1. Introduction
At present, the continued expansion of land use for residential and commercial purposes represents one of the main threats to the availability of agricultural land [
1]. This tendency not only reduces the space available for food production but also increases the demand for water resources and contributes to the deterioration of global food security. This scenario is worsened by uncontrolled deforestation and overgrazing, factors that contribute to soil degradation and biodiversity loss, while also increasing the environmental impact of agricultural production systems. Currently, approximately 38% of the Earth's land surface is dedicated to agricultural activities, and 95% of global food production depends directly on land use, whether for livestock or crop production [
2]. However, the United Nations' demographic projections estimate that the world population will reach 8.5 billion by 2030 and could reach 9.7 billion by 2050 [
3], which will imply an unprecedented demand for food, water, and natural resources, and will force a rethinking of current agricultural practices. Nevertheless, the challenge lies not only in feeding more people, but in doing so in the face of two converging threats: increasing urbanization and the global environmental crisis. Water scarcity, for example, interferes with plants' ability to regulate their internal temperature, inducing heat stress. Likewise, increased soil salinity, the presence of heavy metals, and elevated temperatures alter the soil's microbial balance, reducing plants' natural resistance to diseases and pests [
4]. In this context, plant resilience to climate change has become a key factor in ensuring both agricultural sustainability and environmental security [
5]. One of the most widely used strategies to mitigate these effects has been the application of fertilizers and pesticides, which not only meet the nutritional needs of plants but also offer protection against pathogens and adverse conditions. However, the indiscriminate use of these inputs also carries environmental risks, highlighting the urgent need to adopt more sustainable approaches, such as the use of biofertilizers, clean technologies, and regenerative agricultural practices that balance productivity with environmental conservation [
4].
Over the past six decades, the nutritional quality of food has deteriorated due to the reduction of essential minerals and nutraceutical compounds in fruit, vegetables, and staple crops. Some of the causes of this phenomenon globally include the indiscriminate application of mineral fertilizers, a preference for crops with lower nutritional value, the use of high-yield varieties, and agronomic problems stemming from the shift from traditional to more intensive and chemically intensive agriculture. The area dedicated to nutrient-rich crops has decreased due to their low profitability compared to cash crops such as corn, wheat, rice, potatoes, and tomatoes. This has a significant impact on developing and underdeveloped countries, where large segments of the population suffer from weakened immune systems, severe malnutrition, and multiple nutritional deficiencies. These problems are intensified by poor diets and a lack of awareness of the benefits of traditional nutrient-rich diets, as well as by the abandonment of ecological and sustainable agricultural practices [
6].
Ensuring food security involves not only guaranteeing access to food, but also meeting the growing nutritional needs of an expanding population. Although food production has increased in recent decades, this growth has not been sufficient to keep pace with population growth. Global food demand is projected to increase between 35% and 56% by 2050. This gap between supply and demand has led to nutritional inequality: while one in eight adults suffers from obesity, more than 800 million people suffer from malnutrition, and 2.4 billion have limited access to food. The COVID-19 pandemic aggravated the situation, reversing previous progress and hindering the achievement of the Sustainable Development Goals related to food security [
7].
Leguminous plants have high nutritional and functional value, with proven health benefits and contributions to the prevention of chronic diseases. Their composition includes bioactive compounds such as proteins, dietary fiber, resistant starch, polyphenols, saponins, lectins, and phytic acids, which contribute to antioxidant, prebiotic, and metabolic-regulating effects [
8]. These plants are also notable for their contribution of B vitamins, vitamin C, and minerals such as potassium, calcium, magnesium, zinc, copper, and iron, along with essential fatty acids like linoleic and oleic acids. Their low sodium content and high nutrient density make them a key food for promoting healthy diets. Several studies indicate that regular consumption of leguminous plants helps reduce the risk of chronic diseases such as obesity, type 2 diabetes, cardiovascular disease, and certain types of cancer by improving parameters such as blood pressure, glucose metabolism, and lipid profile [
9].
1.1. Application of Organic Fertilizer
Organic fertilizers increase soil fertility and are free of harmful substances, unlike inorganic fertilizers, which add heavy metals and carcinogens that are absorbed by plants and inevitably enter the human food chain. Similarly, excess of some elements tends to cause illness and poisoning, such as arsenic, mercury, chromium, nickel, lead, cadmium, zinc, and iron. Some species of seaweed are nitrogen-fixing and phosphate-solubilizing agents because they contain live microorganisms, restore pH to normal, and improve the organic content in clay and sandy soils [
10,
11]. The chemical composition of microalgae extract includes carbohydrates, proteins, lipids, vitamins, micronutrients, macronutrients, and phytohormones, including auxins, cytokinins, ethylene, abscisic acid, and gibberellins; this makes them useful for reducing stressful situations that affect crop growth and development with a limited negative impact [
12], making them a versatile and low-emission resource of great importance. Due to increased agricultural production, efforts are underway to guarantee crop quality and yield, thereby increasing production and preserving the shelf life of high-demand products. This has led to an increase in the use of biofertilizers or bio-stimulants to improve the morphological and molecular responses of crops by enhancing germination and the development of seedlings, shoots, and root systems in vegetables, cereals, leguminous plants, and other crops, thus generating greater productivity [
11].
Ascophyllum nodosum is considered a brown algae and its extract has the capacity to improve plant growth and agricultural productivity because it contains bioactive compounds that increase plant tolerance to biotic and abiotic stress by regulating molecular, physiological, and biochemical processes [
13]. Existing studies [
13,
14] indicate improvement in fruit production using
A. nodosum extract, applied as a spray on the foliage, improving photosynthesis, increasing fruit ripening, mitigating stress from low temperatures, lack of water [
15] and decreasing the presence of lesions and number of nematode eggs [
16]; in addition, it increases the number of leaves, stem diameter and leaf area. In broccoli crops, they reduce the early occurrence of white lesion eruptions caused by
Albugo candida [
17]; in bean pods, they generate higher pod quality and yield, increasing their nutritional value and chemical composition [
18]; in corn crops [
19], they improve potassium availability in the soil, increase soil water storage, and enhance soil enzyme activity. In oat crops, they generate a greater number and weight of grains [
20]. The generation of bio-stimulants using microalgae in conjunction with other organic products such as fulvic acids promote plant recovery after water stress, reducing yield losses. Furthermore, these bio-stimulants result in higher photosynthetic rates, more efficient mechanisms for dissipating excess energy, and greater antioxidant enzyme activity, thus favoring leaf hydration under dry soil conditions, root growth, shoot development, and the absorption of nutrients and water [
21]. Similarly, morphological and physiological responses in tomato plants increased with the application of
A. Nodosum extract, regardless of the water deficiency present [
22], and molecular responses to oxidative stress were suppressed in tomato and pepper crops [
23].
Consequently, it can be emphasized that biofertilizers reduce pollution by minimizing the filtering of nitrates and phosphates into water bodies, promote soil microbial biodiversity, which improves soil structure and fertility, and decrease dependence on agrochemicals, aiding in the regeneration of degraded soils. Furthermore, biofertilizers provide essential nutrients and promote microbial activity, leading to better crop growth and development through atmospheric nitrogen fixation, phosphorus solubilization, and phytohormone production. The use of biofertilizers represents an economically viable option for producers, as their cost is lower than chemical fertilization, generating greater profitability while respecting environmental regulations. Thus, the application of biofertilizers results in healthier food with higher nutrient content in crops and less accumulation of heavy metals, thereby reducing contamination in the food chain.
1.2. Aeroponic Crops: A Sustainable Solution for Future Agriculture
Aeroponic crops have emerged as an innovative and sustainable solution for food production in a world where the availability of farmable land and water is increasingly limited. This system is based on growing plants without soil using a nutrient-rich solution applied directly to the roots, offering benefits in terms of efficiency, sustainability, and productivity [
24,
25]. Another benefit of aeroponic crops is the significant water savings; aeroponics uses up to 95% less water, making it an ideal alternative for water-stressed regions. Furthermore, this method minimizes the use of pesticides and fertilizers, as the plants grow in a controlled environment, thus reducing environmental impact and improving the quality of the food produced.
An alternative to the use of chemical fertilizers is liquid organic fertilizers, which provide nutrients more quickly and safely, improving the physiological activities of plants over time without compromising the environment [
26]. In short, the combination of organic fertilizers with aeroponics allows for cultivation in urban environments, such as vertical greenhouses or modular indoor systems, optimizing the use of available space and promoting sustainable urban agriculture. This not only contributes to food security in densely populated cities but also reduces the carbon footprint by decreasing the need to transport food from rural areas [
27]. Furthermore, aeroponic cultivation accelerates plant growth and increases yield compared to traditional methods [
24].
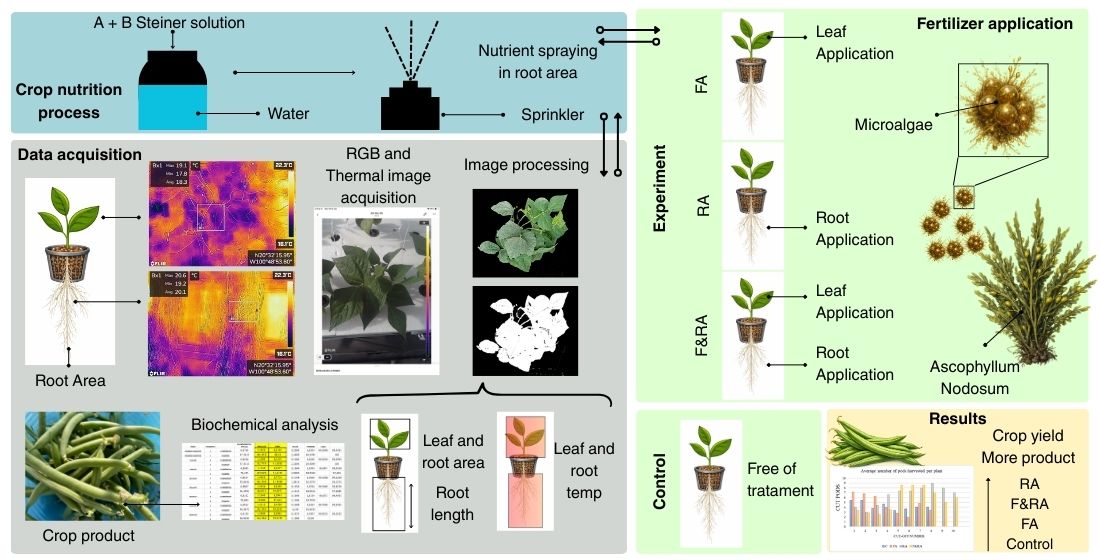
This article presents an experimental design based on three test crops and a control crop of
Phaseolus vulgaris L. var. Opus (green bean) that were studied to evaluate the effect of applying a biofertilizer based on
Ascophyllum nodosum microalgae extract during the vegetative development and fruiting stages and its influence on agronomic and nutritional variables. This leguminous plant was chosen due to its nutritional relevance, as green beans are widely valued vegetables for their high protein content, making them an essential food for meeting dietary requirements. Furthermore, their adaptability in agriculture and consumption in different regions of the world reinforces their importance as a strategic crop in horticultural production [
28].
2. Materials and Methods
The crops were grown in a controlled environment located at coordinates 20°32'16.0"N 100°48'54.4"W in the city of Celaya, Guanajuato, inside a greenhouse with an aeroponic system. The experimental crop was Phaseolus vulgaris L. var. Opus (green bean), acquired from the Seminis company (Seminis México Vegetables by Bayer®). The objective of the experiment was to evaluate the effect of applying a biofertilizer based on Ascophyllum nodosum (microalgae extract) on three aeroponic crops, including one untreated control crop. The biofertilizer was applied to one crop only in the root area, another in the foliar area, and the third in both the root and foliar areas, in order to evaluate the phenological and productive behavior of the crops.
The factors determined for the experiments were three experimental groups, two with 30 replicates each and one with 5 replicates, along with a control group of 5 replicates for experimental validation. The crops were grown on horizontal aeroponic structures with an automatic fertigation system, set to a timer with cycles of 24 minutes of rest followed by 30 seconds of irrigation. This frequency was maintained in all three experimental groups and the control group. The crops were grown in a greenhouse where variable environmental conditions were recorded. The average temperature reached 19.16 °C during the day and rose to 41.23 °C at sunset. Regarding relative humidity, average values of 37.82% were observed during the day and 5.2% at sunset. Likewise, the recorded light radiation was 5878.42 lux during the day and 2576 x 10 lux at sunset. These environmental parameters were key factors in the phenological development of the plants, as well as in the interpretation of the results obtained from the treatments applied.
During the first 15 days after transplanting from the germination stage to the aeroponic chamber, the plants were fertigated with tap water at a pH of 6.5, supplemented with a diluted Steiner universal nutrient solution at a concentration of 2.5 mL/L. Subsequently, for the remainder of the phenological cycle, the concentration of the nutrient solution was increased to 5 mL/L of tap water, ensuring an adequate supply of nutrients throughout the rest of the cycle.
2.1. Description of the Phenological Cycle
Phaseolus vulgaris L., commonly known as green bean, is a shrubby leguminous plant with a phenological cycle of 90 to 100 days from planting to harvest. Factors such as temperature, relative humidity, and radiation significantly influence the crop's phenological development. It is a source of protein that meets the caloric needs of the world's population [
29]. Its cultivation is divided into two main phases: vegetative and reproductive. The vegetative phase extends from seed sowing to the beginning of flowering. During this period, the plant experiences a rapid increase in dry matter, focusing on the synthesis of new tissues. This growth is influenced by environmental factors such as temperature and the availability of nutrients. The developing pods extract essential nutrients from the plant for their growth and subsequent development. The reproductive phase begins with the appearance of the first flowers and culminates when the fruits reach the necessary maturity for harvesting.
2.2. Application of Biofertilizer
The first application of A. nodosum was carried out in 3 crops at the beginning of flowering (at the beginning of the appearance of flower buds), which occurred 30 days after transplanting in all cases (including the control crop). The second application was carried out 15 days after the first application in all 3 crops. Experiment 1, designated F&RA, consisted of supplying A. nodosum extract to the foliar and root areas of the crop in 30 replicates. The foliar application was carried out by aerial spraying, with a single application followed by a second application 15 days after the first. The root application was performed by diluting the A. nodosum extract directly in the main container holding tap water and the nutrient solution. The extract was completely diluted within 5 days of application, and this process was repeated 30 days after the first application. For Experiment 2, designated FA, also with 30 replicates, the biofertilizer was applied by aerial spraying at the beginning of flowering, with a second application 15 days later. For experiment 3, designated RA, with 5 replicates, the biofertilizer was applied by diluting A. nodosum extract in the container with tap water and the nutrient solution. The extract was completely dissolved 5 days after application, and the process was repeated 30 days after the first application. The experiments involved frequent fertigation of nutrients, meaning application was continuous. For the control crop (C), also with 5 replicates, only constant fertigation of Steiner nutrients was carried out throughout the entire phenological cycle.
3. Results
In order to evaluate the feasibility of using a microalgae-based biofertilizer in aeroponic cultivation of Phaseolus vulgaris L. var. Opus, a comprehensive statistical analysis was conducted on the behavior of agronomic and nutritional variables under different treatments. The Kolmogorov-Smirnov test revealed that most variables did not follow a normal distribution. Consequently, the Kruskal-Wallis test was used to compare the medians of the four data sets. This test identified significant differences in the evaluated variables, providing statistical evidence of the biofertilizer's effects on the vegetative and fruiting stages. In this way a solid framework for analyzing the influence of treatments on the quality and productivity of the experimental crop is established.
This section shows the statistical analysis of the study variables considered in the application of microalgae-based biofertilizer in aeroponic crops of Phaseolus vulgaris L. var. Opus; morphological, yield, and nutritional variables were analyzed. The treatments included foliar application (FA), root application (RA) and combined foliar and root application (F&RA), also considering a control (C) as a reference. The main study variables were yield, plant height, leaf area, fruit weight, circularity, pod length, and pod weight, in addition to characterizing the nutritional profile of the final product by evaluating macronutrients (carbohydrates, sugars, fiber, fat, protein, and water) and micronutrients (calcium, iron, magnesium, manganese, phosphorus, potassium, and zinc). These elements are critical indicators of the crop's nutritional quality, as they determine both the energy contribution and the functional value of the product. Furthermore, a correlation was established between the biofertilizer application method and the improvement in nutrient accumulation compared to the control group. In particular, pod length is considered a key parameter for yield, given the reported direct correlation between longer pods and a greater number of grains per pod, as well as an increase in total fruit weight, which translates into an improvement in overall crop yield.
MATLAB R2019a software was used to evaluate the null hypothesis (H0), which states that the medians of the different groups are statistically equal, against the alternative hypothesis (H1), which states that at least one of the medians differs significantly from the others. In this regard, p-values less than 0.05 indicate that the variations in responses are not due to chance, but rather to the effect of the treatments, supporting the hypothesis that their application influences the evaluated agronomic and nutritional characteristics. On the contrary, p-values greater than this threshold suggest that there are no statistically significant differences between the medians of the groups being compared.
3.1. Product Assessment
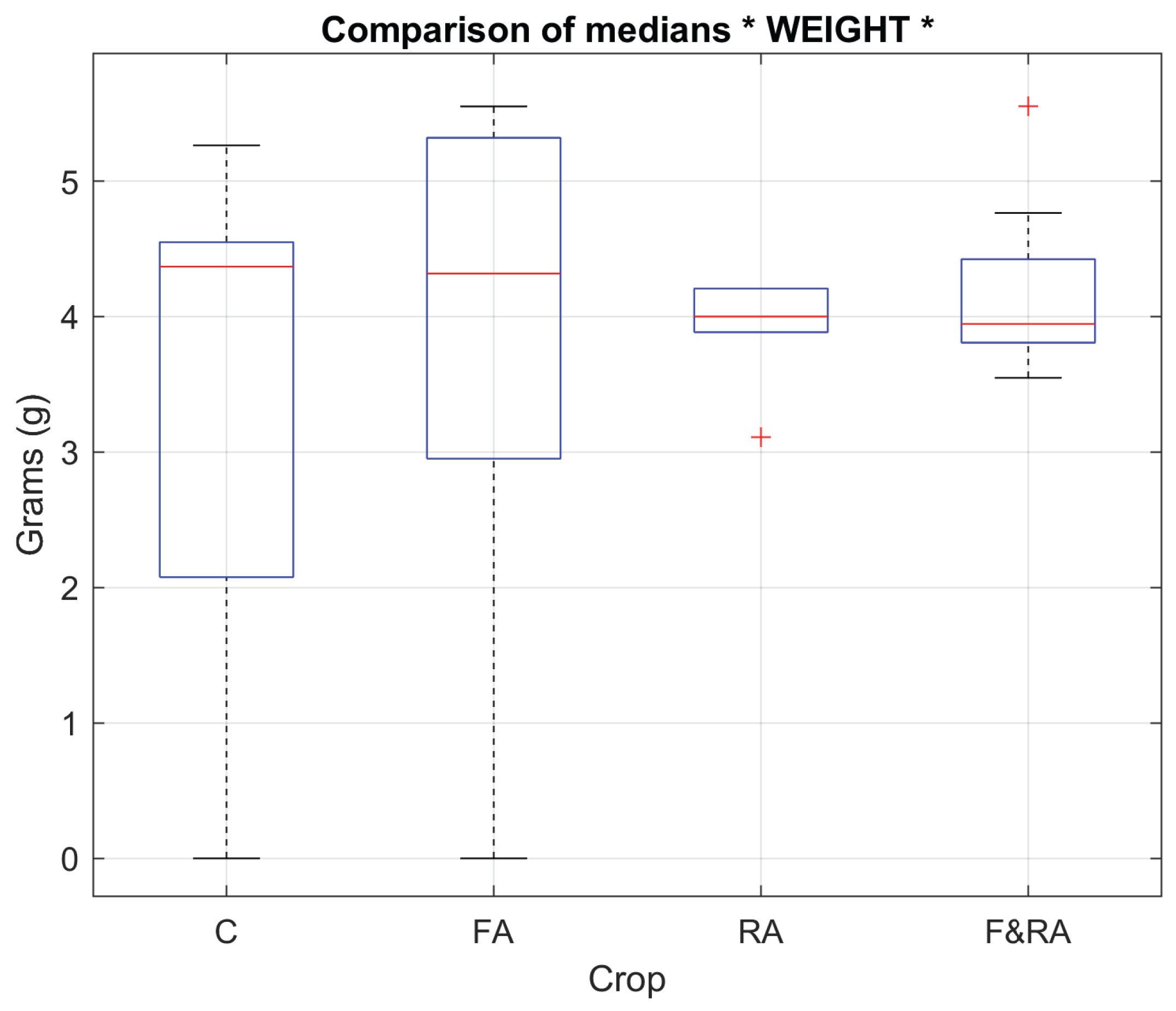
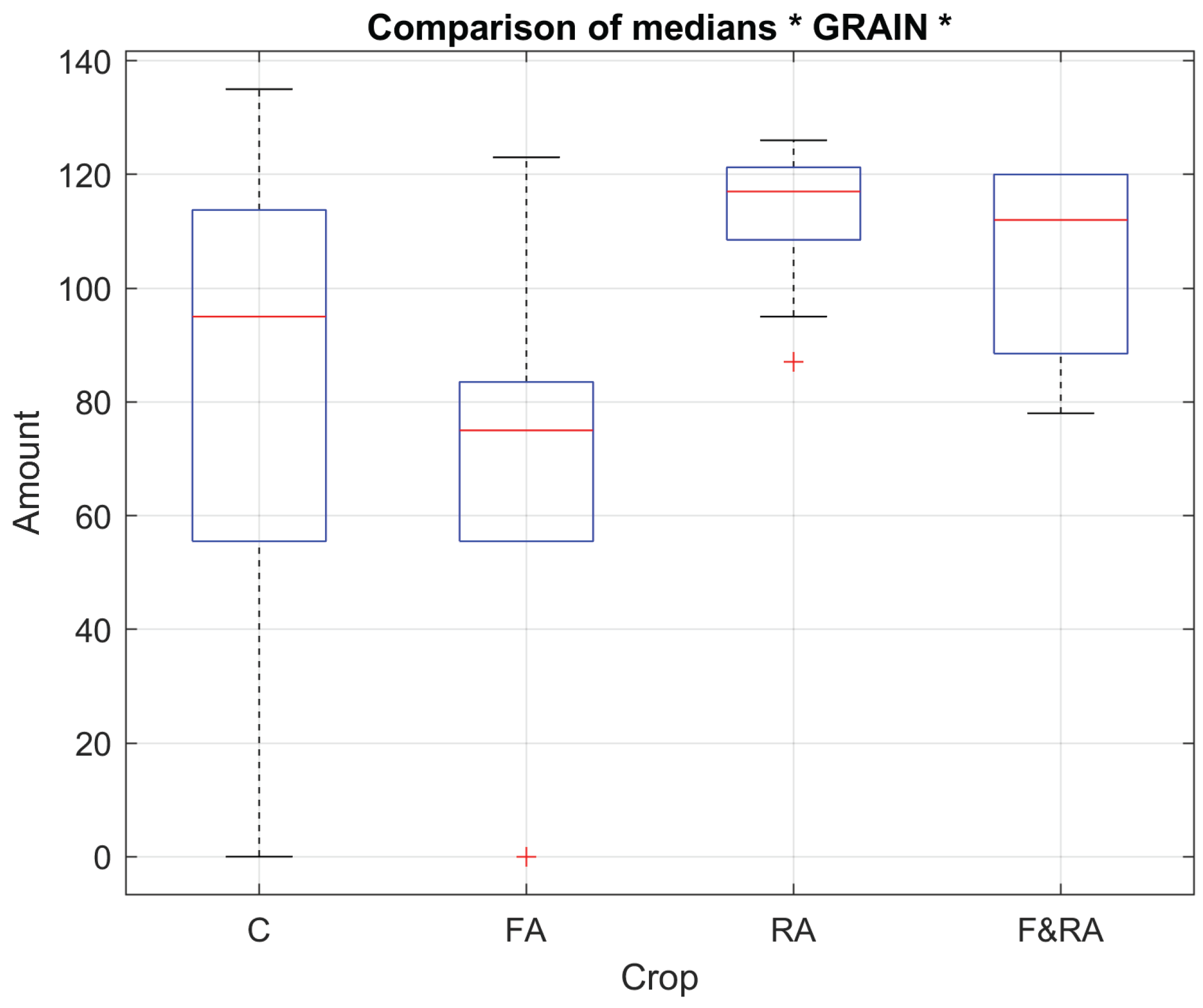
To evaluate product quality in each of the experimental treatments, five key variables were defined: weight, length, width, circularity, and number of grains per pod [
30]. These morphological characteristics represent fundamental indicators of yield and commercial quality of the
Phaseolus vulgaris crop [
31].
Table 1 presents the results of the median comparison test, where statistically significant differences can be observed in some of the evaluated quality variables, reflecting the possible effect of the treatments applied to the experimental crops. In particular, for the variable corresponding to the number of grains, the analysis yielded a
p-value < 0.05, indicating the existence of significant differences between the medians of the experiments and, therefore, a notable influence of the treatment on this characteristic.
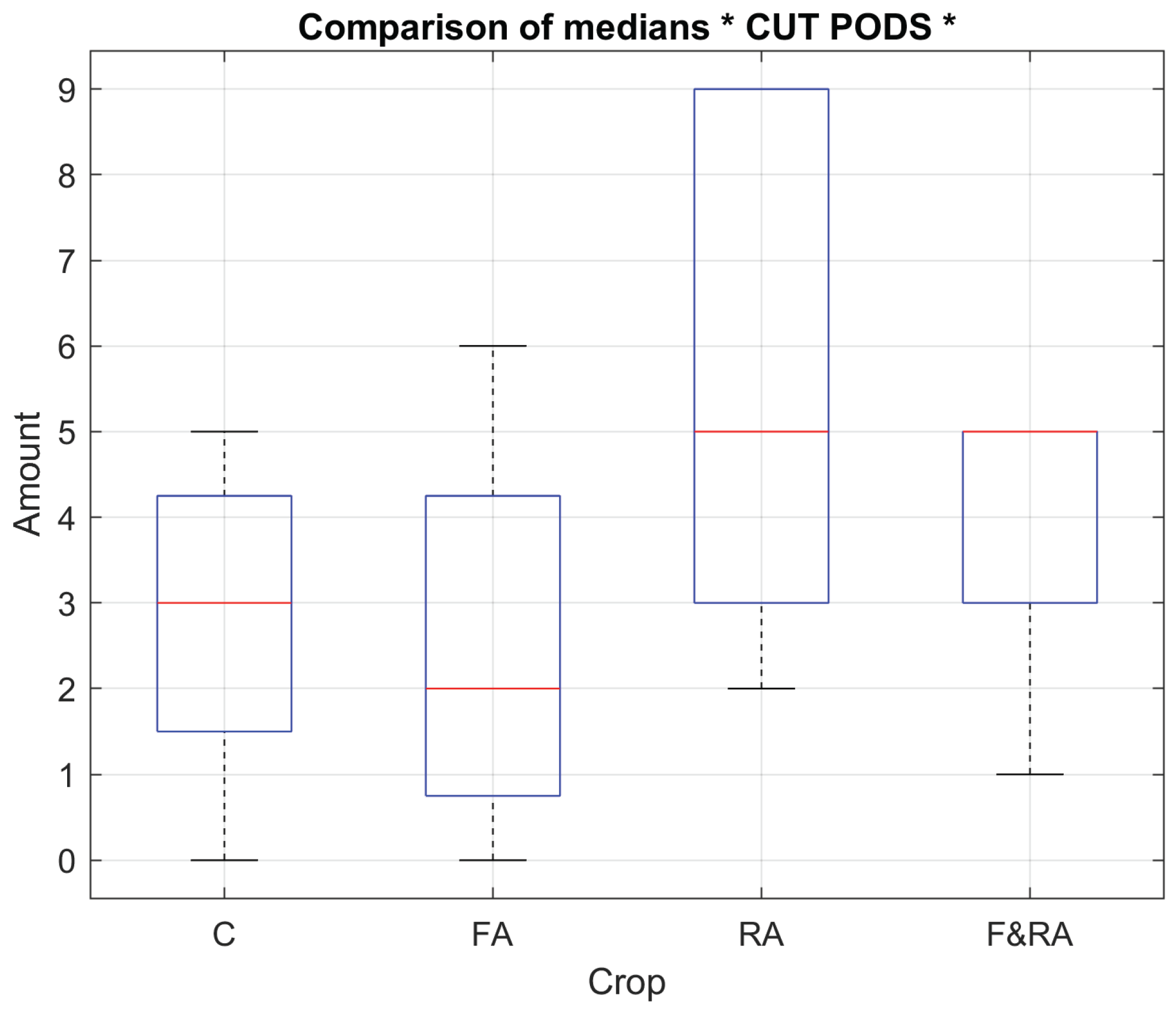
Figure 1 and
Figure 2 show the box plots of 2 of the study variables, the weight of the pods in grams and the number of pods obtained.
3.2. Vegetative State
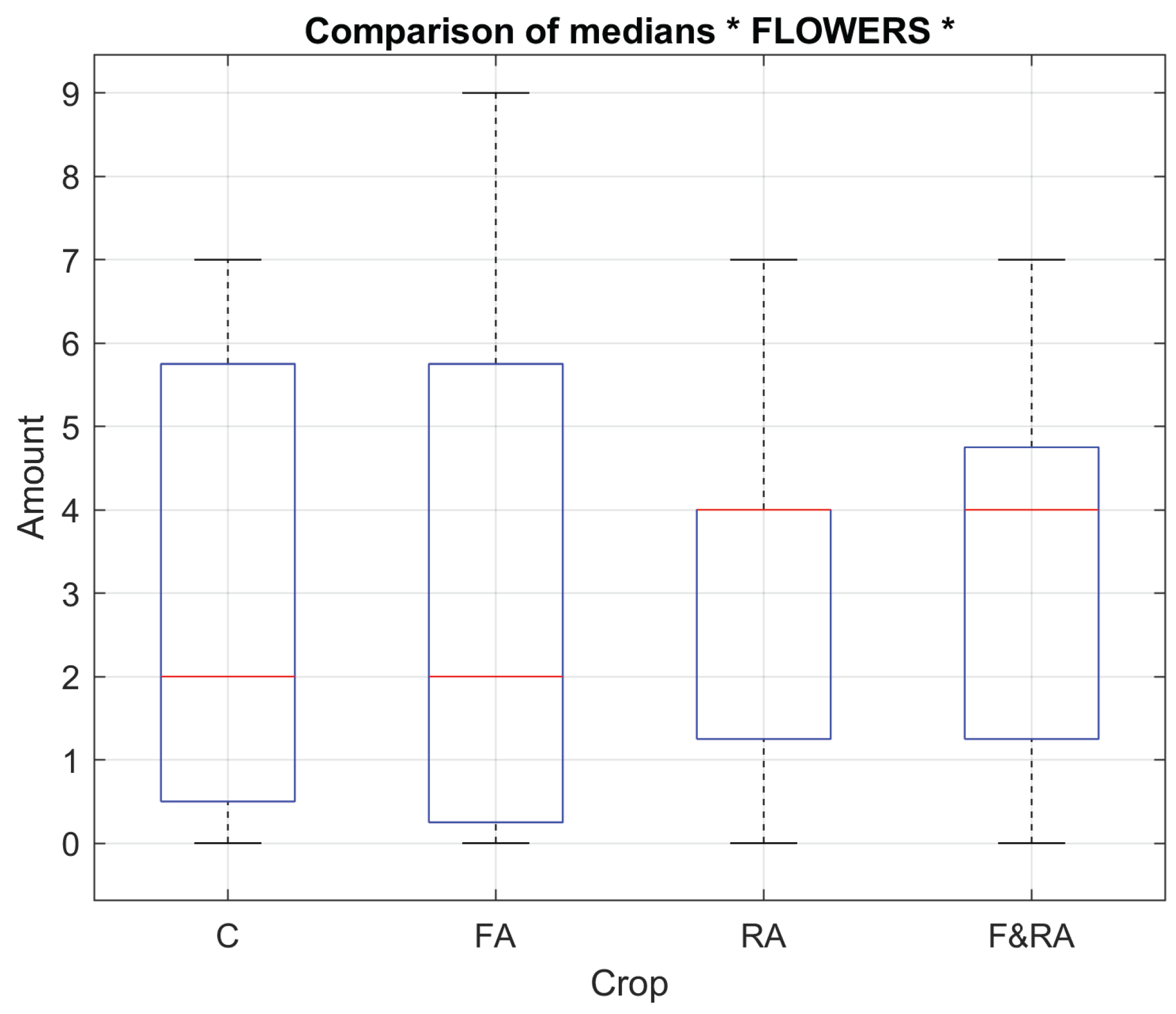
Following the morphological analysis of the plants, variables were defined to quantify aspects associated with the physical quality of the plants' vegetative state during the phenological cycle. These variables were selected for their agronomic relevance and their ability to reflect the differential physiological and morphological behavior among the applied treatments.
The variables evaluated are: number of flowers per plant, number of pods per plant, plant height (cm), leaf temperature (°C), and root temperature (°C). These variables provide information on potential productivity, the structural development of the plants, and their physiological response to the application of the biofertilizer.
Table 2 shows the results of applying the Kruskal-Wallis test.
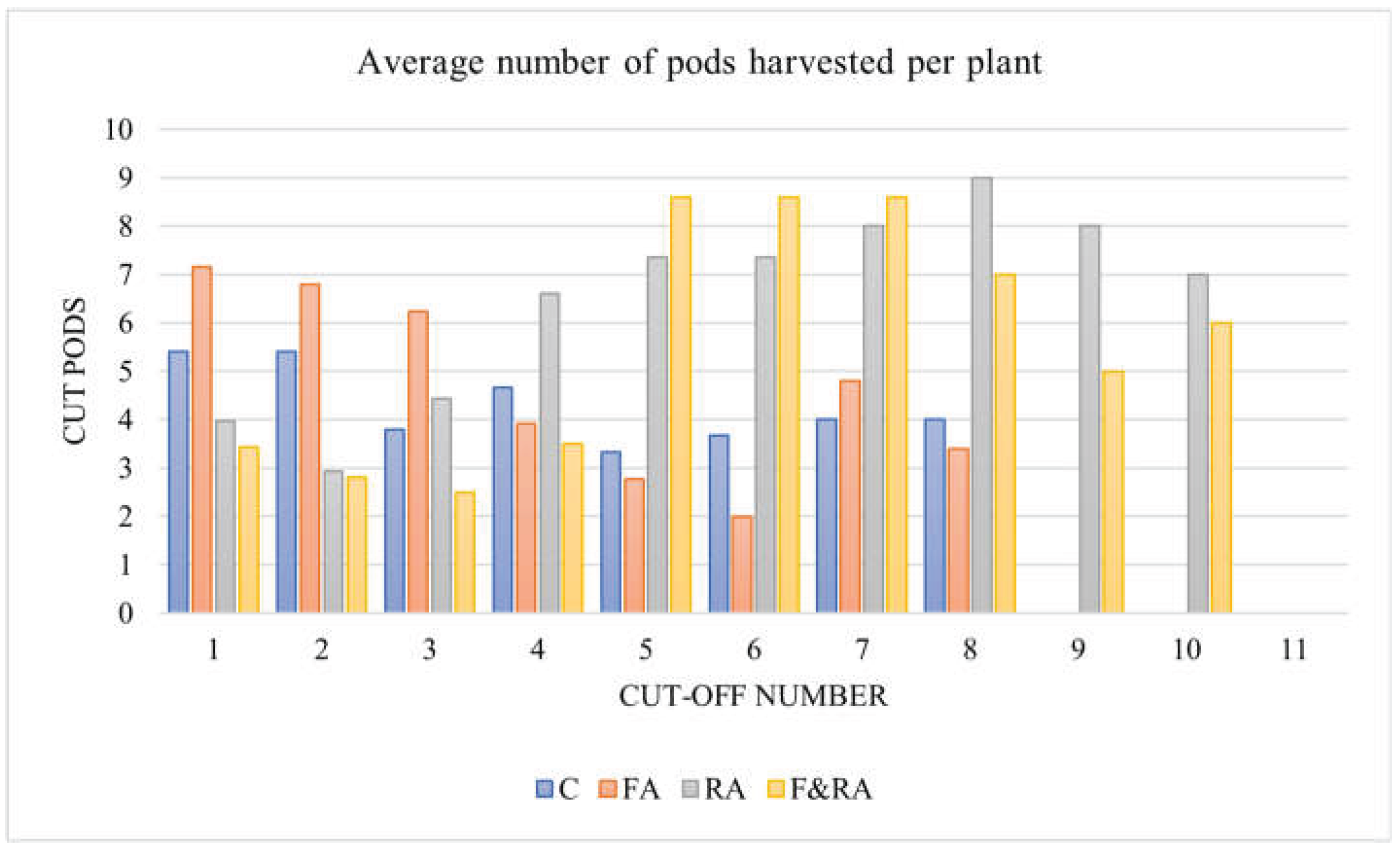
Figure 3 shows the average number of pods harvested per plant during ten harvests for each treatment.
Figure 4 shows that treatments RA (gray) and F&RA (yellow) significantly outperformed the control (blue) and FA (orange) treatments in terms of average pod production per plant. This difference becomes evident from the fourth harvest in treatment RA and from the fifth harvest in F&RA, continuing throughout the rest of the growing season. Furthermore, both RA and F&RA extended the harvest cycle, allowing for two additional harvests compared to treatments C and FA, which showed an early decline in yield.
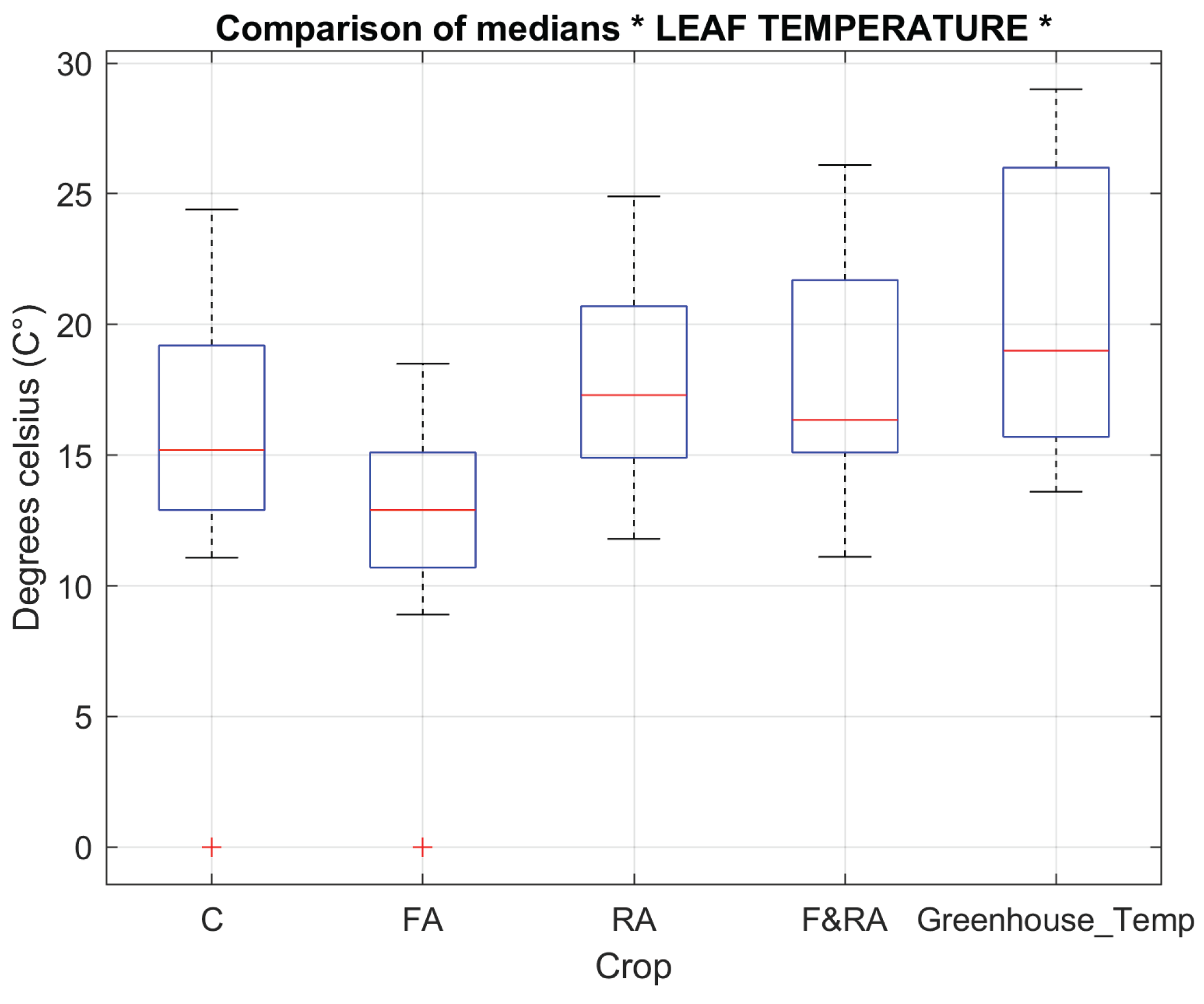
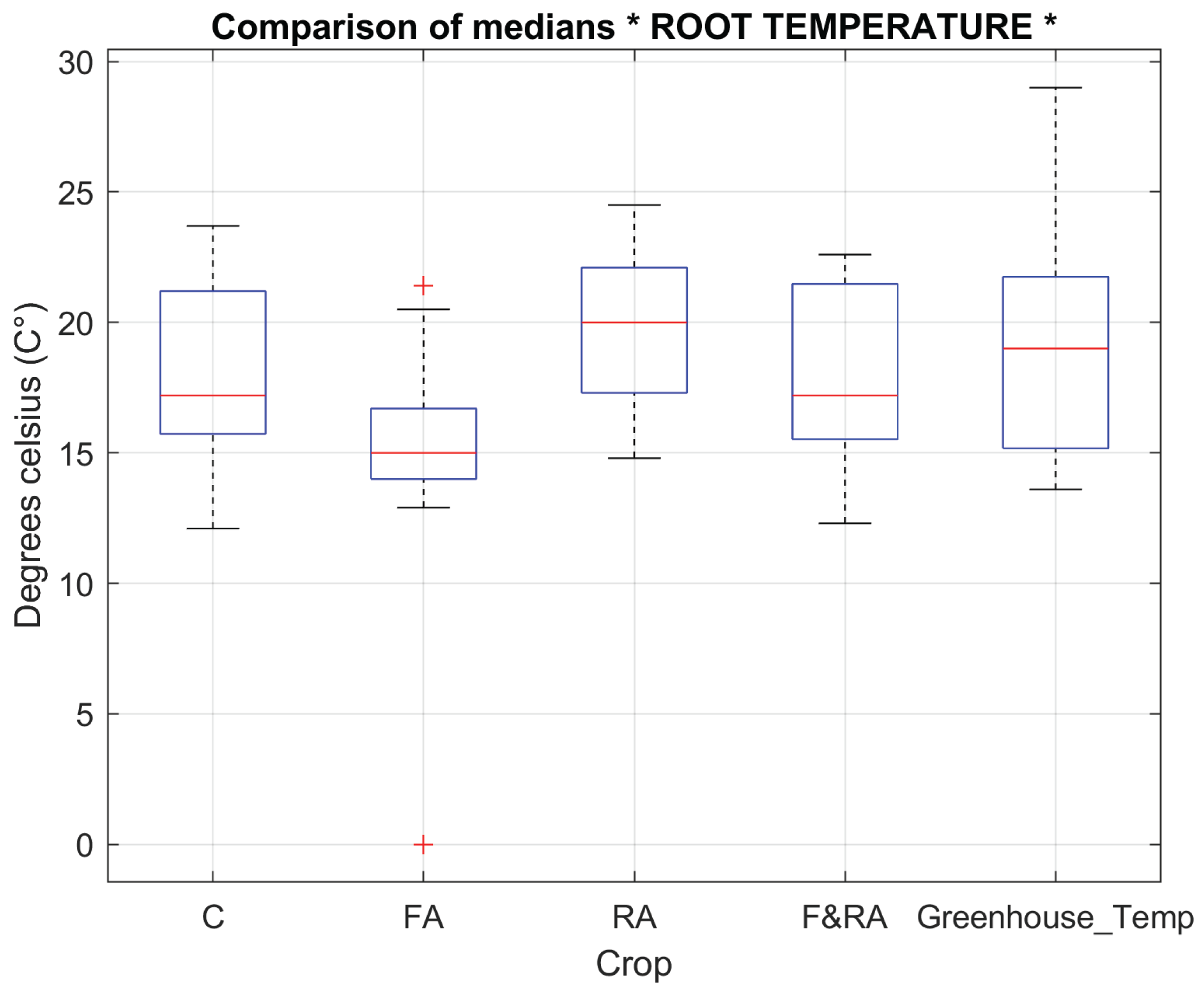
Leaf temperature (
Figure 5) and root temperature (
Figure 6) reflect the plant's ability to adapt to changing environmental conditions and directly influence the efficiency of water and nutrient absorption.
3.3. Agricultural Yield
Agricultural yield allows us to estimate the amount of product obtained in relation to the cultivated area. For this study, agricultural yield was determined using the average weight of pods harvested per plant per square meter (kg/m²). This data allowed us to compare the medians among the different experimental treatments in order to identify any statistically significant differences.
The Kruskal-Wallis test applied to the variable "number of pods harvested" allowed us to evaluate the possible existence of statistical differences among the four experimental treatments (C, FA, RA, and F&RA). The results yielded a significant value of p = 0.1131, which indicates that there are no significant differences among the medians of the treatments evaluated (p > 0.05).
Furthermore, the average agricultural yield values were calculated for each treatment. The results are shown in
Table 3. It can be observed that the RA treatment obtained the highest yield, followed by F&RA, which agrees with the results observed for the duration of the production cycle and the number of cuts performed.
Figure 7 shows the box plot of the average agricultural yield values.
3.4. Nutritional Quality Analysis of Green Beans
To determine if the treatments applied to green beans grown in the aeroponic system had a statistically significant influence on their nutritional composition, the medians of each experimental group were compared for micro and macronutrient variables, identifying statistically significant differences in several of the analyzed parameters. This statistical approach provided a basis for interpreting the effect of the treatments on nutritional quality under controlled aeroponic production conditions.
3.4.1. Macronutrients
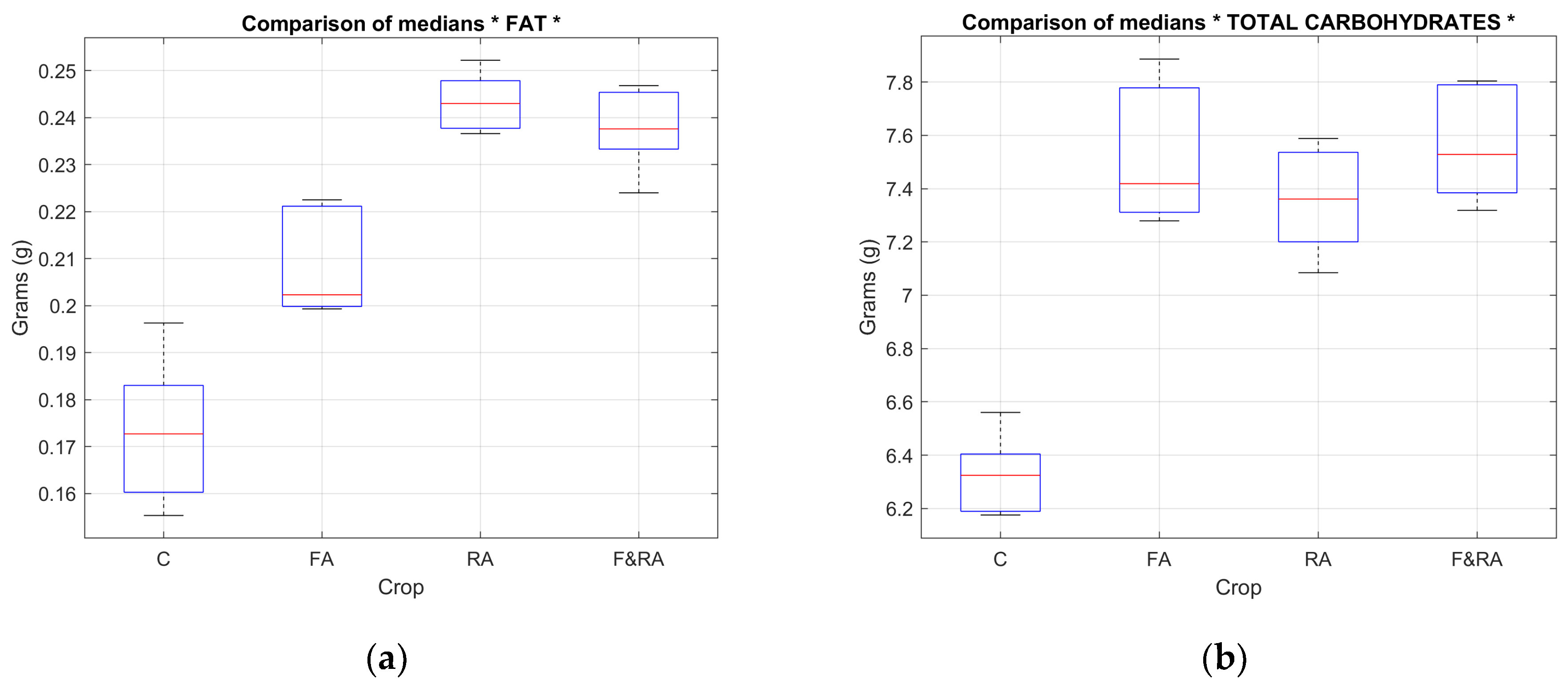
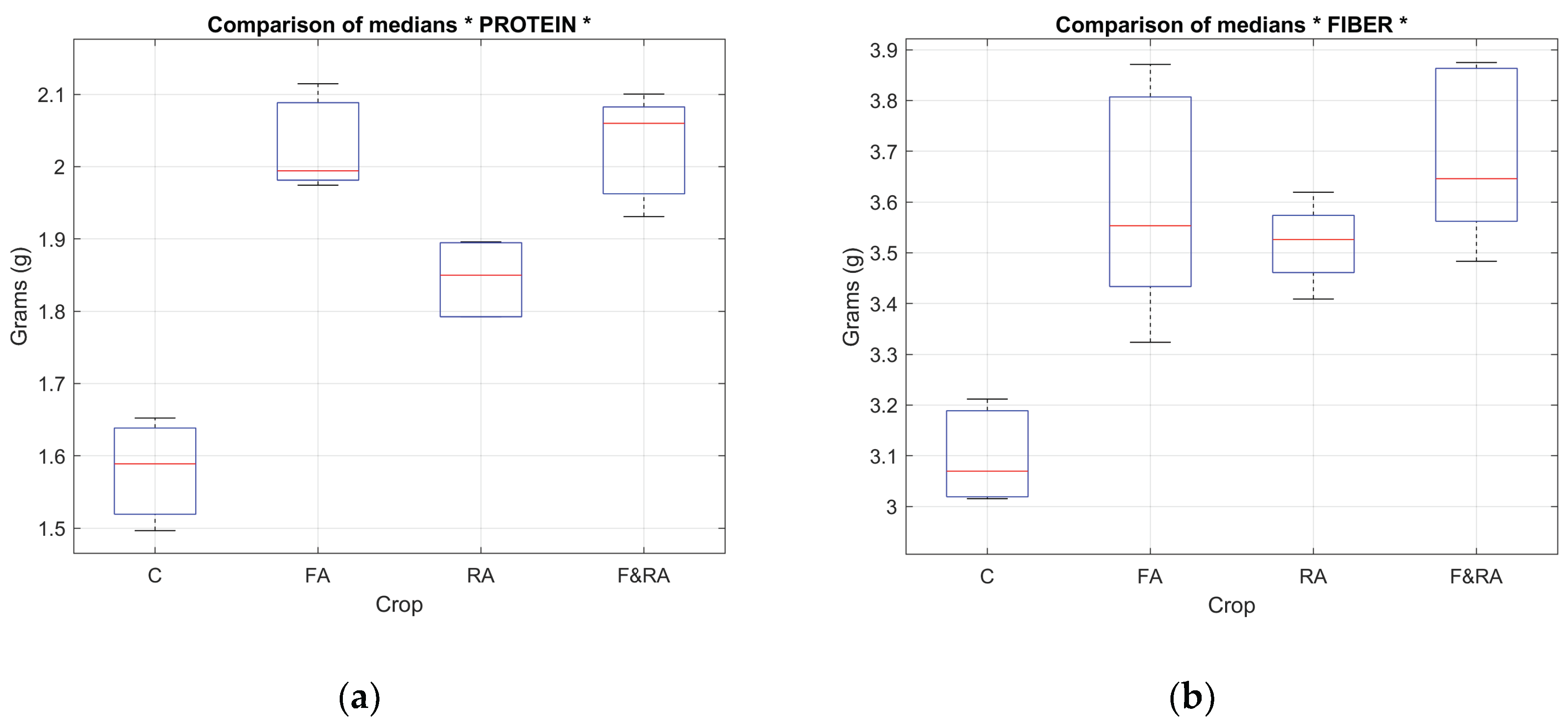
Table 4 shows the results of the median comparison test for the macronutrient variables: total carbohydrates, sugars, fiber, fat, protein, and water content. Significant differences were found in all cases.
3.4.1. Micronutrients
In the case of micronutrients (
Table 5), the results were more heterogeneous. Of the seven variables analyzed, only two showed significant differences.
Calcium (p = 0.0486) and iron (p = 0.0065) are elements directly associated with fruit quality, post-harvest resistance, and nutraceutical value. Magnesium, manganese, phosphorus, potassium, and zinc did not show statistically significant differences (p > 0.05), which may be explained by a homogeneous distribution of these elements among the treatments or by the limited capacity of the applied biofertilizer to significantly influence these micronutrients during the study.
Figure 8 (a) and (b) and
Figure 9 (a) and (b) show the mean box plot for some macro and micronutrient variables.
The behavior of the variables suggests that the application of the biofertilizer can favorably modify the accumulation of essential mineral compounds, contributing not only to agronomic performance but also to an improvement in the crop's nutritional value.
4. Discussion
The evaluation of the phenological evolution and yield of Phaseolus vulgaris L. var. Opus in an experimental aeroponic system allows for the interpretation of the results obtained in terms of the plant's physiological response under controlled conditions. This section discusses the variations observed between treatments, as well as their relationship with previous studies under hydroponic and soil cultivation conditions, with the aim of identifying strengths, limitations, and possible agronomic implications of using aeroponics in green bean cultivation. The possible effects of the interaction between physiological and technical factors on the phenological and productive indicators recorded during the experiment are also analyzed.
4.1. Product Evaluation
Product evaluation is a key element for understanding the impact of the treatments applied on the qualitative and quantitative attributes of yield. This subsection analyzes the effects of the Ascophyllum nodosum-based biofertilizer on variables directly related to the commercial quality of green beans (Phaseolus vulgaris L. var. Opus), such as the number of grains per pod and average pod weight. These variables reflect the physiological and reproductive efficiency of the crop under aeroponic conditions, where the controlled availability of nutrients is stable and allows for isolating the specific influence of each treatment.
The comparative analysis between the root (RA), foliar (FA), combined (F&RA), and control treatments showed how the biofertilizer application route modulates biomass partitioning, grain filling synchrony, and reproductive development uniformity. The biofertilizer treatments, especially RA and F&RA, showed greater uniformity and a tendency toward a higher number of grains (
Figure 1). However, pod weight was slightly lower (
Figure 2), demonstrating an inverse relationship between the quantity and individual grain size. This behavior can be attributed to the redistribution of physiological resources stimulated by the bioactive compounds in the extract, which contains natural phytohormones, complex carbohydrates, and essential minerals that promote cell division, the formation of reproductive structures, and mineral absorption.
In RA and F&RA, root absorption favored a sustained supply of nutrients, increasing the production of reproductive structures (pods and grains), although with less individual biomass accumulation per grain. In contrast, the control and FA groups showed fewer grains, but larger in size, indicating less synchronicity in grain filling. Studies such as of [
32] and [
33] agree that
A. nodosum extracts increase the number of tomato fruits (
Lycopersicum esculentum Mill and
Solanum lycopersicum L., correspondingly) without significantly altering the average weight. From an agronomic perspective, greater uniformity in grain formation is directly related to yield stability and the commercial quality of the product, especially in sustainable systems such as aeroponics.
Previous research has reported positive responses, but also inconsistent ones depending on the crop, dose, and conditions. [
34] observed effects on growth and biomass in
Vigna aconitifolia (Jacq.), while [
35] reported approximately a 13% decrease in yield and number of fruits in cucumber (
Cucumis sativus L.). [
36] observed increases in Alphonse Lavallée grape (
Vitis vinifera L.) but cautioned that the effects depend on the species, dose, and conditions.
4.2. Vegetative State
In order to understand the physiological and morphological response of
Phaseolus vulgaris L. var. Opus to the treatments, vegetative variables such as the number of flowers, pods and plant height which did not show significant differences between treatments, indicating a relatively homogeneous phenotypic response were evaluated. These variables did not show significant differences between treatments, indicating a relatively homogeneous phenotypic response. In contrast, leaf and root temperatures did show significant differences (
p < 0.01), suggesting variations in transpiration, root absorption, and thermal regulation [
37,
38,
39,
40].
In the yield evaluation by cuttings, RA and F&RA significantly outperformed the control and FA treatments from the fourth and fifth cuttings onward, extending production for two additional cuttings, thus demonstrating greater productive persistence. This aligns with the findings of [
41], who highlight that bio-stimulants improve chlorophyll biosynthesis, photosynthetic efficiency, and biomass accumulation. Root application provided a continuous supply of nutrients, while foliar application did not outperform the control, indicating that isolated foliar application may not be sufficient to sustain high productivity throughout the cycle, at least under the experimental conditions of this study.
[
42] report that
A. nodosum improves osmotic regulation, water use efficiency, and physiological stability, promoting more vigorous growth and a longer reproductive cycle. [
43] demonstrated that different formulations of the extract promote root or foliar development depending on their composition. However, [
44] and [
45] indicate that
A. nodosum does not always produce significant improvements, depending on the crop, dosage, and application method.
Factors such as crop type, plant part treated, dosage, formulation, growing conditions, and application route affect the outcome of A. nodosum application. This indicates that yield improvements or increased productivity metrics are not always achieved. While the results of this study show a positive effect for the system used, the literature reveals that this effect is not universal.
On the other hand, temperature played a determining role. The crop experienced morning temperatures of 17°C and temperatures up to 40°C at midday, which affected flowering, height, and grain weight, as indicated by [
46]. Temperatures outside the optimum range generate variability in flowers and pods [
47,
48]. These thermal fluctuations explain some of the variability observed in phenological parameters.
4.3. Agricultural Performance
Performance is a key indicator of the agronomic success of any biotechnological treatment, as it reflects the interaction between plant physiology, nutrient availability, and the environmental conditions of the production system [
49]. In this study, the variable corresponding to the number of pods per plant showed a statistically uniform behavior across treatments, suggesting that the biofertilizer did not generate significant differences in productivity under the evaluated aeroponic conditions. The evaluated variable, corresponding to the number of pods harvested per plant, presented a significance value of
p = 0.0113, indicating that the biofertilizer did not modify productivity in this aeroponic system, probably due to the controlled conditions and the uniform availability of nutrients. While technologies such as nanoparticles or microbial consortia have shown significant increases in the performance of
Phaseolus vulgaris [
28,
50], microalgae-based biofertilizers may require adjustments in formulation, dosage, or application frequency to enhance their effect [
34]. In this study, the stable response suggests that the aeroponic environment minimized differences between treatments. Future research could focus on comparing different concentrations of the biofertilizer or combining its use with other technologies, such as nanoparticles or plant growth-promoting microorganisms, to maximize the productive response in aeroponic systems.
4.4. Analysis of the Nutritional Quality of Green Beans
In order to evaluate the impact of the experimental treatments on the nutritional quality of green beans, a statistical analysis was conducted to determine if there were significant differences between the medians of the nutritional variables measured in the harvested fruits. This analysis allowed us to identify the potential influence of the microalgae-based biofertilizer on the chemical composition and nutritional value of the crop. The variables considered for this evaluation included total carbohydrates, sugars, fiber, fat, protein, and water, selected for their nutritional relevance both for human consumption and for analyzing the biofertilizer's effectiveness in improving the crop's added value [
51,
52]. The results of the statistical analysis are presented in
Table 4, which shows the influence of each treatment on the nutritional quality of the green beans. Wet digestion and spectrophotometry allowed us to quantify macro and micronutrients, which are detailed below.
4.5. Macronutrients
Regarding total carbohydrates (p = 0.008), at least one treatment resulted in a significantly different accumulation of carbohydrates, which may be related to photosynthetic efficiency and sugar translocation under specific nutritional conditions. The sugar content (p = 0.0073) confirms variability among treatments, possibly due to differences in nutrient availability or the biofertilizer application method, affecting the plant's primary metabolism. Fiber (p = 0.0064) and fat (p = 9.8285e-04) content: fiber is linked to cell structure, while fat, although present in low proportions in green beans, can vary depending on physiological maturity. Both results suggest that the treatments directly influence the structural and nutritional quality of the fruit. Protein (p = 0.0011) is one of the most relevant nutrients from a dietary perspective. Its statistical significance implies that the type of biofertilizer application may have favored greater nitrogen fixation or protein synthesis. The water content (p = 9.8285e-04) reflects differences in water retention of the fruit, possibly related to the physiological state of the crop and the type of nutrition received.
These results demonstrate that the macronutrient composition of the green bean was significantly affected by the different treatments, validating the biofertilizer's potential to improve the quality of the harvested product.
4.6. Micronutrients
The statistical analysis for micronutrients allowed us to evaluate the influence of the biofertilizer treatments on the average concentration of micronutrients in the green bean fruits. These results showed statistically significant differences for calcium (
p = 0.0486) and iron (
p = 0.0065), while the other elements, such as magnesium, manganese, phosphorus, potassium, and zinc, did not show significant differences (
p > 0.05). These findings indicate that the biofertilizer that was applied had a positive and differential effect on the absorption or accumulation of calcium and iron in the fruit. Similarly, the work of [
42] reported an improvement in mineral absorption and nutrient availability, which is relevant from a physiological and nutraceutical perspective. Calcium is an essential element for cell wall integrity, fruit firmness, and resistance to mechanical or physiological damage during post-harvest. Iron, for its part, actively participates in key metabolic processes such as chlorophyll synthesis and electron transport, and is also a fundamental component of the crop's nutritional value, given its direct impact on food quality and human health.
The absence of significant differences in the remaining micronutrients could be attributed to a homogeneous distribution of these elements in the aeroponic substrate, or to the biofertilizer's limited capacity to modulate their availability under the established experimental conditions. Furthermore, the uniformity of the fertigation system and environmental control within the greenhouse may have reduced variations in nutrient absorption, contributing to the observed homogeneity.
Studies such as [
53] on green beans (
Phaseolus vulgaris L. cv. Perona) highlight that quality should be considered along with post-harvest handling and storage to preserve vitamins and minerals. Similarly, [
26] showed that liquid biofertilizers improve growth and nutrient absorption in hydroponic systems.
The results suggest that the nutritional quality of green beans can be selectively modulated by the application of biofertilizer, highlighting a particular influence on the elements involved in the structural quality and nutraceutical value of the fruit. This reinforces the hypothesis that certain biological components of the fertilizer, such as growth-promoting microorganisms or bioactive compounds, can favor the differential absorption of specific micronutrients, especially calcium and iron, without significantly altering the concentration of other essential elements.
Finally, this nutritional response is closely linked to yield and the number of pods per plant, since greater availability of calcium and iron could have indirectly contributed to strengthening the vegetative and reproductive structure, favoring the productive persistence observed in the root and combined application treatments. This suggests that nutritional improvement not only has implications for fruit quality but also for the crop's productive efficiency under controlled aeroponic conditions. In this sense, the integration of advanced agricultural technologies with appropriate storage protocols represents an opportunity to improve the crop's nutritional value, closing the production cycle with a comprehensive approach to quality.
5. Conclusions
The results from this study demonstrate that the application of a biofertilizer formulated from Ascophyllum nodosum promoted distinct physiological responses in Phaseolus vulgaris L. var. Opus cultivation under aeroponic conditions. While some variables, such as the number of pods per plant, did not show significant differences between treatments, their uniformity suggests a productive steadiness resulting from the use of the biofertilizer, maintaining yield levels comparable to the control crop. In contrast, positive effects were observed in the absorption and accumulation of calcium and iron, essential elements for the structural integrity, firmness, and nutraceutical quality of the fruit, highlighting the potential of this biotechnological input in improving the mineral composition and nutritional value of green beans.
Furthermore, the variability observed in flowering was mainly associated with thermal and photoperiodic fluctuations within the greenhouse, factors that influenced reproductive synchrony and the phenological expression of the crop. These results confirm the need to consider environmental control as a determining factor for maximizing the physiological efficiency of biotechnological treatments. Finally, the yield was established as a key indicator of agronomic success, integrating physiological response, nutrient availability, and production system conditions. As a whole, the findings suggest that the A. nodosum-based biofertilizer represents a sustainable alternative with agronomic potential whose effect could be optimized through adjustments in formulation, concentration, or application frequency, as well as through its integration with plant growth-promoting microorganisms or complementary technologies in high-efficiency aeroponic systems.
Future research should evaluate interactions such as water stress, temperature variations, and different concentration levels of the biofertilizer.
Author Contributions
Conceptualization, J.A., J.P., and N.R.; methodology, J.A., J.P., N.R., and M.B.; software, J.M., and A.B; validation, J.A., J.P., and M.B.; formal analysis, J.A., and J.P.; investigation, J.A., J.P., N.R., and M.B.; resources, J.M., and A.B; data curation, J.A., and J.P.; writing—original draft preparation, J.A., J.P., N.R., M.B., J.M., and A.B; visualization, J.A., and J.P.; supervision, J.P., and M.B.; project administration, J.A., A.B., and J.P., and M.B.; writing - review and editing by all authors. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
The raw data supporting the conclusions of this study are available from the corresponding author upon reasonable request.
Acknowledgments
We acknowledge the support of the National Technological Institute of Mexico and the Secretariat of Science, Humanities, Technology and Innovation (SECIHTI).
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| L |
Linnaeus |
| var |
Variety |
| cv |
Cultivated variety |
| LD |
Linear dichroism |
References
- Zandalinas, S.I.; Fritschi, F.B.; Mittler, R. Global Warming, Climate Change, and Environmental Pollution: Recipe for a Multifactorial Stress Combination Disaster. Trends Plant Sci. 2021, 26, 588–599. [Google Scholar] [CrossRef]
- Borrelli, P.; Robinson, D.A.; Panagos, P.; Lugato, E.; Yang, J.E.; Alewell, C.; Wuepper, D.; Montanarella, L.; Ballabio, C. Land use and climate change impacts on global soil erosion by water (2015–2070). Proc. Natl. Acad. Sci. U.S.A. 2020, 117(36), 21994–22001. [Google Scholar] [CrossRef] [PubMed]
- United Nations; Department of Economic and Social Affairs; Population Division. World Population Prospects 2022: Summary of Results. 2022. Available online: https://www.un.org/development/desa/pd/sites/www.un.org.development.desa.pd/files/wpp2022_summary_of_results.pdf (accessed on 20 November 2025).
- Fasciolo, B.; Awouda, A.; Bruno, G.; Lombardi, F. A smart aeroponic system for sustainable indoor farming. Procedia CIRP 2023, 116, 636–641. [Google Scholar] [CrossRef]
- Anderson, J.T.; Song, B.-H. Plant adaptation to climate change—Where are we? J. Syst. Evol. 2020, 58, 533–545. [Google Scholar] [CrossRef]
- Bhardwaj, R.; Parashar, A.; Parewa, H.; Vyas, L. An Alarming Decline in the Nutritional Quality of Foods: The Biggest Challenge for Future Generations’ Health. Foods 2023, 12, 1–26. [Google Scholar] [CrossRef]
- Lan, Y.; Xu, B.; Huan, Y.; Guo, J.; Liu, X.; Han, J.; Li, K. Food Security and Land Use under Sustainable Development Goals: Insights from Food Supply to Demand Side and Limited Arable Land in China. Foods 2023, 12, 4168. [Google Scholar] [CrossRef]
- Acquah, C.; Ohemeng, G.; Power, K.; Tosh, S. The Effect of Processing on Bioactive Compounds and Nutritional Qualities of Pulses in Meeting the Sustainable Development Goal 2. Front. Sustain. Food Syst. 2021, 5, 1–16. [Google Scholar] [CrossRef]
- Lisciani, S.; Marconi, S.; Le Donne, C.; Camilli, E.; Aguzzi, A.; Gabrielli, P.; Gambelli, L.; Kunert, K.; Marais, D.; Vorster, B.; Alvarado, K.; Reboul, E.; Comenelli, C.; Preite, C.; Sparvoli, F.; Losa, A.; Sala, T.; Botha, A.; Ferrari, M. Legumes and Common Beans in Sustainable Diets: Nutritional Quality, Environmental Benefits, Spread and Use in Food Preparations. Front. Nutr. 2024, 11, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ammar, E.E.; Aioub, A.A.; Elesawy, A.E.; Karkourd, A.M.; Mouhamedd, M.S.; Amer, A.A.; EL-Shershabye, N.A. Algae as Bio-fertilizers: Between current situation and future. Saudi J. Biol. Sci. 2022, 29, 3083–3096. [Google Scholar] [CrossRef]
- Miranda, A.M.; Hernandez-Tenorio, F.; Villalta, F.; Vargas, G.J.; Sáez, A.A. Advances in the development of Biofertilizers and Biostimulants. Biology 2024, 13(3), 199. [Google Scholar] [CrossRef] [PubMed]
- Bello, A.S.; Saadaoui, I.; Ben-Hamadou, R. Beyond the Source of Bioenergy: Microalgae in Modern Agriculture as a Biostimulant, Biofertilizer, and Anti-Abiotic Stress. Agronomy 2021, 11, 1–25. [Google Scholar] [CrossRef]
- Shukla, P.S.; Mantin, E.G.; Adil, M.; Bajpai, S.; Critchley, A.T.; Prithiviraj, B. Ascophyllum nodosum-Based Biostimulants: Sustainable Applications in Agriculture for the Stimulation of Plant Growth, Stress Tolerance, and Disease Management. Front. Plant Sci. 2019, 10, 1–22. [Google Scholar] [CrossRef]
- Frioni, T.; VanderWeide, J.; Palliotti, A.; Tombesi, S.; Poni, S.; Sabbatini, P. Foliar vs. soil application of Ascophyllum nodosum extracts to improve grapevine water stress tolerance. Sci. Hortic. 2021, 277, 1–7. [Google Scholar] [CrossRef]
- Campobenedetto, C.; Agliassa, C.; Mannino, G.; Vigliante, I.; Contartese, V.; Secchi, F.; Bertea, C.M. A Biostimulant Based on Seaweed (Ascophyllum nodosum and Laminaria digitata) and Yeast Extracts Mitigates Water Stress Effects on Tomato (Solanum lycopersicum L.). Agriculture 2021, 11, 1–16. [Google Scholar] [CrossRef]
- Rinaldi, L.K.; Calandrelli, A.; Miamoto, A.; Rodrigues e Silva, M.T.; Dias-Arieira, C.R. Application of Ascophyllum nodosum extract and its nutrient components for the management of Meloidogyne javanica in soybean. Chil. J. Agric. Res. 2022, 82, 127–137. [Google Scholar] [CrossRef]
- Castronuovo, D.; Comegna, A.; Belviso, C.; Satriani, A.; Lovelli, S. Zeolite and Ascophyllum nodosum-Based Biostimulant Effects on Spinach Gas Exchange and Growth. Agriculture 2023, 13, 1–8. [Google Scholar] [CrossRef]
- Petropoulos, S.A.; Fernandes, Â.; Plexida, S.; Chrysargyris, A.; Tzortzakis, N.; Barreira, J.; Barros, L.; Ferreira, I. Biostimulants Application Alleviates Water Stress Effects on Yield and Chemical Composition of Greenhouse Green Bean (Phaseolus vulgaris L.). Agronomy 2019, 9, 1–26. [Google Scholar] [CrossRef]
- Długosz, J.; Piotrowska-Długosz, A.; Kotwica, K.; Przybyszewska, E. Application of Multi-Component Conditioner with Clinoptilolite and Ascophyllum nodosum Extract for Improving Soil Properties and Zea mays L. Growth and Yield. Agronomy 2020, 10, 1–22. [Google Scholar] [CrossRef]
- Drygás, B.; Depciuch, J.; Puchalski, C. Effect of Ascophyllum nodosum Alga Application on Microgreens, Yield, and Yield Components in Oats (Avena sativa L.). Agronomy 2021, 11, 1–19. [Google Scholar] [CrossRef]
- do Rosário Rosa, V.; Farias dos Santos, A.L.; Alves da Silva, A.; Peduti Vicentini Sab, M.; Germino, G.H.; Barcellos Cardoso, F.; de Almeida Silva, M. Increased soybean tolerance to water deficiency through biostimulant based on fulvic acids and Ascophyllum nodosum (L.) seaweed extract. Plant Physiol. Biochem. 2020, 153, 1–50. [Google Scholar] [CrossRef]
- Villa e Vila, V.; Alves Marques, P.A.; Rezende, R.; Soares Wenneck, G.; de Souza Terassi, D.; Alves Andrean, F.B.; de Faria Nocchi, R.C.; Matumoto-Pintro, P.T. Deficit Irrigation with Ascophyllum nodosum Extract. Agronomy 2023, 13, 1–14. [Google Scholar] [CrossRef]
- Staykov, N.S.; Angelov, M.; Petrov, V.; Minkov, P.; Kanojia, A.; Guinan, K.J.; Alseekh, S.; Fernie, A.R.; Sujeeth, N.; Gechev, T.S. An Ascophyllum nodosum-Derived Biostimulant Protects Model. Metabolites 2021, 11, 1–20. [Google Scholar]
- Garzón, J.; Montes, L.; Garzón, J.; Lampropoulos, G. Systematic Review of Technology in Aeroponics: Introducing the Technology Adoption and Integration in Sustainable Agriculture Model. Agronomy 2023, 13, 1–23. [Google Scholar] [CrossRef]
- Faisal, A.; Rajashekar, V.; Biswal, P.; Mukherjee, A.; Bhowmick, G.D. Advancing Agriculture with Aeroponics: A Critical Review of Methods, Benefits, and Limitations. In Hydroponics; Encyclopedia of Sustainability Science and Technology Series; Springer: New York, NY, USA, 2024; pp. 311–322. [Google Scholar]
- Madushani, G.; Karunarathna, B. Impact of organic liquid fertilizers on growth and yield performance of Green Bean (Phaseolus vulgaris L.) in non-circulating hydroponic system. AGRIEAST 2023, 25, 25–41. [Google Scholar] [CrossRef]
- Zareba, A.; Krzemińska, A.; Adynkiewicz Piragas, M.; Stojanovski, T.; Jia, H.; Privitera, R.; van der Horst, D. Multifunctional Vertical Farming Systems as a Basis for Transforming Urban Food Systems Amid Climate Change. Sustainability 2025, 17, 1–18. [Google Scholar] [CrossRef]
- Bautista, J.; Cruz, O.; Hernández, O.; Sánchez, E.; Jacobo, J.; Preciado, P.; Ávila, G.; Ojeda, D. Zinc sulphate or zinc nanoparticle applications to leaves of green beans. Folia Hortic. 2021, 33, 365–375. [Google Scholar] [CrossRef]
- Nadeem, M.A.; Yeken, M.Z.; Shahid, M.Q.; Habyarimana, E.; Yılmaz, H.; Alsaleh, A.; Hatipoğlu, R.; Çilesiz, Y.; Khawar, K.M.; Ludidi, N.; Ercişli, S.; Aasim, M.; Karaköy, T.; Baloch, F.S. Common Bean as a Potential Crop for Future Food Security: An Overview of Past, Current and Future Contributions in Genomics, Transcriptomics, Transgenics and Proteomics. Biotechnol. Biotechnol. Equip. 2021, 35, 759–787. [Google Scholar] [CrossRef]
- de Paula, E.; Almeida, R.N.; Santos, T.d.O.; Souza Neto, J.D.; Riva-Souza, E.M.; Posse, S.C.P.; Souza, M.N.; Madella de Oliveira, A.d.F.; Santos Júnior, A.C.; Santos, J.O.; Pimenta, S.; Bento, C.d.S.; Moulin, M.M. Genetic Diversity of Common Bean (Phaseolus vulgaris L.) Landraces Based on Morphological Traits and Molecular Markers. Plants 2024, 13, 2584. [Google Scholar] [CrossRef]
- Njau, S.N.; Parker, T.A.; Duitama, J.; Gepts, P.; Arunga, E.E. QTL mapping for pod quality and yield traits in snap bean (Phaseolus vulgaris L.). Front. Plant Sci. 2024, 15, 1372202. [Google Scholar] [CrossRef] [PubMed]
- Ali, N.; Farrell, A.; Ramsubhag, A.; Jayaraman, J. The effect of Ascophyllum nodosum extract on the growth, yield and fruit quality of tomato grown under tropical conditions. J. Appl. Phycol. 2015, 28, 1353–1362. [Google Scholar] [CrossRef]
- Subramaniyan, L.; Veerasamy, R.; Prabhakaran, J.; Selvaraj, A.; Algarswamy, S.; Karuppasami, K.M.; Thangavel, K.; Nalliappan, S. Biostimulation Effects of Seaweed Extract (Ascophyllum nodosum) on Phytomorpho-Physiological, Yield, and Quality Traits of Tomato (Solanum lycopersicum L.). Horticulturae 2023, 9(3), 348. [Google Scholar] [CrossRef]
- Verma, N.; Sehrawat, K.D.; Mundlia, P.; Sehrawat, A.R.; Choudhary, R.; Rajput, V.D.; Minkina, T.; van Hullebusch, E.D.; Siddiqui, M.H.; Alamri, S. Potential Use of Ascophyllum nodosum as a Biostimulant for Improving the Growth Performance of Vigna aconitifolia (Jacq.) Marechal. Plants 2021, 10(11), 2361. [Google Scholar] [CrossRef]
- Zamljen, T.; Šircelj, H.; Veberič, R.; Hudina, M.; Slatnar, A. Impact of Two Brown Seaweed (Ascophyllum nodosum L.) Biostimulants on the Quantity and Quality of Yield in Cucumber (Cucumis sativus L.). Foods 2024, 13, 401. [Google Scholar] [CrossRef] [PubMed]
- Doğan, O.; Yazar, K. Effect of Different Seaweed Extracts on Yield, Quality and Physiological Characteristics of the Alphonse Lavallée (Vitis vinifera L.) Grape Variety. Horticulturae 2025, 11(9), 1118. [Google Scholar] [CrossRef]
- Xia, Z.; Zhang, G.; Zhang, S.; Wang, Q.; Fu, Y.; Lu, H. Efficacy of Root Zone Temperature Increase in Root and Shoot Development and Hormone Changes in Different Maize Genotypes. Agriculture 2021, 11, 1–13. [Google Scholar] [CrossRef]
- Son, K-H.; Sim, H.-S.; Lee, J.-K.; Lee, J. Precise Sensing of Leaf Temperatures for Smart Farm Applications. Horticulturae 2023, 9, 1–16. [Google Scholar] [CrossRef]
- He, F.; Thiele, B.; Santhiraraja-Abresch, S.; Watt, M.; Kraska, T.; Ulbrich, A.; Kuhn, A.J. Effects of Root Temperature on the Plant Growth and Food Quality of Chinese Broccoli (Brassica oleracea var. alboglabra Bailey). Agronomy 2020, 10, 1–18. [Google Scholar] [CrossRef]
- Lam, V.P.; Kim, S.J.; Bok, G.J.; Lee, J.W.; Park, J.S. The Effects of Root Temperature on Growth, Physiology, and Accumulation of Bioactive Compounds of Agastache rugosa. Agriculture 2020, 10, 1–15. [Google Scholar] [CrossRef]
- Bulgari, R.; Franzoni, G.; Ferrante, A. Biostimulants Application in Horticultural Crops under Abiotic Stress Conditions. Agronomy 2019, 9, 1–30. [Google Scholar] [CrossRef]
- Kumari, S.; Sehrawat, K.D.; Phogat, D.; Sehrawat, A.R.; Chaudhary, R.; Sushkova, S.N.; Voloshina, M.S.; Rajput, V.D.; Shmaraeva, A.N.; Marc, R.A.; Shende, S.S. Ascophyllum nodosum (L.) Le Jolis, a Pivotal Biostimulant toward Sustainable Agriculture: A Comprehensive Review. Agriculture 2023, 13, 1–31. [Google Scholar] [CrossRef]
- Shakya, R.; Capilla, E.; Torres-Pagán, N.; Muñoz, M.; Boscaiu, M.; Lupuţ, I.; Vicente, O.; Verdeguer, M. Effect of Two Biostimulants, Based on Ascophyllum nodosum Extracts, on Strawberry Performance under Mild Drought Stress. Agriculture 2023, 13, 1–17. [Google Scholar] [CrossRef]
- Ali, O.; Ramsubhag, A.; Jayaraman, J. Biostimulatory activities of Ascophyllum nodosum extract in tomato and sweet pepper crops in a tropical environment. PLOS ONE 2019, 14, 1–19. [Google Scholar] [CrossRef]
- Ellena, M.; González, A.; Romero, I. Effect of seaweed extracts (Ascophyllum nodosum) on yield and nut quality in hazelnut. Acta Hortic. 2023, 1379, 253–258. [Google Scholar] [CrossRef]
- Urbano, P. Tratado de fitotecnia general; Mundi-Prensa: Madrid, España, 1988. [Google Scholar]
- Mishra, S.; Spaccarotella, K.; Gido, J.; Samanta, I.; Chowdhary, G. Effects of Heat Stress on Plant-Nutrient Relations: An Update on Nutrient Uptake, Transport, and Assimilation. Int. J. Mol. Sci. 2023, 24(21), 15670. [Google Scholar] [CrossRef]
- Tene, T.M.; Sari, H.; Canci, H.; Maaruf, A.; Eker, T.; Toker, C. Traits related to heat stress in Phaseolus species. Agriculture 2023, 13, 953. [Google Scholar] [CrossRef]
- Alotaibi, M.M.; Aljuaid, A.; Alsudays, I.M.; Aloufi, A.S.; AlBalawi, A.N.; Alasmari, A.; Alghanem, S.; Albalawi, B.F.; Alwutayd, K.M.; Gharib, H.S.; Awad-Allah, M.M. Effect of Bio-Fertilizer Application on Agronomic Traits, Yield, and Nutrient Uptake of Barley (Hordeum vulgare) in Saline Soil. Plants 2024, 13, 951. [Google Scholar] [CrossRef]
- Baldi, E.; Gioacchini, P.; Montecchio, D.; Mocali, S.; Antonielli, L.; Masoero, G.; Toselli, M. Effect of Biofertilizers Application on Soil Biodiversity and Litter Degradation in a Commercial Apricot Orchard. Agronomy 2021, 11, 1116. [Google Scholar] [CrossRef]
- Quintana, J.; Harrison, H.; Palta, J.; Nienhuis, J.; Kmiecik, K. Stomatal Density and Calcium Concentration of Six Snap Bean Cultivars. J. Am. Soc. Hortic. Sci. 2001, 126, 110–114. [Google Scholar] [CrossRef]
- Salinas, N.; Escalante, J.A.; Rodríguez, M.T.; Sosa, E. Rendimiento y calidad nutrimental de frijol ejotero (Phaseolus vulgaris L.) en fechas de siembra. Rev. Fitotec. Mex. 2008, 31, 235–241. [Google Scholar] [CrossRef]
- Sanchez Mata, M.C.; Camara, M.; Diez Marques, C. Extending shelf-life and nutritive value of green beans (Phaseolus vulgaris L.) by controlled atmosphere storage: micronutrients. Food Chem. 2003, 3, 309–315. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).