1. Introduction
Transplantation is a life-saving medical procedure that involves the transplantation of cells, tissues, or organs to replace or repair damaged ones. This procedure significantly improves the quality of life in cases of organ failure [
1]. Hematopoietic stem cell transplantation (HSCT), a subset of transplantation therapies, exemplifies this potential, offering hope for patients with malignant and non-malignant diseases [
2]. Stem cells, particularly hematopoietic stem cells (HSCs), play a crucial role in reconstructing the immune system, which also helps to foster immune tolerance and improve post-transplant outcomes [
3]. While advancements in transplant techniques have significantly increased survival rates, they also come with risks. Among the most serious complications is post-transplant lymphoproliferative disorder (PTLD) [
4].
PTLD refers to a heterogeneous group of lymphoid proliferative conditions ranging from benign, self-limiting infectious mononucleosis to aggressive lymphomas [
5]. Usually, after transplantation, patients who experience fever, adenopathy, weight loss, mass lesions, indescribable pain, or dysfunction of the transplanted organ can be recognized as a suspect for PTLD [
6]. Although PTLD in the solid-organ transplant population initiates after 6 months and in HST recipients, 70–90 days, some reports claim that the onset of PTLD can occur even within 1 week or even after 9 years after the transplantation. Symptoms are diverse and may be related to organ dysfunction, mass effect, viremia, or lymphoma-related B symptoms. The reasons for these differences in incidence were not clearly reported; however, depending on the amount of lymphoid tissue present in each organ, immunological reaction, virus, and the immunosuppression treatment, it plausibly may vary [
6,
7].
Despite studies identifying PTLD in various solid organ transplant recipients, the association and prevalence of PTLD in HSCT patients remain unclear. Therefore, in this systematic review and meta-analysis, we determined the incidence of PTLD in HSCT patients along with subgroup analyses of the prevalence of PTLD among different types of HSC transplantation (i.e. allogeneic, autologous, and syngeneic), the prevalence of death cases in HSCT, continent-based analyses of PTLD prevalence in HSCT, and the possible risk factors.
2. Methodology
2.1. Study Design and Selection of Inclusion-Exclusion Criteria
This study followed the PRISMA study design (registration link:
https://osf.io/mp72n), with minor adjustments, and employed a systematic approach to identify relevant studies and conduct a meta-analysis to assess the global prevalence of PTLD among HSCT patients, adhering to established PRISMA guidelines obtained from previous studies [
8,
9,
10]. The study included peer-reviewed articles focusing on the prevalence, incidence, occurrence, or frequency of PTLD in HSCT patients. Excluded materials encompassed narrative or systematic reviews, meta-analyses, book chapters, case reports, editorials, correspondence, conference papers, press releases, and non-original journal articles. Only English-language publications were considered. The study did not involve patients, the public, healthcare institutions, or any third parties in data collection, processing, study design, assessment, analysis, or interpretation of results.
2.2. Search Strategy and Study Inclusion
A comprehensive search strategy was developed to screen relevant studies from three online databases: PubMed, ScienceDirect, and Google Scholar. The search utilized multiple terms such as “prevalence,” “post-transplant lymphoproliferative disorder,” and “hematopoietic stem,” along with their respective related terms or short forms like “incidence”, “epidemiology”, “frequency”; “PTLD”, “bone marrow”, and “transplant”. Boolean operators (AND/OR) were used to refine the search. Specific filters, including “Title/Abstract” for PubMed, “Title, abstract or author-specified keywords” for ScienceDirect, and “allintitle” for Google Scholar, were applied in the advanced search options, following previous studies with slight modifications [
11,
12]. No publication date filters were used. Duplicate search results were carefully managed and excluded from the study.
2.3. Data Extraction, Meta-Analysis and Heterogeneity
The authors independently conducted the initial search and necessary data extraction from the included studies, subsequently validating their findings through discussion. Primarily, the data of PTLD cases and the number of HSCT patients were obtained from each of the individual studies. The extracted information encompassed basic study characteristics such as study ID, study type, HSCT participants number, male and female numbers, age, HSCT type, PTLD type, and study region. Any disagreements regarding data inclusion or exclusion were carefully resolved by the authors, and they addressed missing or unclear information by contacting the corresponding or first author of the study.
The authors employed a meta-analysis approach to ascertain the pooled prevalence of PTLD in HSCT patients, utilizing the collected data. A random-effects model with a 95% confidence interval (CI) was employed to analyze the pooled prevalence. The I2 statistics method was utilized to estimate the pooled heterogeneity of the included studies. The I2 values indicated the level of heterogeneity among the studies, with 25-50% suggesting low heterogeneity, 51-75% indicating moderate heterogeneity, and values exceeding 75% signifying high heterogeneity. Furthermore, outlier studies and sensitivity analysis were implemented, based on previously established methodologies to explore potential outliers and their influence on the overall effect size [
13,
14]. The outlier determination and sensitivity analyses were conducted sequentially through the primary exclusion of outlier studies, identified using funnel and Galbraith plots, and subsequently the reanalysis of the primary meta-plot employing a random-effects model. The meta-analysis, forest plot, funnel plot, and Galbraith plot construction were executed using RStudio software (version 4.3.0) and the “metafor” package (version 4.2-0) of R.
2.4. Evaluation of Study Quality and Analysis of Bias Risk
The researchers evaluated the quality of the included studies using a set of nine questions derived from the Study Quality Assessment Tools, NIH, and Systematic Reviews: Step 6: Assess Quality of Included Studies, UNC [
15,
16]. In this study, the selected questions could be answered with “Yes”, “No”, “Unclear”, “Not Reported” (NR), or “Not Applicable” (NA), which were then converted to numerical scores (e.g., 1 for Yes, 0 for No and Unclear, no score for NR and NA). For each study, these scores were totaled and divided by the number of questions (9 in this case), then converted to a percentage. The resulting percentage indicated the study’s quality and potential publication bias risk: <50% suggested low quality and high bias risk, 50-70% indicated moderate quality, and >80% signified high quality and low bias risk. The scoring method was adapted from previous research with minor modifications [
17,
18].
2.5. Subgroup Analyses and Risk Factors
To evaluate the PTLD prevalence among HSCT patients based on the type of PTLD, death cases, and continent separate subgroup analyses were conducted. Plausible risk factors of the PTLD were also assessed.
3. Results
3.1. Search Results and Study Inclusion
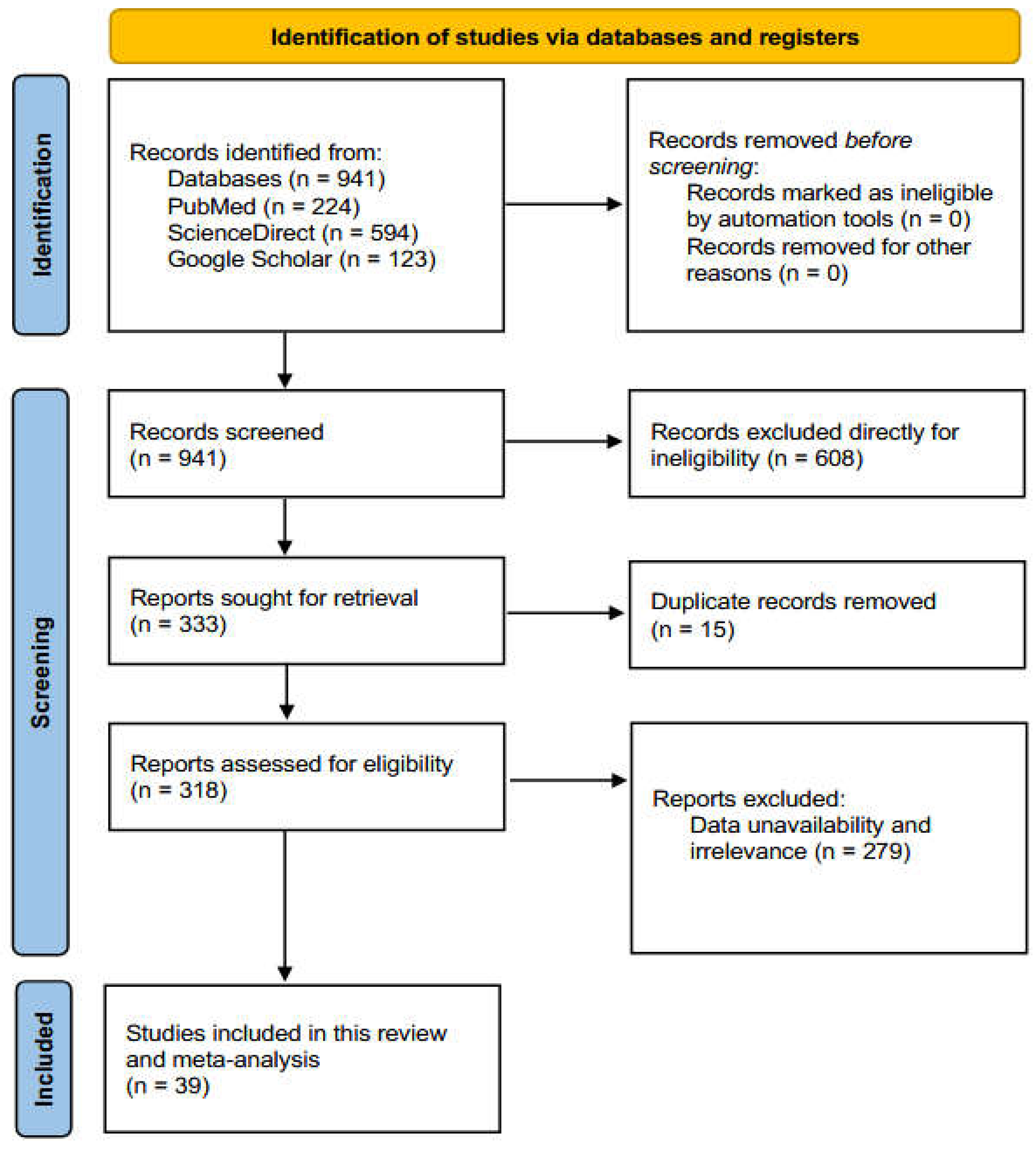
Initially, a total of 941 search results were identified from three electronic databases: PubMed (n=224), ScienceDirect (n=594), and Google Scholar (n=123). These results were obtained through our search strategies, applied keywords, and various filters utilized during the searches. During the study screening phase, 608 articles were directly excluded. These articles included reviews, systematic reviews, mini-reviews, meta-analyses, editorials, case reports, and correspondence, excluding only the full-text original research articles. From the remaining 333 studies, 15 were excluded due to study duplication.
Out of the 50 articles selected for evaluation, after rigorous screening, validation, and excluding studies (n=279) that were deemed irrelevant to the study’s aims and objectives, 39 studies were selected for inclusion in the systematic review and meta-analysis. These studies met the eligibility and inclusion criteria (Figure 1).
Figure 1.
A simplified PRISMA flow diagram of methodology.
Figure 1.
A simplified PRISMA flow diagram of methodology.
3.2. Major Characteristics of the Included Studies
The authors carefully extracted and validated major characteristic data of the included articles (n=39) that completely met the criteria of the current systematic review and meta-analysis [
19,
20,
21,
22,
23,
24,
25,
26,
27,
28,
29,
30,
31,
32,
33,
34,
35,
36,
37,
38,
39,
40,
41,
42,
43,
44,
45,
46,
47,
48,
49,
50,
51,
52,
53,
54,
55,
56,
57]. The 39 included studies were conducted in different countries of different continents including Europe (i.e. Portugal, Italy, Germany, Switzerland, Sweden, Spain, France, Poland, and Denmark), Asia (i.e. India, Japan, China, Korea, and Turkey), and North America (i.e. Canada and USA). Among HSCT types allogenic, autologous, and syngeneic were reported. Among the types of PTLD, such as Monomorphic PTLD (M-PTLD), Polymorphic-PTLD (P-PTLD), Plasmacytic hyperplasia PTLD (PH-PTLD), Classic Hodgkin lymphoma PTLD (CHL-PTLD), Epstein–Barr virus PTLD (EBV-PTLD), and Early lesion PTLD (EL-PTLD). Besides, the type of the studies was cross-sectional or cohort. The number of total participants along with male, and female numbers and their age details were reported in detail in
Table 1.
3.3. Primary Meta-Analysis, Heterogeneity, outliers and Sensitivity
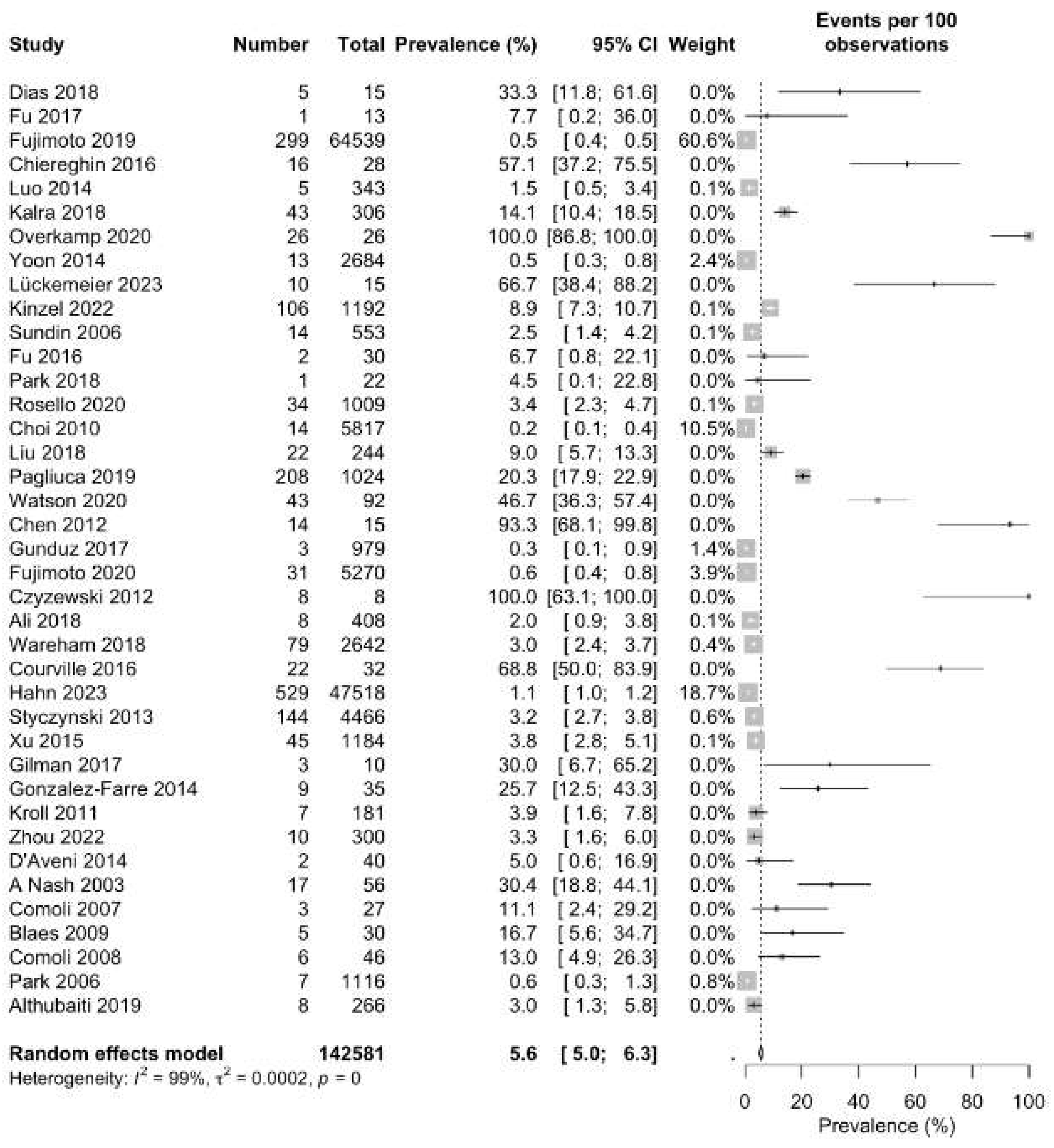
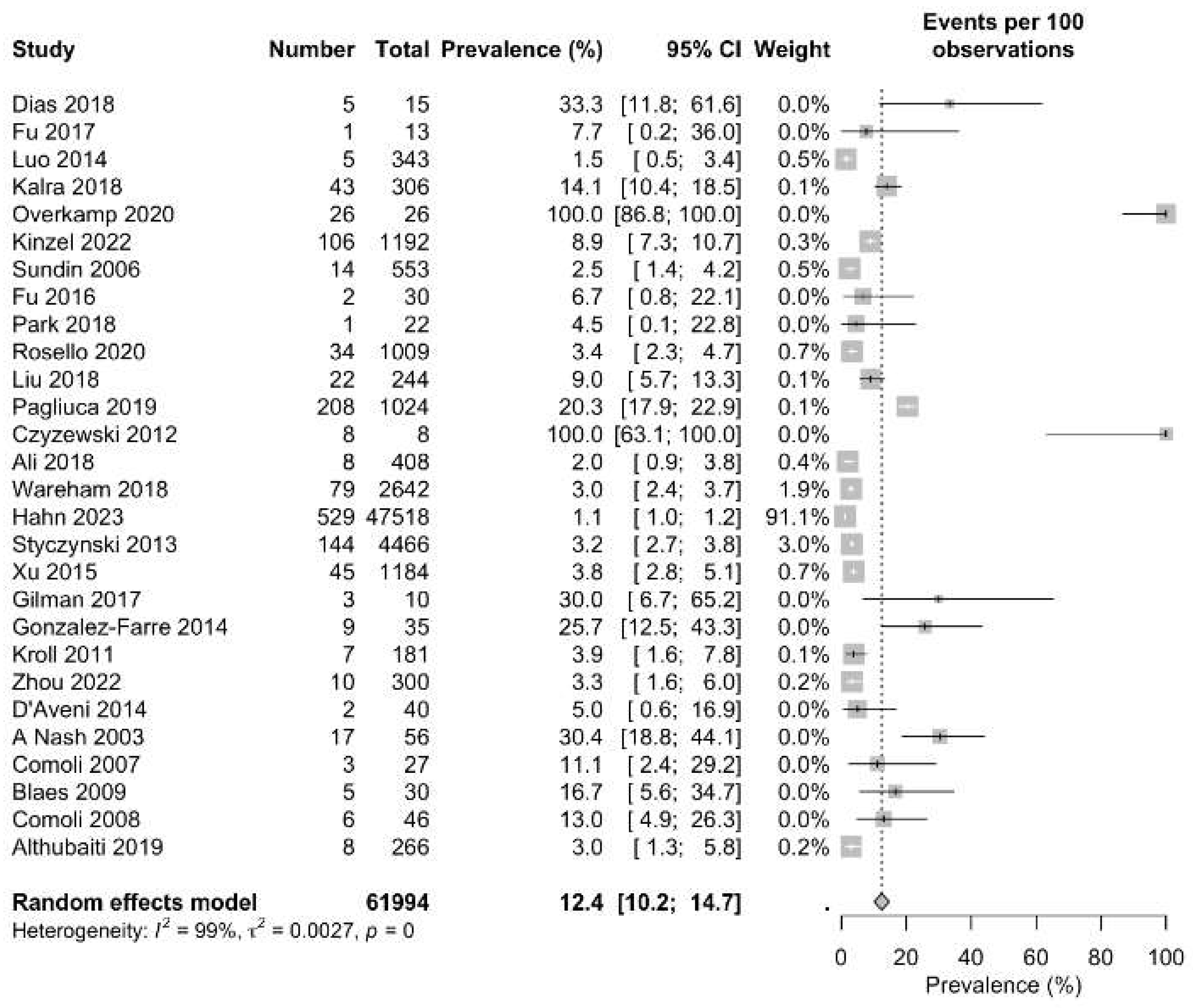
The primary objective of our meta-analysis was to determine the pooled prevalence of PTLD in HSCT patients globally. As a result, 1822 PTLD cases were reported out of 142581 HSCT patients. Based on data extracted from the included studies, an overall pooled prevalence of 5.6% (95% CI: 5.0 to 6.3) with a substantial heterogeneity of 99% (p=0) was found. The highest prevalence was found in Overkamp 2020 (100%, (95% CI: 86.8 to 100.0)) and Czyzewski 2012 (100%, (95% CI: 63.1 to 100.0)) whereas the lowest prevalence was found in Choi 2010 (0.2% (95%CI: 0.1 to 0.4)) (Figure 2).
Figure 2.
Forest plot of the pooled prevalence of PTLD in patients after hematopoietic stem cell transplantation.
Figure 2.
Forest plot of the pooled prevalence of PTLD in patients after hematopoietic stem cell transplantation.
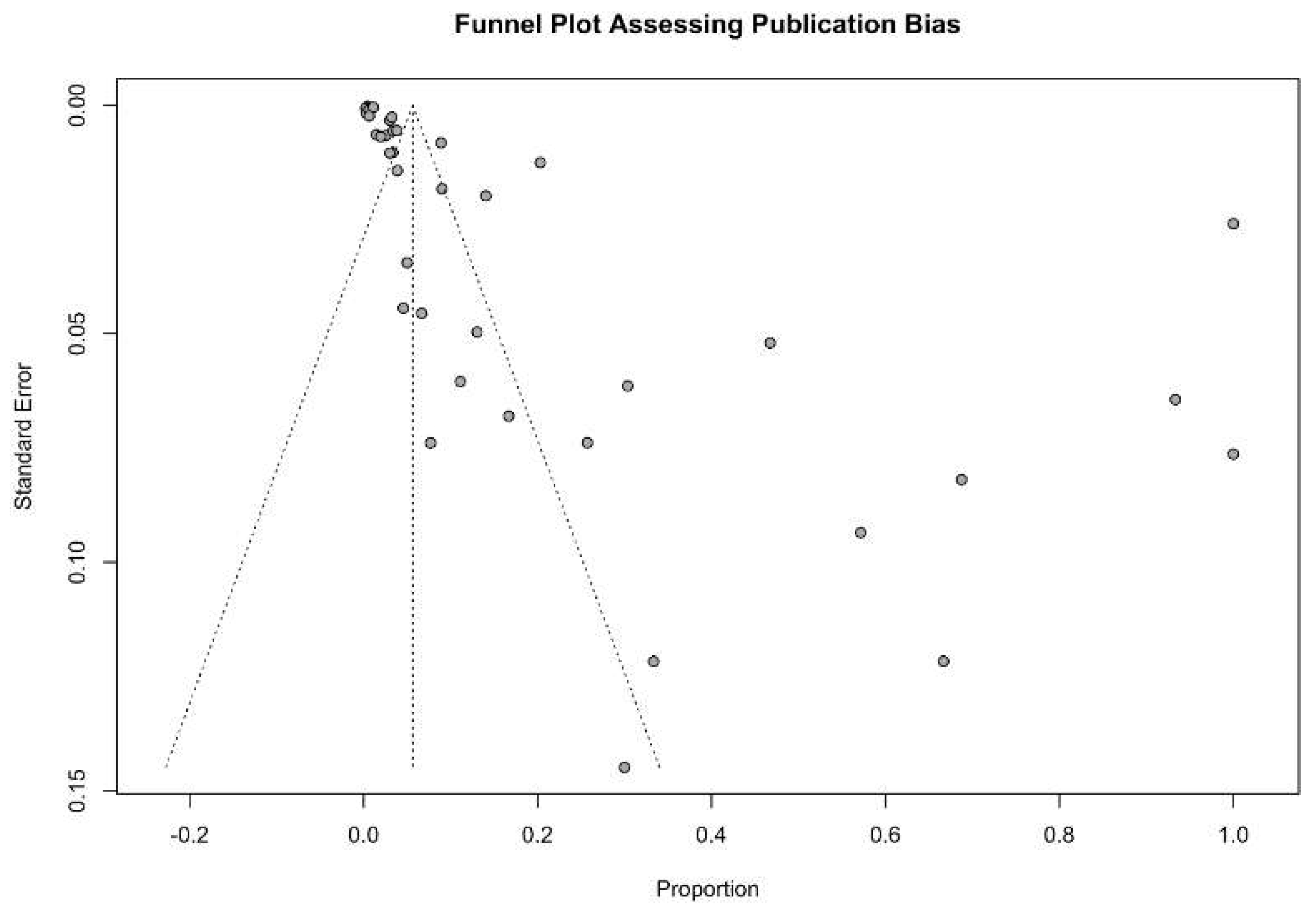
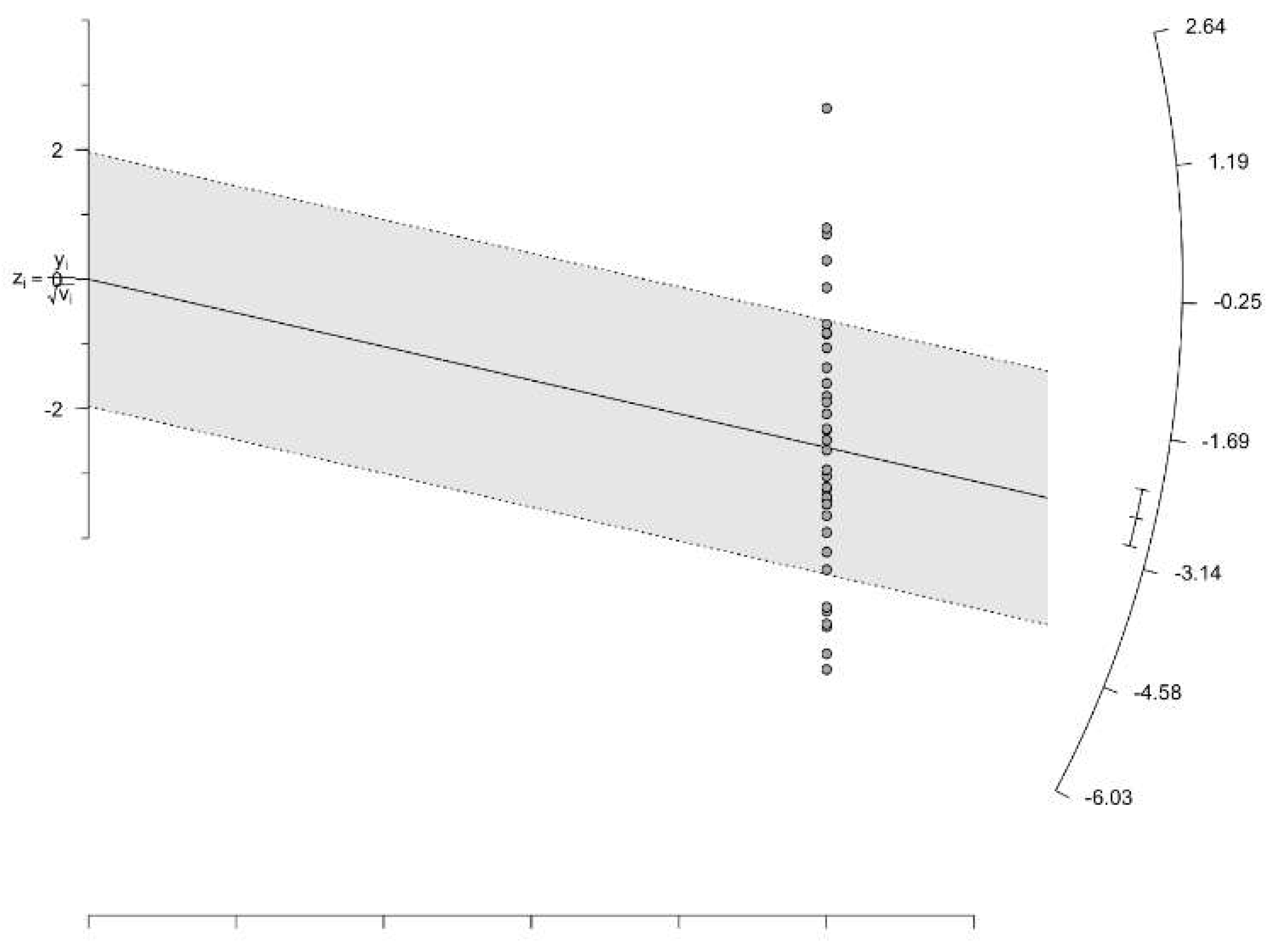
A funnel plot constructed for visualization of the possible sources of publication bias indicated multiple potential outlier studies with high risks of bias (Figure 3). The Galbraith plot which was constructed for visual investigation and confirmation of the potential sources of bias indicated 11 outlier studies (i.e. Chen 2012, Courville 2016, Luckemeier 2023, Chiereghin 2016, Watson 2020, Park 2006, Fujimoto 2020, Yoon 2014, Fujimoto 2019, Gunduz 2017, and Choi 2010) as shown in Figure 6 (Figure 4).
After excluding the outliers, the forest plot was reconstructed using a random effect model to identify the sensitivity. As a result, the prevalence was enhanced to 12.4% (95%CI: 10.2 to 14.7) confirming the primary outcome to be accurate (Figure 5).
Figure 3.
Funnel plot assessing the plausible publication biases of included studies for the prevalence of PTLD in patients after hematopoietic stem cell transplantation.
Figure 3.
Funnel plot assessing the plausible publication biases of included studies for the prevalence of PTLD in patients after hematopoietic stem cell transplantation.
Figure 4.
Galbraith’s plot assessing the outlier studies for the prevalence of PTLD in patients after hematopoietic stem cell transplantation.
Figure 4.
Galbraith’s plot assessing the outlier studies for the prevalence of PTLD in patients after hematopoietic stem cell transplantation.
Figure 5.
Forest plot excluding the outlier studies of the prevalence of PTLD in patients after hematopoietic stem cell transplantation.
Figure 5.
Forest plot excluding the outlier studies of the prevalence of PTLD in patients after hematopoietic stem cell transplantation.
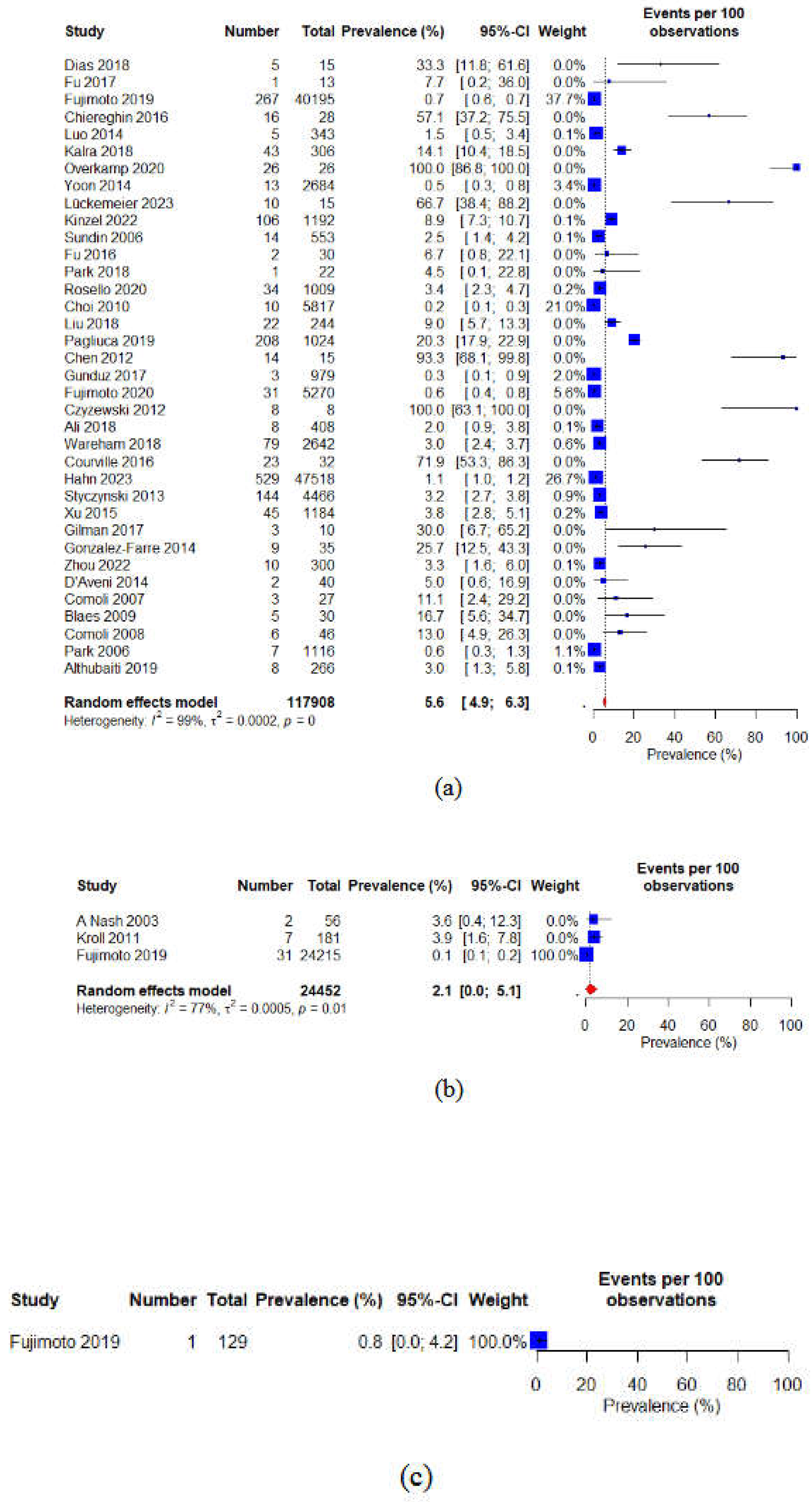
Figure 6.
Forest plot of the prevalence of PTLD in patients after (a) allogeneic (b) autologous and (c) syngeneic hematopoietic stem cell transplantation.
Figure 6.
Forest plot of the prevalence of PTLD in patients after (a) allogeneic (b) autologous and (c) syngeneic hematopoietic stem cell transplantation.
3.4. Assessment of Study Quality and Risk of Bias
Based on the answers to our specified set of quality assessment questions and subsequent scoring strategy, 33 studies scored more than 80% indicating high-quality studies and 6 studies scored more than 50% resembling moderate-quality studies. Interestingly, 17 studies obtained a 100% score. No study was determined to be scored less than 50%, hence there were no low-quality studies with a high-risk of bias (
Table 2).
3.5. Subgroup Analysis and Risk Factors
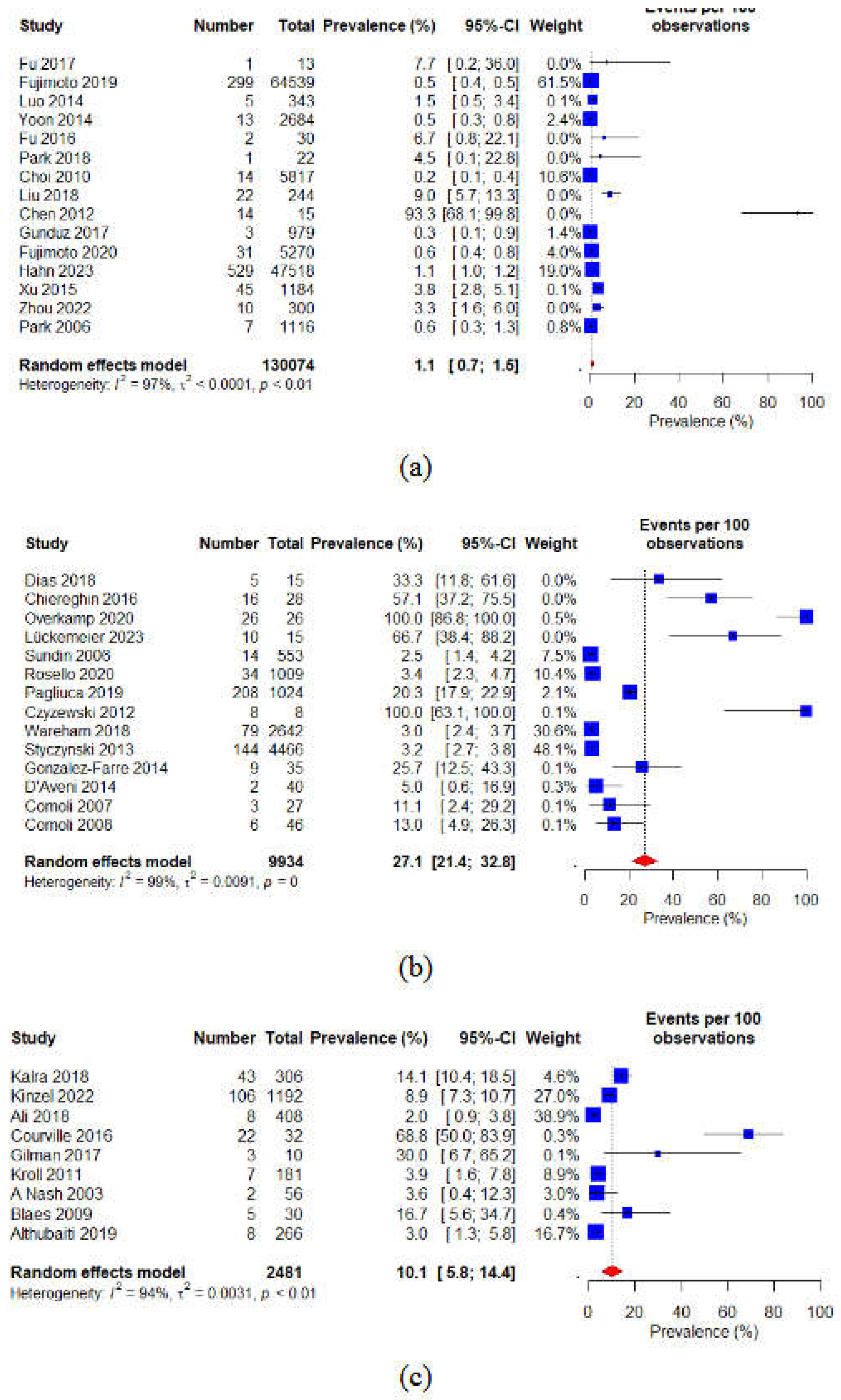
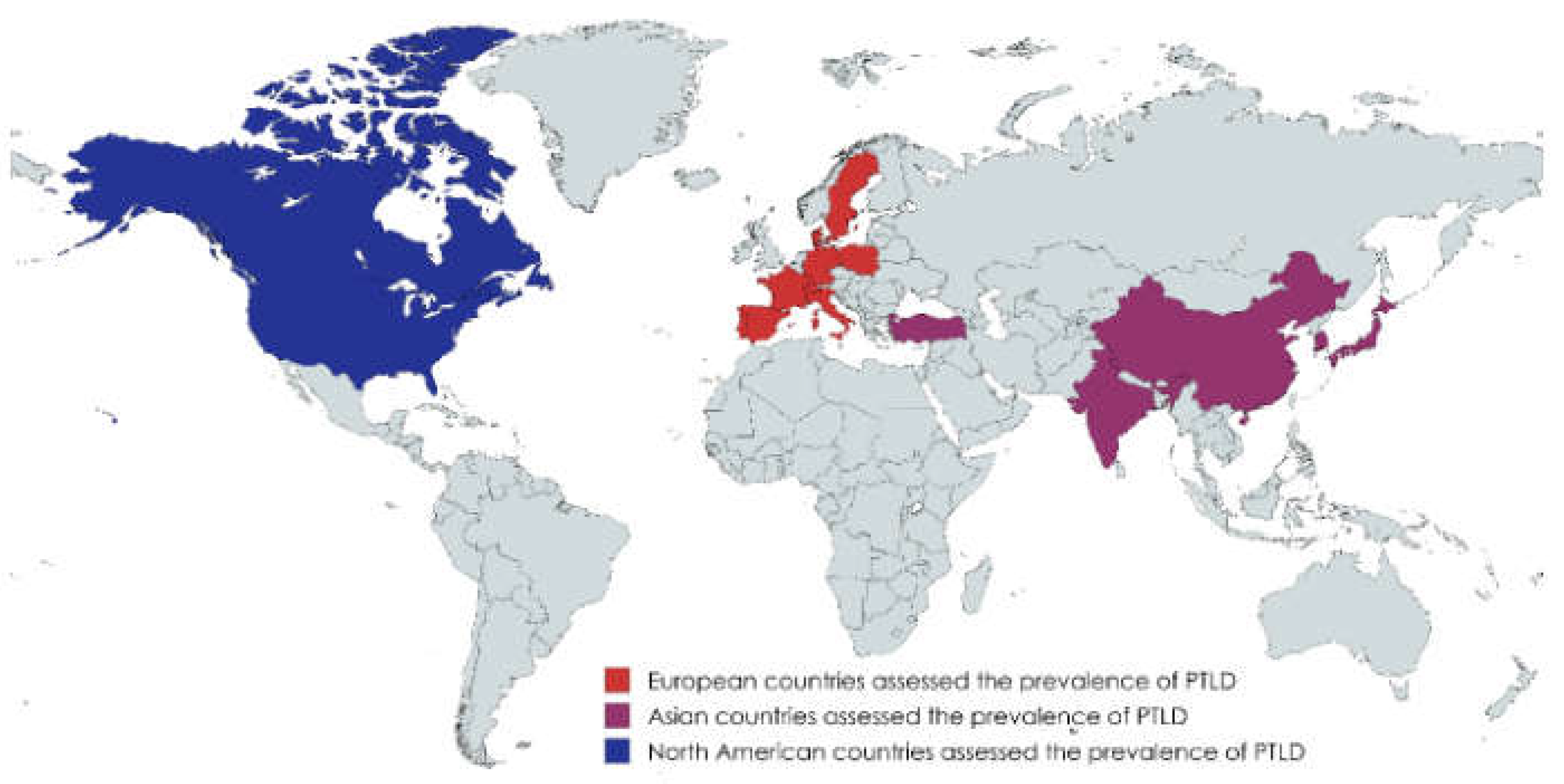
Subgroup analysis was aimed to investigate the pooled prevalence of PTLD in HSCT patients based on the type of HSCT, overall death cases, and patients across different continents. For HSCT type, the highest pooled prevalence was found in allogeneic HSCT (5.6% (95% CI: 4.9 to 6.3)) followed by the autologous (2.1% (95%CI: 0.0 to 5.1)) and syngeneic (0.8% (95%CI: 0.0 to 4.2)) HSCT (Figure 6). The prevalence of death cases among the overall HSCT patients was found 0.6% (95%CI: 0.4 to 0.9) (Figure 7). Regarding the continent-based subgroup analysis, Asia was found to have the lowest prevalence rate (1.1% (95%I: 0.7 to 1.5)), followed by North America (10.1% (95%I: 5.8 to 14.4)); whereas, Europe had the highest prevalence rate of PTLD among HSCT patients (27.1% (95%I: 21.4 to 32.8)) (Figure 8). The geographical locations of the countries within different continents are demonstrated in detail in Figure 9.
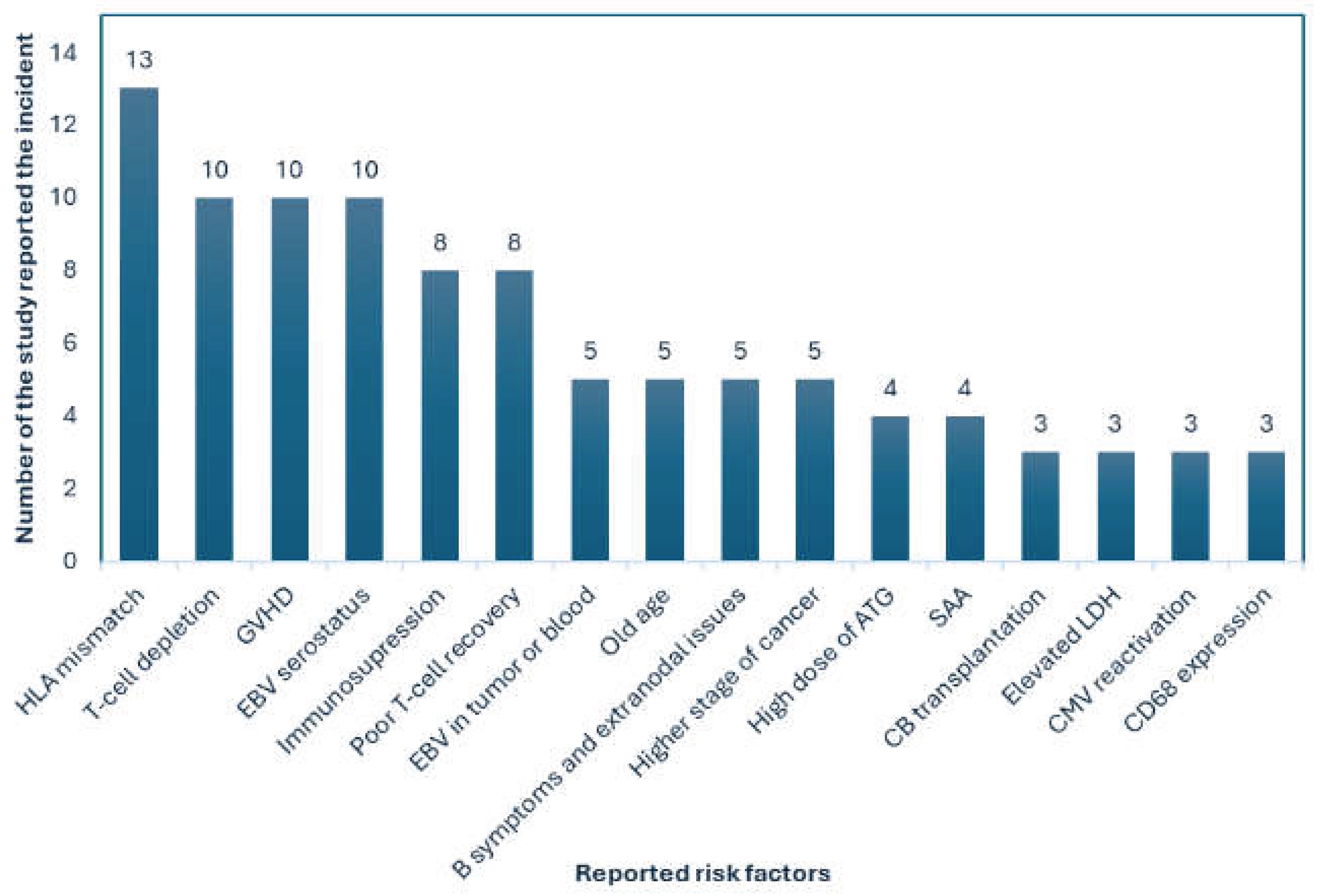
Plausible reported risk factors were also obtained from the included studies. As a result, we observed that HLA mismatch was the highest reported risk factor of PTLD in HSCT patients, followed by the T-cell depletion, Graft-versus-host disease, and EBV seroprevalence or serostatus. All the plausible risk factors are presented in Figure 10.
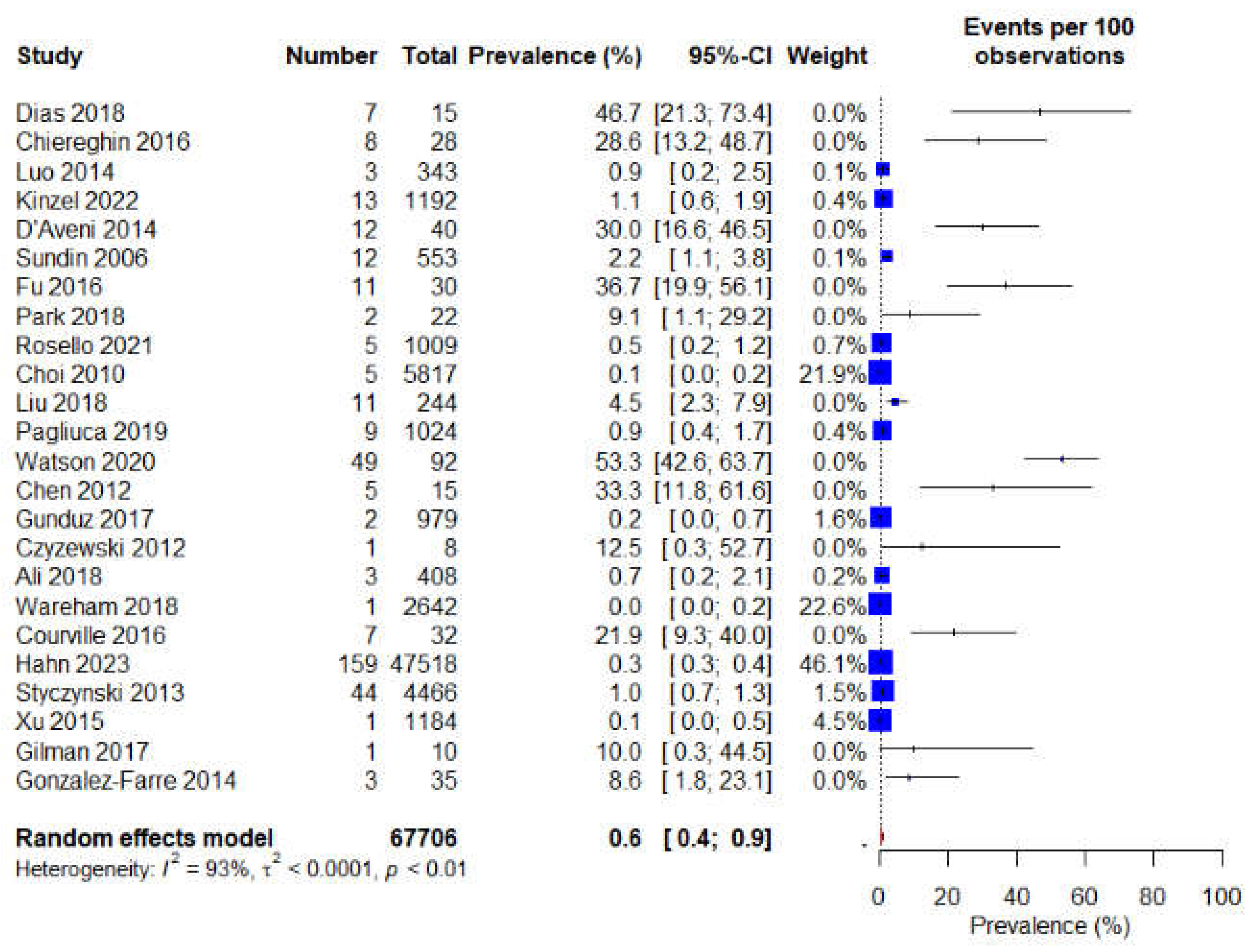
Figure 7.
Forest plot of the prevalence of reported death cases in HSCT patients.
Figure 7.
Forest plot of the prevalence of reported death cases in HSCT patients.
Figure 8.
Forest plot of the pooled prevalence of PTLD in patients after HSTC in (a) Asia (b) Europe and (c) North America.
Figure 8.
Forest plot of the pooled prevalence of PTLD in patients after HSTC in (a) Asia (b) Europe and (c) North America.
Figure 9.
Geographical locations of the studies reported the prevalence of PTLD in HSTC patients.
Figure 9.
Geographical locations of the studies reported the prevalence of PTLD in HSTC patients.
Figure 10.
Plausible risk factors of PTLD reported in different included studies. Here, GVHD= Graft-versus-host disease, EBV= Epstein-Barr virus, ATG= Antithymocyte Globulin, SAA= severe aplastic anemia, CB= cord blood, LDH= lactate dehydrogenase, CMV= Cytomegalovirus.
Figure 10.
Plausible risk factors of PTLD reported in different included studies. Here, GVHD= Graft-versus-host disease, EBV= Epstein-Barr virus, ATG= Antithymocyte Globulin, SAA= severe aplastic anemia, CB= cord blood, LDH= lactate dehydrogenase, CMV= Cytomegalovirus.
4. Discussion
To the best of our knowledge, this is the individual meta-analysis to investigate the global prevalence of PTLD in patients undergoing HSCT. By analyzing data from diverse regions, we provide new insights into the prevalence of PTLD with HSCT, highlighting significant epidemiological and clinical trends. Our findings determined that the pooled prevalence rate of PTLD among HSCT was 5.6% (95% CI: 5.0 to 6.3), which indicates that it is a critical concern for HSCT patients, and they need to be carefully handled after the transplantation to restrict PTLD as the prevalence rate is quite high (Figure 2). Interestingly, after excluding the plausible outlier studies the prevalence rate of PTLD rose to 12.4% (95%CI: 10.2 to 14.7) which further proves it is to be even more serious issue for HSCT patients (Figure 5). According to previous reports, the incidence of PTLD varies depending on the type of transplantation such as 19% for intestinal transplants, 2-10% for heart transplants, 5%–9% for heart-lung transplants, and 2%–8% for liver transplants [
7]. Therefore, other than PTLD in intestinal transplant patients, every other reported transplantation was investigated to have a lower prevalence of PTLD.
In subgroup analyses, we initially investigated that allogenic HSCT was the most prevalent transplantation type as compared to autologous and syngeneic HSCT. However, the prevalence of PTLD was also higher in the allogenic HSCT (5.6% (95%CI: 4.9 to 6.3)) as compared to autologous (2.1% (95%CI: 0.0 to 5.1)) and syngeneic (0.8% (95%CI: 0.0 to 4.2)) HSCT (Figure 6). These differences in PTLD among autologous, syngeneic, and allogeneic HSCT highlight the need for tailored transplant approaches. Autologous HSCT, which uses the patient’s own stem cells, eliminates concerns about donor compatibility and reduces the risk of graft-versus-host disease (GVHD) [
58]. Our findings indicate that autologous HSCT recipients have substantially lower rates of PTLD, with near-zero post-transplant mortality (Figures 6 and7). Syngeneic HSCT on the other hand, performed between identical twins, shows promise but remains underexplored, with limited data available on its efficacy and long-term outcomes [
59,
60]. The prevalence of death cases was less than 1% (i.e. 0.6% (95%CI: 0.4 to 0.9)) among the overall HSCT patients, where not all the patients had PTLD (Figure 7). This phenomenon showed that besides PTLD other risk factors can create critical conditions for patients and can lead to death. Physiological conditions such as age, weakness, comorbidity such as diabetes, cardiovascular diseases, and psychological complications such as anxiety, and depression were some factors that may lead to death [
61,
62,
63].
Depending on the study region, we did a separate subgroup analysis, observing that the studies were from three different continents: Asia, Europe, and North America. Interestingly, we found that the Asian region had the lowest PTLD rate (1.1% (95%I: 0.7 to 1.5)) compared to the European (27.1% (95%I: 21.4 to 32.8)) and North American (10.1% (95%I: 5.8 to 14.4)) regions, respectively (Figures 8 and 9). Plausible risk factors or other physiological conditions and comorbidities may impact these changes in the prevalence rate. Besides, we observed that no autologous HSCT was done in Europe, which may also had an impact on its higher prevalence rate. On the other hand, among the risk factors we determined from our included articles, HLA mismatch was the most prevalent, followed by T-cell depletion, GVHD, and EBV infection. Besides, immunosuppression, poor T-cell recovery, presence of EBV in tumor/blood, old age, B symptoms (i.e. sweats, pyrexia, and weight loss), and extranodal involvements (i.e. lung, bone marrow, gastrointestinal tract (GIT), skin, and central nervous system (CNS)), presence of high stage of cancer, high dose of Antithymocyte Globulin (ATG), severe aplastic anemia (SAA), cord blood (CB) transplantation, elevated level of lactate dehydrogenase (LDH), cytomegalovirus (CMV) reactivation, and CD68 expression are some other reported risk factors. Previous studies also support these risk factors to be crucial [
64,
65]. It was also reported that the development of PTLD is closely linked to immunosuppression, particularly the use of ATG, ex vivo T-cell depletion, and mismatched or unrelated donors [
66]. These factors weaken the immune system’s ability to control EBV-driven B-cell proliferation, significantly increasing the risk of PTLD. Umbilical cord blood grafts further elevate PTLD risk due to their lower T-cell maturity and count [
67].
Advances in transplant techniques, such as improved T-cell depletion protocols, have reduced traditional complications but introduced new challenges, including EBV-related PTLD. Reduction of immunosuppression (RI) remains the first-line treatment for PTLD, aiming to restore CTL function while minimizing allograft rejection. For cases with CD20-positive PTLD, rituximab, and anti-CD20 monoclonal antibodies have shown promising results, achieving remission rates of 40–60% when combined with chemotherapy. However, treatment outcomes vary widely, reflecting the heterogeneity of PTLD. The increasing incidence of PTLD in recent decades highlights the evolving landscape of transplantation. Novel therapies, including chimeric antigen receptor (CAR) T cells targeting CD19 and EBV-specific adoptive T-cell therapies, are being explored as potential game-changers in PTLD management. Factors such as expanded use of HSCT, potent immunosuppressive drugs, and improved diagnostic techniques have contributed to this trend. However, these advances also present opportunities for early detection and intervention, which are critical for improving survival rates [
64,
68,
69,
70]. Nevertheless, routine checks and follow-ups are crucial for diagnosing and managing PTLD, enabling clinicians to initiate preemptive therapies before the onset of clinical symptoms.
5. Limitations
Data from general search engines, news portals, and blogs were excluded from this analysis. Only data from published articles was included.
6. Conclusions
This study represents a significant advancement in the understanding of the prevalence and association of PTLD in HSCT patients. Our findings underscore the imperative of early diagnosis, personalized treatment approaches, and sustained research into the mechanisms underlying PTLD. By addressing these challenges, favorable outcomes for HSCT recipients and the advancement of transplantation medicine can be realized.
Author Contributions
Conceptualization, S.S.K., and D.Z.I.; methodology, S.S.K., A.H.P., A.R.J., M.Z., A.S., and D.S.; software, E.S., S.S.K., R.B. and A.N.M.M-O-R; writing—original draft preparation, A.H.P., A.R.J., M.Z., A.S., D.S., D.Z.I., E.S. and S.S.K.; writing—review and editing, S.S.K., M.A.I., R.B. and A.N.M.M-O-R; visualization, E.S., S.S.K. and M.A.I.; supervision, S.S.K., M.A.I, R.B. and A.N.M.M-O-R. All authors have agreed on the journal to which the article has been submitted and have read and agreed to the published version of the manuscript.
Funding
There are no relevant financial or non-financial competing interests to report.
Data Availability Statement
All data analyzed in this study were obtained from previously published studies and publicly available sources. No new data were generated for this meta-analysis. The datasets supporting the conclusions of this article are available from the corresponding author upon reasonable request.
Conflicts of Interest
No potential competing interest was reported by the author(s). There are no relevant financial or non-financial competing interests to report.
Declaration: This study is not a clinical trial. It is a meta-analysis where secondary data analysis was done. Therefore, no registry, trial registration number, or data of registration was required.
Ethics, Consent to Participate, and Consent to Publish Declarations: Not applicable.
References
- Ullah, A.
Post-transplant Lymphoproliferative Disorder (PTLD) in the US Population: Demographics, Treatment Characteristics, and Survival Analysis
. Cureus 2023, 15(5). [Google Scholar] [CrossRef]
- Sullivan, K.M.; Parkman, R.; Walters, M.C.
Bone marrow transplantation for non-malignant disease
. ASH Education Program Book 2000, 2000(1), 319–338. [Google Scholar]
- Elahimehr, R.; Scheinok, A.T.; McKay, D.B.
Hematopoietic stem cells and solid organ transplantation
. Transplantation reviews 2016, 30(4), 227–234. [Google Scholar] [CrossRef]
- Dierickx, D.; Habermann, T.M.
Post-transplantation lymphoproliferative disorders in adults
. New England Journal of Medicine 2018, 378(6), 549–562. [Google Scholar] [CrossRef] [PubMed]
- Zaffiri, L.
Incidence and outcome of post-transplant lymphoproliferative disorders in lung transplant patients: analysis of ISHLT Registry
. The Journal of Heart and Lung Transplantation 2020, 39(10), 1089–1099. [Google Scholar] [CrossRef]
- Loren, A.
Post-transplant lymphoproliferative disorder: a review
. Bone marrow transplantation 2003, 31(3), 145–155. [Google Scholar] [CrossRef] [PubMed]
- Shroff, R.; Rees, L.
The post-transplant lymphoproliferative disorder—a literature review
. Pediatric Nephrology 2004, 19, 369–377. [Google Scholar] [CrossRef]
- Khandker, S.S.
Diagnostic accuracy of rapid antigen test kits for detecting SARS-CoV-2: a systematic review and meta-analysis of 17,171 suspected COVID-19 patients
. Journal of clinical medicine 2021, 10(16), 3493. [Google Scholar] [CrossRef] [PubMed]
- Khandker, S.S.
A systematic review on COVID-19 vaccine strategies, their effectiveness, and issues
. Vaccines 2021, 9(12), 1387. [Google Scholar] [CrossRef]
- Islam, M.A.
Immunomodulatory effects of diet and nutrients in systemic lupus erythematosus (SLE): a systematic review
. Frontiers in immunology 2020, 11, 1477. [Google Scholar] [CrossRef]
- Islam, M.A.
Prevalence of antiphospholipid antibodies in Behçet’s disease: a systematic review and meta-analysis
. PLoS One 2020, 15(1), e0227836. [Google Scholar] [CrossRef]
- Jamiruddin, M.R.
Microfluidics technology in SARS-CoV-2 diagnosis and beyond: a systematic review
. Life 2022, 12(5), 649. [Google Scholar] [CrossRef] [PubMed]
- Zahra, S.
Prevalence of nephropathy among diabetic patients in North American region: a systematic review and meta-analysis
. Medicine 2024, 103(38), p. e39759. [Google Scholar] [CrossRef]
- Shabbir, N.A.
Prevalence of HIV/AIDS among pregnant women in North American region: A systematic review and meta-analysis
. Medicine 2024, 103(44), p. e40339. [Google Scholar] [CrossRef] [PubMed]
- NIH. Study Quality Assessment Tools. 2021 [cited 2024 22 November]; Available from: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools. 2021.
- UNC. Systematic Reviews: Step 6: Assess Quality of Included Studies. 2023 [cited 2024 22 November]; Available from: https://guides.lib.unc.edu/systematic-reviews/assess-quality.
- Mughal, H.B.
Brain natriuretic peptide in acute heart failure and its association with glomerular filtration rate: A systematic review and meta-analysis
. Medicine 2024, 103(8), p. e36933. [Google Scholar] [CrossRef]
- Khandker, S.S.
Association between glomerular filtration rate and β-thalassemia major: a systematic review and meta-analysis
. Thalassemia Reports 2023, 13(3), 195–205. [Google Scholar] [CrossRef]
- Marinho-Dias, J.
Post-transplant lymphoproliferative disorder in hematopoietic stem cell transplant patients: A single center retrospective study between 2005 and 2012
. Molecular Medicine Reports 2018, 18(5), 4650–4656. [Google Scholar] [CrossRef]
- Fu, L.
Monomorphic post-transplant lymphoproliferative disorder after kidney transplantation and hematopoietic stem cell transplantation: clinicopathological characteristics, treatments and prognostic factors
. Indian Journal of Hematology and Blood Transfusion 2017, 33, 492–499. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, A.
Risk factors and predictive scoring system for post-transplant lymphoproliferative disorder after hematopoietic stem cell transplantation
. Biology of Blood and Marrow Transplantation 2019, 25(7), 1441–1449. [Google Scholar] [CrossRef]
- Chiereghin, A.
Prospective Epstein–Barr virus
-related post
-transplant lymphoproliferative disorder prevention program in pediatric allogeneic hematopoietic stem cell transplant: virological monitoring and first
-line treatment
. Transplant Infectious Disease 2016, 18(1), 44–54. [Google Scholar] [CrossRef]
- Luo, L.
Post-transplant lymphoproliferative disease after allogeneic hematopoietic stem cell transplantation: a single-center experience
. Ann Transplant 2014, 19, 6–12. [Google Scholar]
- Kalra, A.
Epstein-barr virus DNAemia monitoring for the management of post-transplant lymphoproliferative disorder
. Cytotherapy 2018, 20(5), 706–714. [Google Scholar] [CrossRef]
- Overkamp, M.
Comparative analysis of post-transplant lymphoproliferative disorders after solid organ and hematopoietic stem cell transplantation reveals differences in the tumor microenvironment
. Virchows Archiv 2021, 478, 1135–1148. [Google Scholar] [CrossRef]
- Yoon, J.H.
Comparative analysis of post
-transplant lymphoproliferative disorder after kidney transplantation versus hematopoietic stem cell transplantation
. Transplant International 2014, 27(7), 721–732. [Google Scholar] [CrossRef]
- Office, F.P.
Erratum: Characterization and outcome of post-transplant lymphoproliferative disorders within a collaborative study
. Frontiers in Oncology 2023, 13, 1290218. [Google Scholar] [CrossRef]
- Kinzel, M.
Risk factors for the incidence of and the mortality due to post-transplant lymphoproliferative disorder after hematopoietic cell transplantation
. Transplantation and Cellular Therapy 2022, 28(1), 53. e1–53. e10. [Google Scholar] [CrossRef]
- Sundin, M.
The role of HLA mismatch, splenectomy and recipient Epstein-Barr virus seronegativity as risk factors in post-transplant lymphoproliferative disorder following allogeneic hematopoietic stem cell transplantation
. Haematologica 2006, 91(8), 1059–1067. [Google Scholar] [PubMed]
- Fu, L.
Allogeneic hematopoietic stem-cell transplantation for adult and adolescent hemophagocytic lymphohistiocytosis: a single center analysis
. International journal of hematology 2016, 104, 628–635. [Google Scholar] [CrossRef] [PubMed]
- Park, B.-K.
Allogeneic hematopoietic stem cell transplantation in congenital hemoglobinopathies with myeloablative conditioning and rabbit anti-thymocyte globulin
. Blood research 2018, 53(2), p. 145. [Google Scholar] [CrossRef] [PubMed]
- Balaguer-Rosello, A.
Central nervous system involvement in Epstein–Barr virus-related post-transplant lymphoproliferative disorders after allogeneic hematopoietic stem cell transplantation
. Transplantation and Cellular Therapy 2021, 27(3), 261. e1–261. e7. [Google Scholar] [CrossRef]
- Choi, J.-H.
Clinical characteristics of monomorphic post-transplant lymphoproliferative disorders
. Journal of Korean medical science 2010, 25(4), 523–526. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.-X.
Clinical Characteristics, Treatment and Prognosis of PTLD after allogeneic Hematopoietic Stem Cell Transplantation
. Zhongguo shi yan xue ye xue za zhi 2018, 26(2), 535–540. [Google Scholar] [PubMed]
- Pagliuca, S.
Epstein-Barr virus-associated post-transplantation lymphoproliferative disease in patients who received anti-CD20 after hematopoietic stem cell transplantation
. Biology of Blood and Marrow Transplantation 2019, 25(12), 2490–2500. [Google Scholar] [CrossRef]
- Watson, C.
Retrospective database analysis of healthcare resource utilization and costs in patients who develop post-transplant lymphoproliferative disease within the first year following allogeneic hematopoietic stem cell transplants
. Journal of Medical Economics 2020, 23(10), 1159–1167. [Google Scholar] [CrossRef]
- Chen, D.
Post-transplant lymphoproliferative disorder: a clinicopathologic study of 15 cases
. Zhonghua Bing li xue za zhi= Chinese Journal of Pathology 2012, 41(9), 607–612. [Google Scholar]
- Gündüz, M.
Subsequent malignancies after allogeneic hematopoietic stem cell transplantation
. Clinical Transplantation 2017, 31(7), p. e12987. [Google Scholar] [CrossRef]
- Fujimoto, A.
Low incidence of posttransplant lymphoproliferative disorder after allogeneic stem cell transplantation in patients with lymphoma treated with rituximab
. Hematological Oncology 2020, 38(2), 146–152. [Google Scholar] [CrossRef]
- Czyzewski, K.
Intrathecal therapy with rituximab in central nervous system involvement of post-transplant lymphoproliferative disorder
. Leukemia & Lymphoma 2013, 54(3), 503–506. [Google Scholar]
- Ali, S.
Hemophagocytic lymphohistiocytosis is a sign of poor outcome in pediatric Epstein
-Barr virus
-associated post
-transplant lymphoproliferative disease after allogeneic hematopoietic stem cell transplantation
. Pediatric transplantation 2019, 23(1), p. e13319. [Google Scholar] [CrossRef] [PubMed]
- Wareham, N.E.
The value of EBV DNA in early detection of post-transplant lymphoproliferative disorders among solid organ and hematopoietic stem cell transplant recipients
. Journal of Cancer Research and Clinical Oncology 2018, 144, 1569–1580. [Google Scholar] [CrossRef]
- Courville, E.L.
EBV-negative monomorphic B-cell post-transplant lymphoproliferative disorders are pathologically distinct from EBV-positive cases and frequently contain TP53 mutations
. Modern Pathology 2016, 29(10), 1200–1211. [Google Scholar] [CrossRef] [PubMed]
- Hahn, S.M.
Incidence and Features of Lymphoid Proliferation and Lymphomas after Solid Organ or Hematopoietic Stem Cell Transplantation in a National Database Cohort
. Cancer Research and Treatment: Official Journal of Korean Cancer Association 2023, 56(1), 305. [Google Scholar] [CrossRef]
- Styczynski, J.
Response to rituximab-based therapy and risk factor analysis in Epstein Barr virus–related lymphoproliferative disorder after hematopoietic stem cell transplant in children and adults: a study from the Infectious Diseases Working Party of the European Group for Blood and Marrow Transplantation
. Clinical infectious diseases 2013, 57(6), 794–802. [Google Scholar] [PubMed]
- Xu, L.P.
Epstein-Barr Virus-Related Post-Transplantation Lymphoproliferative Disorder after Unmanipulated Human Leukocyte Antigen Haploidentical Hematopoietic Stem Cell Transplantation: Incidence, Risk Factors, Treatment, and Clinical Outcomes
. Biol Blood Marrow Transplant 2015, 21(12), 2185–2191. [Google Scholar] [CrossRef]
- Gilman, A.L.
Alternative donor hematopoietic stem cell transplantation for sickle cell disease
. Blood advances 2017, 1(16), 1215–1223. [Google Scholar] [CrossRef]
- Gonzalez-Farre, B. In vivo intratumoral Epstein–Barr virus replication is associated with XBP1 activation and early-onset post-transplant lymphoproliferative disorders with prognostic implications. Modern Pathology 2014, 27(12), 1599–1611. [Google Scholar] [CrossRef]
- Kroll, J.
Lytic and latent EBV gene expression in transplant recipients with and without post-transplant lymphoproliferative disorder
. Journal of clinical virology 2011, 52(3), 231–235. [Google Scholar] [CrossRef]
- Zhou, X.
Clinical value of plasma and peripheral blood mononuclear cells Epstein–Barr Virus DNA dynamics on prognosis of allogeneic stem cell transplantation
. Frontiers in Cellular and Infection Microbiology 2022, 12, 980113. [Google Scholar] [CrossRef] [PubMed]
- d’Aveni, M.
The clinical value of concomitant Epstein Barr virus (EBV)-DNA load and specific immune reconstitution monitoring after allogeneic hematopoietic stem cell transplantation
. Transplant Immunology 2011, 24(4), 224–232. [Google Scholar] [CrossRef]
- Nash, R.A.
Epstein-Barr virus-associated posttransplantation lymphoproliferative disorder after high-dose immunosuppressive therapy and autologous CD34-selected hematopoietic stem cell transplantation for severe autoimmune diseases
. Biology of Blood and Marrow Transplantation 2003, 9(9), 583–591. [Google Scholar] [CrossRef]
- Comoli, P.
T cell therapy of Epstein–Barr virus and adenovirus infections after hemopoietic stem cell transplant
. Blood Cells, Molecules, and Diseases 2008, 40(1), 68–70. [Google Scholar] [CrossRef] [PubMed]
- Blaes, A.H.
Monitoring and preemptive rituximab therapy for Epstein-Barr virus reactivation after antithymocyte globulin containing nonmyeloablative conditioning for umbilical cord blood transplantation
. Biology of Blood and Marrow Transplantation 2010, 16(2), 287–291. [Google Scholar] [CrossRef]
- Comoli, P.
Preemptive therapy of EBV-related lymphoproliferative disease after pediatric haploidentical stem cell transplantation
. American Journal of Transplantation 2007, 7(6), 1648–1655. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.
Clinical characteristics and outcomes of posttransplant lymphoproliferative disorders following allogeneic hematopoietic stem cell transplantation in Korea
. Journal of Korean Medical Science 2006, 21(2), p. 259. [Google Scholar] [CrossRef] [PubMed]
- Althubaiti, S.
Lymphocyte subset at time of Epstein
-Barr viremia post
-allogeneic hematopoietic stem cell transplantation in children may predict development of post
-transplant lymphoproliferative disease: CD8: CD20 ratio as a sensitive predictor
. Pediatric Transplantation 2019, 23(8), p. e13574. [Google Scholar] [CrossRef]
- Clinic, M. Autologous stem cell transplant. 2025 [cited 2025 1 February ]; Available from: https://www.mayoclinic.org/tests-procedures/autologous-stem-cell-transplant/pyc-20384859.
- Niederwieser, D.
Hematopoietic stem cell transplantation activity worldwide in 2012 and a SWOT analysis of the Worldwide Network for Blood and Marrow Transplantation Group including the global survey
. Bone marrow transplantation 2016, 51(6), 778–785. [Google Scholar] [CrossRef]
- Gale, R.P.
Identical-twin bone marrow transplants for leukemia
. Annals of internal medicine 1994, 120(8), 646–652. [Google Scholar] [CrossRef]
- Briggs, J.D.
Causes of death after renal transplantation
. Nephrology Dialysis Transplantation 2001, 16(8), 1545–1549. [Google Scholar] [CrossRef]
- Washer, G.F.
Causes of death after kidney transplantation
. Jama 1983, 250(1), 49–54. [Google Scholar] [CrossRef]
- Dew, M.A.
Depression and anxiety as risk factors for morbidity and mortality after organ transplantation: a systematic review and meta-analysis
. Transplantation 2016, 100(5), 988–1003. [Google Scholar] [CrossRef]
- Al-Mansour, Z.; Nelson, B.P.; Evens, A.M.
Post-transplant lymphoproliferative disease (PTLD): risk factors, diagnosis, and current treatment strategies
. Current hematologic malignancy reports 2013, 8, 173–183. [Google Scholar] [CrossRef]
- Cockfield, S.
Identifying the patient at risk for post
-transplant lymphoproliferative disorder
. Transplant infectious disease 2001, 3(2), 70–78. [Google Scholar] [CrossRef] [PubMed]
- Landgren, O.
Risk factors for lymphoproliferative disorders after allogeneic hematopoietic cell transplantation
. Blood, The Journal of the American Society of Hematology 2009, 113(20), 4992–5001. [Google Scholar] [CrossRef] [PubMed]
- Styczynski, J.
Management of Epstein-Barr Virus infections and post-transplant lymphoproliferative disorders in patients after allogeneic hematopoietic stem cell transplantation: Sixth European Conference on Infections in Leukemia (ECIL-6) guidelines
. Haematologica 2016, 101(7), p. 803. [Google Scholar] [CrossRef] [PubMed]
- Elstrom, R.
Treatment of PTLD with rituximab or chemotherapy
. American Journal of Transplantation 2006, 6(3), 569–576. [Google Scholar] [CrossRef]
- Cheng, J.
Systematic review and meta
-analysis of post
-transplant lymphoproliferative disorder in lung transplant recipients
. Clinical transplantation 2018, 32(5), p. e13235. [Google Scholar] [CrossRef]
- Dang, B.N.
Treatment of post
-transplant lymphoproliferative disorder (PTLD) in a heart transplant recipient with chimeric antigen receptor T
-cell therapy
. Pediatric Transplantation 2021, 25(5), p. e13861. [Google Scholar] [CrossRef]
Table 1.
Quality assessment of the included studies.
Table 1.
Quality assessment of the included studies.
| Study ID |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
Overall Score (%) |
| Prabodh 2012 [18] |
Y |
Y |
Y |
Y |
Y |
NR |
Y |
Y |
Y |
100 |
| Mirzaii-Dizgah 2012 [19] |
Y |
Y |
Y |
Y |
Y |
N |
Y |
Y |
Y |
88.8 |
| Patil 2013 [20] |
Y |
Y |
Y |
Y |
Y |
Y |
Y |
Y |
Y |
100 |
| Parveen 2015 [21] |
Y |
Y |
Y |
Y |
Y |
NR |
Y |
Y |
Y |
100 |
| Niazi 2021 [22] |
Y |
Y |
Y |
Y |
Y |
NR |
Y |
Y |
Y |
100 |
| Wojtkowska 2021 [23] |
Y |
Y |
Y |
Y |
Y |
NR |
Y |
Y |
Y |
100 |
| Ali 2022 [24] |
Y |
Y |
Y |
Y |
Y |
NR |
N |
Y |
Y |
87.5 |
| Alsaiegh 2023 [25] |
Y |
Y |
Y |
Y |
Y |
NR |
Y |
Y |
Y |
100 |
Table 2.
Major characteristics of the included study.
Table 2.
Major characteristics of the included study.
| Study ID |
Location |
Study Design |
Participant’s Condition |
Demographics of Total Participants |
cTn Measuring Method |
| Case |
Control |
Age
(Mean ± SD) |
Male (%) |
Female (%) |
| Prabodh 2012 [18] |
India |
Case-Control |
AMI |
Healthy |
ND |
ND |
ND |
Chemiluminescence assay |
| Mirzaii-Dizgah 2012 [19] |
Iran |
Case-Control |
AMI |
Healthy |
58.86 ± 12.48 |
71.6 |
28.3 |
ELISA |
| Patil 2013 [20] |
India |
Case-Control |
MI |
Healthy |
48 ± 22 |
65 |
35 |
Immunofluorescence assay |
| Parveen 2015 [21] |
India |
Case-Control |
AMI |
Healthy |
57.28 ± 7.99 |
64 |
36 |
Chemiluminescence |
| Niazi 2021[22] |
Iran |
Case-Control |
AMI |
Healthy |
60.95 ± 14 |
48.75 |
51.25 |
EIA |
| Wojtkowska 2021 [23] |
Poland |
Case-Control |
AMI |
Healthy |
53.11 ± 7.74 |
ND |
ND |
Electro-chemiluminescence |
| Ali 2022 [24] |
United Kingdom |
Cohort |
AMI |
No CVD events for 2 consecutive years |
71 ± 7 |
67 |
33 |
ND |
| Alsaiegh 2023 [25] |
Iraq |
Case-Control |
AMI |
Healthy |
52.37 ± 16.6 |
73.3 |
26.7 |
COBAS C411 Technique |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).