Submitted:
16 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
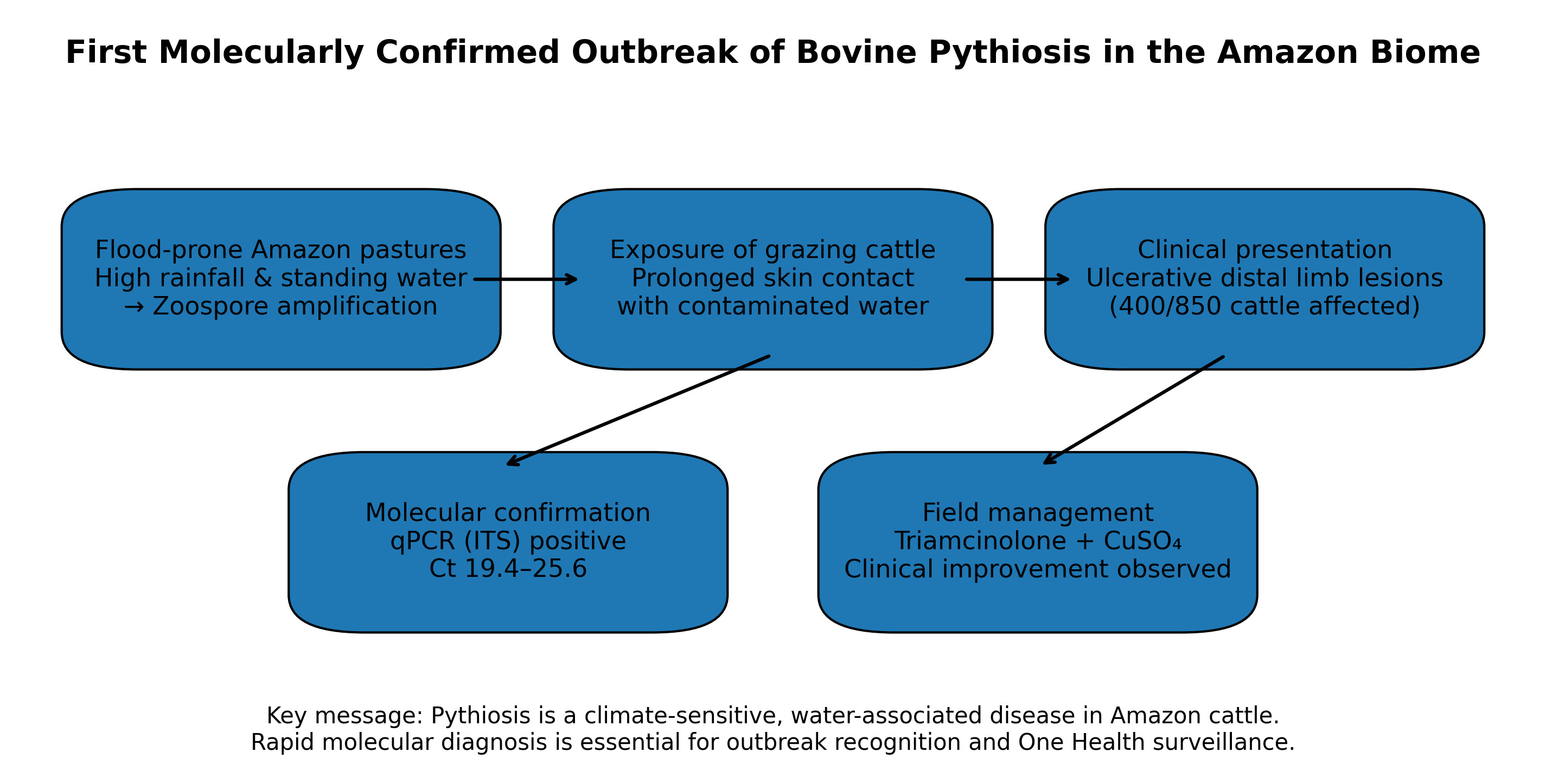
2.1. Study Area and Epidemiological Background
2.2. Clinical Assessment and Case Definition
2.3. Sample Collection, Histopathological and Molecular Diagnostic
2.4. Treatment Protocol
2.5. Ethical Considerations
3. Results
3.1. Epidemiological Characterization of the Outbreak
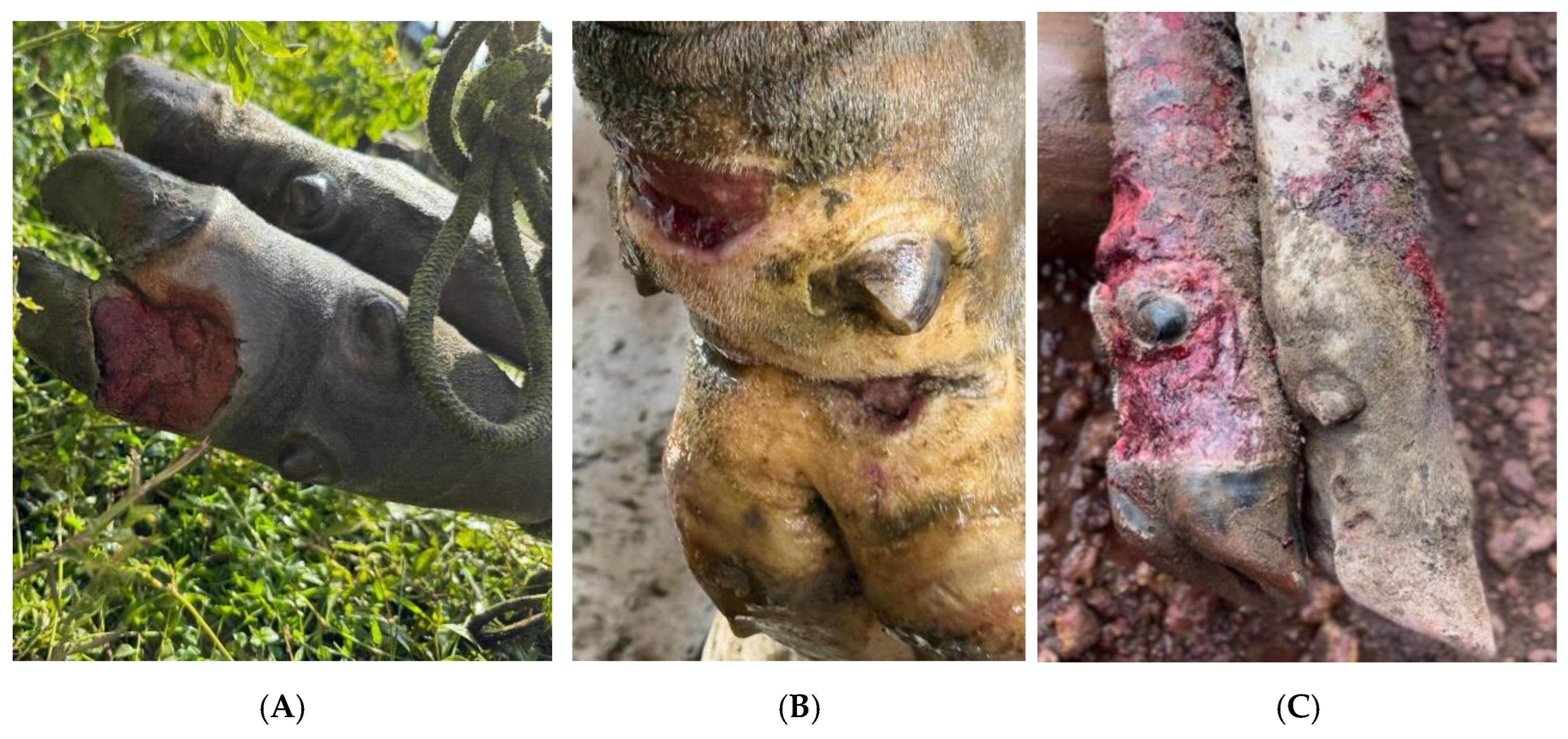
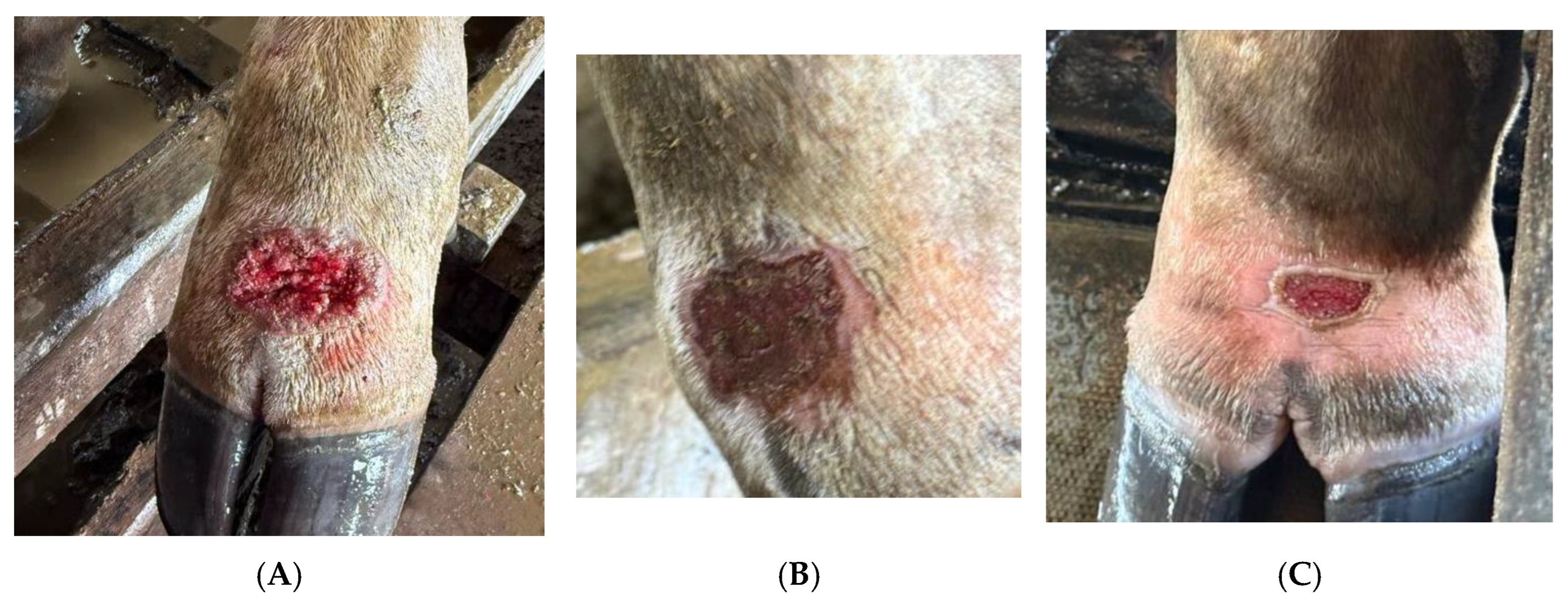
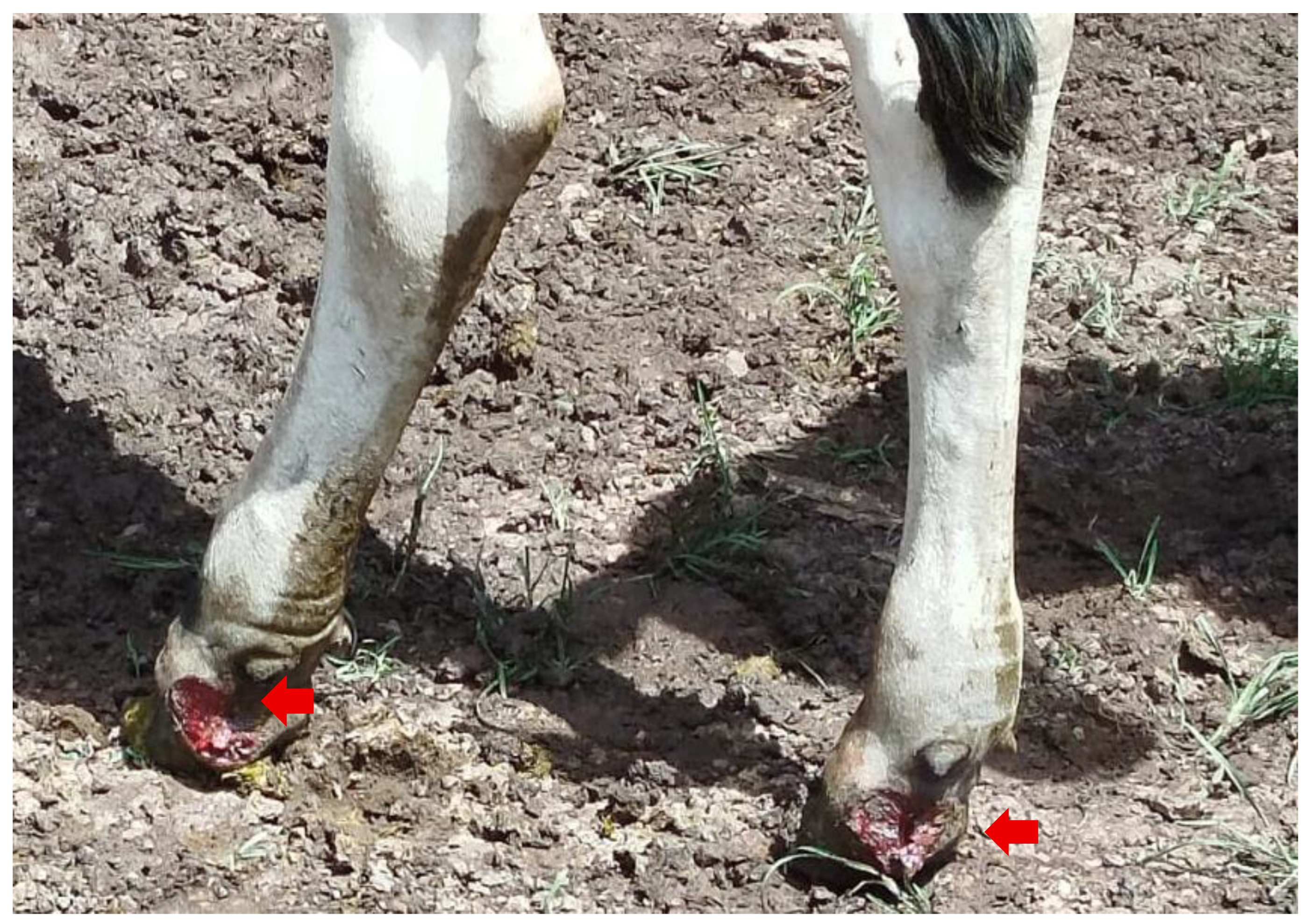
3.2. Clinical Presentation and Lesion Characteristic
3.3. Histopathological Findings
3.4. Molecular Diagnostic
3.5. Therapeutic Management and Clinical Outcomes
4. Discussion
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mendoza, L.; Ajello, L.; McGinnis, M.R. Infections caused by the oomycetous pathogen Pythium insidiosum. J. Mycol. Med. 1996, 6, 151–164.
- Alexopoulos, C.J.; Mims, C.W.; Blackwell, M. Introductory Mycology, 4th ed.; John Wiley & Sons: New York, NY, USA, 1996; pp. 599–611.
- Mendoza, L.; Hernandez, F.; Ajello, L. Life cycle of the human and animal oomycete pathogen Pythium insidiosum. J. Clin. Microbiol. 1993, 31, 2967–2973. [CrossRef]
- Gaastra, W.; Lipman, L.J.A.; De Cock, A.W.A.M.; Exel, T.K.; Pegge, R.B.G.; Scheurwater, J.; Vilela, R.; Mendoza, L. Pythium insidiosum: An overview. Vet. Microbiol. 2010, 146, 1–16. [CrossRef]
- Yolanda, H.; Krajaejun, T. Global distribution and clinical features of pythiosis in humans and animals. J. Fungi 2022, 8, 182. [CrossRef]
- Konradt, G.; Bassuino, D.M.; Bianchi, M.V.; Castro, L.; Caprioli, R.A.; Pavarini, S.P.; Santurio, J.M.; Azevedo, M.I.; Jesus, F.P.; Driemeier, D. Cutaneous pythiosis in calves: An epidemiologic, pathologic, serologic, and molecular characterization. Med. Mycol. Case Rep. 2016, 14, 24–26. [CrossRef]
- Maia, L.A.; Souto, E.P.F.; Frade, M.T.S.; Pimentel, L.A.; Azevedo, E.O.; Kommers, G.D.; Riet-Correa, F.; Dantas, A.F.M. Pythiosis in cattle in Northeastern Brazil. Pesq. Vet. Bras. 2020, 40, 340–345. [CrossRef]
- Gabriel, A.L.; Kommers, G.D.; Trost, M.E.; Barros, C.S.L.; Pereira, D.B.; Schwendler, S.E.; Santurio, J.M. Outbreak of cutaneous pythiosis in cattle. Pesq. Vet. Bras. 2008, 28, 583–587. [CrossRef]
- Grooters, A.M.; Gee, M.K.; Durbin, M.; Foureman, P.; Lovell, J. Development of a nested PCR assay for the detection of Pythium insidiosum DNA in clinical specimens. J. Vet. Intern. Med. 2003, 17, 534–538. [CrossRef]
- Keeratijarut, A.; Lohnoo, T.; Yingyong, W.; Rujirawat, T.; Srichunrusami, C.; Onpeaw, P.; Chongtrakool, P.; Brandhorst, T.T.; Krajaejun, T. Detection of the oomycete Pythium insidiosum by real-time PCR targeting the gene coding for exo-1,3-β-glucanase. J. Med. Microbiol. 2015, 64, 971–977. [CrossRef]
- Worasilchai, N.; Permpalung, N.; Chindamporn, A. High-resolution melting analysis: A novel approach for clade differentiation in Pythium insidiosum and pythiosis. Med. Mycol. 2018, 56, 868–876. [CrossRef]
- Sridapan, T.; Krajaejun, T. Nucleic acid–based detection of Pythium insidiosum: A systematic review. J. Fungi 2023, 9, 27. [CrossRef]
- Azevedo, M.I.; Botton, S.A.; Pereira, D.I.B.; Robe, L.J.; Jesus, F.P.K.; Alves, S.H.; Santurio, J.M. Phylogenetic relationships of Brazilian isolates of Pythium insidiosum based on ITS rDNA and cytochrome oxidase II gene sequences. Vet. Microbiol. 2012, 159, 141–148. [CrossRef]
- Krajaejun, T.; Sathapatayavongs, B.; Pracharktam, R.; Nitiyanant, P.; Leelachaikul, P.; Wanachiwanawin, W.; Chaiprasert, A.; Assanasen, P.; Saipetch, M.; Mootsikapun, P.; et al. Environmental and climatic factors related to outbreaks of pythiosis. Clin. Microbiol. Rev. 2009, 22, 544–558.
- Marengo, J.A.; Souza, C.M., Jr.; Thonicke, K.; Burton, C.; Halladay, K.; Betts, R.A.; Alves, L.M.; Soares, W.R. Changes in climate and land use over the Amazon region: Current and future variability and trends. Weather Clim. Extremes 2018, 22, 1–17. [CrossRef]
- Intergovernmental Panel on Climate Change (IPCC). Climate Change 2023: Synthesis Report; IPCC: Geneva, Switzerland, 2023. [CrossRef]
- Santos, C.E.P.; Santurio, J.M.; Marques, L.C. Pythiosis in production animals in the Pantanal of Mato Grosso. Pesq. Vet. Bras. 2011, 31, 1083–1089.
- Paz, G.S.; Camargo, G.G.; Cury, J.E.; et al. Outbreak of equine pythiosis in a southeastern region of Brazil: Environmental isolation and phylogeny. Transbound. Emerg. Dis. 2022, 69, 1617–1624. [CrossRef]
- do Carmo, P.M.S.; Santurio, J.M.; Kommers, G.D.; Riet-Correa, F.; Dantas, A.F.M. Diseases caused by Pythium insidiosum in sheep and goats: A review. J. Vet. Diagn. Investig. 2021, 33, 20–24. [CrossRef]
- Ribeiro, T.C.; Weiblen, C.; Azevedo, M.I.; Botton, S.A.; Robe, L.J.; Pereira, D.I.B.; Santurio, J.M. Microevolutionary analyses of Pythium insidiosum isolates of Brazil and Thailand based on exo-1,3-β-glucanase gene. Infect. Genet. Evol. 2017, 48, 58–63. [CrossRef]
- Schurko, A.M.; Mendoza, L.; Lévesque, C.A.; Désaulniers, N.L.; de Cock, A.W.A.M.; Klassen, G.R. A molecular phylogeny of Pythium insidiosum. Mycol. Res. 2003, 107, 537–544. [CrossRef]
- Htun, Z.M.; Laikul, A.; Pathomsakulwong, W.; Yurayart, C.; Lohnoo, T.; Yingyong, W.; et al. Identification and biotyping of Pythium insidiosum isolated from urban and rural areas by multiplex PCR, DNA barcoding and proteomic analyses. J. Fungi 2021, 7, 242. [CrossRef]
- Brilhante, R.S.N.; Rodrigues, A.M.; Sidrim, J.J.C.; Rocha, M.F.G.; Cordeiro, R.A.; Castelo-Branco, D.S.C.M.; Pereira, S.A.; Dantas, A.M.M.; Melo, R.S.; Monteiro, A.J.; et al. Use of copper-based compounds in the control of pythiosis: A promising antifungal strategy. J. Med. Microbiol. 2016, 65, 1–7. [CrossRef]
- Mendoza, L. Immunotherapy of pythiosis in animals. Mycopathologia 2015, 179, 213–220.
- Vasconcelos, A.B.; França, D.A.; Prado, A.C.; Yamauchi, D.H.; Silva, A.C.A.; Barros, I.O.; Valença, S.R.F.A.; Lucheis, S.B.; Bosco, S.M.G. Molecular detection of Pythium insidiosum in cutaneous lesions of horses from Northeastern Brazil. Animals 2025, 15, 2863. [CrossRef]
- Znajda, N.R.; Grooters, A.M.; Marsella, R. PCR-based detection of Pythium and Lagenidium DNA in frozen and ethanol-fixed animal tissues. Vet. Dermatol. 2002, 13, 187–194. [CrossRef]
- Hansen, R.D.; Vilela, R.; Mendoza, L. Cutaneous pythiosis in a Red Brangus beef calf cured by immunotherapy. Med. Mycol. Case Rep. 2016, 14, 20–23. [CrossRef]
- Pereira, D.I.B.; Botton, S.A.; Ianiski, L.B.; Braga, C.Q.; Maciel, A.F.; Melo, L.G.; Zambrano, C.G.; Bruhn, F.R.P.; Santurio, J.M. Equidae pythiosis in Brazil and the world: A systematic review of the last 63 years (1960–2023). Braz. J. Microbiol. 2024, 55, 2969–2981. [CrossRef]
- de Souto, E.P.F.; Kommers, G.D.; Souza, A.P.; Miranda Neto, E.G.; Assis, D.M.; Riet-Correa, F.; Galiza, G.J.N.; Dantas, A.F.M. A retrospective study of pythiosis in domestic animals in Northeastern Brazil. J. Comp. Pathol. 2022, 195, 34–50. [CrossRef]
- dos Santos, J.B.; Salvarani, F.M. Impacts of Climate Change on Cattle Health and Production in the Brazilian Amazon Biome. Ruminants 2025, 5, 58. [CrossRef]
- Cheung, C.; Mendoza, L.; Grooters, A.M.; et al. From plants to animals: Pythium, Lagenidium, Aphanomyces, and other oomycete pathogens of animals from a One Health perspective. Aust. Vet. J. 2026. [CrossRef]
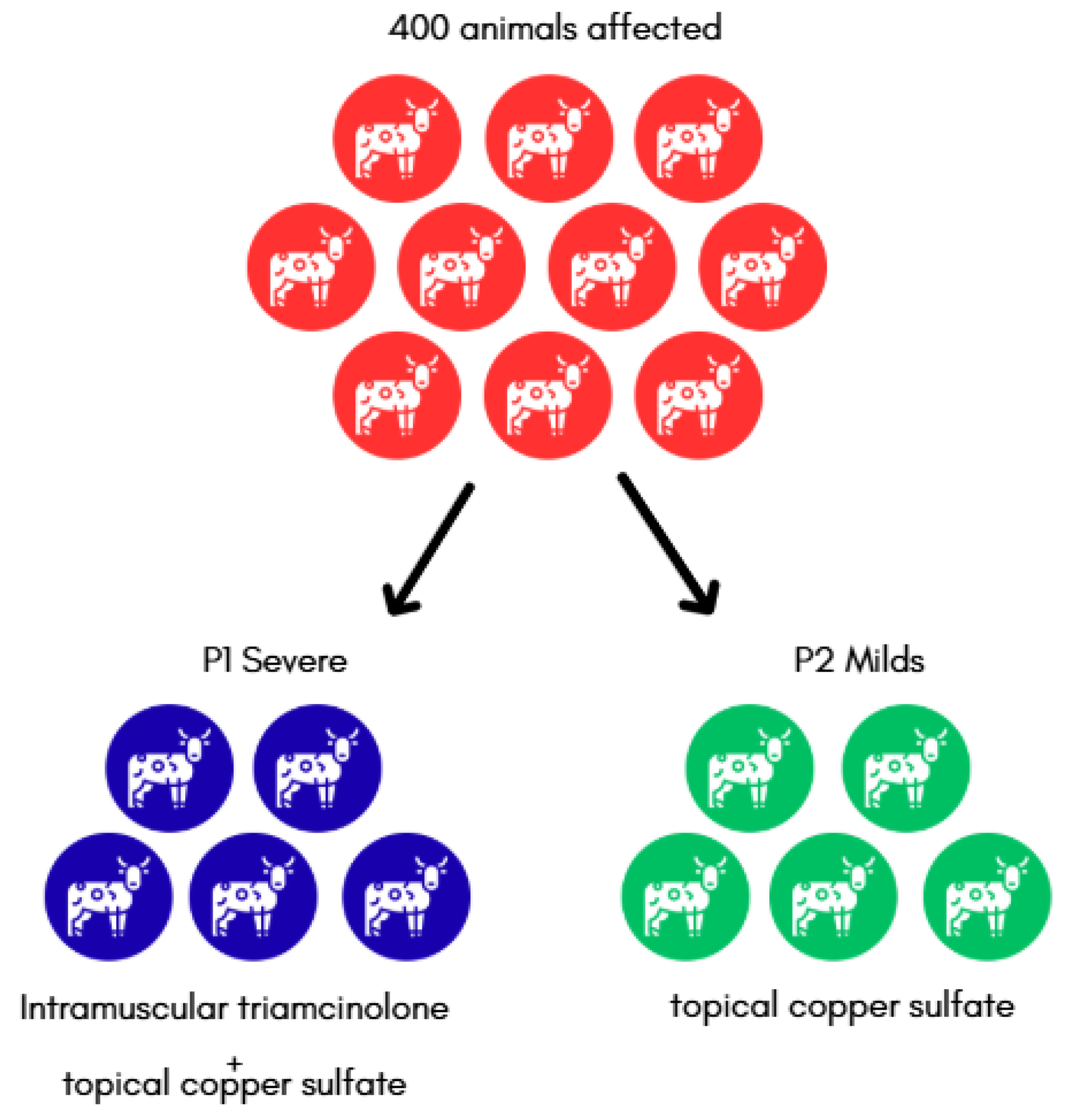

| Outcome | Number of Animals | Retreatment | Recovered |
|---|---|---|---|
| Protocol 1 | 213 | 22 | 213 |
| Protocol 2 | 187 | 0 | 187 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





