
- Curtis, A. Machines of Loving Grace. Television documentary series. 2011. [Google Scholar]
- Munoz, M.A. Colloquium: Criticality and dynamical scaling in living systems. Reviews of Modern Physics 2018, 90, 031001. [Google Scholar] [CrossRef]
- Solé, R.; Levin, S. Ecological complexity and the biosphere: the next 30 years. 2022. [Google Scholar]
- Kemp, L.; Xu, C.; Depledge, J.; Ebi, K.L.; Gibbins, G.; Kohler, T.A.; et al. Climate Endgame: Exploring catastrophic climate change scenarios. Proceedings of the National Academy of Sciences 2022, 119. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Kohler, T.A.; Lenton, T.M.; Svenning, J.C.; Scheffer, M. Future of the human climate niche. Proceedings of the National Academy of Sciences 2020, 117, 11350–11355. [Google Scholar] [CrossRef]
- Clauer, P.; Nou, A.X.; Toth, T.; Yu, Q.; Chemla, Y.; Boo, A.; Yoon, K.; Voigt, C. Synthetic Biology of Plants and Microbes for Agriculture, Environment, and Future Applications This extensive review describes how synthetic biology tools can turn plants and their microbiomes into programmable systems for improved agriculture and future applications like sensing, manufacturing, and remediation. Also highlights major hurdles such as field reliability, community stability, evolution, delivery, and biosafety/containment. Chemical Reviews 2026, 126, 895–1109. [Google Scholar] [CrossRef]
- Artime, O.; De Domenico, M. From the origin of life to pandemics: Emergent phenomena in complex systems. Philosophical Transactions of the Royal Society A 2022, 380, 20200410. [Google Scholar] [CrossRef]
- Pedrolli, D.B.; Ribeiro, N.V.; Squizato, P.N.; de Jesus, V.N.; Cozetto, D.A.; at iGEM 2017, T.A.U. Engineering Microbial Living Therapeutics: The Synthetic Biology Toolbox. Trends in Biotechnology 2019, 37, 100–115. [Google Scholar] [CrossRef] [PubMed]
- Brooks, S.M.; Alper, H.S. Applications, challenges, and needs for employing synthetic biology beyond the lab. Nature Communications 2021, 12, 1390. [Google Scholar] [CrossRef] [PubMed]
- Stirling, F.; Bitzan, L.; O’Keefe, S.; Redfield, E.; Oliver, J.W.; Way, J.; et al. Rational design of evolutionarily stable microbial kill switches. Molecular cell 2017, 68, 686–697. [Google Scholar] [CrossRef]
- Chan, C.T.; Lee, J.W.; Cameron, D.E.; Bashor, C.J.; Collins, J.J. ’Deadman’ and ’Passcode’ microbial kill switches for bacterial containment. Nature Chemical Biology 2016, 12, 82–86. [Google Scholar] [CrossRef]
- Schmidt, M.; de Lorenzo, V. Synthetic bugs on the loose: containment options for deeply engineered (micro)organisms. Current Opinion in Biotechnology 2016, 38, 90–96. [Google Scholar] [CrossRef]
- Lee, J.W.; Chan, C.T.Y.; Slomovic, S.; Collins, J.J. Next-generation biocontainment systems for engineered organisms. Nature Chemical Biology 2018, 14, 530–537. [Google Scholar] [CrossRef]
- Rottinghaus, A.G.; Ferreiro, A.; Fishbein, S.R.S.; Dantas, G.; Moon, T.S. Genetically stable CRISPR-based kill switches for engineered microbes. Nature Communications 2022, 13, 672. [Google Scholar] [CrossRef]
- Adamala, K.P.; Agashe, D.; Belkaid, Y.; et al. Confronting risks of mirror life. Science 2024, 386, 1351–1353. [Google Scholar] [CrossRef]
- van den Berg, N.I.; Machado, D.; Santos, S.; Rocha, I.; Chacón, J.; Harcombe, W. Ecological modelling approaches for predicting emergent properties in microbial communities This review explains how different ecological modelling frameworks can predict emergent properties of microbial communities and compares the strengths and limits of approaches from Lotka–Volterra to genome-scale metabolic models for guiding rational ecosystem modulation. Nature Ecology & Evolution 2022, 6, 855–865. [Google Scholar] [CrossRef]
- Holling, C.S. Engineering resilience versus ecological resilience. In Engineering Within Ecological Constraints; Schulze, P.C., Ed.; National Academy Press: Washington, DC, 1996; pp. 31–44, ** Holling contrasts “engineering resilience” with “ecological resilience”, arguing that sustainable management must design for the latter in dynamic, uncertain systems. [Google Scholar]
- Hui, C.; Richardson, D.M. How to Invade an Ecological Network. Trends in Ecology & Evolution 2019, 34, 121–131. [Google Scholar] [CrossRef]
- Aguadé-Gorgorió, G.; Kéfi, S. Emergent coexistence and the limits of reductionism in ecological communities. 2025. [Google Scholar] [CrossRef]
- Case, T.J. Invasion resistance arises in strongly interacting species-rich model competition communities. Proceedings of the National Academy of Sciences 1990, 87, 9610–9614. [Google Scholar] [CrossRef] [PubMed]
- Maull, V.; Solé, R. Biodiversity as a firewall to engineered microbiomes for restoration and conservation. Royal Society Open Science 2024, 11. * Provides the theoretical basis for biodiversity as a predictable firewall and scaffold for engraftment of resident-derived synthetic strains for ecosystem restoration.. [Google Scholar] [CrossRef] [PubMed]
- Holling, C.S. Resilience and stability of ecological systems. Annual review of ecology and systematics 1973, 4, 1–23. [Google Scholar] [CrossRef]
- Kéfi, S.; Holmgren, M.; Scheffer, M. When can positive interactions cause alternative stable states in ecosystems? Functional Ecology 2016, 30, 88–97. [Google Scholar] [CrossRef]
- Aguadé-Gorgorió, G.; Arnoldi, J.F.; Barbier, M.; Kéfi, S. A taxonomy of multiple stable states in complex ecological communities. Ecology Letters 2024, 27, e14413. [Google Scholar] [CrossRef]
- Walker, B.H.; Ludwig, D.; Holling, C.S.; Peterman, R.M. Stability of Semi-Arid Savanna Grazing Systems. Journal of Ecology 1981, 69, 473–498. [Google Scholar] [CrossRef]
- Sánchez, A.; Vila, J.C.; Chang, C.Y.; Diaz-Colunga, J.; Estrela, S.; Rebolleda-Gomez, M. Directed Evolution of Microbial Communities. Annual Review of Biophysics 2021, 50, 323–341. [Google Scholar] [CrossRef]
- Goldford, J.E.; Lu, N.; Bajić, D.; Estrela, S.; Tikhonov, M.; Sanchez-Gorostiaga, A.; et al. Emergent simplicity in microbial community assembly. Science 2018, 361, 469–474. [Google Scholar] [CrossRef]
- Burke, C.; Steinberg, P.; Rusch, D.; Kjelleberg, S.; Thomas, T. Bacterial community assembly based on functional genes rather than species. Proceedings of the National Academy of Sciences 2011, 108, 14288–14293. [Google Scholar] [CrossRef]
- Stock, M.; Gorochowski, T.E. Open-endedness in synthetic biology: A route to continual innovation for biological design. Science Advances 2024, 10. [Google Scholar] [CrossRef] [PubMed]
- Skerker, J.M.; Lucks, J.B.; Arkin, A.P. Evolution, ecology and the engineered organism: lessons for synthetic biology. Genome Biology 2009, 10, 114. [Google Scholar] [CrossRef]
- Michael-Igolima, U.; Abbey, S.J.; Ifelebuegu, A.O. A Systematic Review on the Effectiveness of Remediation Methods for Oil Contaminated Soils. 9, 100319. [CrossRef]
- Mustafa, M.G.; Khan, M.G.M.; Nguyen, D.; Iqbal, S. Techniques in Biotechnology. In Omics Technologies and Bio-Engineering; Elsevier; pp. 233–249. [CrossRef]
- Cleves, P.A.; Strader, M.E.; Bay, L.K.; Pringle, J.R.; Matz, M.V. CRISPR/Cas9-mediated genome editing in a reef-building coral. Proceedings of the National Academy of Sciences 2018, 115, 5235–5240. [Google Scholar] [CrossRef]
- Maestre, F.T.; Sole, R.; Singh, B.K. Microbial biotechnology as a tool to restore degraded drylands. Microbial biotechnology 2017, 10, 1250–1253. [Google Scholar] [CrossRef] [PubMed]
- Solé, R.V.; Montañez, R.; Duran-Nebreda, S.; Rodriguez-Amor, D.; Vidiella, B.; Sardanyés, J. Population dynamics of synthetic terraformation motifs. Royal Society Open Science 2018, 5, 180121. [Google Scholar] [CrossRef]
- de Lorenzo, V.; Marliere, P.; Sole, R. Bioremediation at a global scale: from the test tube to planet Earth. Microbial biotechnology 2016, 9, 618–625. [Google Scholar] [CrossRef]
- Rosado, P.M.; Leite, D.C.A.; Duarte, G.A.S.; Chaloub, R.M.; Jospin, G.; Nunes da Rocha, U.; Saraiva, J.P.; Dini-Andreote, F.; Eisen, J.A.; Bourne, D.G.; et al. Marine probiotics: increasing coral resistance to bleaching through microbiome manipulation. The ISME Journal 2018, 13, 921–936. [Google Scholar] [CrossRef]
- Coleman, M.A.; Goold, H.D. Harnessing synthetic biology for kelp forest conservation1. Journal of Phycology 2019, 55, 745–751. [Google Scholar] [CrossRef]
- van Oppen, M.J.H.; Blackall, L.L. Coral microbiome dynamics, functions and design in a changing world. Nature Reviews Microbiology 2019, 17, 557–567. [Google Scholar] [CrossRef]
- de Lorenzo, V. Environmental Galenics: large-scale fortification of extant microbiomes with engineered bioremediation agents. Philosophical Transactions of the Royal Society B: Biological Sciences 2022, 377. [Google Scholar] [CrossRef]
- Jansson, J.K.; McClure, R.; Egbert, R.G. Soil microbiome engineering for sustainability in a changing environment. Nature Biotechnology 2023, 41, 1716–1728, * It reviews how microbial ecology and synthetic biology can be used to engineer soil microbiomes that boost plant growth and restore soil health under environmental change.. [Google Scholar] [CrossRef] [PubMed]
- Lea-Smith, D.J.; Hassard, F.; et al. C. Engineering biology applications for environmental solutions: potential and challenges. Nature Communications 2025, 16. * This perspective surveys how engineering biology can tackle environmental problems and what’s needed to deploy it safely and at scale in the real world. [Google Scholar] [CrossRef]
- de Lorenzo, V. Systems Biology Approaches to Bioremediation. 19, 579–589. [CrossRef]
- Jiang, T.; Montgomery, V.A.; Jetty, K.; Ganesan, V.; Incha, M.R.; Gladden, J.M.; Hillson, N.J.; Liu, D. Metabolic Engineering and Synthetic Biology for the Environment: From Perspectives of Biodetection, Bioremediation, and Biomanufacturing. 14. [CrossRef]
- Nandy, S.; Andraskar, J.; Lanjewar, K.; Kapley, A. Challenges in bioremediation: from lab to land. In Bioremediation for Environmental Sustainability; Elsevier, 2021; pp. 561–583. [Google Scholar] [CrossRef]
- Chemla, Y.; Sweeney, C.J.; Wozniak, C.A.; Voigt, C.A. Design and Regulation of Engineered Bacteria for Environmental Release. 10, 281–300. [CrossRef] [PubMed]
- Vidiella, B.; Solé, R. Ecological firewalls for synthetic biology. iScience 2022, 25, 104658. [Google Scholar] [CrossRef]
- Solé, R.; Maull, V.; Amor, D.R.; Pla Mauri, J.; Conde-Pueyo, N. Synthetic Ecosystems: From the Test Tube to the Biosphere. ACS Synthetic Biology 2024, 13, 3812–3826. [Google Scholar] [CrossRef]
- Elton, C.S. The Ecology of Invasions by Animals and Plants; Chapman and Hall: London, 1958. [Google Scholar]
- Maull, V.; Solé, R. Network-level containment of single-species bioengineering. Philosophical Transactions of the Royal Society B 2022, 377, 20210396. [Google Scholar] [CrossRef]
- Aparicio, T.; Silbert, J.; Cepeda, S.; de Lorenzo, V. Propagation of Recombinant Genes through Complex Microbiomes with Synthetic Mini-RP4 Plasmid Vectors. BioDesign Research 2022, 2022, 9850305. [Google Scholar] [CrossRef] [PubMed]
- Maull, V.; Aguadé-Gorgorió, G.; de Lorenzo, V.; Solé, R. Synthetic Horizontal Gene Transfer for Ecosystem Restoration. bioRxiv 2025. [Google Scholar] [CrossRef]
- Lenton, T.; Watson, A. Revolutions that made the Earth; OUP Oxford, 2013. [Google Scholar]
- de Lorenzo, V. Environmental Galenics: large-scale fortification of extant microbiomes with engineered bioremediation agents. Philosophical Transactions of the Royal Society B 2022, 377, 20210395. [Google Scholar] [CrossRef] [PubMed]
| 1 | An excellent analysis of this attempt and its many failures can be found in Adam Curtis documentary Machines of loving grace [9]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




