Submitted:
14 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methodology
2.1. Chemicals
2.2. Deposition of ZnO Thin Film Active Layer on Support MS10/Z90 Using Spin Coating
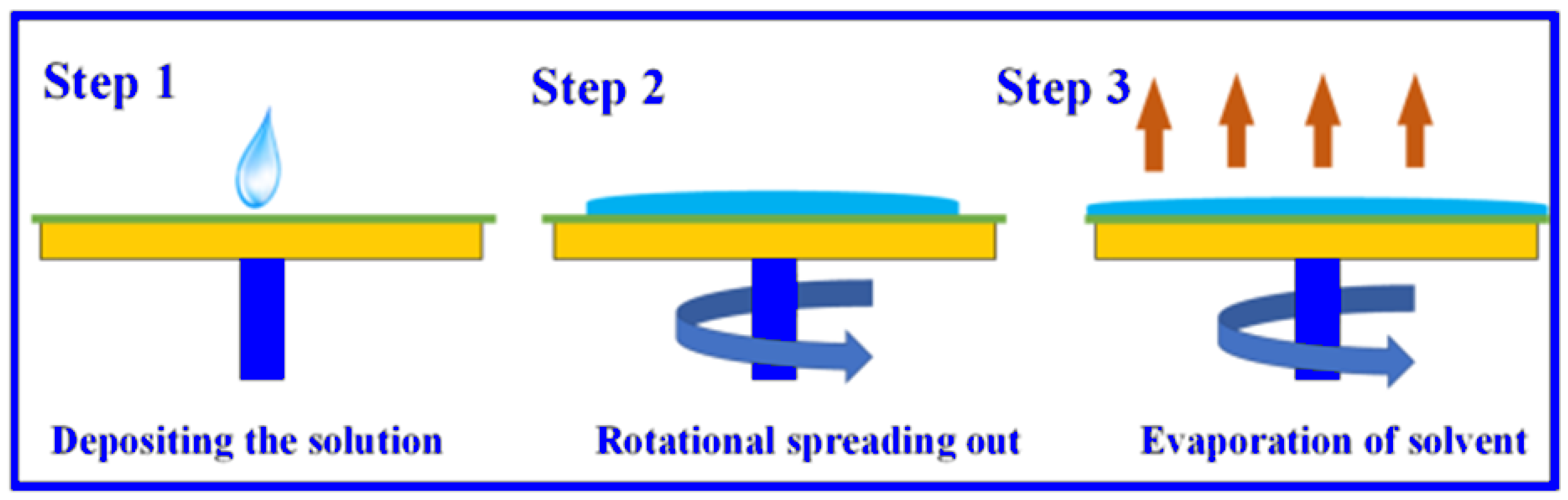
2.3. Membrane Characterization
2.4. Membrane Performance
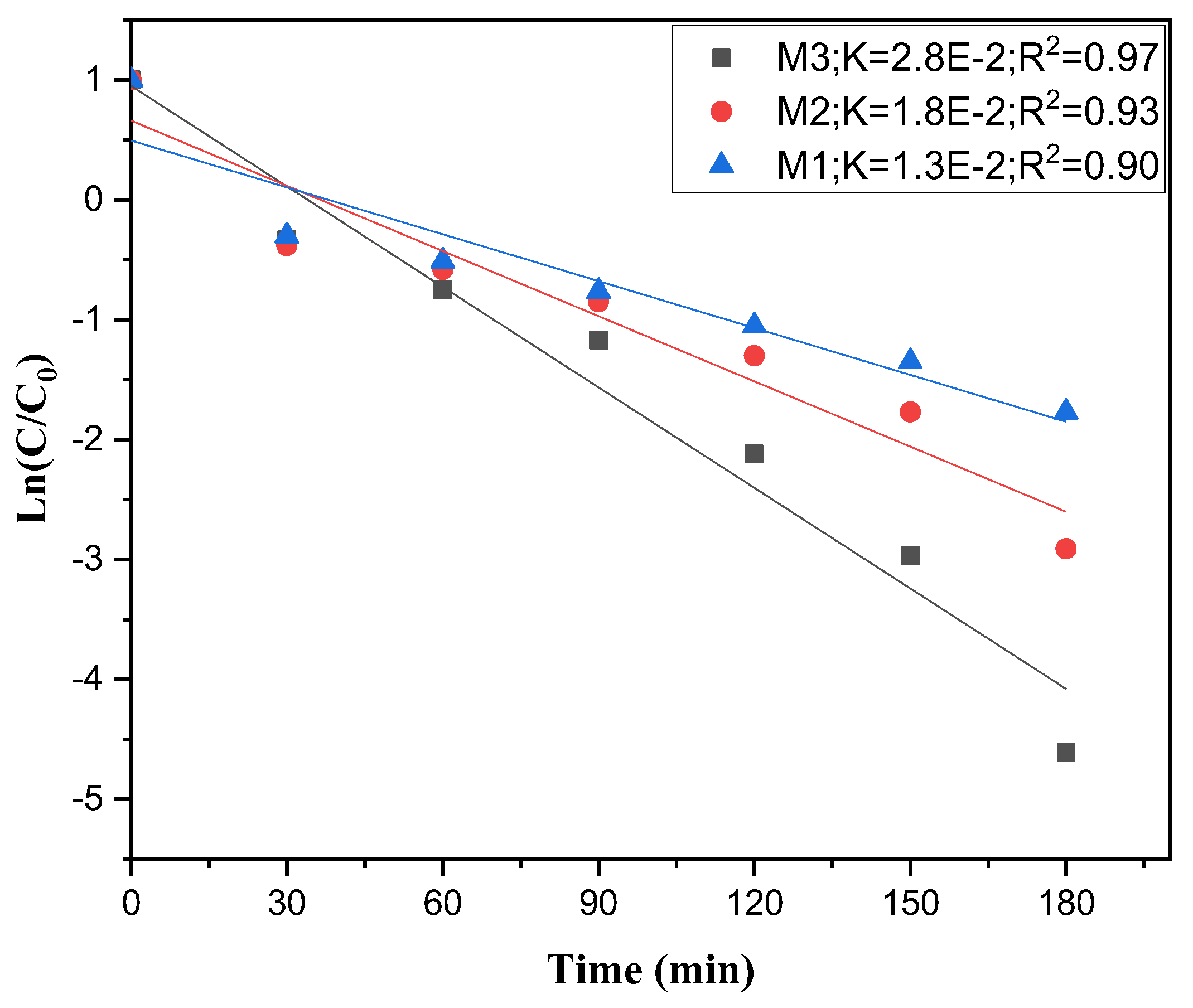
2.5. Kinetics Study
- = apparent rate constant (min⁻¹)
- = initial concentration of the pollutant (mg.L-1)
- = concentration of the pollutant at time (mg.L-1)
- = reaction time (min).
3. Results and Discussion
3.1. Membrane Characterization
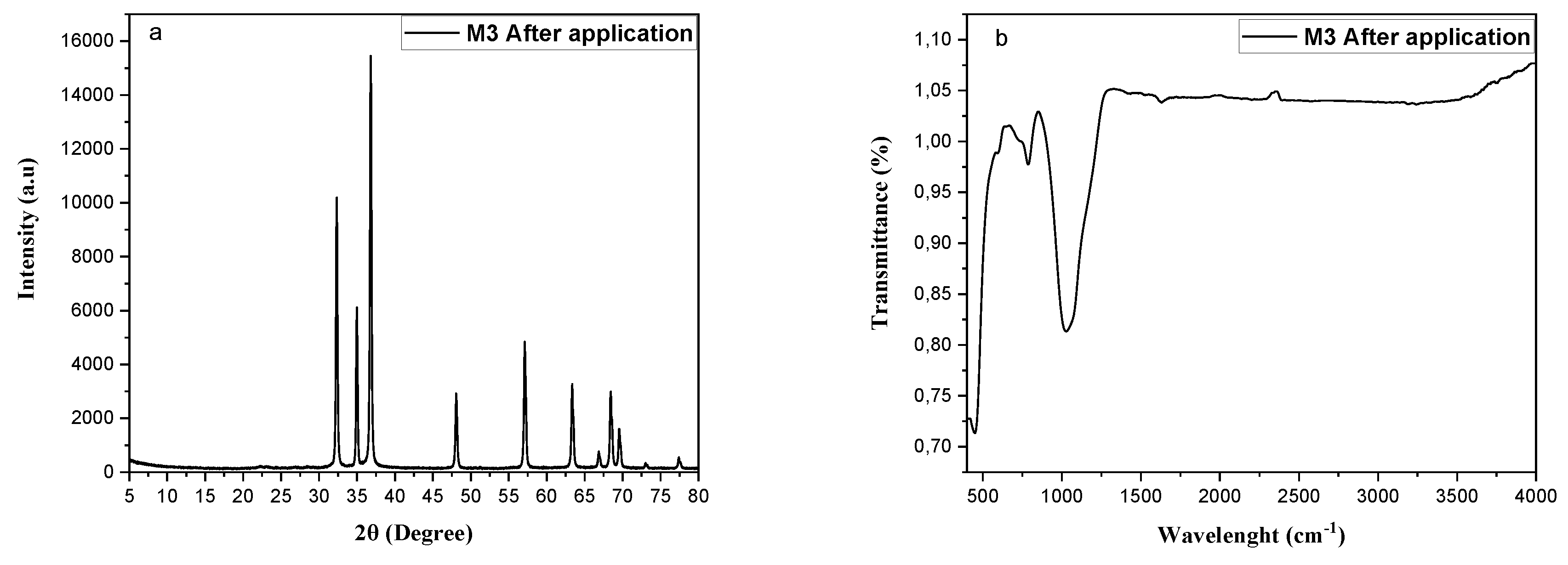
3.1.1. XRD Analysis
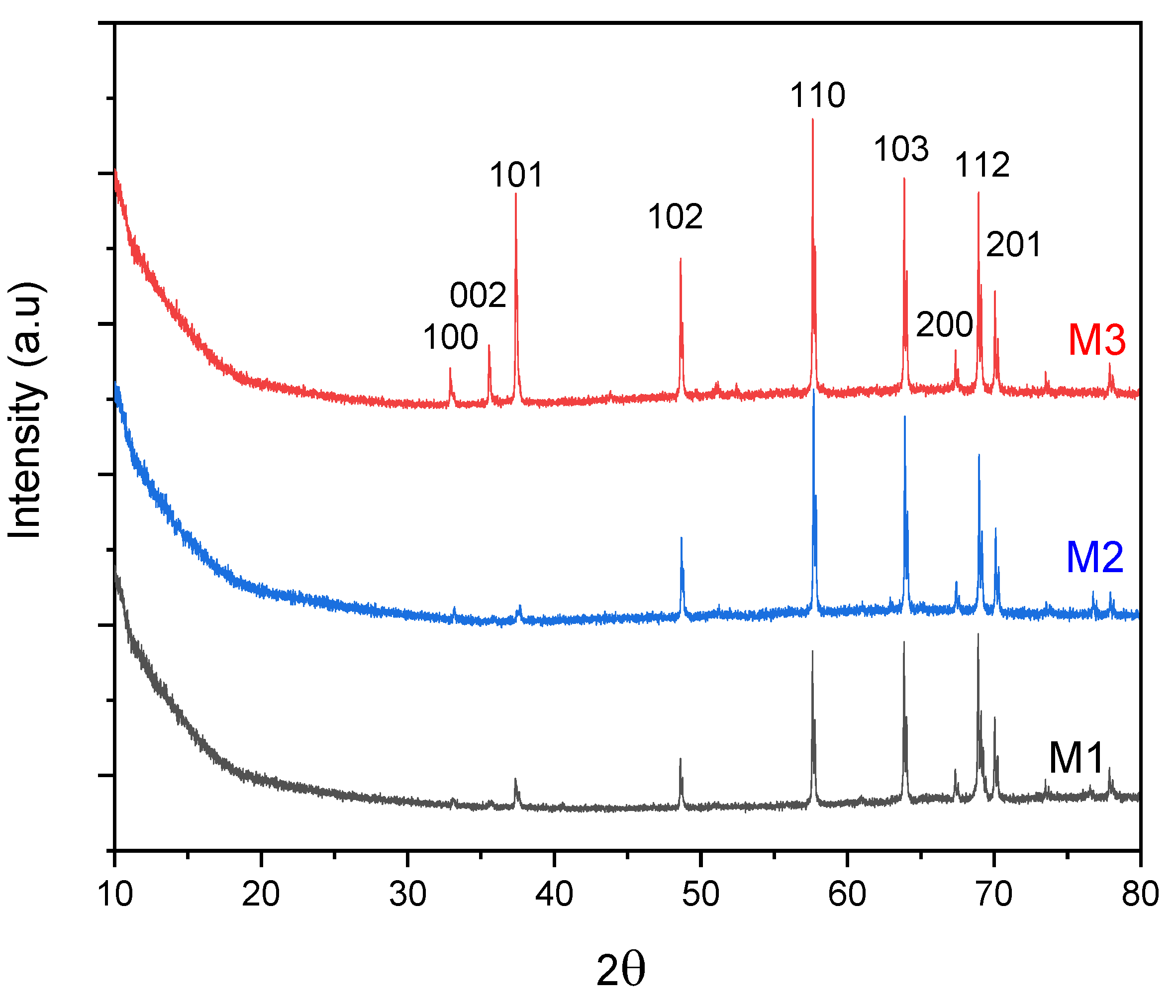
3.1.2. FTIR Analysis
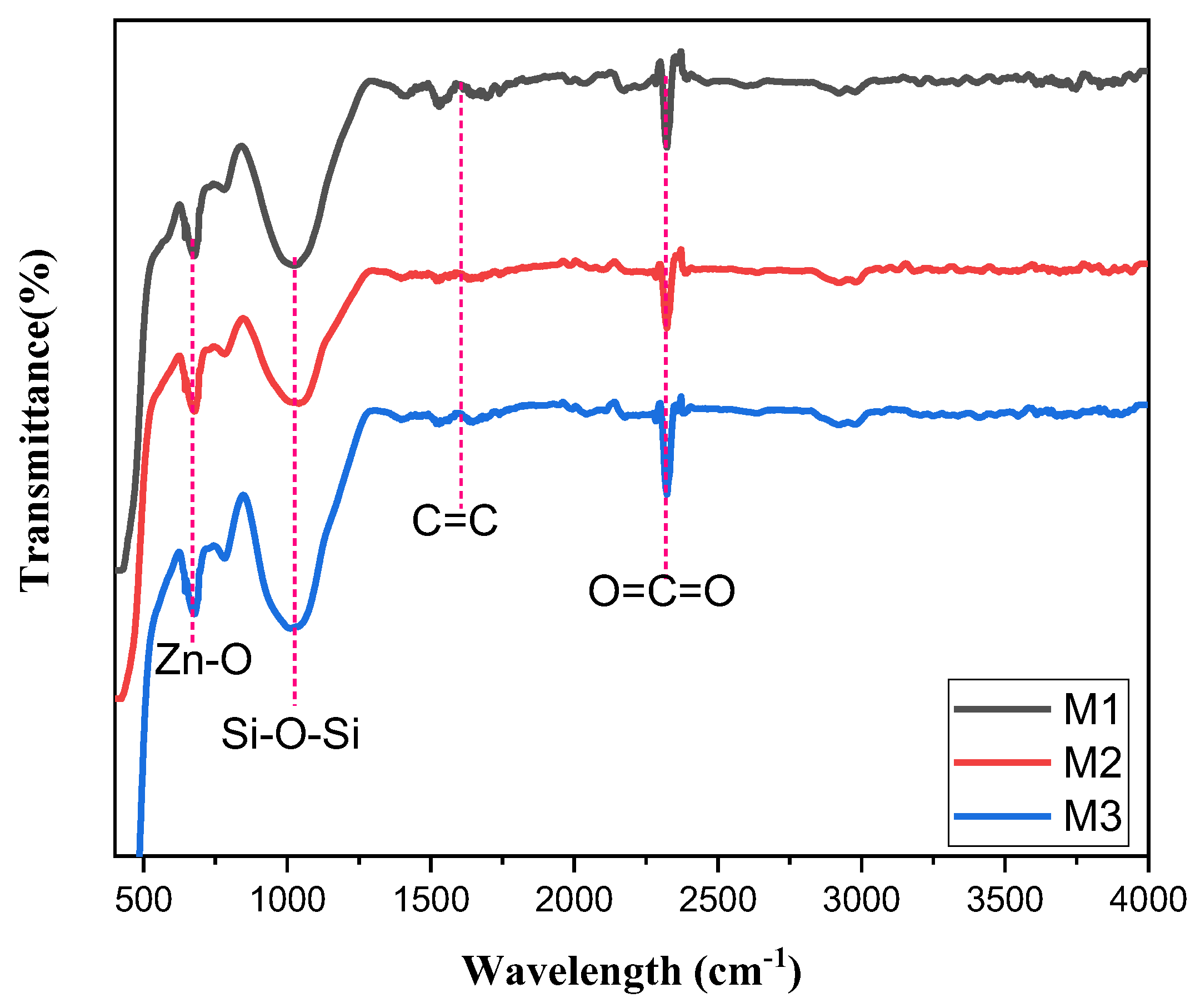
3.1.3. SEM Characterisation


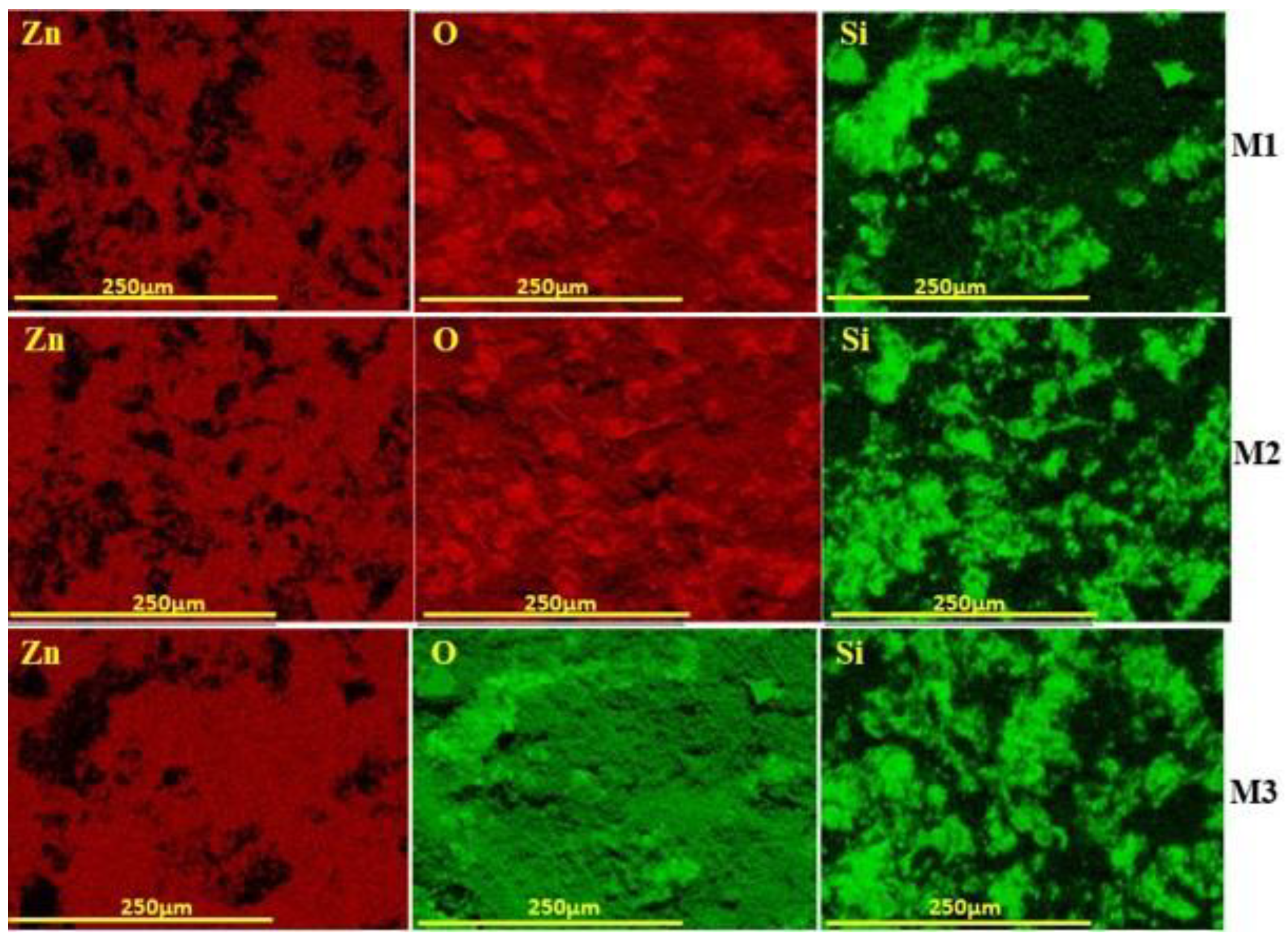
3.1.4. Determination of Membrane Permeability
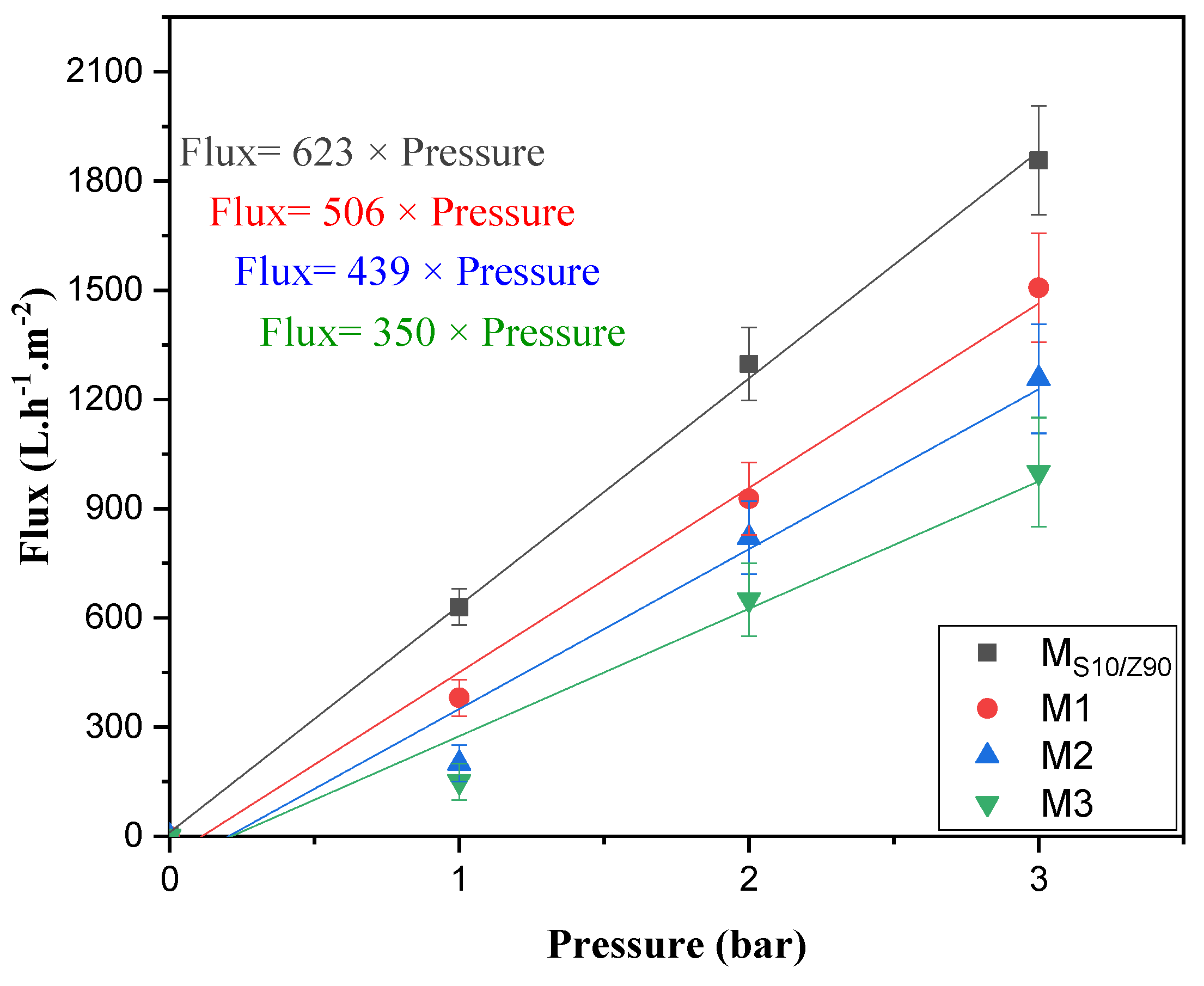
3.2. Photocatalytic Activity
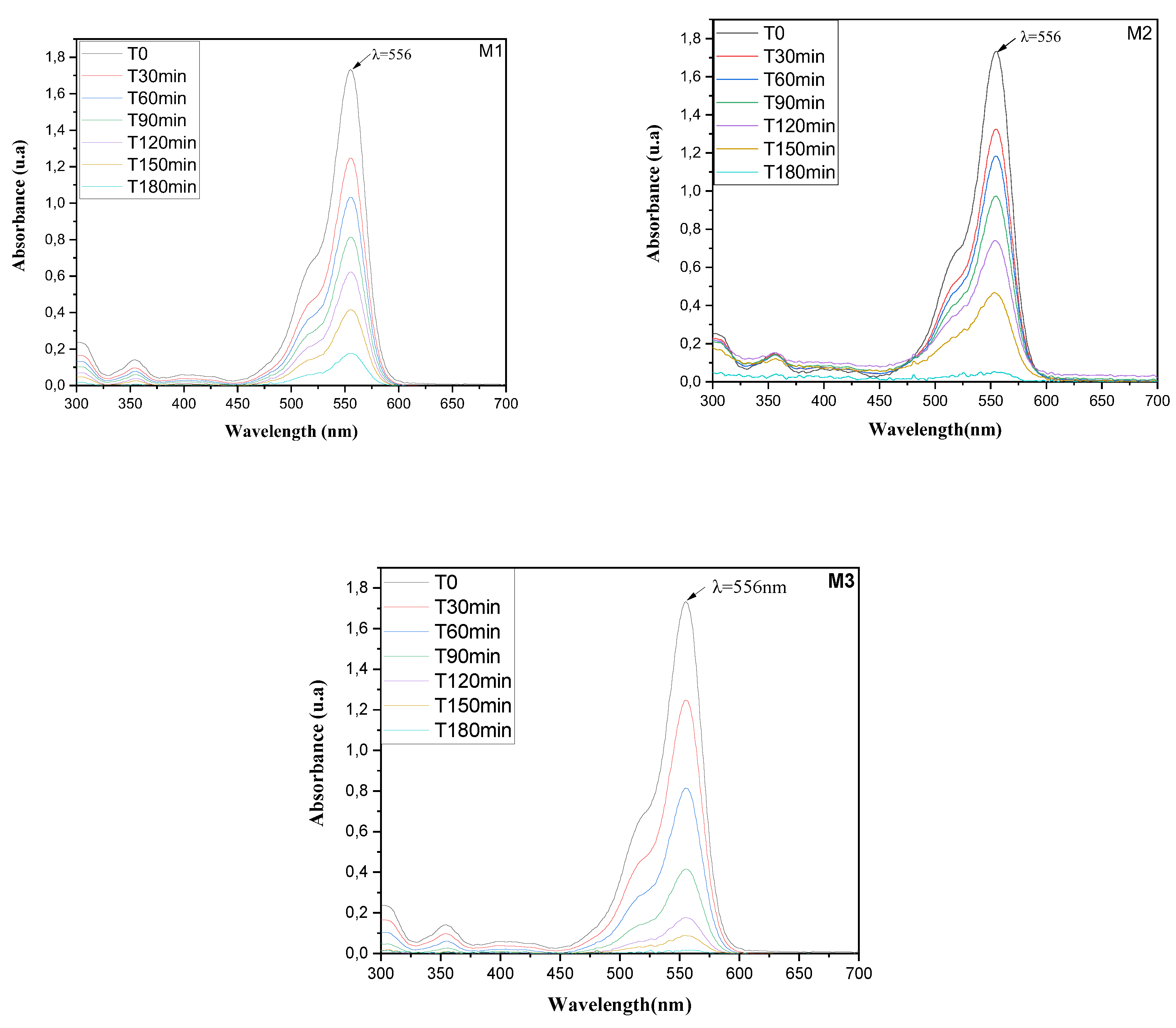
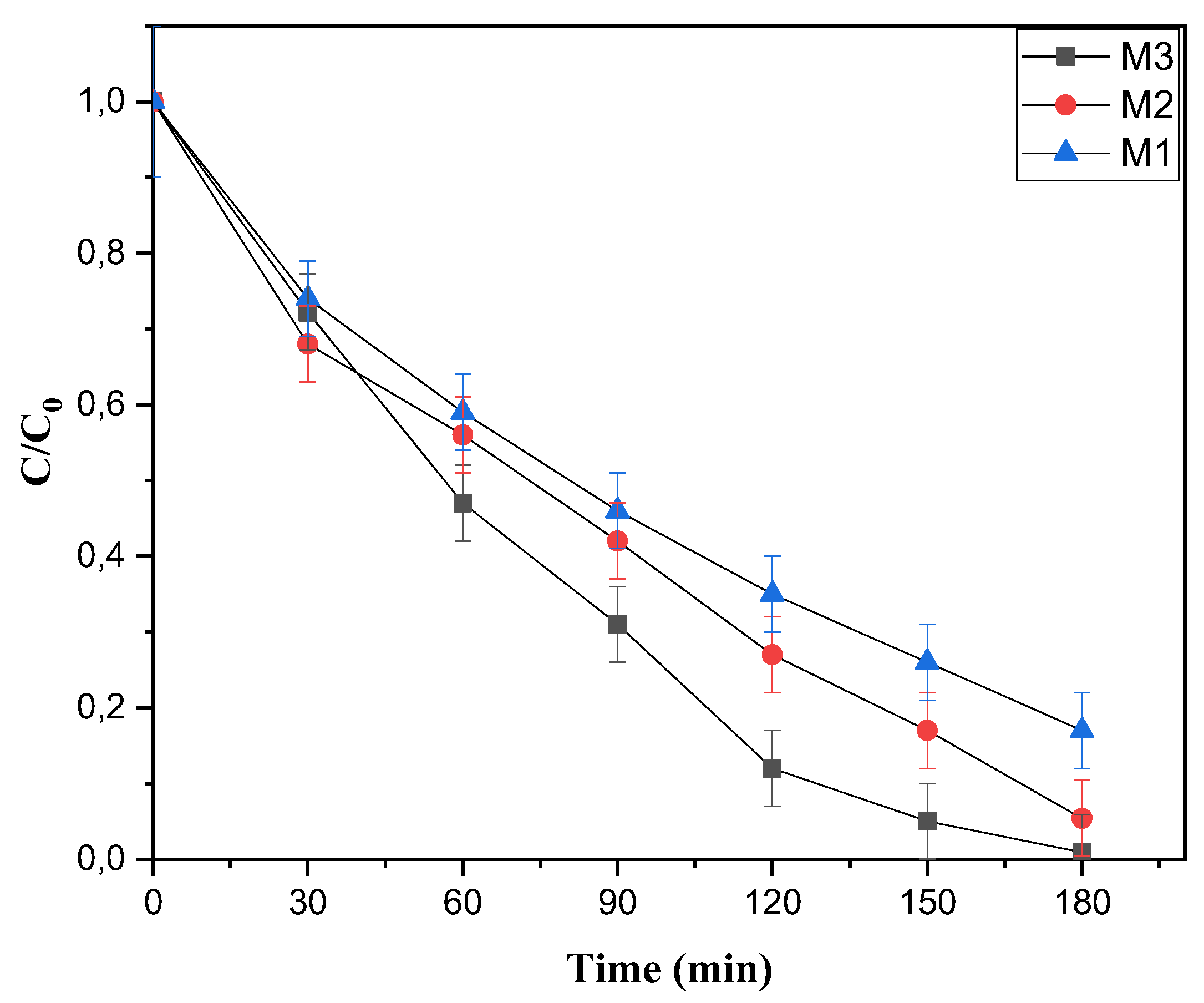
3.3. Kinetic Study
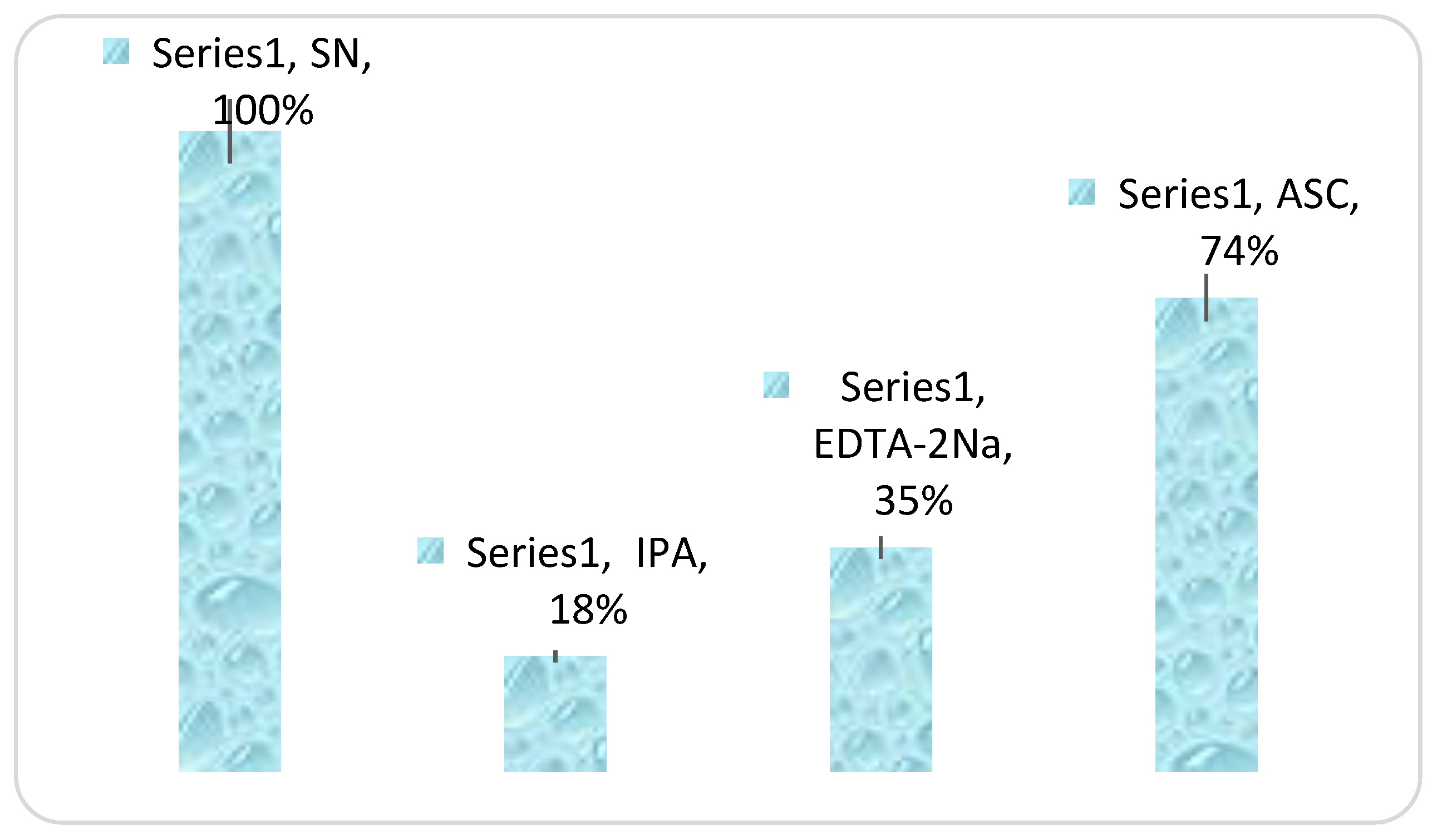
3.3. Self-Cleaning

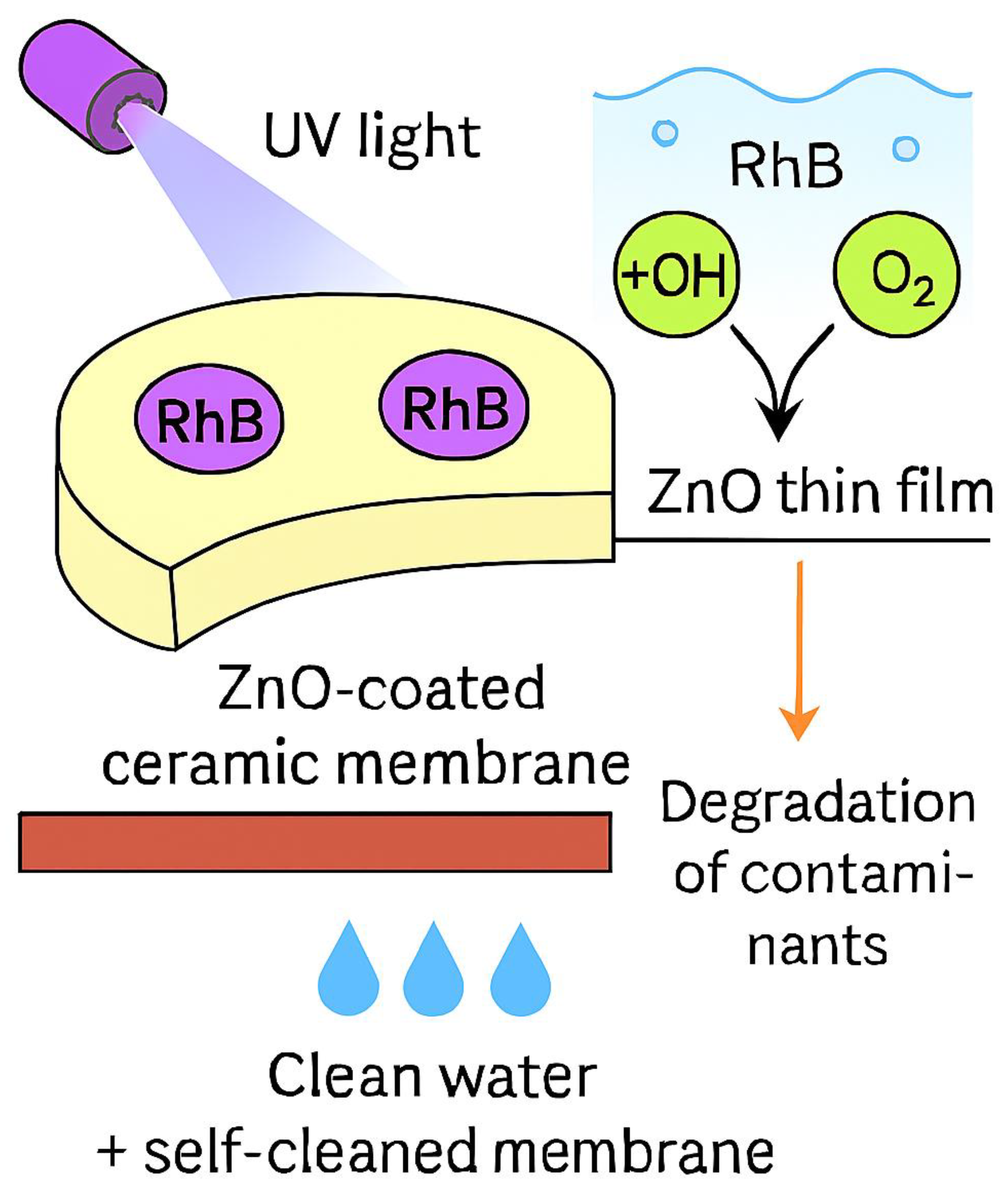
3.5. Rhodamine B Degradation Mechanism Using M3 Membrane
4. Comparative Study
5. Conclusions
Funding
Data availability
References
- Espinosa, M.; Afonso, C.; Saraiva, B.; Vione, D.; Fernandes, A. Textile Wastewater Treatment by Membrane and Electrooxidation Processes: A Critical Review. Clean Technol. 2026, 8, 9. [Google Scholar] [CrossRef]
- Khan, W.U.; Ahmed, S.; Dhoble, Y.; Madhav, S. A Critical Review of Hazardous Waste Generation from Textile Industries and Associated Ecological Impacts. J. Indian Chem. Soc. 2023, 100, 100829. [Google Scholar] [CrossRef]
- Aouay, F.; Attia, A.; Dammak, L.; Ben Amar, R.; Deratani, A. Activated Carbon Prepared from Waste Coffee Grounds: Characterization and Adsorption Properties of Dyes. Materials 2024, 17, 3078. [Google Scholar] [CrossRef]
- Oladimeji, T.E.; Oyedemi, M.; Emetere, M.E.; Agboola, O.; Adeoye, J.B.; Odunlami, O.A. Review on the Impact of Heavy Metals from Industrial Wastewater Effluent and Removal Technologies. Heliyon 2024, 10, e40370. [Google Scholar] [CrossRef]
- Imam, S.; Babamale, H. A Short Review on the Removal of Rhodamine B Dye Using Agricultural Waste-Based Adsorbents. Asian J. Chem. Sci. 2020, 25–37. [Google Scholar] [CrossRef]
- Postai, D.L.; Demarchi, C.A.; Zanatta, F.; Melo, D.C.C.; Rodrigues, C.A. Adsorption of Rhodamine B and Methylene Blue Dyes Using Waste of Seeds of Aleurites Moluccana, a Low Cost Adsorbent. Alex. Eng. J. 2016, 55, 1713–1723. [Google Scholar] [CrossRef]
- Saadi, A.S.; Slimane Ben Ali, D.; Bousba, S. Utilization of a Biosorbent Derived from Plant Residues for the Treatment of Water Contaminated with Rhodamine B: Preparation and Characterization. RSC Adv. 2025, 15, 43038–43052. [Google Scholar] [CrossRef] [PubMed]
- ATTIA, A.; Ahmed, A.B.; Bhattacharyya, S.; Cozzolino, V.; Algieri, C.; Chakraborty, S.; AMAR, R.B. PES/Clay Mixed Matrix Membranes for Efficient Removal of Recalcitrant Chloramphenicol: Experimental and DFT Study. Chem. Eng. J. 2026, 527, 172025. [Google Scholar] [CrossRef]
- Foorginezhad, S.; Zerafat, M.M.; Ismail, A.F.; Goh, P.S. Emerging Membrane Technologies for Sustainable Water Treatment: A Review on Recent Advances. Environ. Sci. Adv. 2025, 4, 530–570. [Google Scholar] [CrossRef]
- Guo, W.; Ngo, H.-H.; Li, J. A Mini-Review on Membrane Fouling. Bioresour. Technol. 2012, 122, 27–34. [Google Scholar] [CrossRef]
- Qi, L.; Li, S.; Duan, L.; Hermanowicz, S.; Ng, H. A Review on Membrane Modification Techniques for Membrane Fouling Control: Mechanisms and Membrane Preparation. Crit. Rev. Environ. Sci. Technol. 2025, 55, 1455–1478. [Google Scholar] [CrossRef]
- Bahrouni, J.; Aloulou, H.; Attia, A.; Dammak, L.; Amar, R.B. Surface Modification of a Zeolite Microfiltration Membrane: Characterization and Application to the Treatment of Colored and Oily Wastewaters. Chem. Afr. 2024, 7, 4513–4527. [Google Scholar] [CrossRef]
- Sak, G.; Taşar, Ş.; Dursun, G. Organic Pollutant Degradation Through Photocatalysis: Progress, Challenges, and Sustainable Solutions (Mini Review). Appl. Sci. 2026, 16, 204. [Google Scholar] [CrossRef]
- Karim, N.; Kyawoo, T.; Jiang, C.; Ahmed, S.; Tian, W.; Li, H.; Feng, Y. Fenton-like Degradation of Methylene Blue on Attapulgite Clay Composite by Loading of Iron–Oxide: Eco-Friendly Preparation and Its Catalytic Activity. Materials 2024, 17, 2615. [Google Scholar] [CrossRef]
- Zhang, D.; Yang, X.; Wang, T.; Wang, H.; Cao, S.; Shi, H. Efficient Adsorption of Typical Antibiotics by Petroleum Coke-Based Ultra-Large Surface Porous Carbon Materials: Mechanism and Cost Analysis. Chem. Eng. Res. Des. 2025, 222, 557–572. [Google Scholar] [CrossRef]
- Ibrahim, I.; Elseman, A.M.; Sadek, H.; Eliwa, E.M.; Abusaif, M.S.; Kyriakos, P.; Belessiotis, G.V.; Mudgal, M.M.; Abdelbasir, S.M.; Elsayed, M.H.; et al. Membrane-Based Photocatalytic and Electrocatalytic Systems: A Review. Catalysts 2025, 15, 528. [Google Scholar] [CrossRef]
- Baig, U.; Al-Kuhaili, M.F.; Dastageer, M.A. Photo-Responsive Zinc Oxide-Coated Alumina Ceramic Membrane with Super-Wettable and Self-Cleaning Features Fabricated by Single Step RF Magnetron Sputtering for Oily Water Treatment. Process Saf. Environ. Prot. 2023, 175, 541–553. [Google Scholar] [CrossRef]
- Kuang, C.; Li, Y.; Liu, D.; Li, Y.; Sun, D.; Chen, J.; Donghai, D.; Xiao, G. An Al2O3@ZnO Membrane for Oil-in-Water Emulsion Separation with Photocatalytic Regeneration Prepared via a Simple Deposition Route. J. Water Process Eng. 2024, 67, 106254. [Google Scholar] [CrossRef]
- Wei, J.; Nian, P.; Wang, Y.; Wang, X.; Wang, Y.; Xu, N.; Wei, Y. Preparation of Superhydrophobic-Superoleophilic ZnO nanoflower@SiC Composite Ceramic Membranes for Water-in-Oil Emulsion Separation. Sep. Purif. Technol. 2022, 292, 121002. [Google Scholar] [CrossRef]
- Huang, A. Fabrication of Zinc Oxide Nanostructure Coated Membranes for Efficient Oil/Water Separation. J. Membr. Sci. 2018. [Google Scholar] [CrossRef]
- Molinari, R.; Pietro, A.; Szymański, K.; Darowna, D.; Mozia, S. Photocatalytic Membrane Reactors for Wastewater Treatment; 2020; pp. 83–116. ISBN 978-0-12-816823-3. [Google Scholar]
- Muscetta, M.; Ganguly, P.; Clarizia, L. Solar-Powered Photocatalysis in Water Purification: Applications and Commercialization Challenges. J. Environ. Chem. Eng. 2024, 12, 113073. [Google Scholar] [CrossRef]
- Rakcho, Y.; Naboulsi, A.; Mansouri, S.; Mouiya, M.; Sehaqui, H.; Bouazizi, A.; Abouliatim, Y.; Benhammou, A.; Abourriche, A.; Alami, J. Sustainable Valorization of Food Waste into a Pore-Forming Agent for Ceramic Membrane Production: Experimental and DFT Studies on Methylene Blue Dye Removal. Sustain. Mater. Technol. 2024, 42, e01181. [Google Scholar] [CrossRef]
- Khmiri, Y.; Attia, A.; Aloulou, H.; Dammak, L.; Baklouti, L.; Ben Amar, R. Preparation and Characterization of New and Low-Cost Ceramic Flat Membranes Based on Zeolite-Clay for the Removal of Indigo Blue Dye Molecules. Membranes 2023, 13, 865. [Google Scholar] [CrossRef]
- Santra, N.; Tudu, M.; Chatterjee, S.; Samanta, A.; Kayal, N. Preparation of ZnO Nanostructures in SiC Ceramic Membrane for Removal of Dyes from Water and Its Antibacterial Activity. Water. Air. Soil Pollut. 2025, 236, 700. [Google Scholar] [CrossRef]
- Khmiri, Y.; Attia, A.; Elboughdiri, N.; Ghernaout, D.; Charcosset, C.; Dammak, L.; Amar, R.B. Preparing Sustainable Membranes Made From Zeolite–Smectite for Treating Textile Wastewater and Pulp Industry Wastewater. ChemistrySelect 2024, 9. [Google Scholar] [CrossRef]
- Xiong, Z.; Cao, J.; Yang, D.; Lai, B.; Yang, P. Coagulation-Flocculation as Pre-Treatment for Micro-Scale Fe/Cu/O3 Process (CF-mFe/Cu/O3) Treatment of the Coating Wastewater from Automobile Manufacturing. Chemosphere 2017, 166, 343–351. [Google Scholar] [CrossRef]
- Nandi, P.; Das, D. ZnO-CuxO Heterostructure Photocatalyst for Efficient Dye Degradation. J. Phys. Chem. Solids 2020, 143, 109463. [Google Scholar] [CrossRef]
- Kayani, Z.; Iqbal, M.; Riaz, S.; Zia, R.; Naseem, S. Fabrication and Properties of Zinc Oxide Thin Film Prepared by Sol-Gel Dip Coating Method. Mater. Sci.-Pol. 2014, 33. [Google Scholar] [CrossRef]
- Uğur, Ş.S.; Sarıışık, M.; Aktaş, A.H.; Uçar, M.Ç.; Erden, E. Modifying of Cotton Fabric Surface with Nano-ZnO Multilayer Films by Layer-by-Layer Deposition Method. Nanoscale Res. Lett. 2010, 5, 1204. [Google Scholar] [CrossRef]
- Zerdali, M.; Hamzaoui, S.; Teherani, F.H.; Rogers, D. Growth of ZnO Thin Film on SiO2/Si Substrate by Pulsed Laser Deposition and Study of Their Physical Properties. Mater. Lett. 2006, 60, 504–508. [Google Scholar] [CrossRef]
- Oliaee, J.; Dehghany, M.; McKellar, A.; Moazzen-Ahmadi, N. High Resolution Infrared Spectroscopy of Carbon Dioxide Clusters up to (CO2)(13). J. Chem. Phys. 2011, 135, 044315. [Google Scholar] [CrossRef] [PubMed]
- Na, J.-S.; Gong, B.; Scarel, G.; Parsons, G.N. Surface Polarity Shielding and Hierarchical ZnO Nano-Architectures Produced Using Sequential Hydrothermal Crystal Synthesis and Thin Film Atomic Layer Deposition. ACS Nano 2009, 3, 3191–3199. [Google Scholar] [CrossRef]
- O’Brien, S.; Nolan, M.; Çopuroğlu, M.; Hamilton, J.; Povey, I.; Pereira, L.; Martins, R.; Fortunato, E.; Pemble, M. Zinc Oxide Thin Films: Characterization and Potential Applications. Thin Solid Films 2010, 518, 4515–4519. [Google Scholar] [CrossRef]
- Venables, J. Introduction to Surface and Thin Film Processes. Introd. Surf. Thin Film Process. John Venables Pp 392 ISBN 0521785006 Camb. UK Camb. Univ. Press Novemb. 2000 2000, 145–146. [Google Scholar] [CrossRef]
- Özgür, Ü.; Alivov, Y.; Liu, C.; Teke, A.; Reshchikov, M.; Dogan, S.; Avrutin, V.; Cho, S.-J.; Morkoç, H. A Comprehensive Review of ZnO Materials and Devices. J. Appl. Phys. 2005, 98, 041301–041301. [Google Scholar] [CrossRef]
- Salih, A.; Irvine, C.; Matar, F.; Aditya, L.; Nghiem, L.; Ton, C. Photocatalytic Self-Cleansing ZnO-Coated Ceramic Membranes for Preconcentrating Microalgae. J. Membr. Sci. 2025, 718, 123700. [Google Scholar] [CrossRef]
- Roshani, R.; Ardeshiri, F.; Peyravi, M.; Jahanshahi, M. Highly Permeable PVDF Membrane with PS/ZnO Nanocomposite Incorporated for Distillation Process. RSC Adv 2018, 8, 23499–23515. [Google Scholar] [CrossRef]
- Shaban, M.; Abdallah, S.; Khalek, A.A. Characterization and Photocatalytic Properties of Cotton Fibers Modified with ZnO Nanoparticles Using Sol–Gel Spin Coating Technique. Beni-Suef Univ. J. Basic Appl. Sci. 2016, 5, 277–283. [Google Scholar] [CrossRef]
- Alrebdi, T.A.; Ahmed, H.A.; Alkallas, F.H.; Pashameah, R.A.; Alrefaee, S.H.; Alsubhe, E.; Mostafa, A.M.; Mwafy, E.A. Ag/ZnO Thin Film Nanocomposite Membrane Prepared by Laser-Assisted Method for Catalytic Degradation of 4-Nitrophenol. Membranes 2022, 12, 732. [Google Scholar] [CrossRef]
- Salih, A.K.; Aditya, L.; Matar, F.; Nghiem, L.D.; Ton-That, C. Improved Flux and Anti-Fouling Performance of a Photocatalytic ZnO Membrane on Porous Stainless Steel Substrate for Microalgae Harvesting. J. Membr. Sci. 2024, 694, 122405. [Google Scholar] [CrossRef]
- Sawunyama, L.; Oyewo, O.A.; Makgato, S.S.; Bopape, M.F.; Onwudiwe, D.C. TiO2–ZnO Functionalized Low-Cost Ceramic Membranes from Coal Fly Ash for the Removal of Tetracycline from Water under Visible Light. Discov. Nano 2025, 20, 1. [Google Scholar] [CrossRef]
- Mukherjee, S.; Ghati, A.; Paul, G. An Ultraviolet–Visible Spectrophotometric Approach to Establish a Method for Determining the Presence of Rhodamine B in Food Articles. ACS Food Sci. Technol. 2021, 1. [Google Scholar] [CrossRef]
- Hu, X.; Mohamood, T.; Ma, W.; Chen, C.; Zhao, J. Oxidative Decomposition of Rhodamine B Dye in the Presence of VO2+ and/or Pt(IV) under Visible Light Irradiation: N-Deethylation, Chromophore Cleavage, and Mineralization. J. Phys. Chem. B 2006, 110, 26012–26018. [Google Scholar] [CrossRef]
- Percivalle, N.M.; Carofiglio, M.; Hernández, S.; Cauda, V. Ultra-Fast Photocatalytic Degradation of Rhodamine B Exploiting Oleate-Stabilized Zinc Oxide Nanoparticles. Discov. Nano 2024, 19, 126. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, J.-M. Photocatalysis Fundamentals Revisited to Avoid Several Misconceptions. Appl. Catal. B Environ. 2010, 99, 461–468. [Google Scholar] [CrossRef]
- Feng, D.-M.; Zhu, Y.-P.; Chen, P.; Ma, T.-Y. Recent Advances in Transition-Metal-Mediated Electrocatalytic CO2 Reduction: From Homogeneous to Heterogeneous Systems. Catalysts 2017, 7, 373. [Google Scholar] [CrossRef]
- Garcia, J.; Iborra-Clar, M.I.; Miranda, M.-I.; Van der Bruggen, B. Comparison between Hydrophilic and Hydrophobic Metal Nanoparticles on the Phase Separation Phenomena during Formation of Asymmetric Polyethersulphone Membranes. J. Membr. Sci. 2015, 493, 709. [Google Scholar] [CrossRef]
- Chang, N.; Liu, J.; Ji, Y.; Liu, J.; Chen, Y. Kinetic Study of Rhodamine B Degradation of Electro-Catalysis by TiO2/Activated Semi-Coke Composite as Tiny Electrode. J. Sol-Gel Sci. Technol. 2023, 106, 1–15. [Google Scholar] [CrossRef]
- Binazadeh, M.; Rasouli, J.; Sabbaghi, S.; Mousavi, S.M.; Hashemi, S.A.; Lai, C.W. An Overview of Photocatalytic Membrane Degradation Development. Materials 2023, 16, 3526. [Google Scholar] [CrossRef]
- Sarabyar, S.; Farahbakhsh, A.; Tahmasebi, H.; Vaziri, B.; Khosroyar, S. Enhancing Photocatalytic Degradation of Beta-Blocker Drugs Using TiO2 NPs/Zeolite and ZnO NPs/Zeolite as Photocatalysts: Optimization and Kinetic Investigations. Sci. Rep. 2024, 14. [Google Scholar] [CrossRef]
- Kabadayi, O.; Altintig, E.; Ballai, G. Zeolite Supported Zinc Oxide Nanoparticles Composite: Synthesis, Characterization, and Photocatalytic Activity for Methylene Blue Dye Degradation. Desalination Water Treat. 2024, 319, 100433. [Google Scholar] [CrossRef]
- Cahyanti, R.; Sumari, S.; Fajaroh, F.; Asrori, M.R.; Prakasa, Y. Fe-TiO2/Zeolite H-A Photocatalyst for Degradation of Waste Dye (Methylene Blue) under UV Irradiation. AIMS Mater. Sci. 2023, 10, 40–54. [Google Scholar] [CrossRef]
- Park, K.-H.; Sun, P.-F.; Kang, E.H.; Han, G.D.; Kim, B.J.; Jang, Y.; Lee, S.-H.; Shim, J.H.; Park, H.-D. Photocatalytic Anti-Biofouling Performance of Nanoporous Ceramic Membranes Treated by Atomic Layer Deposited ZnO. Sep. Purif. Technol. 2021, 272, 118935. [Google Scholar] [CrossRef]
- Mousa, S.A.; Abdallah, H.; Khairy, S.A. Low-Cost Photocatalytic Membrane Modified with Green Heterojunction TiO2/ZnO Nanoparticles Prepared from Waste. Sci. Rep. 2023, 13, 22150. [Google Scholar] [CrossRef]
- Khmiri, Y.; Attia, A.; Jallouli, N.; Chabanon, E.; Charcosset, C.; Mahouche-Chergui, S.; Dammak, L.; Algieri, C.; Chakraborty, S.; Amar, R.B. Synthesis of a Cost-Effective ZnO/Zeolite Photocatalyst for Paracetamol Removal. Emergent Mater. 2025. [Google Scholar] [CrossRef]
- Khalid, A.; Ahmad, P.; Memon, R.; Gado, L.F.; Khandaker, M.U.; Almukhlifi, H.A.; Modafer, Y.; Bashir, N.; Abida, O.; Alshammari, F.A.; et al. Structural, Optical, and Renewable Energy-Assisted Photocatalytic Dye Degradation Studies of ZnO, CuZnO, and CoZnO Nanostructures for Wastewater Treatment. Separations 2023, 10, 184. [Google Scholar] [CrossRef]
- Patil, S.P.; Shrivastava, V.S.; Sonawane, G.H. Photocatalytic Degradation of Rhodamine 6G Using ZnO-Montmorillonite Nanocomposite: A Kinetic Approach. Desalination Water Treat. 2015, 54, 374–381. [Google Scholar] [CrossRef]
- Attia, A.; Elboughdiri, N.; Ghernaout, D.; Carbonnier, B.; Amar, R.B.; Mahouche-Chergui, S. Enhancing the Generation and Stabilization of ZnO Nanoparticles on Modified Clay with Polyethylenimine to Improve the Photodegradation of Dyes in Textile Wastewater. J. Water Process Eng. 2025, 73, 107711. [Google Scholar] [CrossRef]
- Haounati, R.; Ighnih, H.; Malekshah, R.E.; Alahiane, S.; Alakhras, F.; Alabbad, E.; Alghamdi, H.; Ouachtak, H.; Addi, A.A.; Jada, A. Exploring ZnO/Montmorillonite Photocatalysts for the Removal of Hazardous RhB Dye: A Combined Study Using Molecular Dynamics Simulations and Experiments. Mater. Today Commun. 2023, 35, 105915. [Google Scholar] [CrossRef]
- Saja, S.; Bouazizi, A.; Achiou, B.; Ouaddari, H.; Karim, A.; Ouammou, M.; Aaddane, A.; Bennazha, J.; Younssi, S.A. Fabrication of Low-Cost Ceramic Ultrafiltration Membrane Made from Bentonite Clay and Its Application for Soluble Dyes Removal. J. Eur. Ceram. Soc. 2020, 40, 2453–2462. [Google Scholar] [CrossRef]
- Yadav, D.; Kishore, K.; Bethi, B.; Sonawane, S.; Bhagawan, D. ZnO Nanophotocatalysts Coupled with Ceramic Membrane Method for Treatment of Rhodamine-B Dye Waste Water. Environ. Dev. Sustain. 2018, 20. [Google Scholar] [CrossRef]
- Pradeev Raj, K.; Sadaiyandi, K.; Kennedy, A.; Sagadevan, S. Photocatalytic and Antibacterial Studies of Indium-Doped ZnO Nanoparticles Synthesized by Co-Precipitation Technique. J. Mater. Sci. Mater. Electron. 2017, 28, 19025–19037. [Google Scholar] [CrossRef]
| Dye | Molecular Formula |
Molecular Weight (g.mol-1) |
Wavelength (λmax) (nm) |
Charge | Chemical structure |
|
RhB |
C28H31N2O3Cl |
479.01 |
556 |
Cationic |
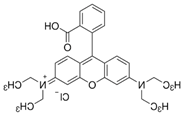 |
| Parameters | Values |
| Water permeability (L.h-1.m-2.bar-1) | 623 |
| Mechanical strength (MPa) | 23 |
| Average pore size (µm) | 0.98 |
| Permeability (%) | 39.2 |
| Membrane | Coating layers |
|---|---|
| M1 | 1 |
| M2 | 2 |
| M3 | 3 |
| Structured membrane | RB concentration |
Performances | Reference |
| Bentonite coated perlite mem- brane |
50 ppm | -Dye removal Efficiency 80.1% Permeability 21 L.m-2.h-1.bar-1 |
[61] |
| ZnO photocatalyst coupled with ceramic Nanoporous membrane (hybrid system) |
500 ppm | Dye removal Efficiency 96 % water flux Permeability 31-21 L.m-2.h-1.bar-1 |
[62] |
| ZnO layered polytetrafluorethylene membrane |
10-100 ppm | Removal Efficiency 42-97 % |
[27] |
| ZnO-Cu0.5 O hetro composite structure (photocatalyst) |
10 ppm | Removal Efficiency 73 % |
[28] |
| 6% In-doped ZnO nano par- ticles (under UV radiation) ––– RB |
20 ppm | -Removal Efficiency 73 % |
[63] |
| ZnO layered porous SiC support |
4-50 ppm | Removal Efficiency 91-95% Permeability 306-205 L.m-2.h-1.bar-1 |
[25] |
| ZnO layered porous support MS10/Z90 |
10 ppm | Removal Efficiency 99.1% |
This work |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





