Submitted:
27 February 2026
Posted:
28 February 2026
You are already at the latest version
Abstract
Keywords:
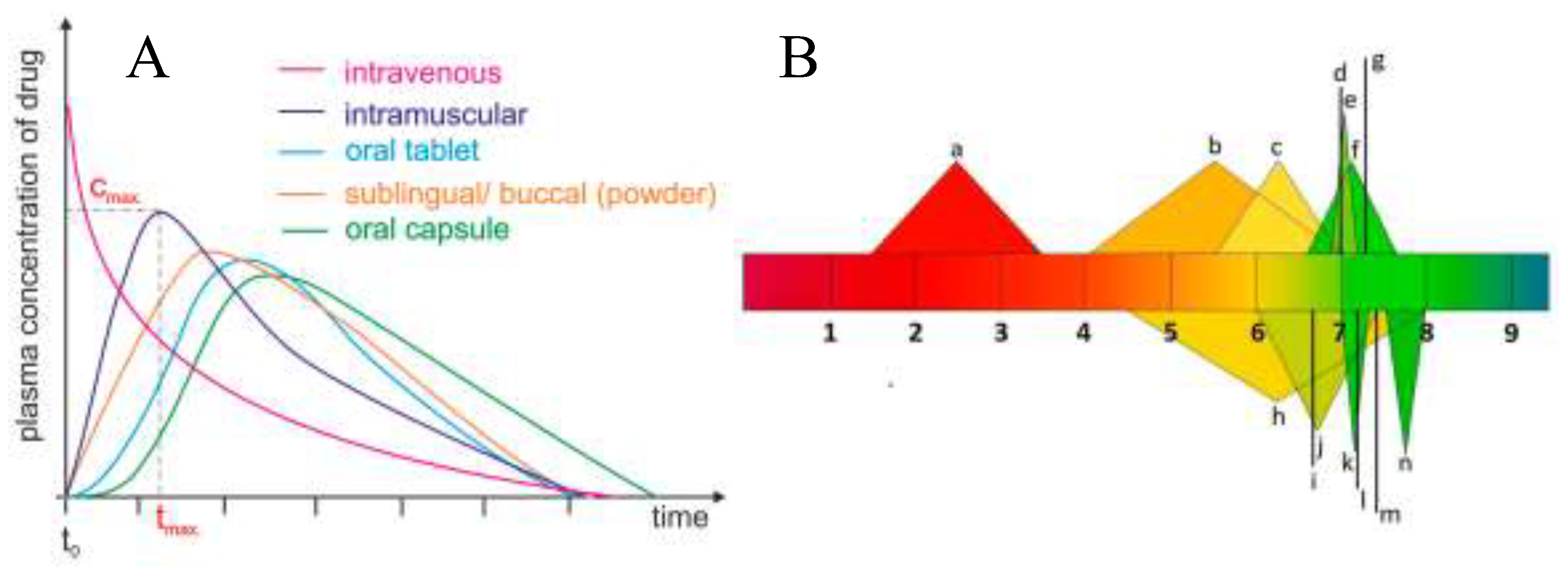
Funding
Ethical approval number
Data Availability Statement
Acknowledgments
Conflicts of Interest
Artificial intelligence (AI) usage statement
References
- Chunarkar-Patil, P.; Kaleem, M.; Mishra, R.; Ray, S.; Ahmad, A.; Verma, D.; Bhayye, S.; Dubey, R.; Singh, H.N.; Kumar, S. Anticancer Drug Discovery Based on Natural Products: From Computational Approaches to Clinical Studies. Biomedicines 2024, 12, 201. [Google Scholar] [CrossRef]
- Asano, T. Investigating the Pharmacokinetics of Natural Products: Challenges and Advances in Bioavailability. J Pharmacogn Nat Prod. 2024, 10, 326. [Google Scholar]
- Alqahtani, M.S.; Kazi, M.; Alsenaidy, M.A.; Ahmad, M.Z. Advances in Oral Drug Delivery. Front. Pharmacol. 2021, 12. [Google Scholar] [CrossRef]
- Zhang, Y; Wang, Y; Lu, Y.; et al. Advanced oral drug delivery systems for gastrointestinal targeted delivery: the design principles and foundations. J Nanobiotechnol 2025, 23, 400. [Google Scholar] [CrossRef] [PubMed]
- Stielow, M.; Witczyńska, A.; Kubryń, N.; Fijałkowski, Ł.; Nowaczyk, J.; Nowaczyk, A. The Bioavailability of Drugs—The Current State of Knowledge. Molecules 2023, 28, 8038. [Google Scholar] [CrossRef] [PubMed]
- Gao, S; Basu, S; Yang, G; Deb, A; Hu, M. Oral Bioavailability Challenges of Natural Products Used in Cancer Chemoprevention. Progress in Chemistry 2013, 25(9), 1553–1574. [Google Scholar]
- Gao, S.; Hu, M. Bioavailability Challenges Associated with Development of Anti-Cancer Phenolics. Mini-Rev. Med. Chem. 2010, 10, 550–567. [Google Scholar] [CrossRef]
- Fu, W.; Liu, L.; Tong, S. Berberine inhibits the progression of breast cancer by regulating METTL3-mediated m6A modification of FGF7 mRNA. Thorac. Cancer 2024, 15, 1357–1368. [Google Scholar] [CrossRef]
- Chen, W.; Miao, Y.-Q.; Fan, D.-J.; Yang, S.-S.; Lin, X.; Meng, L.-K.; Tang, X. Bioavailability Study of Berberine and the Enhancing Effects of TPGS on Intestinal Absorption in Rats. Aaps Pharmscitech 2011, 12, 705–711. [Google Scholar] [CrossRef]
- Liu, Y.-T.; Hao, H.-P.; Xie, H.-G.; Lai, L.; Wang, Q.; Liu, C.-X.; Wang, G.-J. Extensive Intestinal First-Pass Elimination and Predominant Hepatic Distribution of Berberine Explain Its Low Plasma Levels in Rats. Drug Metab. Dispos. 2010, 38, 1779–1784. [Google Scholar] [CrossRef]
- Hua, S. Advances in Oral Drug Delivery for Regional Targeting in the Gastrointestinal Tract - Influence of Physiological, Pathophysiological and Pharmaceutical Factors. Front. Pharmacol. 2020, 11, 524. [Google Scholar] [CrossRef]
- Timofeeva, S.V.; Zlatnik, E.Y.; Vaschenko, L.N.; Enin, Y.S.; Nepomnyashchaya, E.M. Molecular mechanisms of berberine action on tumor cells. Kazan Med J. 2025, 106, 267–276. [Google Scholar] [CrossRef]
- Orzetti, S.; Baldo, P. Toxicity Derived from Interaction between Natural Compounds and Cancer Therapeutic Drugs Metabolized by CYP3A4: Lessons Learned from Two Clinical Case Reports. Int. J. Mol. Sci. 2023, 24, 15976. [Google Scholar] [CrossRef]
- Currie, GM. Pharmacology, Part 2: Introduction to Pharmacokinetics. J Nucl Med Technol. 2018, 46(3), 221–230. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Tavassolian, N.; Haddad, W.M.; Bailey, J.M.; Gholami, B. A Fast Parameter Identification Framework for Personalized Pharmacokinetics. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef]
- Molarity and concentration calculators. Novus Biologicals- A Biotechne brand. Available online: https://www.novusbio.com/resources/calculators (accessed on 29 December 2025).
- Song, J.S.; Chae, J.-W.; Lee, K.-R.; Lee, B.H.; Choi, E.J.; Ahn, S.H.; Kwon, K.-I.; Bae, M.A. Pharmacokinetic characterization of decursinol derived fromAngelica gigasNakai in rats. Xenobiotica 2011, 41, 895–902. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zhang, J.; Xing, C.; Kim, S.-H.; Lü, J. Single Oral Dose Pharmacokinetics of Decursin, Decursinol Angelate, and Decursinol in Rats. Planta Medica 2013, 79, 275–280. [Google Scholar] [CrossRef]
- Cheng, ZX; Wu, JJ; Liu, ZQ; Lin, N. Development of a hydrophilic interaction chromatography-UPLC assay to determine trigonelline in rat plasma and its application in a pharmacokinetic study. Chin J Nat Med. 2013, 11(2), 164–70. [Google Scholar] [CrossRef] [PubMed]
- Wadhwa, G; Krishna, KV; Taliyan, R; et al. Preclinical pharmacokinetics of trigonelline using ultra-performance liquid chromatography–tandem mass spectrometry and pharmacological studies targeting type 2 diabetes. Sep Sci plus 2021, 4, 185–194. [Google Scholar] [CrossRef]
- Konsue, N.; Kirkpatrick, J.; Kuhnert, N.; King, L.J.; Ioannides, C. Repeated oral administration modulates the pharmacokinetic behavior of the chemopreventive agent phenethyl isothiocyanate in rats. Mol. Nutr. Food Res. 2010, 54, 426–432. [Google Scholar] [CrossRef]
- Narueporn, S; Dunyaporn, T; Aroonwan, L; Thitima, W. Pharmacokinetic, safety and tolerability studies after single and multiple oral administration of Phenethyl isothiocyanate in Nutri Jelly. Chulalongkorn Medical Journal 2015, 59(6), Article 3. [Google Scholar]
- Shively, C.A.; Tarka, S.M. Theobromine metabolism and pharmacokinetics in pregnant and nonpregnant Sprague-Dawley rats. Toxicol. Appl. Pharmacol. 1983, 67, 376–382. [Google Scholar] [CrossRef]
- Resman, B.H.; Blumenthal, H.P.; Jusko, W.J. Breast milk distribution of theobromine from chocolate. J. Pediatr. 1977, 91, 477–480. [Google Scholar] [CrossRef] [PubMed]
- Latini, R.; Bonati, M.; Gaspari, F.; Traina, G.; Jiritano, L.; Bortolotti, A.; Borzelleca, J.; Tarka, S.; Arnaud, M.; Garattini, S., II. Kinetics and metabolism of theobromine in male and female non-pregnant and pregnant rabbits. Toxicology 1984, 30, 343–354. [Google Scholar] [CrossRef] [PubMed]
- A Shively, C.; Tarka, S.M.; Arnaud, M.J.; Dvorchik, B.H.; Passananti, G.T.; Vesell, E.S. High levels of methylxanthines in chocolate do not alter theobromine disposition. Clin. Pharmacol. Ther. 1985, 37, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-W.; Shin, K.-H.; Yi, S.; Kim, T.-E.; Oh, S.-U.; Yoon, S.H.; Cho, J.-Y.; Yu, K.-S.; Jang, I.-J.; Shin, S.-G. Safety and Pharmacokinetics of Theobromine after Multiple Adminstrations in Healthy Volunteers. J. Korean Soc. Clin. Pharmacol. Ther. 2010, 18, 15–22. [Google Scholar] [CrossRef]
- Kapetanovic, I.M.; Muzzio, M.; Huang, Z.; Thompson, T.N.; McCormick, D.L. Pharmacokinetics, oral bioavailability, and metabolic profile of resveratrol and its dimethylether analog, pterostilbene, in rats. Cancer Chemother. Pharmacol. 2011, 68, 593–601. [Google Scholar] [CrossRef]
- Deng, L.; Li, Y.; Zhang, X.; Chen, B.; Deng, Y.; Li, Y. UPLC–MS method for quantification of pterostilbene and its application to comparative study of bioavailability and tissue distribution in normal and Lewis lung carcinoma bearing mice. J. Pharm. Biomed. Anal. 2015, 114, 200–207. [Google Scholar] [CrossRef]
- Azzolini, M.; La Spina, M.; Mattarei, A.; Paradisi, C.; Zoratti, M.; Biasutto, L. Pharmacokinetics and tissue distribution of pterostilbene in the rat. Mol. Nutr. Food Res. 2014, 58, 2122–2132. [Google Scholar] [CrossRef]
- Yeo, S.C.M.; Ho, P.C.; Lin, H. Pharmacokinetics of pterostilbene inSprague-Dawley rats: The impacts of aqueous solubility, fasting, dose escalation, and dosing route on bioavailability. Mol. Nutr. Food Res. 2013, 57, 1015–1025. [Google Scholar] [CrossRef]
- Choo, Q.-Y.; Yeo, S.C.M.; Ho, P.C.; Tanaka, Y.; Lin, H.-S. Pterostilbene surpassed resveratrol for anti-inflammatory application: Potency consideration and pharmacokinetics perspective. J. Funct. Foods 2014, 11, 352–362. [Google Scholar] [CrossRef]
- Chang, L.-S.; Oblinger, J.L.; Burns, S.S.; Huang, J.; Anderson, L.W.; Hollingshead, M.G.; Shen, R.; Pan, L.; Agarwal, G.; Ren, Y.; et al. Targeting Protein Translation by Rocaglamide and Didesmethylrocaglamide to Treat MPNST and Other Sarcomas. Mol. Cancer Ther. 2020, 19, 731–741. [Google Scholar] [CrossRef]
- Liu, Z.-Q.; Chan, K.; Zhou, H.; Jiang, Z.-H.; Wong, Y.-F.; Xu, H.-X.; Liu, L. The pharmacokinetics and tissue distribution of sinomenine in rats and its protein binding ability in vitro. Life Sci. 2005, 77, 3197–3209. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.H.; Li, H.D.; Peng, W.X.; Liu, F.Q.; Shao, Y.; He, Y.Q. Determination of sinomenine HCl in serum and urine by HPLC and its pharmacokinetics in normal volunteers. 1997, 32, 620–4. [Google Scholar]
- Zhang, M.-F.; Zhao, Y.; Jiang, K.-Y.; Han, L.; Lu, X.-Y.; Wang, X.; Zuo, L.; Meng, S.-N. Comparative Pharmacokinetics Study of Sinomenine in Rats after Oral Administration of Sinomenine Monomer and Sinomenium Acutum Extract. Molecules 2014, 19, 12065–12077. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Yan, M.; Li, H.; Jiang, P.; Deng, Y.; Cai, H. Pharmacokinetics and penetration into synovial fluid of systemical and electroporation administered sinomenine to rabbits. Biomed. Chromatogr. 2014, 29, 883–889. [Google Scholar] [CrossRef]
- Huang, H.; Zhang, E.-B.; Yi, O.-Y.; Wu, H.; Deng, G.; Huang, Y.-M.; Liu, W.-L.; Yan, J.-Y.; Cai, X. Sex-related differences in safety profiles, pharmacokinetics and tissue distribution of sinomenine hydrochloride in rats. Arch. Toxicol. 2022, 96, 3245–3255. [Google Scholar] [CrossRef]
- Hanlon, N.; Coldham, N.; Gielbert, A.; Kuhnert, N.; Sauer, M.J.; King, L.J.; Ioannides, C. Absolute bioavailability and dose-dependent pharmacokinetic behaviour of dietary doses of the chemopreventive isothiocyanate sulforaphane in rat. Br. J. Nutr. 2008, 99, 559–564. [Google Scholar] [CrossRef]
- Son, E.S.; Fei, X.; Yoon, J.-H.; Seo, S.-Y.; Maeng, H.-J.; Jeong, S.H.; Kim, Y.C. Comparison of Pharmacokinetics and Anti-Pulmonary Fibrosis-Related Effects of Sulforaphane and Sulforaphane N-acetylcysteine. Pharmaceutics 2021, 13, 958. [Google Scholar] [CrossRef]
- Veeranki, O.L.; Bhattacharya, A.; Marshall, J.R.; Zhang, Y. Organ-specific exposure and response to sulforaphane, a key chemopreventive ingredient in broccoli: implications for cancer prevention. Br. J. Nutr. 2012, 109, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Ren, T.; Wang, Q.; Li, C.; Yang, M.; Zuo, Z. Efficient brain uptake of piperine and its pharmacokinetics characterization after oral administration. Xenobiotica 2017, 48, 1249–1257. [Google Scholar] [CrossRef]
- Bajad, S.; A.K.; Bedi, K.L. Liquid chromatographic method for determination of piperine in rat plasma: application to pharmacokinetics. J. Chromatogr. B 2002, 776, 245–249. [Google Scholar] [CrossRef]
- Ramesh, B.; Vadaparthi, P.R.R.; Sukumar, G.; Manjula, N.; Babu, K.S.; Devi, P.S. LC–HRMS determination of piperine on rat dried blood spots: A pharmacokinetic study. J. Pharm. Anal. 2016, 6, 18–23. [Google Scholar] [CrossRef]
- Liu, J.; Bi, Y.; Luo, R.; Wu, X. Simultaneous UFLC–ESI–MS/MS determination of piperine and piperlonguminine in rat plasma after oral administration of alkaloids from Piper longum L.: Application to pharmacokinetic studies in rats. J. Chromatogr. B 2011, 879, 2885–2890. [Google Scholar] [CrossRef]
- Sahu, P.; Sharma, A.; Rayees, S.; Kour, G.; Singh, A.; Khullar, M.; Magotra, A.; Paswan, S.; Gupta, M.; Ahmed, I.; et al. Pharmacokinetic study of piperine in wistar rats after oral and intravenous administration. 2014. [Google Scholar]
- Roy, S; Gupta, A; Chopra, H; et al. Pharmacokinetic study of piperine in mice plasma after orally and intravenous administration. Int J Drug Deliv. 2012, 4(1), 107. [Google Scholar]
- Ren, T.; Zuo, Z. Role of piperine in CNS diseases: pharmacodynamics, pharmacokinetics and drug interactions. Expert Opin. Drug Metab. Toxicol. 2019, 15, 849–867. [Google Scholar] [CrossRef] [PubMed]
- Doolaege, E.H.A.; Raes, K.; De Vos, F.; Verhé, R.; De Smet, S. Absorption, Distribution and Elimination of Carnosic Acid, A Natural Antioxidant from Rosmarinus officinalis, in Rats. Plant Foods Hum. Nutr. 2011, 66, 196–202. [Google Scholar] [CrossRef]
- Yan, H.; Wang, L.; Li, X.; Yu, C.; Zhang, K.; Jiang, Y.; Wu, L.; Lu, W.; Tu, P. High-performance liquid chromatography method for determination of carnosic acid in rat plasma and its application to pharmacokinetic study. Biomed. Chromatogr. 2009, 23, 776–781. [Google Scholar] [CrossRef] [PubMed]
- Wang, F; Yang, G; ZhouY; Song, H; Xiong, L; Wang, L; et al. Pharmacokinetics of niazirin from Moringa oleifera Lam in rats by UPLC-MS/MS: Absolute bioavailability and dose proportionality. eFood 2022, 3, e39. [Google Scholar] [CrossRef]
- Wang, QL; Li, H; Li, XX; Cui, CY; Wang, R; Yu, NX; Chen, LX. Acute and 30-day oral toxicity studies of administered carnosic acid. Food Chem Toxicol. 2012, 50(12), 4348–55. [Google Scholar] [CrossRef]
- Manoharan, S. Improving Laboratory-Based Cancer Drug Discovery Study Designs for Better Research Translations. Methods Protoc. 2026, 9, 38. [Google Scholar] [CrossRef]
- Manoharan, S.; Ying, L.Y. Pyrimethamine reduced tumour growth in pre-clinical cancer models: a systematic review to identify potential pre-clinical studies for subsequent human clinical trials. Biol. Methods Protoc. 2024, 9, bpae021. [Google Scholar] [CrossRef]
- Jo, J.H.; Lee, J.-M.; Lee, S. Identification of absolute conversion to geraldol from fisetin and pharmacokinetics in mouse. J. Chromatogr. B 2016, 1038, 95–100. [Google Scholar] [CrossRef]
- Krishnakumar, I.M.; Jaja-Chimedza, A.; Joseph, A.; Balakrishnan, A.; Maliakel, B.; Swick, A. Enhanced bioavailability and pharmacokinetics of a novel hybrid-hydrogel formulation of fisetin orally administered in healthy individuals: a randomised double-blinded comparative crossover study. J. Nutr. Sci. 2022, 11, e74. [Google Scholar] [CrossRef] [PubMed]
- Sari, E.N.; Soysal, Y. Molecular and Therapeutic Effects of Fisetin Flavonoid in Diseases. J. Basic Clin. Heal. Sci. 2020, 4, 190–196. [Google Scholar] [CrossRef]
- Tashkandi, H.; Chaparala, A.; Peng, S.; Nagarkatti, M.; Nagarkatti, P.; Alexander; Chumanevich, A.A.; Hofseth, L.J. Pharmacokinetics of Panaxynol in Mice. J. Cancer Sci. Clin. Ther. 2020, 04, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Dong, X.; Fan, Y.; Wang, Y.; Xu, J.; Ma, J.; Yu, X. Determination and Pharmacokinetics of Acetylcorynoline in Mouse Plasma by UPLC–MS/MS. Int. J. Anal. Chem. 2025, 2025, 5319104. [Google Scholar] [CrossRef]
- Wu, Y.; Du, Y.; Wang, Z.; Wu, Y.; Li, Z.; Wang, A.; Sun, Y. Alisol B 23-acetate as a promising natural bioactive tetracyclic triterpenoid: A review of its extraction methods, pharmacokinetics, metabolism, pharmacology, and toxicity. Chem. Interactions 2026, 426, 111928. [Google Scholar] [CrossRef]
- Wang, Q.-L.; Zhang, P.-X.; Shen, R.; Xu, M.; Han, L.; Shi, X.; Zhou, Z.-R.; Yang, J.-Y.; Liu, J.-Q. Determination of arbutin in vitro and in vivo by LC-MS/MS: Pre-clinical evaluation of natural product arbutin for its early medicinal properties. J. Ethnopharmacol. 2024, 330, 118232. [Google Scholar] [CrossRef]
- Ju, X.H.; Shi, Y.; Liu, N.; Guo, D.M.; Cui, X. Determination and pharmacokinetics of gastrodin in human plasma by HPLC coupled with photodiode array detector. J. Chromatogr. B 2010, 878, 1982–1986. [Google Scholar] [CrossRef]
- Tang, C.; Wang, L.; Liu, X.; Cheng, M.; Qu, Y.; Xiao, H. Comparative pharmacokinetics of gastrodin in rats after intragastric administration of free gastrodin, parishin and Gastrodia elata extract. J. Ethnopharmacol. 2015, 176, 49–54. [Google Scholar] [CrossRef]
- Jiang, L.; Dai, J.; Huang, Z.; Du, Q.; Lin, J.; Wang, Y. Simultaneous determination of gastrodin and puerarin in rat plasma by HPLC and the application to their interaction on pharmacokinetics. J. Chromatogr. B 2013. 915-916, 8–12. [Google Scholar] [CrossRef]
- Jia, Y.; Li, X.; Xie, H.; Shen, J.; Luo, J.; Wang, J.; Wang, K.D.; Liu, Q.; Kong, L. Analysis and pharmacokinetics studies of gastrodin and p-hydroxybenzyl alcohol in dogs using ultra fast liquid chromatography–tandem mass spectrometry method. J. Pharm. Biomed. Anal. 2014, 99, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Bezerra, D.P.; Pessoa, C.; de Moraes, M.O.; Saker-Neto, N.; Silveira, E.R.; Costa-Lotufo, L.V. Overview of the therapeutic potential of piplartine (piperlongumine). Eur. J. Pharm. Sci. 2013, 48, 453–463. [Google Scholar] [CrossRef]
- Yu, X.-A.; Azietaku, J.T.; Li, J.; An, M.; He, J.; Hao, J.; Cao, J.; Chang, Y.-X. The pharmacokinetics, bioavailability and excretion of bergapten after oral and intravenous administration in rats using high performance liquid chromatography with fluorescence detection. BMC Chem. 2016, 10, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Peng, C.; Du, W.; Wang, S. Simultaneous determination of imperatorin and its metabolites in vitro and in vivo by a GC-MS method: application to a bioavailability and protein binding ability study in rat plasma. Biomed. Chromatogr. 2013, 28, 947–956. [Google Scholar] [CrossRef]
- Guthrie, S.K.; Hariharan, M.; Grunhaus, L.J. Yohimbine bioavailability in humans. Eur. J. Clin. Pharmacol. 1990, 39, 409–411. [Google Scholar] [CrossRef]
- Reyes-Hernández, O.D.; Figueroa-González, G.; Quintas-Granados, L.I.; Gutiérrez-Ruíz, S.C.; Hernández-Parra, H.; Romero-Montero, A.; Del Prado-Audelo, M.L.; Bernal-Chavez, S.A.; Cortés, H.; Peña-Corona, S.I.; et al. 3,3′-Diindolylmethane and indole-3-carbinol: potential therapeutic molecules for cancer chemoprevention and treatment via regulating cellular signaling pathways. Cancer Cell Int. 2023, 23, 1–21. [Google Scholar] [CrossRef]
- Gómez-García, R.; Vilas-Boas, A.A.; Machado, M.; Campos, D.A.; Aguilar, C.N.; Madureira, A.R.; Pintado, M. Impact of simulated in vitro gastrointestinal digestion on bioactive compounds, bioactivity and cytotoxicity of melon (Cucumis melo L. inodorus) peel juice powder. Food Biosci. 2022, 47. [Google Scholar] [CrossRef]
- Biswas, A.; Verma, S.K.; Kumar, S.; Mishra, T.; Kumar, M.; Choudhury, A.D.; Agrawal, S.; Sanap, S.N.; Bisen, A.C.; Mishra, A.; et al. Preclinical Pharmacokinetics and CYP Modulation Activity of Chebulinic Acid: A Potent Molecule Against Metabolic Disease. Curr. Drug Metab. 2023, 24, 587–598. [Google Scholar] [CrossRef] [PubMed]
- Ganesan, A. The impact of natural products upon modern drug discovery. Curr. Opin. Chem. Biol. 2008, 12, 306–317. [Google Scholar] [CrossRef]
- Beheshtizadeh, N.; Amiri, Z.; Tabatabaei, S.Z.; Seraji, A.A.; Gharibshahian, M.; Nadi, A.; Saeinasab, M.; Sefat, F.; Azar, H.K. Boosting antitumor efficacy using docetaxel-loaded nanoplatforms: from cancer therapy to regenerative medicine approaches. J. Transl. Med. 2024, 22, 1–34. [Google Scholar] [CrossRef]
- Schwahn, B.C.; Hafner, D.; Hohlfeld, T.; Balkenhol, N.; Laryea, M.D.; Wendel, U. Pharmacokinetics of oral betaine in healthy subjects and patients with homocystinuria. Br. J. Clin. Pharmacol. 2003, 55, 6–13. [Google Scholar] [CrossRef]
- Schwab, U.; Törrönen, A.; Meririnne, E.; Saarinen, M.; Alfthan, G.; Aro, A.; Uusitupa, M. Orally Administered Betaine Has an Acute and Dose-Dependent Effect on Serum Betaine and Plasma Homocysteine Concentrations in Healthy Humans. J. Nutr. 2006, 136, 34–38. [Google Scholar] [CrossRef]
- Kim, B.H.; Lee, M.J.; Lee, W.-Y.; Pyo, J.; Shin, M.-S.; Hwang, G.S.; Shin, D.; Kim, C.E.; Park, E.-S.; Kang, K.S. Hair Growth Stimulation Effect of Centipeda minima Extract: Identification of Active Compounds and Anagen-Activating Signaling Pathways. Biomolecules 2021, 11, 976. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Lin, H.; Chen, Y.; Huang, X. Pharmacokinetic and bioavailability study of kurarinone in dog plasma by UHPLC–MS/MS. Biomed. Chromatogr. 2020, 34, e4945. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Chan, K.; Budd, T. Pharmacokinetics of d-limonene in the rat by GC–MS assay. J. Pharm. Biomed. Anal. 1998, 17, 631–640. [Google Scholar] [CrossRef]
- Croce, N.; Pitaro, M.; Gallo, V.; Antonini, G. Toxicity of Usnic Acid: A Narrative Review. J. Toxicol. 2022, 2022, 1–12. [Google Scholar] [CrossRef]
- Loftsson, T.; Moya-Ortega, M.D.; Alvarez-Lorenzo, C.; Concheiro, A. Pharmacokinetics of cyclodextrins and drugs after oral and parenteral administration of drug/cyclodextrin complexes. J. Pharm. Pharmacol. 2015, 68, 544–555. [Google Scholar] [CrossRef]
- Gómez-García, R.; Vilas-Boas, A.A.; Machado, M.; Campos, D.A.; Aguilar, C.N.; Madureira, A.R.; Pintado, M. Impact of simulated in vitro gastrointestinal digestion on bioactive compounds, bioactivity and cytotoxicity of melon (Cucumis melo L. inodorus) peel juice powder. Food Biosci. 2022, 47. [Google Scholar] [CrossRef]
- Hsu, S.-J.; Chung, H.-C.; Chang, C.-H.; Liao, Y.-C.; Liu, T.-W.; Lin, S.-M.; Lee, C.-K. Rapid evaluation of apigenin bioavailability and hypouricemic bioactivity by targeted metabolomics study in enterohepatic microenvironment mimetic cell culture model. Food Res. Int. 2025, 209, 116281. [Google Scholar] [CrossRef]
- Manoharan, S.; Ying, L.Y. Baricitinib statistically significantly reduced COVID-19-related mortality: a systematic review and meta-analysis of five phase III randomized, blinded and placebo-controlled clinical trials. Biol. Methods Protoc. 2024, 9, bpae002. [Google Scholar] [CrossRef]
- Kawasaki, Y.; Freire, E. Finding a better path to drug selectivity. Drug Discov. Today 2011, 16, 985–990. [Google Scholar] [CrossRef]
- Marshall, L.J.; Bailey, J.; Cassotta, M.; Herrmann, K.; Pistollato, F. Poor Translatability of Biomedical Research Using Animals — A Narrative Review. Altern. Lab. Anim. 2023, 51, 102–135. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Macedonia, C.; Chen, Z.; Chandrasekaran, S.; Najarian, K.; Zhou, S.; Cernak, T.; Ellingrod, V.L.; Jagadish, H.V.; Marini, B.; et al. Can Machine Learning Overcome the 95% Failure Rate and Reality that Only 30% of Approved Cancer Drugs Meaningfully Extend Patient Survival? J. Med. Chem. 2024, 67, 16035–16055. [Google Scholar] [CrossRef] [PubMed]
- Thomas, DW; Burns, J; Audette, J; Caroll, A; Dow-Hygelund, C; Hay, M. Clinical Development Success Rates 2006-2015. Biomedtracker, BIO, Amplion. 2016. Available online: https://go.bio.org/rs/490-EHZ-999/images/Clinical%20Development%20Success%20Rates%202006-2015%20-%20BIO%2C%20Biomedtracker%2C%20Amplion%202016.pdf (accessed on 11 February 2026).
- Worldwide Clinical Trials. 6 reasons early phase oncology trials fail. Available online: https://www.worldwide.com/wp-content/uploads/2022/05/EarlyPhase-eBook-6ReasonsEPOncoTrialsFail-20200924.pdf#:~:text=Only%2015%25%20of%20oncology%20drugs%20that%20get,if%20the%20odds%20of%20success%20were%20greater (accessed on 11 February).
- Begley, CG; Ellis, LM. Drug development: Raise standards for preclinical cancer research. Nature 2012, 483, 531–533. [Google Scholar] [CrossRef] [PubMed]
- Bernard, R.; Gerlach, B.; Haas, M.; Macleod, M.; Potschka, H.; Rice, A.S.; Steckler, T.; Voikar, V.; Wever, K.E. Testing the usefulness of a quality system in preclinical research. Neurosci. Appl. 2024, 4, 105395. [Google Scholar] [CrossRef]
- Percie du Sert, N; Hurst, V; Ahluwalia, A; Alam, S; Avey, MT; Baker, M; et al. The ARRIVE guidelines 2.0: Updated guidelines for reporting animal research. PLoS Biol. 2020, 18, e3000410. [Google Scholar]
- Akhtar, A. The Flaws and Human Harms of Animal Experimentation. Camb. Q. Heal. Ethic- 2015, 24, 407–419. [Google Scholar] [CrossRef]
- Mahalmani, V.; Prakash, A.; Medhi, B. Do alternatives to animal experimentation replace preclinical research? Indian J. Pharmacol. 2023, 55, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Poh, W.T.; Stanslas, J. The new paradigm in animal testing – “3Rs alternatives”. Regul. Toxicol. Pharmacol. 2024, 153, 105705. [Google Scholar] [CrossRef] [PubMed]
| No | Compound | Dose (mg/kg) | Cmax** (μM) | t1/2 (h) | F(%) | Vd (L/Kg) | Animal | Ref |
| 1 | Decursinol | 5 10 20 |
5.81 18.52 15.8 |
0.74 0.87 2.07 |
45.1 66.6 81.0 |
NA |
Male SD rats | [17] |
| 37.5 | 43.3 | ~3* | NA | 2.5 | Male CD rats | [18] | ||
| 2 | Trigonelline | 10 | 89.98 | 3.6 | 57.37 | NA | Male SD rats | [19] |
| 10 | 78.59 | NA | NA | 0.778V1/ 0.851V2 |
Wistar rats | [20] | ||
| 3 | PEITC |
0.5 (single) 1.0 (single) 5.0 (single) 0.5 (repeated) 1.0 (repeated) 5.0 (repeated) |
1.95 2.37 4.71 2.47 3.96 13.10 |
2.62 2.78 3.19 2.55 1.86 1.62 |
77 61 23 83 86 43 |
1.53 1.75 2.15 1.50 1.05 0.88 |
Male Wistar rats |
[21] [21] |
| 40 mg (single) 40 mg (repeated) |
2.19 3.01 |
1.75 1.88 |
NA | 1.13 0.78 |
Humans | [22] | ||
| 4 | Theobromine | 5 10 50 100 5 10 50 100 |
44.96 84.37 404.64 929.17 41.63 73.82 444.60 941.94 |
4.96 4.83 7.51 5.88 5.27 4.38 6.00 5.98 |
NA NA |
0.716 0.735 0.749 0.598 0.685 0.694 0.632 0.589 |
Pregnant SD rats Non-pregnant SD rats |
[23] [23] |
| 4# (240 mg) | 31.64 | 7.1 | NA | 0.62 | Humans (nursing women) |
[24] | ||
| 10 100 |
6.66Day 15^ 58.45Day 15^ |
NA | NA | NA | Male rabbits | [25] | ||
| 6 solid 6 liquid |
35.69 49.23 |
9 10.17 |
80.67 100 |
0.783 0.72 |
Human (Men) |
[26] | ||
| 8.33 (500 mg) | 52.181st day 122.117th day |
13.0 13.4 |
NA | NA | Human (Men) |
[27] | ||
| 5 | Pterostilbene | 56 1 dose 56 14 doses 168 1 dose 168 14 doses |
11.00 9.95 30.75 21.69 |
1.5 1.6 1.9 1.9 |
66.9 73.2 94.2 80.8 |
NA |
Male CD rats |
[28] |
| 14 28 56 14 28 56 |
39.41 85.06 127.58 67.89 108.08 269.22 |
1.7 1.46 0.95 0.73 1.69 0.86 |
11.9 13.9 26.4 20.7 21.2 26.7 |
4.9 3.76 1.57 1.16 1.40 1.51 |
Normal C57/BL6mice C57/BL6 mice with cancer |
[29] |
||
| 22.5 | NA | NA | 35 | NA | Male Wistar rats | [30] | ||
| 15 Fasting/suspension 15 Suspension 15 Solution 30 Solution 60 Solution |
0.29 0.92 1.62 4.05 6.43 |
NA |
<5.5 15.9 59.2 105.8 115.9 |
NA |
Male SD rats |
[31] |
||
| 25 Single 25 8 doses |
2.96 2.44 |
NA | NA | NA | Male SD rats | [32] | ||
| 6 | Rocaglamide | 5+ | ~0.8 | 2.4 | 50 | NA | NSG mice | [33] |
| 7 | Sinomenine | 90 | 42.20 | 5.54 | 79.6 | 17.07 | Male SD rats | [34] |
| 1.33 (80 mg) | 0.750 | 9.4 | NA | NA | Human | [35] | ||
| 30 60 |
15.89 35.16 |
4.88 5.77 |
NA | NA | Male Wistar rats | [36] | ||
| 50 | 8.71 | 3.27 | NA | NA | Rabbits | [37] | ||
| 25 male*** 25 female |
5.98 8.91 |
1.27 1.78 |
NA | 13.90 7.6 |
Both sexes of SD rats | [38] |
||
| 8 | Sulforaphane | 0.5 1 5 |
0.262 0.474 1.19 |
62.2 50.5 27.3 |
82.4 51.8 21.0 |
95 77 42 |
Male Wistar rats | [39] |
| 0.1 0.2 0.5 |
0.203 0.367 0.807 |
0.7 3.3 5.33 |
77.8 72.4 251.4 |
NA |
Male SD rats | [40] | ||
| 26.6$ | 15.21.5h++ 5.66h++ 0.524h++ |
NA |
NA |
NA |
Male F344/NHsd rats | [41] | ||
| 9 | Piperine | 35 | 5.43 | 2.26 | NA | NA | Male SD rats | [42] |
| 20 | 9.92 | ~2.5& | NA | NA | Rats | [43] | ||
| 50 | 5.81 | 2.6 | NA | NA | Male Wistar rats | [44] | ||
| 54.1 | 15.04 | 4.1 | NA | NA | Male SD rats | [45] | ||
| 10 | 3.44 | 1.22 | NA | 4.692 | Male Wistar rats | [46] | ||
| 20 | 3.77 | 1.27 | 23.2 | 5.078 | Swiss albino mice | [47] | ||
| 20 mg 1 dose 20 mg 7 doses |
1.02 2.10 |
13.3 15.8 |
NA NA |
NA NA |
Human | [48] |
||
| 10 | Carnosic acid^^ | 64.3 | 105.29 39.1124th h |
NA | 40.1 | NA | Male SD rats | [49] |
| 90 | 127.91 27.0724th h |
16< | 65.09 | 3.23 | Male SD rats | [50] | ||
| 11 | Niazirin | 5 20 40 |
1.363 4.308 9.236 |
3.36 3.15 2.74 |
46.78 52.61 48.28 |
28.98 25.20 22.91 |
Male SD rats |
[51] |
| 12 | Falcarinol | 20 | ~6.55 | 5.89 | 50.4 | NA | CD-1 mice | [58] |
| 13 | Acetylcorynoline | 20 | 470.1 ng/ml\ | 2.7 | 58.9 | NA | mice | [59] |
| 14 | Gastrodin | 200 mg | 5.184 | 6.06 | NA | NA | Human | [62] |
| 100 | 156.63 | 1.13 | NA | NA | SD rats | [63] | ||
| 40 | 75.80 | 2.81 | 40.8 | NA | Male Wistar rats | [64] | ||
| 40 | 80.34 | 4.8 | NA | 1.57 | Dog | [65] | ||
| 15 | Bergapten | 5 10 15 |
3.975 6.462 6.046 |
6-7* 6-8* 7-9* |
80.1 94 69.5 |
NA NA NA |
Male SD rats |
[67] |
| 16 | Imperatorin | 6.25 12.5 25 |
0.137 2.124 3.922 |
1.46 6.01 7.901 |
3.85 33.51 34.76 |
6.617 10.237 16.017 |
SD rats |
[68] |
| 17 | Betaine | 50 (3 g/60kg) | 939 | 14.38 | NA | 1.324 | Human | [75] |
| 1 g 3 g 6 g |
284 599 1015 |
25.92 19.95 12.98 |
NA |
NA |
Human |
[76] |
||
| 18 | Kurarinone | 20 | 0.206 | 4.56 | 38.19 | NA | Dog | [78] |
| 19 | d-limonene | 200 | 82.948 | 5.62 | 43 | NA | Male SD rats | [79] |
| 20 | Usnic acid+^ | 20 | 94.389 | 18.9 | 77.8 | NA | Rabbit | [80] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




