Submitted:
23 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
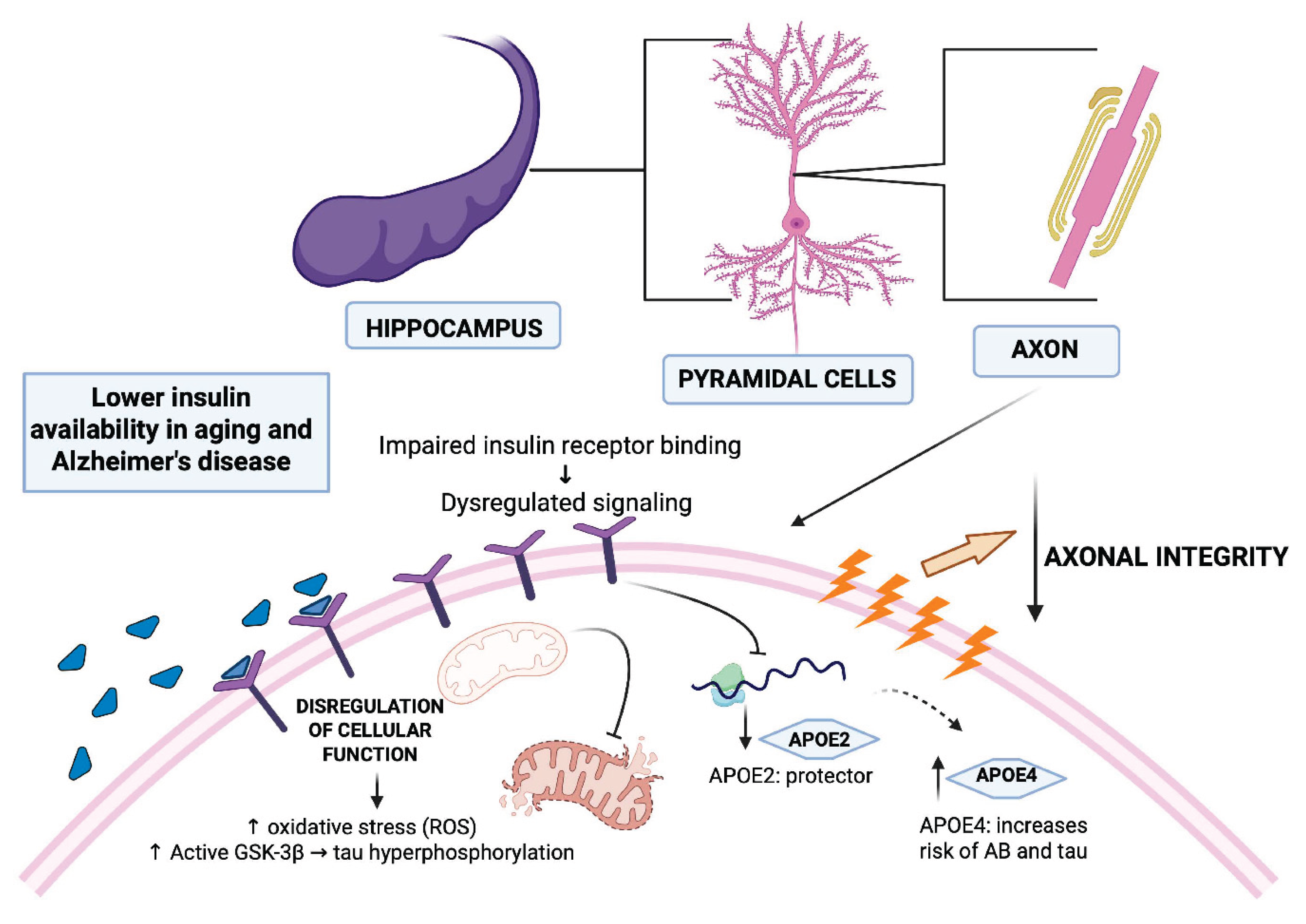
1. Introduction
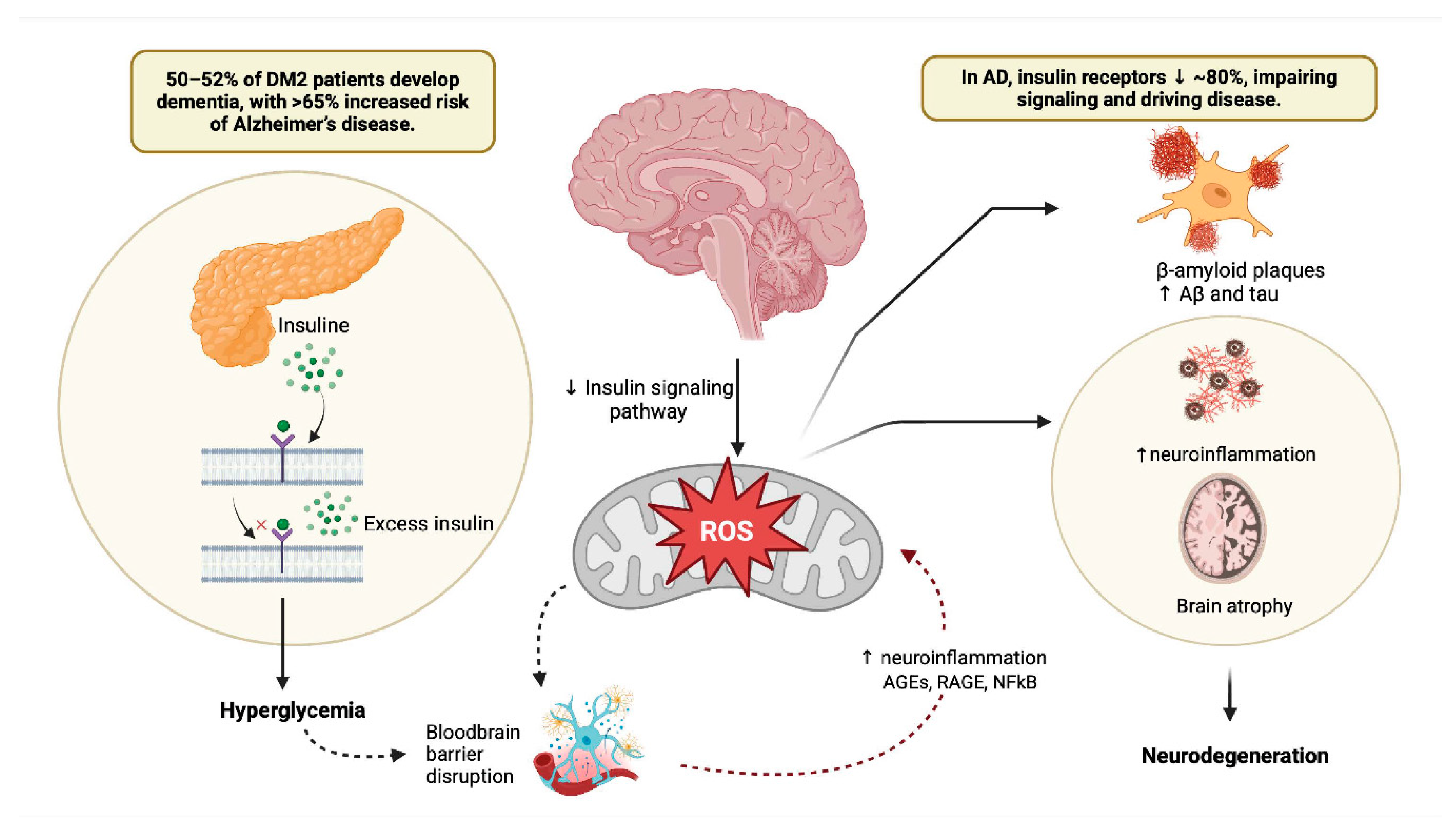
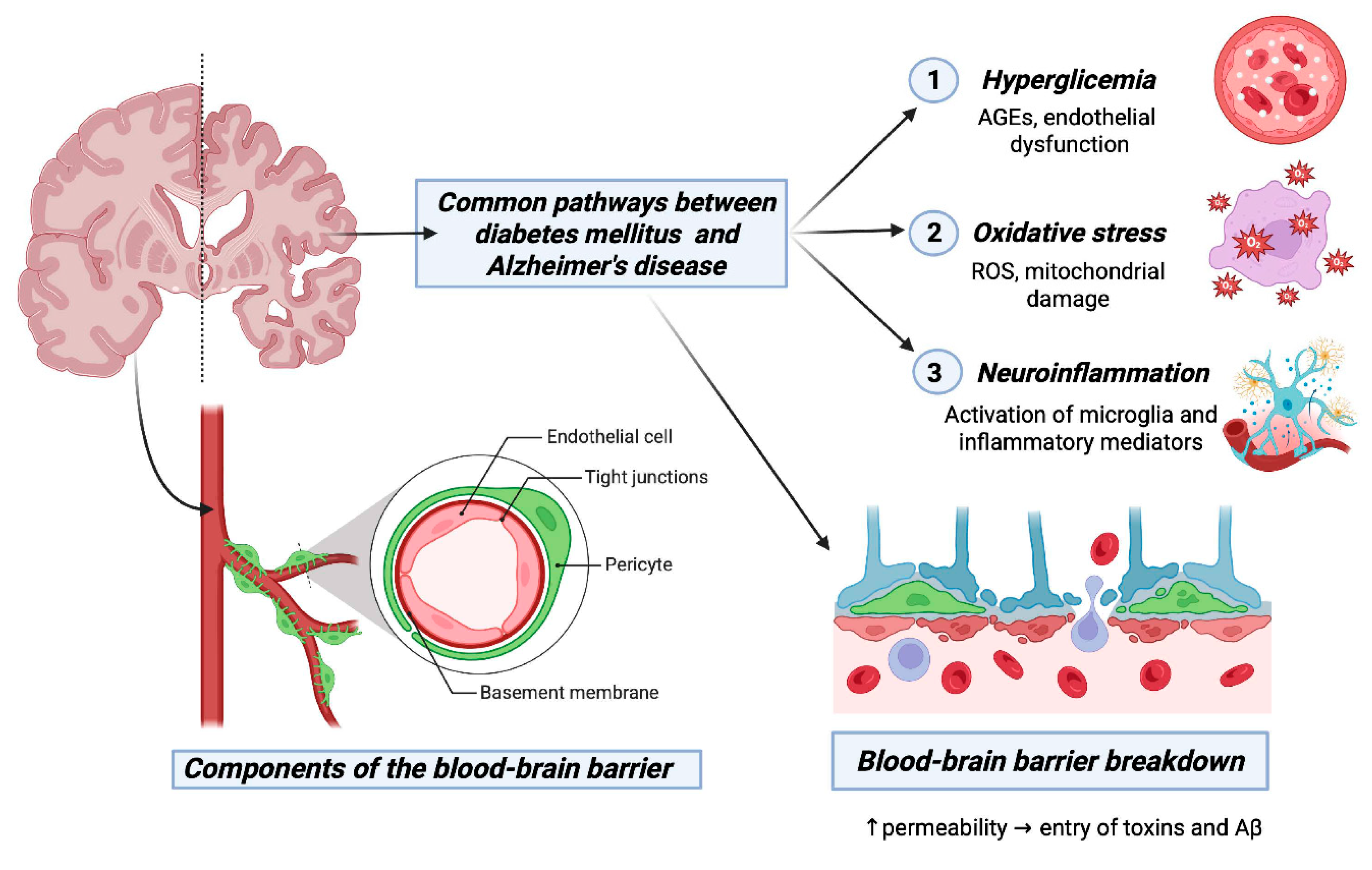
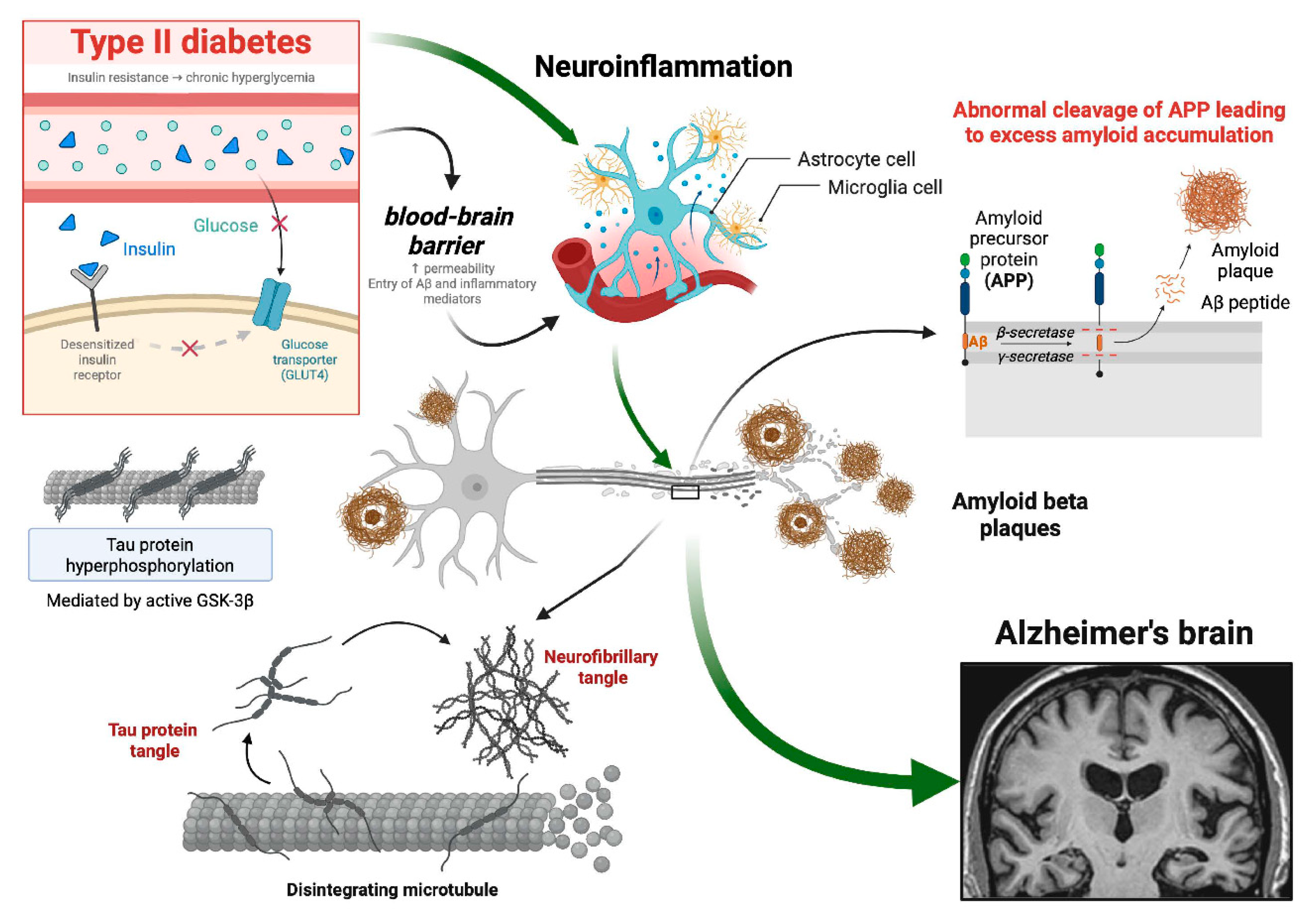
2. Unraveling the Molecular Nexus Between Type 2 Diabetes Mellitus and Alzheimer's Disease: Insights Into Shared Pathways and Therapeutic Implications.
3. Bridging the Gap: Exploring the Interplay Between Insulin Signaling, Diabetes, and Alzheimer's Disease.
4. Therapeutic Opportunities and Pharmacological Interventions
5. Biomarkers Related to Neurodegeneration in Diabetes Mellitus and Alzheimer's Disease
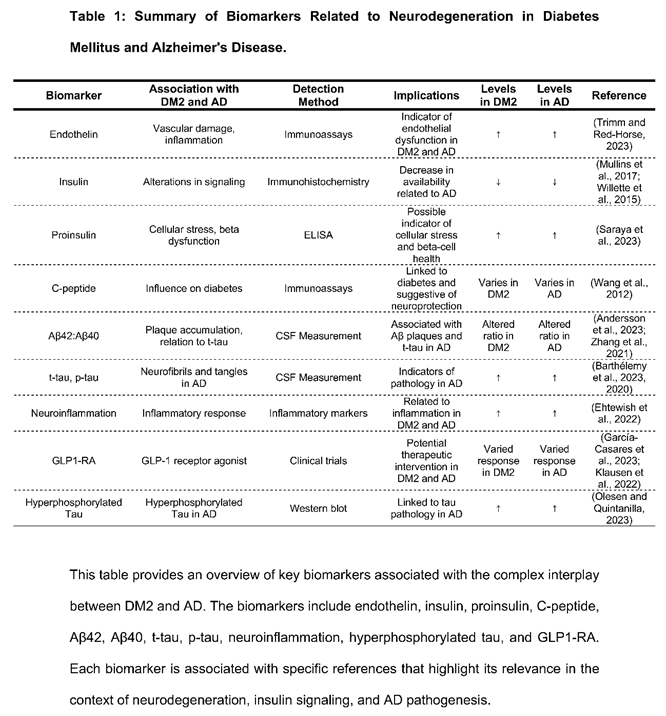
6. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Aβ | Amyloid-beta |
| AD | Alzheimer's Disease |
| AGEs | Advanced Glycation End Products |
| AKT | Protein Kinase B |
| APOE | Aprolipoprotein E |
| APP | Amyloid Precursor Protein |
| BBB | Blood-Brain Barrier |
| BMECs | Brain Microvascular Endothelial Cells |
| CNS | Central Nervous System |
| CSF | Cerebrospinal Fluid |
| DM2 | Type 2 Diabetes Mellitus |
| GLP1-RA | Glucagon-Like Peptide-1 Receptor Agonist |
| GLUT1 and GLUT3 | Glucose Transporters |
| GSK-3β | Glycogen Synthase Kinase 3 beta |
| IL-1β | Interleukin-1 beta |
| IL-6 | Interleukin-6 |
| IR | Insulin resistance |
| MAPK | Mitogen-Activated Protein Kinase |
| NFT | Neurofibrillary Tangles |
| PET-FDG | Positron Emission Tomography with Fluorodeoxyglucose |
| PI3K | Phosphoinositide 3-Kinase |
| PS1/PS2 | Presenilin-1/Presenilin-2 |
| p-tau | Phosphorylated Tau Protein |
| PTM | Posttranslational Modifications |
| RAGEs | Receptor for AGEs |
| ROS | Reactive Oxygen Species |
| t-tau | Total Tau Protein |
| VEGF | Vascular Endothelial Growth Factor |
References
- Adolfsson, R.; Bucht, G.; Lithner, F.; Winblad, B. Hypoglycemia in Alzheimer’s disease. Acta Med Scand 1980, 208, 387–8. [Google Scholar] [CrossRef]
- Andersson, E.; Schultz, N.; Saito, T.; Saido, T.C.; Blennow, K.; Gouras, G.K.; Zetterberg, H.; Hansson, O. Cerebral Aβ deposition precedes reduced cerebrospinal fluid and serum Aβ42/Aβ40 ratios in the AppNL−F/NL−F knock-in mouse model of Alzheimer’s disease. Alzheimers Res Ther 2023, 15, 64. [Google Scholar] [CrossRef]
- Arvanitakis, Z.; Wang, H.; Capuano, A.W.; Khan, A.; Taïb, B.; Anokye-Danso, F.; Schneider, J.A.; Bennett, D.A.; Ahima, R.S.; Arnold, S.E. Brain Insulin Signaling, Alzheimer Disease Pathology, and Cognitive Function. Ann Neurol 2020, 88, 513–525. [Google Scholar] [CrossRef]
- Arvanitakis, Z.; Wilson, R.S.; Bienias, J.L.; Evans, D.A.; Bennett, D.A. Diabetes Mellitus and Risk of Alzheimer Disease and Decline in Cognitive Function. Arch Neurol 2004, 61, 661. [Google Scholar] [CrossRef] [PubMed]
- Bakker, W.; Eringa, E.C.; Sipkema, P.; van Hinsbergh, V.W.M. Endothelial dysfunction and diabetes: roles of hyperglycemia, impaired insulin signaling and obesity. Cell Tissue Res 2009, 335, 165–189. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, M. Type 2 diabetes mellitus and Alzheimer’s disease. World J Diabetes 2014, 5, 889. [Google Scholar] [CrossRef]
- Barbier, P.; Zejneli, O.; Martinho, M.; Lasorsa, A.; Belle, V.; Smet-Nocca, C.; Tsvetkov, P.O.; Devred, F.; Landrieu, I. Role of Tau as a Microtubule-Associated Protein: Structural and Functional Aspects. Front Aging Neurosci 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Barthélemy, N.R.; Li, Y.; Joseph-Mathurin, N.; Gordon, B.A.; Hassenstab, J.; Benzinger, Tammie.L.S.; Buckles, V.; Fagan, A.M.; Perrin, R.J.; Goate, A.M.; Morris, J.C.; Karch, C.M.; Xiong, C.; Allegri, R.; Mendez, P.C.; Berman, S.B.; Ikeuchi, T.; Mori, H.; Shimada, H.; Shoji, M.; Suzuki, K.; Noble, J.; Farlow, M.; Chhatwal, J.; Graff-Radford, N.R.; Salloway, S.; Schofield, P.R.; Masters, C.L.; Martins, R.N.; O’Connor, A.; Fox, N.C.; Levin, J.; Jucker, M.; Gabelle, A.; Lehmann, S.; Sato, C.; Bateman, R.J.; McDade, E. A soluble phosphorylated tau signature links tau, amyloid and the evolution of stages of dominantly inherited Alzheimer’s disease. Nat Med 2020, 26, 398–407. [Google Scholar] [CrossRef]
- Barthélemy, N.R.; Saef, B.; Li, Y.; Gordon, B.A.; He, Y.; Horie, K.; Stomrud, E.; Salvadó, G.; Janelidze, S.; Sato, C.; Ovod, V.; Henson, R.L.; Fagan, A.M.; Benzinger, T.L.S.; Xiong, C.; Morris, J.C.; Hansson, O.; Bateman, R.J.; Schindler, S.E. CSF tau phosphorylation occupancies at T217 and T205 represent improved biomarkers of amyloid and tau pathology in Alzheimer’s disease. Nat Aging 2023, 3, 391–401. [Google Scholar] [CrossRef]
- Bedse, G.; Di Domenico, F.; Serviddio, G.; Cassano, T. Aberrant insulin signaling in Alzheimer’s disease: current knowledge. Front Neurosci 2015, 9. [Google Scholar] [CrossRef]
- Behl, T.; Arora, A.; Sehgal, A.; Singh, S.; Sharma, N.; Bhatia, S.; Al-Harrasi, A.; Bungau, S.; Mostafavi, E. Molecular and Biochemical Pathways Encompassing Diabetes Mellitus and Dementia. CNS Neurol Disord Drug Targets 2022, 21, 542–556. [Google Scholar] [CrossRef] [PubMed]
- Biessels, G.J.; Despa, F. Cognitive decline and dementia in diabetes mellitus: mechanisms and clinical implications. Nat Rev Endocrinol 2018, 14, 591–604. [Google Scholar] [CrossRef]
- Biessels, G.-J.; Kamal, A.; Urban, I.J.A.; Spruijt, B.M.; Erkelens, D.W.; Gispen, W.H. Water maze learning and hippocampal synaptic plasticity in streptozotocin-diabetic rats: effects of insulin treatment. Brain Res 1998, 800, 125–135. [Google Scholar] [CrossRef]
- Biessels, G.J.; Kappelle, L.J. Increased risk of Alzheimer’s disease in Type II diabetes: insulin resistance of the brain or insulin-induced amyloid pathology? Biochem Soc Trans 2005, 33, 1041–1044. [Google Scholar] [CrossRef] [PubMed]
- Biju, M.P.; Paulose, C.S. Brain glutamate dehydrogenase changes in streptozotocin diabetic rats as a function of age. Biochem Mol Biol Int 1998, 44, 1–7. [Google Scholar] [CrossRef]
- Bogush, M.; Heldt, N.A.; Persidsky, Y. Blood Brain Barrier Injury in Diabetes: Unrecognized Effects on Brain and Cognition. Journal of Neuroimmune Pharmacology 2017, 12, 593–601. [Google Scholar] [CrossRef]
- Bohórquez Moreno, C.E.; Barreto Vásquez, M.; Muvdi Muvdi, Y.P.; Rodríguez Sanjuán, A.; Badillo Viloria, M.A.; Martínez de la Rosa, W.Á.; Mendoza Sánchez, X. FACTORES MODIFICABLES Y RIESGO DE DIABETES MELLITUS TIPO 2 EN ADULTOS JÓVENES: UN ESTUDIO TRANSVERSAL. Ciencia y Enfermería 2020, 26. [Google Scholar] [CrossRef]
- Boucher, J.; Kleinridders, A.; Kahn, C.R. Insulin Receptor Signaling in Normal and Insulin-Resistant States. Cold Spring Harb Perspect Biol 2014, 6, a009191–a009191. [Google Scholar] [CrossRef]
- Burillo, J.; Marqués, P.; Jiménez, B.; González-Blanco, C.; Benito, M.; Guillén, C. Insulin Resistance and Diabetes Mellitus in Alzheimer’s Disease. Cells 2021, 10, 1236. [Google Scholar] [CrossRef]
- Burns, J.M.; Honea, R.A.; Vidoni, E.D.; Hutfles, L.J.; Brooks, W.M.; Swerdlow, R.H. Insulin is differentially related to cognitive decline and atrophy in Alzheimer’s disease and aging. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 2012, 1822, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Busiguina, S.; Fernandez, A.M.; Barrios, V.; Clark, R.; Tolbert, D.L.; Berciano, J.; Torres-Aleman, I. Neurodegeneration Is Associated to Changes in Serum Insulin-like Growth Factors. Neurobiol Dis 2000, 7, 657–665. [Google Scholar] [CrossRef]
- Calabrò, M.; Rinaldi, C.; Santoro, G.; Crisafulli, C. The biological pathways of Alzheimer disease: a review. AIMS Neurosci 2021, 8, 86–132. [Google Scholar] [CrossRef]
- Chelombitko, M.A. Role of Reactive Oxygen Species in Inflammation: A Minireview. Moscow Univ Biol Sci Bull 2018, 73, 199–202. [Google Scholar] [CrossRef]
- Chen, Y.; Sillaire, A.R.; Dallongeville, J.; Skrobala, E.; Wallon, D.; Dubois, B.; Hannequin, D.; Pasquier, F. Low Prevalence and Clinical Effect of Vascular Risk Factors in Early-Onset Alzheimer’s Disease. Journal of Alzheimer’s Disease 2017, 60, 1045–1054. [Google Scholar] [CrossRef]
- Cheng, Y.; Ren, J.-R.; Jian, J.-M.; He, C.-Y.; Xu, M.-Y.; Zeng, G.-H.; Tan, C.-R.; Shen, Y.-Y.; Jin, W.-S.; Chen, D.-W.; Li, H.-Y.; Yi, X.; Zhang, Y.; Bu, X.-L.; Wang, Y.-J. Associations of plasma angiostatin and amyloid-β and tau levels in Alzheimer’s disease. Transl Psychiatry 2022, 12, 194. [Google Scholar] [CrossRef]
- Cholerton, B.; Baker, L.D.; Craft, S. Insulin, cognition, and dementia. Eur J Pharmacol 2013, 719, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Chrem Mendez, P.; Surace, E.; Bérgamo, Y.; Calandri, I.; Vázquez, S.; Sevlever, G.; Allegri, R.F. Biomarkers for Alzheimer’s disease. Where we stand and where we are headed. Medicina (B Aires) 2019, 79, 546–551. [Google Scholar] [PubMed]
- Ciudin, A. Diabetes mellitus tipo 2 y enfermedad de Alzheimer: una relación para no olvidar. Endocrinología y Nutrición 2016, 63, 191–193. [Google Scholar] [CrossRef]
- Clark, I.A.; Vissel, B. Therapeutic implications of how TNF links apolipoprotein E, phosphorylated tau, α-synuclein, amyloid-β and insulin resistance in neurodegenerative diseases. Br J Pharmacol 2018, 175, 3859–3875. [Google Scholar] [CrossRef] [PubMed]
- Costache, A.D.; Ignat, B.E.; Grosu, C.; Mastaleru, A.; Abdulan, I.; Oancea, A.; Roca, M.; Leon, M.M.; Badescu, M.C.; Luca, S.; Jigoranu, A.R.; Chetran, A.; Mitu, O.; Costache, I.I.; Mitu, F. Inflammatory Pathways in Overweight and Obese Persons as a Potential Mechanism for Cognitive Impairment and Earlier Onset Alzeihmer’s Dementia in the General Population: A Narrative Review. Biomedicines 2023, 11, 3233. [Google Scholar] [CrossRef]
- Craft, S. Insulin resistance syndrome and Alzheimer disease: pathophysiologic mechanisms and therapeutic implications. Alzheimer Dis Assoc Disord 2006, 20, 298–301. [Google Scholar] [CrossRef] [PubMed]
- Craft, S. Insulin resistance and cognitive impairment: a view through the prism of epidemiology. Arch Neurol 2005, 62, 1043–4. [Google Scholar] [CrossRef] [PubMed]
- Craft, S.; Peskind, E.; Schwartz, M.W.; Schellenberg, G.D.; Raskind, M.; Porte, D. Cerebrospinal fluid and plasma insulin levels in Alzheimer’s disease. Neurology 1998, 50, 164–168. [Google Scholar] [CrossRef]
- Craft, S.; Raman, R.; Chow, T.W.; Rafii, M.S.; Sun, C.-K.; Rissman, R.A.; Donohue, M.C.; Brewer, J.B.; Jenkins, C.; Harless, K.; Gessert, D.; Aisen, P.S. Safety, Efficacy, and Feasibility of Intranasal Insulin for the Treatment of Mild Cognitive Impairment and Alzheimer Disease Dementia. JAMA Neurol 2020, 77, 1099. [Google Scholar] [CrossRef]
- Cukierman-Yaffe, T.; Kasher-Meron, M.; Fruchter, E.; Gerstein, H.C.; Afek, A.; Derazne, E.; Tzur, D.; Karasik, A.; Twig, G. Cognitive Performance at Late Adolescence and the Risk for Impaired Fasting Glucose Among Young Adults. J Clin Endocrinol Metab 2015, 100, 4409–4416. [Google Scholar] [CrossRef]
- De Felice, F.G.; Gonçalves, R.A.; Ferreira, S.T. Impaired insulin signalling and allostatic load in Alzheimer disease. Nat Rev Neurosci 2022, 23, 215–230. [Google Scholar] [CrossRef]
- de la Monte, S.M.; Wands, J.R. Alzheimer’s disease is type 3 diabetes-evidence reviewed. J Diabetes Sci Technol 2008, 2, 1101–13. [Google Scholar] [CrossRef]
- DeFronzo, R.A.; Hompesch, M.; Kasichayanula, S.; Liu, X.; Hong, Y.; Pfister, M.; Morrow, L.A.; Leslie, B.R.; Boulton, D.W.; Ching, A.; LaCreta, F.P.; Griffen, S.C. Characterization of Renal Glucose Reabsorption in Response to Dapagliflozin in Healthy Subjects and Subjects With Type 2 Diabetes. Diabetes Care 2013, 36, 3169–3176. [Google Scholar] [CrossRef]
- Delikkaya, B.; Moriel, N.; Tong, M.; Gallucci, G.; de la Monte, S.M. Altered expression of insulin-degrading enzyme and regulator of calcineurin in the rat intracerebral streptozotocin model and human apolipoprotein E-ε4–associated Alzheimer’s disease. Alzheimer’s & Dementia: Diagnosis, Assessment & Disease Monitoring 2019, 11, 392–404. [Google Scholar] [CrossRef]
- Demetrius, L.A.; Simon, D.K. An inverse-Warburg effect and the origin of Alzheimer’s disease. Biogerontology 2012, 13, 583–594. [Google Scholar] [CrossRef]
- Desouza, C.V.; Shivaswamy, V. Pioglitazone in the Treatment of Type 2 Diabetes: Safety and Efficacy Review. Clin Med Insights Endocrinol Diabetes 2010, 3, CMED.S5372. [Google Scholar] [CrossRef]
- DeTure, M.A.; Dickson, D.W. The neuropathological diagnosis of Alzheimer’s disease. Mol Neurodegener 2019, 14, 32. [Google Scholar] [CrossRef] [PubMed]
- Dolan, H.; Crain, B.; Troncoso, J.; Resnick, S.M.; Zonderman, A.B.; Obrien, R.J. Atherosclerosis, dementia, and Alzheimer disease in the Baltimore Longitudinal Study of aging cohort. Ann Neurol 2010, 68, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Domínguez, R.O.; Pagano, M.A.; Marschoff, E.R.; González, S.E.; Repetto, M.G.; Serra, J.A. Enfermedad de Alzheimer y deterioro cognitivo asociado a la diabetes mellitus de tipo 2: relaciones e hipótesis. Neurología 2014, 29, 567–572. [Google Scholar] [CrossRef]
- Donoso, S.A.; Behrens, P.M.I. Variabilidad y variantesde la enfermedad de Alzheimer. Rev Med Chil 2005, 133. [Google Scholar] [CrossRef]
- Duarte, A.I.; Moreira, P.I.; Oliveira, C.R. Insulin in Central Nervous System: More than Just a Peripheral Hormone. J Aging Res 2012, 2012, 1–21. [Google Scholar] [CrossRef]
- Duarte, J. Metabolic Alterations Associated to Brain Dysfunction in Diabetes. Aging Dis 2014. [Google Scholar] [CrossRef] [PubMed]
- Duarte, J.M.N. Loss of brain energy metabolism control as a driver for memory impairment upon insulin resistance. Biochem Soc Trans 2023, 51, 287–301. [Google Scholar] [CrossRef]
- Ehtewish, H.; Arredouani, A.; El-Agnaf, O. Diagnostic, Prognostic, and Mechanistic Biomarkers of Diabetes Mellitus-Associated Cognitive Decline. Int J Mol Sci 2022, 23, 6144. [Google Scholar] [CrossRef]
- El Khoury, N.B.; Gratuze, M.; Papon, M.-A.; Bretteville, A.; Planel, E. Insulin dysfunction and Tau pathology. Front Cell Neurosci 2014, 8. [Google Scholar] [CrossRef]
- Fava, S. Glycaemic Control: A Balancing Act or A Different Approach? Curr Diabetes Rev 2014, 10, 124–130. [Google Scholar] [CrossRef]
- Formiga, F.; Reñe, R.; Pérez-Maraver, M. Demencia y diabetes: ¿relación casual o causal? Med Clin (Barc) 2015, 144, 176–180. [Google Scholar] [CrossRef]
- Freychet, P. Insulin receptors and insulin actions in the nervous system. Diabetes Metab Res Rev 2000, 16, 390–392. [Google Scholar] [CrossRef] [PubMed]
- Gabbouj, S.; Ryhänen, S.; Marttinen, M.; Wittrahm, R.; Takalo, M.; Kemppainen, S.; Martiskainen, H.; Tanila, H.; Haapasalo, A.; Hiltunen, M.; Natunen, T. Altered Insulin Signaling in Alzheimer’s Disease Brain – Special Emphasis on PI3K-Akt Pathway. Front Neurosci 2019, 13. [Google Scholar] [CrossRef]
- Galea, I. The blood–brain barrier in systemic infection and inflammation. Cell Mol Immunol 2021, 18, 2489–2501. [Google Scholar] [CrossRef]
- García-Casares, N.; González-González, G.; de la Cruz-Cosme, C.; Garzón-Maldonado, F.J.; de Rojas-Leal, C.; Ariza, M.J.; Narváez, M.; Barbancho, M.Á.; García-Arnés, J.A.; Tinahones, F.J. Effects of GLP-1 receptor agonists on neurological complications of diabetes. Rev Endocr Metab Disord 2023, 24, 655–672. [Google Scholar] [CrossRef]
- Garcia-Serrano, A.M.; Duarte, J.M.N. Brain Metabolism Alterations in Type 2 Diabetes: What Did We Learn From Diet-Induced Diabetes Models? Front Neurosci 2020, 14, 229. [Google Scholar] [CrossRef] [PubMed]
- Gerritsen, A.A.J.; Bakker, C.; Verhey, F.R.J.; de Vugt, M.E.; Melis, R.J.F.; Koopmans, R.T.C.M.; Oosterveld, S.M.; Kessels, R.P.; Olde Rikkert, M.G.; Hamel, R.; Ramakers, I.H.; Aalten, P.; Sistermans, N.; Smits, L.L.; Pijnenburg, Y.A.; van der Flier, W.M. Prevalence of Comorbidity in Patients With Young-Onset Alzheimer Disease Compared With Late-Onset: A Comparative Cohort Study. J Am Med Dir Assoc 2016, 17, 318–323. [Google Scholar] [CrossRef]
- Gonçalves, R.A.; Wijesekara, N.; Fraser, P.E.; De Felice, F.G. The Link Between Tau and Insulin Signaling: Implications for Alzheimer’s Disease and Other Tauopathies. Front Cell Neurosci 2019, 13. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Luo, H.; Li, Z.; Feng, Y.; Liu, Z.; Chang, J. Metabolic Profile of Alzheimer’s Disease: Is 10-Hydroxy-2-decenoic Acid a Pertinent Metabolic Adjuster? Metabolites 2023, 13, 954. [Google Scholar] [CrossRef]
- Götz, J.; Ittner, L.M.; Lim, Y.-A. Common features between diabetes mellitus and Alzheimer’s disease. Cell Mol Life Sci 2009, 66, 1321–5. [Google Scholar] [CrossRef] [PubMed]
- Groot, C.; Cicognola, C.; Bali, D.; Triana-Baltzer, G.; Dage, J.L.; Pontecorvo, M.J.; Kolb, H.C.; Ossenkoppele, R.; Janelidze, S.; Hansson, O. Diagnostic and prognostic performance to detect Alzheimer’s disease and clinical progression of a novel assay for plasma p-tau217. Alzheimers Res Ther 2022, 14, 67. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Rodelo, C.; Roura-Guiberna, A.; Olivares-Reyes, J.A. Molecular Mechanisms of Insulin Resistance: An Update. Gac Med Mex 2017, 153, 214–228. [Google Scholar]
- Haass, C.; Selkoe, D.J. Soluble protein oligomers in neurodegeneration: lessons from the Alzheimer’s amyloid β-peptide. Nat Rev Mol Cell Biol 2007, 8, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Hameed, I.; Masoodi, S.R.; Mir, S.A.; Nabi, M.; Ghazanfar, K.; Ganai, B.A. Type 2 diabetes mellitus: From a metabolic disorder to an inflammatory condition. World J Diabetes 2015, 6, 598. [Google Scholar] [CrossRef]
- Han, Y.; Kim, S.Y. Endothelial senescence in vascular diseases: current understanding and future opportunities in senotherapeutics. Exp Mol Med 2023, 55, 1–12. [Google Scholar] [CrossRef]
- Hansson Petersen, C.A.; Alikhani, N.; Behbahani, H.; Wiehager, B.; Pavlov, P.F.; Alafuzoff, I.; Leinonen, V.; Ito, A.; Winblad, B.; Glaser, E.; Ankarcrona, M. The amyloid β-peptide is imported into mitochondria via the TOM import machinery and localized to mitochondrial cristae. Proceedings of the National Academy of Sciences 2008, 105, 13145–13150. [Google Scholar] [CrossRef]
- Hernández-Contreras, K.A.; Martínez-Díaz, J.A.; Hernández-Aguilar, M.E.; Herrera-Covarrubias, D.; Rojas-Durán, F.; Aranda Abreu, G.E. Mecanismos de asociación entre Enfermedad de Alzheimer y Diabetes Mellitus: La paradoja de la insulina. Archivos de Neurociencias 2021, 25. [Google Scholar] [CrossRef]
- Hobday, A.L.; Parmar, M.S. The Link Between Diabetes Mellitus and Tau Hyperphosphorylation: Implications for Risk of Alzheimer’s Disease; Cureus, 2021. [Google Scholar] [CrossRef]
- Horie, K.; Salvadó, G.; Barthélemy, N.R.; Janelidze, S.; Li, Y.; He, Y.; Saef, B.; Chen, C.D.; Jiang, H.; Strandberg, O.; Pichet Binette, A.; Palmqvist, S.; Sato, C.; Sachdev, P.; Koyama, A.; Gordon, B.A.; Benzinger, T.L.S.; Holtzman, D.M.; Morris, J.C.; Mattsson-Carlgren, N.; Stomrud, E.; Ossenkoppele, R.; Schindler, S.E.; Hansson, O.; Bateman, R.J. CSF MTBR-tau243 is a specific biomarker of tau tangle pathology in Alzheimer’s disease. Nat Med 2023, 29, 1954–1963. [Google Scholar] [CrossRef]
- Huang, C.-C.; Chung, C.-M.; Leu, H.-B.; Lin, L.-Y.; Chiu, C.-C.; Hsu, C.-Y.; Chiang, C.-H.; Huang, P.-H.; Chen, T.-J.; Lin, S.-J.; Chen, J.-W.; Chan, W.-L. Diabetes Mellitus and the Risk of Alzheimer’s Disease: A Nationwide Population-Based Study. PLoS One 2014, 9, e87095. [Google Scholar] [CrossRef]
- Huang, R.; Tian, S.; Zhang, H.; Zhu, W.; Wang, S. Chronic hyperglycemia induces tau hyperphosphorylation by downregulating OGT-involved O-GlcNAcylation in vivo and in vitro. Brain Res Bull 2020, 156, 76–85. [Google Scholar] [CrossRef]
- Jabeen, K.; Rehman, K.; Akash, M.S.H. Genetic mutations of APOEε4 carriers in cardiovascular patients lead to the development of insulin resistance and risk of Alzheimer’s disease. J Biochem Mol Toxicol 2022, 36. [Google Scholar] [CrossRef]
- Jais, A.; Brüning, J.C. Hypothalamic inflammation in obesity and metabolic disease. Journal of Clinical Investigation 2017, 127, 24–32. [Google Scholar] [CrossRef]
- Ke, Y.D.; Delerue, F.; Gladbach, A.; Götz, J.; Ittner, L.M. Experimental Diabetes Mellitus Exacerbates Tau Pathology in a Transgenic Mouse Model of Alzheimer’s Disease. PLoS One 2009, 4, e7917. [Google Scholar] [CrossRef]
- Kellar, D.; Craft, S. Brain insulin resistance in Alzheimer’s disease and related disorders: mechanisms and therapeutic approaches. Lancet Neurol 2020, 19, 758–766. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Backus, C.; Oh, S.; Feldman, E.L. Hyperglycemia-Induced Tau Cleavage in vitro and in vivo: A Possible Link Between Diabetes and Alzheimer’s Disease. Journal of Alzheimer’s Disease 2013, 34, 727–739. [Google Scholar] [CrossRef] [PubMed]
- Kimura, N.; Aota, T.; Aso, Y.; Yabuuchi, K.; Sasaki, K.; Masuda, T.; Eguchi, A.; Maeda, Y.; Aoshima, K.; Matsubara, E. Predicting positron emission tomography brain amyloid positivity using interpretable machine learning models with wearable sensor data and lifestyle factors. Alzheimers Res Ther 2023, 15, 212. [Google Scholar] [CrossRef]
- Kivisäkk, P.; Fatima, H.A.; Cahoon, D.S.; Otieno, B.; Chacko, L.; Minooei, F.; Demos, C.; Stengelin, M.; Sigal, G.; Wohlstadter, J.; Arnold, S.E. Clinical evaluation of a novel plasma pTau217 electrochemiluminescence immunoassay in Alzheimer’s disease. Sci Rep 2024, 14, 629. [Google Scholar] [CrossRef] [PubMed]
- Klausen, M.K.; Thomsen, M.; Wortwein, G.; Fink-Jensen, A. The role of glucagon-like peptide 1 (GLP-1) in addictive disorders. Br J Pharmacol 2022, 179, 625–641. [Google Scholar] [CrossRef]
- Kleinridders, A.; Ferris, H.A.; Cai, W.; Kahn, C.R. Insulin Action in Brain Regulates Systemic Metabolism and Brain Function. Diabetes 2014, 63, 2232–2243. [Google Scholar] [CrossRef]
- Knopman, D.S.; Jack, C.R.; Wiste, H.J.; Lundt, E.S.; Weigand, S.D.; Vemuri, P.; Lowe, V.J.; Kantarci, K.; Gunter, J.L.; Senjem, M.L.; Mielke, M.M.; Roberts, R.O.; Boeve, B.F.; Petersen, R.C. 18F-fluorodeoxyglucose positron emission tomography, aging, and apolipoprotein E genotype in cognitively normal persons. Neurobiol Aging 2014, 35, 2096–2106. [Google Scholar] [CrossRef]
- Kong, Y.; Wang, F.; Wang, J.; Liu, C.; Zhou, Y.; Xu, Z.; Zhang, C.; Sun, B.; Guan, Y. Pathological Mechanisms Linking Diabetes Mellitus and Alzheimer’s Disease: the Receptor for Advanced Glycation End Products (RAGE). Front Aging Neurosci 2020, 12. [Google Scholar] [CrossRef]
- Könner, A.C.; Hess, S.; Tovar, S.; Mesaros, A.; Sánchez-Lasheras, C.; Evers, N.; Verhagen, L.A.W.; Brönneke, H.S.; Kleinridders, A.; Hampel, B.; Kloppenburg, P.; Brüning, J.C. Role for Insulin Signaling in Catecholaminergic Neurons in Control of Energy Homeostasis. Cell Metab 2011, 13, 720–728. [Google Scholar] [CrossRef]
- Lamport, D.J.; Dye, L.; Mansfield, M.W.; Lawton, C.L. Acute glycaemic load breakfast manipulations do not attenuate cognitive impairments in adults with type 2 diabetes. Clinical Nutrition 2013, 32, 265–272. [Google Scholar] [CrossRef]
- Lamport, D.J.; Lawton, C.L.; Mansfield, M.W.; Moulin, C.A.J.; Dye, L. Type 2 diabetes and impaired glucose tolerance are associated with word memory source monitoring recollection deficits but not simple recognition familiarity deficits following water, low glycaemic load, and high glycaemic load breakfasts. Physiol Behav 2014, 124, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Langella, S.; Barksdale, N.G.; Vasquez, D.; Aguillon, D.; Chen, Y.; Su, Y.; Acosta-Baena, N.; Acosta-Uribe, J.; Baena, A.Y.; Garcia-Ospina, G.; Giraldo-Chica, M.; Tirado, V.; Muñoz, C.; Ríos-Romenets, S.; Guzman-Martínez, C.; Oliveira, G.; Yang, H.-S.; Vila-Castelar, C.; Pruzin, J.J.; Ghisays, V.; Arboleda-Velasquez, J.F.; Kosik, K.S.; Reiman, E.M.; Lopera, F.; Quiroz, Y.T. Effect of apolipoprotein genotype and educational attainment on cognitive function in autosomal dominant Alzheimer’s disease. Nat Commun 2023, 14, 5120. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.C.; Choe, Y.M.; Suh, G.-H.; Choi, I.-G.; Lee, J.H.; Kim, H.S.; Hwang, J.; Yi, D.; Kim, J.W. A combination of midlife diabetes mellitus and the apolipoprotein E ε4 allele increase risk for cognitive decline. Front Aging Neurosci 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Hölscher, C. Common pathological processes in Alzheimer disease and type 2 diabetes: a review. Brain Res Rev 2007, 56, 384–402. [Google Scholar] [CrossRef]
- Lim, Y.Y.; Yassi, N.; Bransby, L.; Ayton, S.; Buckley, R.F.; Eratne, D.; Velakoulis, D.; Li, Q.-X.; Fowler, C.; Masters, C.L.; Maruff, P. CSF Aβ42 and tau biomarkers in cognitively unimpaired Aβ- middle-aged and older APOE ε4 carriers. Neurobiol Aging 2023, 129, 209–218. [Google Scholar] [CrossRef]
- Liu, C.; Wang, Z.; Song, Y.; Wu, D.; Zheng, X.; Li, P.; Jin, J.; Xu, N.; Li, L. Effects of Berberine on Amelioration of Hyperglycemia and Oxidative Stress in High Glucose and High Fat Diet-Induced Diabetic Hamsters In Vivo. Biomed Res Int 2015, 2015, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Chu, B.; Jin, S.; Li, M.; Xu, Y.; Yang, H.; Feng, Z.; Bi, J.; Wang, P. Vascular endothelial growth factor alleviates mitochondrial dysfunction and suppression of mitochondrial biogenesis in models of Alzheimer’s disease. International Journal of Neuroscience 2021, 131, 154–162. [Google Scholar] [CrossRef]
- Liu, Y.; Jiang, Y.; Du, W.; Gao, B.; Gao, J.; Hu, S.; Song, Q.; Wang, W.; Miao, Y. White matter microstructure alterations in type 2 diabetes mellitus and its correlation with cerebral small vessel disease and cognitive performance. Sci Rep 2024, 14, 270. [Google Scholar] [CrossRef]
- Luna, R.; Talanki Manjunatha, R.; Bollu, B.; Jhaveri, S.; Avanthika, C.; Reddy, N.; Saha, T.; Gandhi, F. A Comprehensive Review of Neuronal Changes in Diabetics. Cureus. [CrossRef] [PubMed]
- Madhusudhanan, J.; Suresh, G.; Devanathan, V. Neurodegeneration in type 2 diabetes: Alzheimer’s as a case study. Brain Behav 2021, 2020, 10. [Google Scholar] [CrossRef]
- Majid, H.; Islam, S.U.; Kohli, S.; Nidhi. Neuroinflammation and metabolic dysregulation as predictors of cognitive impairment, depression, and quality of life in type 2 diabetes mellitus patients on SGLT2 inhibitors and sulfonylureas. Inflammopharmacology 2025. [Google Scholar] [CrossRef] [PubMed]
- Malenka, R.C. Synaptic plasticity in the hippocampus: LTP and LTD. Cell 1994, 78, 535–538. [Google Scholar] [CrossRef]
- Mancinetti, F.; Xenos, D.; De Fano, M.; Mazzieri, A.; Porcellati, F.; Boccardi, V.; Mecocci, P. Diabetes-Alzheimer’s connection in older age: SGLT2 inhibitors as promising modulators of disease pathways. Ageing Res Rev 2023, 90, 102018. [Google Scholar] [CrossRef]
- Martínez, S.; Ochoa, B.; Pérez, M.R.; Torrico, F.; García, I.; Garcia, C.C. Polimorfismos del gen de la apolipoproteína E en adultos mayores de 60 años con disminución de la memoria cognitiva y enfermedad de Alzheimer en diferentes poblaciones venezolanas. Biomédica 2022, 42, 116–129. [Google Scholar] [CrossRef]
- McNay, E.C.; Ong, C.T.; McCrimmon, R.J.; Cresswell, J.; Bogan, J.S.; Sherwin, R.S. Hippocampal memory processes are modulated by insulin and high-fat-induced insulin resistance. Neurobiol Learn Mem 2010, 93, 546–553. [Google Scholar] [CrossRef] [PubMed]
- Messier, C.; Gagnon, M. Glucose regulation and cognitive functions: relation to Alzheimer’s disease and diabetes. Behavioural Brain Research 1996, 75, 1–11. [Google Scholar] [CrossRef]
- Mestizo Gutiérrez, S.L.; Herrera Rivero, M.; Cruz Ramírez, N.; Hernández, E.; Aranda-Abreu, G.E. Decision trees for the analysis of genes involved in Alzheimer׳s disease pathology. J Theor Biol 2014, 357, 21–25. [Google Scholar] [CrossRef]
- Mietelska-Porowska, A.; Wasik, U.; Goras, M.; Filipek, A.; Niewiadomska, G. Tau Protein Modifications and Interactions: Their Role in Function and Dysfunction. Int J Mol Sci 2014, 15, 4671–4713. [Google Scholar] [CrossRef]
- Milà-Alomà, M.; Ashton, N.J.; Shekari, M.; Salvadó, G.; Ortiz-Romero, P.; Montoliu-Gaya, L.; Benedet, A.L.; Karikari, T.K.; Lantero-Rodriguez, J.; Vanmechelen, E.; Day, T.A.; González-Escalante, A.; Sánchez-Benavides, G.; Minguillon, C.; Fauria, K.; Molinuevo, J.L.; Dage, J.L.; Zetterberg, H.; Gispert, J.D.; Suárez-Calvet, M.; Blennow, K. Plasma p-tau231 and p-tau217 as state markers of amyloid-β pathology in preclinical Alzheimer’s disease. Nat Med 2022. [Google Scholar] [CrossRef]
- Milà-Alomà, M.; Salvadó, G.; Gispert, J.D.; Vilor-Tejedor, N.; Grau-Rivera, O.; Sala-Vila, A.; Sánchez-Benavides, G.; Arenaza-Urquijo, E.M.; Crous-Bou, M.; González-de-Echávarri, J.M.; Minguillon, C.; Fauria, K.; Simon, M.; Kollmorgen, G.; Zetterberg, H.; Blennow, K.; Suárez-Calvet, M.; Molinuevo, J.L. Amyloid beta, tau, synaptic, neurodegeneration, and glial biomarkers in the preclinical stage of the Alzheimer’s continuum. Alzheimer’s & Dementia 2020, 16, 1358–1371. [Google Scholar] [CrossRef]
- Mittal, M.; Siddiqui, M.R.; Tran, K.; Reddy, S.P.; Malik, A.B. Reactive Oxygen Species in Inflammation and Tissue Injury. Antioxid Redox Signal 2014, 20, 1126–1167. [Google Scholar] [CrossRef] [PubMed]
- Mullins, R.J.; Diehl, T.C.; Chia, C.W.; Kapogiannis, D. Insulin Resistance as a Link between Amyloid-Beta and Tau Pathologies in Alzheimer’s Disease. Front Aging Neurosci 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Naia, L.; Shimozawa, M.; Bereczki, E.; Li, X.; Liu, J.; Jiang, R.; Giraud, R.; Leal, N.S.; Pinho, C.M.; Berger, E.; Falk, V.L.; Dentoni, G.; Ankarcrona, M.; Nilsson, P. Mitochondrial hypermetabolism precedes impaired autophagy and synaptic disorganization in App knock-in Alzheimer mouse models. Mol Psychiatry 2023, 28, 3966–3981. [Google Scholar] [CrossRef]
- Olesen, M.A.; Quintanilla, R.A. Pathological Impact of Tau Proteolytical Process on Neuronal and Mitochondrial Function: a Crucial Role in Alzheimer’s Disease. Mol Neurobiol 2023, 60, 5691–5707. [Google Scholar] [CrossRef]
- Palasí, A.; Gutiérrez-Iglesias, B.; Alegret, M.; Pujadas, F.; Olabarrieta, M.; Liébana, D.; Quintana, M.; Álvarez-Sabín, J.; Boada, M. Differentiated clinical presentation of early and late-onset Alzheimer’s disease: is 65 years of age providing a reliable threshold? J Neurol 2015, 262, 1238–1246. [Google Scholar] [CrossRef]
- Park, S.A. A common pathogenic mechanism linking type-2 diabetes and Alzheimer’s disease: evidence from animal models. J Clin Neurol 2011, 7, 10–8. [Google Scholar] [CrossRef] [PubMed]
- Pasquier, F.; Boulogne, A.; Leys, D.; Fontaine, P. Diabetes mellitus and dementia. Diabetes Metab 2006, 32, 403–14. [Google Scholar] [CrossRef]
- Pérez, M.J.; Jara, C.; Quintanilla, R.A. Contribution of Tau Pathology to Mitochondrial Impairment in Neurodegeneration. Front Neurosci 2018, 12. [Google Scholar] [CrossRef]
- Petermann, F.; Troncoso-Pantoja, C.; Martínez, M.A.; Leiva, A.M.; Ramírez-Campillo, R.; Poblete-Valderrama, F.; Garrido-Méndez, A.; Díaz-Martínez, X.; Ulloa, N.; Concha, Y.; Celis-Morales, C. Asociación entre diabetes mellitus tipo 2018, 2, historia familiar de diabetes y deterioro cognitivo en adultos mayores chilenos. Rev Med Chil 146, 872–881. [CrossRef] [PubMed]
- Polsinelli, A.J.; Logan, P.E.; Lane, K.A.; Manchella, M.K.; Nemes, S.; Sanjay, A.B.; Gao, S.; Apostolova, L.G. APOE ε4 carrier status and sex differentiate rates of cognitive decline in early- and late-onset Alzheimer’s disease. Alzheimer’s & Dementia 2023, 19, 1983–1993. [Google Scholar] [CrossRef]
- Pomytkin, I.; Costa-Nunes, J.P.; Kasatkin, V.; Veniaminova, E.; Demchenko, A.; Lyundup, A.; Lesch, K.; Ponomarev, E.D.; Strekalova, T. Insulin receptor in the brain: Mechanisms of activation and the role in the CNS pathology and treatment. CNS Neurosci Ther 2018, 24, 763–774. [Google Scholar] [CrossRef] [PubMed]
- Pooja Naik, L.C. Diabetes Mellitus and Blood-Brain Barrier Dysfunction: An Overview. J Pharmacovigil 2014, 02. [Google Scholar] [CrossRef]
- Ramírez Rincón, A.; Saldarriaga Betancur, S.; García Ramos, A.F.; González Arango, J.; Estupiñán Vargas, V. Tratamiento farmacológico del paciente que vive con diabetes mellitus tipo 2. CES Medicina 2022, 36, 81–105. [Google Scholar] [CrossRef]
- Rasool, M.; Malik, A.; Waquar, S.; Zaheer, A.; Asif, M.; Iqbal, Z.; Gauthaman, K.; Kamal, M.A.; Pushparaj, P.N. Cellular and Molecular Mechanisms of Dementia: Decoding the Causal link of Diabetes Mellitus in Alzheimer’s Disease. CNS Neurol Disord Drug Targets 2021, 20, 602–612. [Google Scholar] [CrossRef]
- Raulin, A.-C.; Doss, S.V.; Trottier, Z.A.; Ikezu, T.C.; Bu, G.; Liu, C.-C. ApoE in Alzheimer’s disease: pathophysiology and therapeutic strategies. Mol Neurodegener 2022, 17, 72. [Google Scholar] [CrossRef]
- Ravipati, K.; Chen, Y.; Manns, J.R. Reassessing Diabetes and APOE Genotype as Potential Interacting Risk Factors for Alzheimer’s Disease. Am J Alzheimers Dis Other Demen 2022, 37, 153331752110709. [Google Scholar] [CrossRef]
- Razay, G.; Wilcock, G.K. Hyperinsulinaemia and Alzheimer’s disease. Age Ageing 1994, 23, 396–9. [Google Scholar] [CrossRef] [PubMed]
- Rebeck, G.W.; Kindy, M.; LaDu, M.J. Apolipoprotein E and Alzheimer’s disease: The protective effects of ApoE2 and E3. Journal of Alzheimer’s Disease 2002, 4, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Meza, M.; Muñoz, D.; Jerez, E.; Quintanilla, M.E.; Salinas-Luypaert, C.; Fernandez, K.; Karahanian, E. Fenofibrate Administration Reduces Alcohol and Saccharin Intake in Rats: Possible Effects at Peripheral and Central Levels. Front Behav Neurosci 2017, 11. [Google Scholar] [CrossRef] [PubMed]
- Roda, A.R.; Montoliu-Gaya, L.; Villegas, S. The Role of Apolipoprotein E Isoforms in Alzheimer’s Disease. Journal of Alzheimer’s Disease 2019, 68, 459–471. [Google Scholar] [CrossRef]
- Saini, V. Molecular mechanisms of insulin resistance in type 2 diabetes mellitus. World J Diabetes 2010, 1, 68–75. [Google Scholar] [CrossRef]
- Sánchez-Zúñiga, M. de J.; Carrillo-Esper, R.; Sánchez-Pérez, H.; González-Chávez, A.; Elizondo-Argueta, S. Circuito insulinérgico cerebral. De las bases a su impacto en la clínica. Cir Cir 2020, 88. [Google Scholar] [CrossRef]
- Saraya, A.W.; Tunvirachaisakul, C.; Sonpee, C.; Katasrila, P.; Sathaporn, T.; Tepmongkol, S.; Tangwongchai, S. Serum proinsulin levels as peripheral blood biomarkers in patients with cognitive impairment. Sci Rep 2023, 13, 22436. [Google Scholar] [CrossRef]
- Scheltens, P.; De Strooper, B.; Kivipelto, M.; Holstege, H.; Chételat, G.; Teunissen, C.E.; Cummings, J.; van der Flier, W.M. Alzheimer’s disease. The Lancet 2021, 397, 1577–1590. [Google Scholar] [CrossRef]
- Sebastian, M.J.; Khan, S.K.; Pappachan, J.M.; Jeeyavudeen, M.S. Diabetes and cognitive function: An evidence-based current perspective. World J Diabetes 2023, 14, 92–109. [Google Scholar] [CrossRef]
- Shinohara, M.; Suzuki, K.; Bu, G.; Sato, N. Interaction Between APOE Genotype and Diabetes in Longevity. Journal of Alzheimer’s Disease 2021, 82, 719–726. [Google Scholar] [CrossRef]
- Sible, I.J.; Yew, B.; Jang, J.Y.; Alitin, J.P.M.; Li, Y.; Gaubert, A.; Nguyen, A.; Dutt, S.; Blanken, A.E.; Ho, J.K.; Marshall, A.J.; Kapoor, A.; Shenasa, F.; Rodgers, K.E.; Sturm, V.E.; Head, E.; Martini, A.; Nation, D.A. Blood pressure variability and plasma Alzheimer’s disease biomarkers in older adults. Sci Rep 2022, 12, 17197. [Google Scholar] [CrossRef] [PubMed]
- Sienes Bailo, P.; Llorente Martín, E.; Calmarza, P.; Montolio Breva, S.; Bravo Gómez, A.; Pozo Giráldez, A.; Sánchez-Pascuala Callau, J.J.; Vaquer Santamaría, J.M.; Dayaldasani Khialani, A.; Cerdá Micó, C.; Camps Andreu, J.; Sáez Tormo, G.; Fort Gallifa, I. Implicación del estrés oxidativo en las enfermedades neurodegenerativas y posibles terapias antioxidantes. Advances in Laboratory Medicine / Avances en Medicina de Laboratorio 2022, 3, 351–360. [Google Scholar] [CrossRef]
- Sima, A.A.F.; Zhang, W. Mechanisms of diabetic neuropathy; 2014; pp. 429–442. [Google Scholar] [CrossRef]
- Sims-Robinson, C.; Kim, B.; Rosko, A.; Feldman, E.L. How does diabetes accelerate Alzheimer disease pathology? Nat Rev Neurol 2010, 6, 551–559. [Google Scholar] [CrossRef] [PubMed]
- Snyder, P.J. Introducing Alzheimer’s & Dementia: Diagnosis, Assessment & Disease Monitoring, an open access journal of the Alzheimer’s Association. Alzheimer’s & Dementia: Diagnosis, Assessment & Disease Monitoring 2015, 1, 1–4. [Google Scholar] [CrossRef]
- Sonar, S.A.; Lal, G. Blood–brain barrier and its function during inflammation and autoimmunity. J Leukoc Biol 2018, 103, 839–853. [Google Scholar] [CrossRef]
- Stanciu, G.D.; Bild, V.; Ababei, D.C.; Rusu, R.N.; Cobzaru, A.; Paduraru, L.; Bulea, D. Link between Diabetes and Alzheimer’s Disease Due to the Shared Amyloid Aggregation and Deposition Involving Both Neurodegenerative Changes and Neurovascular Damages. J Clin Med 2020, 9, 1713. [Google Scholar] [CrossRef]
- Steen, E.; Terry, B.M.; Rivera, E.J.; Cannon, J.L.; Neely, T.R.; Tavares, R.; Xu, X.J.; Wands, J.R.; de la Monte, S.M. Impaired insulin and insulin-like growth factor expression and signaling mechanisms in Alzheimer’s disease – is this type 3 diabetes? Journal of Alzheimer’s Disease 2005, 7, 63–80. [Google Scholar] [CrossRef]
- Tabeshmehr, P.; Eftekharpour, E. Tau; One Protein, So Many Diseases. Biology (Basel) 2023, 12, 244. [Google Scholar] [CrossRef]
- Takeishi, J.; Tatewaki, Y.; Nakase, T.; Takano, Y.; Tomita, N.; Yamamoto, S.; Mutoh, T.; Taki, Y. Alzheimer’s Disease and Type 2 Diabetes Mellitus: The Use of MCT Oil and a Ketogenic Diet. Int J Mol Sci 2021, 22, 12310. [Google Scholar] [CrossRef]
- Talbot, K.; Wang, H.-Y.; Kazi, H.; Han, L.-Y.; Bakshi, K.P.; Stucky, A.; Fuino, R.L.; Kawaguchi, K.R.; Samoyedny, A.J.; Wilson, R.S.; Arvanitakis, Z.; Schneider, J.A.; Wolf, B.A.; Bennett, D.A.; Trojanowski, J.Q.; Arnold, S.E. Demonstrated brain insulin resistance in Alzheimer’s disease patients is associated with IGF-1 resistance, IRS-1 dysregulation, and cognitive decline. Journal of Clinical Investigation 2012, 122, 1316–1338. [Google Scholar] [CrossRef]
- Tobon-Velasco, J.; Cuevas, E.; Torres-Ramos, M. Receptor for AGEs (RAGE) as Mediator of NF-kB Pathway Activation in Neuroinflammation and Oxidative Stress. CNS Neurol Disord Drug Targets 2014, 13, 1615–1626. [Google Scholar] [CrossRef] [PubMed]
- Trimm, E.; Red-Horse, K. Vascular endothelial cell development and diversity. Nat Rev Cardiol 2023, 20, 197–210. [Google Scholar] [CrossRef] [PubMed]
- Tumminia, A.; Vinciguerra, F.; Parisi, M.; Frittitta, L. Type 2 Diabetes Mellitus and Alzheimer’s Disease: Role of Insulin Signalling and Therapeutic Implications. Int J Mol Sci 2018, 19, 3306. [Google Scholar] [CrossRef] [PubMed]
- Uribarri, J.; Buchman, A.S.; Cai, W.; Haroutunian, V.; Beeri, M.S. Human brain and serum advanced glycation end products are highly correlated: Preliminary results of their role in Alzheimer’s disease and type 2 diabetes. Alzheimer’s & Dementia 2020, 16. [Google Scholar] [CrossRef]
- Vanhandsaeme, G.; Benhalima, K. The long-term metabolic and neurocognitive risks in offspring of women with type 1 diabetes mellitus. Acta Diabetol 2021, 58, 845–858. [Google Scholar] [CrossRef]
- Verdelho, A.; Madureira, S.; Ferro, J.M.; Basile, A.-M.; Chabriat, H.; Erkinjuntti, T.; Fazekas, F.; Hennerici, M.; O’Brien, J.; Pantoni, L.; Salvadori, E.; Scheltens, P.; Visser, M.C.; Wahlund, L.-O.; Waldemar, G.; Wallin, A.; Inzitari, D.; LADIS Study. Differential impact of cerebral white matter changes, diabetes, hypertension and stroke on cognitive performance among non-disabled elderly. The LADIS study. J Neurol Neurosurg Psychiatry 2007, 78, 1325–30. [Google Scholar] [CrossRef]
- Verdile, G.; Fuller, S.J.; Martins, R.N. The role of type 2 diabetes in neurodegeneration. Neurobiol Dis 2015, 84, 22–38. [Google Scholar] [CrossRef]
- von Bernhardi, R. La Barrera Hemato-Encefálica en la patología del Sistema Nervioso Central: su importancia en la Respuesta Inflamatoria. Rev Chil Neuropsiquiatr 2004, 42. [Google Scholar] [CrossRef]
- Wang, S.; Wei, W.; Zheng, Y.; Hou, J.; Dou, Y.; Zhang, S.; Luo, X.; Cai, X. The Role of Insulin C-Peptide in the Coevolution Analyses of the Insulin Signaling Pathway: A Hint for Its Functions. PLoS One 2012, 7, e52847. [Google Scholar] [CrossRef]
- Watson, D.; Castaño, E.; Kokjohn, T.A.; Kuo, Y.-M.; Lyubchenko, Y.; Pinsky, D.; Connolly, E.S.; Esh, C.; Luehrs, D.C.; Stine, W.B.; Rowse, L.M.; Emmerling, M.R.; Roher, A.E. Physicochemical characteristics of soluble oligomeric A β and their pathologic role in Alzheimer’s disease. Neurol Res 2005, 27, 869–881. [Google Scholar] [CrossRef]
- Willette, A.A.; Modanlo, N.; Kapogiannis, D.; Alzheimer’s Disease Neuroimaging Initiative. Insulin resistance predicts medial temporal hypermetabolism in mild cognitive impairment conversion to Alzheimer disease. Diabetes 2015, 64, 1933–40. [Google Scholar] [CrossRef]
- Winocur, G.; Greenwood, C.E. Studies of the effects of high fat diets on cognitive function in a rat model. Neurobiol Aging 2005, 26 1, 46–9. [Google Scholar] [CrossRef]
- Woodfield, A.; Gonzales, T.; Helmerhorst, E.; Laws, S.; Newsholme, P.; Porter, T.; Verdile, G. Current Insights on the Use of Insulin and the Potential Use of Insulin Mimetics in Targeting Insulin Signalling in Alzheimer’s Disease. Int J Mol Sci 2022, 23, 15811. [Google Scholar] [CrossRef] [PubMed]
- Xourgia, E.; Papazafiropoulou, A.; Melidonis, A. Antidiabetic treatment on memory and spatial learning: From the pancreas to the neuron. World J Diabetes 2019, 10, 169–180. [Google Scholar] [CrossRef]
- Yaffe, K. The Metabolic Syndrome, Inflammation, and Risk of Cognitive Decline. JAMA 2004, 292, 2237. [Google Scholar] [CrossRef]
- Yang, C.; Wei, M.; Zhao, Y.; Yang, Z.; Song, M.; Mi, J.; Yang, X.; Tian, G. Regulation of insulin secretion by the post-translational modifications. Front Cell Dev Biol 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Maji, S.; Sanghera, N.; Gopalasingam, P.; Gorbunov, E.; Tarasov, S.; Epstein, O.; Klein-Seetharaman, J. Structure and dynamics of the insulin receptor: implications for receptor activation and drug discovery. Drug Discov Today 2017, 22, 1092–1102. [Google Scholar] [CrossRef] [PubMed]
- Zapata-Tragodara, D.; Roque, H.; Parodi, J.F. Relación entre trastornos neurocognitivos,Diabetes Mellitus Tipo 2 y otros factores en adultos mayores del Centro Médico Naval del Perú, entre los años 2010 a 2015. Rev Neuropsiquiatr 2020, 83, 87–96. [Google Scholar] [CrossRef]
- Zhang, H.; Wei, W.; Zhao, M.; Ma, L.; Jiang, X.; Pei, H.; Cao, Y.; Li, H. Interaction between Aβ and Tau in the Pathogenesis of Alzheimer’s Disease. Int J Biol Sci 2021, 17, 2181–2192. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




