Submitted:
02 March 2026
Posted:
03 March 2026
You are already at the latest version
Abstract
Ecological vulnerability of coral reefs contrasts sharply with their persistence through geologic time, creating a paradox from mis-scaled assumptions of time, mortality and organismal dimensionality, namely bleaching susceptibility, mortality, and recovery are treated as linear or sequential outcomes. Recursive definitions built on such mis-scaled assumptions generate straw-man inferences by conflating vulnerability with fragility and obscuring cryptic recovery dynamics. Using post hoc meta-analyses integrating datasets on coral bleaching, life history, reproductive strategy, morphology, and taxonomy, I evaluate system behavior across matrixed categories of thermal exposure and observation timing. Susceptibility emerges as a graded physiological response with weak coupling between predictor importance and variance, whereas mortality exhibits thresholded dynamics consistent with collapse behavior. Partial overlap in predictor structure indicates that bleaching does not represent a direct trajectory toward death, but rather a regulated buffering phase preceding potential tissue-level failure. Skeletal architecture consistently appears as a strong predictor across susceptibility and mortality, while taxonomic identity shows weak and variable effects. Recovery dynamics further indicate host–symbiont restructuring consistent with recursive evolutionary filtering rather than deterministic trait replacement. Together, these findings reframe coral bleaching as a regulated physiological state decoupled from mortality and demonstrate how recursive logic frameworks resolve paradoxes of timing, scale, and resilience in coral bleaching dynamics.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Zooxanthellae Density Meta-Database
- i)
- Select a Sample Size (method SS): branch, colony, core, fragment, nubbin, and sample
- ii)
- Method for Tissue Removal (method TR): studies either decalcified the skeleton (decalcification) or stripped tissue from the skeleton (airbrushing and waterpiking). Data were first examined without pooled categories.
- iii)
- Method to determine the Surface Area (method SA); e.g. measured or inferred, were various: aluminum foil, calculated, vernier callipers, graph paper, imaging analyses, measured, paraffin wax, modification of paraffin wax, photography, spectrophotometry, submerged weight, normalized to weight, colony volume, normalized to polyps.
2.2. Defining Phases of Health by Fluorometric Analyses
| Healthy | ±SE | n | Pale | ±SE | n | Bleached | ±SE | n | |||||
| 21.37 | 1.86 | 349 | 56.26 | 1.23 | 195 | 8.15 | 1.01 | 163 | |||||
| |||||||||||||
| Healthy | Sublethal bleaching | Bleached | |||||||||||
| Source | Average | n | ±SE | Average | n | ±SE | Average | n | ±SE | ||||
| Within-colony | 16.42 | 37 | 6.43 | 63.45 | 11 | 2.45 | |||||||
| Temperature | 25.34 | 195 | 2.70 | 56.16 | 141 | 1.49 | 82.76 | 129 | 1.13 | ||||
| Habitat | 8.99 | 75 | 3.43 | 66.13 | 18 | 3.10 | 83 | 3 | 0 | ||||
| Pollutant | 28.54 | 39 | 2.07 | 43.18 | 22 | 2.62 | 74.17 | 27 | 2.32 | ||||
| |||||||||||||
| Source | SS Type III | df | n | MS | F | p | |||||||
| Natural Variation | 76.548 | 3 | 345 | 0.216 | 3.863 | <0.05* | |||||||
| Sublethal bleaching | 9.248 | 3 | 191 | 0.044 | 6.876 | <0.001*** | |||||||
| Sublethal, pollution removed | 8.200 | 2 | 169 | 0.047 | 2.858 | >0.05 | |||||||
| Lethal bleaching | 9.312 | 1 | 155 | 0.056 | 11.292 | <0.01** | |||||||
2.3. Mass Bleaching Response Meta-Database
2.4. Aims and Statistics
2.4.1. Examine Healthy Fluctuations in Zooxanthellae Densities to Define a Bleached State
Examine a Bleaching Definition Through Absolute Zooxanthellae Densities
Dynamic Equations for Absolute Healthy and Bleached Zooxanthellae Densities
Proportional Loss in Zooxanthellae Densities as a Metric to Define Bleaching
Exploratory Analysis of Proportional Variation in Zooxanthellae Densities
Proportional Loss as a Metric to Define Bleaching Lethality
Statistical Exploration of Proportional Loss
Variability in the Proportional Loss Associated with Bleaching Due to Causes
2.4.2. Quantitative Fluorometric Definitions for the Phases of Health
2.4.3. Exploration of Bleaching Responses with Regards to New Perspective of Bleaching
Exploratory Statistics for Bleaching Responses
Exploratory Statistics on Recovery
Post Hoc Groupings
Map Bleaching Through a Matrix of Temperature and Time
Compare and Contrast Bleaching Definitions with Bleaching Responses
Survival of Genetic Material and Disturbance Guilds
3. Results
3.1. The Architecture of Resilience
Limits of a Stable Symbiosis
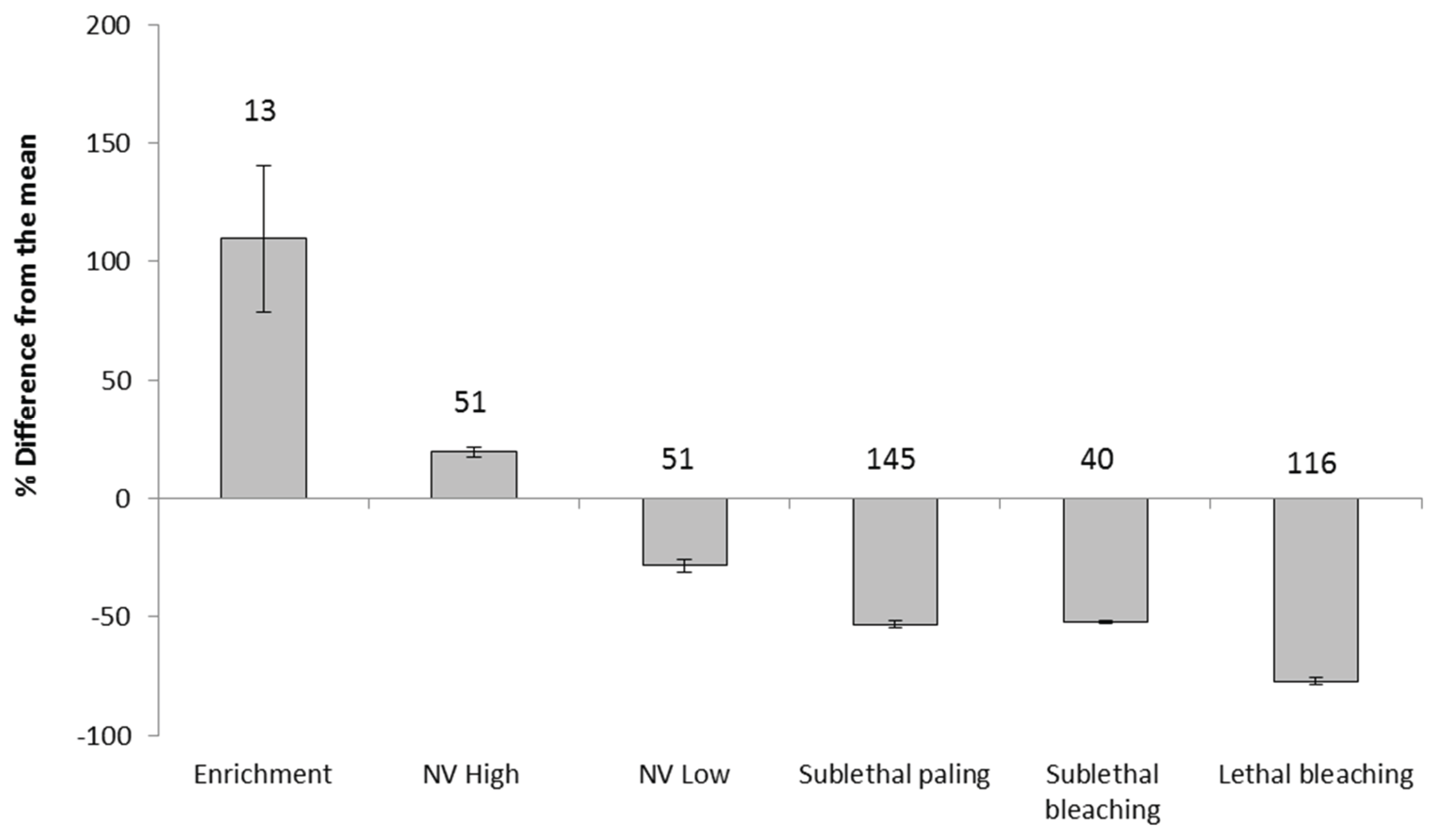
| |||||||
| Factor | Source | Recovery (months) | ±SE | n | |||
| Shelf Position | Inner | 8.00 | 0.00 | 3 | |||
| Middle | 2.29 | 0.33 | 11 | ||||
| Outer | 1.94 | 0.30 | 16 | ||||
| Relief from Weather | No | 7.00 | 0.53 | 43 | |||
| Yes | 3.59 | 0.31 | 80 | ||||
| Thermal Stress Accumulation | Low | 3.98 | 0.49 | 82 | |||
| Medium | 8.20 | 0.75 | 54 | ||||
| High | 10.33 | 1.69 | 6 | ||||
| Anthropogenic Input | Low | 6.72 | 0.44 | 32 | |||
| Medium | 6.67 | 1.33 | 4 | ||||
| High | 12.00 | 0 | 5 | ||||
| Locations | Caribbean | 5.00 | 1.00 | 8 | |||
| Centre of Biodiversity | 10.33 | 1.71 | 20 | ||||
| Eastern Pacific | 6.72 | 0.44 | 32 | ||||
| French Polynesia | 3.29 | 0.27 | 66 | ||||
| Great Barrier Reef | 8.05 | 1.66 | 23 | ||||
| Habitat | Bay | 5.62 | 0.21 | 13 | |||
| Reef Flat | 3.00 | 1.73 | 4 | ||||
| Reef Crest | 1.50 | 0.00 | 14 | ||||
| Reef Slope | 4.15 | 1.04 | 20 | ||||
| Sex | Gonochoric | 4.99 | 0.67 | 30 | |||
| Hermaphroditic | 3.44 | 0.35 | 46 | ||||
| Genus | Acropora | 3.18 | 0.49 | 20 | |||
| Astrea | 4.20 | 1.10 | 5 | ||||
| Montipora | 13.00 | 4.02 | 7 | ||||
| Pavona | 6.43 | 1.15 | 7 | ||||
| Platygyra | 2.85 | 1.10 | 3 | ||||
| Pocillopora | 3.72 | 0.56 | 16 | ||||
| Porites | 3.46 | 0.57 | 11 | ||||
| Psammocora | 7.58 | 1.85 | 6 | ||||
| Bleaching Occurrence | First | 7.15 | 1.25 | 33 | |||
| Second | 4.71 | 0.86 | 55 | ||||
| Third | 6.00 | 0.00 | 26 | ||||
| Fourth | 5.60 | 1.60 | 5 | ||||
| Fifth | 5.85 | 0.10 | 27 | ||||
| Species | Acropora gemmifera | 4.50 | 1.50 | 3 | |||
| Acropora hyacinthus | 3.08 | 0.78 | 7 | ||||
| Acropora millepora | 2.19 | 0.48 | 3 | ||||
| Astrea curta | 4.20 | 1.10 | 5 | ||||
| Pavona cactus | 7.25 | 1.97 | 4 | ||||
| Platygyra daedalea | 2.85 | 1.10 | 3 | ||||
| Pocillopora damicornis | 6.00 | 0.00 | 3 | ||||
| Pocillopora elegans | 6.00 | 0.00 | 3 | ||||
| Pocillopora meandrina | 3.00 | 1.50 | 3 | ||||
| Porites lobata | 2.51 | 0.85 | 4 | ||||
| Transmission of Algae to Offspring | No | 4.58 | 0.53 | 44 | |||
| Yes | 3.42 | 0.43 | 23 | ||||
| Level of Colony Integration | Low | 3.60 | 1.16 | 10 | |||
| Medium | 4.28 | 0.44 | 31 | ||||
| High | 5.96 | 0.81 | 51 | ||||
| Generalization | Generalist | 3.78 | 0.38 | 49 | |||
| Specialist | 4.49 | 0.65 | 29 | ||||
| Reproductive Mode | Brooder | 4.44 | 0.55 | 8 | |||
| Spawner | 4.04 | 0.39 | 67 | ||||
| Family | Acroporidae | 6.14 | 1.24 | 37 | |||
| Agariciidae | 6.08 | 0.68 | 12 | ||||
| Fungiidae | 4.45 | 1.18 | 10 | ||||
| Merulinidae | 5.58 | 1.17 | 20 | ||||
| Mussidae | 4.00 | 0.00 | 3 | ||||
| Pocilloporidae | 4.94 | 0.95 | 24 | ||||
| Poritidae | 4.21 | 0.53 | 17 | ||||
| Psammocoridae | 7.58 | 1.85 | 6 | ||||
| Dominant Topography | Acropora and Montipora | 8.25 | 1.33 | 16 | |||
| Acropora hyacinthus | 6.00 | 0.00 | 16 | ||||
| Montastrea | 4.00 | 0.00 | 4 | ||||
| Pavonids and Pocilloporids | 5.77 | 0.13 | 22 | ||||
| Pocilloporidae, Fungiidae, Acroporidae, and Poritidae | 6.00 | 0.00 | 5 | ||||
| Porites lobata, Pocillopora damicornis, and Pocillopora elegans | 6.00 | 0.00 | 6 | ||||
| Complexity of Skeleton | Complex | 5.67 | 0.72 | 66 | |||
| Robust | 5.48 | 0.58 | 65 | ||||
| |||||||
| Source | SS (Type III) | df | n | MS | F | p | |
| Shelf Position | 8.501 | 2 | 29 | 0.108 | 25.908 | <0.001*** | |
| Relief from Weather | 86.023 | 1 | 122 | 0.593 | 23.972 | <0.001*** | |
| Thermal Stress Accumulation | 144.314 | 2 | 141 | 0.828 | 17.655 | <0.001*** | |
| Anthropogenic Input | 10.106 | 2 | 40 | 0.171 | 10.623 | <0.001*** | |
| Location | 147.165 | 4 | 148 | 0.817 | 9.042 | <0.001*** | |
| Habitat | 57.530 | 3 | 92 | 0.495 | 5.826 | <0.001*** | |
| Sex | 36.405 | 1 | 75 | 0.463 | 4.648 | <0.05* | |
| Genus | 57.856 | 7 | 74 | 0.592 | 4.392 | <0.001*** | |
| Bleaching Occurrence | 145.510 | 4 | 145 | 0.966 | 2.418 | >0.05 | |
| Species | 14.550 | 9 | 37 | 0.309 | 2.127 | >0.05 | |
| Transmission of Algae to Offspring | 33.485 | 1 | 66 | 0.502 | 1.735 | >0.05 | |
| Level of Colony Integration | 73.084 | 2 | 91 | 0.795 | 1.490 | >0.05 | |
| Generalization | 37.160 | 1 | 77 | 0.484 | 0.803 | >0.05 | |
| Reproductive Mode | 35.957 | 1 | 74 | 0.489 | 0.550 | >0.05 | |
| Family | 119.113 | 7 | 128 | 0.955 | 0.536 | >0.05 | |
| Dominant Topography | 35.463 | 6 | 73 | 0.518 | 0.252 | >0.05 | |
| Complexity | 121.788 | 1 | 130 | 0.944 | 0.026 | >0.05 | |
| |||||||
| Source | n | Pearson’s Correlation Coefficient | p | ||||
| Shelf Position | 30 | -0.654 | <0.001*** | ||||
| Anthropogenic Pressure | 41 | 0.533 | <0.001*** | ||||
| Degree Heating Index | 142 | 0.446 | <0.001*** | ||||
| Relief from Weather | 123 | -0.407 | <0.001*** | ||||
| Sex | 76 | -0.243 | <0.05* | ||||
| Location | 149 | -0.173 | <0.05* | ||||
| *Significant, **strongly significant, ***highly significant. | |||||||
| |||||||
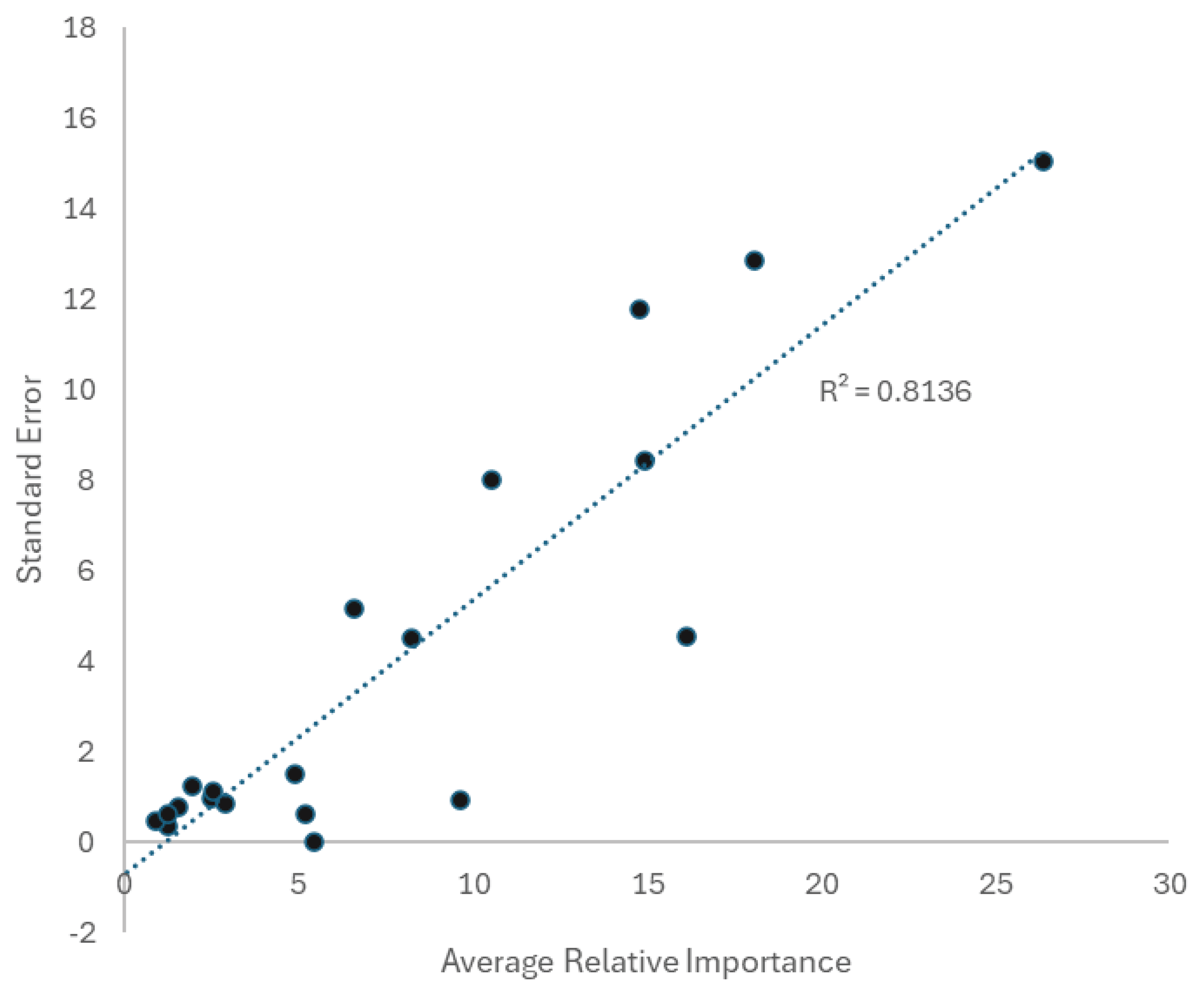
| Factor | Importance | Source | F | Relative Importance | |||
| Weather | 63.5% | Relief from Weather | 23.972 | 36.58% | |||
| Degree Heating Index | 17.655 | 26.94% | |||||
| Biology and Ecology | 16.0% | Habitat | 5.826 | 8.89% | |||
| Sex | 4.648 | 7.09% | |||||
| Location | 13.8% | Location | 9.042 | 13.80% | |||
| Taxonomy | 6.7% | Genus | 4.392 | 6.70% | |||
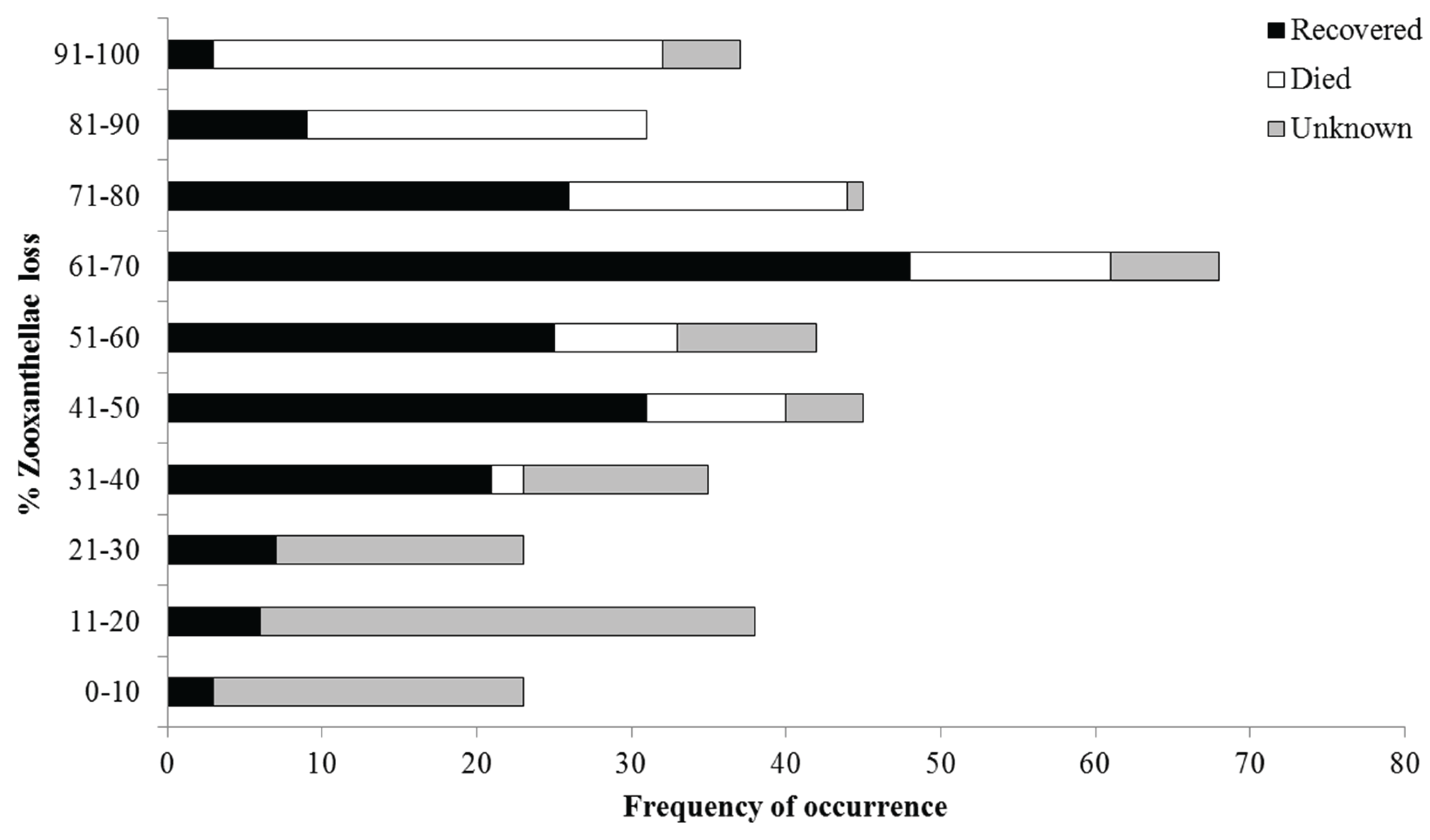
3.2. Defining Bleaching
3.3. The System Architecture of Mass Bleaching Responses
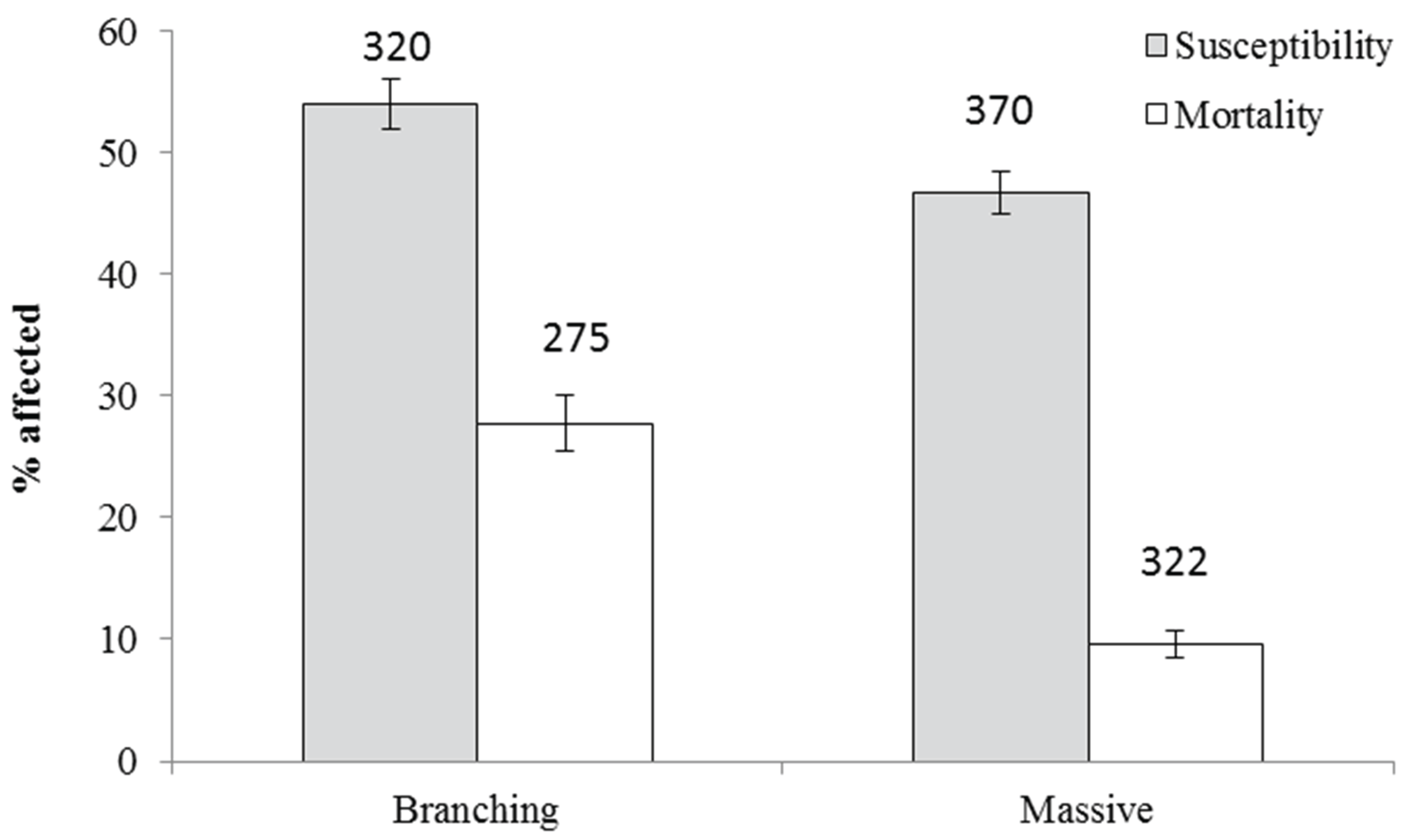
3.3.1. Factored Components of Mass Bleaching Susceptibility and Mortality
| Field | Factor | Average Cladal Count | Counts | SE | Upper Range |
|---|---|---|---|---|---|
| Complexity of Skeleton | Complex | 1.26 | 111 | 0.073 | 5 |
| Robust | 1.17 | 156 | 0.037 | 4 | |
| Reproductive Mode | Brooder | 1.60 | 5 | 0.245 | 2 |
| Spawner | 1.37 | 68 | 0.115 | 5 | |
| Sexual Reproduction |
Gonochoric | 1.06 | 17 | 0.059 | 2 |
| Hermaphroditic | 1.47 | 59 | 0.131 | 5 | |
| Transmission of Algae to Offspring | No | 1.42 | 60 | 0.126 | 5 |
| Yes | 1.43 | 7 | 0.297 | 3 | |
| Morphology | Branching | 1.19 | 27 | 0.085 | 3 |
| Free living | 1 | 6 | 0 | 1 | |
| Massive | 1.31 | 13 | 0.133 | 2 | |
| Sub-massive | 1.43 | 7 | 0.202 | 2 | |
| Tabular | 1 | 3 | 0 | 1 | |
| Genera | Acropora | 1.57 | 37 | 0.2 | 5 |
| Cyphastrea | 1 | 5 | 0 | 1 | |
| Echinophyllia | 1 | 6 | 0 | 1 | |
| Echinopora | 1.25 | 8 | 0.164 | 2 | |
| Favia | 1.29 | 7 | 0.184 | 2 | |
| Fungia | 1 | 9 | 0 | 1 | |
| Galaxea | 1.57 | 7 | 0.202 | 2 | |
| Goniastrea | 1.25 | 8 | 0.164 | 2 | |
| Goniopora | 1 | 6 | 0 | 1 | |
| Hydnophora | 1.6 | 5 | 0.245 | 2 | |
| Leptoseris | 1 | 5 | 0 | 1 | |
| Lobophyllia | 1.17 | 6 | 0.167 | 2 | |
| Montastrea | 1.33 | 6 | 0.211 | 2 | |
| Montipora | 1 | 17 | 0 | 1 | |
| Pachyseris | 1 | 5 | 0 | 1 | |
| Pavona | 1 | 6 | 0 | 1 | |
| Platygyra | 1 | 7 | 0 | 1 | |
| Pocillopora | 2 | 6 | 0.516 | 4 | |
| Porites | 1 | 10 | 0 | 1 | |
| Psammocora | 1 | 9 | 0 | 1 | |
| Symphyllia | 1 | 5 | 0 | 1 | |
| Turbinaria | 1.5 | 6 | 0.224 | 2 |
| Thermal stress accumulation | Timing of observations | Complexity of skeleton | Susceptibility | ±SE | n | Mortality | ±SE | BEQ (S-M)/S |
|---|---|---|---|---|---|---|---|---|
| Low | Beginning | Complex | 39.51 | 2.07 | 280 | 7.10 | 1.16 | 0.82 |
| Robust | 40.60 | 2.00 | 271 | 4.95 | 0.96 | 0.88 | ||
| Middle | Complex | 48.63 | 3.76 | 87 | 9.85 | 2.58 | 0.80 | |
| Robust | 35.67 | 3.52 | 89 | 3.09 | 0.77 | 0.91 | ||
| End | Complex | 37.50 | 3.46 | 99 | 16.41 | 3.04 | 0.56 | |
| Robust | 33.76 | 3.07 | 108 | 7.74 | 1.62 | 0.77 | ||
| Medium | Beginning | Complex | 51.22 | 5.19 | 54 | 8.48 | 3.12 | 0.83 |
| Robust | 37.78 | 3.25 | 107 | 3.01 | 0.87 | 0.92 | ||
| Middle | Complex | 33.20 | 5.45 | 25 | 14.52 | 4.26 | 0.56 | |
| Robust | 29.86 | 5.06 | 36 | 3.28 | 1.14 | 0.89 | ||
| End | Complex | 68.10 | 3.86 | 94 | 48.94 | 4.48 | 0.28 | |
| Robust | 64.34 | 3.90 | 80 | 41.70 | 4.54 | 0.35 | ||
| High | Beginning | Robust | 55.73 | 13.50 | 10 | 17.93 | 3.57 | 0.68 |
| Complex | 42.45 | 14.12 | 11 | 3.57 | 9.28 | 0.92 | ||
| End | Complex | 80.45 | 7.50 | 20 | 70.85 | 9.57 | 0.12 | |
| Robust | 100.00 | 0.00 | 20 | 58.54 | 10.81 | 0.41 |
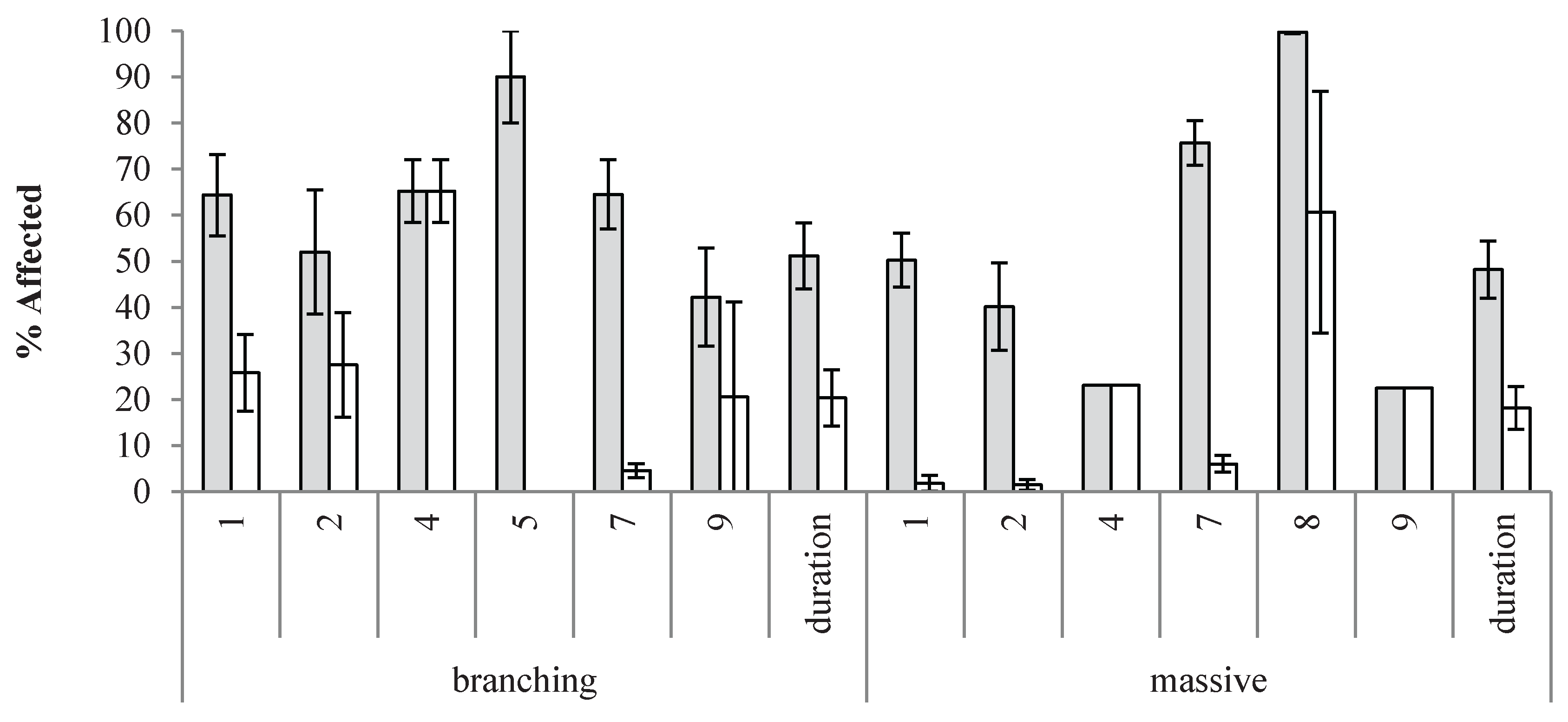
3.3.2. A Thermal and Time-Relative Matrixed View of Bleaching Responses
Overview of Matrixed Descriptive Statistics
Systems Architecture of Bleaching Responses
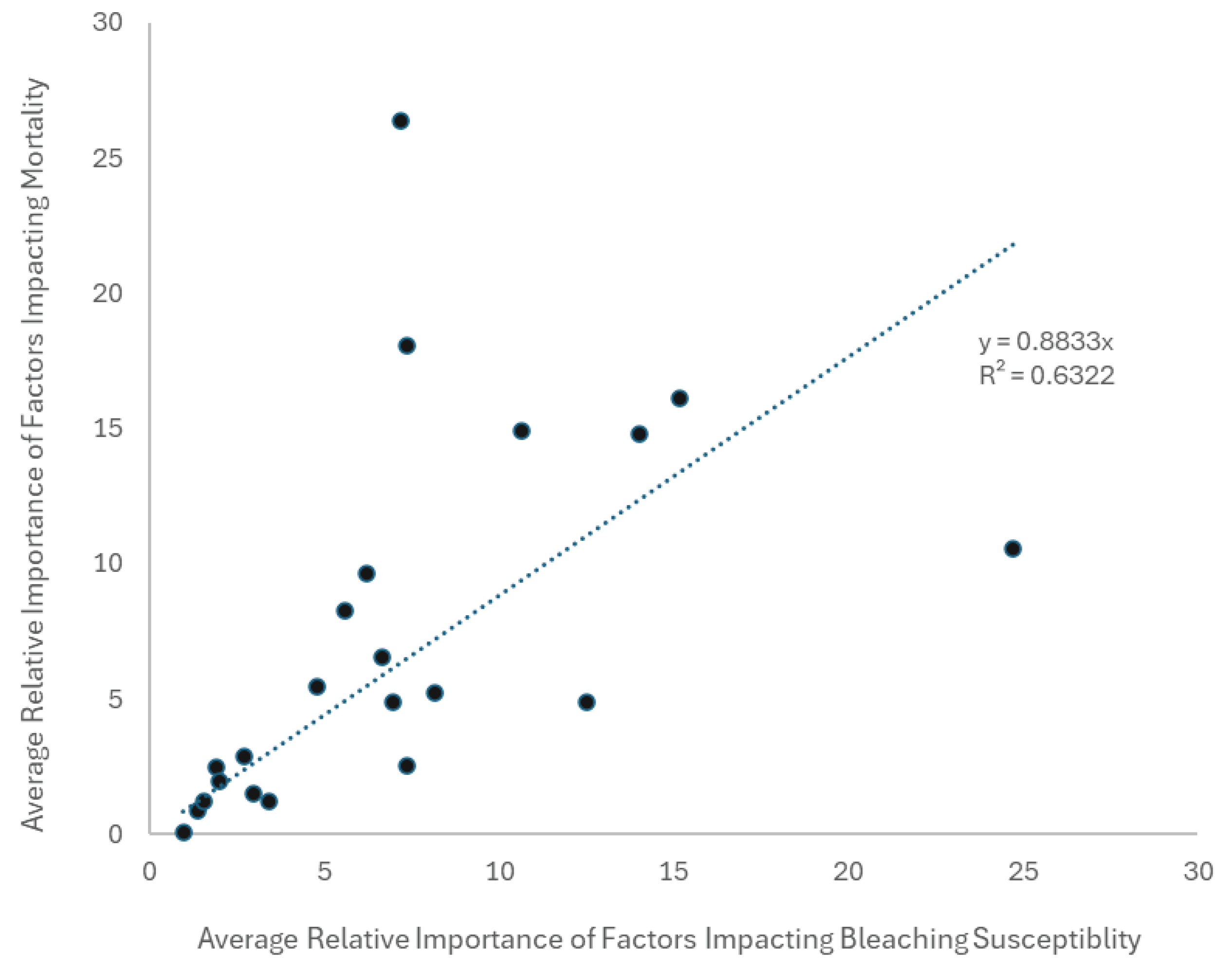
3.3.3. Unravelling the Mechanisms of Bleaching Recovery
3.3.4. Quantitative Resolution of the Paradox of Efficiency vs. Mortality
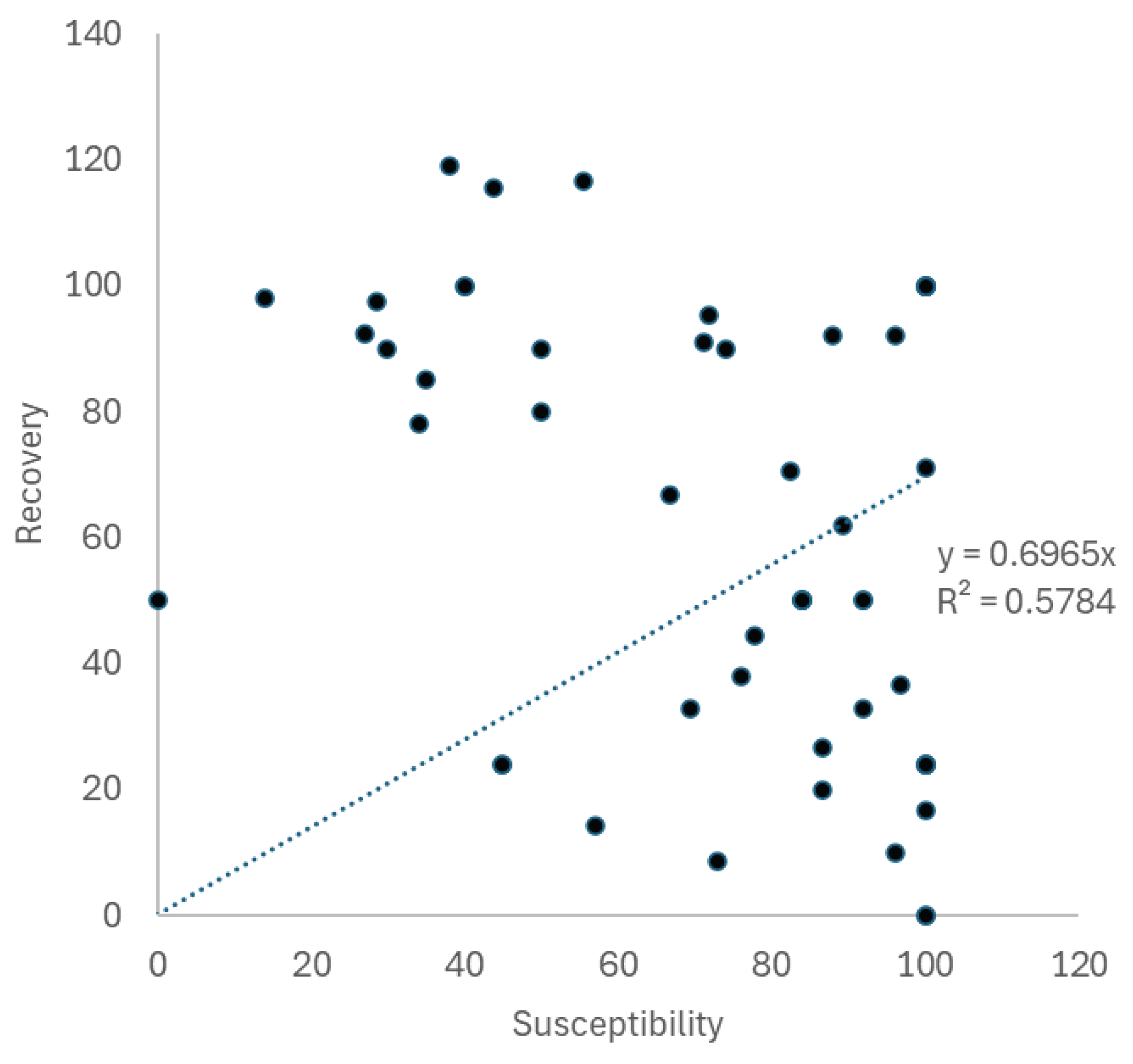
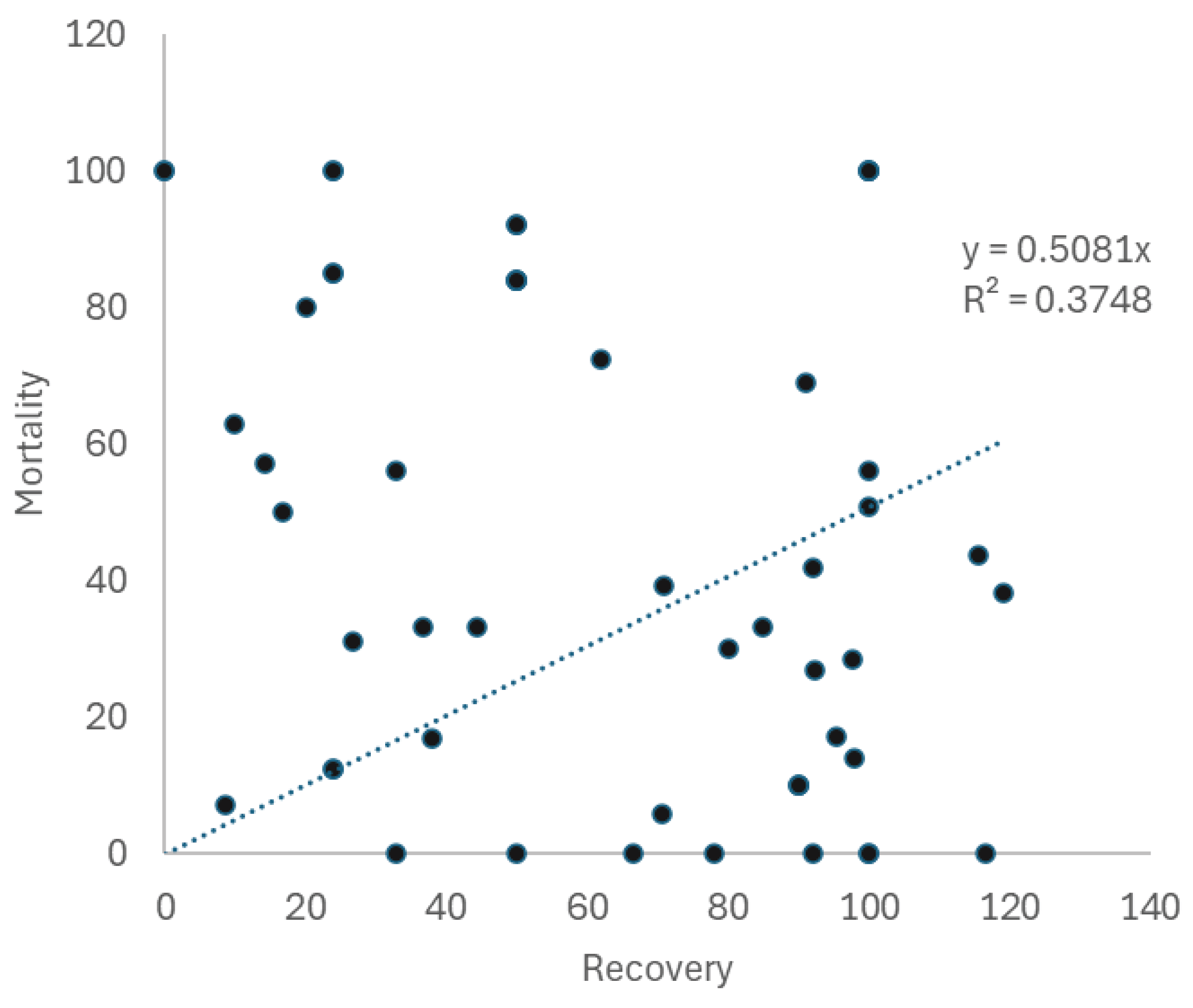
| Thermal stress accumulation | Timing of observations | Reproductive mode | Susceptibility | ±SE | n | Mortality | ±SE | BEQ (S-M)/S |
|---|---|---|---|---|---|---|---|---|
| Low | Beginning | Spawner | 39.36 | 2.84 | 157 | 8.10 | 1.50 | 0.79 |
| Brooder | 39.16 | 4.06 | 62 | 2.88 | 1.24 | 0.93 | ||
| Middle | Spawner | 44.40 | 5.47 | 41 | 8.46 | 2.91 | 0.81 | |
| Brooder | 50.49 | 6.33 | 29 | 5.09 | 1.39 | 0.90 | ||
| Both | 14 | 6.67 | 4 | 2.75 | 1.89 | 0.80 | ||
| End | Brooder | 47.20 | 10.68 | 12 | 33.76 | 10.17 | 0.28 | |
| Spawner | 48.47 | 3.99 | 86 | 18.52 | 3.09 | 0.62 | ||
| Medium | Beginning | Spawner | 54.20 | 3.55 | 89 | 5.37 | 1.77 | 0.90 |
| Brooder | 26.74 | 5.21 | 31 | 4.05 | 1.82 | 0.85 | ||
| Both | 66.20 | 21.20 | 5 | 2.00 | 2.00 | 0.97 | ||
| End | Spawner | 63.63 | 4.02 | 87 | 44.13 | 4.59 | 0.31 | |
| Brooder | 70.75 | 5.84 | 25 | 32.67 | 7.05 | 0.54 | ||
| High | Beginning | Spawner | 48.07 | 10.76 | 18 | 12.14 | 5.68 | 0.75 |
| Brooder | 53.00 | 24.95 | 3 | 0.00 | 0.00 | 1 | ||
| End | Brooder | 100.00 | 0.00 | 8 | 75.50 | 16.04 | 0.24 | |
| Spawner | 90.22 | 4.99 | 23 | 64.74 | 9.72 | 0.28 |
| Thermal stress accumulation | Timing of observations | Sex | Susceptibility | ±SE | n | Mortality | ±SE | BEQ (S-M)/S |
|---|---|---|---|---|---|---|---|---|
| Low | Beginning | Hermaphroditic | 40.34 | 2.99 | 142 | 8.99 | 1.64 | 0.78 |
| Gonochoric | 37.38 | 3.71 | 77 | 2.26 | 1.04 | 0.94 | ||
| Middle | Gonochoric | 50.07 | 6.02 | 33 | 7.47 | 3.16 | 0.85 | |
| Hermaphroditic | 42.20 | 5.40 | 40 | 6.47 | 1.81 | 0.85 | ||
| End | Gonochoric | 40.44 | 6.45 | 32 | 24.16 | 6.01 | 0.40 | |
| Hermaphroditic | 51.36 | 4.52 | 67 | 18.36 | 3.38 | 0.64 | ||
| Medium | Beginning | Hermaphroditic | 46.79 | 4.37 | 70 | 6.50 | 2.22 | 0.86 |
| Gonochoric | 46.58 | 4.55 | 55 | 2.36 | 0.99 | 0.95 | ||
| End | Hermaphroditic | 73.53 | 4.27 | 56 | 48.46 | 5.66 | 0.34 | |
| Gonochoric | 56.47 | 5.13 | 55 | 35.18 | 5.49 | 0.38 | ||
| High | Beginning | Gonochoric | 47.64 | 11.80 | 16 | 13.66 | 6.30 | 0.71 |
| Hermaphroditic | 52.40 | 16.91 | 5 | 0.00 | 0.00 | 1 | ||
| End | Hermaphroditic | 100.00 | 0.00 | 16 | 75.50 | 10.96 | 0.24 | |
| Gonochoric | 85.00 | 7.38 | 15 | 59.00 | 12.35 | 0.31 |
| Thermal stress accumulation | Timing of observations | Transmission of algae to offspring | Susceptibility | ±SE | n | Mortality | ±SE | BEQ (S-M)/S | |
|---|---|---|---|---|---|---|---|---|---|
| Low | Beginning | Yes | 45.21 | 11.39 | 8 | 9.36 | 4.18 | 0.79 | |
| No | 61.32 | 3.67 | 68 | 7.20 | 2.42 | 0.88 | |||
| Both | 66.20 | 21.20 | 5 | 2.00 | 2.00 | 0.97 | |||
| Middle | No | 44.29 | 6.10 | 33 | 5.76 | 1.88 | 0.87 | ||
| Yes | 48.35 | 7.59 | 22 | 2.39 | 0.60 | 0.95 | |||
| End | No | 50.79 | 4.69 | 61 | 18.73 | 3.38 | 0.63 | ||
| Yes | 39.76 | 7.27 | 23 | 14.29 | 5.49 | 0.64 | |||
| Medium | Beginning | Yes | 45.21 | 11.39 | 8 | 9.36 | 4.18 | 0.79 | |
| No | 61.32 | 3.67 | 68 | 7.20 | 2.42 | 0.88 | |||
| Both | 66.20 | 21.20 | 5 | 2.00 | 2.00 | 0.97 | |||
| End | No | 56.45 | 5.83 | 42 | 27.18 | 4.79 | 0.52 | ||
| Yes | 50.30 | 6.54 | 25 | 19.38 | 6.25 | 0.61 | |||
| High | End | No | 82.50 | 13.89 | 6 | 63.17 | 18.49 | 0.23 | |
| Yes | 92.50 | 5.12 | 16 | 57.06 | 12.57 | 0.38 | |||
| Thermal stress accumulation | Timing of observations | Colony integration | Susceptibility | ±SE | n | Mortality | ±SE | BEQ (S-M)/S |
|---|---|---|---|---|---|---|---|---|
| Low | Beginning | Low | 25.45 | 4.39 | 43 | 0.95 | 0.46 | 0.96 |
| Medium | 38.92 | 2.77 | 148 | 3.13 | 0.65 | 0.92 | ||
| High | 37.71 | 2.75 | 169 | 13.50 | 1.97 | 0.64 | ||
| Middle | Low | 30.79 | 13.94 | 7 | 2.69 | 1.34 | 0.91 | |
| Medium | 42.07 | 5.03 | 46 | 5.45 | 1.43 | 0.87 | ||
| High | 55.62 | 5.95 | 37 | 20.00 | 5.53 | 0.64 | ||
| End | Low | 31.70 | 12.53 | 9 | 16.52 | 9.00 | 0.48 | |
| Medium | 39.13 | 4.76 | 52 | 12.45 | 2.68 | 0.68 | ||
| High | 55.33 | 5.61 | 50 | 28.60 | 5.19 | 0.48 | ||
| Medium | Beginning | Low | 38.39 | 4.87 | 18 | 1.28 | 0.76 | 0.97 |
| Medium | 56.35 | 3.58 | 83 | 3.95 | 1.00 | 0.93 | ||
| High | 34.70 | 6.33 | 37 | 10.08 | 4.27 | 0.71 | ||
| Middle | Medium | 21.71 | 4.59 | 7 | 13.43 | 4.95 | 0.38 | |
| High | 42.25 | 12.71 | 8 | 42.25 | 12.71 | 0 | ||
| End | Medium | 53.45 | 4.61 | 68 | 28.61 | 4.37 | 0.47 | |
| High | 72.62 | 4.40 | 60 | 51.73 | 5.70 | 0.29 | ||
| High | Beginning | Medium | 23.86 | 13.60 | 7 | 0.00 | 0.00 | 1 |
| High | 58.61 | 10.47 | 17 | 21.32 | 8.92 | 0.64 | ||
| End | Medium | 55.00 | 15.33 | 5 | 16.60 | 11.92 | 0.70 | |
| High | 97.22 | 2.78 | 36 | 82.49 | 6.17 | 0.15 |
| Thermal stress accumulation | Timing of observations | Bleaching occurrence | Susceptibility | ±SE | n | Mortality | ±SE | (S-M)/S | |
|---|---|---|---|---|---|---|---|---|---|
| Low | Beginning | First | 47.04 | 1.97 | 270 | 9.60 | 1.38 | 0.80 | |
| Second | 37.67 | 2.46 | 204 | 6.88 | 1.32 | 0.82 | |||
| Third | 9.98 | 2.02 | 54 | 4.19 | 1.04 | 0.58 | |||
| Fourth | 50.55 | 3.26 | 91 | 0.06 | 0.06 | 1 | |||
| Middle | First | 54.35 | 3.02 | 124 | 8.09 | 1.97 | 0.85 | ||
| Second | 14.28 | 2.51 | 38 | 5.43 | 1.72 | 0.62 | |||
| Fourth | 28.13 | 5.90 | 25 | 3.48 | 1.62 | 0.88 | |||
| End | First | 41.71 | 3.17 | 97 | 11.71 | 2.44 | 0.72 | ||
| Second | 25.67 | 3.60 | 64 | 19.07 | 3.49 | 0.26 | |||
| Third | 28.54 | 5.35 | 44 | 3.94 | 2.76 | 0.86 | |||
| Fourth | 80.33 | 7.99 | 9 | 10.89 | 7.30 | 0.86 | |||
| Medium | Beginning | First | 56.53 | 3.91 | 58 | 8.76 | 2.89 | 0.84 | |
| Second | 24.00 | 4.06 | 31 | 9.42 | 2.27 | 0.61 | |||
| Third | 76.42 | 5.44 | 33 | 8.94 | 4.39 | 0.88 | |||
| Middle | First | 22.47 | 3.08 | 57 | 12.70 | 3.00 | 0.43 | ||
| Second | 68.44 | 6.04 | 18 | 9.44 | 3.41 | 0.86 | |||
| End | First | 73.18 | 8.24 | 22 | 66.65 | 8.68 | 0.09 | ||
| Second | 52.40 | 11.25 | 17 | 42.29 | 10.03 | 0.19 | |||
| Third | 99.17 | 0.83 | 6 | 68.33 | 10.78 | 0.31 | |||
| Fourth | 60.10 | 4.65 | 53 | 30.43 | 4.09 | 0.49 | |||
| Fifth | 36.40 | 3.95 | 39 | 7.13 | 3.00 | 0.80 | |||
| High | Beginning | First | 41.62 | 9.21 | 21 | 9.52 | 6.56 | 0.77 | |
| Second | 97.33 | 2.67 | 4 | 54.65 | 6.88 | 0.44 | |||
| Middle | First | 87.10 | 4.72 | 20 | 27.65 | 6.13 | 0.68 | ||
| End | First | 100.00 | 0.00 | 31 | 98.74 | 0.90 | 0.01 | ||
| Second | 58.75 | 11.87 | 12 | 34.00 | 12.46 | 0.42 | |||
| Fifth | 100.00 | 0.00 | 7 | 2.14 | 0.51 | 0.98 |
| Thermal stress accumulation | Timing of observations | Location | Susceptibility | ±SE | n | Mortality | ±SE | (S-M)/S |
|---|---|---|---|---|---|---|---|---|
| Low | Beginning | N. Indian | 70.71 | 8.84 | 15 | 24.33 | 6.79 | 0.66 |
| N. Pacific | 52.57 | 4.03 | 72 | 12.44 | 3.39 | 0.76 | ||
| S. Atlantic | 45.26 | 5.99 | 28 | 2.11 | 1.79 | 0.95 | ||
| S. Pacific | 27.95 | 3.09 | 121 | 0.05 | 0.04 | 1 | ||
| W. Indian | 42.68 | 2.66 | 139 | 1.04 | 0.47 | 0.98 | ||
| W. Pacific | 47.72 | 3.27 | 86 | 13.49 | 2.66 | 0.72 | ||
| Middle | N. Indian | 24.81 | 4.64 | 49 | 17.09 | 4.64 | 0.31 | |
| S. Atlantic | 68.52 | 2.34 | 34 | 5.17 | 0.86 | 0.92 | ||
| S. Indian | 54.07 | 4.38 | 54 | 0.30 | 0.24 | 0.99 | ||
| W. Atlantic | 30.06 | 4.90 | 49 | 4.58 | 1.44 | 0.85 | ||
| End | E. Pacific | 61.87 | 24.99 | 3 | 61.87 | 24.99 | 0 | |
| N. Atlantic | 30.21 | 6.04 | 15 | 15.54 | 2.62 | 0.49 | ||
| N. Indian | 38.47 | 10.91 | 17 | 29.65 | 11.36 | 0.23 | ||
| N. Pacific | 5.72 | 0.93 | 29 | 1.07 | 0.48 | 0.81 | ||
| S. Atlantic | 0.00 | 0.00 | 4 | 0.00 | 0.00 | *** | ||
| S. Indian | 30.98 | 2.92 | 65 | 2.48 | 1.17 | 0.92 | ||
| S. Pacific | 37.34 | 5.64 | 46 | 1.96 | 1.96 | 0.95 | ||
| W. Atlantic | 57.33 | 7.92 | 18 | 26.02 | 6.07 | 0.55 | ||
| W. Indian | 55.79 | 6.65 | 21 | 52.59 | 7.19 | 0.06 | ||
| W. Pacific | 71.40 | 9.65 | 13 | 27.29 | 9.38 | 0.62 | ||
| Medium | Beginning | E. Indian | 0.00 | 0.00 | 4 | 0.00 | 0.00 | *** |
| N. Pacific | 31.84 | 10.49 | 13 | 5.38 | 2.34 | 0.83 | ||
| S. Atlantic | 72.25 | 13.23 | 4 | 0.00 | 0.00 | 1 | ||
| S. Pacific | 38.82 | 6.68 | 11 | 0.00 | 0.00 | 1 | ||
| W. Atlantic | 32.43 | 2.79 | 114 | 2.85 | 0.73 | 0.91 | ||
| W. Indian | 87.58 | 3.55 | 34 | 20.88 | 5.89 | 0.76 | ||
| Middle | E. Indian | 19.23 | 2.67 | 40 | 6.35 | 1.97 | 0.67 | |
| N. Pacific | 61.35 | 6.46 | 23 | 13.35 | 4.51 | 0.78 | ||
| W. Indian | 33.30 | 10.49 | 10 | 33.30 | 10.49 | 0 | ||
| End | E. Pacific | 38.97 | 4.27 | 42 | 9.74 | 3.32 | 0.75 | |
| N. Pacific | 88.13 | 4.54 | 16 | 82.19 | 7.02 | 0.07 | ||
| S. Atlantic | 74.75 | 4.92 | 4 | 0.00 | 0.00 | 1 | ||
| S. Pacific | 74.00 | 17.11 | 4 | 51.00 | 18.89 | 0.31 | ||
| W. Indian | 26.67 | 13.33 | 3 | 0.00 | 0.00 | 1 | ||
| High | Beginning | E. Pacific | 50.53 | 8.77 | 25 | 16.74 | 6.52 | 0.67 |
| Middle | E. Indian | 87.10 | 4.72 | 20 | 27.65 | 6.13 | 0.68 | |
| End | E. Pacific | 91.13 | 4.56 | 31 | 60.77 | 8.22 | 0.33 | |
| N. Pacific | 100.00 | 0.00 | 16 | 100.00 | 0.00 | 0 |
| ||||||||||||||||
| Factor | n | Correlation coefficient | p | |||||||||||||
| Healthy and bleached | 10 | 0.893 | <0.001*** | |||||||||||||
| Healthy and susceptibility | 10 | 0.346 | >0.05 | |||||||||||||
| Healthy and mortality | 10 | 0.431 | >0.05 | |||||||||||||
| Bleached and susceptibility | 10 | 0.059 | >0.05 | |||||||||||||
| Bleached and mortality | 10 | 0.191 | >0.05 | |||||||||||||
| Susceptibility and mortality | 10 | 0.500 | >0.05 | |||||||||||||
| ||||||||||||||||
| Location | Healthy | Bleached | ||||||||||||||
| Average | ±SE | n | Average | ±SE | n | |||||||||||
| East Indian | 7.06 | 1.02 | 20 | 1.34 | 0.32 | 12 | ||||||||||
| East Pacific | 4.63 | 0.71 | 57 | 1.32 | 0.27 | 42 | ||||||||||
| North Atlantic | 1.89 | 0.16 | 13 | 0.44 | 0.12 | 10 | ||||||||||
| North Indian | 2.81 | 0.52 | 38 | 0.33 | 0.19 | 3 | ||||||||||
| North Pacific | 2.29 | 0.17 | 38 | 0.33 | 0.19 | 3 | ||||||||||
| South Atlantic | 3.29 | 0.28 | 18 | 0.82 | 0.26 | 11 | ||||||||||
| Southern Pacific | 2.28 | 0.48 | 23 | 1.03 | 0.19 | 4 | ||||||||||
| West Atlantic | 2.43 | 0.20 | 63 | 0.48 | 0.08 | 48 | ||||||||||
| West Indian | 11.09 | 1.13 | 40 | 3.03 | 0.46 | 25 | ||||||||||
| West Pacific | 2.58 | 0.26 | 103 | 0.93 | 0.15 | 79 | ||||||||||
| ||||||||||||||||
| Location | Susceptibility | Mortality | ||||||||||||||
| Average | ±SE | n | Average | ±SE | n | |||||||||||
| East Indian | 56.13 | 3.53 | 118 | 22.69 | 3.58 | 78 | ||||||||||
| East Pacific | 56.63 | 3.49 | 126 | 28.39 | 3.85 | 112 | ||||||||||
| North Atlantic | 58.89 | 5.90 | 34 | 13.71 | 2.62 | 17 | ||||||||||
| North Indian | 61.25 | 4.54 | 62 | 38.07 | 6.44 | 43 | ||||||||||
| North Pacific | 44.15 | 1.94 | 347 | 24.31 | 2.59 | 223 | ||||||||||
| South Atlantic | 58.76 | 3.56 | 59 | 3.51 | 0.68 | 50 | ||||||||||
| Southern Pacific | 35.30 | 2.16 | 254 | 1.65 | 0.82 | 182 | ||||||||||
| West Atlantic | 45.73 | 1.62 | 463 | 8.81 | 1.02 | 389 | ||||||||||
| West Indian | 56.23 | 2.19 | 288 | 30.65 | 2.36 | 277 | ||||||||||
| West Pacific | 49.65 | 2.65 | 144 | 20.50 | 2.90 | 116 | ||||||||||
| ||||||||||||||||
| Factor | Natural variation | Sublethal bleaching | Lethal bleaching | |||||||||||||
| Location | LCI | Average | n | ±SE | Average | n | ±SE | Average | n | ±SE | ||||||
| East Indian | Low | 24.12 | 8 | 1.5 | 62 | 3 | 0 | 81.00 | 3 | 0.00 | ||||||
| Medium | 29.00 | 9 | 7.5 | 53.75 | 6 | 3.91 | 85.00 | 6 | 0.00 | |||||||
| East Pacific | Medium | 25.64 | 36 | 2.24 | 51.14 | 21 | 3.28 | 80.52 | 21 | 3.36 | ||||||
| High | 34.82 | 17 | 9.61 | 47.00 | 13 | 4.07 | 82.80 | 13 | 1.68 | |||||||
| North Atlantic | Medium | 45.40 | 5 | 12.00 | 51.83 | 9 | 3.92 | 78.69 | 9 | 4.77 | ||||||
| North Indian | High | 21.67 | 16 | 3.77 | ||||||||||||
| North Pacific | Medium | 26.00 | 3 | 0 | 48.06 | 5 | 3.64 | 83.38 | 5 | 6.66 | ||||||
| High | 32.38 | 29 | 2.81 | 64.20 | 10 | 5.81 | 94.5 | 10 | 0.67 | |||||||
| South Atlantic | High | 8.71 | 14 | 2.68 | 55.00 | 9 | 10.87 | 78.9 | 9 | 6.21 | ||||||
| South Pacific | High | 19.06 | 17 | 4.92 | 56.50 | 4 | 3.50 | 83.75 | 4 | 0.25 | ||||||
| West Atlantic | Medium | 33.84 | 44 | 4.28 | 47.72 | 11 | 1.56 | 87.45 | 11 | 2.34 | ||||||
| High | 20.40 | 15 | 5.44 | 37.47 | 7 | 2.24 | 77.17 | 7 | 4.24 | |||||||
| West Indian | Medium | 68.77 | 33 | 5.87 | 67.78 | 28 | 2.67 | |||||||||
| High | 38.57 | 7 | 7.67 | 45.00 | 3 | 0.00 | 68.00 | 3 | 0.00 | |||||||
| West Pacific | Low | 3.00 | 9 | 1.50 | 57.00 | 6 | 12.52 | 73.00 | 6 | 8.94 | ||||||
| Medium | 34.50 | 4 | 7.50 | 52.00 | 3 | 0.00 | 63.75 | 3 | 2.75 | |||||||
| High | 19.01 | 78 | 2.24 | 59.26 | 52 | 2.23 | 79.04 | 52 | 1.29 | |||||||
| Skeletal Complexity | Reproductive Mode | Sexual Reproduction | Transmission of Algae to Offspring | Level of Colony Integration | |
|---|---|---|---|---|---|
| Mechanism of Influence | Provides micro-habitats and light-scattering properties within the calcium carbonate structure. | Distinguishes between different strategies of larval development and settlement. | Involves the specific methods of gamete exchange and genetic recombination. | Defines whether symbionts are passed directly from the parent (vertical) or acquired from the environment (horizontal). | The degree of physiological connectivity and resource sharing between individual polyps. |
| Why it Matters | It acts as an underlying expression mechanism of bleaching survival by creating internal refugia for zooxanthellae. | This trait serves as a biological driver that determines how a colony expressed resilience or vulnerability during a bleaching event. | It is a key component in the system architecture of mass bleaching responses, influencing long-term recovery patterns. | This is a critical survival mechanism that dictates the stability of the symbiosis across generations | High integration allows the colony to function as a unified system, pushing the "death" of the organism into a perpendicular state through resource redistribution. |
3.3.5. Biological Drivers Undermining Bleaching Response Patterns
Skeletal Complexity as an Underlying Expression Mechanism of Bleaching Survival Mechanisms
Reproductive Mode as an Underlying Expression Mechanism of Bleaching Survival Mechanisms
Mode of Sexual Reproduction as an Underlying Expression Mechanism of Bleaching Survival Mechanisms
Transmission of Algae to Offspring as an Underlying Expression Mechanism of Bleaching Survival Mechanisms
Level of Colony Integration as an Underlying Expression Mechanism of Bleaching Survival Mechanisms
3.3.6. The Impact of the Variability of the Underlying System Components of Time and Location on Bleaching Responses
4. Discussion
4.1. From Variability to System Architecture
Interpreting Zooxanthellae Population Densities
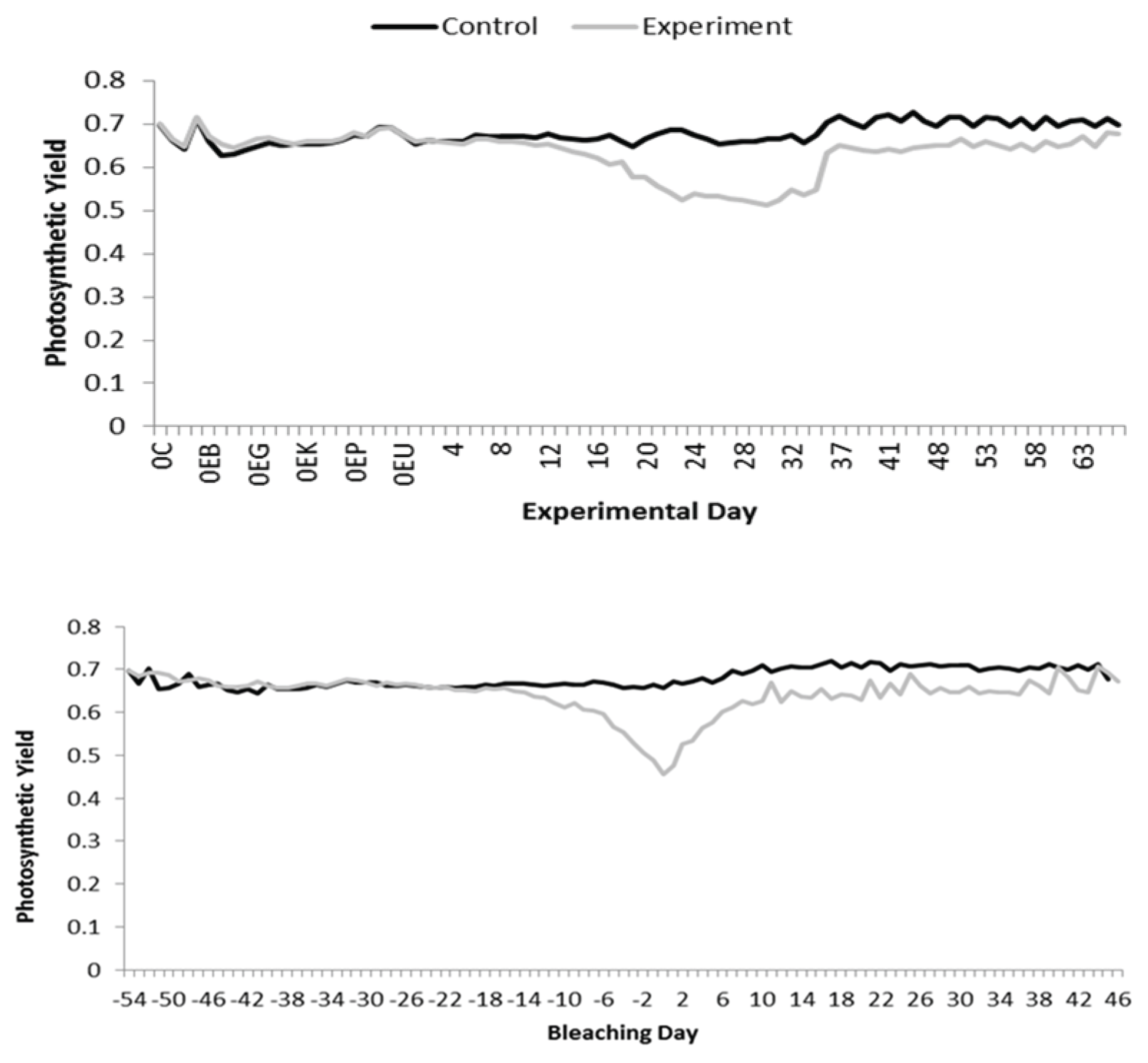
4.2. Bleaching as a Functional, Not Terminal, State
New Framework Monitoring Protocol
- (1)
- Measurement consistency within individuals is required. Comparisons of zooxanthellae population densities, symbiont assemblage composition and associated metrics must be conducted on comparable regions of the same organism across time. Sampling different branches, tissues, or colony regions without accounting for internal heterogeneity introduces variance that cannot be biologically interpreted as response. Functional assessment therefore requires repeated measurements anchored to consistent sampling locations; such that proportional change, recovery, or suppression reflects system behavior rather than spatial artifact.
- (2)
- Temporal alignment between stress exposure and biological response must be assumed explicitly. Full bleaching responses are a temporally structured process, not instantaneous events. Monitoring protocols treat stress response exposure and observed response as synchronous collapse dynamic trajectories into static classifications. Assessments must therefore account for lagged responses, delayed recovery, and asynchronous expression across individuals. Failure to define the temporal window of observation renders comparisons of ‘bleached’ vs. ‘unbleached’ states biologically ambiguous [e.g. 50% successful estimate of mortality based on color alone, 42].
- (3)
- Chronic stressors must be distinguished from acute bleaching responses. Long-term exposure to pollutants or sublethal stressors can suppress baseline zooxanthellae population densities and reduce population variability, producing conditions that superficially resemble bleaching. In these cases, low variance and reduced deviation from the mean should not be interpreted as stability or health – healthy is variable. Rather, chronically suppressed states represent energetically constrained systems that may be highly vulnerable to subsequent disturbance. Protocols that rely solely on absolute density thresholds risk conflating chronic suppression with acute bleaching.
- (4)
- Life history context constrains valid interpretations. Monitoring protocols must be scaled to the life history and evolutionary context of the study organism. For long-lived, modular organisms such as corals, short observation windows are insufficient to resolve functional outcomes. Assumptions regarding recovery potential, persistence, and failure must therefore be stated explicitly, including the temporal scale over which observations are considered biologically meaningful.
- (5)
- Definitions of mortality must be operationally specific. In modular organisms, visual tissue loss does not necessarily constitute organismal death. Remnant tissue reservoirs and endoskeletal refugia may retain the capacity for regeneration long after apparent bleaching or partial mortality. Monitoring protocols that do not account for these recovery pathways systematically underestimate persistence and misclassify functional states. Absence of visible tissue should not be assumed to indicate terminal failure without explicit justification (e.g. Robust or solitary coral).
4.3. Exploring Bleaching Response Architectures
4.3.1. Sources of Bleaching Response Variability
4.3.2. Architectural Determinants of Deep Time Adaptations
4.3.3. Sentinel Disturbance Guilds
4.3.4. Phase Shifts Along Functional Disturbance Guilds
4.3.5. Scope, Constraints, and Interpretive Limits
4.3.6. Deep Time Ecology and Future Implications
4.3.7. Formal Synthesis: A Unifying Interpretive Framework
4.3.8. Speculative Synthesis: A Unifying Interpretive Framework
5. Conclusions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| COTS | Crown of Thorn Seastars |
| ZDD | Zooxanthellae Density Database |
| ZDM | Zooxanthellae Density Meta-database |
| BRD | Bleaching Response Database |
| BRM | Bleaching Response Meta-database |
Appendix A. Figures
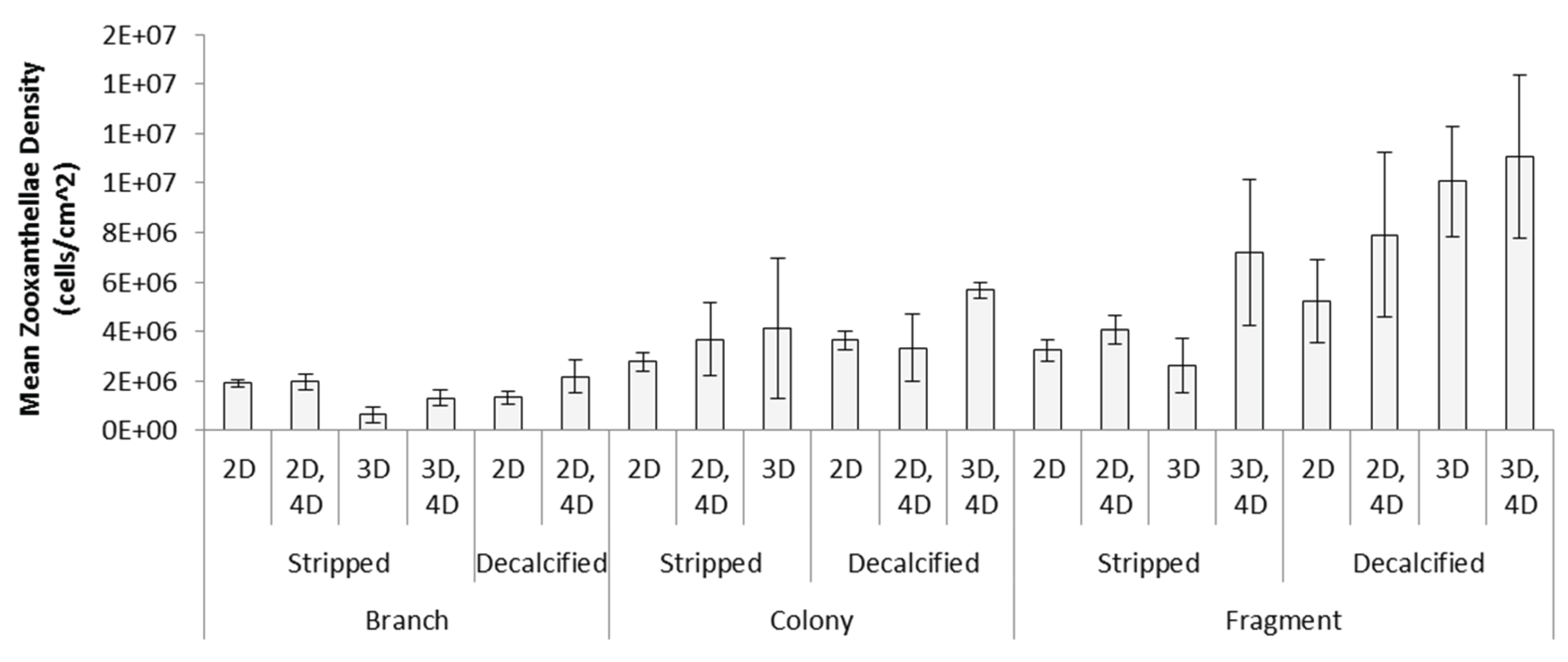
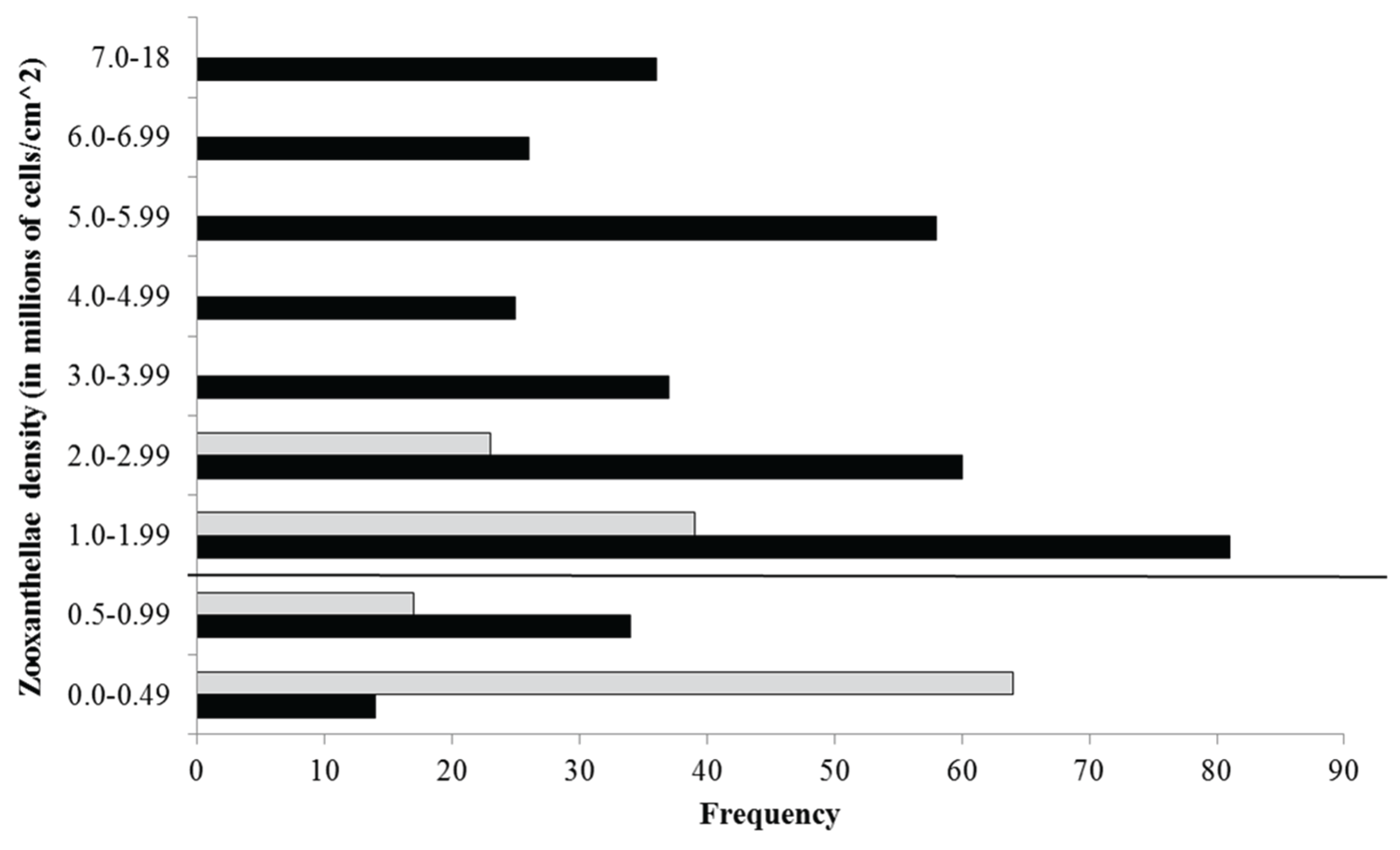
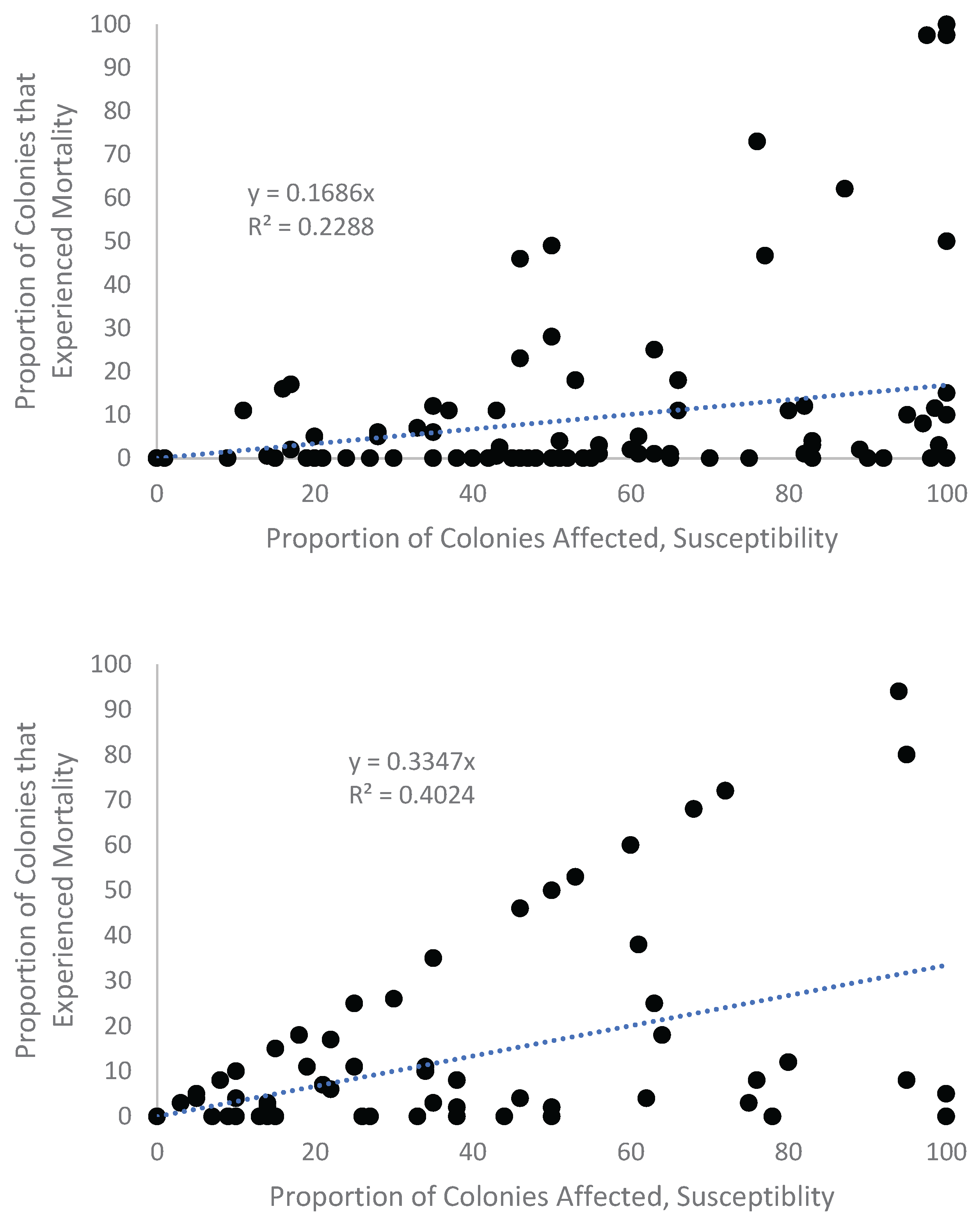
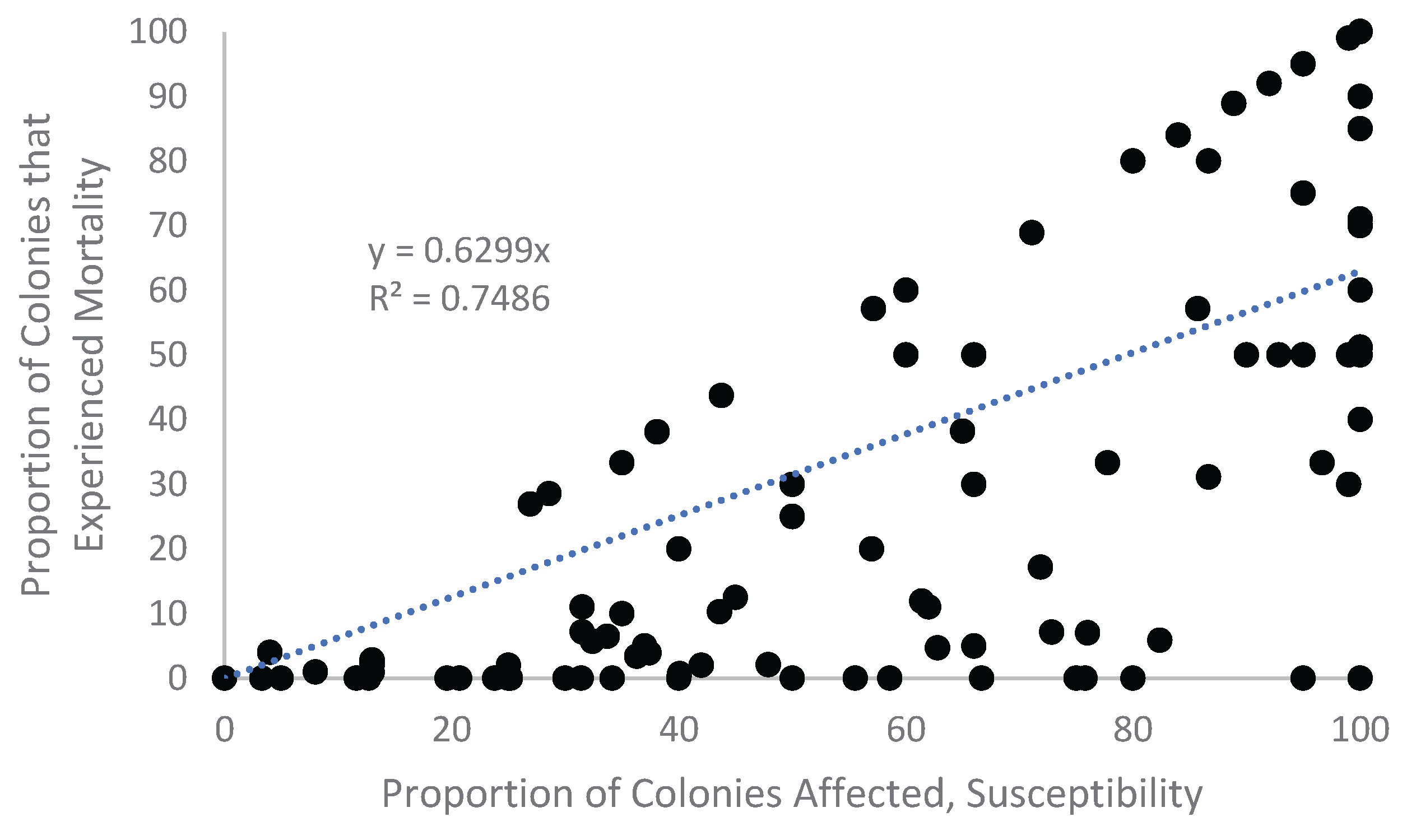
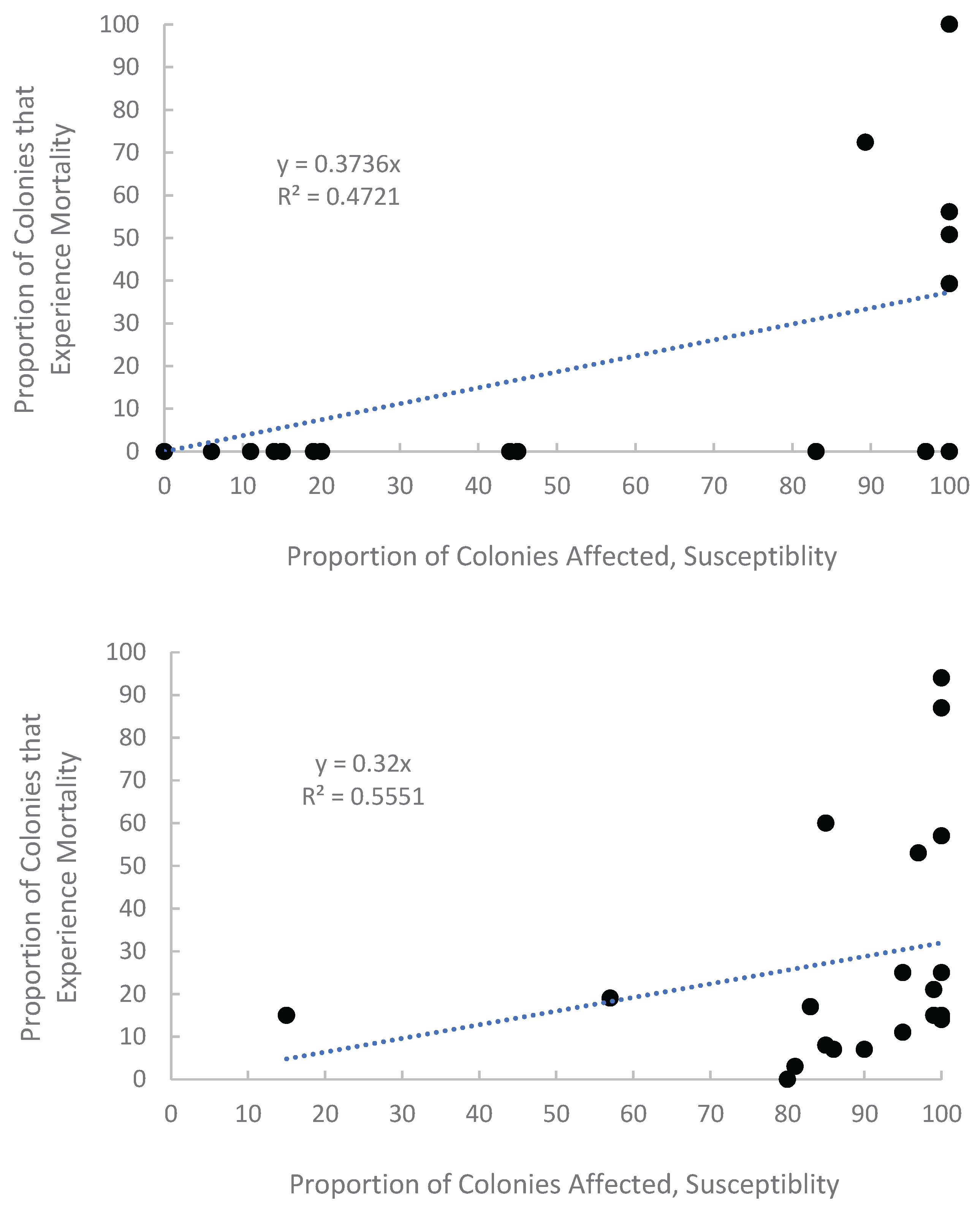
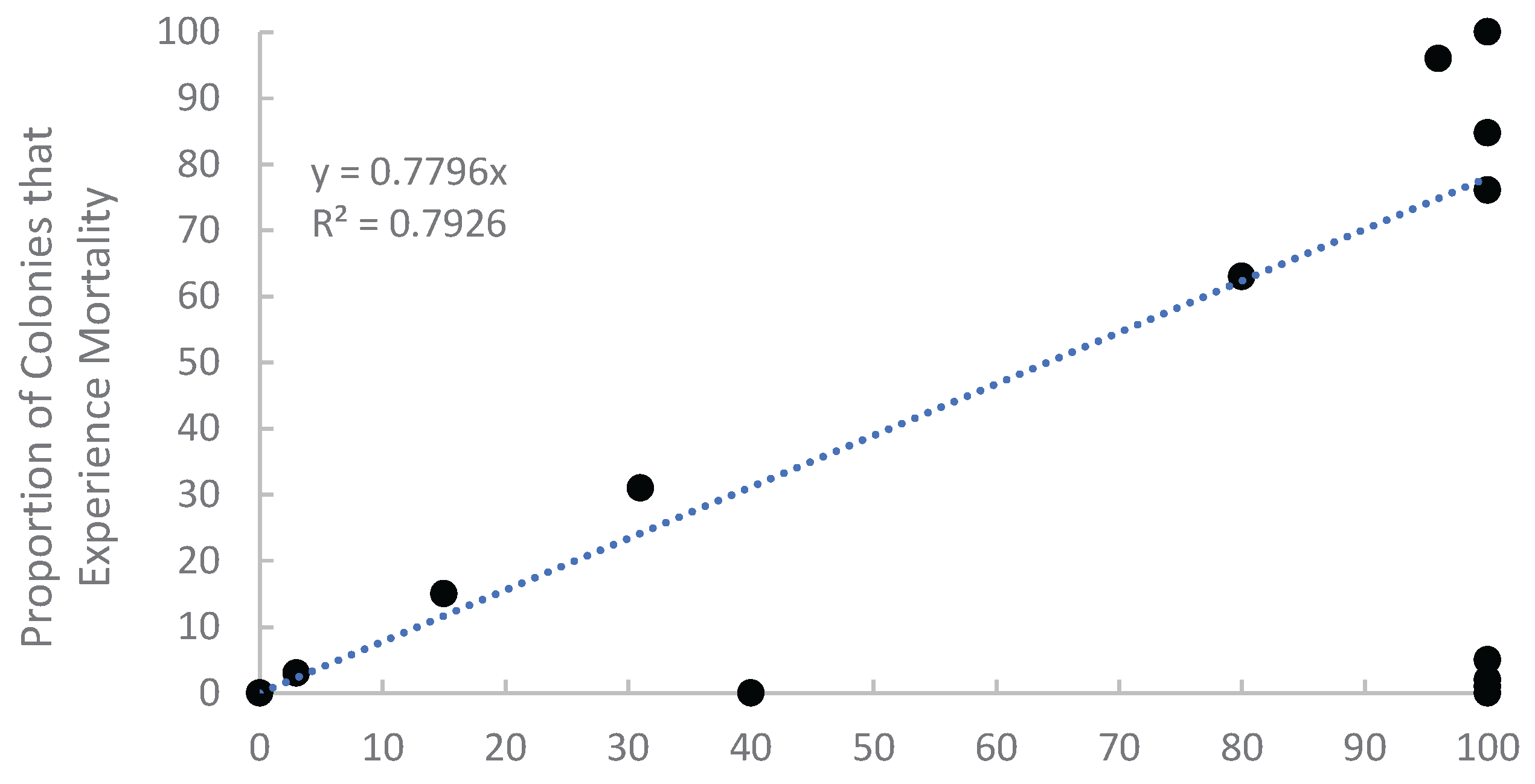
Appendix A.1. Examination of Absolute Healthy and Bleached Zooxanthellae Population Densities
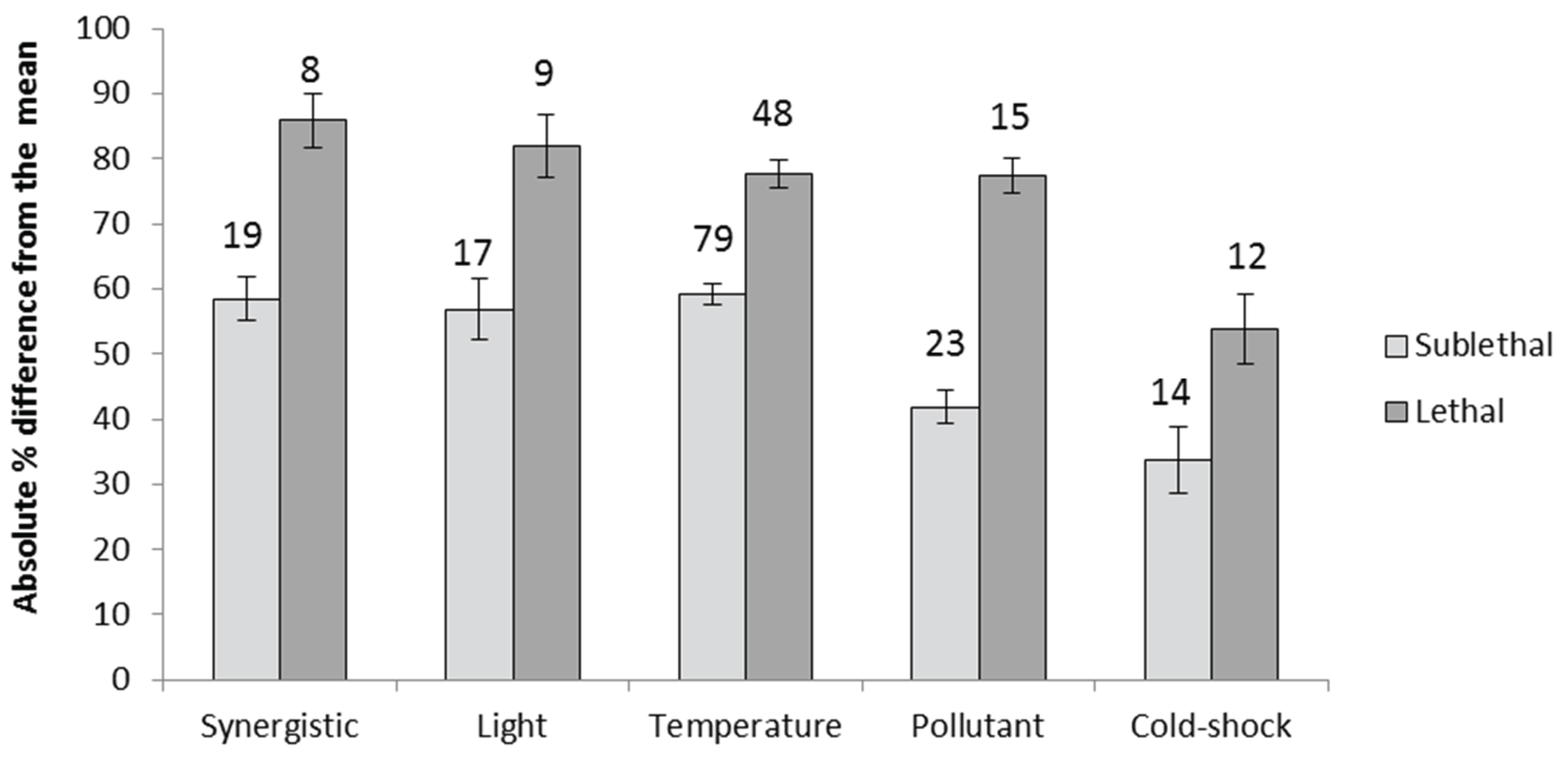
Appendix A.2. Proportional Loss in Zooxanthellae Population Density by Cause of Bleaching
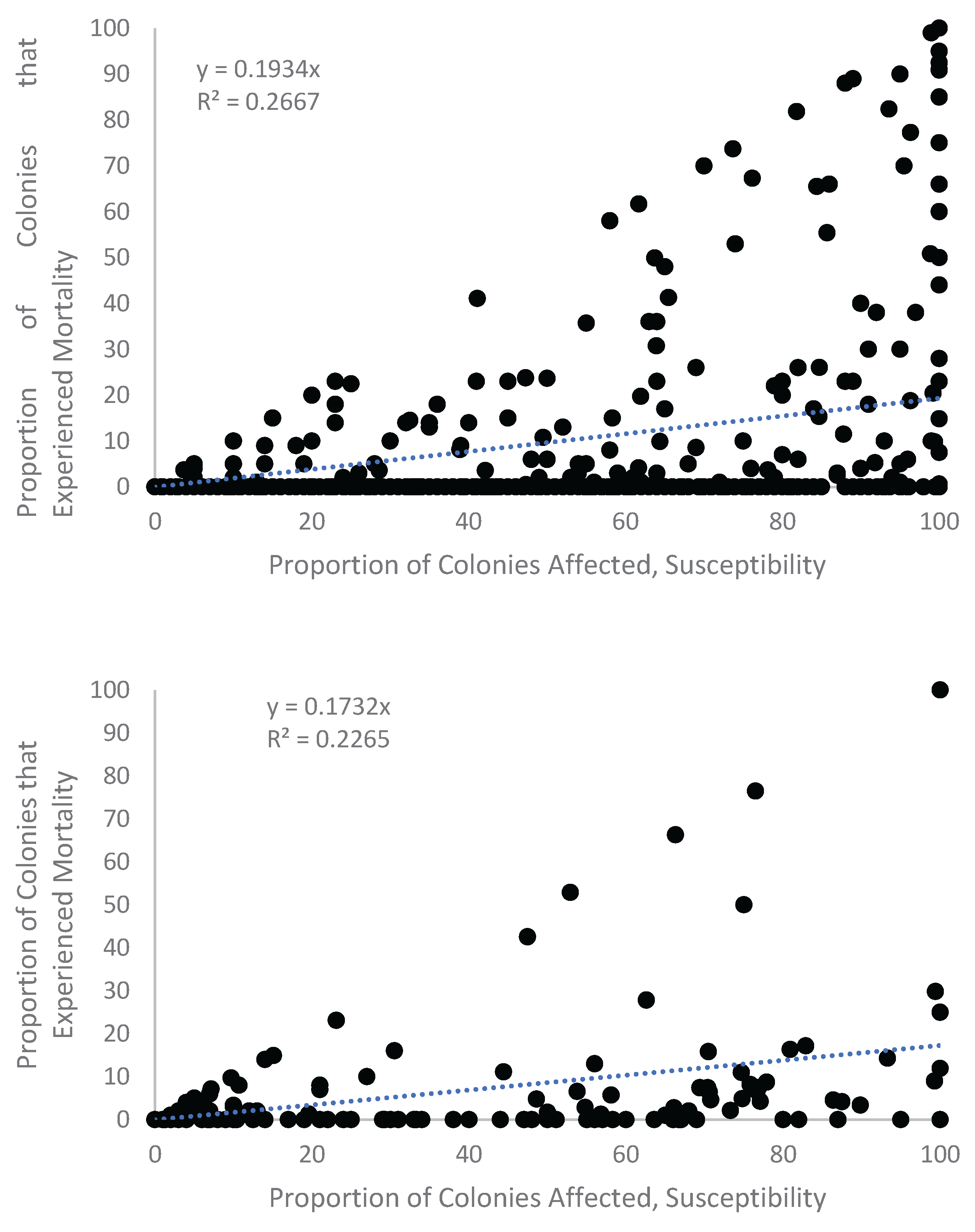
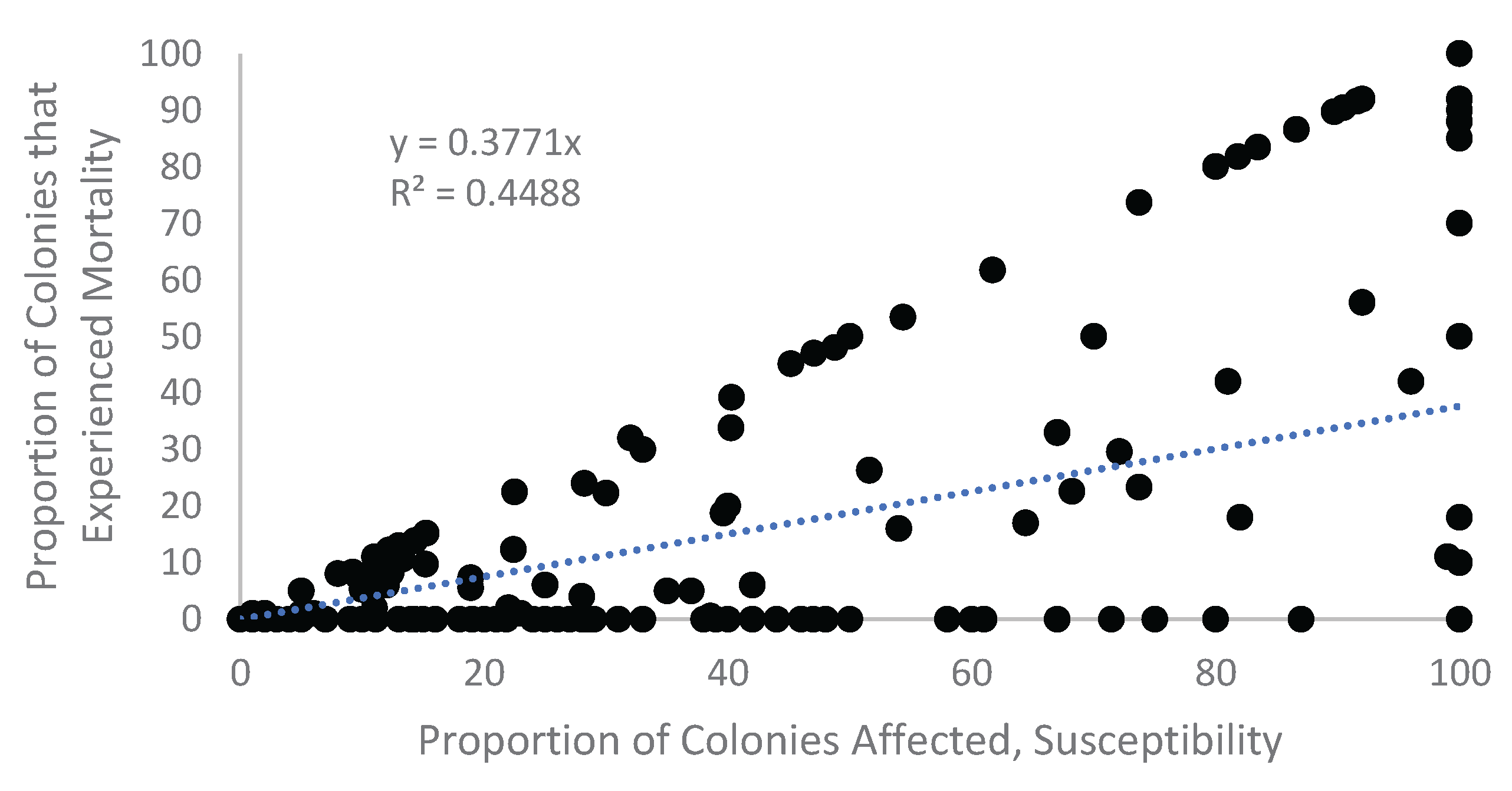
Appendix B. Tables
Dynamic Equations for Absolute Zooxanthellae Densities
| ||||||||||
| Test | Coefficient | n | p | |||||||
| Correlation | 0.804 | 246 | <0.001*** | |||||||
| T-test | 24.039 | 245 | <0.001*** 1 | |||||||
| 1 ***Highly significant interaction. | ||||||||||
| ||||||||||
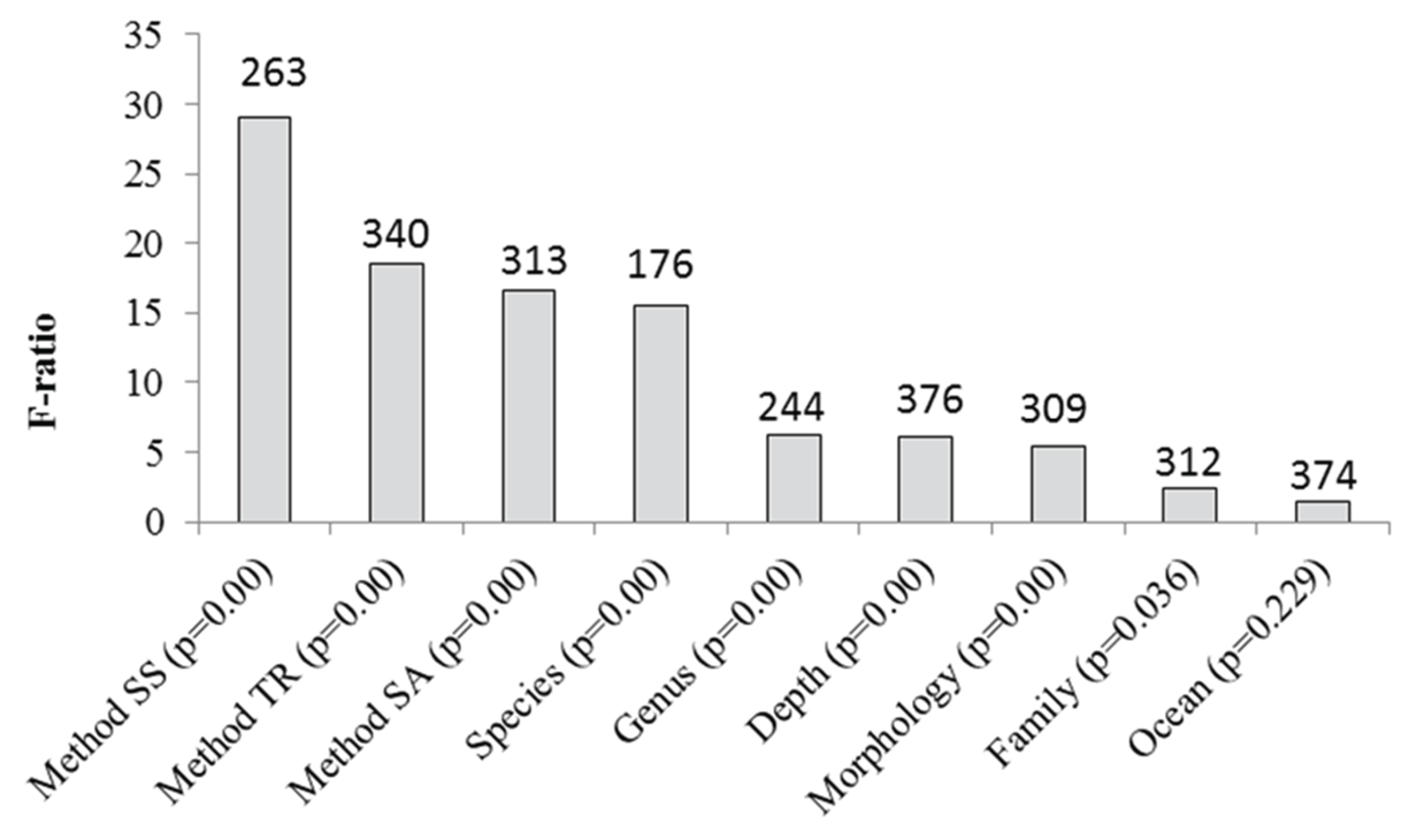
| Source | SS Type III | df | n | MS | F | p | ||||
| Method for Tissue Removal | 18.83 | 1 | 233 | 6.92 | 43.917 | <0.001*** | ||||
| Ocean Basin | 2.07 | 2 | 242 | 2.40 | 36.643 | <0.001*** | ||||
| Source Author | 16.53 | 13 | 178 | 2.74 | 33.700 | <0.001*** | ||||
| Surface Area Determination | 17.64 | 3 | 202 | 18.09 | 29.476 | <0.001*** | ||||
| Subregion | 20.70 | 3 | 245 | 6.67 | 22.716 | <0.001*** | ||||
| Genus | 15.00 | 12 | 230 | 6.44 | 19.130 | <0.001*** | ||||
| Location | 18.77 | 10 | 228 | 7.37 | 14.080 | <0.001*** | ||||
| Replication | 10.07 | 2 | 64 | 11.38 | 13.216 | <0.001*** | ||||
| Transmission of Algae | 1.45 | 2 | 210 | 6.21 | 12.886 | <0.001*** | ||||
| Sex | 18.17 | 1 | 227 | 7.61 | 12.620 | <0.001*** | ||||
| Sample Size | 19.22 | 6 | 242 | 6.30 | 11.512 | <0.001*** | ||||
| Species | 10.25 | 15 | 191 | 3.03 | 10.798 | <0.001*** | ||||
| Morphology | 1.92 | 2 | 242 | 7.41 | 9.759 | <0.001*** | ||||
| Generalization | 1.92 | 1 | 241 | 7.71 | 9.058 | <0.01** | ||||
| Reproductive Mode | 16.39 | 2 | 221 | 6.93 | 8.793 | <0.001*** | ||||
| Depth | 14.29 | 3 | 154 | 8.20 | 7.716 | <0.001*** | ||||
| Sampling Time | 19.22 | 10 | 242 | 6.29 | 7.466 | <0.001*** | ||||
| Family | 18.78 | 6 | 238 | 6.79 | 7.438 | <0.001*** | ||||
| Complexity of Skeleton | 19.22 | 1 | 242 | 7.78 | 6.045 | <0.05* | ||||
| Decade | 19.22 | 2 | 242 | 7.67 | 5.300 | <0.01** | ||||
| *Significant, **strongly significant, ***highly significant. | ||||||||||
| ||||||||||
| Grouped Factor | Grouped Relative Importance | Source | F | Relative Importance | ||||||
| Methods | 43.2 | Method for Tissue Removal | 43.917 | 13.6 | ||||||
| Source Author | 33.700 | 10.5 | ||||||||
| Surface Area Determination | 29.476 | 9.1 | ||||||||
| Replication | 13.216 | 4.1 | ||||||||
| Sample Size | 11.512 | 3.6 | ||||||||
| Sampling Time | 7.466 | 2.3 | ||||||||
| Location | 26.8 | Ocean Basin | 36.643 | 11.4 | ||||||
| Subregion | 22.716 | 7.0 | ||||||||
| Location | 14.080 | 4.4 | ||||||||
| Depth | 7.716 | 2.4 | ||||||||
| Decade | 5.300 | 1.6 | ||||||||
| Biology and Ecology | 18.4 | Transmission of Algae | 12.886 | 4.0 | ||||||
| Sex | 12.620 | 3.9 | ||||||||
| Morphology | 9.759 | 3.0 | ||||||||
| Generalization | 9.058 | 2.8 | ||||||||
| Reproductive Mode | 8.793 | 2.7 | ||||||||
| Complexity of Skeleton | 6.045 | 1.9 | ||||||||
| Taxonomy | 11.6 | Genus | 19.130 | 5.9 | ||||||
| Species | 10.798 | 3.4 | ||||||||
| Family | 7.438 | 2.3 | ||||||||
| ||||||||||
| Source | n | Pearson’s Correlation Coefficient | p | |||||||
| Location | 229 | 0.471 | <0.001*** | |||||||
| Method for Tissue Removal | 234 | 0.399 | <0.001*** | |||||||
| Subregion | 246 | -0.370 | <0.001*** | |||||||
| Replication | 65 | -0.351 | <0.001*** | |||||||
| Sampling Time | 243 | 0.317 | <0.001*** | |||||||
| Sex | 228 | -0.230 | <0.001*** | |||||||
| Depth | 155 | -0.287 | <0.001*** | |||||||
| Morphology | 243 | 0.237 | <0.001*** | |||||||
| Transmission of Algae | 211 | -0.217 | <0.001*** | |||||||
| Sample Size | 243 | 0.199 | <0.01** | |||||||
| Generalization | 242 | 0.191 | <0.01** | |||||||
| Source Author | 179 | 0.162 | <0.01** | |||||||
| Complexity of Skeleton | 243 | 0.156 | <0.05* | |||||||
| Ocean Basin | 246 | -0.150 | >0.05* | |||||||
| Reproductive Mode | 222 | -0.112 | >0.05 | |||||||
| Surface Area Determination | 203 | 0.096 | >0.05 | |||||||
| Genus | 231 | 0.087 | >0.05 | |||||||
| Species | 192 | -0.063 | >0.05 | |||||||
| Family | 239 | 0.030 | >0.05 | |||||||
| Decade | 243 | 0.026 | >0.05 | |||||||
| *Significant, **strongly significant, ***highly significant. | ||||||||||
| ||||||||||
| Source | SS Type III | df | n | MS | F | p | ||||
| Method for Tissue Removal | 6.97 | 1 | 233 | 2.57 | 39.928 | <0.001*** | ||||
| Morphology | 3.61 | 3 | 473 | 6.51 | 28.142 | <0.001*** | ||||
| Source Author | 5.94 | 13 | 178 | 1.24 | 24.256 | <0.001*** | ||||
| Transmission of Algae | 0.56 | 2 | 210 | 2.23 | 21.238 | <0.001*** | ||||
| Ocean Basin | 0.72 | 2 | 242 | 2.61 | 18.494 | <0.001*** | ||||
| Genus | 5.90 | 12 | 230 | 1.38 | 17.608 | <0.001*** | ||||
| Surface Area Determination | .046 | 3 | 202 | 2.47 | 15.341 | <0.001*** | ||||
| Subregion | 8.50 | 3 | 246 | 3.08 | 11.158 | <0.001*** | ||||
| Location | 6.94 | 10 | 228 | 2.24 | 9.166 | <0.001*** | ||||
| Sex | 6.94 | 1 | 227 | 2.95 | 8.980 | <0.01** | ||||
| Species | 4.01 | 15 | 191 | 1.35 | 8.072 | <0.001*** | ||||
| Depth | 5.14 | 3 | 154 | 2.96 | 7.610 | <0.001*** | ||||
| Sample Size | 7.22 | 6 | 242 | 2.59 | 7.117 | <0.001*** | ||||
| Family | 6.15 | 6 | 238 | 2.29 | 6.534 | <0.001*** | ||||
| Sampling Time | 7.22 | 10 | 242 | 2.54 | 5.266 | <0.001*** | ||||
| Generalization | 7.19 | 1 | 241 | 2.93 | 5.179 | <0.05* | ||||
| Decade | 7.22 | 2 | 242 | 2.90 | 4.615 | <0.05* | ||||
| Reproductive Mode | 6.70 | 2 | 221 | 2.94 | 4.485 | <0.05* | ||||
| Complexity of Skeleton | 7.22 | 1 | 242 | 2.94 | 4.232 | <0.05* | ||||
| Replication | 2.81 | 2 | 64 | 4.29 | 1.821 | >0.05 | ||||
| *Significant, **strongly significant, ***highly significant. | ||||||||||
| ||||||||||
| Grouped Factor | Grouped Relative Importance | Source | F | Relative Importance | ||||||
| Methods | 37.1 | Method for Tissue Removal | 39.928 | 16.1 | ||||||
| Source Author | 24.256 | 9.8 | ||||||||
| Surface Area Determination | 15.341 | 6.2 | ||||||||
| Samples Size | 7.117 | 2.9 | ||||||||
| Sample Time | 5.266 | 2.1 | ||||||||
| Biology and Ecology | 29.2 | Morphology | 28.142 | 11.4 | ||||||
| Transmission of Algae | 21.348 | 8.6 | ||||||||
| Sex | 8.980 | 3.6 | ||||||||
| Generalization | 5.179 | 2.1 | ||||||||
| Reproductive Mode | 4.485 | 1.8 | ||||||||
| Complexity of Skeleton | 4.232 | 1.7 | ||||||||
| Location% | 20.6 | Subregion | 11.158 | 4.5 | ||||||
| Ocean Basin | 18.494 | 7.5 | ||||||||
| Location | 9.166 | 3.7 | ||||||||
| Depth | 7.610 | 3.1 | ||||||||
| Decade | 4.615 | 1.9 | ||||||||
| Taxonomy | 13.0 | Genus | 17.608 | 7.1 | ||||||
| Species | 8.072 | 3.3 | ||||||||
| Family | 6.534 | 2.6 | ||||||||
| ||||||||||
| Source | n | Pearson’s Correlation Coefficient | p | |||||||
| Location | 229 | 0.426 | <0.001*** | |||||||
| Method for Tissue Removal | 234 | 0.379 | <0.001*** | |||||||
| Source Author | 179 | 0.276 | <0.001*** | |||||||
| Subregion | 246 | -0.260 | <0.001*** | |||||||
| Transmission of Algae | 211 | -0.220 | <0.001*** | |||||||
| Replication | 65 | -0.206 | <0.001*** | |||||||
| Sex | 228 | -0.196 | <0.001*** | |||||||
| Surface Area Determination | 203 | 0.185 | <0.001*** | |||||||
| Depth | 155 | -0.185 | <0.05* | |||||||
| Morphology | 243 | 0.167 | <0.01** | |||||||
| Reproductive Mode | 222 | -0.157 | <0.01** | |||||||
| Generalization) | 242 | 0.145 | <0.01** | |||||||
| Complexity of Skeleton | 243 | 0.131 | >0.05 | |||||||
| Ocean Basin | 246 | 0.114 | <0.05* | |||||||
| Species | 192 | -0.092 | >0.05 | |||||||
| Decade | 242 | -0.054 | >0.05 | |||||||
| Genus | 231 | 0.039 | >0.05 | |||||||
| Family | 239 | -0.031 | >0.05 | |||||||
| Sampling Time | 243 | 0.014 | >0.05 | |||||||
| Sample Size | 243 | -0.003 | >0.05 | |||||||
| |||||||||
| Healthy | ±SE | n | Bleached | ±SE | n | ||||
| 3.70 | 2.69 | 243 | 1.00 | 0.83 | 243 | ||||
| 1 ***Highly significant interaction. | |||||||||
| |||||||||
| Factor | Source | Healthy | ±SE | n | Bleached | ±SE | n | ||
| Family | Acroporidae | 1.89 | 2.12 | 55 | 0.50 | 0.60 | 55 | ||
| Agariciidae | 7.57 | 1.69 | 16 | 2.51 | 5.45 | 16 | |||
| Faviidae | 4.04 | 4.28 | 74 | 1.28 | 1.80 | 74 | |||
| Oculinidae | 11.24 | 16.76 | 7 | 1.63 | 4.91 | 7 | |||
| Pocilloporidae | 3.20 | 4.16 | 58 | 0.84 | 1.11 | 58 | |||
| Poritidae | 3.45 | 9.85 | 31 | 0.64 | 1.66 | 31 | |||
| Genus | Acropora | 1.49 | 1.00 | 45 | 0.39 | 0.48 | 45 | ||
| Agaricia | 1.30 | 4.25 | 8 | 0.52 | 1.93 | 8 | |||
| Cladocora | 3.32 | 3.68 | 6 | 0.95 | 2.46 | 6 | |||
| Coelastrea | 8.15 | 10.66 | 11 | 3.00 | 2.44 | 11 | |||
| Montastrea | 1.82 | 4.71 | 6 | 0.75 | 1.57 | 6 | |||
| Montipora | 3.73 | 9.02 | 10 | 1.02 | 1.85 | 10 | |||
| Oculina | 11.12 | 19.77 | 6 | 1.73 | 5.68 | 6 | |||
| Orbicella | 2.40 | 1.46 | 45 | 0.53 | 0.83 | 45 | |||
| Pavona | 12.92 | 8.31 | 6 | 5.00 | 0.45 | 6 | |||
| Pocillopora | 4.28 | 6.90 | 31 | 0.82 | 1.36 | 31 | |||
| Porites | 2.93 | 8.66 | 30 | 0.63 | 1.71 | 30 | |||
| Seriatopora | 2.95 | 3.81 | 13 | 1.31 | 3.39 | 13 | |||
| Stylophora | 1.03 | 1.61 | 14 | 0.43 | 0.69 | 14 | |||
| Species | Acropora millepora | 1.58 | 1.31 | 24 | 0.54 | 0.07 | 24 | ||
| Acropora muricata | 1.25 | 2.77 | 7 | 0.21 | 0.00 | 7 | |||
| Agaricia lamarcki | 1.48 | 6.95 | 5 | 0.60 | 3.18 | 5 | |||
| Cladocora caespita | 3.32 | 3.68 | 6 | 0.95 | 2.46 | 6 | |||
| Coelastrea aspera | 8.15 | 10.66 | 11 | 3.00 | 2.44 | 11 | |||
| Montastrea cavernosa | 1.82 | 4.71 | 6 | 0.75 | 1.57 | 6 | |||
| Oculina patagonica | 11.12 | 19.77 | 6 | 1.73 | 5.68 | 6 | |||
| Orbicella annularis | 2.30 | 1.63 | 27 | 0.54 | 1.15 | 27 | |||
| Orbicella faveolata | 3.08 | 4.01 | 10 | 0.45 | 1.51 | 10 | |||
| Orbicella franksi | 1.86 | 1.84 | 8 | 0.59 | 1.97 | 8 | |||
| Pocillopora damicornis | 3.34 | 4.24 | 26 | 0.57 | 0.98 | 26 | |||
| Porites cylindrica | 3.36 | 3.06 | 5 | 1.06 | 0.85 | 5 | |||
| Porites lobata | 3.26 | 1.35 | 19 | 0.64 | 2.60 | 19 | |||
| Porites lutea | 1.45 | 6.87 | 5 | 0.22 | 1.20 | 5 | |||
| Seriatopora hystrix | 2.95 | 3.81 | 13 | 1.31 | 3.39 | 13 | |||
| Stylophora pistillata | 1.03 | 1.61 | 14 | 0.43 | 0.69 | 14 | |||
| |||||||||
| Factor | Source | Healthy | ±SE | n | Bleached | ±SE | n | ||
| Decade | 1980’s | 1.45 | 3.11 | 14 | |||||
| 1990’s | 4.98 | 7.40 | 54 | 1.35 | 2.14 | 54 | |||
| 2000’s | 3.49 | 2.86 | 175 | 0.94 | 0.91 | 175 | |||
| Ocean Basin | Atlantic | 2.29 | 1.36 | 69 | 0.53 | 0.70 | 69 | ||
| Indian | 7.67 | 8.26 | 37 | 1.82 | 1.98 | 37 | |||
| Pacific | 3.35 | 3.67 | 137 | 1.02 | 1.23 | 137 | |||
| Subregion | East | 5.59 | 8.06 | 54 | 1.32 | 2.22 | 54 | ||
| North | 1.72 | 2.77 | 25 | 0.47 | 1.66 | 25 | |||
| South | 4.18 | 4.91 | 15 | 0.88 | 1.93 | 15 | |||
| West | 3.54 | 3.24 | 152 | 1.13 | 1.30 | 152 | |||
| Location | Bahamas | 2.61. | 2.16 | 27 | 0.39 | 1.16 | 27 | ||
| Costa Rica | 0.88 | 0.69 | 9 | 0.11 | 0.37 | 9 | |||
| Florida | 1.81 | 1.91 | 10 | 0.44 | 1.19 | 10 | |||
| Great Barrier Reef | 2.56 | 3.32 | 79 | 0.93 | 1.47 | 79 | |||
| Jamaica | 1.51 | 1.63 | 21 | 0.59 | 0.89 | 21 | |||
| Japan | 0.72 | 1.27 | 10 | 0.20 | 0.66 | 10 | |||
| Kenya | 3.36 | 3.06 | 5 | 1.06 | 0.85 | 5 | |||
| Mediterranean Sea | 7.22 | 1.52 | 12 | 1.34 | 3.18 | 12 | |||
| Mexico | 3.38 | 3.08 | 9 | 0.75 | 2.88 | 9 | |||
| Panama | 6.63 | 1.21 | 30 | 1.70 | 3.54 | 30 | |||
| Thailand | 10.06 | 1.13 | 17 | 2.65 | 2.30 | 17 | |||
| |||||||||
| Factor | Source | Healthy | ±SE | n | Bleached | ±SE | n | ||
| Depth | <3m | 2.90 | 2.39 | 36 | 1.04 | 1.75 | 36 | ||
| <6m | 5.51 | 9.76 | 31 | 0.85 | 1.51 | 31 | |||
| <9m | 6.37 | 10.53 | 40 | 1.77 | 3.54 | 40 | |||
| <12m | 1.91 | 3.61 | 5 | 0.38 | 0.58 | 5 | |||
| <15m | 1.76 | 2.02 | 19 | 0.21 | 0.76 | 19 | |||
| >15m | 1.82 | 2.37 | 24 | 0.78 | 1.25 | 24 | |||
| Transmission of Algae to Offspring | Both | 8.15 | 10.66 | 11 | 3.00 | 2.44 | 11 | ||
| No | 3.12 | 3.38 | 108 | 0.82 | 1.14 | 108 | |||
| Yes | 3.08 | 3.96 | 92 | 0.78 | 0.88 | 92 | |||
| Morphology | Branching | 2.54 | 2.34 | 119 | 0.68 | 0.620 | 119 | ||
| Encrusting | 6.24 | 15.90 | 13 | 1.20 | 3.30 | 13 | |||
| Massive | 4.65 | 4.74 | 111 | 1.32 | 1.57 | 111 | |||
| Generalization | Generalist | 3.10 | 2.85 | 150 | 0.90 | 1.05 | 150 | ||
| Specialist | 4.70 | 5.22 | 92 | 1.18 | 1.32 | 92 | |||
| Sex | Gonochoric | 5.65 | 7.46 | 65 | 1.57 | 2.35 | 65 | ||
| Hermaphroditic | 2.92 | 2.28 | 163 | 0.80 | 0.70 | 163 | |||
| Reproductive Mode | Botha | 5.13 | 6.50 | 31 | 1.51 | 2.31 | 31 | ||
| Brooder | 1.80 | 2.25 | 34 | 0.73 | 1.53 | 34 | |||
| Spawner | 3.54 | 3.43 | 157 | 0.96 | 1.09 | 157 | |||
| Complexity of Skeleton | Complex | 3.33 | 4.53 | 104 | 0.86 | 1.22 | 104 | ||
| Robust | 3.98 | 3.25 | 139 | 1.11 | 1.11 | 139 | |||
| |||||||||
| Factor | Source | Healthy | ±SE | n | Bleached | ±SE | n | ||
| Method for Tissue Removal from skeleton | Pick | 3.29 | 2.63 | 213 | 0.90 | 0.85 | 213 | ||
| Decalcify | 8.83 | 1.07 | 21 | 2.42 | 2.20 | 21 | |||
| Sample Size | Branch | 1.46 | 0.96 | 50 | 0.49 | 0.52 | 50 | ||
| Colony | 3.32 | 6.91 | 36 | 1.23 | 2.98 | 36 | |||
| Core | 8.96 | 1.31 | 18 | 2.06 | 3.14 | 18 | |||
| Fragment | 4.27 | 7.00 | 59 | 1.27 | 2.08 | 59 | |||
| Nubbin | 4.87 | 5.61 | 12 | 1.10 | 1.14 | 12 | |||
| Sample | 2.76 | 1.82 | 46 | 0.52 | 1.21 | 46 | |||
| Subsample | 4.94 | 9.89 | 22 | 1.15 | 2.00 | 22 | |||
| Sampling Time | <1 day | 2.15 | 2.27 | 38 | 0.63 | 0.97 | 38 | ||
| <1 week | 2.92 | 4.07 | 45 | 1.15 | 1.77 | 45 | |||
| <2 weeks | 1.54 | 2.32 | 12 | 0.57 | 1.25 | 12 | |||
| <1 month | 2.25 | 2.58 | 27 | 0.94 | 1.96 | 27 | |||
| <1.5 months | 2.95 | 6.22 | 17 | 0.77 | 2.49 | 17 | |||
| <3 months | 4.01 | 12.59 | 18 | 0.84 | 2.46 | 18 | |||
| <4 months | 7.06 | 26.02 | 8 | 2.76 | 1.11 | 8 | |||
| <6 months | 3.02 | 4.00 | 9 | 0.94 | 2.13 | 9 | |||
| <1 year | 4.93 | 9.06 | 11 | 0.69 | 1.09 | 11 | |||
| <2 years | 9.10 | 12.24 | 31 | 2.03 | 3.24 | 31 | |||
| >2 years | 2.43 | 2.00 | 27 | 0.24 | 0.64 | 27 | |||
| Surface Area Determination | Calipers | 4.30 | 5.51 | 25 | 1.00 | 2.39 | 25 | ||
| Foil | 2.38 | 2.93 | 89 | 0.61 | 0.83 | 89 | |||
| Image Analyzer | 13.91 | 12.11 | 11 | 3.14 | 6.81 | 11 | |||
| Wax | 4.05 | 5.35 | 78 | 1.15 | 1.57 | 78 | |||
| Replication | <6 | 7.42 | 9.60 | 36 | 2.01 | 2.94 | 36 | ||
| <8 | 13.25 | 15.66 | 8 | 1.87 | 2.80 | 8 | |||
| ≥8 | 3.24 | 7.70 | 31 | 1.25 | 3.23 | 31 | |||
| Source Author | Berkelmans | 1.39 | 0.99 | 18 | 0.36 | 0.39 | 18 | ||
| Brown | 10.06 | 11.28 | 17 | 2.65 | 2.30 | 17 | |||
| Centeno | 0.88 | 0.69 | 9 | 0.11 | 0.37 | 9 | |||
| D’Croz | 1.27 | 2.14 | 17 | 0.30 | 1.11 | 17 | |||
| Fitt | 1.96 | 1.83 | 37 | 0.25 | 0.47 | 37 | |||
| Flores-Ramirez | 3.38 | 3.08 | 9 | 0.75 | 2.88 | 9 | |||
| Hill | 4.59 | 2.50 | 7 | 1.19 | 1.60 | 7 | |||
| Hoegh-Guldberg | 1.09 | 2.07 | 12 | 0.36 | 0.66 | 12 | |||
| Hueerkamp | 13.65 | 8.92 | 13 | 3.54 | 4.30 | 13 | |||
| Jones | 4.78 | 1.16 | 19 | 2.04 | 5.13 | 19 | |||
| Mercurio | 0.98 | 0.43 | 6 | 0.21 | 0.02 | 6 | |||
| Rodolfo-Metalpa | 7.32 | 2.03 | 9 | 1.63 | 3.77 | 9 | |||
| Visram | 3.36 | 3.06 | 5 | 1.06 | 0.85 | 5 | |||
| Warner | 1.88 | 2.32 | 9 | 0.77 | 1.52 | 9 | |||
| A) Average values for factors that cause inter and intra-specific variation | ||||
| Source | Healthy | ±SE | n | |
| Species | Acropora millepora | 2.06 | 1.59 | 40 |
| Coelastrea aspera | 10.23 | 1.21 | 20 | |
| Orbicella annularis | 2.91 | 2.92 | 34 | |
| Pocillopora damicornis | 2.52 | 2.77 | 49 | |
| Stylophora pistillata | 1.92 | 4.12 | 36 | |
| Ocean | Atlantic | 2.91 | 2.51 | 40 |
| Indian | 6.93 | 1.01 | 35 | |
| Pacific | 2.11 | 1.63 | 104 | |
| Replication level | Branch | 1.81 | 1.58 | 63 |
| Colony | 2.75 | 3.09 | 32 | |
| Core | 9.54 | 1.59 | 17 | |
| Fragment | 2.48 | 6.94 | 16 | |
| Sample | 2.65 | 2.34 | 27 | |
| Location | Bahamas | 2.57 | 2.95 | 12 |
| Florida | 2.07 | 2.48 | 6 | |
| Great Barrier Reef | 2.11 | 1.70 | 61 | |
| Hawaii | 1.70 | 3.21 | 13 | |
| Jamaica | 3.49 | 6.25 | 14 | |
| Japan | 1.55 | 5.60 | 8 | |
| Mexico | 2.92 | 3.16 | 6 | |
| Pacific | 7.73 | 1.60 | 4 | |
| Panama | 2.74 | 6.55 | 16 | |
| Red Sea | 2.96 | 7.99 | 17 | |
| Thailand | 10.68 | 1.30 | 18 | |
| Method for tissue removal | Airbrush | 2.53 | 2.29 | 60 |
| Decalcification | 6.24 | 9.36 | 38 | |
| Waterpik | 2.52 | 2.54 | 72 | |
| Method for surface area | Calculated | 2.31 | 3.13 | 10 |
| Callipers | 5.19 | 1.00 | 29 | |
| Foil | 2.41 | 3.74 | 41 | |
| Image analyser | 5.11 | 2.09 | 7 | |
| Paraffin Wax | 2.11 | 1.99 | 64 | |
| Modified Paraffin Wax | 3.60 | 6.24 | 9 | |
| Time | < 1 day | 2.81 | 3.51 | 45 |
| < 1 week | 5.13 | 8.64 | 29 | |
| < 2 weeks | 4.30 | 1.82 | 4 | |
| < 1 month | 1.62 | 1.91 | 26 | |
| < 2 months | 1.63 | 1.93 | 18 | |
| < 3 months | 1.85 | 5.27 | 4 | |
| < 6 months | 3.98 | 7.33 | 11 | |
| < 9 months | 3.25 | 3.03 | 10 | |
| < 1 year | 2.84 | 9.54 | 11 | |
| < 2 years | 6.64 | 2.60 | 5 | |
| > 2 years | 5.13 | 1.62 | 16 | |
| Cause | Aerial exposure | 9.77 | 18.95 | 3 |
| Ammonium enrichment | 1.45 | 2.35 | 16 | |
| Depth | 3.53 | 5.45 | 28 | |
| High light and temperature | 4.98 | 7.91 | 18 | |
| Light | 2.75 | 7.26 | 19 | |
| One sample | 3.27 | 12.07 | 5 | |
| Pollutant | 1.38 | 8.87 | 6 | |
| Seasonal variation | 4.59 | 16.04 | 15 | |
| Temperature | 2.81 | 3.92 | 68 | |
| Subregion | East | 2.74 | 6.55 | 16 |
| North | 2.54 | 4.62 | 33 | |
| South | 1.90 | 2.43 | 24 | |
| West | 3.82 | 3.96 | 106 | |
| Depth | < 3m | 2.38 | 4.07 | 43 |
| < 6m | 2.38 | 2.99 | 43 | |
| > 6m | 3.18 | 3.81 | 25 | |
| Replicate counts | < 6 | 4.53 | 1.03 | 15 |
| > 6 | 2.95 | 6.55 | 25 | |
| B) Variation in healthy zooxanthellae population densities of Acropora millepora | ||||
| Source | Healthy | ±SE | n | |
| Replication level | Branch | 1.69 | 1.29 | 27 |
| Colony | 3.25 | 3.03 | 10 | |
| Population | 1.39 | 1.99 | 3 | |
| Cause | Depth | 1.62 | 1.51 | 15 |
| High temperature and light | 2.87 | 3.48 | 3 | |
| Pollutant | 1.49 | 0.78 | 3 | |
| Seasonal variation | 3.25 | 3.03 | 10 | |
| Temperature | 1.39 | 1.47 | 9 | |
| Method for surface area | Callipers | 2.49 | 1.74 | 3 |
| Foil | 1.47 | 2.33 | 3 | |
| Wax | 2.07 | 1.82 | 34 | |
| Method for tissue removal | Airbrush | 2.07 | 1.83 | 34 |
| Decalcification | 2.49 | 1.74 | 3 | |
| Waterpik | 1.49 | 0.78 | 3 | |
| C) Variation in healthy zooxanthellae population densities of Coelastrea aspera | ||||
| Source | Healthy | ±SE | n | |
| Replication level | Colony | 6.07 | 6.05 | 4 |
| Core | 11.63 | 16.80 | 13 | |
| Time | < 1 month | 8.85 | 11.34 | 14 |
| > 1 year | 13.46 | 27.60 | 6 | |
| Cause | Aerial Exposure | 9.77 | 18.85 | 3 |
| High light and temperature | 8.70 | 19.28 | 5 | |
| Temperature | 10.56 | 22.63 | 6 | |
| Replicate counts | 3 | 9.77 | 18.85 | 3 |
| 6 | 10.00 | 31.39 | 3 | |
| D) Variation in healthy zooxanthellae population densities of Orbicella annularis | ||||
| Source | Healthy | ±SE | n | |
| Method for surface area | Callipers | 5.51 | 9.20 | 6 |
| Foil | 2.31 | 1.96 | 21 | |
| Cause | Depth | 4.99 | 6.52 | 9 |
| High light and temperature | 2.16 | 3.62 | 4 | |
| Temperature | 2.08 | 1.45 | 20 | |
| Depth | < 3m | 2.27 | 4.91 | 3 |
| < 6m | 2.36 | 3.99 | 4 | |
| < 15m | 2.11 | 1.56 | 14 | |
| < 18m | 1.53 | 2.73 | 3 | |
| < 24m | 3.93 | 1.76 | 3 | |
| > 24m | 5.51 | 9.20 | 6 | |
| Time | < 1 week | 1.86 | 1.91 | 6 |
| < 1 month | 3.93 | 1.76 | 3 | |
| < 6 months | 4.72 | 8.63 | 8 | |
| < 1 year | 2.37 | 4.18 | 3 | |
| > 2years | 2.16 | 2.01 | 12 | |
| Replicate counts | < 8 | 3.93 | 1.76 | 3 |
| > 8 | 2.59 | 4.17 | 5 | |
| Subregion | North | 2.07 | 2.48 | 6 |
| West | 3.10 | 3.55 | 27 | |
| Location | Bahamas | 2.57 | 2.95 | 12 |
| Florida | 2.07 | 2.48 | 6 | |
| Jamaica | 3.49 | 6.25 | 14 | |
| Method for tissue removal | Airbrush | 2.16 | 3.62 | 4 |
| Decalcification | 3.93 | 1.76 | 3 | |
| Waterpik | 2.91 | 3.55 | 27 | |
| Replication level | Colony | 1.97 | 3.31 | 5 |
| Core | 2.74 | 4.76 | 4 | |
| Sample | 2.40 | 2.32 | 17 | |
| E) Variation in healthy zooxanthellae population densities of Pocillopora damicornis | ||||
| Source | Healthy | ±SE | n | |
| Method for surface area | Calipers | 1.64 | 3.83 | 11 |
| Wax | 2.56 | 5.06 | 21 | |
| Subregion | East | 2.74 | 6.55 | 16 |
| North | 1.54 | 2.27 | 3 | |
| South | 1.48 | 2.64 | 17 | |
| West | 4.59 | 2.50 | 7 | |
| Cause | Ammonium Enrichment | 1.41 | 2.29 | 10 |
| High light and temperature | 4.83 | 0.96 | 6 | |
| Light | 2.03 | 6.03 | 6 | |
| Seasonal Variation | 0.77 | 1.74 | 3 | |
| Temperature | 2.82 | 6.53 | 16 | |
| Method for tissue removal | Airbrush | 3.45 | 5.59 | 20 |
| Decalcification | 1.64 | 3.83 | 11 | |
| Waterpik | 1.77 | 2.27 | 9 | |
| Time | < 1 day | 2.75 | 5.50 | 10 |
| < 1 week | 3.97 | 6.79 | 5 | |
| < 2 months | 1.70 | 2.40 | 14 | |
| < 3 months | 1.85 | 5.27 | 4 | |
| < 1 year | 3.02 | 1.32 | 8 | |
| Depth | < 3m | 1.79 | 3.03 | 13 |
| < 6m | 2.16 | 1.73 | 5 | |
| < 9m | 2.77 | 8.17 | 13 | |
| Replication level | Branch | 2.57 | 3.26 | 24 |
| Colony | 2.93 | 8.05 | 13 | |
| F) Variation in healthy zooxanthellae population densities of Stylophora pistillata | ||||
| Source | Healthy | ±SE | n | |
| Ocean | Indian | 2.96 | 7.99 | 17 |
| Pacific | 0.98 | 1.20 | 19 | |
| Depth | < 3m | 1.10 | 1.30 | 15 |
| < 6m | 0.97 | 3.29 | 5 | |
| < 9m | 0.61 | 1.64 | 4 | |
| > 9m | 2.81 | 1.03 | 3 | |
| Location | Great Barrier Reef | 1.03 | 1.61 | 14 |
| Japan | 0.85 | 0.71 | 5 | |
| Red Sea | 2.96 | 7.99 | 17 | |
| Subregion | North | 2.48 | 6.43 | 22 |
| West | 1.03 | 1.61 | 14 | |
| Time | < 1 day | 2.95 | 8.57 | 16 |
| < 1 week | 0.91 | 1.32 | 14 | |
| < 2 weeks | 1.50 | 2.46 | 6 | |
| Cause | Ammonium enrichment | 1.04 | 3.04 | 5 |
| Light | 3.08 | 1.03 | 13 | |
| Pollutant | 1.27 | 1.45 | 3 | |
| Temperature | 0.96 | 1.99 | 11 | |
| Method for surface area | Calculated | 1.41 | 1.71 | 4 |
| Foil | 2.48 | 9.49 | 15 | |
| Image Analyser | 2.70 | 7.13 | 4 | |
| Wax | 1.21 | 1.89 | 9 | |
| |||||||||||||
| Factor | Source | (S-M)/S | Susceptibility | Mortality | |||||||||
| Average | ±SE | n | Average | ±SE | n | ||||||||
| Family | Acroporidae | 0.50 | 54.7 | 1.89 | 367 | 27.2 | 2.27 | 290 | |||||
| Agariciidae | 0.69 | 51.7 | 3.31 | 137 | 16.2 | 2.93 | 112 | ||||||
| Astrocoeniidae | 0.53 | 36.1 | 9.44 | 13 | 17.0 | 9.39 | 11 | ||||||
| Dendrophyllidae | 41.1 | 13.96 | 11 | ||||||||||
| Diploastraeidae | 0.58 | 35.7 | 7.68 | 27 | 15.0 | 7.90 | 20 | ||||||
| Euphyllidae | 0.65 | 42.8 | 5.04 | 46 | 15.1 | 5.24 | 36 | ||||||
| Fungiidae | 0.77 | 40.0 | 3.82 | 77 | 9.2 | 3.08 | 66 | ||||||
| Lobophyllidae | 0.97 | 36.1 | 4.51 | 45 | 1.16 | 0.54 | 38 | ||||||
| Meandrinidae | 0.86 | 30.7 | 4.96 | 35 | 4.4 | 2.18 | 30 | ||||||
| Merulinidae | 0.80 | 47.8 | 1.87 | 333 | 9.7 | 1.28 | 268 | ||||||
| Milleporidae | 0.33 | 56.4 | 4.96 | 60 | 37.7 | 6.53 | 48 | ||||||
| Montastraeidae | 0.94 | 42.0 | 6.32 | 27 | 2.7 | 1.52 | 21 | ||||||
| Mussidae | 0.78 | 45.4 | 2.83 | 144 | 10.2 | 2.12 | 120 | ||||||
| Pocilloporidae | 0.57 | 47.3 | 2.45 | 229 | 20.4 | 2.80 | 164 | ||||||
| Poritidae | 0.64 | 41.4 | 2.15 | 273 | 14.9 | 2.05 | 214 | ||||||
| Psammocoridae | 0.84 | 54.7 | 8.58 | 21 | 8.6 | 5.66 | 15 | ||||||
| Siderastreidae | 0.93 | 57.7 | 4.02 | 58 | 4.0 | 1.73 | 48 | ||||||
| Genus | Acanthastrea | 0.97 | 44.7 | 7.08 | 16 | 1.4 | 1.00 | 12 | |||||
| Acropora | 0.44 | 60.1 | 2.45 | 230 | 33.8 | 3.06 | 188 | ||||||
| Agaricia | 0.80 | 60.4 | 4.42 | 60 | 11.9 | 3.51 | 49 | ||||||
| Astreopora | 0.94 | 38.7 | 5.08 | 17 | 2.5 | 1.83 | 13 | ||||||
| Colpopyllia | 0.78 | 52.1 | 6.41 | 21 | 11.3 | 4.31 | 16 | ||||||
| Cyphastrea | 0.41 | 28.1 | 8.92 | 17 | 16.6 | 8.61 | 13 | ||||||
| Diploastrea | 0.58 | 35.7 | 7.68 | 27 | 15.0 | 7.90 | 20 | ||||||
| Diploria | 0.67 | 56.4 | 8.67 | 20 | 18.7 | 7.45 | 15 | ||||||
| Echinophyllia | 1 | 20.6 | 7.20 | 10 | 0 | 0 | 10 | ||||||
| Echinopora | 0.61 | 35.3 | 7.07 | 25 | 13.7 | 5.96 | 21 | ||||||
| Favia | 0.90 | 47.1 | 5.04 | 37 | 4.7 | 3.28 | 30 | ||||||
| Favites | 0.75 | 51.5 | 5.79 | 33 | 12.7 | 5.00 | 23 | ||||||
| Fungia | 0.83 | 56.5 | 5.73 | 31 | 9.5 | 5.23 | 23 | ||||||
| Galaxea | 0.74 | 36.2 | 5.71 | 29 | 9.5 | 4.58 | 24 | ||||||
| Goniastrea | 0.84 | 47.8 | 7.08 | 27 | 7.7 | 4.38 | 24 | ||||||
| Goniopora | 0.87 | 38.1 | 8.03 | 25 | 5.0 | 4.66 | 21 | ||||||
| Hydnophora | 0.68 | 40.7 | 6.68 | 24 | 13.0 | 5.19 | 21 | ||||||
| Leptoria | 27.5 | 9.64 | 11 | ||||||||||
| Lobophyllia | 43.0 | 11.06 | 11 | ||||||||||
| Madracis | 0.39 | 10.9 | 3.40 | 21 | 6.6 | 3.90 | 17 | ||||||
| Meandrina | 0.92 | 36.0 | 7.82 | 13 | 2.7 | 2.24 | 12 | ||||||
| Millepora | 0.35 | 55.6 | 5.00 | 59 | 36.4 | 6.54 | 47 | ||||||
| Montastrea | 0.94 | 44.4 | 4.89 | 43 | 2.8 | 1.17 | 31 | ||||||
| Montipora | 0.65 | 44.9 | 3.79 | 91 | 15.8 | 3.64 | 64 | ||||||
| Mycetophyllia | 0.50 | 32.6 | 12.32 | 11 | 16.4 | 11.06 | 11 | ||||||
| Orbicella | 0.84 | 65.6 | 3.85 | 50 | 10.4 | 2.11 | 44 | ||||||
| Pavona | 0.52 | 43.4 | 5.59 | 51 | 20.9 | 5.85 | 40 | ||||||
| Platygyra | 0.75 | 51.3 | 5.07 | 45 | 13.0 | 4.33 | 35 | ||||||
| Pocillopora | 0.54 | 47.2 | 2.82 | 162 | 21.8 | 3.41 | 114 | ||||||
| Porites | 0.60 | 42.4 | 2.32 | 235 | 16.8 | 2.31 | 183 | ||||||
| Psammocora | 0.85 | 55.7 | 8.58 | 21 | 8.6 | 5.66 | 15 | ||||||
| Pseudodiploria | 0.88 | 48.9 | 4.80 | 34 | 5.9 | 2.19 | 30 | ||||||
| Seriatopora | 75.1 | 9.39 | 12 | ||||||||||
| Siderastrea | 0.92 | 56.1 | 4.15 | 53 | 4.7 | 1.93 | 44 | ||||||
| Stephanocoenia | 0.54 | 35.0 | 10.19 | 12 | 16.17 | 10.34 | 10 | ||||||
| Stylophora | 0.73 | 61.4 | 8.18 | 23 | 16.4 | 9.67 | 14 | ||||||
| Symphyllia | 0.95 | 37.9 | 9.50 | 15 | 2.0 | 1.37 | 12 | ||||||
| Turbinaria | 41.1 | 13.96 | 11 | ||||||||||
| Species | Acropora cervicornis | 0.77 | 36.6 | 7.90 | 21 | 8.3 | 5.62 | 16 | |||||
| Acropora hyacinthus | 0.25 | 68.1 | 13.78 | 10 | 51.3 | 14.22 | 10 | ||||||
| Acropora palmata | 0.78 | 39.7 | 8.49 | 18 | 8.6 | 4.64 | 14 | ||||||
| Agaricia agaricites | 0.80 | 68.9 | 6.62 | 22 | 13.8 | 6.24 | 19 | ||||||
| Agaricia tenuifolia | 0.58 | 37.9 | 8.74 | 12 | 16.0 | 9.51 | 11 | ||||||
| Colpophyllia natans | 0.73 | 48.5 | 7.32 | 17 | 12.9 | 4.78 | 14 | ||||||
| Diploria labyrinthiformis | 0.64 | 51.5 | 8.91 | 18 | 18.7 | 7.45 | 15 | ||||||
| Favia fragum | 0.98 | 46.9 | 7.77 | 13 | 0.9 | 0.63 | 12 | ||||||
| Meandrina meandrites | 0.91 | 34.8 | 8,40 | 12 | 3.0 | 2.44 | 11 | ||||||
| Montastrea cavernosa | 0.94 | 42.0 | 6.32 | 27 | 2.7 | 1.52 | 21 | ||||||
| Montipora capitata | 31.4 | 11.14 | 13 | ||||||||||
| Orbicella annularis | 0.87 | 66.9 | 4.36 | 35 | 8.9 | 2.16 | 32 | ||||||
| Orbicella faveolata | 70.2 | 7.29 | 12 | ||||||||||
| Pavona varians | 52.0 | 11.32 | 11 | ||||||||||
| Platygyra daedalea | 0.69 | 69.9 | 6.54 | 14 | 21.4 | 7.69 | 14 | ||||||
| Pocillopora damicornis | 0.38 | 48.3 | 6.57 | 36 | 30.0 | 8.28 | 25 | ||||||
| Pocillopora elegans | 62.5 | 12.26 | 10 | ||||||||||
| Pocillopora meandrina | 19.8 | 5.40 | 21 | ||||||||||
| Pocillopora verrucosa | 0.56 | 71.2 | 9.55 | 10 | 31.2 | 13.25 | 10 | ||||||
| Porites astreoides | 0.95 | 45.1 | 6.19 | 30 | 2.2 | 0.78 | 26 | ||||||
| Porites compressa | 2.5 | 1.29 | 10 | ||||||||||
| Porites lobata | 0.79 | 23.3 | 6.32 | 26 | 4.9 | 3.19 | 24 | ||||||
| Porites porites | 0.77 | 56.8 | 6.92 | 24 | 13.3 | 4.15 | 21 | ||||||
| Pseudodiploria strigosa | 0.91 | 48.9 | 6.04 | 25 | 4.4 | 1.72 | 22 | ||||||
| Siderastrea siderea | 0.93 | 60.7 | 4.98 | 34 | 4.4 | 2.47 | 31 | ||||||
| |||||||||||||
| Factor | Source | (S-M)/S | Susceptibility | Mortality | |||||||||
| Average | ±SE | n | Average | ±SE | n | ||||||||
| Ocean Basin | Indian | 0.82 | 47.9 | 1.46 | 556 | 8.4 | 0.88 | 456 | |||||
| Pacific | 0.84 | 53.4 | 1.46 | 556 | 8.4 | 0.88 | 456 | ||||||
| Atlantic | 0.60 | 44.2 | 1.22 | 872 | 17.8 | 1.34 | 634 | ||||||
| Subregion | Central | 43.6 | 11.47 | 10 | |||||||||
| East | 0.62 | 43.0 | 1.72 | 450 | 16.4 | 1.80 | 309 | ||||||
| Northeast | 0.49 | 63.7 | 7.59 | 24 | 32.6 | 9.67 | 18 | ||||||
| North | 0.55 | 70.6 | 4.64 | 36 | 31.9 | 9.88 | 15 | ||||||
| Northwest | 0.16 | 67.3 | 3.76 | 108 | 56.3 | 4.39 | 94 | ||||||
| Southeast | 1 | 15.3 | 2.61 | 101 | 0.0 | 0.00 | 58 | ||||||
| South | 0.95 | 45.7 | 1.89 | 286 | 2.4 | 0.77 | 258 | ||||||
| Southwest | 0.94 | 58.8 | 3.56 | 59 | 3.5 | 0.68 | 50 | ||||||
| West | 0.64 | 50.4 | 1.12 | 970 | 18.2 | 1.09 | 825 | ||||||
| Country | Andaman Sea | 0 | 90.9 | 2.48 | 53 | 90.9 | 2.48 | 53 | |||||
| Barbados | 61.7 | 6.44 | 28 | ||||||||||
| Belize | 0.89 | 35.8 | 2.75 | 99 | 4.0 | 1.57 | 82 | ||||||
| Brazil | 0.93 | 57.8 | 3.70 | 55 | 3.8 | 0.72 | 46 | ||||||
| Costa Rica | 0.78 | 52.7 | 4.21 | 60 | 11.8 | 3.56 | 50 | ||||||
| Curacao | 0.98 | 36.9 | 8.82 | 15 | 0.7 | 0.73 | 15 | ||||||
| Dominica | 0.80 | 46.5 | 4.20 | 44 | 9.4 | 2.69 | 44 | ||||||
| Florida Keys | 79.4 | 5.62 | 17 | ||||||||||
| French Polynesia | 0.96 | 48.9 | 2.84 | 138 | 1.9 | 1.08 | 111 | ||||||
| Great Barrier Reef | 0.59 | 49.7 | 2.65 | 144 | 20.5 | 2.90 | 116 | ||||||
| Grand Cayman | 0.94 | 83.2 | 11.22 | 12 | 4.9 | 2.60 | 12 | ||||||
| Hawaii | 1 | 15.8 | 2.63 | 103 | 0.0 | 0.00 | 58 | ||||||
| Indonesia | 0.73 | 46.1 | 2.78 | 172 | 12.5 | 2.09 | 172 | ||||||
| Jamaica | 0.96 | 27.8 | 4.07 | 67 | 1.2 | 0.77 | 63 | ||||||
| Japan | 0.13 | 76.8 | 5.13 | 49 | 66.5 | 6.07 | 45 | ||||||
| Kenya | 0.56 | 57.8 | 3.05 | 93 | 25.7 | 3.31 | 91 | ||||||
| Maldives | 72.9 | 4.05 | 14 | ||||||||||
| Martinique | 0.52 | 49.7 | 8.13 | 15 | 24.0 | 6.84 | 15 | ||||||
| Mauritius | 0.79 | 14.9 | 2.30 | 71 | 3.2 | 0.82 | 71 | ||||||
| Meso-American Reef | 0.60 | 38.4 | 7.73 | 17 | 15.5 | 2.62 | 15 | ||||||
| Palau | 0.52 | 31.8 | 2.44 | 133 | 15.2 | 7.16 | 23 | ||||||
| Panama | 0.48 | 54.2 | 7.83 | 32 | 28.0 | 7.99 | 30 | ||||||
| Persian Gulf | 71.4 | 5.39 | 43 | ||||||||||
| Papua New Guinea | 0.63 | 51.4 | 7.66 | 16 | 18.8 | 7.72 | 11 | ||||||
| Puerto Rico | 63.6 | 11.08 | 14 | ||||||||||
| Rodrigues | 0.97 | 43.7 | 7.10 | 15 | 1.3 | 0.91 | 15 | ||||||
| Seychelles | 0.32 | 52.7 | 7.09 | 24 | 35.6 | 7.73 | 21 | ||||||
| Singapore | 0.69 | 20.1 | 3.51 | 21 | 6.3 | 2.59 | 21 | ||||||
| South Africa | 0.96 | 42.2 | 2.49 | 147 | 1.6 | 0.63 | 147 | ||||||
| Thailand | 0.70 | 90.9 | 3.04 | 35 | 27.0 | 6.66 | 32 | ||||||
| Tioman Island | 0.65 | 18.3 | 4.17 | 19 | 6.4 | 3.07 | 19 | ||||||
| Tobago | 1 | 53.4 | 7.72 | 16 | 0.0 | 0.00 | 16 | ||||||
| United Arabian Emirates | 0.37 | 29.4 | 11.04 | 14 | 18.4 | 12.03 | 11 | ||||||
| US Virgin Islands | 0.69 | 48.1 | 3.19 | 120 | 15.0 | 2.37 | 115 | ||||||
| Location | Eastern Indian | 0.60 | 56.1 | 3.53 | 118 | 22.7 | 3.58 | 78 | |||||
| Eastern Pacific | 0.50 | 56.6 | 3.49 | 126 | 28.4 | 3.85 | 112 | ||||||
| Northern Atlantic | 0.77 | 58.9 | 5.90 | 34 | 13.7 | 2.62 | 17 | ||||||
| Northern Indian | 0.38 | 61.3 | 4.54 | 62 | 38.1 | 6.44 | 43 | ||||||
| Northern Pacific | 0.45 | 44.1 | 1.94 | 347 | 24.3 | 2.59 | 223 | ||||||
| Southern Atlantic | 0.94 | 58.8 | 3.56 | 59 | 3.5 | 0.68 | 50 | ||||||
| Southern Indian | 0.96 | 42.3 | 2.48 | 148 | 1.6 | 0.63 | 147 | ||||||
| Southern Pacific | 0.95 | 35.3 | 2.16 | 254 | 1.6 | 0.82 | 182 | ||||||
| Western Atlantic | 0.81 | 45.7 | 1.62 | 463 | 8.8 | 1.02 | 389 | ||||||
| Western Indian | 0.46 | 56.2 | 2.19 | 288 | 30.6 | 2.36 | 277 | ||||||
| Western Pacific | 29.2 | 49.7 | 2.65 | 144 | 20.5 | 2.90 | 116 | ||||||
| Continental Shelf Position | Inner | 0.73 | 61.6 | 3.66 | 70 | 16.5 | 3.69 | 61 | |||||
| Middle | 0.41 | 68.1 | 6.95 | 26 | 40.5 | 7.74 | 24 | ||||||
| Outer | 0.86 | 41.0 | 3.22 | 99 | 5.6 | 2.72 | 65 | ||||||
| |||||||||||||
| Factor | Source | (S-M)/S | Susceptibility | Mortality | |||||||||
| Average | ±SE | n | Average | ±SE | n | ||||||||
| Complexity of Skeleton | Complex | 0.60 | 49.2 | 1.22 | 902 | 19.5 | 1.27 | 715 | |||||
| Robust | 0.75 | 45.0 | 1.14 | 940 | 11.4 | 0.94 | 745 | ||||||
| Level of Colony Integration | High | 0.45 | 52.0 | 1.72 | 518 | 28.8 | 1.91 | 425 | |||||
| Medium | 0.79 | 46.9 | 1.57 | 491 | 10.0 | 1.03 | 430 | ||||||
| Low | 0.78 | 37.2 | 3.20 | 103 | 8.2 | 2.28 | 88 | ||||||
| Transmission of Algae to Offspring | Both | 0.75 | 48.7 | 9.87 | 20 | 12.1 | 6.77 | 20 | |||||
| No | 0.67 | 54.2 | 1.77 | 421 | 18.1 | 1.60 | 374 | ||||||
| Yes | 0.67 | 44.6 | 2.25 | 260 | 14.5 | 2.05 | 216 | ||||||
| Morphology | Branching | 0.44 | 50.2 | 1.84 | 443 | 28.0 | 2.05 | 359 | |||||
| Columnar | 0.63 | 55.9 | 9.33 | 22 | 20.6 | 8.24 | 20 | ||||||
| Encrusting | 0.80 | 36.3 | 3.60 | 80 | 7.2 | 2.32 | 66 | ||||||
| Free-living | 0.72 | 40.3 | 7.07 | 23 | 11.2 | 5.98 | 22 | ||||||
| Massive | 0.79 | 47.3 | 1.66 | 434 | 10.1 | 1.09 | 378 | ||||||
| Submassive | 0.79 | 43.8 | 4.88 | 57 | 9.3 | 3.12 | 52 | ||||||
| Tabular | 0.40 | 65.1 | 5.38 | 53 | 39.2 | 6.32 | 46 | ||||||
| Generalization | Generalist | 0.71 | 47.6 | 1.45 | 629 | 13.7 | 1.17 | 531 | |||||
| Specialist | 0.49 | 50.6 | 2.40 | 286 | 25.9 | 2.50 | 250 | ||||||
| Sex | Gonochoric | 0.68 | 49.4 | 335 | 2.00 | 15.6 | 1.78 | 294 | |||||
| Hermaphroditic | 0.63 | 51.6 | 471 | 1.71 | 19.3 | 1.62 | 407 | ||||||
| Reproductive Mode | Both | 0.75 | 44.3 | 9.46 | 22 | 11.0 | 6.19 | 22 | |||||
| Brooder | 0.71 | 49.3 | 2.45 | 211 | 14.3 | 2.15 | 175 | ||||||
| Spawner | 0.63 | 51.7 | 1.53 | 589 | 19.2 | 1.44 | 518 | ||||||
| |||||||||||||
| Factor | Source | (S-M)/S | Susceptibility | Mortality | |||||||||
| Average | ±SE | n | Average | ±SE | n | ||||||||
| Anthropogenic Input | No | 0.87 | 44.1 | 2.20 | 200 | 5.8 | 1.13 | 195 | |||||
| Yes, but protected | 0.90 | 38.5 | 4.30 | 60 | 4.0 | 1.86 | 58 | ||||||
| Yes | 0.39 | 60.8 | 6.46 | 27 | 37.0 | 7.06 | 27 | ||||||
| Anthropogenic Pressure | No | 0.54 | 53.7 | 7.07 | 22 | 24.5 | 5.91 | 22 | |||||
| Yes | 0.87 | 44.1 | 2.20 | 200 | 5.8 | 1.13 | 195 | ||||||
| Dominant Topography | Acropora | 0.67 | 36.5 | 6.32 | 37 | 11.9 | 4.67 | 36 | |||||
| Acropora and Montipora | 0.32 | 80.3 | 4.62 | 30 | 54.5 | 7.26 | 30 | ||||||
| Acropora cervicornis | 0.58 | 74.7 | 7.34 | 19 | 31.3 | 6.70 | 19 | ||||||
| Acropora hyacinthus | 1 | 50.2 | 7.92 | 16 | 0.0 | 0.00 | 16 | ||||||
| Montastrea | 0.58 | 57.4 | 7.36 | 19 | 24.1 | 6.00 | 19 | ||||||
| Pavona and Pocillopora | 0.83 | 31.3 | 3.96 | 31 | 5.2 | 2.57 | 31 | ||||||
| Pocillopora | 0.56 | 58.5 | 7.77 | 22 | 25.8 | 9.44 | 19 | ||||||
| Porites | 0.65 | 60.5 | 7.47 | 17 | 21.2 | 7.71 | 17 | ||||||
| Porites and others | 0.38 | 49.4 | 5.98 | 33 | 30.7 | 5.66 | 31 | ||||||
| Depth | <3 m | 0.79 | 27.2 | 4.07 | 57 | 5.6 | 2.30 | 53 | |||||
| <6 m | 0.42 | 60.3 | 3.50 | 131 | 34.9 | 3.84 | 129 | ||||||
| <9 m | 0.95 | 43.4 | 2.44 | 165 | 2.4 | 0.76 | 165 | ||||||
| <12 m | 0.71 | 49.9 | 1.96 | 283 | 14.6 | 1.63 | 260 | ||||||
| <15 m | 0.75 | 59.8 | 5.13 | 43 | 15.1 | 4.60 | 38 | ||||||
| <18 m | 0.80 | 46.5 | 4.20 | 44 | 9.4 | 2.69 | 44 | ||||||
| >18 m | 0.72 | 38.7 | 3.31 | 100 | 10.9 | 2.33 | 961 | ||||||
| Habitat | Bay | 0.40 | 35.0 | 4.06 | 64 | 21.0 | 6.03 | 35 | |||||
| Lagoon | 0.34 | 56.3 | 7.30 | 23 | 36.9 | 7.59 | 21 | ||||||
| Reef Crest | 0.93 | 24.3 | 3.96 | 60 | 1.6 | 1.07 | 57 | ||||||
| Reef Flat | 0.09 | 75.6 | 6.12 | 36 | 68.6 | 7.34 | 34 | ||||||
| Reef Slope | 0.86 | 42.8 | 3.15 | 121 | 5.8 | 1.83 | 103 | ||||||
| Polluted | No | 0.92 | 45.0 | 2.69 | 144 | 3.7 | 1.11 | 142 | |||||
| Yes | 0.7 | 43.0 | 3.78 | 82 | 12.9 | 2.95 | 82 | ||||||
| Reef Type | Barrier Reef | 0.59 | 49.7 | 2.65 | 144 | 20.5 | 2.90 | 116 | |||||
| Patch Reef | 0.61 | 45.7 | 1.58 | 456 | 17.8 | 1.59 | 370 | ||||||
| Subtropical | 0.96 | 41.9 | 2.66 | 133 | 1.8 | 0.69 | 133 | ||||||
| Supratropical | 1 | 45.2 | 6.98 | 14 | 0.0 | 0.00 | 14 | ||||||
| |||||||||||||
| Factor | Source | (S-M)/S | Susceptibility | Mortality | |||||||||
| Average | ±SE | n | Average | ±SE | n | ||||||||
| Timing of Observations | 0-3 months | 0.83 | 42.2 | 1.03 | 1068 | 7.0 | 0.67 | 825 | |||||
| 4-7 months | 0.74 | 47.1 | 1.90 | 346 | 12.2 | 1.42 | 294 | ||||||
| >7 months | 0.40 | 54.9 | 1.73 | 484 | 32.8 | 1.90 | 465 | ||||||
| Month of observations relative to the onset of thermal anomalies | <1 month | 0.83 | 53.9 | 2.90 | 128 | 9.1 | 2.11 | 114 | |||||
| 1-2 months | 0.86 | 38.2 | 1.26 | 655 | 5.4 | 0.73 | 504 | ||||||
| 2-3 months | 0.83 | 53.0 | 2.92 | 145 | 9.0 | 2.27 | 111 | ||||||
| 3-4 months | 0.73 | 39.2 | 3.04 | 140 | 10.4 | 2.20 | 96 | ||||||
| 4-5 months | 0.41 | 45.8 | 3.72 | 81 | 27.2 | 4.75 | 55 | ||||||
| 5-6 months | 0.86 | 39.5 | 3.57 | 97 | 5.4 | 2.03 | 97 | ||||||
| 6-7 months | 0.77 | 47.4 | 3.38 | 118 | 11.1 | 1.98 | 92 | ||||||
| 8-9 months | 0.38 | 44.5 | 5.06 | 73 | 27.8 | 4.66 | 71 | ||||||
| 9-10 months | 0.85 | 31.9 | 2.90 | 68 | 4.7 | 1.77 | 68 | ||||||
| 10-11 months | 0.36 | 52.2 | 8.12 | 22 | 33.3 | 6.62 | 22 | ||||||
| >11 months | 0.35 | 62.1 | 2.09 | 313 | 40.2 | 2.49 | 296 | ||||||
| Decade | 1980’s | 0.40 | 67.1 | 3.60 | 121 | 40.2 | 4.78 | 88 | |||||
| 1990’s | 0.66 | 51.9 | 1.13 | 910 | 17.5 | 1.12 | 776 | ||||||
| 2000’s | 0.81 | 38.2 | 1.38 | 584 | 7.3 | 0.87 | 476 | ||||||
| 2010’s | 0.53 | 47.6 | 1.84 | 429 | 22.5 | 2.16 | 295 | ||||||
| Recovery time (months) | <3 months | 0.74 | 43.1 | 5.41 | 55 | 11.3 | 3.63 | 55 | |||||
| 3-12 months | 0.85 | 47.2 | 3.39 | 74 | 7.0 | 2.32 | 70 | ||||||
| 12-24 months | 0.19 | 87.9 | 2.73 | 22 | 71.0 | 5.70 | 22 | ||||||
| Bleaching Occurrence | First bleaching | 0.65 | 54.9 | 1.07 | 932 | 19.0 | 1.14 | 757 | |||||
| Second | 0.67 | 35.8 | 1.21 | 655 | 11.7 | 1.06 | 470 | ||||||
| Third | 0.81 | 41.1 | 2.93 | 165 | 7.9 | 1.75 | 139 | ||||||
| Fourth | 0.56 | 59.9 | 2.14 | 234 | 26.3 | 2.5 | 220 | ||||||
| Fifth | 0.81 | 46.0 | 4.01 | 58 | 8.8 | 2.96 | 49 | ||||||
| |||||||||||||
| Factor | Source | (S-M)/S | Susceptibility | Mortality | |||||||||
| Average | ±SE | n | Average | ±SE | n | ||||||||
| Thermal Stress Accumulation | 0-30 DHW | 0.75 | 42.2 | 0.84 | 1601 | 10.7 | 0.65 | 1315 | |||||
| >30 DHW | 0.52 | 79.5 | 2.53 | 158 | 38.1 | 3.55 | 147 | ||||||
| Degree Heating Week Index (DHW) | 0-10 | 0.91 | 43.6 | 3.12 | 126 | 4.0 | 1.70 | 96 | |||||
| 11-20 | 0.78 | 40.0 | 1.00 | 1074 | 8.6 | 0.68 | 931 | ||||||
| 21-30 | 0.58 | 47.7 | 1.73 | 401 | 19.8 | 1.82 | 288 | ||||||
| 31-40 | 0.74 | 81.3 | 3.22 | 59 | 20.9 | 4.64 | 50 | ||||||
| 41-50 | 0.50 | 71.0 | 4.86 | 62 | 35.4 | 5.36 | 60 | ||||||
| >50 | 0.28 | 91.1 | 4.28 | 37 | 65.7 | 7.37 | 37 | ||||||
| Annual Temperature Range | ≤3 | 0.92 | 56.8 | 3.93 | 54 | 4.4 | 2.10 | 47 | |||||
| 3.1-4.0 | 0.77 | 25.2 | 2.20 | 106 | 5.9 | 5.88 | 17 | ||||||
| 4.1-5.0 | 0.79 | 43.1 | 2.75 | 142 | 9.4 | 1.56 | 142 | ||||||
| 5.1-6.0 | 0.72 | 48.7 | 2.63 | 166 | 13.4 | 2.52 | 166 | ||||||
| >6.0 | 0.40 | 83.6 | 10.15 | 11 | 49.9 | 11.81 | 11 | ||||||
| Peak Temperature (above mean monthly, oC) | <1 | 0.93 | 28.3 | 4.92 | 45 | 2.1 | 1.19 | 45 | |||||
| 1-1.9 | 0.66 | 46.7 | 3.44 | 75 | 15.7 | 3.00 | 67 | ||||||
| 2-2.9 | 0.53 | 54.3 | 2.23 | 274 | 25.5 | 2.18 | 248 | ||||||
| 3-3.9 | 0.91 | 49.6 | 2.04 | 209 | 4.4 | 0.92 | 209 | ||||||
| 4-5.9 | 0.50 | 59.0 | 7.40 | 22 | 29.6 | 9.82 | 20 | ||||||
| >6 | 0.50 | 73.9 | 4.77 | 54 | 36.6 | 10.38 | 15 | ||||||
| Relief or exacerbation of physical stress | No relief | 0.68 | 73.1 | 3.66 | 79 | 23.6 | 4.16 | 76 | |||||
| No relief and Doldrums | 0.39 | 46.8 | 3.35 | 142 | 28.5 | 3.36 | 137 | ||||||
| Relief | 0.80 | 45.4 | 1.85 | 335 | 9.3 | 1.31 | 319 | ||||||
| Relief and Doldrums | 0.84 | 52.8 | 3.29 | 68 | 8.7 | 2.18 | 68 | ||||||
| Cloud Cover/Storm | No | 0.55 | 53.9 | 2.45 | 247 | 24.1 | 2.37 | 239 | |||||
| Yes | 0.76 | 44.9 | 2.56 | 179 | 10.8 | 1.89 | 163 | ||||||
| Upwelling | No | 0.53 | 48.3 | 3.21 | 144 | 23.0 | 2.97 | 142 | |||||
| Yes | 0.83 | 48.1 | 2.11 | 224 | 8.1 | 1.43 | 224 | ||||||
| Doldrums | No | 0.52 | 68.3 | 7.51 | 27 | 32.8 | 7.69 | 24 | |||||
| Yes | 0.59 | 47.5 | 2.26 | 256 | 19.3 | 2.11 | 251 | ||||||
| Leeward/Windward | Leeward | 0.70 | 42.1 | 2.59 | 112 | 12.8 | 2.09 | 112 | |||||
| Windward | 0.61 | 63.1 | 7.26 | 13 | 24.4 | 8.18 | 10 | ||||||
| |||||||
| Source | SS Type III | df | n | MS | F | p | |
| Thermal Stress Accumulation* | 267.066 | 1 | 1758 | 0.138 | 172.769 | <0.001*** | |
| Degree Heating Index** | 267.466 | 5 | 1758 | 0.137 | 39.994 | <0.001*** | |
| Bleaching Occurrence* | 324.008 | 4 | 2043 | 0.148 | 38.448 | <0.001*** | |
| Decade** | 324.008 | 3 | 2043 | 0.151 | 33.735 | <0.001*** | |
| Subregion** | 324.008 | 8 | 2043 | 0.146 | 23.095 | <0.001*** | |
| Timing of Observations* | 295.925 | 2 | 1897 | 0.153 | 21.189 | <0.001*** | |
| Country** | 296.466 | 31 | 1914 | 0.118 | 20.182 | <0.001*** | |
| Relief from Physical Stress* | 105.637 | 3 | 669 | 0.148 | 15.520 | <0.001*** | |
| Annual Temperature Range** | 65.407 | 4 | 478 | 0.122 | 15.307 | <0.001*** | |
| Habitat | 53.116 | 4 | 303 | 0.148 | 15.098 | <0.001*** | |
| Recovery | 22.650 | 2 | 150 | 0.128 | 14.193 | <0.001*** | |
| Months of Observations** | 294.000 | 11 | 1889 | 0.146 | 12.668 | <0.001*** | |
| Shelf Position | 25.790 | 2 | 194 | 0.119 | 12.051 | <0.001*** | |
| Ocean Basin** | 324.008 | 2 | 2043 | 0.157 | 9.982 | <0.001*** | |
| Peak Temperature** | 102.300 | 5 | 678 | 0.142 | 9.725 | <0.001*** | |
| Depth | 119.339 | 6 | 822 | 0.137 | 9.513 | <0.001*** | |
| Location* | 323.562 | 10 | 2042 | 0.152 | 9.147 | <0.001*** | |
| Transmission of Algae to Offspring | 118.282 | 1 | 700 | 0.167 | 8.950 | <0.01** | |
| Level of Colony Integration* | 192.431 | 2 | 1111 | 0.171 | 7.919 | <0.001*** | |
| Clouds/Stormy Weather** | 71.90 | 1 | 425 | 0.167 | 6.483 | <0.05* | |
| Doldrums** | 47.764 | 1 | 282 | 0.166 | 6.335 | <0.05* | |
| Dominant taxa | 34.475 | 8 | 223 | 0.130 | 6.254 | <0.001*** | |
| Species | 71.165 | 24 | 480 | 0.122 | 5.340 | <0.01** | |
| Complexity of Skeleton | 292.282 | 1 | 1841 | 0.158 | 5.220 | <0.05* | |
| Morphology** | 192.341 | 6 | 1111 | 0.170 | 4.058 | <0.001*** | |
| Genus | 259.843 | 37 | 1632 | 0.149 | 3.899 | <0.001*** | |
| Anthropogenic Input* | 38.114 | 2 | 286 | 0.131 | 3.545 | <0.05* | |
| Family | 302.905 | 16 | 1902 | 0.156 | 3.456 | <0.001*** | |
| Reproductive Mode | 139.803 | 1 | 821 | 0.170 | 3.199 | >0.05 | |
| Sex | 118.804 | 1 | 821 | 0.144 | 2.650 | >0.05 | |
| Reef Type | 104.075 | 3 | 787 | 0.132 | 1.660 | >0.05 | |
| Generalization | 168.780 | 1 | 914 | 0.185 | 0.748 | >0.05 | |
| Anthropogenic Pressure** | 28.085 | 1 | 221 | 0.127 | 0.710 | >0.05 | |
| Pollutants** | 32.422 | 1 | 225 | 0.144 | 0.471 | >0.05 | |
| Upwelling** | 56.378 | 1 | 367 | 0.154 | 0.064 | >0.05 | |
| |||||||
| Source | SS Type III | df | n | MS | F | p | |
| Thermal Stress Accumulation* | 178.988 | 1 | 1461 | 0.112 | 144.065 | <0.001*** | |
| Timing of Observations* | 227.967 | 2 | 1583 | 0.124 | 130.470 | <0.001*** | |
| Recovery Time | 23.176 | 2 | 146 | 0.085 | 64.911 | <0.001*** | |
| Degree Heating Index** | 178.988 | 5 | 1461 | 0.103 | 56.784 | <0.001*** | |
| Habitat | 43.400 | 4 | 249 | 0.098 | 54.519 | <0.001*** | |
| Subregion** | 249.592 | 8 | 1634 | 0.128 | 40.393 | <0.001*** | |
| Level of Colony Integration* | 152.856 | 2 | 942 | 0.150 | 39.704 | <0.001*** | |
| Anthropogenic Pressure* | 21.477 | 2 | 279 | 0.061 | 38.522 | <0.001*** | |
| Months of Observations** | 224.857 | 11 | 1575 | 0.113 | 38.024 | <0.001*** | |
| Decade** | 249.592 | 3 | 1634 | 0.144 | 35.630 | <0.001*** | |
| Anthropogenic Input** | 13.962 | 1 | 216 | 0.057 | 30.969 | <0.001*** | |
| Location* | 249.530 | 10 | 1633 | 0.130 | 29.397 | <0.001*** | |
| Upwelling** | 47.361 | 1 | 365 | 0.1320 | 29.871 | <0.001*** | |
| Country** | 215.033 | 31 | 1539 | 0.088 | 29.807 | <0.001*** | |
| Ocean Basin*** | 249.592 | 2 | 1634 | 0.149 | 22.211 | <0.001*** | |
| Complexity of Skeleton | 209.574 | 1 | 1459 | 0.141 | 25.103 | <0.001*** | |
| Reef Type | 88.178 | 3 | 666 | 0.122 | 20.862 | <0.001*** | |
| Generalization | 125.061 | 1 | 780 | 0.156 | 20.282 | <0.001*** | |
| Peak Temperature** | 80.630 | 5 | 603 | 0.115 | 20.186 | <0.001*** | |
| Shelf Position | 20.995 | 2 | 149 | 0.113 | 19.360 | <0.001*** | |
| Depth | 100.204 | 6 | 784 | 0.112 | 19.716 | <0.001*** | |
| Clouds/Stormy Weather** | 66.379 | 1 | 425 | 0.167 | 18.830 | <0.001*** | |
| Pollutants | 15.868 | 1 | 223 | 0.171 | 14.440 | <0.001*** | |
| Morphology** | 152.856 | 6 | 942 | 0.150 | 14.387 | <0.01** | |
| Relief from Physical Stress* | 90.368 | 3 | 645 | 0.133 | 12.686 | <0.001*** | |
| Bleaching Occurrence* | 249.592 | 4 | 1634 | 0.149 | 10.511 | <0.001*** | |
| Dominant Taxa | 36.509 | 8 | 217 | 0.129 | 9.340 | <0.001*** | |
| Annual Temperature Range** | 45.074 | 4 | 382 | 0.110 | 7.629 | <0.001*** | |
| Family | 227.860 | 16 | 1507 | 0.142 | 6.822 | <0.01** | |
| Genus | 200.483 | 37 | 1276 | 0.146 | 3.573 | <0.01** | |
| Species | 39.719 | 24 | 394 | 0.088 | 3.342 | <0.01** | |
| Reproductive Mode | 113.360 | 1 | 714 | 0.158 | 3.199 | >0.05 | |
| Doldrums** | 47.134 | 1 | 274 | 0.171 | 2.841 | >0.05 | |
| Transmission of Algae to Offspring | 90.930 | 1 | 609 | 0.149 | 2.770 | >0.05 | |
| Sex | 4.908 | 1 | 714 | 0.007 | 0.0477 | >0.05 | |
| |||||||
| Grouped Factor | Grouped Relative Importance | Source | F | Relative Importance | |||
| Timing | 30.1% | Decade | 33.735 | 12.43% | |||
| Recovery | 14.193 | 5.23% | |||||
| Months of Observations | 12.668 | 4.67% | |||||
| Weather | 28.7% | Degree Heating Index | 39.994 | 14.74 | |||
| Annual Temperature Range | 15.307 | 5.64% | |||||
| Peak Temperature | 9.725 | 3.58% | |||||
| Cloudy/Stormy Weather | 6.483 | 2.39% | |||||
| Doldrums | 6.335 | 2.33% | |||||
| Location | 21.4% | Subregion | 23.095 | 8.51% | |||
| Country | 20.182 | 7.44% | |||||
| Ocean Basin | 9.982 | 3.68% | |||||
| Biology and Ecology | 15.1% | Complexity of Skeleton | 5.22 | 1.92% | |||
| Depth | 9.513 | 3.51% | |||||
| Transmission of Algae to Offspring | 8.950 | 3.30% | |||||
| Habitat | 5.444 | 2.01% | |||||
| Morphology | 4.058 | 1.50% | |||||
| Taxonomy | 1.7 % | Species | 5.340 | 1.97% | |||
| Genus | 3.899 | 1.44% | |||||
| Family | 3.456 | 1.27% | |||||
| |||||||
| Grouped Factors | Grouped Relative Importance | Source | F | Relative Importance | |||
| Timing | 21.1% | Recovery Time | 64.911 | 9.56% | |||
| Months of Observations | 38.024 | 5.60% | |||||
| Decade | 35.630 | 5.25% | |||||
| Weather | 19.6% | Degree Heating Index | 56.784 | 8.36% | |||
| Upwelling | 29.871 | 4.40% | |||||
| Peak Temperature | 20.186 | 2.97% | |||||
| Clouds and Stormy Weather | 18.830 | 2.77% | |||||
| Annual Temperature Range | 7.629 | 1.12% | |||||
| Biology and Ecology | 22.7% | Complexity of Skeleton | 25.103 | 3.70% | |||
| Habitat | 20.825 | 3.07% | |||||
| Generalization | 20.282 | 2.99% | |||||
| Depth | 19.716 | 2.90% | |||||
| Pollutants | 14.440 | 2.13% | |||||
| Morphology | 14.387 | 2.12% | |||||
| Location | 16.0% | Subregion | 40.393 | 5.95% | |||
| Country | 29.807 | 4.39% | |||||
| Ocean Basin | 22.211 | 3.27% | |||||
| Taxonomy | 2.0% | Family | 6.822 | 1.00% | |||
| Genus | 3.573 | 0.53% | |||||
| Species | 3.342 | 0.49% | |||||
| |||||||
| Source | n | Pearson’s Correlation Coefficient | p | ||||
| Recovery | 151 | 0.355 | <0.001*** | ||||
| Degree Heating Index** | 1759 | 0.284 | <0.001*** | ||||
| Shelf position** | 195 | -0.282 | <0.001*** | ||||
| Thermal Stress Accumulation* | 1759 | 0.258 | <0.001*** | ||||
| Peak Temperature | 679 | 0.212 | <0.001*** | ||||
| Months of Observations* | 1890 | 0.166 | <0.001*** | ||||
| Doldrums** | 283 | -0.148 | <0.05* | ||||
| Timing of Observations* | 1898 | 0.147 | <0.001*** | ||||
| Relief from Physical Stress* | 670 | -0140 | <0.001*** | ||||
| Decade** | 2044 | -0.127 | <0.001*** | ||||
| Annual Temperature Range** | 479 | 0.123 | <0.01** | ||||
| Weather** | 426 | -0.123 | <0.05* | ||||
| Country** | 1915 | -0.121 | <0.001*** | ||||
| Transmission of Algae to Offspring | 701 | -0.112 | <0.01** | ||||
| Level of Colony Integration* | 1112 | 0.111 | >0.001*** | ||||
| Habitat | 304 | 0.088 | >0.05 | ||||
| Genus | 1633 | -0.072 | <0.01** | ||||
| Reef Type | 788 | -0.070 | >0.05 | ||||
| Sex | 806 | 0.067 | >0.05 | ||||
| Family | 1903 | -0.063 | <0.01** | ||||
| Anthropogenic Pressure** | 222 | 0.057 | >0.05 | ||||
| Complexity of Skeleton | 1842 | -0.053 | <0.05* | ||||
| Ocean | 2044 | -0.051 | <0.05* | ||||
| Subregion | 2044 | 0.049 | <0.05* | ||||
| Anthropogenic Input* | 287 | 0.049 | >0.05 | ||||
| Depth | 823 | -0.047 | >0.05 | ||||
| Pollution | 226 | -0.046 | >0.05 | ||||
| Species | 481 | -0.038 | >0.05 | ||||
| Dominant Taxa | 224 | -0.034 | >0.05 | ||||
| Location* | 2043 | -0.033 | >0.05 | ||||
| Bleaching Occurrence* | 2044 | -0.032 | >0.05 | ||||
| Generalization/Specialization | 915 | 0.029 | >0.05 | ||||
| Reproductive Mode | 822 | 0.025 | >0.05 | ||||
| Upwelling | 368 | 0.013 | >0.05 | ||||
| Morphology | 1112 | 0.006 | >0.05 | ||||
| |||||||
| Source | n | Pearson’s Correlation Coefficient | p | ||||
| Recovery | 147 | 0.490 | <0.001*** | ||||
| Months of Observations** | 1576 | 0.393 | <0.001*** | ||||
| Degree Heating Index** | 1462 | 0.391 | <0.001*** | ||||
| Thermal Stress Accumulation* | 1462 | 0.371 | <0.001*** | ||||
| Timing of Observations | 1576 | 0.370 | <0.001*** | ||||
| Anthropogenic Pressure** | 217 | 0.355 | <0.001*** | ||||
| Anthropogenic Input* | 280 | 0.343 | <0.001*** | ||||
| Upwelling** | 366 | -0.275 | <0.001*** | ||||
| Shelf Position | 150 | -0.256 | <0.01** | ||||
| Pollution** | 224 | 0.247 | <0.001*** | ||||
| Weather** | 402 | -0.212 | <0.001*** | ||||
| Relief from Physical Stress* | 646 | -0.182 | <0.001*** | ||||
| Annual Temperature Range** | 383 | 0.162 | <0.01** | ||||
| Level of Colony Integration* | 943 | -0.160 | <0.001*** | ||||
| Morphology** | 943 | -0.160 | <0.001*** | ||||
| Generalization/Specialization | 781 | 0.159 | <0.001*** | ||||
| Habitat | 250 | -0.155 | <0.05* | ||||
| Country** | 1540 | -0.135 | <0.001*** | ||||
| Location* | 1634 | -0.131 | <0.001*** | ||||
| Complexity of Skeleton | 1460 | -0.130 | <0.001*** | ||||
| Family | 1508 | -0.119 | <0.001*** | ||||
| Doldrums** | 275 | -.101 | >0.05 | ||||
| Species | 395 | -0.098 | >0.05 | ||||
| Ocean** | 1635 | 0.091 | <0.001*** | ||||
| Genus | 1277 | -0.085 | <0.001*** | ||||
| Decade** | 1635 | -0.071 | <0.01** | ||||
| Reproductive Mode | 715 | 0.067 | >0.05 | ||||
| Transmission of Algae to Offspring | 610 | -0.067 | >0.05 | ||||
| Depth | 785 | -0.065 | >0.05 | ||||
| Bleaching Occurrence* | 1635 | -0.032 | >0.05 | ||||
| Sex | 715 | -0.025 | >0.05 | ||||
| Peak Temperature** | 604 | 0.013 | >0.05 | ||||
| Dominant taxa | 218 | -0.003 | >0.05 | ||||
| A) ANOVAs (i and iii) and descriptive statistics (ii and iv) for inter-familial variation in bleaching susceptibility (i and ii) and mortality (iii and iv). | |||||||
| i) Bleaching susceptibility -where n>50 for each family and n ≥ 5 for factors | |||||||
| Source | SS(Type III) | df | n | MS | F | p | |
| Thermal stress accumulation | 182.053 | 2 | 1095 | 0.156 | 35.905 | <0.001*** | |
| Bleaching occurrence | 213.121 | 4 | 1227 | 0.161 | 24.720 | <0.001*** | |
| Ocean_subregion | 212.574 | 11 | 1222 | 0.147 | 21.231 | <0.001*** | |
| Timing of observations | 205.053 | 2 | 1193 | 0.167 | 16.978 | <0.001*** | |
| Relief from weather | 60.127 | 3 | 406 | 0.139 | 9.570 | <0.001*** | |
| Habitat | 48.313 | 7 | 250 | 0.160 | 8.498 | <0.001*** | |
| Level of colony integration | 138.745 | 2 | 726 | 0.187 | 8.130 | <0.001*** | |
| Species | 87.156 | 44 | 469 | 0.126 | 6.095 | <0.001*** | |
| Transmission of algae to offspring | 90.565 | 2 | 505 | 0.176 | 5.228 | <0.01** | |
| Genus | `199.376 | 38 | 1111 | 0.162 | 4.083 | <0.001*** | |
| FAMILY | 213.121 | 6 | 1227 | 0.171 | 3.674 | <0.01** | |
| Dominant taxa | 22.823 | 7 | 142 | 0.149 | 2.645 | <0.05* | |
| Shelf position | 65.446 | 6 | 462 | 0.139 | 2.210 | <0.05* | |
| Sex | 103.911 | 1 | 570 | 0.182 | 1.663 | >0.05 | |
| Complexity of skeleton | 212.751 | 1 | 1226 | 0.173 | 1.445 | >0.05 | |
| Anthropogenic Input | 25.554 | 3 | 173 | 0.157 | 1.303 | >0.05 | |
| Depth | 70.637 | 4 | 469 | 0.150 | 1.284 | >0.05 | |
| Generalisation | 129.022 | 1 | 643 | 0.20-1 | 1.241 | >0.05 | |
| Reproductive mode | 105.887 | 2 | 578 | 0.183 | 1.228 | >0.05 | |
| *Significant, **strongly significant, ***highly significant. | |||||||
| ii) Bleaching-related mortality- where n>50 for each family and n ≥ 5 for factors | |||||||
| Source | SS(Type III) | df | n | MS | F | p | |
| Timing of observations | 172.975 | 2 | 1193 | 0.123 | 109.773 | <0.001*** | |
| Thermal stress accumulation | 127.812 | 2 | 1095 | 0.101 | 85.998 | <0.001*** | |
| Habitat | 38.317 | 7 | 250 | 0.080 | 33.614 | <0.001*** | |
| Ocean_subregion | 189.248 | 1 | 1222 | 0.120 | 33.054 | <0.001*** | |
| Bleaching occurrence | 189.565 | 4 | 1227 | 0.143 | 26.125 | <0.001*** | |
| Level of colony integration | 120.787 | 2 | 726 | 0.156 | 24.858 | <0.001*** | |
| Anthropogenic Input | 13.428 | 3 | 173 | 0.056 | 23.419 | <0.001** | |
| Relief from weather | 44.711 | 3 | 406 | 0.096 | 21.318 | <0.001*** | |
| Complexity of skeleton | 189.502 | 1 | 1226 | 0.152 | 18.628 | <0.001*** | |
| Generalisation | 110.218 | 1 | 643 | 0.168 | 14.564 | <0.001*** | |
| Shelf position | 57.561 | 6 | 462 | 0.108 | 12.672 | <0.001*** | |
| FAMILY | 189.565 | 6 | 1227 | 0.148 | 9.277 | <0.001*** | |
| Depth | 62.523 | 4 | 469 | 0.129 | 5.091 | <0.01** | |
| Dominant taxa | 22.332 | 7 | 142 | 0.133 | 4.658 | <0.001*** | |
| Reproductive mode | 100.046 | 2 | 578 | 0.1722 | 3.222 | <0.05* | |
| Transmission of algae to offspring | 80.916 | 2 | 505 | 0.159 | 2.700 | >0.05 | |
| Species | 60.851 | 44 | 469 | 0.112 | 2.734 | <0.001*** | |
| Genus | 176.381 | 38 | 1111 | 0.151 | 2.589 | <0.001*** | |
| Sex | 99.134 | 1 | 570 | 0.174 | 0.081 | >0.05 | |
| A) Averages for inter-familial variation in bleaching susceptibility (i) and mortality (ii), ordered by F values of ANOVAs | |||||||
| iii) Bleaching-related mortality- where n>50 for each family and n ≥ 5 for factors | |||||||
| Source | SS(Type III) | df | n | MS | F | p | |
| Timing of observations | 172.975 | 2 | 1193 | 0.123 | 109.773 | <0.001*** | |
| Thermal stress accumulation | 127.812 | 2 | 1095 | 0.101 | 85.998 | <0.001*** | |
| Habitat | 38.317 | 7 | 250 | 0.080 | 33.614 | <0.001*** | |
| Ocean_subregion | 189.248 | 1 | 1222 | 0.120 | 33.054 | <0.001*** | |
| Bleaching occurrence | 189.565 | 4 | 1227 | 0.143 | 26.125 | <0.001*** | |
| Level of colony integration | 120.787 | 2 | 726 | 0.156 | 24.858 | <0.001*** | |
| Anthropogenic Input | 13.428 | 3 | 173 | 0.056 | 23.419 | <0.001** | |
| Relief from weather | 44.711 | 3 | 406 | 0.096 | 21.318 | <0.001*** | |
| Complexity of skeleton | 189.502 | 1 | 1226 | 0.152 | 18.628 | <0.001*** | |
| Generalisation | 110.218 | 1 | 643 | 0.168 | 14.564 | <0.001*** | |
| Shelf position | 57.561 | 6 | 462 | 0.108 | 12.672 | <0.001*** | |
| FAMILY | 189.565 | 6 | 1227 | 0.148 | 9.277 | <0.001*** | |
| Depth | 62.523 | 4 | 469 | 0.129 | 5.091 | <0.01** | |
| Dominant taxa | 22.332 | 7 | 142 | 0.133 | 4.658 | <0.001*** | |
| Reproductive mode | 100.046 | 2 | 578 | 0.1722 | 3.222 | <0.05* | |
| Transmission of algae to offspring | 80.916 | 2 | 505 | 0.159 | 2.700 | >0.05 | |
| Species | 60.851 | 44 | 469 | 0.112 | 2.734 | <0.001*** | |
| Genus | 176.381 | 38 | 1111 | 0.151 | 2.589 | <0.001*** | |
| Sex | 99.134 | 1 | 570 | 0.174 | 0.081 | >0.05 | |
| B) ANOVAs for inter-generic variation in bleaching (i) susceptibility and (ii) mortality | |||||||
| i) Bleaching susceptibility -where n>40 for each genus and n ≥ 5 for factors | |||||||
| Source | SS(Type III) | df | n | MS | F | p | |
| Bleaching occurrence | 127.755 | 4 | 728 | 0.150 | 31.678 | <0.001*** | |
| Thermal stress accumulation | 108.071 | 2 | 635 | 0.153 | 31.519 | <0.001*** | |
| Ocean_subregion | 127.755 | 12 | 728 | 0.128 | 23.602 | <0.001*** | |
| Timing of observations | 122.590 | 2 | 702 | 0.165 | 21.635 | <0.001*** | |
| Level of colony integration | 102.982 | 2 | 549 | 0.177 | 17.241 | <0.001*** | |
| Habitat | 36.723 | 6 | 179 | 0.139 | 15.097 | <0.001*** | |
| Transmission of algae to offspring | 58.713 | 1 | 338 | 0.167 | 13.570 | <0.001*** | |
| Species | 64.990 | 29 | 353 | 0.116 | 8.082 | <0.001*** | |
| Relief from weather | 31.971 | 3 | 215 | 0.139 | 6.166 | <0.001*** | |
| GENUS | 127.400 | 7 | 726 | 0.169 | 5.146 | <0.001*** | |
| Dominant taxa | 13.548 | 5 | 89 | 0.126 | 4.639 | <0.01** | |
| Depth | 42.484 | 5 | 312 | 0.130 | 3.717 | <0.01** | |
| Shelf position | 33.815 | 5 | 251 | 0.133 | 1.635 | >0.05 | |
| Anthropogenic input | 8.753 | 3 | 78 | 0.110 | 1.582 | >0.05 | |
| Reproductive mode | 69.176 | 1 | 395 | 0.175 | 0.870 | >0.05 | |
| Sex | 68.715 | 1 | 395 | 0.174 | 0.404 | >0.05 | |
| Generalisation | 87.553 | 1 | 442 | 0.198 | 0.176 | >0.05 | |
| ii) Bleaching mortality - where n>40 for each genus and n ≥ 5 for factors | |||||||
| Source | SS(Type III) | df | n | MS | F | p | |
| Thermal stress accumulation | 99.721 | 2 | 635 | 0.122 | 91.828 | <0.001*** | |
| Habitat | 32.078 | 6 | 179 | 0.049 | 80.867 | <0.001*** | |
| Timing of observations | 127.290 | 2 | 702 | 0.149 | 75.719 | <0.001*** | |
| Generalisation | 81.014 | 1 | 442 | 0.171 | 32.106 | <0.001*** | |
| Ocean_subregion | 138.023 | 12 | 728 | 0.132 | 27.512 | <0.001*** | |
| Level of colony integration | 103.917 | 2 | 549 | 0.175 | 23.467 | <0.001*** | |
| Relief from weather | 36.411 | 3 | 215 | 0.144 | 13.596 | <0.001*** | |
| Bleaching occurrence | 138.023 | 4 | 728 | 0.177 | 13.640 | <0.001*** | |
| Depth | 53.287 | 5 | 312 | 0.145 | 12.273 | <0.001*** | |
| Anthropogenic input | 7.958 | 3 | 78 | 0.082 | 7.478 | <0.001*** | |
| GENUS | 137.823 | 7 | 726 | 0.179 | 7.122 | <0.001*** | |
| Reproductive mode | 74.791 | 1 | 395 | 0.187 | 6.973 | <0.01** | |
| Shelf position | 44.202 | 5 | 251 | 0.1598 | 6.724 | <0.001*** | |
| Dominant taxa | 15.100 | 5 | 89 | 0.131 | 6.311 | <0.001*** | |
| Transmission of algae to offspring | 57.933 | 1 | 338 | 0.169 | 6.096 | <0.05* | |
| Sex | 74.791 | 1 | 395 | 0.188 | 4.143 | <0.05* | |
| Species | 48.129 | 29 | 353 | 0.110 | 3.886 | <0.001*** | |
| C) ANOVAs for inter-species variation in bleaching (i) susceptibility and (ii) mortality | |||||||
| i) Bleaching susceptibility (where n>20) for each species and n ≥ 5 for individual records) | |||||||
| Source | SS(Type III) | df | n | MS | F | p | |
| Transmission of algae to offspring | 21.933 | 1 | 156 | 0.126 | 18.592 | <0.001*** | |
| Ocean_subregion | 25.818 | 6 | 192 | 0.091 | 16.081 | <0.001*** | |
| Bleaching occurrence | 26.753 | 4 | 198 | 0.116 | 9.059 | <0.001*** | |
| Relief from weather | 5.588 | 2 | 50 | 0.090 | 6.980 | <0.01** | |
| Thermal stress accumulation | 24.528 | 2 | 184 | 0.126 | 6.233 | <0.01** | |
| SPECIES | 26.753 | 7 | 198 | 0.115 | 5.833 | <0.001*** | |
| Timing of observations | 26.300 | 2 | 195 | 0.129 | 5.211 | <0.01** | |
| Sex | 26.753 | 1 | 198 | 0.133 | 4.195 | <0.05* | |
| Complexity of skeleton | 26.753 | 1 | 198 | 0.134 | 3.200 | >0.05 | |
| Anthropogenic input | 6.737 | 3 | 100 | 0.064 | 2.790 | <0.05* | |
| Depth | 8.299 | 4 | 101 | 0.080 | 1.632 | >0.05 | |
| Shelf position | 6.752 | 1 | 84 | 0.080 | 1.095 | >0.05 | |
| Dominant taxa | 1.288 | 2 | 18 | 0.075 | 0.564 | >0.05 | |
| Habitat | 1.473 | 1 | 57 | 0.026 | 0.780 | >0.05 | |
| Level of colony integration | 26.753 | 1 | 198 | 0.136 | 0.166 | >0.05 | |
| Reproductive mode | 26.753 | 1 | 198 | 0.136 | 0.014 | >0.05 | |
| ii) Bleaching mortality -where n>20 for each species and n ≥ 5 for factors | |||||||
| Source | SS(Type III) | df | n | MS | F | p | |
| Timing of observations | 15.062 | 2 | 195 | 0.054 | 42.565 | <0.001*** | |
| Level of colony integration | 15.481 | 1 | 198 | 0.069 | 28.246 | <0.001*** | |
| Thermal stress accumulation | 13.777 | 2 | 184 | 0.068 | 10.936 | <0.001*** | |
| Reproductive mode | 14.705 | 1 | 198 | 0.075 | 10.789 | <0.01** | |
| Relief from weather | 3.419 | 2 | 50 | 0.053 | 8.534 | <0.01** | |
| SPECIES | 15.841 | 7 | 198 | 0.066 | 6.016 | <0.001**** | |
| Anthropogenic input | 6.874 | 3 | 100 | 0.060 | 5.932 | <0.01** | |
| Location | 14.003 | 6 | 192 | 0.063 | 5.880 | <0.001*** | |
| Complexity of skeleton | 15.481 | 1 | 198 | 0.076 | 5.810 | <0.05* | |
| Sex | 15.481 | 1 | 198 | 0.076 | 5.499 | <0.05* | |
| Bleaching occurrence | 15.481 | 4 | 198 | 0.076 | 2.401 | <0.05* | |
| Depth | 7.395 | 4 | 101 | 0.071 | 1.844 | >0.05 | |
| Transmission of algae to offspring | 13.048 | 1 | 156 | 0.084 | 1.156 | >0.05 | |
| Habitat | 1.473 | 4 | 57 | 0.026 | 0.780 | >0.05 | |
| Dominant taxa | 1.476 | 2 | 18 | 0.090 | 0.227 | >0.05 | |
| Shelf position | 4.985 | 1 | 84 | 0.060 | 0.173 | >0.05 | |
| ||||||
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 235.725 | 2 | 1449 | 0.151 | 57.743 | <0.001*** |
| Mortality | 174.188 | 2 | 1449 | 0.103 | 121.648 | <0.001*** |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 149.950 | 1 | 1019 | 0.341 | 0.341 | >0.05 |
| Mortality | 74.552 | 1 | 1019 | 0.073 | 10.261 | <0.01** |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 53.651 | 1 | 334 | 0.136 | 62.518 | <0.001*** |
| Mortality | 47.720 | 1 | 334 | 0.143 | 0.184 | >0.05 |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 14.701 | 1 | 94 | 0.141 | 11.114 | <0.01** |
| Mortality | 26.844 | 1 | 94 | 0.256 | 11.973 | <0.01** |
| ||||||
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 237.725 | 3 | 1449 | 0.161 | 5.568 | <0.01** |
| Mortality | 174.188 | 3 | 1449 | 0.109 | 51.418 | <0.001*** |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 129.953 | 3 | 824 | 0.153 | 9.469 | <0.001*** |
| Mortality | 54.999 | 3 | 824 | 0.066 | 3.503 | <0.05* |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 43.908 | 3 | 293 | 0.144 | 4.873 | <0.01** |
| Mortality | 28.758 | 3 | 293 | 0.088 | 11.650 | <0.001*** |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 85.133 | 4 | 464 | 0.172 | 8.691 | <0.001*** |
| Mortality | 112.005 | 4 | 464 | 0.217 | 14.211 | <0.001*** |
| ||||||
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 323.562 | 10 | 2042 | 0.152 | 9.147 | <0.0001*** |
| Mortality | 249.530 | 10 | 1633 | 0.130 | 29.397 | <0.001*** |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 20.845 | 5 | 117 | 0.103 | 18.088 | <0.001*** |
| Mortality | 11.427 | 5 | 77 | 0.064 | 21.347 | <0.001*** |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 23.828 | 5 | 125 | 0.156 | 6.612 | <0.001*** |
| Mortality | 26.291 | 5 | 111 | 0.157 | 12.306 | <0.001*** |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 3.508 | 1 | 32 | 0.077 | 14.585 | <0.01** |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 9.558 | 6 | 57 | 0.111 | 5.822 | <0.001*** |
| Mortality | 9.898 | 5 | 40 | 0.196 | 3.118 | <0.05* |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 52.525 | 4 | 345 | 0.125 | 19.418 | <0.001*** |
| Mortality | 48.449 | 4 | 222 | 0.143 | 30.464 | <0.001*** |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 66.861 | 13 | 454 | 0.132 | 5.127 | <0.001*** |
| Mortality | 29.618 | 13 | 387 | 0.065 | 6.051 | <0.001*** |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 40.794 | 5 | 247 | 0.111 | 24.866 | <0.001*** |
| Mortality | 2.533 | 5 | 179 | 0.012 | 6.520 | <0.01** |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 47.656 | 7 | 285 | 0.067 | 61.703 | <0.001*** |
| Mortality | 60.043 | 7 | 274 | 0.093 | 54.061 | <00.001*** |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 15.159 | 2 | 143 | 0.102 | 0.038 | >0.05 |
| Mortality | 16.565 | 2 | 114 | 0.134 | 5.476 | <0.01** |
| ||||||
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 192.341 | 6 | 111 | 0.170 | 4.058 | <0.001*** |
| Mortality | 152.704 | 6 | 940 | 0.1650 | 14.564 | <0.001*** |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 90.835 | 1 | 464 | 0.196 | 0.254 | >0.05 |
| Mortality | 85.834 | 1 | 378 | 0.227 | 0.930 | >0.05 |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 71.473 | 1 | 490 | 0.146 | 0.971 | >0.05 |
| Mortality | 35.442 | 1 | 428 | 0.083 | 0.475 | >0.05 |
| ||||||
| Factor | SS (Type III) | df | n | MS | F | p |
| Susceptibility | 15.234 | 1 | 102 | 0.150 | 0.320 | >0.05 |
| Mortality | 6.707 | 1 | 87 | 0.078 | 0.075 | >0.05 |
| |||||||||
| Thermal stress accumulation | Timing of observations | Family | Susceptibility | ±SE | n | Mortality | ±SE | (S-M)/S | |
| Low | Beginning | Milleporidae | 37.51 | 8.75 | 13 | 19.26 | 10.00 | 0.49 | |
| Acroporidae | 44.04 | 3.28 | 122 | 10.73 | 2.08 | 0.76 | |||
| Pocilloporidae | 42.95 | 4.40 | 76 | 10.07 | 2.78 | 0.98 | |||
| Euphyllidae | 37.36 | 9.19 | 10 | 8.80 | 6.66 | 0.76 | |||
| Agariciidae | 42.26 | 5.99 | 33 | 6.06 | 4.02 | 0.86 | |||
| Merulinidae | 43.76 | 2.93 | 105 | 5.18 | 1.30 | 0.88 | |||
| Montastraeidae | 28.37 | 0.00 | 7 | 4.35 | 0.00 | 0.85 | |||
| Poritidae | 31.81 | 3.27 | 95 | 3.94 | 1.36 | 0.88 | |||
| Mussidae | 37.89 | 5.55 | 34 | 0.74 | 0.68 | 0.98 | |||
| Siderastreidae | 60.63 | 8.27 | 13 | 0.65 | 0.58 | 0.99 | |||
| Alcyonacae | 40.40 | 14.88 | 5 | 0.40 | 0.24 | 0.99 | |||
| Lobophyllidae | 38.83 | 8.46 | 12 | 0.25 | 0.25 | 0.99 | |||
| Fungiidae | 40.52 | 7.09 | 23 | 0.22 | 0.22 | 0.99 | |||
| Diploastraeidae | 18.53 | 10.83 | 7 | 0.00 | 0.00 | 1 | |||
| Meandrinidae | 15.54 | 9.08 | 7 | 0.00 | 0.00 | 1 | |||
| Dendrophylliidae | 0.75 | 0.75 | 4 | 0.00 | 0.00 | 1 | |||
| Middle | Milleporidae | 54.90 | 18.35 | 5 | 29.41 | 17.91 | 0.46 | ||
| Acroporidae | 49.74 | 5.62 | 32 | 13.04 | 5.21 | 0.74 | |||
| Poritidae | 49.90 | 7.00 | 26 | 11.07 | 4.35 | 0.78 | |||
| Alcyonacae | 51.00 | 28.88 | 3 | 6.00 | 6.00 | 0.88 | |||
| Mussidae | 45.26 | 6.83 | 21 | 5.05 | 2.38 | 0.89 | |||
| Agariciidae | 24.00 | 12.06 | 3 | 3.67 | 3.67 | 0.85 | |||
| Fungiidae | 23.25 | 11.52 | 8 | 4.50 | 1.93 | 0.81 | |||
| Merulinidae | 34.45 | 5.71 | 35 | 2.71 | 1.17 | 0.92 | |||
| Siderastreidae | 55.91 | 9.90 | 12 | 2.05 | 0.71 | 0.96 | |||
| Pocilloporidae | 27.71 | 10.95 | 7 | 1.14 | 0.46 | 0.96 | |||
| Lobophyllidae | 34.50 | 13.81 | 8 | 0.75 | 0.75 | 0.98 | |||
| Euphyllidae | 26.17 | 11.44 | 6 | 0.67 | 0.42 | 0.97 | |||
| End | Agariciidae | 38.33 | 11.39 | 12 | 22.46 | 9.71 | 0.41 | ||
| Acroporidae | 45.59 | 5.31 | 47 | 17.51 | 4.69 | 0.62 | |||
| Poritidae | 30.86 | 7.24 | 23 | 15.30 | 6.14 | 0.50 | |||
| Mussidae | 28.35 | 10.35 | 11 | 13.39 | 4.79 | 0.53 | |||
| Euphyllidae | 30.39 | 9.16 | 9 | 12.49 | 9.80 | 0.59 | |||
| Merulinidae | 39.43 | 4.58 | 49 | 9.64 | 2.27 | 0.76 | |||
| Pocilloporidae | 38.37 | 7.98 | 20 | 9.45 | 5.95 | 0.75 | |||
| Milleporidae | 9.40 | 9.40 | 3 | 8.00 | 8.00 | 0.15 | |||
| Siderastreidae | 37.78 | 6.24 | 5 | 7.10 | 3.38 | 0.81 | |||
| Fungiidae | 12.32 | 3.69 | 11 | 1.00 | 1.00 | 0.92 | |||
| Lobophyllidae | 29.55 | 8.01 | 11 | 0.00 | 0.00 | 1 | |||
| Medium | Beginning | Acroporidae | 58.21 | 10.41 | 17 | 22.15 | 9.09 | 0.62 | |
| Milleporidae | 37.31 | 13.49 | 8 | 13.06 | 12.08 | 0.65 | |||
| Pocilloporidae | 7.00 | 2.98 | 9 | 6.94 | 5.19 | 0.01 | |||
| Meandrinidae | 33.08 | 6.14 | 12 | 5.50 | 4.07 | 0.83 | |||
| Merulinidae | 64.93 | 6.66 | 29 | 4.21 | 1.94 | 0.94 | |||
| Poritidae | 35.95 | 9.87 | 11 | 3.41 | 1.69 | 0.91 | |||
| Mussidae | 28.42 | 4.31 | 31 | 2.13 | 1.02 | 0.93 | |||
| Siderastreidae | 63.80 | 8.18 | 10 | 1.60 | 0.91 | 0.97 | |||
| Montastreidae | 65.17 | 13.59 | 6 | 1.00 | 1.00 | 0.98 | |||
| Agariciidae | 45.38 | 11.47 | 13 | 0.77 | 0.77 | 0.98 | |||
| Fungiidae | 32.85 | 6.91 | 13 | 0.00 | 0.00 | 1 | |||
| Middle | Acroporidae | 34.73 | 8.93 | 11 | 21.73 | 7.44 | 0.37 | ||
| Poritidae | 36.13 | 8.57 | 8 | 14.13 | 7.53 | 0.61 | |||
| Pocilloporidae | 55.00 | 17.09 | 4 | 13.25 | 8.44 | 0.76 | |||
| Fungiidae | 48.67 | 14.50 | 3 | 6.00 | 2.52 | 0.88 | |||
| Agariciidae | 24.00 | 12.06 | 3 | 3.67 | 3.67 | 0.85 | |||
| Euphyllidae | 40.40 | 20.28 | 5 | 3.00 | 3.00 | 0.93 | |||
| Lobophyllidae | 22.75 | 6.26 | 4 | 2.50 | 2.50 | 0.89 | |||
| Merulinidae | 24.63 | 6.37 | 19 | 1.84 | 0.65 | 0.93 | |||
| Mussidae | 37.50 | 23.94 | 4 | 0.50 | 0.50 | 0.99 | |||
| End | Diploastraeidae | 96.67 | 3.33 | 3 | 96.67 | 3.33 | 0 | ||
| Fungiidae | 83.77 | 8.20 | 6 | 78.22 | 13.68 | 0.07 | |||
| Acroporidae | 84.79 | 5.34 | 30 | 74.93 | 6.78 | 0.12 | |||
| Euphyllidae | 86.00 | 12.00 | 4 | 73.50 | 24.50 | 0.15 | |||
| Merulinidae | 75.87 | 7.99 | 20 | 50.26 | 9.01 | 0.34 | |||
| Mussidae | 70.83 | 6.95 | 16 | 44.47 | 9.44 | 0.37 | |||
| Agariciidae | 65.28 | 7.63 | 25 | 37.19 | 7.86 | 0.43 | |||
| Poritidae | 54.46 | 8.67 | 23 | 33.26 | 8.88 | 0.39 | |||
| Pocilloporidae | 57.87 | 7.04 | 23 | 32.30 | 8.54 | 0.44 | |||
| Milleporidae | 48.67 | 12.74 | 5 | 31.56 | 19.58 | 0.35 | |||
| Siderastreidae | 46.16 | 14.75 | 7 | 15.48 | 10.98 | 0.66 | |||
| Meandrinidae | 27.98 | 15.50 | 6 | 10.84 | 7.00 | 0.61 | |||
| Psammocoridae | 43.90 | 16.66 | 3 | 1.13 | 1.13 | 0.97 | |||
| High | Beginning | Milleporidae | 59.75 | 23.24 | 4 | 50.00 | 28.87 | 0.16 | |
| Psammocoridae | 48.83 | 26.58 | 4 | 30.80 | 18.32 | 0.37 | |||
| Agariciidae | 48.86 | 18.72 | 7 | 5.61 | 5.61 | 0.89 | |||
| Pocilloporidae | 52.40 | 16.91 | 5 | 0.00 | 0.00 | 1 | |||
| Poritidae | 31.25 | 23.11 | 4 | 0.00 | 0.00 | 1 | |||
| Middle | Poritidae | 87.00 | 1.53 | 3 | 24.67 | 17.67 | 0.72 | ||
| Merulinidae | 83.87 | 10.87 | 8 | 24.62 | 7.07 | 0.71 | |||
| End | Acroporidae | 100.00 | 0.00 | 4 | 100.00 | 0.00 | 0 | ||
| Milleporidae | 100.00 | 0.00 | 8 | 100.00 | 0.00 | 0 | |||
| Pocilloporidae | 100.00 | 0.00 | 16 | 73.05 | 0.00 | 0.27 | |||
| Poritidae | 85.46 | 7.68 | 13 | 72.00 | 8.42 | 0.16 | |||
| Agariciidae | 32.67 | 23.9 | 3 | 27.00 | 4.93 | 0.17 | |||
| Psammocoridae | 100.00 | 0 | 4 | 0.50 | 0.29 | 0.99 | |||
| Thermal stress accumulation | Timing of observations | Genus | Susceptibility | ±SE | n | Mortality | ±SE | (S-M)/S | |
| Low | Beginning | Millepora | 37.51 | 8.75 | 13 | 19.26 | 10.00 | 0.49 | |
| Pavona | 34.71 | 11.62 | 11 | 18.19 | 11.55 | 0.48 | |||
| Stylophora | 74.50 | 11.76 | 9 | 14.22 | 11.16 | 0.81 | |||
| Acropora | 47.82 | 4.29 | 71 | 13.52 | 3.17 | 0.72 | |||
| Pocillopora | 38.11 | 4.93 | 54 | 11.48 | 3.43 | 0.70 | |||
| Dipsastraea | 48.36 | 22.18 | 3 | 9.47 | 5.14 | 0.80 | |||
| Galaxea | 37.36 | 9.19 | 10 | 8.80 | 6.66 | 0.76 | |||
| Hydnophora | 37.65 | 8.38 | 13 | 8.78 | 6.31 | 0.77 | |||
| Echinopora | 39.68 | 9.58 | 9 | 8.66 | 5.89 | 0.78 | |||
| Lobophyllia | 25.33 | 15.34 | 3 | 8.33 | 7.36 | 0.67 | |||
| Cyphastrea | 10.28 | 10.28 | 6 | 7.20 | 3.64 | 0.30 | |||
| Montipora | 35.90 | 6.39 | 37 | 6.49 | 2.76 | 0.82 | |||
| Favites | 37.38 | 7.10 | 8 | 6.08 | 3.19 | 0.84 | |||
| Platygyra | 33.26 | 7.94 | 11 | 6.12 | 3.15 | 0.82 | |||
| Orbicella | 76.49 | 5.48 | 16 | 5.85 | 3.70 | 0.92 | |||
| Porites | 32.29 | 3.61 | 80 | 4.68 | 1.60 | 0.86 | |||
| Astreopora | 41.09 | 8.79 | 7 | 4.64 | 3.29 | 0.89 | |||
| Seriatopora | 97.75 | 2.25 | 4 | 4.50 | 4.50 | 0.95 | |||
| Leptoria | 51.00 | 15.79 | 5 | 2.00 | 2.00 | 0.96 | |||
| Goniastrea | 30.20 | 8.30 | 10 | 1.50 | 0.97 | 0.95 | |||
| Montastraea | 27.55 | 4.79 | 12 | 0.83 | 0.83 | 0.97 | |||
| Siderastrea | 60.63 | 8.27 | 13 | 0.65 | 0.58 | 0.99 | |||
| Fungia | 60.90 | 10.87 | 10 | 0.50 | 0.50 | 0.99 | |||
| Favia | 40.84 | 7.03 | 13 | 0.46 | 0.39 | 0.99 | |||
| Diploria | 66.70 | 23.61 | 3 | 0.00 | 0.00 | 1 | |||
| Colpophyllia | 62.55 | 12.92 | 4 | 0.00 | 0.00 | 1 | |||
| Agaricia | 52.26 | 6.94 | 19 | 0.00 | 0.00 | 1 | |||
| Acanthastrea | 51.67 | 13.54 | 3 | 0.00 | 0.00 | 1 | |||
| Symphyllia | 34.80 | 17.00 | 5 | 0.00 | 0.00 | 1 | |||
| Goniopora | 34.71 | 10.39 | 11 | 0.00 | 0.00 | 1 | |||
| Pseudodiploria | 32.33 | 9.52 | 10 | 0.00 | 0.00 | 1 | |||
| Echinophyllia | 26.00 | 13.45 | 3 | 0.00 | 0.00 | 1 | |||
| Diploastrea | 18.53 | 10.83 | 7 | 0.00 | 0.00 | 1 | |||
| Madracis | 5.40 | 2.77 | 5 | 0.00 | 0.00 | 1 | |||
| Turbinaria | 0.75 | 0.75 | 4 | 0.00 | 0.00 | 1 | |||
| Middle | Millepora | 54.90 | 18.35 | 5 | 29.41 | 17.91 | 0.46 | ||
| Montipora | 53.33 | 7.67 | 4 | 16.83 | 16.49 | 0.68 | |||
| Acropora | 46.97 | 7.96 | 21 | 16.67 | 7.26 | 0.65 | |||
| Porites | 47.74 | 7.90 | 21 | 13.70 | 5.24 | 0.71 | |||
| Orbicella | 47.00 | 19.65 | 4 | 11.46 | 7.19 | 0.76 | |||
| Pseudodiploria | 61.10 | 10.54 | 7 | 5.02 | 1.59 | 0.92 | |||
| Coelastrea | 18.67 | 6.74 | 3 | 3.67 | 2.33 | 0.80 | |||
| Platygyra | 36.29 | 13.36 | 7 | 3.57 | 3.57 | 0.90 | |||
| Agaricia | 59.86 | 16.38 | 7 | 3.09 | 1.42 | 0.95 | |||
| Siderastrea | 47.59 | 9.87 | 10 | 2.46 | 0.79 | 0.95 | |||
| Diploastrea | 17.68 | 6.15 | 4 | 2.43 | 2.43 | 0.86 | |||
| Montastraea | 45.54 | 17.93 | 5 | 2.86 | 2.86 | 0.94 | |||
| Diploria | 23.40 | 6.58 | 5 | 2.20 | 2.20 | 0.91 | |||
| Acanthastrea | 58.67 | 27.34 | 3 | 2.00 | 2.00 | 0.97 | |||
| Coeloseris | 33.00 | 15.71 | 4 | 2.00 | 2.00 | 0.94 | |||
| Colpophyllia | 33.00 | 15.71 | 4 | 2.00 | 2.00 | 0.94 | |||
| Favia | 48.64 | 13.69 | 7 | 1.55 | 1.04 | 0.97 | |||
| Pocillopora | 37.80 | 12.73 | 5 | 1.20 | 0.58 | 0.97 | |||
| Galaxea | 23.33 | 21.36 | 3 | 0.67 | 0.67 | 0.97 | |||
| Echinopora | 17.50 | 16.19 | 4 | 0.50 | 0.50 | 0.97 | |||
| Goniopora | 52.00 | 27.15 | 3 | 0.00 | 0.00 | 1 | |||
| Astreopora | 49.00 | 10.69 | 3 | 0.00 | 0.00 | 1 | |||
| Echinophyllia | 25.00 | 15.54 | 4 | 0.00 | 0.00 | 1 | |||
| End | Pavona | 56.68 | 21.35 | 5 | 35.73 | 20.89 | 0.37 | ||
| Hydnophora | 32.92 | 16.46 | 3 | 32.68 | 16.35 | 0.01 | |||
| Agaricia | 45.50 | 19.17 | 3 | 30.30 | 10.33 | 0.33 | |||
| Acropora | 56.08 | 7.09 | 31 | 26.06 | 6.62 | 0.54 | |||
| Porites | 36.93 | 8.65 | 18 | 19.55 | 7.58 | 0.47 | |||
| Orbicella | 65.89 | 10.33 | 7 | 18.96 | 2.51 | 0.71 | |||
| Pseudodiploria | 36.10 | 15.48 | 3 | 17.53 | 7.87 | 0.51 | |||
| Galaxea | 32.64 | 11.05 | 7 | 16.06 | 12.45 | 0.51 | |||
| Echinopora | 13.59 | 13.59 | 4 | 13.34 | 13.34 | 0.02 | |||
| Platygyra | 52.32 | 9.81 | 7 | 13.17 | 8.50 | 0.75 | |||
| Pocillopora | 46.90 | 9.58 | 14 | 12.92 | 8.40 | 0.72 | |||
| Millepora | 9.40 | 9.40 | 3 | 8.00 | 8.00 | 0.15 | |||
| Favites | 23.59 | 3.20 | 4 | 4.96 | 3.28 | 0.79 | |||
| Montipora | 21.71 | 6.64 | 7 | 2.14 | 1.83 | 0.90 | |||
| Astrea | 64.00 | 20.22 | 5 | 0.00 | 0.00 | 1 | |||
| Goniastrea | 58.33 | 22.05 | 3 | 0.00 | 0.00 | 1 | |||
| Acanthastrea | 35.67 | 5.93 | 3 | 0.00 | 0.00 | 1 | |||
| Astreopora | 22.33 | 10.71 | 3 | 0.00 | 0.00 | 1 | |||
| Favia | 21.67 | 5.84 | 3 | 0.00 | 0.00 | 1 | |||
| Fungia | 16.64 | 8.24 | 4 | 0.00 | 0.00 | 1 | |||
| Echinophyllia | 9.33 | 4.70 | 3 | 0.00 | 0.00 | 1 | |||
| Goniopora | 1.67 | 0.88 | 3 | 0.00 | 0.00 | 1 | |||
| Mussismilia | 0.00 | 0.00 | 3 | 0.00 | 0.00 | *** | |||
| Medium | Beginning | Acropora | 59.88 | 12.77 | 13 | 24.73 | 11.82 | 0.59 | |
| Eusmilia | 38.33 | 14.24 | 3 | 16.33 | 16.33 | 0.57 | |||
| Montipora | 52.75 | 18.07 | 4 | 13.75 | 5.30 | 0.74 | |||
| Millepora | 37.31 | 13.49 | 8 | 13.06 | 12.08 | 0.65 | |||
| Favites | 86.25 | 13.75 | 4 | 12.50 | 12.50 | 0.86 | |||
| Madracis | 7.88 | 3.23 | 8 | 7.81 | 5.80 | 0.01 | |||
| Stephanocoenia | 51.00 | 28.88 | 3 | 6.00 | 6.00 | 0.88 | |||
| Colpophyllia | 34.25 | 5.53 | 4 | 4.50 | 2.87 | 0.87 | |||
| Orbicella | 49.11 | 8.64 | 9 | 4.00 | 2.66 | 0.92 | |||
| Porites | 35.95 | 9.87 | 11 | 3.41 | 1.69 | 0.91 | |||
| Goniastrea | 86.20 | 8.57 | 5 | 3.20 | 2.96 | 0.96 | |||
| Coeloseris | 100.00 | 0.00 | 4 | 2.50 | 2.50 | 0.97 | |||
| Platygyra | 83.75 | 11.43 | 4 | 2.50 | 2.50 | 0.97 | |||
| Coelastrea | 82.50 | 17.50 | 4 | 2.50 | 2.50 | 0.97 | |||
| Siderastrea | 63.80 | 8.18 | 10 | 1.60 | 0.91 | 0.97 | |||
| Meandrina | 46.00 | 6.16 | 4 | 1.50 | 1.19 | 0.97 | |||
| Pseudodiploria | 46.71 | 6.19 | 7 | 1.29 | 0.64 | 0.97 | |||
| Montastraea | 65.17 | 13.59 | 6 | 1.00 | 1.00 | 0.98 | |||
| Agaricia | 23.75 | 7.07 | 8 | 0.00 | 0.00 | 1 | |||
| Mycetophyllia | 4.00 | 4.00 | 5 | 0.00 | 0.00 | 1 | |||
| Middle | Montipora | 45.33 | 24.85 | 3 | 36.00 | 22.07 | 0.21 | ||
| Porites | 39.25 | 5.44 | 4 | 24.75 | 13.46 | 0.37 | |||
| Acropora | 19.83 | 7.61 | 6 | 14.67 | 8.63 | 0.26 | |||
| End | Diploastrea | 96.67 | 3.33 | 3 | 96.67 | 3.33 | 0 | ||
| Acropora | 86.72 | 6.70 | 22 | 79.33 | 7.45 | 0.09 | |||
| Montipora | 93.50 | 6.50 | 4 | 77.50 | 22.50 | 0.17 | |||
| Stephanocoenia | 56.25 | 23.66 | 3 | 47.92 | 28.94 | 0.15 | |||
| Agaricia | 86.84 | 6.14 | 9 | 37.26 | 10.94 | 0.57 | |||
| Favites | 71.56 | 14.07 | 3 | 36.00 | 31.13 | 0.50 | |||
| Millepora | 48.67 | 12.74 | 5 | 31.56 | 19.58 | 0.35 | |||
| Porites | 52.48 | 8.84 | 22 | 30.32 | 8.77 | 0.42 | |||
| Pavona | 45.43 | 11.34 | 10 | 28.63 | 13.82 | 0.37 | |||
| Pocillopora | 57.44 | 7.90 | 18 | 27.72 | 9.17 | 0.52 | |||
| Pseudodiploria | 61.43 | 7.69 | 3 | 26.44 | 15.75 | 0.57 | |||
| Orbicella | 64.73 | 13.91 | 6 | 18.52 | 6.55 | 0.71 | |||
| Siderastrea | 46.64 | 12.78 | 8 | 16.67 | 9.58 | 0.64 | |||
| Psammocora | 43.90 | 16.66 | 3 | 1.13 | 1.13 | 0.97 | |||
| High | Beginning | Millepora | 59.75 | 23.24 | 4 | 50.00 | 28.87 | 0.16 | |
| Psammocora | 48.83 | 26.58 | 4 | 30.80 | 18.32 | 0.92 | |||
| Pavona | 59.40 | 24.26 | 5 | 7.86 | 7.86 | 0.87 | |||
| Pocillopora | 52.40 | 16.91 | 5 | 0.00 | 0.00 | 1 | |||
| Porites | 31.25 | 23.11 | 4 | 0.00 | 0.00 | 1 | |||
| End | Millepora | 100.00 | 0.00 | 8 | 100.00 | 0.00 | 0 | ||
| Acropora | 100.00 | 0.00 | 3 | 100.00 | 0.00 | 0 | |||
| Porites | 85.46 | 7.68 | 13 | 72.00 | 12.30 | 0.16 | |||
| Pocillopora | 100.00 | 0.00 | 14 | 69.20 | 11.94 | 0.31 | |||
| Psammocora | 100.00 | 0.00 | 4 | 0.50 | 0.29 | 0.99 | |||
| |||||||||
| |||||||||
| Thermal stress accumulation | Timing of observations | Species | Susceptibility | ±SE | n | Mortality | ±SE | (S-M)/S | |
| Low | Beginning | Diploastrea heliopora | 43.88 | 22.98 | 5 | 41.94 | 23.77 | 0.04 | |
| Pocillopora verrucosa | 58.98 | 13.52 | 6 | 21.35 | 13.93 | 0.64 | |||
| Orbicella faveolata | 97.67 | 2.33 | 3 | 20.00 | 20.00 | 0.80 | |||
| Platygyra daedalea | 47.36 | 8.48 | 3 | 17.44 | 7.85 | 0.63 | |||
| Pocillopora damicornis | 28.24 | 11.57 | 11 | 11.87 | 5.75 | 0.58 | |||
| Hydnophora exesa | 92.50 | 7.50 | 4 | 3.75 | 3.75 | 0.96 | |||
| Acropora hyacinthus | 24.13 | 6.71 | 7 | 3.57 | 2.83 | 0.85 | |||
| Orbicella annularis | 75.83 | 4.57 | 12 | 2.80 | 1.02 | 0.96 | |||
| Dipsastraea favus | 23.40 | 6.58 | 5 | 2.20 | 2.20 | 0.91 | |||
| Montipora digitata | 51.50 | 26.86 | 4 | 1.65 | 1.46 | 0.97 | |||
| Siderastrea siderea | 60.90 | 8.72 | 11 | 0.77 | 0.68 | 0.99 | |||
| Porites astreoides | 33.79 | 7.88 | 11 | 0.18 | 0.18 | 0.99 | |||
| Diploria labyrinthiformis | 66.70 | 23.61 | 3 | 0.00 | 0.00 | 1 | |||
| Colpophyllia natans | 62.55 | 12.92 | 4 | 0.00 | 0.00 | 1 | |||
| Agaricia agaricites | 58.63 | 10.65 | 8 | 0.00 | 0.00 | 1 | |||
| Porites porites | 40.52 | 11.31 | 7 | 0.00 | 0.00 | 1 | |||
| Agaricia tenuifolia | 38.68 | 12.75 | 5 | 0.00 | 0.00 | 1 | |||
| Favia fragum | 34.82 | 7.69 | 6 | 0.00 | 0.00 | 1 | |||
| Pseudodiploria strigosa | 32.22 | 10.65 | 9 | 0.00 | 0.00 | 1 | |||
| Montastraea cavernosa | 28.37 | 4.35 | 7 | 0.00 | 0.00 | 1 | |||
| Meandrina meandrites | 25.70 | 14.35 | 4 | 0.00 | 0.00 | 1 | |||
| Acropora palmata | 8.93 | 7.03 | 3 | 0.00 | 0.00 | 1 | |||
| Madracis mirabilis | 5.33 | 3.93 | 3 | 0.00 | 0.00 | 1 | |||
| Montipora flabellata | 0.65 | 0.38 | 4 | 0.00 | 0.00 | 1 | |||
| Porites lobata | 0.27 | 0.19 | 11 | 0.00 | 0.00 | 1 | |||
| Montipora capita | 0.00 | 0.00 | 5 | 0.00 | 0.00 | *** | |||
| Montipora patula | 0.00 | 0.00 | 5 | 0.00 | 0.00 | *** | |||
| Pocillopora ligulata | 0.00 | 0.00 | 7 | 0.00 | 0.00 | *** | |||
| Pocillopora meandrina | 0.00 | 0.00 | 6 | 0.00 | 0.00 | *** | |||
| Porites compressa | 0.00 | 0.00 | 7 | 0.00 | 0.00 | *** | |||
| Porites evermanni | 0.00 | 0.00 | 6 | 0.00 | 0.00 | *** | |||
| Middle | Porites porites | 47.45 | 13.67 | 6 | 11.91 | 5.29 | 0.75 | ||
| Orbicella annularis | 47.00 | 19.65 | 4 | 11.46 | 7.19 | 0.76 | |||
| Pseudodiploria strigosa | 57.30 | 14.85 | 5 | 4.62 | 2.22 | 0.92 | |||
| Coelastrea aspera | 18.67 | 6.74 | 3 | 3.67 | 2.33 | 0.80 | |||
| Agaricia agaricites | 88.91 | 6.68 | 3 | 3.62 | 2.16 | 0.96 | |||
| Montastraea cavernosa | 40.25 | 22.12 | 4 | 3.57 | 3.57 | 0.91 | |||
| Acropora palmata | 41.48 | 14.56 | 4 | 3.48 | 2.63 | 0.92 | |||
| Siderastrea radians | 43.60 | 14.39 | 4 | 3.14 | 1.10 | 0.93 | |||
| Favia fragum | 50.88 | 19.00 | 4 | 2.71 | 1.64 | 0.95 | |||
| Porites astreoides | 42.44 | 16.36 | 7 | 2.37 | 1.28 | 0.94 | |||
| Siderastrea siderea | 50.25 | 14.35 | 6 | 2.00 | 1.14 | 0.96 | |||
| Acropora cervicornis | 16.38 | 11.52 | 4 | 0.00 | 0.00 | 1 | |||
| End | Acropora digitifera | 80.33 | 19.67 | 3 | 74.33 | 25.67 | 0.07 | ||
| Porites nigrescens | 85.21 | 10.53 | 4 | 60.69 | 19.28 | 0.29 | |||
| Galaxea astreata | 85.90 | 4.90 | 3 | 54.08 | 25.27 | 0.37 | |||
| Acropora aspera | 94.17 | 5.83 | 6 | 50.00 | 22.36 | 0.47 | |||
| Acropora eurystoma | 79.82 | 16.34 | 3 | 49.79 | 22.57 | 0.38 | |||
| Acropora hyacinthus | 64.56 | 14.89 | 9 | 45.89 | 14.70 | 0.29 | |||
| Stylophora pistillata | 66.67 | 33.33 | 3 | 42.67 | 29.78 | 0.36 | |||
| Acropora valida | 66.00 | 22.68 | 3 | 38.00 | 31.26 | 0.42 | |||
| Porites panamensis | 58.80 | 19.17 | 6 | 33.45 | 21.05 | 0.43 | |||
| Isophyllia sinuosa | 78.57 | 14.87 | 3 | 28.38 | 16.50 | 0.64 | |||
| Pocillopora damicornis | 52.75 | 8.72 | 24 | 27.09 | 8.08 | 0.49 | |||
| Montipora aequituberculata | 32.50 | 22.87 | 4 | 26.25 | 24.61 | 0.19 | |||
| Psammocora stellata | 59.06 | 22.99 | 5 | 24.84 | 15.39 | 0.58 | |||
| Pavona gigantea | 34.58 | 20.24 | 4 | 24.75 | 22.33 | 0.28 | |||
| Pocillopora elegans | 67.81 | 12.37 | 9 | 24.30 | 14.33 | 0.64 | |||
| Pocillopora verrucosa | 68.03 | 10.06 | 9 | 23.50 | 12.10 | 0.65 | |||
| Acropora millepora | 77.33 | 22.67 | 3 | 20.00 | 6.43 | 0.74 | |||
| Madracis decactis | 19.20 | 11.77 | 5 | 19.20 | 11.77 | 0 | |||
| Acropora gemmifera | 75.00 | 14.60 | 4 | 18.75 | 18.75 | 0.75 | |||
| Agaricia tenuifolia | 41.97 | 9.70 | 10 | 17.59 | 10.37 | 0.58 | |||
| Favia pentagona | 40.89 | 13.49 | 3 | 17.50 | 3.09 | 0.57 | |||
| Favites abdita | 74.17 | 17.34 | 6 | 16.67 | 10.54 | 0.78 | |||
| Orbicella faveolata | 70.77 | 9.64 | 9 | 15.83 | 6.31 | 0.78 | |||
| Platygyra daedalea | 68.02 | 6.75 | 13 | 15.73 | 5.61 | 0.77 | |||
| Diploria labyrinthiformis | 47.82 | 10.64 | 13 | 15.44 | 6.89 | 0.68 | |||
| Coelastrea aspera | 60.75 | 15.06 | 8 | 15.13 | 12.21 | 0.75 | |||
| Porites porites | 54.43 | 7.39 | 20 | 14.00 | 4.31 | 0.74 | |||
| Colpophyllia natans | 50.63 | 7.87 | 13 | 13.92 | 5.05 | 0.72 | |||
| Coeloseris mayeri | 83.33 | 16.67 | 6 | 13.33 | 11.45 | 0.85 | |||
| Gardineroseris planulata | 23.88 | 13.21 | 6 | 13.00 | 10.30 | 0.46 | |||
| Dipsastraea favus | 43.77 | 16.34 | 4 | 12.68 | 4.85 | 0.71 | |||
| Eusmilia fastigiata | 26.25 | 15.19 | 4 | 12.25 | 12.25 | 0.53 | |||
| Acropora cytherea | 21.00 | 21.00 | 3 | 12.00 | 12.00 | 0.43 | |||
| Pavona varians | 40.78 | 13.65 | 8 | 11.40 | 10.09 | 0.72 | |||
| Acropora palmata | 36.98 | 9.50 | 12 | 10.03 | 5.33 | 0.73 | |||
| Pseudodiploria clivosa | 48.69 | 7.17 | 7 | 11.35 | 7.72 | 0.77 | |||
| Stephanocoenia intersepta | 41.13 | 14.30 | 6 | 10.29 | 7.31 | 0.75 | |||
| Orbicella franksi | 31.03 | 21.41 | 3 | 9.87 | 9.87 | 0.68 | |||
| Dichoenia stokesii | 22.47 | 8.05 | 5 | 9.82 | 7.38 | 0.56 | |||
| Orbicella annularis | 66.37 | 4.63 | 30 | 9.49 | 2.26 | 0.86 | |||
| Goniastrea retiformis | 78.33 | 16.41 | 6 | 9.17 | 6.64 | 0.88 | |||
| Acropora cervicornis | 29.57 | 8.62 | 15 | 8.89 | 5.98 | 0.70 | |||
| Agaricia agaricites | 66.21 | 8.21 | 14 | 8.04 | 4.40 | 0.88 | |||
| Galaxea fascicularis | 25.07 | 6.17 | 4 | 7.55 | 4.21 | 0.70 | |||
| Pavona cactus | 45.67 | 17.93 | 6 | 7.20 | 6.45 | 0.84 | |||
| Siderastrea radians | 46.81 | 11.19 | 7 | 6.56 | 4.54 | 0.86 | |||
| Porites lutea | 31.82 | 8.15 | 8 | 6.48 | 4.17 | 0.80 | |||
| Porites furcata | 67.17 | 8.86 | 4 | 5.92 | 4.05 | 0.91 | |||
| Siderastrea siderea | 54.91 | 5.35 | 28 | 4.89 | 2.72 | 0.91 | |||
| Pseudodiploria strigosa | 46.66 | 6.47 | 21 | 4.60 | 1.79 | 0.90 | |||
| Montipora digitata | 43.80 | 22.18 | 5 | 3.91 | 2.53 | 0.91 | |||
| Porites lobata | 21.92 | 7.09 | 23 | 3.55 | 3.04 | 0.84 | |||
| Montastraea cavernosa | 31.56 | 6.33 | 17 | 3.36 | 1.85 | 0.89 | |||
| Meandrina meandrites | 32.67 | 10.00 | 10 | 3.29 | 2.67 | 0.90 | |||
| Madracis mirabilis | 7.67 | 3.04 | 6 | 2.75 | 2.65 | 0.64 | |||
| Pocillopora eydouxi | 21.32 | 7.88 | 6 | 2.33 | 2.33 | 0.89 | |||
| Porites astreoides | 40.79 | 6.67 | 25 | 2.29 | 0.81 | 0.94 | |||
| Favia fragum | 43.55 | 8.15 | 11 | 0.98 | 0.68 | 0.98 | |||
| Pocillopora meandrina | 14.85 | 8.83 | 9 | 0.23 | 0.23 | 0.98 | |||
| Astrea curta | 64.00 | 20.22 | 5 | 0.00 | 0.00 | 1 | |||
| Manicina areolata | 49.03 | 4.96 | 3 | 0.00 | 0.00 | 1 | |||
| Dendrogyra cylindrus | 30.00 | 20.00 | 5 | 0.00 | 0.00 | 1 | |||
| Montipora flabellata | 0.65 | 0.38 | 4 | 0.00 | 0.00 | 1 | |||
| Montastraea capitata | 0.00 | 0.00 | 5 | 0.00 | 0.00 | *** | |||
| Montipora patula | 0.00 | 0.00 | 5 | 0.00 | 0.00 | *** | |||
| Pocillopora ligulata | 0.00 | 0.00 | 7 | 0.00 | 0.00 | *** | |||
| Porites compressa | 0.00 | 0.00 | 7 | 0.00 | 0.00 | *** | |||
| Porites evermanni | 0.00 | 0.00 | 6 | 0.00 | 0.00 | *** | |||
| Medium | Beginning | Acropora aspera | 100.00 | 0.00 | 4 | 50.00 | 28.87 | 0.5 | |
| Isophyllia. sinuosa | 56.67 | 23.33 | 3 | 9.33 | 9.33 | 0.84 | |||
| Madracis decactis | 15.33 | 15.33 | 3 | 7.67 | 7.67 | 0.50 | |||
| Madracis mirabilis | 13.33 | 1.76 | 3 | 5.50 | 5.25 | 0.59 | |||
| Porites astreoides | 39.75 | 15.62 | 4 | 5.25 | 4.03 | 0.87 | |||
| Porites porites | 57.67 | 23.85 | 3 | 4.67 | 3.28 | 0.92 | |||
| Colpophyllia natans | 34.25 | 5.53 | 4 | 4.50 | 2.87 | 0.87 | |||
| Dichocoenia stokesii | 29.33 | 12.13 | 3 | 3.67 | 3.67 | 0.87 | |||
| Coeloseris mayeri | 100.00 | 0.00 | 4 | 2.50 | 2.50 | 0.97 | |||
| Platygyra daedalea | 83.75 | 11.43 | 4 | 2.50 | 2.50 | 0.97 | |||
| Meandrina meandrites | 44.67 | 8.51 | 3 | 2.00 | 1.53 | 0.96 | |||
| Siderastrea siderea | 64.22 | 9.14 | 9 | 1.78 | 1.00 | 0.97 | |||
| Pseudodiploria strigosa | 51.50 | 7.73 | 4 | 1.50 | 0.96 | 0.97 | |||
| Orbicella annularis | 44.86 | 9.96 | 7 | 1.71 | 1.55 | 0.96 | |||
| Montastraea cavernosa | 65.17 | 13.59 | 6 | 1.00 | 1.00 | 0.98 | |||
| Pseudodiploria clivosa | 40.33 | 10.65 | 3 | 1.00 | 1.00 | 0.98 | |||
| Acropora palmata | 17.50 | 9.19 | 4 | 0.63 | 0.47 | 0.96 | |||
| End | Diploastrea heliopora | 96.67 | 3.33 | 3 | 96.67 | 3.33 | 0 | ||
| Pocillopora damicornis | 51.48 | 19.00 | 4 | 31.25 | 23.02 | 0.39 | |||
| Porites lutea | 30.33 | 30.33 | 3 | 30.33 | 30.33 | 0 | |||
| Orbicella annularis | 78.09 | 8.03 | 4 | 26.03 | 6.99 | 0.67 | |||
| Agaricia agaricites | 88.75 | 5.15 | 4 | 25.00 | 14.43 | 0.72 | |||
| Siderastrea siderea | 69.72 | 18.30 | 3 | 25.00 | 25.00 | 0.64 | |||
| Porites lobata | 51.30 | 17.14 | 4 | 17.50 | 17.50 | 0.66 | |||
| Pocillopora elegans | 35.77 | 14.53 | 3 | 4.90 | 1.27 | 0.86 | |||
| Pavona cactus | 24.67 | 7.45 | 3 | 1.30 | 1.30 | 0.95 | |||
| High | Beginning | Psammocora stellata | 48.83 | 26.58 | 4 | 30.80 | 18.32 | 0.37 | |
| Pocillopora damicornis | 53.00 | 24.95 | 3 | 0.00 | 0.00 | 1 | |||
| End | Pocillopora damicornis | 100.00 | 0.00 | 6 | 67.33 | 20.66 | 0.33 | ||
| Pocillopora elegans | 100.00 | 0.00 | 4 | 51.00 | 28.29 | 0.49 | |||
| A) Depth (in descending order) | ||||||||
| Thermal stress accumulation | Timing of observations | Depth (metres) | Susceptibility | ±SE | n | Mortality | ±SE | (S-M)/S |
| Low | Beginning | 0-3m | 47.00 | 5.84 | 26 | 0.31 | 0.22 | 0.99 |
| 3-6m | 69.80 | 8.17 | 19 | 31.45 | 7.28 | 0.55 | ||
| 6-9m | 43.31 | 5.43 | 35 | 1.69 | 1.43 | 0.96 | ||
| 9-12m | 53.67 | 3.33 | 79 | 10.53 | 2.42 | 0.80 | ||
| 12-15m | 43.50 | 6.60 | 19 | 0.02 | 0.02 | 1 | ||
| >15m | 43.40 | 6.52 | 28 | 2.34 | 2.34 | 0.95 | ||
| Middle | 0-3m | 26.50 | 7.31 | 26 | 18 | 7.06 | 0.32 | |
| 6-9m | 58.07 | 3.58 | 69 | 1.36 | 0.44 | 0.98 | ||
| 9-12m | 42.38 | 5.07 | 34 | 4.08 | 0.78 | 0.90 | ||
| >15m | 27.08 | 5.76 | 26 | 3.34 | 1.56 | 0.88 | ||
| End | 0-3m | 6.93 | 1.53 | 14 | 1.36 | 0.63 | 0.80 | |
| 3-6m | 42.40 | 5.45 | 54 | 9.92 | 3.38 | 0.77 | ||
| 6-9m | 30.98 | 2.92 | 65 | 2.48 | 1.17 | 0.92 | ||
| 9-12m | 18.72 | 7.74 | 18 | 10.56 | 5.55 | 0.70 | ||
| 12-15m | 45.92 | 8.92 | 13 | 21.87 | 7.74 | 0.52 | ||
| Medium | Beginning | 3-6m | 20.33 | 3.53 | 3 | 0.00 | 0.00 | 1 |
| 6-9m | 74.80 | 8.84 | 12 | 35.40 | 10.93 | 0.53 | ||
| 9-12m | 57.91 | 6.36 | 12 | 5.83 | 2.50 | 0.90 | ||
| 15-18m | 45.47 | 4.16 | 43 | 7.33 | 1.71 | 0.84 | ||
| >18m | 46.67 | 2.91 | 3 | 0.00 | 0.00 | 1 | ||
| End | 0-3m | 72.50 | 15.88 | 4 | 48.75 | 20.93 | 0.33 | |
| 3-6m | 55.54 | 10.38 | 7 | 13.19 | 9.61 | 0.76 | ||
| 9-12m | 47.32 | 4.73 | 49 | 16.25 | 3.61 | 0.66 | ||
| 12-15m | 95.00 | 2.89 | 4 | 95.00 | 3.54 | 0 | ||
| >15m | 48.87 | 5.82 | 34 | 29.89 | 5.37 | 0.39 | ||
| High | Beginning | 9-12m | 68.38 | 11.24 | 13 | 19.29 | 10.66 | 0.72 |
| 12-15m | 96.43 | 3.57 | 3 | 55.93 | 9.56 | 0.42 | ||
| >15m | 9.44 | 3.13 | 9 | 0.00 | 0.00 | 1 | ||
| End | 3-6m | 100.00 | 0.00 | 20 | 89.44 | 6.73 | 0.11 | |
| 9-12m | 78.75 | 13.74 | 8 | 50.76 | 16.56 | 0.36 | ||
| 12-15m | 72.50 | 14.11 | 8 | 1.38 | 0.60 | 0.98 | ||
| B) Habitat | ||||||||
| Thermal stress accumulation | Timing of observations | Habitat | Susceptibility | ±SE | n | Mortality | ±SE | (S-M)/S |
| Low | Beginning | Lagoon | 44.50 | 8.78 | 15 | 26.17 | 7.62 | 0.41 |
| Fringing reef | 55.30 | 4.09 | 51 | 12.99 | 3.13 | 0.77 | ||
| Subtropical | 45.36 | 10.01 | 14 | 4.21 | 3.55 | 0.91 | ||
| Fore reef | 22.51 | 4.93 | 40 | 2.04 | 1.51 | 0.91 | ||
| Back reef | 23.58 | 4.34 | 39 | 0.26 | 0.15 | 0.99 | ||
| Reef slope | 59.44 | 4.69 | 36 | 0.03 | 0.02 | 1 | ||
| Patch reef | 29.00 | 5.51 | 39 | 0.00 | 0.00 | 1 | ||
| Middle | Reef flat | 52.91 | 13.81 | 11 | 40.09 | 14.47 | 0.24 | |
| Fringing reef | 68.52 | 2.34 | 34 | 5.17 | 0.86 | 0.92 | ||
| Patch | 28.13 | 5.90 | 25 | 3.48 | 1.62 | 0.88 | ||
| Subtropical | 54.07 | 4.38 | 54 | 0.30 | 0.24 | 0.99 | ||
| End | Fringing | 54.24 | 5.90 | 40 | 21.85 | 4.77 | 0.60 | |
| Subtropical | 30.98 | 2.92 | 65 | 2.48 | 1.17 | 0.92 | ||
| Reef Crest | 30.26 | 8.51 | 14 | 0.00 | 0.00 | 1 | ||
| Reef Slope | 23.02 | 8.77 | 14 | 0.00 | 0.00 | 1 | ||
| Medium | End | Reef flat | 72.50 | 15.88 | 4 | 48.75 | 20.93 | 0.33 |
| Lagoon | 76.00 | 7.08 | 20 | 39.25 | 7.79 | 0.48 | ||
| Patch reef | 49.44 | 5.08 | 40 | 25.75 | 4.92 | 0.48 | ||
| Gulf | 43.82 | 5.41 | 19 | 12.71 | 5.92 | 0.71 | ||
| Fringing reef | 24.19 | 7.53 | 14 | 4.29 | 3.62 | 0.83 | ||
| High | End | Reef flat | 100.00 | 0.00 | 16 | 100.00 | 0.00 | 0 |
| Gulf | 90.00 | 6.00 | 4 | 80.40 | 3.04 | 0.11 | ||
| Bay | 75.71 | 15.47 | 7 | 47.14 | 18.66 | 0.38 | ||
| Reef slope | 26.67 | 13.33 | 3 | 0.00 | 0.00 | 1 | ||
| C) Anthropogenic input | ||||||||
| Thermal stress accumulation | Timing of observations | Pollution | Susceptibility | ±SE | n | Mortality | ±SE | (S-M)/S |
| Low | Beginning | No | 57.26 | 8.15 | 22 | 11.14 | 4.02 | 0.81 |
| Yes | 46.09 | 6.05 | 34 | 5.26 | 2.98 | 0.89 | ||
| Middle | Yes | 28.13 | 5.90 | 25 | 3.48 | 1.62 | 0.88 | |
| No | 54.07 | 4.38 | 54 | 0.30 | 0.24 | 0.99 | ||
| End | Yes | 45.92 | 8.92 | 13 | 21.87 | 7.74 | 0.52 | |
| No | 30.98 | 2.92 | 65 | 2.48 | 1.17 | 0.92 | ||
| D) Shelf position | ||||||||
| Thermal stress accumulation | Timing of observations | Shelf position | Susceptibility | ±SE | n | Mortality | ±SE | (S-M)/S |
| Low | Beginning | Middle | 60.25 | 14.56 | 6 | 54.75 | 22.35 | 0.09 |
| Lagoon | 62.29 | 14.19 | 7 | 25.57 | 9.67 | 0.59 | ||
| Gulf | 78.09 | 11.19 | 8 | 23.25 | 8.22 | 0.70 | ||
| Inner | 55.94 | 4.22 | 47 | 10.56 | 1.54 | 0.81 | ||
| Fringing | 38.79 | 3.44 | 64 | 3.34 | 0.42 | 0.91 | ||
| Outer | 59.44 | 4.69 | 36 | 0.03 | 0.00 | 1 | ||
| High Latitude | 45.17 | 6.98 | 14 | 0.00 | 0.00 | 1 | ||
| ||||||||||
| ||||||||||
| ||||||||||
| Factor | SS (Type III) | df | n | MS | F | p | ||||
| Relief from Weather | 25.543 | 2 | 180 | 0.094 | 41.271 | <0.001*** | ||||
| Bleaching Occurrence | 91.851 | 3 | 618 | 0.131 | 28.900 | <0.001*** | ||||
| Location | 90.222 | 18 | 607 | 0.086 | 25.651 | <0.001*** | ||||
| Species | 25.534 | 18 | 146 | 0.058 | 15.329 | <0.001*** | ||||
| Generalization | 44.546 | 1 | 257 | 0.166 | 13.095 | <0.001*** | ||||
| Habitat | 37.089 | 6 | 233 | 0.128 | 10.502 | <0.001*** | ||||
| Ocean_subregion | 90.669 | 6 | 614 | 0.136 | 10.100 | <0.001*** | ||||
| Months of Observations | 91.168 | 3 | 616 | 0.144 | 7.344 | <0.001*** | ||||
| Transmission of Algae to Offspring | 35.151 | 1 | 192 | 0.759 | 4.679 | <0.05* | ||||
| Level of Colony Integrations | 58.041 | 2 | 359 | 0.959 | 3.478 | <0.05* | ||||
| Shelf Position | 20.798 | 7 | 195 | 0.142 | 3.357 | <0.01** | ||||
| Dominant Topography | 8.958 | 5 | 53 | 0.139 | 3.326 | <0.05* | ||||
| Genus | 73.722 | 27 | 479 | 0.138 | 3.061 | <0.001*** | ||||
| Depth | 24.165 | 5 | 205 | 0.114 | 2.389 | <0.05* | ||||
| Degree Heating Index | 91.851 | 1 | 618 | 0.148 | 1.995 | >0.05 | ||||
| Family | 83.758 | 14 | 562 | 0.147 | 1.628 | >0.05 | ||||
| Anthropogenic Input | 9.641 | 2 | 55 | 0.173 | 1.321 | >0.05 | ||||
| Sex | 34.428 | 1 | 218 | 0.158 | 0.407 | >0.05 | ||||
| Complexity | 83.034 | 1 | 550 | 0.151 | 0.139 | >0.05 | ||||
| Reproductive Mode | 34.428 | 1 | 218 | 0.159 | 0.088 | >0.05 | ||||
| ||||||||||
| Factor | n | Perason’s Correlation Coefficient | p | |||||||
| Habitat | 234 | 0.235 | <0.001*** | |||||||
| Generalization | 258 | -0.221 | <0.001*** | |||||||
| Relief from Weather | 181 | 0.199 | <0.01** | |||||||
| Transmission of Algae to Offspring | 192 | -0.155 | <0.05* | |||||||
| Bleaching Occurrence | 619 | -0.087 | <0.05* | |||||||
| ||||||||||
| Factor | SS (Type III) | df | n | MS | F | p | ||||
| Ocean_subregion | 39.813 | 6 | 614 | 0.055 | 19.112 | <0.001*** | ||||
| Habitat | 10.39 | 6 | 233 | 0.032 | 18.720 | <0.001*** | ||||
| Shelf Position | 12.740 | 7 | 195 | 0.055 | 17.715 | <0.001*** | ||||
| Depth | 13.693 | 5 | 205 | 0.049 | 15.907 | <0.001*** | ||||
| Level of Colony Integration | 24.579 | 2 | 359 | 0.064 | 14.353 | <0.001*** | ||||
| Sex | 12.527 | 1 | 218 | 218 | 12.023 | <0.01** | ||||
| Anthropogenic Input | 3.617 | 2 | 55 | 0.047 | 11.728 | <0.001*** | ||||
| Months of Observations | 39.832 | 3 | 616 | 0.062 | 11.052 | <0.001*** | ||||
| Dominant Topography | 4.482 | 5 | 53 | 0.050 | 8.264 | <0.001*** | ||||
| Reproductive Mode | 12.527 | 1 | 218 | 0.056 | 7.221 | <0.01** | ||||
| Transmission of Algae to Offspring | 11.294 | 1 | 191 | 0.160 | 4.679 | <0.05* | ||||
| Species | 3.168 | 18 | 146 | 0.018 | 2.778 | <0.001*** | ||||
| Family | 34.557 | 14 | 562 | 0.059 | 2.632 | <0.01** | ||||
| Complexity | 32.168 | 1 | 550 | 0.058 | 2.243 | >0.05 | ||||
| Genus | 32.809 | 27 | 479 | 0.066 | 1.553 | <0.05* | ||||
| Degree Heating Index | 39.860 | 1 | 618 | 0.065 | 0.517 | >0.05 | ||||
| Generalization | 13.024 | 1 | 257 | 0.051 | 0.397 | >0.05 | ||||
| ||||||||||
| Factor | n | Pearson’s Correlation Coefficient | p | |||||||
| Doldrums | 79 | -0.504 | <0.001*** | |||||||
| Decade | 615 | -0.273 | <0.001*** | |||||||
| Level of Colony Integration | 360 | 0.264 | <0.001*** | |||||||
| Sex | 219 | 0.229 | <0.01** | |||||||
| Reproductive Mode | 219 | 0.179 | <0.01** | |||||||
| Transmission of Algae to Offspring | 192 | -0.155 | <0.05* | |||||||
| Ocean | 619 | 0.100 | <0.05* | |||||||
| Family | 563 | -0.099 | <0.05* | |||||||
| Subregion | 617 | 0.086 | <0.05* | |||||||
| Location | 608 | -0.080 | <0.05* | |||||||
| ||||||||||
| ||||||||||
| Factor | SS (Type III) | df | n | MS | F | p | ||||
| Months of Observations | 27.022 | 3 | 186 | 0.119 | 14.729 | <0.001*** | ||||
| Ocean_subregion | 27.014 | 3 | 185 | 0.120 | 14.627 | <0.001*** | ||||
| Habitat | 27.022 | 3 | 186 | 0.125 | 11.226 | <0.001*** | ||||
| Anthropogenic Input | 12.008 | 1 | 78 | 0.140 | 9.075 | <0.01** | ||||
| Depth | 7.956 | 3 | 154 | 0.121 | 8.072 | <0.001*** | ||||
| Complexity | 25.868 | 1 | 175 | 0.144 | 5.448 | <0.05* | ||||
| Degree Heating Index | 27.022 | 1 | 186 | 0.142 | 5.376 | <0.05* | ||||
| Relief from Weather | 14.177 | 2 | 109 | 0.122 | 4.728 | <0.05* | ||||
| Level of Colony Integration | 13.856 | 2 | 89 | 0.149 | 1.985 | >0.05 | ||||
| Reproductive Mode | 10.218 | 2 | 73 | 0.317 | 1.865 | >0.05 | ||||
| Family | 25.339 | 10 | 171 | 0.145 | 1.419 | >0.05 | ||||
| Sex | 9.803 | 1 | 72 | 0.136 | 0.942 | >0.05 | ||||
| Genus | 13.445 | 10 | 95 | 0.148 | 0.607 | >0.05 | ||||
| Species | 3.368 | 4 | 29 | 0.128 | 0.349 | >0.05 | ||||
| Transmission of Algae to Offspring | 7.672 | 1 | 54 | 0.144 | 0.230 | >0.05 | ||||
| Generalization | 10.939 | 1 | 77 | 0.144 | 0.050 | >0.05 | ||||
| ||||||||||
| Factor | n | Pearson’s Correlation Coefficient | p | |||||||
| Ocean-subregion | 186 | -0.326 | <0.001*** | |||||||
| Anthropogenic Input | 79 | -0.325 | <0.01** | |||||||
| Months of Observations | 187 | 0.324 | <0.001*** | |||||||
| Relief from Weather | 110 | 0.199 | <0.05* | |||||||
| Complexity | 176 | -0.174 | <0.05* | |||||||
| Degree Heating Index | 187 | 0.168< | <0.05* | |||||||
| ||||||||||
| Factor | SS (Type III) | df | n | MS | F | p | ||||
| Relief from Weather | 7.056 | 2 | 109 | 0.037 | 42.001 | <0.001*** | ||||
| Habitat | 7.607 | 3 | 123 | 0.039 | 25.630 | <0.001*** | ||||
| Ocean_subregion | 11.915 | 3 | 185 | 0.054 | 12.890 | <0.001*** | ||||
| Depth | 7.956 | 3 | 154 | 0.044 | 9.959 | <0.001*** | ||||
| Level of Colony Integration | 8.477 | 2 | 89 | 0.90 | 3.466 | <0.05* | ||||
| Anthropogenic Input | 7.961 | 1 | 79 | 0.098 | 2.957 | >0.05 | ||||
| Months of Observations | 12.185 | 3 | 186 | 0.064 | 2.806 | <0.05* | ||||
| Generalization | 3.638 | 1 | 77 | 0.049 | 2.654 | >0.05 | ||||
| Family | 11.853 | 10 | 171 | 0.066 | 1.966 | <0.05* | ||||
| Species | 0.698 | 4 | 29 | 0.023 | 1.388 | >0.05 | ||||
| Transmission of Algae to Offspring | 1.499 | 1 | 54 | 0.028 | 0.648 | >0.05 | ||||
| Degree Heating Index | 12.185 | 1 | 186 | 0.066 | 0.640 | >0.05 | ||||
| Genus | 8.161 | 10 | 95 | 0.082 | 1.426 | >0.05 | ||||
| Reproductive Mode | 3.638 | 2 | 73 | 0.051 | 0.137 | >0.05 | ||||
| Sex | 3.610 | 1 | 72 | 0.051 | 0.043 | >0.05 | ||||
| ||||||||||
| Factor | n | Pearson’s Correlation Coefficient | p | |||||||
| Anthropogenic Input | 79 | 0.327 | <0.01** | |||||||
| Level of Colony Integration | 90 | 0.260 | <0.05* | |||||||
| Habitat | 124 | -0.239 | <0.01** | |||||||
| Complexity | 176 | -0.171 | <0.05* | |||||||
| Location | 184 | -0.167 | <0.05* | |||||||
| Depth | 155 | -0.161 | <0.05* | |||||||
| ||||||||||
| ||||||||||
| Factor | SS (Type III) | df | n | MS | F | p | ||||
| Bleaching Occurrence | 30.513 | 3 | 213 | 0.127 | 9.795 | <0.001*** | ||||
| Ocean_subregion | 30.513 | 8 | 213 | 0.108 | 9.793 | <0.001*** | ||||
| Reef Types | 14.624 | 2 | 107 | 0.118 | 9.247 | <0.001*** | ||||
| Relief from Weather | 18.463 | 1 | 122 | 0.142 | 9.108 | <0.01** | ||||
| Recovery (months) | 12.929 | 4 | 57 | 0.150 | 8.274 | <0.001*** | ||||
| Shelf Position | 13.701 | 2 | 70 | 0.164 | 7.753 | <0.01** | ||||
| Species | 103.222 | 75 | 582 | 0.128 | 4.124 | <0.001*** | ||||
| Months of Observations | 30.513 | 2 | 213 | 0.139 | 3.013 | <0.05* | ||||
| Level of Colony Integration | 20.918 | 2 | 110 | 0.185 | 2.649 | >0.05 | ||||
| Genus | 23.245 | 25 | 155 | 0.133 | 1.815 | <0.05* | ||||
| Depth | 11.156 | 3 | 167 | 0.144 | 1.617 | >0.05 | ||||
| Family | 28.951 | 10 | 200 | 0.140 | 1.486 | >0.05 | ||||
| Sex | 17.052 | 1 | 96 | 0.177 | 1.437 | >0.05 | ||||
| Habitat | 4.624 | 2 | 30 | 0.150 | 1.368 | >0.05 | ||||
| Transmission of Algae to Offspring | 14.121 | 1 | 83 | 0.170 | 1.285 | >0.05 | ||||
| Complexity of Skeleton | 29.026 | 1 | 203 | 0.143 | 0.883 | >0.05 | ||||
| Generalization | 13.055 | 1 | 99 | 0.187 | 0.211 | >0.05 | ||||
| Reproductive Mode | 16.607 | 1 | 95 | 0.177 | 0.001 | >0.05 | ||||
| ||||||||||
| Factor | n | Pearson’s Correlation Coefficient | p | |||||||
| Recovery (months) | 58 | 0.383 | <0.01** | |||||||
| Reef Type | 108 | -0.367 | <0.001*** | |||||||
| Relief from Weather | 123 | -0.265 | <0.01** | |||||||
| Complexity | 214 | 0.197 | <0.01** | |||||||
| Species | 583 | -0.088 | <0.05* | |||||||
| ||||||||||
| Factor | SS (TypeIII) | df | n | MS | F | p | ||||
| Habitat | 1.564 | 2 | 39 | 0.150 | 196.399 | <0.001*** | ||||
| Anthropogenic Input | 4.725 | 1 | 79 | 0.040 | 39.372 | <0.001*** | ||||
| Ocean-subregion | 21.507 | 8 | 213 | 0.108 | 34.933 | <0.001*** | ||||
| Reef Type | 10.148 | 2 | 107 | 0.073 | 16.991 | <0.001*** | ||||
| Recovery (months) | 3.948 | 4 | 57 | 0.034 | 16.056 | <0.001*** | ||||
| Months of Observations | 21.507 | 3 | 213 | 0.092 | 7.737 | <0.001*** | ||||
| Bleaching Occurrence | 21.507 | 3 | 213 | 0.093 | 7.247 | <0.001*** | ||||
| Complexity of Skeleton | 20.148 | 1 | 203 | 0.097 | 5.321 | <0.05* | ||||
| Depth | 11.156 | 3 | 167 | 0.063 | 5.746 | <0.01** | ||||
| Shelf Position | 8.417 | 2 | 7 | 0.108 | 4.850 | <0.05* | ||||
| Reproductive Mode | 12.662 | 1 | 95 | 0.128 | 3.896 | <0.05* | ||||
| Species | 65.683 | 75 | 582 | 0.095 | 2.509 | <0.001*** | ||||
| Level of Colony Integration | 14.957 | 2 | 100 | 0.134 | 1.981 | >0.05 | ||||
| Genus | 17.777 | 25 | 155 | 0.112 | 1.279 | >0.05 | ||||
| Family | 20.117 | 10 | 200 | 0.100 | 1.216 | >0.05 | ||||
| Transmission of Algae to Offspring | 9.316 | 1 | 83 | 0.112 | 0.934 | >0.05 | ||||
| Relief From Weather | 5.069 | 1 | 122 | 0.042 | 0.873 | >0.05 | ||||
| Sex | 12.763 | 1 | 96 | 0.133 | 0.740 | >0.05 | ||||
| Generalization | 13.055 | 1 | 99 | 0.153 | 0.005 | >0.05 | ||||
| ||||||||||
| Factor | n | Pearson’s Correlation Coefficient | p | |||||||
| Habitat | 31 | -0.69 | ||||||||
| Anthropogenic Input | 80 | 0.579 | <0.001*** | |||||||
| Reef Type | 108 | -0.469 | <0.001*** | |||||||
| Location | 214 | 0.291 | <0.001*** | |||||||
| Months of Observations | 214 | 0.240 | <0.001*** | |||||||
| Depth | 164 | 0.211 | <0.01** | |||||||
| Complexity of Skeleton | 204 | -0.160 | <0.05* | |||||||
| Species | 583 | -0.122 | <0.01** | |||||||
| ||||||||||
| ||||||||||
| ||||||||||
| Factor | SS (Type III) | df | n | MS | F | p | ||||
| Bleaching Occurrence | 31.317 | 2 | 179 | 0.105 | 61.133 | <0.001*** | ||||
| Ocean_subregion | 31.372 | 5 | 179 | 0.110 | 22.132 | <0.001*** | ||||
| Relief from Weather | 5.456 | 2 | 52 | 0.072 | 12.674 | <0.001*** | ||||
| Months of Observations | 31.740 | 3 | 180 | 0.149 | 11.941 | <0.001*** | ||||
| Reproductive Mode | 18.644 | 2 | 124 | 0.131 | 10.136 | <0.001*** | ||||
| Level of Colony Integration | 19.780 | 2 | 137 | 0.132 | 7.395 | <0.01** | ||||
| Species | 10.007 | 19 | 84 | 0.053 | 6.546 | <0.001*** | ||||
| Transmission of Algae to Offspring | 15.083 | 2 | 104 | 0.133 | 5.664 | <0.01** | ||||
| Complexity of Skeleton | 27.845 | 1 | 160 | 0.170 | 4.556 | <0.05* | ||||
| Family | 28.079 | 12 | 165 | 0.137 | 4.373 | <0.001*** | ||||
| Genus | 21.113 | 22 | 137 | 0.101 | 4.233 | <0.001*** | ||||
| Depth | 6.324 | 3 | 72 | 0.079 | 3.598 | <0.05* | ||||
| Generalization | 23.446 | 1 | 141 | 0.164 | 2.895 | >0.05 | ||||
| Sex | 19.304 | 1 | 124 | 0.1577 | 0.025 | >0.05 | ||||
| ||||||||||
| Factor | n | Pearson’s Correlation Coefficient | p | |||||||
| Relief from Weather | 53 | -0.396 | <0.01** | |||||||
| Months of Observations | 181 | -0.381 | <0.001*** | |||||||
| Ocean_subregion | 180 | 0.373 | <0.001*** | |||||||
| Transmission of Algae to Offspring | 105 | -0.307 | <0.01** | |||||||
| Reproductive Mode | 125 | 0.216 | <0.05* | |||||||
| Family | 166 | -0.169 | <0.05 | |||||||
| Complexity of Skeleton | 161 | -0.167 | <0.05* | |||||||
| ||||||||||
| Factor | SS (Type III) | df | n | MS | F | p | ||||
| Ocean_subregion | 10.611 | 5 | 179 | 0.110 | 22.132 | <0.001*** | ||||
| Relief from Weather | 6.847 | 2 | 52 | 0.109 | 6.357 | <0.01** | ||||
| Complexity of Skeleton | 7.774 | 1 | 160 | 0.047 | 5.808 | <0.05* | ||||
| Months of Observations | 10.625 | 3 | 180 | 0.055 | 5.244 | <0.01** | ||||
| Depth | 5.145 | 3 | 72 | 0.066 | 2.849 | <0.05* | ||||
| Family | 8.928 | 1 | 65 | 0.049 | 2.340 | <0.001*** | ||||
| Level of Colony Integration | 6.831 | 2 | 137 | 0.049 | 1.903 | >0.05* | ||||
| Reproductive Mode | 5.996 | 2 | 124 | 0.049 | 0.203 | >0.05* | ||||
| Bleaching Occurrence | 10.586 | 2 | 179 | 0.059 | 1.919 | >0.05* | ||||
| Species | 4.522 | 19 | 84 | 0.050 | 1.302 | >0.05* | ||||
| Genus | 8.546 | 22 | 137 | 0.064 | 0.877 | >0.05* | ||||
| ||||||||||
| Factor | n | Pearson’s Correlation Coefficient | p | |||||||
| Relief from Weather | 53 | 0.316 | <0.05* | |||||||
| Months of Observations | 181 | -0.238 | <0.01** | |||||||
| Ocean-subregion | 180 | 0.197 | <0.01** | |||||||
| Complexity of Skeleton | 161 | -0.188 | <0.05* | |||||||
| ||||||||||
| ||||||||||
| Factor | SS (Type III) | df | n | MS | F | p | ||||
| Bleaching Occurrence | 9.085 | 1 | 74 | 0.078 | 43.055 | <0.001*** | ||||
| Ocean_subregion | 8.529 | 2 | 72 | 0.079 | 18.933 | <0.001*** | ||||
| Genus | 0.990 | 3 | 13 | 0.053 | 2.880 | >0.05 | ||||
| Depth | 0.929 | 1 | 8 | 0.104 | 1.904 | >0.05 | ||||
| Level of Colony Integration | 1.454 | 1 | 14 | 0.107 | 0.534 | >0.05 | ||||
| Months of Observations | 9.033 | 2 | 72 | 0.128 | 0.363 | >0.05 | ||||
| Complexity of Skeleton | 6.765 | 1 | 60 | 0.114 | 0.328 | >0.05 | ||||
| Family | 6.820 | 8 | 60 | 0.118 | 0.725 | >0.05 | ||||
| Relief from Weather | 1.655 | 1 | 14 | 0.127 | 0.056 | >0.05 | ||||
| ||||||||||
| Factor | n | Pearson’s Correlation Coefficient | p | |||||||
| Bleaching Occurrence | 75 | 0.609 | <0.001*** | |||||||
| Ocean_subregion | 73 | 0.352 | <0.01** | |||||||
| ||||||||||
| Factor | SS (Type III) | df | n | MS | F | p | ||||
| Level of Colony Integration | 5.608 | 2 | 72 | 0.058 | 13.306 | <0.001*** | ||||
| Ocean_subregion | 5.608 | 2 | 72 | 0.064 | 8.739 | >0.001*** | ||||
| Complexity of Skeleton | 3.188 | 1 | 60 | 0.047 | 8.146 | <0.01** | ||||
| Family | 3.201 | 8 | 60 | 0.043 | 2.905 | <0.01** | ||||
| Genus | 1.380 | 3 | 13 | 0.108 | 0.943 | >0.05 | ||||
| Relief from Weather | 1.963 | 1 | 14 | 0.146 | 0.412 | >0.05 | ||||
| Depth | 1.017 | 1 | 8 | 0.141 | 0.222 | >0.05 | ||||
| Bleaching Occurrence | 5.716 | 1 | 74 | 0.078 | 0.005 | >0.05 | ||||
| ||||||||||
| Factor | n | Pearson’s Correlation Coefficient | p | |||||||
| Ocean_subregion | 73 | 0.435 | <0.001*** | |||||||
| Complexity of Skeleton | 61 | -0.348 | <0.01** | |||||||
| ||||||||||
| ||||||||||
| Factor | SS (Type III) | df | n | MS | F | p | ||||
| Relief from Weather | 10.071 | 2 | 78 | 0.071 | 32.847 | <0.001*** | ||||
| Ocean_subregion | 30.800 | 6 | 198 | 0.104 | 17.265 | <0.001*** | ||||
| Degree Heating Weeks | 23.168 | 1 | 136 | 0.157 | 12.834 | <0.001*** | ||||
| Dominant Taxa | 18.981 | 6 | 113 | 0.106 | 11.877 | <0.001*** | ||||
| Shelf Position | 14.660 | 2 | 96 | 0.126 | 11.002 | <0.001*** | ||||
| Bleaching Occurrence | 30.800 | 4 | 198 | 0.131 | 10.184 | <0.001*** | ||||
| Anthropogenic Input | 4.479 | 2 | 48 | 0.068 | 10.175 | <0.001*** | ||||
| Recovery | 5.392 | 5 | 54 | 0.061 | 7.883 | <0.001*** | ||||
| Sex | 17.356 | 1 | 110 | 0.149 | 7.650 | <0.01** | ||||
| Generalization | 19.780 | 1 | 125 | 0.151 | 7.404 | <0.01** | ||||
| Level of Colony Integration | 23.630 | 2 | 136 | 0.164 | 5.199 | <0.01** | ||||
| Depth | 12.024 | 4 | 91 | 0.112 | 5.026 | <0..01** | ||||
| Months of Observations | 30.800 | 3 | 198 | 0.147 | 4.726 | <0.01** | ||||
| Species | 3.953 | 8 | 30 | 0.092 | 2.629 | <0.05* | ||||
| Family | 28.576 | 13 | 176 | 0.145 | 2.607 | <0.01** | ||||
| Genus | 18.579 | 13 | 118 | 0.136 | 2.394 | <0.01** | ||||
| Reproductive Mode | 17.293 | 1 | 110 | 0.156 | 0.510 | >0.05 | ||||
| Transmission of Algae to Offspring | 14.899 | 1 | 96 | 0.156 | 0.510 | >0.05 | ||||
| Complexity of Skeleton | 28.268 | 1 | 173 | 0.164 | 0.446 | >0.05 | ||||
| ||||||||||
| Factor | n | Pearson’s Correlation Coefficient | p | |||||||
| Relief from Weather | 79 | -0.680 | <0.001*** | |||||||
| Anthropogenic Input | 49 | 0.552 | <0.001*** | |||||||
| Species | 31 | -0.430 | <0.05* | |||||||
| Location | 199 | 0.422 | <0.001*** | |||||||
| Shelf Position | 97 | -0.399 | <0.001*** | |||||||
| Genus | 119 | 0.422 | <0.001*** | |||||||
| Recovery | 55 | 0.310 | <0.05* | |||||||
| Degree Heating Weeks | 137 | 0.295 | <0.01** | |||||||
| Family | 177 | -0.257 | <0.01** | |||||||
| Sex | 111 | 0.256 | <0.01** | |||||||
| Level of Colony Integration | 137 | 0.250 | <0.01** | |||||||
| Generalization | 126 | 0.237 | <0.01** | |||||||
| Months of Observations | 199 | -0.216 | <0.05* | |||||||
| Depth | 92 | -0.215 | <0.05* | |||||||
| Dominant Taxa | 114 | -0.191 | <0.05* | |||||||
| ||||||||||
| Factor | SS (Type III) | df | n | MS | F | p | ||||
| Ocean_subregion | 48.146 | 6 | 198 | 0.113 | 39.002 | <0.001*** | ||||
| Anthropogenic Input | 7.213 | 2 | 48 | 0.066 | 31.311 | <0.001*** | ||||
| Recovery | 11.352 | 5 | 54 | 0.087 | 16.257 | <0.001*** | ||||
| Bleaching Occurrence | 48.146 | 4 | 198 | 0.187 | 15.858 | <0.01** | ||||
| Dominant Taxa | 20.743 | 6 | 113 | 0.105 | 15.242 | <0.001*** | ||||
| Generalization | 31.185 | 1 | 125 | 0.226 | 13.697 | <0.001*** | ||||
| Degree Heating Weeks | 26.455 | 1 | 136 | 0.181 | 10.996 | <0.01** | ||||
| Depth | 11.826 | 4 | 91 | 0.107 | 5.913 | <0.001*** | ||||
| Level of Colony Integration | 33.428 | 2 | 136 | 0.230 | 5.519 | <0.01** | ||||
| Sex | 27.557 | 1 | 110 | 0.242 | 4.875 | <0.05* | ||||
| Months of Observations | 48.146 | 3 | 198 | 0.231 | 4.600 | <0.01** | ||||
| Family | 43.792 | 13 | 176 | 0.199 | 4.397 | <0.001*** | ||||
| Transmission of Algae to Offspring | 24.100 | 1 | 96 | 0.244 | 3.931 | <0.05* | ||||
| Genus | 29.940 | 13 | 118 | 0.193 | 3.860 | <0.001*** | ||||
| Species | 5.873 | 8 | 30 | 0.1557 | 2.114 | >0.05 | ||||
| Reproductive Mode | 27.156 | 1 | 111 | 0.245 | 1.003 | >0.05 | ||||
| Complexity of Skeleton | 42.057 | 1 | 173 | 0.246 | 0.791 | >0.05 | ||||
| ||||||||||
| Factor | n | Pearson’s Correlation Coefficient | p | |||||||
| Anthropogenic Input | 49 | 0.745 | <0.001*** | |||||||
| Relief from Weather | 79 | -0.680 | <0.001*** | |||||||
| Location | 199 | 0.488 | <0.01** | |||||||
| Genus | 119 | -0.428 | <0.001*** | |||||||
| Dominant Taxa | 114 | -0.343 | <0.001*** | |||||||
| Family | 177 | -0.331 | <0.001*** | |||||||
| Generalization | 126 | 0.315 | <0.001*** | |||||||
| Recovery | 55 | 0.310 | <0.05* | |||||||
| Degree Heating Weeks | 137 | 0.222 | <0.01** | |||||||
| Months of Observations | 199 | -0.235 | <0.05* | |||||||
| Level of Colony Integration | 137 | 0.222 | <0.01** | |||||||
| Sex | 111 | 0.207 | <0.05 | |||||||
| Transmission of Algae to Offspring | 97 | -0.199 | <0.05* | |||||||
| Bleaching Occurrence | 199 | -0.188 | <0.01** | |||||||
| C) High thermal stress accumulation (>40 DHW) | ||||||||||
| a) The beginning of the high thermal stress accumulation | ||||||||||
| i) Factors associated with variation in estimates of susceptibility to mass bleaching events | ||||||||||
| Source | SS(Type III) | df | n | MS | F | P | ||||
| Depth | 5.718 | 2 | 23 | 0.129 | 11.686 | <0.001*** | ||||
| Months | 5.359 | 1 | 22 | 0.182 | 8.370 | <0.01** | ||||
| Degree heating weeks | 6.049 | 1 | 24 | 0.209 | 5.999 | <0.05* | ||||
| Ocean_subregion | 6.049 | 1 | 24 | 0.209 | 5.999 | <0.05* | ||||
| Bleaching occurrence | 6.049 | 1 | 24 | 0.209 | 5.999 | <0.05* | ||||
| Level of colony integration | 5.718 | 1 | 23 | 0.219 | 4.166 | >0.05 | ||||
| Generalization | 5.273 | 1 | 20 | 0.262 | 1.138 | >0.05 | ||||
| Complexity of skeleton | 5.273 | 1 | 20 | 0.268 | 0.653 | >0.05 | ||||
| Family | 5.718 | 4 | 23 | 0.285 | 0.273 | >0.05 | ||||
| Genus | 5.255 | 4 | 21 | 0.292 | 0.255 | >0.05 | ||||
| Sex | 5.273 | 1 | 20 | 0.275 | 0.198 | >0.05 | ||||
| Reproductive mode | 5.273 | 1 | 20 | 0.275 | 0.160 | >0.05 | ||||
| Species | 1.530 | 1 | 6 | 0.300 | 0.104 | >0.05 | ||||
| Transmission of algae to offspring | 4.485 | 1 | 18 | 0.263 | 0.029 | >0.05 | ||||
| 1) Correlation of factors with average susceptibility to mass bleaching events (only significant results reported) | ||||||||||
| Source | n | Pearson’s correlation coefficient | p | |||||||
| Months of observations | 23 | 0.534 | <0.01** | |||||||
| Depth | 24 | -0.528 | <0.01** | |||||||
| Degree heating weeks | 25 | -0.455 | <0.05* | |||||||
| Bleaching occurrence | 25 | 0.455 | >0.05 | |||||||
| Location | 25 | -0.455 | >0.05 | |||||||
| ii) Factors associated with variation in estimates of mortality associated with mass bleaching | ||||||||||
| Source | SS(Type III) | df | n | MS | F | p | ||||
| Months of observations | 1.941 | 1 | 22 | 0.002 | 1109.104 | <0.001*** | ||||
| Degree heating weeks | 4.254 | 1 | 24 | 0.125 | 10.993 | <0.001*** | ||||
| Ocean_subregion | 4.254 | 1 | 24 | 0.125 | 10.993 | <0.001*** | ||||
| Bleaching occurrence | 4.254 | 1 | 24 | 0.055 | 17.715 | <0.001*** | ||||
| Depth | 3.985 | 2 | 23 | 0.126 | 5.273 | <0.05* | ||||
| Transmission of algae to offspring | 1.856 | 1 | 18 | 0.093 | 3.060 | >0.05 | ||||
| Generalisation | 1.903 | 1 | 20 | 0.087 | 2.848 | >0.05 | ||||
| Family | 3.939 | 4 | 23 | 0.137 | 2.459 | >0.05 | ||||
| Level of colony integration | 3.939 | 1 | 23 | 0.161 | 2.422 | >0.05 | ||||
| Genus | 3.853 | 4 | 21 | 0.151 | 2.116 | >0.05 | ||||
| Species | 0.963 | 1 | 6 | 0.136 | 2.097 | >0.05 | ||||
| Complexity of skeleton | 1.903 | 1 | 20 | 0.091 | 1.915 | >0.05 | ||||
| Sex | 1.903 | 1 | 20 | 0.093 | 1.477 | >0.05 | ||||
| Reproductive mode | 1.903 | 1 | 20 | 0.096 | 0.760 | >0.05 | ||||
| 1) Pearson’s correlation tests for significant results of ANOVA factors versus mortality (only significant results reported) | ||||||||||
| Source | n | Pearson’s correlation coefficient | p | |||||||
| Months of observations | 23 | 0.991 | <0.001*** | |||||||
| Degree heating weeks | 25 | -0.569 | <0.001*** | |||||||
| Bleaching occurrence | 25 | 0.569 | <0.001*** | |||||||
| Location (country) | 25 | -0.569 | <0.01** | |||||||
| Depth | 24 | 0.146 | >0.05 | |||||||
| b) The middle of the high thermal stress | ||||||||||
| i) Factors associated with variation in estimates of susceptibility to mass bleaching events | ||||||||||
| Source | SS(Type III) | df | n | MS | F | p | ||||
| Complexity | 0.935 | 1 | 19 | 0.01 | 0.313 | >0.05 | ||||
| Family | 0.683 | 1 | 10 | 0.076 | 0.007 | >0.05 | ||||
| ii) Factors associated with variation in estimates of mortality associated with mass bleaching events | ||||||||||
| Source | SS(Type III) | df | n | MS | F | p | ||||
| Complexity | 1.478 | 1 | 19 | 0.080 | 0.482 | >0.05 | ||||
| Family | 0.503 | 1 | 10 | 0.056 | 0.018 | >0.05 | ||||
| c) The end of the high degree heating week category | ||||||||||
| i) Factors associated with variation in estimates of susceptibility to mass bleaching events | ||||||||||
| Source | SS(Type III) | df | n | MS | F | p | ||||
| Bleaching occurrence | 4.202 | 2 | 49 | 0.048 | 20.007 | <0.001*** | ||||
| Level of colony integration | 2.831 | 1 | 40 | 0.051 | 16.849 | <0.001*** | ||||
| Ocean_subregion | 4.202 | 2 | 49 | 0.052 | 16.510 | <0.001*** | ||||
| Family | 2.868 | 5 | 47 | 0.030 | 10.442 | <0.001*** | ||||
| Depth | 3.449 | 3 | 35 | 0.065 | 7.096 | <0.01 | ||||
| Sex | 1.399 | 1 | 30 | 0.042 | 4.691 | <0.05* | ||||
| Generalization | 1.405 | 1 | 31 | 0.042 | 3.750 | >0.05 | ||||
| Relief from weather | 2.161 | 2 | 27 | 0.069 | 3.257 | >0.05 | ||||
| Months of observations | 4.202 | 1 | 49 | 0.083 | 2.493 | >0.05 | ||||
| Genus | 1.140 | 4 | 41 | 0.026 | 1.907 | >0.05 | ||||
| Reproductive mode | 1.399 | 1 | 30 | 0.046 | 1.379 | >0.05 | ||||
| Transmission of algae to offspring | 1.324 | 1 | 21 | 0.063 | 0.930 | >0.05 | ||||
| Thermal stress accumulation | 4.202 | 1 | 49 | 0.086 | 0.887 | >0.05 | ||||
| 1) Correlation of factors with average susceptibility to mass bleaching events | ||||||||||
| Source | n | Pearson’s correlation coefficient | p | |||||||
| Level of colony integration | 41 | 0.549 | <0.001*** | |||||||
| Depth | 36 | -0.508 | <0.01** | |||||||
| Complexity of skeleton | 40 | 0.393 | <0.05* | |||||||
| Sex | 31 | -0.373 | <0.05* | |||||||
| Ocean_subregion | 50 | -0.288 | <0.05* | |||||||
| i) Factors associated with variation in estimates of mortality to mass bleaching events | ||||||||||
| Source | SS(Type III) | df | n | MS | F | p | ||||
| Bleaching occurrence | 12.936 | 22 | 49 | 0.067 | 72.448 | <0.001*** | ||||
| Depth | 10.771 | 3 | 35 | 0.137 | 15.479 | <0.001*** | ||||
| Level of colony integration | 9.483 | 1 | 40 | 0.178 | 14.215 | <0.001*** | ||||
| Ocean_subregion | 12.936 | 2 | 49 | 0.174 | 13.571 | <0.001*** | ||||
| Relief from weather | 4.498 | 2 | 27 | 0.103 | 9.396 | <0.05 | ||||
| Genus | 10.887 | 4 | 41 | 0.179 | 5.945 | <0.01** | ||||
| Family | 12.066 | 5 | 47 | 0.168 | 5.929 | <0.05* | ||||
| Months of observations | 12.936 | 1 | 49 | 0.257 | 2.345 | >0.05 | ||||
| Generalization | 8.546 | 1 | 31 | 0.267 | 1.979 | >0.05 | ||||
| Sex | 8.411 | 1 | 30 | 0.280 | 1.083 | >0.05 | ||||
| Complexity of skeleton | 10.940 | 1 | 39 | 0.281 | 0.929 | >0.05 | ||||
| Reproductive mode | 8.411 | 1 | 30 | 0.287 | 0.344 | >0.05 | ||||
| Transmission of algae to offspring | 6.464 | 1 | 21 | 0.321 | 0.113 | >0.05 | ||||
| Thermal stress accumulation | 12.936 | 1 | 49 | 0.269 | 0.080 | >0.05 | ||||
| 1) Correlation of factors with average mortality to mass bleaching events | ||||||||||
| Source | n | Pearson’s correlation coefficient | p | |||||||
| Bleaching occurrence | 50 | -0.764 | <0.001*** | |||||||
| Depth | 36 | -0.702 | <0.001*** | |||||||
| Level of colony integration | 41 | 0.517 | <0.01** | |||||||
| Family | 48 | -0.339 | <0.05* | |||||||
References
- Wood, R. The Ecological Evolution of Reefs. Ann. Rev. Eco. Syst. 2003 29 p. 179-206. [CrossRef]
- Himmler, T.; Smrzka, D.; Zwicker, J.; Kasten, S.; Shapiro, R.D.; Bohrmann, G. Stromatolites below the photic zone in the northern Arabian Sea formed by calcifying chemotrophic microbial mats Geology 2018 46 (4): 339-342. [CrossRef]
- Stanley G.D. Jr. Recovery of Corals and Reefs after the End-Permian and the “Naked Coral” Lazarus Effect Journal of Earth Science 2012 21(1): 161-164. [CrossRef]
- Darroch, S.A.F.; Casey, M.M.; Cribb, A.T.; Bates, A.E.; Clapham, M.E.; Contreras, D.L.; Craffey, M.; Duijnstee, I.A.P.; Gearty, W.; Gotelli, N.J.; Hamilton, M.J.; Hayes R.F.; Hull, P.M.; Ibarra, D.E.; Korasidis, V.A.; Lee, J.; Looy, C.V.; Lyson, T.R.; Muddiman, B.; Roopnarine, P.D.; Shupinski, A.B.; Smith, F.A.; Stigall, A.L.; Tomé, C.P.; Turk, K.A.; Villaseñor, A.; Wagner, P.J.; Wang, S.C.; Lyons, S.K. 'Earth system engineers' and the cumulative impact of organisms in deep time Trends Ecol. Evol. 2025 40(11): 1066-1076. PMID: 40993023. [CrossRef]
- Birkeland, C. Geographic Comparison of Coral-Reef Community Processes. Proc. of the 6th ICRS, Australia 1988 1.
- Hall, M.S.; Bulinkski K.V., Mazur MC; Challener R. Devonian Stromatoporid Interactions at the Falls of the Ohio State Park, Clarksville, Indiana Thesis submitted for the Honors program and Bachelors of Science at Bellarmine University 2018 p. 63.
- Muscatine, L., Porter, J.W. Reef Corals: Mutualistic Symbioses Adapted to Nutrient-Poor Environments. BioScience 1977 27(7): 454-460. [CrossRef]
- Stanley, G. D. Jr.; Lipps J.H. Photosymbiosis: The driving force for reef success and failure. The Paleontological Society Papers 2011 17: 33-59. [CrossRef]
- Connell, J.H. Diversity in Tropical Rainforests and Coral Reefs: High diversity of trees and corals is maintained only in a nonequilibrium state. Science 1978 199(4335): 1302-1310. [CrossRef]
- Hoegh-Guldberg, O. Climate Change, Coral Bleaching, and the Future of the World’s Reefs. Mar Freshwater Res 1999 50(8): 839-866. [CrossRef]
- Hughes, T.P.; Baird, A.H.; Bellwood, D.R.; Card, M.; Connolly, S.R.; Folke, C.; Grosberg, R.;, Hoegh-Guldberg, O.; Jackson, J.B.; Kleypas, J.; Lough, J.M.; Marshall, P.; Nyström, M.; Palumbi, S.R.; Pandolfi, J.M.; Rosen, B.; Roughgarden, J. Climate change, human impacts, and the resilience of coral reefs. Science 2003 5635: 929-33. PMID: 12920289. [CrossRef]
- Nyström M.; Folke, C. Spatial Resilience of Coral Reefs Ecosystems 2001 4: 406-417.
- Riegl, B.; Purkis S. Model of Population Response to Accelerated Bleaching and Mass Mortality in a Changing Climate. Ecological Modelling 2009 220(2): 192-208. [CrossRef]
- Pearse, V.B.; Muscatine, L. Role of Algae (Zooxanthellae) in coral calcification The Biological Bulletin 1971 141(2):. [CrossRef]
- Stolarski, J.; Kitahara, M.V. Miller, D.J.; Cairns, S.D. , A.; Mazur, M.; Meibom, A. Cretaceous Scleractinian coral with a calcitic skeleton. B.M.C. Evolutionary Biology 2011 11: 316. PMID: 22034946. [CrossRef]
- Veron, J.E.N. A Reef in Time: The Great Barrier Reef from Beginning to End. Belknap Press of Harvard University Press (Cambridge, MA) 2008.
- Trench R.K. Diversity of symbiotic dinoflagellates and the evolution of micro-algal-invertebrate symbioses. Proc. 8th ICRS 1997 2: 1275-1286.
- Wooldridge, S. Is the coral-algae symbiosis really ‘mutually beneficial’ for the partners? BioEssays 2010 32(7): 615-625. [CrossRef]
- Fine, M; Tchernov, D. Scleractinian Coral Species Survive and Recover from Decalcification Science 2007 30(315): 1811. PMID: 17395821. [CrossRef]
- Berkelmans, R.; Oliver J.K. Large-scale Bleaching of Corals on the Great Barrier Reef Coral Reefs 1999 18: 55-60. [CrossRef]
- McClanahan, T. R.; Ateweberhan, M.; Ruiz Sebastian, C.; Graham, N.A.J.; Wilson, S.K.; Bruggemann J.H.; Guillaume M.M.M. Predictability of coral bleaching from synoptic satellite and in situ temperature observations. Coral Reefs 2007 26: 695-701. [CrossRef]
- West, J.M; Salm, R.V. Resistance and Resilience to Coral Bleaching: Implications for Coral Reef Conservation and Management Conservation Biology 2003 17(4): 956-967. [CrossRef]
- Loya, Y.; Salkai K.; Yamazato R.; Nakano Y.; Sambali, H.; Van Woesik, R. Coral bleaching: The winners and the losers Ecology Letters 2001 4(2): 122-131. [CrossRef]
- Rowan, R.; Knowlton, N. Baker, A.; Jara, J. Landscape ecology algal symbionts creates variation in episodes of coral bleaching. Nature 1997 388: 6639: 265-9. PMID: 9230434. [CrossRef]
- Shenkar, N.; Fine, M.; Loya, Y. Size matters: - bleaching dynamics of the coral Oculina patagonica. Mar. Ecol. Prog. Ser. 2005 294: 181-188. [CrossRef]
- Salih, A.; Larkum, A.; Cox, G.; Kuhl, M.; Hoegh-Guldberg, O. Fluorescent pigments in corals are photoprotective. Nature 2000 14(408): 850-3. PMID: 11130722. [CrossRef]
- Salih, A.; Cox, G.; Szymczak, R.; Coles, S.L.; Baird, A.H.; Dunstan, A. The role of host-based color and fluorescent pigments in photoprotection and in reducing bleaching stress in corals. Proc. 10th Int. Coral Reef Symp. 2006 75: 46-756.
- Arceo, H.; Alino, P.; Lim, G.; Licuanan, W.; Coral bleaching in Philippine Reefs: Coincident evidences with mesoscale thermal anomalies Bulletin of Marine Science 2001 69(2): 579-593.
- Fitt, W.K.; Brown B.E.; Warner, M.E.; Dunne R.P. Coral bleaching: interpretation of thermal tolerance limits and thermal thresholds in tropical corals Coral Reefs 2001 20: p. 51-65. [CrossRef]
- Brown, B.E. Coral Bleaching: Causes and Mechanisms Coral Reefs 1997 16: S129-S138. [CrossRef]
- Siebeck, U.; Marshall N.J.; Kluter, A.; Hoegh-Guldberg, O. Monitoring Coral Bleaching Using a Colour Reference Card Coral Reefs 2006 25(3): 453-460. [CrossRef]
- Baird, A.H.; Marshall P.A. Mortality, Growth, and Reproduction in Scleractinian Coral Following Bleaching on the Great Barrier Reef. Maine Ecology Progress Series 2002 237: 133-141. [CrossRef]
- Suggett, D.J.; Smith, D.J. Interpreting the Sign of Coral Bleaching as Friend vs. Foe. Global Change Biology 2010 7(1): 45-55. [CrossRef]
- Schreiber, U.; Schliwa, U.; Bilger, W. Continuous Recording of Photochemical and Nonphotochemical Chlorophyll Fluorescence Quenching with a New Type of Modulation. Photosynthetic Research 1986 10(1-2): 51-62. PMID: 24435276. [CrossRef]
- Demmig-Adams, B.; Adams W.W. III. Photoprotection and Other Responses of Plants to High Light Stress Annu. Rev. Plant Physiol. Plant Mol. Biol. 1992 43: p. 599-626. [CrossRef]
- Jones, R. Hoegh-Guldberg, O.; Larkum, A.W.L.; Schreiber, U. Temperature-induced bleaching of corals begins with impairment of the dark metabolism in zooxanthellae. Plant, Cell, and Environment 1998 21: 1219-30.
- Genty, B.; Briantis, J-M.; Baker, N.R. The Relationship Between the Quantum Yield of Photosynthetic Electron Transport and Quenching of Chlorophyll Fluorescence. Biochimica et Biophysica Acta 1989 990(1): 87-92. [CrossRef]
- Lesser, M.P. Elevated Temperatures and UltraViolet Radiation Cause Oxidative Stress and Inhibit Photosynthesis in Symbiotic Dinoflagellates. Limnology and Oceanography 1996 41: 271-283. [CrossRef]
- Ralph, P. J.; Gademann, R.; Larkum, A.W.D. Zooxanthellae expelled from bleached corals at 33◦C are photosynthetically competent. Marine Ecology Progress Series 2001 220: 63–168. [CrossRef]
- Berkelmans, R.; Willis B.L. Seasonal and local spatial patterns in the upper thermal limits of corals on the inshore Central Great Barrier Reef. Coral Reefs 1999 18: 219-228. [CrossRef]
- Hueerkamp, C.; Glynn P.W.; D’Croz, L.; Mate, J.L.; Colley S.B. Bleaching and Recovery of Five Eastern Pacific Corals in an El-Nino-Related Temperature Experiment. Bulletin of Marine Science 2001 69(1): 215-236.
- Bradbury, D.M. Inter- and Intra-specific Variation in Bleaching Susceptibility Among Scleractinian Corals. Thesis submitted for PhD at the Australian Research Council Centre of Excellence for Coral Reef Studies at James Cook University, p. 211. [CrossRef]
- Riegl, B. Phoenix from the Ashes? Repetitive Mass Mortality and the Biogeology of Southern Arabian Gulf (United Arab Emirates) Coral Systems In The Vienna School of Marine Biology: a tribute to Jorg Ott Bright, M.; Dworschak, P.C.; Stachowitsch M. (Eds),; Facultas Universitatsverlag: Vienna, Austria, 2002 p. 137-159.
- Hoey, A.S.; Howells, E.; Johansen, J.L.; Hobbs, J. P. A., Messmer, V., McCowan, D. M.; Pratchett, M.S. Recent advances in understanding the effects of climate change on coral reefs. Diversity, 2016 8(2): 12. [CrossRef]
- Swain, T.D.; Vega-Perkins J.B.; Oestrich W.K.; Triebold, C.; DuBois E.; Henss, J.; Baird A.; Siple, M.; Backman, V.; Marcelino L. Coral Bleaching Response Index: a New Tool to Standardize and Compare Susceptibility to Thermal Bleaching Global Change Biology 2016 22(7): 2475-2488. [CrossRef]
- Brown, B. E.; Suharsono Damage and recovery of coral reefs in Indonesia. Coral Reefs 1990, 8: 163-170.
- Glynn, P.W.; Colgan, M.W. Tropical Pacific coral reefs as records of environmental change. 2005 In The Geologic History of the Reefs (pp. 132-161).
- Gleason M.G. 1993 Effects of disturbance on coral communities: Bleaching in Moorea, French Polynesia. Coral Reefs 1993 12: 193-201. [CrossRef]
- Obura D. Can Corals Survive Climate Change? Copeia 2001 4: 1161-1163.
- Foden, J.; Rogers, S.I.; Jones, A.P. A critical review of the use of indicators for the management of the marine environment. Marine Environmental Research 2008 65(2): p. 139-163.
- Grigg, R.W. Coral reef environmental science: truth versus the Cassandra syndrome. Coral Reefs 1992 11: 183–186. [CrossRef]
- GBRMPA Great Barrier Reef Outlook Report 2009 Great Barrier Reef Marine Park Authority, Townsville.
- Maynard, J.A.; Baird, A.H.; Pratchett, M.S. Revisiting the model of the coral bleaching process. Coral Reefs 2008 27(3): 515-525.
- McCowan , D.M.; Pratchett, M.S.; Paley, A.S.; Seeley, M.; Baird, A.H. A Comparison of Two Methods of Obtaining Densities of Zooxanthellae in Acropora millepora. Galaxea, Journal of Coral Reef Studies 2011 13: 29-34. [CrossRef]
- Veron J.E.N. Corals of the World. Vol. 1-3, Australian Institute of Marine Science, Townville 2000 1382.
- Bridge, T.C.; Baird, A.H.; Pandolfi, J.M.; McWilliam, R.K.; Pratchett, M.S. Depth-dependent mortality of reef corals following a severe bleaching event: implications for thermal refuges and population recovery. F1000Research 2014 2: 187.
- Baird, A.H.; Bhagooli, R.; Ralph, P.J.; Takahashi, S. Coral Bleaching: The Role of the Host Trends in Ecology and Evolution 2009 24(1): 16-20.
- Dunstan, P. Distribution of zooxanthellae and photosynthetic chloroplast pigments of the reef-building coral Montastrea annularis Ellis and Solander in relation to depth on a West Indian coral reef. Bulletin of Marine Science 1979 29: p. 79-95.
- Li, S.; Yu, K.; Shi, Q.; Chen, T.; Zhao, M.; Zhao, J. Interspecies and spatial diversity in the symbiotic zooxanthellae density in corals from northern South China Sea and its relationship to coral reef bleaching. Chinese Science Bulletin 2008 53: 295-303. [CrossRef]
- Fagoonee, I.; Wilson, H.B.; Hassell, M.P.; Turner, J.R. The population dynamics of zooxanthellae populations; a long-term study in the field. Science 1999 283:843-845. [CrossRef]
- Fitt, W.K.; McFarland, F.K.; Warner, M.E.; Chilcoat, G.C. Seasonal Patterns of Tissue Biomass and Densities of Symbiotic Dinoflagellates in Reef Corals and Relation to Coral Bleaching. Limnology and Oceanography 2000 45: 677-685. [CrossRef]
- Warner, M.E.; Chilcoat, G.C.; McFarland, F.K.; Fitt, W.K. Seasonal fluctuations in the photosynthetic capacity of photosystem II in symbiotic dinoflagellates in the Caribbean reef-building coral Montastraea. Marine Biology 2002 141: 31-38. [CrossRef]
- Toller, W.W.; Rowan, R.; Knowlton, N. Repopulation of Zooxanthellae in the Caribbean Corals Montastraea annularis and M. faveolata following Experimental and Disease-Associated Bleaching. Biological Bulletin 2001 201: 360-373. [CrossRef]
- Walz Underwater Fluorometer Diving-PAM Submersible Photosynthesis Yield Analyzer, Handbook of Operation 1998 Effeltrich, Germany.
- McCowan, D.M.; Pratchett, M.S.; Baird, A.H. Bleaching susceptibility and mortality among corals with differing growth forms. Proc. of the 12th ICRS 2012.
- Pratchett, M.S.; McCowan, D.M.; Heron, S.; Maynard, J.A. Changes in bleaching susceptibility among corals subject to ocean warming and recurrent bleaching in Moorea, French Polynesia. PLoS One 2013 8(7): p, e70443. [CrossRef]
- Goreau, T.J.; Hayes, R.L. Coral Bleaching and Ocean “Hot Spots”. Ambio 1994 23: 176-180.
- Aeby, G.S.; Kenyon, J.C.; Maragos, J.E.; Potts, D.C. First record of mass coral bleaching in the Northwestern Hawaiian Islands Coral Reefs 2003 22: 256. [CrossRef]
- Rowan, R.; Knowlton, N.; Baker, A.C., Jara, J. Landscape ecology of algal symbionts creates variation in episodes of coral bleaching. Nature 1997 388: 265-269. [CrossRef]
- Berkelmans, R.; Van Oppen, M.J.H. The role of zooxanthella in the thermal tolerance of corals: a 'nugget of hope' for coral reefs in an era of climate change. Proceedings of the Royal Society, B 2006 273: 2305-2312.
- Tonks, L. Unpublished database on Zooxanthellae Genetic (Cladal) Diversity.
- Clark, R.; Jeffrey, C.; Woody, K.; Hillis-Starr, Z. Monaco, M. Spatial and temporal patterns of coral bleaching around Buck Island reef national monument, St. Croix, US Virgin Islands. Bulletin of Marine Science 2009 84(2): 167-182.
- Centeno, C.J. Effects of recent warming events on coral reef communities of Costa Rica (Central America). PhD thesis, University of Bremen 2002 p 182.
- Ying, H.; Cooke, I.; Sprungula S.; Wang, W.; Hayward, D.C.; Tang, Y.; Huttley G.; Ball, E.E.; Foret, S.; Miller, D.J. Comparative genomics reveals the distinct evolutionary trajectories of the robust and complex lineages. Genome Biology 2018 19(175). [CrossRef]
- Moothien-Pillay, R.M.; Willis, B.L.; Terashima, H. Trends in the density of zooxanthellae in Acropora millepora (Ehrenberg, 1834) at the Palm Island Group, Great Barrier Reef, Australia. Symbiosis 2005 38: 209-226.
- Fitt, W.K.; Hesley, W.K.; Belda, C.A.; Yellowlees, D. Seasonal patterns of tissue biomass and densities of symbiotic dinoflagellates in reef corals. Coral Reefs 2005 24(1): p. 67-75.
- Stimson, J.S. The annual cycle of density of zooxanthellae in the tissues of field and laboratory-held Pocillopora damicornis (Linnaeus). Journal of Experimental Marine Biology and Ecology 1997 214: 35-48. [CrossRef]
- Mwaura, J.; Grimsditch, G.; Kilonzo, J.; Amiyo, N.; Obura, D. Zooxanthellae densities are highest in summer months in equatorial corals in Kenya, Western Indian Ocean. Journal of Marine Science 2010 8: 193-202. [CrossRef]
- Jones, R.J.; Kildea, T.; Hoegh-Guldberg O. PAM Fluorometry as a tool to monitor light-use efficiency and relevant parameters in corals. Marine Ecology Progress Series 1997 152: 145-152.
- Kleppel, G.S.; Dodge, R.E.; Reese, C.J. Changes in pigmentation associated with bleaching in stony corals. Limnology and Oceanography 1989 34: 1331-1335. [CrossRef]
- Okamoto, M.; Nojima, S.; Furushima, Y.; Nojima, H. Evaluation of coral bleaching condition in situ using an underwater pulse amplitude modulated fluorometer. Fisheries Science 2005 71: 847-854. [CrossRef]
- Lang, J.C.; Lasker, H.R.; Gladfelter, E.H.; Hallock, P.; Jaap, W.C.; Losada, F.J.; Muller, R.G. Spatial and temporal variability during periods of “recovery” after mass bleaching on Western Atlantic coral reefs. American Zoologist 1992 32: 696-706. [CrossRef]
- D’Angelo, C.D.; Denzel, A.; Vogt, A.; Matz, M.V.; Oswald, F.; Salih, A.; Nienhaus, U.; Wiedenmann, J. Blue light regulation of host pigment in reef-building corals. Marine Ecology Progress Series 2008 364: 97-106. [CrossRef]
- Bucher, D.J.; Fischer, E. A possible role for urea in mediating coral responses to external ammonium supply Proc. of the 10th ICRS 2006 831-836.
- Meyer, J.L.; Schulz, E.T. Tissue condition and growth rate of corals associated with schooling fish. Limnology and Oceanography 1985 30: 157-166.
- Fitt, W.K.; Cook, C.B. The effects of feeding or addition of dissolved inorganic nutrients in maintaining the symbiosis between dinoflagellates and a tropical marine cnidarian. Marine Biology 2001 139: 507-517.
- McGuire, M.P.; Szmant A.M. Time course of physiological responses to NH4 enrichment by a coral-zooxanthellae symbiosis. Proc. of the 8th ICRS 1997 1: 909-914.
- Schloder, C.; D'Croz, L. Responses of massive and branching coral species to the combined effects of water temperature and nitrate enrichment. Journal of Experimental Marine Biology and Ecology 2004 313: 255-268. [CrossRef]
- Stambler, N.; Cox, E.F.; Vago, R. Effect of ammonium enrichment on respiration, zooxanthellar densities, and pigment concentration in two species of Hawaiian corals. Pacific Science 1994 48: 284-290.
- Nese, B.; Hidaka, M. High zooxanthella density shortens the survival time of coral cell aggregates under thermal stress. Journal of Experimental Marine Biology and Ecology 2009 368: 81-87.
- Le Tissier, M.D.A.A; Brown, B.E. Dynamics of solar bleaching in the intertidal reef coral Goniastrea aspera at Ko Phuket, Thailand. Marine Ecology Progress Series 1996 136: 235-244. [CrossRef]
- Drollet, J.H.; Faucon, M.; Maritorena, S.M.; Martin, P.M.V. A survey of environmental physico-chemical parameters during a minor coral mass bleaching event in Tahiti in 1993. Australian Journal of Freshwater Research 1994 45: 1149-1156. [CrossRef]
- Drollet, J.H.; Faucon, M.; Martin, P.M.V. Elevated sea-water temperature and solar UV-B flux associated with two successive coral mass bleaching events in Tahiti. Marine and Freshwater Research 1995 46: 1153-1157. [CrossRef]
- Jimenez, C.; Cortes, J.; Leon, A.; Ruiz, E. Coral bleaching and mortality associated with the 1997-1998 El Nino in an upwelling environment in the Eastern Pacific (Gulf of Papagayo, Costa Rica). Bulletin of Marine Science 2001 69: 151-169.
- Hoegh-Guldberg, O.; Smith GJ Influence of the population density of zooxanthellae and supply of ammonium on the biomass and metabolic characteristics of the reef corals Seriatopora hystrix and Stylophora pistillata. Marine Ecology Progress Series 1989 57: 173-186.
- Smith, D.J.; Suggett, D.J.; Baker, N.R. Is photoinhibition of zooxanthellae photosynthesis the primary cause of thermal bleaching in corals? Global Change Biology 2005 11: 1-11. [CrossRef]
- Gladfelter, E.H.; Michel, G.; Sanfelici, A. Metabolic gradients along a branch of the reef coral Acropora palmata. Bulletin of Marine Science 1989 44: 1166-1173.
- Jones, R.J.; Yellowlees D. Regulation and control of intracellular algae (=zooxanthellae) in hard corals. Philosophical Transactions of the Royal Society of London, B. 1997 352: 457-468. [CrossRef]
- Franklin, D.J.; Cedres, C.M.M.; Hoegh-Guldberg, O. Increased mortality and photoinhibition in the symbiotic dinoflagellates of the Indo-Pacific coral Stylophora pistillata (Esper) after summer bleaching. Marine Biology 2006 149: 633-642. [CrossRef]
- Glynn, P.W.; D’Croz L. Experimental evidence for high temperature tolerance of algal endosymbionts of reef-building corals. Coral Reefs 2001 20(2): 113-116.
- Jones, R.J. Zooxanthellae loss as a bioassay for assessing stress in corals. Marine Ecology Progress Series 1997 149: 163-171.
- Hoegh-Guldberg, O. A method for determining the surface area of corals. Coral Reefs 1988 7: 113-116. [CrossRef]
- Jones, R.J. Coral bleaching, bleaching-induced mortality, and the adaptive significance of the bleaching response Marine Biology 2008. [CrossRef]
- Diaz-Pulido, G.; McCook, L.J.; Dove, S.; Berkelmans, R.; Holliday, G.; Beer, M; Hoegh-Guldberg, O. Doom and Boom on a Resilient Reef: Climate Change, Algal Overgrowth and Coral Recovery. PLoS One 2009 44(4): e5239.
- Rinkevich, B. Coral chimerism as an evolutionary rescue mechanism to mitigate climate change impacts Global Change Biology 2019 25(4): 1198-1206.
- Anthony K.R.N.; Connolly, S.R.;, Hoegh-Guldberg, O. Bleaching, energetics, and coral mortality risk: effects of temperature, light, and sediment regime. Limnology and Oceanography 2007 52: 716-736. [CrossRef]
- Yonge, C,M,; Nicholls, A.G. Studies on the physiology of corals: V. The effect of starvation in light and in darkness on the relationship between corals and zooxanthellae. Scientific Report of the Great Barrier Reef Expedition 1931 1: 177-211.
- McClanahan, T.R.; Weil, E.; Maina, J. Strong geographic differences in coral bleaching in response to the 1998-2001 thermal events in the Indian and western Pacific Oceans. Marine Ecology Progress Series 2009 378: 119-125.
- Glynn , P.W.; Mate, J.L.; Baker, A.C.; Calderon, M.O. Coral bleaching and mortality in Panama and Ecuador during the 1997-1998 El-Nino Southern Oscillation Event: spatial/temporal patterns and comparisons with the 1982-1983 event. Bulletin of Marine Science 2001 69: 79-109.
- Riegl, B. Climate change and coral reefs: different effects in two high-latitude areas (Arabian Gulf , South Africa). Coral Reefs 2003 22(4): 433-446. [CrossRef]
- Wilkinson, C.R. Status of Coral Reefs of the World: 2004. Australian Institute of Marine Science, Townsville, QLD 2004.
- Wilkinson, C.R. Status of Coral Reefs of the World: 2008. Global Coral Reef Monitoring Network and Australian Institute of Marine Science, Townsville, QLD Australia 2008.
- Schmidt-Roach, S.; Lundgren, P.; Miller, K.J.; Geriach, G.; Noreen, A.M.E.; Andreakis, N. Assessing hidden species diversity in the coral Pocillopora damicornis from Eastern Australia. Coral Reefs 2013 32(1): 161-172. [CrossRef]
- Vollmer, S.B. Palumbi, S.R. Hybridization and the Evolution of Reef Coral Diversity. Science 2002 296(5575): 2023-5. [CrossRef]
- Buddemeier, R.W.; Fautin, D.G Coral bleaching as an adaptive mechanism. BioScience 1993 43: 320-326. [CrossRef]
- Baker, A.C. Reef corals bleach to survive change. Nature 2001 411: 765-766. [CrossRef]
- Berkelmans, R. Bleaching and mortality thresholds: how much is too much? In: Coral bleaching- Patterns, processes, causes, and consequences. Van Oppen, M.J.H.; Lough, J.M.; (Eds) Springer Berlin, Heidelberg 2009 p.103-119.
- Wilkinson, C.R. Status of coral reefs of the world. Australian Institute of Marine Science, Townsville 1998.
- Baker, A.C.; Glynn, P.W. Riegl, B. Climate change and coral reef bleaching: An ecological assessment of long-term impacts, recovery capacity and adaptation strategy. Ocean and Costal Management 2008 51(12): 801-817. [CrossRef]
- McClanahan, T.R.; Maina, J. Marine Protected Areas and Resilience to Coral Bleaching. Western Indian Ocean Journal of Marine Science 2003 2(2): 113-124.
- Thompson, D.M.; van Woesik, R. Corals escape bleaching in regions that recently and historically experienced frequent thermal stress. Proceedings of the Royal Society, B: Biological Sciences 2009 276(1669): 2893-2901. [CrossRef]
- Thornhill, D.J.; Fitt, W.K; Schmidt, G.W. Highly stable symbioses among Western Atlantic brooding corals. Coral Reefs 2006 25: 515-519. [CrossRef]
- Glynn, P.W.; Colley, S,B. Survival of Eastern Pacific Pocillopora corals (Scleractinia) through extreme El-Nino-Southern Oscillation warming events: A potential for coral reef resilience. Marine Ecology Progress Series 2008 367: 131-143.
- Penin, L. Adjeroud, M.; Pratchett, M.S. Tsuchiya, M. Spatial distribution of juvenile corals among different life history groups and habitats on Tiahura Reef, Moorea, French Polynesia. Marine Ecology Progress Series 2007 349: 127-139.
- Berumen, M.L.; Pratchett, M.S. Recovery without resilience: persistent disturbance and long-term shifts in the structure of fish and coral communities at Tiahura Reef, Moorea. Coral Reefs 2006 25: 647-653. [CrossRef]
- Hoegh-Guldberg, O.; Mumby, P.J.; Hooten, A.J.; Steneck, R.;S.; Greenfield, P.; Gomez, E.; Harvell, C.D.; Sale, P.F.; Edwards, A.J.; Caldeira, K.; Knowlton, N.; Eakin, C.M.; Iglesias-Prieto, R.; Muthiga, N.A.; Bradbury, R.H.; Dubi, A.; Hatziolos, M.E. Coral Reefs Under Rapid Climate Change and Ocean Acidification. Science 2007 318: 1737-1742.
- Pratchett, M.S.; Wilson S.K.; Graham, N.A.J.; Cinner, J.E.; Bellwood, D.R.; Jones, G.P.; Polunin, N.V.C.; McClanahan, T.R. Effects of climate=induced coral bleaching on coral reef fishes – ecological and economic consequences. Oceanography and Marine Biology: An Annual Review 2008 46: 251-296.
- Kramer, P.A.; Kramer, P. R. Transient and lethal effects of the 1998 coral bleaching event on the MesoAmerican reef system. Proc. of the 9th ICRS 2000 2: 1175-1180.
- Wilkinson, C.R. Status of Coral Reefs of the World:2000. Global Coral Reef Monitoring Network (GCRMN) and Australian Institute of Marine Science 2000.
- Guest, J.R.; Baird, A.H.; Maynard, J.A.; Muttaqin, E.; Edwards, A.J.; Campbell, S.J.; Yewdall, K.; Affendi, Y.A,; Chou, L.M. Contrasting patterns of coral bleaching susceptibility suggest an adaptive response to thermal stress. PLoS One 2012 7: e33353. [CrossRef]
- Brandt, M.E. The effect of species and colony size on the bleaching response of reef-building corals in the Florida Keys during the 2005 mass bleaching event. Coral Reefs 2009 28(4): 911-924. [CrossRef]
- Goorah, B.; Rathacharen, B.D.; Kultputeea, D. Occurrence of coral bleaching in the marine parks of Mauritius. AMAS 1998 135-141.
- Guzman, H.M.; Cortes, J. Changes in reef community structure after fifteen years of natural disturbances in the eastern Pacific (Costa Rica). Bulletin of Marine Science 2001 69(1): 133-139.
- Mohammed, T.A.E.-A.A.; Mohamed, A.E.-W. Some ecological factors affecting coral reef assemblages off of Hurghada, Red Sea, Egypt. Egyptian Journal of Aquatic Research 2005 31(1): 133-142.
- Dunne, R.P.; Brown, B.E.; The influence of solar radiation on bleaching of shallow water reef corals in the Andaman Sea, 1993-1998. Coral Reefs 2001 20: 201-210. [CrossRef]
- Castillo, K.D.; Helmuth, B.S.T. Influence of thermal history on the response of Montastraea annularis to short-term temperature exposure. Marine Biology 2005 148(2): 261-270. [CrossRef]
- Oliver, T.A.; Palumbi , S.R. Do corals care about record highs or high-frequency heat? Coral Reefs 2011 30(1): 195-206. [CrossRef]
- Pandolfi, J.M.; Connolly, S.R.; Marshall, D.J.l Cohen, A.L. Protected Coral Reef Futures Under Global Warming and Ocean Acidification. Science 2011 333(6041): 418-422.
- Kramer, P.A.; Kramer P.R. Ecological status of the Caribbean: a regional overview of coral reef health. United Nations Environment Programme 2000.
- Huppert, A.; Stone, L. Chaos in the Marine Deep: The Dynamics of phytoplankton-zooplankton system. The American Naturalist 1998 152(5): 723-738.
- McWilliams, J.P.; Cote, I.M.; Gill, AJ.A.; Watkinson, A.R.; Sutherland, W.J. Accelerating impacts of temperature-induced coral bleaching in the Caribbean. Ecology 2005 86(8): 2055-2060. [CrossRef]
- Stone, L.; Gabric, A.; Berman, T. Ecosystem resilience, biodiversity and canopy structure. Oikos 1999 86(3): 569-574.
- Stanley, Jr, G.D.; Fautin, D.G. Paleontology and evolution: The origins of modern corals. Science 2001 291(5510); 1913-1914.
- Wood, R. Chapter 22: Using the past to understand the future: Paleoecology of coral reefs. In Coral Reefs: An ecosystem in Transition. Dubinsky, Z.; Stambler, N. (Eds). Springer, Dordrecht. 2001 p. 313-324.
- Kinzie III, R.A.; Takayama, M.; Satos S.R.; Coffroth, M.A. The Adaptive Bleaching Hypothesis: Experimental Tests of Critical Assumptions. Biological Bulletin 2001 200: 51-58. [CrossRef]
- Ulstrup, K.E.; van Oppen, M.J. Geographic and within-colony variation in bacterial compositions and diversity in Acropora valida. Coral Reefs 2003 22(3): 253-264.
- Baker, A.C.; Starger, S.J.; McClanahan, T.R.; Glynn, P.W. Corals’ adaptive response to climate change: Shifting to new algal symbionts may safeguard devastated reefs from extinction. Nature 2004 430: 741.
- Buddemeier R.W.; Baker, A.C.; Fautin, D.G.; Jacobs, J.R. The adaptive hypothesis of bleaching. In Coral Health and Disease Rosenberg E and Loya Eds 2004 427-444.
- Fabricius, K.E.; Mieog, J.C.; Colin, P.L.; Idip, D.; van Oppen, M.J.H. Identity and diversity of coral endosymbionts (zooxanthellae) form three Palauan reefs with contrasting bleaching, temperature and shading histories. Molecular Ecology 2004 13(8): 2445-2458. [CrossRef]
- Rowan, R. Coral bleaching: thermal adaptation in reef coral symbionts. Nature 2004 430: 742.
- Ulstrup, K.E.; Berkelmans, R.; Ralph, P.J.; Van Oppen, M.J.H. Variation in bleaching sensitivity of two coral species across a latitudinal gradient on the Great Barrier Reef: the role of zooxanthellae. Marine Ecology Progress Series 2006 314: 135-148. [CrossRef]
- Wood, R. Reef Evolution. Oxford University Press, Oxford 1999.
- Brown, B.E.; Dunne, R.P.; Goodson, M.S.; Douglas, A.E. Experience shapes the susceptibility of a reef coral to bleaching. Coral Reefs 2002 21: 119-126. [CrossRef]
- Donner, S.D.; Skirving, Q.J.; Little, C.M.; Oppenheimer, M.; Hoegh-Guldberg, O. Global assessment of corals bleaching and required rates of adaptation under climate change. Global Change Biology 2005 11: 2251-2265. [CrossRef]
- Enriques, S.; Mendez, E.R., Iglesias-Prieto, R. Multiple light scattering and absorption in reef-building corals. Applied Optics 2005 44(1): 2218-2225. [CrossRef]
- Bongaerts P. Mesophotic Coral Ecosystems Current Biology 2022 32(8): R345-R346.
- Tilman, D. Causes, Consequences, and Ethics of Biodiversity. Nature 2000 405: 208-211. [CrossRef]
- Myers, N.; Knoll, A.H. The Biotic Crisis and the Future of Evolution Proc Natl Acad Sci U.S.A 2001 98(10): 5389-92.
- Riegl, B. Inhibition of reef framework by frequent disturbance: examples from the Arabian Gulf, South Africa, and the Cayman Islands. Palaeogeography, Palaeoclimatology, Palaeoecology 2001 175: 79-101. [CrossRef]
- Hughes, T.P.; Reed, D.C.; Boyle, M.J. Herbivory on coral reefs: community structure following mass mortalities of sea urchins. Journal of Experimental Marine Biology and Ecology 1987 113(1): 39-59. [CrossRef]
- Hughes, T.P. Catastrophes, Phase Shifts, and Large-Scale Degradation of a Caribbean Coral Reef. Science 1994 265(5178): 1547-1551. [CrossRef]
- Lapointe, B.E. Nutrient Thresholds for Bottom-Up Control of Macroalgal Blooms on Coral Reefs in Jamaica and Southeast Florida. Limnology and Oceanography 1997 42: 1119-1131. [CrossRef]
- Birkeland, C.C. Ratcheting down the coral reefs. BioScience 2004 54: 1021-1029.
- Feary, D.A.; Almany, G.R.; McCormick, M.I.; Jones, G.P. Habitat choice, recruitment and the response of coral reef fishes to coral degradation. Oecologia 2007 153: 727-737. [CrossRef]
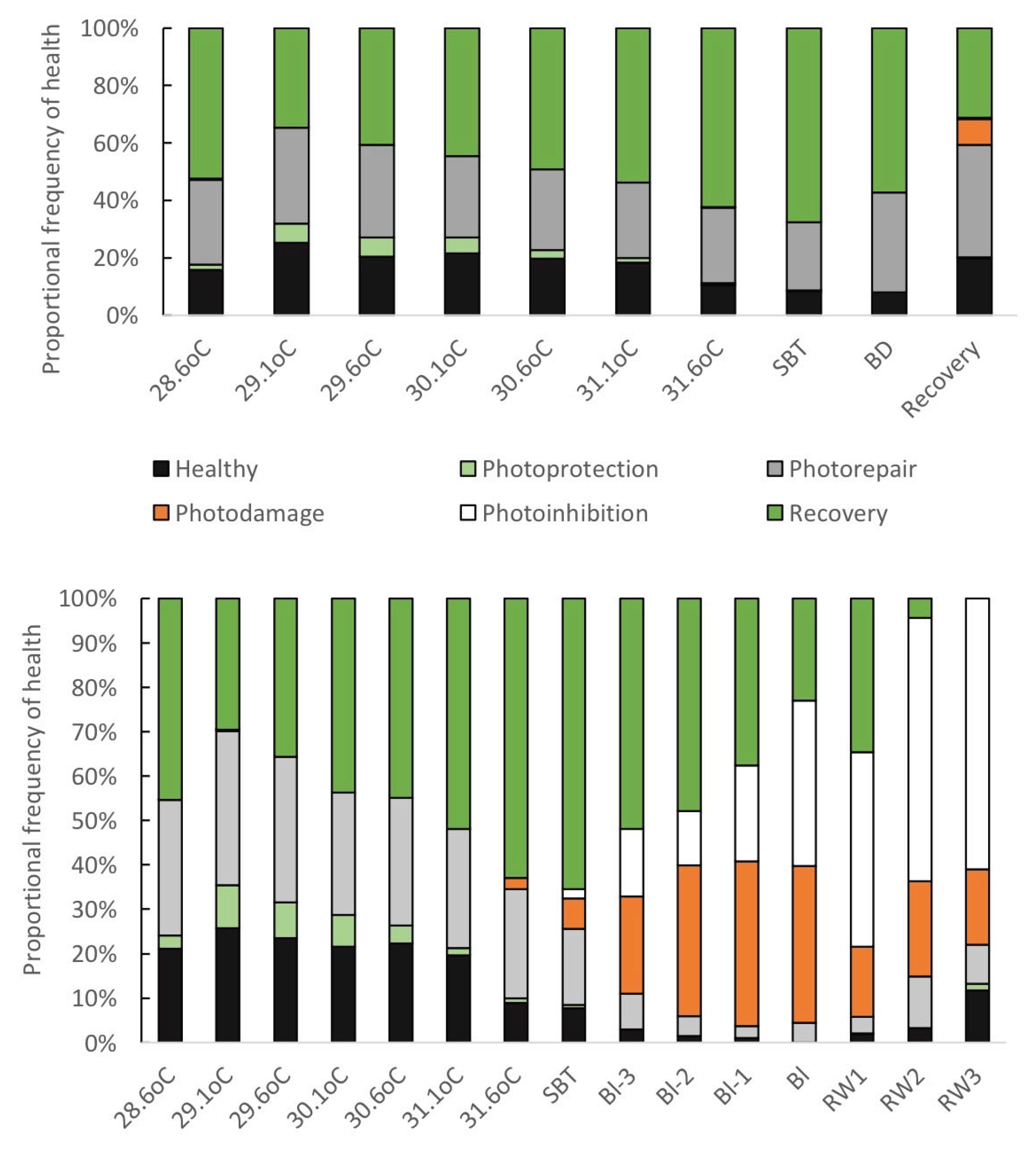
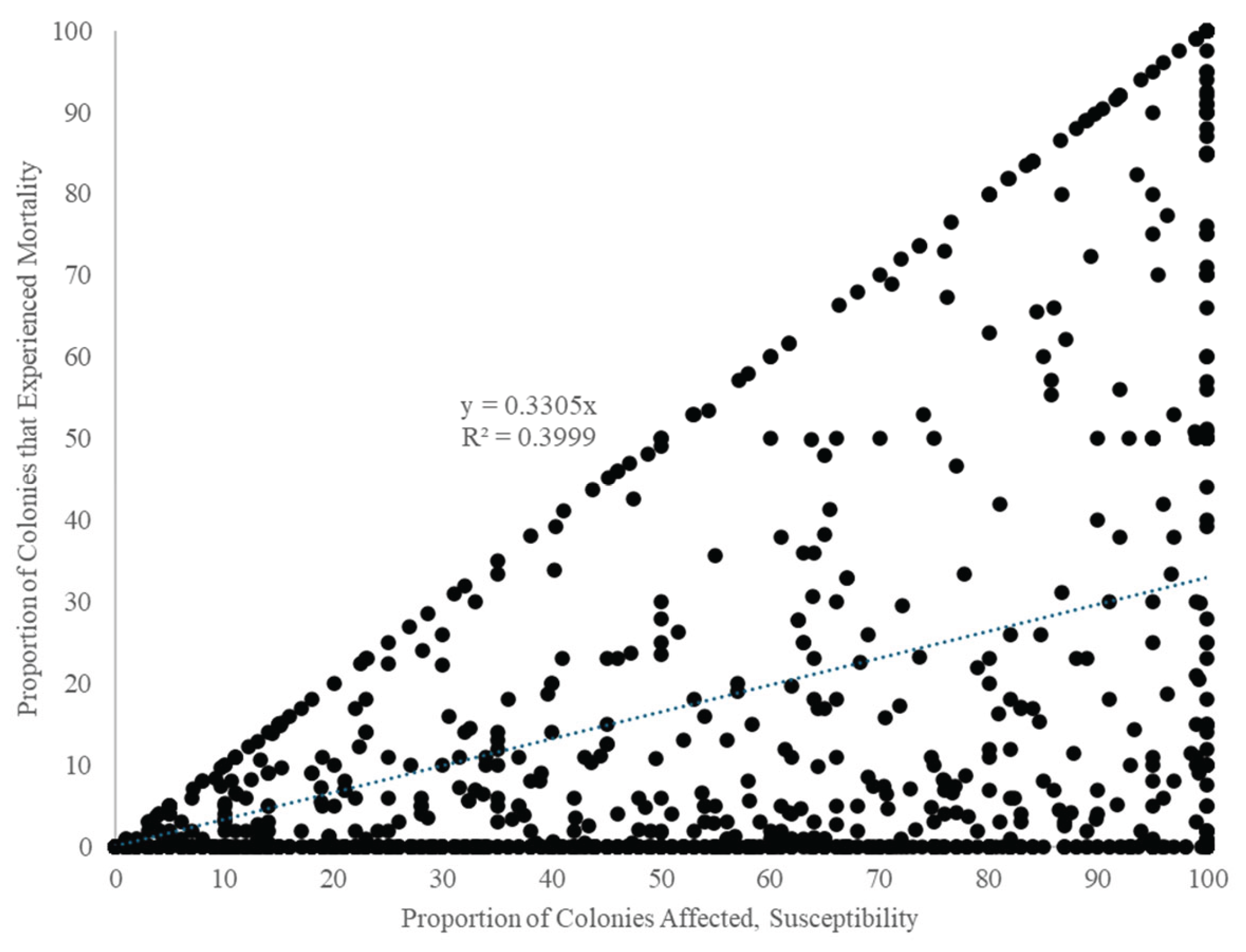
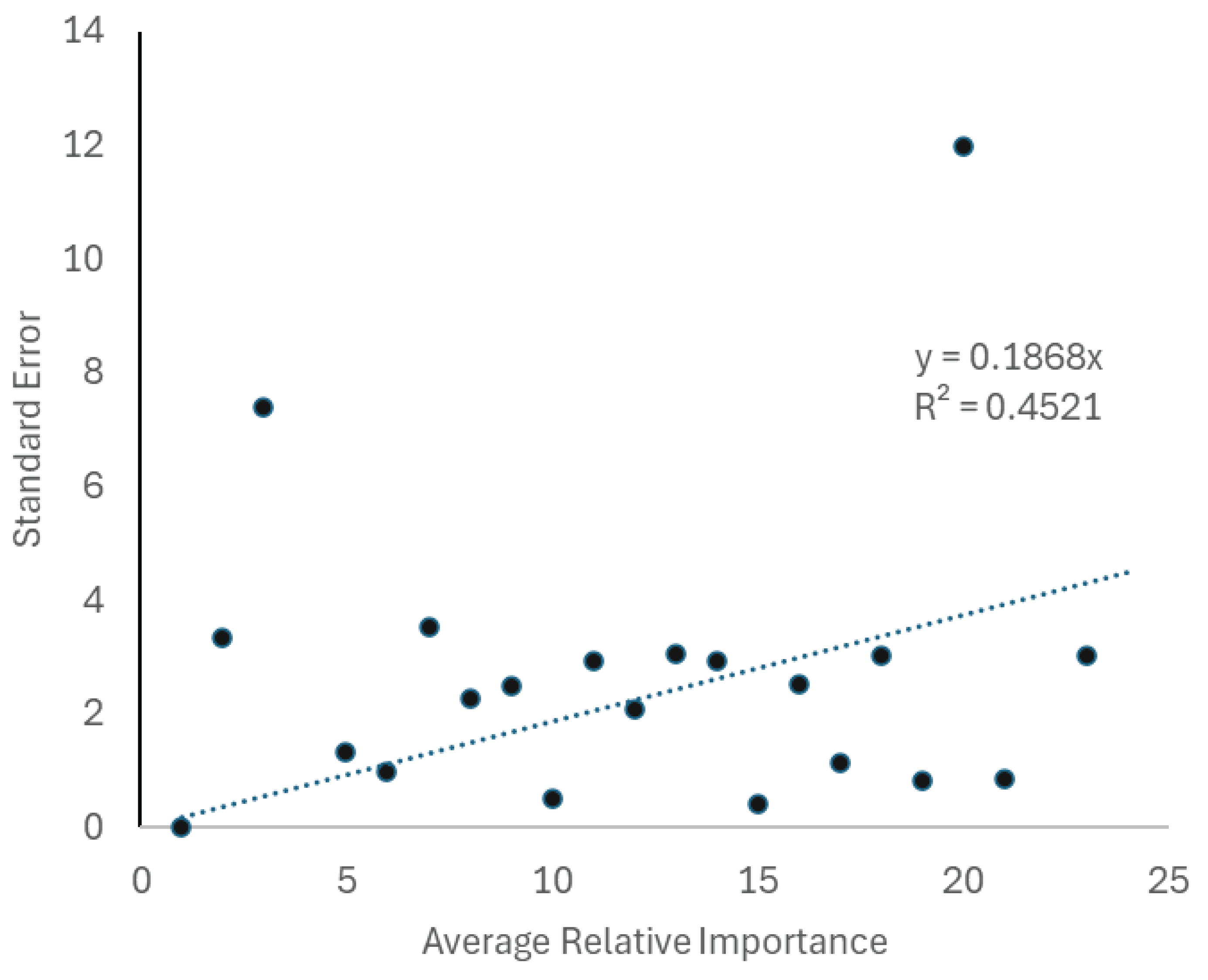
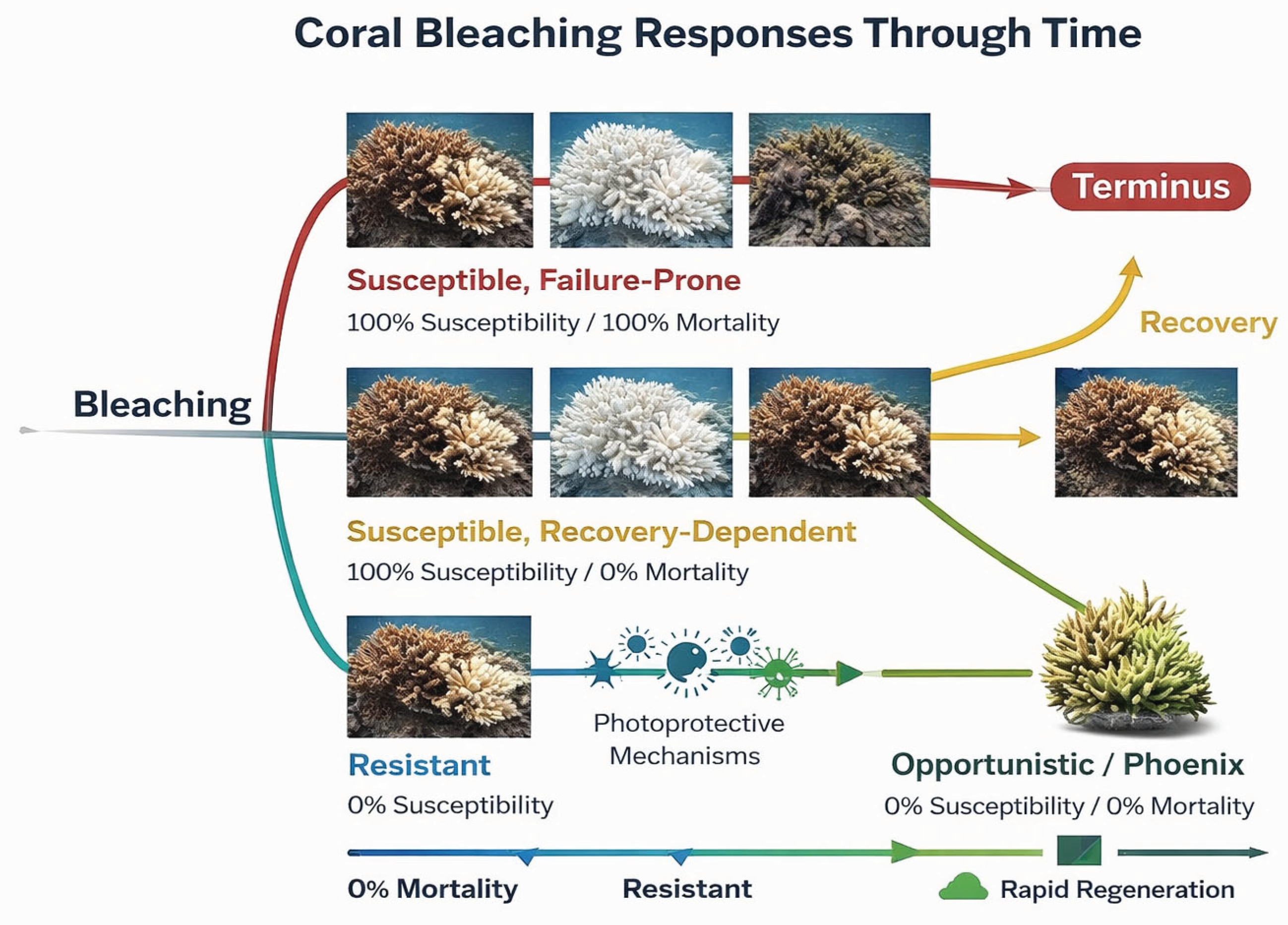
| A) Comparison of healthy and bleached colonies using fluorometric parameters (F′, Fm′, effective quantum yield, qP, qN, NPQ) and quenching dynamics. Thresholds are based on short-term temporal patterns (3-day means) to distinguish sustained dysfunction from transient fluctuation. | ||||||||||||
| Variable | F’ | Fm’ | Yield | qP | qN | NPQ | ||||||
| Control | 130-300; mean 185 | 400 – 700, mean 600 | 0.61-0.7, mean 0.66 | 1 | 0 | 0 | ||||||
| Bleached | Significant increase followed by a rapid decline, for 3 days | 3-day mean of 200 (within SE) | 3 consecutive decreasing days: 3-day mean <0.45 (within SE) | 3-day mean of 0 or >2 (within SE) | 3-day mean of 0 or >2 (within SE) | 3-day mean of 0 or >2 (within SE) | ||||||
| B) Objective phase classifications of photosynthetic health derived from PAM metrics, defining functional states including photoprotection, photorepair, photodamage, photoinhibition, and recovery. Together, these criteria translate continuous photophysiological measures into discrete, repeatable health states, providing a standardized framework for identifying phase transitions in coral bleaching dynamics. | ||||||||||||
| Phase of Health | F’ | Fm’ | Quantum yield | qP | qN | NPQ | ||||||
| Healthy | 200-325 | 600-800 | 0.6-0.8 | 1 | 0 | 0 | ||||||
| Photoprotection | 250+ | 800+ | 0.6+ | 1 | 0 | 0 | ||||||
| Photorepair | 150-250 | 500-600 | 0.5-0.6 | >1 | <1 | 1 | ||||||
| Photodamaged | 100-200 | <500 | <0.5 | 0 | >1 | >1 | ||||||
| Photoinhibited | <100 | <300 | <0.5 | 0 | >1 | >5 | ||||||
| Recovering | 50-150 | >400 | 0.6+ | >0 | <1 | <1 | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





