1. Introduction
Chili pepper (
Capsicum annuum L.) transcends its horticultural classification to become consolidated as a product of global strategic importance, with significant economic impact and deep cultural rootedness [
1,
2]. Belonging to the Solanaceae family,
C. annuum is the most cultivated and traded species globally, characterized by a notable genetic diversity that confers it essential phenotypical plasticity for its adaptation to diverse edaphoclimatic conditions [
3]. This adaptive capacity positions it as a pillar for the food security and economy of many regions, with a global production that consistently exceeds 44 million annual tons [
4].
The value of chili pepper extends beyond its dietary use and possesses a rich biochemical matrix in bioactive compounds with nutraceutical, functional and therapeutic properties [
5,
6]. Among these compounds, capsaicinoids, carotenoids, flavonoids, polyphenols and vitamins stand out, which act as strong antioxidants [
7,
8,
9]. These traits have driven its use in the pharmaceutical, cosmetic and functional foods industries [
10,
11], generating intense research directed at agronomic manipulation to potentiate these secondary metabolites.
Productive optimization requires farming strategies that maximize yield and uniformity in post-harvest quality [
12,
13,
14], and includes advances in genomics, genetic improvement, and fundamentally, the development of production systems [
15,
16,
17,
18]. Traditionally, chili pepper production has been based on open field cultivation; although it is an accessible system, it presents significant limitations due to its vulnerability to environmental fluctuations, exposure to extreme climatic events, and variability in temperature and moisture, which result in limited efficiency of resources, low yields, and inconsistent quality [
19,
20].
As a response to these challenges, protected agriculture, particularly the use of greenhouses, has transformed the productive outlook of chili pepper cultivation [
21,
22]. These systems allow the modification and control of the microclimate, making it possible to extend the growing season and create optimal conditions for phenological development. Hydroponics represents the most advanced technology, by replacing the soil with inert substrates and applying precise nutritional solutions and eliminating edaphic restrictions [
23,
24,
25]. This system optimizes the use of water resources and fertilizers, which facilitate the maximum expression of the varietal genetic potential with superior yields, shorter cycles, and uniform quality [
26,
27]. However, the efficacy of the protected systems is subject to accurate climatic management. In regions with high solar impact, greenhouses can experience extreme thermal increases, generating conditions of abiotic stress that cancel out their potential benefits [
28,
29]. The excessive heat within the greenhouse has been identified as a critical limiting factor for crops that are sensitive to high temperatures such as
Capsicum annuum, which negatively affect key physiological processes like photosynthesis, viability of pollen, and fruit setting [
30,
31]. This paradox underlines the need to evaluate not only the productive potential of the genotypes in different systems, but also their physiological resilience in the presence of specific microclimatic conditions, often suboptimal, which can be generated in protected environments.
The productive success in contrasting systems like open-field and hydroponic is intrinsically linked to the genotype-environment (G×E) interaction [
32,
33]. This concept defines the differential variation of the phenological, physiological and morphological performance of different genotypes under different environmental conditions. Phenological behavior constitutes a key indicator of the genotypical response to the environment, with critical stages such as blooming, fruiting and development of the fruit that are highly sensitive to environmental factors [
34]. A genotype with accelerated or stable phenology in hydroponics can translate into better harvest efficiency and superior profitability [
35].
Mexico, recognized as center of origin and domestication of
Capsicum annuum [
36,
37], is home to a vast genetic diversity represented in native landraces. This genetic wealth constitutes a reservoir of desirable traits, including tolerance to biotic and abiotic stress, adaptability to marginal conditions, and unique organoleptic profiles [
38,
39,
40]. However, modern agriculture has favored commercial cultivars, generating displacement of traditional landrace and creating a significant gap in knowledge regarding the phenological behavior and the productive potential of native landraces.
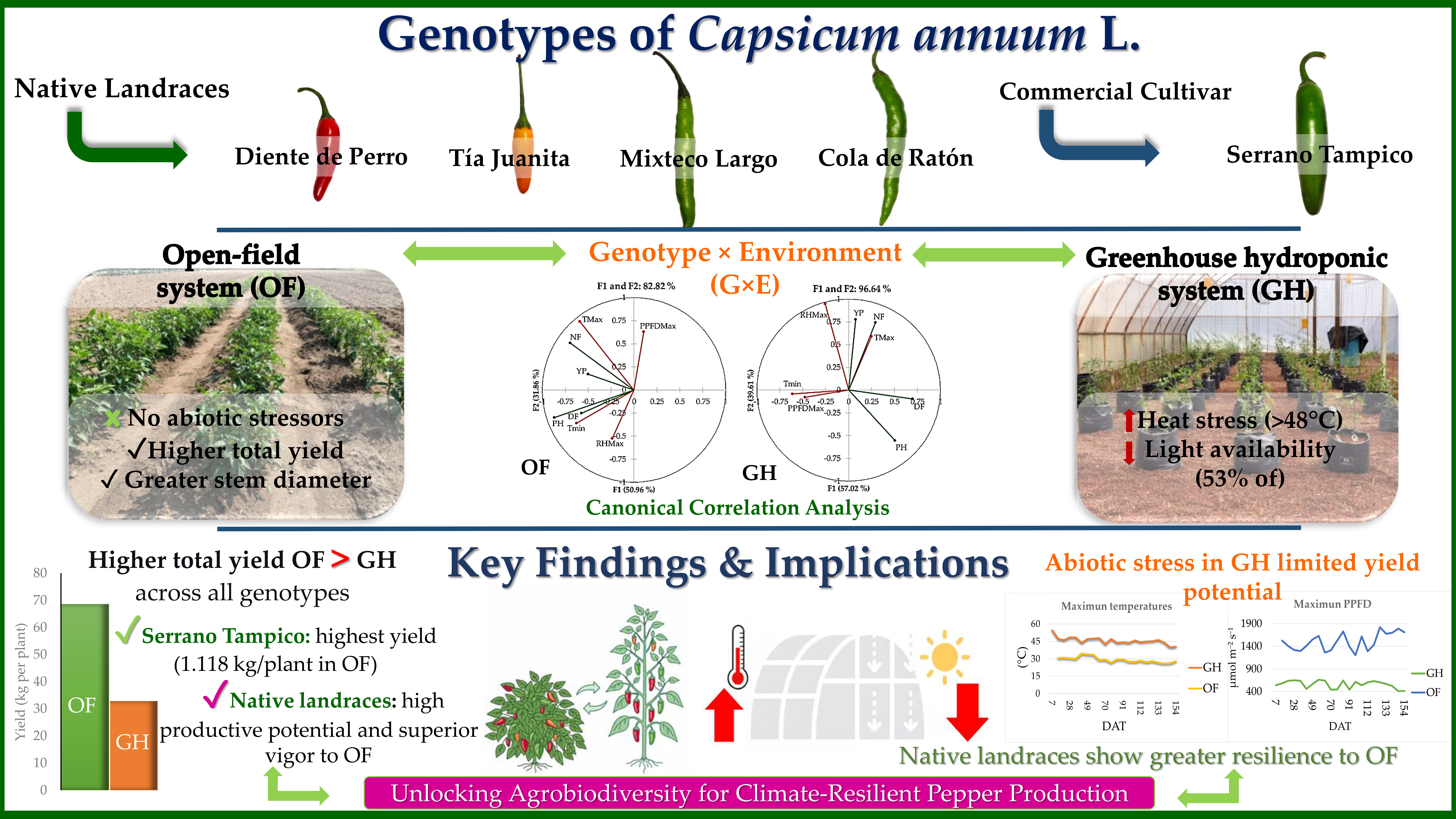
In face of this knowledge gap, the objective of this study was to determine the phenological and productive behavior of five genotypes of Capsicum annuum L. (four native landraces and one commercial cultivar), grown under two contrasting production systems: open-field traditional and greenhouse hydroponic. The working hypothesis postulated that the production system would have a significant effect in the performance of the genotypes, and that the greenhouse hydroponic system would substantially improve the phenological parameters, of growth and yield, compared to the open field. The results of this study, which include the analysis of the genotype-environment (G×E) interaction, provide fundamental scientific evidence for the selection of suitable native landraces and for the definition of specific management strategies, contributing to productive diversification and to the valuation of the economic potential of Mexican chili pepper agrobiodiversity.
2. Materials and Methods
2.1. Experimental Site and Plant Material
The study was conducted in San Agustín Calvario, San Pedro Cholula, Puebla, Mexico, during the spring-fall agricultural cycle in 2023. Two production systems were evaluated (
Figure 1):
Open-Field (OF): The experiment was established in the plot of a cooperating farmer located at 19°03'18''N, 98°20'08''W, 2164 masl. The soil presented a pH of 7.30, Electrical Conductivity (EC) of 0.75 dS m-1, and low organic matter content (0.74%).
Greenhouse Hydroponic (GH): It was conducted in a low-technology greenhouse measuring 100 m2, tunnel type, and protected with anti-aphid netting. Its location was at 19°02'59''N, 98°20'03''W, 2158 masl. The polyethylene cover, caliber 800 (200 μm), was characterized by reducing luminosity in 15% and filtering 76% of the ultraviolet radiation.
Five genotypes of
Capsicum annuum L. were used: four native landraces (Diente de Perro, Tía Juanita, Mixteco Largo, Cola de Ratón) and one commercial cultivar (Serrano Tampico). The taxonomic identity was confirmed by the Botanical Garden of the BUAP (herbarium vouchers 88725-88729). The plant morphology of these genotypes is illustrated in detail in
Figure 2.
2.2. Experimental Design and Crop Management
2.2.1. Open-Field System
The experiment was set up under a completely randomized block design (CRBD) with four repetitions. The transplant was carried out 80 days after sowing in furrows 6.0 m long and 0.80 m wide, with a distance between plants of 0.50 m, which resulted in a population density of 25,000 plants·ha-1. Each experimental unit consisted of six furrows, considering as a useful plot the five central plants of thirteen, from each furrow for measurements.
Fertilization: A fertilization formula of 180-100-30 (N-P2O5-K2O) was applied, using urea (46% N), diammonium phosphate (DAP, 18% N and 46% P2O5), and potassium chloride (60% K₂O). A third of the nitrogen and all the phosphorus and potassium were applied at the time of transplant. The remaining nitrogen was fractioned into two applications at 45 and 90 days after transplanting (dat).
Irrigation: The irrigation was programmed according to the local calendar of the farmer. Initially, furrow irrigation was applied, and it was changed to spray irrigation starting at 50 dat. A measuring system for soil moisture was not used.
Phytosanitary control: Pest and disease management was carried out in a preventive and healing manner. To control whitefly (Bemisia tabaci), imidacloprid was applied in a dose of 0.75 L·ha-1. To prevent bacterial diseases, copper sulfate was used. The management of fungal soil pathogens was done by alternating the application of the biofungicide Trichoderma harzianum (Natucontrol®) at a concentration of 15 mL per plant, and systemic fungicides. The latter included propamocarb + fosetyl (Previcur®) dosed at 6 L·ha-1, azoxystrobin + difenoconazole (Quadris Opti®) at 3 L·ha-1 and chlorothalonil + carbendazim (Prozicar®), applied at 2.5 L·ha-1.
Cultural practices: Periodical manual weeding and one plow pass were conducted at 45 dat to favor root development and to control weeds.
2.2.2. Greenhouse Hydroponic System
The experiment was conducted during the same agricultural cycle under a Completely Random Design (CRD) with 12 repetitions per genotype. Each experimental unit consisted in a black polyethylene bag with 11 kg of disinfected red tezontle sand, containing two seedlings, which resulted in a density of 6.67 plants·m-2.
Nutritional solution: The universal Steiner nutritional solution (1984) was used, with a final ionic concentration of 12, 1 and 7 meq·L-1 of NO₃⁻, H₂PO₄⁻ and SO₄²⁻ (anions), and 7, 9 and 4 meq·L-1 of K⁺, Ca²⁺ and Mg²⁺ (cations). The solution was prepared with soluble fertilizers: KNO3, Ca(NO3)2, KH2PO4, MgSO4, K2SO4, and a micronutrient complex (Fe, Mn, B, Zn, Cu, Mo).
Irrigation management and nutritional solution: The solution concentration was adjusted according to the phenological stage: 25% in post-transplanting, 50% during the initial plant development, and 100% starting at 15 dat. The irrigation volumes varied from 250 mL to 1.5 L per plant during flowering and fruiting. The electrical conductivity (EC) was kept at 2.5 dS·m-1 and the pH stabilized at 6.0 through the addition of phosphoric acid. Weekly, washing irrigation was carried out with just water to avoid the accumulation of salts.
Phytosanitary control: During flowering, systemic insecticides were applied with imidacloprid (Confidor®) and abamectin (Confinal®) in doses of 0.75 L·ha-1, for the control of whitefly, aphids and thrips. For fungal diseases, 200g/100 L of elemental sulfur water (Kumulus®) were applied preventively.
Cultural practices: Support was provided to plants through tutoring with agricultural raffia.
2.3. Variables Evaluated
Phenological variables. The phenological variables included time of emergence, defined as the number of days that passes from planting until 50% of the seeds of each genotype achieved emergence; and percentage of emergence, calculated as the production of seeds that emerged successfully from the total planted. Likewise, the time that passed from transplant to the appearance of key reproductive events was recorded: days to flowering (until anthesis of the first flower) and days to fruiting (until the appearance of the first fruit), both quantified when 50% of the population reached each stage per treatment.
Growth variables. The growth variables, recorded weekly since transplant, were plant height (vertical distance in cm from the base of the stem to the apex) and stem diameter (mm), measured with a digital gauge at 2 cm above the substrate level.
Yield variables. The number of fruits per plant was evaluated, which was quantified by recording the total of fruits developed and harvested once they reached physiological maturity; and total yield, which was determined as the total fresh weight (kg) of the fruits harvested per plant at the end of the experimental period, and the results were expressed in kg per plant.
Agroclimatic variables. The agroclimatic variables were monitored continually, and simultaneously. The air temperature (°C), relative humidity (%), and Photosynthetically Active Radiation, expressed as the Photosynthetic Photon Flux Density (PPFD, μmol m⁻² s⁻¹), were recorded. For these measurements, HOBO® dataloggers (models MX2301A, MX2202 and MX1104) were used, configured to record data with an hourly frequency during the experimental period.
2.4. Statistical Analysis
The data of phenological, growth and yield variables were analyzed through a two-way analysis of variance (ANOVA), considering the main effects of genotypes and production systems, as well as their interaction. The differences between means were compared with Tukey’s test (α≤0.05) using the statistical SAS® Studio package (Platform SAS® OnDemand for Academics, Version 3.1.0).
Additionally, a Canonical Correlation Analysis (CCA) was performed to explore the multivariate relationships between a set of agronomic variables (plant height, days to flowering, number of fruits per plant, yield), and a set of agroclimatic variables (maximum and minimum temperature, maximum relative humidity, and PPFD), grouped by growth stage. This analysis allowed elucidating the influence of the specific environmental conditions of each system on the agronomic performance of the genotypes. The CCA was executed with the XLSTAT software (version 2019.2.2) and the SAS® Studio statistical package (Version 3.1.0.).
3. Results
3.1. Agroclimatic Conditions
3.1.1. Air Temperature
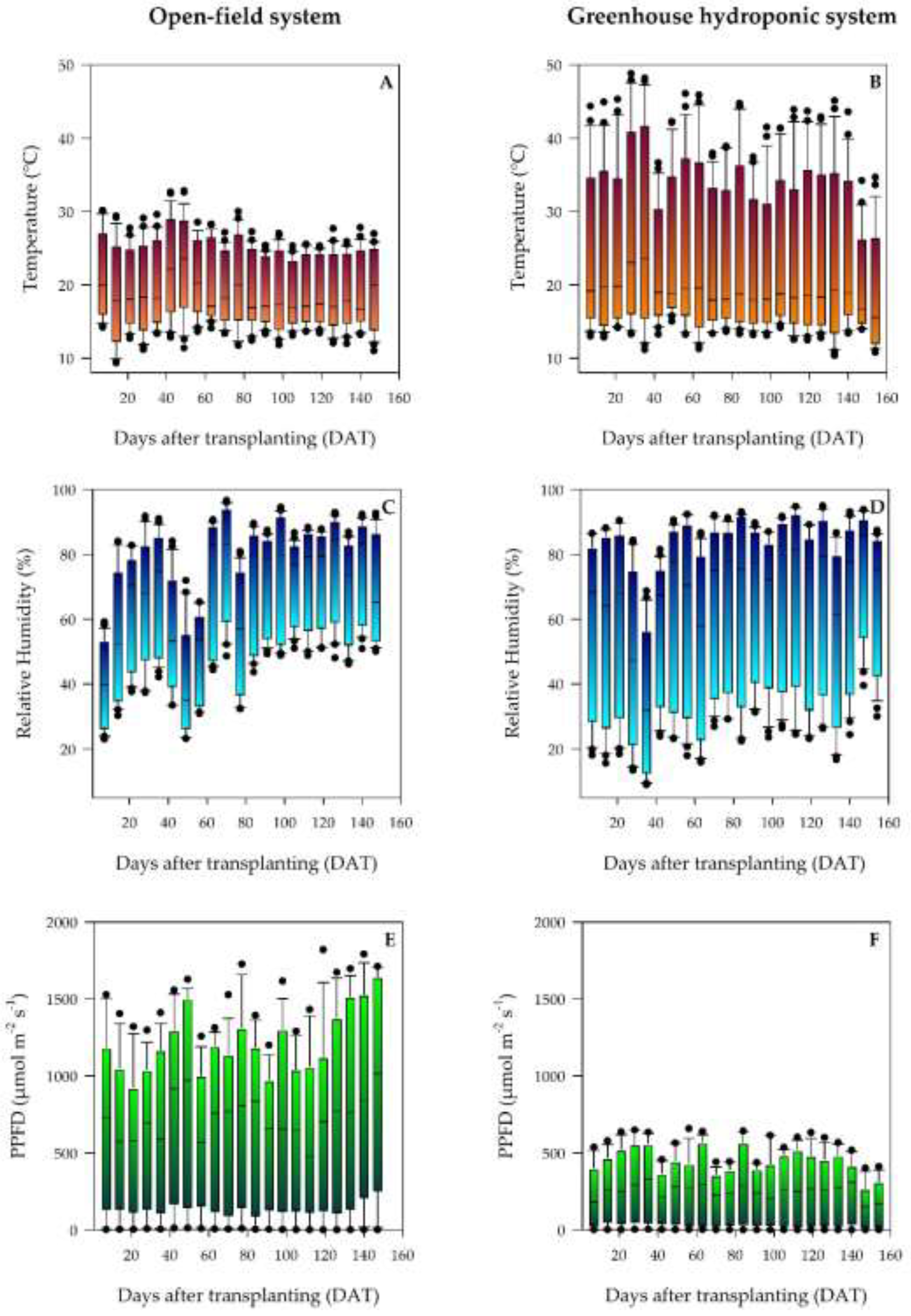
The thermal patterns were contrasting between the production systems (
Figure 3A, B). In open field (OF), a marked seasonal evolution was present. The initial cultivation period (14-35 dat) exhibited the greatest thermal amplitude, characterized by critical minimums of 9.36 °C and daytime maximums of 30.15 °C. The absolute maximum peak of the cycle was concentrated during the vegetative phase, reaching 32.80 °C at 56 dat (
Figure 3A). On the contrary, the greenhouse (GH) revealed extreme thermal stress from heat. The maximum temperatures consistently exceeded the optimal thresholds (18-30 °C), recording peaks of 48.15 °C in the initial period and an absolute maximum of 48.78 °C during the vegetative development.
3.1.2. Relative Humidity
The patterns of relative humidity (RH) markedly differed between the systems (
Figure 3C and D). In open field there was greater daytime variation, with morning maximums of 87-96% and minimums at noon of 23-35%. The initial cultivation period presented the most variable conditions (average 72.4 %, range 23.85-90.88%). On the other hand, the greenhouse kept a higher average humidity (78.2-83.7% through the phases), although it exhibited extreme daytime variations, with minimum values as low as 8.93% at noon and maximums that reached 94.78 % at dawn. Despite these initial ranges, GH presented a progressive stabilization of RH in the reproductive and maturation phases, reflecting better environmental control toward the end of the cycle.
3.1.3. Photosynthetic Photon Flux Density (PPFD)
The analysis exhibited differences in light availability, and PPFD was substantially higher in open field than in the greenhouse (
Figure 3E, F). The average value in OF was 523.4 μmol m
-2 s
-1. In this environment, the vegetative growth phase recorded the maximum average intensity (712.8 μmol m
-2 s
-1), and the absolute peak (1818.8 μmol m
-2 s
-1), while a progressive reduction of the PPFD was seen during maturation (543.6 μmol m
-2 s
-1) associated with seasonal progress. In contrast, the average PPFD in the greenhouse was 247.3 μmol m
-2 s
-1, which is equivalent to 53% of the available radiation in open field and is directly attributable to the shade effect generated by the cover and the structure. The maximum values under GH were reached during the vegetative phase (657.0 μmol m⁻² s⁻¹) and the attenuation exhibited a marked seasonal variation, with the proportion of blocked light being more pronounced in the summer (56%-64%, from June to September), than in the fall (40%-45%, from September to October), period when natural radiation was in itself limited.
3.2. Phenological Development
3.2.1. Time and Percentage of Emergence
Significant differences were observed (
p≤0.05) in the parameters of emergence between genotypes (
Table 1). Serrano Tampico required the shortest time of emergence (15.25 ± 1.26 days) followed by Diente de Perro (16.25 ± 1.26 days). Tía Juanita and Cola de Ratón exhibited the longest periods of emergence (24.75 ± 0.96 and 22.75 ± 2.22 days, respectively).
The percentage of emergence were higher for Serrano Tampico (98.50 ± 1.08%), Tía Juanita (97.75 ± 1.55%), and Diente de Perro (93.50 ± 5.15%). Mixteco Largo and Cola de Ratón presented a significantly lower emergence (78.63 ± 6.69% and 75.13 ± 2.66%, respectively).
3.2.2. Growth Dynamics
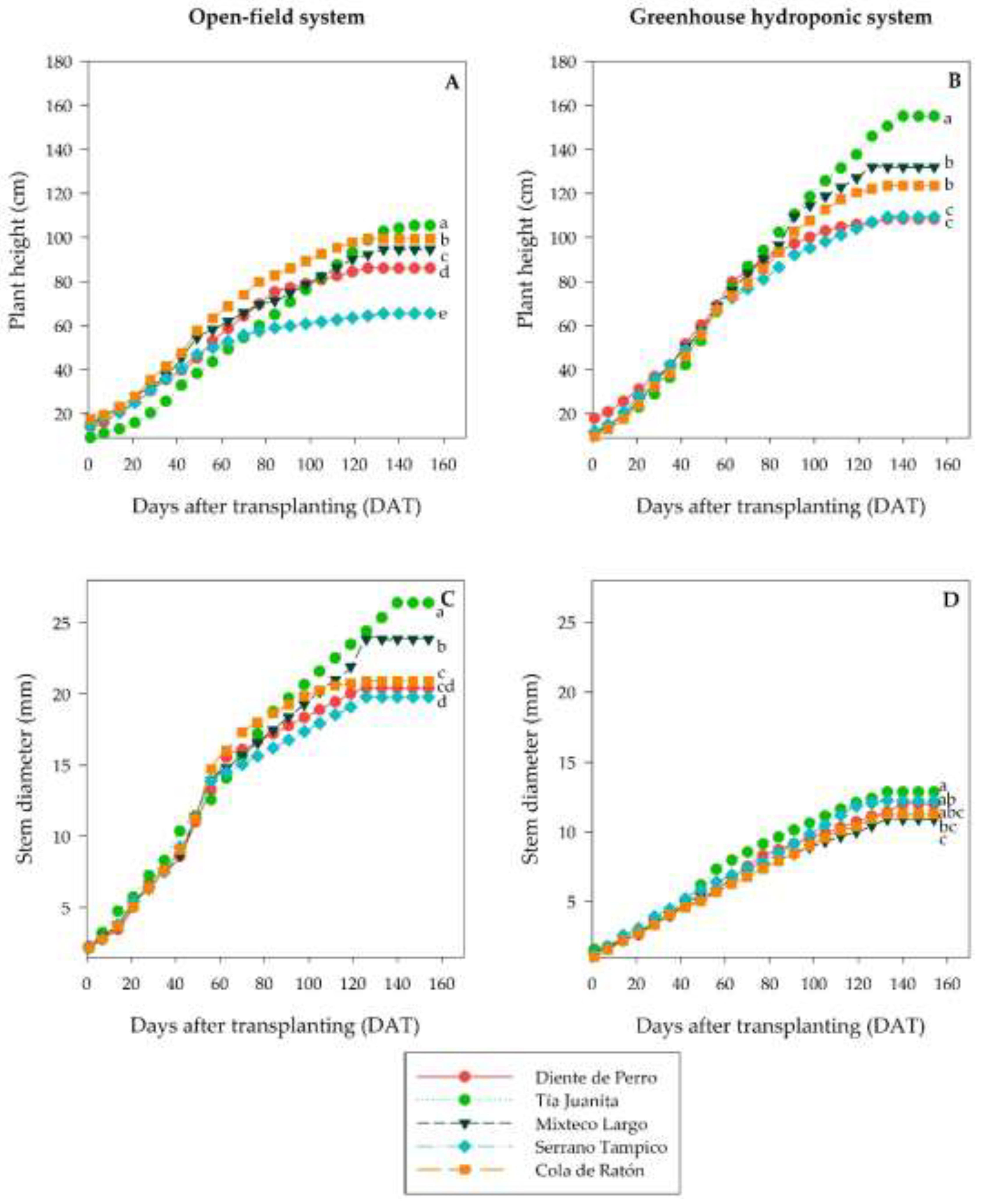
The analysis revealed significant differences (
p≤0.05) in the height and diameter of the stem between genotypes and systems (
Figure 4). The greenhouse hydroponic system promoted greater development in height, while a greater stem diameter was obtained in open field in all genotypes. The genotype Tía Juanita reached the greatest final height in both systems (OF, 105.44 cm; 155.04 cm), also highlighting the thickening of the stem in OF (26.41 mm). On the contrary, Serrano Tampico exhibited the lowest height increase in OF (65.45 cm) and a reduced final height in GH (109.42 cm), although this genotype demonstrated a more efficient growing pattern, prioritizing the allotment of resources toward the yield.
The temporal analysis of the development demonstrated differentiated genotypical growth patterns between genotypes. The genotype Cola de Ratón presented a fast initial growth with later stabilization of the height, suggesting a strategy of vigorous establishment. On the contrary, Tía Juanita exhibited late development, although sustained throughout the cycle. Finally, Serrano Tampico, a commercial cultivar of short stature, demonstrated the lowest vegetative growth (stem height and diameter). However, its efficient allotment of photoassimilates was reflected in the yield obtained in both production systems (Table 3).
3.2.3. Time of Flowering and Fruiting
The analysis of phenological times exhibited highly significant differences (
p≤0.05) between genotypes and production systems (
Table 2). The greenhouse hydroponic system accelerated both phenological phases, reducing the cycle between 4-13 days depending on the genotype.
Diente de Perro presented the most precocious trait, with the shortest times to flowering (51 dat in open-field; 45 dat in greenhouse) and fruiting (OF 67 dat; GH 54 dat). Serrano Tampico also stood out for its precociousness, particularly in the greenhouse (50 dat flowering and 59 dat fruiting), with the second genotype being more precocious. In the field, their times were slightly higher (57 dat flowering; 68 dat fruiting), although keeping its early character. The genotypes Mixteco Largo and Cola de Ratón presented intermediate phenologies, although significantly different: Mixteco Largo flowered at 59 dat (OF) and 52 dat (GH), while Cola de Ratón did it at 62 dat and 57 dat, respectively. Both genotypes presented a reduction of 7 days in time to fruiting under greenhouse conditions. Lastly, Tía Juanita exhibited the latest phenology, requiring 82 dat for flowering in open field and 77 dat in greenhouse, with fruiting at 92 and 85 dat, respectively, a trait that agrees with its vigorous late vegetative growth.
3.3. Components of the Yield
Significant differences (
p≤0.05) were observed in the yield and number of fruits between genotypes and production systems (
Table 3), which reflects different productive strategies.
The commercial cultivar Serrano Tampico stood out as the most productive in both systems, with 1.118 kg per plant in open field and 0.603 kg per plant in greenhouse. This high yield was explained by its high individual fruit weight, which compensates its lower number of fruits per plant (371.25 in OF; 152.42 in GH). The genotypes Mixteco Largo and Cola de Ratón demonstrated high productive potential in the field, with 642.40 and 628.60 fruits per plant, respectively, and yields of 0.759 and 0.872 kg per plant. However, in greenhouse both genotypes experienced significant reductions, maintaining high fruit production (240.25 and 279.92 fruits per plant) although with intermediate yields (0.273 and 0.332 kg per plant).
Tía Juanita presented a contrasting pattern: although it produced abundant fruits (402.00 in open field; 288.33 in greenhouse), exhibited the lowest yields (0.294 and 0.181 kg per plant, respectively). Diente de Perro presented the lowest values in both parameters, with 289.65 fruits and 0.387 kg per plant in open field, decreasing to 130.00 fruits and 0.255 kg per plant in the greenhouse. Overall, the open-field system exceeded the greenhouse in yield for all genotypes, with reductions that varied between 32% (Serrano Tampico) and 54% (Diente de Perro).
3.4. Canonical Correlation Analysis
The analysis revealed the existence of significant and specific genotype-environment (G×E) interactions for each genotype (
Table S1 of the Supplementary Material). The canonical correlations (r) varied from 0.6002 (Cola de Ratón in open field) to 0.9607 (Diente de Perro in greenhouse), indicating a strong association between the environmental conditions and the phenotypical response in most cases (
Table S2, Supplementary Material).
Table 4 summarizes the key environmental factors and their most relevant structural correlations (≥ 0.29) with the agronomic variables for each genotype and system.
3.4.1. Patterns of Interaction per Genotype
Diente de Perro presented the highest canonical correlations in both systems (r = 0.8789 in OF CA; r = 0.9607 in GH). In open field, the maximum and minimum temperatures exhibited positive correlations (TMax: r = 0.52; TMin: r = 0.55) with plant height and number of fruits. Under greenhouse, the pattern changed drastically: the minimum temperature (r = 0.59) and the maximum PPFD (r = 0.46) were negatively correlated with days to flowering and plant height, suggesting an acceleration of the cycle and a reduction of the plant development as a response to stress.
Tía Juanita exhibited strong and similar correlations in both environments (r= 0.9034 in OF; r = 0.8760 in GH). In open field, the maximum PPFD was the key environmental factor (r = 0.41), correlating positively with a higher number of fruits (r= 0.79) and yield (r= 0.51). In contrast, under greenhouse, the environmental vector was made up by the maximum PPFD (r= 0.57) and the maximum relative humidity (r= 0.43), showing a limited and negative correlation with the yield (r= -0.29).
Mixteco Largo presented a moderate G×E interaction in the field (r= 0.7095) and stronger in greenhouse (r= 0.8421). In open field, the maximum temperature presented a positive correlation (r=0.55) with the yield. A strong negative correlation with the maximum relative humidity (r= -0.53) stood out, indicating sensitivity to high humidity conditions. In the greenhouse, the minimum temperature was the dominating factor (r= 0.78), associated positively with the plant height (r= 0.52) but negatively with the yield (r= -0.53) and the number of fruits (r= -0.47).
Serrano Tampico recorded significant canonical correlations in both systems (r= 0.7699 in OF; r = 0.7582 in GH). In open field, the combination of high maximum temperature (r=0.64) and high maximum PPFD (r= 0.50) was associated with a delay in days to flowering (r= 0.73). In the greenhouse hydroponic system, the minimum temperature was the most impactful environmental factor (r= 0.69), correlating positively with the yield (r= 0.32) and the number of fruits (r= 0.40), but negatively with the plant height (r= -0.59).
Cola de Ratón presented lower canonical correlation in open field (r= 0.6002) and high correlation in the greenhouse (r= 0.8690). In open field, the maximum relative humidity (r= 0.47) and the minimum temperature (r= 0.43) were negatively correlated with the yield (r= -0.46) and days to flowering (r= -0.42). In the greenhouse, the maximum PPFD emerged as the key factor (r= 0.69), showing a positive correlation with plant height (r= 0.34) and, to a greater degree, with days to flowering (r= 0.69).
The structural correlation coefficients of the first three canonical factors for all the variables and genotypes are represented graphically in
Figures S1 to S5 of the Supplementary Material, providing an integral visualization of these multivariate interrelations.
4. Discussion
The results from this study do not fully support the initial hypothesis, which postulated a superiority of the greenhouse hydroponic system. Instead, they reveal that the phenological and productive performance of Capsicum annuum is fundamentally regulated by a complex genotype-environment (G×E) interaction, where the specific agroclimatic conditions of each system modulated the expression of genetic potential in a differential manner for each genotype. The central finding—an accelerated phenology but a consistently lower yield in the greenhouse—is explained by the thermal stress conditions found in this environment. Although the high temperatures promoted a fast accumulation of degrees-day that accelerated flowering and fruiting, the maximum extreme temperatures (frequently >40 °C) compromised the reproductive physiology of the plants, which limited fruit setting and filling.
4.1. Microclimatic Conditions as Limiting Factor in the Protected System
The microclimatic conditions of the greenhouse, characterized by extreme maximum temperatures (>48 °C) and attenuated photosynthetic photon flux density (PPFD) by 47% compared to the open field, constituted the main limiting factors. These extreme temperatures are directly related to the characteristics of the greenhouse design: low height (maximum 3 m to the center) and absence of zenithal ventilation, which limited the capacity of the greenhouse to dissipate heat and renovate the accumulated warm air on the top cover. Therefore, the temperatures recorded consistently exceeded the optimal range of 18–30 °C for
C. annuum [
41,
42,
43], in alignment with studies that report negative effects on key processes such as photosynthesis, pollen viability, and fruit setting when 32 °C is exceeded [
30,
31,
44,
45,
46]. This severe thermal stress, induced by deficient passive ventilation, resulted in a significant reduction of the yield, which varied between 32 and 54% according to the genotype, a direct impact on the limitations imposed by the protected environment.
In a complementary way, this significant reduction in radiation (PPFD) compared to the open field (
Figure 3E, F) limited the capacity for carbon assimilation. Average values of PPFD of 247.3 μmol m⁻² s⁻¹ are below the optimal range reported (1200 to 1500 μmol m⁻² s⁻¹) [
47] to maximize photosynthesis in
Capsicum, compromising the availability of photoassimilates for fruit filling [
48]. In addition, the extreme amplitudes of relative humidity (8.93–94.78%), outside the optimal range of 65–85% [
49], could affect the transpiration and create risk windows for pathogens [
50].
4.2. Phenological and Growth Responses: Genotypical Plasticity and Adaptation Strategies
The genotypes exhibited a broad diversity in their phenological and morphological behavior, demonstrating different adaptation strategies. The precociousness of Serrano Tampico and Diente de Perro (
Table 2) is a valuable trait for short cycles and an efficient early allotment of resources [
51,
52]. In contrast, the prolonged cycle and greater vegetative development of Tía Juanita reflect a strategy that prioritizes the accumulation of structural biomass before reproduction, typical of genotypes adapted to long cycles or competition for light [
53,
54]. The late but sustained growth pattern results in high biomass volume and high yield potential, desirable characteristic in long-cycle crops [
55,
56].
The differences in percentages of emergence between genotypes (75.13-98.50%) reflect variability in seed quality and seedling vigor [
57,
58]. The low percentages of Mixteco Largo and Cola de Ratón suggested the need for seed pre-treatment practices [
59,
60,
61] or adjustments in seed density to guarantee a successful establishment.
The generalized phenological acceleration in the greenhouse (4–13 days) did not translate into higher yield but rather seems to be an escape response to stress. The most revealing was the asymmetry in growth: while the plant height was higher in the greenhouse (etiolation), the stem diameter was significantly lower (
Figure 4). This morphological imbalance, induced by the low PPFD and high temperatures [
62], compromises the robustness of the plants and their capacity to sustain a productive load, in part explaining the low reproductive efficiency in this system [
63,
64,
65].
4.3. Genotype-Environment Interaction of the Yield
The Canonical Correlation Analysis (CCA) allowed robust and specific quantification of the G×E interaction, revealing contrasting adaptation strategies between genotypes. The superiority of the open-field yield for most of the native landraces (Mixteco Largo, Cola de Ratón) challenges the paradigm of generalized automatic superiority of protected systems, indicating that these materials are finely adapted to the environmental conditions of their zones of origin [
66,
72]. For example, Diente de Perro presented a positive correlation between the maximum temperature and its field yield (r= 0.52), suggesting a trait of productive thermo-tolerance [
67,
68]. On the contrary, in the greenhouse, the minimum temperatures were key for Serrano Tampico (r= 0.69 with yield), highlighting the importance of management of the nighttime temperature in controlled environments [
69].
The CCA also demonstrated physiological compensation. In the greenhouse, factors such as the high PPFD which promoted vegetative growth in Cola de Ratón (r= 0.69 with PH) did not translate into higher yield and even presented negative correlations in Mixteco Largo (r= -0.53). This highlights the balance between the vegetative development and the allotment of resources to fruits, a key principle under stress conditions [
70]. The different productive strategies —Serrano Tampico prioritizing the fruit weight, and Mixteco Largo/Cola de Ratón the number of fruits— reflect “domestication syndromes” of the genetic resource adapted to different niches [
71].
4.4. Agronomic Implications and Perspectives
The findings have immediate practical consequences:
Varietal selection: There is no universally superior genotype. For open-field systems under similar conditions, the native landraces Mixteco Largo and Cola de Ratón, along with the commercial Serrano Tampico, are high-yield options. Their transference to greenhouses without strict climatic control (especially of maximum temperature) is not advisable.
Greenhouse management in warm climates: The study alerts about the risk of heat stress from passive structures. The use of ventilation systems, shading and/or evaporative cooling becomes imperative to maintain temperatures within the optimal range [
28,
29,
72]. In a similar way, light attenuation should be critically monitored: it requires guaranteeing a PPFD that approaches the optimal range to promote high fruit production.
Native germplasm value: The native landraces demonstrated a high productive potential and resilience in the traditional system for which they were selected, constituting an invaluable genetic reservoir for breeding programs focused on tolerance to abiotic stress [
70,
73].
Future studies should: (i) replicate the study in greenhouses with active climatic control to isolate the effect of hydroponic from thermal stress, (ii) evaluate fruit quality parameters (capsacinoids, soluble solids) in this G×E interaction, and (iii) perform detailed physiological studies (photosynthetic rates, water use efficiency), to elucidate the mechanisms behind the adaptation strategies observed, and (iv) investigate seed pre-treatment methods to optimize the germination and emergence of native landraces.
5. Conclusions
This study demonstrated that the phenological and productive performance of Capsicum annuum is governed by a strong genotype-environment interaction, which was differentially modulated by the specific agroclimatic conditions of each production system.
The critical limiting factor was the greenhouse microclimate, not the hydroponics technique. The deficient architecture of the greenhouse (low height and absence of zenithal ventilation) caused extreme thermal stress and light attenuation, which negatively affected the plants and resulted in a severe loss of yield despite accelerating its phenology.
The open-field system consistently exceeded the yield of the protected system. This finding underlines that inadequate ventilation is a greater productive risk than the advantages that hydroponics can offer.
This study confirms the critical need to implement active climatic control strategies (ventilation, shading or cooling) in tunnel-type greenhouses was confirmed. It is imperative to maintain the PPFD and temperatures in the optimal range during the reproductive phase.
The canonical correlation analysis confirmed specific responses from each genotype to key environmental factors. The commercial cultivar Serrano Tampico was the most efficient in precociousness and yield under the conditions evaluated. However, the study validates the high potential and productive resilience of the native germplasm (Mixteco Largo and Cola de Ratón) under open-field conditions, making it a valuable genetic resource for the selection of genotypes adapted to stress.
This study provides essential evidence for decision making in the selection of genotypes and microclimatic management, contributing to the sustainability of chili pepper production under scenarios of climate change.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on Preprints.org. Table S1. Average values of agroclimatic and agronomic variables recorded for five Capsicum annuum L genotypes grown in two production systems. Table S2. Summary of Canonical Correlations among environmental variables for five Capsicum annuum L. genotypes in two production systems. Figure S1. Graphical representation of the structural correlation coefficients of the first three canonical factors (F1, F2, and F3) for the environmental and agronomic variables of the Diente de Perro pepper genotype, grown in open-field and greenhouse hydroponic systems: plant height (PH), days to flowering (DF), number of fruits per plant (NF), yield per plant (YP), maximum temperature (TMax), minimum temperature (TMin), maximum relative humidity (RHMax), and maximum photosynthetic photon flux density (PPFDMax). Figure S2. Graphical representation of the structural correlation coefficients of the first three canonical factors (F1, F2, and F3) for the environmental and agronomic variables of the Tía Juanita pepper genotype, grown in open-field and greenhouse hydroponic systems: plant height (PH), days to flowering (DF), number of fruits per plant (NF), yield per plant (YP), maximum temperature (TMax), minimum temperature (TMin), maximum relative humidity (RHMax), and maximum photosynthetic photon flux density (PPFDMax). Figure S3. Graphical representation of the structural correlation coefficients of the first three canonical factors (F1, F2, and F3) for the environmental and agronomic variables of the Mixteco Largo pepper genotype, grown in open-field and greenhouse hydroponic systems: plant height (PH), days to flowering (DF), number of fruits per plant (NF), yield per plant (YP), maximum temperature (TMax), minimum temperature (TMin), maximum relative humidity (RHMax), and maximum photosynthetic photon flux density (PPFDMax). Figure S4. Graphical representation of the structural correlation coefficients of the first three canonical factors (F1, F2, and F3) for the environmental and agronomic variables of the Serrano Tampico pepper genotype, grown in open-field and greenhouse hydroponic systems: plant height (PH), days to flowering (DF), number of fruits per plant (NF), yield per plant (YP), maximum temperature (TMax), minimum temperature (TMin), maximum relative humidity (RHMax), and maximum photosynthetic photon flux density (PPFDMax). Figure S5. Graphical representation of the structural correlation coefficients of the first three canonical factors (F1, F2, and F3) for the environmental and agronomic variables of the Cola de Ratón pepper genotype, grown in open-field and greenhouse hydroponic systems: plant height (PH), days to flowering (DF), number of fruits per plant (NF), yield per plant (YP), maximum temperature (TMax), minimum temperature (TMin), maximum relative humidity (RHMax), and maximum photosynthetic photon flux density (PPFDMax).
Author Contributions
Conceptualization, A.D.-A.; Investigation, B.N.H.-H.; methodology, A.D.-A., B.N.H.-H and M.A.T-C.; data curation and statistical analysis, B.N.H.-H. and B.E.H.-C.; resources, A.D.-A.; funding acquisition, A.D.-A. and B.N.H.-H.; writing—original draft preparation, B.N.H.-H.; supervision, M.A.T.-C., writing-review and editing, B.E.H.-C., M.A.T.-C., L.C.L.-E. and J.L.J.-V. All authors have read and agreed to the published version of the manuscript.
Funding
This research was primarily supported by Colegio de Postgraduados, Campus Puebla, through the Student Activity Support Program (AAE, Apoyo a la Actividad de Estudiantes). Additional funding was provided by Colegio de Postgraduados under Grant CONV_RGAA_2024_68 (Research and Incidence Projects for the Conservation and Sustainable Use of Genetic Resources for Food and Agriculture).
Data Availability Statement
The original contributions presented in this study are included in the article/
Supplementary Materials. Further inquiries can be directed to the corresponding author.
Acknowledgments
This study is product of the doctoral dissertation of the first, who wishes to thank the Ministry of Science, Humanities, Technology and Innovation (Secretaría de Ciencia, Humanidades, Tecnología e Innovación, SECIHTI), for scholarship number 784576 in support of her doctoral studies. The authors express their sincere gratitude to the local farmers for providing the native variety seeds used in this study. Special thanks are extended to Mr. Tomás Cuautle for providing the farmland and logistical support for the field experiment. The authors also acknowledge Colegio de Postgraduados, Campus Puebla, for providing greenhouse facilities and technical support.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Bijalwan, P.; Shukla, Y.R. Bell pepper (Capsicum annuum) productivity and economics as influenced by different weed management strategies. Indian J. Weed Sci. 2024, 56, 91–94. [Google Scholar] [CrossRef]
- Hossain, M.M.; Azad, M.A.K.; Soren, E.B.; Alam, M.N.; Ahmed, M.S.; Islam, M.S.; Monira, S. Enhancing growth, yield, and nutritional value of Capsicum annuum: Evaluating micronutrient efficiency and varietal performance. J. Agric. Food Res. 2025, 101945. [Google Scholar] [CrossRef]
- Organización de las Naciones Unidas para la Alimentación y la Agricultura (FAO). Estadísticas sobre alimentación y agricultura. FAOSTAT. 2023. Available online: https://www.fao.org/faostat/es/#data (Accessed on 15 July 2023).
- Penella, C.; Calatayud, A. Pepper Crop under Climate Change: Grafting as an Environmental Friendly Strategy. In Pepper Crops: Production and Management; Calatayud, A., Barreto, M.G., Eds.; IntechOpen: London, UK, 2018. [CrossRef]
- Hudáková, T.; Šuleková, M.; Tauchen, J.; Šemeláková, M.; Várady, M.; Popelka, P. Bioactive compounds and antioxidant activities of selected types of chilli peppers. Czech J. Food Sci. 2023, 41, 3:204–211. [Google Scholar] [CrossRef]
- Mandal, S.K.; Rath, S.K.; Logesh, R.; Mishra, S.K.; Devkota, H.P.; Das, N. Capsicum annuum L. and its bioactive constituents: A critical review of a traditional culinary spice in terms of its modern pharmacological potentials with toxicological issues. Phytother. Res. 2023, 37, 965–1002. [Google Scholar] [CrossRef]
- Akhter, M.J.; Akhter, S.; Islam, S.; Sarker, M.S.H.; Hasan, S.K. Varietal influence on bioactive compounds and antioxidant activity in chilies during development stages. Heliyon 2024, 10, e17441. [Google Scholar] [CrossRef]
- Bulle, M.; Rahman, M.M.; Islam, M.R.; Abbagani, S. Strategies to develop climate-resilient chili peppers: transcription factor optimization through genome editing. Planta 2025, 262, 30. [Google Scholar] [CrossRef] [PubMed]
- Baenas, N.; Belović, M.; Ilic, N.; Moreno, D.A.; García-Viguera, C. Industrial use of pepper (Capsicum annuum L.) derived products: Technological benefits and biological advantages. Food Chem. 2019, 274, 872–885. [Google Scholar] [CrossRef]
- Romero-Luna, H.E.; Colina, J.; Guzmán-Rodríguez, L.; Velazquez-Lozada, L. Capsicum fruits as functional ingredients with antimicrobial activity: An emphasis on mechanisms of action. J. Food Sci. Technol. 2023, 60, 2725–2735. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.M.; Khalid, N.I.; Wondi, M.H.; Haris, N.I.N.; Azman, P.N.M.A. Exploring the nutritional values, volatile compounds, health benefits, and potential food products of chilli (Capsicum annuum): A comprehensive review. Food Chem. 2025, 145091. [Google Scholar] [CrossRef]
- Wise, K.; Selby-Pham, J.; Simovich, T.; Gill, H. Enhancement of capsicum (Capsicum annuum L.) functional food value through nutrient supplementation with a biostimulant complex comprising triacontanol, phosphate, and potassium. N. Z. J. Crop Hortic. Sci. 2025, 53, 202–213. [Google Scholar] [CrossRef]
- Zanchini, R.; Spina, D.; De Pascale, A.; Buzi, G. Shaping consumer preferences for sweet peppers: Exploring the role of social, environmental, and sensory attributes in the era of health consciousness and local sourcing. Agric. Econ. 2025, 13, 42. [Google Scholar] [CrossRef]
- Selwal, N.; Rahayu, F.; Herwati, A.; Latifah, E.; Suhara, C.; Suastika, I.B.K.; Wani, A.K. Enhancing secondary metabolite production in plants: Exploring traditional and modern strategies. J. Agric. Food Res. 2023, 14, 100702. [Google Scholar] [CrossRef]
- Darko, E.; Hamow, K.A.; Marček, T.; Dernovics, M.; Ahres, M.; Galiba, G. Modulated Light Dependence of Growth, Flowering, and the Accumulation of Secondary Metabolites in Chilli. Front. Plant Sci. 2022, 13, 801656. [Google Scholar] [CrossRef]
- Islam, K.; Rawoof, A.; Kumar, A.; Momo, J.; Ahmed, I.; Dubey, M.; Ramchiary, N. Genetic Regulation, Environmental Cues, and Extraction Methods for Higher Yield of Secondary Metabolites in Capsicum. J. Agric. Food Chem. 2023, 71, 9213–9242. [Google Scholar] [CrossRef]
- Arya, M.; Kumar, G.; Giridhar, P. Serotonin-Salt-Stress Model-Induced Cell Growth via Promoting an Antioxidant System and Secondary Metabolites in Capsicum annuum Cell Suspension Culture. ACS Omega 2024, 9, 37330–37342. [Google Scholar] [CrossRef] [PubMed]
- Thuy, T.L.; Kenji, M. Effect of High Temperature on Fruit Productivity and Seed-Set of Sweet Pepper (Capsicum annuum L.) in the Field Condition J. Agric. Sci. Technol. A B Hue Univ. J. Sci. 2015, 5, 515–520. [Google Scholar] [CrossRef]
- Park, B.; Kim, S.; Kim, S. Simulation of the impacts of high temperature stress on pepper (Capsicum annum L.) yields. Front. Plant Sci. 2025, 16, 1590193. [Google Scholar] [CrossRef]
- Yang, H.; Bae, Y.; Kim, Y.; Hyeon, S.; Choi, M.; Yang, S.; Kim, D.; Jang, D. Effects of Irrigation Methods on Growth and Water Productivity in Bell Pepper Cultivation in Northern South Korea. Horticulturae 2024, 10, 1353. [Google Scholar] [CrossRef]
- Abdelhafez, A.A.; Al Dhumri, S.A.; Shaban, M.S.; Elgeheny, O.K.; Saleh, A.S.; Metwally, A.S. Exploring Future Cultivation Strategies in Greenhouses. EntechOpen 2025. [CrossRef]
- Velazquez-Gonzalez, R.S.; Garcia-Garcia, A.L.; Ventura-Zapata, E.; Barceinas-Sanchez, J.D.O.; Sosa-Savedra, J.C. A Review on Hydroponics and the Technologies Associated for Medium- and Small-Scale Operations. Agriculture 2022, 12, 646. [Google Scholar] [CrossRef]
- Rajaseger, G.; Chan, K.L.; Tan, K.Y.; Ramasamy, S.; Khin, M.C.; Amaladoss, A.; Kadamb Haribhai, P. Hydroponics: current trends in sustainable crop production. Bioinformation 2023, 19, 925–938. [Google Scholar] [CrossRef]
- Sangeetha, T.; Periyathambi, E. Automatic nutrient estimator: distributing nutrient solution in hydroponic plants based on plant growth. PeerJ Comput. Sci. 2024, 10, e1871. [Google Scholar] [CrossRef]
- Naresh, R.; Jadav, S.K.; Singh, M.; Patel, A.; Singh, B.; Beese, S.; Pandey, S.K. Role of Hydroponics in Improving Water-Use Efficiency and Food Security. Int. J. Environ. Clim. Change 2024, 14, 608–633. [Google Scholar] [CrossRef]
- Korsa, G.; Ayele, A.; Haile, S.; Alemu, D. Hydroponic Farming: Innovative Solutions for Sustainable and Modern Cultivation Technique. EntechOpen 2025. [CrossRef]
- Sran, T.S.; Jindal, S.K.; Chawla, N. Genotype by environment interaction for quality traits in chilli pepper (Capsicum annuum L.). Genetika 2021, 53, 23–49. [Google Scholar] [CrossRef]
- Shamshiri, R.R.; Jones, J.W.; Thorp, K.R.; Ahmad, D.; Man, H.C.; Taheri, S. Review of optimum temperature, humidity, and vapour pressure deficit for microclimate evaluation and control in greenhouse cultivation of tomato: a review. Int. Agrophys. 2018, 32, 287–302. [Google Scholar] [CrossRef]
- Gruda, N.; Tanny, J. Protected crops—recent advances, innovative technologies and future challenges. Horticulturae 2022, 8, 1087. [Google Scholar] [CrossRef]
- Oh, S.Y.; Koh, S.C. Fruit Development and Quality of Hot Pepper (Capsicum annuum L.) under Various Temperature Regimes. Hortic. Sci. Technol. 2019, 37, 313–321. [Google Scholar] [CrossRef]
- Jang, Y.; Schafleitner, R.; Barchenger, D.W.; Lin, Y.P.; Lee, J. Evaluation of heat stress response in pepper (Capsicum annuum L.) seedlings under controlled environmental conditions using a high-throughput 3D multispectral phenotyping. Sci. Hortic. 2025, 345, 114136. [Google Scholar] [CrossRef]
- Karim, K.M.R.; Rafii, M.Y.; Misran, A.; Ismail, M.F.; Harun, A.R.; Ridzuan, R.; Haque, M.A. Genetic Diversity Analysis among Capsicum annuum Mutants Based on Morpho-Physiological and Yield Traits. Agronomy 2022, 12, 2436. [Google Scholar] [CrossRef]
- Feldmann, F.; Rutikanga, A. Phenological growth stages and BBCH-identification keys of chilli (Capsicum annuum L., Capsicum chinense JACQ., Capsicum baccatum L.). J. Plant Dis. Prot. 2021, 128, 549–555. [Google Scholar] [CrossRef]
- Deepashri, K.M.; Kumar, J.S.; Santhosh, K.V. Harnessing Technological Advancements for Enhanced Crop Management: A Study on Capsicum Phenology and Automation in Agriculture. F1000Research 2025, 13, 1516. [Google Scholar] [CrossRef]
- Pickersgill, B. Relationships Between Weedy and Cultivated Forms in Some Species of Chili Peppers (Genus capsicum). Evolution 1971, 683–691. [Google Scholar] [CrossRef]
- García-Vásquez, R.; Vera-Guzmán, A.M.; Carrillo-Rodríguez, J.C.; Pérez-Ochoa, M.L.; Aquino-Bolaños, E.N.; Alba-Jiménez, J.E.; Chávez-Servia, J.L. Bioactive and nutritional compounds in fruits of pepper (Capsicum annuum L.) landraces conserved among indigenous communities from Mexico. AIMS Agric. Food 2023, 8, 3. [Google Scholar] [CrossRef]
- Taitano, N.; Bernau, V.; Jardón-Barbolla, L.; Leckie, B.; Mazourek, M.; Mercer, K.; McHale, L.; Michel, A.; Baumler, D.; Kantar, M.; van der Knaap, E. Genome-wide genotyping of a novel Mexican Chile Pepper collection illuminates the history of landrace differentiation after Capsicum annuum L. domestication. Evol. Appl. 2018, 12, 78–92. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Martínez, A.L.; Eguiarte, L.E.; Mercer, K.L.; Martínez-Ainsworth, N.E.; McHale, L.; van der Knaap, E.; Jardón-Barbolla, L. Genetic diversity, gene flow, and differentiation among wild, semiwild, and landrace chile pepper (Capsicum annuum) populations in Oaxaca, Mexico. Am. J. Bot. 2022, 109, 1157–1176. [Google Scholar] [CrossRef]
- Chouikhi, M.; Tlili, I.; Henane, I.; Takács, S.; Daood, H.; Pék, Z.; Helyes, L.; Montefusco, A.; De Caroli, M.; Di Sansebastiano, G.P.; Azam, M.; Siddiqui, M.W.; Ilahy, R.; Lenucci, M.S.; R’him, T. Agronomic and Functional Quality Traits in Various Underutilized Hot Pepper Landraces. Horticulturae 2024, 10, 710. [Google Scholar] [CrossRef]
- Rodríguez-Campos, E. La diversidad genética de Capsicum annuum de México. In Los chiles que le dan sabor al mundo; Aguilar-Meléndez, A., Vásquez-Levin, M.A., Katz, E., Hernández-Colorado, V., Eds.; IRD Éditions: Marseille, France, 2018. [Google Scholar] [CrossRef]
- Yamazaki, A.; Kitade, T.; Takezawa, A.; Nishimura, K.; Maki, T.; Nakano, R.; Hosokawa, M. Identification of a novel quantitative trait locus improving fruit set in chili pepper (Capsicum annuum) under high-temperature greenhouse conditions. Sci. Hortic. 2025, 353, 114471. [Google Scholar] [CrossRef]
- Bello, A.S.; Ahmed, T.; Saadaoui, I.; Ben-Hamadou, R.; Hamdi, H. Heat-stress-induced changes in enzymatic antioxidant activities and biochemicalprocesses in bell pepper (Capsicum annuum L.) seedlings. Turk. J. Agric. For. 2023, 47, 1165–1173. [Google Scholar] [CrossRef]
- Sajjad, N.; Kang, Y.; Khattak, M.; Lu, M. Molecular Cascades of Heat Stress Responses in Solanaceae with Emphasis on Capsicum annuum L., Integrating Heat Shock Transcription Factors and Proteins. Horticulturae 2025, 11, 1038. [Google Scholar] [CrossRef]
- Moore, CE; Meacham-Hensold, K; Lemonnier, P; Slattery, RA; Benjamin, C; Bernacchi, CJ; Lawson, T; Cavanagh, AP. The effect of increasing temperature on crop photosynthesis: from enzymes to ecosystems. J. Exp. Bot. 2021, 72, 2822–2844. [Google Scholar] [CrossRef]
- Kim, M.K.; Jeong, H.B.; Yu, N.; Park, B.M.; Chae, W.B.; Lee, O.J.; Lee, H.E.; Kim, S. Comparative heat stress responses of three hot pepper (Capsicum annuum L.) genotypes differing temperature sensitivity. Sci. Rep. 2023, 13, 14203. [Google Scholar] [CrossRef]
- Kaur, N.; Dhaliwal, M.S.; Jindal, S.; Singh, P. Evaluation of Hot Pepper (Capsicum annuum L.) Genotypes for Heat Tolerance during Reproductive Phase. Int. J. Bio-Resour. Stress Manag. 2023, 7, 126–129. [Google Scholar] [CrossRef]
- Wittmann, S.; Wittmann, I.; Mewis, I.; Förster, N.; Asseng, S.; Mempel, H. Capsicum annuum in vertical indoor farming: yield and capsaicinoid responses to reduced light and additional UV-A. Scientia Horticulturae 2025, 350, 114364. [Google Scholar] [CrossRef]
- Nie, W.F.; Li, Y.; Chen, Y.; Zhou, Y.; Yu, T.; Zhou, Y.; Yang, Y. Spectral light quality regulates the morphogenesis, architecture, and flowering in pepper (Capsicum annuum L.). J. Photochem. Photobiol. B Biol. 2023, 241, 112673. [Google Scholar] [CrossRef]
- López-Bravo, E.; Placeres-Remior, A.; Carbonell-Saavedra, E.; Martínez-Rodríguez, A.; González Cueto, O. Variabilidad de factores agroclimáticos y gasto de riego en cultivo protegido del pimiento (Capsicum annuum). Rev. Cienc. Téc. Agropecu. 2023, 32, 4. Available online: https://cu-id.com/2177/v32n4e05.
- Méndez-Melo, G.S.; Tornero-Campante, M.A.; Paniagua-Solar, L.A. Design and implementation of an electronic system to monitor and record agroclimatic variables in greenhouses. AgroProductividad 2024. [Google Scholar] [CrossRef]
- Olatunji, T.L.; Afolayan, A.J. Variability in seed germination characteristics of Capsicum annuum L. and Capsicum frutescens L. Pak. J. Bot. 2019, 51, 561–565. [Google Scholar] [CrossRef]
- Corozo-Quiñónez, L.; López-Cedeño, J.; Lascano-Borja, G.; Monteros-Altamirano, Á.; Arteaga-Alcívar, F.; Jaimez, R.E.; García-Dávila, M.A. Seed viability, germination, and seedling growth of capsicum cultivated species. Chilean J. Agric. Anim. Sci. 2023, 39, 401–412. [Google Scholar] [CrossRef]
- Poorter, H.; Niklas, K.J.; Reich, P.B.; Oleksyn, J.; Poot, P.; Mommer, L. Biomass allocation to leaves, stems and roots: meta-analyses of interspecific variation and environmental control. New Phytol. 2012, 193, 30–50. [Google Scholar] [CrossRef]
- Raihan, T.; Geneve, R.L.; Perry, S.E.; Rodriguez Lopez, C.M. The Regulation of Plant Vegetative Phase Transition and Rejuvenation: miRNAs, a Key Regulator. Epigenomes 2021, 5, 24. [Google Scholar] [CrossRef]
- Chen, S.; Kerstens, T.; Zepeda, B.; Ouzounis, T.; Olschowski, S.; Marcelis, L.F.M.; Heuvelink, E. Additional far-red increases fruit yield of greenhouse sweet pepper mainly through enhancing plant source strength. Scientia Horticulturae 2025, 339, 113787. [Google Scholar] [CrossRef]
- Rosado-Souza, L.; Yokoyama, R.; Sonnewald, U.; Fernie, A.R. Understanding source–sink interactions: Progress in model plants and translational research to crops. Mol. Plant 2023, 16, 96–121. [Google Scholar] [CrossRef]
- Dagne, B.A.; Duga, J.N.; Hirpa, G.D. Response of Hot Pepper (Capsicum annuum) to Potassium Nitrate Seed Priming. Adv. Agric. 2024, 2024, 6881808. [Google Scholar] [CrossRef]
- Reed, R.C.; Bradford, K.J.; Khanday, I. Seed germination and vigor: ensuring crop sustainability in a changing climate. Heredity 2022, 128, 450–459. [Google Scholar] [CrossRef] [PubMed]
- Garruña-Hernández, R.; Latournerie-Moreno, L.; Ayala-Garay, O.; Santamaría, J.M.; Pinzón-López, L. Acondicionamiento pre-siembra: una opción para incrementar la germinación de semillas de chile habanero (Capsicum chinense Jacq.). Agrociencia 2014, 48, 413–423. Available online: http://www.scielo.org.mx/scielo.php?script=sci_arttext&pid=S1405-31952014000400006&lng=es&tlng=es.
- Brondo-Ricárdez, R.; Domínguez-Angulo, S.; Pérez-Hernández, I.; D’Artola-Barceló, A.L. Tratamientos pregerminativos a semillas y desarrollo inicial de plántulas de chile amashito (Capsicum annuum L. var. glabriusculum). Agro Productividad. 2020, 13, 53–59. [Google Scholar] [CrossRef]
- Antúnez-Ocampo, O.M.; Croseños-Palazin, M.I.; Espinosa-Rodríguez, M.; Vázquez-Villamar, M.; Rojas-García, A.R.; Sabino-López, J.E. Germinación de la semilla y vigor de plántula de chiles apaxtlecos en respuesta a la radiación gamma. Bio Cienc. 2024, 11, e1591. [Google Scholar] [CrossRef]
- He, X.; Maier, C.; Chavan, S.G.; Zhao, C.C.; Alagoz, Y.; Cazzonelli, C.; Ghannoum, O.; Tissue, D.T.; Chen, Z.H. Light-altering cover materials and sustainable greenhouse production of vegetables: A review. Plant Growth Regul. 2021, 95, 1–17. [Google Scholar] [CrossRef]
- Islam, M.; Satyaranjan, S.A.H.A.; Akand, H.; Rahim, A. Effect of spacing on the growth and yield of sweet pepper (Capsicum annuum L.). J. Cent. Eur. Agric. 2011. [Google Scholar] [CrossRef]
- Jadama, L.; Jammeh, P.T.; Cham, A.K.; Diedhiou, I. Effect of Different Spacing on the Growth and Yield of California Wonder Bell Pepper (Capsicum annuum) on Sandy Loam Soil in the Gambia. Asian J. Biol. 2021, 12, 1–9. [Google Scholar] [CrossRef]
- Thakur, P.; Singh, S. Effect of different spacing on growth and yield of bell pepper (Capsicum annuum L.) hybrid Bomby grown under protected conditions. Int. J. Plant Sci. 2023, 18, 160–165. [Google Scholar] [CrossRef]
- Gerakari, M.; Ralli, P.; Giannakoula, A.; Ouzounidou, G.; Anagnostou, A.; Antoniadis, C.; Avdikos, I.D. Agronomic, Morphological, and Nutritional Characterization of Greek Traditional Pepper (Capsicum annuum L.) Landraces at Commercial and Physiological Maturity for Sustainable and Climate-Smart Vegetable Systems. Plants 2025, 14, 3164. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Wang, J.; Ren, C.; Chen, Y.; Yang, S.; Yin, Q.; Liu, W.; Wang, J.; Wang, P.; Zou, X. Genome-Wide Identification of CaGA20ox Gene Family Members Related to Floral Organ Development in Pepper (Capsicum annuum) at Different Temperatures. Horticulturae 2025, 11, 469. [Google Scholar] [CrossRef]
- Yamazaki, A.; Takezawa, A.; Nishimura, K.; Motoki, K.; Nagasaka, K.; Nakano, R.; Yagi, N.; Hosokawa, M. Pollen Dispersion is a Key Factor for Autonomous Fruit Set under High Temperatures in the Capsicum annuum ‘Takanotsume’. Hortic. J. 2024, 93, 49–57. [Google Scholar] [CrossRef]
- Roudbari, Z.; Sarhadi, J.; Azadvar, M.; Alavi-Siney, S. M.; Jalali, A. Evaluation of the Effect of Day and Night Temperature Fluctuations in different Seasons of the Year on the Fruit Formation of Sweet Pepper (Capsicum annuum L.) Lines. Journal of Horticultural Science 2023, 37(1), 121–133. [Google Scholar] [CrossRef]
- Mahmood, T.; Rana, R. M.; Ahmar, S.; Saeed, S.; Gulzar, A.; Khan, M. A.; Ali, A.; Alghamdi, A. A.; Du, X. Effect of Drought Stress on Capsaicin and Antioxidant Contents in Pepper Genotypes at Reproductive Stage. Plants 2021, 10, 1286. [Google Scholar] [CrossRef]
- Sood, T.; Sood, S.; Sood, V.K.; Badiyal, A.; Anuradha; Kapoor, S.; Sood, V.; Kumar, N. Characterisation of bell pepper (Capsicum annuum L. var. grossum Sendt.) accessions for genetic diversity and population structure based on agro-morphological and microsatellite markers. Sci. Hortic. 2023, 321, 112308. [Google Scholar] [CrossRef]
- Lozano-Fernández, J.; Orozco-Orozco, L.F.; Grisales-Vásquez, N.Y. Comportamiento agronómico de cultivares de pimentón (Capsicum annuum L.) cultivados en campo abierto y en condiciones protegidas. Terra Latinoamericana 2022, 40, e1459. [Google Scholar] [CrossRef]
- Ahmad, F.; Kusumiyati, K.; Arief Soleh, M.; Rabnawaz Khan, M.; Siti Sundari, R. Microclimates growing and watering volumes influences the physiological traits of chili pepper cultivars in combating abiotic stress. Sci. Rep. 2025, 15, 4183. [Google Scholar] [CrossRef]
Figure 1.
Production systems of Capsicum annuum L. evaluated in San Agustín Calvario, Puebla: (A) Open-Field and (B) Greenhouse Hydroponic System.
Figure 1.
Production systems of Capsicum annuum L. evaluated in San Agustín Calvario, Puebla: (A) Open-Field and (B) Greenhouse Hydroponic System.
Figure 2.
Morphology of the plants from the five genotypes of Capsicum annuum L. evaluated in the study: (A) Diente de Perro, (B) Tía Juanita, (C) Mixteco Largo, (E) Cola de Ratón (native landraces) and (D) Serrano Tampico (commercial cultivar).
Figure 2.
Morphology of the plants from the five genotypes of Capsicum annuum L. evaluated in the study: (A) Diente de Perro, (B) Tía Juanita, (C) Mixteco Largo, (E) Cola de Ratón (native landraces) and (D) Serrano Tampico (commercial cultivar).
Figure 3.
Dynamics of agroclimatic variables in (A, C, E) open-field and (B, D, F) greenhouse hydroponic systems: (A, B) air temperature, (C, D) relative humidity, and (E, F) photosynthetic photon flux density (PPFD). Each boxplot represents 24 daily data points. The first and third quartiles represent the interquartile range, while the horizontal line within the box indicates the mean. The whiskers represent the minimum and maximum values, and the individual points represent outliers.
Figure 3.
Dynamics of agroclimatic variables in (A, C, E) open-field and (B, D, F) greenhouse hydroponic systems: (A, B) air temperature, (C, D) relative humidity, and (E, F) photosynthetic photon flux density (PPFD). Each boxplot represents 24 daily data points. The first and third quartiles represent the interquartile range, while the horizontal line within the box indicates the mean. The whiskers represent the minimum and maximum values, and the individual points represent outliers.
Figure 4.
Growth dynamics of plant height (A, B) and stem diameter (C, D) of five Capsicum annuum L. genotypes grown in open-field (A, C) and greenhouse hydroponic (B, D) systems. Data points represent mean values. Different letters at the final point of each curve indicate statistically significant differences between genotypes within each system and variable, according to Tukey's test (p ≤ 0.05).
Figure 4.
Growth dynamics of plant height (A, B) and stem diameter (C, D) of five Capsicum annuum L. genotypes grown in open-field (A, C) and greenhouse hydroponic (B, D) systems. Data points represent mean values. Different letters at the final point of each curve indicate statistically significant differences between genotypes within each system and variable, according to Tukey's test (p ≤ 0.05).
Table 1.
Emergence parameters of five Capsicum annuum L. genotypes grown under greenhouse conditions.
Table 1.
Emergence parameters of five Capsicum annuum L. genotypes grown under greenhouse conditions.
| Genotype |
Germination
(days)
|
Emergence
(%)
|
| Diente de Perro |
16.25 ab ± 1.26 |
93.50 a ± 5.15 |
| Tía Juanita |
24.75 c ± 0.96 |
97.75 a ± 1.55 |
| Mixteco Largo |
19.25 b ± 1.50 |
78.63 b ± 6.69 |
| Serrano Tampico |
15.25 a ± 1.26 |
98.50 a ± 1.08 |
| Cola de Ratón |
22.75 c ± 2.22 |
75.13 b ± 2.66 |
| HSD |
3.28 |
8.84 |
Table 2.
Flowering and fruiting times (days after transplanting, DAT) of five Capsicum annuum L. genotypes grown in open-field and greenhouse hydroponic systems.
Table 2.
Flowering and fruiting times (days after transplanting, DAT) of five Capsicum annuum L. genotypes grown in open-field and greenhouse hydroponic systems.
| Genotype |
Flowering (DAT) |
Fruiting (DAT) |
| Open-field |
Greenhouse |
Open-field |
Greenhouse |
| Diente de Perro |
51 a ± 1.76 |
45 a ± 3.16 |
67 a ± 2.24 |
54 a ± 4.39 |
| Tía Juanita |
82 e ± 1.76 |
77 d ± 3.09 |
92 d ± 1.97 |
85 d ± 4.39 |
| Mixteco Largo |
59 c ± 1.76 |
52 b ± 3.09 |
69 b ± 1.96 |
61 b ± 4.39 |
| Serrano Tampico |
57 b ± 2.01 |
50 b ± 2.81 |
68 ab ± 2.16 |
59 ab ± 4.39 |
| Cola de Ratón |
62 d ± 2.01 |
57 c ± 1.88 |
74 c ± 3.50 |
68 c ± 4.39 |
| HSD |
1.47 |
2.53 |
2.14 |
3.95 |
Table 3.
Yield components of five Capsicum annuum L. genotypes grown in open-field and greenhouse hydroponic systems.
Table 3.
Yield components of five Capsicum annuum L. genotypes grown in open-field and greenhouse hydroponic systems.
| Genotype |
Number of fruits per plant |
Yield (kg per plant) |
| Open-field |
Greenhouse |
Open-field |
Greenhouse |
| Diente de Perro |
289.65 c ± 50.70 |
130.00 c ± 10.79 |
0.387 c ± 0.09 |
0.255 c ± 0.02 |
| Tía Juanita |
402.00 b ± 70.61 |
288.33 a ± 39.75 |
0.294 c ± 0.05 |
0.181 d ± 0.03 |
| Mixteco Largo |
642.40 a ± 152.07 |
240.25 b ± 21.68 |
0.759 b ± 0.14 |
0.273 c ± 0.03 |
| Serrano Tampico |
371.25 bc ± 85.71 |
152.42 c ± 25.23 |
1.118 a ± 0.27 |
0.603 a ± 0.09 |
| Cola de Ratón |
628.60 a ± 148.03 |
279.92 a ± 46.86 |
0.872 b ± 0.19 |
0.332 b ± 0.05 |
| HSD |
93.11 |
35.37 |
0.141 |
0.055 |
Table 4.
Key environmental factors and their correlation with agronomic variables in five Capsicum annuum L. genotypes grown in two production systems.
Table 4.
Key environmental factors and their correlation with agronomic variables in five Capsicum annuum L. genotypes grown in two production systems.
| Genotype |
System |
Canonical correlation
(r)
|
Key environmental factors
(correlation)
|
Key agronomic variables
(correlation)
|
| Diente de Perro |
OF |
0.8789 |
TMax (+0.52), TMin (+0.55) |
PH (+0.76), NF (+0.61), DF (+0.50), YP (+0.44) |
| GH |
0.9607 |
TMin (+0.59), PPFDMax (+0.46) |
PH (-0.49), DF (-0.67) |
| Tía Juanita |
OF |
0.9034 |
PPFDMax (+0.41) |
NF (+0.79), YP (+0.51) |
| GH |
0.8760 |
RHMax (+0.43), PPFDMax (+0.57) |
YP (-0.29) |
| Mixteco Largo |
OF |
0.7095 |
TMax (+0.55) |
YP (+0.36) |
| GH |
0.8421 |
TMin (+0.78) |
PH (+0.52), YP (-0.53), NF (-0.47) |
Serrano
Tampico |
OF |
0.7699 |
TMax (+0.64), PPFDMax (+0.50) |
DF (+0.73) |
| GH |
0.7582 |
TMin (+0.69) |
PH (-0.59), YP (+0.32), NF (+0.40) |
| Cola de Ratón |
OF |
0.6002 |
TMin (+0.43), RHMax (+0.47) |
YP (-0.46), DF (-0.42) |
| GH |
0.8690 |
PPFDMax (+0.69) |
PH (+0.34), DF (+0.69) |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).