Submitted:
28 February 2026
Posted:
04 March 2026
You are already at the latest version
Abstract
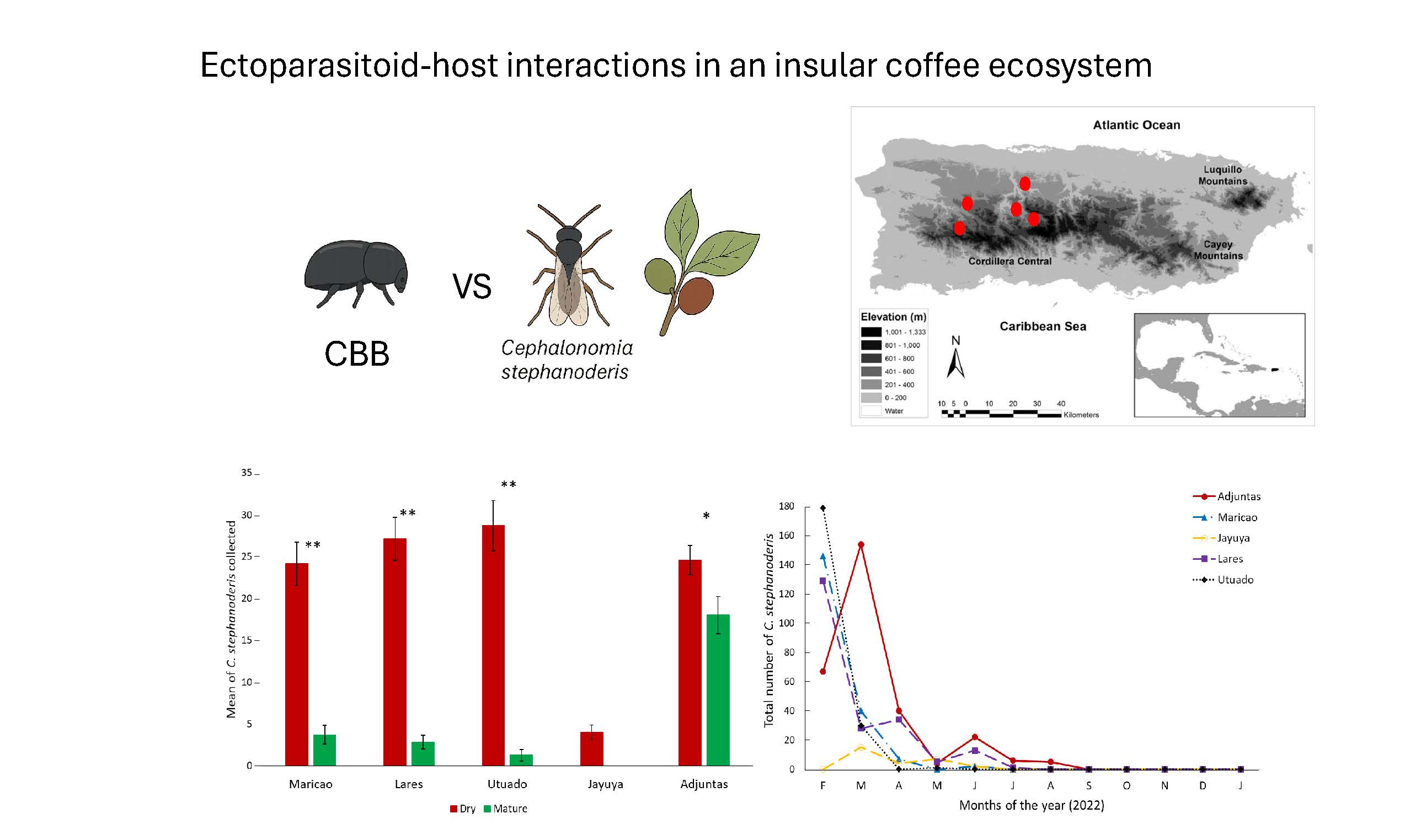
Keywords:
1. Introduction
2. Materials and Methods
2.1. Sampling Dry Versus Mature Berries for Parasitoids
2.2. Sampling to Estimate Percent Parasitism
2.3. Statistical Analysis
3. Results
3.1. Cephalonomia Stephanoderis from Dry Versus Mature Coffee Berries
3.2. Total of Parasitoids Produced per Location
3.2.1. Parasitoids Produced per Location in Dry and Mature Coffee Berries
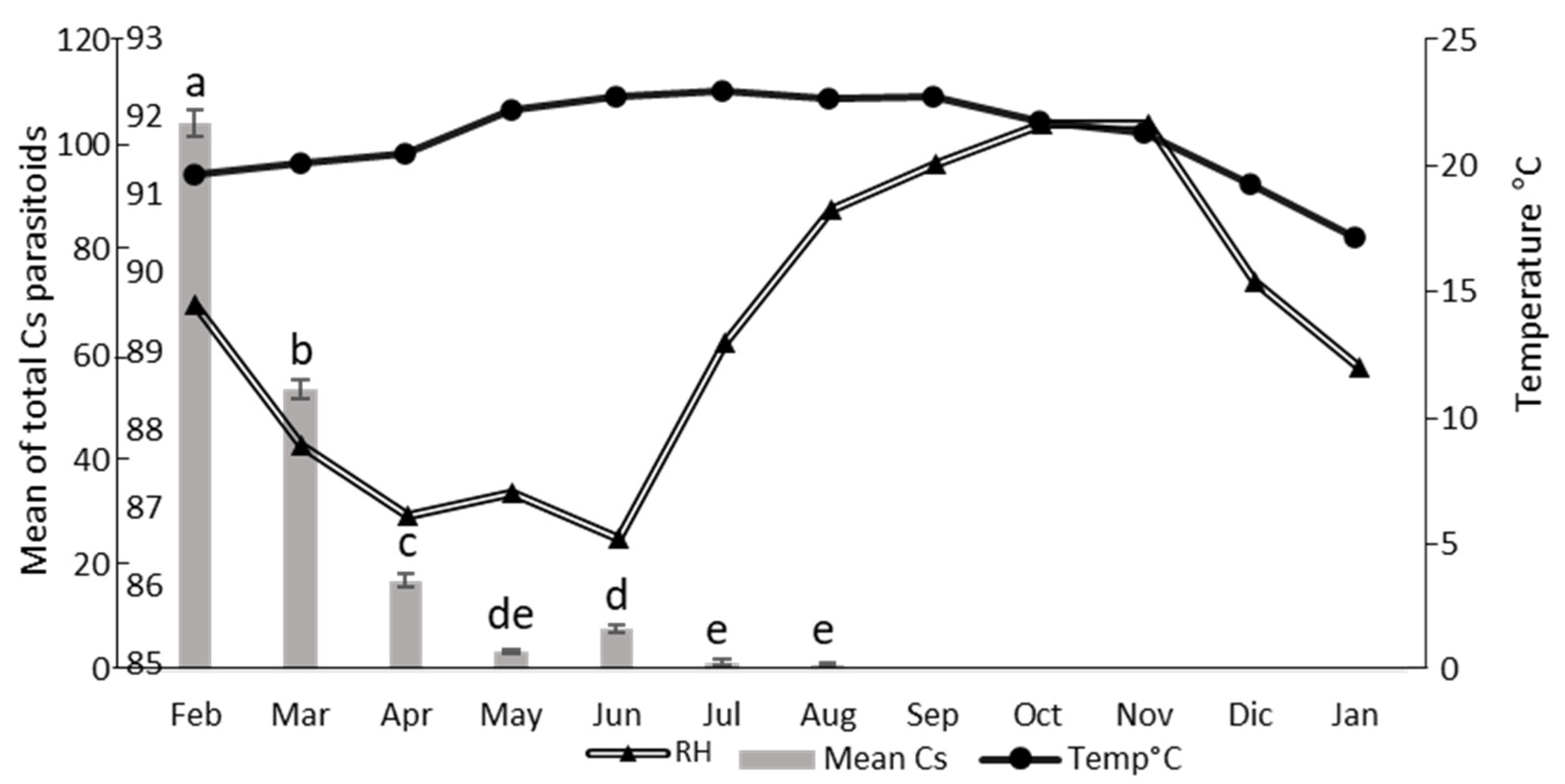
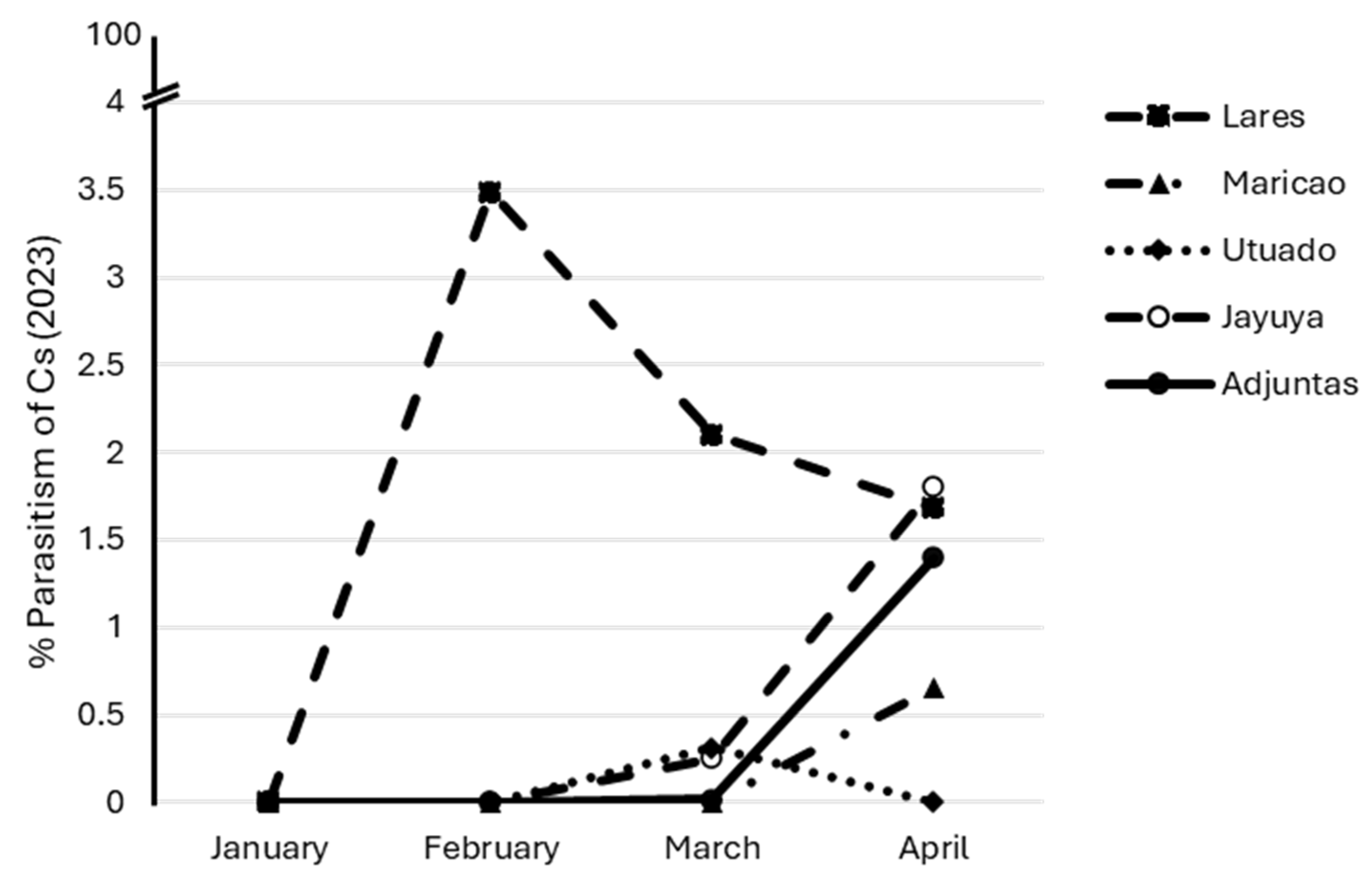
3.3. Parasitoids Abundance over Time at Each Location
3.4. Parasitoids Abundance over Time
3.5. Correlation Between Parasitoid Abundance and Environmental Factors
3.6. Percentage of Parasitism by Cephalonomia stephanoderis
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vega F. E., Franqui R.A., Benavides P. Scientific note: The presence of the coffee berry borer, Hypothenemus hampei, in Puerto Rico: fact or fiction? J. Insect Sci. 2002, 2,1-3. [CrossRef]
- NAPPO- Norteamerican Organization of Plant Protection 2007. Detecciones de la Broca del Café Hypothenemus hampei in Puerto Rico, Estados Unidos. Oficial Pest Report. https://pestalerts.org/es/official-pest-report/detecciones-de-la-broca-del-caf-hypothenemus-hampei-en-puerto-rico-estados. (May 2023).
- Bayman P., Mariño Y.A., Garcia-Rodriguez N.M., Oduardo-Sierra O.F., Rehner S. Local isolates of Bauveria bassiana for control of the coffee berry borer Hypothenemus hampei in Puerto Rico: Virulence, efficacy and persistence. J. Biol.Control 2021,155,1-10. [CrossRef]
- Aristizabal A.L.F., Johnson M., Shriner S., Hollingsworth R., Manoukis N.C., Myers R., Bayman P., Arthus S.P. Integrated Pest Management of Coffee Berry Borer in Hawaii and Puerto Rico: current Status and Prospects. Insects 2017, 8,1-16. [CrossRef]
- Infante F. Pest Management Strategies Against the coffee Berry Borer (Coleoptera: Curculionidae: Scolytinae). J. Agric. Food Chem. 2018, 66, 5275-5280. [CrossRef]
- Orozco-Hoyos J. A guide to the rearing of the parasitoid Phymastichus coffea for control of the coffee berry borer. The Commodities Press, Cali, Colombia, 2002; pp.19.
- Portilla R.M. & Bustillo P.A. Nuevas investigaciones de Hypothenemus hampei y de sus parasitoides Cephalonomia stephanoderis y Prorops nasuta. Rev. Colomb. Entomol. 1995, 21, 25-33.
- Portilla M & Streeett D. Producción masiva automatizada de la Broca del Café Hypothenemus hampei (Ferrari) (Coleoptera: Scolytidae) y de sus parasitoides sobre dietas artificiales. Sist. Agroeco. Mod. Biomatematic 2008,1, 9-24.
- Portilla M. (1999). Mass rearing technique for Cephalonomia stephanoderis (Hymenoptera: Bethylidae) on Hypothenemus hampei (Coleoptera: Scolytidae) developed using Cenibroca artificial diet. Rev. Colomb. Entomol 1999, 25, 57–66. [CrossRef]
- Bustillo A.E., Orozco H.J., Benavides M.P., Portilla R.M. Producción masiva y uso de parasitoides para el control de la broca del café, Hypothenemus hampei, en Colombia. Cenicafé, 1996; 47, 215-230.
- Brun L.O., Gaudichon V., Wigley P.J. An artificial diet for continuous rearing of the coffee berry borer, Hypothenemus hampei (Ferrari) (Coleoptera: Scolytidae). Insect Sci. Appl.1993, 14,585-587. [CrossRef]
- Ticheler J. Etude analytique del’épidémiologie du scolyte des graines de café Stephanoderis hampei Ferr. en Côte d’Ivoire. Mededelingen Landbouwhogeschool Wageningen 1961, 61, 1-49.
- Infante F., Valdez J., Penagos D.I., Barrera J.F. Description of the life stages of Cephalonomia stephanoderis (Hymenoptera: Bethylidae), a parasitoid of Hypothenemus hampei (Coleoptera: Scolytidae). Vedalia 1994, 1, 13-18.
- Abraham Y.J., Moore D., Godwin G. Rearing and aspects of biology of Cephalonomia stephanoderis and Prorops nasuta (Hymenoptera: Bethylidae) parasitoids of the coffee berry borer, Hypothenemus hampei (Coleoptera: Scolytidae). Bull. Entom. Res. 1990, 80, 121-128. [CrossRef]
- Garcia H.M., Duque E.T.C., Rodriguez V.Y., Borrero R.Y., Cabrera R.D., Campo C.Y. Reproducción, ciclo biológico y relación sexual de Cephalonomia stephanoderis Betrem en condiciones de laboratorio en Cuba. Fitosanidad 2008, 12, 227-232.
- Alonso J., Cabrera M., Barrera J.F. The importance of the Parasitoid Egg in Host Discrimination by Cephaolnomia stephanoderis, and Ectoparasitoid of the Coffe Berry Borer Hypothenemus hampei. Biocontrol Sci. Techn. 1998, 8, 153-162.
- Barrera J.F., Gomez J., Alauzet C. Evidence for a marking pheromone in host discrimination by Cephalonomia stephanoderis (Hym.: Bethylidae), Short Note. Entomophaga 1994, 39, 363-366. [CrossRef]
- Lauziere I., Perez-Lachaud G., Brodeur J. Behaviour and Activity Pattern of Cephalonomia stephanoderis (Hymenoptera: Bethylidae) attacking the Coffee Berry Borer, Hypothenemus hampei (Coleoptera: Scolytinae). J. Insect Behav 2000, 13, 1-21. [CrossRef]
- Alonso C.P.L., Rodriguez A.L., Monzon R.C.E., Delgado T.R., Perez R.Y. Evaluation of the parameters of quality and biological effectiveness of Cephalonomia stephanoderis Betrem under Cienfuegos province conditions. Rev. Científ. Agroecosist. 2022, 10, 130-138.
- Gomez J., Barrera J. F., Leido P., Valle J. Influence of age and diet on the performance of Cephalonomia stephanoderis (Hymenoptera, Bethylidae) a parasitoid of the coffee berry borer, Hypothenemus hampei (Coleoptera, Curculionidae). Rev. Brasil. Entom. 2012, 56, 95-100. [CrossRef]
- Lauziere I., Perez-Lachaud G., Brodeur J. Importance of nutrition and host availability on oogenesis and oviposition of Cephalonomia stephanoderis (Hymenoptera: Bethylidae). Bull. Entom. Res. 2001, 91, 185-191. [CrossRef]
- Lauziere I., Perez-Lachaud G., Brodeur J. Effect of female body size and adult feeding on the fecundity and longevity of the parasitoid Cephalonomia stephanoderis Betrem (Hymenoptera: Bethylidae). Ann. Entomol. Soc. Am. 2000, 93, 103-109. [CrossRef]
- Chiu-Alvarado P. & Rojas J.C. Behavioural responses of bethylid parasitoid species of the coffee Berry borer to chemicals cues from host and non-host dust/frass. Biocontrol 2011, 56,45-53. [CrossRef]
- Gomez J., Barrera J.F., Rojas J.C., Macias-Samano J., Liedo J.P., Cruz-Lopez L., Badii M.H. Volatile compounds released by disturbed females of Cephalonomia stephanoderis (Hymenoptera: Bethylidae): a parasitoid of the coffee berry borer Hypothenemus hampei (Coleoptera: Scolytidae). Fla. Entomol. 2005, 88, 180-187. [CrossRef]
- Infante F., Luis J.H., Barrera J.F., Gomez J., Castillo A. Thermal constants for preimaginal development of the parasitoid Cephalonomia stephanoderis Betrem (Hymenoptera: Bethylidae). Can. Entomol 1992, 124, 935-941. [CrossRef]
- Infante F., Mumford J., Baker P., Barrera J., Fowler S. Interspecific competition between Cephalonomia stephanoderis and Prorops nasuta (Hym., Bethylidae), parasitoids of the coffee berry borer, Hypothenemus hampei (Col., Scolytidae). J. Appl. Entomol. 2001,125, 63-70. [CrossRef]
- Damon A. & Valle J. Comparison of two release techniques for the use of Cephalonomia stephanoderis (Hymenoptera: Bethylidae), to control the coffee berry borer Hypothenemus hampei (Coleoptera: Scolytidae) in Soconusco, southeastern Mexico. Biol. Control 2002, 24, 117-127.
- Lauziere I., Brodeur J., Perez-Lachaud G. Host stage selection and suitability in Cephalonomia stephanoderis Betrem (Hymenoptera: Bethylidae), a parasitoid of the coffee Berry borer. Biol. Control 2001, 21, 128-133. [CrossRef]
- Aristizabal A.L.F., Bustillo P.A.E., Baker P.S., Orozco J.H., Chaves B.C. Depredatory effects of the parasitoid Cephalonomia stephanoderis on the immature stages of Hypothenemus hampei under field conditions. Rev. Colomb. Entomol. 1998, 24, 35-41. [CrossRef]
- Vijayalakshmi C.K., Rahiman P.A., Sreedharan K., Selvakumar M. Field evaluation of coffee Berry Borer parasitoid Cephalonomia stephanoderis (Betrem) in the coffee tracts of Wayanad, Kerala. International Scientific Association of Coffee in British Library Conference Proceedings, Bangalore, India, pp. 1282-1283.
- Aristizabal A.L.F., Baker P.S., Orozco J.H., Chaves B.C. Parasitism of Cephalonomia stephanoderis Betrem on populations of Hypothenemus hampei (Ferrari) at infestation low levels in the field. Rev. Colomb. Entomol. 1997, 23, 157-164. [CrossRef]
- Aristizabal A.L.F., Salazar H.M., Mejía C.G., Jimenez M., Bustillo A.E. and Arthurs S.P. Establishment of exotic parasitoids of the coffee berry borer Hypothenemus hampei (Coleoptera: Curculionidae: Scolytinae) in Colombia through farmer participatory research. Int. J. Trop. Insect Sci. 2012, 32, 24-31. [CrossRef]
- Souza M.S., Texeira C.A.D, Azevedo C.O., Costa V.A., Costa J.N.M. Occurrence of Cephalonomia stephanoderis Betrem (Hymenoptera: Bethylidae) in Coffee Plantations of the Brazilian Amazonia, Scientific note. Neotrop. Entomol. 2006, 35, 560-562. [CrossRef]
- Bustillo A.E., Cárdenas R., Posada F.J. Natural Enemies and Competitors of Hypothenemus hampei (Ferrari) (Coleoptera: Scolytidae) in Colombia. Neotrop. Entomol. 2002, 31, 635-639. [CrossRef]
- Quintero H.C., Bustillo P.A.E., Benavides M.P., Chaves C.B. Evidences of the establishment of Cephalonomia stephanoderis and Prorops nasuta (Hymenoptera: Bethylidae) in coffee plantations in the department of Nariño, Colombia. Rev. Colomb. Entomol. 1998, 24, 141-147. [CrossRef]
- Irulandi S., Samuel S.D., Kumar P.K.V., Sreedharan K. Parasitism by the exotic parasitoid, Cephalonomia stephanoderis Betrem (Hymenoptera: Bethylidae) on coffee berry borer, Hypothenemus hampei (F.) on Pulney Hills, Tamil Nadu. Hexapoda, 2009, 16, 32-35.
- Mailafiya D.M., Le Ru B.P., Kairu E.W., Calatayud P.A., Dupas S. Factors affecting stem borer parasitoid species diversity and parasitism in cultivated and natural habitats. Environ. Entomol. 2010, 39, 57-67. [CrossRef]
- Bourchier R.S., & Smith, S.M. Influence of environmental conditions and parasitoid quality on field performance of Trichogramma minutum. Entomol. Exp. Appl. 1996, 80, 461-468. [CrossRef]
- Abreu R.E. & Gallardo C.F. Presencia de Cephalonomia stephanoderis (Hymenoptera: Bethylidae) parasitoide de la broca del café, Hypothenemus hampei (Coleoptera: Curculionidae) en Puerto Rico. Proceedings of the Sociedad Puertoriqueña de Ciencias Agrícolas 2011, Ponce, Puerto Rico, pp. 9.
- Gallardo-Covas F. Biological control of insect pests in Puerto Rico. J. Agric Univ. Puerto Rico 2017, 101, 153-163. [CrossRef]
- González-Cardona O.P. Dinámica Poblacional de la Broca del Café Hypothenemus hampei (Ferrari) y sus Enemigos Naturales en el Cultivo de Café Coffea arabica L. en Puerto Rico. Master thesis, Master degree, Mayaguez, Puerto Rico, 2013.
- Gallardo-Covas F. (2019). Augmentation and conservation of Cephalonomia stephanoderis Betrem for the biological suppression of the coffee berry borer, Hypothenemus hampei Ferrari in Puerto Rico. Memorias Reunión de la Empresa de Café. Puerto Rico, 46p.
- Bustillo A.E., Cárdenas R., Villalba D., Benavides P., Orozco J., Posada F.J. (1998). Manejo Integrado de la Broca del café Hypothenemus hampei (Ferrari) en Colombia. Cenicafe-Boletín informativo sobre la broca del café, 1998, 134p.
- Mathieu F., Brun L.O., Frerot B., Suckling D.M., Frampton C. Progression in field infestation is linked with trapping of coffee berry borer, Hypothenemus hampei (Col., Scolytinae). J. Appl. Entomol. 1999, 123, 535-540. [CrossRef]
- Balakrishnan M.M., & Sreedharan, K. Establishment of Cephalonomia stephanoderis, the introduced parasitoid of coffee berry borer in Coorg District, Karnataka. In Proceedings of the 15th Plantation Crops Symposium Placrosym XV, Mysore, India, 10-13 December, 2002 (pp. 504-508).
- Bernaschini M.L., Rossetti M.R., Valladares G., Salvo A. Microclimatic edge effects in a fragmented forest: disentangling the drivers of ecological processes in plant-leafminer-parasitoid food webs. Ecol. Entomol. 2021, 46, 1047-1058. [CrossRef]
- Landis D. & Menalled F.D. Ecological considerations in the conservation of effective parasitoid communities in agricultural systems. In Conservation biological control, Barbosa P. Ed..Academic Press, London, UK, 1998; pp. 101-121.
- Hassell, M. The spatial and temporal dynamics of host-parasitoid interactions. 1 st ed.; Oxford University Press: New York, USA, 2000; p.198.
- Gallardo-Covas F. & Gonzalez-Cardona O.P., (2015). Manejo Integrado de la Broca del Café en Puerto Rico. Guía Tecnica, Departamento de Agricultura: San Juan, Puerto Rico , USA, 2015; p. 12.
- Mariño Y.A., Vega V.J., Garcia J.M., Verle R. J.C., Garcia N.M., Bayman P. The Coffee Berry Borer (Coleoptera: Curculionidae) in Puerto Rico: Distribution, Infestation, and Population per Fruit. J. Insect Sci. 2017, 17, 1-8. [CrossRef]
- Ijala A.R., Kyamanywa S., Cherukut S., Sebatta C., Karungi J. Parasitism of Hypothenemus hampei (Coleoptera: Scolytidae) in different farming systems and altitudes of Mount Elgon, Uganda. J. Appl. Entomol. 2019, 89,1-10. [CrossRef]
- Maldonado L.C.E. & Benavides M.P. Evaluación del Establecimiento de Cephalonomia stephanoderis y Prorops nasuta, Controladores de Hypothenemus hampei, en Colombia. Cenicafe 2007, 58, 333-339.
- Souza M.S., Almeida S.A., Teixeira C.A.D., Costa J.N.M. Parasitism in the population of the coffee berry borer Hypothenemus hampei (Ferrari) (Coleoptera: Curculionidae) by the parasitoid Cephalonomia stephanoderis Betrem (Hymenoptera: Bethylidae). EntomoBrasilis 2014, 7, 178-182. [CrossRef]
- Salazar H.M. & Baker P.S. Impacto de liberaciones de Cephalonomia stephanoderis sobre poblaciones de Hypothenemus hampei. Cenicafe 2002, 53, 306-316.
- Benavides M.P., Bustillo P.A.E., Montoya E.C. Advances on the use of the parasitoid Cephalonomia stephanoderis for the control of the coffee berry borer Hypothenemus hampei. Rev. Colomb. Entomol. 1994, 20, 247-253. [CrossRef]
- Lauziere I., Perez-Lachaud G., Brodeur J. Influence of host density on the reproductive strategy of Cephalonomia stephanoderis, a parasitoid of the coffee berry borer. Entomol. Exp. Appl. 1999, 92, 21-28. [CrossRef]
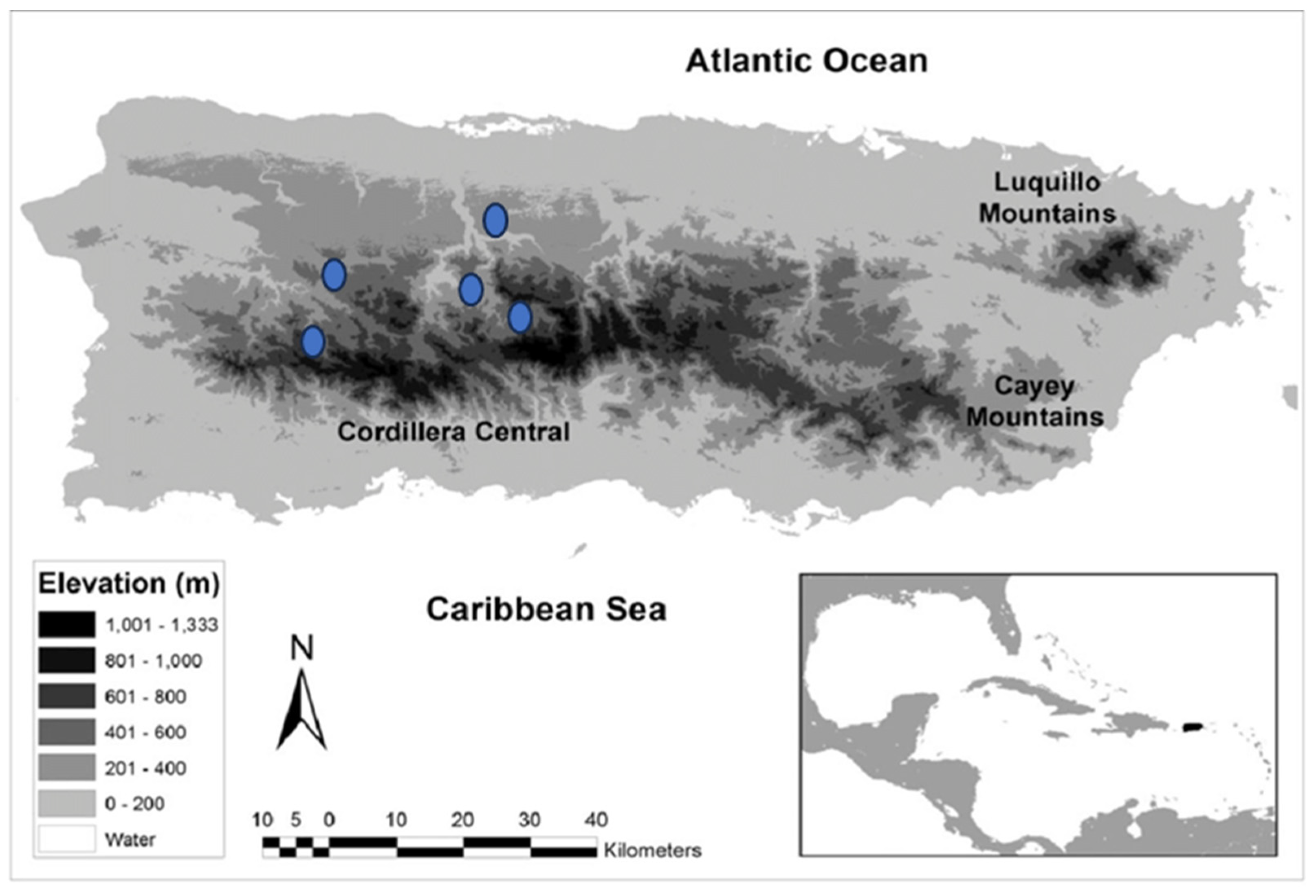
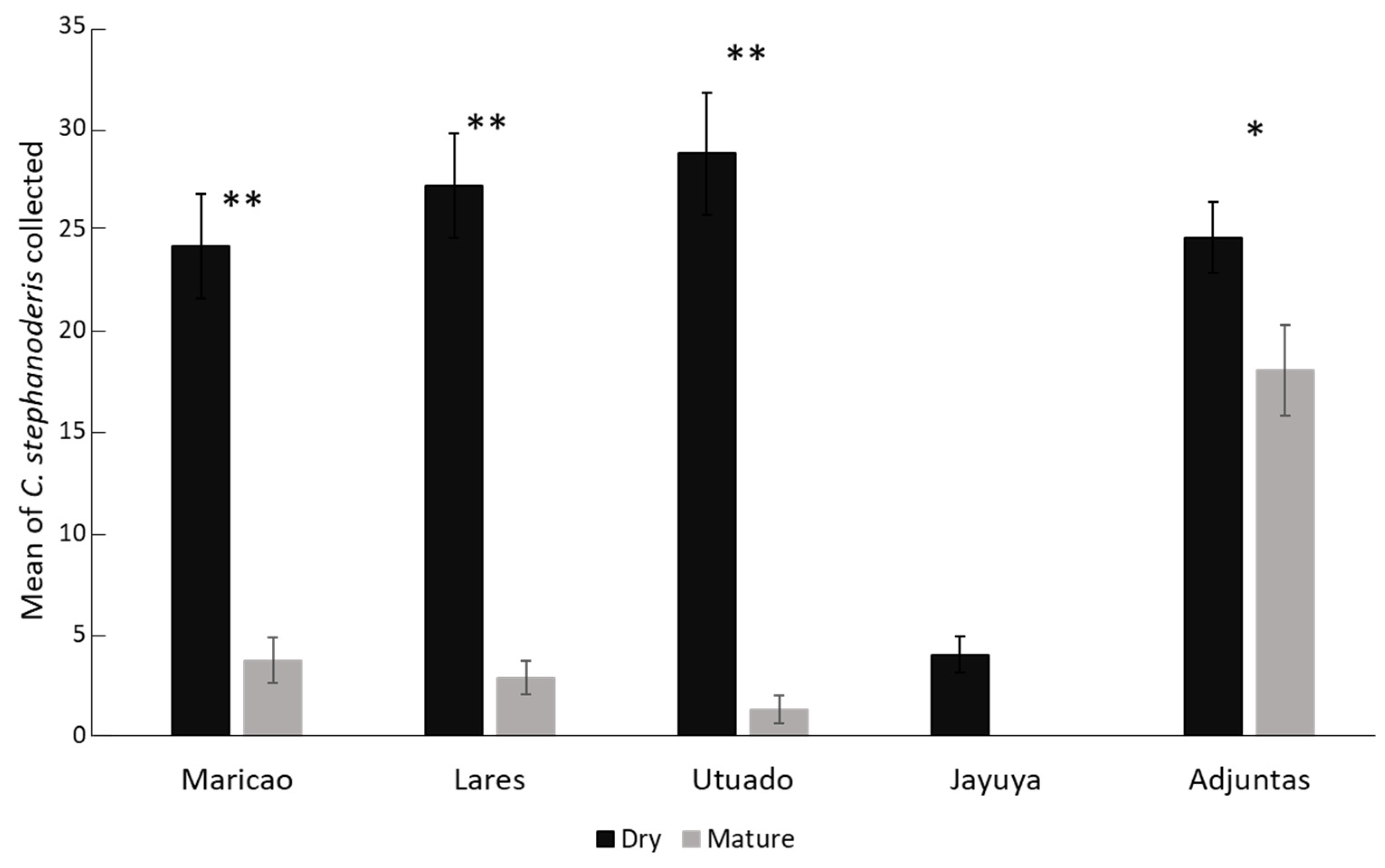
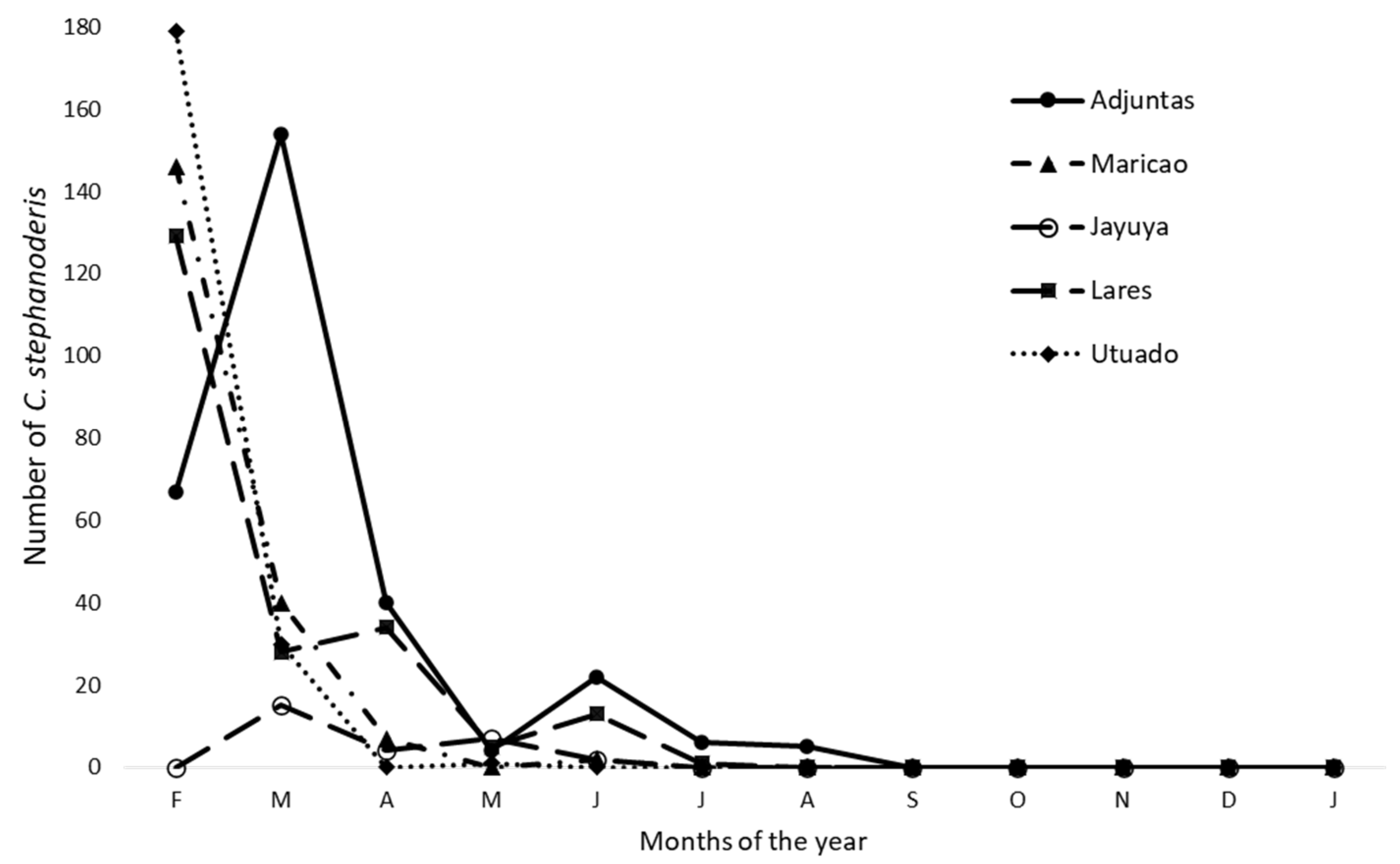
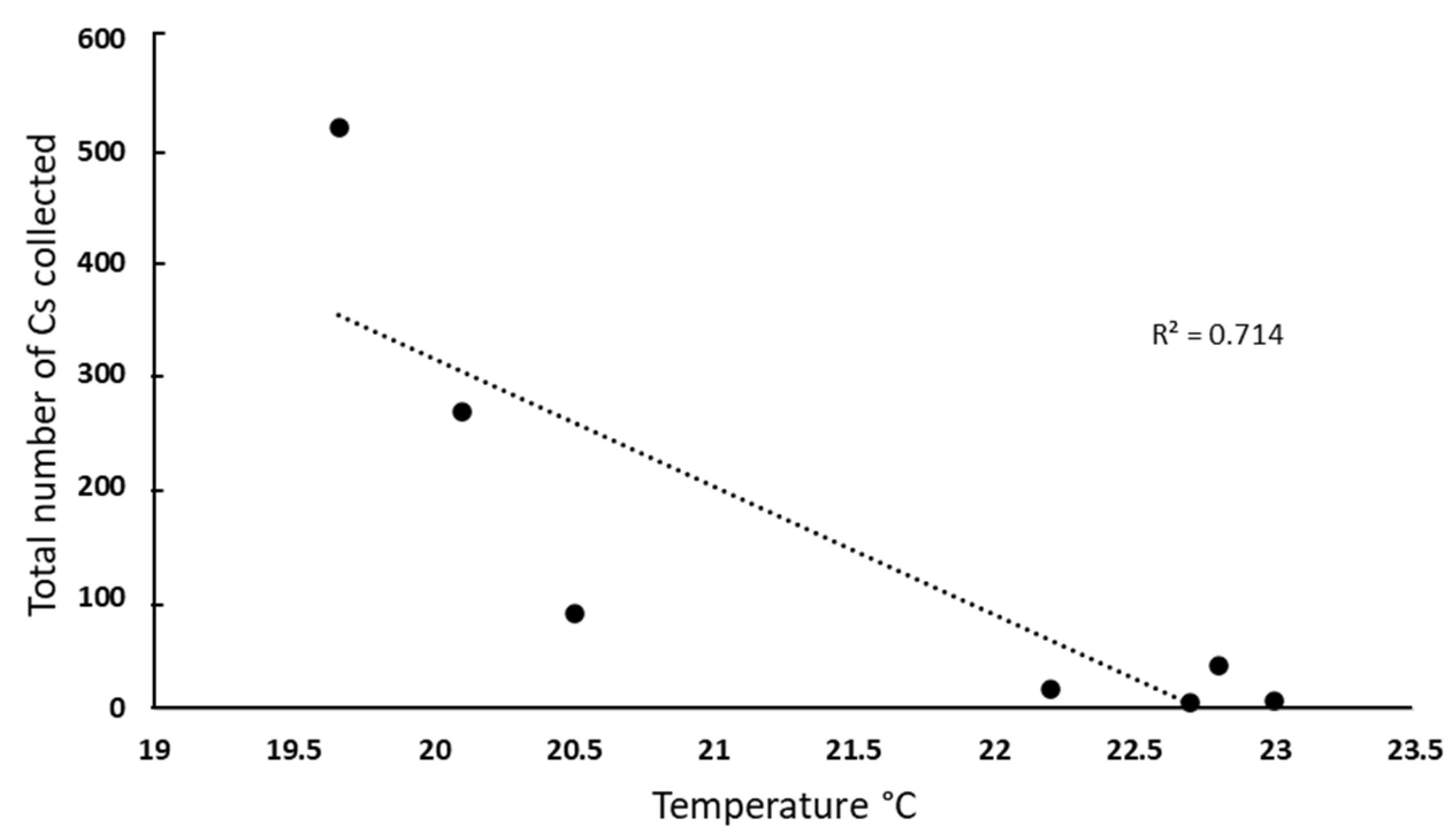
| Climatic variables | Correlation coefficient (r) | Determination coefficient (r2) | Pvalue1 |
| Temperature | -0.84 | 0.714 | 0.01 |
| Relative Humidity | 0.202 | 0.041 | 0.66 |
| Altitude | 0.07 | 0.005 | 0.73 |
| Month |
% parasitism |
Total number of Cs collected | Average number of Cs collected per site | Total number of CBB | Max. CBB per coffee berry | Average of CBB per coffee berry |
| January | 0 | 0 | 0 | 1393 | 47 | 5.5 |
| February | 3.49 | 92 | 18.4 | 2538 | 65 | 10.5 |
| March | 2.79 | 98 | 19.6 | 3404 | 87 | 13.6 |
| April | 4.97 | 168 | 33.6 | 3207 | 57 | 12.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





