Submitted:
15 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. The Avogadro History
- Avogadro gets the Name (despite no calculation).
- Perrin gets the Nobel Prize (1926, for proving the atomic nature of matter).
- Loschmidt gets Neither, remaining a niche figure primarily known for the “Loschmidt Constant”- a value often treated as a secondary gas-physics parameter rather than a primary universal constant in standard pedagogy.
1.1. Experts Opinion on Avogadro Number and Avogadro Constant
- The Core Argument: The “amount of substance” is essentially a count of entities. Defining it through a dimensioned constant (the Avogadro constant) is seen as circular reasoning because the constant itself is derived from the unit.
- The Analogy: The mole is viewed as a scaling tool, similar to a “dozen” or “gross,” but applied specifically to elementary entities like atoms or molecules. From this perspective, the constant is a consequence of the definition, not the source of it.
- The Core Argument: In physics, a “number of entities” is dimensionless, whereas “amount of substance” has a specific unit. To maintain mathematical consistency in science (dimensional analysis), a bridge is required to convert counts into amounts.
- The Function of the Constant: The Avogadro constant is seen as a fundamental proportionality factor. Without it, the dimensional integrity of chemistry, such as the units for molar mass or the gas constant-would collapse into simple ratios, losing their physical meaning.
- The Core Argument: The constant is defined as the reciprocal of the “elementary amount” (the amount of substance contained in exactly one atom).
- The Comparison: Just as a “second” is defined by a fixed number of periods of radiation, a mole is defined by a fixed number of these “elementary amounts.” By establishing the atom itself as the base reference, this view argues that the definition is not circular, but a logical progression from the microscopic to the macroscopic.
1.2. The Silicon Sphere Experiment – The kg Tension
1.3. Objectives of This Paper
- 1)
- Demonstrate nuclear binding origin of atomic mass unit via QCD saturation (~8 MeV/nucleon peak) and SEMF analysis.
- 2)
- Demonstrate Avogadro number (~6.02×1026 atoms/kg) as inverse of atomic mass unit from computational nuclear data.
- 3)
- Validate kg-scale Faraday constant (F ≈ 9.6435×10^7 C/kg), and to show that, 1/F ≅ Mpl*sin θW bridge to Electro-Chemistry and Quantum Gravity.
- 4)
- Derive Dulong-Petit specific heat (~25/A kJ/kg·K) as independent thermodynamic origin of ‘kg’ Avogadro scale.
- 5)
- Presenting 4G micro-macro gravity ratio [(Gweak*Gnuclear)/(Gelectromag*GNewton)] ≅ 6.1088×1023 as universal scale predictor.
- 6)
- To rename ‘Avogadro constant’ as “Einstein-Perrin-Loschmidt-Avogadro-Newton” Ratio. (EPLAN Ratio)
2. Nuclear Binding Energy Formula with Single Set of Energy Coefficients for Z=2 to 118
- 1)
- Global Accuracy: Fixed coefficients achieve <1% errors across chart of nuclides (light to superheavy), reproducing iron peak (BE/A~8.7 MeV at ⁵⁶Fe), magic shells (Z/N=2,8,20,28,50,82), and driplines.
- 2)
- Physical Interpretability: Each term maps directly to nuclear physics (volume saturation, surface tension, proton repulsion, isospin imbalance, quantum pairing, neutron-skin damping), enabling stability predictions (fission barriers, β-decay Q-values).
- 3)
- Light Nuclei Robustness: [1-(1/A)] correction prevent divergences at low A; exponential damping handles extreme N/Z ratios better than standard asymmetry.
- 4)
- Computational Simplicity: Single set works for all Z- no piecewise tuning required; vectorizable for rapid A-loops (2Z-3.5Z), enabling batch processing Z=1-140 with CSV/PNG outputs.
- 5)
- Predictive Power: Estimates unmeasured masses, fusion yields, and Avogadro implications (your extension); competitive with AME2020 least-squares fits (~0.6 MeV RMS).
- 6)
- Easy Fine tuning: With machine learning techniques and AI, above formula can be fine-tuned for a better accuracy [27].
2.1. Unified Atomic Energy Unit (UAEU) and The Avogadro Number
2.2. The Inverse of Binding-Limited Atomic Mass
- The Limit: Every atom is subject to a “mass defect”, where a portion of the constituent nucleons’ mass is converted into binding energy to hold the nucleons together.
- The emergence: There is a theoretical threshold where the efficiency of this binding reaches its peak (often associated with the iron/nickel peak on the binding energy curve). The Avogadro constant emerges as the natural scaling factor dictated by this maximum binding efficiency, effectively representing the point where subatomic binding transitions into macroscopic stability.
- Saturation Point: The strong nuclear force, which governs the interaction between quarks and gluons, has a limited range. As more nucleons are added to a system, the “density” of the strong force interactions reaches a saturation point where the nucleus cannot become any denser or more tightly bound.
- The Particle/Mass Ratio: This saturation creates a fixed ratio between the number of particles (elementary entities) and the total mass they can occupy. This ratio is what we measure as the Avogadro constant. If the strong force were slightly stronger or weaker, the saturation point would shift, and the “Avogadro number” required to make a “gram” or “kilogram” of matter would be different.
- Bottom-Up Reality: Instead of humans deciding that 6.022x1023 is the definition, this insight proposes that the universe enforces this number through the equilibrium of forces.
- Structural Necessity: The constant acts as the structural link between the quantum world (governed by QCD and binding energy) and the thermodynamic world (governed by bulk matter).
2.3. Variation of Avogadro Number for Z=2 to 118
3. 4G Micro-Macro Gravity Unification Scale
3.1. Three Assumptions and Two Applications of Our 4G Model of Final Unification
- 1)
- There exists a characteristic electroweak fermion of rest energy, . It can be considered as the zygote of all elementary particles.
- 2)
- There exists a nuclear elementary charge in such a way that, = Strong coupling constant and .
- 3)
- Each atomic interaction is associated with a characteristic large gravitational coupling constant. Their fitted magnitudes are,
- 1)
- Recent high-precision astrophysical observations lend growing support to our first assumption of a characteristic electroweak fermion with rest energy near 585 GeV. In particular, the sharp spectral break at 1.17 TeV in the all-electron cosmic-ray spectrum reported by H.E.S.S., and independently confirmed by DAMPE and CALET, coincides precisely with twice the proposed fermion mass, suggesting the presence of bound or resonant fermion–antifermion states. This correspondence is further reinforced by Galactic gamma-ray excess studies, which infer neutral particles in the 500–800 GeV range, consistent with the neutral component of our 4G fermion doublet. Together, these converging astrophysical signatures provide empirical motivation for the 585 GeV fermion hypothesis, strengthening its role as a unifying microscopic origin for both nuclear phenomenology and TeV-scale cosmic-ray features [23,24,25].
- 2)
- In the 4G model, the strong coupling constant [31] acquires a simple, physically transparent definition: , where is the fundamental electromagnetic charge and is the nuclear elementary charge. This relation reveals that strong interaction strength arises directly from the ratio of these fundamental charges, eliminating arbitrary empirical parameters. With nearly three times , the formula naturally yields 2, matching low-energy experimental values (–) and elegantly unifying electromagnetic and nuclear forces. In the context of the 4G model of nuclear charge, if one assigns a nuclear elementary charge of 3e to quarks, then the electromagnetic charges of the quark families can be expressed in a simple and unified manner. Specifically, the up-series quarks (u, c, t) carry an effective electromagnetic charge of 2e, while the down-series quarks (d, s, b) carry an effective charge of e. This formulation, provides a charge-based reinterpretation of quark structure [32]. It highlights how quark charges may be understood as scaled fractions of a fundamental nuclear charge, offering a natural bridge between electromagnetic and nuclear interactions within the 4G framework. The universal nuclear energy scale is set by Important point to be noted is that, the strong attraction between protons is about times stronger than the repulsive Coulomb energy, ensuring nuclear stability. Coming to the Bohr radius of Hydrogen atom, it is very interesting to note that, where
- 3)
- In our 4G framework, the necessity of large gravitational couplings arises from the fundamental requirement that point particles must sustain non-trivial spacetime curvature at quantum scales. If gravity were as weak as the classical Newtonian constant, the immense energy density of point-like particles would fail to generate meaningful curvature, undermining the geometric foundation of quantum structure. By assigning enhanced gravitational constants to the strong, electromagnetic, and weak interactions, curvature is preserved at the femtometer–picometer domain. Moreover, as particle mass increases, the effective gravitational influence decreases with the square of the mass, ensuring that heavier particles and nuclei do not collapse under excessive curvature. This dual principle-that high gravity is essential for point particles, yet naturally weakens with increasing mass-provides a coherent explanation for the observed hierarchy of forces and the emergence of atomic radii consistent with experimental bond lengths.
- 4)
-
In a unified approach, most important point to be noted is that,Clearly speaking, based on the electroweak interaction, the well believed quantum constant seems to have a deep inner meaning. Following this kind of relation, there is a possibility to understand the integral nature of quantum mechanics with a relation of the form,
- 5)
- Weak interaction point of view [35], following our assumptions, Fermi’s weak coupling constant can be fitted with the following relations.
- 6)
-
Our theoretical GN = 6.679851×10⁻¹¹ from unified gravitational constants agrees with the latest precision measurements: Brack et al. [6.67559(27)×10⁻¹¹] and Tobias et al. [6.682(17)×10⁻¹¹].
3.2. Dimensionless Hierarchy Ratio
4. Discussion
4.1. Theoretical Superiority
4.2. Practical Stability
- Measurement Consistency: The numerical value of the Avogadro constant was fixed based on the most precise measurements available at the time of the 2019 revision. Therefore, results obtained before and after the change remain identical in practice.
- Instrumentation: Molar masses and gas constant values used in software and laboratory equipment do not require recalibration. The precision of the “fixed” value exceeds the requirements of almost all practical applications, providing a stable platform for global industry and research.
Nomenclature as a Conceptual Foundation
- Defining the Entity: By distinguishing between the Avogadro number (the fixed value) and the Avogadro constant (the value with units), the SI brochure provides a more rigorous logical framework.
- Eliminating Ambiguity: The current nomenclature clarifies that the mole is not a measure of mass, but a measure of the number of entities. This prevents the conceptual “drift” where the mole was historically tied to the mass of carbon-12.
- For a laboratory technician, the mole remains a tool for stoichiometry.
- For the metrologist, the nomenclature ensures that the ‘International System of Units’ is logically sound and ‘future-proof’.
4.3. Testable Predictions
- The Test: As laboratories synthesize increasingly heavy or neutron-rich isotopes, their measured binding energies must align with the predicted curves.
- The Significance: If isotopic stability and mass defects fluctuate according to the model’s specific geometric or coupling requirements, it confirms that the Avogadro constant is fundamentally linked to nuclear structure rather than being an arbitrary scaling factor.
- The Test: Utilizing Lattice QCD, a computational approach that simulates quark and gluon interactions on a space-time grid, researchers can calculate the ratio of the strong coupling saturation to the gravitational/electromagnetic hierarchy.
- The Significance: If these high-precision simulations independently yield the value of 6.022x1023, it provides mathematical proof that the constant is a “built-in” feature of the Standard Model’s scaling laws.
- The Test: By calculating how gravitational and subatomic force couplings evolve from laboratory energy scales up to the Planck scale 1019 GeV, the model predicts a convergence point matching the value 6.1088x1023.
- The Significance: Matching this specific value 6.1088x1023 through force unification formulas would link the Avogadro number directly to the 4G Model of Final Unification. This would elevate the constant from a chemistry “counting tool” to a fundamental “coupling ratio” of the universe itself.
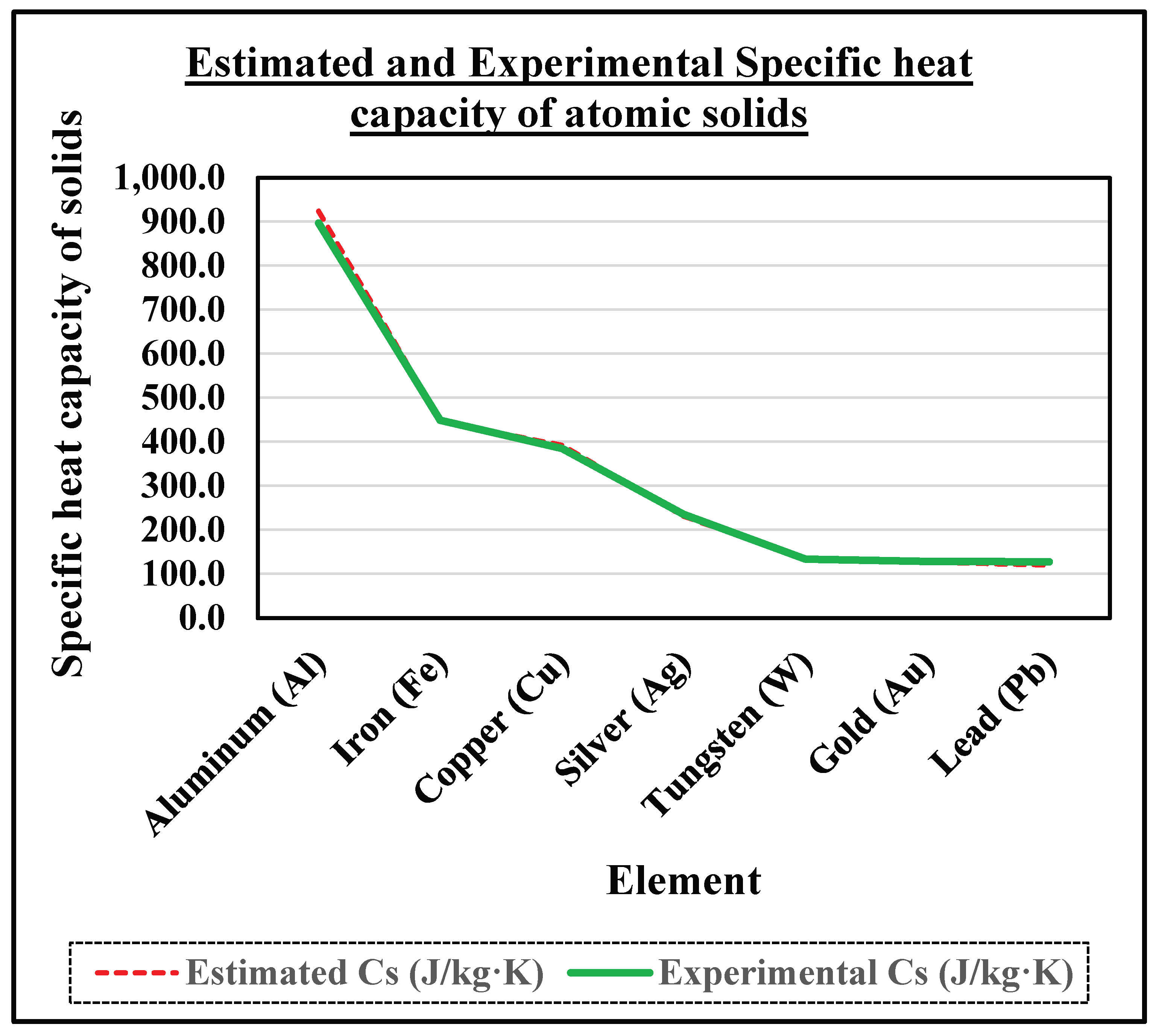
4.4. Specific Heat Confirmation via Kg-Scale Avogadro:
- 1)
- Molar Heat Capacity: Defined as the heat required to raise one mole of a substance by 1 K, with units typically given as J/mol·K.
- 2)
- Specific Heat Capacity: Defined as the heat required to raise one kilogram of a substance by 1 K, with units J/kg·K.
- 3)
- The Dulong–Petit law [42] approximates the molar heat capacity of many solids as about 3RU, where RU is the universal gas constant (~8.314 J/mol·K). Numerically, this is approximately 24.94 J/mol·K.
- 5)
- To convert molar heat capacity Cm to specific heat capacity C, the molar mass of the substance plays a crucial role:

4.5. Faraday Charge Confirmation via Kg-Scale Avogadro
4.6. Faraday Constant as a Low-Energy Window into Planck-Scale Physics
5. Conclusions
Data availability statement
Conflicts of Interest
Acknowledgments
Appendix A. Python Code for the Estimated Nuclear Binding Energy and Avogadro Number
| import os import csv import math import gc from datetime import datetime import numpy as np import matplotlib.pyplot as plt import matplotlib as mpl from matplotlib.patches import Rectangle graph_header = [ "Z", "N", "A", "A-2Z", "<Estimated Stable A>", "r1", "r2", "r3", "r4", "r5", "r6", "r7", "EBEPN (MeV)" ] avogadro_header = [ "Z", "A_Low", "A_Upper", "Stable Mass Number", "No. of Isotopes", "EABEPN (MeV)", "EUAEU (MeV)", "EUAMU (kg)", "EAN (No. of atoms/kg)" ] def ensure_folder(folder_path): if not os.path.exists(folder_path): os.makedirs(folder_path) def process_Z(z, folder_path, all_diff_be_values): ebe = [] xl = [] tl = [] graph_fn = os.path.join(folder_path, f"{z}_GraphData.csv") with open(graph_fn, 'w', newline='') as fgraph: writer = csv.writer(fgraph) writer.writerow(graph_header) sebepn = 0 al = round(2.0 * z) - 1 if z == 1: al = 2 au = round(3.5 * z) sm = round((2 * z) + (0.00642 * z * z), 0) rc = au - al + 1 smc = int(sm) for a in range(al, au + 1): n = a - z i = float(((n - z) / a)) beta = 1.0 - pow(i, 2) # REQUIRED: EXACT REFERENCE BLOCK RETAINED r1 = 16.0 * a r2 = beta * a ** (2 / 3) * 19.4 x2 = 0.75 - (0.5 * z / a) r3 = 0.71 * z ** 2 / (a ** (1 / 3) * beta ** x2) r4 = (1 - 1 / a) * 24.5 * (n - z) ** 2 / a r5 = 0.5 * ((-1) ** z + (-1) ** n) * 10.0 / (a ** 0.5) r6 = 10.0 * math.exp(-4.2 * abs(n - z) / a) r7 = r1 - r2 - r3 - r4 + r5 + r6 ebe.append(r7) ebepn = r7 / a sebepn += ebepn row = [ z, n, a, n - z, smc, round(r1, 2), round(r2, 2), round(r3, 2), round(r4, 2), round(r5, 4), round(r6, 2), round(r7, 2), round(ebepn, 2), ] for i in range(11, len(row)): row[i] = "{:.5E}".format(float(row[i])) writer.writerow(row) # Tick marks tg = int(round(0.5 * pow(z, 0.5), 0)) if z == 1: tg = 1 if (len(ebe) - 1) % tg == 0: tl.append(len(ebe) - 1) xl.append(a) aebepn = sebepn / rc # SIMPLIFIED AVOGADRO LOGIC (using only reference values) euaeu = ((939.56563 + 938.27231) / 2.0 - aebepn + 0.5109906) - ( (1.44381e-5 * z ** 1.39) + (1.55468e-12 * z ** 4.35)) euamu = 1.602176565e-13 * euaeu / pow(2.99792458e8, 2) ean = 1 / euamu # PLOT REFERENCE CURVE ONLY crbe = np.array(ebe, dtype=np.float64) cxl = np.array(xl, dtype=np.int64) ctl = np.array(tl, dtype=np.int64) plt.figure(figsize=(8, 7)) plt.plot(crbe, ls='-', color='g', label='Single set of energy coefficients for Z=1 to 118') xmin, xmax = 0, len(crbe) - 1 ymin = np.min(crbe) ymax = np.max(crbe) ymargin = 0.05 * (ymax - ymin) ymin -= ymargin ymax += ymargin plt.xlim(xmin, xmax) plt.ylim(ymin, ymax) mpl.style.use('ggplot') mpl.rc('text', color='blue') plt.title(f"Estimated Nuclear Binding Energy of isotopes of Z={z}", size=14, color='red', weight='bold', fontweight='bold') plt.xlabel("Mass number (A)", fontweight='bold') plt.ylabel("Binding Energy of Isotopes (MeV)", fontweight='bold') plt.grid() plt.xticks(ctl, cxl, rotation=90) ax = plt.gca() for label in ax.get_xticklabels(): label.set_fontweight('bold') for label in ax.get_yticklabels(): label.set_fontweight('bold') plt.legend(loc='best') fig = plt.gcf() plt.subplots_adjust(left=0.18, right=0.92, top=0.94, bottom=0.17) outer = Rectangle((0, 0), 1, 1, transform=fig.transFigure, fill=False, edgecolor='blue', linewidth=6, zorder=10000) fig.add_artist(outer) x0, x1 = ax.get_xlim() y0, y1 = ax.get_ylim() inner = Rectangle((x0, y0), x1 - x0, y1 - y0, linewidth=3, edgecolor='black', facecolor='none', zorder=1000) ax.add_patch(inner) fn = os.path.join(folder_path, f"Z={z}_Graph_RBE_{datetime.now().strftime('%Y-%m-%d_%H-%M-%S')}.pdf") plt.savefig(fn) plt.clf() plt.cla() plt.close() gc.collect() print(f"Processed Z={z}, plot saved as {fn}") return [z, al, au, smc, rc, aebepn, euaeu, euamu, ean] def write_avogadro_csv(avogadro_data, all_diff_be_values, folder_path): avocsv = os.path.join(folder_path, "Data_Estimated_Avogadro.csv") with open(avocsv, 'w', newline='') as f: writer = csv.writer(f) writer.writerow(avogadro_header) for row in avogadro_data: outrow = [row[0], row[1], row[2], row[3], row[4], "{:.5E}".format(row[5])] outrow += ["{:.5E}".format(v) for v in row[6:]] writer.writerow(outrow) print(f"Estimated Avogadro CSV saved: {avocsv}") def plot_avogadro_graph(avogadro_data, folder_path): zs = [row[0] for row in avogadro_data] ran_vals = [row[[-1] for row in avogadro_data] plt.figure(figsize=(10, 6)) plt.plot(zs, ran_vals, 'go-', label='Estimated Avogadro Number (EAN)') plt.title("Estimated Avogadro Number vs Z", fontweight='bold') plt.xlabel("Atomic Number (Z)", fontweight='bold') plt.ylabel("Avogadro Number (No. of atoms/kg)", fontweight='bold') plt.legend() plt.grid() ax = plt.gca() for label in ax.get_xticklabels(): label.set_fontweight('bold') for label in ax.get_yticklabels(): label.set_fontweight('bold') fig = plt.gcf() x0, x1 = ax.get_xlim() y0, y1 = ax.get_ylim() inner_rect = Rectangle((x0, y0), x1 - x0, y1 - y0, linewidth=2, edgecolor='black', facecolor='none', zorder=1000) ax.add_patch(inner_rect) external_rect = Rectangle((0, 0), 1, 1, transform=fig.transFigure, fill=False, edgecolor='magenta', linewidth=3, zorder=10000) fig.add_artist(external_rect) plt.tight_layout() fn = os.path.join(folder_path, f"Graph_Estimated_Avogadro_{datetime.now().strftime('%Y-%m-%d_%H-%M-%S')}.pdf") plt.savefig(fn) plt.clf() plt.cla() plt.close() print(f"Reference Avogadro graph saved as {fn}") def main_loop(): base_folder = "D:/Nuclear_Binding_Energy_Results" zl = int(input("Enter Z lower value (>=1): ")) zi = int(input("Enter Z increment value (>=1): ")) zu = int(input("Enter Z upper value (<=140): ")) timestamp = datetime.now().strftime("%Y-%m-%d_%H-%M-%S") folder_name = f"{zl}_{zu}_{timestamp}" folder_path = os.path.join(base_folder, folder_name) ensure_folder(folder_path) avogadro_data = [] all_diff_be_values = [] for z in range(zl, zu + 1, zi): summary = process_Z(z, folder_path, all_diff_be_values) avogadro_data.append(summary) write_avogadro_csv(avogadro_data, all_diff_be_values, folder_path) plot_avogadro_graph(avogadro_data, folder_path) print(f"All data files saved in: {folder_path}") if __name__ == "__main__": main_loop() |
References
- SI Brochure. The International System of Units (SI) (Bureau International des Poids et Mesures (BIPM), 2019.
- Taylor, B.N. Quantity calculus, fundamental constants, and SI units. J. Res. NIST 2018, 123 123008. [Google Scholar] [CrossRef]
- Mohr, P.J.; Newell, D.B.; Taylor, B.N.; Tiesinga, E. Data and analysis for the CODATA 2017 special fundamental constants adjustment. Metrologia 2018, 55, 125–146. [Google Scholar] [CrossRef]
- Tiesinga, E; Mohr, P.J; Newell, D.B.; Taylor, B.N. CODATA recommended values of the fundamental physical constants: 2018. Rev Mod Phys. 2021, 93, 025010. [Google Scholar] [CrossRef]
- Jean Perrin, M. Brownian Movement and Molecular Reality. Nature 1911, 86, 105. [Google Scholar] [CrossRef]
- Millikan, R. A. A new determination of e, N, and related constants. Phil. Mag. 1917, 34, 1–30. [Google Scholar] [CrossRef]
- Schuster, Peter. From Curiosity to Passion: Loschmidt’s Route from Philosophy to Natural Science (PDF), 1st ed.; Springer US: Boston, MA, 1997; pp. 269–276. [Google Scholar]
- Leonard, B. P. Note on invariant redefinitions of SI base units for both mass and amount of substance. Metrologia 2006, 43, L3–L5. [Google Scholar] [CrossRef]
- Leonard, B P. The atomic-scale unit, entity: key to a direct and easily understood definition of the SI base unit for amount of substance. Metrologia 2007, 44, 402–406. [Google Scholar] [CrossRef]
- Milton, M.J.T. A new definition for the mole based on the Avogadro constant: a journey from physics to chemistry. Philosoph. Trans. Royal Socie. A 2011, 369, 3993–4003. [Google Scholar] [CrossRef] [PubMed]
- Güttler, B.; Bettin, H.; Brown, R.J.C.; Davis, R.S.; Mester, Z. Amount of substance and the mole in the SI. Metrologia 2019, 56, 044002. [Google Scholar] [CrossRef]
- Brown, R J C; Brewer, Paul J. What is a mole? Metrologia 2020, 57, 065002. [Google Scholar] [CrossRef]
- Brown, R. J. C. On the distinction between SI base units and SI derived units Metrologia. 2024, 61, 013001. [Google Scholar]
- Brown, Richard J C. Comment on ‘The Avogadro constant is not the defining constant of the mole’. Metrologia 2025, 62, 058004. [Google Scholar]
- Kacker, Raghu N.; Irikura, Karl K. The SI unit mole and Avogadro constant. Measurement: Sensors 2025, 38, 101767. [Google Scholar] [CrossRef]
- Seshavatharam, U.V.S; Lakshminarayana, S. Computing unified atomic mass unit and Avogadro number with various nuclear binding energy formulae coded in Python. Int. J. Chem. Stud. 2025, 13(1), 24–30. [Google Scholar] [CrossRef]
- Seshavatharam, U.V.S; Gunavardhana, T. N.; Lakshminarayana, S. Avogadro’s Number: History, Scientific Role, State-of-the-Art, and Frontier Computational Perspectives. Curr. Trends. Mass. Comm 2025, 4(3), 01–10, Preprints2025, 2025080338. [Google Scholar] [CrossRef]
- Fujii, K.; et al. Present State of the Avogadro Constant Determination from Silicon Crystals with Natural Isotopic Compositions. IEEE Transactions on Instrumentations Measurements 2005, 54(2), 854–859. [Google Scholar] [CrossRef]
- Becker, P. Determination of the Avogadro constant – A contribution to the new definition of the mass unit kilogram. Eur. Phys. J. Special Topics 2009, 172(1), 343–352. [Google Scholar] [CrossRef]
- Luis Márquez-Jaime, Guillaume Gondre and Sergio Márquez-Gamiño. The first and a recent experimental determination of Avogadro's number. REVISTA ENLACE QUÍMICO, UNIVERSIDAD DE GUANAJUATO. 2(8), 2009.
- Becker, P; Bettin, H. The Avogadro constant: determining the number of atoms in a single-crystal 28Si sphere. Phil Trans R Soc A. 2011, 369, 3925–3935. [Google Scholar] [CrossRef]
- Seshavatharam, U. V. S.; Lakshminarayana, S. A Unified 6-Term Formula for Nuclear Binding Energy with a Single Set of Energy Coefficients for Z = 1–140. International Journal of Advance Research and Innovative Ideas in Education 2025, 2(6), 1716–1731. [Google Scholar]
- Seshavatharam U.V.S, Gunavardhana Naidu T and Lakshminarayana S. 4G Model of Heavy Electroweak Charged 585 GeV Fermions as the Supposed Microscopic Origin of the 1.17 TeV All-Electron Spectral Break. International Journal of Advance Research and Innovative Ideas in Education. 11(6), 2116-2140, 2025.
- Seshavatharam, U. V. S; Gunavardhana Naidu, T; Lakshminarayana, S. Nuclear evidences for confirming the physical existence of 585 GeV weak fermion and galactic observations of TeV radiation. International Journal of Advanced Astronomy 2025, 13(1), 1–17. [Google Scholar]
- Seshavatharam, U.V.S; Gunavardhana, T. N.; Lakshminarayana, S. Advancing String Theory with 4G Model of Final Unification. J. Phys.: Theor. Appl. 2025, 9(2), 158–197. [Google Scholar]
- Seshavatharam, U.V.S; Gunavardhana, T. N.; Lakshminarayana, S. Quarks-Higgs Resonances in the 4G Model of Final Unification: Precision Mass Predictions and Observational Targets. Zenodo 2026. [Google Scholar] [CrossRef]
- Gao, ZP; Wang, YJ; Lü, HL; et al. Machine learning the nuclear mass. Nucl Sci Tech. 2021, 32, 109. [Google Scholar] [CrossRef]
- Royer, G. On the coefficients of the liquid drop model mass formulae and nuclear radii. Nuclear Physics A 2008, 807(3–4), 105–118. [Google Scholar] [CrossRef]
- Nordén, B. The Mole, Avogadro’s Number and Albert Einstein. Mol Front J. 2021, 5, 66–78. [Google Scholar] [CrossRef]
- Siafarikas, M; Stylos, G; Chatzimitakos, T; Georgopoulos, K; Kosmidis, C; Kotsis, KT. Experimental teaching of the Avogadro constant. Phys Educ. 2023, 58, 065026. [Google Scholar] [CrossRef]
- d’Enterria, D; et al. The strong coupling constant: state of the art and the decade ahead. J. Phys. G: Nucl. Part. Phys. 2024, 51 090501. [Google Scholar] [CrossRef]
- Seshavatharam, U.V.S.; Lakshminarayana, S. Understanding the Origins of Quark Charges, Quantum of Magnetic Flux, Planck’s Radiation Constant and Celestial Magnetic Moments with the 4G Model of Nuclear Charge. Current Physics 2024, 1(e090524229812), 122–147. [Google Scholar] [CrossRef]
- Patel, Apoorva D. EPR Paradox, Bell Inequalities and Peculiarities of Quantum Correlations. arXiv 2025, arXiv:2502.06791v1. [Google Scholar] [CrossRef]
- Clifford Cheung, Aaron Hillman, Grant N. Remmen. String Theory May Be Inevitable as a Unified Theory of Physics. Physics World, 2025.
- Abokhalil, Ahmed. The Higgs Mechanism and Higgs Boson: Unveiling the Symmetry of the Universe. arXiv arXiv:2306.01019v2 [hep-ph]. [CrossRef]
- Brack, T.; Zybach, B.; Balabdaoui, F.; et al. Dynamic measurement of gravitational coupling between resonating beams in the hertz regime. Nat. Phys. 2022, 18, 952–957. [Google Scholar] [CrossRef]
- Tobias, B.; Jonas, F.; Bernhard, Z.; et al. Dynamic gravitational excitation of structural resonances in the hertz regime using two rotating bars. Commun Phys. 2023, 6, 270. [Google Scholar] [CrossRef]
- Schiller, Christoph. From Maximum Force Via the Hoop Conjecture to Inverse Square Gravity. Gravit. Cosmol. 2022, 28, 305–307. [Google Scholar] [CrossRef]
- Schiller, Christoph. Tests for maximum force and maximum power. Phys. Rev. D 2021, 104, 124079. [Google Scholar] [CrossRef]
- Seshavatharam U. V. S, Gunavardhana Naidu T and Lakshminarayana S. Empirical formula for specific heat of solids based on atomic constants and a universal subzero limiting temperature. EPJ Web Conf. Volume 345, Article Number 01029, 2026. 4th International Conference & Exposition on Materials, Manufacturing and Modelling Techniques (ICE3MT2025).
- Gusev, Y.V. Experimental verification of the field theory of specific heat with the scaling in crystalline matter. Sci Rep 2021, 11, 18155. [Google Scholar] [CrossRef]
- Piazza, Roberto. The strange case of Dr. Petit and Mr. Dulong. 2018, arXiv:1807.02270v1 [physics.hist-ph]. [Google Scholar]
- Resta, Raffaele. Faraday law, oxidation numbers, and ionic conductivity: The role of topology. arXiv 2021, arXiv:2104.06026v2. [Google Scholar] [CrossRef]
- Kenneth Barbalace. Periodic Table of Elements. EnvironmentalChemistry.com. 1995 - 2024. (Complied references there in).
- Seshavatharam, U. V. S.; Lakshminarayana, S. Inferring and confirming the rest mass of electron neutrino with neutron life time and strong coupling constant via 4G model of final unification. World Scientific News 2024, 191, 127–156. [Google Scholar]
- The LHCb collaboration; Aaij, R.; Adeva, B.; et al. Measurement of the forward-backward asymmetry in Z/γ∗ → μ + μ − decays and determination of the effective weak mixing angle. J. High Energ. Phys. 2015, 190. [Google Scholar] [CrossRef]
- “Weak mixing angle”. The NIST reference on constants, units, and uncertainty. 2022 CODATA value. National Institute of Standards and Technology.
- Erler, Jens; Ferro-Hernández, Rodolfo; Kuberski, Simon. Theory-Driven Evolution of the Weak Mixing Angle. Phys. Rev. Lett. 2024, 133, 171801. [Google Scholar] [CrossRef] [PubMed]
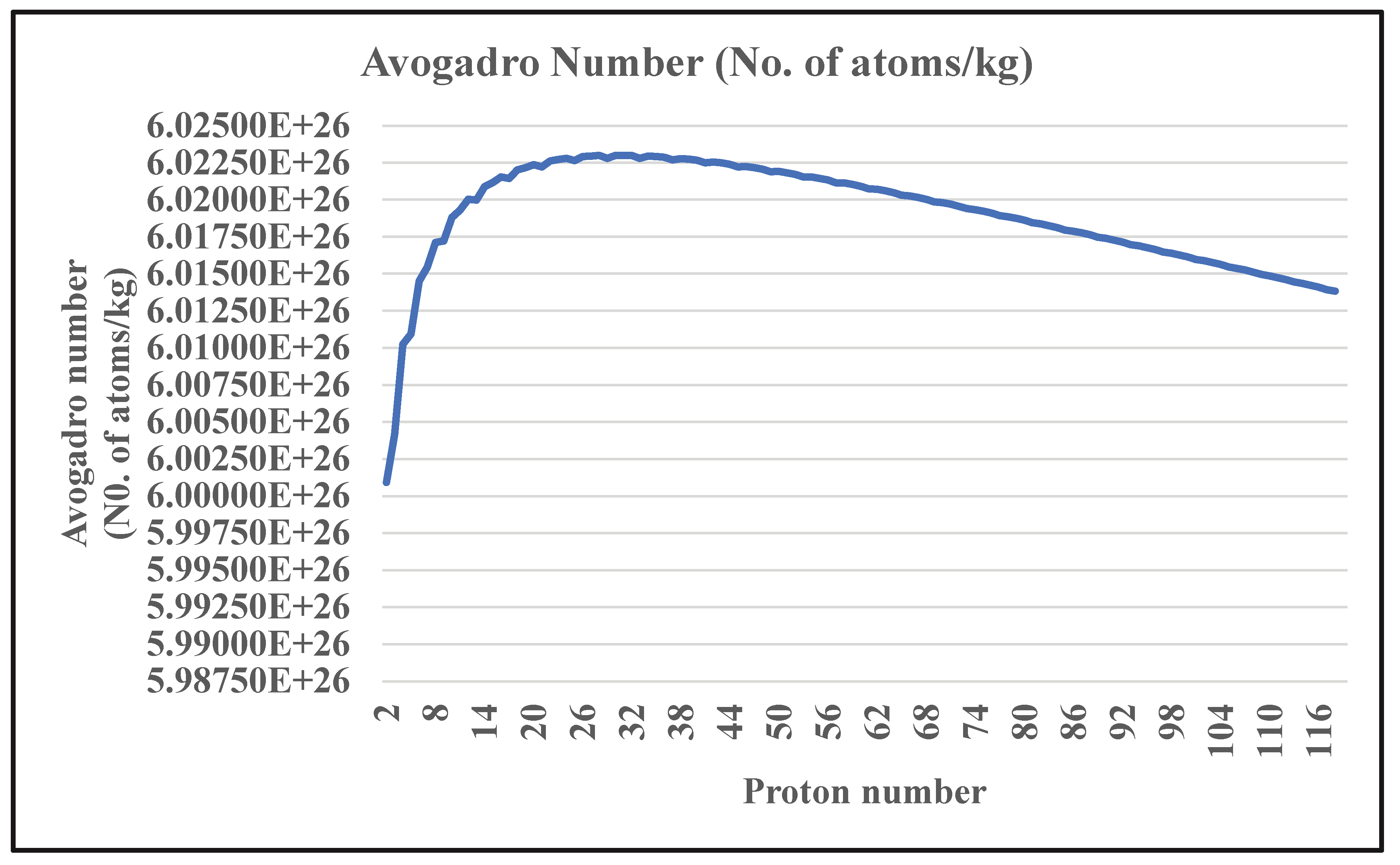
| Z | Lower mass number | Upper mass number | No. of Isotopes | Avogadro number (No. of atoms/kg) |
|---|---|---|---|---|
| 2 | 3 | 7 | 5 | 6.00092E+26 |
| 3 | 5 | 10 | 6 | 6.00417E+26 |
| 4 | 7 | 14 | 8 | 6.01024E+26 |
| 5 | 9 | 18 | 10 | 6.01093E+26 |
| 6 | 11 | 21 | 11 | 6.01450E+26 |
| 7 | 13 | 24 | 12 | 6.01547E+26 |
| 8 | 15 | 28 | 14 | 6.01711E+26 |
| 9 | 17 | 32 | 16 | 6.01721E+26 |
| 10 | 19 | 35 | 17 | 6.01881E+26 |
| 11 | 21 | 38 | 18 | 6.01930E+26 |
| 12 | 23 | 42 | 20 | 6.02003E+26 |
| 13 | 25 | 46 | 22 | 6.01998E+26 |
| 14 | 27 | 49 | 23 | 6.02090E+26 |
| 15 | 29 | 52 | 24 | 6.02117E+26 |
| 16 | 31 | 56 | 26 | 6.02155E+26 |
| 17 | 33 | 60 | 28 | 6.02143E+26 |
| 18 | 35 | 63 | 29 | 6.02203E+26 |
| 19 | 37 | 66 | 30 | 6.02218E+26 |
| 20 | 39 | 70 | 32 | 6.02238E+26 |
| 21 | 41 | 74 | 34 | 6.02223E+26 |
| 22 | 43 | 77 | 35 | 6.02263E+26 |
| 23 | 45 | 80 | 36 | 6.02272E+26 |
| 24 | 47 | 84 | 38 | 6.02281E+26 |
| 25 | 49 | 88 | 40 | 6.02264E+26 |
| 26 | 51 | 91 | 41 | 6.02292E+26 |
| 27 | 53 | 94 | 42 | 6.02296E+26 |
| 28 | 55 | 98 | 44 | 6.02299E+26 |
| 29 | 57 | 102 | 46 | 6.02281E+26 |
| 30 | 59 | 105 | 47 | 6.02301E+26 |
| 31 | 61 | 108 | 48 | 6.02301E+26 |
| 32 | 63 | 112 | 50 | 6.02299E+26 |
| 33 | 65 | 116 | 52 | 6.02281E+26 |
| 34 | 67 | 119 | 53 | 6.02295E+26 |
| 35 | 69 | 122 | 54 | 6.02292E+26 |
| 36 | 71 | 126 | 56 | 6.02288E+26 |
| 37 | 73 | 130 | 58 | 6.02269E+26 |
| 38 | 75 | 133 | 59 | 6.02278E+26 |
| 39 | 77 | 136 | 60 | 6.02274E+26 |
| 40 | 79 | 140 | 62 | 6.02267E+26 |
| 41 | 81 | 144 | 64 | 6.02249E+26 |
| 42 | 83 | 147 | 65 | 6.02255E+26 |
| 43 | 85 | 150 | 66 | 6.02249E+26 |
| 44 | 87 | 154 | 68 | 6.02240E+26 |
| 45 | 89 | 158 | 70 | 6.02222E+26 |
| 46 | 91 | 161 | 71 | 6.02225E+26 |
| 47 | 93 | 164 | 72 | 6.02218E+26 |
| 48 | 95 | 168 | 74 | 6.02208E+26 |
| 49 | 97 | 172 | 76 | 6.02190E+26 |
| 50 | 99 | 175 | 77 | 6.02191E+26 |
| 51 | 101 | 178 | 78 | 6.02183E+26 |
| 52 | 103 | 182 | 80 | 6.02173E+26 |
| 53 | 105 | 186 | 82 | 6.02154E+26 |
| 54 | 107 | 189 | 83 | 6.02153E+26 |
| 55 | 109 | 192 | 84 | 6.02145E+26 |
| 56 | 111 | 196 | 86 | 6.02134E+26 |
| 57 | 113 | 200 | 88 | 6.02115E+26 |
| 58 | 115 | 203 | 89 | 6.02113E+26 |
| 59 | 117 | 206 | 90 | 6.02104E+26 |
| 60 | 119 | 210 | 92 | 6.02092E+26 |
| 61 | 121 | 214 | 94 | 6.02074E+26 |
| 62 | 123 | 217 | 95 | 6.02071E+26 |
| 63 | 125 | 220 | 96 | 6.02061E+26 |
| 64 | 127 | 224 | 98 | 6.02049E+26 |
| 65 | 129 | 228 | 100 | 6.02031E+26 |
| 66 | 131 | 231 | 101 | 6.02026E+26 |
| 67 | 133 | 234 | 102 | 6.02016E+26 |
| 68 | 135 | 238 | 104 | 6.02004E+26 |
| 69 | 137 | 242 | 106 | 6.01986E+26 |
| 70 | 139 | 245 | 107 | 6.01981E+26 |
| 71 | 141 | 248 | 108 | 6.01970E+26 |
| 72 | 143 | 252 | 110 | 6.01957E+26 |
| 73 | 145 | 256 | 112 | 6.01940E+26 |
| 74 | 147 | 259 | 113 | 6.01934E+26 |
| 75 | 149 | 262 | 114 | 6.01923E+26 |
| 76 | 151 | 266 | 116 | 6.01910E+26 |
| 77 | 153 | 270 | 118 | 6.01893E+26 |
| 78 | 155 | 273 | 119 | 6.01886E+26 |
| 79 | 157 | 276 | 120 | 6.01875E+26 |
| 80 | 159 | 280 | 122 | 6.01862E+26 |
| 81 | 161 | 284 | 124 | 6.01845E+26 |
| 82 | 163 | 287 | 125 | 6.01838E+26 |
| 83 | 165 | 290 | 126 | 6.01826E+26 |
| 84 | 167 | 294 | 128 | 6.01813E+26 |
| 85 | 169 | 298 | 130 | 6.01796E+26 |
| 86 | 171 | 301 | 131 | 6.01788E+26 |
| 87 | 173 | 304 | 132 | 6.01777E+26 |
| 88 | 175 | 308 | 134 | 6.01764E+26 |
| 89 | 177 | 312 | 136 | 6.01747E+26 |
| 90 | 179 | 315 | 137 | 6.01739E+26 |
| 91 | 181 | 318 | 138 | 6.01727E+26 |
| 92 | 183 | 322 | 140 | 6.01714E+26 |
| 93 | 185 | 326 | 142 | 6.01697E+26 |
| 94 | 187 | 329 | 143 | 6.01689E+26 |
| 95 | 189 | 332 | 144 | 6.01677E+26 |
| 96 | 191 | 336 | 146 | 6.01664E+26 |
| 97 | 193 | 340 | 148 | 6.01647E+26 |
| 98 | 195 | 343 | 149 | 6.01639E+26 |
| 99 | 197 | 346 | 150 | 6.01627E+26 |
| 100 | 199 | 350 | 152 | 6.01613E+26 |
| 101 | 201 | 354 | 154 | 6.01597E+26 |
| 102 | 203 | 357 | 155 | 6.01588E+26 |
| 103 | 205 | 360 | 156 | 6.01576E+26 |
| 104 | 207 | 364 | 158 | 6.01563E+26 |
| 105 | 209 | 368 | 160 | 6.01547E+26 |
| 106 | 211 | 371 | 161 | 6.01537E+26 |
| 107 | 213 | 374 | 162 | 6.01525E+26 |
| 108 | 215 | 378 | 164 | 6.01512E+26 |
| 109 | 217 | 382 | 166 | 6.01496E+26 |
| 110 | 219 | 385 | 167 | 6.01486E+26 |
| 111 | 221 | 388 | 168 | 6.01474E+26 |
| 112 | 223 | 392 | 170 | 6.01461E+26 |
| 113 | 225 | 396 | 172 | 6.01445E+26 |
| 114 | 227 | 399 | 173 | 6.01435E+26 |
| 115 | 229 | 402 | 174 | 6.01423E+26 |
| 116 | 231 | 406 | 176 | 6.01410E+26 |
| 117 | 233 | 410 | 178 | 6.01394E+26 |
| 118 | 235 | 413 | 179 | 6.01384E+26 |
| Average | 10764 | 6.01899E+26 |
| S.No | Interaction | String Tension | String energy |
| 1 | Weak | ||
| 2 | Strong | ||
| 3 | Electromagnetic |
| S.No | Interaction | String Tension | String energy |
| 1 | Weak | ||
| 2 | Strong | ||
| 3 | Electromagnetic |
| Element | Atomic Mass Number | Estimated Cs (J/kg·K) | Experimental Cs (J/kg·K) | Difference (J/kg·K) |
% Error |
|---|---|---|---|---|---|
| Aluminium (Al) | 26.98 | 924.5 | 897 | -27.5 | -3.1 |
| Iron (Fe) | 55.85 | 446.6 | 449 | 2.4 | 0.5 |
| Copper (Cu) | 63.55 | 392.5 | 385 | -7.5 | -1.9 |
| Silver (Ag) | 107.87 | 231.2 | 235 | 3.8 | 1.6 |
| Tungsten (W) | 183.84 | 135.7 | 134 | -1.7 | -1.3 |
| Gold (Au) | 196.97 | 126.6 | 129 | 2.4 | 1.9 |
| Lead (Pb) | 207.2 | 120.4 | 128 | 7.6 | 5.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





