1. Introduction
Rheumatoid arthritis (RA) is a chronic, systemic autoimmune disease characterized by persistent synovial inflammation that may lead to progressive joint destruction, functional impairment, and reduced quality of life if inadequately controlled [
1]. Over the past two decades, the implementation of treat-to-target strategies has substantially improved clinical outcomes [
2]. In parallel, the expansion of available therapies—including conventional synthetic disease-modifying antirheumatic drugs (csDMARDs), biologic DMARDs (bDMARDs), and targeted synthetic DMARDs (tsDMARDs)—has further enhanced disease control and long-term prognosis [
3]. Nevertheless, therapeutic response remains heterogeneous [
4], and a considerable proportion of patients require treatment escalation or switching due to inadequate response or loss of efficacy [
5].
Serological biomarkers play a central role in RA diagnosis and prognostication [
6]. Rheumatoid factor (RF) and anti–cyclic citrullinated peptide (anti-CCP) antibodies are well-established markers associated with disease classification and structural progression [
7]. However, these antibodies do not fully explain variability in treatment response or the need for advanced therapies [
8]. Antinuclear antibodies (ANA), although traditionally associated with other systemic autoimmune rheumatic diseases, are also detected in a substantial subset of patients with RA [
9], with reported prevalence ranging from 15% to over 30% depending on the population studied and laboratory methodology [
10].
The clinical significance of ANA in RA remains incompletely understood [
9]. Some studies have suggested associations between ANA positivity and higher inflammatory burden, broader autoantibody repertoires, extra-articular manifestations, or features of polyautoimmunity [
11]. Emerging immunologic data further indicate that subsets of RA characterized by distinct molecular signatures—such as enhanced type I interferon pathway activation—may exhibit differential clinical trajectories and therapeutic responses [
12]. These observations raise the possibility that ANA positivity could reflect a distinct immunophenotype with implications for treatment outcomes [
13].
Despite these considerations, most prior investigations have focused on cross-sectional associations between ANA and disease activity or correlations with classical serological markers [
14]. Data examining the longitudinal relationship between ANA status and transition to advanced therapies remain limited [
15]. In particular, whether ANA positivity independently predicts earlier initiation of biologic treatment—beyond established prognostic markers such as RF and anti-CCP—has not been clearly established [
16]. Furthermore, the potential clinical relevance of ANA titer levels, rather than simple binary positivity, has been insufficiently explored [
17].
In the context of precision medicine and treat-to-target management, identifying biomarkers associated with treatment escalation is of practical importance. Early recognition of patients at higher risk of inadequate response to csDMARDs may facilitate timely therapeutic adjustment and minimize prolonged exposure to uncontrolled inflammation [
17].
Accordingly, the present study was designed to evaluate the association between ANA status and treatment trajectories in a well-characterized RA cohort. Specifically, we aimed to (1) compare baseline clinical characteristics according to ANA status, (2) assess the relationship between ANA positivity and time to biologic therapy initiation, adjusting for conventional serological markers, and (3) explore whether ANA titer levels are associated with differential treatment patterns. By addressing these questions, we sought to clarify the potential role of ANA as an adjunct prognostic biomarker in RA management.
2. Materials and Methods
Study Design and Ethical Approval
This retrospective cohort study was conducted at the Department of Rheumatology, Dicle University Faculty of Medicine. The study was performed in accordance with the ethical principles of the Declaration of Helsinki and was approved by the Dicle University Medical Faculty Ethics Committee for Non-Interventional Studies (Approval No: 62; February 4, 2026). Given the retrospective design and the use of anonymized clinical records, the requirement for written informed consent was waived.
Study Population
We screened adult patients (≥18 years) diagnosed with rheumatoid arthritis (RA) between January 2020 and December 2025. RA diagnosis was established according to the 2010 American College of Rheumatology/European Alliance of Associations for Rheumatology (ACR/EULAR) classification criteria [
18]. Eligible patients were required to have documented antinuclear antibody (ANA) testing performed at the time of diagnosis and a minimum follow-up duration of 12 months. Patients were excluded if they had overlapping systemic autoimmune rheumatic diseases, including systemic lupus erythematosus, Sjögren’s syndrome, systemic sclerosis, or inflammatory myositis, in order to ensure that outcomes were attributable specifically to RA. Additional exclusion criteria included active malignancy at diagnosis or during follow-up, as well as incomplete baseline serological or longitudinal treatment data.
Data Collection and Baseline Variables
Demographic, clinical, and laboratory data were systematically retrieved from electronic medical records and standardized rheumatology follow-up forms. Baseline variables were defined at the time of RA diagnosis (time zero). Demographic characteristics included age, sex, body mass index (BMI), and smoking status. Clinical variables comprised symptom duration prior to diagnosis, pattern of joint involvement, presence of extra-articular manifestations, and comorbidities such as hypertension, diabetes mellitus, and cardiovascular disease. Disease activity was assessed using the Disease Activity Score in 28 joints (DAS28). Systemic inflammation was evaluated through erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) levels.
Serological Assessment
ANA status was determined at diagnosis using indirect immunofluorescence (IIF) on HEp-2 cells in accordance with international consensus recommendations. A titer of ≥1:80 was considered positive. To assess the potential impact of antibody concentration, ANA-positive patients were stratified into low–moderate titer (1:80–1:320) and high-titer (>1:320) subgroups. Rheumatoid factor (RF) and anti–cyclic citrullinated peptide (anti-CCP) antibodies were measured at diagnosis using standard laboratory techniques, including nephelometry and enzyme-linked immunosorbent assay (ELISA). All serological assessments were performed prior to the initiation of biologic or targeted synthetic therapies.
Treatment Exposure and Outcome Definition
Longitudinal treatment data were collected throughout the follow-up period and included the use of conventional synthetic DMARDs (csDMARDs), documentation of treatment failure, initiation of biologic DMARDs (bDMARDs), and initiation of targeted synthetic DMARDs (tsDMARDs). Biologic switching was defined as transition to a second or subsequent biologic agent during follow-up. Corticosteroid exposure was recorded as baseline dose at diagnosis, mean daily dose during follow-up, and cumulative dose.
The primary outcome was time to initiation of biologic or targeted synthetic DMARD therapy. Time zero was defined as the date of RA diagnosis. Time-to-event was calculated from diagnosis to the date of first biologic or targeted synthetic DMARD initiation. Patients who did not initiate advanced therapy during follow-up were censored at the date of their last documented clinical visit.
Statistical Analysis
All statistical analyses were performed using SPSS Statistics version 27.0 (IBM Corp., Armonk, NY, USA). Continuous variables were expressed as mean ± standard deviation (SD) for normally distributed data or median with interquartile range (IQR) for non-normally distributed data, while categorical variables were presented as counts and percentages. Normality was assessed using the Shapiro–Wilk or Kolmogorov–Smirnov test, as appropriate. Between-group comparisons were conducted using Student’s t-test or Mann–Whitney U test for continuous variables and Chi-square or Fisher’s exact test for categorical variables. All statistical tests were two-sided, and a p-value <0.05 was considered statistically significant.
Time to biologic initiation was evaluated using Kaplan–Meier survival analysis, and differences between groups were assessed using the log-rank test. Univariate Cox proportional hazards regression analyses were performed to identify potential predictors of biologic initiation. Variables with p < 0.10 in univariate analysis, along with clinically relevant covariates (age, sex, symptom duration, RF positivity, and anti-CCP positivity), were entered into the multivariate Cox regression model. Hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated. The proportional hazards assumption was evaluated using Schoenfeld residuals and graphical inspection of log-minus-log survival plots, and no significant violations were detected.
An exploratory subgroup analysis was conducted within the ANA-positive cohort to evaluate differences in time to biologic initiation according to ANA titer category (low–moderate versus high). Given the limited sample size of the high-titer subgroup, these analyses were considered hypothesis-generating.
The proportion of missing data was below 10% for all variables included in the analyses. Given the low degree of missingness, pairwise deletion was applied. Although no a priori sample size calculation was performed due to the retrospective design, post hoc assessment demonstrated adequate statistical power. Based on the observed event rate and hazard ratio estimates, the study had greater than 80% power to detect a hazard ratio of approximately 2.0 at a two-sided alpha level of 0.05. Additionally, the number of biologic initiation events exceeded the recommended threshold of 10 events per variable included in the multivariate Cox model, supporting the stability and reliability of the regression estimates.
3. Results
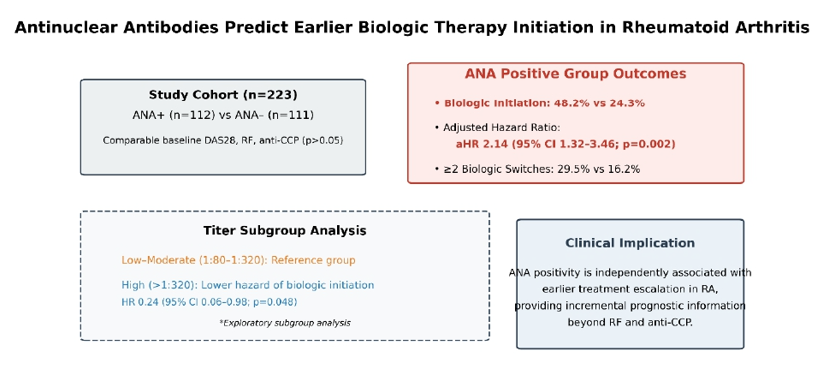
The final analysis included 223 patients, of whom 112 were ANA-positive and 111 were ANA-negative. Baseline demographic and clinical characteristics are summarized in
Table 1. Mean age was 54.2 ± 11.4 years in the ANA-positive group and 53.8 ± 12.1 years in the ANA-negative group (p = 0.78). Female predominance was 83.9% versus 80.1% (p = 0.45), and body mass index was similar across groups. Smoking status and major comorbidities, including hypertension, diabetes mellitus, and cardiovascular disease, did not differ significantly. Median disease duration was 114 months in ANA-positive patients and 108 months in ANA-negative patients (p = 0.61). Serological profiles, including RF and anti-CCP positivity, were also comparable (RF: 73.2% vs. 70.3%, p = 0.62; anti-CCP: 68.8% vs. 66.7%, p = 0.71). Baseline inflammatory markers (ESR, CRP) and disease activity scores (DAS28) did not significantly differ between groups, indicating homogeneous baseline profiles (
Table 1).
Distinct patterns in treatment escalation were observed according to ANA status (
Table 2). ANA-positive patients were significantly more likely to initiate biologic therapy compared with ANA-negative patients (48.2% vs. 24.3%; p < 0.001). The median number of biologic agents used during follow-up was higher in ANA-positive patients (1.0 vs. 0.0; p = 0.004), and they were more likely to experience multiple biologic switches (≥2 agents) (29.5% vs. 16.2%; p = 0.028).
Differences were also noted in the choice of first-line biologic agents (p = 0.026). TNF inhibitors were the most frequently prescribed in both groups, while IL-6 inhibitors were more commonly used in the ANA-negative cohort (25.9% vs. 3.7%). Corticosteroid-related parameters—including mean daily dose, cumulative dose, and total duration—were similar between groups. Conventional synthetic DMARD use and failure rates did not differ significantly.
Kaplan–Meier survival analysis demonstrated a significantly shorter time to biologic initiation in ANA-positive patients compared with ANA-negative patients (log-rank p < 0.001;
Figure 1). In univariate Cox regression, ANA positivity, longer disease duration, and lower baseline corticosteroid dose were associated with earlier biologic initiation.
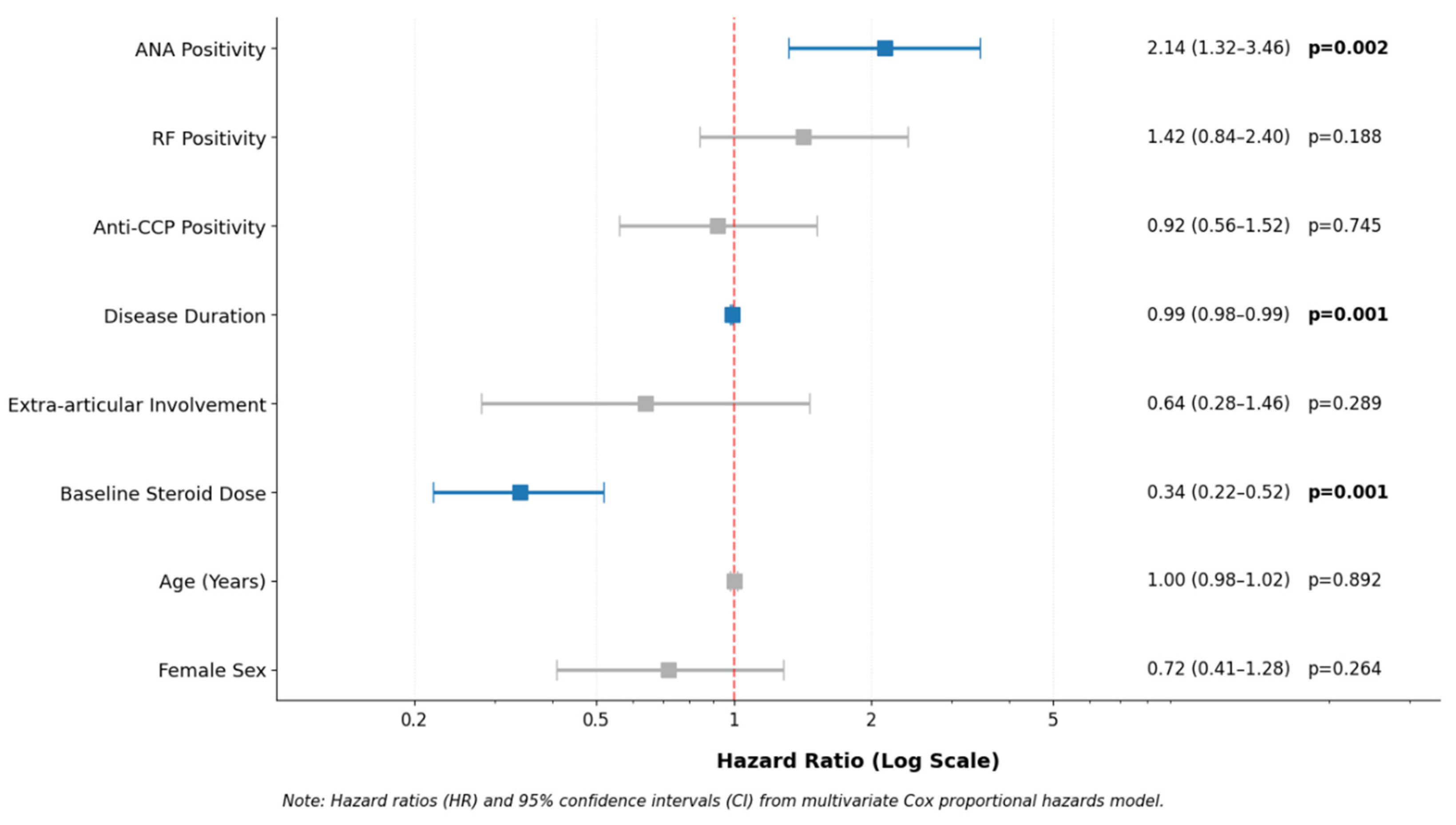
Multivariate Cox proportional hazards analysis identified ANA positivity as an independent predictor of earlier biologic therapy (adjusted HR: 2.14; 95% CI: 1.32–3.46; p = 0.002) after controlling for age, sex, disease duration, RF, and anti-CCP status (
Table 3,
Figure 2). RF and anti-CCP positivity were not independently predictive (p = 0.188 and p = 0.457, respectively). Additional independent predictors included longer disease duration and lower baseline corticosteroid dose (both p < 0.001).
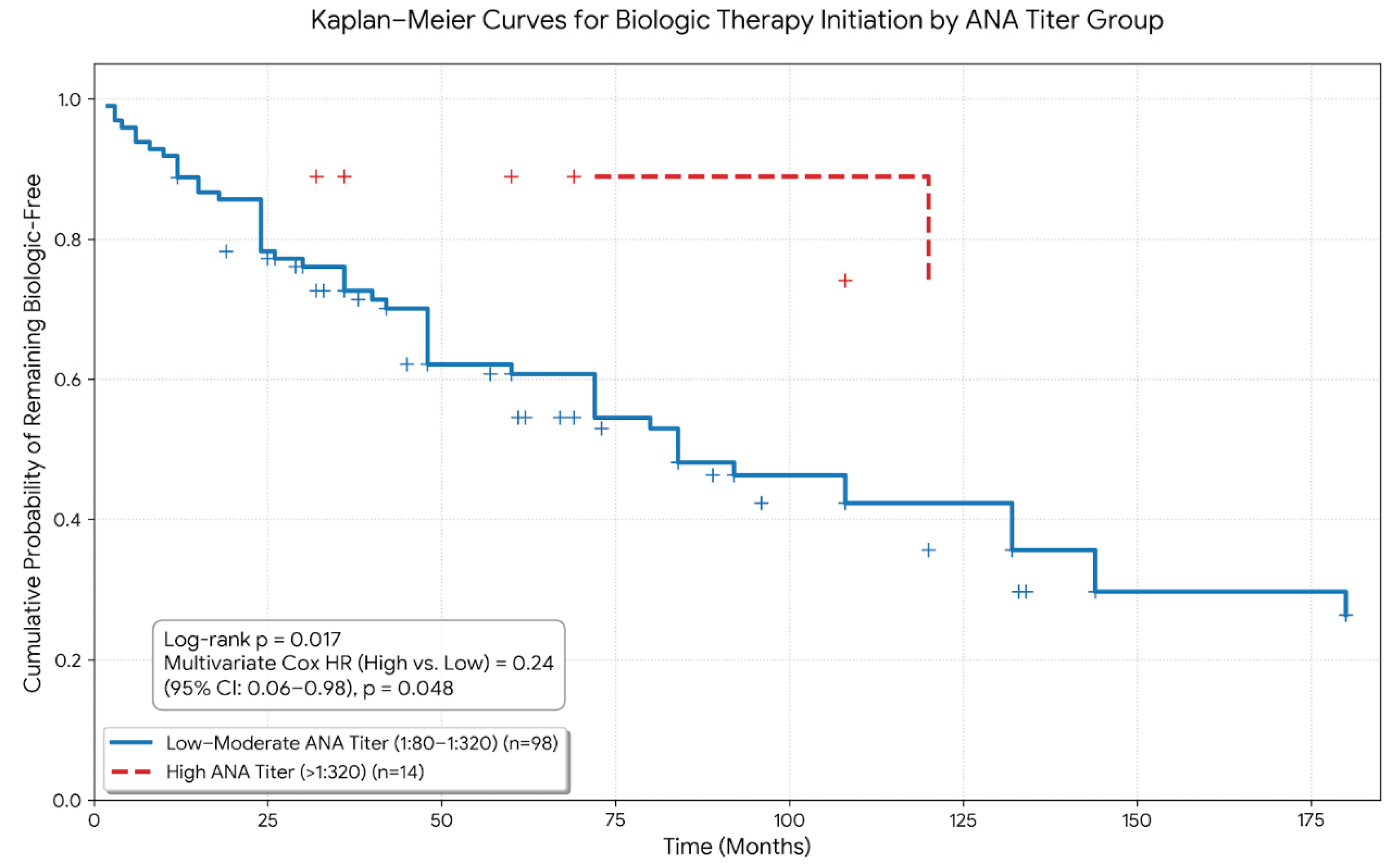
Within the ANA-positive cohort, 98 patients had low–moderate titers (1:80–1:320) and 14 patients had high titers (>1:320) (Supplementary
Table 1). Baseline demographic and clinical characteristics were similar between titer subgroups. Kaplan–Meier survival analysis revealed a significant difference in biologic-free survival according to ANA titer (log-rank p = 0.017;
Figure 1).
Interestingly, multivariate Cox regression indicated that high ANA titers were associated with a lower hazard of initiating biologic therapy compared with low–moderate titers (HR: 0.24; 95% CI: 0.06–0.98; p = 0.048), illustrating a non-linear “titer paradox” in which higher ANA concentrations did not necessarily correlate with more aggressive or synovial-dominant disease.
4. Discussion
In this retrospective cohort of patients with rheumatoid arthritis (RA), antinuclear antibody (ANA) positivity independently predicted earlier initiation of biologic therapy after adjustment for conventional serological markers and relevant clinical covariates. By defining time zero as the date of RA diagnosis and applying Cox proportional hazards modeling, we demonstrated that ANA-positive patients underwent more rapid escalation to biologic therapy. This observation suggests that ANA status may reflect immune processes associated with reduced responsiveness to conventional synthetic DMARDs, rather than being a mere epiphenomenon of autoimmunity.
Previous epidemiological studies have reported ANA positivity in RA cohorts with prevalence estimates ranging from 15% to 30%, though the clinical implications have remained inconsistent [
9,
15]. Our findings align with recent real-world registry data indicating that ANA-positive RA patients, especially those exhibiting triple seropositivity (RF+/ACPA+/ANA+), demonstrate higher disease activity indices and reduced likelihood of sustained remission [
19]. These patterns persisted even after accounting for RF and anti-CCP status, suggesting that ANA contributes independent prognostic information beyond classical markers.
While RF and anti-CCP antibodies are well-established predictors of structural progression, they did not independently predict time to biologic initiation in our adjusted models. Prior work has similarly shown that seropositivity alone does not uniformly stratify risk for treatment escalation, particularly for targeted therapies; rather, a broader autoantibody repertoire correlates more strongly with refractory disease patterns [
20,
21]. This may reflect immunological heterogeneity within RA, whereby conventional serological profiles inadequately capture complex B-cell activation and epitope spreading phenomena increasingly recognized in chronic disease evolution [
22,
23].
A particularly striking and novel finding in our study was the non-linear association between ANA titer and biologic initiation. Although ANA positivity overall predicted earlier escalation, high ANA titers (>1:320) were paradoxically associated with a lower hazard of biologic initiation compared with low–moderate titers. This “titer paradox” should be interpreted cautiously given the small size of the high-titer subgroup and borderline statistical significance. Nonetheless, similar non-linear autoantibody effects have been suggested in recent immunophenotyping studies, where high ANA titers correlated more with systemic or interferon-enriched immunological signatures than with synovial inflammation per se [
27,
28].
Mounting evidence highlights distinct molecular endotypes within RA, including type I interferon–dominant subgroups characterized by unique transcriptional profiles and distinct cytokine milieus. Such interferon-high RA endotypes may exhibit differential responses to biologic therapy, as opposed to TNF-driven inflammation where TNF inhibitors are more effective [
29,
30]. It is plausible that very high ANA concentrations identify such interferon-enriched immunophenotypes, which may initially respond differently to conventional DMARDs and biologics.
Our observation associating lower baseline corticosteroid dose with earlier biologic initiation may reflect evolving clinical practices favoring steroid-sparing strategies or more aggressive treat-to-target approaches rather than a direct biological effect. In line with recent European Alliance of Associations for Rheumatology (EULAR) recommendations advocating minimal long-term glucocorticoid use, clinicians may prefer early biologic intervention in patients with suboptimal inflammation control [
31,
32]; however, residual confounding cannot be excluded.
Differences in first-line biologic selection between ANA groups were also observed. These variations likely reflect clinician preference, temporal prescribing trends, or unmeasured clinical characteristics rather than a direct mechanistic effect of ANA itself, particularly in the absence of randomized allocation.
This study has several limitations. The retrospective, single-center design introduces potential selection bias and limits generalizability. Despite verification of proportional hazards assumptions, residual confounding cannot be entirely excluded. The exploratory titer analysis — especially in the high-titer subgroup — warrants validation in larger, multicenter cohorts with comprehensive immunophenotyping. Nonetheless, the study’s strengths include a robust time-to-event framework, extended follow-up, systematic ANA titer stratification, and multivariate adjustment for established prognostic factors.
Future research should prioritize mechanistic studies investigating the immunological basis of ANA-associated treatment refractoriness, particularly focusing on type I interferon pathways, B-cell regulation, and autoantibody epitope expansion. Prospective biomarker-driven clinical trials are needed to determine whether integrating ANA status — alone or in combination with multi-biomarker disease activity scores and genomic risk profiles — can improve patient stratification and long-term outcomes. Such integrative approaches may advance precision medicine strategies in RA
5. Conclusions
In summary, ANA positivity serves as an independent predictor of earlier and more intensive escalation to biologic therapy in patients with rheumatoid arthritis. This prognostic value appears to extend beyond traditional serological markers such as RF and anti-CCP antibodies, highlighting the potential utility of ANA testing in informing individualized treatment strategies. Notably, the observed “titer paradox,” wherein high ANA levels were associated with delayed treatment escalation, underscores the complex, non-linear relationship between antibody concentration and therapy intensification, and warrants further mechanistic investigation. Collectively, these findings support the integration of comprehensive autoantibody profiling, including ANA status and titer, into rheumatoid arthritis management strategies to optimize early risk stratification and personalized therapeutic decision-making.
Supplementary Materials
Supplementary Table 1 presents baseline characteristics of ANA-positive patients stratified by titer level. Comparisons were conducted using non-parametric and exact tests due to the small size of the high-titer subgroup (n = 14). This exploratory analysis should be interpreted cautiously.
Author Contributions
Conceptualization, Z.A.A., and D.Y.; methodology, R.Ç.; software, M.Ç.; validation, Z.I.S, .M.K., formal analysis, R.Ç.; investigation, S.K.; resources, İ.B.; data curation, S.E.; writing—original draft preparation, Z.A.A; writing—review and editing, Z.A.A; visualization, P.O.; supervision, R.Ç.; project administration, M.K.; funding acquisition, M.Ç. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was performed in accordance with the ethical principles of the Declaration of Helsinki and was approved by the Dicle University Medical Faculty Ethics Committee for Non-Interventional Studies (Approval No: 62; February 4, 2026). Given the retrospective design and the use of anonymized clinical records, the requirement for written informed consent was waived.
Informed Consent Statement
Given the retrospective design and the use of anonymized clinical records, the requirement for written informed consent was waived.
Data Availability Statement
Data are available from the corresponding author upon reasonable request, and will be shared in a manner that ensures the confidentiality of the data.
Acknowledgments
The authors would like to thank Dicle University Scientific Research Projects Unit (DUBAP) for their support.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
| ACR/EULAR |
American College of Rheumatology / European Alliance of Associations for Rheumatology |
| ANA |
Antinuclear Antibody |
| bDMARD |
Biologic Disease-Modifying Antirheumatic Drug |
| BMI |
Body Mass Index |
| CRP |
C-Reactive Protein |
| csDMARD |
tsDMARD |
| DAS28 |
Disease Activity Score in 28 joints |
| DMARD |
Disease-Modifying Antirheumatic Drug |
| ESR |
Erythrocyte Sedimentation Rate |
| IIF |
Indirect Immunofluorescence |
| IL-6 |
Interleukin-6 |
| JAK |
Janus Kinase |
| RF |
Rheumatoid Factor |
| RA |
Rheumatoid Arthritis |
| tsDMARD |
Targeted Synthetic Disease-Modifying Antirheumatic Drug
|
References
- Scott DL, Wolfe F, Huizinga TW. Rheumatoid arthritis. Lancet. 2010 Sep 25;376(9746):1094-108. doi: 10.1016/S0140-6736(10)60826-4.
- Salomon-Escoto K, Kay J. The “Treat to Target” Approach to Rheumatoid Arthritis. Rheum Dis Clin North Am. 2019 Nov;45(4):487-504. doi: 10.1016/j.rdc.2019.06.001.
- Kerschbaumer A, Sepriano A, Bergstra SA, Smolen JS, van der Heijde D, Caporali R, Edwards CJ, Verschueren P, de Souza S, Pope JE, Takeuchi T, Hyrich KL, Winthrop KL, Aletaha D, Stamm TA, Schoones JW, Landewé RBM. Efficacy of synthetic and biological DMARDs: a systematic literature review informing the 2022 update of the EULAR recommendations for the management of rheumatoid arthritis. Ann Rheum Dis. 2023 Jan;82(1):95-106. doi: 10.1136/ard-2022-223365.
- Ibrahim F, Scott IC, Scott DL, Ayis SA. Heterogeneity of treatment responses in rheumatoid arthritis using group based trajectory models: secondary analysis of clinical trial data. BMC Rheumatol. 2023 Sep 25;7(1):33. doi: 10.1186/s41927-023-00348-5.
- Taylor PC, Matucci Cerinic M, Alten R, Avouac J, Westhovens R. Managing inadequate response to initial anti-TNF therapy in rheumatoid arthritis: optimising treatment outcomes. Ther Adv Musculoskelet Dis. 2022 Aug 16;14:1759720X221114101. doi: 10.1177/1759720X221114101.
- Matuszewska A, Madej M, Wiland P. Markery immunologiczne reumatoidalnego zapalenia stawów [Immunological markers of rheumatoid arthritis]. Postepy Hig Med Dosw (Online). 2016 Mar 25;70:251-7. Polish. doi: 10.5604/17322693.1198270.
- Taylor P, Gartemann J, Hsieh J, Creeden J. A systematic review of serum biomarkers anti-cyclic citrullinated Peptide and rheumatoid factor as tests for rheumatoid arthritis. Autoimmune Dis. 2011;2011:815038. doi: 10.4061/2011/815038.
- Zhao SS, Moots RJ. Biomarkers for Treatment Response in Rheumatoid Arthritis: Where are they? Rheumatol Immunol Res. 2020 Dec 1;1(1):1-3. doi: 10.2478/rir-2020-0008.
- Nakano K, Fujita S, Hiramatsu-Asano S, Nagasu A, Tsuji S, Koide Y, Yamada M, Mizuta Y, Ikeda M, Hirano H, Morita Y. Clinical Significance of Antinuclear Antibodies in Patients with Rheumatoid Arthritis: From SETOUCHI-RA Registry. J Clin Med. 2025 Feb 26;14(5):1553. doi: 10.3390/jcm14051553.
- Nishimura S, Nishiya K, Hisakawa N, Chikazawa H, Ookubo S, Nakatani K, Hashimoto K. Positivity for antinuclear antibody in patients with advanced rheumatoid arthritis. Acta Med Okayama. 1996 Oct;50(5):261-5. doi: 10.18926/AMO/30501.
- Liu F, Wang XQ, Zou JW, Li M, Pan CC, Si YQ. Association between serum antinuclear antibody and rheumatoid arthritis. Front Immunol. 2024 Apr 22;15:1358114. doi: 10.3389/fimmu.2024.1358114.
- Castañeda-Delgado JE, Bastián-Hernandez Y, Macias-Segura N, Santiago-Algarra D, Castillo-Ortiz JD, Alemán-Navarro AL, Martínez-Tejada P, Enciso-Moreno L, Garcia-De Lira Y, Olguín-Calderón D, Trouw LA, Ramos-Remus C, Enciso-Moreno JA. Type I Interferon Gene Response Is Increased in Early and Established Rheumatoid Arthritis and Correlates with Autoantibody Production. Front Immunol. 2017 Mar 20;8:285. doi: 10.3389/fimmu.2017.00285.
- Wither J, Johnson SR, Liu T, Noamani B, Bonilla D, Lisnevskaia L, Silverman E, Bookman A, Landolt-Marticorena C. Presence of an interferon signature in individuals who are anti-nuclear antibody positive lacking a systemic autoimmune rheumatic disease diagnosis. Arthritis Res Ther. 2017 Feb 28;19(1):41. doi: 10.1186/s13075-017-1243-y.
- Phillips EA, Moehling TJ, Ejendal KFK, Hoilett OS, Byers KM, Basing LA, Jankowski LA, Bennett JB, Lin LK, Stanciu LA, Linnes JC. Microfluidic rapid and autonomous analytical device (microRAAD) to detect HIV from whole blood samples. Lab Chip. 2019 Oct 9;19(20):3375-3386. doi: 10.1039/c9lc00506d.
- Paknikar SS, Crowson CS, Davis JM, Thanarajasingam U. Exploring the Role of Antinuclear Antibody Positivity in the Diagnosis, Treatment, and Health Outcomes of Patients With Rheumatoid Arthritis. ACR Open Rheumatol. 2021 Jun;3(6):422-426. doi: 10.1002/acr2.11271.
- Anaparti V, Smolik I, Meng X, O’Neil L, Jantz MA, Fritzler MJ, El-Gabalawy H. Expansion of Alternative Autoantibodies Does Not Follow the Evolution of Anti-Citrullinated Protein Antibodies in Preclinical Rheumatoid Arthritis: An Analysis in At-Risk First Degree Relatives. Arthritis Rheumatol. 2021 May;73(5):740-749. doi: 10.1002/art.41675.
- Sahin D, Di Matteo A, Emery P. Biomarkers in the diagnosis, prognosis and management of rheumatoid arthritis: A comprehensive review. Annals of Clinical Biochemistry: International Journal of Laboratory Medicine. 2024;62(1):3-21. doi:10.1177/00045632241285843.
- Kay J, Upchurch KS. ACR/EULAR 2010 rheumatoid arthritis classification criteria. Rheumatology (Oxford). 2012 Dec;51 Suppl 6:vi5-9. doi: 10.1093/rheumatology/kes279.
- Maldonado-Cañón K, Coral-Alvarado P, Méndez-Patarroyo P, Bautista-Molano W, Quintana-López G. ANAs and triple positivity effect on disease activity and sustained remission in rheumatoid arthritis: a retrospective real-world machine learning approach. Clin Rheumatol. 2025 Dec;44(12):4895-4907. doi: 10.1007/s10067-025-07683-7.
- Courvoisier DS, Chatzidionysiou K, Mongin D, Lauper K, Mariette X, Morel J, Gottenberg JE, Bergstra SA, Suarez MP, Codreanu C, Kvien TK, Santos MJ, Pavelka K, Hetland ML, Askling J, Turesson C, Kubo S, Tanaka Y, Iannone F, Choquette D, Nordström DC, Rotar Z, Lukina G, Gabay C, Van Vollenhoven R, Finckh A. The impact of seropositivity on the effectiveness of biologic anti-rheumatic agents: results from a collaboration of 16 registries. Rheumatology (Oxford). 2021 Feb 1;60(2):820-828. doi: 10.1093/rheumatology/keaa393.
- Mikuls, T., Thiele, G., Curtis, J., Robinson, W., Pappas, D., Kremer, J., Hunter, C., Duryee, M., Sayles, H., England, B., Dougherty, J., & Petro, A. (2021). Associations between an expanded autoantibody profile and treatment responses to biologic therapies in patients with rheumatoid arthritis. International Immunopharmacology, 91, 107260. [CrossRef]
- Sokolova MV, Schett G, Steffen U. Autoantibodies in Rheumatoid Arthritis: Historical Background and Novel Findings. Clin Rev Allergy Immunol. 2022 Oct;63(2):138-151. doi: 10.1007/s12016-021-08890-1.
- Guo, Q., Wang, Y., Xu, D. et al. Rheumatoid arthritis: pathological mechanisms and modern pharmacologic therapies. Bone Res 6, 15 (2018). [CrossRef]
- Ishikawa Y, Hashimoto M, Ito H, Tanaka M, Yukawa N, Fujii T, Yamamoto W, Mimori T, Terao C. Anti-nuclear antibody development is associated with poor treatment response to biological disease-modifying anti-rheumatic drugs in patients with rheumatoid arthritis. Semin Arthritis Rheum. 2019 Oct;49(2):204-210. doi: 10.1016/j.semarthrit.2019.02.003.
- Martins A, Oliveira D, Martins FR, Rato MS, Pinheiro FO, Fonseca D, Garcia S, Fernandes BM, Pimenta S, Vaz C, Costa L, Bernardes M. The impact of antinuclear antibodies seroconversion induced by anti-tumor necrosis factor α agents on the clinical outcomes in rheumatic patients. ARP Rheumatol. 2023 Apr 12. English. Epub ahead of print. PMID: 37178156.
- Atzeni F, Turiel M, Capsoni F, Doria A, Meroni P, Sarzi-Puttini P. Autoimmunity and anti-TNF-alpha agents. Ann N Y Acad Sci. 2005 Jun;1051:559-69. doi: 10.1196/annals.1361.100.
- Rodríguez-Carrio J, Burska A, Conaghan PG, Dik WA, Biesen R, Eloranta ML, Cavalli G, Visser M, Boumpas DT, Bertsias G, Wahren-Herlenius M, Rehwinkel J, Frémond ML, Crow MK, Ronnblom L, Vital E, Versnel M. Association between type I interferon pathway activation and clinical outcomes in rheumatic and musculoskeletal diseases: a systematic literature review informing EULAR points to consider. RMD Open. 2023 Mar;9(1):e002864. doi: 10.1136/rmdopen-2022-002864.
- Rodríguez-Carrio J, Burska A, Conaghan PG, Dik WA, Biesen R, Eloranta ML, Cavalli G, Visser M, Boumpas DT, Bertsias G, Wahren-Herlenius M, Rehwinkel J, Frémond ML, Crow MK, Rönnblom L, Versnel MA, Vital EM. 2022 EULAR points to consider for the measurement, reporting and application of IFN-I pathway activation assays in clinical research and practice. Ann Rheum Dis. 2023 Jun;82(6):754-762. doi: 10.1136/ard-2022-223628.
- van der Pouw Kraan TC, Wijbrandts CA, van Baarsen LG, Voskuyl AE, Rustenburg F, Baggen JM, Ibrahim SM, Fero M, Dijkmans BA, Tak PP, Verweij CL. Rheumatoid arthritis subtypes identified by genomic profiling of peripheral blood cells: assignment of a type I interferon signature in a subpopulation of patients. Ann Rheum Dis. 2007 Aug;66(8):1008-14. doi: 10.1136/ard.2006.063412.
- de Jong, T.D., Lübbers, J., Turk, S. et al. The type I interferon signature in leukocyte subsets from peripheral blood of patients with early arthritis: a major contribution by granulocytes. Arthritis Res Ther 18, 165 (2016). [CrossRef]
- Misra DP, Agarwal V, Sharma A, Wakhlu A, Negi VS. 2016 update of the EULAR recommendations for the management of rheumatoid arthritis: a utopia beyond patients in low/middle income countries? Ann Rheum Dis. 2017 Nov;76(11):e47. doi: 10.1136/annrheumdis-2017-211446.
- Smolen JS, Landewé RBM, Bergstra SA, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2022 update. Ann Rheum Dis. 2023 Jan;82(1):3-18. doi: 10.1136/ard-2022-223356. Epub 2022 Nov 10. Erratum in: Ann Rheum Dis. 2023 Mar;82(3):e76. doi: 10.1136/ard-2022-223356corr1.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).