Submitted:
24 February 2026
Posted:
28 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
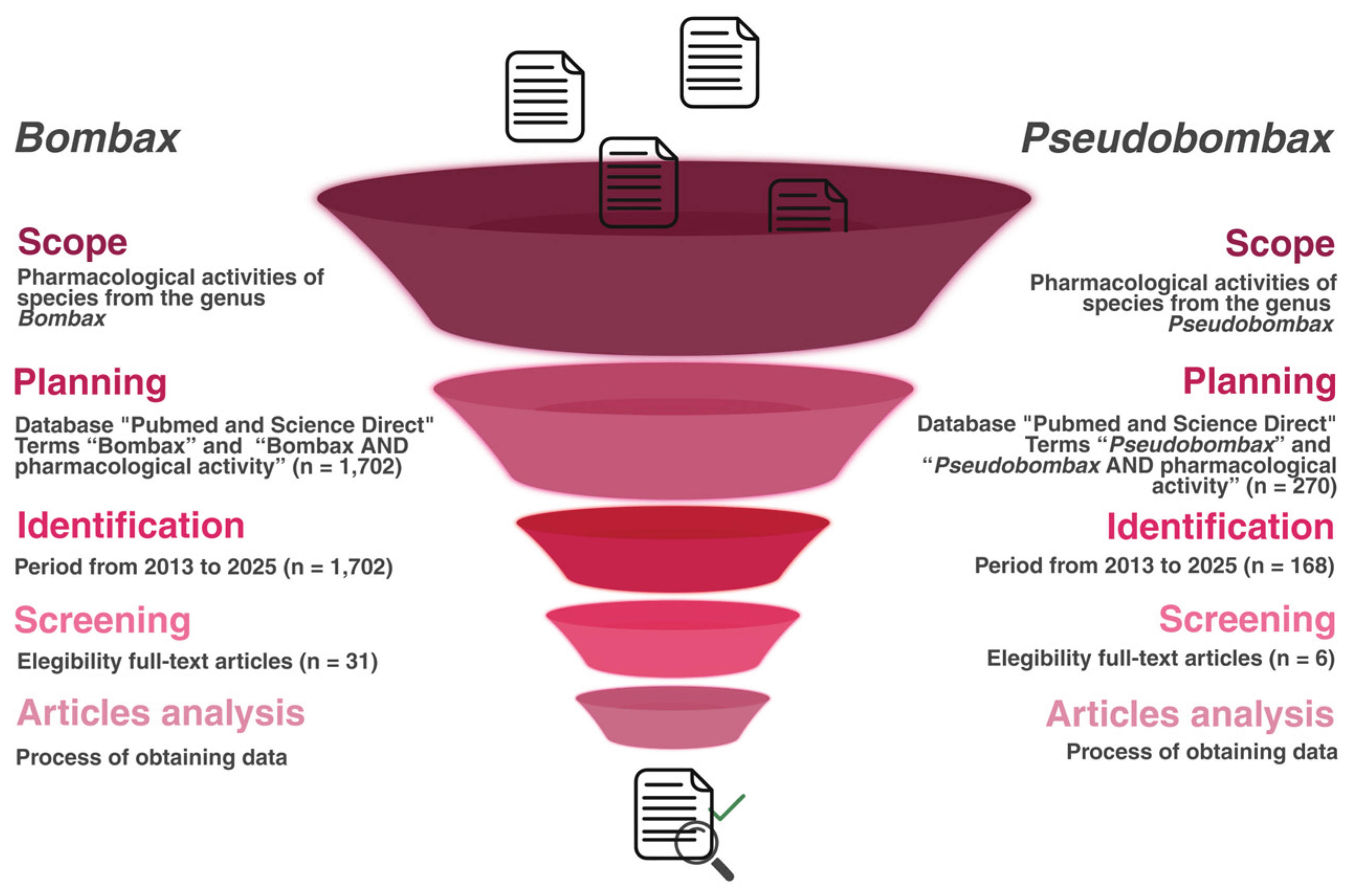
2.1. Study Design
2.2. Literature Search Strategy
- “Bombax”
- “Bombax AND pharmacological activity”
- “Pseudobombax”
- “Pseudobombax AND pharmacological activity”
2.3. Eligibility Criteria
2.4. Study Selection Process
2.5. Data Extraction and Synthesis
- Botanical species investigated;
- Plant part(s) utilized;
- Type of extract, fraction, or isolated compound;
- Biological or pharmacological activity evaluated;
- Experimental model employed (in vitro, in vivo, or in silico);
- Identified bioactive compounds (when applicable).
2.6. Assessment of Evidence Quality
2.7. Hierarchical Classification of Evidence Strength
3. Results
3.1. Study Identification and Selection
3.2. General Characteristics of the Evidence Base
3.3. Phytochemical Characterization: Analytical Scope and Depth
- Lack of comprehensive quantitative profiling: Studies often focused on qualitative identification without determining compound concentrations.
- Non-standardized extraction protocols: High variability in solvent systems and extraction parameters limits inter-study comparability.
- Absence of batch reproducibility assessments: Minimal data exists regarding the chemical consistency of plant materials across different harvests or geographic locations.
- Underutilization of bioassay-guided fractionation: Systematic isolation strategies to identify active principles remain scarce.
3.3. Genus Bombax
3.3.1. Biological Activities: Critical Appraisal of Experimental Evidence
3.3.2. Antioxidant Activity
3.3.3. Antimicrobial and Antiviral Activities
3.3.4. Cytotoxic and Anticancer Effects
- Lack of selectivity indices: Failure to evaluate toxicity in non-tumoral (healthy) cell lines.
- Absence of mechanistic confirmation: Lack of data regarding apoptosis induction or cell cycle arrest pathways.
- Inadequate validation: Absence of in vivo tumor xenograft models.
- Pharmacological gaps: No assessment of pharmacodynamic or pharmacokinetic profiles.
3.3.5. Metabolic and Organ-Protective Effects
3.3.6. Hierarchical Assessment of Evidence Strength
3.4. Genus Pseudobombax
3.4.1. Biological Activities: Critical Appraisal of Experimental Evidence
3.4.2. Antioxidant Activity
3.4.3. Antimicrobial Activity
3.4.4. Anti-Inflammatory and Antinociceptive Effects
3.4.5. Other Biological Properties
3.4.6. Hierarchical Assessment of Evidence Strength
- Level II: Predominates in studies concerning anti-inflammatory and antinociceptive effects, where in vivo functional efficacy is demonstrated but mechanistic details are sparse.
- Level III: Comprises the bulk of antioxidant and antimicrobial research, restricted to in vitro systems with low translational predictability.
- Level IV: Includes preliminary reports on anti-sickling and specific cellular protective effects lacking systemic validation.
4. Discussion
5. Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| A549 | Human alveolar basal epithelial cell line |
| ABTS | 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid |
| AGE | Advanced Glycation End-products |
| ALT | Alanine Aminotransferase |
| ARE | Antioxidant Response Element pathway |
| AST | Aspartate Aminotransferase |
| BSA | Bovine Serum Albumin |
| CD56 | Cluster of Differentiation 56 |
| COX-2 | Cyclooxygenase-2 |
| DNA | Deoxyribonucleic Acid |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| EGFR | Epidermal Growth Factor Receptor |
| ESR1 | Estrogen Receptor 1 |
| FRAP | Ferric Reducing Antioxidant Power |
| GC-MS | Gas Chromatography–Mass Spectrometry |
| GPx | Glutathione Peroxidase |
| HepG2 | Human hepatocellular carcinoma cell line |
| HIO180 | Human intestinal organoid 180 |
| HPLC | High-Performance Liquid Chromatography |
| Huh7 | Human hepatocellular carcinoma cell line |
| LC-MS | Liquid Chromatography–Mass Spectrometry |
| MCF-7 | Human breast adenocarcinoma cell line |
| MDA | Malondialdehyde |
| MIC | Minimum Inhibitory Concentration |
| MMPs | Matrix Metalloproteinases |
| Nrf2 | Nuclear factor erythroid 2–related factor 2 pathway |
| PK | Pharmacokinetics |
| RSV | Respiratory syncytial virus |
| SDF-1 | Stromal cell–Derived Factor 1 |
| SOD | Superoxide Dismutase |
| SRC | Proto-oncogene tyrosine-protein kinase Src |
| STZ | Streptozotocin |
| T2DM | Type 2 Diabetes Mellitus |
| TAC | Total Antioxidant Capacity |
References
- Shao, L.; Jin, S.; Chen, J.; Yang, G.; Fan, R.; Zhang, Z.; Deng, Q.; Han, J.; Ma, X.; Dong, Z.; et al. High-Quality Genomes of Bombax ceiba and Ceiba pentandra Provide Insights into the Evolution of Malvaceae Species and Differences in Their Natural Fiber Development. Plant Commun. 2024, 5, 100832. [Google Scholar] [CrossRef] [PubMed]
- Das, G.; Shin, H.-S.; Ningthoujam, S.S.; Talukdar, A.D.; Upadhyaya, H.; Tundis, R.; Das, S.K.; Patra, J.K. Systematics, Phytochemistry, Biological Activities and Health Promoting Effects of the Plants from the Subfamily Bombacoideae (Family Malvaceae). Plants 2021, 10, 651. [Google Scholar] [CrossRef]
- Posinasetty, B.; Chikatipalli, R.; Chenchula, S.; Kuttiappan, A.; G, S.; G N A, L.; Audinarayana, N. Anti-Arthritic Efficacy of Bombax Ceiba Ethanolic Extract in a Murine Model for Rheumatoid Arthritis Using in Vivo, in Vitro and Radiological Analysis. Bioinformation 2023, 19, 833–839. [Google Scholar] [CrossRef]
- Diab, K.A.; El-Shenawy, R.; Helmy, N.M.; El-Toumy, S.A. Polyphenol Content, Antioxidant, Cytotoxic, and Genotoxic Activities of Bombax ceiba Flowers in Liver Cancer Cells Huh7. Asian Pac. J. Cancer Prev. 2022, 23, 1345–1350. [Google Scholar] [CrossRef]
- de Senes-Lopes, T.F.; da Luz, J.R.D.; Guterres, Z. da R.; Barbosa, E.A.; Batista, D.; Galdino, O.A.; Ururahy, M.A.G.; Gomes Dos Santos, E.C.; López, J.A.; Araujo-Silva, G.; et al. Pseudobombax parvifolium Hydroalcoholic Bark Extract: Chemical Characterisation and Cytotoxic, Mutagenic, and Preclinical Aspects Associated with a Protective Effect on Oxidative Stress. Metabolites 2023, 13, 748. [Google Scholar] [CrossRef]
- Collares, L.J.; Turchen, L.M.; Guedes, R.N.C. Research Trends, Biases, and Gaps in Phytochemicals as Insecticides: Literature Survey and Meta-Analysis. Plants 2023, 12, 318. [Google Scholar] [CrossRef]
- Pacheco, G.; Vianna-Almeida, M.G.; Oliveira Garcia, R.; Mansur, E. Challenges and Recent Progress on the Use of Cryobiotechnology for Conserving Brazilian Native Plants. Cryo Letters 2025, 46, 143–163. [Google Scholar] [CrossRef]
- Noguera, N.H.; Noguera, D.C.L.H.; Machado, A.P. da F.; Reguengo, L.M.; Nascimento, R. de P. do Emerging Berries from the Brazilian Amazon and Atlantic Forest Biomes: New Sources of Bioactive Compounds with Potential Health Benefits. Food Funct. 2024, 15, 5752–5784. [Google Scholar] [CrossRef] [PubMed]
- Taher, M.A.; Hossain, M.J.; Zahan, M.S.; Hasan, M.M.; Ferdous, J.; Rahman, A.; Khan, M.; Hosain, M.K.; Rashid, M.A. Phyto-Pharmacological and Computational Profiling of Bombax ceiba Linn. Leaves Revealed Pharmacological Properties against Oxidation, Hyperglycemia, Pain, and Diarrhea. Heliyon 2024, 10, e35422. [Google Scholar] [CrossRef] [PubMed]
- Komati, A.; Anand, A.; Nagendla, N.K.; Madhusudana, K.; Mudiam, M.K.R.; Babu, K.S.; Tiwari, A.K. Bombax ceiba Calyx Displays Antihyperglycemic Activity via Improving Insulin Secretion and Sensitivity: Identification of Bioactive Phytometabolomes by UPLC-QTof-MS/MS. J. Food Sci. 2022, 87, 1865–1881. [Google Scholar] [CrossRef]
- Haque, M.U.; Alam, A.K.; Islam Shovon, M.T.; Sujon, K.M.; Hasan Maruf, M.M.; Kabir, S.R.; Faisal Hoque, K.M.; Reza, M.A. Unveiling the Apoptotic Potential of Antioxidant-Rich Bangladeshi Medicinal Plant Extractives and Computational Modeling to Identify Antitumor Compounds. Heliyon 2024, 10, e38885. [Google Scholar] [CrossRef]
- Ullah, M.N.; Ali, I.; Hanif, S.; Ali Syed, M.; Talha, M.; Samran, M.A.; Haider, A.; Huraira, M. Phytochemical Screening and Physicochemical Analysis of Oil Extracted from Seeds of Bombax ceiba and Determination of Antioxidant Activity. Pak. J. Pharm. Sci. 2024, 37, 743–751. [Google Scholar]
- Xu, G.-K.; Qin, X.-Y.; Wang, G.-K.; Xie, G.-Y.; Li, X.-S.; Sun, C.-Y.; Liu, B.-L.; Qin, M.-J. Antihyperglycemic, Antihyperlipidemic and Antioxidant Effects of Standard Ethanol Extract of Bombax ceiba Leaves in High-Fat-Diet- and Streptozotocin-Induced Type 2 Diabetic Rats. Chin. J. Nat. Med. 2017, 15, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Masood-Ur-Rehman; Akhtar, N.; Mustafa, R. Antibacterial and Antioxidant Potential of Stem Bark Extract of Bombax ceiba Collected Locally from South Punjab Area of Pakistan. Afr. J. Tradit. Complement. Altern. Med. 2017, 14, 9–15. [Google Scholar] [CrossRef]
- Laoung-On, J.; Ounjaijean, S.; Sudwan, P.; Boonyapranai, K. Phytochemical Screening, Antioxidant Effect and Sperm Quality of the Bombax ceiba Stamen Extracts on Charolais Cattle Sperm Induced by Ferrous Sulfate. Plants 2024, 13, 960. [Google Scholar] [CrossRef] [PubMed]
- Amat-Ur-Rasool, H.; Symes, F.; Tooth, D.; Schaffert, L.-N.; Elmorsy, E.; Ahmed, M.; Hasnain, S.; Carter, W.G. Potential Nutraceutical Properties of Leaves from Several Commonly Cultivated Plants. Biomolecules 2020, 10, 1556. [Google Scholar] [CrossRef]
- Sinha, S.; Kumar, B.; Luqman, S.; Singh, D.K. Neuroprotective Potential of Cucurbita Maxima Duchesne Ex Poir, Caeselpenia bunduc (L.) Roxb and Bombax Ceiba Linn Extracts. S. Afr. J. Bot. 2019, 120, 319–325. [Google Scholar] [CrossRef]
- Zhang, Y.-B.; Wu, P.; Zhang, X.-L.; Xia, C.; Li, G.-Q.; Ye, W.-C.; Wang, G.-C.; Li, Y.-L. Phenolic Compounds from the Flowers of Bombax malabaricum and Their Antioxidant and Antiviral Activities. Molecules 2015, 20, 19947–19957. [Google Scholar] [CrossRef]
- Yasien, S.; Iqbal, M.M.; Javed, M.; Alnuwaiser, M.A.; Iqbal, S.; Mahmood, Q.; Elkaeed, E.B.; Dera, A.A.; Alrbyawi, H.; Pashameah, R.A.; et al. Comparative Evaluation of Various Extraction Techniques for Secondary Metabolites from Bombax ceiba L. Flowering Plants along with in Vitro Anti-Diabetic Performance. Bioengineering (Basel) 2022, 9, 486. [Google Scholar] [CrossRef]
- Gupta, P.; Goyal, R.; Chauhan, Y.; Sharma, P.L. Possible Modulation of FAS and PTP-1B Signaling in Ameliorative Potential of Bombax ceiba against High Fat Diet Induced Obesity. BMC Complement. Altern. Med. 2013, 13, 281. [Google Scholar] [CrossRef]
- Safdar, M.; Aslam, S.; Akram, M.; Khaliq, A.; Ahsan, S.; Liaqat, A.; Mirza, M.; Waqas, M.; Qureshi, W.A. Bombax ceiba Flower Extract Mediated Synthesis of Se Nanoparticles for Antibacterial Activity and Urea Detection. World J. Microbiol. Biotechnol. 2023, 39, 80. [Google Scholar] [CrossRef]
- Aziz, N.; Alhajouj, S.A.; Basit, A.; Khan, I.A.; Alaida, M.F.; Alzayed, R.M.; Albalawi, M.A.; Alshareef, S.A.; Al-Duais, M.A.; Sakran, M.; et al. Green Synthesis and Antibacterial Activity of Silver and Gold Nanoparticles Using Crude Flavonoids Extracted from Bombax ceiba Flowers. Cell. Mol. Biol. (Noisy-le-grand) 2025, 71, 127–135. [Google Scholar] [CrossRef]
- Hossain, E.; Sarkar, D.; Chatterjee, M.; Chakraborty, S.; Mandal, S.C.; Gupta, J.K. Effect of Methanol Extract of Bombax malabaricum Leaves on Nitric Oxide Production during Inflammation. Acta Pol. Pharm. 2013, 70, 255–260. [Google Scholar]
- Xu, G.-K.; Sun, C.-Y.; Qin, X.-Y.; Han, Y.; Li, Y.; Xie, G.-Y.; Min-Jian, Q. Effects of Ethanol Extract of Bombax ceiba Leaves and Its Main Constituent Mangiferin on Diabetic Nephropathy in Mice. Chin. J. Nat. Med. 2017, 15, 597–605. [Google Scholar] [CrossRef]
- Arafa, A.F.; Foda, D.S.; Mahmoud, A.H.; Metwally, N.S.; Farrag, A.R.H. Bombax Ceiba Flowers Extract Ameliorates Hepatosteatosis Induced by Ethanol and Relatively Moderate Fat Diet in Rats. Toxicol. Rep. 2019, 6, 401–408. [Google Scholar] [CrossRef]
- Tundis, R.; Rashed, K.; Said, A.; Menichini, F.; Loizzo, M.R. In Vitro Cancer Cell Growth Inhibition and Antioxidant Activity of Bombax ceiba (Bombacaceae) Flower Extracts. Nat. Prod. Commun. 2014, 9, 691–694. [Google Scholar] [CrossRef]
- Yin, K.; Yang, J.; Wang, F.; Wang, Z.; Xiang, P.; Xie, X.; Sun, J.; He, X.; Zhang, X. A Preliminary Study of the Chemical Composition and Bioactivity of Bombax ceiba L. Flower and Its Potential Mechanism in Treating Type 2 Diabetes Mellitus Using Ultra-Performance Liquid Chromatography Quadrupole-Time-Flight Mass Spectrometry and Network Pharmacology Analysis. Front. Nutr. 2022, 9, 1018733. [Google Scholar] [CrossRef]
- Gavade, N.L.; Deshmukh, S.P.; Serena Dossou, A.; Raut, S.; Garadkar, K.M. Phytosynthesis of Anisotropic Silver and Gold Nanoparticles: Characterization and Anticancer Activity towards HIO180 and HeyA8 Cells. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2025, 333, 125905. [Google Scholar] [CrossRef]
- Dolsophon, K.; Nak-On, S.; Chontananarth, T. Tegumental Surface Change in Paramphistomum epiclitum Caused by Bombax ceiba Flowers and Black Pepper Seed Extract. Exp. Parasitol. 2024, 260, 108724. [Google Scholar] [CrossRef]
- Komati, A.; Anand, A.; Shaik, H.; Mudiam, M.K.R.; Suresh Babu, K.; Tiwari, A.K. Bombax ceiba (Linn.) Calyxes Ameliorate Methylglyoxal-Induced Oxidative Stress via Modulation of RAGE Expression: Identification of Active Phytometabolites by GC-MS Analysis. Food Funct. 2020, 11, 5486–5497. [Google Scholar] [CrossRef]
- Wang, L.; Xie, S.; Jiang, X.; Xu, C.; Wang, Y.; Feng, J.; Yang, B. Therapeutic Effects of Bombax ceiba Flower Aqueous Extracts against Loperamide-Induced Constipation in Mice. Pharm. Biol. 2023, 61, 125–134. [Google Scholar] [CrossRef]
- Anandhan, P.; Hayagreva Kumar, M.; Elshafey, S.H.; J.C., S; Jones, S.; Sheriff, D.; Pramod Roy, A.; Ayyaswamy, G.; Tk, B.; K, P. Evaluation of In-Vitro Studies of the Shalmali Extract on Human Endometrial Stromal Cells. Cureus 2024, 16, e60699. [Google Scholar] [CrossRef]
- Chauhan, S.; Sharma, A.; Upadhyay, N.K.; Singh, G.; Lal, U.R.; Goyal, R. In-Vitro Osteoblast Proliferation and in-Vivo Anti-Osteoporotic Activity of Bombax ceiba with Quantification of Lupeol, Gallic Acid and β-Sitosterol by HPTLC and HPLC. BMC Complement. Altern. Med. 2018, 18, 233. [Google Scholar] [CrossRef]
- Upadhyay, S.S.; Parate, S.S.; D’Souza, U.P.; Kurian, A.T.; Biradar, S.; Bhat, G.S.; Sadananda, V.; Prasad, T.S.K. An Integrative Omics and Network Pharmacology Approach to Evaluate the Antiulcer Activity of Bombax ceiba L. Gum in Rats. ACS Omega 2025, 10, 59044–59055. [Google Scholar] [CrossRef]
- Antwi-Adjei, M.; Yeboah, K.O.; Oppong-Kyekyeku, J.; Osafo, N. Inflammation Modulating Activity of the Hydroethanol Stem Bark Extract of Bombax costatum in Murine Models. Scientifica (Cairo) 2022, 2022, 6882147. [Google Scholar] [CrossRef]
- Léa Blondelle, K.D.; Simplice, F.H.; Hervé Hervé, N.A.; Eglantine, K.W.; Roland, R.N.; Jorelle Linda, D.K.; Balbine, K.N.; Simon Désiré, G.N.; Guillaume, C.W.; Alin, C. Antidepressant, Anti-Amnesic and Vasoprotective Effect of Bombax costatum Pellegr. & Vuillet Aqueous Stem Bark Extract on Chronic Mild Unpredictable Stress Induced in Rat. J. Ethnopharmacol. 2022, 293, 115315. [Google Scholar] [CrossRef]
- Christian, A.G.; Thecla, E.C.; Dick, E.A.; Chile, A.E.; Chimsorom, C.K.; Ckukwu, N.D.; Nwobodo, N.N. In vivo antiplasmodial activity of Bombax buonopozense root bark aqueous extract in mice infected by Plasmodium berghei. Journal of Traditional Chinese Medicine 2017, 37, 431–435. [Google Scholar] [CrossRef]
- Fikry, E.; Mahdi, I.; Buğra Ortaakarsu, A.; Tawfeek, N.; Adhiambo Ochieng, M.; Ben Bakrim, W.; Ao Abdelfattah, M.; Omari, K.W.; Mahmoud, M.F.; Sobeh, M. Dermato-Cosmeceutical Properties of Pseudobombax ellipticum (Kunth) Dugand: Chemical Profiling, in Vitro and in Silico Studies. Saudi Pharm. J. 2023, 31, 101778. [Google Scholar] [CrossRef]
- Mohamed, A.S.; Abd El Dayem, O.Y.; El Shamy, A.M.; El Sakhawy, F.S.; El Gedaily, R.A. Comparative Antisickling and Antioxidant Activities of Pseudobombax ellipticum Cultivars in Relation to Their Metabolite Profiling Using LC/MS. RSC Adv. 2023, 13, 21327–21335. [Google Scholar] [CrossRef]
- Santos, M.E.N.; Queiroz, W.A.M.; Santos, W.M.; Veras, B.O.; Silva, M.V.; Filho, A.A.O.; Gomes, M.A.; Torres, M.C.M. Phytochemical Characterization, Antioxidant, and Photoprotective Activities of Pseudobombax simplicifolium A. Robyns. Chem. Biodivers. 2026, 23, e01237. [Google Scholar] [CrossRef]
- Paiva, D.C.C.; dos Santos, C.A.; Diniz, J.C.; Viana, F.A.; Thomazzi, S.M.; Falcão, D.A. Anti-Inflammatory and Antinociceptive Effects of Hydroalcoholic Extract from Pseudobombax marginatum Inner Bark from Caatinga Potiguar. J. Ethnopharmacol. 2013, 149, 416–421. [Google Scholar] [CrossRef] [PubMed]
- da Silva Santana, K.T.; Do Nascimento Marinho, K.S.; de Melo Alcântara, L.F.; da Silva Carvalho, C.M.; Alves Viturino da Silva, W.; Assunção Ferreira, M.R.; da Silva, M.M.; Dos Santos Souza, T.G.; Soares, L.A.L.; Chagas, C.A.; et al. Phytochemical Profile and Determination of Cytotoxicity, Acute Oral Toxicity, Genotoxicity, and Mutagenicity of Aqueous and Ethanolic Extracts of Pseudobombax marginatum (A. St.-Hil.) A. Robyns. J. Toxicol. Environ. Health Part A. 2024, 87, 662–673. [Google Scholar] [CrossRef] [PubMed]
- Penido, A.B.; de Morais, S.M.; Ribeiro, A.B.; Silva, A.Z. Ethnobotanical study of medicinal plants in Imperatriz, State of Maranhão, Northeastern Brazil. Acta Amazon. 2016, 46, 345–354. [Google Scholar] [CrossRef]
- Dötterl, S.; Gershenzon, J. Chemistry, Biosynthesis and Biology of Floral Volatiles: Roles in Pollination and Other Functions. Nat. Prod. Rep. 2023, 40, 1901–1937. [Google Scholar] [CrossRef]
- Beltrán-Rodríguez, L.; Valdez-Hernández, J.I.; Saynes-Vásquez, A.; Blancas, J.; Sierra-Huelsz, J.A.; Cristians, S.; Martínez-Ballesté, A.; Romero-Manzanares, A.; Luna-Cavazos, M.; Borja de la Rosa, M.A.; et al. Sustaining Medicinal Barks: Survival and Bark Regeneration of Amphipterygium adstringens (Anacardiaceae), a Tropical Tree under Experimental Debarking. Sustainability 2021, 13, 2860. [Google Scholar] [CrossRef]
| Evidence Level | Definition | Minimum Experimental Criteria | Typical Study Design Observed | Main Limitations Identified |
|---|---|---|---|---|
| Level I | In vivo validation with biochemical and/or molecular mechanistic markers | Established disease model + biomarker quantification + dose–response analysis | Rodent disease models with biochemical endpoints | Limited molecular pathway validation; absence of clinical follow-up |
| Level II | Functional in vivo evidence without detailed mechanistic elucidation | Disease model + functional outcome measurement | Animal models without molecular confirmation | Lack of target-specific validation |
| Level III | In vitro cellular or biochemical assays | Cell viability, enzyme inhibition, or radical-scavenging assays | Cell culture models, DPPH/ABTS assays | No in vivo confirmation; limited physiological relevance |
| Level IV | Exploratory or limited evidence | Qualitative or preliminary assays | Screening assays without replication or controls | Insufficient experimental rigor |
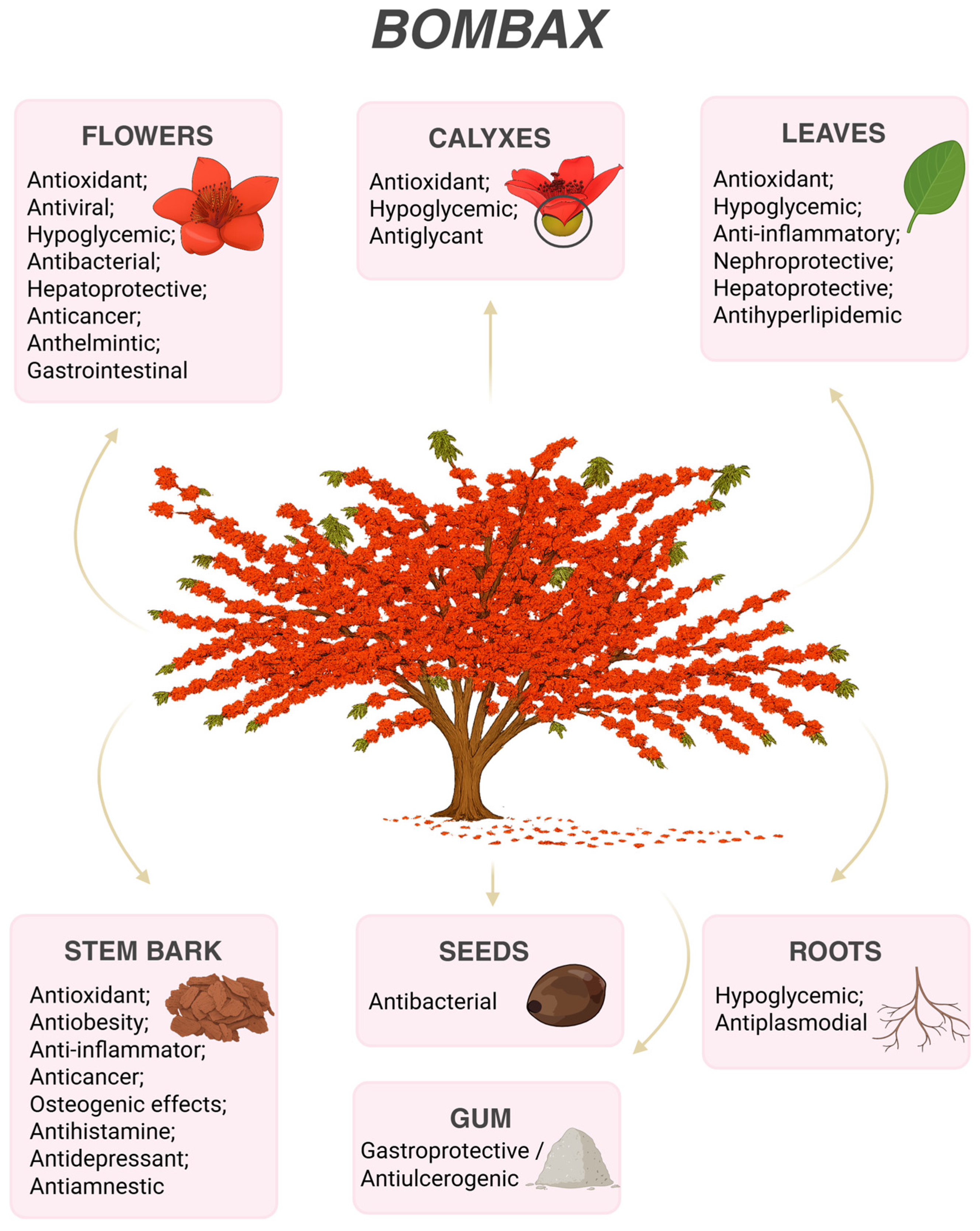
| Bombax ceibaL. | ||||
| Biological activity | Experimental model | Plant part / Extract | Main associated metabolites | Reference |
| Antioxidant | In vitro (DPPH, ABTS, FRAP); in vivo (MDA reduction) | Leaves (methanolic), flowers, calyces, stamens, bark | β-Sitosterol; gallic acid; rutin; scopoletin; mangiferin; flavonoids; tannins | [4,9,10,11,12,13,14,15,16,17,18] |
| Antiviral | In vitro (RSV cytopathic effect and plaque reduction assays) | Flowers | Mangiferin | [18] |
| Hypoglycemic / Antidiabetic | In vivo (diabetic rodent models; α-glucosidase/α-amylase inhibition) | Leaves, calyces, roots, flowers | Mangiferin; protocatechuic acid; quercetin; isovitexin | [10,19] |
| Anti-obesity | In vivo (high-fat diet model) | Stem bark | Lupeol; flavonoids | [20] |
| Antibacterial | In vitro (MIC assays against Gram-positive and Gram-negative bacteria) | Flowers, roots, seeds | Not chemically characterized | [14,21,22] |
| Anti-inflammatory | In vivo (carrageenan-induced edema); in vitro (NO inhibition) | Leaves | Not chemically characterized | [23] |
| Anti-arthritic | In vivo (adjuvant-induced arthritis; IL-6, TNF-α reduction) | Aerial parts | Not chemically characterized | [3] |
| Nephroprotective | In vivo (STZ-induced nephropathy) | Leaves | Mangiferin | [24] |
| Hepatoprotective | In vivo (ethanol-induced liver injury; ALT/AST reduction) | Leaves, flowers | Polyphenols; flavonoids; saponins | [25] |
| Cytotoxic / Anticancer | In vitro (MCF-7, HepG2, A549, HIO180, Huh7 cell lines) | Flowers, bark | β-Sitosterol; gallic acid; flavonoids; phenolics | [4,26,27,28] |
| Anthelmintic | Ex vivo helminth model | Flowers | Not chemically characterized | [29] |
| Antihyperglycemic | In vivo (T2DM rodent model) | Leaves | Mangiferin; isoorientin; vitexin; isomangiferin; isovitexin; quercetin hexoside; 2′-trans-O-coumaroyl mangiferin; nigricanoside | [13] |
| Antihyperlipidemic | In vivo (lipid profile modulation) | Leaves | Same as above | [13] |
| Antiglycation | In vitro (AGE formation inhibition; BSA–methylglyoxal model) | Flower calyx | Myo-inositol; scopoletin; D-sedoheptulose; succinic acid; xylitol | [30] |
| Gastrointestinal (laxative effect) | In vivo (intestinal motility assays) | Flowers (aqueous extract) | Chlorogenic acid; rutin | [31] |
| Anti-hemorrhagic | In vitro (human endometrial stromal cells; ESR1, CD56, SDF-1 expression) | Dried resin | Not chemically characterized | [32] |
| Osteogenic / Anti-osteoporotic | In vivo (bone mineral density in Wistar rats) | Bark | Lupeol; β-sitosterol; gallic acid | [33] |
| Gastroprotective / Antiulcerogenic activity | In vivo study in rats with experimentally induced gastric ulcers | Gum (plant exudate) | Alkaloids, flavonoids, glycosides; interaction with targets such as EGFR, SRC, COX2, MMPs | [34] |
| Bombax costatumPellegr. & Vuillet | ||||
| Anti-inflammatory / Anti-arthritic | In vivo (prostaglandin E₂-induced edema; Freund’s adjuvant arthritis model) | Stem bark | Not chemically characterized | [35] |
| Antihistaminic | In vivo (clonidine-induced catalepsy) | Stem bark | Not chemically characterized | [35] |
| Antidepressant | In vivo (sucrose preference, forced swim test; corticosterone, serotonin, dopamine levels) | Stem bark | Not chemically characterized | [36] |
| Antiamnesic | In vivo (Morris water maze; object recognition test) | Stem bark | Not chemically characterized | [36] |
| Bombax buonopozenseP. Beauv. | ||||
| Antiplasmodial | In vivo (Plasmodium berghei-infected mice) | Root bark | Not chemically characterized | [37] |
| Pseudobombax parvifoliumA.DC. | ||||
| Activity | Model | Plant part | Key metabolites | Ref. |
| Antioxidant | In vivo (lipid peroxidation reduction; SOD and GPx increase in rodents) | Stem bark | Loliolide | [5] |
| Pseudobombax ellipticum(Kunth) Dugand | ||||
| Activity | Model | Plant part | Key metabolites | Ref. |
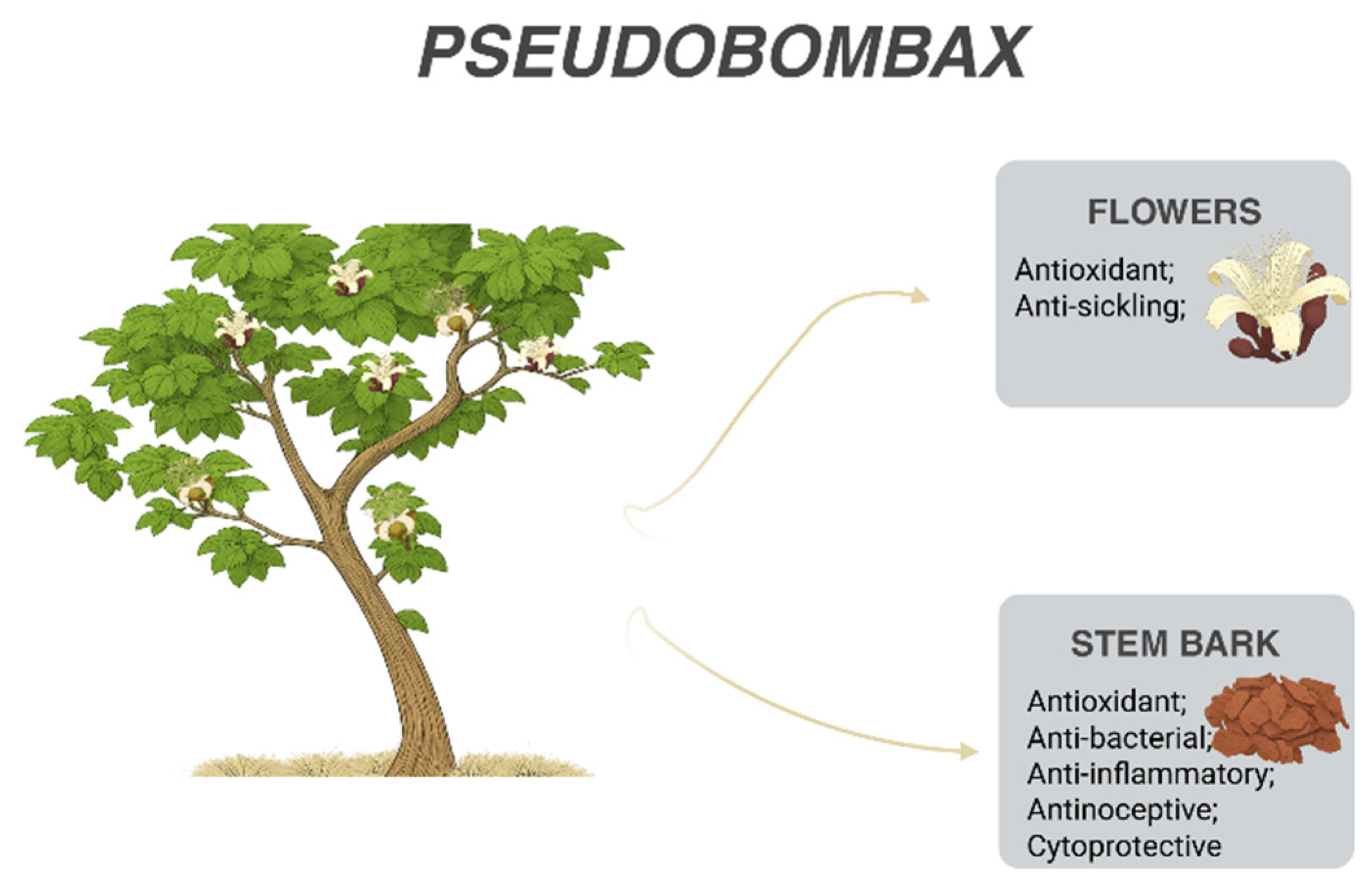
| Antioxidant | In vitro (DPPH, FRAP, iron chelation assays) | Stem bark; fresh flowers | Phenolic acids; flavonoids; pelargonidin-3-O-glucoside; cyanidin-3-O-rutinoside; rutin; kaempferol-3-O-glucoside | [38,39] |
| Antibacterial | In vitro (biofilm inhibition against Pseudomonas aeruginosa) | Stem bark | Phenolic acids; flavonoids | [38] |
| Antisickling | In vitro (reduction of sickled erythrocytes) | Fresh flowers | Pelargonidin-3-O-glucoside; cyanidin-3-O-rutinoside; rutin; kaempferol-3-O-glucoside | [39] |
| Pseudobombax simplicifoliumA. Robyns | ||||
| Antioxidant | In vitro (DPPH, ABTS, TAC) | Stem bark | Phenolic compounds, flavonoids, tannins | [40] |
| Pseudobombax ellipticumcultivar alba Hort. | ||||
| Activity | Model | Plant part | Key metabolites | Ref. |
| Antisickling | In vitro (erythrocyte sickling assay) | Fresh flowers | Pelargonidin-3-O-glucoside; cyanidin-3-O-rutinoside; rutin; kaempferol-3-O-glucoside | [39] |
| Antioxidant | In vitro (DPPH, iron chelation assays) | Stem bark; fresh flowers | Pelargonidin-3-O-glucoside; cyanidin-3-O-rutinoside; rutin; kaempferol-3-O-glucoside | [39] |
| Pseudobombax marginatum(A. St.-Hil.) A. Robyns | ||||
| Activity | Model | Plant part | Key metabolites | Ref. |
| Anti-inflammatory | In vivo (carrageenan-induced paw edema in Wistar rats) | Stem bark | Not chemically characterized | [41] |
| Antinociceptive | In vivo (acetic acid-induced writhing test) | Stem bark | Not chemically characterized | [41] |
| Cytoprotective / Genoprotective | In vitro (comet assay; DNA damage reduction) | Stem bark | Flavonoids; tannins; coumarins | [42] |
| Species | Biological Activity | Evidence Level | Critical Appraisal | Major Methodological Gaps |
|---|---|---|---|---|
| Bombax ceiba | Antioxidant | III | Activity consistently demonstrated in chemical and cellular assays; physiological relevance remains to be fully established | Limited in vivo confirmation; insufficient redox pathway characterization; extract standardization inconsistently reported |
| Bombax ceiba | Antiviral | III | Promising in vitro antiviral effect | Absence of in vivo validation; limited host–virus interaction analysis |
| Bombax ceiba | Hypoglycemic / Antidiabetic | I | Supported by established metabolic models with biochemical endpoints | Molecular target identification and pharmacokinetic profiling remain limited |
| Bombax ceiba | Antiobesity | II | Functional efficacy demonstrated in diet-induced models | Lack of mechanistic investigation of metabolic signaling pathways |
| Bombax ceiba | Antibacterial | III | Reproducible in vitro inhibition observed | In vivo infection models and toxicity evaluation not reported |
| Bombax ceiba | Anti-inflammatory | I | Demonstrated activity in validated inflammatory models | Deeper cytokine profiling and pathway-level analyses are warranted |
| Bombax ceiba | Antiarthritic | II | Functional improvement reported in induced arthritis models | Limited molecular mediator assessment |
| Bombax ceiba | Nephroprotective | I | Biochemical and functional renal protection observed | Mechanistic renal signaling pathways remain underexplored |
| Bombax ceiba | Hepatoprotective | I | Activity supported by biochemical and histological parameters | Further molecular-level validation desirable |
| Bombax ceiba | Anticancer / Cytotoxic | III | Cytotoxic potential demonstrated in tumor cell lines | Selectivity in normal cells and in vivo tumor validation lacking |
| Bombax ceiba | Anti-helminthic | IV | Preliminary biological activity observed | Requires in vivo confirmation and pharmacodynamic assessment |
| Bombax ceiba | Antihyperglycemic | I | Consistent metabolic improvements reported | Direct causative linkage with identified metabolites requires clarification |
| Bombax ceiba | Antihyperlipidemic | I | Classical lipid biomarkers modulated | Gene-level pathway investigation limited |
| Bombax ceiba | Antiglycation | III | In vitro AGE inhibition demonstrated | Physiological validation absent |
| Bombax ceiba | Gastrointestinal | II | Functional improvement observed in vivo | Mechanistic basis remains insufficiently explored |
| Bombax ceiba | Anti-hemorrhagic | IV | Activity suggested in cellular systems | Systemic hemostatic validation required |
| Bombax ceiba | Osteogenic / Antiosteoporotic | I | Bone density improvements suggest translational relevance | Molecular osteogenic signaling requires further investigation |
| Bombax ceiba | Gastroprotective / Antiulcerogenic activity | I | Robust dual-model preclinical design; integrated metabolomics + network pharmacology | No clinical data; small sample (n=6); mechanism not experimentally validated; no PK or biomarker analysis |
| Bombax costatum | Anti-inflammatory / Antiarthritic | II | Functional in vivo evidence reported | Mediator-level confirmation limited |
| Bombax costatum | Antihistaminic | II | Activity supported in pharmacological models | Receptor-level validation not reported |
| Bombax costatum | Antidepressant | II | Behavioral improvements observed | Neurochemical biomarker evaluation limited |
| Bombax costatum | Antiamnesic | II | Cognitive benefits demonstrated | Synaptic and neuroplasticity markers not investigated |
| Bombax buonopozense | Antiplasmodial | II | Activity demonstrated in relevant infectious model | Expanded replication and molecular target characterization desirable |
| Species | Biological Activity | Evidence Level | Critical Appraisal | Major Methodological Gaps |
|---|---|---|---|---|
| Pseudobombax parvifolium | Antioxidant | I | In vivo antioxidant modulation supported by reduced lipid peroxidation and increased SOD and GPx activity | Absence of pathway-level redox signaling analysis; lack of standardized extract characterization |
| Pseudobombax ellipticum | Antioxidant | III | Consistent radical scavenging and metal chelation activity in chemical assays | No in vivo confirmation; limited mechanistic elucidation of cellular antioxidant pathways |
| Pseudobombax ellipticum | Antibacterial | III | Demonstrated inhibition of biofilm formation against Pseudomonas aeruginosa | Lack of in vivo infection models; absence of toxicity and pharmacokinetic evaluation |
| Pseudobombax ellipticum | Antisickling | III | Reduction of erythrocyte sickling observed in vitro, suggesting hematological relevance | No in vivo validation; mechanism of hemoglobin stabilization not investigated |
| Pseudobombax ellipticum (cv. alba) | Antioxidant | III | Reproducible antioxidant activity in chemical assays | Limited comparative phytochemical profiling; no biological validation beyond in vitro assays |
| Pseudobombax ellipticum (cv. alba) | Antisickling | III | In vitro erythrocyte stabilization demonstrated | Translational hematological assessment and systemic validation lacking |
| Pseudobombax simplicifolium | Antioxidant | IV | Multiple complementary antioxidant assays; phytochemical screening and phenolic/flavonoid quantification; exclusively in vitro | No in vivo/clinical validation; no cytotoxicity or dermatological safety tests; no photostability/formulation studies; no compound isolation or mechanistic assays |
| Pseudobombax marginatum | Anti-inflammatory | II | Functional reduction of carrageenan-induced edema in vivo | Limited cytokine profiling; absence of molecular inflammatory pathway analysis |
| Pseudobombax marginatum | Antinociceptive | II | Significant reduction in nociceptive responses in validated rodent models | Mechanistic differentiation between central and peripheral pathways not performed |
| Pseudobombax marginatum | Cytoprotective / Genoprotective | III | DNA damage reduction demonstrated in comet assay | No systemic confirmation; responsible bioactive compounds not fully isolated |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




