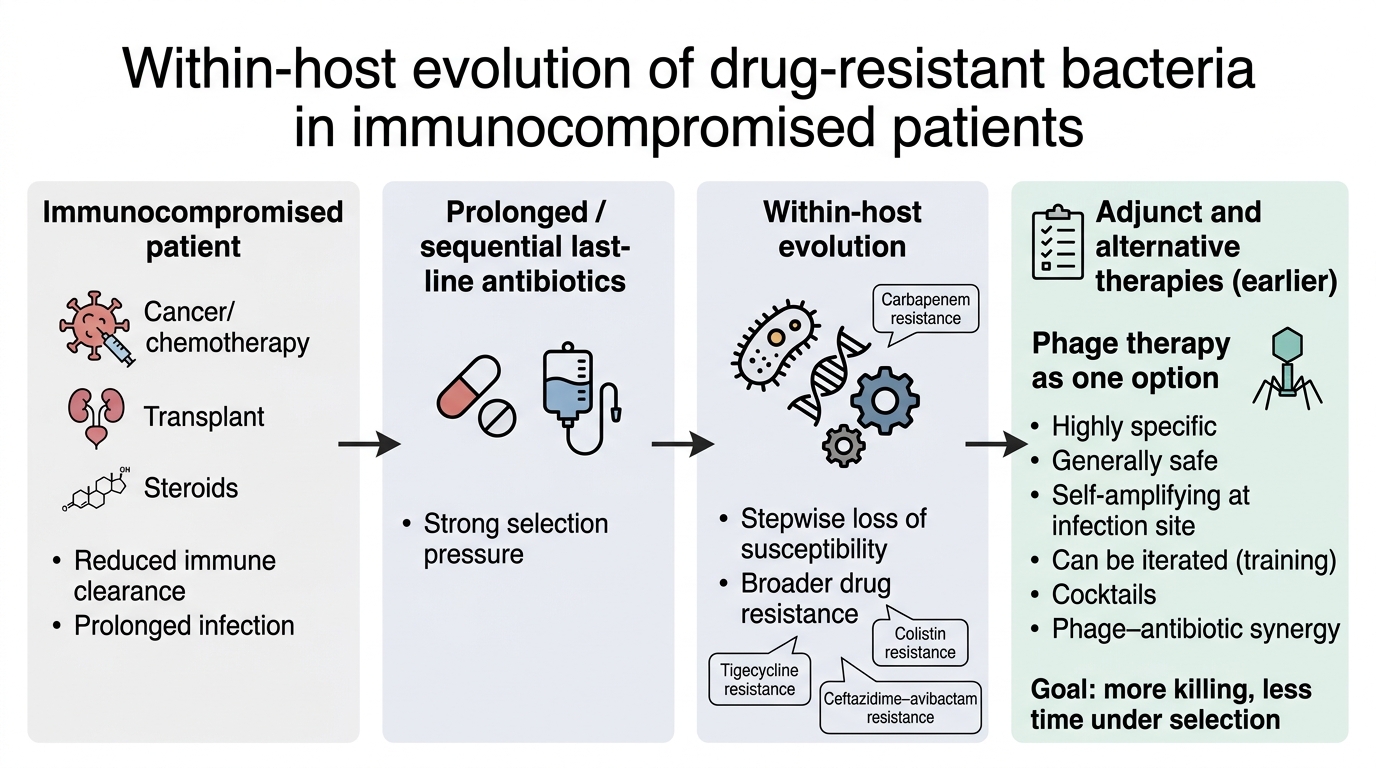
Antimicrobial resistance is often framed as a problem acquired outside the patient through transmission of resistant strains and genes. This view is important, but it is incomplete for immunocompromised patients, where there is substantial evidence that drug-resistant bacteria can evolve within the host during therapy. In haematological malignancy, transplantation, and other states of impaired immunity, infections persist longer, immune clearance is reduced, and prolonged use of last-line antibiotics creates repeated selection events. These conditions favour stepwise evolution toward the hardest-to-treat phenotypes, including carbapenem resistance, tigecycline resistance, colistin resistance, and resistance to ceftazidime–avibactam, often alongside persistence in reservoirs such as the gastrointestinal tract. This essay argues that antibiotic escalation alone is therefore an incomplete strategy in these settings and that care should be explicitly evolution-aware. Adjunct and alternative approaches should be prioritised earlier to reduce bacterial burden, shorten time under selection, and limit reliance on prolonged sequential antibiotic regimens. Bacteriophages are highlighted as one promising adjunct because they are highly specific, generally well tolerated, can self-amplify at sites where susceptible bacteria are present, and can be iterated through approaches such as training and rational cocktails. Phage–antibiotic synergy is also discussed as a practical strategy to improve killing and reduce escape.