1. Introduction
Three-dimensional (3D) bioprinting has profoundly impacted personalized medicine by enabling the development of patient-specific devices, drug delivery systems, and tissue constructs, thus facilitating a shift from standardized to customized healthcare solutions [
1,
2,
3]. This technological innovation is particularly significant in the fields of regenerative medicine and tissue engineering, where there is a pressing demand for structures that replicate the structural and functional complexities of native tissues [
4,
5]. Polysaccharide-based hydrogels have emerged as exemplary bioink candidates due to their sustainability, biocompatibility, and adjustable physicochemical properties [
6,
7]. These hydrogels are derived from abundant and renewable sources, including seaweed, plants, bacteria, and animals [
8,
9]. Their high-water content and structural similarity to glycosaminoglycans in the native extracellular matrix (ECM) provide a conducive microenvironment for cell encapsulation, viability, and function, aligning with principles of green manufacturing [
6,
7,
10].
Despite these compelling advantages, translating polysaccharide hydrogels into reliable bioinks remains challenging. Issues of mechanical robustness, precise rheological control, batch-to-batch variability, and often inadequate bioactivity complicate their adaptation to diverse printing techniques such as extrusion, inkjet, and stereolithography [
11,
12,
13]. While extensive research has optimized individual formulations, such as alginate blends for extrusion or methacrylated derivatives for vat photopolymerization, the field lacks integrative and systematic analyses. There is a pressing need to quantify the complex interrelationships between polysaccharide chemistry, ink rheology, printability parameters, and the ultimate biological outcomes. This gap hinders the establishment of generalizable design rules and the development of next-generation bioinks.
This comprehensive review endeavors to synthesize and critically evaluate the current state of polysaccharide-based bioinks for 3D bioprinting in the medical field. We present a detailed classification of polysaccharides according to their sources, examine their fundamental properties within the context of bioprinting, and assess their performance across both established and emerging fabrication platforms. Furthermore, we analyze the crucial roles of crosslinking methodologies and rheological designs in the development of functional constructs. By identifying key challenges and synergies, this review aims to serve as a comprehensive resource that informs the rational design of advanced polysaccharide bioinks, thereby facilitating their successful integration into clinically viable tissue engineering and regenerative medicine strategies.
2. Polysaccharide-Based Bioinks: Fundamentals and Properties
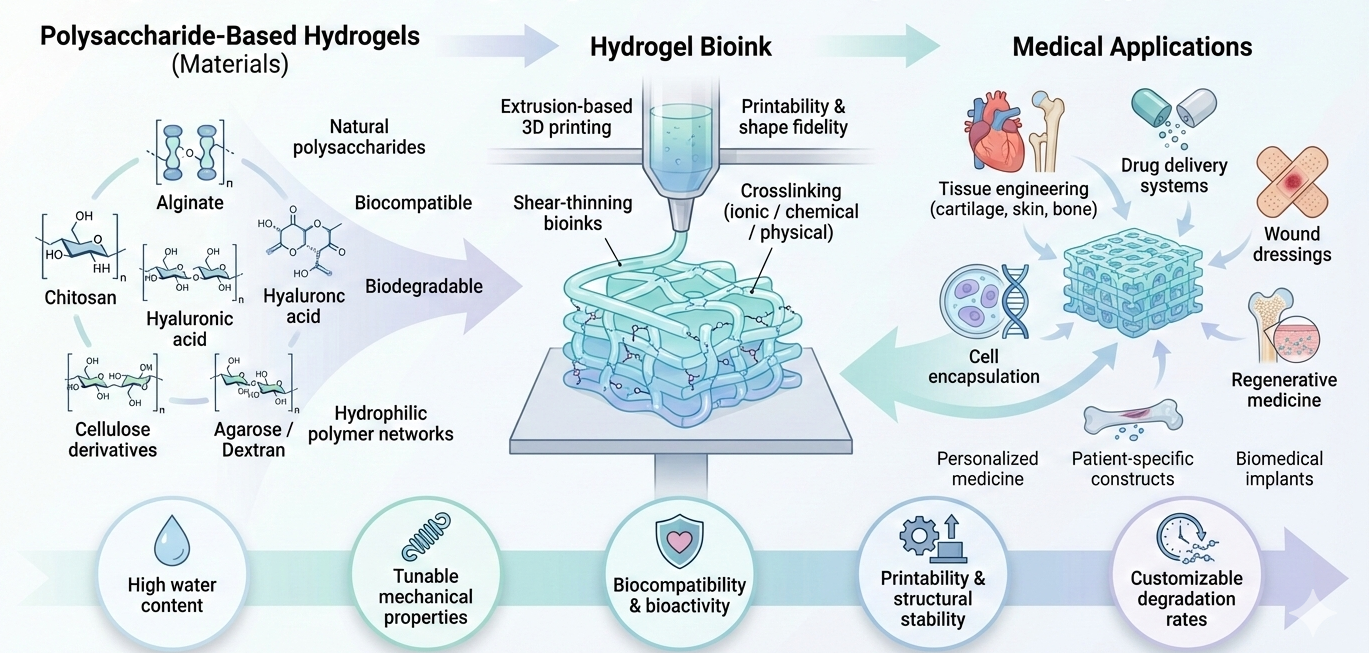
2.1. 3D Bioprinting with Hydrogels
Hydrogels are polymer networks characterized by their ability to swell in water and by a three-dimensional crosslinked structure that preserves their integrity despite substantial absorption of aqueous solutions [
14,
15]. In the context of medical 3D bioprinting, hydrogels are frequently employed as bioink matrices because of their soft, moisture-rich composition, which effectively mimics the essential physical properties of the native extracellular matrix (ECM) surrounding the cells (as shown in
Figure 1) [
16,
17,
18].
2.1.1. Hydration and Diffusion
Hydrogels are distinguished by their substantial water content, often surpassing 90% [
6,
19]. This degree of hydration is critical for their biological efficacy. The porous, hydrated biopolymer structure emulates the native extracellular matrix (ECM), thereby creating an environment conducive to essential cellular activities [
9,
20]. It facilitates the inward flow of oxygen, nutrients, and regulatory biomolecules to the embedded cells while permitting the efflux of metabolic waste products. Such passive mass transport is vital for maintaining cell viability, particularly in large, thick 3D bioprinted structures intended for clinical applications [
9,
21,
22]. In the absence of adequate diffusivity, cells located deep within a printed scaffold are susceptible to rapid necrosis. However, the highly hydrated and open architecture that enhances diffusion generally compromises mechanical strength. Consequently, a primary challenge in bioink development is to achieve an appropriate balance between maintaining sufficient porosity for effective mass transport and ensuring adequate mechanical stability to withstand printing stresses and functional loads in vivo [
17,
23].
2.1.2. Biocompatibility
Biocompatibility is a critical criterion for materials intended for implantation [
24]. Natural hydrogels derived from polysaccharides, such as alginate, chitosan, hyaluronic acid, and cellulose-based systems, are often preferred due to their biological origins, including seaweeds, crustacean shells, and connective tissues [
20,
25]. These materials generally exhibit chemical properties that are compatible with living tissues, leading to minimal cytotoxic effects and a reduced likelihood of significant inflammatory or immune responses [
24,
26]. This inherent compatibility supports essential cellular processes, including adhesion, proliferation, and lineage-specific differentiation [
9]. However, the term “natural” should not be misconstrued as synonymous with biological inertness, as impurities from raw material extraction, reactive groups introduced during functionalization for printing (such as methacrylation), and residual crosslinking agents can provoke adverse biological responses [
6]. Therefore, achieving genuine biocompatibility in the context of 3D bioprinting is an engineered attribute that relies on stringent purification methods and the judicious selection of mild, cytocompatible crosslinking techniques, rather than being an assured outcome of utilizing natural polymers [
27].
2.1.3. Tunability
Hydrogels are highly adaptable for biomedical applications, with their mechanical properties, such as stiffness, elasticity, and toughness, and degradation rates being modifiable through adjustments in parameters like polymer concentration, degree of crosslinking, and network structure [
9]. This flexibility facilitates the creation of scaffolds tailored to the specific needs of various tissues and organs. For instance, hydrogels with low moduli ranging from 0.1 to 1 kPa can be engineered to replicate brain tissue for neural regeneration, whereas stiffer hydrogels in the range of 10–100 kPa are more appropriate for supporting cartilage or early bone formation [
28]. Additionally, the degradation rate can be fine-tuned to align with the pace of new tissue development, enabling the scaffold to offer temporary support before safely degrading [
9,
29]. In the realm of 3D bioprinting, this adaptability permits variations in processing parameters to enhance printability. Modifications in factors such as viscosity and gel stiffness can improve material extrusion, shape retention, and post-printing stability [
9,
11]. The primary challenge lies in adjusting these properties without hindering cell growth and differentiation.
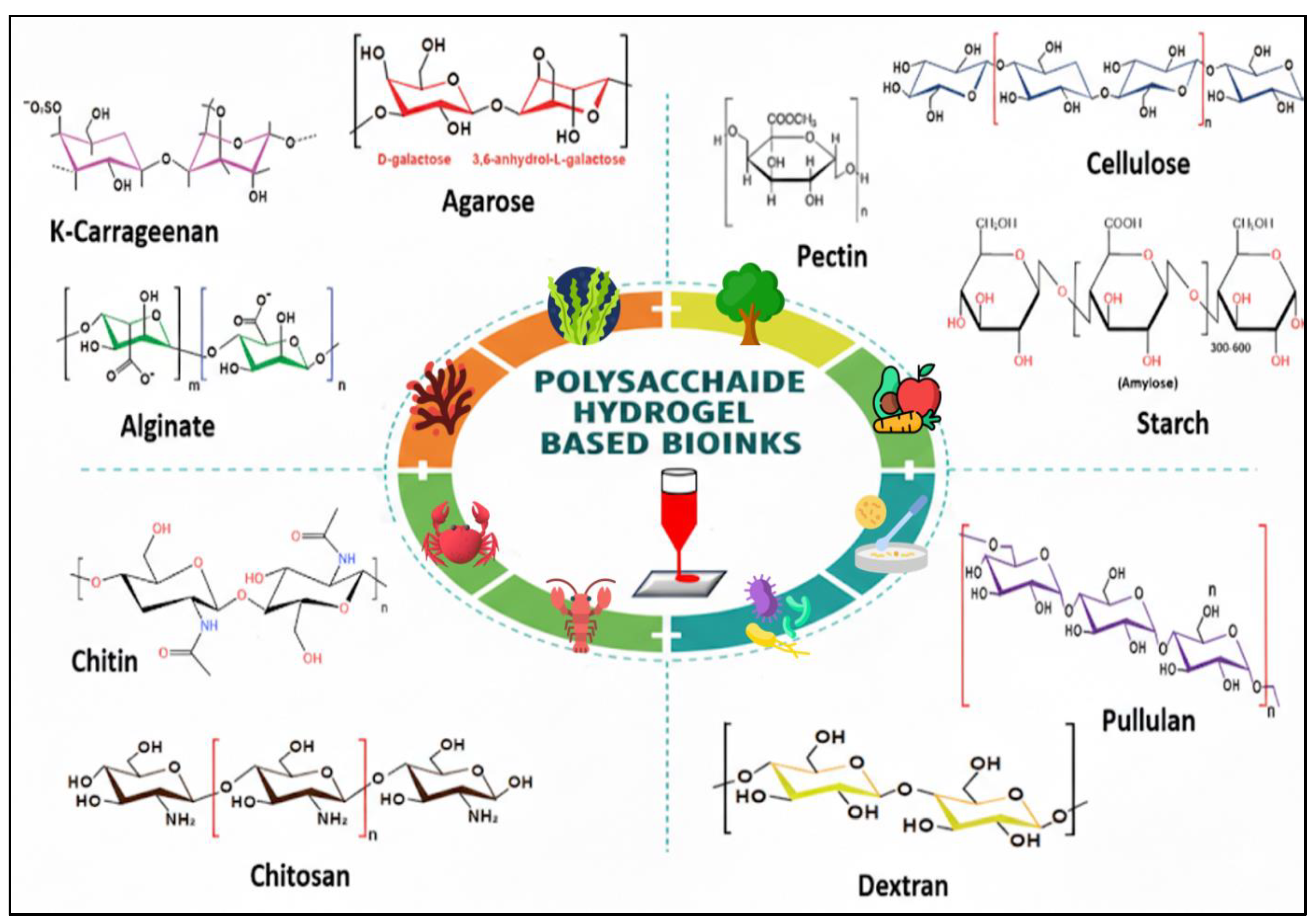
2.2. Classification of Polysaccharides
Polysaccharides are macromolecular carbohydrates composed of extensive chains of monosaccharide units connected by glycosidic bonds. The diversity in their chemical structures, which is determined by the type of monomers, the nature of the linkages (α or β), and the degree of branching, results in a wide range of advantageous physicochemical properties for 3D bioprinting [
9,
30,
31]. Polysaccharides are categorized based on their source (As illustrated in
Figure 2), which influences the available functional groups, purity, and intrinsic properties, as the origin affects the specific functional groups available for chemical modification and the inherent mechanical properties of the resultant hydrogel networks [
32].
2.2.1. Seaweed-Derived Polysaccharides
Polysaccharides extracted from seaweed, such as alginate, agarose, and carrageenan, are among the most frequently utilized materials in extrusion-based bioprinting due to their rapid gelation rates and high versatility [
33,
34].
Alginate, an economical polysaccharide derived from brown algae, is frequently utilized in tissue engineering due to its biocompatibility, ability to mimic certain characteristics of the extracellular matrix (ECM), and adjustable properties [
35,
36]. Its molecular structure consists of linear sequences of guluronic (G) and mannuronic (M) acids, which facilitate ionic gelation upon interaction with divalent cations, such as Ca
2+. This mild ionic crosslinking is advantageous for drug delivery systems and wound treatment [
37]. However, in the context of 3D bioprinting, alginate’s relatively low viscosity and insufficient viscoelastic properties in its uncrosslinked form often limit its printability. Consequently, strategies such as in situ ionic crosslinking, blending with other polymers such as gelatin or hydroxyapatite, or employing coaxial extrusion techniques, where a calcium-rich sheath phase promotes rapid gelation, are frequently employed to enhance shear-thinning behavior and shape fidelity [
36,
38]. These approaches have facilitated the effective creation of constructs for cartilage and vascularized tissues; however, their mechanical strength remains inadequate for load-bearing applications without additional reinforcement [
36].
Owing to its versatile properties, alginate remains the predominant natural bioink employed in extrusion-based 3D bioprinting. Tarassoli et al. [
39] observed that it is the most frequently utilized material in extrusion research, whether used independently or in combination with other substances. However, this prevalence is attributed more to convenience than to optimal performance in specific tissues. Similarly, González-Fernández et al. [
40] systematically adjusted the molecular weight of alginate, type of crosslinker, and concentration, demonstrating that well-optimized formulations can yield printable filaments with high post-printing cell viability. However, rapid diffusion-limited gelation can lead to uneven crosslinking and suboptimal mechanical properties. Furthermore, multiple research groups have emphasized that unmodified alginate is bioinert, exhibits poor adhesive qualities, and undergoes slow and sometimes unpredictable degradation in vivo [
41,
42]. For instance, some studies have noted that creating complex, cell-directing structures generally requires incorporating gelatin, collagen, decellularized ECM, or nanocellulose to enhance cell adhesion and achieve mechanical properties more akin to physiological conditions [
42,
43]. Recent approaches, such as RGD- or peptide-modified alginate bioinks and double-network alginate–GelMA systems, suggest that chemical modifications can significantly enhance cell spreading and tissue-specific differentiation [
43]. However, these innovations often involve trade-offs, such as reduced viscosity due to alterations in polymer chemistry, compromised structural integrity, and diminished long-term architectural stability. Overall, this development underscores that the primary role of alginate in 3D bioprinting is evolving into that of a customizable ionic scaffold within more advanced composite or engineered systems, rather than serving as a standalone matrix [
44].
Agarose is a thermoreversible polysaccharide that undergoes physical gelation, characterized by a rapid sol-gel transition, excellent shape retention post-deposition, and inherent biocompatibility. These properties have prompted various research teams to consider it a promising structural foundation for bioinks in medical three-dimensional (3D) printing applications [
45,
46]. López-Marcial et al. [
47] demonstrated that composites of 5% (w/v) agarose–alginate can achieve rheological properties comparable to Pluronic, support long-term cell viability, and function effectively as bioinks for cartilage. In contrast, unmodified agarose did not achieve the desired balance between print quality and biological efficacy. Wenger et al. [
48] systematically evaluated low-melt agarose and agar inks, concluding that a minimum concentration of 4.5% was necessary to produce reproducible lattice structures. However, the resulting hydrogels exhibit limited mechanical and thermal stability, restricting their effectiveness as durable functional constructs. More recent thermoresponsive formulations, such as the carboxymethyl cellulose–agarose–gelatin system developed by Sekar et al. [
49], specifically utilize agarose to modulate rheology and gelation. This polysaccharide blend outperformed single-component agarose in terms of printability and long-term stability. Collectively, these findings underscore the primary role of agarose in 3D bioprinting, which is to facilitate rapid and gentle gelation. They also highlighted the limitation that agarose is bioinert, mechanically weak, and possesses a relatively narrow range of printability unless modified or blended with other materials.
Carrageenan, a sulfated polysaccharide derived from marine sources, is increasingly utilized as a hydrogel component and bioink in 3D printing for medical applications, particularly in tissue engineering, drug delivery, and wound healing [
50,
51]. Recent investigations into κ-carrageenan bioinks underscore their potential as alternatives to alginate derived from algae, while also recognizing their limited processing range. Marques et al. [
50] demonstrated that unmodified κ-carrageenan can be 3D bioprinted into highly hydrated, soft scaffolds (with water content exceeding 97% and a modulus of 20–100 kPa) that support good fibroblast viability, albeit within a narrow range of temperature and composition that affects viscosity and gelation. This reliance on physical and ionic crosslinking raises concerns regarding long-term stability under physiological conditions.
Various research groups have developed modified or composite systems to address these challenges. In visible-light printing systems, methacrylated κ-carrageenan produces scaffolds that exhibit enhanced strength and stability, closely resembling bone. Nevertheless, this method necessitates the use of photoinitiators and denser networks, which may potentially compromise the biofunctionality of native polysaccharides [
52,
53]. Inks formulated with κ-carrageenan, collagen, and magnetic nanoparticles have demonstrated improved mechanical strength and cellular interactions compared to κ-carrageenan alone, thereby establishing the polysaccharide as a structural and rheological enhancer within a multicomponent bioink [
54]. Granular κ-carrageenan-based support baths, such as CarGrow, further illustrate this transformation by utilizing κ-carrageenan microgels to create a dynamic support environment rather than serving as the primary scaffold [
55,
56].
2.2.2. Plant-Derived Polysaccharides
Plant-derived polysaccharides, such as cellulose, pectin, and starch, are among the most abundant renewable sources for developing bioinks [
43,
57].
Cellulose, the most abundant natural organic polymer, is renowned for its sustainability, biodegradability, biocompatibility, and renewability. Structurally, it consists of linear β (1→4)-linked D-glucose units, each containing three hydroxyl groups per monomer unit. This configuration imparts significant hydrophilicity while restricting solubility in standard solvents owing to strong hydrogen bonding. This limitation can be addressed through chemical modification [
58,
59]. Cellulose, particularly in the form of nanocellulose and bacterial cellulose, has been extensively studied as a structural component of 3D printable hydrogels. These forms can impart shear-thinning, yield stress, and dimensional stability to otherwise fragile formulations while maintaining high water content and cytocompatibility [
60,
61]. Some studies [
43,
62] have demonstrated that nanofibrillated cellulose-based inks enable the high-fidelity extrusion of self-supporting scaffolds and sustain the viability of fibroblasts or chondrocytes. However, both research teams noted that unmodified cellulose is largely bioinert and requires blending with more bioactive polymers (such as alginate or gelatin) to provide specific cell-adhesive and differentiation signals. Similarly, Grunberg et al. [
63] found that bacterial nanocellulose enhanced the mechanical strength and print accuracy of composite bioinks; however, its limited degradability and intrinsic bioactivity restricted its use as a standalone matrix for dynamic remodeling of tissues.
Cellulose derivatives modify this context by introducing charged or hydrophilic substituents, rendering cellulose water-soluble and significantly enhancing its viscosity. This transformation shifts the role of cellulose in bioinks from an insoluble reinforcement to a genuine continuous phase [
64,
65]. For instance, Habib et al. [
66] demonstrated that carboxymethyl cellulose (CMC) can be employed to adjust the shear-thinning and filament stability of extrusion bioinks. In contrast, Wang et al. [
67] established that methacrylated cellulose ethers facilitate photocrosslinkable networks with improved elastic moduli and shape fidelity compared to physically entangled cellulose systems. Concurrently, Lee et al. [
68] and others observed that CMC- or methylcellulose-based inks largely lack adhesion and primarily function as printable “vehicles,” necessitating a blend with proteins or other bioactive polysaccharides to promote cell spreading in the bioink. They noted that excessively high substitution levels could result in inks with high viscosities, thereby impeding extrusion and nutrient diffusion.
Pectin, a naturally occurring heteropolysaccharide derived from plant cell walls, has garnered increasing attention in the field of biomaterials due to its robust safety profile, biodegradability, and structural attributes. Composed primarily of α-1-4-linked galacturonic acid residues with varying degrees of methylation, pectin exhibits significant and adjustable gelling properties [
23]. Consequently, this galacturonic acid-rich polysaccharide, traditionally employed in food products and wound care, is being recognized as a sustainable bioink component. However, it is often perceived as a specialized additive rather than a primary material for 3D bioprinting in the medical field.
Pitton et al. [
69] demonstrated that the integration of pectin with TEMPO-oxidized cellulose nanofibers markedly improved the viscosity, shear-thinning behavior, and filament stability, facilitating the extrusion of cell-laden structures with adequate viability. However, they highlighted that pectin alone lacks the requisite viscosity and mechanical strength for effective printing. In the realm of biological applications, Merli et al. [
70] developed crosslinked pectin bioinks for neural application. They illustrated that precise pH control and the utilization of CaCO
3 for gelation can produce printable networks that support the survival and morphological changes of neurons, astrocytes, and microglia. However, the narrow and formulation-sensitive processing window poses challenges in terms of reproducibility and scalability.
Recent research has focused on exploiting specific structural regions of pectin to augment its functionality. Chang et al. [
71] developed hydrogels from RG-I–rich pectin, a structural variant characterized by abundant branched, bioactive side chains, for the construction of chondrocyte-laden models. This approach yielded favorable shape fidelity and swelling-regulated degradation, capitalizing on the inherent anti-inflammatory properties associated with the RG-I domain. In a related study, Cernencu et al. engineered stiffer scaffolds inspired by nature using pectin–nanocellulose inks [
72]. Collectively, these studies underscore the primary advantage of pectin, which lies in its incorporation into tailored, multifunctional composites rather than serving as a basic, stand-alone matrix. Its future in 3D bioprinting will likely depend on the strategic application of its gelling properties and bioactive regions to enhance complex and application-specific formulations.
Starch, a neutral polysaccharide comprising linear amylose and extensively branched amylopectin, is the primary energy reserve in plants. Its intricate branched structure can undergo chemical modifications to form highly porous networks that facilitate nutrient diffusion and transport, which is a critical feature in tissue engineering [
73]. Starch-based hydrogels are increasingly recognized as promising plant-derived materials for 3D bioprinting because of their cost-effectiveness, biodegradability, and inherent shear-thinning and thixotropic properties, which enable high-fidelity extrusion. Biologically enhanced formulations reveal both the potential and limitations of these approaches. Zhuang et al. [
73] developed a nanocomposite starch bioink incorporating gelatin nanoparticles and collagen, reporting significantly improved cell attachment, migration, and proliferation compared to pure starch, which, despite excellent printability, remained largely bioinert and could not support 3D cell spreading. Similarly, a recent study [
74] demonstrated that gelatin–starch–halloysite nanocomposite hydrogels exhibited self-healing properties and robust filament formation, but primarily utilized starch as a rheology and water-retention modifier within a mechanically reinforced structure rather than as an independent structural matrix. Recent investigations into methacrylated-starch bioinks have shown that partial chemical modification can yield printable, cytocompatible gels with enhanced mechanical stability. However, these systems sacrifice simplicity for more complex synthesis processes and raise concerns regarding residual monomers and initiators in the final product [
75]. Overall, these studies underscore a critical perspective: while starch can significantly enhance printability and degradability in composite bioinks, unmodified starch hydrogels are inadequate in terms of sustained bioactivity and long-term mechanical strength for rigorous medical applications unless combined with proteins, nanoparticles, or covalent crosslinking techniques [
73,
75,
76].
2.2.3. Microbial Polysaccharides
Microbial polysaccharides, such as xanthan gum, gellan gum, and dextran, are synthesized through controlled fermentation processes, ensuring consistency in batch production that frequently surpasses that of plant and animal extracts [
23,
77]. These microbial polysaccharides are increasingly utilized as supplementary components to modify rheological properties and mechanical characteristics, rather than serving as standalone bioinks. Xanthan gum is commonly used as a robust shear-thinning modifier. Taniguchi et al. [
78] demonstrated that augmenting the xanthan concentration in alginate–xanthan mixtures significantly improved filament stability and shape accuracy, thereby facilitating the formation of crosslinked structures with favorable cytocompatibility. Recent formulations combining agarose and xanthan by Teixeira et al. [
8] achieved extrusion without necessitating post-printing gelation, although they still relied on agarose for structural support and encountered challenges owing to the inherent lack of adhesion and degradability of xanthan. Gellan gum followed a similar trajectory. Lameirinhas et al. showed that composites of nanofibrillated cellulose and gellan produced printable constructs resembling skin, with approximately 90% viability of keratinocytes by day 7 [
79]. In contrast, Phromviyo et al. required the addition of methylcellulose, cellulose nanocrystals, and ionic crosslinkers to address the brittleness and limited gelation window of pure gellan [
80]. Dextran is predominantly utilized in chemically modified forms, such as dextran-methacrylate and dextran–PCL photoinks, which enable high-resolution light-based printing and adjustable stiffness [
81]. Recent systems employing alginate/gelatin/dextran-aldehyde (AGDA), as reported by Lim et al. [
82], demonstrated enhanced cell engraftment compared to commercial GelXA, although this was accompanied by increased crosslinking complexity and potential cytotoxicity due to the aldehyde chemistry. These studies suggest that while microbial polysaccharides are valuable for enhancing printability and mechanical properties, their limited bioactivity and sometimes challenging degradation or crosslinking characteristics indicate that they are most effective when incorporated into composite structures rather than as primary cell-instructive scaffolds.
2.2.4. Animal-Derived Polysaccharides
Polysaccharides sourced from animals, notably chitosan and hyaluronic acid (HA), offer biologically pertinent characteristics and adaptable chemistries; however, they have not yet achieved the same level of extensive application and routine use as alginate [
83]. Chitosan-based systems leverage their cationic charge, antimicrobial properties, and mild gelation for osteogenesis and wound healing [
84,
85]. Nevertheless, Teixeira et al. noted that native chitosan frequently requires acidic conditions, complex multicomponent crosslinking, or blending with nanocellulose or nano-hydroxyapatite to achieve satisfactory print fidelity and mechanical strength [
8]. For instance, Maturavongsadit et al. introduced thermogelling chitosan–glycerophosphate–nanocellulose inks that demonstrated rapid gelation and good shape retention; however, this was confined to a limited temperature and composition range and still lacked the rigidity required for load-bearing bone constructs [
86]. Concurrently, HA-based bioinks are commended for their capacity to mimic the extracellular matrix (ECM) and their excellent cytocompatibility [
87]. Mouser et al. [
88] and more recent studies [
89] have shown that methacrylated or thiol–ene modified HA can be printed into constructs containing cells that facilitate chondrogenic or neural differentiation. Nonetheless, these studies also concur with the consensus that unmodified HA is excessively soluble and mechanically weak for extrusion, and that chemical functionalization, while essential for crosslinking and structural integrity, results in trade-offs in viscosity, degradation characteristics, and potential toxicity from photoinitiators. This has led to the more frequent use of HA as a bioactive component within composite or double-network inks rather than as a standalone structural matrix.
2.3. Properties of Polysaccharide-Based Hydrogels
Polysaccharide-based hydrogels have emerged as a significant class of bioinks owing to their capacity to modulate physicochemical properties across a wide design spectrum while maintaining compatibility with living cells and tissues for clinical applications [
10,
31]. In the context of extrusion 3D bioprinting, their efficacy is determined not only by general characteristics such as stiffness and swelling but also by rheological properties, including yield stress, shear-thinning, and rapid recovery. These factors are essential for filament formation and preservation of shape accuracy during printing, ultimately influencing the cell functionality within the resultant structures [
90,
91].
Native polysaccharide networks, including alginate, hyaluronic acid, chitosan, and xanthan, typically exhibit characteristics of softness and brittleness, with moduli comparable to those of the brain or soft connective tissues rather than cartilage or bone. This limits their applicability in environments that require mechanical robustness [
92,
93]. To address this limitation, research has indicated that effective polysaccharide bioinks often rely on strategies such as ionic–covalent double networks, nanofiller enhancement ( nanocellulose, silica, and hydroxyapatite), or the incorporation of synthetic networks to enhance compressive strength and toughness while preserving printability [
94,
95]. Furthermore, methods such as dual-enzyme and post-polymerization techniques have shown that in situ secondary crosslinking can convert initially soft and easily extrudable inks into more rigid, load-bearing hydrogels after the printing process [
95].
Polysaccharides, including alginate, chitosan, hyaluronic acid, and cellulose derivatives, are highly valued for their minimal cytotoxicity, reduced protein adsorption, and structural resemblance to glycosaminoglycans and extracellular matrix (ECM) fibers. These characteristics contribute to enhanced cell viability and reduced inflammatory responses in both in vitro and in vivo studies [
96]. However, numerous studies have highlighted that biocompatibility is not inherently assured. Residual solvents, initiators, unreacted crosslinkers, and contaminants from marine or animal sources can elicit adverse reactions. Therefore, rigorous purification and the application of mild crosslinking techniques are essential for the safe utilization of these materials as clinical bioinks [
97].
The swelling behavior of polysaccharides is determined by their hydrophilic backbones and the presence of ionic or hydrogen-bonding groups, which facilitate high equilibrium water retention [
6]. This characteristic promotes the diffusion of nutrients and oxygen but also results in considerable swelling that may compromise the integrity of the printed structures [
95,
99]. Gebeyehu et al. highlighted the necessity of optimizing the crosslink density and network structure: gels that are under-crosslinked tend to swell excessively and lose their geometric integrity, whereas those that are over-crosslinked restrict diffusion and may adversely affect the cell viability and matrix deposition. Consequently, controlling swelling is critical for drug delivery and tissue development [
100].
Biodegradability is a critical characteristic of polysaccharide hydrogels, which typically undergo degradation via hydrolysis and enzymatic processes. For instance, chitosan is degraded by lysozyme, hyaluronic acid by hyaluronidases, and alginate by alginate lyases, respectively. The degradation rate is significantly influenced by factors such as the substitution pattern, type of crosslinking, and presence of secondary networks [
8,
24,
98]. Recent studies on modular enzyme-responsive hydrogels suggest that degradation can occur independently of the initial mechanical properties or printing requirements by incorporating specific cleavage sites and enzyme-sensitive crosslinkers [
100,
101]. However, numerous studies have emphasized that precisely aligning degradation rates with the formation of new tissue and vascularization remains a significant challenge for polysaccharide-based bioinks intended for implantation [
9,
102].
Polysaccharides are indispensable in 3D bioprinting because of their biocompatibility, biodegradability, and ECM-mimetic properties. However, their application is not uniform, each polysaccharide presents a unique profile of advantages and constraints that dictate its role in bioink formulation, compatibility with specific bioprinting modalities, and ultimate suitability for targeted tissue engineering applications (as summarized in
Table 1).
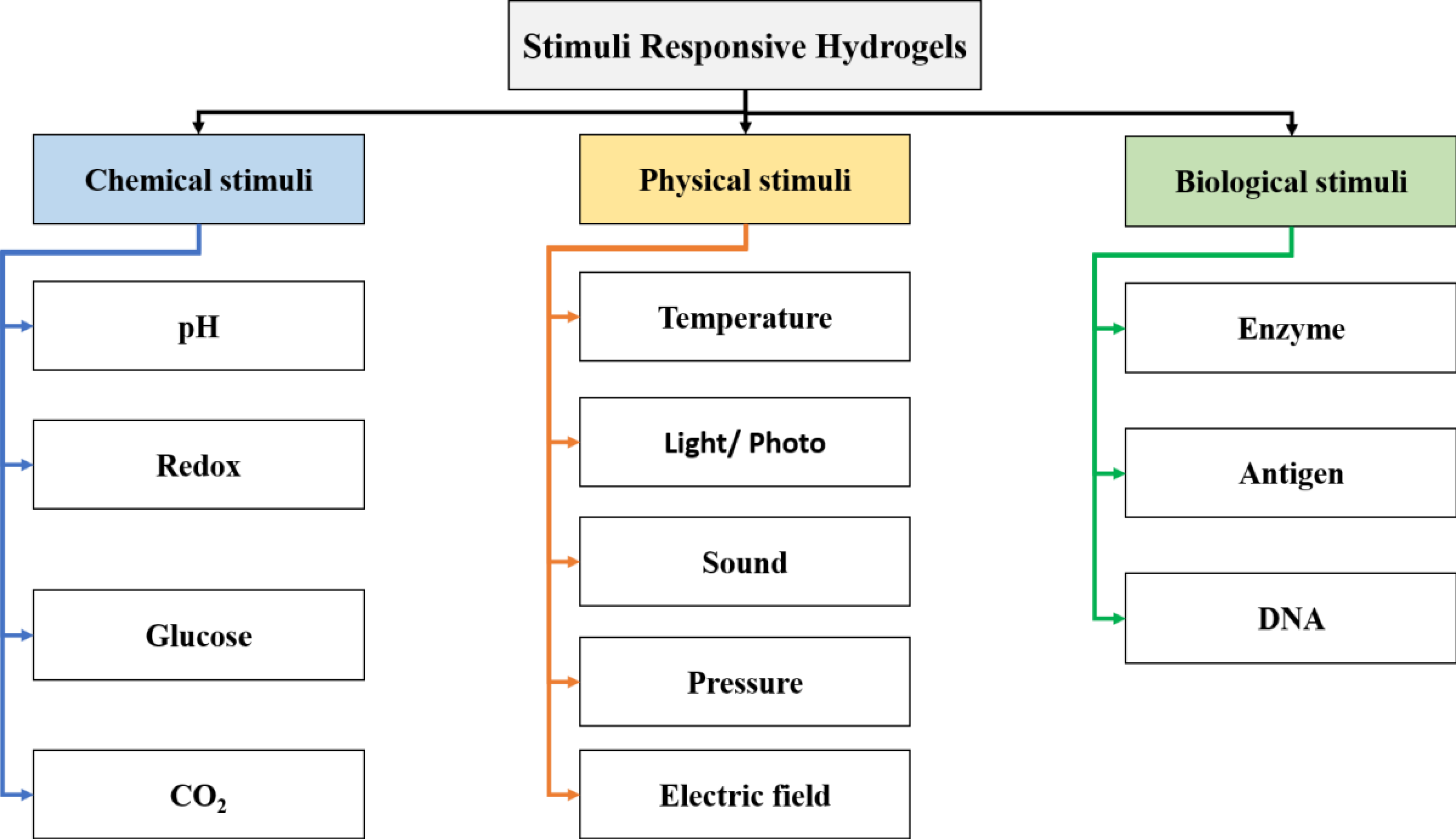
Stimuli-Responsive and Smart Hydrogels
A transformative advancement beyond static material properties is the engineering of stimuli-responsive or smart polysaccharide hydrogels [
117,
118]. These dynamic materials are designed to sense and respond to specific environmental cues, such as temperature, pH, light, or enzymatic activity (
Figure 3), with predictable changes in their physical or chemical states, such as swelling, stiffness, degradation kinetics, or bioactivity [
6,
119]. This intrinsic adaptability allows them to closely mimic the dynamic and reciprocal interactions found in native tissues [
120].
In 3D bioprinting, this property enables several paradigm-shifting capabilities to be achieved. First, it enhances printability and structural fidelity through mechanisms such as thermally triggered gelation (e.g., agarose or modified chitosan systems that gel at physiological temperature) or photocrosslinking of methacrylated derivatives (e.g., HA-Me or alginate-Me), which provides spatial and temporal control over solidification [
121]. Second, it forms the cornerstone of 4D bioprinting, where printed 3D constructs are programmed to change shape or function over time in response to a trigger, such as a self-folding vascular graft activated by a temperature shift [
119,
122]. Third, smart hydrogels facilitate the creation of dynamic cell-instructive microenvironments. Post-printing, their properties can be modulated on-demand to guide cell behavior (e.g., softening a matrix to encourage migration or stiffening it to promote differentiation) or to provide localized therapeutic release triggered by disease-specific signals (e.g., pH-sensitive drug delivery in tumor models) [
123].
While often achieved through chemical modification (grafting of light-sensitive or enzyme-cleavable groups) and integrated crosslinking strategies, the core appeal lies in leveraging the versatile chemistry of polysaccharides, such as the carboxyl groups of alginate or the hydroxyl groups of cellulose, to create bioinks that are not merely structural but also interactively biological [
117,
120]. This shift from passive scaffolds to active, communicative matrices represents a critical frontier in the development of truly biomimetic and clinically functional bioprinted tissues.
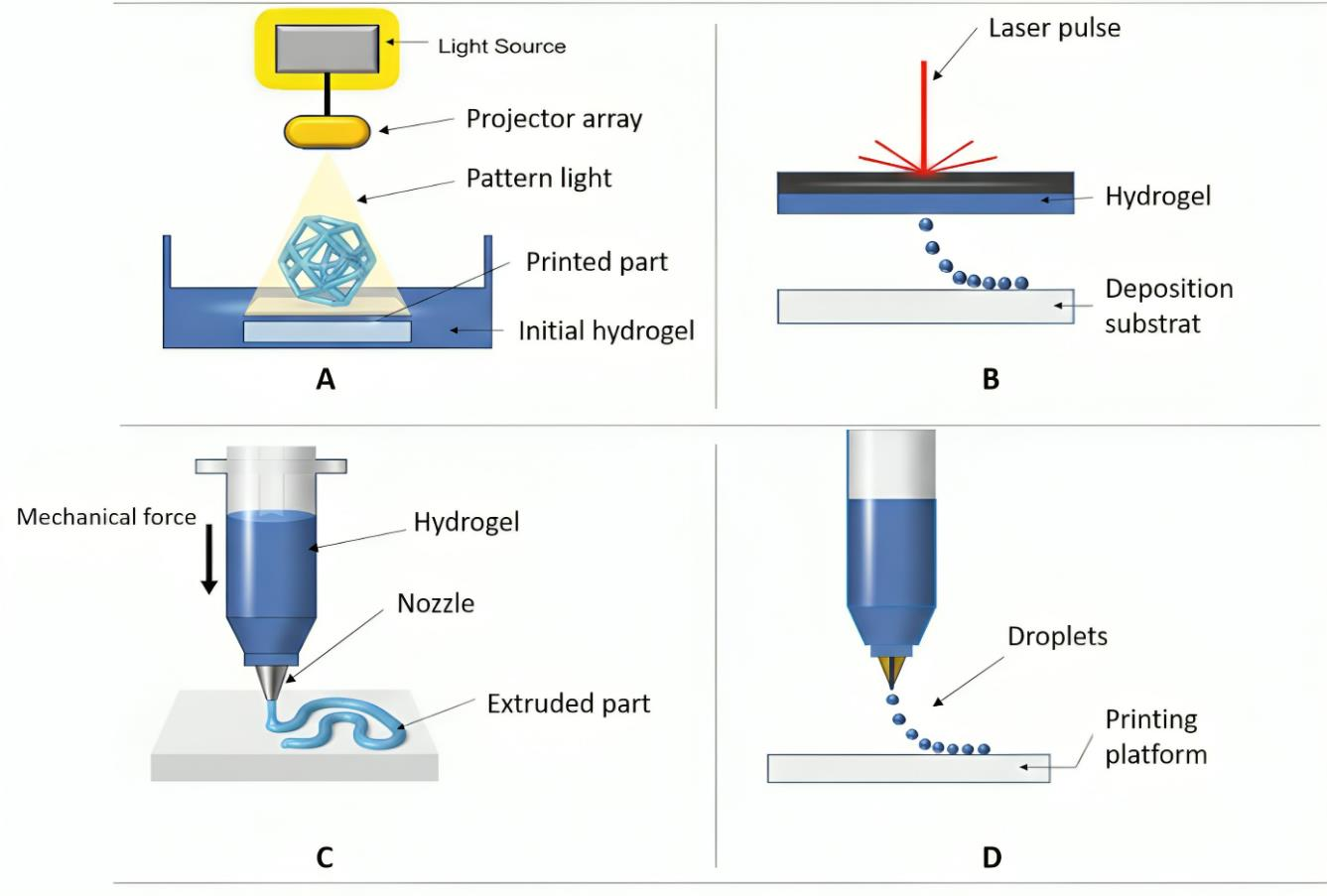
2.4. Bioprinting Techniques
The selection of a 3D bioprinting method (depicted in
Figure 4) significantly limits the capabilities of polysaccharide-based hydrogels, as each technique functions within a unique range of viscosity, crosslinking rates, and cellular compatibility [
114]. Recent research has highlighted that factors, including printability (shear-thinning, yield stress, and recovery) and the ability to maintain shape, must be evaluated differently across extrusion, droplet, and vat photopolymerization technologies, which clarifies why the same polysaccharide composition may be effective in one platform but fail in another [
7,
43].
2.4.1. Extrusion-Based Bioprinting
Extrusion is the predominant technique employed for polysaccharide bioinks because of its ability to process highly viscous, shear-thinning compositions, including alginate, chitosan blends, xanthan, and nanocellulose systems [
124]. Gillispie et al. and Cooke et al. suggested that polysaccharide inks deemed “successful” in extrusion typically operate within a specific rheological range, they must exhibit a finite yield stress, substantial shear-thinning to minimize nozzle pressure, and rapid viscosity recovery to maintain the filament shape [
125,
126]. However, excessive viscosity or crosslinking can elevate the shear stress, thereby compromising cell viability. To mitigate this issue, numerous research teams have utilized multicomponent formulations, such as gelatin–alginate–methylcellulose, or adopted core–shell and granular methods, wherein a more fluid cell-laden phase is printed within a stiffer polysaccharide support or carrier [
124,
127]. This strategy facilitates the separation of cell-friendly rheology from the structural requirements. Nonetheless, the resolution attainable through extrusion remains constrained to approximately 100–200 µm, and repeated exposure to shear can pose risks to sensitive cell types, particularly when high viscosities or small nozzle diameters are used [
128].
2.4.2. Inkjet-Based Bioprinting
Inkjet bioprinting offers high spatial resolution with droplet sizes ranging from 10 to 100 µm, rapid patterning capabilities, and minimal shear stress, all of which are advantageous for cell viability [
129]. However, this technique necessitates the use of extremely low-viscosity, nearly Newtonian fluids (typically less than 10 mPa·s), which is incompatible with the rheological properties required for unmodified polysaccharide hydrogels to be printable and self-supporting [
130]. As noted in several studies, unmodified alginate, hyaluronic acid, or chitosan must be substantially diluted or chemically modified to meet jetting standards, which reduces the solid content and mechanical strength while increasing the risk of satellite droplets and nozzle blockages owing to partial gelation [
131]. Consequently, polysaccharides are more commonly employed as minor components in inkjet formulations, serving as viscosity enhancers or bioactive additives, rather than as the primary structural element, thereby limiting the applicability of this method for bioinks that are predominantly polysaccharide-based [
103,
129].
2.4.3. Vat Photopolymerization (SLA/DLP)
Stereolithography (SLA) and digital light processing (DLP) can achieve exceptional resolution and intricate geometries by solidifying thin layers of photocurable bioinks using patterned light [
132,
133]. For polysaccharides, this process requires the incorporation of photoreactive groups, such as methacrylated hyaluronic acid (HA), alginate, and κ-carrageenan, which enable precise control over the crosslink density and facilitate the formation of finely detailed, mechanically robust structures, as evidenced in MeHA–GelMA and methacrylated κ-carrageenan systems [
52]. The primary challenges include the potential reduction in inherent bioactivity and alteration of degradation characteristics due to chemical modifications, as well as the reliance on photoinitiators for light-based curing processes. The type, dosage, and wavelength of these photoinitiators must be meticulously calibrated to prevent cytotoxicity and DNA damage while ensuring complete polymerization [
134,
135]. Additionally, light scattering and absorption in cell-laden, often opaque polysaccharide inks complicate uniform curing in thicker constructs, prompting ongoing research into visible-light initiators, hybrid resin–hydrogel systems, and co-printing with reinforcing thermoplastics [
133].
2.4.4. Laser-Assisted Bioprinting (LAB)
Laser-assisted bioprinting (LAB) employs focused laser beams to eject droplets from a donor ribbon onto a receiving surface, thereby obviating the need for nozzles and circumventing clogging issues associated with high-viscosity inks or those containing particulates [
136]. LAB can process higher viscosities than inkjet printing and achieve single-cell precision. However, it requires complex optical systems, metallic absorbing layers, and precise laser energy management to prevent local overheating and cellular damage, which has constrained its widespread adoption to specialized laboratories [
137]. In the realm of polysaccharide hydrogels, the extant literature presents relatively few examples compared with extrusion and vat photopolymerization techniques. Current studies predominantly utilize LAB to create patterns with cell suspensions or low-viscosity fluids, with the primary structural framework established through subsequent bulk crosslinking rather than during the jetting phase. This suggests that the practical application of constructing polysaccharide-rich, mechanically intricate structures remains more theoretical than empirical [
7,
136].
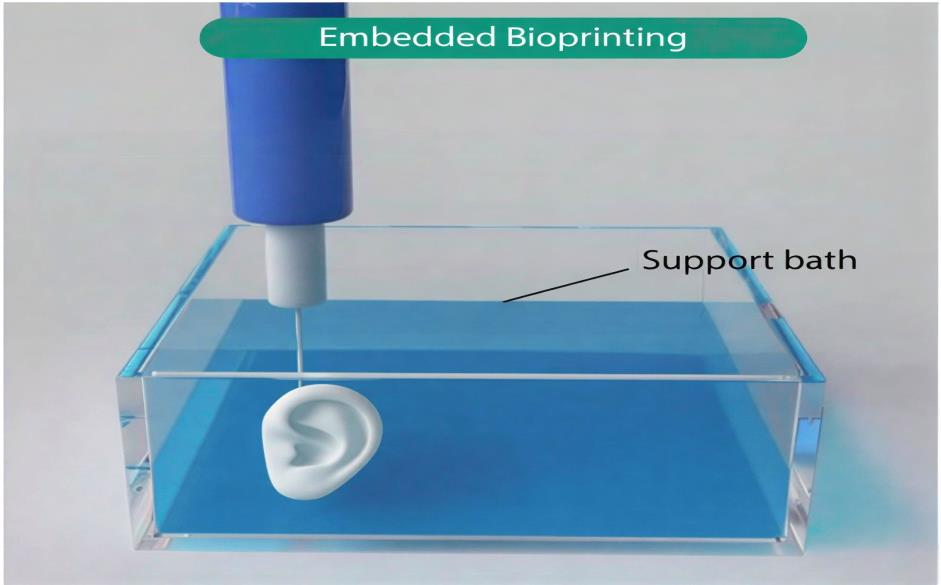
2.5. Embedded Techniques in 3D Bioprinting
Traditional bioprinting methods, including extrusion-based, droplet-based, and vat photopolymerization-based techniques, have advanced significantly; however, they often struggle to replicate the intricate architectures of native tissues, particularly those involving overhanging structures, vascular networks, and high cellular densities [
33,
102]. Embedded bioprinting has emerged as a pivotal innovation to overcome these limitations, allowing printing within a supportive matrix that mitigates gravitational constraints and enables freeform fabrication (As illustrated in
Figure 5) [
139,
140]. Embedded bioprinting, often described as a “gel-in-gel” approach, involves extruding bioinks directly into a supportive suspension medium, such as hydrogels or microgels, which provides mechanical stability during the printing process. This method addresses the key drawbacks of conventional bioprinting, including structural collapse due to low-viscosity bioinks and the inability to create overhanging or hollow features without additional support [
140,
141]. The support bath typically exhibits shear-thinning and self-healing properties; under the shear stress of the printing nozzle, it locally fluidizes, enabling bioink deposition, and rapidly recovers its solid-like state upon stress removal, thereby encapsulating the printed structure [
141,
142]. Several embedded techniques have been developed, each tailored to specific fabrication needs (summarized in
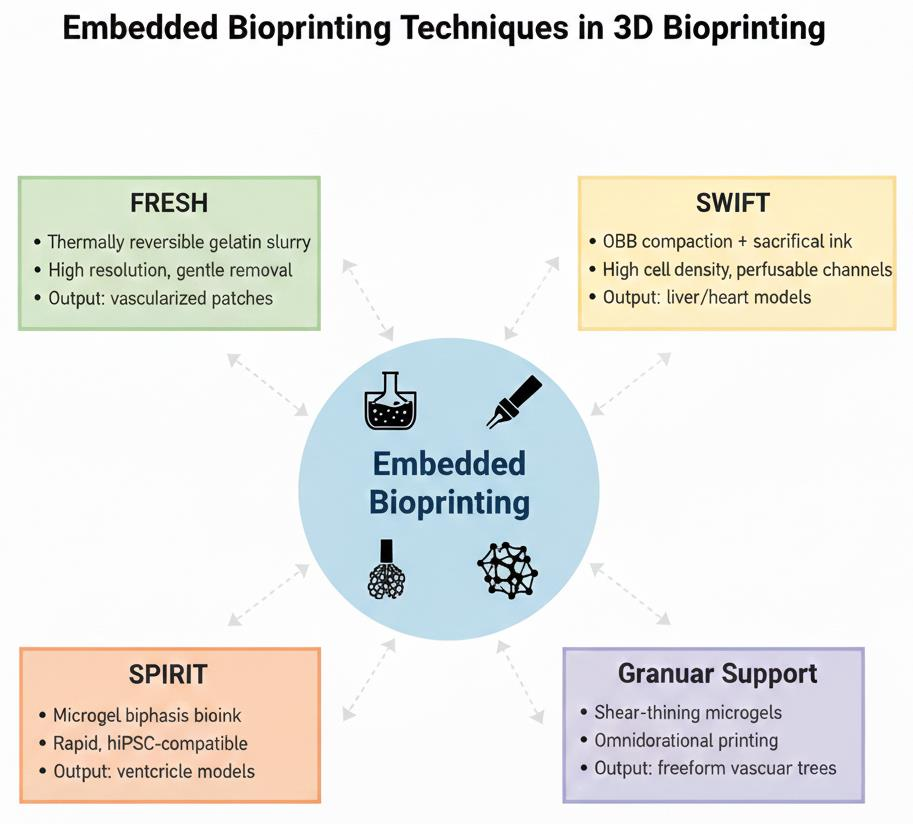
Figure 6).
Freeform Reversible Embedding of Suspended Hydrogels (FRESH): this method uses a gelatin slurry or microgel bath to support the printing of soft, low-viscosity bioinks, such as collagen or alginate [
143,
144]. The bath is thermoreversible, allowing easy removal by gentle heating after printing. FRESH has enabled the creation of vascularized heart models and branched airway structures with resolutions of 20-50 μm. It is particularly suited for tissues requiring high fidelity to native extracellular matrix (ECM) composition [
145].
Sacrificial Writing into Functional Tissue (SWIFT): involves compacting organ-building blocks (OBBs), such as spheroids or organoids, into a dense matrix and embedding sacrificial inks to create perfusable channels [
142,
146]. Upon ink removal, vascular-like networks are formed, supporting nutrient diffusion in high-cell-density constructs. This technique has been applied to cardiac tissue and liver models, achieving cell densities comparable to those of native organs (up to 10
8 cells/mL) [
146].
Granular Gel Medium and Omnidirectional Printing: these variants employ granular hydrogels (e.g., carbomer or agarose microparticles) as the support, enabling omnidirectional nozzle movement for complex geometries. Omnidirectional printing, for instance, facilitates the fabrication of freeform vascular trees without layer-by-layer constraints, which is ideal for engineering branched networks in non-yield-stress fluids [
140,
141].
Other Hybrid Approaches: techniques such as CLASS (Constructs Laid in Agarose Slurry Suspension) and guest-host writing integrate supramolecular chemistry for reversible bonding in the support medium, enhancing print fidelity for delicate structures [
142,
147]. These methods collectively expand bioprinting capabilities, allowing micron-scale resolution (down to 1-10 μm in some cases) and the incorporation of multiple cell types for biomimetic tissues [
148]. Bioinks in embedded systems often include natural polymers (gelatin methacryloyl and hyaluronic acid) or synthetic polymers (polyethylene glycol) that are optimized for biocompatibility, printability, and post-print crosslinking via UV light, enzymes, or ionic bonds.
The SPIRIT Technique: introduced in 2023, the SPIRIT (Supportive Perfusable Integrated Reversible Template) technique represents a generalizable embedded bioprinting strategy that utilizes a microgel-based biphasic (MB) bioink that functions dually as a printable material and a reversible suspension medium [
154]. This MB bioink, typically composed of gelatin or similar polymers, exhibits shear-thinning and self-healing behaviors, enabling sequential printing steps in which structures are built layer-by-layer within the medium without collapse. Unlike traditional embedded methods, SPIRIT allows for the direct incorporation of human-induced pluripotent stem cells (hiPSCs), promoting their proliferation and differentiation into functional tissues, such as cardiac organoids. A key innovation is the facilitation of perfusable vascular networks with complex geometries. For instance, SPIRIT has been used to print a ventricle model with integrated freeform vessels that support nutrient perfusion, a feat unattainable with conventional extrusion or laser-assisted bioprinting. The process involves preparing the MB bioink, embedding the nozzle for sequential deposition, crosslinking the construct, and removing the template via reversible mechanisms, such as temperature shifts. This results in faster fabrication (up to 10x compared to some methods) and enhanced replication of organ intricacies, including external shapes and internal architectures [
154]. Compared to other embedded techniques, SPIRIT excels in versatility; it combines elements of FRESH (reversible template) and SWIFT (vascular integration) while minimizing material waste and improving speed. However, it requires precise rheological tuning of the MB bioink to balance the printability and cell viability. The SPIRIT technique exemplifies how advanced embedded bioprinting can leverage polysaccharide-based microgel systems to create complex, perfusable tissue constructs, highlighting the role of polysaccharides in next-generation bioprinting strategies.
Table 2 summarizes embedded bioprinting techniques.
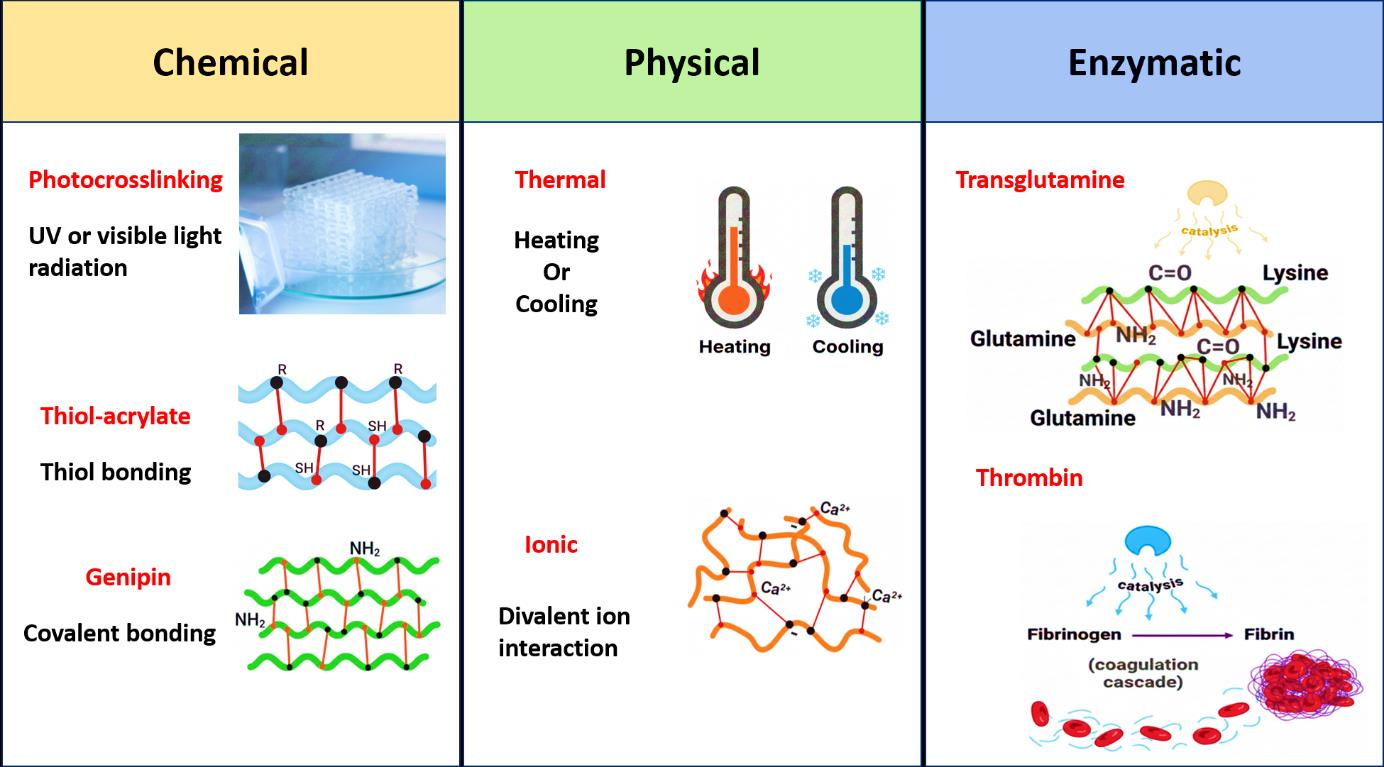
2.6. Crosslinking Methods
Crosslinking is a crucial design factor in 3D bioprinting. It controls the shift from a low-viscosity, cell-laden ink to a mechanically robust structure, thus being at the heart of the balance between printability, structural strength, and cell viability [
155]. Current trends in bioink development favor multimodal crosslinking strategies over single-method approaches. By integrating chemical, physical, and radiation-based mechanisms (
Figure 7), researchers can independently tune the extrusion behavior and the mechanical and degradation properties of the printed construct [
156].
2.6.1. Chemical Crosslinking
Chemical crosslinking methods utilize covalent bonding through processes such as photopolymerization, enzyme-mediated reactions, or click-type chemistries, resulting in networks that exhibit greater stiffness, toughness, and dissolution resistance than purely physical gels [
155]. Traditional small-molecule crosslinkers, such as epoxides, isocyanates, and aldehydes, are now approached with caution or completely avoided in systems containing cells because of concerns regarding their cytotoxicity [
98]. Instead, modern bioinks prefer functionalization with methacrylate, norbornene, or tyramine, paired with cytocompatible initiators and enzymes. Photocrosslinkable systems, such as GelMA and methacrylated HA, provide spatiotemporal control over the gelation process and are frequently incorporated into dual-crosslinking strategies [
156]. These strategies involve a quick, mild mechanism to ensure the stability of the filaments during the printing phase, followed by a secondary light-triggered step that finalizes the architecture of the printed object [
155]. Formulations such as GelMA–alginate and GelMA–gellan illustrate this idea: ionic crosslinking through Ca
2+ or pre-chelation offers immediate yield stress and shape fidelity, while the subsequent application of UV or visible-light curing results in a more robust double network [
157]. However, this comes with the trade-off of increased network density, which may lead to reduced nutrient transport. Enzyme-mediated crosslinking strategies, particularly those involving transglutaminase or horseradish peroxidase (HRP)/tyramine systems, have emerged as prominent bio-orthogonal pathways because of their ability to function under mild physiological conditions [
155]. These methods are especially advantageous for specialized fabrication techniques, such as coaxial extrusion and in situ gelation. However, precise control over the kinetic parameters, oxygen availability, and localized H
2O
2 levels is critical for mitigating potential oxidative stress and ensuring cytocompatibility [
156].
2.6.2. Physical Crosslinking
Physical crosslinking involves hydrogels that utilize noncovalent interactions, such as ionic coordination, hydrogen bonding, hydrophobic associations, crystallite formation, and chain entanglements [
158]. The main benefit is that gelation can occur quickly, reversibly, and without the need for additional reagents. In the case of polysaccharide-based inks, commonly employed methods for achieving the required shear-thinning and yield stress behavior necessary for filament creation and self-supporting layers include ionic crosslinking of alginate or κ-carrageenan, thermogelation of methylcellulose and certain chitosan systems, and crystallite formation in PVA through freeze-thaw cycles [
159,
160]. Despite their cellular compatibility, these materials are prone to creep and dissolution when exposed to physiological ionic strength. Due to their mechanical fragility, they typically serve as temporary scaffolds, providing an initial structure before being strengthened or partially substituted with permanent covalent networks [
161]. The integration of dynamic covalent bonds, such as disulfides, boronate esters, and reversible Schiff bases, effectively bridges the gap between chemical and physical crosslinking processes. These linkages provide the dual benefits of reversibility and mechanical robustness, facilitating self-healing and the dissipation of printing-induced stresses while maintaining structural integrity. However, a primary hurdle in their application is the ability to fine-tune the reaction kinetics without compromising the survival of encapsulated cells [
162].
2.6.3. Radiation-Based Crosslinking
Radiation-based crosslinking techniques employ high-energy photons or particles, traditionally gamma and electron beams, and more frequently UV/visible light in bioprinting, to produce radicals and link polymer chains without requiring additional small-molecule crosslinkers [
163]. For polysaccharides, the direct use of gamma or electron beam crosslinking is appealing for sterilization and the creation of bulk hydrogels; however, its application in constructs containing cells is restricted because of DNA damage and the generation of reactive oxygen species (ROS), making it primarily useful for acellular scaffolds or for the pre-formulation of bioink components [
164]. Conversely, lithography-based bioprinting systems utilize UV/visible-initiated crosslinking of methacrylated or acrylated polysaccharides with specifically chosen photoinitiators (such as LAP or alternatives to Irgacure), enabling layer-by-layer curing with high spatial precision and adjustable stiffness based on the dose and exposure duration. However, concerns remain regarding light penetration, heat generation, and the long-term safety of residual initiator fragments [
165].
2.6.4. Hybrid and Advanced Strategies
Hybrid and advanced strategies are also being explored. Hybrid crosslinking has emerged as a characteristic feature of high-performance bioinks, where dual- and multi-network hydrogels serve to differentiate functions: one network is tailored for printing rheology, another for long-term mechanical properties, and occasionally a third for degradability or biological activity [
150,
166]. Systems such as GelMA/alginate, GelMA/κ-carrageenan, and HA–dextran–tyramine illustrate how sequential or orthogonal crosslinking methods (ionic, photo, and enzymatic) can be integrated to independently adjust the viscosity, shape precision, stiffness, and degradation rate [
167,
168]. However, the increased complexity of formulation and processing can impede reproducibility and clinical application. Stimuli-responsive crosslinking activated by pH, temperature, redox conditions, or specific enzymes further broadens the design possibilities, allowing for constructs that harden in situ, soften during remodeling, or degrade locally in response to enzymes secreted by cells [
67]. However, current research emphasizes that achieving precise synchronization of these responses with tissue growth and functionality remains challenging.
2.7. Rheological Requirements for 3D Bioprinting
Rheology is currently considered a critical design factor for polysaccharide-based bioinks, as it determines whether a formulation can transition from a low-resistance fluid in the syringe to a self-supporting filament that maintains the desired shape without harming cell viability [
126]. The ability to print through extrusion is influenced by a combination of factors, including shear-thinning, yield stress, viscoelastic properties, and thixotropic recovery, which collectively create a limited “printability window” in which smooth flow, accurate shape retention, and careful handling of cells can be achieved simultaneously [
170].
Shear-Thinning is a phenomenon in which viscosity decreases as the shear rate increases, and is widely recognized as essential for extrusion-based bioprinting, as it lowers the pressure required for ink extrusion while reducing shear damage to encapsulated cells [
66]. Schwab et al. and Amorim et al. demonstrated that bioinks with a low power-law index (n<1) are easier to extrude and produce more continuous filaments, and that the flow consistency index (k) can be directly associated with the extrusion pressure required, allowing for rational adjustments of molecular weight, concentration, and blending to achieve a targeted operational range [
171,
172]. On a molecular level, this behavior results from the reversible breaking of polysaccharide chain entanglements and weak interactions under shear stress. Recent parametric investigations of inks based on alginate and CMC have shown that increasing the polymer concentration or incorporating associative fillers enhances the shear-thinning effect but may also raise the low-shear viscosity to a level that causes clogging if not carefully managed [
171].
Yield stress indicates the minimum stress required to initiate flow and is crucial for preventing issues such as spreading, filament fusion, and layer collapse after deposition [
27,
173]. Yield stresses that are too low can lead to a loss of pore structure and poor resolution, whereas excessively high values may hinder extrusion and cell mixing. Methods to introduce or enhance yield stress in polysaccharide systems include controlled pre-crosslinking, such as partial ionic crosslinking of alginate or κ-carrageenan, creating weak physical networks such as thermogelation of methylcellulose, and integrating nanofillers such as nanocellulose, which forms a percolating network that can support the weight of additional layers [
155]. For instance, nanocellulose-reinforced HA bioinks demonstrate how a small amount of fibrils can significantly increase the yield stress and storage modulus, enabling stable vertical structures while maintaining high cell viability, thereby representing a transition from basic thickening to more intentional rheological engineering [
174].
In addition to static shear-thinning and yield behavior, the time-dependent restoration of structure known as thixotropy is increasingly acknowledged as critical for multilayer printing [
27]. Rheological investigations and “printability maps” show that rapid viscosity recovery and the restoration of the elastic modulus after shear stress removal are vital to prevent sagging of layers and support subsequent layers [
175]. However, the recovery must not be overly rapid to avoid premature gelation within the nozzle or during prolonged printing sessions [
176]. Recent advancements in nanocellulose-based and dynamic-network inks demonstrate this delicate balance: their reversible network reconstruction enables constructs to provide self-support and self-repair after deposition [
177]. However, networks that are too robust or slow to rearrange can entrap stress and hinder cell spreading [
176]. These insights emphasize that the rheology involved in polysaccharide-based bioprinting is not a single property but a multi-faceted design space, in which factors such as shear-thinning indices, yield stress, viscoelastic characteristics, and thixotropic recovery need to be optimized together to harmonize printing performance with the biological needs of the intended tissue [
8].
The optimization of these rheological properties is directly influenced by a set of key processing parameters during printing.
Table 3 summarizes the impact of these critical parameters on polysaccharide bioinks.
2.8. A Roadmap for Polysaccharide Bioink Development
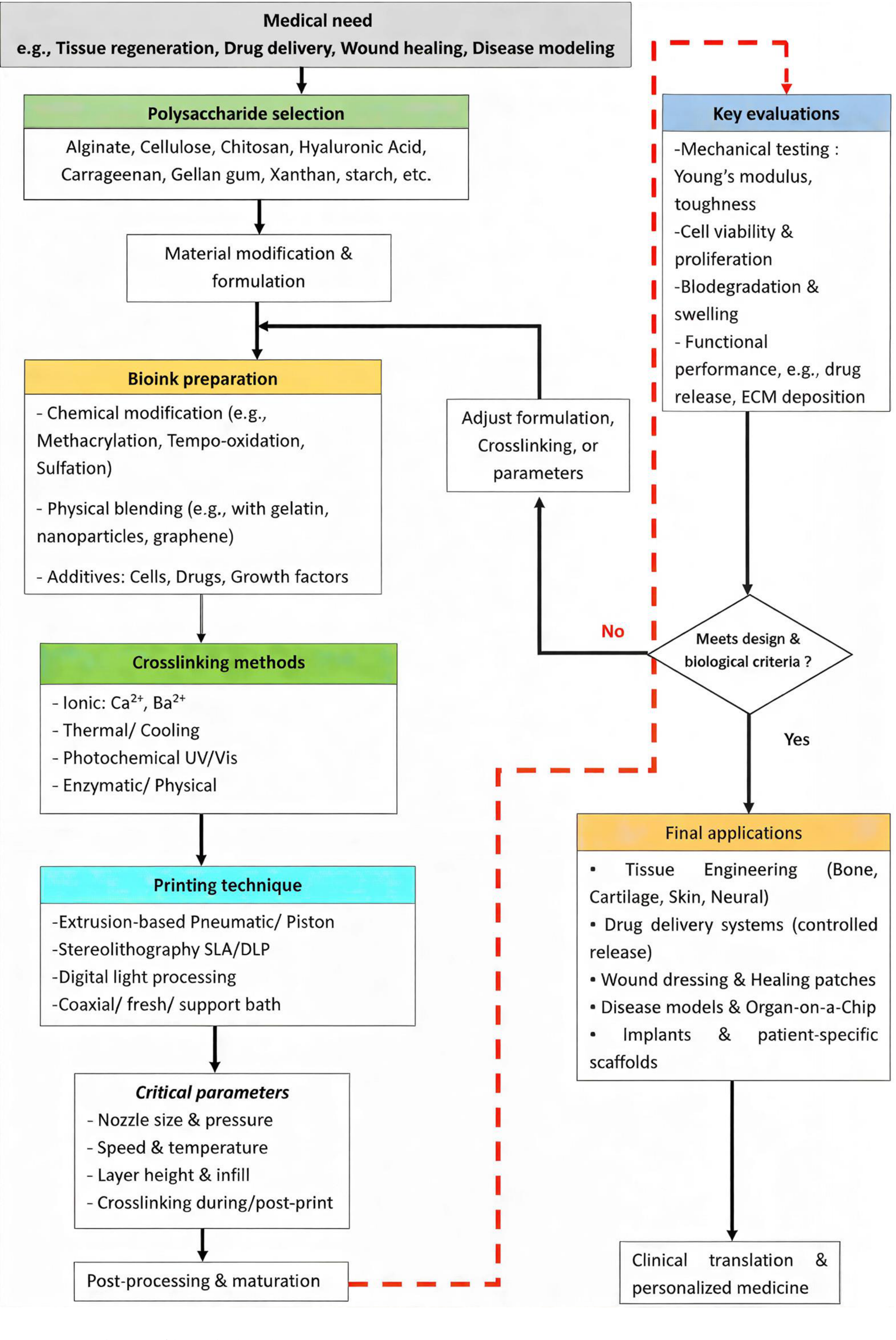
To systematically guide the design and optimization of polysaccharide-based bioinks, this review proposes a conceptual roadmap (
Figure 8) that integrates the key aspects discussed throughout. This iterative framework begins with identifying a specific clinical need (e.g., tissue regeneration, drug delivery, wound healing), which dictates the target bioink specifications. The subsequent steps involve:
Polysaccharide selection: choosing one or more base polysaccharides (e.g., alginate, chitosan, cellulose derivatives) based on their inherent properties and suitability for the intended application and printing modality.
Material modification & formulation: chemically modifying (e.g., methacrylation, sulfation) or physically blending the selected polysaccharides with complementary polymers (e.g., gelatin), nanoparticles, or bioactive additives (cells, drugs, growth factors) to achieve the desired rheological, mechanical, and biological functionality.
Crosslinking strategy selection: determining an appropriate crosslinking method (ionic, thermal, photochemical, enzymatic) or a hybrid approach to balance printability with post-printing stability.
Printing technique application: employing a suitable bioprinting technique (extrusion, vat photopolymerization, embedded) based on the formulated bioink’s properties and the architectural complexity required.
Post-processing & maturation: applying necessary post-printing crosslinking, conditioning, or culture protocols to mature the construct.
Key evaluations: rigorously assessing the printed construct through mechanical testing (Young’s modulus, toughness), biological assays (cell viability, proliferation, differentiation), and functional performance (degradation, swelling, drug release, ECM deposition).
Iterative refinement: the results from these evaluations feed back into the modification and formulation stage, allowing for iterative refinement of the bioink until it meets the predefined design and biological criteria.
Only after successfully passing these iterative cycles should the bioink proceed to application-specific testing and clinical translation considerations. This roadmap underscores that successful bioink development is not a linear process but a dynamic, interdisciplinary cycle of design, fabrication, and evaluation, central to advancing personalized medicine solutions.
3. Conclusions
This review highlights the central role of polysaccharide-based hydrogels as versatile and sustainable materials in the rapidly advancing field of 3D bioprinting for medical applications. A comparative analysis of polysaccharides derived from seaweed, plants, bacteria, and animals demonstrates that no single material provides a universal solution for all applications. Instead, each class offers distinct advantages, such as the rapid ionic gelation of alginate, mechanical reinforcement imparted by cellulose, or intrinsic bioactivity of hyaluronic acid, while simultaneously exhibiting inherent limitations, including mechanical fragility, bioinertness, or constrained processing windows. The progression from single-component bioinks to multi-network composites and hybrid material systems reflects the growing recognition that printability, mechanical integrity, and biological functionality must be independently engineered and subsequently integrated.
The performance of polysaccharide bioinks is intrinsically linked to the choice of bioprinting modality and crosslinking strategy used. While extrusion-based printing remains dominant, embedded approaches such as Freeform Reversible Embedding of Suspended Hydrogels (FRESH) have substantially expanded the range of printable, cell-compatible materials by decoupling structural support from ink rheology. In parallel, the adoption of orthogonal and multimodal crosslinking strategies has enabled temporal control over gelation, allowing bioinks to satisfy the competing demands of cytocompatible processing and post-printing mechanical stability. Nevertheless, this review reveals a critical imbalance: advances in geometric precision and architectural complexity have frequently outpaced progress in long-term biological performance. Persistent challenges, including limited cell survival, insufficient vascularization at physiologically relevant length scales, and inadequate integration with host tissues, continue to impede clinical translation.
Polysaccharide bioinks are evolving from passive structural matrices to dynamic, cell-instructive components within complex, multifunctional formulations. Their future lies not in standalone use but in rationally engineered hybrid systems that combine the sustainability and biocompatibility of polysaccharides with complementary bioactive, synthetic, or nanoscale elements. Addressing the intertwined trade-offs between material properties, processing constraints, and biological requirements is essential to realize bioprinted constructs that are not only precisely fabricated but also biologically functional and clinically viable.
4. Future Directions
Based on the critical analysis presented, several targeted research avenues are essential to propel polysaccharide-based bioinks from promising laboratory platforms to robust clinical solutions.
Development of predictive design frameworks: There is an urgent need for computational and experimental models to predict bioink performance. Integrating machine learning with high-throughput rheological and biological screening could establish quantitative relationships between molecular parameters (degree of substitution and molecular weight), ink properties (shear thinning index and yield stress), printing outcomes, and cellular responses. This would shift the field from empirical formulation to rational design.
Hybrid bioinks with enhanced biofunctionality: future efforts must prioritize the creation of “cell-instructive” bioinks. This involves moving beyond blending for rheology towards the strategic incorporation of engineered ECM components, peptide motifs (e.g., RGD and IKVAV), and controlled release systems for growth factors. The goal is to direct specific cellular behaviors, such as adhesion, proliferation, differentiation, and matrix deposition, in a spatially and temporally controlled manner.
Confronting the vascularization challenge: achieving perfusion-ready, capillary-scale (<50 µm) vascular networks within bioprinted constructs is a non-negotiable milestone. Research must focus on advanced techniques, such as sacrificial writing into functional tissue (SWIFT) or in-bath printing of co-cultured endothelial and supportive cells within specifically designed bioinks that facilitate rapid anastomosis and stability under flow.
Sustainability and scalability: as the field matures, the environmental impact and economic viability of bioink components become critical. Leveraging underutilized biorefinery streams (e.g., bacterial cellulose and pectin from agro-waste) and developing energy-efficient, aqueous-based processing and crosslinking methods will be vital for sustainable scale-up and widespread adoption.
In vivo validation and standardization: finally, rigorous long-term in vivo studies are required to evaluate the integration, degradation, immune response, and functional efficacy of polysaccharide-based constructs. Concurrently, the community must develop standardized protocols for characterizing printability, bioactivity, and safety to enable meaningful comparisons between studies and accelerate regulatory approval pathways.
By addressing these interdisciplinary challenges, the next generation of polysaccharide bioinks can fulfill their potential, enabling the 3D printing of living tissues that are structurally accurate, biologically viable, and therapeutically effective.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Acknowledgments
Not applicable.
Glossary of Key Terms and Abbreviations
| AGDA |
Alginate/Gelatin/Dextran-Aldehyde |
| CMC |
Carboxymethyl Cellulose |
| ECM |
Extracellular Matrix |
| FRESH |
Freeform Reversible Embedding of Suspended Hydrogels |
| GelMA |
Gelatin Methacryloyl |
| HA |
Hyaluronic Acid |
| HRP |
Horseradish Peroxidase |
| LAB |
Laser-Assisted Bioprinting |
| MB Bioink |
Microgel-Based Biphasic Bioink |
| MeHA |
Methacrylated Hyaluronic Acid |
| OBBs |
Organ Building Blocks |
| PVA |
Polyvinyl Alcohol |
| RGD |
Arginine-Glycine-Aspartate (cell-adhesive peptide) |
| ROS |
Reactive Oxygen Species |
| SPIRIT |
Supportive Perfusable Integrated Reversible Template |
| SWIFT |
Sacrificial Writing into Functional Tissue |
References
- Khizar, M., Zaib, M., Haider, S., Karimi, H., & Aminpoor, H. (2026). 3D-printed patient-specific surgical instruments and implants: a technological leap in precision medicine. Annals of Medicine and Surgery, 88(1), 969-971. [CrossRef]
- Prządka, M., Pająk, W., Kleinrok, J., Pec, J., Michno, K., Karpiński, R., & Baj, J. (2025). Advances in 3D Printing Applications for Personalized Orthopedic Surgery: From Anatomical Modeling to Patient-Specific Implants. Journal of Clinical Medicine, 14(11), 3989. [CrossRef]
- Kapoor, D. U., Pareek, A., Uniyal, P., Prajapati, B. G., Thanawuth, K., & Sriamornsak, P. (2025). Innovative applications of 3D printing in personalized medicine and complex drug delivery systems. iScience, 28(10). [CrossRef]
- Singh, S., Kumar, M., Kumar, D., Kumar, S., Chopra, S., & Bhatia, A. (2025). Therapeutic precision: unveiling the potential of 3D printing in drug delivery, tissue engineering, and regenerative medicine. 3D Printing and Additive Manufacturing, 12(5), 447-473. [CrossRef]
- Datta, S. (2025). State-of-art of 3D printing and bioprinting technology in various domains of biomedicine, tissue engineering and regenerative medicine. Frontiers in Bioengineering and Biotechnology, 13, 1703915. [CrossRef]
- Rumon, M. M. H., Akib, A. A., Sarkar, S. D., Khan, M. A. R., Uddin, M. M., Nasrin, D., & Roy, C. K. (2024). Polysaccharide-based hydrogels for advanced biomedical engineering applications. ACS Polymers Au, 4(6), 463-486. [CrossRef]
- Wu, X., Cheng, X., Kang, M., Dong, R., Zhao, J., & Qu, Y. (2023). Natural polysaccharide-based hydrogel bioprinting for articular cartilage repair. Frontiers in Materials, 10, 1204318. [CrossRef]
- Teixeira, M. C., Lameirinhas, N. S., Carvalho, J. P., Silvestre, A. J., Vilela, C., & Freire, C. S. (2022). A guide to polysaccharide-based hydrogel bioinks for 3D bioprinting applications. International journal of molecular sciences, 23(12), 6564. [CrossRef]
- Damiri, F., Fatimi, A., Liu, Y., Musuc, A. M., Fajardo, A. R., Gowda, B. J., ... & Okoro, O. V. (2025). Recent advances in 3D bioprinted polysaccharide hydrogels for biomedical applications: A comprehensive review. Carbohydrate polymers, 348, 122845. [CrossRef]
- Nagaraja, K., Dhokare, P., Bhattacharyya, A., & Noh, I. (2024). Recent advances in 3D bioprinting of polysaccharide-based bioinks for fabrication of bioengineered tissues. Molecular Systems Design & Engineering, 9(10), 977-999. [CrossRef]
- Picado-Tejero, D., Mendoza-Cerezo, L., Rodríguez-Rego, J. M., Macías-García, A., & Marcos-Romero, A. C. (2026). 3D Bioprinted Natural Hydrogels: Rheological Characterization, Cytotoxicity, and Printability Assessment of a Polysaccharide-Based Bioink. ACS Omega. [CrossRef]
- Jiang, T., Yang, Y., Lin, Z., Hong, Y., & Luo, Z. (2025). Modified Polysaccharides: Potential Biomaterials for Bioprinting. Journal of Functional Biomaterials, 16(9), 338. [CrossRef]
- Aghajani, M., Garshasbi, H. R., Naghib, S. M., & Mozafari, M. R. (2025). 3D Printing of Hydrogel Polysaccharides for Biomedical Applications: A Review. Biomedicines, 13(3), 731. [CrossRef]
- Berradi, A., Aziz, F., Achaby, M. E., Ouazzani, N., & Mandi, L. (2023). A comprehensive review of polysaccharide-based hydrogels as promising biomaterials. Polymers, 15(13), 2908. [CrossRef]
- Ho, T. C., Chang, C. C., Chan, H. P., Chung, T. W., Shu, C. W., Chuang, K. P., ... & Tyan, Y. C. (2022). Hydrogels: properties and applications in biomedicine. Molecules, 27(9), 2902. [CrossRef]
- Xie, T., Wang, H., Li, J., Xuan, L., Nazir, F., Chen, L., ... & Tang, G. (2025). Microgels for 3D Biofabrication. Aggregate, 6(11), e70166. [CrossRef]
- Mathur, V., Agarwal, P., Kasturi, M., Srinivasan, V., Seetharam, R. N., & Vasanthan, K. S. (2025). Innovative bioinks for 3D bioprinting: Exploring technological potential and regulatory challenges. Journal of Tissue Engineering, 16, 20417314241308022. [CrossRef]
- Muñoz-Castiblanco, T., Moreno-Marín, J. P., & Osorio, M. (2025). Natural macromolecule-based bioinks for 3D bioprinting: A systematic review of composition, physicochemical characterization, and biomedical applications. Bioprinting, e00407. [CrossRef]
- Ma, C., Du, L., Guo, Y., & Yang, X. (2024). A review of polysaccharide hydrogels as materials for skin repair and wound dressing: Construction, functionalization and challenges. International Journal of Biological Macromolecules, 280, 135838. [CrossRef]
- Li, S., Yao, F., Liu, Q., Tang, C., Zhuo, Y., Dai, M., ... & Zhong, X. (2025). Natural polysaccharide hydrogels: Design, preparation, and tissue engineering applications. Materials & Design, 114876. [CrossRef]
- Uysal, B., Madduma-Bandarage, U. S., Jayasinghe, H. G., & Madihally, S. (2025). 3D-Printed Hydrogels from Natural Polymers for Biomedical Applications: Conventional Fabrication Methods, Current Developments, Advantages, and Challenges. Gels, 11(3), 192. [CrossRef]
- Fang, W., Yang, M., Wang, L., Li, W., Liu, M., Jin, Y., ... & Fu, Q. (2023). Hydrogels for 3D bioprinting in tissue engineering and regenerative medicine: Current progress and challenges. International journal of bioprinting, 9(5), 759. [CrossRef]
- Ullah, M. W., Ul-Islam, M., Shehzad, A., Manan, S., Islam, S. U., Fatima, A., ... & Sebakhy, K. O. (2025). From bioinks to functional tissues and organs: advances, challenges, and the promise of 3D bioprinting. Macromolecular Materials and Engineering, 310(12), e00251. [CrossRef]
- Segneanu, A. E., Bejenaru, L. E., Bejenaru, C., Blendea, A., Mogoşanu, G. D., Biţă, A., & Boia, E. R. (2025). Advancements in hydrogels: a comprehensive review of natural and synthetic innovations for biomedical applications. Polymers, 17(15), 2026. [CrossRef]
- Santhamoorthy, M., & Kim, S. C. (2025). A Review of the Development of Biopolymer Hydrogel-Based Scaffold Materials for Drug Delivery and Tissue Engineering Applications. Gels, 11(3), 178. [CrossRef]
- Sanjanwala, D., Londhe, V., Trivedi, R., Bonde, S., Sawarkar, S., Kale, V., & Patravale, V. (2024). Polysaccharide-based hydrogels for medical devices, implants and tissue engineering: A review. International Journal of Biological Macromolecules, 256, 128488. [CrossRef]
- Elango, J., & Zamora-Ledezma, C. (2025). Rheological, structural, and biological trade-offs in bioink design for 3d bioprinting. Gels, 11(8), 659. [CrossRef]
- Liu, G., Wei, X., Li, J., Zhai, Y., Zhang, J., Jin, M., ... & Zhao, D. (2024). Hydrogel composition and mechanical stiffness of 3D bioprinted cell-loaded scaffolds promote cartilage regeneration. Frontiers in Materials, 11, 1501505. [CrossRef]
- Maillard, M., Dujardin, C., Aprile, P., Aid, R., Letourneur, D., & Simon-Yarza, T. (2025). Double-network polysaccharide hydrogel for guided tissue repair. Biomaterials Science, 13(20), 5741-5754. [CrossRef]
- Mohammed, A. S. A., Naveed, M., & Jost, N. (2021). Polysaccharides; classification, chemical properties, and future perspective applications in fields of pharmacology and biological medicine (a review of current applications and upcoming potentialities). Journal of Polymers and the Environment, 29(8), 2359-2371. [CrossRef]
- Xue, H., Wang, P., Ji, L., Zhang, K., Ge, S., & Tan, J. (2025). Polysaccharide-based hydrogels: Materials, preparation, and applications in medicine, food, adsorption, and agriculture. Journal of Agriculture and Food Research, 102395. [CrossRef]
- Berradi, A., Aziz, F., Achaby, M. E., Ouazzani, N., & Mandi, L. (2023). A comprehensive review of polysaccharide-based hydrogels as promising biomaterials. Polymers, 15(13), 2908. [CrossRef]
- Rossi, A., Pescara, T., Gambelli, A. M., Gaggia, F., Asthana, A., Perrier, Q., ... & Calafiore, R. (2024). Biomaterials for extrusion-based bioprinting and biomedical applications. Frontiers in Bioengineering and Biotechnology, 12, 1393641. [CrossRef]
- Beaumont, M., Tran, R., Vera, G., Niedrist, D., Rousset, A., Pierre, R., ... & Forget, A. (2021). Hydrogel-forming algae polysaccharides: From seaweed to biomedical applications. Biomacromolecules, 22(3), 1027-1052. [CrossRef]
- Datta, S. (2023). Advantage of alginate bioinks in biofabrication for various tissue engineering applications. International Journal of Polymer Science, 2023(1), 6661452. [CrossRef]
- Axpe, E., & Oyen, M. L. (2016). Applications of alginate-based bioinks in 3D bioprinting. International journal of molecular sciences, 17(12), 1976. [CrossRef]
- Abka-Khajouei, R., Tounsi, L., Shahabi, N., Patel, A. K., Abdelkafi, S., & Michaud, P. (2022). Structures, properties and applications of alginates. Marine drugs, 20(6), 364. [CrossRef]
- Liu, W., Zhong, Z., Hu, N., Zhou, Y., Maggio, L., Miri, A. K., ... & Zhang, Y. S. (2018). Coaxial extrusion bioprinting of 3D microfibrous constructs with cell-favorable gelatin methacryloyl microenvironments. Biofabrication, 10(2), 024102. [CrossRef]
- Tarassoli, S. P., Jessop, Z. M., Jovic, T., Hawkins, K., & Whitaker, I. S. (2021). Candidate bioinks for extrusion 3D bioprinting—A systematic review of the literature. Frontiers in bioengineering and biotechnology, 9, 616753. [CrossRef]
- Gonzalez-Fernandez, T., Tenorio, A. J., Campbell, K. T., Silva, E. A., & Leach, J. K. (2021). Alginate-based bioinks for 3D bioprinting and fabrication of anatomically accurate bone grafts. Tissue Engineering Part A, 27(17-18), 1168-1181. [CrossRef]
- Gao, Q., Kim, B. S., & Gao, G. (2021). Advanced strategies for 3D bioprinting of tissue and organ analogs using alginate hydrogel bioinks. Marine Drugs, 19(12), 708. [CrossRef]
- Neves, M. I., Moroni, L., & Barrias, C. C. (2020). Modulating alginate hydrogels for improved biological performance as cellular 3D microenvironments. Frontiers in bioengineering and biotechnology, 8, 665. [CrossRef]
- Cavallo, A., Radaelli, G., Al Kayal, T., Mero, A., Mezzetta, A., Guazzelli, L., ... & Losi, P. (2025). Optimization of gelatin and crosslinker concentrations in a gelatin/alginate-based bioink with potential applications in a simplified skin model. Molecules, 30(3), 649. [CrossRef]
- Jahani Kadousaraei, M., Yamada, S., Aydin, M. S., Rashad, A., Cabeza, N. M., Mohamed-Ahmed, S., ... & Mustafa, K. (2025). Bioprinting of mesenchymal stem cells in low concentration gelatin methacryloyl/alginate blends without ionic crosslinking of alginate. Scientific Reports, 15(1), 6609. [CrossRef]
- Tripathi, S., Mandal, S. S., Bauri, S., & Maiti, P. (2023). 3D bioprinting and its innovative approach for biomedical applications. MedComm, 4(1), e194. [CrossRef]
- Bartolo, P., Malshe, A., Ferraris, E., & Koc, B. (2022). 3D bioprinting: Materials, processes, and applications. CIRP Annals, 71(2), 577-597. [CrossRef]
- López-Marcial, G. R., Zeng, A. Y., Osuna, C., Dennis, J., García, J. M., & O’Connell, G. D. (2018). Agarose-based hydrogels as suitable bioprinting materials for tissue engineering. ACS biomaterials science & engineering, 4(10), 3610-3616. [CrossRef]
- Wenger, L., Radtke, C. P., Gerisch, E., Kollmann, M., Niemeyer, C. M., Rabe, K. S., & Hubbuch, J. (2022). Systematic evaluation of agarose-and agar-based bioinks for extrusion-based bioprinting of enzymatically active hydrogels. Frontiers in Bioengineering and Biotechnology, 10, 928878. [CrossRef]
- Sekar, M. P., Budharaju, H., Sethuraman, S., & Sundaramurthi, D. (2023). Carboxymethyl cellulose-agarose-gelatin: A thermoresponsive triad bioink composition to fabricate volumetric soft tissue constructs. SLAS technology, 28(3), 183-198. [CrossRef]
- Marques, D. M., Silva, J. C., Serro, A. P., Cabral, J. M., Sanjuan-Alberte, P., & Ferreira, F. C. (2022). 3D bioprinting of novel κ-carrageenan bioinks: An algae-derived polysaccharide. Bioengineering, 9(3), 109. [CrossRef]
- Muscolino, E., Di Stefano, A. B., Trapani, M., Sabatino, M. A., Giacomazza, D., Alessi, S., ... & Dispenza, C. (2022). κ-Carrageenan and PVA blends as bioinks to 3D print scaffolds for cartilage reconstruction. International Journal of Biological Macromolecules, 222, 1861-1875. [CrossRef]
- Kumari, S., Mondal, P., Tyeb, S., & Chatterjee, K. (2024). Visible light-based 3D bioprinted composite scaffolds of κ-carrageenan for bone tissue engineering applications. Journal of Materials Chemistry B, 12(7), 1926-1936. [CrossRef]
- Kansız, S., Vurat, M. T., Parmaksiz, M., Elçin, A. E., & Elçin, Y. M. (2025). Methacrylated-κ-carrageenan/hydroxyapatite composite bioinks for extrusion-based bioprinting: Physicochemical, rheological, mechanical, and in vitro biological investigations. International Journal of Biological Macromolecules, 144745. [CrossRef]
- Almeida, D., Küppers, F., Gusmão, A., Manjua, A. C., Ferreira, C. F., Portugal, C. A., ... & Ferreira, F. C. (2024). Design of magnetic kappa-carrageenan-collagen bioinks for 3D bioprinting. Journal of Materials Science, 59(31), 14573-14592. [CrossRef]
- Zhang, H., Zhu, T., Luo, Y., Xu, R., Li, G., Hu, Z., ... & Wu, K. (2024). Embedded Bioprinting of Tissue-like Structures Using κ-Carrageenan Sub-Microgel Medium. Journal of Visualized Experiments (JoVE), (207), e66806. [CrossRef]
- Machour, M., Hen, N., Goldfracht, I., Safina, D., Davidovich-Pinhas, M., Bianco-Peled, H., & Levenberg, S. (2022). Print-and-Grow within a novel support material for 3D bioprinting and post-printing tissue growth. Advanced Science, 9(34), 2200882. [CrossRef]
- Shahbazi, M., & Jäger, H. (2020). Current status in the utilization of biobased polymers for 3D printing process: a systematic review of the materials, processes, and challenges. ACS Applied Bio Materials, 4(1), 325-369. [CrossRef]
- Huang, J., & Li, L. (2025). 3D printing of cellulose-based biomaterials: A review. BME Horizon, 3(1), 138-138. [CrossRef]
- Patel, D. K., Cha, J., Won, S. Y., & Han, S. S. (2025). 3D printing of cellulose-based biopolymers: Materials, processing strategies, and biomedical applications. International Journal of Biological Macromolecules, 149077. [CrossRef]
- Wang, X., Wang, Q., & Xu, C. (2020). Nanocellulose-based inks for 3d bioprinting: Key aspects in research development and challenging perspectives in applications—A mini review. Bioengineering, 7(2), 40. [CrossRef]
- Utoiu, E., Manoiu, V. S., Oprita, E. I., & Craciunescu, O. (2024). Bacterial cellulose: A sustainable source for hydrogels and 3D-Printed scaffolds for tissue engineering. Gels, 10(6), 387. [CrossRef]
- Jovic, T. H., Gazze, A., Morgan, B., Hawkins, K., Francis, L., Arora, H., ... & Whitaker, I. S. (2025). Development of nanocellulose-hyaluronic acid bioinks for 3D bioprinting facial cartilages. Carbohydrate Polymer Technologies and Applications, 100929. [CrossRef]
- Grunberg, N., Mcmeeking, A., Dieckmann, E., & Cheeseman, C. (2024). Development of printable bacterial nanocellulose bioinks for bioprinting applications. Biotechnology for Sustainable Materials, 1(1), 14. [CrossRef]
- Carvalho, J. P., Lameirinhas, N. S., Teixeira, M. C., Luís, J. L., Oliveira, H., Oliveira, J. M., ... & Freire, C. S. (2025). All-Cellulose Hydrogel-Based Bioinks for the Versatile 3D Bioprinting of Different Cell Lines. Biomacromolecules, 26(3), 1761-1770. [CrossRef]
- Sharma, C., Raza, M. A., Purohit, S. D., Pathak, P., Gautam, S., Corridon, P. R., & Han, S. S. (2025). Cellulose-based 3D printing bio-inks for biomedical applications: A review. International Journal of Biological Macromolecules, 141174. [CrossRef]
- Habib, M. A., & Khoda, B. (2022). Rheological analysis of bio-ink for 3D bio-printing processes. Journal of manufacturing processes, 76, 708-718. [CrossRef]
- Wang, Y., Yue, H., Liu, A., Cui, Y., Hou, Y., Ni, X., ... & Bartolo, P. (2025). Dual crosslinkable bioink for direct and embedded 3D bioprinting at physiological temperature. Materials Today, 85, 1-16. [CrossRef]
- Lee, J., Lee, S., Lim, J. W., Byun, I., Jang, K. J., Kim, J. W., ... & Seonwoo, H. (2023). Development of plum seed-derived carboxymethylcellulose bioink for 3D bioprinting. Polymers, 15(23), 4473. [CrossRef]
- Pitton, M., Fiorati, A., Buscemi, S., Melone, L., Farè, S., & Contessi Negrini, N. (2021). 3D bioprinting of pectin-cellulose nanofibers multicomponent bioinks. Frontiers in Bioengineering and Biotechnology, 9, 732689. [CrossRef]
- Merli, M., Sardelli, L., Baranzini, N., Grimaldi, A., Jacchetti, E., Raimondi, M. T., ... & Tunesi, M. (2022). Pectin-based bioinks for 3D models of neural tissue produced by a pH-controlled kinetics. Frontiers in Bioengineering and Biotechnology, 10, 1032542. [CrossRef]
- Chang, H. C., Jørgensen, B., Di Silvio, L., & Gurzawska-Comis, K. (2023). 3D bioprinted pectin-based hydrogel as sustainable biomaterials for musculoskeletal tissue engineering. Sustainable Materials and Technologies, 38, e00732. [CrossRef]
- Cernencu, A. I., Lungu, A., Stancu, I. C., Serafim, A., Heggset, E., Syverud, K., & Iovu, H. (2019). Bioinspired 3D printable pectin-nanocellulose ink formulations. Carbohydrate Polymers, 220, 12-21. [CrossRef]
- Zhuang, P., Greenberg, Z., & He, M. (2021). Biologically enhanced starch bio-ink for promoting 3D cell growth. Advanced materials technologies, 6(12), 2100551. [CrossRef]
- Mollaheydaralimoazzen, M., Sheikholeslam, M., Poursamar, S. A., Farzan, M., Farzan, M., & Rafienia, M. (2025). 3D-printing of shear-thinning and self-healing gelatin/starch/halloysite-nanotube hydrogels for soft tissue engineering: An in vitro and in vivo assessment. International Journal of Biological Macromolecules, 144502. [CrossRef]
- Zhou, X., Wu, S., Liu, P., Wang, L., & Xie, F. (2025). Hybrid bioink of methyacrylated starch with minimal methacrylated chitosan enables high-precision 3D printing for complex tissue scaffolds. Carbohydrate Polymers, 367, 124023. [CrossRef]
- Tipa, C., Cidade, M. T., Borges, J. P., Costa, L. C., Silva, J. C., & Soares, P. I. (2022). Clay-based nanocomposite hydrogels for biomedical applications: a review. Nanomaterials, 12(19), 3308. [CrossRef]
- Su, C., Chen, Y., Tian, S., Lu, C., & Lv, Q. (2022). Natural materials for 3D printing and their applications. Gels, 8(11), 748. [CrossRef]
- Taniguchi Nagahara, M. H., Caiado Decarli, M., Inforcatti Neto, P., Lopes da Silva, J. V., & Moraes, A. M. (2022). Crosslinked alginate-xanthan gum blends as effective hydrogels for 3D bioprinting of biological tissues. Journal of Applied Polymer Science, 139(28), e52612. [CrossRef]
- Lameirinhas, N. S., Teixeira, M. C., Carvalho, J. P., Valente, B. F., Pinto, R. J., Oliveira, H., ... & Freire, C. S. (2023). Nanofibrillated cellulose/gellan gum hydrogel-based bioinks for 3D bioprinting of skin cells. International Journal of Biological Macromolecules, 229, 849-860. [CrossRef]
- Ouiyangkul, P., Hirun, N., Suknuntha, K., & Tantishaiyakul, V. (2024). Development and characterization of 3D bioprintable and mechanically reinforced hydrogel based on gellan gum/methylcellulose/cellulose nanocrystals. Polymers for Advanced Technologies, 35(1), e6206. [CrossRef]
- Patrocinio, D., Galván-Chacón, V., Gómez-Blanco, J. C., Miguel, S. P., Loureiro, J., Ribeiro, M. P., ... & Sanchez-Margallo, F. M. (2023). Biopolymers for tissue engineering: crosslinking, printing techniques, and applications. Gels, 9(11), 890. [CrossRef]
- Lim, H., Seo, D., Park, K. D., Shim, H. E., Park, J. H., Ann, E. J., ... & Kang, S. W. (2025). Optimization of alginate/gelatin/dextran-aldehyde bioink for 3D bioprinting and cell engraftment. International Journal of Bioprinting, 8440. [CrossRef]
- Gopinathan, J., & Noh, I. (2018). Recent trends in bioinks for 3D printing. Biomaterials research, 22(1), 11. [CrossRef]
- Taghizadeh, M., Taghizadeh, A., Yazdi, M. K., Zarrintaj, P., Stadler, F. J., Ramsey, J. D., ... & Schubert, U. S. (2022). Chitosan-based inks for 3D printing and bioprinting. Green Chemistry, 24(1), 62-101. [CrossRef]
- Apăvăloaiei, I., Nacu, I., Cojocaru, F. D., Balan, V., Bercea, M., Niță, L. E., & Vereștiuc, L. (2025). Effect of chitosan on 3D printed scaffolds with gelatin–hyaluronic acid, hydroxyapatite and magnetic nanoparticles for bone tissues defects repair. Reactive and Functional Polymers, 106422. [CrossRef]
- Maturavongsadit, P., Narayanan, L. K., Chansoria, P., Shirwaiker, R., & Benhabbour, S. R. (2021). Cell-laden nanocellulose/chitosan-based bioinks for 3D bioprinting and enhanced osteogenic cell differentiation. ACS Applied Bio Materials, 4(3), 2342-2353. [CrossRef]
- Tsanaktsidou, E., Kammona, O., Labude, N., Neuss, S., Krüger, M., Kock, L., & Kiparissides, C. (2020). Biomimetic cell-laden MeHA hydrogels for the regeneration of cartilage tissue. Polymers, 12(7), 1598. [CrossRef]
- Mouser, V. H. M., Abbadessa, A., Levato, R., Hennink, W. E., Vermonden, T., Gawlitta, D., & Malda, J. (2017). Development of a thermosensitive HAMA-containing bio-ink for the fabrication of composite cartilage repair constructs. Biofabrication, 9(1), 015026. [CrossRef]
- Bebiano, L. B., Presa, R., Vieira, F., Lourenço, B. N., & Pereira, R. F. (2024). Bioinspired and photo-clickable thiol-ene bioinks for the extrusion bioprinting of mechanically tunable 3D skin models. Biomimetics, 9(4), 228. [CrossRef]
- Calafel, M. I., Criado-Gonzalez, M., Aguirresarobe, R., Fernández, M., & Mijangos, C. (2025). From rheological concepts to additive manufacturing assessment of hydrogel-based materials for advanced bioprinting applications. Materials Advances, 6(14), 4566-4597. [CrossRef]
- Sánchez-Sánchez, R., Rodríguez-Rego, J. M., Macías-García, A., Mendoza-Cerezo, L., & Díaz-Parralejo, A. (2023). Relationship between shear-thinning rheological properties of bioinks and bioprinting parameters. International Journal of Bioprinting, 9(2), 687. [CrossRef]
- Kruczkowska, W., Gałęziewska, J., Grabowska, K. H., Gromek, P., Czajkowska, K., Rybicki, M., ... & Kłosiński, K. K. (2025). From Molecules to Mind: The Critical Role of Chitosan, Collagen, Alginate, and Other Biopolymers in Neuroprotection and Neurodegeneration. Molecules, 30(5), 1017. [CrossRef]
- Zhang, H., Shi, L. W. E., & Zhou, J. (2023). Recent developments of polysaccharide-based double-network hydrogels. Journal of Polymer Science, 61(1), 7-43. [CrossRef]
- Greco, I., Machrafi, H., & Iorio, C. S. (2024). Double-network hydrogel 3D bioprinting biocompatible with fibroblast cells for tissue engineering applications. Gels, 10(11), 684. [CrossRef]
- Shen, S., Shen, J., Shen, H., Wu, C., Chen, P., & Wang, Q. (2020). Dual-enzyme crosslinking and post-polymerization for printing of polysaccharide-polymer hydrogel. Frontiers in Chemistry, 8, 36. [CrossRef]
- Psarrou, M., Mitraki, A., Vamvakaki, M., & Kokotidou, C. (2023). Stimuli-responsive polysaccharide hydrogels and their composites for wound healing applications. Polymers, 15(4), 986. [CrossRef]
- Khiari, Z. (2024). Recent developments in bio-ink formulations using marine-derived biomaterials for three-dimensional (3D) bioprinting. Marine Drugs, 22(3), 134. [CrossRef]
- Unagolla, J. M., & Jayasuriya, A. C. (2020). Hydrogel-based 3D bioprinting: A comprehensive review on cell-laden hydrogels, bioink formulations, and future perspectives. Applied materials today, 18, 100479. [CrossRef]
- Bittner, S. M., Pearce, H. A., Hogan, K. J., Smoak, M. M., Guo, J. L., Melchiorri, A. J., ... & Mikos, A. G. (2021). Swelling behaviors of 3D printed hydrogel and hydrogel-microcarrier composite scaffolds. Tissue Engineering Part A, 27(11-12), 665-678. [CrossRef]
- Gebeyehu, A., Surapaneni, S. K., Huang, J., Mondal, A., Wang, V. Z., Haruna, N. F., ... & Singh, M. (2021). Polysaccharide hydrogel based 3D printed tumor models for chemotherapeutic drug screening. Scientific reports, 11(1), 372. [CrossRef]
- Kronemberger, G. S., Spagnuolo, F. D., Karam, A. S., Chattahy, K., Storey, K. J., & Kelly, D. J. (2024). Rapidly degrading hydrogels to support biofabrication and 3D bioprinting using cartilage microtissues. ACS Biomaterials Science & Engineering, 10(10), 6441-6450. [CrossRef]
- Son, J., Li, S., & Jeong, W. (2025). Bioprinting vascularized constructs for clinical relevance: Engineering hydrogel systems for biological maturity. Gels, 11(8), 636. [CrossRef]
- Prakash, A., Malviya, R., Sridhar, S. B., Wadhwa, T., Shareef, J., & Tripathy, D. B. (2025). Polysaccharides as functional biomaterials in 3D bioprinting: Strategies for antimicrobial applications. Next Materials, 9, 101255. [CrossRef]
- Rijal, G. (2023). Bioinks of natural biomaterials for printing tissues. Bioengineering, 10(6), 705. [CrossRef]
- Lupu, A., Gradinaru, L. M., Gradinaru, V. R., & Bercea, M. (2023). Diversity of bioinspired hydrogels: From structure to applications. Gels, 9(5), 376. [CrossRef]
- Jin, A., Shao, Y., Wang, F., Feng, J., Lei, L., & Dai, M. (2024). Designing polysaccharide materials for tissue repair and regeneration. APL Materials, 12(8). [CrossRef]
- Träger, A., Naeimipour, S., Jury, M., Selegård, R., & Aili, D. (2023). Nanocellulose reinforced hyaluronan-based bioinks. Biomacromolecules, 24(7), 3086-3093. [CrossRef]
- Tavakoli, S., Krishnan, N., Mokhtari, H., Oommen, O. P., & Varghese, O. P. (2024). Fine-tuning dynamic cross–linking for enhanced 3D bioprinting of hyaluronic acid hydrogels. Advanced Functional Materials, 34(4), 2307040. [CrossRef]
- Tavakoli, S., Kocatürkmen, A., Oommen, O. P., & Varghese, O. P. (2025). Ultra-fine 3D bioprinting of dynamic hyaluronic acid hydrogel for in vitro modeling. Advanced Materials, 2500315. [CrossRef]
- Pereira, I., Lopez-Martinez, M. J., Villasante, A., Introna, C., Tornero, D., Canals, J. M., & Samitier, J. (2023). Hyaluronic acid-based bioink improves the differentiation and network formation of neural progenitor cells. Frontiers in Bioengineering and Biotechnology, 11, 1110547. [CrossRef]
- Kim, M. H., Lee, Y. W., Jung, W. K., Oh, J., & Nam, S. Y. (2019). Enhanced rheological behaviors of alginate hydrogels with carrageenan for extrusion-based bioprinting. Journal of the mechanical behavior of biomedical materials, 98, 187-194. [CrossRef]
- Lim, W., Kim, G. J., Kim, H. W., Lee, J., Zhang, X., Kang, M. G., ... & Bae, H. (2020). Kappa-carrageenan-based dual crosslinkable bioink for extrusion type bioprinting. Polymers, 12(10), 2377. [CrossRef]
- Sahranavard, M., Zamanian, A., Ghader, A. B., & Shahrezaee, M. (2025). Optimization of gellan gum-based bioink printability for precision 3D bioprinting in tissue engineering. International Journal of Biological Macromolecules, 145800. [CrossRef]
- Jongprasitkul, H., Turunen, S., Parihar, V. S., & Kellomäki, M. (2022). Two-step crosslinking to enhance the printability of methacrylated gellan gum biomaterial ink for extrusion-based 3D bioprinting. Bioprinting, 25, e00185. [CrossRef]
- Iervolino, F., Belgio, B., Bonessa, A., Potere, F., Suriano, R., Boschetti, F., ... & Levi, M. (2023). Versatile and non-cytotoxic GelMA-xanthan gum biomaterial ink for extrusion-based 3D bioprinting. Bioprinting, 31, e00269. [CrossRef]
- Garcia-Cruz, M. R., Postma, A., Frith, J. E., & Meagher, L. (2021). Printability and bio-functionality of a shear thinning methacrylated xanthan–gelatin composite bioink. Biofabrication, 13(3), 035023. [CrossRef]
- El-Husseiny, H. M., Mady, E. A., Hamabe, L., Abugomaa, A., Shimada, K., Yoshida, T., ... & Tanaka, R. (2022). Smart/stimuli-responsive hydrogels: Cutting-edge platforms for tissue engineering and other biomedical applications. Materials Today Bio, 13, 100186. [CrossRef]
- Pardeshi, S., Damiri, F., Zehravi, M., Joshi, R., Kapare, H., Prajapati, M. K., ... & Barai, H. R. (2022). Functional thermoresponsive hydrogel molecule to material design for biomedical applications. Polymers, 14(15), 3126. [CrossRef]
- Neumann, M., di Marco, G., Iudin, D., Viola, M., van Nostrum, C. F., van Ravensteijn, B. G., & Vermonden, T. (2023). Stimuli-responsive hydrogels: the dynamic smart biomaterials of tomorrow. Macromolecules, 56(21), 8377-8392. [CrossRef]
- Mohamed, M. A., Fallahi, A., El-Sokkary, A. M., Salehi, S., Akl, M. A., Jafari, A., ... & Cheng, C. (2019). Stimuli-responsive hydrogels for manipulation of cell microenvironment: From chemistry to biofabrication technology. Progress in polymer science, 98, 101147. [CrossRef]
- Arif, Z. U., Khalid, M. Y., Tariq, A., Hossain, M., & Umer, R. (2024). 3D printing of stimuli-responsive hydrogel materials: Literature review and emerging applications. Giant, 17, 100209. [CrossRef]
- Tran, T. S., Balu, R., Mettu, S., Roy Choudhury, N., & Dutta, N. K. (2022). 4D printing of hydrogels: innovation in material design and emerging smart systems for drug delivery. Pharmaceuticals, 15(10), 1282. [CrossRef]
- Patroklou, G., Triantafyllopoulou, E., Goula, P. E., Karali, V., Chountoulesi, M., Valsami, G., ... & Pippa, N. (2025). pH-Responsive Hydrogels: Recent Advances in Pharmaceutical Applications. Polymers, 17(11), 1451. [CrossRef]
- Chen, X. B., Anvari-Yazdi, A. F., Duan, X., Zimmerling, A., Gharraei, R., Sharma, N. K., ... & Ning, L. (2023). Biomaterials/bioinks and extrusion bioprinting. Bioactive Materials, 28, 511-536. [CrossRef]
- Gillispie, G. J., Copus, J., Uzun-Per, M., Yoo, J. J., Atala, A., Niazi, M. K. K., & Lee, S. J. (2023). The correlation between rheological properties and extrusion-based printability in bioink artifact quantification. Materials & design, 233, 112237. [CrossRef]
- Cooke, M. E., & Rosenzweig, D. H. (2021). The rheology of direct and suspended extrusion bioprinting. APL bioengineering, 5(1). [CrossRef]
- Gao, T., Gillispie, G. J., Copus, J. S., Pr, A. K., Seol, Y. J., Atala, A., ... & Lee, S. J. (2018). Optimization of gelatin–alginate composite bioink printability using rheological parameters: A systematic approach. Biofabrication, 10(3), 034106. [CrossRef]
- Kim, M. S., Choi, Y., & Lee, K. Y. (2025). Three-Dimensional Printing and Bioprinting Strategies for Cardiovascular Constructs: From Printing Inks to Vascularization. Polymers, 17(17), 2337. [CrossRef]
- Barui, S. (2021). 3D inkjet printing of biomaterials: Principles and applications. Medical Devices & Sensors, 4(1), e10143. [CrossRef]
- Tharakan, S., Khondkar, S., & Ilyas, A. (2021). Bioprinting of stem cells in multimaterial scaffolds and their applications in bone tissue engineering. Sensors, 21(22), 7477. [CrossRef]
- Guida, L., Cavallaro, M., & Levi, M. (2024). Advancements in high-resolution 3D bioprinting: exploring technological trends, bioinks and achieved resolutions. Bioprinting, e00376. [CrossRef]
- Fang, W., Yu, Z., Gao, G., Yang, M., Du, X., Wang, Y., & Fu, Q. (2024). Light-based 3D bioprinting technology applied to repair and regeneration of different tissues: A rational proposal for biomedical applications. Materials Today Bio, 27, 101135. [CrossRef]
- Zheng, Z., Eglin, D., Alini, M., Richards, G. R., Qin, L., & Lai, Y. (2021). Visible light-induced 3D bioprinting technologies and corresponding bioink materials for tissue engineering: a review. Engineering, 7(7), 966-978. [CrossRef]
- Wang, M., Li, W., Hao, J., Gonzales III, A., Zhao, Z., Flores, R. S., ... & Zhang, Y. S. (2022). Molecularly cleavable bioinks facilitate high-performance digital light processing-based bioprinting of functional volumetric soft tissues. Nature communications, 13(1), 3317. [CrossRef]
- Ghosh, R. N., Thomas, J., Janardanan, A., Namboothiri, P. K., & Peter, M. (2023). An insight into synthesis, properties and applications of gelatin methacryloyl hydrogel for 3D bioprinting. Materials Advances, 4(22), 5496-5529. [CrossRef]
- Dou, C., Perez, V., Qu, J., Tsin, A., Xu, B., & Li, J. (2021). A state-of-the-art review of laser-assisted bioprinting and its future research trends. ChemBioEng Reviews, 8(5), 517-534.v . [CrossRef]
- Ventura, R. D. (2021). An overview of laser-assisted bioprinting (LAB) in tissue engineering applications. Medical Lasers; Engineering, Basic Research, and Clinical Application, 10(2), 76-81. [CrossRef]
- Zeng, X., Meng, Z., He, J., Mao, M., Li, X., Chen, P., ... & Li, D. (2022). Embedded bioprinting for designer 3D tissue constructs with complex structural organization. Acta biomaterialia, 140, 1-22. [CrossRef]
- Yuan, T. Y., Zhang, J., Yu, T., Wu, J. P., & Liu, Q. Y. (2022). 3D bioprinting for spinal cord injury repair. Frontiers in bioengineering and biotechnology, 10, 847344. [CrossRef]
- Öztürk-Öncel, M. Ö., Leal-Martínez, B. H., Monteiro, R. F., Gomes, M. E., & Domingues, R. M. (2023). A dive into the bath: embedded 3D bioprinting of freeform in vitro models. Biomaterials Science, 11(16), 5462-5473. [CrossRef]
- Brunel, L. G., Hull, S. M., & Heilshorn, S. C. (2022). Engineered assistive materials for 3D bioprinting: support baths and sacrificial inks. Biofabrication, 14(3), 032001. [CrossRef]
- Budharaju, H., Sundaramurthi, D., & Sethuraman, S. (2024). Embedded 3D bioprinting–An emerging strategy to fabricate biomimetic & large vascularized tissue constructs. Bioactive Materials, 32, 356-384. [CrossRef]
- Hinton, T. J., Jallerat, Q., Palchesko, R. N., Park, J. H., Grodzicki, M. S., Shue, H. J., ... & Feinberg, A. W. (2015). Three-dimensional printing of complex biological structures by freeform reversible embedding of suspended hydrogels. Science advances, 1(9), e1500758. [CrossRef]
- Wu, C. A., Zhu, Y., Venkatesh, A., Stark, C. J., Lee, S. H., & Woo, Y. J. (2023). Optimization of freeform reversible embedding of suspended hydrogel microspheres for substantially improved three-dimensional bioprinting capabilities. Tissue Engineering Part C: Methods, 29(3), 85-94. [CrossRef]
- Lee, A. R. H. A., Hudson, A. R., Shiwarski, D. J., Tashman, J. W., Hinton, T. J., Yerneni, S., ... & Feinberg, A. W. (2019). 3D bioprinting of collagen to rebuild components of the human heart. Science, 365(6452), 482-487. [CrossRef]
- Skylar-Scott, M. A., Uzel, S. G., Nam, L. L., Ahrens, J. H., Truby, R. L., Damaraju, S., & Lewis, J. A. (2019). Biomanufacturing of organ-specific tissues with high cellular density and embedded vascular channels. Science advances, 5(9), eaaw2459. [CrossRef]
- Mirdamadi, E., Muselimyan, N., Koti, P., Asfour, H., & Sarvazyan, N. (2019). Agarose slurry as a support medium for bioprinting and culturing freestanding cell-laden hydrogel constructs. 3D printing and additive manufacturing, 6(3), 158-164. [CrossRef]
- You, S., Xiang, Y., Hwang, H. H., Berry, D. B., Kiratitanaporn, W., Guan, J., ... & Chen, S. (2023). High cell density and high-resolution 3D bioprinting for fabricating vascularized tissues. Science advances, 9(8), eade7923. [CrossRef]
- Seiti, M., Mazzoldi, E. L., Pandini, S., Giliani, S., Ferraris, E., Ginestra, P. S., & Ceretti, E. (2024). FRESH 3D Bioprinting of Alginate–Cellulose–Gelatin Constructs for Soft Tissue Biofabrication. Procedia CIRP, 125, 42-47. [CrossRef]
- Asim, S., Tabish, T. A., Liaqat, U., Ozbolat, I. T., & Rizwan, M. (2023). Advances in gelatin bioinks to optimize bioprinted cell functions. Advanced healthcare materials, 12(17), 2203148. [CrossRef]
- Shiwarski, D. J., Hudson, A. R., Tashman, J. W., Bakirci, E., Moss, S., Coffin, B. D., & Feinberg, A. W. (2025). 3D bioprinting of collagen-based high-resolution internally perfusable scaffolds for engineering fully biologic tissue systems. Science Advances, 11(17), eadu5905. [CrossRef]
- Wolf, K. J., Weiss, J. D., Uzel, S. G., Skylar-Scott, M. A., & Lewis, J. A. (2022). Biomanufacturing human tissues via organ building blocks. Cell Stem Cell, 29(5), 667-677. [CrossRef]
- Shao, L., Jiang, J., Yuan, C., Zhang, X., Gu, L., & Wang, X. (2024). Omnidirectional anisotropic embedded 3D bioprinting. Materials Today Bio, 27, 101160. [CrossRef]
- Fang, Y., Guo, Y., Wu, B., Liu, Z., Ye, M., Xu, Y., ... & Xiong, Z. (2023). Expanding embedded 3D bioprinting capability for engineering complex organs with freeform vascular networks. Advanced Materials, 35(22), 2205082. [CrossRef]
- GhavamiNejad, A., Ashammakhi, N., Wu, X. Y., & Khademhosseini, A. (2020). Crosslinking strategies for 3D bioprinting of polymeric hydrogels. Small, 16(35), 2002931. [CrossRef]
- Zennifer, A., Manivannan, S., Sethuraman, S., Kumbar, S. G., & Sundaramurthi, D. (2022). 3D bioprinting and photocrosslinking: emerging strategies & future perspectives. Biomaterials advances, 134, 112576. [CrossRef]
- Gungor-Ozkerim, P. S., Inci, I., Zhang, Y. S., Khademhosseini, A., & Dokmeci, M. R. (2018). Bioinks for 3D bioprinting: an overview. Biomaterials science, 6(5), 915-946. [CrossRef]
- Ribeiro, M. M., Simões, M., Vitorino, C., & Mascarenhas-Melo, F. (2025). Physical crosslinking of hydrogels: The potential of dynamic and reversible bonds in burn care. Coordination Chemistry Reviews, 542, 216868. [CrossRef]
- Lazaridou, M., Bikiaris, D. N., & Lamprou, D. A. (2022). 3D bioprinted chitosan-based hydrogel scaffolds in tissue engineering and localised drug delivery. Pharmaceutics, 14(9), 1978. [CrossRef]
- Stojkov, G., Niyazov, Z., Picchioni, F., & Bose, R. K. (2021). Relationship between structure and rheology of hydrogels for various applications. Gels, 7(4), 255. [CrossRef]
- Bu, N., Li, L., & Hu, X. (2023). Recent trends in natural polymer-based hydrogels for biomedical applications. Biofunctional Materials, 2(1), 1-46. [CrossRef]
- Douglas, A., Chen, Y., Elloso, M., Levschuk, A., & Jeschke, M. G. (2025). Bioprinting-By-Design of Hydrogel-Based Biomaterials for In Situ Skin Tissue Engineering. Gels, 11(2), 110. [CrossRef]
- Raza, M. A., Jeong, J. O., & Park, S. H. (2021). State-of-the-art irradiation technology for polymeric hydrogel fabrication and application in drug release system. Frontiers in Materials, 8, 769436. [CrossRef]
- Ahmed, M. S., Islam, M., Hasan, M. K., & Nam, K. W. (2024). A Comprehensive review of radiation-induced hydrogels: Synthesis, properties, and multidimensional applications. Gels, 10(6), 381. [CrossRef]
- Lai, Y., Xiao, X., Huang, Z., Duan, H., Yang, L., Yang, Y., ... & Feng, L. (2024). Photocrosslinkable biomaterials for 3D bioprinting: mechanisms, recent advances, and future prospects. International Journal of Molecular Sciences, 25(23), 12567. [CrossRef]
- Zhang, X., Kim, G. J., Kang, M. G., Lee, J. K., Seo, J. W., Do, J. T., ... & Bae, H. (2018). Marine biomaterial-based bioinks for generating 3D printed tissue constructs. Marine drugs, 16(12), 484. [CrossRef]
- Gan, X., Li, C., Sun, J., Zhang, X., Zhou, M., Deng, Y., & Xiao, A. (2023). GelMA/κ-carrageenan double-network hydrogels with superior mechanics and biocompatibility. RSC advances, 13(3), 1558-1566. [CrossRef]
- Li, J., Moeinzadeh, S., Kim, C., Pan, C. C., Weale, G., Kim, S., ... & Yang, Y. P. (2023). Development and systematic characterization of GelMA/alginate/PEGDMA/xanthan gum hydrogel bioink system for extrusion bioprinting. Biomaterials, 293, 121969. [CrossRef]
- Branco, F., Cunha, J., Mendes, M., Sousa, J. J., & Vitorino, C. (2025). 3D Bioprinting Models for Glioblastoma: From Scaffold Design to Therapeutic Application. Advanced Materials, 37(18), 2501994. [CrossRef]
- Karvinen, J., & Kellomäki, M. (2023). Design aspects and characterization of hydrogel-based bioinks for extrusion-based bioprinting. Bioprinting, 32, e00274. [CrossRef]
- Schwab, A., Levato, R., D’este, M., Piluso, S., Eglin, D., & Malda, J. (2020). Printability and shape fidelity of bioinks in 3D bioprinting. Chemical reviews, 120(19), 11028-11055. [CrossRef]
- Amorim, P. A., d’Ávila, M. A., Anand, R., Moldenaers, P., Van Puyvelde, P., & Bloemen, V. (2021). Insights on shear rheology of inks for extrusion-based 3D bioprinting. Bioprinting, 22, e00129. [CrossRef]
- Zhao, L., Yao, D., Xia, Z., Wang, L., & Xu, M. (2025). Biological 3D-printed viscoelastic scaffolds with controllable stress relaxation rates. International Journal of Bioprinting, 11(3), 261-276. [CrossRef]
- Baniasadi, H., Polez, R. T., Kimiaei, E., Madani, Z., Rojas, O. J., Österberg, M., & Seppälä, J. (2021). 3D printing and properties of cellulose nanofibrils-reinforced quince seed mucilage bio-inks. International Journal of Biological Macromolecules, 192, 1098-1107. [CrossRef]
- Perin, F., Spessot, E., Famà, A., Bucciarelli, A., Callone, E., Mota, C., ... & Maniglio, D. (2023). Modeling a dynamic printability window on polysaccharide blend inks for extrusion bioprinting. ACS Biomaterials Science & Engineering, 9(3), 1320-1331. [CrossRef]
- Morgan, F. L., Moroni, L., & Baker, M. B. (2020). Dynamic bioinks to advance bioprinting. Advanced healthcare materials, 9(15), 1901798. [CrossRef]
- Lin, L., Jiang, S., Yang, J., Qiu, J., Jiao, X., Yue, X., ... & Zhang, L. (2022). Application of 3D-bioprinted nanocellulose and cellulose derivative-based bio-inks in bone and cartilage tissue engineering. International Journal of Bioprinting, 9(1), 637. [CrossRef]
- Malekpour, A., & Chen, X. (2022). Printability and cell viability in extrusion-based bioprinting from experimental, computational, and machine learning views. Journal of functional biomaterials, 13(2), 40. [CrossRef]
- McCauley, P. J., Fromen, C. A., & Bayles, A. V. (2025). Cell viability in extrusion bioprinting: the impact of process parameters, bioink rheology, and cell mechanics. Rheologica Acta, 1-19. [CrossRef]
- Lombardi, L., Scalzone, A., Ausilio, C., Gentile, P., & Tammaro, D. (2025). Optimizing nozzle design in extrusion-based 3D bioprinting to minimize mechanical stress and enhance cell viability. [CrossRef]
- Krishna, D. V., & Sankar, M. R. (2023). Persuasive factors on the bioink printability and cell viability in the extrusion-based 3D bioprinting for tissue regeneration applications. Engineered Regeneration, 4(4), 396-410. [CrossRef]
- Geevarghese, R., Żur-Pińska, J., Parisi, D., & Włodarczyk-Biegun, M. K. (2025). A comprehensive protocol for hydrogel-based bioink design: balancing printability, stability, and biocompatibility. Journal of Materials Chemistry B, 13(42), 13750-13768. [CrossRef]
- Boularaoui, S., Shanti, A., Khan, K. A., Iacoponi, S., Christoforou, N., & Stefanini, C. (2022). Harnessing shear stress preconditioning to improve cell viability in 3D post-printed biostructures using extrusion bioprinting. Bioprinting, 25, e00184. [CrossRef]
- Arjoca, S., Bojin, F., Neagu, M., Păunescu, A., Neagu, A., & Păunescu, V. (2024). Hydrogel extrusion speed measurements for the optimization of bioprinting parameters. Gels, 10(2), 103. [CrossRef]
- Ahmadi Soufivand, A., Faber, J., Hinrichsen, J., & Budday, S. (2023). Multilayer 3D bioprinting and complex mechanical properties of alginate-gelatin mesostructures. Scientific Reports, 13(1), 11253. [CrossRef]
- Adhikari, J., Roy, A., Das, A., Ghosh, M., Thomas, S., Sinha, A., ... & Saha, P. (2021). Effects of processing parameters of 3D bioprinting on the cellular activity of bioinks. Macromolecular bioscience, 21(1), 2000179. [CrossRef]
- Shyam, R., & Palaniappan, A. (2024). Development and optimization of starch-based biomaterial inks and the effect of infill patterns on the mechanical, physicochemical, and biological properties of 3D printed scaffolds for tissue engineering. International Journal of Biological Macromolecules, 258, 128986. [CrossRef]
- Khajehmohammadi, M., Azizi Tafti, R., & Nikukar, H. (2023). Effect of porosity on mechanical and biological properties of bioprinted scaffolds. Journal of Biomedical Materials Research Part A, 111(2), 245-260. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).