Submitted:
02 March 2026
Posted:
04 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
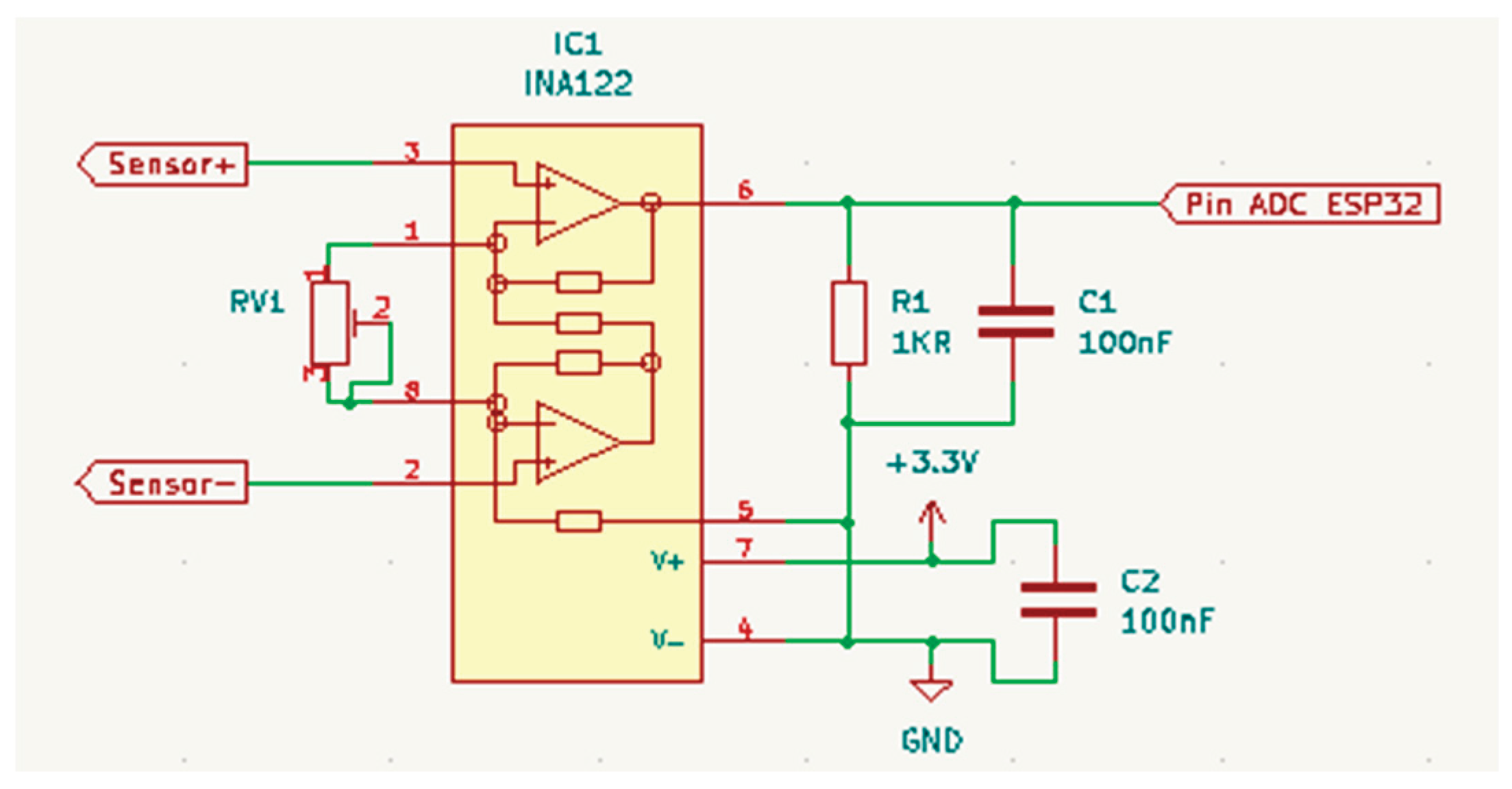
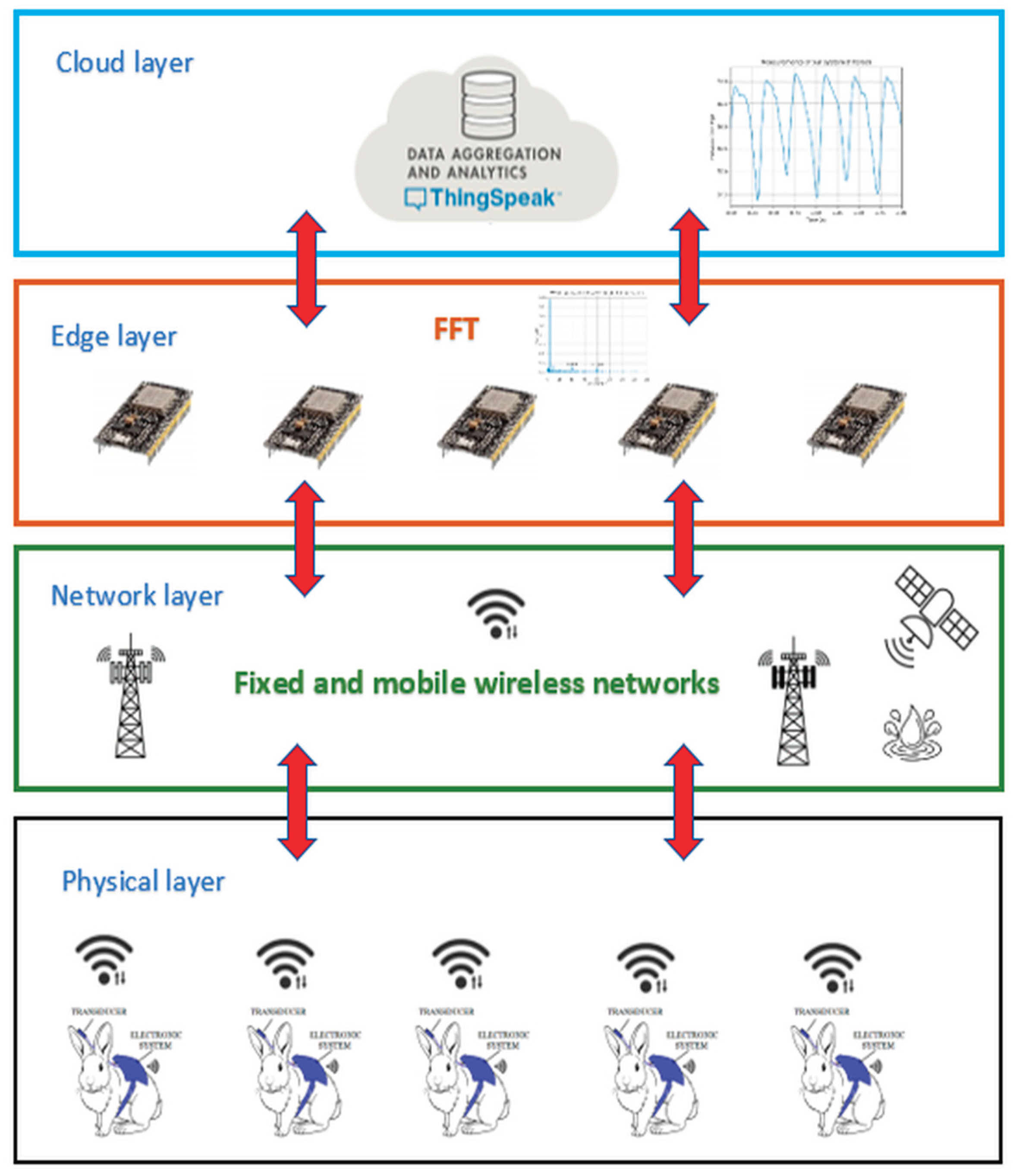
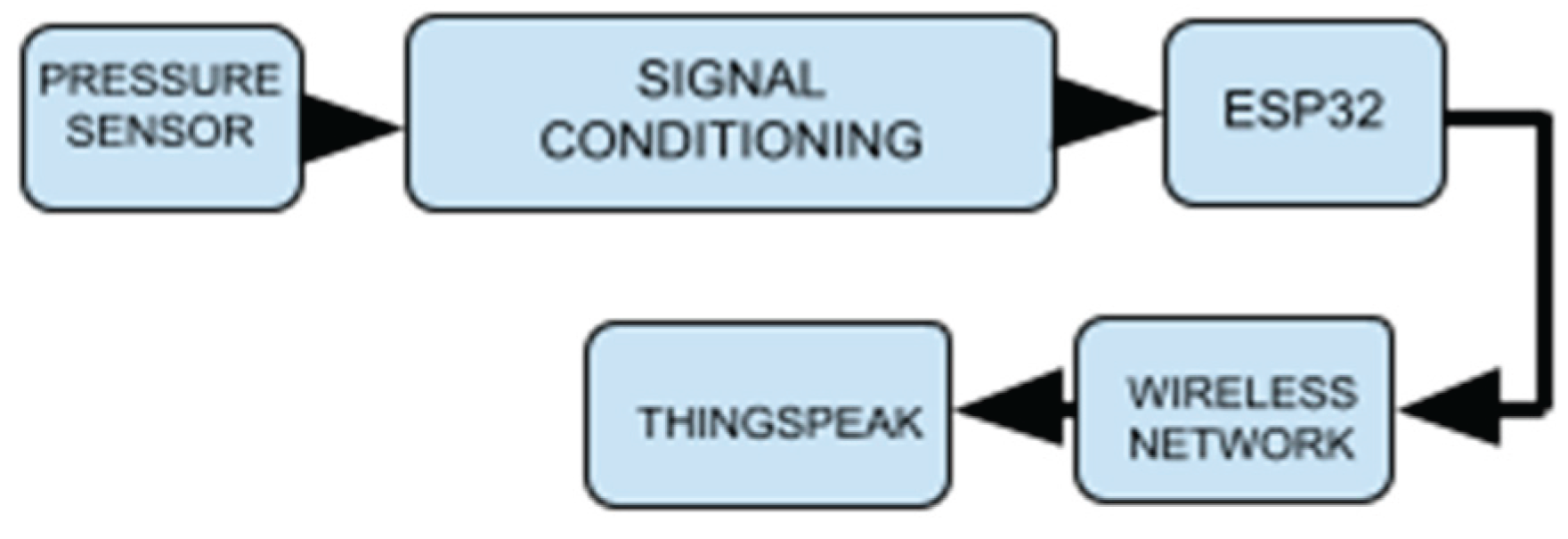
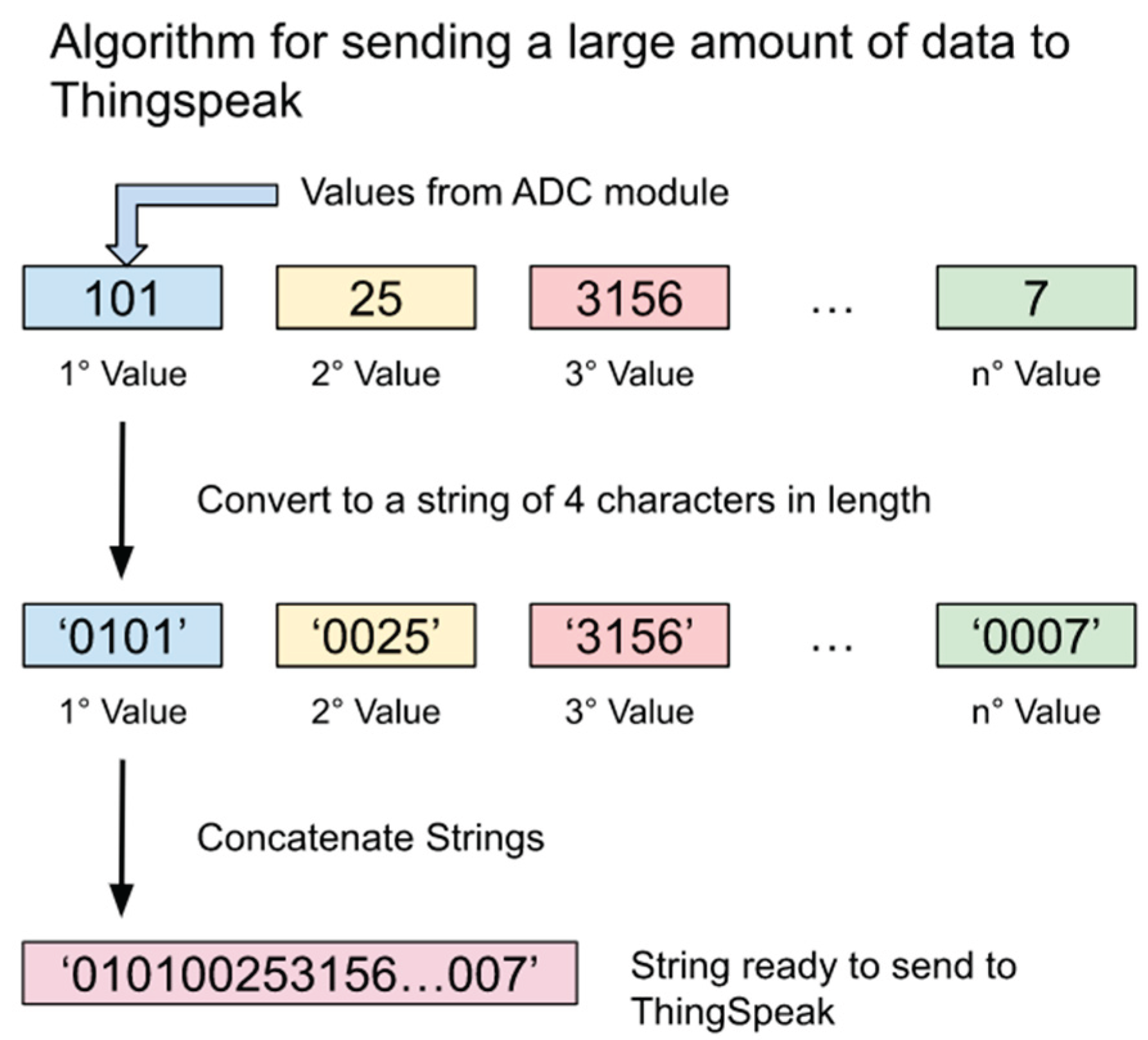
2.1. System Architecture
2.2. System Development
2.3. Rabbits Preparation
2.4. Experimental Procedures
3. Results
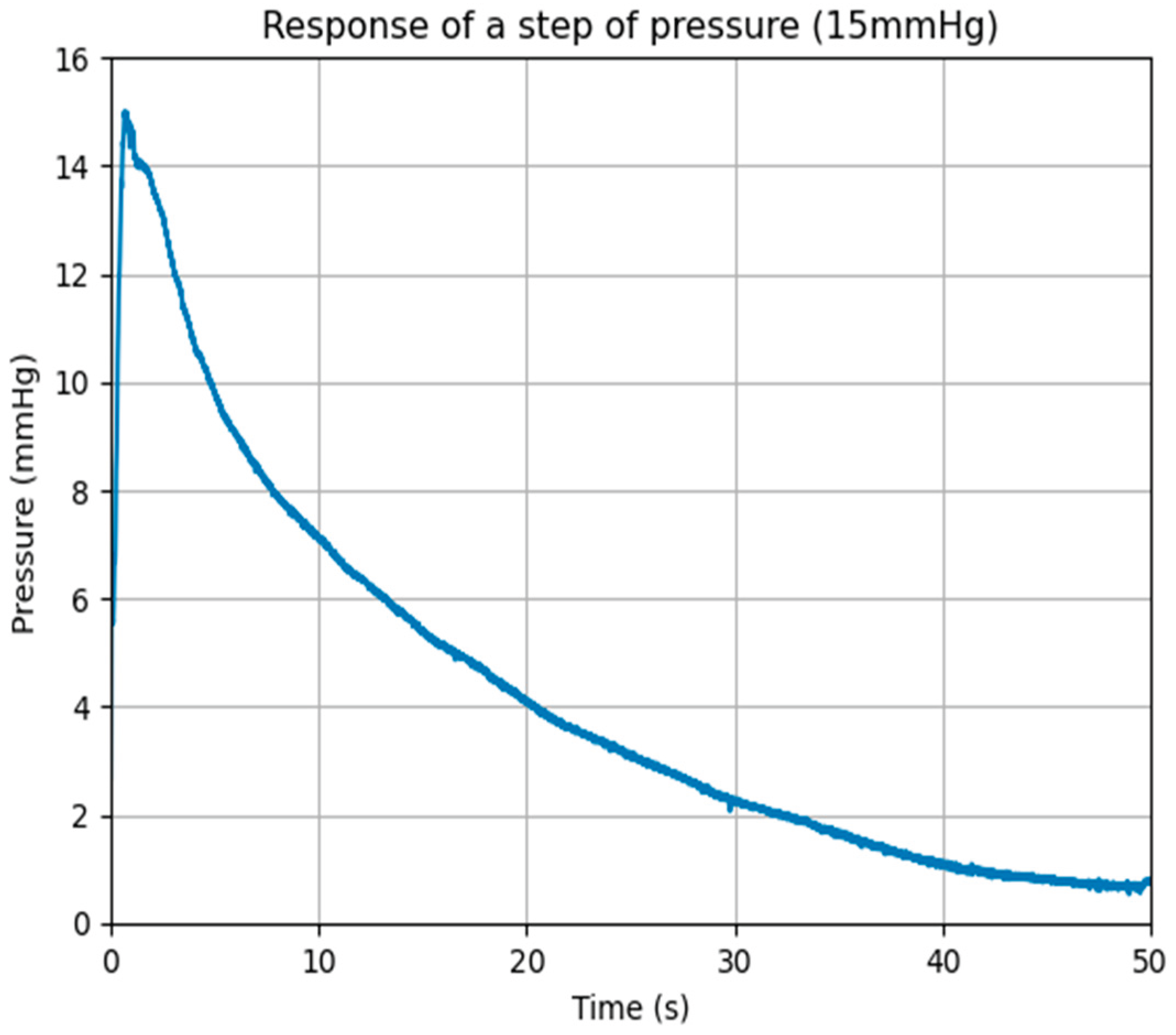
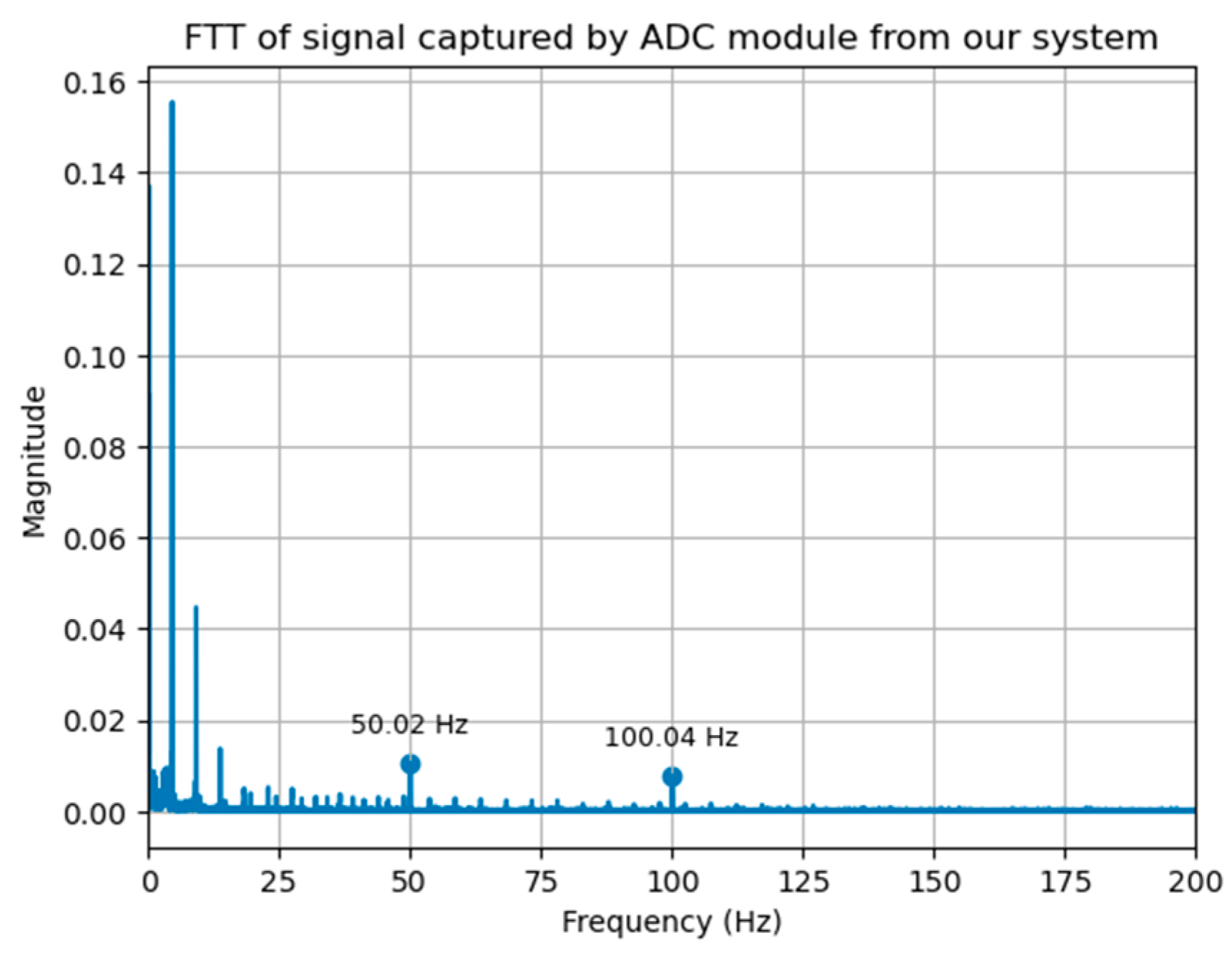
3.1. System Calibration
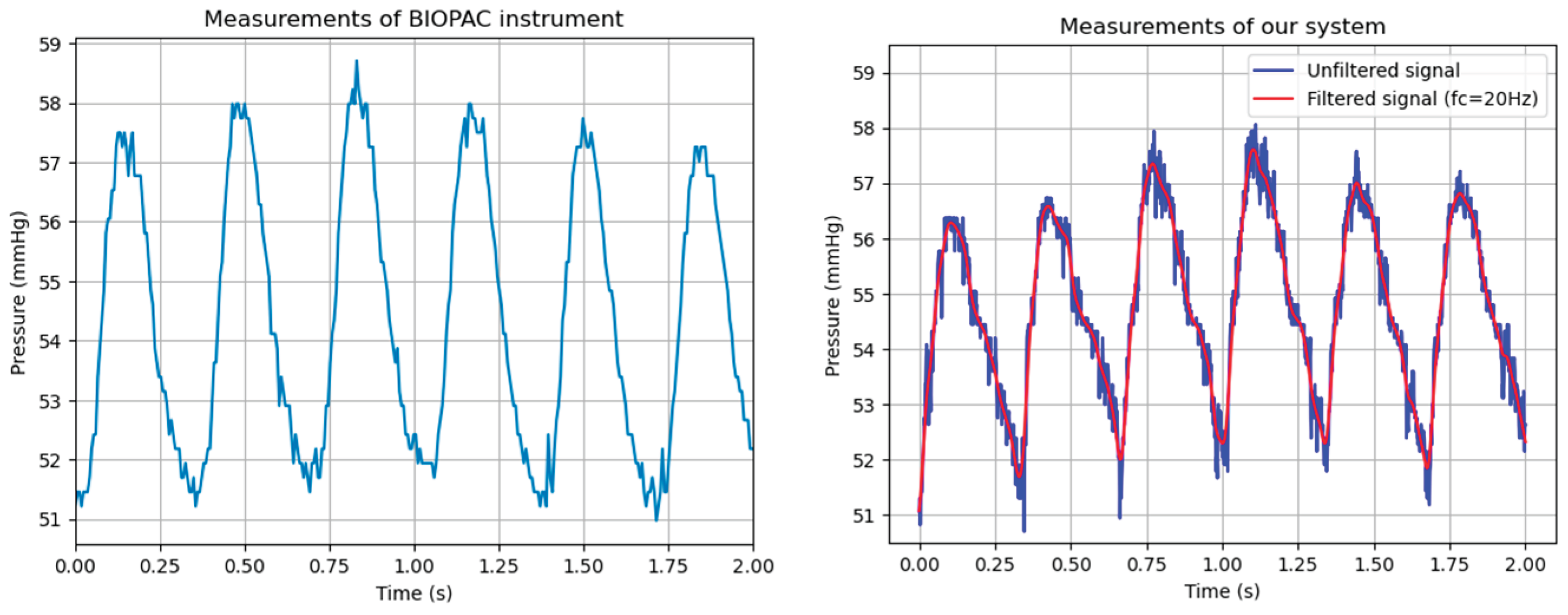
3.2. Data Acquisition and Processing
4. Discussion

5. Conclusions
Acknowledgments
References
- Han, Y.; Wei, T.; Chen, Z.; Wang, H.; Wang, L.; Li, C.; Mei, X.; Kuang, L.; Gong, J. LIVEMOS-G: A High Throughput Gantry Monitoring System with Multi-Source Imaging and Environmental Sensing for Large-Scale Commercial Rabbit Farming. Animals 2025, 15(21), 3177. [CrossRef]
- Ashton, K. That ‘Internet of Things’ Thing. RFID J. 2009, 22, 97–114.
- Vailshery, L.S. Number of Internet of Things (IoT) Connected Devices Worldwide from 2019 to 2021, with Forecasts from 2022 to 2034; Statista: Hamburg, Germany, 2022; Available online: https://www.statista.com/statistics/1183457/iot-connected-devices-worldwide/ (accessed on 15 February 2026).
- Yan, H.; Xu, L.D.; Bi, Z.; Pang, Z.; Zhang, J.; Chen, Y. An Emerging Technology – Wearable Wireless Sensor Networks with Applications in Human Health Condition Monitoring. Journal of Management Analytics 2015, 2, 121–137. [CrossRef]
- Tomaszewski, L.; Kołakowski, R. Mobile Services for Smart Agriculture and Forestry, Biodiversity Monitoring, and Water Management: Challenges for 5G/6G Networks. Telecom 2023, 4, 67–99. [CrossRef]
- 5G Americas. Mobile Communications Beyond 2020—The Evolution of 5G Towards the Next G; 5G Americas: Bellevue, WA, USA, 2020; Available online: https://www.5gamericas.org/wp-content/uploads/2020/12/Future-Networks-2020-InDesign-PDF.pdf (accessed on 16 February 2026).
- Giordani, M.; Polese, M.; Mezzavilla, M.; Rangan, S.; Zorzi, M. Towards 6G Networks: Use Cases and Technologies 2020.
- Karthick, G.S.; Sridhar, M.; Pankajavalli, P.B. Internet of Things in Animal Healthcare (IoTAH): Review of Recent Advancements in Architecture, Sensing Technologies and Real-Time Monitoring. SN COMPUT. SCI. 2020, 1, 301. [CrossRef]
- Bello, S.A.; Passaglia, C.L. A Wireless Pressure Sensor for Continuous Monitoring of Intraocular Pressure in Conscious Animals. Ann Biomed Eng 2017, 45, 2592–2604. [CrossRef]
- Chen, Y.; Niimi, M.; Zhang, L.; Tang, X.; Lu, J.; Fan, J. A Simple Telemetry Sensor System for Monitoring Body Temperature in Rabbits—A Brief Report. Animals 2023, 13, 1677. [CrossRef]
- Pariaut, R. Cardiovascular Physiology and Diseases of the Rabbit. Veterinary Clinics of North America: Exotic Animal Practice 2009, 12, 135–144. [CrossRef]
- Williams, P.B.; Schapiro, H.; Yeiser, P.E. Noninvasive Blood Pressure Determination in the Rabbit with a Doppler Ultrasound Probe. Proceedings of the Society for Experimental Biology and Medicine 1979, 161, 417–420. [CrossRef]
- Jekl, V.; Agudelo, C.F.; Hauptman, K. Cardiology in Rodents, Rabbits, and Small Exotic Mammals—Diagnostic Workup. Veterinary Clinics: Exotic Animal Practice 2022, 25, 503–524. [CrossRef]
- Calero Rodriguez, A.; Van Zeeland, Y.Ra.; Schoemaker, N.J.; De Grauw, J.C. Agreement between Invasive and Oscillometric Arterial Blood Pressure Measurement Using a High-Definition Oscillometric Device in Normotensive New Zealand White Rabbits Using Two Different Anaesthetic Protocols. Veterinary Anaesthesia and Analgesia 2021, 48, 679–687. [CrossRef]
- Barter LS, Epstein SE. Comparison of Doppler, Oscillometric, Auricular and Carotid Arterial Blood Pressure Measurements in Isoflurane Anesthetized New Zealand White Rabbits. Vet Anaesth Analg. 2014 Jul;41(4):393-7. Epub 2014 Feb 27. PMID: 24571422. [CrossRef]
- González-González, E.; González-Alonso-Alegre, E.; Montesinos-Barceló, A.; Caro-Vadillo, A. Non-invasive Blood Pressure Measurement in Conscious Rabbits: A Comparison of Doppler Ultrasonic and Oscillometric Devices. Veterinary Record 2024, 195, e4399. [CrossRef]
- Akhigbe, B.I.; Munir, K.; Akinade, O.; Akanbi, L.; Oyedele, L.O. IoT Technologies for Livestock Management: A Review of Present Status, Opportunities, and Future Trends. BDCC 2021, 5, 10. [CrossRef]
- The MathWorks, Inc. Learn More About ThingSpeak; The MathWorks, Inc.: Natick, MA, USA, 2026; Available online: https://thingspeak.mathworks.com/pages/learn_more (accessed on 16 February 2026).
- OpenFog, C. Openfog Reference Architecture for Fog Computing. URL: https://www. openfogconsortium. org/ra 2017.
- Markakis, E.; Mastorakis, G.; Mavromoustakis, C.X.; Pallis, E. Cloud and Fog Computing in 5G Mobile Networks: Emerging Advances and Applications; Institution of Engineering and Technology, 2017; ISBN 1-78561-083-X.
- Rahmani, A.M.; Gia, T.N.; Negash, B.; Anzanpour, A.; Azimi, I.; Jiang, M.; Liljeberg, P. Exploiting Smart E-Health Gateways at the Edge of Healthcare Internet-of-Things: A Fog Computing Approach. Future Generation Computer Systems 2018, 78, 641–658. [CrossRef]
- Texas Instruments. INA122 Datasheet. Available online: https://www.ti.com/lit/ds/symlink/ina122.pdf (accessed on 16 February 2026).
- Espressif Systems. ADC API Reference (ESP-IDF v4.4). Available online: https://docs.espressif.com/projects/esp-idf/en/v4.4/esp32/api-reference/peripherals/adc.html (accessed on 16 February 2026).
- Harrenstien, K. Time Server; RFC 738; Internet Engineering Task Force: Fremont, CA, USA, 1977. Disponible en línea: https://www.rfc-editor.org/rfc/rfc738.html (accedido el 16 de febrero de 2026).
- Mills, D.; Martin, J.; Burbank, J.; Kasch, W. Network Time Protocol Version 4: Protocol and Algorithms Specification; RFC 5905; Internet Engineering Task Force: Fremont, CA, USA, 2010. Disponible en línea: https://www.rfc-editor.org/rfc/rfc5905 (accedido el 16 de febrero de 2026).
- Espressif Systems. Sleep Modes API Reference. Available online: https://docs.espressif.com/projects/esp-idf/en/stable/esp32/api-reference/system/sleep_modes.html (accessed on 16 February 2026).
- INSIBIO. Apoyo a la Investigación—Conejos. Available online: http://insibio.org.ar/apoyo-a-la-investigacion/conejos/ (accessed on 16 February 2026).
- Comolli, J.; d’Ovidio, D.; Adami, C.; Schnellbacher, R. Technological Advances in Exotic Pet Anesthesia and Analgesia. Veterinary Clinics: Exotic Animal Practice 2019, 22, 419–439.
- BIOPAC Systems, Inc. AcqKnowledge Software Guide. Available online: https://www.biopac.com/wp-content/uploads/acqknowledge_software_guide.pdf (accessed on 16 February 2026).
- Limaye, H.; Deshmukh, V.V. ECG Noise Sources and Various Noise Removal Techniques: A Survey. International Journal of Application or Innovation in Engineering & Management 2016, 5, 86–92.
- Mewett, D.T.; Nazeran, H.; Reynolds, K.J. Removing Power Line Noise from Recorded EMG.; IEEE, 2001; Vol. 3, pp. 2190–2193.
- Tompkins, W.J. Biomedical Digital Signal Processing; Prentice Hall New Jersey, 1993; Vol. 237.
- Lyons, R.G. Understanding Digital Signal Processing; 3rd ed.; Prentice Hall: Upper Saddle River, NJ, 2011; ISBN 978-0-13-702741-5.
- Gustafsson, F. Determining the Initial States in Forward-Backward Filtering. IEEE Transactions on signal processing 2002, 44, 988–992.
- De Oliveira, M.A.; Da Rocha, A.M.; Puntel, F.E.; Cavalheiro, G.G.H. Time Series Compression for IoT: A Systematic Literature Review. Wireless Communications and Mobile Computing 2023, 2023, 1–23. [CrossRef]
- Smith, S.W. The Scientist and Engineer’s Guide to Digital Signal Processing; 2nd edition.; California Technical Pub.: San Diego (Calif.), 1999; ISBN 978-0-9660176-7-0.
- Chiarot, G.; Silvestri, C. Time Series Compression Survey. ACM Computing Surveys 2023, 55, 1–32.
- Azar, J.; Makhoul, A.; Barhamgi, M.; Couturier, R. An Energy Efficient IoT Data Compression Approach for Edge Machine Learning. Future Generation Computer Systems 2019, 96, 168–175. [CrossRef]


| Metric | BIOPAC PA (mean ± SD) |
Proposed system (all beats, mean ± SD) |
Relative error (%) |
Proposed system (session mean ± SD, N=10) |
Range (min–max) |
|---|---|---|---|---|---|
| Detected beats (n) | 230 | 230 | — | — | — |
| SBP | 62.24 ± 1.46 | 56.90 ± 2.07 | -8.58 | 56.71 ± 1.74 | 54.14–58.41 |
| DBP | 54.00 ± 1.04 | 55.31 ± 2.21 | +2.43 | 55.10 ± 1.78 | 52.00–56.72 |
| MAP | 56.75 ± 1.17 | 55.84 ± 2.11 | -1.60 | 55.64 ± 1.72 | 52.72–57.28 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





